Specific Features of the Proteomic Response of Thermophilic Bacterium Geobacillus icigianus to Terahertz Irradiation
Abstract
:1. Introduction
2. Results
2.1. Changes in the Proteomic Profile of the Thermophilic Bacterium G. icigianus in Response to Irradiation with THz Electromagnetic Waves (2.3 THz) for 15 Min
| Protein/Reaction | Gene/Locus | Process/Function | ∆ |
|---|---|---|---|
| Electron transfer flavoprotein subunit beta/FixA family protein (ETFb) Electron transfer flavoprotein subunit alpha/FixB family protein (ETFa) | EP10_09770 EP10_09765 | ETF:heterodimer, a key component of the electron transfer chain | 1.7 1.4 |
| NADH dehydrogenase (EC 7.1.1.2), NADH:ubiquinone reductase (H+-translocating) NADH + Q + 5H+-> QH2 + NAD +4H+ | EP10_04625 | Respiration, generates PMF | 1.6 |
| D-3-phosphoglycerate dehydrogenase (EC 1.1.1.95) G3P + NAD+ -> 3-phosphooxypyruvate + NADH + H+ | EP10_12110 | NADH synthesis, serine biosynthesis | 1.8 |
| Type I glyceraldehyde-3-phosphate dehydrogenase (NAD+), (EC 1.2.1.12) G3P + Pi + NAD -> NADH + 1, 3PDG | gap/ EP10_12960 | Key enzyme of the glycolytic pathway, NADH synthesis | 1.2 |
| Class II fructose-bisphosphatase Fructose-1,6-bisphosphatase (FBPase) (EC 3.1.3.11) FDP+ H2O + ADP -> F6P + ATP | glpX/ EP10_10310 | ATP biosynthesis, peptidoglycan biosynthesis | 1.2 |
| Elongation factor Tu | tuf/ EP10_11775 | Translation elongation factor, GTPase | 1.6 |
| Ribosome recycling factor | frr/EP10_17125 | Translation | 1.4 |
| Cysteine-tRNA ligase (EC:6.1.1.16) | CysS/ EP10_11880 | Translation | 1.35 |
| Transcription elongation factor GreA | EP10_01545 | Transcription, chaperone activity | 1.8 |
| Bifunctional 3,4-dihydroxy-2-butanone 4-phosphate synthase, DHBP synthase (EC 4.1.99.12)/GTP cyclohydrolase II (EC 3.5.4.25) | EP10_18260 | Riboflavin biosynthesis, protects DNA from oxygen radicals | 1.4 |
| Thioredoxin reductase (EC 1.8.1.9) Thr + NADP+ -> Thr disulfide + NADPH + H+ | EP10_04630 | Antioxidant system, NADPH synthesis | 1.6 |
| Thiol peroxidase, Tpx-type (EC:1.11.1.24) [thioredoxin]-dithiol + a hydroperoxide -> [thioredoxin]-disulfide + alcohol + H2O | EP10_18085 | Thiol peroxidase is part of an oxidative stress defense system | 1.6 |
| Deacetylase BshB2 (deacetylase of the LmbE family protein) | bshB2/ EP10_10565 | Bacillithiol synthesis, resistance during oxidative stress | 1.55 |
| Pantoate-beta-alanine ligase (EC 6.3.2.1) ATP + pantoate + alanine -> AMP + diphosphate + pantothenate | EP10_07480 | Synthesis of pantothenate from alanine | 1.3 |
| L-serine deaminase, L-serine ammonia-lyase, iron-sulfur-dependent, subunit beta (EC 4.3.1.17) L-serine -> pyruvate + NH3 | sdaAB/ EP10_16780 | Pyruvate synthesis | 1.4 |
| Acetyl-CoA acetyltransferase (EC 2.3.1.9) 2 Acetyl-CoA -> CoA + acetoacetyl-CoA | thlA/ EP10_03775 | CoA biosynthesis | 1.7 |
| Acetate kinase (EC 2.7.2.1) ATP + acetate -> ADP + acetyl phosphate | ackA/EP10_18095 | АсСоА biosynthesis | 1.4 |
| Pyridoxal 5′-phosphate synthase (PLP synthase) Glutaminase subunit PdxT (EC 2.4.2.-) | pdxT/ EP10_02655 | De novo pyridoxal 5′-phosphate biosynthesis | 1.5 |
| Inorganic pyrophosphatase (EC 3.6.1.1) Diphosphate + H2O <-> 2 phosphate | EP10_12120 | Cell growth | 1.3 |
| Protein/Reaction | Gene/Locus | Process/Function | ∆ |
|---|---|---|---|
| ATP synthase alpha chain (EC 7.1.2.2) ADP + PI + [H+]ex <-> ATP + H2O + [H+]in | atpA/ EP10_10185 | ATP biosynthesis; when the PMF is decreased, ATP synthase catalyzes the reverse reaction, working as an ATP-dependent proton pump | 1.3 |
| Predicted L-lactate dehydrogenase (multisubunits, lldEFG, syn ykgEFG), PYR + NAD+ <-> LAC + NADH + H+ | EP10_13275 | NADH and lactate biosynthesis; iron–sulfur cluster-binding subunit YkgF [51] | 1.6 |
| Malic enzyme, NAD-dependent Mal + NAD -> Pyr + NADH + CO2 | EP10_15870 | NADH and pyruvate biosynthesis | 1.8 |
| Oxidoreductase, aldo/keto reductase family | EP10_11090 | NADH and NADPH biosynthesis | 1.55 |
| 2-Oxo acid dehydrogenase complex subunit E2 Dihydrolipoamide acyltransferase component of branched-chain alpha-keto acid dehydrogenase complex (EC 2.3.1.168) | EP10_06390 | Complex catalyze-irreversible oxidation of 2-oxoacids with production of acyl-CoA and NADH | 2.15 |
| Aspartate-semialdehyde dehydrogenase (EC 1.2.1.11) L-aspartate 4-semialdehyde + phosphate + NADP+ -> L-4-aspartyl phosphate + NADPH + H+ | Asd/ EP10_17245 | NADPH biosynthesis | 2.0 |
| Isocitrate dehydrogenase [NADP] (EC 1.1.1.42) ICIT + NADP -> AKG + CO2 + NADPH | EP10_07280 | NADPH biosynthesis | 2.2 |
| HslU—subunit of the ATP-dependent HslUV protease | EP10_16935 | Molecular chaperone | 1.3 |
| Trehalose-6-phosphate hydrolase (EC 3.2.1.93) Trehalose 6-phosphate + H2O -> glucose + glucose 6-phosphate | EP10_08195/treA | Trehalose—osmoprotectant and carbon source [52] | 2.0 |
| Tyrosine-tRNA ligase (EC 6.1.1.1) | EP10_13885 | Translation | 1.9 |
| Tripeptide aminopeptidase (EC 3.4.11.4) | pepT EP10_11320 | Protease | 1.6 |
| Excinuclease ABC subunit UvrB, ATP-dependent | EP10_00715 | DNA reparation | 3.3 |
| Cysteine methyltransferase Methylated-DNA-protein-cysteine methyltransferase (EC 2.1.1.63) | EP10_00085 | Repair of methylated DNA | 5.4 |
| Flagellar motor switch phosphatase FliY | EP10_17020 | Chemotaxis disorder | 1.8 |
2.2. The Reaction of G. icigianus Cells 10 Min after the End of the THz Irradiation (2.3 THz)
3. Discussion
4. Materials and Methods
4.1. Cell Culture Conditions
4.2. Irradiation Conditions
- Wavelength, mm 0.05–0.340
- Pulse duration, ps 50
- Pulse repetition frequency, MHz 2.8–11.2
- Average power, W up to 400
- Peak power, MW up to 1
- Minimum relative line width 3 × 10−3
4.3. Two-Dimensional Electrophoresis of Proteins
4.4. Determination of the Proteomic Profile
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Peltek, S.; Meshcheryakova, I.; Kiseleva, E.; Oshchepkov, D.; Rozanov, A.; Serdyukov, D.; Demidov, E.; Vasiliev, G.; Vinokurov, N.; Bryanskaya, A.; et al. E. coli aggregation and impaired cell division after terahertz irradiation. Sci. Rep. 2021, 11, 20464. [Google Scholar] [CrossRef] [PubMed]
- Weightman, P. Prospects for the study of biological systems with high power sources of terahertz radiation. Phys. Biol. 2012, 9, 053001. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhao, X.; Yang, K.; Liu, Y.; Liu, Y.; Fu, W.; Luo, Y. Biomedical Applications of Terahertz Spectroscopy and Imaging. Trends Biotechnol. 2016, 34, 810–824. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, S.S.; Vitiello, M.S.; Linfield, E.H.; Davies, A.G.; Hoffmann, M.C.; Booske, J.; Paoloni, C.; Gensch, M.; Weightman, P.; Williams, G.P.; et al. The 2017 terahertz science and technology roadmap. J. Phys. D 2017, 50, 043001. [Google Scholar] [CrossRef]
- Sun, L.; Zhao, L.; Peng, R.Y. Research progress in the effects of terahertz waves on biomacromolecules. Mil. Med. Res. 2021, 8, 28. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; He, Y.; Liu, K.; Fan, S.; Parrott, E.P.J.; Pickwell-MacPherson, E. Recent advances in terahertz technology for biomedical applications. Quant. Imaging Med. Surg. 2017, 7, 345–355. [Google Scholar] [CrossRef] [Green Version]
- Wu, K.; Qi, C.; Zhu, Z.; Wang, C.; Song, B.; Chang, C. Terahertz Wave Accelerates DNA Unwinding: A Molecular Dynamics Simulation Study. J. Phys. Chem. Lett. 2020, 11, 7002–7008. [Google Scholar] [CrossRef]
- Cheon, H.; Paik, J.H.; Choi, M.; Yang, H.J.; Son, J.H. Detection and manipulation of methylation in blood cancer DNA using terahertz radiation. Sci. Rep. 2019, 9, 6413. [Google Scholar] [CrossRef] [Green Version]
- Cheon, H.; Yang, H.J.; Choi, M.; Son, J.H. Effective demethylation of melanoma cells using terahertz radiation. Biomed. Opt. Express 2019, 10, 4931–4941. [Google Scholar] [CrossRef]
- Titova, L.V.; Ayesheshim, A.K.; Golubov, A.; Fogen, D.; Rodriguez-Juarez, R.; Hegmann, F.A.; Kovalchuk, O. Intense THz pulses cause H2AX phosphorylation and activate DNA damage response in human skin tissue. Biomed. Opt. Express 2013, 4, 559–568. [Google Scholar] [CrossRef]
- De Amicis, A.; Sanctis, S.D.; Cristofaro, S.D.; Franchini, V.; Lista, F.; Regalbuto, E.; Giovenale, E.; Gallerano, G.P.; Nenzi, P.; Bei, R.; et al. Biological effects of in vitro THz radiation exposure in human foetal fibroblasts. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2015, 793, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Bogomazova, A.N.; Vassina, E.M.; Goryachkovskaya, T.N.; Popik, V.M.; Sokolov, A.S.; Kolchanov, N.A.; Lagarkova, M.A.; Kiselev, S.L.; Peltek, S.E. No DNA damage response and negligible genome-wide transcriptional changes in human embryonic stem cells exposed to terahertz radiation. Sci. Rep. 2015, 5, 7749. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sergeeva, S.; Demidova, E.; Sinitsyna, O.; Goryachkovskaya, T.; Bryanskaya, A.; Semenovm, A.; Meshcheryakova, I.; Dianov, G.; Popik, V.; Peltek, S. 2.3 THz radiation: Absence of genotoxicity/mutagenicity in Escherichia coli and Salmonella typhimurium. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2016, 803–804, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Tachizaki, T.; Sakaguchi, R.; Terada, S.; Kamei, K.I.; Hirori, H. Terahertz pulse-altered gene networks in human induced pluripotent stem cells. Opt. Lett. 2020, 45, 6078–6081. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, S.; Harata, M.; Idehara, T.; Konagaya, K.; Yokoyama, G.; Hoshina, H.; Ogawa, Y. Actin polymerization is activated by terahertz irradiation. Sci. Rep. 2018, 8, 9990. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamazaki, S.; Harata, M.; Ueno, Y.; Tsubouchi, M.; Konagaya, K.; Ogawa, Y.; Isoyama, G.; Otani, C.; Hoshina, H. Propagation of THz irradiation energy through aqueous layers: Demolition of actin filaments in living cells. Sci. Rep. 2020, 10, 9008. [Google Scholar] [CrossRef]
- Yamazaki, S.; Ueno, Y.; Hosoki, R.; Saito, T.; Idehara, T.; Yamaguchi, Y.; Otani, C.; Ogawa, Y.; Harata, M.; Hoshina, H. THz irradiation inhibits cell division by affecting actin dynamics. PLoS ONE 2021, 16, e0248381. [Google Scholar] [CrossRef]
- Perera, V.R.; Newton, G.L.; Pogliano, K. Bacillithiol: A key protective thiol in Staphylococcus aureus. Expert Rev. Anti Infect. Ther. 2015, 13, 1089–1107. [Google Scholar] [CrossRef] [Green Version]
- Bock, J.; Fukuyo, Y.; Kang, S.; Phipps, M.L.; Alexandrov, L.B.; Rasmussen, K.Ø.; Bishop, A.R.; Rosen, E.D.; Martinez, J.S.; Chen, H.T.; et al. Mammalian stem cells reprogramming in response to terahertz radiation. PLoS ONE 2010, 5, e15806. [Google Scholar] [CrossRef]
- Alexandrov, B.S.; Phipps, M.L.; Alexandrov, L.B.; Booshehri, L.G.; Erat, A.; Zabolotny, J.; Mielke, C.H.; Chen, H.T.; Rodriguez, G.; Rasmussen, K.Ø.; et al. Specificity and heterogeneity of terahertz radiation effect on gene expression in mouse mesenchymal stem cells. Sci. Rep. 2013, 3, 1184. [Google Scholar] [CrossRef]
- Alexandrov, B.S.; Rasmussen, K.Ø.; Bishop, A.R.; Usheva, A.; Alexandrov, L.B.; Chong, S.; Dagon, Y.; Booshehri, L.G.; Mielke, C.H.; Phipps, M.L.; et al. Non-thermal effects of terahertz radiation on gene expression in mouse stem cells. Biomed. Opt. Express 2011, 2, 2679–2689. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Serdyukov, D.S.; Goryachkovskaya, T.N.; Mescheryakova, I.A.; Bannikova, S.V.; Kuznetsov, S.A.; Cherkasova, O.P.; Popik, V.M.; Peltek, S.E. Study on the effects of terahertz radiation on gene networks of Escherichia coli by means of fluorescent biosensors. Biomed. Opt. Express 2020, 11, 5258–5273. [Google Scholar] [CrossRef] [PubMed]
- Serdyukov, D.S.; Goryachkovskaya, T.N.; Mescheryakova, I.A.; Kuznetsov, S.A.; Popik, V.M.; Peltek, S.E. Fluorescent bacterial biosensor E. coli/pTdcR-TurboYFP sensitive to terahertz radiation. Biomed. Opt. Express 2021, 12, 705–721. [Google Scholar] [CrossRef] [PubMed]
- Demidova, E.V.; Goryachkovskaya, T.N.; Malup, T.K.; Bannikova, S.V.; Semenov, A.I.; Vinokurov, N.A.; Kolchanov, N.A.; Popik, V.M.; Peltek, S.E. Studying the non-thermal effects of terahertz radiation on E. coli/pKatG-GFP biosensor cells. Bioelectromagnetics 2013, 34, 15–21. [Google Scholar] [CrossRef]
- Demidova, E.V.; Goryachkovskaya, T.N.; Mescheryakova, I.A.; Malup, T.K.; Semenov, A.I.; Vinokurov, N.A.; Kolchanov, N.A.; Popik, V.M.; Peltek, S.E. Impact of terahertz radiation on stress-sensitive genes of E. coli cell. IEEE Transact. Terahertz Sci. Technol. 2016, 6, 435–441. [Google Scholar] [CrossRef]
- Bryanskaya, A.V.; Rozanov, A.S.; Slynko, N.M.; Shekhovtsov, S.V.; Peltek, S.E. Geobacillus icigianus sp. nov., a thermophilic bacterium isolated from a hot spring. Int. J. Syst. Evol. Microbiol. 2015, 65 Pt 3, 864–869. [Google Scholar] [CrossRef]
- Ferreira, E.; Giménez, R.; Aguilera, L.; Guzmán, K.; Aguilar, J.; Badia, J.; Baldomà, L. Protein interaction studies point to new functions for Escherichia coli glyceraldehyde-3-phosphate dehydrogenase. Res. Microbiol. 2013, 164, 145–154. [Google Scholar] [CrossRef] [Green Version]
- Ferreira, E.; Giménez, R.; Cañas, M.A.; Aguilera, L.; Aguilar, J.; Badia, J.; Baldomà, L. Glyceraldehyde-3-phosphate dehydrogenase is required for efficient repair of cytotoxic DNA lesions in Escherichia coli. Int. J. Biochem. Cell Biol. 2015, 60, 202–212. [Google Scholar] [CrossRef] [Green Version]
- Ul Haq, I.; Brantl, S. Moonlighting in Bacillus subtilis: The Small Proteins SR1P and SR7P Regulate the Moonlighting Activity of Glyceraldehyde 3-Phosphate Dehydrogenase A (GapA) and Enolase in RNA Degradation. Microorganisms 2021, 9, 1046. [Google Scholar] [CrossRef]
- Prüss, B.M.; Nelms, J.M.; Park, C.; Wolfe, A.J. Mutations in NADH:ubiquinone oxidoreductase of Escherichia coli affect growth on mixed amino acids. J. Bacteriol. 1994, 176, 2143–2150. [Google Scholar] [CrossRef]
- McFall, E.; Newman, E.B. Amino acids as carbon sources. In Escherichia coli and Salmonella: Cellular and Molecular Biology; Neidhardt, F.C., Currtiss, R., III, Ingraham, J.L., Lin, E.C.C., Low, K.B., Magasanik, B., Reznikoff, W.S., Riley, M., Schaechter, M., Umbarger, H.E., Eds.; ASM Press: Washington, DC, USA, 1996; pp. 358–379. [Google Scholar]
- Janosi, L.; Ricker, R.; Kaji, A. Dual functions of ribosome recycling factor in protein biosynthesis: Disassembling the termination complex and preventing translational errors. Biochimie 1996, 78, 959–969. [Google Scholar] [CrossRef] [PubMed]
- Kusuya, Y.; Kurokawa, K.; Ishikawa, S.; Ogasawara, N.; Oshima, T. Transcription factor GreA contributes to resolving promoter-proximal pausing of RNA polymerase in Bacillus subtilis cells. J. Bacteriol. 2011, 193, 3090–3099. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, K.; Jiang, T.; Yu, B.; Wang, L.; Gao, C.; Ma, C.; Xu, P.; Ma, Y. Transcription elongation factor GreA has functional chaperone activity. PLoS ONE 2012, 7, e47521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Imashimizu, M.; Tanaka, M.; Hoshina, H. Gre factors prevent thermal and mechanical stresses induced by terahertz irradiation during transcription. Genes Cells 2021, 26, 56–64. [Google Scholar] [CrossRef]
- Newton, G.L.; Rawat, M.; La Clair, J.J.; Jothivasan, V.K.; Budiarto, T.; Hamilton, C.J.; Claiborne, A.; Helmann, J.D.; Fahey, R.C. Bacillithiol is an antioxidant thiol produced in Bacilli. Nat. Chem. Biol. 2009, 5, 625–627. [Google Scholar] [CrossRef]
- Gaballa, A.; Newton, G.L.; Antelmann, H.; Parsonage, D.; Upton, H.; Rawat, M.; Claiborne, A.; Fahey, R.C.; Helmann, J.D. Biosynthesis and functions of bacillithiol, a major low-molecular-weight thiol in Bacilli. Proc. Natl. Acad. Sci. USA 2010, 107, 6482–6486. [Google Scholar] [CrossRef] [Green Version]
- Lu, J.; Holmgren, A. The thioredoxin antioxidant system. Free Radic. Biol. Med. 2014, 66, 75–87. [Google Scholar] [CrossRef]
- Lu, J.; Yang, F.; Li, Y.; Zhang, X.; Xia, B.; Jin, C. Reversible conformational switch revealed by the redox structures of Bacillus subtilis thiol peroxidase. Biochem. Biophys. Res. Commun. 2008, 373, 414–418. [Google Scholar] [CrossRef]
- Kobayashi, M.; Ohara-Nemoto, Y.; Kaneko, M.; Hayakawa, H.; Sekiguchi, M.; Yamamoto, K. Potential of Escherichia coli GTP cyclohydrolase II for hydrolyzing 8-oxo-dGTP, a mutagenic substrate for DNA synthesis. J. Biol. Chem. 1998, 273, 26394–26399. [Google Scholar] [CrossRef] [Green Version]
- Averianova, L.A.; Balabanova, L.A.; Son, O.M.; Podvolotskaya, A.B.; Tekutyeva, L.A. Production of Vitamin B2 (Riboflavin) by Microorganisms: An Overview. Front. Bioeng. Biotechnol. 2020, 8, 570828. [Google Scholar] [CrossRef]
- Belitsky, B.R. Physical and enzymological interaction of Bacillus subtilis proteins required for de novo pyridoxal 5′-phosphate biosynthesis. J. Bacteriol. 2004, 186, 1191–1196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burns, K.E.; Xiang, Y.; Kinsland, C.L.; McLafferty, F.W.; Begley, T.P. Reconstitution and biochemical characterization of a new pyridoxal-5′-phosphate biosynthetic pathway. J. Am. Chem. Soc. 2005, 127, 3682–3683. [Google Scholar] [CrossRef] [PubMed]
- Raschle, T.; Amrhein, N.; Fitzpatrick, T.B. On the two components of pyridoxal 5′-phosphate synthase from Bacillus subtilis. J. Biol. Chem. 2005, 280, 32291–32300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Richts, B.; Rosenberg, J.; Commichau, F.M. A Survey of pyridoxal 5′-phosphate-dependent proteins in the gram-positive model bacterium Bacillus subtilis. Front. Mol. Biosci. 2019, 6, 32. [Google Scholar] [CrossRef] [Green Version]
- Hoffarth, E.R.; Rothchild, K.W.; Ryan, K.S. Emergence of oxygen- and pyridoxal phosphate-dependent reactions. FEBS J. 2020, 287, 1403–1428. [Google Scholar] [CrossRef]
- Jules, M.; Le Chat, L.; Aymerich, S.; Le Coq, D. The Bacillus subtilis ywjI (glpX) gene encodes a class II fructose-1,6-bisphosphatase, functionally equivalent to the class III Fbp enzyme. J. Bacteriol. 2009, 191, 3168–3171. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; Brevet, A.; Fromant, M.; Lévêque, F.; Schmitter, J.M.; Blanquet, S.; Plateau, P. Pyrophosphatase is essential for growth of Escherichia coli. J. Bacteriol. 1990, 172, 5686–5689. [Google Scholar] [CrossRef] [Green Version]
- Baykov, A.A.; Cooperman, B.S.; Goldman, A.; Lahti, R. Cytoplasmic inorganic pyrophosphatase. Prog. Mol. Subcell Biol. 1999, 23, 127–150. [Google Scholar] [CrossRef]
- Serrano-Bueno, G.; Hernández, A.; López-Lluch, G.; Pérez-Castiñeira, J.R.; Navas, P.; Serrano, A. Inorganic pyrophosphatase defects lead to cell cycle arrest and autophagic cell death through NAD+ depletion in fermenting yeast. J. Biol. Chem. 2013, 288, 13082–13092. [Google Scholar] [CrossRef] [Green Version]
- Pinchuk, G.E.; Rodionov, D.A.; Yang, C.; Li, X.; Osterman, A.L.; Dervyn, E.; Geydebrekht, O.V.; Reed, S.B.; Romine, M.F.; Collart, F.R.; et al. Genomic reconstruction of Shewanella oneidensis MR-1 metabolism reveals a previously uncharacterized machinery for lactate utilization. Proc. Natl. Acad. Sci. USA 2009, 106, 2874–2879. [Google Scholar] [CrossRef]
- Helfert, C.; Gotsche, S.; Dahl, M.K. Cleavage of trehalose-phosphate in Bacillus subtilis is catalysed by a phospho-alpha-(1-1)-glucosidase encoded by the treA gene. Mol. Microbiol. 1995, 16, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Strøm, A.R.; Kaasen, I. Trehalose metabolism in Escherichia coli: Stress protection and stress regulation of gene expression. Mol. Microbiol. 1993, 8, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Phadtare, S. Recent developments in bacterial cold-shock response. Curr. Issues Mol. Biol. 2004, 6, 125–136. [Google Scholar]
- Dębski, J.; Stojowska-Swędrzyńska, K.; Sominka, H.; Bukrejewska, M.; Laskowska, E. Trehalose protects Escherichia coli against carbon stress manifested by protein acetylation and aggregation. Mol. Microbiol. 2019, 112, 866–880. [Google Scholar] [CrossRef]
- Sauer, U.; Hatzimanikatis, V.; Hohmann, H.P.; Manneberg, M.; van Loon, A.P.; Bailey, J.E. Physiology and metabolic fluxes of wild-type and riboflavin-producing Bacillus subtilis. Appl. Environ. Microbiol. 1996, 62, 3687–3696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Typas, A.; Banzhaf, M.; Gross, C.A.; Vollmer, W. From the regulation of peptidoglycan synthesis to bacterial growth and morphology. Nat. Rev. Microbiol. 2012, 10, 123–136. [Google Scholar] [CrossRef] [Green Version]
- Sircar, R.; Greenswag, A.R.; Bilwes, A.M.; Gonzalez-Bonet, G.; Crane, B.R. Structure and activity of the flagellar rotor protein FliY: A member of the CheC phosphatase family. J. Biol. Chem. 2013, 288, 13493–13502. [Google Scholar] [CrossRef] [Green Version]
- Chiang, P.K.; Gordon, R.K.; Tal, J.; Zeng, G.C.; Doctor, B.P.; Pardhasaradhi, K.; McCann, P.P. S-Adenosylmethionine and methylation. FASEB J. 1996, 10, 471–480. [Google Scholar] [CrossRef] [Green Version]
- Schuch, R.; Garibian, A.; Saxild, H.H.; Piggot, P.J.; Nygaard, P. Nucleosides as a carbon source in Bacillus subtilis: Characterization of the drm-pupG operon. Microbiology 1999, 145 Pt 10, 2957–2966. [Google Scholar] [CrossRef] [Green Version]
- Seong, I.S.; Oh, J.Y.; Lee, J.W.; Tanaka, K.; Chung, C.H. The HslU ATPase acts as a molecular chaperone in prevention of aggregation of SulA, an inhibitor of cell division in Escherichia coli. FEBS Lett. 2000, 477, 224–229. [Google Scholar] [CrossRef] [Green Version]
- Hernández, V.M.; Arteaga, A.; Dunn, M.F. Diversity, properties and functions of bacterial arginases. FEMS Microbiol. Rev. 2021, 23, fuab034. [Google Scholar] [CrossRef] [PubMed]
- Goryachkovskaya, T.N.; Konstantinova, S.H.; Meshcheriakova, I.A.; Bannikova, S.V.; Demidov, E.A.; Bryanskaya, A.V.; Scheglov, M.A.; Semenov, A.I.; Oshchepkov, D.Y.; Popik, V.M.; et al. The impact of terahertz radiation on an extremophilic archaean Halorubrum saccharovorum proteome. Vavilovskii Zhurnal Genet. Selektsii. 2016, 20, 869–875. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; El-Hajj, Z.W.; Newman, E. Deficiency in L-serine deaminase interferes with one-carbon metabolism and cell wall synthesis in Escherichia coli K-12. J. Bacteriol. 2010, 192, 5515–5525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, C.Y.; Weng, Y.T.; Hwang, L.Y.; Hu, H.T.; Shih, P.S.; Kuan, J.E.; Wu, K.F.; Wu, W.F. Specific regions of the SulA protein recognized and degraded by the ATP-dependent ClpYQ (HslUV) protease in Escherichia coli. Microbiol. Res. 2019, 220, 21–31. [Google Scholar] [CrossRef]
- Thomas, C.M.; Nielsen, K.M. Mechanisms of, and barriers to, horizontal gene transfer between bacteria. Nat. Rev. Microbiol. 2005, 3, 711–721. [Google Scholar] [CrossRef]
- Wion, D.; Casadesus, J. N(6)-methyl-adenine: An epigenetic signal for DNA-protein interactions. Nat. Rev. Microbiol. 2006, 4, 183–192. [Google Scholar] [CrossRef]
- Lobner-Olesen, A.; Skovgaard, O.; Marinus, M.G. Dam methylation: Coordinating cellular processes. Curr. Opin. Microbiol. 2005, 8, 154–160. [Google Scholar] [CrossRef]
- Topanurak, S.; Sinchaikul, S.; Sookkheo, B.; Phutrakul, S.; Chen, S.T. Functional proteomics and correlated signaling pathway of the thermophilic bacterium Bacillus stearothermophilus TLS33 under cold-shock stress. Proteomics 2005, 5, 4456–4471. [Google Scholar] [CrossRef]
- Wang, Q.; Cen, Z.; Zhao, J. The survival mechanisms of thermophiles at high temperatures: An angle of omics. Physiology 2015, 30, 97–106. [Google Scholar] [CrossRef]
- Ranawat, P.; Rawat, S. Stress response physiology of thermophiles. Arch. Microbiol. 2017, 199, 391–414. [Google Scholar] [CrossRef]
- Bradford, M.M. A Rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Bryanskaya, A.V.; Rozanov, A.S.; Logacheva, M.D.; Kotenko, A.V.; Peltek, S.E. Draft genome sequence of Geobacillus icigianus strain G1w1T isolated from hot springs in the Valley of geysers, Kamchatka (Russian Federation). Genome Announc. 2014, 2, e01098-14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kulyashov, M.; Peltek, S.E.; Akberdin, I.R. A Genome-scale metabolic model of 2,3-butanediol production by thermophilic bacteria Geobacillus icigianus. Microorganisms 2020, 8, 1002. [Google Scholar] [CrossRef] [PubMed]
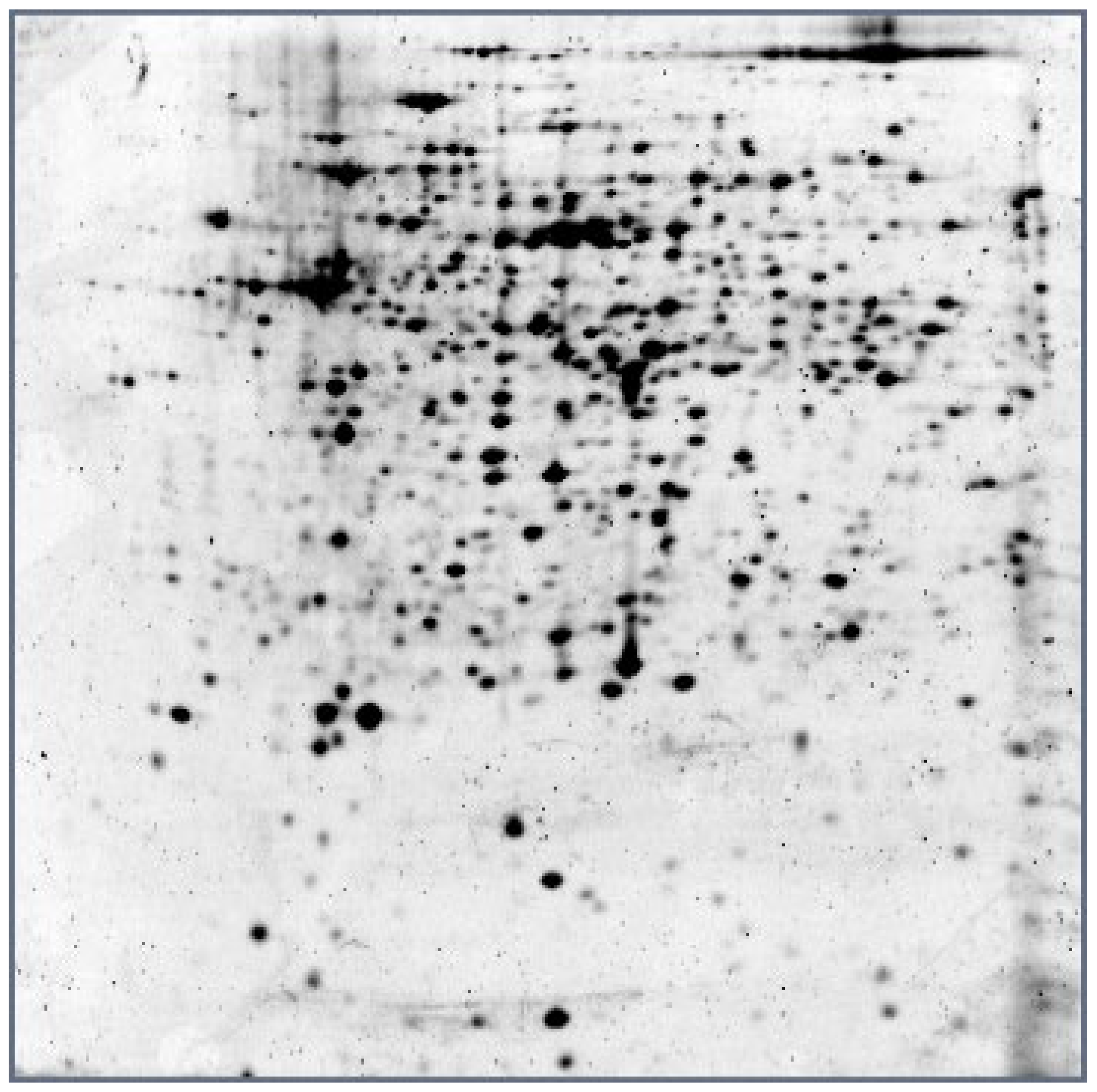
| Protein/Reaction | Gene/Locus | Process/Function | ∆ |
|---|---|---|---|
| Electron transfer flavoprotein subunit alpha/FixB family protein ETFb Electron transfer flavoprotein subunit beta/FixA family protein, ETFa | EP10_09770 EP10_09765 | ETF—heterodimer, a key component of the electron transfer chain | 1.7 1.3 |
| Elongation factor Tu | tuf/EP10_11775 | Translation elongation factor, GTPase | 1.4 |
| DNA-directed RNA polymerase subunit alpha | rpoA/EP10_11630 | Transcription | 2.2 * |
| NAD-dependent malic enzyme Mal + NAD -> Pyr + NADH + CO2 | EP10_15870 | NADH and pyruvate biosynthesis | 1.3 * |
| Acetyl-CoA acetyltransferase (EC 2.3.1.9) 2 Acetyl-CoA -> CoA + acetoacetyl-CoA | thlA/EP10_03775 | Acetyl-CoA degradation | 1.5 * |
| Molecular chaperone DnaK | EP10_05360 | Molecular chaperone | 1.35 * |
| Phosphopentomutase (EC 5.4.2.7) (2-Deoxy)-alpha-D-ribose 1-phosphate -> (2-deoxy)-D-ribose 5-phosphate | EP10_18365 | Ribonucleoside catabolism | 1.6 |
| Methionine adenosyltransferase (EC 2.5.1.6) ATP + L-methionine + H2O -> phosphate + diphosphate + S-adenosyl-L-methionine | metK/EP10_09910 | S-Adenosyl-L-methionine biosynthesis, SAM—methyl donor | 1.4 |
| Acid sugar phosphatase (TIGR01457 family HAD-type hydrolase) | EP10_04705 | 1.5 | |
| Amino acid ABC transporter ATP-binding protein | EP10_06290 | Amino acid transport | 1.5 |
| Bifunctional 3,4-dihydroxy-2-butanone 4-phosphate (DHBP) synthase (EC 4.1.99.12)/GTP cyclohydrolase II (EC 3.5.4.25) | EP10_18260 | Riboflavin biosynthesis, protects DNA from oxygen radicals | 2.1 * |
| Protein/Reaction | Gene/Locus | Process/Function | ∆ |
|---|---|---|---|
| ATP synthase alpha chain (EC 7.1.2.2) ADP + PI + [H+]ex <-> ATΦ + H2O + [H+]in | atpA/EP10_10185 | ATP biosynthesis | 1.1 ± 0.04 |
| Type I glyceraldehyde-3-phosphate dehydrogenase (NAD+), EC 1.2.1.12 G3P + PI + NAD -> NADH + 1, 3PDG | gapA/ EP10_12960 | NADH synthesis | 1.3 |
| Pyruvate dehydrogenase (Acetyl-transferring) E1 component subunit alpha (EC 1.2.4.1) PYR + NAD + COA -> ACCOA + NADH + CO2 | pdhA/EP10_14290 | Complex production of NADH and AcCoA | 2.5 |
| Dihydrolipoamide acetyltransferase component of pyruvate dehydrogenase complex, subunit E2 (EC 2.3.1.12) ACLIPO + COA -> ACCOA + DLIPO | EP10_14300 | Complex production of NADH and AcCoA | 2.8 |
| 2-Oxo acid dehydrogenase complex subunit E2 Dihydrolipoamide acyltransferase component of branched-chain alpha-keto acid dehydrogenase complex (EC 2.3.1.168) | EP10_06390 | Complex production of acyl-CoA and NADH | 1.6 |
| Leucine dehydrogenase, NAD (+)-dependent (EC 1.4.1.9) L-leucine + H2O + NAD+ -> 4-methyl-2-oxopentanoate + NH3 + NADH + H+ | EP10_06415 | NADH biosynthesis | 1.3 |
| Glutamate dehydrogenase, NAD-specific (EC 1.4.1.2) L-glutamate + H2O + NAD+ -> 2-oxoglutarate + NH3 + NADH + H+. | EP10_12170 | NADH biosynthesis | 1.3 |
| Glycine dehydrogenase (decarboxylating) (glycine cleavage system P1 protein) (EC 1.4.4.2) Gly + THF + NAD+ -> MetTHF + CO2 + NH3 + NADH | EP10_05755 | NADH biosynthesis | 1.6 |
| Aspartate-semialdehyde dehydrogenase (EC 1.2.1.11) L-aspartate 4-semialdehyde + phosphate + NADP+ -> L-4-aspartyl phosphate + NADPH + H+ | Asd/EP10_17245 | NADPH biosynthesis | 1.2 |
| 6-Phosphogluconate dehydrogenase, NADP (+)-dependent (EC 1.1.1.44) 6PG + NADP -> D-ribulose 5-phosphate + CO2 + NADPH | EP10_18520 | PPP, NADPH biosynthesis | 2.2 |
| Phosphoenolpyruvate carboxykinase (ATP) (EC 4.1.1.49) ATP + oxaloacetate -> ADP + phosphoenolpyruvate + CO2 | pckA/EP10_09915 | Enhances pyruvate pool | 1.3 |
| Serine hydroxymethyltransferase (EC 2.1.2.1) MetTHF + glycine + H2O -> THF + L-serine | EP10_10230 | L-serine biosynthesis | 1.8 |
| UDP-N-acetylmuramoyl(AM)-tripeptide-D-alanyl-D-alanine ligase, ATP-dependent (EC 6.3.2.10) ATΦ + UDP-N-AM-tripeptide + D-alanyl-D-alanine -> ADP + Pi + N-AM-pentapeptide | MurF/EP10_18840 | Peptidoglycan biosynthesis | 1.4 |
| Arginase (L-arginine amidinohydrolase, EC 3.5.3.1) L-arginine + H2O -> L-ornithine + urea | rocF/EP10_17840 | Urea and ornithine can be further utilized as nitrogen and carbon sources | 1.3 |
| Aspartate-tRNA ligase, Aspartyl-tRNA synthetase (EC 6.1.1.12) | EP10_01675 | Translation | 1.3 |
| Proline-tRNA ligase, Prolyl-tRNA synthetase (EC 6.1.1.15) | EP10_17150 | Translation | 1.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bannikova, S.; Khlebodarova, T.; Vasilieva, A.; Mescheryakova, I.; Bryanskaya, A.; Shedko, E.; Popik, V.; Goryachkovskaya, T.; Peltek, S. Specific Features of the Proteomic Response of Thermophilic Bacterium Geobacillus icigianus to Terahertz Irradiation. Int. J. Mol. Sci. 2022, 23, 15216. https://doi.org/10.3390/ijms232315216
Bannikova S, Khlebodarova T, Vasilieva A, Mescheryakova I, Bryanskaya A, Shedko E, Popik V, Goryachkovskaya T, Peltek S. Specific Features of the Proteomic Response of Thermophilic Bacterium Geobacillus icigianus to Terahertz Irradiation. International Journal of Molecular Sciences. 2022; 23(23):15216. https://doi.org/10.3390/ijms232315216
Chicago/Turabian StyleBannikova, Svetlana, Tamara Khlebodarova, Asya Vasilieva, Irina Mescheryakova, Alla Bryanskaya, Elizaveta Shedko, Vasily Popik, Tatiana Goryachkovskaya, and Sergey Peltek. 2022. "Specific Features of the Proteomic Response of Thermophilic Bacterium Geobacillus icigianus to Terahertz Irradiation" International Journal of Molecular Sciences 23, no. 23: 15216. https://doi.org/10.3390/ijms232315216
APA StyleBannikova, S., Khlebodarova, T., Vasilieva, A., Mescheryakova, I., Bryanskaya, A., Shedko, E., Popik, V., Goryachkovskaya, T., & Peltek, S. (2022). Specific Features of the Proteomic Response of Thermophilic Bacterium Geobacillus icigianus to Terahertz Irradiation. International Journal of Molecular Sciences, 23(23), 15216. https://doi.org/10.3390/ijms232315216






