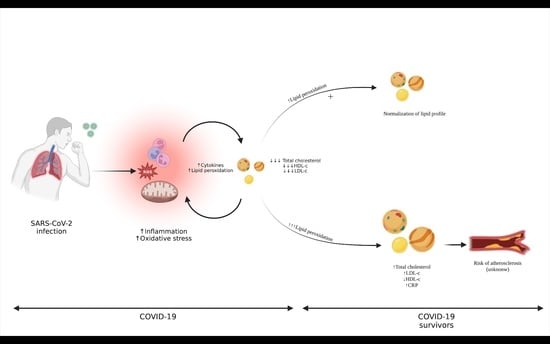
Dyslipidemia and Inflammation as Hallmarks of Oxidative Stress in COVID-19: A Follow-Up Study
Abstract
:1. Introduction
2. Results
2.1. The Inflammatory, Redox, and Lipid Profile of COVID-19 vs. Controls
2.2. Association of Lipid Peroxidation with Lipid and Inflammatory Markers
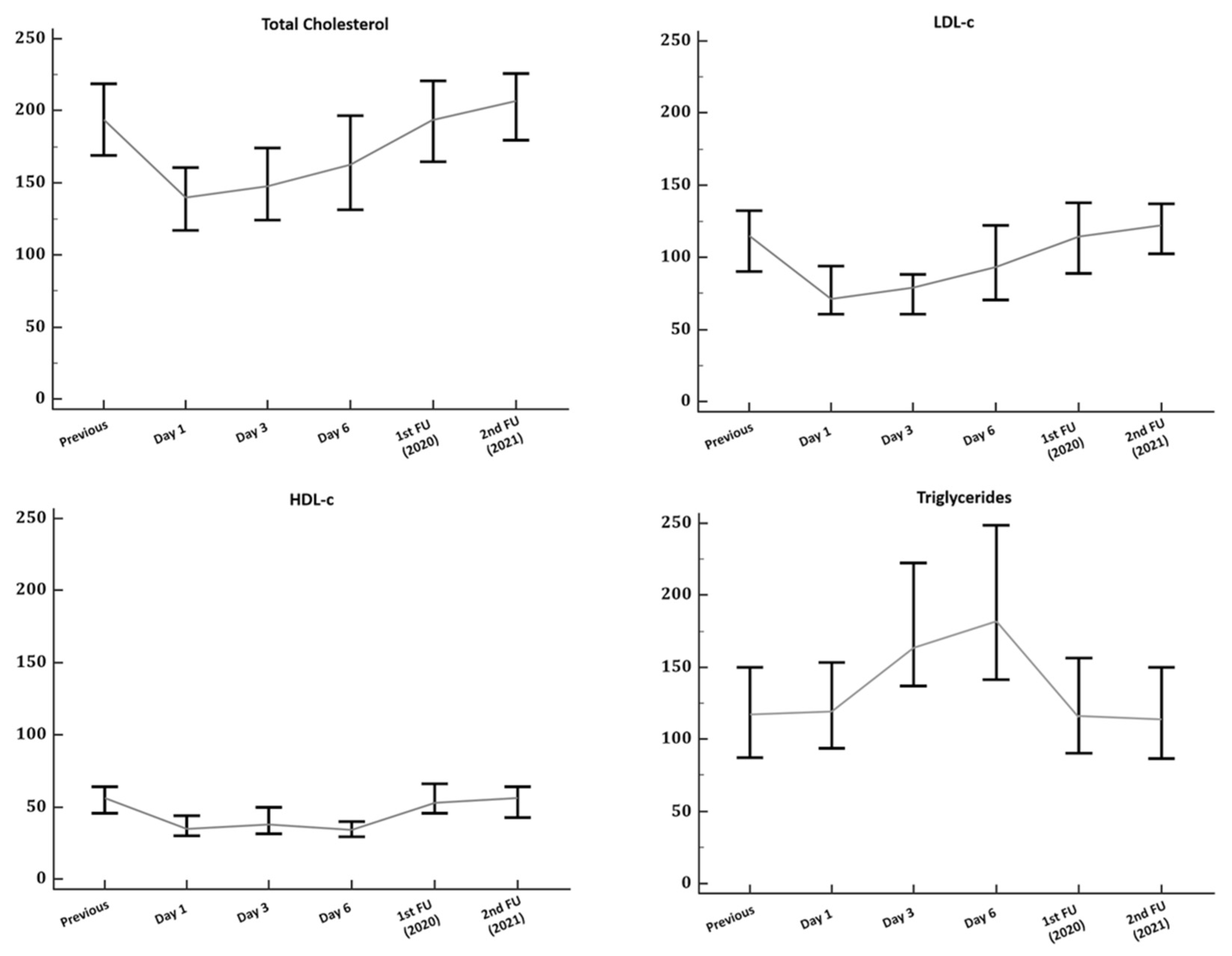
2.3. Follow-Up
2.4. Clinical Outcomes and Mortality
3. Discussion
Limitations
4. Materials and Methods
4.1. Study Design and Patient Inclusion Criteria
4.2. Biological Samples
4.3. Lipid Profile Analysis
4.4. Cytokine Analysis
4.5. Oxidant Cell Damage and Antioxidant Capacity Determination
4.6. Endpoints
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| COVID-19 | Coronavirus disease 2019 |
| CI | Confidence interval |
| CRP | C-reactive protein |
| IL | Interleukin |
| HDL-c | High-density lipoprotein cholesterol |
| LDL-c | Low-density lipoprotein cholesterol |
| LPO | Lipid peroxidation |
| NLRP3 | Pyrin domain-containing 3 |
| SARS-CoV-2 | Severe acute respiratory syndrome coronavirus 2 |
| TC | Total cholesterol |
| TG | Tryglycerides |
References
- Singh, A.K.; Gillies, C.L.; Singh, R.; Singh, A.; Chudasama, Y.; Coles, B.; Seidu, S.; Zaccardi, F.; Davies, M.J.; Khunti, K. Prevalence of Comorbidities and Their Association with Mortality in Patients with COVID-19: A Systematic Review and Meta-analysis. Diabetes Obes. Metab. 2020, 22, 1915–1924. [Google Scholar] [CrossRef] [PubMed]
- Dalleau, S.; Baradat, M.; Guéraud, F.; Huc, L. Cell Death and Diseases Related to Oxidative Stress:4-Hydroxynonenal (HNE) in the Balance. Cell Death Differ. 2013, 20, 1615–1630. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Forman, H.J.; Zhang, H. Targeting Oxidative Stress in Disease: Promise and Limitations of Antioxidant Therapy. Nat. Rev. Drug Discov. 2021, 20, 689–709. [Google Scholar] [CrossRef]
- Binder, C.J.; Papac-Milicevic, N.; Witztum, J.L. Innate Sensing of Oxidation-Specific Epitopes in Health and Disease. Nat. Rev. Immunol. 2016, 16, 485–497. [Google Scholar] [CrossRef] [PubMed]
- Kalem, A.K.; Kayaaslan, B.; Neselioglu, S.; Eser, F.; Hasanoglu, İ.; Aypak, A.; Akinci, E.; Akca, H.N.; Erel, O.; Guner, R. A Useful and Sensitive Marker in the Prediction of COVID-19 and Disease Severity: Thiol. Free Radic. Biol. Med. 2021, 166, 11–17. [Google Scholar] [CrossRef]
- Biagini, D.; Franzini, M.; Oliveri, P.; Lomonaco, T.; Ghimenti, S.; Bonini, A.; Vivaldi, F.; Macera, L.; Balas, L.; Durand, T.; et al. MS-Based Targeted Profiling of Oxylipins in COVID-19: A New Insight into Inflammation Regulation. Free Radic. Biol. Med. 2022, 180, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Žarković, N.; Orehovec, B.; Milković, L.; Baršić, B.; Tatzber, F.; Wonisch, W.; Tarle, M.; Kmet, M.; Mataić, A.; Jakovčević, A.; et al. Preliminary Findings on the Association of the Lipid Peroxidation Product 4-Hydroxynonenal with the Lethal Outcome of Aggressive COVID-19. Antioxidants 2021, 10, 1341. [Google Scholar] [CrossRef] [PubMed]
- Sharif-Askari, N.S.; Sharif-Askari, F.S.; Mdkhana, B.; Alsayed, H.A.H.; Alsafar, H.; Alrais, Z.F.; Hamid, Q.; Halwani, R. Upregulation of Oxidative Stress Gene Markers during SARS-COV-2 Viral Infection. Free Radic. Biol. Med. 2021, 172, 688–698. [Google Scholar] [CrossRef]
- Golabi, S.; Ghasemi, S.; Adelipour, M.; Bagheri, R.; Suzuki, K.; Wong, A.; Seyedtabib, M.; Naghashpour, M. Oxidative Stress and Inflammatory Status in COVID-19 Outpatients: A Health Center-Based Analytical Cross-Sectional Study. Antioxidants 2022, 11, 606. [Google Scholar] [CrossRef]
- Martín-Fernández, M.; Aller, R.; Heredia-Rodríguez, M.; Gómez-Sánchez, E.; Martínez-Paz, P.; Gonzalo-Benito, H.; Prada, L.S.; Gorgojo, Ó.; Carnicero-Frutos, I.; Tamayo, E.; et al. Lipid Peroxidation as a Hallmark of Severity in COVID-19 Patients. Redox Biol. 2021, 48, 102181. [Google Scholar] [CrossRef]
- Zinellu, A.; Paliogiannis, P.; Fois, A.G.; Solidoro, P.; Carru, C.; Mangoni, A.A. Cholesterol and Triglyceride Concentrations, COVID-19 Severity, and Mortality: A Systematic Review and Meta-Analysis With Meta-Regression. Front. Public Health 2021, 9, 705916. [Google Scholar] [CrossRef] [PubMed]
- Mahat, R.K.; Rathore, V.; Singh, N.; Singh, N.; Singh, S.K.; Shah, R.K.; Garg, C. Lipid Profile as an Indicator of COVID-19 Severity: A Systematic Review and Meta-Analysis. Clin. Nutr. ESPEN 2021, 45, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid Peroxidation: Production, Metabolism, and Signaling Mechanisms of Malondialdehyde and 4-Hydroxy-2-Nonenal. Oxidative Med. Cell. Longev. 2014, 2014, 360438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Serhan, C.N.; Chiang, N.; Dyke, T.E.V. Resolving Inflammation: Dual Anti-Inflammatory and pro-Resolution Lipid Mediators. Nat. Rev. Immunol. 2008, 8, 349–361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Serhan, C.N. Pro-Resolving Lipid Mediators Are Leads for Resolution Physiology. Nature 2014, 510, 92–101. [Google Scholar] [CrossRef] [Green Version]
- Superko, H.R. Advanced Lipoprotein Testing and Subfractionation Are Clinically Useful. Circulation 2009, 119, 2383–2395. [Google Scholar] [CrossRef] [Green Version]
- Khovidhunkit, W.; Kim, M.-S.; Memon, R.A.; Shigenaga, J.K.; Moser, A.H.; Feingold, K.R.; Grunfeld, C. Thematic Review Series: The Pathogenesis of Atherosclerosis. Effects of Infection and Inflammation on Lipid and Lipoprotein Metabolism Mechanisms and Consequences to the Host 1. J. Lipid Res. 2004, 45, 1169–1196. [Google Scholar] [CrossRef] [Green Version]
- Guirgis, F.W.; Black, L.P.; Henson, M.; Labilloy, G.; Smotherman, C.; Hopson, C.; Tfirn, I.; DeVos, E.L.; Leeuwenburgh, C.; Moldawer, L.; et al. A Hypolipoprotein Sepsis Phenotype Indicates Reduced Lipoprotein Antioxidant Capacity, Increased Endothelial Dysfunction and Organ Failure, and Worse Clinical Outcomes. Crit. Care 2021, 25, 341. [Google Scholar] [CrossRef]
- Pincemail, J.; Cavalier, E.; Charlier, C.; Cheramy–Bien, J.-P.; Brevers, E.; Courtois, A.; Fadeur, M.; Meziane, S.; Goff, C.L.; Misset, B.; et al. Oxidative Stress Status in COVID-19 Patients Hospitalized in Intensive Care Unit for Severe Pneumonia. A Pilot Study. Antioxidants 2021, 10, 257. [Google Scholar] [CrossRef]
- Aparisi, Á.; Iglesias-Echeverría, C.; Ybarra-Falcón, C.; Cusácovich, I.; Uribarri, A.; García-Gómez, M.; Ladrón, R.; Fuertes, R.; Candela, J.; Tobar, J.; et al. Low-Density Lipoprotein Cholesterol Levels Are Associated with Poor Clinical Outcomes in COVID-19. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 2619–2627. [Google Scholar] [CrossRef]
- Fajgenbaum, D.C.; June, C.H. Cytokine Storm. N. Engl. J. Med. 2020, 383, 2255–2273. [Google Scholar] [CrossRef] [PubMed]
- Lekkou, A.; Mouzaki, A.; Siagris, D.; Ravani, I.; Gogos, C.A. Serum Lipid Profile, Cytokine Production, and Clinical Outcome in Patients with Severe Sepsis. J. Crit. Care 2014, 29, 723–727. [Google Scholar] [CrossRef]
- Golucci, A.P.B.S.; Marson, F.A.L.; Ribeiro, A.F.; Nogueira, R.J.N. Lipid Profile Associated with the Systemic Inflammatory Response Syndrome and Sepsis in Critically Ill Patients. Nutrition 2018, 55–56, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Tschopp, J.; Schroder, K. NLRP3 Inflammasome Activation: The Convergence of Multiple Signalling Pathways on ROS Production? Nat. Rev. Immunol. 2010, 10, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Lage, S.L.; Amaral, E.P.; Hilligan, K.L.; Laidlaw, E.; Rupert, A.; Namasivayan, S.; Rocco, J.; Galindo, F.; Kellogg, A.; Kumar, P.; et al. Persistent Oxidative Stress and Inflammasome Activation in CD14highCD16− Monocytes From COVID-19 Patients. Front. Immunol. 2022, 12, 799558. [Google Scholar] [CrossRef] [PubMed]
- Münzel, T.; Camici, G.G.; Maack, C.; Bonetti, N.R.; Fuster, V.; Kovacic, J.C. Impact of Oxidative Stress on the Heart and Vasculature Part 2 of a 3-Part Series. J. Am. Coll. Cardiol. 2017, 70, 212–229. [Google Scholar] [CrossRef]
- Deakin, S.; Moren, X.; James, R.W. HDL Oxidation Compromises Its Influence on Paraoxonase-1 Secretion and Its Capacity to Modulate Enzyme Activity. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 1146–1152. [Google Scholar] [CrossRef] [Green Version]
- Yeon, S.H.; Yang, G.; Lee, H.E.; Lee, J.Y. Oxidized Phosphatidylcholine Induces the Activation of NLRP3 Inflammasome in Macrophages. J. Leukoc. Biol. 2017, 101, 205–215. [Google Scholar] [CrossRef] [Green Version]
- Li, W.L.; Hua, L.G.; Qu, P.; Yan, W.H.; Ming, C.; Jun, Y.D.; Yuan, L.D.; Nan, N. NLRP3 Inflammasome: A Novel Link between Lipoproteins and Atherosclerosis. Arch. Med. Sci. 2016, 12, 950–958. [Google Scholar] [CrossRef] [Green Version]
- Papotti, B.; Macchi, C.; Favero, C.; Iodice, S.; Adorni, M.P.; Zimetti, F.; Corsini, A.; Aliberti, S.; Blasi, F.; Carugo, S.; et al. HDL in COVID-19 Patients: Evidence from an Italian Cross-Sectional Study. J. Clin. Med. 2021, 10, 5955. [Google Scholar] [CrossRef]
- Surendran, A.; Zhang, H.; Winter, T.; Edel, A.; Aukema, H.; Ravandi, A. Oxylipin Profile of Human Low-Density Lipoprotein Is Dependent on Its Extent of Oxidation. Atherosclerosis 2019, 288, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Mazzucchelli, R.; Durum, S.K. Interleukin-7 Receptor Expression: Intelligent Design. Nat. Rev. Immunol. 2007, 7, 144–154. [Google Scholar] [CrossRef] [PubMed]
- Mills, K.H.G.; Dungan, L.S.; Jones, S.A.; Harris, J. The Role of Inflammasome-derived IL-1 in Driving IL-17 Responses. J. Leukoc. Biol. 2013, 93, 489–497. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Von Stebut, E.; Boehncke, W.-H.; Ghoreschi, K.; Gori, T.; Kaya, Z.; Thaci, D.; Schäffler, A. IL-17A in Psoriasis and Beyond: Cardiovascular and Metabolic Implications. Front. Immunol. 2020, 10, 3096. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Varshney, P.; Narasimhan, A.; Mittal, S.; Malik, G.; Sardana, K.; Saini, N. Transcriptome Profiling Unveils the Role of Cholesterol in IL-17A Signaling in Psoriasis. Sci. Rep. 2016, 6, 19295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pietrowski, E.; Bender, B.; Huppert, J.; White, R.; Luhmann, H.J.; Kuhlmann, C.R.W. Pro-Inflammatory Effects of Interleukin-17A on Vascular Smooth Muscle Cells Involve NAD(P)H- Oxidase Derived Reactive Oxygen Species. J. Vasc. Res. 2010, 48, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Webster, K.E.; Kim, H.-O.; Kyparissoudis, K.; Corpuz, T.M.; Pinget, G.V.; Uldrich, A.P.; Brink, R.; Belz, G.T.; Cho, J.-H.; Godfrey, D.I.; et al. IL-17-Producing NKT Cells Depend Exclusively on IL-7 for Homeostasis and Survival. Mucosal Immunol. 2014, 7, 1058–1067. [Google Scholar] [CrossRef] [Green Version]
- Kalinowska, M.; Bazdar, D.A.; Lederman, M.M.; Funderburg, N.; Sieg, S.F. Decreased IL-7 Responsiveness Is Related to Oxidative Stress in HIV Disease. PLoS ONE 2013, 8, e58764. [Google Scholar] [CrossRef] [Green Version]
- Angriman, F.; Rosella, L.C.; Lawler, P.R.; Ko, D.T.; Wunsch, H.; Scales, D.C. Sepsis hospitalization and risk of subsequent cardiovascular events in adults: A population-based matched cohort study. Intensive Care Med. 2022, 48, 448–457. [Google Scholar] [CrossRef]
- Xie, Y.; Xu, E.; Bowe, B.; Al-Aly, Z. Long-Term Cardiovascular Outcomes of COVID-19. Nat. Med. 2022, 28, 583–590. [Google Scholar] [CrossRef]
- Felici, N.; Liu, D.; Maret, J.; Restrepo, M.; Borovskiy, Y.; Hajj, J.; Chung, W.; Laudanski, K. Long-Term Abnormalities of Lipid Profile After a Single Episode of Sepsis. Front. Cardiovasc. Med. 2021, 8, 674248. [Google Scholar] [CrossRef] [PubMed]
- Gazi, I.F.; Apostolou, F.A.; Liberopoulos, E.N.; Filippatos, T.D.; Tellis, C.C.; Elisaf, M.S.; Tselepis, A.D. Leptospirosis Is Associated with Markedly Increased Triglycerides and Small Dense Low-Density Lipoprotein and Decreased High-Density Lipoprotein. Lipids 2011, 46, 953–960. [Google Scholar] [CrossRef]
- Apostolou, F.; Gazi, I.F.; Lagos, K.; Tellis, C.C.; Tselepis, A.D.; Liberopoulos, E.N.; Elisaf, M. Acute Infection with Epstein–Barr Virus Is Associated with Atherogenic Lipid Changes. Atherosclerosis 2010, 212, 607–613. [Google Scholar] [CrossRef]
- Vazzana, N.; Ganci, A.; Cefalù, A.B.; Lattanzio, S.; Noto, D.; Santoro, N.; Saggini, R.; Puccetti, L.; Averna, M.; Davì, G. Enhanced Lipid Peroxidation and Platelet Activation as Potential Contributors to Increased Cardiovascular Risk in the Low-HDL Phenotype. J. Am. Heart Assoc. Cardiovasc. Cerebrovasc. Dis. 2013, 2, e000063. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsuura, E.; Hughes, G.R.V.; Khamashta, M.A. Oxidation of LDL and Its Clinical Implication. Autoimmun. Rev. 2008, 7, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Tamayo-Velasco, Á.; Peñarrubia-Ponce, M.J.; Álvarez, F.J.; Gonzalo-Benito, H.; de la Fuente, I.; Martín-Fernández, M.; Eiros, J.M.; Martínez-Paz, P.; Miramontes-González, J.P.; Fiz-López, A.; et al. Evaluation of Cytokines as Robust Diagnostic Biomarkers for COVID-19 Detection. J. Pers. Med. 2021, 11, 681. [Google Scholar] [CrossRef]
| Global COVID-19 (n = 108) | Controls (n = 28) | p-Value | |
|---|---|---|---|
| Inflammatory markers | |||
| CRP, mg/L | 83.5 [38–151.2] | 10 [7–10] | <0.001 |
| D-dimer, ng/mL | 800 [470–1713] | 255 [224–330] | <0.001 |
| Ferritin, ng/mL | 731 [412–1500] | NA | NA |
| Lymphocytes, cells/mm3 | 945 [630–1221] | 2255 [1545–2485] | <0.001 |
| Cytokines and chemokines | |||
| IL-1β, pg/mL | 6.62 [2.71–13.2] | 2.49 [1.76–2.81] | <0.001 |
| IL-1RA, pg/mL | 604.7 [258.6–1282.5] | 72.5 [45.9–87.1] | <0.001 |
| IL-6, pg/mL | 13.07 [6.57–29.45] | 8.69 [5.95–10.01] | 0.010 |
| IL-7, pg/mL | 1.67 [0.72–3.58] | 0.72 [0.50–0.82] | <0.001 |
| IL-17A, pg/mL | 7.03 [3.11–18.33] | 2.03 [1.59–2.32] | <0.001 |
| IL-18, pg/mL | 47.2 [25–76.55] | 14.53 [11.89–32.3] | <0.001 |
| IP-10, pg/mL | 45.55 [29.07–71.30] | 5.56 [3.07–7.34] | <0.001 |
| Oxidative markers | |||
| 8-OHdG a, pg/mL | 8373 [5445–12,497] | 7925 [5130–9722] | 0.246 |
| ABTS b, µM | 2264 [1938–2462] | 2510 [2371–2768] | <0.001 |
| FRAP b, µM | 453.8 [385.7–576.1] | 700.7 [554.1–796.4] | <0.001 |
| LPO c, µM | 2123 [1250–3249] | 284 [174–509] | <0.001 |
| Protein carbonyl d, nmol/mg prot. | 10.8 [7.2–14.6] | 5.6 [4.4–8.07] | <0.001 |
| Lipid profile | |||
| Total cholesterol, mg/dL | 140 [117.5–159.5] | 175 [164–197] | <0.001 |
| LDL-c, mg/dL | 71.3 [60.4–93.5] | 98 [84.4–125.6] | 0.002 |
| HDL-c, mg/dL | 35 [29.2–44] | 59 [50.4–65.3] | <0.001 |
| TG, mg/dL | 119 [94.5–153.5] | 89.5 [83–127] | 0.027 |
| TC/HDL-c | 3.68 [3.18–4.66] | 2.9 [2.52–3.83] | 0.008 |
| Variable | Univariable | Multivariable | ||
|---|---|---|---|---|
| OR (95% CI) | p-Value | OR (95% CI) | p-Value | |
| Age | 1.007 (0.971–1.045) | 0.700 | ||
| Male gender | 0.883 (0.320–2.435) | 0.810 | ||
| Total cholesterol | 0.986 (0.969–1.002) | 0.092 | 0.982 (0.969–0.996) | 0.012 |
| IL1RA | 0.999 (0.998–0.999) | 0.017 | 0.999 (0.998–0.999) | 0.021 |
| IL-2 | 0.961 (0.862–1.070) | 0.467 | ||
| IL-4 | 0.991 (0.825–1.191) | 0.927 | ||
| IL-5 | 0.996 (0.976–1.017) | 0.703 | ||
| IL-6 | 1.094 (1.026–1.166) | 0.006 | 1.062 (1.017–1.110) | 0.007 |
| IL-7 | 0.582 (0.311–1.087) | 0.089 | 0.653 (0.433–0.986) | 0.042 |
| IL-8 | 0.983 (0.952–1.015) | 0.302 | ||
| IL-9 | 0.797 (0.512–1.240) | 0.314 | ||
| IL-12p70 | 0.856 (0.480–1.528) | 0.600 | ||
| IL-15 | 1.018 (0.931–1.112) | 0.702 | ||
| IL-17α | 1.200 (0.998–1.442) | 0.052 | 1.098 (1.010–1.193) | 0.028 |
| IL-18 | 1.010 (0.996–1.023) | 0.164 | ||
| IL-23 | 1.449 (0.951–2.208) | 0.084 | ||
| IL-31 | 0.735 (0.535–1.009) | 0.057 | ||
| IP-10 | 0.995 (0.987–1.003) | 0.225 | ||
| LPO ≤ 1948 μM a | LPO > 1948 μM a | ANCOVA Adjusted p-Value d | |
|---|---|---|---|
| First follow-up b | |||
| TC, mg/dL | 186 [163–210] | 195 [157–215] | 0.763 |
| LDL-c, mg/dL | 123 [102–139] | 107 [102–135] | 0.475 |
| HDL-c, mg/dL | 58 [47–66] | 49 [36–67] | 0.392 |
| TG, mg/dL | 107 [83–139] | 115 [101–168] | 0.372 |
| TC/HDL-c | 3.4 [2.9–4.5] | 3.5 [2.8–5.2] | 0.306 |
| CRP, mg/L | 1.6 [1–4.3] | 1.7 [1–4.3] | 0.703 |
| Second follow-upc | |||
| TC, mg/dL | 192 [174–211] | 207 [197–230] | 0.026 |
| LDL-c, mg/dL | 114 [99–128] | 131 [120–147] | 0.025 |
| HDL-c, mg/dL | 57 [45–65] | 48 [38–62] | 0.031 |
| TG, mg/dL | 109 [86–127] | 127 [107–150] | 0.002 |
| TC/HDL-c | 3.4 [2.9–3.6] | 3.9 [3.4–4.8] | 0.003 |
| CRP, mg/L | 2.5 [1–5.25] | 5.8 [2–15] | 0.072 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aparisi, Á.; Martín-Fernández, M.; Ybarra-Falcón, C.; Gil, J.F.; Carrasco-Moraleja, M.; Martínez-Paz, P.; Cusácovich, I.; Gonzalo-Benito, H.; Fuertes, R.; Marcos-Mangas, M.; et al. Dyslipidemia and Inflammation as Hallmarks of Oxidative Stress in COVID-19: A Follow-Up Study. Int. J. Mol. Sci. 2022, 23, 15350. https://doi.org/10.3390/ijms232315350
Aparisi Á, Martín-Fernández M, Ybarra-Falcón C, Gil JF, Carrasco-Moraleja M, Martínez-Paz P, Cusácovich I, Gonzalo-Benito H, Fuertes R, Marcos-Mangas M, et al. Dyslipidemia and Inflammation as Hallmarks of Oxidative Stress in COVID-19: A Follow-Up Study. International Journal of Molecular Sciences. 2022; 23(23):15350. https://doi.org/10.3390/ijms232315350
Chicago/Turabian StyleAparisi, Álvaro, Marta Martín-Fernández, Cristina Ybarra-Falcón, José Francisco Gil, Manuel Carrasco-Moraleja, Pedro Martínez-Paz, Iván Cusácovich, Hugo Gonzalo-Benito, Raúl Fuertes, Marta Marcos-Mangas, and et al. 2022. "Dyslipidemia and Inflammation as Hallmarks of Oxidative Stress in COVID-19: A Follow-Up Study" International Journal of Molecular Sciences 23, no. 23: 15350. https://doi.org/10.3390/ijms232315350
APA StyleAparisi, Á., Martín-Fernández, M., Ybarra-Falcón, C., Gil, J. F., Carrasco-Moraleja, M., Martínez-Paz, P., Cusácovich, I., Gonzalo-Benito, H., Fuertes, R., Marcos-Mangas, M., Iglesias-Echeverría, C., San Román, J. A., Tamayo, E., Andaluz-Ojeda, D., & Tamayo-Velasco, Á. (2022). Dyslipidemia and Inflammation as Hallmarks of Oxidative Stress in COVID-19: A Follow-Up Study. International Journal of Molecular Sciences, 23(23), 15350. https://doi.org/10.3390/ijms232315350