HPV-Negative Adenocarcinomas of the Uterine Cervix: From Molecular Characterization to Clinical Implications
Abstract
1. Introduction
2. Critical Issues in Diagnosing Non-HPV-Related Cervical Lesions
HPV-Negative Cervical Cancer
- Real HPV-negative cervical cancer;
- False HPV-negative case;
- Incorrect classification of a non-cervical cancer.
- a.
- Real HPV-negative cervical cancer
- b.
- False HPV-negative case
- -
- Loss of targeted HPV DNA fragment
- -
- Low viral load in latent HPV infection
- -
- Cervical cancer caused by low-risk HPV-genotype
- -
- False negative HPV-test (incorrect sampling/pre-analytical errors)
- c.
- Incorrect classification of a non-cervical cancer
3. Classification of Endocervical Adenocarcinoma
4. Clinical Features of Human Papillomavirus-Negative Adenocarcinoma
4.1. Gastric-Type Adenocarcinoma (GCA)
4.2. Clear Cell Adenocarcinoma (CCC)
4.3. Mesonephric Adenocarcinoma
4.4. Endometrioid Adenocarcinoma (ENAC)
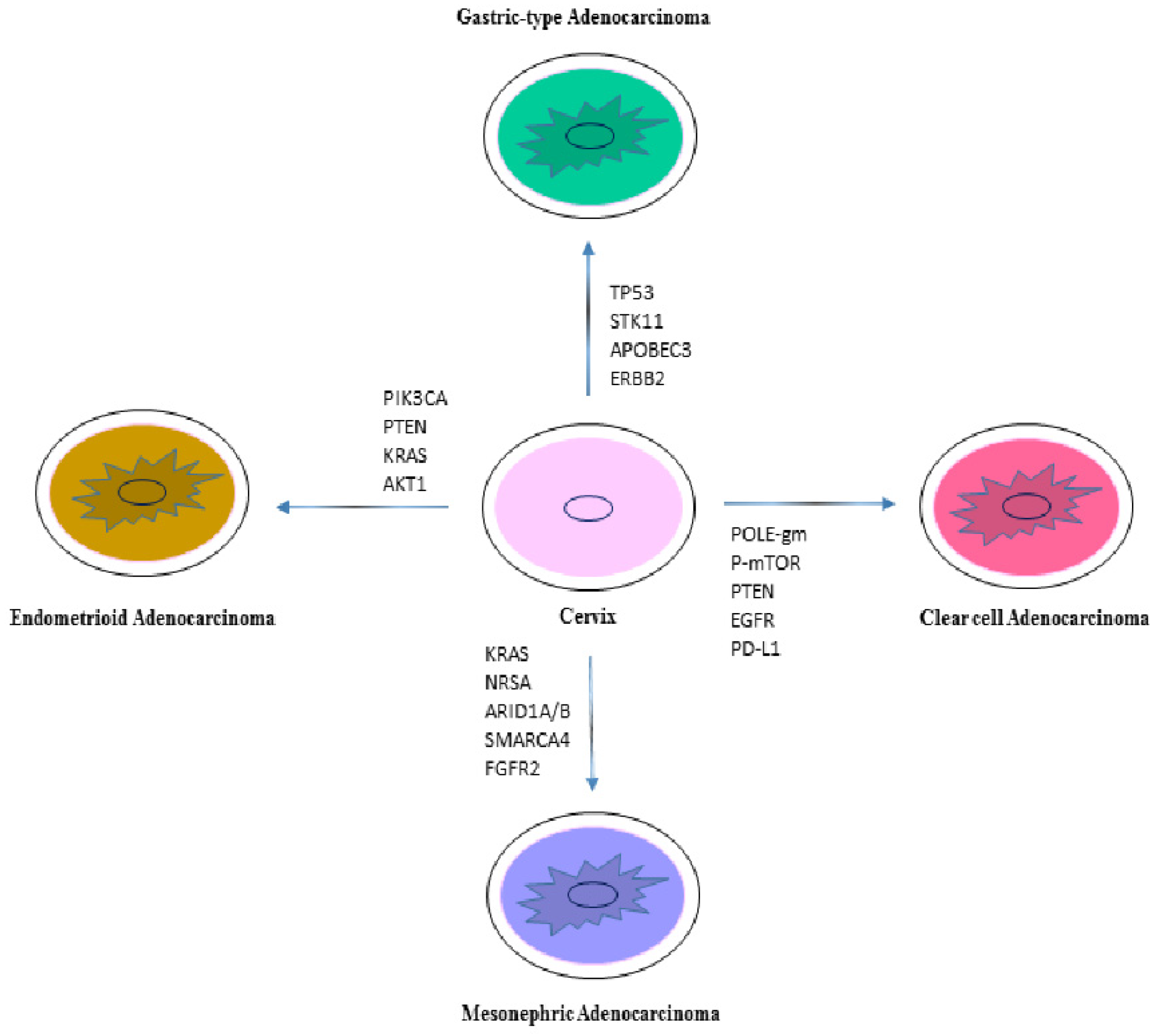
5. Molecular Characterization of Non-HPV Associated Adenocarcinoma
5.1. Gastric-Type Adenocarcinoma
5.2. Clear Cell Adenocarcinoma (CCC)
5.3. Mesonephric Adenocarcinoma
5.4. Endometrioid Adenocarcinoma
5.5. PD1 and PDL1 Expression in NHPVA
6. Treatment of Human Papillomavirus-Negative Cervical Cancer
6.1. Staging
- TNM and FIGO stage, including maximum tumor size and detailed description of extracervical tumor extension and nodal involvement.
- Pathological tumor type.
- Depth of cervical stromal invasion and a minimum thickness of uninvolved cervical stroma.
- Presence (or absence) of lymphovascular space involvement (LVSI).
- Presence or absence of distant metastases.
- In the early stage (T1a, T1b1, T2a1), surgical/pathological staging of pelvic lymph nodes is the gold standard to assess the prognosis and guide treatment (except for T1a1 and no LVSI).
- In locally advanced cervical cancer T1b2 and higher (except T2a1) or early stage disease with suspicious lymph nodes on imaging, positron emission tomography-computed tomography (PET-CT) or chest/abdomen computed tomography (CT) is recommended for assessment of nodal and distant disease.
- Paraaortic lymph node dissection, at least up to the inferior mesenteric artery, may be considered in locally advanced cervical cancer with negative paraaortic lymph nodes on imaging for staging purposes.
6.2. Mangement of Stage T1a
6.3. Management of Other Stages (T1b/TIV)
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| AKT1 | Serine-threonine protein kinase AKT1 |
| APOBEC3 | Apolipoprotein B mRNA editing enzyme, catalytic subunit 3 |
| ARID1A | AT-rich interactive domain-containing protein 1A |
| ATM | Ataxia telangiectasia mutated |
| BCOR | BCL6 corepressor |
| BRCA2 | Breast cancer gene 2 |
| CCC | Clear cell adenocarcinoma |
| CDK12 | Cyclin dependent kinase 12 |
| CDKN2A | Cyclin dependent kinase inhibitor 2 |
| CIN | Cervical intraepithelial neoplasia |
| CTNNB1 | Catenin Beta 1 |
| DES | Diethylstilbestrol |
| DNA | Deoxyribonucleic acid |
| ECA | endocervical adenocarcinoma |
| EGFR | Epidermal growth factor receptor |
| ELF | Ets domain transcription factor |
| ENAC | Endometrioid adenocarcinoma |
| ERB2 | Erb-B2 receptor tyrosine kinase 2 |
| FBXW7 | F-box and WD repeat domain containing 7 |
| FDA | Food and Drug Administration |
| FGFR2 | fibroblast growth factor receptor 2 |
| FGFR4 | Fibroblast growth factor receptor 4 |
| GAIS | Gastric-type adenocarcinoma in situ |
| GCA | Gastric-type adenocarcinoma |
| GNAS | Guanine nucleotide binding protein |
| H&E | hematoxylin eosin |
| HER2 | Human epidermal growth factor receptor 2 |
| HLA-B | Human leukocyte antigen-B |
| HPV | Human papillomavirus |
| IECC | International endocervical criteria and classification |
| IHC | immunohistochemistry |
| ISGyP | International Society of Gynecological Pathologists |
| KMT2D | Lysine methyltransferase 2D |
| KRAS | Kirsten rat sarcoma |
| LEGH | Lobular endocervical glandular hyperplasia |
| MDA | Minimal-deviation adenocarcinoma |
| MDM2 | Mouse double minute 2 |
| MECOM | MDS1 and EVI1 complex locus |
| MMA | DNA mismatch repair |
| MRI | Magnetic resonance imaging |
| MSH2 | MutS homolog 2 |
| MSH6 | MutS homolog 6 |
| MSI-H | microsatellite instability-high |
| NGS | Next generation sequencing |
| NHPVA | non-HPV adenocarcinoma |
| NTRK3 | Neurotrophic receptor tyrosine kinase 3 |
| p-mTOR | Phosphorylated mammalian target of rapamycin |
| PCR | Polymerase chain reaction |
| PD-L1 | programmed cell death ligand 1 |
| PI3K-AKT | Phosphatidylinositol-3-kinase |
| PIK3CA | Phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha |
| POLE | polymerase epsilon |
| PTEN | Phosphatase and tensin homolog |
| PTPRS | Protein tyrosine phosphatase receptor type S |
| RISH | RNA in-situ hybridization |
| RNA | Ribonucleic acid |
| SCC | Squamous cell carcinoma |
| SLX4 | Structure-specific endonuclease subunit |
| SMAD4 | Mothers against decapentaplegic homolog 4 |
| SMARCA4 | SWI/SNF related, matrix associated, actin dependent regulator of chromatin, subfamily A, member 4 |
| STK11 | Serine/threonine kinase 11 |
| TP53 | Tumor protein p53 |
| WHO | World Health Organization |
References
- Molijn, A.; Jenkins, D.; Chen, W.; Zhang, X.; Pirog, E.; Enqi, W.; Liu, B.; Schmidt, J.; Cui, J.; Qiao, Y.; et al. The complex relationship between human papillomavirus and cervical adenocarcinoma. Int. J. Cancer 2016, 138, 409–416. [Google Scholar] [CrossRef]
- Massad, L.S.; Einstein, M.H.; Huh, W.K.; Katki, H.A.; Kinney, W.K.; Schiffman, M.; Solomon, D.; Wentzensen, N.; Lawson, H.W. 2012 updated consensus guidelines for the management of abnormal cervical cancer screening tests and cancer precursors. Obstet. Gynecol. 2013, 121, 829–846. [Google Scholar] [CrossRef] [PubMed]
- Liverani, C.A.; Di Giuseppe, J.; Giannella, L.; Delli Carpini, G.; Ciavattini, A. Cervical Cancer Screening Guidelines in the Postvaccination Era: Review of the Literature. J. Oncol. 2020, 2020, 8887672. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, H.; Shiraishi, K.; Kato, T. Molecular Pathology of Human Papilloma Virus-Negative Cervical Cancers. Cancers 2021, 13, 6351. [Google Scholar] [CrossRef] [PubMed]
- Xing, B.; Guo, J.; Sheng, Y.; Wu, G.; Zhao, Y. Human Papillomavirus-Negative Cervical Cancer: A Comprehensive Review. Front. Oncol. 2021, 10, 606335. [Google Scholar] [CrossRef] [PubMed]
- Giannella, L.; Giorgi Rossi, P.; Delli Carpini, G.; Di Giuseppe, J.; Bogani, G.; Gardella, B.; Monti, E.; Liverani, C.A.; Ghelardi, A.; Insinga, S.; et al. Age-related distribution of uncommon HPV genotypes in cervical intraepithelial neoplasia grade 3. Gynecol. Oncol. 2021, 161, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Pirog, E.C. Cervical Adenocarcinoma: Diagnosis of Human Papillomavirus-Positive and Human Papillomavirus-Negative Tumors. Arch. Pathol. Lab. Med. 2017, 141, 1653–1667. [Google Scholar] [CrossRef]
- Jenkins, D.; Molijn, A.; Kazem, S.; Pirog, E.C.; Alemany, L.; de Sanjosé, S.; Dinjens, W.; Quint, W. Molecular and pathological basis of HPV-negative cervical adenocarcinoma seen in a global study. Int. J. Cancer 2020, 147, 2526–2536. [Google Scholar] [CrossRef]
- Seoud, M.; Tjalma, W.A.; Ronsse, V. Cervical adenocarcinoma: Moving towards better prevention. Vaccine 2011, 29, 9148–9158. [Google Scholar] [CrossRef]
- Rettig, E.M.; D’Souza, G. Epidemiology of head and neck cancer. Surg. Oncol. Clin. N. Am. 2015, 24, 379–396. [Google Scholar] [CrossRef]
- Ciavattini, A.; Giannella, L.; De Vincenzo, R.; Di Giuseppe, J.; Papiccio, M.; Lukic, A.; Delli Carpini, G.; Perino, A.; Frega, A.; Sopracordevole, F.; et al. HPV Vaccination: The Position Paper of the Italian Society of Colposcopy and Cervico-Vaginal Pathology (SICPCV). Vaccines 2020, 8, 354. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Franceschi, S.; Howell-Jones, R.; Snijders, P.J.; Clifford, G.M. Human papillomavirus type distribution in 30,848 invasive cervical cancers worldwide: Variation by geographical region, histological type and year of publication. Int. J. Cancer 2011, 128, 927–935. [Google Scholar] [CrossRef] [PubMed]
- Pirog, E.C.; Lloveras, B.; Molijn, A.; Tous, S.; Guimerà, N.; Alejo, M.; Clavero, O.; Klaustermeier, J.; Jenkins, D.; Quint, W.G.; et al. HPV prevalence and genotypes in different histological subtypes of cervical adenocarcinoma, a worldwide analysis of 760 cases. Mod. Pathol. 2014, 27, 1559–1567. [Google Scholar] [CrossRef] [PubMed]
- Cancer Genome Atlas Research Network; Albert Einstein College of Medicine; Analytical Biological Services. Integrated genomic and molecular characterization of cervical cancer. Nature 2017, 543, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Petry, K.U.; Liebrich, C.; Luyten, A.; Zander, M.; Iftner, T. Surgical staging identified false HPV-negative cases in a large series of invasive cervical cancers. Papillomavirus Res. 2017, 4, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Blatt, A.J.; Kennedy, R.; Luff, R.D.; Austin, R.M.; Rabin, D.S. Comparison of cervical cancer screening results among 256,648 women in multiple clinical practices. Cancer Cytopathol. 2015, 123, 282–288, Corrected in Cancer Cytopathol. 2016, 124, 362–363. [Google Scholar] [CrossRef]
- Guan, P.; Howell-Jones, R.; Li, N.; Bruni, L.; De Sanjosé, S.; Franceschi, S.; Clifford, G.M. Human papillomavirus types in 115,789 HPV-positive women: A meta-analysis from cervical infection to cancer. Int. J. Cancer 2012, 131, 2349–2359. [Google Scholar] [CrossRef]
- Clifford, G.M.; Smith, J.S.; Plummer, M.; Muñoz, N.; Franceschi, S. Human papillomavirus types in invasive cervical cancer worldwide: A meta-analysis. Br. J. Cancer 2003, 88, 63–73. [Google Scholar] [CrossRef]
- Rodríguez-Carunchio, L.; Soveral, I.; Steenbergen, R.; Torné, A.; Martinez, S.; Fusté, P.; Pahisa, J.; Marimon, L.; Ordi, J.; Del Pino, M. HPV-negative carcinoma of the uterine cervix: A distinct type of cervical cancer with poor prognosis. BJOG 2015, 122, 119–127. [Google Scholar] [CrossRef]
- Holl, K.; Nowakowski, A.M.; Powell, N.; McCluggage, W.G.; Pirog, E.C.; Collas De Souza, S.; Tjalma, W.A.; Rosenlund, M.; Fiander, A.; Sánchez, M.C.; et al. Human papillomavirus prevalence and type-distribution in cervical glandular neoplasias: Results from a European multinational epidemiological study. Int. J. Cancer 2015, 137, 2858–2868. [Google Scholar] [CrossRef]
- Ueno, S.; Sudo, T.; Oka, N.; Wakahashi, S.; Yamaguchi, S.; Fujiwara, K.; Mikami, Y.; Nishimura, R. Absence of human papillomavirus infection and activation of PI3K-AKT pathway in cervical clear cell carcinoma. Int. J. Gynecol. Cancer 2013, 23, 1084–1091. [Google Scholar] [CrossRef] [PubMed]
- Pirog, E.C.; Park, K.J.; Kiyokawa, T.; Zhang, X.; Chen, W.; Jenkins, D.; Quint, W. Gastric-type Adenocarcinoma of the Cervix: Tumor With Wide Range of Histologic Appearances. Adv. Anat. Pathol. 2019, 26, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Giorgi Rossi, P.; Ronco, G.; Dillner, J.; Elfström, K.M.; Snijders, P.J.F.; Arbyn, M.; Berkhof, J.; Carozzi, F.; Del Mistro, A.; De Sanjosè, S.; et al. Why follow-back studies should be interpreted cautiously: The case of an HPV-negative cervical lesion. Cancer Cytopathol. 2016, 124, 66–67. [Google Scholar] [CrossRef] [PubMed]
- Tjalma, W.A.; Depuydt, C.E. Cervical cancer screening: Which HPV test should be used--L1 or E6/E7? Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 170, 45–46. [Google Scholar] [CrossRef] [PubMed]
- Banister, C.E.; Liu, C.; Pirisi, L.; Creek, K.E.; Buckhaults, P.J. Identification and characterization of HPV-independent cervical cancers. Oncotarget 2017, 8, 13375–13386. [Google Scholar] [CrossRef] [PubMed]
- Katki, H.A.; Kinney, W.K.; Fetterman, B.; Lorey, T.; Poitras, N.E.; Cheung, L.; Demuth, F.; Schiffman, M.; Wacholder, S.; Castle, P.E. Cervical cancer risk for women undergoing concurrent testing for human papillomavirus and cervical cytology: A population-based study in routine clinical practice. Lancet Oncol. 2011, 12, 663–672, Corrected in Lancet Oncol. 2011, 12, 722. [Google Scholar] [CrossRef] [PubMed]
- Houghton, O.; Jamison, J.; Wilson, R.; Carson, J.; McCluggage, W.G. p16 Immunoreactivity in unusual types of cervical adenocarcinoma does not reflect human papillomavirus infection. Histopathology 2010, 57, 342–350. [Google Scholar] [CrossRef]
- Guimerà, N.; Lloveras, B.; Alemany, L.; Iljazovic, E.; Shin, H.-R.; Jung-Il, S.; José, F.X.B.; Jenkins, D.; Bosch, F.X.; Quint, W. Laser capture microdissection shows HPV11 as both a causal and a coincidental infection in cervical cancer specimens with multiple HPV types. Histopathology 2013, 63, 287–292. [Google Scholar] [CrossRef]
- Guimerà, N.; Lloveras, B.; Lindeman, J.; Alemany, L.; van de Sandt, M.; Alejo, M.; Hernandez-Suarez, G.; Bravo, I.G.; Molijn, A.; Jenkins, D.; et al. The occasional role of low-risk human papillomaviruses 6, 11, 42, 44, and 70 in anogenital carcinoma defined by laser capture microdissection/PCR methodology: Results from a global study. Am. J. Surg. Pathol. 2013, 37, 1299–1310. [Google Scholar] [CrossRef]
- Dong, L.; Li, T.; Li, L.; Wang, M.Z.; Wu, Z.; Cui, J.; Liu, B.; Zhang, X.; Qiao, Y.; Chen, W. Clustering patterns of type-type combination in multiple genotypes infections of human papillomavirus in cervical adenocarcinoma. J. Med. Virol. 2019, 91, 2001–2008. [Google Scholar] [CrossRef]
- An, H.J.; Kim, K.R.; Kim, I.S.; Kim, D.W.; Park, M.H.; Park, I.A.; Suh, K.S.; Seo, E.J.; Sung, S.H.; Sohn, J.H.; et al. Prevalence of human papillomavirus DNA in various histological subtypes of cervical adenocarcinoma: A population-based study. Mod. Pathol. 2005, 18, 528–534. [Google Scholar] [CrossRef] [PubMed]
- Mazur, M.T.; Hsueh, S.; Gersell, D.J. Metastases to the female genital tract. Analysis of 325 cases. Cancer 1984, 53, 1978–1984. [Google Scholar] [CrossRef] [PubMed]
- Park, K.J. Cervical adenocarcinoma: Integration of HPV status, pattern of invasion, morphology and molecular markers into classification. Histopathology 2020, 76, 112–127. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, A.; Park, K.J.; Djordjevic, B.; Howitt, B.; Nucci, M.R.; Oliva, E.; Stolnicu, S.; Xu, B.; Soslow, R.A.; Parra-Herran, C. International Endocervical Adenocarcinoma Criteria and Classification: Validation and Interobserver Reproducibility. Am. J. Surg. Pathol. 2019, 43, 75–83. [Google Scholar] [CrossRef]
- Ondič, O.; Němcová, J.; Alaghehbandan, R.; Černá, K.; Gomolčáková, B.; Kinkorová-Luňáčková, I.; Chytra, J.; Šidlová, H.; Hósová, M.; Bouda, J. The newly proposed International Endocervical Adenocarcinoma Criteria and Classification and its relevance to cervical cytology screening assessed in a prospective 2-year study of 118 cases. Cytopathology 2020, 31, 288–291. [Google Scholar] [CrossRef]
- Stolnicu, S.; Barsan, I.; Hoang, L.; Patel, P.; Terinte, C.; Pesci, A.; Aviel-Ronen, S.; Kiyokawa, T.; Alvarado-Cabrero, I.; Pike, M.C.; et al. International Endocervical Adenocarcinoma Criteria and Classification (IECC): A New Pathogenetic Classification for Invasive Adenocarcinomas of the Endocervix. Am. J. Surg. Pathol. 2018, 42, 214–226. [Google Scholar] [CrossRef]
- Stolnicu, S.; Park, K.J.; Kiyokawa, T.; Oliva, E.; McCluggage, W.G.; Soslow, R.A. Tumor Typing of Endocervical Adenocarcinoma: Contemporary Review and Recommendations From the International Society of Gynecological Pathologists. Int. J. Gynecol. Pathol. 2021, 40 (Suppl. S1), S75–S91. [Google Scholar] [CrossRef]
- Kurman, R.J.; Carcangiu, M.L.; Herrington, C.S.; Young, R.H. WHO Classification of Tumours of the Female Reproductive Organs, 4th ed.; IARC: Lyon, France, 2014. [Google Scholar]
- WHO Classification of Tumours Editorial Board. WHO Classification of Female Genital Tumours, 5th ed.; International Agency for Research on Cancer: Lyon, France, 2020; Volume 4. [Google Scholar]
- Park, K.J.; Kiyokawa, T.; Soslow, R.A.; Lamb, C.A.; Oliva, E.; Zivanovic, O.; Juretzka, M.M.; Pirog, E.C. Unusual endocervical adenocarcinomas: An immunohistochemical analysis with molecular detection of human papillomavirus. Am. J. Surg. Pathol. 2011, 35, 633–646. [Google Scholar] [CrossRef]
- Kusanagi, Y.; Kojima, A.; Mikami, Y.; Kiyokawa, T.; Sudo, T.; Yamaguchi, S.; Nishimura, R. Absence of high-risk human papillomavirus (HPV) detection in endocervical adenocarcinoma with gastric morphology and phenotype. Am. J. Pathol. 2010, 177, 2169–2175. [Google Scholar] [CrossRef]
- Mikami, Y.; Hata, S.; Melamed, J.; Fujiwara, K.; Manabe, T. Lobular endocervical glandular hyperplasia is a metaplastic process with a pyloric gland phenotype. Histopathology 2001, 39, 364–372. [Google Scholar] [CrossRef]
- McCluggage, W.G. New developments in endocervical glandular lesions. Histopathology 2013, 62, 138–160. [Google Scholar] [CrossRef] [PubMed]
- Selenica, P.; Alemar, B.; Matrai, C.; Talia, K.L.; Veras, E.; Hussein, Y.; Oliva, E.; Beets-Tan, R.G.H.; Mikami, Y.; McCluggage, W.G.; et al. Massively parallel sequencing analysis of 68 gastric-type cervical adenocarcinomas reveals mutations in cell cycle-related genes and potentially targetable mutations. Mod. Pathol. 2021, 34, 1213–1225. [Google Scholar] [CrossRef] [PubMed]
- Giardiello, F.M.; Brensinger, J.D.; Tersmette, A.C.; Goodman, S.N.; Petersen, G.M.; Booker, S.V.; Cruz–Correa, M.; Offerhaus, J.A. Very high risk of cancer in familial Peutz-Jeghers syndrome. Gastroenterology 2000, 119, 1447–1453. [Google Scholar] [CrossRef] [PubMed]
- Kido, A.; Mikami, Y.; Koyama, T.; Kataoka, M.; Shitano, F.; Konishi, I.; Togashi, K. Magnetic resonance appearance of gastric-type adenocarcinoma of the uterine cervix in comparison with that of usual-type endocervical adenocarcinoma: A pitfall of newly described unusual subtype of endocervical adenocarcinoma. Int. J. Gynecol. Cancer 2014, 24, 1474–1479. [Google Scholar] [CrossRef] [PubMed]
- Bonin, L.; Devouassoux-Shisheboran, M.; Golfier, F. Clinicopathological characteristics of patients with mucinous adenocarcinoma of the uterine cervix: A retrospective study of 21 cases. J. Gynecol. Obstet. Hum. Reprod. 2019, 48, 319–327. [Google Scholar] [CrossRef]
- Garg, S.; Nagaria, T.S.; Clarke, B.; Freedman, O.; Khan, Z.; Schwock, J.; Bernardini, M.Q.; Oza, A.; Han, K.; Smith, A.C.; et al. Molecular characterization of gastric-type endocervical adenocarcinoma using next-generation sequencing. Mod. Pathol. 2019, 32, 1823–1833. [Google Scholar] [CrossRef]
- Nishio, S.; Mikami, Y.; Tokunaga, H.; Yaegashi, N.; Satoh, T.; Saito, M.; Okamoto, A.; Kasamatsu, T.; Miyamoto, T.; Shiozawa, T.; et al. Analysis of gastric-type mucinous carcinoma of the uterine cervix—An aggressive tumor with a poor prognosis: A multi-institutional study. Gynecol. Oncol. 2019, 153, 13–19. [Google Scholar] [CrossRef]
- Park, C.M.; Koh, H.M.; Park, S.; Kang, H.S.; Shim, S.S.; Kim, S.Y. Gastric type mucinous endocervical adenocarcinoma of the uterine cervix: Very rare and interesting case. Obstet. Gynecol. Sci. 2018, 61, 165–169. [Google Scholar] [CrossRef]
- Nucci, M.R.; Clement, P.B.; Young, R.H. Lobular endocervical glandular hyperplasia, not otherwise specified: A clinicopathologic analysis of thirteen cases of a distinctive pseudoneoplastic lesion and comparison with fourteen cases of adenoma malignum. Am. J. Surg. Pathol. 1999, 23, 886–891. [Google Scholar] [CrossRef]
- Matsubara, A.; Sekine, S.; Ogawa, R.; Yoshida, M.; Kasamatsu, T.; Tsuda, H.; Kanai, Y. Lobular endocervical glandular hyperplasia is a neoplastic entity with frequent activating GNAS mutations. Am. J. Surg. Pathol. 2014, 38, 370–376. [Google Scholar] [CrossRef]
- Nishio, S.; Tsuda, H.; Fujiyoshi, N.; Ota, S.-I.; Ushijima, K.; Sasajima, Y.; Kasamatsu, T.; Kamura, T.; Matsubara, O. Clinicopathological significance of cervical adenocarcinoma associated with lobular endocervical glandular hyperplasia. Pathol. Res. Pract. 2009, 205, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, A.; Howitt, B.E.; Park, K.J.; Lindeman, N.; Nucci, M.R.; Parra-Herran, C. Genomic Characterization of HPV-related and Gastric-type Endocervical Adenocarcinoma: Correlation With Subtype and Clinical Behavior. Int. J. Gynecol. Pathol. 2020, 39, 578–586. [Google Scholar] [CrossRef] [PubMed]
- Talia, K.L.; Stewart, C.J.R.; Howitt, B.E.; Nucci, M.R.; McCluggage, W.G. HPV-negative Gastric Type Adenocarcinoma In Situ of the Cervix: A Spectrum of Rare Lesions Exhibiting Gastric and Intestinal Differentiation. Am. J. Surg. Pathol. 2017, 41, 1023–1033. [Google Scholar] [CrossRef] [PubMed]
- Kawauchi, S.; Kusuda, T.; Liu, X.P.; Suehiro, Y.; Kaku, T.; Mikami, Y.; Takeshita, M.; Nakao, M.; Chochi, Y.; Sasaki, K. Is lobular endocervical glandular hyperplasia a cancerous precursor of minimal deviation adenocarcinoma?: A comparative molecular-genetic and immunohistochemical study. Am. J. Surg. Pathol. 2008, 32, 1807–1815. [Google Scholar] [CrossRef]
- Karamurzin, Y.S.; Kiyokawa, T.; Parkash, V.; Jotwani, A.R.; Patel, P.; Pike, M.C.; Soslow, R.A.; Park, K.J. Gastric-type Endocervical Adenocarcinoma: An Aggressive Tumor With Unusual Metastatic Patterns and Poor Prognosis. Am. J. Surg. Pathol. 2015, 39, 1449–1457. [Google Scholar] [CrossRef]
- Jung, H.; Bae, G.E.; Kim, H.M.; Kim, H.S. Clinicopathological and Molecular Differences Between Gastric-type Mucinous Carcinoma and Usual-type Endocervical Adenocarcinoma of the Uterine Cervix. Cancer Genom. Proteom. 2020, 17, 627–641. [Google Scholar] [CrossRef]
- Kawakami, F.; Mikami, Y.; Sudo, T.; Fujiwara, K.; Hirose, T.; Itoh, T. Cytologic features of gastric-type adenocarcinoma of the uterine cervix. Diagn. Cytopathol. 2015, 43, 791–796. [Google Scholar] [CrossRef]
- Omori, M.; Hashi, A.; Ishii, Y.; Yuminamochi, T.; Nara, M.; Kondo, T.; Hirata, S.; Katoh, R.; Hoshi, K. Clinical impact of preoperative screening for gastric mucin secretion in cervical discharge by HIK1083-labeled latex agglutination test. Am. J. Clin. Pathol. 2008, 130, 585–594. [Google Scholar] [CrossRef][Green Version]
- Hanselaar, A.; van Loosbroek, M.; Schuurbiers, O.; Helmerhorst, T.; Bulten, J.; Bernhelm, J. Clear cell adenocarcinoma of the vagina and cervix. An update of the central Netherlands registry showing twin age incidence peaks. Cancer 1997, 79, 2229–2236. [Google Scholar] [CrossRef]
- Ahrens, W.A.; Barrón-Rodriguez, L.P.; McKee, M.; Rivkees, S.; Reyes-Múgica, M. Clear cell adenocarcinoma of the cervix in a child without in utero exposure to diethylstilbestrol: A case report and review of the literature. Pediatr. Dev. Pathol. 2005, 8, 690–695. [Google Scholar] [CrossRef]
- Liebrich, C.; Brummer, O.; Von Wasielewski, R.; Wegener, G.; Meijer, C.; Iftner, T.; Petry, K.U. Primary cervical cancer truly negative for high-risk human papillomavirus is a rare but distinct entity that can affect virgins and young adolescents. Eur. J. Gynaecol. Oncol. 2009, 30, 45–48. [Google Scholar] [PubMed]
- Robboy, S.J.; Kaufman, R.H.; Prat, J.; Welch, W.R.; Gaffey, T.; Scully, R.E.; Richart, R.; Fenoglio, C.M.; Virata, R.; Tilley, B.C. Pathologic findings in young women enrolled in the National Cooperative Diethylstilbestrol Adenosis (DESAD) project. Obstet. Gynecol. 1979, 53, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Huo, D.; Anderson, D.; Herbst, A.L. Follow-up of Patients with Clear-Cell Adenocarcinoma of the Vagina and Cervix. N. Engl. J. Med. 2018, 378, 1746–1748. [Google Scholar] [CrossRef] [PubMed]
- Limaiem, F.; Mahdy, H. Cervical Clear Cell Carcinoma. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Jiang, X.; Jin, Y.; Li, Y.; Huang, H.-F.; Wu, M.; Shen, K.; Pan, L.-Y. Clear cell carcinoma of the uterine cervix: Clinical characteristics and feasibility of fertility-preserving treatment. Onco Targets Ther. 2014, 7, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Tantitamit, T.; Hamontri, S.; Rangsiratanakul, L. Clear cell adenocarcinoma of the cervix in second generation young women who are without maternal exposure to diethylstilbestrol: A case report. Gynecol. Oncol. Rep. 2017, 20, 34–36. [Google Scholar] [CrossRef]
- Hiromura, T.; Tanaka, Y.O.; Nishioka, T.; Satoh, M.; Tomita, K. Clear cell adenocarcinoma of the uterine cervix arising from a background of cervical endometriosis. Br. J. Radiol. 2009, 82, e20–e22. [Google Scholar] [CrossRef]
- Talia, K.L.; Arora, R.; McCluggage, W.G. Precursor Lesions of Cervical Clear Cell Carcinoma: Evidence For Origin From Tubo-Endometrial Metaplasia. Int. J. Gynecol. Pathol. 2022, 41, 105–112. [Google Scholar] [CrossRef]
- Wang, D.; Zhao, C.; Fu, L.; Liu, Y.; Zhang, W.; Xu, T. Primary Clear Cell Adenocarcinoma of the Cervix: A Clinical Analysis of 18 Cases without Exposure to Diethylstilbestrol. Obstet. Gynecol. Int. 2019, 2019, 9465375. [Google Scholar] [CrossRef]
- Adani-Ifè, A.; Goldschmidt, E.; Innominato, P.; Ulusakarya, A.; Errihani, H.; Bertheau, P.; Morère, J.F. Very late recurrence of Diethylstilbestrol-related clear cell carcinoma of the cervix: Case report. Gynecol. Oncol. Res. Pract. 2015, 2, 3. [Google Scholar] [CrossRef][Green Version]
- Jones, W.B.; Koulos, J.P.; Saigo, P.E.; Lewis, J.L., Jr. Clear-cell adenocarcinoma of the lower genital tract: Memorial Hospital 1974–1984. Obstet. Gynecol. 1987, 70, 573–577. [Google Scholar]
- Reich, O.; Tamussino, K.; Lahousen, M.; Pickel, H.; Haas, J.; Winter, R. Clear cell carcinoma of the uterine cervix: Pathology and prognosis in surgically treated stage IB-IIB disease in women not exposed in utero to diethylstilbestrol. Gynecol. Oncol. 2000, 76, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Kenny, S.L.; McBride, H.A.; Jamison, J.; McCluggage, W.G. Mesonephric adenocarcinomas of the uterine cervix and corpus: HPV-negative neoplasms that are commonly PAX8, CA125, and HMGA2 positive and that may be immunoreactive with TTF1 and hepatocyte nuclear factor 1-β. Am. J. Surg. Pathol. 2012, 36, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Silver, S.A.; Devouassoux-Shisheboran, M.; Mezzetti, T.P.; Tavassoli, F.A. Mesonephric adenocarcinomas of the uterine cervix: A study of 11 cases with immunohistochemical findings. Am. J. Surg. Pathol. 2001, 25, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Devarashetty, S.; Chennapragada, S.S.; Mansour, R. Not Your Typical Adenocarcinoma: A Case of Mesonephric Adenocarcinoma of the Cervix With Fibroblast Growth Factor Receptor 2 (FGFR2) Mutation. Cureus 2022, 14, e25098. [Google Scholar] [CrossRef] [PubMed]
- Dierickx, A.; Göker, M.; Braems, G.; Tummers, P.; Van den Broecke, R. Mesonephric adenocarcinoma of the cervix: Case report and literature review. Gynecol. Oncol. Rep. 2016, 17, 7–11. [Google Scholar] [CrossRef]
- Pirog, E.C.; Kleter, B.; Olgac, S.; Bobkiewicz, P.; Lindeman, J.; Quint, W.G.; Richart, R.M.; Isacson, C. Prevalence of human papillomavirus DNA in different histological subtypes of cervical adenocarcinoma. Am. J. Pathol. 2000, 157, 1055–1062. [Google Scholar] [CrossRef]
- Quint, K.D.; de Koning, M.N.; Geraets, D.T.; Quint, W.G.; Pirog, E.C. Comprehensive analysis of Human Papillomavirus and Chlamydia trachomatis in in-situ and invasive cervical adenocarcinoma. Gynecol. Oncol. 2009, 114, 390–394. [Google Scholar] [CrossRef]
- Jones, M.W.; Onisko, A.; Dabbs, D.J.; Elishaev, E.; Chiosea, S.; Bhargava, R. Immunohistochemistry and HPV in situ hybridization in pathologic distinction between endocervical and endometrial adenocarcinoma: A comparative tissue microarray study of 76 tumors. Int. J. Gynecol. Cancer 2013, 23, 380–384. [Google Scholar] [CrossRef]
- Ansari-Lari, M.A.; Staebler, A.; Zaino, R.J.; Shah, K.V.; Ronnett, B.M. Distinction of endocervical and endometrial adenocarcinomas: Immunohistochemical p16 expression correlated with human papillomavirus (HPV) DNA detection. Am. J. Surg. Pathol. 2004, 28, 160–167. [Google Scholar] [CrossRef]
- Wang, S.S.; Sherman, M.E.; Silverberg, S.G.; Carreon, J.D.; Lacey, J.V., Jr.; Zaino, R.; Kurman, R.J.; Hildesheim, A. Pathological characteristics of cervical adenocarcinoma in a multi-center US-based study. Gynecol. Oncol. 2006, 103, 541–546. [Google Scholar] [CrossRef]
- Chang, S.H.; Maddox, W.A. Adenocarcinoma arising within cervical endometriosis and invading the adjacent vagina. Am. J. Obstet. Gynecol. 1971, 110, 1015–1017. [Google Scholar] [CrossRef] [PubMed]
- Uehara, T.; Yoshida, H.; Kondo, A.; Kato, T. A case of cervical adenocarcinoma arising from endometriosis in the absence of human papilloma virus infection. J. Obstet. Gynaecol. Res. 2020, 46, 536–541. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Jin, B.; Yan, H.; Zhu, T.; Ding, H.; Chen, X.; Guan, Y. Is there different prognosis between cervical endometrioid adenocarcinoma and ordinary cervical adenocarcinoma in a propensity score matching study based on the surveillance, epidemiology, and end results (SEER) database? Transl. Cancer Res. 2022, 11, 1652–1664. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Shi, J.; Zhang, X.; Kong, F.; Liu, L.; Dong, X.; Wang, K.; Shen, D. Comprehensive genomic profiling and prognostic analysis of cervical gastric-type mucinous adenocarcinoma. Virchows Arch. 2021, 479, 893–903. [Google Scholar] [CrossRef] [PubMed]
- Park, E.; Kim, S.W.; Kim, S.; Kim, H.S.; Lee, J.Y.; Kim, Y.T.; Cho, N.H. Genetic characteristics of gastric-type mucinous carcinoma of the uterine cervix. Mod. Pathol. 2021, 34, 637–646. [Google Scholar] [CrossRef]
- Vogelstein, B.; Lane, D.; Levine, A.J. Surfing the p53 network. Nature 2000, 408, 307–310. [Google Scholar] [CrossRef]
- Hirose, S.; Murakami, N.; Takahashi, K.; Kuno, I.; Takayanagi, D.; Asami, Y.; Matsuda, M.; Shimada, Y.; Yamano, S.; Sunami, K.; et al. Genomic alterations in STK11 can predict clinical outcomes in cervical cancer patients. Gynecol. Oncol. 2020, 156, 203–210. [Google Scholar] [CrossRef]
- Shi, H.; Shao, Y.; Lu, W.; Lu, B. An analysis of HER2 amplification in cervical adenocarcinoma: Correlation with clinical outcomes and the International Endocervical Adenocarcinoma Criteria and Classification. J. Pathol. Clin. Res. 2021, 7, 86–95. [Google Scholar] [CrossRef]
- Zeng, Q.; Chen, J.; Li, Y.; Werle, K.D.; Zhao, R.-X.; Quan, C.-S.; Wang, Y.-S.; Zhai, Y.-X.; Wang, J.-W.; Youssef, M.; et al. LKB1 inhibits HPV-associated cancer progression by targeting cellular metabolism. Oncogene 2017, 36, 1245–1255. [Google Scholar] [CrossRef]
- Kuragaki, C.; Enomoto, T.; Ueno, Y.; Sun, H.; Fujita, M.; Nakashima, R.; Ueda, Y.; Wada, H.; Murata, Y.; Toki, T.; et al. Mutations in the STK11 gene characterize minimal deviation adenocarcinoma of the uterine cervix. Lab. Investig. 2003, 83, 35–45. [Google Scholar] [CrossRef]
- McCluggage, W.G.; Harley, I.; Houghton, J.P.; Geyer, F.C.; MacKay, A.; Reis-Filho, J.S. Composite cervical adenocarcinoma composed of adenoma malignum and gastric type adenocarcinoma (dedifferentiated adenoma malignum) in a patient with Peutz Jeghers syndrome. J. Clin. Pathol. 2010, 63, 935–941. [Google Scholar] [CrossRef] [PubMed]
- Liao, X.; Xia, X.; Su, W.; Yan, H.; Ma, Y.; Xu, L.; Luo, H.; Liu, W.; Yin, D.; Zhang, W.-H.; et al. Association of recurrent APOBEC3B alterations with the prognosis of gastric-type cervical adenocarcinoma. Gynecol. Oncol. 2022, 165, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Boyd, J.; Takahashi, H.; Waggoner, S.E.; Jones, L.A.; Hajek, R.A.; Wharton, J.T.; Liu, F.-s.; Fujino, T.; Barrett, J.C.; McLachlan, J.A. Molecular genetic analysis of clear cell adenocarcinomas of the vagina and cervix associated and unassociated with diethylstilbestrol exposure in utero. Cancer 1996, 77, 507–513. [Google Scholar] [CrossRef]
- Lee, E.K.; Lindeman, N.I.; Matulonis, U.A.; Konstantinopoulos, P.A. POLE-mutated clear cell cervical cancer associated with in-utero diethylstilbestrol exposure. Gynecol. Oncol. Rep. 2019, 28, 15–17. [Google Scholar] [CrossRef] [PubMed]
- Mills, A.M.; Liou, S.; Kong, C.S.; Longacre, T.A. Are women with endocervical adenocarcinoma at risk for lynch syndrome? Evaluation of 101 cases including unusual subtypes and lower uterine segment tumors. Int. J. Gynecol. Pathol. 2012, 31, 463–469. [Google Scholar] [CrossRef]
- Nakamura, K.; Nakayama, K.; Minamoto, T.; Ishibashi, T.; Ohnishi, K.; Yamashita, H.; Ono, R.; Sasamori, H.; Razia, S.; Hossain, M.M.; et al. Lynch Syndrome-Related Clear Cell Carcinoma of the Cervix: A Case Report. Int. J. Mol. Sci. 2018, 19, 979. [Google Scholar] [CrossRef]
- Mirkovic, J.; Sholl, L.M.; Garcia, E.; Lindeman, N.I.; Macconaill, L.E.; Hirsch, M.S.; Cin, P.D.; Gorman, M.; Barletta, J.A.; Nucci, M.R.; et al. Targeted genomic profiling reveals recurrent KRAS mutations and gain of chromosome 1q in mesonephric carcinomas of the female genital tract. Mod. Pathol. 2015, 28, 1504–1514. [Google Scholar] [CrossRef]
- Mirkovic, J.; McFarland, M.; Garcia, E.; Sholl, L.M.; Lindeman, N.; MacConaill, L.; Dong, F.; Hirsch, M.; Nucci, M.R.; Quick, C.M.; et al. Targeted Genomic Profiling Reveals Recurrent KRAS Mutations in Mesonephric-like Adenocarcinomas of the Female Genital Tract. Am. J. Surg. Pathol. 2018, 42, 227–233. [Google Scholar] [CrossRef]
- Pandey, R.K.; Shukla, S.; Hadi, R.; Husain, N.; Islam, M.H.; Singhal, A.; Tripathi, S.K.; Garg, R. Kirsten rat sarcoma virus protein overexpression in adenocarcinoma lung: Association with clinicopathological and histomorphological features. J. Carcinog. 2020, 19, 9. [Google Scholar]
- Yang, W.; Lu, Y.P.; Yang, Y.Z.; Kang, J.R.; Jin, Y.D.; Wang, H.W. Expressions of programmed death (PD)-1 and PD-1 ligand (PD-L1) in cervical intraepithelial neoplasia and cervical squamous cell carcinomas are of prognostic value and associated with human papillomavirus status. J. Obstet. Gynaecol. Res. 2017, 43, 1602–1612. [Google Scholar] [CrossRef]
- Marincola, F.M.; Jaffee, E.M.; Hicklin, D.J.; Ferrone, S. Escape of human solid tumors from T-cell recognition: Molecular mechanisms and functional significance. Adv. Immunol. 2000, 74, 181–273. [Google Scholar] [PubMed]
- Iwai, Y.; Ishida, M.; Tanaka, Y.; Okazaki, T.; Honjo, T.; Minato, N. Involvement of PD-L1 on tumor cells in the escape from host immune system and tumor immunotherapy by PD-L1 blockade. Proc. Natl. Acad. Sci. USA 2002, 99, 12293–12297. [Google Scholar] [CrossRef]
- Davis, A.A.; Patel, V.G. The role of PD-L1 expression as a predictive biomarker: An analysis of all US Food and Drug Administration (FDA) approvals of immune checkpoint inhibitors. J. Immunother. Cancer 2019, 7, 278. [Google Scholar] [CrossRef] [PubMed]
- Song, F.; Jia, M.; Yu, S.; Cao, L.; Sun, P.L.; Gao, H. PD-L1 expression and immune stromal features in HPV-independent cervical adenocarcinoma. Histopathology 2021, 79, 861–871. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Lucas, E.; Zhang, X.; Liu, Q.; Zhuang, Y.; Lin, W.; Chen, H.; Zhou, F. Programmed death-ligand 1 expression in human papillomavirus-independent cervical adenocarcinoma and its prognostic significance. Histopathology 2022, 80, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Cibula, D.; Pötter, R.; Planchamp, F.; Avall-Lundqvist, E.; Fischerova, D.; Meder, C.H.; Köhler, C.; Landoni, F.; Lax, S.; Lindegaard, J.C.; et al. The European Society of Gynaecological Oncology/European Society for Radiotherapy and Oncology/European Society of Pathology Guidelines for the Management of Patients With Cervical Cancer. Int. J. Gynecol. Cancer 2018, 28, 641–655. [Google Scholar] [CrossRef]
- The NCCN Guidelines for Cervical Cancer, Version 1. 2022. Available online: https://www.nccn.org/login?ReturnURL=https://www.nccn.org/professionals/physician_gls/pdf/cervical.pdf (accessed on 26 October 2021).
- Teoh, D.; Musa, F.; Salani, R.; Huh, W.; Jimenez, E. Diagnosis and Management of Adenocarcinoma in Situ: A Society of Gynecologic Oncology Evidence-Based Review and Recommendations. Obstet. Gynecol. 2020, 135, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Ciavattini, A.; Giannella, L.; Delli Carpini, G.; Tsiroglou, D.; Sopracordevole, F.; Chiossi, G.; Di Giuseppe, J.; Italian Society of Colposcopy and Cervico-Vaginal Pathology (SICPCV). Adenocarcinoma in situ of the uterine cervix: Clinical practice guidelines from the Italian society of colposcopy and cervical pathology (SICPCV). Eur. J. Obstet. Gynecol. Reprod. Biol. 2019, 240, 273–277. [Google Scholar] [CrossRef]
- ACOG Practice Bulletin. Diagnosis and treatment of cervical carcinomas. Number 35, May 2002. American College of Obstetricians and Gynecologists. Int. J. Gynaecol. Obstet. 2002, 78, 79–91. [Google Scholar]
- Gaffney, D.K.; Erickson-Wittmann, B.A.; Jhingran, A.; Mayr, N.A.; Puthawala, A.A.; Moore, D.; Rao, G.G.; Small, W.; Varia, M.A.; Wolfson, A.H.; et al. ACR Appropriateness Criteria® on Advanced Cervical Cancer Expert Panel on Radiation Oncology-Gynecology. Int. J. Radiat. Oncol. Biol. Phys. 2011, 81, 609–614. [Google Scholar] [CrossRef]
- Monk, B.J.; Tewari, K.S.; Koh, W.J. Multimodality therapy for locally advanced cervical carcinoma: State of the art and future directions. J. Clin. Oncol. 2007, 25, 2952–2965. [Google Scholar] [CrossRef] [PubMed]
- Gien, L.T.; Beauchemin, M.C.; Thomas, G. Adenocarcinoma: A unique cervical cancer. Gynecol. Oncol. 2010, 116, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Baalbergen, A.; Veenstra, Y.; Stalpers, L.L.; Ansink, A.C. Primary surgery versus primary radiation therapy with or without chemotherapy for early adenocarcinoma of the uterine cervix. Cochrane Database Syst. Rev. 2010, 1, CD006248. [Google Scholar] [CrossRef]
- Park, J.Y.; Kim, D.Y.; Kim, J.H.; Kim, Y.M.; Kim, Y.T.; Nam, J.H. Outcomes after radical hysterectomy in patients with early-stage adenocarcinoma of uterine cervix. Br. J. Cancer 2010, 102, 1692–1698. [Google Scholar] [CrossRef]
- Ryu, S.Y.; Kim, M.H.; Nam, B.H.; Lee, T.S.; Song, E.S.; Park, C.Y.; Kim, J.W.; Kim, Y.B.; Ryu, H.S.; Park, S.Y.; et al. Intermediate-risk grouping of cervical cancer patients treated with radical hysterectomy: A Korean Gynecologic Oncology Group study. Br. J. Cancer 2014, 110, 278–285. [Google Scholar] [CrossRef]
- Noh, J.M.; Park, W.; Kim, Y.S.; Kim, J.-Y.; Kim, H.J.; Kim, J.; Kim, J.H.; Yoon, M.S.; Choi, J.H.; Yoon, W.S.; et al. Comparison of clinical outcomes of adenocarcinoma and adenosquamous carcinoma in uterine cervical cancer patients receiving surgical resection followed by radiotherapy: A multicenter retrospective study (KROG 13-10). Gynecol. Oncol. 2014, 132, 618–623. [Google Scholar] [CrossRef]
| HPVA | NHPVA | Ref. | |
|---|---|---|---|
| Adenocarcinoma Histology | HPV Positivity Rate (%) | ||
| Usual type | 72–100 | - | [7,13,20] |
| Mucinous | 83–100 | - | [7] |
| Gastric type | - | 0 | [20] |
| Clear cells type | - | 0–28 | [13,20,21] |
| Mesonephric type | - | 0 | [7] |
| Endometrioid type | - | 0–27 | [7,13,20] |
| WHO 2014 | IECC 2018/WHO 2020 | |
|---|---|---|
| HPV-Associated (HPVA) | Non-HPV-Associated (NHPVA) | |
| Usual type | Usual type | Gastric type |
| Mucinous carcinoma, NOS | Villoglandular | Clear cells |
| Gastric type | Mucinous, NOS | Mesonephric |
| Intestinal type | Mucinous, intestinal | Endometrioid |
| Signet ring cell | Invasive stratified mucin-producing | |
| Villoglandular | Micropapillary | |
| Endometrioid | Serous’-like | |
| Clear cells | ||
| Serous | ||
| Mesonephric | ||
| Genetic Mutation | Sample Size (Number of Patients) | Cases with Genetic Mutation (%) | Ref. |
|---|---|---|---|
| AKT1 | 11 | 33 | [54] |
| ARID1A | 14 | 29 | [48] |
| ARID1A | 15 | 20 | [87] |
| ATM | 11 | 18 | [54] |
| BRCA2 | 14 | 21 | [48] |
| BRCA2 | 21 | 10 | [88] |
| CDKN2A | 3 | 67 | [8] |
| CDKN2A | 14 | 36 | [48] |
| CDKN2A | 15 | 27 | [87] |
| CDKN2A | 68 | 18 | [44] |
| ELF | 11 | 18 | [54] |
| ERBB2 | 68 | 9 | [44] |
| ERBB3 | 21 | 10 | [88] |
| ERBB3 | 68 | 10 | [44] |
| FGFR4 | 21 | 14 | [88] |
| GNAS | 21 | 10 | [88] |
| GNAS | 68 | 9 | [44] |
| HLA-B | 21 | 19 | [88] |
| KMT2D | 11 | 18 | [54] |
| KRAS | 3 | 33 | [8] |
| KRAS | 11 | 36 | [54] |
| KRAS | 68 | 17 | [44] |
| MSH2 | 14 | 21 | [48] |
| MSH6 | 14 | 43 | [48] |
| NTRK3 | 11 | 18 | [54] |
| PIK3CA | 11 | 18 | [54] |
| PIK3CA | 68 | 7 | [44] |
| POLE | 14 | 36 | [48] |
| PTEN | 15 | 20 | [87] |
| PTPRS | 21 | 19 | [88] |
| SLX4 | 14 | 36 | [48] |
| SLX4 | 14 | 36 | [48] |
| SLX4 | 21 | 10 | [88] |
| SMAD4 | 68 | 9 | [44] |
| STK11 | 3 | 33 | [8] |
| STK11 | 14 | 29 | [48] |
| STK11 | 15 | 33 | [88] |
| STK11 | 19 | 21 | [88] |
| STK11 | 68 | 10 | [44] |
| TP53 | 3 | 67 | [8] |
| TP53 | 11 | 46 | [54] |
| TP53 | 14 | 50 | [48] |
| TP53 | 15 | 53 | [87] |
| TP53 | 21 | 52,4 | [88] |
| TP53 | 68 | 41 | [44] |
| CDK12 | 15 | 7 | [87] |
| ERBB2 | 15 | 13 | [87] |
| MDM2 | 14 | 14 | [48] |
| MECOM | 15 | 7 | [87] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giannella, L.; Di Giuseppe, J.; Delli Carpini, G.; Grelloni, C.; Fichera, M.; Sartini, G.; Caimmi, S.; Natalini, L.; Ciavattini, A. HPV-Negative Adenocarcinomas of the Uterine Cervix: From Molecular Characterization to Clinical Implications. Int. J. Mol. Sci. 2022, 23, 15022. https://doi.org/10.3390/ijms232315022
Giannella L, Di Giuseppe J, Delli Carpini G, Grelloni C, Fichera M, Sartini G, Caimmi S, Natalini L, Ciavattini A. HPV-Negative Adenocarcinomas of the Uterine Cervix: From Molecular Characterization to Clinical Implications. International Journal of Molecular Sciences. 2022; 23(23):15022. https://doi.org/10.3390/ijms232315022
Chicago/Turabian StyleGiannella, Luca, Jacopo Di Giuseppe, Giovanni Delli Carpini, Camilla Grelloni, Mariasole Fichera, Gianmarco Sartini, Serena Caimmi, Leonardo Natalini, and Andrea Ciavattini. 2022. "HPV-Negative Adenocarcinomas of the Uterine Cervix: From Molecular Characterization to Clinical Implications" International Journal of Molecular Sciences 23, no. 23: 15022. https://doi.org/10.3390/ijms232315022
APA StyleGiannella, L., Di Giuseppe, J., Delli Carpini, G., Grelloni, C., Fichera, M., Sartini, G., Caimmi, S., Natalini, L., & Ciavattini, A. (2022). HPV-Negative Adenocarcinomas of the Uterine Cervix: From Molecular Characterization to Clinical Implications. International Journal of Molecular Sciences, 23(23), 15022. https://doi.org/10.3390/ijms232315022








