Experimentally Induced Hyperinsulinemia Fails to Induce Polycystic Ovary Syndrome-like Traits in Female Rhesus Macaques
Abstract
1. Introduction
2. Results
2.1. Metabolic Observations
Glucoregulatory Effects of Chronic Insulin Therapy
2.2. Metabolic Challenge Tests
Frequently Sampled Intravenous Glucose Tolerance Tests (FSIGTT) and Minimal Modeling
2.3. Body Weights and Blood Chemistry Panels
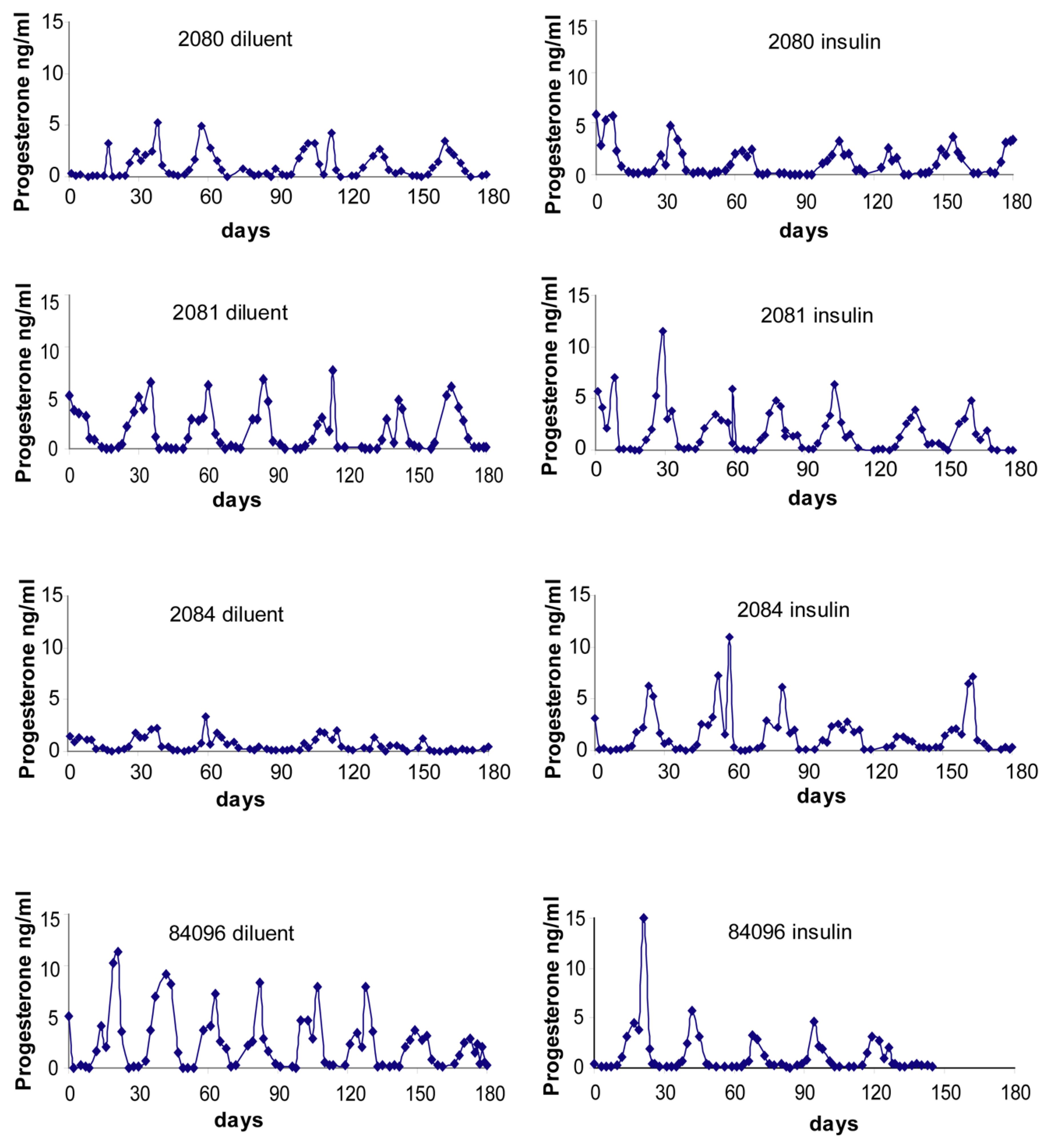
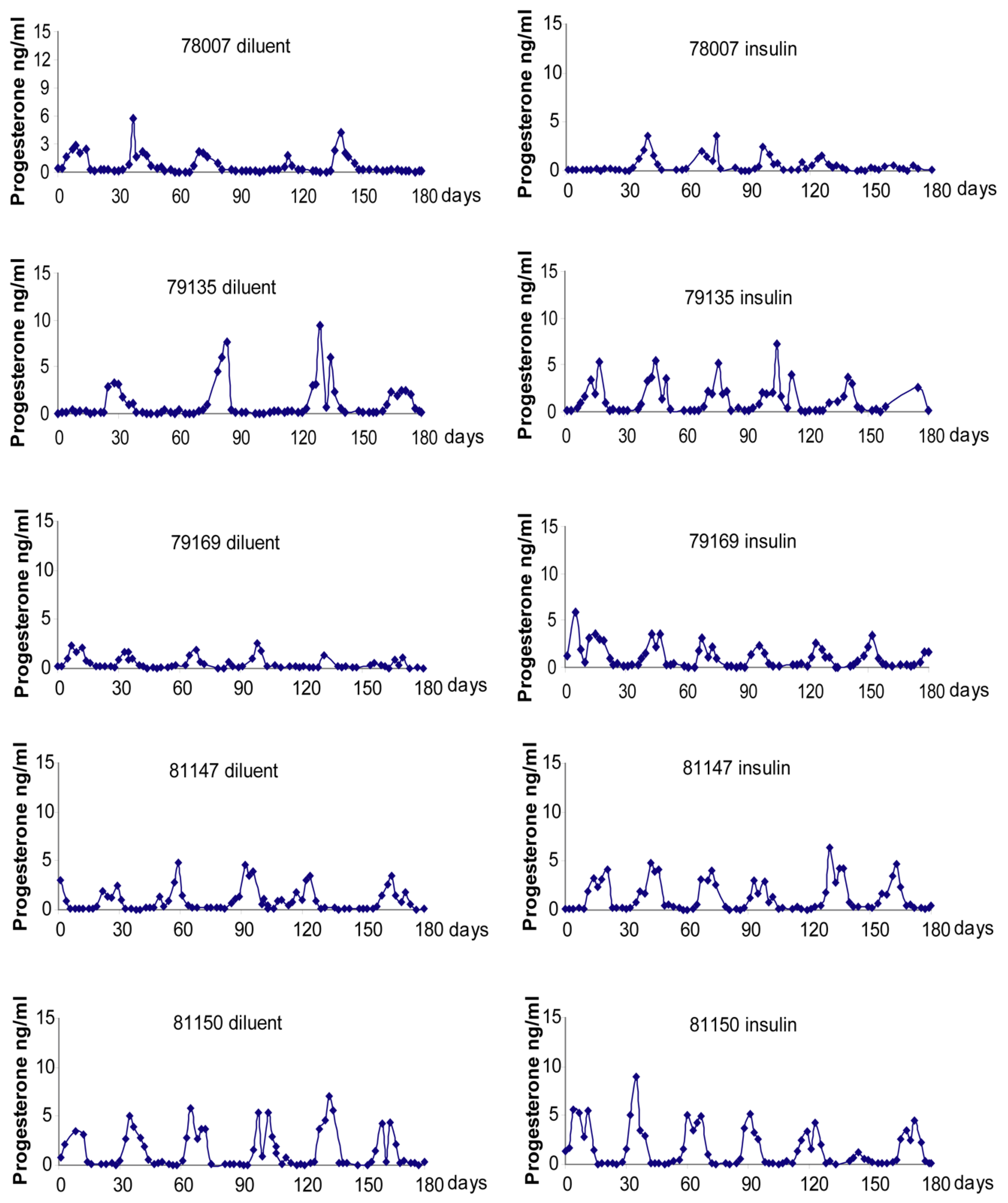
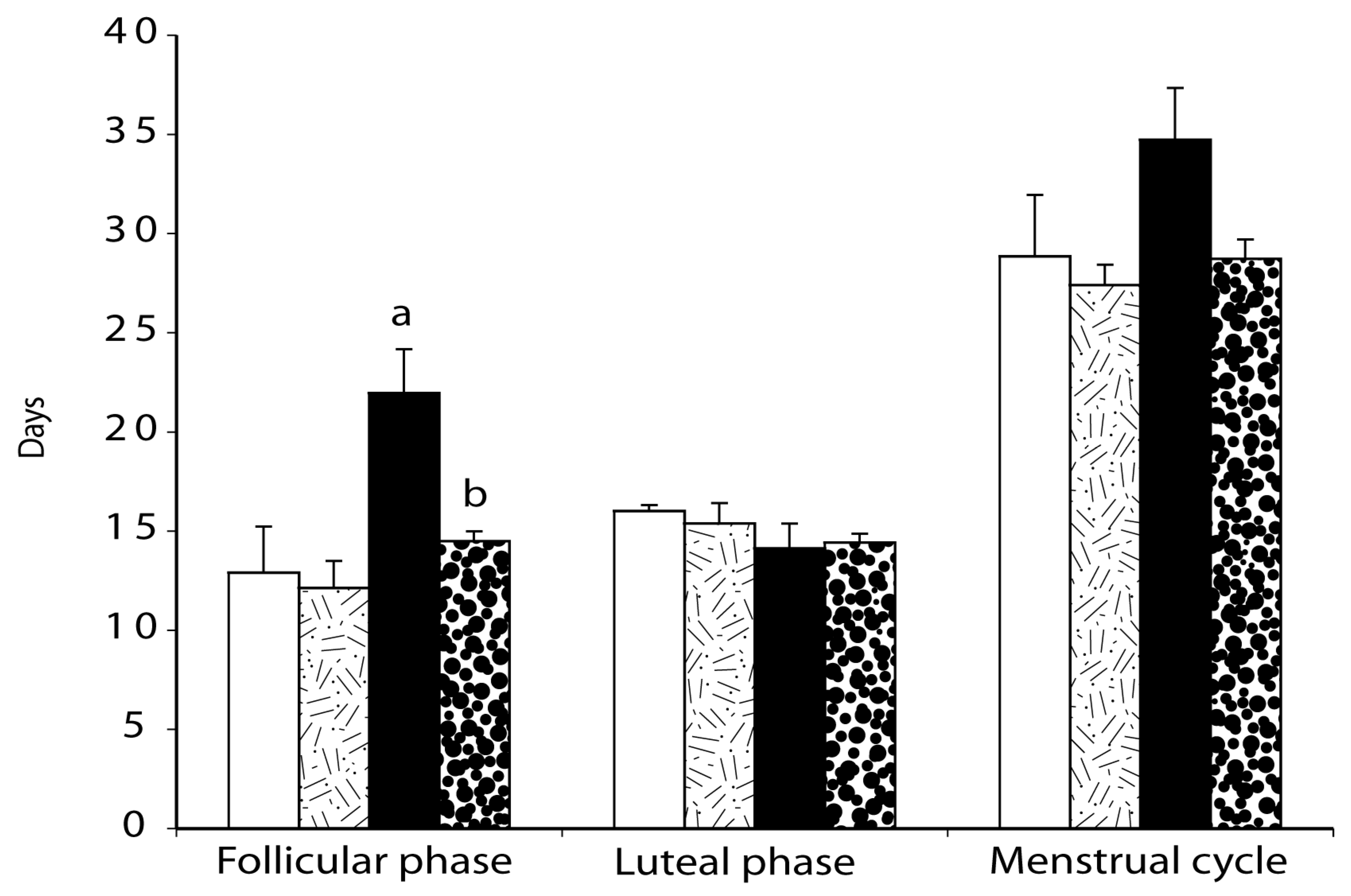
2.4. Menstrual Cycle
2.5. Ovarian Androgen Stimulation Test (at ~3–5 Months of Treatment)
2.6. Gonadotropin-Releasing Hormone (GnRH) Test of Pituitary Gonadotropin Release
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Experimental Design
4.3. Metabolic Observations
4.3.1. Body Weight, and Select Glucoregulatory, Lipid and Hepatic Parameters
4.3.2. Assessment of Serum Binding of Insulin after 6–7 Months of Exogenous Insulin Treatment
4.4. Metabolic Challenge Test
Frequently Sampled Intravenous Glucose Tolerance Test (FSIGTT) at 3–4 Months of Treatment
4.5. Reproductive Observations
Menstrual Cycle Assessment
4.6. Endocrine Challenge Tests
4.6.1. Ovarian Androgen Stimulation Test at 3–5 Months of Treatment
4.6.2. GnRH Test at 4–6 Months of Treatment
4.7. Assay Procedures
4.8. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Franks, S. Polycystic ovary syndrome. N. Engl. J. Med. 1995, 333, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Franks, S.; McCarthy, M.; Hardy, K. Development of polycystic ovary syndrome: Involvement of genetic and environmental factors. Int. J. Androl. 2006, 29, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum. Reprod. 2004, 19, 41–47. [Google Scholar] [CrossRef]
- Fauser, B.C.; Tarlatzis, B.C.; Rebar, R.W.; Legro, R.; Balen, A.H.; Lobo, R.; Carmina, E.; Chang, J.; Yildiz, B.O.; Laven, J.S.; et al. Consensus on women’s health aspects of polycystic ovary syndrome (PCOS): The Amsterdam ESHRE/ASRM-Sponsored 3rd PCOS Consensus Workshop Group. Fertil. Steril. 2012, 97, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Teede, H.J.; Misso, M.L.; Costello, M.F.; Dokras, A.; Laven, J.; Moran, L.; Piltonen, T.; Norman, R.J.; International PCOS Network. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Hum. Reprod. 2018, 33, 1602–1618. [Google Scholar] [CrossRef] [PubMed]
- Burghen, G.A.; Givens, J.R.; Kitabchi, A.E. Correlation of hyperandrogenism with hyperinsulinism in polycystic ovarian disease. J. Clin. Endocrinol. Metab. 1980, 50, 113–116. [Google Scholar] [CrossRef] [PubMed]
- Dunaif, A.; Graf, M.; Mandeli, J.; Laumas, V.; Drobrjansky, A. Characterization of groups of hyperandrogenic women with acanthosis nigricans, impaired glucose tolerance and/or hyperinsulinemia. J. Clin. Endocrinol. Metab. 1987, 65, 499–507. [Google Scholar] [CrossRef]
- Vrbíková, J.; Cibula, D.; Dvoráková, K.; Stanická, S.; Sindelka, G.; Hill, M.; Fanta, M.; Vondra, K.; Skrha, J. Insulin sensitivity in women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2004, 89, 2942–2945. [Google Scholar] [CrossRef]
- Dumesic, D.A.; Phan, J.D.; Leung, K.L.; Grogan, T.R.; Ding, X.; Li, X.; Hoyos, L.R.; Abbott, D.H.; Chazenbalk, G.D. Adipose Insulin Resistance in Normal-Weight Women with Polycystic Ovary Syndrome. J. Clin. Endocrinol. Metab. 2019, 104, 2171–2183. [Google Scholar] [CrossRef]
- Pasquali, R.; Venturoli, S.; Paradis, R.; Capelli, M.; Parenti, M.; Melchionda, N. Insulin and C-Peptide Levels in Obese Patients with Polycystic Ovaries. Horm. Metab. Res. 1982, 14, 284–287. [Google Scholar] [CrossRef]
- Shoupe, D.; Kumar, D.D.; Lobo, R.A. Insulin resistance in polycystic ovary syndrome. Am. J. Obstet. Gynecol. 1983, 147, 588–592. [Google Scholar] [CrossRef]
- Chang, R.J.; Nakamura, R.M.; Judd, H.L.; Kaplan, S.A. Insulin Resistance in Nonobese Patients with Polycystic Ovarian Disease. J. Clin. Endocrinol. Metab. 1983, 57, 356–359. [Google Scholar] [CrossRef] [PubMed]
- Ezeh, U.; Pisarska, M.D.; Azziz, R. Association of severity of menstrual dysfunction with hyperinsulinemia and dysglycemia in polycystic ovary syndrome. Hum. Reprod. 2022, deac001. [Google Scholar] [CrossRef]
- Diamanti-Kandarakis, E.; Dunaif, A. Insulin Resistance and the Polycystic Ovary Syndrome Revisited: An Update on Mechanisms and Implications. Endocr. Rev. 2012, 33, 981–1030. [Google Scholar] [CrossRef] [PubMed]
- Teede, H.J.; Joham, A.E.; Paul, E.; Moran, L.; Loxton, D.; Jolley, D.; Lombard, C. Longitudinal weight gain in women identified with polycystic ovary syndrome: Results of an observational study in young women. Obesity 2013, 21, 1526–1532. [Google Scholar] [CrossRef] [PubMed]
- Rosenfield, R.L.; Ehrmann, D.A. The Pathogenesis of Polycystic Ovary Syndrome (PCOS): The Hypothesis of PCOS as Functional Ovarian Hyperandrogenism Revisited. Endocr. Rev. 2016, 37, 467–520. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Xu, Y.; Wang, X.; Xu, L.; Chen, J.; Gao, C.; Wu, C.; Pan, D.; Zhang, Q.; Zhou, J.; et al. Body Mass Index and Polycystic Ovary Syndrome: A 2-Sample Bidirectional Mendelian Randomization Study. J. Clin. Endocrinol. Metab. 2020, 105, 1778–1784. [Google Scholar] [CrossRef] [PubMed]
- Dapas, M.; Lin, F.T.J.; Nadkarni, G.N.; Sisk, R.; Legro, R.S.; Urbanek, M.; Hayes, M.G.; Dunaif, A. Distinct subtypes of polycystic ovary syndrome with novel genetic associations: An unsupervised, phenotypic clustering analysis. PLoS Med. 2020, 17, e1003132. [Google Scholar] [CrossRef] [PubMed]
- Rice, S.; Christoforidis, N.; Gadd, C.; Nikolaou, D.; Seyani, L.; Donaldson, A.; Margara, R.; Hardy, K.; Franks, S. Impaired insulin-dependent glucose metabolism in granulosa-lutein cells from anovulatory women with polycystic ovaries. Hum. Reprod. 2005, 20, 373–381. [Google Scholar] [CrossRef]
- Ehrman, D.A.; Barnes, R.B.; Rosenfield, R.L. Polycystic Ovary Syndrome as a Form of FunctionalOvarian Hyperandrogenism Due to Dysregulation ofAndrogen Secretion. Endocr. Rev. 1995, 16, 322–353. [Google Scholar] [CrossRef]
- Baillargeon, J.P.; Nestler, J.E. Commentary: Polycystic ovary syndrome: A syndrome of ovarian hypersensitivity to insulin? J. Clin. Endocrinol. Metab. 2006, 91, 22–24. [Google Scholar] [CrossRef]
- Poretsky, L.; Cataldo, N.A.; Rosenwaks, Z.; Giudice, L.C. The insulin-related ovarian regulatory system in health and disease. Endocr. Rev. 1999, 20, 535–582. [Google Scholar] [CrossRef]
- Franks, S.; Hardy, K. Androgen Action in the Ovary. Front. Endocrinol. 2018, 9, 452. [Google Scholar] [CrossRef]
- Nestler, J.E.; Jakubowicz, D.J.; De Vargas, A.F.; Brik, C.; Quintero, N.; Medina, F. Insulin Stimulates Testosterone Biosynthesis by Human Thecal Cells from Women with Polycystic Ovary Syndrome by Activating Its Own Receptor and Using Inositolglycan Mediators as the Signal Transduction System. J. Clin. Endocrinol. Metab. 1998, 83, 2001–2005. [Google Scholar] [CrossRef]
- Kiddy, D.S.; Hamilton-Fairley, D.; Bush, A.; Short, F.; Anyaoku, V.; Reed, M.J.; Franks, S. Improvement in endocrine and ovarian function during dietary treatment of obese women with polycystic ovary syndrome. Clin. Endocrinol. 1992, 36, 105–111. [Google Scholar] [CrossRef]
- Holte, J.; Bergh, T.; Berne, C.; Wide, L.; Lithell, H. Restored insulin sensitivity but persistently increased early insulin secretion after weight loss in obese women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 1995, 80, 2586–2593. [Google Scholar] [CrossRef]
- Huber-Buchholz, M.-M.; Carey, D.G.P.; Norman, R.J. Restoration of Reproductive Potential by Lifestyle Modification in Obese Polycystic Ovary Syndrome: Role of Insulin Sensitivity and Luteinizing Hormone. J. Clin. Endocrinol. Metab. 1999, 84, 1470–1474. [Google Scholar] [CrossRef]
- Lee, R.; Mathew, C.J.; Jose, M.T.; Elshaikh, A.O.; Shah, L.; Cancarevic, I. A Review of the Impact of Bariatric Surgery in Women With Polycystic Ovary Syndrome. Cureus 2020, 12, e10811. [Google Scholar] [CrossRef]
- Dunaif, A.; Scott, D.; Finegood, D.; Quintana, B.; Whitcomb, R. The insulin-sensitizing agent troglitazone improves metabolic and reproductive abnormalities in the polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 1996, 81, 3299–3306. [Google Scholar] [CrossRef][Green Version]
- Ehrmann, D.A.; Schneider, D.J.; Sobel, B.E.; Cavaghan, M.K.; Imperial, J.; Rosenfield, R.L.; Polonsky, K.S. Troglitazone Improves Defects in Insulin Action, Insulin Secretion, Ovarian Steroidogenesis, and Fibrinolysis in Women with Polycystic Ovary Syndrome. J. Clin. Endocrinol. Metab. 1997, 82, 2108–2116. [Google Scholar] [CrossRef]
- Lord, J.M.; Flight, I.; Norman, R. Metformin in polycystic ovary syndrome: Systematic review and meta-analysis. BMJ 2003, 327, 951–953. [Google Scholar] [CrossRef] [PubMed]
- Siamashvili, M.; Davis, S.N. Update on the effects of GLP-1 receptor agonists for the treatment of polycystic ovary syndrome. Expert Rev. Clin. Pharmacol. 2021, 14, 1081–1089. [Google Scholar] [CrossRef] [PubMed]
- Blank, S.K.; McCartney, C.R.; Helm, K.D.; Marshall, J.C. Neuroendocrine Effects of Androgens in Adult Polycystic Ovary Syndrome and Female Puberty. Semin. Reprod. Med. 2007, 25, 352–359. [Google Scholar] [CrossRef] [PubMed]
- Paradisi, R.; Fabbri, R.; Battaglia, C.; Venturoli, S. Ovulatory effects of flutamide in the polycystic ovary syndrome. Gynecol. Endocrinol. 2013, 29, 391–395. [Google Scholar] [CrossRef]
- Barber, T.M.; Hanson, P.; Weickert, M.O.; Franks, S. Obesity and Polycystic Ovary Syndrome: Implications for Pathogenesis and Novel Management Strategies. Clin. Med. Insights Reprod. Health 2019, 13, 1179558119874042. [Google Scholar] [CrossRef]
- Duggal, P.S.; Van der Hoek, K.; Milner, C.R.; Ryan, N.K.; Armstrong, D.T.; Magoffin, D.A.; Norman, R.J. The in Vivo and in Vitro Effects of Exogenous Leptin on Ovulation in the Rat. Endocrinology 2000, 141, 1971–1976. [Google Scholar] [CrossRef]
- De Medeiros, S.F.; Rodgers, R.J.; Norman, R.J. Adipocyte and steroidogenic cell cross-talk in polycystic ovary syndrome. Hum. Reprod. Update 2021, 27, 771–796. [Google Scholar] [CrossRef]
- Mitchell, M.; Armstrong, D.T.; Robker, R.L.; Norman, R.J. Adipokines: Implications for female fertility and obesity. Reproduction 2005, 130, 583–597. [Google Scholar] [CrossRef]
- Thong, E.P.; Codner, E.; Laven, J.S.E.; Teede, H. Diabetes: A metabolic and reproductive disorder in women. Lancet Diabetes Endocrinol. 2019, 8, 134–149. [Google Scholar] [CrossRef]
- Soldat-Stanković, V.; Popović-Pejičić, S.; Stanković, S.; Prtina, A.; Malešević, G.; Bjekić-Macut, J.; Livadas, S.; Ognjanović, S.; Mastorakos, G.; Micić, D.; et al. The effect of metformin and myoinositol on metabolic outcomes in women with polycystic ovary syndrome: Role of body mass and adiponectin in a randomized controlled trial. J. Endocrinol. Investig. 2021, 45, 583–595. [Google Scholar] [CrossRef]
- Dupont, J.; Scaramuzzi, R.J. Insulin signalling and glucose transport in the ovary and ovarian function during the ovarian cycle. Biochem. J. 2016, 473, 1483–1501. [Google Scholar] [CrossRef] [PubMed]
- Unfer, V.; DiNicola, S.; Laganà, A.S.; Bizzarri, M. Altered Ovarian Inositol Ratios May Account for Pathological Steroidogenesis in PCOS. Int. J. Mol. Sci. 2020, 21, 7157. [Google Scholar] [CrossRef]
- Laganà, A.S.; Rossetti, P.; Buscema, M.; La Vignera, S.; Condorelli, R.A.; Gullo, G.; Granese, R.; Triolo, O. Metabolism and Ovarian Function in PCOS Women: A Therapeutic Approach with Inositols. Int. J. Endocrinol. 2016, 2016, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Paul, C.; Laganà, A.S.; Maniglio, P.; Triolo, O.; Brady, D.M. Inositol’s and other nutraceuticals’ synergistic actions counteract insulin resistance in polycystic ovarian syndrome and metabolic syndrome: State-of-the-art and future perspectives. Gynecol. Endocrinol. 2016, 32, 431–438. [Google Scholar] [CrossRef]
- Sacchi, S.; Marinaro, F.; Tondelli, D.; Lui, J.; Xella, S.; Marsella, T.; Tagliasacchi, D.; Argento, C.; Tirelli, A.; Giulini, S.; et al. Modulation of gonadotrophin induced steroidogenic enzymes in granulosa cells by d-chiroinositol. Reprod. Biol. Endocrinol. 2016, 14, 52. [Google Scholar] [CrossRef] [PubMed]
- Seto-Young, D.; Paliou, M.; Schlosser, J.; Avtanski, D.; Park, A.; Patel, P.; Holcomb, K.; Chang, P.; Poretsky, L. Direct Thiazolidinedione Action in the Human Ovary: Insulin-Independent and Insulin-Sensitizing Effects on Steroidogenesis and Insulin-Like Growth Factor Binding Protein-1 Production. J. Clin. Endocrinol. Metab. 2005, 90, 6099–6105. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhao, H.; Xing, C.; Zhang, J.; He, B.; Zhao, H.; Xing, C.; Zhang, J.; He, B. Comparative efficacy of oral insulin sensitizers metformin, thiazolidinediones, inositol, and berberine in improving endocrine and metabolic profiles in women with PCOS: A network meta-analysis. Reprod. Health 2021, 18, 171. [Google Scholar] [CrossRef] [PubMed]
- Hostalek, U.; Campbell, I. Metformin for diabetes prevention: Update of the evidence base. Curr. Med. Res. Opin. 2021, 37, 1705–1717. [Google Scholar] [CrossRef]
- Mansfield, R.; Galea, R.; Brincat, M.; Hole, D.; Mason, H. Metformin has direct effects on human ovarian steroidogenesis. Fertil. Steril. 2003, 79, 956–962. [Google Scholar] [CrossRef]
- Hirsch, A.; Hahn, D.; Kempná, P.; Hofer, G.; Nuoffer, J.-M.; Mullis, P.E.; Flück, C.E. Metformin Inhibits Human Androgen Production by Regulating Steroidogenic Enzymes HSD3B2 and CYP17A1 and Complex I Activity of the Respiratory Chain. Endocrinology 2012, 153, 4354–4366. [Google Scholar] [CrossRef]
- Attia, G.R.; Rainey, W.E.; Carr, B.R. Metformin directly inhibits androgen production in human thecal cells. Fertil. Steril. 2001, 76, 517–524. [Google Scholar] [CrossRef]
- Dumesic, D.A.; Oberfield, S.E.; Stener-Victorin, E.; Marshall, J.C.; Laven, J.S.; Legro, R.S. Scientific Statement on the Diagnostic Criteria, Epidemiology, Pathophysiology, and Molecular Genetics of Polycystic Ovary Syndrome. Endocr. Rev. 2015, 36, 487–525. [Google Scholar] [CrossRef]
- Legro, R.S. Ovulation induction in polycystic ovary syndrome: Current options. Best Prac. Res. Clin. Obstet. Gynaecol. 2016, 37, 152–159. [Google Scholar] [CrossRef]
- Kim, C.; Miller, R.; Braffett, B.; Pan, Y.; Arends, V.; Saenger, A.; Barnie, A.; Sarma, A. The EDIC Research Group Ovarian markers and irregular menses among women with type 1 diabetes in the Epidemiology of Diabetes Interventions and Complications study. Clin. Endocrinol. 2018, 88, 453–459. [Google Scholar] [CrossRef]
- Fonseca, V.A.; Haggar, M.A. Achieving glycaemic targets with basal insulin in T2DM by individualizing treatment. Nat. Rev. Endocrinol. 2014, 10, 276–281. [Google Scholar] [CrossRef]
- Escobar-Morreale, H.F.; Bayona, A.; Nattero-Chávez, L.; Luque-Ramírez, M. Type 1 diabetes mellitus and polycystic ovary syndrome. Nat. Rev. Endocrinol. 2021, 17, 701–702. [Google Scholar] [CrossRef]
- Ovalle, F.; Azziz, R. Insulin resistance, polycystic ovary syndrome, and type 2 diabetes mellitus. Fertil. Steril. 2002, 77, 1095–1105. [Google Scholar] [CrossRef]
- Escobar-Morreale, H.F.; Roldán-Martín, M.B. Type 1 Diabetes and Polycystic Ovary Syndrome: Systematic Review and Meta-analysis. Diabetes Care 2016, 39, 639–648. [Google Scholar] [CrossRef]
- Escobar-Morreale, H.F.; Roldán, B.; Barrio, R.; Alonso, M.; Sancho, J.; De La Calle, H.; García-Robles, R. High Prevalence of the Polycystic Ovary Syndrome and Hirsutism in Women with Type 1 Diabetes Mellitus. J. Clin. Endocrinol. Metab. 2000, 85, 4182–4187. [Google Scholar] [CrossRef] [PubMed]
- Roldán, B.; Escobar-Morreale, H.F.; Barrio, R.; de la Calle, H.; Alonso, M.; García-Robles, R.; Sancho, J. Identification of the Source of Androgen Excess in Hyperandrogenic Type 1 Diabetic Patients. Diabetes Care 2001, 24, 1297–1299. [Google Scholar] [CrossRef]
- Abbott, D.H.; Dumesic, D.A.; Eisner, J.R.; Colman, R.J.; Kemnitz, J.W. Insights into the development of polycystic ovary syndrome (PCOS) from studies of prenatally androgenized female rhesus monkeys. Trends Endocrinol. Metab. 1998, 9, 62–67. [Google Scholar] [CrossRef]
- Dumesic, D.A.; Abbott, D.H.; Eisner, J.R.; Goy, R.W. Prenatal exposure of female rhesus monkeys to testosterone propionate increases serum luteinizing hormone levels in adulthood. Fertil. Steril. 1997, 67, 155–163. [Google Scholar] [CrossRef]
- Abbott, D.H.; Barnett, D.K.; Bruns, C.M.; Dumesic, D.A. Androgen excess fetal programming of female reproduction: A developmental aetiology for polycystic ovary syndrome? Hum. Reprod. Update 2005, 11, 357–374. [Google Scholar] [CrossRef] [PubMed]
- Abbott, D.H.; Rogers, J.; Dumesic, D.A.; Levine, J.E. Naturally Occurring and Experimentally Induced Rhesus Macaque Models for Polycystic Ovary Syndrome: Translational Gateways to Clinical Application. Med. Sci. 2019, 7, 107. [Google Scholar] [CrossRef]
- Abbott, D.H.; Dumesic, D.A.; Franks, S. Developmental origin of polycystic ovary syndrome—A hypothesis. J. Endocrinol. 2002, 174, 1–5. [Google Scholar] [CrossRef]
- Stener-Victorin, E.; Padmanabhan, V.; Walters, K.A.; Campbell, R.E.; Benrick, A.; Giacobini, P.; Dumesic, D.A.; Abbott, D.H. Animal Models to Understand the Etiology and Pathophysiology of Polycystic Ovary Syndrome. Endocr. Rev. 2020, 41, bnaa010. [Google Scholar] [CrossRef]
- Thomas, F.T.; Ricordi, C.; Contreras, J.L.; Hubbard, W.J.; Jiang, X.L.; Eckhoff, D.E.; Cartner, S.; Bilbao, G.; Neville, D.M.; Thomas, J.M. Reversal of Naturally Occurring Diabetes in Primates by Unmodified Islet Xenografts without Chronic Immunosuppression. Transplantation 1999, 67, 846–854. [Google Scholar] [CrossRef]
- Contreras, J.L.; Eckhoff, D.E.; Cartner, S.; Bilbao, G.; Ricordi, C.; Neville, D.M.; Thomas, F.T.; Thomas, J.M. Long-Term Functional Islet Mass and Metabolic Function after Xenoislet Transplantation in Primates 1. Transplantation 2000, 69, 195–201. [Google Scholar] [CrossRef]
- Bruns, C.M.; Kemnitz, J.W. Sex hormones, insulin sensitivity, and diabetes mellitus. ILAR J. 2004, 45, 160–169. [Google Scholar] [CrossRef]
- Abbott, D.; Rayome, B.; Dumesic, D.; Lewis, K.; Edwards, A.; Wallen, K.; Wilson, M.; Appt, S.; Levine, J. Clustering of PCOS-like traits in naturally hyperandrogenic female rhesus monkeys. Hum. Reprod. 2017, 32, 923–936. [Google Scholar] [CrossRef]
- Puglisi, S.C.; Mackiewicz, A.L.; Ardeshir, A.; Garzel, L.M.; Christe, K.L. Comparison of Insulins Glargine and Degludec in Diabetic Rhesus Macaques (Macaca mulatta) with CGM Devices. Comp. Med. 2021, 71, 247–255. [Google Scholar] [CrossRef]
- Qiao, C.; Tian, B.; Mai, G.; Wei, L.; Jin, X.; Ren, Y.; Chen, Y.; Li, H.; Li, Y.; Wang, L.; et al. Induction of Diabetes in Rhesus Monkeys and Establishment of Insulin Administration Strategy. Transplant. Proc. 2009, 41, 413–417. [Google Scholar] [CrossRef]
- Kemnitz, J.W. Data Collected as Part of Aging Studies of Rhesus Macaques at the Wisconsin National Primate Research Center, 1989–2020; University of Wisconsin: Madison, WI, USA, unpublishedwork.
- Martens, T.; Beck, R.W.; Bailey, R.; Ruedy, K.J.; Calhoun, P.; Peters, A.L.; Pop-Busui, R.; Philis-Tsimikas, A.; Bao, S.; Umpierrez, G.; et al. MOBILE Study Group. Effect of Continuous Glucose Monitoring on Glycemic Control in Patients with Type 2 Diabetes Treated With Basal Insulin: A Randomized Clinical Trial. JAMA 2021, 325, 2262–2272. [Google Scholar]
- Elting, M.W.; Kwee, J.; Schats, R.; Rekers-Mombarg, L.T.M.; Schoemaker, J. The Rise of Estradiol and Inhibin B after Acute Stimulation with Follicle-Stimulating Hormone Predict the Follicle Cohort Size in Women with Polycystic Ovary Syndrome, Regularly Menstruating Women with Polycystic Ovaries, and Regularly Menstruating Women with Normal Ovaries. J. Clin. Endocrinol. Metab. 2001, 86, 1589–1595. [Google Scholar] [CrossRef]
- Adams, J.M.; Taylor, A.E.; Crowley, W.F.; Hall, J.E. Polycystic Ovarian Morphology with Regular Ovulatory Cycles: Insights into the Pathophysiology of Polycystic Ovarian Syndrome. J. Clin. Endocrinol. Metab. 2004, 89, 4343–4350. [Google Scholar] [CrossRef]
- Kemnitz, J.W.; Colman, R.J. Data Collected as Part of Aging Studies of Rhesus Macaques at the Wisconsin National Primate Research Center, 1989–2022; University of Wisconsin: Madison, WI, USA, unpublished work.
- Bergman, R.N. Lilly lecture Toward physiological understanding of glucose tolerance: Minimal-model approach. Diabetes 1989, 38, 1512–1527. [Google Scholar] [CrossRef]
- Eisner, J.R.; Dumesic, D.A.; Kemnitz, J.W.; Abbott, D.H. Timing of prenatal androgen excess determines differential impairment in insulin secretion and action in adult female rhesus monkeys. J. Clin. Endocrinol. Metab. 2000, 85, 1206–1210. [Google Scholar] [CrossRef]
- Nicol, L.E.; O’Brien, T.; Dumesic, D.A.; Grogan, T.; Tarantal, A.F.; Abbott, D.H. Abnormal Infant Islet Morphology Precedes Insulin Resistance in PCOS-Like Monkeys. PLoS ONE 2014, 9, e106527. [Google Scholar] [CrossRef]
- Hudson, J.C.; Baum, S.T.; Frye, D.M.D.; Roecker, E.B.; Kemnitz, J.W. Age and sex differences in body size and composition during Rhesus monkey adulthood. Aging Clin. Exp. Res. 1996, 8, 197–204. [Google Scholar] [CrossRef]
- Phy, J.L.; Conover, C.A.; Abbott, D.H.; Zschunke, M.A.; Walker, D.L.; Session, D.R.; Tummon, I.S.; Thornhill, A.R.; Lesnick, T.G.; Dumesic, D.A. Insulin and Messenger Ribonucleic Acid Expression of Insulin Receptor Isoforms in Ovarian Follicles from Nonhirsute Ovulatory Women and Polycystic Ovary Syndrome Patients. J. Clin. Endocrinol. Metab. 2004, 89, 3561–3566. [Google Scholar] [CrossRef][Green Version]
- Munir, I.; Yen, H.W.; Geller, D.H.; Torbati, D.; Bierden, R.M.; Weitsman, S.R.; Agarwal, S.K.; Magoffin, D.A. Insulin augmentation of 17alpha-hydroxylase activity is mediated by phosphatidyl inositol 3-kinase but not extracellular signal-regulated kinase-1/2 in human ovarian theca cells. Endocrinology 2004, 145, 175–183. [Google Scholar] [CrossRef]
- Willis, D.; Mason, H.; Gilling-Smith, C.; Franks, S. Modulation by insulin of follicle-stimulating hormone and luteinizing hormone actions in human granulosa cells of normal and polycystic ovaries. J. Clin. Endocrinol. Metab. 1996, 81, 302–309. [Google Scholar] [CrossRef][Green Version]
- Dumesic, D.A. Data Collected as Part of Infertility Studies of Female Rhesus Macaques at the Wisconsin National Primate Research Center, 1997–2004; University of California: Los Angeles, CA, USA, unpublished work.
- Zhou, R.; Bruns, C.M.; Bird, I.M.; Kemnitz, J.W.; Goodfriend, T.L.; Dumesic, D.A.; Abbott, D.H. Pioglitazone improves insulin action and normalizes menstrual cycles in a majority of prenatally androgenized female rhesus monkeys. Reprod. Toxicol. 2007, 23, 438–448. [Google Scholar] [CrossRef][Green Version]
- Dumesic, D.A.; Padmanabhan, V.; Chazenbalk, G.D.; Abbott, D.H. Polycystic ovary syndrome as a plausible evolutionary outcome of metabolic adaptation. Reprod. Biol. Endocrinol. 2022, 20, 12. [Google Scholar] [CrossRef]
- Nestler, J.E.; Clore, J.N.; Strauss, J.F., 3rd; Blackard, W.G. The effects of hyperinsulinemia on serum testosterone, progesterone, dehydroepiandrosterone sulfate, and cortisol levels in normal women and in a woman with hyperandrogenism, insulin resistance, and acanthosis nigricans. J. Clin. Endocrinol. Metab. 1987, 64, 180–184. [Google Scholar] [CrossRef]
- Dunaif, A.; Graf, M. Insulin administration alters gonadal steroid metabolism independent of changes in gonadotropin secretion in insulin-resistant women with the polycystic ovary syndrome. J. Clin. Investig. 1989, 83, 23–29. [Google Scholar] [CrossRef]
- Poretsky, L.; Glover, B.; Laumas, V.; Kalin, M.; Dunaif, A. The Effects of Experimental Hyperinsulinemia on Steroid Secretion, Ovarian [125I]Insulin Binding, and Ovarian [125I]Insulin-Like Growth-Factor I Binding in the Rat. Endocrinology 1988, 122, 581–585. [Google Scholar] [CrossRef]
- Stuart, C.A.; Prince, M.J.; Peters, E.J.; Meyer, W.J., 3rd. Hyperinsulinemia and hyperandrogenemia: In vivo androgen response to insulin infusion. Obstet. Gynecol. 1987, 69, 921–925. [Google Scholar]
- Poretsky, L.; Kalin, M.F. The Gonadotropic Function of Insulin. Endocr. Rev. 1987, 8, 132–141. [Google Scholar] [CrossRef]
- Adashi, E.Y.; Resnick, C.E.; D’Ercole, A.J.; Svoboda, M.E.; Van Wyk, J.J. Insulin-Like Growth Factors as Intraovarian Regulators of Granulosa Cell Growth and Function. Endocr. Rev. 1985, 6, 400–420. [Google Scholar] [CrossRef]
- Adams, T.E.; Epa, V.C.; Garrett, T.P.; Ward, C.W. Structure and function of the type 1 insulin-like growth factor receptor. Cell. Mol. Life Sci. 2000, 57, 1050–1093. [Google Scholar] [CrossRef] [PubMed]
- Nakae, J.; Kido, Y.; Accili, D. Distinct and overlapping functions of insulin and IGF-I receptors. Endocr. Rev. 2001, 22, 818–835. [Google Scholar] [CrossRef] [PubMed]
- Siddle, K.; Soos, M.A.; Field, C.E.; Navé, B.T. Hybrid and atypical insulin/insulin-like growth factor I receptors. Horm. Res. 1994, 41 (Suppl. S2), 56–64. [Google Scholar] [CrossRef] [PubMed]
- Pessin, J.E.; Frattali, A.L. Molecular dynamics of insulin/IGF-I receptor transmembrane signaling. Mol. Reprod. Dev. 1993, 35, 339–345. [Google Scholar] [CrossRef]
- Vendola, K.; Zhou, J.; Wang, J.; Famuyiwa, O.A.; Bievre, M.; Bondy, C.A. Androgens Promote Oocyte Insulin-Like Growth Factor I Expression and Initiation of Follicle Development in the Primate Ovary. Biol. Reprod. 1999, 61, 353–357. [Google Scholar] [CrossRef]
- Martinez-Chequer, J.; Stouffer, R.; Hazzard, T.; Patton, P.; Molskness, T. Insulin-Like Growth Factors-1 and -2, but not Hypoxia, Synergize with Gonadotropin Hormone to Promote Vascular Endothelial Growth Factor-A Secretion by Monkey Granulosa Cells from Preovulatory Follicles. Biol. Reprod. 2003, 68, 1112–1118. [Google Scholar] [CrossRef]
- Zeleznik, A.J.; Little-Ihrig, L.; Ramasawamy, S. Administration of Insulin-Like Growth Factor I to Rhesus Monkeys Does Not Augment Gonadotropin-Stimulated Ovarian Steroidogenesis. J. Clin. Endocrinol. Metab. 2002, 87, 5722–5729. [Google Scholar] [CrossRef]
- Wilson, M.E. Premature elevation in serum insulin-like growth factor-I advances first ovulation in rhesus monkeys. J. Endocrinol. 1998, 158, 247–257. [Google Scholar] [CrossRef]
- Wilson, M.E. IGF-I administration advances the decrease in hypersensitivity to oestradiol negative feedback inhibition of serum LH in adolescent female rhesus monkeys. J. Endocrinol. 1995, 145, 121–130. [Google Scholar] [CrossRef]
- Steiner, R.A.; Clifton, D.K.; Spies, H.G.; Resko, J.A. Sexual Differentiation and Feedback Control of Luteinizing Hormone Secretion in the Rhesus Monkey. Biol. Reprod. 1976, 15, 206–212. [Google Scholar] [CrossRef]
- Klinger, B.; Anin, S.; Silbergeld, A.; Eshet, R.; Laron, Z. Development of hyperandrogenism during treatment with insulin-like growth factor-I (IGF-I) in female patients with Laron syndrome. Clin. Endocrinol. 1998, 48, 81–87. [Google Scholar] [CrossRef]
- Codner, E.; Iñíguez, G.; Villarroel, C.; Lopez, P.; Soto, N.; Sir-Petermann, T.; Cassorla, F.; Rey, R.A. Hormonal Profile in Women with Polycystic Ovarian Syndrome with or without Type 1 Diabetes Mellitus. J. Clin. Endocrinol. Metab. 2007, 92, 4742–4746. [Google Scholar] [CrossRef]
- Franks, S.; Mason, H.; White, D.; Willis, D. Etiology of Anovulation in Polycystic Ovary Syndrome. Steroids 1998, 63, 306–307. [Google Scholar] [CrossRef]
- Coffler, M.S.; Patel, K.; Dahan, M.; Yoo, R.Y.; Malcom, P.J.; Chang, R.J. Enhanced Granulosa Cell Responsiveness to Follicle-Stimulating Hormone during Insulin Infusion in Women with Polycystic Ovary Syndrome Treated with Pioglitazone. J. Clin. Endocrinol. Metab. 2003, 88, 5624–5631. [Google Scholar] [CrossRef]
- Dahlgren, E.; Johansson, S.; Lindstedt, G.; Knutsson, F.; Odén, A.; Janson, P.O.; Mattson, L.; Crona, N.; Lundberg, P.-A. Women with polycystic ovary syndrome wedge resected in 1956 to 1965: A long-term follow-up focusing on natural history and circulating hormones. Fertil. Steril. 1992, 57, 505–513. [Google Scholar] [CrossRef]
- Poretsky, L.; Clemons, J.; Bogovich, K. Hyperinsulinemia and human chorionic gonadotropin synergistically promote the growth of ovarian follicular cysts in rats. Metabolism 1992, 41, 903–910. [Google Scholar] [CrossRef]
- Weng, J.; Li, Y.; Xu, W.; Shi, L.; Zhang, Q.; Zhu, D.; Hu, Y.; Zhou, Z.; Yan, X.; Tian, H.; et al. Effect of intensive insulin therapy on beta-cell function and glycaemic control in patients with newly diagnosed type 2 diabetes: A multicentre randomized parallel-group trial. Lancet 2008, 371, 1753–1760. [Google Scholar] [CrossRef]
- Retnakaran, R.; Drucker, D.J. Intensive insulin therapy in newly diagnosed type 2 diabetes. Lancet 2008, 371, 1725–1726. [Google Scholar] [CrossRef]
- Barbieri, R.L.; Makris, A.; Randall, R.W.; Daniels, G.; Kistner, R.W.; Ryan, K.J. Insulin Stimulates Androgen Accumulation in Incubations of Ovarian Stroma Obtained from Women with Hyperandrogenism. J. Clin. Endocrinol. Metab. 1986, 62, 904–910. [Google Scholar] [CrossRef]
- De Leo, V.; la Marca, A.; Petraglia, F. Insulin-lowering agents in the management of polycystic ovary syndrome. Endocr. Rev. 2003, 24, 633–667. [Google Scholar] [CrossRef]
- Brettenthaler, N.; De Geyter, C.; Huber, P.R.; Keller, U. Effect of the Insulin Sensitizer Pioglitazone on Insulin Resistance, Hyperandrogenism, and Ovulatory Dysfunction in Women with Polycystic Ovary Syndrome. J. Clin. Endocrinol. Metab. 2004, 89, 3835–3840. [Google Scholar] [CrossRef] [PubMed]
- Nelson, V.L.; Legro, R.S.; Strauss, J.F., 3rd; McAllister, J.M. Augmented androgen production is a stable steroidogenic phenotype of propagated theca cells from polycystic ovaries. Mol. Endocrinol. 1999, 13, 946–957. [Google Scholar] [CrossRef] [PubMed]
- Willis, D.; Franks, S. Insulin action in human granulosa cells from normal and polycystic ovaries is mediated by the insulin receptor and not the type-I insulin-like growth factor receptor. J. Clin. Endocrinol. Metab. 1995, 80, 3788–3790. [Google Scholar] [CrossRef] [PubMed]
- Abbott, D.H.; Tarantal, A.F.; Dumesic, D.A. Fetal, infant, adolescent and adult phenotypes of polycystic ovary syndrome in prenatally androgenized female rhesus monkeys. Am. J. Primatol. 2009, 71, 776–784. [Google Scholar] [CrossRef]
- Abbott, D.H.; Vepraskas, S.H.; Horton, T.H.; Terasawa, E.; Levine, J.E. Accelerated Episodic Luteinizing Hormone Release Accompanies Blunted Progesterone Regulation in PCOS-like Female Rhesus Monkeys (Macaca Mulatta) Exposed to Testosterone during Early-to-Mid Gestation. Neuroendocrinology 2018, 107, 133–146. [Google Scholar] [CrossRef]
- Goy, R.W.; Robinson, J.A. Prenatal exposure of rhesus monkeys to patent androgens: Morphological, behavioral, and physiological consequences. In Banbury Report II: Environmental Factors in Human Growth and Development; Hunt, V.R., Smith, M.K., Worth, D., Eds.; Cold Spring Harbor Laboratory: Cold Spring Harbor, NY, USA, 1982; pp. 355–378. [Google Scholar]
- Kemnitz, J.W.; Elson, D.F.; Roecker, E.B.; Baum, S.T.; Bergman, R.N.; Meglasson, M.D. Pioglitazone increases insulin sensitivity, reduces blood glucose, insulin, and lipid levels, and lowers blood pressure, in obese, insulin-resistant rhesus monkeys. Diabetes 1994, 43, 204–211. [Google Scholar] [CrossRef]
- Cheung, B.W.; Cartier, L.L.; Russlie, H.Q.; Sawchuk, R.J. The application of sample pooling methods for determining AUC, AUMC and mean residence times in pharmacokinetic studies. Fundam. Clin. Pharmacol. 2005, 19, 347–354. [Google Scholar] [CrossRef]
- Ibanez, L.; Hall, J.E.; Potau, N.; Carrascosa, A.; Prat, N.; E Taylor, A. Ovarian 17-hydroxyprogesterone hyperresponsiveness to gonadotropin-releasing hormone (GnRH) agonist challenge in women with polycystic ovary syndrome is not mediated by luteinizing hormone hypersecretion: Evidence from GnRH agonist and human chorionic gonadotropin stimulation testing. J. Clin. Endocrinol. Metab. 1996, 81, 4103–4107. [Google Scholar] [CrossRef]
- Eisner, J.R.; Barnett, M.A.; Dumesic, D.A.; Abbott, D.H. Ovarian hyperandrogenism in adult female rhesus monkeys exposed to prenatal androgen excess. Fertil. Steril. 2002, 77, 167–172. [Google Scholar] [CrossRef]
- Ferin, M.; Warren, M.; Dyrenfurth, I.; Wiele, R.L.V.; White, W.F. Response of Rhesus Monkeys to LRH Throughout the Ovarian Cycle. J. Clin. Endocrinol. Metab. 1974, 38, 231–237. [Google Scholar] [CrossRef][Green Version]
- Dumesic, D.A.; Schramm, R.D.; Peterson, E.; Paprocki, A.M.; Zhou, R.; Abbott, D.H. Impaired developmental competence of oocytes in adult prenatally androgenized female rhesus monkeys undergoing gonadotropin stimulation for in vitro fertilization. J. Clin. Endocrinol Metab. 2002, 87, 1111–1119. [Google Scholar] [CrossRef][Green Version]
- Saltzman, W.; Schultz-Darken, N.J.; Scheffler, G.; Wegner, F.H.; Abbott, D.H. Social and reproductive influences on plasma cortisol in female marmoset monkeys. Physiol. Behav. 1994, 56, 801–810. [Google Scholar] [CrossRef][Green Version]
- Sokal, R.R.; Rohlf, F.J. Biometry: The Principles and Practice of Statistics in Biological Research, 3rd ed.; W.H. Freeman and Co.: New York, NY, USA, 1995. [Google Scholar]

| Hormone Treatment | Time (Months) Following Study Onset | ||||||
|---|---|---|---|---|---|---|---|
| Female Group | 0 | 1 | 2 | 3 | 4 | 5 | 6 |
| Fasted insulin (pmol/L) | |||||||
| Diluent group | |||||||
| Control | 160 ± 12 | 333 ± 35 | 397 ± 30 | 374 ± 66 | 270 ± 25 | 215 ± 3 | 220 ± 4 |
| PCOS-like | 375 ± 71 | 305 ± 40 | 371 ± 92 | 410 ± 78 | 337 ± 48 | 220 ± 2 | 216 ± 3 |
| Insulin group a | |||||||
| Control | 182 ± 49 | 338 ± 106 | 557 ± 98 | 622 ± 130 | 841 ± 195 | 880 ± 115 | 890 ± 106 |
| PCOS-like | 267 ± 91 | 403 ± 105 | 442 ± 123 | 610 ± 102 | 793 ± 151 | 926 ± 52 | 996 ± 36 |
| Fasted glucose (mmol/L) | |||||||
| Diluent group | |||||||
| Control | 3.1 ± 0.1 | 3.1 ± 0.1 | 3.0 ± 0.1 | 3.0 ± 0.1 | 3.0 ± 0.1 | 3.2 ± 0.1 | 3.2 ± 0.1 |
| PCOS-like | 2.9 ± 0.1 | 3.2 ± 0.2 | 3.0 ± 0.2 | 3.1 ± 0.2 | 3.3 ± 0.2 | 3.4 ± 0.2 | 3.3 ± 0.2 |
| Insulin group b | |||||||
| Control | 2.9 ± 0.2 | 3.0 ± 0.1 | 2.8 ± 0. | 2.8 ± 0.2 | 3.2 ± 0.1 | 3.2 ± 0.1 | 3.3 ± 0.1 |
| PCOS-like | 5.3 ± 1.5 | 5.1 ± 1.3 | 4.1 ± 0.8 | 4.6 ± 1.0 | 5.4 ± 1.3 | 5.1 ± 0.8 | 4.7 ± 0.9 |
| Afternoon glucose (mmol/L) | |||||||
| Diluent group | |||||||
| Control | 3.2 ± 0.3 | 3.0 ± 0.1 | 3.0 ± 0.1 | 3.1 ± 0.1 | 3.1 ± 0.1 | 3.3 ± 0.1 | 3.4 ± 0.1 |
| PCOS-like | 3.0 ± 0.1 | 3.3 ± 0.1 | 3.3 ± 0.2 | 3.3 ± 0.1 | 3.4 ± 0.2 | 3.4 ± 0.2 | 3.5 ± 0.2 |
| Insulin group c | |||||||
| Control | 3.2 ± 0.3 | 3.2 ± 0.3 | 3.1 ± 0.1 | 3.2 ± 0.1 | 3.3 ± 0.1 | 3.2 ± 0.1 | 3.4 ± 0.1 |
| PCOS-like | 5.7 ± 1.3 | 6.3 ± 2.2 | 6.0 ± 2.1 | 6.2 ± 2.3 | 5.8 ± 1.9 | 5.4 ± 0.2 | 5.5 ± 1.9 |
| Female Treatment Groups | Hours 0 h | Following 2 h | Daily 4 h | Injection 24 h |
|---|---|---|---|---|
| Control diluent (n = 4) | 373 ± 79 | 743 ± 240 | 961 ± 261 | 476 ± 110 |
| Control insulin a (n = 4) | 972 ± 321 | 672 ± 44 | 3966 ± 1340 b | 964 ± 244 c |
| PCOS-like diluent (n = 5) | 522 ± 193 | 925 ± 192 | 826 ± 214 | 517 ± 152 |
| PCOS-like insulin a (n = 5) | 760 ± 90 | 2125 ± 830 | 1345 ± 387 b | 927 ± 325 c |
| Parameters | Control Monkeys | PCOS-like Monkeys | ||
|---|---|---|---|---|
| Diluent | Insulin | Diluent | Insulin | |
| Gb (mmol/L) | 3.2 ± 0.2 | 3.6 ± 0.6 | 3.5 ± 0.2 | 2.2 ± 0.6 |
| Ib (pmol/L) | 294 ± 117 | 478 ± 139 a | 204 ± 117 | 557 ± 139 a |
| Kg (%/min) | 9.8 ± 1.5 | 4.9 ± 0.9 | 5.0 ± 1.5 | 4.4 ± 0.9 |
| Sg (×10−2/min) | 4.1 ± 1.1 | 5.4 ± 1.0 | 4.1 ± 1.1 | 4.8 ± 1.0 |
| SI (×10−5/min/pmol/L) | 2.0 ± 1.8 | 1.6 ± 0.9 | 3.9 ± 1.8 | 2.2 ± 0.9 |
| DI (×102/min) | 4.8 ± 2.3 | 3.0 ± 0.7 b | 4.8 ± 2.3 | 0.6 ± 0.7 b |
| AIRg (pmol/L) | 251 ± 58 | 233 ± 48 c | 134 ± 58 | 37 ± 48 c |
| AIRtol (pmol/L) | 276 ± 72 | 520 ± 117 | 242 ± 72 | 143 ± 58 |
| AUCINS (0–19) (pmol/L × 19 min × 103) | 38 ± 91 | 60 ± 11 | 27 ± 91 d | 18 ± 11 d |
| AUCINS (22–180) (pmol/L × 158 min × 103) | 84 ± 38 | 183 ± 58 | 63 ± 38 | 97 ± 58 |
| AUCINS (0–180) (pmol/L × 180 min × 103) | 123 ± 44 | 244 ± 69 e | 90 ± 44 | 115 ± 69 e |
| AUCGLU (0–19) (mmol/L × 19 min) | 204 ± 11 | 237 ± 20 | 206 ± 11 | 211 ± 20 |
| AUCGLU (22–180) (mmol/L × 158 min) | 503 ± 48 | 585 ± 100 | 547 ± 48 | 585 ± 100 |
| AUCGLU (0–180) (mmol/L × 180 min) | 716 ± 50 | 819 ± 115 | 752 ± 50 | 796 ± 115 |
| Basal C-peptide (nmol/L) × 10−1 | 1.9 ± 0.6 | 1.1 ± 1.4 | 1.6 ± 0.6 | 5.3 ± 1.4 |
| AUCc-pep (0–180) (nmol/L × 180 min) | 33.3 ± 11.2 | 31.7 ± 2.7 | 29.3 ± 7.3 | 98.3 ± 29.0 |
| Diluent Baseline | Diluent 6–7 mo | Insulin Baseline | Insulin 6–7 mo | |
|---|---|---|---|---|
| Body weight Control (kg) | 8.4 ± 0.6 | 8.8 ± 0.9 | 8.5 ± 0.3 | 9.0 ± 0.6 |
| Body weight PCOS-like (kg) | 8.8 ± 0.3 | 8.4 ± 0.4 | 9.1 ± 0.6 | 9.5 ± 0.5 |
| Cholesterol Control (mg/mL) | 150 ± 13 | 137 ± 4 | 172 ± 10 | 151 ± 7 |
| Cholesterol PCOS-like (mg/mL) | 157 ± 14 | 162 ± 14 | 116 | 140 ± 9 |
| Triglyceride Control (mg/mL) | 158 ± 55 | 55 ± 19 | 130 ± 36 | 235 ± 32 |
| Triglyceride PCOS-like (mg/mL) | 191 ± 28 | 88 ± 42 | 363 | 185 ± 95 |
| AST Control (mU/mL) | 31 ± 5 | 36 ± 10 | 42 ± 7 | 32 ± 14 |
| AST PCOS-like (mU/mL) | 25 | 22 | 29 | 42 ± 2 |
| ALT Control (mU/mL) | 33 ± 6 | 32 ± 4 | 51 ± 21 | 33 ± 7 |
| ALT PCOS-like (mU/mL) | 43 | 27 | 9 | 20 ± 6 |
| Hormone Ratio Treatment | Time (Hours) Following 200IU Intramuscular Injection of rhCG | |||
|---|---|---|---|---|
| Female Group | 0 | 24 | 48 | 72 |
| Androstenedione:Dehydroepiandrosterone | ||||
| Diluent | ||||
| Control | 6.8 ± 1.6 | 6.5 ± 2.9 | 7.7 ± 2.1 | 17.4 ± 11.0 |
| PCOS-like | 2.7 ± 0.2 | 6.3 ± 1.4 | 4.5 ± 0.9 | 1.8 ± 0.5 |
| Insulin | ||||
| Control | 2.4 ± 0.8 | 3.5 ± 1.3 | 2.8 ± 0.4 | 2.3 ± 0.5 |
| PCOS-like | 7.2 ± 2.1 | 6.6 ± 2.6 | 7.8 ± 2.2 | 5.6 ± 2.4 |
| Testosterone:Androstenedione × 10−2 | ||||
| Diluent | ||||
| Control | 1.6 ± 0.2 | 1.9 ± 0.5 | 2.3 ± 0.6 | 2.1 ± 0.3 |
| PCOS-like | 1.5 ± 0.2 | 1.2 ± 0.1 | 1.6 ± 0.4 | 1.4 ± 0.2 |
| Insulin | ||||
| Control | 1.7 ± 0.5 | 1.1 ± 0.2 | 1.1 ± 0.2 | 1.1 ± 0.1 |
| PCOS-like | 1.0 ± 0.4 | 0.7 ± 0.4 | 0.9 ± 0.5 | 0.8 ± 0.4 |
| Testosterone:Estradiol | ||||
| Diluent | ||||
| Control | 15.4 ± 2.9 | 11.3 ± 3.2 | 27.8 ± 12.5 | 19.8 ± 2.7 |
| PCOS-like | 12.4 ± 6.5 | 7.3 ± 1.5 | 14.3 ± 1.3 | 12.3 ± 1.1 |
| Insulin | ||||
| Control | 18.7 ± 5.9 | 8.3 ± 2.6 | 14.5 ± 5.5 | 11.9 ± 3.5 |
| PCOS-like | 22.3 ± 6.8 | 15.5 ± 8.3 | 19.1 ± 6.6 | 25.3 ± 10.9 |
| Estradiol:Progesterone × 10−2 | ||||
| Diluent | ||||
| Control | 23.5 ± 11.5 | 2.7 ± 0.9 | 10.0 ± 8.3 | 8.0 ± 4.4 |
| PCOS-like | 38.9 ± 17.3 | 7.9 ± 2.5 | 4.5 ± 1.0 | 4.0 ± 0.3 |
| Insulin | ||||
| Control | 11.3 ± 6.4 | 4.3 ± 1.4 | 3.0 ± 0.9 | 5.9 ± 1.5 |
| PCOS-like | 4.3 ± 1.3 | 4.4 ± 1.4 | 3.6 ± 1.0 | 3.3 ± 1.6 |
| Hormone Treatment | Time (Minutes) Following GnRH Injection | |||||||
|---|---|---|---|---|---|---|---|---|
| Female Group | 0 | 2 | 5 | 10 | 20 | 30 | 40 | 60 |
| LH (ng/mL) | ||||||||
| Diluent | ||||||||
| Control | 1.2 ± 0.4 a | 1.2 ± 0.3 | 1.3 ± 0.4 | 1.4 ± 0.4 c | 1.3 ± 0.4 | 1.4 ± 0.4 | 1.3 ± 0.4 | 1.3 ± 0.4 |
| PCOS-like | 1.3 ± 0.4 a | 2.3 ± 0.8 | 2.7 ± 1.0 | 2.7 ± 1.0 c | 2.8 ± 0.9 | 2.6 ± 0.8 | 2.5 ± 0.8 | 2.4 ± 1.0 |
| Insulin | ||||||||
| Control | 1.1 ± 0.3 a | 1.1 ± 0.3 | 1.4 ± 0.4 | 1.5 ± 0.3 c | 1.6 ± 0.4 | 1.6 ± 0.4 | 1.7 ± 0.5 | 1.5 ± 0.4 |
| PCOS-like | 1.0 ± 0.2 a | 1.3 ± 0.1 | 1.6 ± 0.1 | 1.6 ± 0.1 c | 1.6 ± 0.1 | 1.7 ± 0.1 | 1.7 ± 0.1 | 1.6 ± 0.1 |
| FSH (ng/mL) | ||||||||
| Diluent | ||||||||
| Control | 1.8 ± 0.3 b | 1.6 ± 0.2 | 1.9 ± 0.4 | 1.7 ± 0.4 c | 1.8 ± 0.4 | 1.7 ± 0.3 | 1.8 ± 0.5 | 1.8 ± 0.4 |
| PCOS-like | 3.9 ± 1.3 b | 4.5 ± 1.5 | 4.5 ± 1.5 | 4.8 ± 1.6 c | 5.4 ± 1.6 | 5.2 ± 1.4 | 5.3 ± 1.5 | 5.3 ± 1.5 |
| Insulin | ||||||||
| Control | 2.8 ± 1.6 b | 2.8 ± 1.5 | 2.7 ± 1.5 | 2.8 ± 1.4 c | 2.7 ± 1.0 | 2.7 ± 1.1 | 2.9 ± 1.5 | 2.9 ± 1.4 |
| PCOS-like | 3.0 ± 1.0 b | 2.9 ± 0.8 | 3.3 ± 1.0 | 3.2 ± 0.8 c | 3.3 ± 0.8 | 3.2 ± 0.6 | 3.5 ± 0.9 | 3.5 ± 0.9 |
| LH:FSH ratio | ||||||||
| Diluent | ||||||||
| Control | 0.8 ± 0.2 | 0.8 ± 0.2 | 0.7 ± 0.2 | 0.8 ± 0.2 | 0.8 ± 0.2 | 0.8 ± 0.2 | 0.8 ± 0.2 | 0.7 ± 0.2 |
| PCOS-like | 0.4 ± 0.1 | 0.5 ± 0.1 | 0.6 ± 0.1 | 0.6 ± 0.1 | 0.5 ± 0.1 | 0.5 ± 0.1 | 0.5 ± 0.1 | 0.4 ± 0.1 |
| Insulin | ||||||||
| Control | 0.5 ± 0.1 | 0.5 ± 0.1 | 0.7 ± 0.2 | 0.7 ± 0.1 | 0.7 ± 0.1 | 0.7 ± 0.1 | 0.8 ± 0.1 | 0.6 ± 0.1 |
| PCOS-like | 0.6 ± 0.2 | 0.7 ± 0.3 | 0.8 ± 0.3 | 0.7 ± 0.3 | 0.7 ± 0.2 | 0.7 ± 0.2 | 0.7 ± 0.2 | 0.6 ± 0.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, R.; Bruns, C.M.; Bird, I.M.; Kemnitz, J.W.; Dumesic, D.A.; Abbott, D.H. Experimentally Induced Hyperinsulinemia Fails to Induce Polycystic Ovary Syndrome-like Traits in Female Rhesus Macaques. Int. J. Mol. Sci. 2022, 23, 2635. https://doi.org/10.3390/ijms23052635
Zhou R, Bruns CM, Bird IM, Kemnitz JW, Dumesic DA, Abbott DH. Experimentally Induced Hyperinsulinemia Fails to Induce Polycystic Ovary Syndrome-like Traits in Female Rhesus Macaques. International Journal of Molecular Sciences. 2022; 23(5):2635. https://doi.org/10.3390/ijms23052635
Chicago/Turabian StyleZhou, Rao, Cristin M. Bruns, Ian M. Bird, Joseph W. Kemnitz, Daniel A. Dumesic, and David H. Abbott. 2022. "Experimentally Induced Hyperinsulinemia Fails to Induce Polycystic Ovary Syndrome-like Traits in Female Rhesus Macaques" International Journal of Molecular Sciences 23, no. 5: 2635. https://doi.org/10.3390/ijms23052635
APA StyleZhou, R., Bruns, C. M., Bird, I. M., Kemnitz, J. W., Dumesic, D. A., & Abbott, D. H. (2022). Experimentally Induced Hyperinsulinemia Fails to Induce Polycystic Ovary Syndrome-like Traits in Female Rhesus Macaques. International Journal of Molecular Sciences, 23(5), 2635. https://doi.org/10.3390/ijms23052635







