The Challenges of O2 Detection in Biological Fluids: Classical Methods and Translation to Clinical Applications
Abstract
1. Introduction
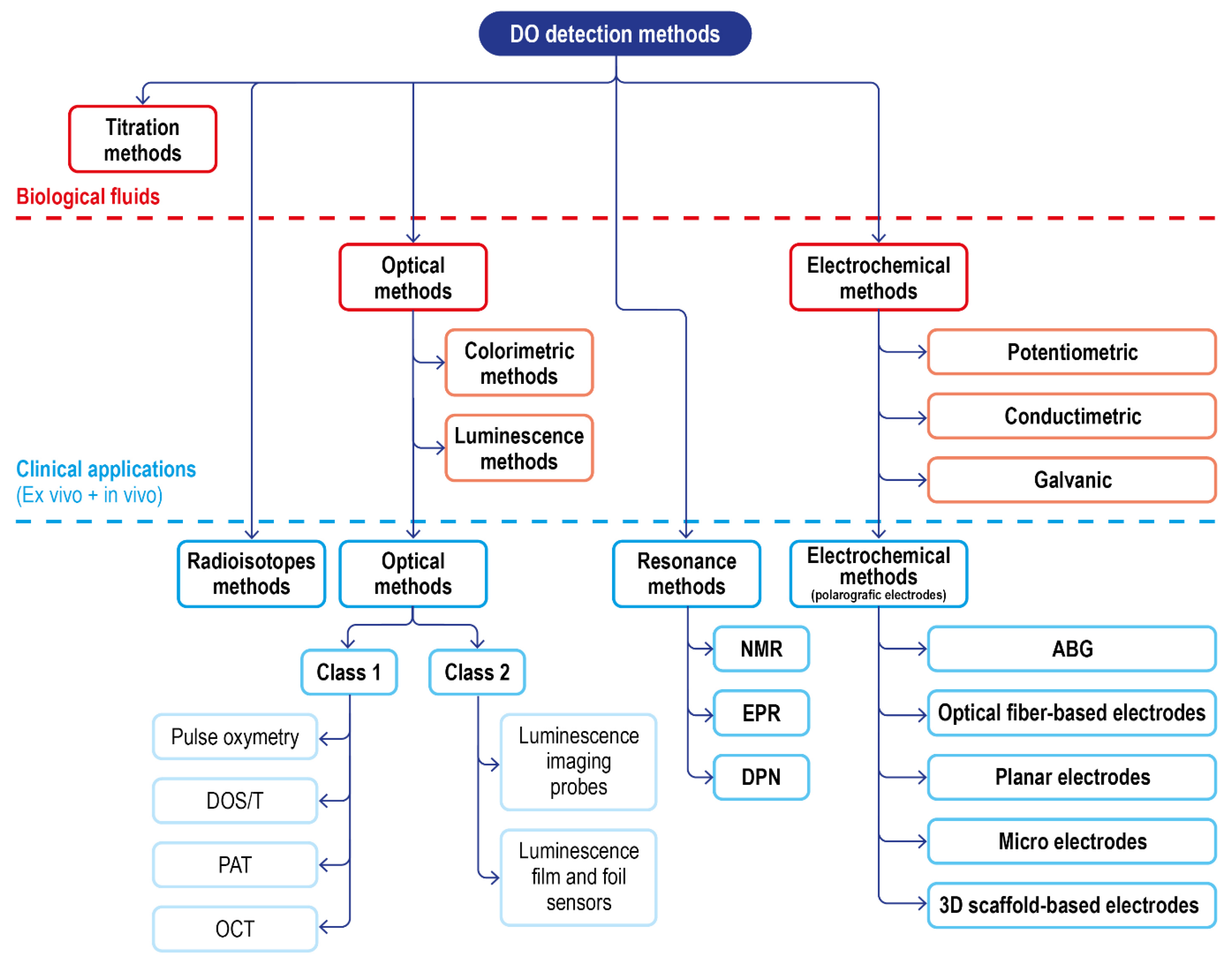
2. Classical Methods for DO Monitoring in Biological Fluids
2.1. Titration Method (Winkler Method)
Advantages, Disadvantages and Applications
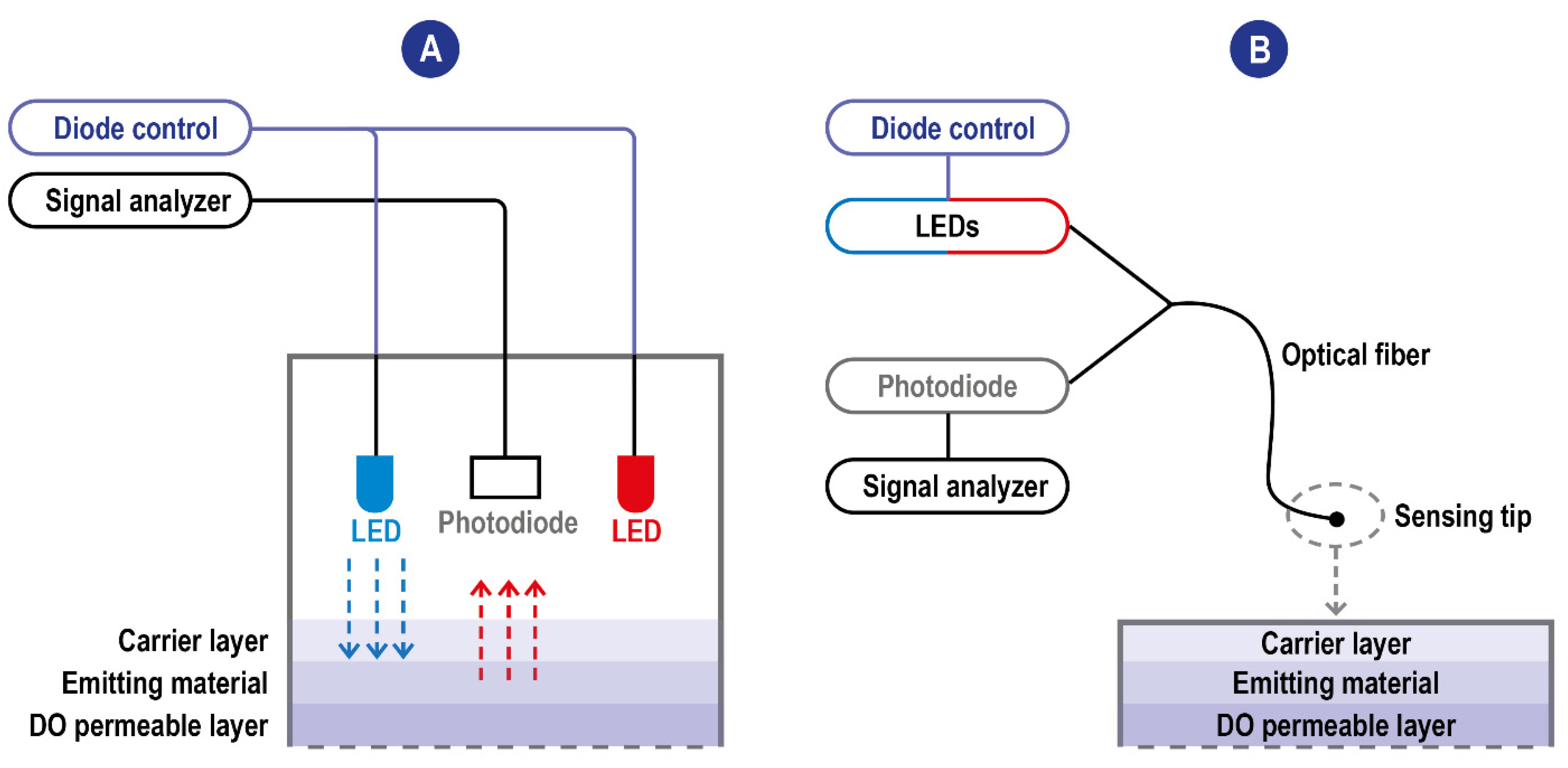
2.2. Optical Methods
2.2.1. Colorimetric Methods
2.2.2. Luminescence Methods
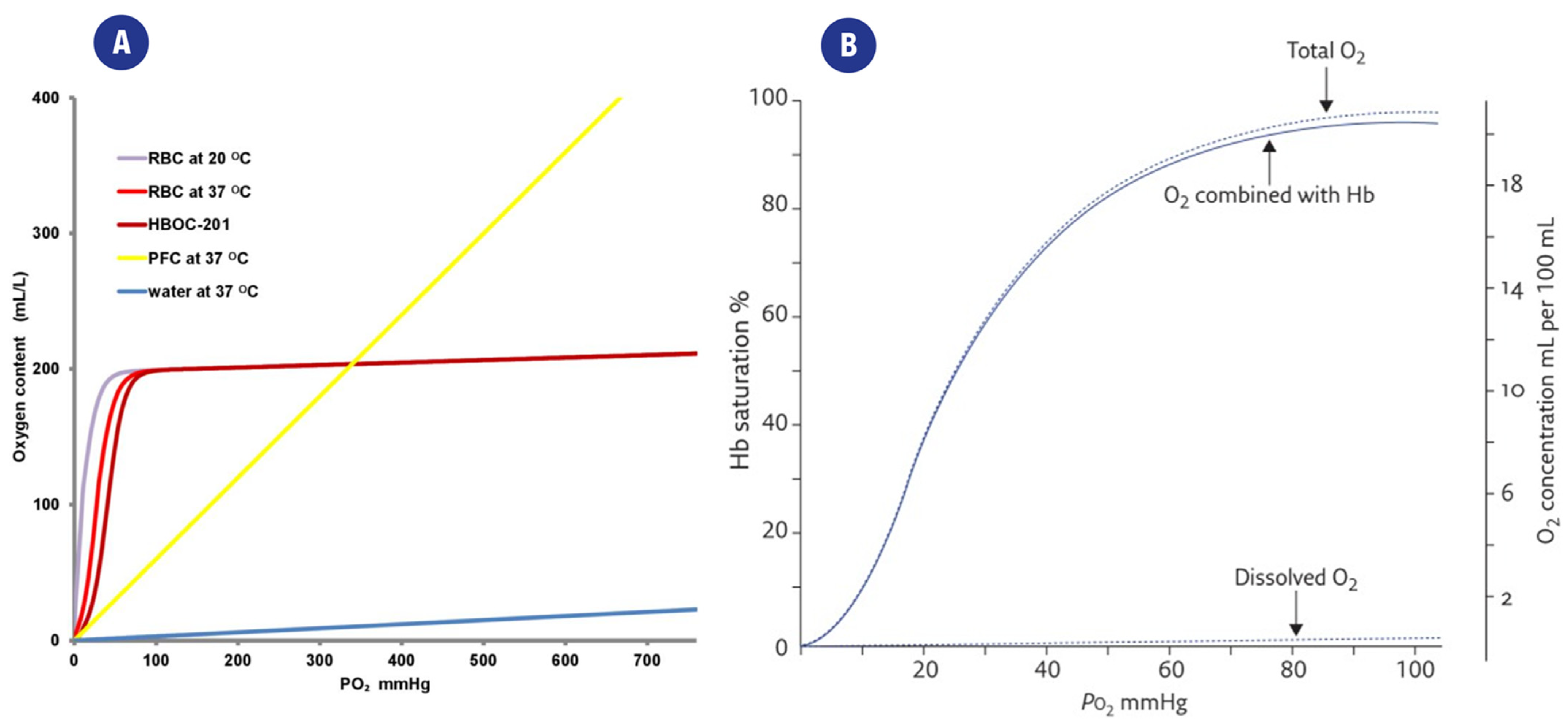
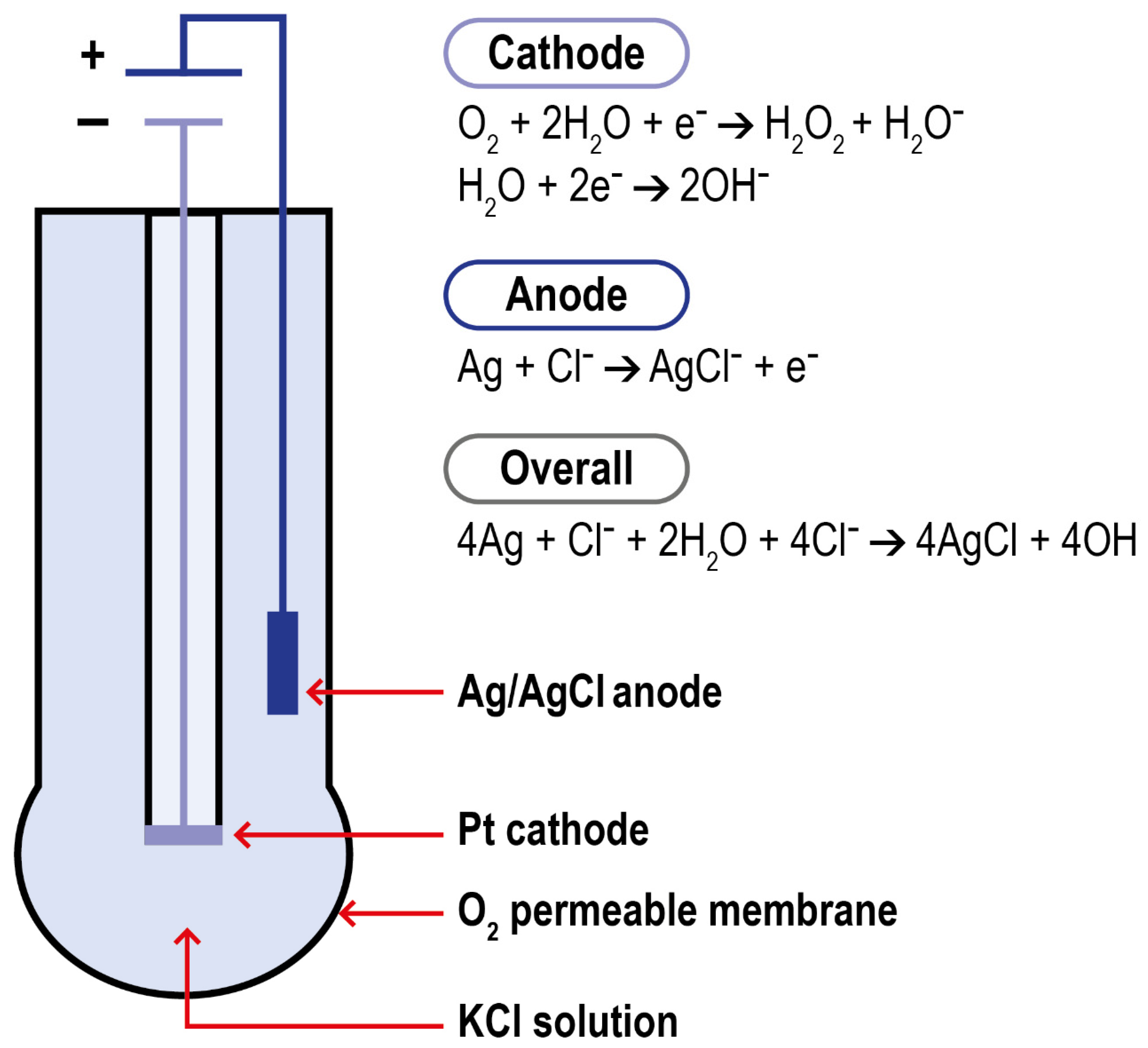
2.3. Electrochemical Methods
2.3.1. Polarographic Type Electrodes
2.3.2. Galvanic Type Electrodes
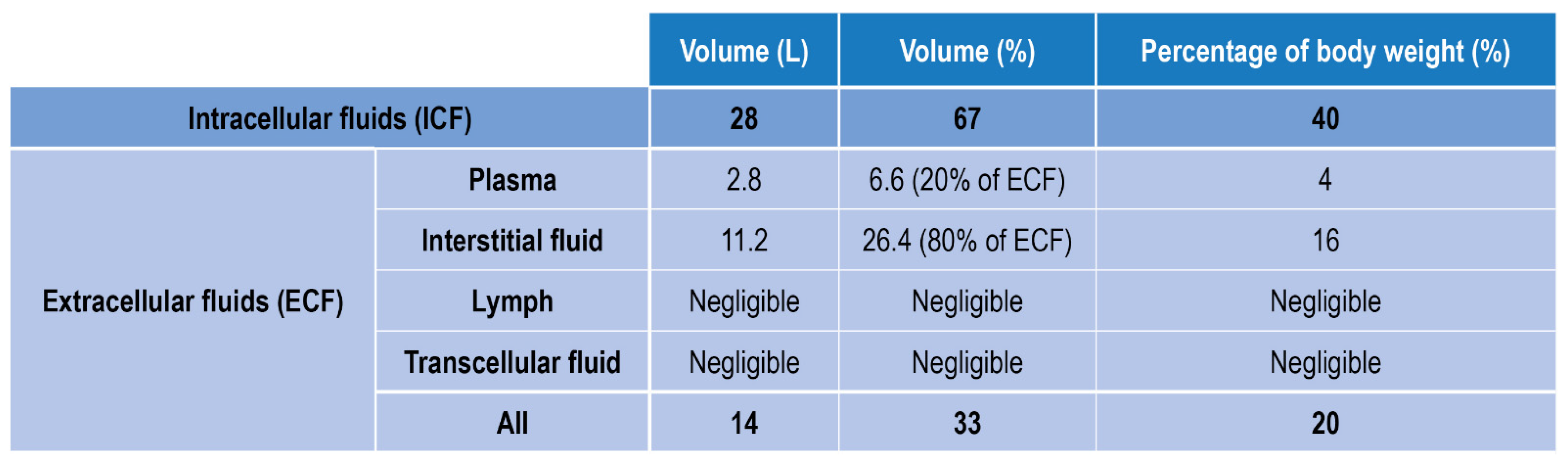
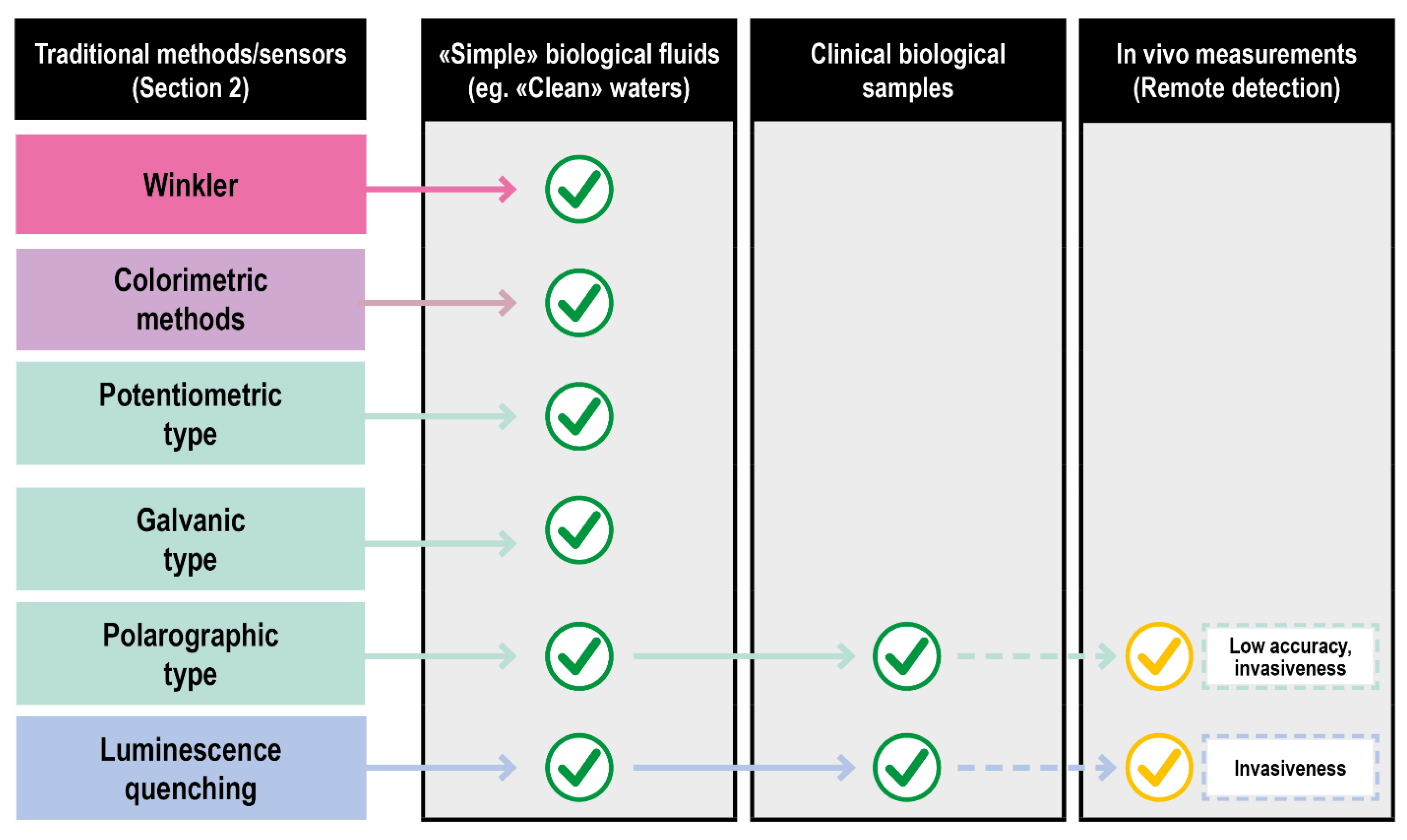
3. DO Detection in Biological Fluids and Physiological Environment
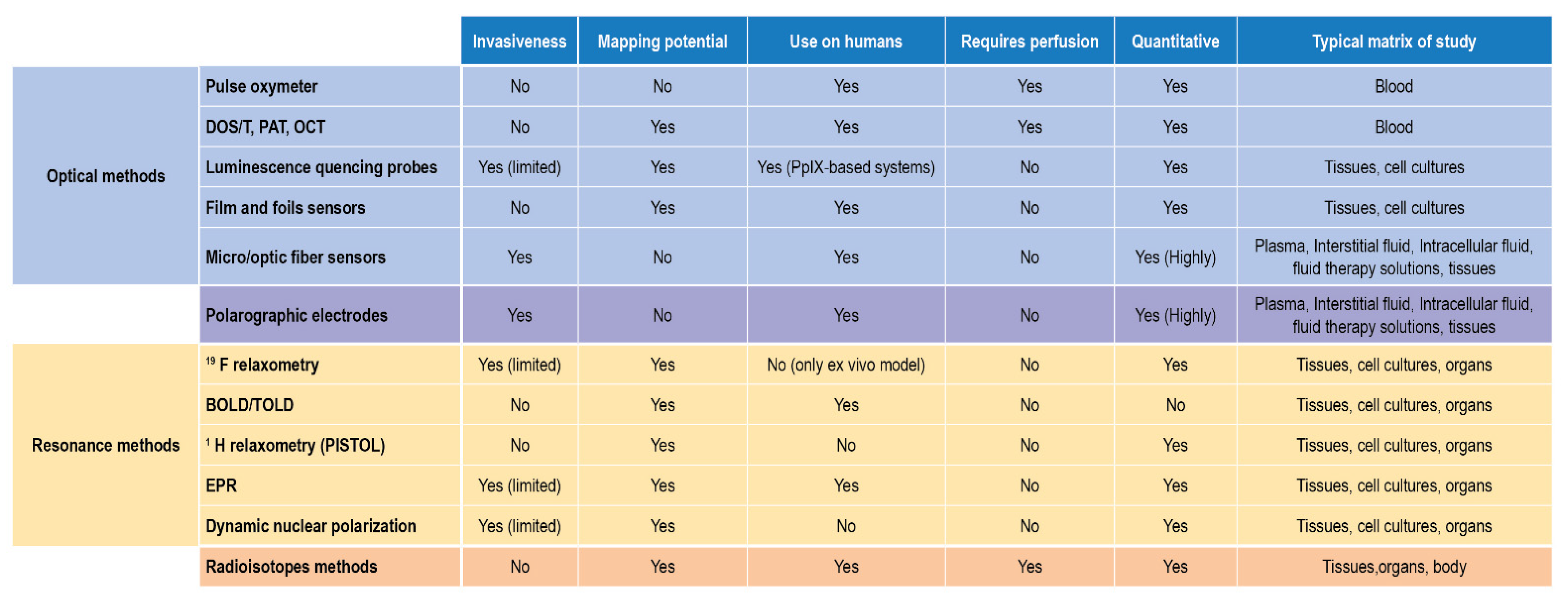
3.1. Translation of Classical Methods to Clinical Settings: Optical Methods
3.1.1. Class 1
3.1.2. Class 2
3.2. Translation of Classical Methods to Clinical Settings: Luminescence Quenching Imaging Probes
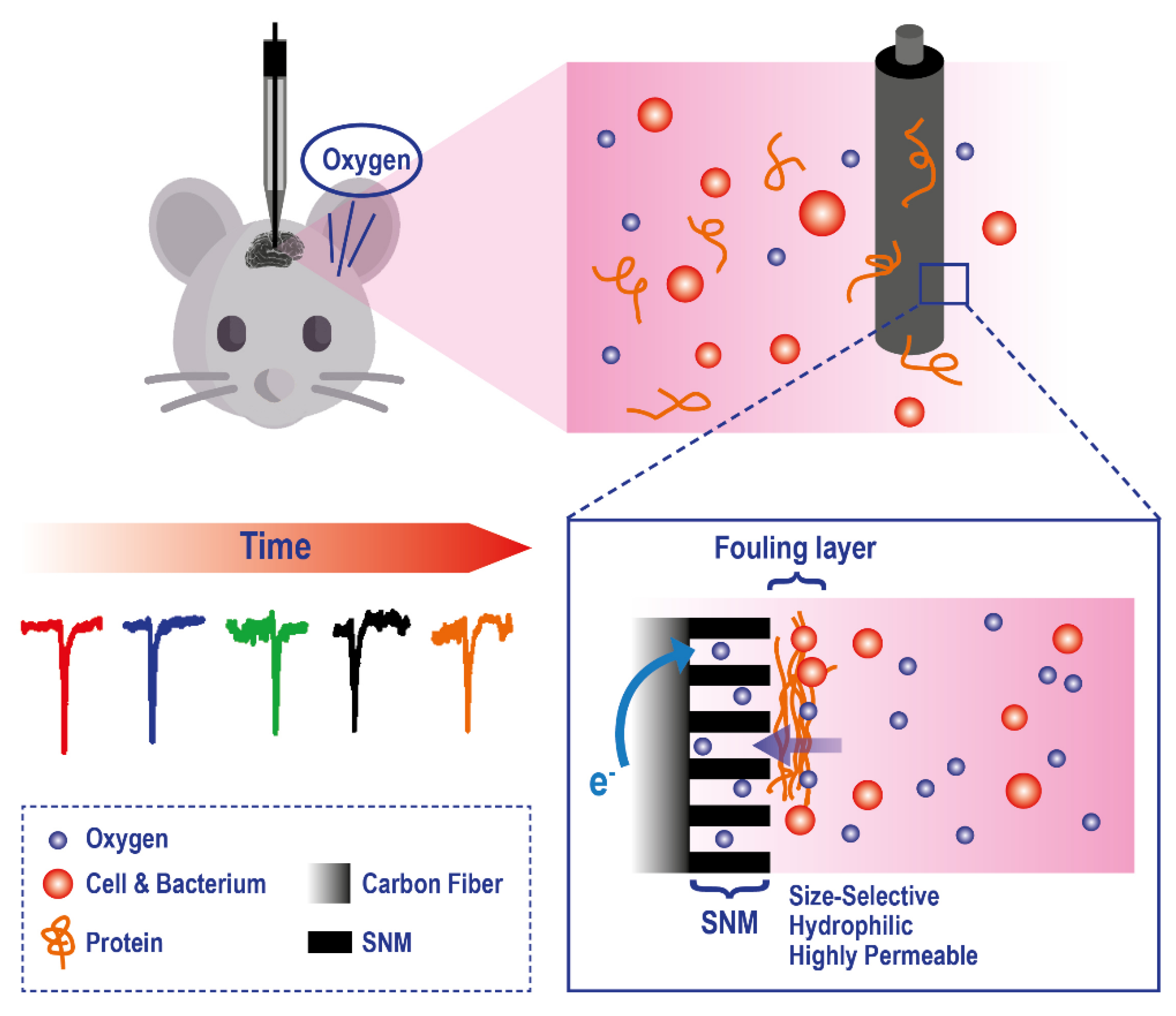
3.3. Translation of Classical Methods to Clinical Settings: Electrochemical Methods
3.4. Translation of Classical Methods to Clinical Settings: DO Sensing Techniques in Biomedicine
3.4.1. Radioisotopes Techniques
3.4.2. Resonance Techniques
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bartz, R.R.; Piantadosi, C.A. Clinical review: Oxygen as a signaling molecule. Crit. Care 2010, 14, 234. [Google Scholar] [CrossRef] [PubMed]
- Rolfe, D.F.; Brown, G.C. Cellular energy utilization and molecular origin of standard metabolic rate in mammals. Physiol. Rev. 1997, 77, 731–758. [Google Scholar] [CrossRef] [PubMed]
- Babcock, G.T. How oxygen is activated and reduced in respiration. Proc. Natl. Acad. Sci. USA 1999, 96, 12971–12973. [Google Scholar] [CrossRef] [PubMed]
- Carreau, A.; El Hafny-Rahbi, B.; Matejuk, A.; Grillon, C.; Kieda, C. Why is the partial oxygen pressure of human tissues a crucial parameter? Small molecules and hypoxia. J. Cell. Mol. Med. 2011, 15, 1239–1253. [Google Scholar] [CrossRef] [PubMed]
- McKeown, S.R. Defining normoxia, physoxia and hypoxia in tumours-implications for treatment response. Br. J. Radiol. 2014, 87, 20130676. [Google Scholar] [CrossRef]
- Roussakis, E.; Li, Z.; Nichols, A.J.; Evans, C.L. Oxygen-Sensing Methods in Biomedicine from the Macroscale to the Microscale. Angew. Chem. Int. Ed. 2015, 54, 8340–8362. [Google Scholar] [CrossRef]
- Sun, L.; Marin de Evsikova, C.; Bian, K.; Achille, A.; Telles, E.; Pei, H.; Seto, E. Programming and Regulation of Metabolic Homeostasis by HDAC11. EBioMedicine 2018, 33, 157–168. [Google Scholar] [CrossRef]
- Ratcliffe, P.J. Oxygen sensing and hypoxia signalling pathways in animals: The implications of physiology for cancer. J. Physiol. 2013, 591, 2027–2042. [Google Scholar] [CrossRef]
- Pischke, S.E.; Hyler, S.; Tronstad, C.; Bergsland, J.; Fosse, E.; Halvorsen, P.S.; Skulstad, H.; Tønnessen, T.I. Myocardial tissue CO2 tension detects coronary blood flow reduction after coronary artery bypass in real-time. Br. J. Anaesth. 2015, 114, 414–422. [Google Scholar] [CrossRef]
- Jordan, B.F.; Sonveaux, P. Targeting tumor perfusion and oxygenation to improve the outcome of anticancer therapy. Front Pharm. 2012, 3, 94. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Mach, W.J.; Thimmesch, A.R.; Pierce, J.T.; Pierce, J.D. Consequences of hyperoxia and the toxicity of oxygen in the lung. Nurs. Res. Pract. 2011, 2011, 260482. [Google Scholar] [CrossRef] [PubMed]
- Auten, R.L.; Davis, J.M. Oxygen Toxicity and Reactive Oxygen Species: The Devil Is in the Details. Pediatric Res. 2009, 66, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.; Shulaev, V.; Mittler, R. Reactive oxygen signaling and abiotic stress. Physiol. Plant. 2008, 133, 481–489. [Google Scholar] [CrossRef]
- Imlay, J.A. Pathways of oxidative damage. Annu. Rev. Microbiol. 2003, 57, 395–418. [Google Scholar] [CrossRef]
- Sherwood, L. Human Physiology: From Cells to Systems, 9th ed.; Brooks/Cole, Cengage Learning: Belmont, CA, USA, 2013. [Google Scholar]
- Giannakopoulos, X.; Evangelou, A.; Kalfakakou, V.; Grammeniatis, E.; Papandropoulos, I.; Charalambopoulos, K. Human bladder urine oxygen content: Implications for urinary tract diseases. Int. Urol. Nephrol. 1997, 29, 393–401. [Google Scholar] [CrossRef]
- Vadakedath, S.; Kandi, V. Dialysis: A Review of the Mechanisms Underlying Complications in the Management of Chronic Renal Failure. Cureus 2017, 9, e1603. [Google Scholar] [CrossRef]
- Smulowitz, P.B.; Serna, D.L.; Beckham, G.E.; Milliken, J.C. Ex vivo cardiac allograft preservation by continuous perfusion techniques. ASAIO J. 2000, 46, 389–396. [Google Scholar] [CrossRef]
- Kwiatkowski, A.; Wszola, M.; Kosieradzki, M.; Danielewicz, R.; Ostrowski, K.; Domagala, P.; Lisik, W.; Nosek, R.; Fesolowicz, S.; Trzebicki, J.; et al. Machine perfusion preservation improves renal allograft survival. Am. J. Transplant. Off. J. Am. Soc. Transplant. Am. Soc. Transpl. Surg. 2007, 7, 1942–1947. [Google Scholar] [CrossRef]
- Bodewes, S.B.; van Leeuwen, O.B.; Thorne, A.M.; Lascaris, B.; Ubbink, R.; Lisman, T.; Monbaliu, D.; De Meijer, V.E.; Nijsten, M.W.N.; Porte, R.J. Oxygen Transport during Ex Situ Machine Perfusion of Donor Livers Using Red Blood Cells or Artificial Oxygen Carriers. Int. J. Mol. Sci. 2021, 22, 235. [Google Scholar] [CrossRef]
- Severinghaus, J.W. Simple, accurate equations for human blood O2 dissociation computations. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1979, 46, 599–602. [Google Scholar] [CrossRef] [PubMed]
- Breuer, H.W.; Groeben, H.; Breuer, J.; Worth, H. Oxygen saturation calculation procedures: A critical analysis of six equations for the determination of oxygen saturation. Intensive Care Med. 1989, 15, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Singer, M.; Young, P.J.; Laffey, J.G.; Asfar, P.; Taccone, F.S.; Skrifvars, M.B.; Meyhoff, C.S.; Radermacher, P. Dangers of hyperoxia. Crit. Care 2021, 25, 440. [Google Scholar] [CrossRef] [PubMed]
- Marassi, V.; Giordani, S.; Reschiglian, P.; Roda, B.; Zattoni, A. Tracking Heme-Protein Interactions in Healthy and Pathological Human Serum in Native Conditions by Miniaturized FFF-Multidetection. Appl. Sci. 2022, 12, 6762. [Google Scholar] [CrossRef]
- Collins, J.A.; Rudenski, A.; Gibson, J.; Howard, L.; O’Driscoll, R. Relating oxygen partial pressure, saturation and content: The haemoglobin-oxygen dissociation curve. Breathe 2015, 11, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Papkovsky, D.B.; Dmitriev, R.I. Biological detection by optical oxygen sensing. Chem. Soc. Rev. 2013, 42, 8700–8732. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.T.; Heindel, T.J. A Review of Dissolved Oxygen Concentration Measurement Methods for Biological Fermentations. In Proceedings of the 2007 ASAE Annual Meeting, Minneapolis, MN, USA, 17–20 June 2007. [Google Scholar]
- Helm, I.; Jalukse, L.; Vilbaste, M.; Leito, I. Micro-Winkler titration method for dissolved oxygen concentration measurement. Anal. Chim. Acta 2009, 648, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Jiao, Y.; An, D.; Li, D.; Li, W.; Wei, Q. Review of Dissolved Oxygen Detection Technology: From Laboratory Analysis to Online Intelligent Detection. Sensors 2019, 19, 3995. [Google Scholar] [CrossRef] [PubMed]
- Wong, G.T.F. Removal of nitrite interference in the Winkler determination of dissolved oxygen in seawater. Mar. Chem. 2012, 130–131, 28–32. [Google Scholar] [CrossRef]
- Wong, G.T.F.; Li, K.-Y. Winkler’s method overestimates dissolved oxygen in seawater: Iodate interference and its oceanographic implications. Mar. Chem. 2009, 115, 86–91. [Google Scholar] [CrossRef]
- Null, S.E.; Mouzon, N.R.; Elmore, L.R. Dissolved oxygen, stream temperature, and fish habitat response to environmental water purchases. J. Environ. Manag. 2017, 197, 559–570. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Wang, Z.; Li, Y.; Zhuang, Q.; Gu, J. Real-Time Monitoring of Dissolved Oxygen with Inherent Oxygen-Sensitive Centers in Metal–Organic Frameworks. Chem. Mater. 2016, 28, 2652–2658. [Google Scholar] [CrossRef]
- Lehner, P.; Staudinger, C.; Borisov, S.M.; Klimant, I. Ultra-sensitive optical oxygen sensors for characterization of nearly anoxic systems. Nat. Commun. 2014, 5, 4460. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Liu, L.; Luo, T.; Hong, L.; Peng, X.; Austin, R.H.; Qu, J. A platinum-porphine/poly(perfluoroether) film oxygen tension sensor for noninvasive local monitoring of cellular oxygen metabolism using phosphorescence lifetime imaging. Sens. Actuators B Chem. 2018, 269, 88–95. [Google Scholar] [CrossRef]
- Müller, B.J.; Burger, T.; Borisov, S.M.; Klimant, I. High performance optical trace oxygen sensors based on NIR-emitting benzoporphyrins covalently coupled to silicone matrixes. Sens. Actuators B Chem. 2015, 216, 527–534. [Google Scholar] [CrossRef]
- Camas-Anzueto, J.L.; Gómez-Valdéz, J.A.; Meza-Gordillo, R.; Pérez-Patricio, M.; Hernández de León, H.R.; León-Orozco, V. Sensitive layer based on Lophine and calcium hydroxide for detection of dissolved oxygen in water. Measurement 2015, 68, 280–285. [Google Scholar] [CrossRef]
- Dmitriev, R.I.; Papkovsky, D.B. Optical probes and techniques for O2 measurement in live cells and tissue. Cell. Mol. Life Sci. 2012, 69, 2025–2039. [Google Scholar] [CrossRef]
- Shehata, N.; Meehan, K.; Ashry, I.; Kandas, I.; Xu, Y. Lanthanide-doped ceria nanoparticles as fluorescence-quenching probes for dissolved oxygen. Sens. Actuators B Chem. 2013, 183, 179–186. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, J.-M.; Li, S. Minreview: Recent advances in the development of gaseous and dissolved oxygen sensors. Instrum. Sci. Technol. 2019, 47, 19–50. [Google Scholar] [CrossRef]
- Sun, Z.; Cai, C.; Guo, F.; Ye, C.; Luo, Y.; Ye, S.; Luo, J.; Zhu, F.; Jiang, C. Oxygen sensitive polymeric nanocapsules for optical dissolved oxygen sensors. Nanotechnology 2018, 29, 145704. [Google Scholar] [CrossRef]
- Pensieri, S.; Bozzano, R.; Schiano, M.E.; Ntoumas, M.; Potiris, E.; Frangoulis, C.; Podaras, D.; Petihakis, G. Methods and Best Practice to Intercompare Dissolved Oxygen Sensors and Fluorometers/Turbidimeters for Oceanographic Applications. Sensors 2016, 16, 702. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, T.O. Luminescence Based Measurement of Dissolved Oxygen in Natural Waters; Hach Company: Loveland, CO, USA, 2006. [Google Scholar]
- Holst, G.; Grunwald, B. Luminescence lifetime imaging with transparent oxygen optodes. Sens. Actuators B Chem. 2001, 74, 78–90. [Google Scholar] [CrossRef]
- Xiong, X.; Xiao, D.; Choi, M.M.F. Dissolved oxygen sensor based on fluorescence quenching of oxygen-sensitive ruthenium complex immobilized on silica–Ni–P composite coating. Sens. Actuators B Chem. 2006, 117, 172–176. [Google Scholar] [CrossRef]
- Chu, C.-S.; Lo, Y.-L. Optical fiber dissolved oxygen sensor based on Pt(II) complex and core-shell silica nanoparticles incorporated with sol–gel matrix. Sens. Actuators B Chem. 2010, 151, 83–89. [Google Scholar] [CrossRef]
- Li, X.-M.; Ruan, F.-C.; Wong, K.-Y. Optical characteristics of a ruthenium(II) complex immobilized in a silicone rubber film for oxygen measurement. Analyst 1993, 118, 289–292. [Google Scholar] [CrossRef]
- Choi, M.M.F.; Xiao, D. Single standard calibration for an optical oxygen sensor based on luminescence quenching of a ruthenium complex. Anal. Chim. Acta 2000, 403, 57–65. [Google Scholar] [CrossRef]
- Lee, Y.C.; Cheng, S.W.; Cheng, C.L.; Fang, W. Design and implementation of gas sensor array based on fluorescence quenching detection using CMOS-MEMS process. In Proceedings of the 2017 19th International Conference on Solid-State Sensors, Actuators and Microsystems (TRANSDUCERS), Kaohsiung, Taiwan, 18–22 June 2017; pp. 672–675. [Google Scholar]
- Chen, R.; Fioroni, G.; McPeak, H.; Hahn, C.E.W.; Farmery, A.D. Chariacteristics of carbon nanotube based nanocomposite oxygen sensing matrices. In Proceedings of the 2016 IEEE SENSORS, Orlando, FL, USA, 30 October–3 November 2016; pp. 1–3. [Google Scholar]
- Chakravorty, K.; Poole, J.A. The effect of dissolved molecular oxygen on the fluorescence of 9,10-dimethylanthracene and 9,10-diphenylanthracene. J. Photochem. 1984, 26, 25–31. [Google Scholar] [CrossRef]
- Välimäki, H.; Verho, J.; Kreutzer, J.; Kattipparambil Rajan, D.; Ryynänen, T.; Pekkanen-Mattila, M.; Ahola, A.; Tappura, K.; Kallio, P.; Lekkala, J. Fluorimetric oxygen sensor with an efficient optical read-out for in vitro cell models. Sens. Actuators B Chem. 2017, 249, 738–746. [Google Scholar] [CrossRef]
- Jin, P.; Chu, J.; Miao, Y.; Tan, J.; Zhang, S.; Zhu, W. A NIR luminescent copolymer based on platinum porphyrin as high permeable dissolved oxygen sensor for microbioreactors. AIChE J. 2013, 59, 2743–2752. [Google Scholar] [CrossRef]
- Peng, L.R.; Yang, X.H.; Zhang, J.; Dai, L.; Ma, Z.L.; Peng, S.J. Fluorescence Detection Method of Dissolved Oxygen Based on Fiber. Chin. J. Lumin. 2014, 35, 382–386. [Google Scholar] [CrossRef]
- Pospíšilová, M.; Kuncová, G.; Trögl, J. Fiber-Optic Chemical Sensors and Fiber-Optic Bio-Sensors. Sensors 2015, 15, 25208–25259. [Google Scholar] [CrossRef] [PubMed]
- Zolkapli, M.; Mahmud, Z.; Herman, S.H.; Abdullah, W.F.H.; Noorl, U.M.; Saharudin, S. Fluorescence characteristic of ruthenium nanoparticles as a dissolved oxygen sensing material in gas and aqueos phase. In Proceedings of the 2014 IEEE 10th International Colloquium on Signal Processing and its Applications, Kuala Lumpur, MA, USA, 7–9 March 2014; pp. 195–198. [Google Scholar]
- Chu, C.-S.; Chuang, C.-Y. Ratiometric optical fiber dissolved oxygen sensor based on metalloporphyrin and CdSe quantum dots embedded in sol–gel matrix. J. Lumin. 2015, 167, 114–119. [Google Scholar] [CrossRef]
- Xu, H.; Aylott, J.W.; Kopelman, R.; Miller, T.J.; Philbert, M.A. A Real-Time Ratiometric Method for the Determination of Molecular Oxygen Inside Living Cells Using Sol−Gel-Based Spherical Optical Nanosensors with Applications to Rat C6 Glioma. Anal. Chem. 2001, 73, 4124–4133. [Google Scholar] [CrossRef] [PubMed]
- Park, E.J.; Reid, K.R.; Tang, W.; Kennedy, R.T.; Kopelman, R. Ratiometric fiber optic sensors for the detection of inter- and intra-cellular dissolved oxygen. J. Mater. Chem. 2005, 15, 2913–2919. [Google Scholar] [CrossRef]
- Jiang, Z.; Yu, X.; Zhai, S.; Hao, Y. Ratiometric Dissolved Oxygen Sensors Based on Ruthenium Complex Doped with Silver Nanoparticles. Sensors 2017, 17, 548. [Google Scholar] [CrossRef]
- Mao, Y.; Gao, Y.; Wu, S.; Wu, S.; Shi, J.; Zhou, B.; Tian, Y. Highly enhanced sensitivity of optical oxygen sensors using microstructured PtTFPP/PDMS-pillar arrays sensing layer. Sens. Actuators B Chem. 2017, 251, 495–502. [Google Scholar] [CrossRef]
- Navarro-Villoslada, F.; Orellana, G.; Moreno-Bondi, M.C.; Vick, T.; Driver, M.; Hildebrand, G.; Liefeith, K. Fiber-Optic Luminescent Sensors with Composite Oxygen-Sensitive Layers and Anti-Biofouling Coatings. Anal. Chem. 2001, 73, 5150–5156. [Google Scholar] [CrossRef]
- Bambot, S.B.; Holavanahali, R.; Lakowicz, J.R.; Carter, G.M.; Rao, G. Phase fluorometric sterilizable optical oxygen sensor. Biotechnol. Bioeng. 1994, 43, 1139–1145. [Google Scholar] [CrossRef]
- Bukowski, R.M.; Ciriminna, R.; Pagliaro, M.; Bright, F.V. High-Performance Quenchometric Oxygen Sensors Based on Fluorinated Xerogels Doped with [Ru(dpp)3]2+. Anal. Chem. 2005, 77, 2670–2672. [Google Scholar] [CrossRef]
- Silva, E.L.; Bastos, A.C.; Neto, M.A.; Silva, R.F.; Zheludkevich, M.L.; Ferreira, M.G.S.; Oliveira, F.J. Boron doped nanocrystalline diamond microelectrodes for the detection of Zn2+ and dissolved O2. Electrochim. Acta 2012, 76, 487–494. [Google Scholar] [CrossRef]
- Martı́nez-Máñez, R.; Soto, J.; Garcia-Breijo, E.; Gil, L.; Ibáñez, J.; Llobet, E. An “electronic tongue” design for the qualitative analysis of natural waters. Sens. Actuators B Chem. 2005, 104, 302–307. [Google Scholar] [CrossRef]
- Zhuiykov, S.; Kalantar-zadeh, K. Development of antifouling of electrochemical solid-state dissolved oxygen sensors based on nanostructured Cu0.4Ru3.4O7+RuO2 sensing electrodes. Electrochim. Acta 2012, 73, 105–111. [Google Scholar] [CrossRef]
- Zhang, D.; Fang, Y.; Miao, Z.; Ma, M.; Chen, Q. Electrochemical determination of dissolved oxygen based on three dimensional electrosynthesis of silver nanodendrites electrode. J. Appl. Electrochem. 2014, 44, 419–425. [Google Scholar] [CrossRef]
- Fu, L.I.; Zheng, Y.; Fu, Z.; Wang, A.; Cai, W.E.N. Dissolved oxygen detection by galvanic displacement-induced graphene/silver nanocomposite. Bull. Mater. Sci. 2015, 38, 611–616. [Google Scholar] [CrossRef]
- Damos, F.S.; Luz, R.C.S.; Tanaka, A.A.; Kubota, L.T. Dissolved oxygen amperometric sensor based on layer-by-layer assembly using host–guest supramolecular interactions. Anal. Chim. Acta 2010, 664, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Guan, J.; Lv, B.; Zhou, Z.; Hou, X.; Xiao, D. An Oxygen Sensor Based on Ru(bpy)2+3/Agar Gel Modified Electrode for the Determination Dissolved Oxygen in Organic Solvents. Sens. Lett. 2006, 4, 455–459. [Google Scholar] [CrossRef]
- Zheng, R.-J.; Fang, Y.-M.; Qin, S.-F.; Song, J.; Wu, A.-H.; Sun, J.-J. A dissolved oxygen sensor based on hot electron induced cathodic electrochemiluminescence at a disposable CdS modified screen-printed carbon electrode. Sens. Actuators B Chem. 2011, 157, 488–493. [Google Scholar] [CrossRef]
- Glasspool, W.; Atkinson, J. A screen-printed amperometric dissolved oxygen sensor utilising an immobilised electrolyte gel and membrane. Sens. Actuators B Chem. 1998, 48, 308–317. [Google Scholar] [CrossRef]
- Wang, P.; Liu, Y.; Abruña, H.D.; Spector, J.A.; Olbricht, W.L. Micromachined dissolved oxygen sensor based on solid polymer electrolyte. Sens. Actuators B Chem. 2011, 153, 145–151. [Google Scholar] [CrossRef]
- Lee, H.-j.; Kim, H.-M.; Park, J.-H.; Lee, S.-K. Fabrication and characterization of micro dissolved oxygen sensor activated on demand using electrolysis. Sens. Actuators B Chem. 2017, 241, 923–930. [Google Scholar] [CrossRef]
- Durduran, T.; Choe, R.; Baker, W.B.; Yodh, A.G. Diffuse Optics for Tissue Monitoring and Tomography. Rep. Prog. Phys. Phys. Soc. 2010, 73, 076701. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.V. Prospects of photoacoustic tomography. Med. Phys. 2008, 35, 5758–5767. [Google Scholar] [CrossRef] [PubMed]
- Yi, J.; Wei, Q.; Liu, W.; Backman, V.; Zhang, H.F. Visible-light optical coherence tomography for retinal oximetry. Opt. Lett. 2013, 38, 1796–1798. [Google Scholar] [CrossRef] [PubMed]
- Pretto, J.J.; Roebuck, T.; Beckert, L.; Hamilton, G. Clinical use of pulse oximetry: Official guidelines from the Thoracic Society of Australia and New Zealand. Respirology 2014, 19, 38–46. [Google Scholar] [CrossRef]
- Driscoll, B.R.; Howard, L.S.; Earis, J.; Mak, V. British Thoracic Society Guideline for oxygen use in adults in healthcare and emergency settings. BMJ Open Respir. Res. 2017, 4, e000170. [Google Scholar] [CrossRef]
- Villringer, A.; Chance, B. Non-invasive optical spectroscopy and imaging of human brain function. Trends Neurosci. 1997, 20, 435–442. [Google Scholar] [CrossRef]
- Lu, C.-W.; Lee, C.-K.; Tsai, M.-T.; Wang, Y.-M.; Yang, C.C. Measurement of the hemoglobin oxygen saturation level with spectroscopic spectral-domain optical coherence tomography. Opt. Lett. 2008, 33, 416–418. [Google Scholar] [CrossRef]
- Kuranov, R.V.; Qiu, J.; McElroy, A.B.; Estrada, A.; Salvaggio, A.; Kiel, J.; Dunn, A.K.; Duong, T.Q.; Milner, T.E. Depth-resolved blood oxygen saturation measurement by dual-wavelength photothermal (DWP) optical coherence tomography. Biomed. Opt. Express 2011, 2, 491–504. [Google Scholar] [CrossRef][Green Version]
- Wang, X.-D.; Wolfbeis, O.S. Optical methods for sensing and imaging oxygen: Materials, spectroscopies and applications. Chem. Soc. Rev. 2014, 43, 3666–3761. [Google Scholar] [CrossRef]
- Woods, R.J.; Scypinski, S.; Love, L.J.C. Transient digitizer for the determination of microsecond luminescence lifetimes. Anal. Chem. 1984, 56, 1395–1400. [Google Scholar] [CrossRef]
- Vinogradov, S.A.; Fernandez-Seara, M.A.; Dupan, B.W.; Wilson, D.F. A method for measuring oxygen distributions in tissue using frequency domain phosphorometry. Comp Biochem Physiol A Mol. Integr Physiol 2002, 132, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Dobrucki, J.W. Interaction of oxygen-sensitive luminescent probes Ru(phen)32+ and Ru(bipy)32+ with animal and plant cells in vitro: Mechanism of phototoxicity and conditions for non-invasive oxygen measurements. J. Photochem. Photobiol. B Biol. 2001, 65, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Paul, U.; Mary-Ann, M. Imaging fluorescence lifetime modulation of a ruthenium-based dye in living cells: The potential for oxygen sensing. J. Phys. D Appl. Phys. 2003, 36, 1689. [Google Scholar] [CrossRef]
- Liu, S.; Wei, L.; Guo, S.; Jiang, J.; Zhang, P.; Han, J.; Ma, Y.; Zhao, Q. Anionic iridium(III) complexes and their conjugated polymer soft salts for time-resolved luminescent detection of intracellular oxygen levels. Sens. Actuators B Chem. 2018, 262, 436–443. [Google Scholar] [CrossRef]
- Zhang, K.Y.; Gao, P.; Sun, G.; Zhang, T.; Li, X.; Liu, S.; Zhao, Q.; Lo, K.K.-W.; Huang, W. Dual-Phosphorescent Iridium(III) Complexes Extending Oxygen Sensing from Hypoxia to Hyperoxia. J. Am. Chem. Soc. 2018, 140, 7827–7834. [Google Scholar] [CrossRef] [PubMed]
- Esipova, T.V.; Barrett, M.J.P.; Erlebach, E.; Masunov, A.E.; Weber, B.; Vinogradov, S.A. Oxyphor 2P: A High-Performance Probe for Deep-Tissue Longitudinal Oxygen Imaging. Cell Metab. 2019, 29, 736–744.e737. [Google Scholar] [CrossRef]
- Ma, X.; Tian, H. Photochemistry and Photophysics. Concepts, Research, Applications. By Vincenzo Balzani, Paola Ceroni and Alberto Juris. Angew. Chem. Int. Ed. 2014, 53, 8817. [Google Scholar] [CrossRef]
- Zhu, H.; Li, Q.; Shi, B.; Ge, F.; Liu, Y.; Mao, Z.; Zhu, H.; Wang, S.; Yu, G.; Huang, F.; et al. Dual-Emissive Platinum(II) Metallacage with a Sensitive Oxygen Response for Imaging of Hypoxia and Imaging-Guided Chemotherapy. Angew. Chem. Int. Ed. 2020, 59, 20208–20214. [Google Scholar] [CrossRef]
- Hiesinger, W.; Vinogradov, S.A.; Atluri, P.; Fitzpatrick, J.R.; Frederick, J.R.; Levit, R.D.; McCormick, R.C.; Muenzer, J.R.; Yang, E.C.; Marotta, N.A.; et al. Oxygen-dependent quenching of phosphorescence used to characterize improved myocardial oxygenation resulting from vasculogenic cytokine therapy. J. Appl. Physiol. 2011, 110, 1460–1465. [Google Scholar] [CrossRef][Green Version]
- Esipova, T.V.; Karagodov, A.; Miller, J.; Wilson, D.F.; Busch, T.M.; Vinogradov, S.A. Two New “Protected” Oxyphors for Biological Oximetry: Properties and Application in Tumor Imaging. Anal. Chem. 2011, 83, 8756–8765. [Google Scholar] [CrossRef]
- Nichols, A.J.; Roussakis, E.; Klein, O.J.; Evans, C.L. Click-Assembled, Oxygen-Sensing Nanoconjugates for Depth-Resolved, Near-Infrared Imaging in a 3 D Cancer Model. Angew. Chem. Int. Ed. 2014, 53, 3671–3674. [Google Scholar] [CrossRef] [PubMed]
- Finikova, O.S.; Chen, P.; Ou, Z.; Kadish, K.M.; Vinogradov, S.A. Dynamic Quenching of Porphyrin Triplet States by Two-Photon Absorbing Dyes: Towards Two-Photon-Enhanced Oxygen Nanosensors. J. Photochem Photobiol A Chem 2008, 198, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Lebedev, A.Y.; Troxler, T.; Vinogradov, S.A. Design of metalloporphyrin-based dendritic nanoprobes for two-photon microscopy of oxygen. J. Porphyr. Phthalocyanines 2008, 12, 1261–1269. [Google Scholar] [CrossRef] [PubMed]
- Roussakis, E.; Spencer, J.A.; Lin, C.P.; Vinogradov, S.A. Two-Photon Antenna-Core Oxygen Probe with Enhanced Performance. Anal. Chem. 2014, 86, 5937–5945. [Google Scholar] [CrossRef]
- Lecoq, J.; Parpaleix, A.; Roussakis, E.; Ducros, M.; Houssen, Y.G.; Vinogradov, S.A.; Charpak, S. Simultaneous two-photon imaging of oxygen and blood flow in deep cerebral vessels. Nat. Med. 2011, 17, 893–898. [Google Scholar] [CrossRef]
- Park, J.; Lee, J.; Kwag, J.; Baek, Y.; Kim, B.; Yoon, C.J.; Bok, S.; Cho, S.-H.; Kim, K.H.; Ahn, G.O.; et al. Quantum Dots in an Amphiphilic Polyethyleneimine Derivative Platform for Cellular Labeling, Targeting, Gene Delivery, and Ratiometric Oxygen Sensing. ACS Nano 2015, 9, 6511–6521. [Google Scholar] [CrossRef]
- Amelia, M.; Lavie-Cambot, A.; McClenaghan, N.D.; Credi, A. A ratiometric luminescent oxygen sensor based on a chemically functionalized quantum dot. Chem. Commun. 2011, 47, 325–327. [Google Scholar] [CrossRef]
- Lin, F.; Li, F.; Lai, Z.; Cai, Z.; Wang, Y.; Wolfbeis, O.S.; Chen, X. MnII-Doped Cesium Lead Chloride Perovskite Nanocrystals: Demonstration of Oxygen Sensing Capability Based on Luminescent Dopants and Host-Dopant Energy Transfer. ACS Appl. Mater. Interfaces 2018, 10, 23335–23343. [Google Scholar] [CrossRef]
- McKinlay, A.C.; Morris, R.E.; Horcajada, P.; Férey, G.; Gref, R.; Couvreur, P.; Serre, C. BioMOFs: Metal–Organic Frameworks for Biological and Medical Applications. Angew. Chem. Int. Ed. 2010, 49, 6260–6266. [Google Scholar] [CrossRef]
- Xu, R.; Wang, Y.; Duan, X.; Lu, K.; Micheroni, D.; Hu, A.; Lin, W. Nanoscale Metal–Organic Frameworks for Ratiometric Oxygen Sensing in Live Cells. J. Am. Chem. Soc. 2016, 138, 2158–2161. [Google Scholar] [CrossRef]
- Wang, X.-d.; Gorris, H.H.; Stolwijk, J.A.; Meier, R.J.; Groegel, D.B.M.; Wegener, J.; Wolfbeis, O.S. Self-referenced RGB colour imaging of intracellular oxygen. Chem. Sci. 2011, 2, 901–906. [Google Scholar] [CrossRef]
- Mik, E.G.; Johannes, T.; Zuurbier, C.J.; Heinen, A.; Houben-Weerts, J.H.P.M.; Balestra, G.M.; Stap, J.; Beek, J.F.; Ince, C. In Vivo Mitochondrial Oxygen Tension Measured by a Delayed Fluorescence Lifetime Technique. Biophys. J. 2008, 95, 3977–3990. [Google Scholar] [CrossRef] [PubMed]
- Bodmer, S.I.A.; Balestra, G.M.; Harms, F.A.; Johannes, T.; Raat, N.J.H.; Stolker, R.J.; Mik, E.G. Microvascular and mitochondrial PO2 simultaneously measured by oxygen-dependent delayed luminescence. J. Biophotonics 2012, 5, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Hynes, J.; Floyd, S.; Soini, A.E.; O’Connor, R.; Papkovsky, D.B. Fluorescence-based cell viability screening assays using water-soluble oxygen probes. J. Biomol. Screen. 2003, 8, 264–272. [Google Scholar] [CrossRef]
- O’Donovan, C.; Hynes, J.; Yashunski, D.; Papkovsky, D.B. Phosphorescent oxygen-sensitive materials for biological applications. J. Mater. Chem. 2005, 15, 2946–2951. [Google Scholar] [CrossRef]
- O’Riordan, T.C.; Fitzgerald, K.; Ponomarev, G.V.; Mackrill, J.; Hynes, J.; Taylor, C.; Papkovsky, D.B. Sensing intracellular oxygen using near-infrared phosphorescent probes and live-cell fluorescence imaging. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2007, 292, R1613–R1620. [Google Scholar] [CrossRef]
- Dmitriev, R.I.; Zhdanov, A.V.; Ponomarev, G.V.; Yashunski, D.V.; Papkovsky, D.B. Intracellular oxygen-sensitive phosphorescent probes based on cell-penetrating peptides. Anal. Biochem. 2010, 398, 24–33. [Google Scholar] [CrossRef]
- Koren, K.; Dmitriev, R.I.; Borisov, S.M.; Papkovsky, D.B.; Klimant, I. Complexes of IrIII-Octaethylporphyrin with Peptides as Probes for Sensing Cellular O2. ChemBioChem 2012, 13, 1184–1190. [Google Scholar] [CrossRef]
- Dmitriev, R.I.; Kondrashina, A.V.; Koren, K.; Klimant, I.; Zhdanov, A.V.; Pakan, J.M.P.; McDermott, K.W.; Papkovsky, D.B. Small molecule phosphorescent probes for O2 imaging in 3D tissue models. Biomater. Sci. 2014, 2, 853–866. [Google Scholar] [CrossRef]
- Patrycja, N.-S.; Eddy, F.; Hubert van den, B.; Georges, W. In vitro and in vivo studies of new photoluminescent oxygen sensors for non-invasive intravascular pO2 measurements. In Photodynamic Therapy: Back to the Future; SPIE: Bellingham, WA, USA, 2009; Volume 7380, pp. 1011–1021. [Google Scholar]
- Xu, S.; Yu, Y.; Gao, Y.; Zhang, Y.; Li, X.; Zhang, J.; Wang, Y.; Chen, B. Mesoporous silica coating NaYF4:Yb,Er@NaYF4 upconversion nanoparticles loaded with ruthenium(II) complex nanoparticles: Fluorometric sensing and cellular imaging of temperature by upconversion and of oxygen by downconversion. Microchim. Acta 2018, 185, 454. [Google Scholar] [CrossRef]
- Ray, A.; Kopelman, R. Hydrogel nanosensors for biophotonic imaging of chemical analytes. Nanomedicine 2013, 8, 1829–1838. [Google Scholar] [CrossRef] [PubMed]
- Ashokkumar, P.; Adarsh, N.; Klymchenko, A.S. Ratiometric Nanoparticle Probe Based on FRET-Amplified Phosphorescence for Oxygen Sensing with Minimal Phototoxicity. Small 2020, 16, 2002494. [Google Scholar] [CrossRef]
- Hao, C.; Wu, X.; Sun, M.; Zhang, H.; Yuan, A.; Xu, L.; Xu, C.; Kuang, H. Chiral Core–Shell Upconversion Nanoparticle@MOF Nanoassemblies for Quantification and Bioimaging of Reactive Oxygen Species in Vivo. J. Am. Chem. Soc. 2019, 141, 19373–19378. [Google Scholar] [CrossRef] [PubMed]
- Lyu, Y.; Pu, K. Recent Advances of Activatable Molecular Probes Based on Semiconducting Polymer Nanoparticles in Sensing and Imaging. Adv. Sci. 2017, 4, 1600481. [Google Scholar] [CrossRef] [PubMed]
- Kellner, K.; Liebsch, G.; Klimant, I.; Wolfbeis, O.S.; Blunk, T.; Schulz, M.B.; Göpferich, A. Determination of oxygen gradients in engineered tissue using a fluorescent sensor. Biotechnol. Bioeng. 2002, 80, 73–83. [Google Scholar] [CrossRef]
- Tsytsarev, V.; Akkentli, F.; Pumbo, E.; Tang, Q.; Chen, Y.; Erzurumlu, R.S.; Papkovsky, D.B. Planar implantable sensor for in vivo measurement of cellular oxygen metabolism in brain tissue. J. Neurosci. Methods 2017, 281, 1–6. [Google Scholar] [CrossRef]
- Koolen, P.G.L.; Li, Z.; Roussakis, E.; Paul, M.A.; Ibrahim, A.M.S.; Matyal, R.; Huang, T.; Evans, C.L.; Lin, S.J. Oxygen-Sensing Paint-On Bandage: Calibration of a Novel Approach in Tissue Perfusion Assessment. Plast. Reconstr. Surg. 2017, 140, 89–96. [Google Scholar] [CrossRef]
- Schreml, S.; Meier, R.J.; Kirschbaum, M.; Kong, S.C.; Gehmert, S.; Felthaus, O.; Küchler, S.; Sharpe, J.R.; Wöltje, K.; Weiß, K.T.; et al. Luminescent Dual Sensors Reveal Extracellular pH-Gradients and Hypoxia on Chronic Wounds That Disrupt Epidermal Repair. Theranostics 2014, 4, 721–735. [Google Scholar] [CrossRef]
- Yazgan, G.; Dmitriev, R.I.; Tyagi, V.; Jenkins, J.; Rotaru, G.-M.; Rottmar, M.; Rossi, R.M.; Toncelli, C.; Papkovsky, D.B.; Maniura-Weber, K.; et al. Steering surface topographies of electrospun fibers: Understanding the mechanisms. Sci. Rep. 2017, 7, 158. [Google Scholar] [CrossRef]
- Xue, R.; Nelson, M.T.; Teixeira, S.A.; Viapiano, M.S.; Lannutti, J.J. Cancer cell aggregate hypoxia visualized in vitro via biocompatible fiber sensors. Biomaterials 2016, 76, 208–217. [Google Scholar] [CrossRef]
- Chisholm, K.I.; Ida, K.K.; Davies, A.L.; Tachtsidis, I.; Papkovsky, D.B.; Dyson, A.; Singer, M.; Duchen, M.R.; Smith, K.J. Hypothermia protects brain mitochondrial function from hypoxemia in a murine model of sepsis. J. Cereb. Blood Flow Metab. Off. J. Int. Soc. Cereb. Blood Flow Metab. 2016, 36, 1955–1964. [Google Scholar] [CrossRef] [PubMed]
- Babilas, P.; Liebsch, G.; Schacht, V.; Klimant, I.; Wolfbeis, O.S.; Szeimies, R.M.; Abels, C. In vivo phosphorescence imaging of pO2 using planar oxygen sensors. Microcirculation 2005, 12, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Schreml, S.; Meier, R.J.; Wolfbeis, O.S.; Maisch, T.; Szeimies, R.M.; Landthaler, M.; Regensburger, J.; Santarelli, F.; Klimant, I.; Babilas, P. 2D luminescence imaging of physiological wound oxygenation. Exp. Dermatol. 2011, 20, 550–554. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, J.; Meier, R.J.; Mahnke, A.; Schatz, V.; Brackmann, F.; Trollmann, R.; Bogdan, C.; Liebsch, G.; Wang, X.D.; Wolfbeis, O.S.; et al. Ratiometric luminescence 2D in vivo imaging and monitoring of mouse skin oxygenation. Methods Appl. Fluoresc. 2013, 1, 045002. [Google Scholar] [CrossRef] [PubMed]
- Mahnke, A.; Meier, R.J.; Schatz, V.; Hofmann, J.; Castiglione, K.; Schleicher, U.; Wolfbeis, O.S.; Bogdan, C.; Jantsch, J. Hypoxia in Leishmania major skin lesions impairs the NO-dependent leishmanicidal activity of macrophages. J. Investig. Dermatol. 2014, 134, 2339–2346. [Google Scholar] [CrossRef] [PubMed]
- Zieger, S.E.; Steinegger, A.; Klimant, I.; Borisov, S.M. TADF-Emitting Zn(II)-Benzoporphyrin: An Indicator for Simultaneous Sensing of Oxygen and Temperature. ACS Sens. 2020, 5, 1020–1027. [Google Scholar] [CrossRef] [PubMed]
- Kilani, R.T.; Mackova, M.; Davidge, S.T.; Guilbert, L.J. Effect of oxygen levels in villous trophoblast apoptosis. Placenta 2003, 24, 826–834. [Google Scholar] [CrossRef]
- Newby, D.; Marks, L.; Lyall, F. Dissolved oxygen concentration in culture medium: Assumptions and pitfalls. Placenta 2005, 26, 353–357. [Google Scholar] [CrossRef]
- Kofstad, J. Blood gases and hypothermia: Some theoretical and practical considerations. Scand. J. Clin. Lab. Investig. Suppl. 1996, 224, 21–26. [Google Scholar] [CrossRef]
- Kim, S.-H.; Shin, B.; Lee, S.J.; Lee, M.K.; Lee, W.-Y.; Yong, S.J. Comparison of pleural fluid pH measurements: Blood gas analyzer, pH indicator stick, litmus paper, and point-of-care testing for blood gases. Eur. Respir. J. 2018, 52, PA2872. [Google Scholar] [CrossRef]
- Nozue, M.; Lee, I.; Yuan, F.; Teicher, B.A.; Brizel, D.M.; Dewhirst, M.W.; Milross, C.G.; Milas, L.; Song, C.W.; Thomas, C.D.; et al. Interlaboratory variation in oxygen tension measurement by Eppendorf “Histograph” and comparison with hypoxic marker. J. Surg. Oncol. 1997, 66, 30–38. [Google Scholar] [CrossRef]
- Lyng, H.; Sundfør, K.; Rofstad, E.K. Oxygen tension in human tumours measured with polarographic needle electrodes and its relationship to vascular density, necrosis and hypoxia. Radiother. Oncol. J. Eur. Soc. Ther. Radiol. Oncol. 1997, 44, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Xiang, L.; Yu, P.; Zhang, M.; Hao, J.; Wang, Y.; Zhu, L.; Dai, L.; Mao, L. Platinized Aligned Carbon Nanotube-Sheathed Carbon Fiber Microelectrodes for In Vivo Amperometric Monitoring of Oxygen. Anal. Chem. 2014, 86, 5017–5023. [Google Scholar] [CrossRef] [PubMed]
- Bolger, F.B.; Bennett, R.; Lowry, J.P. An in vitro characterisation comparing carbon paste and Pt microelectrodes for real-time detection of brain tissue oxygen. Analyst 2011, 136, 4028–4035. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Hou, H.; Wei, H.; Yao, L.; Sun, L.; Yu, P.; Su, B.; Mao, L. In Vivo Monitoring of Oxygen in Rat Brain by Carbon Fiber Microelectrode Modified with Antifouling Nanoporous Membrane. Anal. Chem. 2019, 91, 3645–3651. [Google Scholar] [CrossRef]
- Jang, K.-I.; Han, S.Y.; Xu, S.; Mathewson, K.E.; Zhang, Y.; Jeong, J.-W.; Kim, G.-T.; Webb, R.C.; Lee, J.W.; Dawidczyk, T.J.; et al. Rugged and breathable forms of stretchable electronics with adherent composite substrates for transcutaneous monitoring. Nat. Commun. 2014, 5, 4779. [Google Scholar] [CrossRef]
- Oliveira, M.; Conceição, P.; Kant, K.; Ainla, A.; Diéguez, L. Electrochemical Sensing in 3D Cell Culture Models: New Tools for Developing Better Cancer Diagnostics and Treatments. Cancers 2021, 13, 1381. [Google Scholar] [CrossRef]
- Weltin, A.; Hammer, S.; Noor, F.; Kaminski, Y.; Kieninger, J.; Urban, G.A. Accessing 3D microtissue metabolism: Lactate and oxygen monitoring in hepatocyte spheroids. Biosens Bioelectron 2017, 87, 941–948. [Google Scholar] [CrossRef]
- Krohn, K.A.; Link, J.M.; Mason, R.P. Molecular imaging of hypoxia. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2008, 49 (Suppl. 2), 129s–148s. [Google Scholar] [CrossRef]
- Magata, Y.; Temma, T.; Iida, H.; Ogawa, M.; Mukai, T.; Iida, Y.; Morimoto, T.; Konishi, J.; Saji, H. Development of Injectable O-15 Oxygen and Estimation of Rat OEF. J. Cereb. Blood Flow Metab. 2003, 23, 671–676. [Google Scholar] [CrossRef]
- Sette, G.; Baron, J.C.; Mazoyer, B.; Levasseur, M.; Pappata, S.; Crouzel, C. Local brain haemodynamics and oxygen metabolism in cerebrovascular disease: Positron emission tomography. Brain 1989, 112, 931–951. [Google Scholar] [CrossRef] [PubMed]
- Hino, A.; Imahori, Y.; Tenjin, H.; Mizukawa, N.; Ueda, S.; Hirakawa, K.; Nakahashi, H. Metabolic and hemodynamic aspects of peritumoral low-density areas in human brain tumor. Neurosurgery 1990, 26, 615–621. [Google Scholar] [CrossRef] [PubMed]
- Abate, M.G.; Trivedi, M.; Fryer, T.D.; Smielewski, P.; Chatfield, D.A.; Williams, G.B.; Aigbirhio, F.; Carpenter, T.A.; Pickard, J.D.; Menon, D.K.; et al. Early derangements in oxygen and glucose metabolism following head injury: The ischemic penumbra and pathophysiological heterogeneity. Neurocritical Care 2008, 9, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Oku, N.; Kashiwagi, T.; Hatazawa, J. Nuclear neuroimaging in acute and subacute ischemic stroke. Ann. Nucl. Med. 2010, 24, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Golan, H.; Makogon, B.; Volkov, O.; Smolyakov, Y.; Hadanny, A.; Efrati, S. Imaging-based predictors for hyperbaric oxygen therapy outcome in post-stroke patients. Report 1. Med. Hypotheses 2020, 136, 109510. [Google Scholar] [CrossRef]
- Harch, P.G.; Andrews, S.R.; Fogarty, E.F.; Lucarini, J.; Van Meter, K.W. Case control study: Hyperbaric oxygen treatment of mild traumatic brain injury persistent post-concussion syndrome and post-traumatic stress disorder. Med. Gas Res. 2017, 7, 156–174. [Google Scholar] [CrossRef]
- Saito, H.; Ogasawara, K.; Suzuki, T.; Kuroda, H.; Kobayashi, M.; Yoshida, K.; Kubo, Y.; Ogawa, A. Adverse effects of intravenous acetazolamide administration for evaluation of cerebrovascular reactivity using brain perfusion single-photon emission computed tomography in patients with major cerebral artery steno-occlusive diseases. Neurol. Med. -Chir. 2011, 51, 479–483. [Google Scholar] [CrossRef]
- Tatum, J.L.; Kelloff, G.J.; Gillies, R.J.; Arbeit, J.M.; Brown, J.M.; Chao, K.S.; Chapman, J.D.; Eckelman, W.C.; Fyles, A.W.; Giaccia, A.J.; et al. Hypoxia: Importance in tumor biology, noninvasive measurement by imaging, and value of its measurement in the management of cancer therapy. Int. J. Radiat. Biol. 2006, 82, 699–757. [Google Scholar] [CrossRef]
- Christen, T.; Bolar, D.S.; Zaharchuk, G. Imaging brain oxygenation with MRI using blood oxygenation approaches: Methods, validation, and clinical applications. AJNR. Am. J. Neuroradiol. 2013, 34, 1113–1123. [Google Scholar] [CrossRef]
- Redler, G.; Epel, B.; Halpern, H.J. Principal component analysis enhances SNR for dynamic electron paramagnetic resonance oxygen imaging of cycling hypoxia in vivo. Magn. Reson. Med. 2014, 71, 440–450. [Google Scholar] [CrossRef]
- Wu, L.; Liu, F.; Liu, S.; Xu, X.; Liu, Z.; Sun, X. Perfluorocarbons-Based (19)F Magnetic Resonance Imaging in Biomedicine. Int. J. Nanomed. 2020, 15, 7377–7395. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.X.; Hallac, R.R.; Chiguru, S.; Mason, R.P. New frontiers and developing applications in 19F NMR. Prog. Nucl. Magn. Reson. Spectrosc. 2013, 70, 25–49. [Google Scholar] [CrossRef] [PubMed]
- Mason, R.P.; Nunnally, R.L.; Antich, P.P. Tissue oxygenation: A novel determination using 19F surface coil NMR spectroscopy of sequestered perfluorocarbon emulsion. Magn. Reson. Med. 1991, 18, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.R.; Pratt, R.G.; Millard, R.W.; Samaratunga, R.C.; Shiferaw, Y.; Clark, L.C.; Hoffmann, R.E. Evaluation of the influence of the aqueous phase bioconstituent environment on the F-19 T1 of perfluorocarbon blood substitute emulsions. J. Magn Reson Imaging 1994, 4, 631–635. [Google Scholar] [CrossRef]
- Simkins, J.W.; Stewart, P.S.; Codd, S.L.; Seymour, J.D. Non-invasive imaging of oxygen concentration in a complex in vitro biofilm infection model using (19) F MRI: Persistence of an oxygen sink despite prolonged antibiotic therapy. Magn. Reson. Med. 2019, 82, 2248–2256. [Google Scholar] [CrossRef]
- Jordan, B.F.; Cron, G.O.; Gallez, B. Rapid monitoring of oxygenation by 19F magnetic resonance imaging: Simultaneous comparison with fluorescence quenching. Magn. Reson. Med. 2009, 61, 634–638. [Google Scholar] [CrossRef]
- Liu, S.; Shah, S.J.; Wilmes, L.J.; Feiner, J.; Kodibagkar, V.D.; Wendland, M.F.; Mason, R.P.; Hylton, N.; Hopf, H.W.; Rollins, M.D. Quantitative tissue oxygen measurement in multiple organs using 19F MRI in a rat model. Magn. Reson. Med. 2011, 66, 1722–1730. [Google Scholar] [CrossRef]
- Diepart, C.; Karroum, O.; Magat, J.; Feron, O.; Verrax, J.; Calderon, P.B.; Grégoire, V.; Leveque, P.; Stockis, J.; Dauguet, N.; et al. Arsenic trioxide treatment decreases the oxygen consumption rate of tumor cells and radiosensitizes solid tumors. Cancer Res. 2012, 72, 482–490. [Google Scholar] [CrossRef]
- Matsumoto, K.; Bernardo, M.; Subramanian, S.; Choyke, P.; Mitchell, J.B.; Krishna, M.C.; Lizak, M.J. MR assessment of changes of tumor in response to hyperbaric oxygen treatment. Magn. Reson. Med. 2006, 56, 240–246. [Google Scholar] [CrossRef]
- Padhani, A.R.; Krohn, K.A.; Lewis, J.S.; Alber, M. Imaging oxygenation of human tumours. Eur. Radiol. 2007, 17, 861–872. [Google Scholar] [CrossRef]
- Gallez, B.; Baudelet, C.; Jordan, B.F. Assessment of tumor oxygenation by electron paramagnetic resonance: Principles and applications. NMR Biomed. 2004, 17, 240–262. [Google Scholar] [CrossRef] [PubMed]
- Hallac, R.R.; Ding, Y.; Yuan, Q.; McColl, R.W.; Lea, J.; Sims, R.D.; Weatherall, P.T.; Mason, R.P. Oxygenation in cervical cancer and normal uterine cervix assessed using blood oxygenation level-dependent (BOLD) MRI at 3T. NMR Biomed. 2012, 25, 1321–1330. [Google Scholar] [CrossRef] [PubMed]
- Colliez, F.; Gallez, B.; Jordan, B.F. Assessing Tumor Oxygenation for Predicting Outcome in Radiation Oncology: A Review of Studies Correlating Tumor Hypoxic Status and Outcome in the Preclinical and Clinical Settings. Front. Oncol. 2017, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Gulaka, P.K.; Rastogi, U.; Kodibagkar, V.D. More bullets for PISTOL: Linear and cyclic siloxane reporter probes for quantitative 1H MR oximetry. Sci. Rep. 2020, 10, 1399. [Google Scholar] [CrossRef] [PubMed]
- Liu, V.H.; Vassiliou, C.C.; Imaad, S.M.; Cima, M.J. Solid MRI contrast agents for long-term, quantitative in vivo oxygen sensing. Proc. Natl. Acad. Sci. USA 2014, 111, 6588–6593. [Google Scholar] [CrossRef] [PubMed]
- Vidya Shankar, R.; Kodibagkar, V.D. A faster PISTOL for (1) H MR-based quantitative tissue oximetry. NMR Biomed. 2019, 32, e4076. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Williams, B.B.; Swartz, H.M. Clinical applications of in vivo EPR: Rationale and initial results. Appl. Magn. Reson. 2006, 30, 185–199. [Google Scholar] [CrossRef]
- Hou, H.; Grinberg, O.Y.; Taie, S.; Leichtweis, S.; Miyake, M.; Grinberg, S.; Xie, H.; Csete, M.; Swartz, H.M. Electron paramagnetic resonance assessment of brain tissue oxygen tension in anesthetized rats. Anesth Analg 2003, 96, 1467–1472. [Google Scholar] [CrossRef]
- He, G.; Shankar, R.A.; Chzhan, M.; Samouilov, A.; Kuppusamy, P.; Zweier, J.L. Noninvasive measurement of anatomic structure and intraluminal oxygenation in the gastrointestinal tract of living mice with spatial and spectral EPR imaging. Proc. Natl. Acad. Sci. USA 1999, 96, 4586–4591. [Google Scholar] [CrossRef]
- Elas, M.; Magwood, J.M.; Butler, B.; Li, C.; Wardak, R.; DeVries, R.; Barth, E.D.; Epel, B.; Rubinstein, S.; Pelizzari, C.A.; et al. EPR oxygen images predict tumor control by a 50% tumor control radiation dose. Cancer Res. 2013, 73, 5328–5335. [Google Scholar] [CrossRef]
- Khan, N.; Williams, B.B.; Hou, H.; Li, H.; Swartz, H.M. Repetitive Tissue pO2 Measurements by Electron Paramagnetic Resonance Oximetry: Current Status and Future Potential for Experimental and Clinical Studies. Antioxid. Redox Signal. 2007, 9, 1169–1182. [Google Scholar] [CrossRef] [PubMed]
- Gertsenshteyn, I.; Epel, B.; Barth, E.; Leoni, L.; Markiewicz, E.; Tsai, H.M.; Fan, X.; Giurcanu, M.; Bodero, D.; Zamora, M.; et al. Improving Tumor Hypoxia Location in (18)F-Misonidazole PET with Dynamic Contrast-enhanced MRI Using Quantitative Electron Paramagnetic Resonance Partial Oxygen Pressure Images. Radiol. Imaging Cancer 2021, 3, e200104. [Google Scholar] [CrossRef] [PubMed]
- Swartz, H.M.; Hou, H.; Khan, N.; Jarvis, L.A.; Chen, E.Y.; Williams, B.B.; Kuppusamy, P. Advances in probes and methods for clinical EPR oximetry. Adv. Exp. Med. Biol. 2014, 812, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.B.; Khan, N.; Zaki, B.; Hartford, A.; Ernstoff, M.S.; Swartz, H.M. Clinical electron paramagnetic resonance (EPR) oximetry using India ink. Adv. Exp. Med. Biol. 2010, 662, 149–156. [Google Scholar] [CrossRef]
- Schaner, P.E.; Pettus, J.R.; Flood, A.B.; Williams, B.B.; Jarvis, L.A.; Chen, E.Y.; Pastel, D.A.; Zuurbier, R.A.; diFlorio-Alexander, R.M.; Swartz, H.M.; et al. OxyChip Implantation and Subsequent Electron Paramagnetic Resonance Oximetry in Human Tumors Is Safe and Feasible: First Experience in 24 Patients. Front. Oncol. 2020, 10, 572060. [Google Scholar] [CrossRef]
- Grucker, D.; Chambron, J. Oxygen imaging in perfused hearts by dynamic nuclear polarization. Magn. Reson. Imaging 1993, 11, 691–696. [Google Scholar] [CrossRef]
- Koch, A.; Kähler, W.; Klapa, S.; Grams, B.; van Ooij, P.A.M. The conundrum of using hyperoxia in COVID-19 treatment strategies: May intermittent therapeutic hyperoxia play a helpful role in the expression of the surface receptors ACE2 and Furin in lung tissue via triggering of HIF-1α? Intensive Care Med. Exp. 2020, 8, 53. [Google Scholar] [CrossRef]



| Oxygen Indicator | Matrix | Sample Type | Signals | T(°C) | O2 Range | Sensor Type | Ref |
|---|---|---|---|---|---|---|---|
| P(Pt- TPPTFEMA) P(Pt-TPP-EMA) | quartz substrate | cephalosporin C | Luminescence intensity | 20–28 °C | 0–100% (0–35 ppm) | Optical fiber | [54] |
| Pd (II) TFPP Pd(II) TCPP Pt(II) TFPP Pt(II) OEP CdSe QDs | sol-gel | aqueous oxygen | Ratiometric Luminescence intensity | 0–40 mg/L | Optical fiber | [58] | |
| [Ru-(dpp)3]2+ Oregon green 488-dextran | sol-gel | Rat C6 Glioma Living Cells | Ratiometric Luminescence intensity | 21 °C | 0–30 mg/L LOD 7.9 ±2.1 ppm | Silica nanosensor | [59] |
| PtOEPK OEP | PVC | inter- and intra-cellular | Ratiometric Luminescence intensity | 22 ± 0.5 °C | 0–43 ppm LOD 19 ppb inter 22 ppb intra | Unpulled PVC fiber sensor | [60] |
| Ag NPs doped with Ru(DPP)3Cl2 Coumarin6 | PMMA | aqueous oxygen Chlorella vulgaris | Ratiometric Luminescence intensity | 0–13 mg/L. | Optical sensor LOD = 0.1–0.6 mg/L | [61] | |
| PtTFPP | PDMS pillar arrays | aqueous oxygen enzymatic oxidation of β-d-glucose | Luminescence intensity | 23 °C | 0.00–1.28 μmol/L LOD = 0.1 μmol/L | Optical sensor | [62] |
| Ruthenium (II) dichloride (RD3) | silicone layers plus PC-coating | aqueous media (DO in water) | Luminescence intensity | 25.0± 0.5°C | LOD = 0.0 4 mg/L | Optical sensor | [63] |
| Ruthenium complex | Sol-gel | waste-water monitoring | Luminescence lifetime | 5–30 °C | (LOD) 6 ppb | optoelectronic sensor | [64] |
| [Ru(dpp)3]2+ | sol-gel oxides | microenvironments | Luminescence intensity | 25°C | IN2/IO2 from 3 to 35 | Optical chemical O2 sensors | [65] |
| Winkler Method | Polarographic Methods | Fluorescence Methods | |
|---|---|---|---|
| Remote monitoring | Cannot achieve remote measurement, and samples must be analyzed in the laboratory. | Can achieve remote detection, but the signal transmission will be distorted; thus, the detection results are not accurate. | Can use an optical fiber to transmit signals and achieve remote detection. |
| Analysis/response time | Longest time required | 30–180 s; however, the polarization of the electrode requires about 15 additional minutes, so the response time is longer [30]. | 41 ms-694 s. The fluorescence quenching method has the fastest response time (up to the ms level) [30]. |
| Oxygen consumption | Yes (titration process) | Yes (redox reaction at the electrode) | No (quenching process is reversible) |
| Maintenance | No | Yes | No |
| Application | Laboratory and water samples. | Biological medicine, forestry, fishing. | Life sciences, harsh environments. |
| Interference | Turbidity, nitrite, free chlorine, iron ions, colored solution. | Chlorine, sulfur dioxide, Iodine, Bromine, Electrical interference. | Fluorescence quenching and the stability of organic molecules can be influenced by factors that include pH and temperature. |
| Accuracy | ±0.1% [30] | ±0.01–0.1 mg/L [30] | / |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marassi, V.; Giordani, S.; Kurevija, A.; Panetta, E.; Roda, B.; Zhang, N.; Azzolini, A.; Dolzani, S.; Manko, D.; Reschiglian, P.; et al. The Challenges of O2 Detection in Biological Fluids: Classical Methods and Translation to Clinical Applications. Int. J. Mol. Sci. 2022, 23, 15971. https://doi.org/10.3390/ijms232415971
Marassi V, Giordani S, Kurevija A, Panetta E, Roda B, Zhang N, Azzolini A, Dolzani S, Manko D, Reschiglian P, et al. The Challenges of O2 Detection in Biological Fluids: Classical Methods and Translation to Clinical Applications. International Journal of Molecular Sciences. 2022; 23(24):15971. https://doi.org/10.3390/ijms232415971
Chicago/Turabian StyleMarassi, Valentina, Stefano Giordani, Andjela Kurevija, Emilio Panetta, Barbara Roda, Nan Zhang, Andrea Azzolini, Sara Dolzani, Dmytro Manko, Pierluigi Reschiglian, and et al. 2022. "The Challenges of O2 Detection in Biological Fluids: Classical Methods and Translation to Clinical Applications" International Journal of Molecular Sciences 23, no. 24: 15971. https://doi.org/10.3390/ijms232415971
APA StyleMarassi, V., Giordani, S., Kurevija, A., Panetta, E., Roda, B., Zhang, N., Azzolini, A., Dolzani, S., Manko, D., Reschiglian, P., Atti, M., & Zattoni, A. (2022). The Challenges of O2 Detection in Biological Fluids: Classical Methods and Translation to Clinical Applications. International Journal of Molecular Sciences, 23(24), 15971. https://doi.org/10.3390/ijms232415971









