Adipsin Serum Concentrations and Adipose Tissue Expression in People with Obesity and Type 2 Diabetes
Abstract
1. Introduction
2. Results
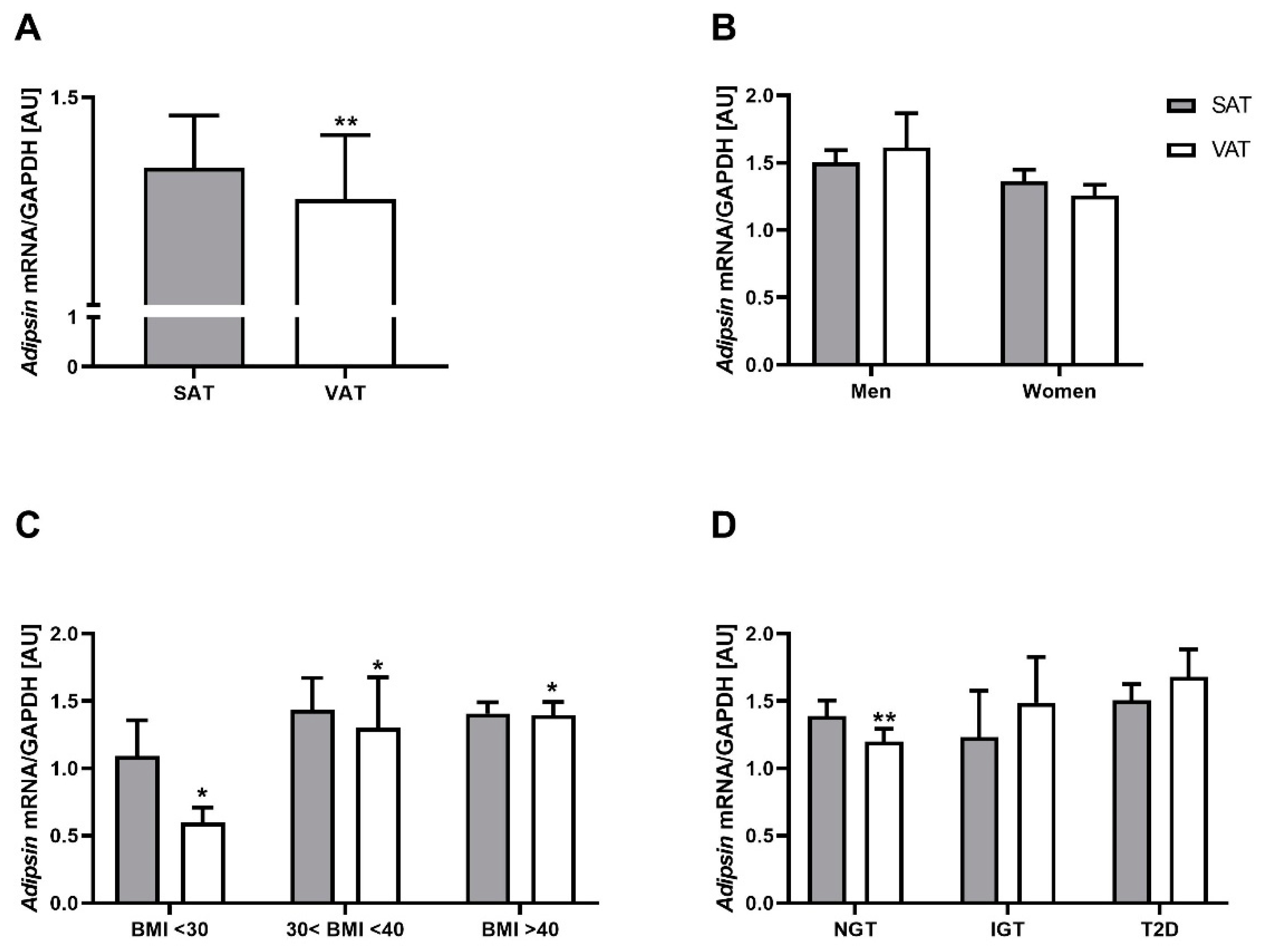
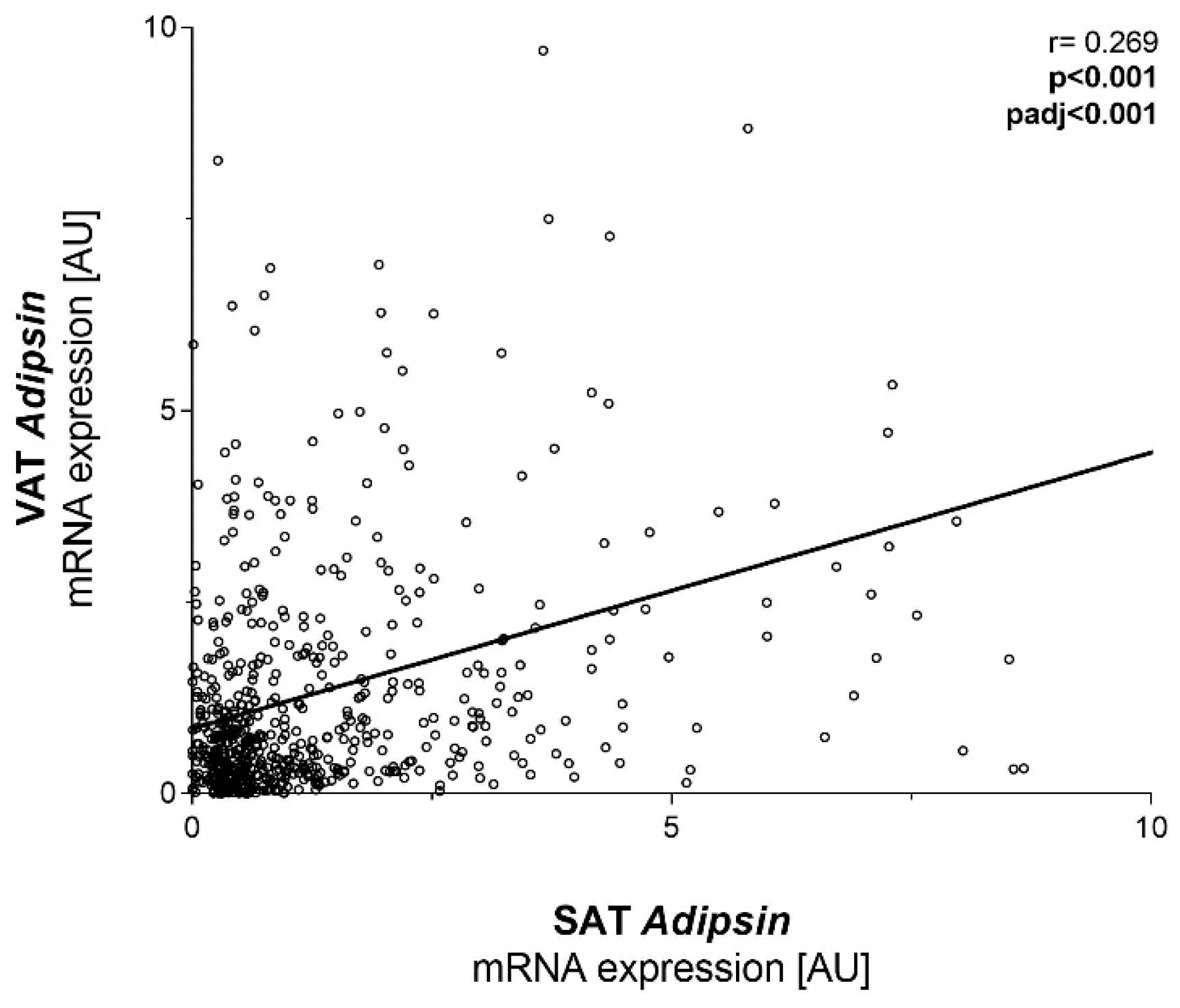
2.1. Adipsin mRNA Is Higher in SAT Compared to VAT but Not Related to Obesity and T2D
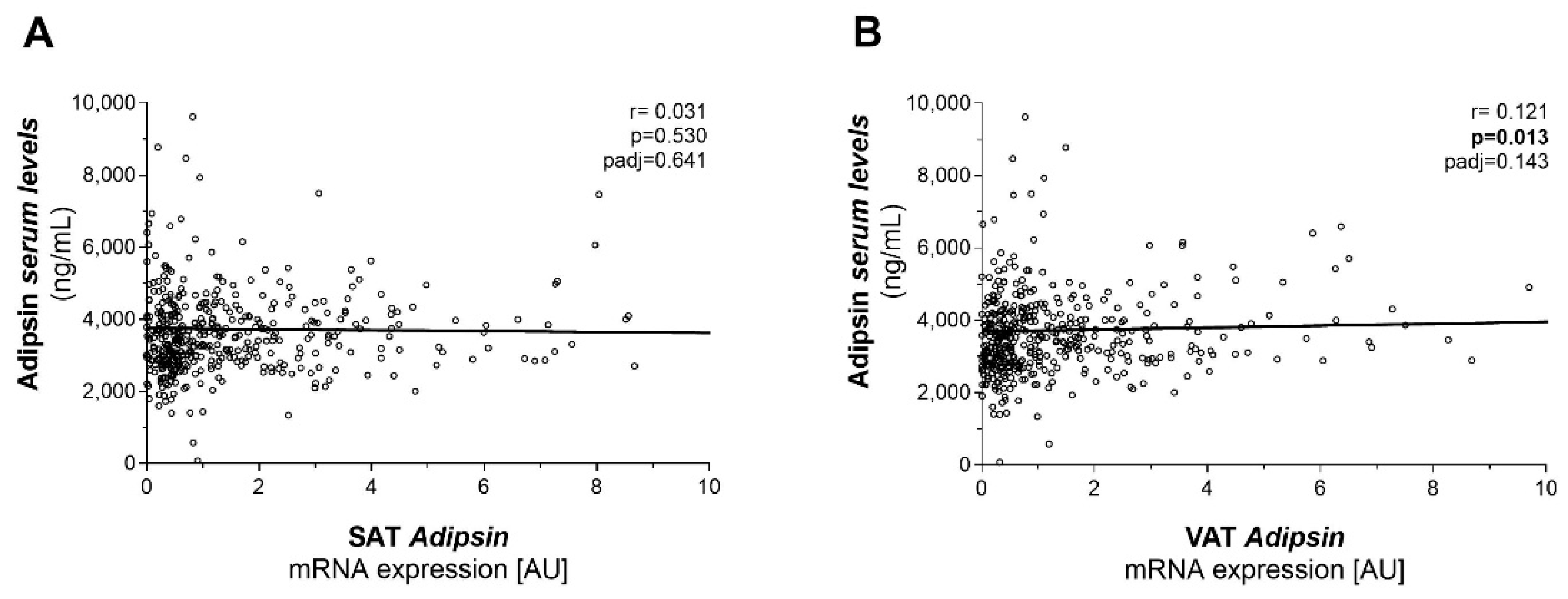
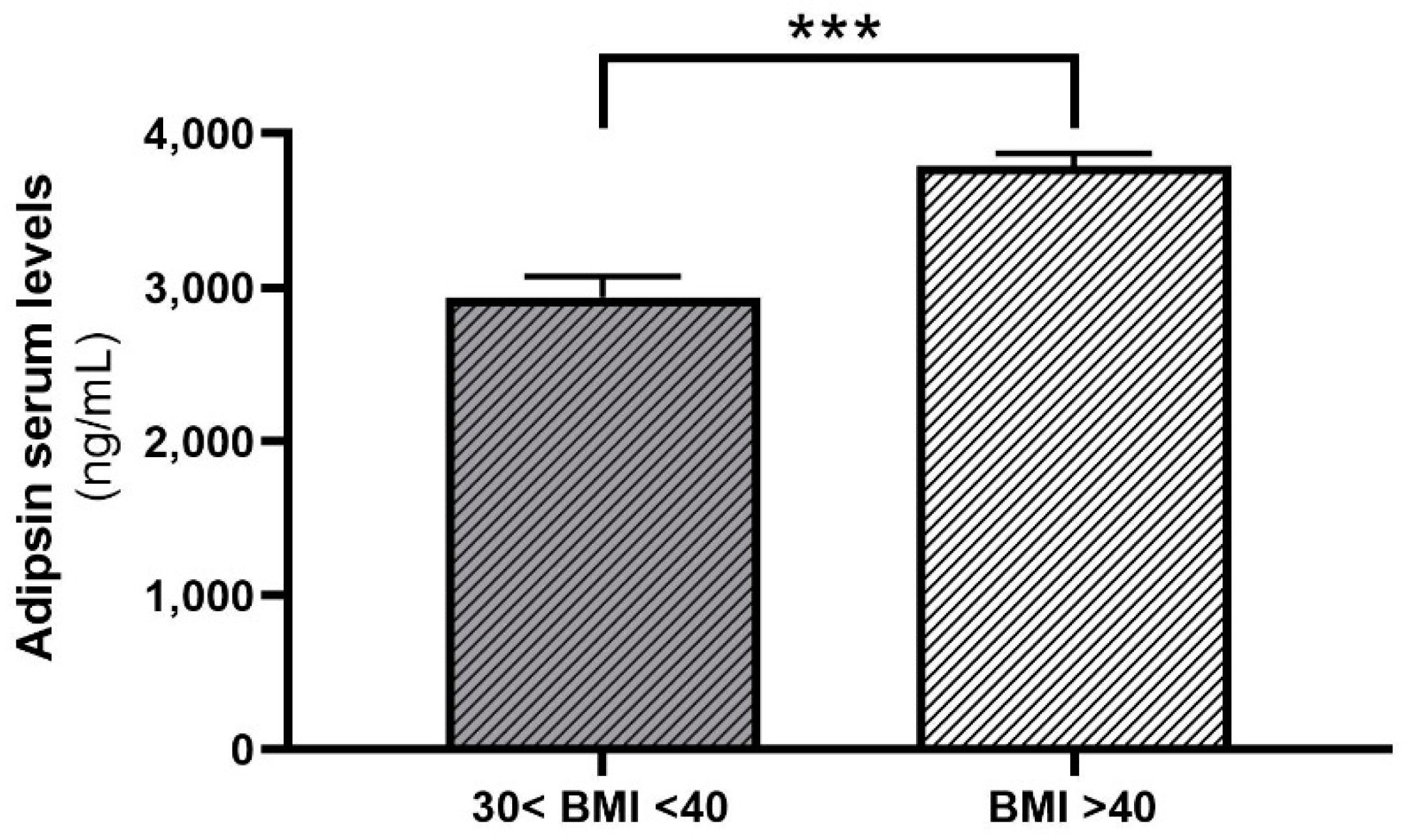
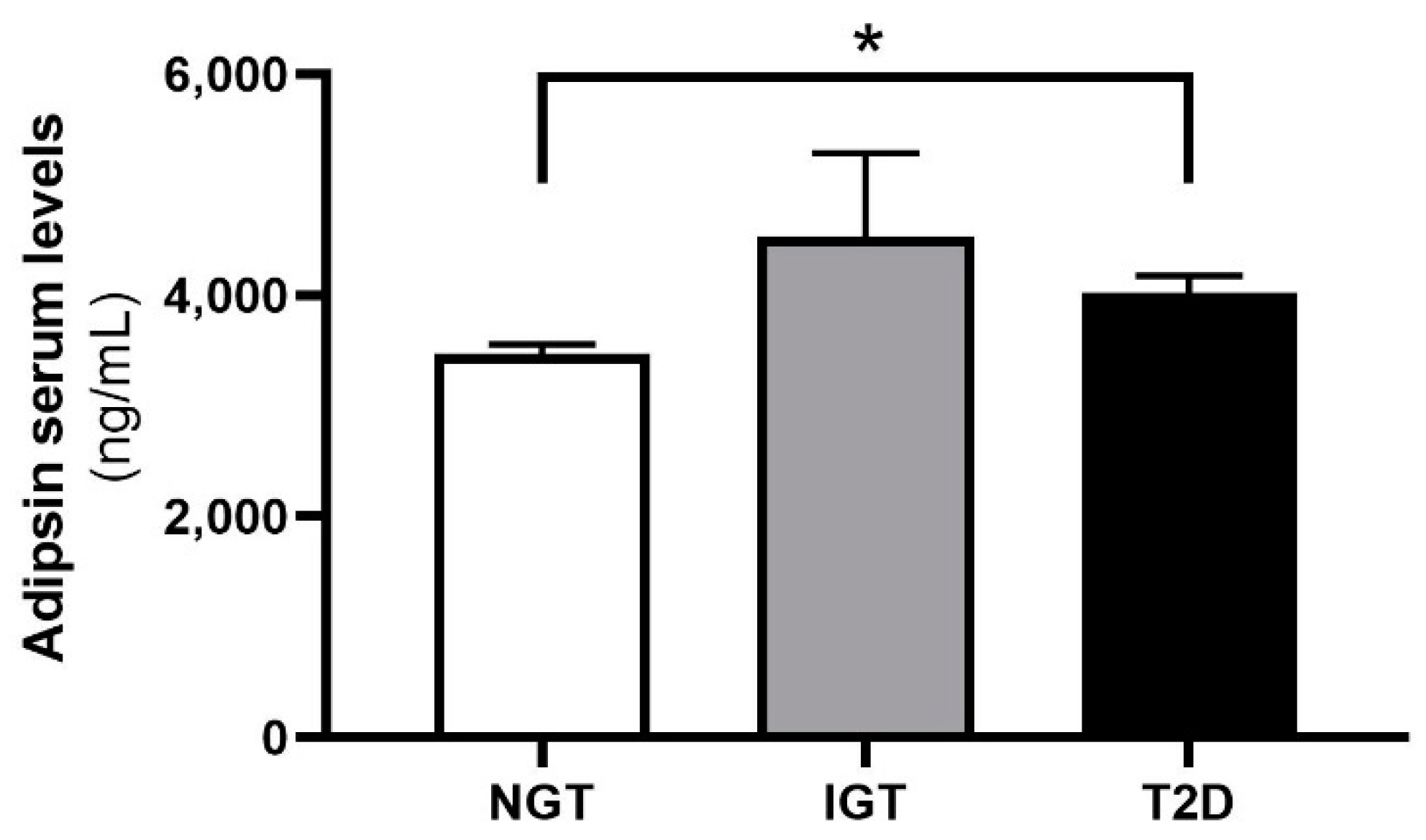
2.2. Adipsin Serum Concentrations Are Higher in Patients with Obesity and T2D
3. Discussion
4. Materials and Methods
4.1. Subjects
4.2. Measurement of Adipsin Serum Concentrations
4.3. Adipsin mRNA Expression Analysis in AT
4.4. Statistical Analyses
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 15 June 2021).
- Blüher, M. Obesity: Global epidemiology and pathogenesis. Nat. Rev. Endocrinol. 2019, 15, 288–298. [Google Scholar] [CrossRef] [PubMed]
- NCD Risk Factor Collaboration (NCD-RisC). Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: A pooled analysis of 2416 population-based measurement studies in 128·9 million children, adolescents, and adults. Lancet 2017, 390, 2627–2642. [Google Scholar] [CrossRef]
- van Gaal, L.F.; Mertens, I.L.; de Block, C.E. Mechanisms linking obesity with cardiovascular disease. Nature 2006, 444, 875–880. [Google Scholar] [CrossRef] [PubMed]
- Afshin, A.; Forouzanfar, M.H.; Reitsma, M.B.; Sur, P.; Estep, K.; Lee, A.; Marczak, L.; Mokdad, A.H.; Moradi-Lakeh, M.; Naghavi, M.; et al. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Klöting, N.; Blüher, M. Adipocyte dysfunction, inflammation and metabolic syndrome. Rev. Endocr. Metab. Disord. 2014, 15, 277–287. [Google Scholar] [CrossRef] [PubMed]
- Klöting, N.; Fasshauer, M.; Dietrich, A.; Kovacs, P.; Schön, M.R.; Kern, M.; Stumvoll, M.; Blüher, M. Insulin-sensitive obesity. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E506–E515. [Google Scholar] [CrossRef]
- Ye, R.Z.; Richard, G.; Gévry, N.; Tchernof, A.; Carpentier, A.C. Fat Cell Size: Measurement Methods, Pathophysiological Origins, and Relationships with Metabolic Dysregulations. Endocr. Rev. 2021, 43, 35–60. [Google Scholar] [CrossRef]
- Rutkowski, J.M.; Stern, J.H.; Scherer, P.E. The cell biology of fat expansion. J. Cell Biol. 2015, 208, 501–512. [Google Scholar] [CrossRef]
- Schleinitz, D.; Böttcher, Y.; Blüher, M.; Kovacs, P. The genetics of fat distribution. Diabetologia 2014, 57, 1276–1286. [Google Scholar] [CrossRef]
- Zhao, S.; Kusminski, C.M.; Scherer, P.E. Adiponectin, Leptin and Cardiovascular Disorders. Circ. Res. 2021, 128, 136–149. [Google Scholar] [CrossRef]
- Scherer, P.E. The many secret lives of adipocytes: Implications for diabetes. Diabetologia 2019, 62, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Dahlman, I.; Elsen, M.; Tennagels, N.; Korn, M.; Brockmann, B.; Sell, H.; Eckel, J.; Arner, P. Functional annotation of the human fat cell secretome. Arch. Physiol. Biochem. 2012, 118, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Blüher, M. Adipokines-removing road blocks to obesity and diabetes therapy. Mol. Metab. 2014, 3, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Cook, K.S.; Min, H.Y.; Johnson, D.; Chaplinsky, R.J.; Flier, J.S.; Hunt, C.R.; Spiegelman, B.M. Adipsin: A circulating serine protease homolog secreted by adipose tissue and sciatic nerve. Science 1987, 237, 402–405. [Google Scholar] [CrossRef] [PubMed]
- Rosen, B.S.; Cook, K.S.; Yaglom, J.; Groves, D.L.; Volanakis, J.E.; Damm, D.; White, T.; Spiegelman, B.M. Adipsin and complement factor D activity: An immune-related defect in obesity. Science 1989, 244, 1483–1487. [Google Scholar] [CrossRef]
- White, R.T.; Damm, D.; Hancock, N.; Rosen, B.S.; Lowell, B.B.; Usher, P.; Flier, J.S.; Spiegelman, B.M. Human adipsin is identical to complement factor D and is expressed at high levels in adipose tissue. J. Biol. Chem. 1992, 267, 9210–9213. [Google Scholar] [CrossRef]
- Xu, Y.; Ma, M.; Ippolito, G.C.; Schroeder, H.W.; Carroll, M.C.; Volanakis, J.E. Complement activation in factor D-deficient mice. Proc. Natl. Acad. Sci. USA 2001, 98, 14577–14582. [Google Scholar] [CrossRef]
- Ansorge, S.; Täger, M. Immunologie. In Löffler/Petrides Biochemie und Pathobiochemie; Heinrich, P.C., Müller, M., Graeve, L., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 893–930. ISBN 978-3-642-17971-6. [Google Scholar]
- Lo, J.C.; Ljubicic, S.; Leibiger, B.; Kern, M.; Leibiger, I.B.; Moede, T.; Kelly, M.E.; Chatterjee Bhowmick, D.; Murano, I.; Cohen, P.; et al. Adipsin is an adipokine that improves β cell function in diabetes. Cell 2014, 158, 41–53. [Google Scholar] [CrossRef]
- Gómez-Banoy, N.; Guseh, J.S.; Li, G.; Rubio-Navarro, A.; Chen, T.; Poirier, B.; Putzel, G.; Rosselot, C.; Pabón, M.A.; Camporez, J.P.; et al. Adipsin preserves beta cells in diabetic mice and associates with protection from type 2 diabetes in humans. Nat. Med. 2019, 25, 1739–1747. [Google Scholar] [CrossRef]
- Napolitano, A.; Lowell, B.B.; Damm, D.; Leibel, R.L.; Ravussin, E.; Jimerson, D.C.; Lesem, M.D.; van Dyke, D.C.; Daly, P.A.; Chatis, P. Concentrations of adipsin in blood and rates of adipsin secretion by adipose tissue in humans with normal, elevated and diminished adipose tissue mass. Int. J. Obes. Relat. Metab. Disord. 1994, 18, 213–218. [Google Scholar]
- Pomeroy, C.; Mitchell, J.; Eckert, E.; Raymond, N.; Crosby, R.; Dalmasso, A.P. Effect of body weight and caloric restriction on serum complement proteins, including Factor D/adipsin: Studies in anorexia nervosa and obesity. Clin. Exp. Immunol. 1997, 108, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Litvinova, L.S.; Vasilenko, M.A.; Zatolokin, P.A.; Aksenova, N.N.; Fattakhov, N.S.; Vaysbeyn, I.Z.; Mironyuk, N.I.; Kirienkova, E.V. Adipokines in metabolic processes regulating during obesity treatment. Diabetes Mellit. 2014, 17, 51–59. [Google Scholar] [CrossRef]
- Flier, J.S.; Cook, K.S.; Usher, P.; Spiegelman, B.M. Severely impaired adipsin expression in genetic and acquired obesity. Science 1987, 237, 405–408. [Google Scholar] [CrossRef] [PubMed]
- Vasilenko, M.A.; Kirienkova, E.V.; Skuratovskaia, D.A.; Zatolokin, P.A.; Mironyuk, N.I.; Litvinova, L.S. The role of production of adipsin and leptin in the development of insulin resistance in patients with abdominal obesity. Dokl. Biochem. Biophys. 2017, 475, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Corvillo, F.; González-Sánchez, L.; López-Lera, A.; Arjona, E.; Ceccarini, G.; Santini, F.; Araújo-Vilar, D.; Brown, R.J.; Villarroya, J.; Villarroya, F.; et al. Complement Factor D (adipsin) Levels Are Elevated in Acquired Partial Lipodystrophy (Barraquer-Simons syndrome). Int. J. Mol. Sci. 2021, 22, 6608. [Google Scholar] [CrossRef] [PubMed]
- Ricklin, D.; Hajishengallis, G.; Yang, K.; Lambris, J.D. Complement: A key system for immune surveillance and homeostasis. Nat. Immunol. 2010, 11, 785–797. [Google Scholar] [CrossRef]
- Lim, J.; Iyer, A.; Suen, J.Y.; Seow, V.; Reid, R.C.; Brown, L.; Fairlie, D.P. C5aR and C3aR antagonists each inhibit diet-induced obesity, metabolic dysfunction, and adipocyte and macrophage signaling. FASEB J. 2013, 27, 822–831. [Google Scholar] [CrossRef]
- Mamane, Y.; Chung Chan, C.; Lavallee, G.; Morin, N.; Xu, L.-J.; Huang, J.; Gordon, R.; Thomas, W.; Lamb, J.; Schadt, E.E.; et al. The C3a anaphylatoxin receptor is a key mediator of insulin resistance and functions by modulating adipose tissue macrophage infiltration and activation. Diabetes 2009, 58, 2006–2017. [Google Scholar] [CrossRef]
- Vlaicu, S.I.; Tatomir, A.; Boodhoo, D.; Vesa, S.; Mircea, P.A.; Rus, H. The role of complement system in adipose tissue-related inflammation. Immunol. Res. 2016, 64, 653–664. [Google Scholar] [CrossRef]
- GTEx Analysis Release V8. Gene Expression for CFD. Available online: https://www.gtexportal.org/home/gene/CFD (accessed on 27 April 2021).
- Xia, Z.; Cianflone, K. Acylation-stimulating protein precursor proteins in adipose tissue in human obesity. Metab. Clin. Exp. 2003, 52, 1360–1366. [Google Scholar] [CrossRef]
- Haim, Y.; Blüher, M.; Konrad, D.; Goldstein, N.; Klöting, N.; Harman-Boehm, I.; Kirshtein, B.; Ginsberg, D.; Tarnovscki, T.; Gepner, Y.; et al. ASK1 (MAP3K5) is transcriptionally upregulated by E2F1 in adipose tissue in obesity, molecularly defining a human dys-metabolic obese phenotype. Mol. Metab. 2017, 6, 725–736. [Google Scholar] [CrossRef] [PubMed]
- Blüher, M.; Bashan, N.; Shai, I.; Harman-Boehm, I.; Tarnovscki, T.; Avinaoch, E.; Stumvoll, M.; Dietrich, A.; Klöting, N.; Rudich, A. Activated Ask1-MKK4-p38MAPK/JNK stress signaling pathway in human omental fat tissue may link macrophage infiltration to whole-body Insulin sensitivity. J. Clin. Endocrinol. Metab. 2009, 94, 2507–2515. [Google Scholar] [CrossRef] [PubMed]
- Bashan, N.; Dorfman, K.; Tarnovscki, T.; Harman-Boehm, I.; Liberty, I.F.; Blüher, M.; Ovadia, S.; Maymon-Zilberstein, T.; Potashnik, R.; Stumvoll, M.; et al. Mitogen-activated protein kinases, inhibitory-kappaB kinase, and insulin signaling in human omental versus subcutaneous adipose tissue in obesity. Endocrinology 2007, 148, 2955–2962. [Google Scholar] [CrossRef] [PubMed]
- Klimontov, V.V.; Bulumbaeva, D.M.; Fazullina, O.N.; Lykov, A.P.; Bgatova, N.P.; Orlov, N.B.; Konenkov, V.I.; Pfeiffer, A.F.H.; Pivovarova-Ramich, O.; Rudovich, N. Circulating Wnt1-inducible signaling pathway protein-1 (WISP-1/CCN4) is a novel biomarker of adiposity in subjects with type 2 diabetes. J. Cell Commun. Signal. 2020, 14, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Klimontov, V.V.; Bulumbaeva, D.M.; Bgatova, N.P.; Taskaeva, I.S.; Orlov, N.B.; Fazullina, O.N.; Soluyanov, M.Y.; Savchenko, S.V.; Konenkov, V.I. Serum adipokine concentrations in patients with type 2 diabetes: The relationships with distribution, hypertrophy and vascularization of subcutaneous adipose tissue. Diabetes Mellit. 2019, 22, 336–347. [Google Scholar] [CrossRef]
- Cox, A.J.; Zhang, P.; Bowden, D.W.; Devereaux, B.; Davoren, P.M.; Cripps, A.W.; West, N.P. Enteroendocrine and adipokine associations with type 2 diabetes: Phenotypic risk scoring approaches. J. Gastroenterol. Hepatol. 2018, 33, 1357–1364. [Google Scholar] [CrossRef]
- Zhou, Q.; Ge, Q.; Ding, Y.; Qu, H.; Wei, H.; Wu, R.; Yao, L.; Wei, Q.; Feng, Z.; Long, J.; et al. Relationship between serum adipsin and the first phase of glucose-stimulated insulin secretion in individuals with different glucose tolerance. J. Diabetes Investig. 2018, 9, 1128–1134. [Google Scholar] [CrossRef]
- Karajibani, M.; Montazerifar, F.; Sadeghi, M.B.; Keikhaie, M.A.; Dashipour, A. Serum Fetuin-A and Adipsin Levels in Type II Diabetes Patients. Int. J. High Risk Behav. Addict. 2019, 8, e91963. [Google Scholar] [CrossRef]
- Labruna, G.; Pasanisi, F.; Nardelli, C.; Caso, R.; Vitale, D.F.; Contaldo, F.; Sacchetti, L. High leptin/adiponectin ratio and serum triglycerides are associated with an “at-risk” phenotype in young severely obese patients. Obes. Silver Spring 2011, 19, 1492–1496. [Google Scholar] [CrossRef]
- Martínez-García, M.Á.; Moncayo, S.; Insenser, M.; Álvarez-Blasco, F.; Luque-Ramírez, M.; Escobar-Morreale, H.F. Metabolic Cytokines at Fasting and During Macronutrient Challenges: Influence of Obesity, Female Androgen Excess and Sex. Nutrients 2019, 11, 2566. [Google Scholar] [CrossRef]
- Derosa, G.; Fogari, E.; D’Angelo, A.; Bianchi, L.; Bonaventura, A.; Romano, D.; Maffioli, P. Adipocytokine levels in obese and non-obese subjects: An observational study. Inflammation 2013, 36, 914–920. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Hutson, I.; Akk, A.M.; Mascharak, S.; Pham, C.T.N.; Hourcade, D.E.; Brown, R.; Atkinson, J.P.; Harris, C.A. Contribution of Adipose-Derived Factor D/Adipsin to Complement Alternative Pathway Activation: Lessons from Lipodystrophy. J. Immunol. 2018, 200, 2786–2797. [Google Scholar] [CrossRef] [PubMed]
- Gursoy Calan, O.; Calan, M.; Yesil Senses, P.; Unal Kocabas, G.; Ozden, E.; Sari, K.R.; Kocar, M.; Imamoglu, C.; Senses, Y.M.; Bozkaya, G.; et al. Increased adipsin is associated with carotid intima media thickness and metabolic disturbances in polycystic ovary syndrome. Clin. Endocrinol. Oxf. 2016, 85, 910–917. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-S.; Lee, W.-J.; Lee, I.-T.; Lin, S.-Y.; Lee, W.-L.; Liang, K.-W.; Sheu, W.H.-H. Association Between Serum Adipsin Levels and Insulin Resistance in Subjects With Various Degrees of Glucose Intolerance. J. Endocr. Soc. 2019, 3, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Tafere, G.G.; Wondafrash, D.Z.; Zewdie, K.A.; Assefa, B.T.; Ayza, M.A. Plasma Adipsin as a Biomarker and Its Implication in Type 2 Diabetes Mellitus. Diabetes Metab. Syndr. Obes. 2020, 13, 1855–1861. [Google Scholar] [CrossRef]
- Taşdemir, E. The Relationship of Plasma Adipsin, Adiponectin, Vaspin, Visfatin and Leptin Levels with Glucose Metabolism and Diabetes Parameters. Haydarpasa Numune Med. J. 2019, 59, 95–103. [Google Scholar] [CrossRef]
- Langhardt, J.; Flehmig, G.; Klöting, N.; Lehmann, S.; Ebert, T.; Kern, M.; Schön, M.R.; Gärtner, D.; Lohmann, T.; Dressler, M.; et al. Effects of Weight Loss on Glutathione Peroxidase 3 Serum Concentrations and Adipose Tissue Expression in Human Obesity. Obes. Facts 2018, 11, 475–490. [Google Scholar] [CrossRef]
- Rolle-Kampczyk, U.; Gebauer, S.; Haange, S.-B.; Schubert, K.; Kern, M.; Moulla, Y.; Dietrich, A.; Schön, M.R.; Klöting, N.; von Bergen, M.; et al. Accumulation of distinct persistent organic pollutants is associated with adipose tissue inflammation. Sci. Total Environ. 2020, 748, 142458. [Google Scholar] [CrossRef]
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2014, 37 (Suppl. S1), S81–S90. [Google Scholar] [CrossRef]
- Mardinoglu, A.; Heiker, J.T.; Gärtner, D.; Björnson, E.; Schön, M.R.; Flehmig, G.; Klöting, N.; Krohn, K.; Fasshauer, M.; Stumvoll, M.; et al. Extensive weight loss reveals distinct gene expression changes in human subcutaneous and visceral adipose tissue. Sci. Rep. 2015, 5, 14841. [Google Scholar] [CrossRef]
- Gesta, S.; Blüher, M.; Yamamoto, Y.; Norris, A.W.; Berndt, J.; Kralisch, S.; Boucher, J.; Lewis, C.; Kahn, C.R. Evidence for a role of developmental genes in the origin of obesity and body fat distribution. Proc. Natl. Acad. Sci. USA 2006, 103, 6676–6681. [Google Scholar] [CrossRef] [PubMed]
| SAT Adipsin mRNA r (p-Value; p-Value adj) | VAT Adipsin mRNA r (p-Value; p-Value adj) | Adipsin Serum Levels r (p-Value; p-Value adj) | |
|---|---|---|---|
| Age (years) | 0.078 (0.057; 0.064) | 0.074 (0.071; 0.025) | 0.282 (<0.001; <0.001) |
| Body weight (kg) | −0.018 (0.664; 0.219) | 0.058 (0.165; 0.460) | 0.264 (<0.001; <0.001) |
| Height (m) | −0.058 (0.166; 0.137) | −0.017 (0.687; 0.623) | 0.037 (0.444; 0.534) |
| BMI (kg/m2) | 0.008 (0.849; 0.482) | 0.077 (0.060; 0.170) | 0.359 (<0.001; <0.001) |
| Waist circumference (cm) | −0.234 (0.080; 0.576) | 0.089 (0.504; 0.670) | 0.137 (0.439; 0.398) |
| Hip circumference (cm) | 0.039 (0.803; 0.971) | 0.175 (0.262; 0.051) | 0.099 (0.66; 0.45) |
| WHR | −0.49 (0.001; <0.001) | 0.03 (0.849; 0.666) | −0.108 (0.633; 0.170) |
| Body fat (%) | 0.048 (0.569; 0.290) | 0.081 (0.330; 0.561) | 0.073 (0.423; 0.101) |
| FPG (mmol/L) | 0.025 (0.576; 0.338) | 0.022 (0.624; 0.748) | 0.136 (0.006; 0.948) |
| FPI (pmol/L) | −0.048 (0.588; 0.911) | −0.100 (0.256; 0.875) | 0.126 (0.176; 0.53) |
| HbA1c (%) | 0.018 (0.770; 0.272) | 0.036 (0.547; 0.727) | 0.044 (0.470; 0.095) |
| HOMA-IR | −0.074 (0.402; 0.777) | −0.100 (0.253; 0.574) | 0.076 (0.410; 0.794) |
| Total Cholesterol (mmol/L) | 0.003 (0.955; 1.00) | 0.019 (0.751; 0.278) | −0.067 (0.326; 0.896) |
| HDL-C (mmol/L) | 0.042 (0.476; 0.462) | −0.050 (0.401; 0.615) | −0.012 (0.858; 0.486) |
| LDL-C (mmol/L) | 0.012 (0.842; 0.907) | −0.025 (0.681; 0.455) | 0.007 (0.919; 0.508) |
| Triglycerides (mmol/L) | 0.018 (0.753; 0.671) | 0.033 (0.571; 0.753) | −0.068 (0.313; 0.694) |
| CrP (mg/L) | −0.065 (0.115; 0.158) | −0.049 (0.238; 0.297) | 0.039 (0.411; 0.825) |
| Leptin serum levels (ng/mL) | 0.008 (0.875; 0.44) | 0.135 (0.001; 0.021) | 0.362 (<0.001; <0.001) |
| BMI < 30 kg/m2 (n = 21) | BMI 30–40 kg/m2 (n = 52) | BMI > 40 kg/m2 (n = 564) | |
|---|---|---|---|
| Age (years) | 66.13 ± 10.56 | 48.41 ± 11.34 *** | 46.60 ± 11.93 *** |
| Men/Women (n) | 14/7 | 14/38 | 148/416 |
| T2D (n) | 4 | 22 | 211 |
| Body weight (kg) | 75.90 ± 12.28 | 107.41 ± 14.19 *** | 142.81 ± 26.36 ***,### |
| Height (m) | 1.74 ± 0.97 | 1.70 ± 0.084 | 1.70 ± 0.098 |
| BMI (kg/m2) | 25.05 ± 2.33 | 36.70 ± 2.84 *** | 49.52 ± 7.21 ***,### |
| Body fat (%) | 22.91 ± 5.01 | 42.97 ± 9.40 *** | 48.42 ± 10.09 *** |
| Waist circumference (cm) | 96.39 ± 14.95 | 122.50 ± 9.20 * | 143.667 ± 14.22 *** |
| Hip circumference (cm) | 97.11 ± 10.64 | 124 ± 11.32 * | 149.42 ± 14.50 ***,# |
| WHR | 0.99 ± 0.11 | 0.99 ± 0.17 | 0.99 ± 0.09 |
| FPG (mmol/L) | 5.72 ± 0.66 | 6.12 ± 1.96 | 6.40 ± 2.43 |
| FPI (pmol/L) | 47.33 ± 29.99 | 115.78 ± 88.57 | 145.47 ± 104.87 |
| HbA1c (%) | 5.73 ± 0.45 | 6.25 ± 1.46 | 5.99 ± 1.139 |
| HOMA-Index | 1.67 ± 1,08 | 5.00 ± 4.16 | 5.90 ± 6.00 |
| Total Cholesterol (mmol/L) | 5.40 ± 1.25 | 5.30 ± 1.27 | 5.99 ± 1.139 |
| HDL-Cholesterol (mmol/L) | 1.25 ± 0.25 | 1.26 ± 0.30 | 1.16 ± 0.61 |
| LDL-Cholesterol (mmol/L) | 3.31 ± 0.98 | 3.46 ± 1.15 | 3.09 ± 0.93 |
| Triglycerides (mmol/L) | 1.26 ± 0.57 | 1.98 ± 1.22 | 2.06 ± 2.24 |
| CrP (mg/L) | 9.75 ± 11.77 | 7.00 ± 10.48 | 12.57 ± 17.58 |
| AT adipsin mRNA (n) | 21 | 48 | 538 |
| Adipsin serum levels (n) | 4 | 38 | 414 |
| Parallel adipsin mRNA and serum levels data (n) | 4 | 32 | 388 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Milek, M.; Moulla, Y.; Kern, M.; Stroh, C.; Dietrich, A.; Schön, M.R.; Gärtner, D.; Lohmann, T.; Dressler, M.; Kovacs, P.; et al. Adipsin Serum Concentrations and Adipose Tissue Expression in People with Obesity and Type 2 Diabetes. Int. J. Mol. Sci. 2022, 23, 2222. https://doi.org/10.3390/ijms23042222
Milek M, Moulla Y, Kern M, Stroh C, Dietrich A, Schön MR, Gärtner D, Lohmann T, Dressler M, Kovacs P, et al. Adipsin Serum Concentrations and Adipose Tissue Expression in People with Obesity and Type 2 Diabetes. International Journal of Molecular Sciences. 2022; 23(4):2222. https://doi.org/10.3390/ijms23042222
Chicago/Turabian StyleMilek, Margarete, Yusef Moulla, Matthias Kern, Christine Stroh, Arne Dietrich, Michael R Schön, Daniel Gärtner, Tobias Lohmann, Miriam Dressler, Peter Kovacs, and et al. 2022. "Adipsin Serum Concentrations and Adipose Tissue Expression in People with Obesity and Type 2 Diabetes" International Journal of Molecular Sciences 23, no. 4: 2222. https://doi.org/10.3390/ijms23042222
APA StyleMilek, M., Moulla, Y., Kern, M., Stroh, C., Dietrich, A., Schön, M. R., Gärtner, D., Lohmann, T., Dressler, M., Kovacs, P., Stumvoll, M., Blüher, M., & Guiu-Jurado, E. (2022). Adipsin Serum Concentrations and Adipose Tissue Expression in People with Obesity and Type 2 Diabetes. International Journal of Molecular Sciences, 23(4), 2222. https://doi.org/10.3390/ijms23042222






