Antidepressive Effect of Natural Products and Their Derivatives Targeting BDNF-TrkB in Gut–Brain Axis
Abstract
1. Introduction
2. Results
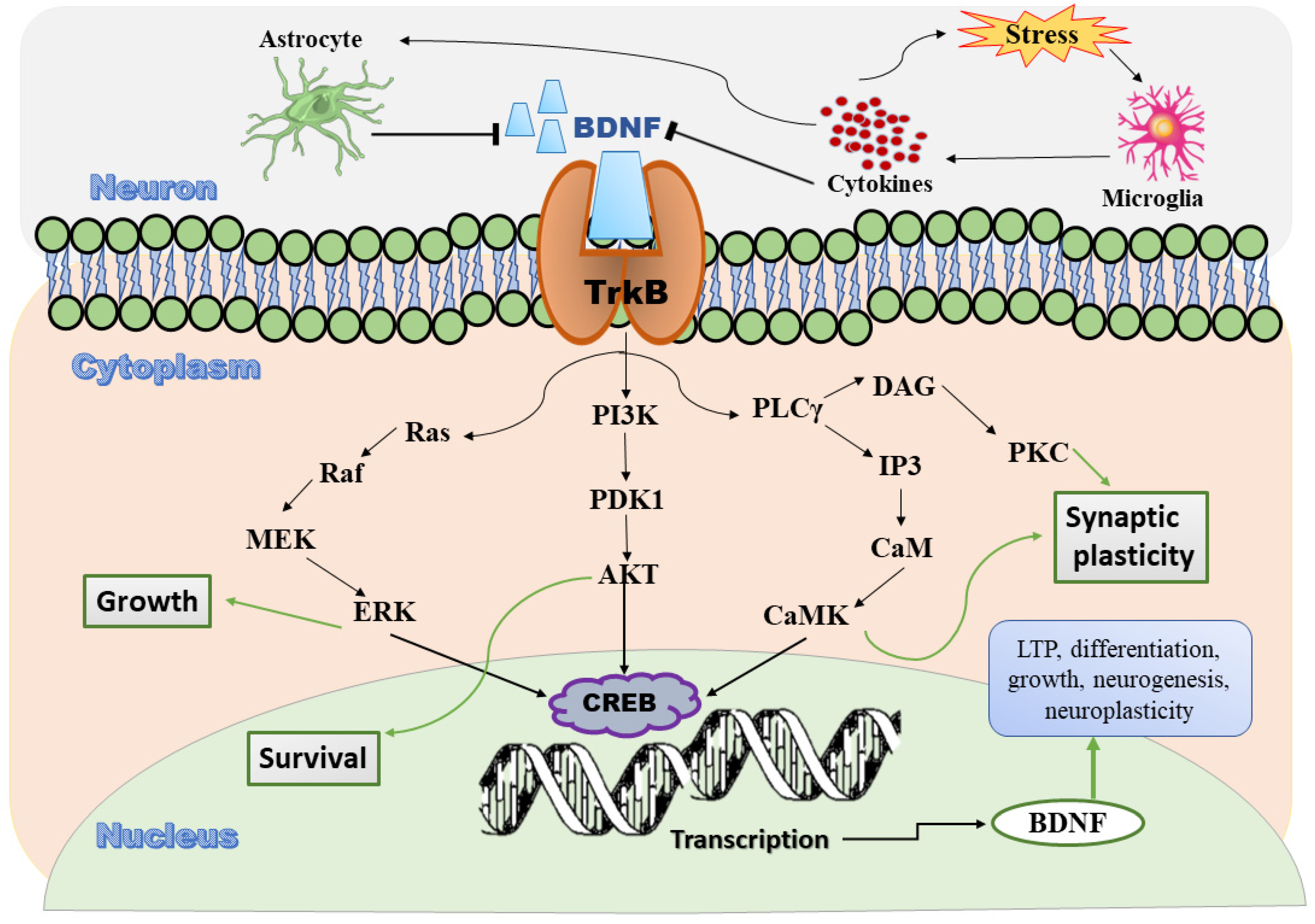
2.1. Brain-Derived Neurotrophic Factor
2.2. Signaling Pathway of BDNF
2.3. Role of Gut Microbiota in Depression
2.3.1. Gut Microbiota–Brain Axis in Depression
2.3.2. Role of BDNF in Irritable Bowel Syndrome Induced Depression
2.3.3. Findings Related to Gut Microbiota in Patients with Depression
2.4. Role of BDNF-mTORC1 Signaling Pathway in Depression
2.5. Effects of Antidepressants on BDNF and Their Related Side Effects
2.6. Modulation of BDNF Signaling by Different Natural Products and Their Derivatives
2.6.1. Flavonoids, Flavanones, and Flavanol
Apigenin
Baicalein
Hesperidin
Luteolin-7-O-glucuronide
Naringenin
Quercetin
Sanggenon G
Silibinin
Orange Juice
2.6.2. Phenol, Polyphenol, and Phenolic Acid Derivatives
Chlorogenic Acid
Curcumin
Ellagic Acid
Epigallocatechin-3-gallate
Eleutheroside E
Eugenol
Resveratrol
Thymol
2,3,5,4′-Tetrahydroxystilbene-2-O-β-d-glucoside
2.6.3. Fatty Acids, Lignan, Steroids, and Terpenoids
Ginsenoside Rd
Limonene
Omega-3 Fatty Acids
Oleanolic Acid
Poria cocos
Sesamol
2.6.4. Anthraquinone and Anthocyanin
Cyandin-3-glucoside and Cyanidin-3-sophoroside-5-glucoside
Hypericin
2.6.5. Carotenoids
Fucoxanthin
Lycopene
2.6.6. Bodily Fluids
Nicotinamide Riboside
Honey
2.6.7. Medicinal Plants and Herbal Formulations
Cinnamomum zeylanicum
Dendrobium officinale
Huanglian-Jie-Du-Tang
Cocos nucifera Husk Fiber
Semen Sojae Praeparatum
2.6.8. Phthalide
3-n-Butylphthalide
2.6.9. Probiotics
Clostridium butyricum (C. butyricum)
Porphyromonas gingivalis
Bifidobacterium longum NCC3001
Lactobacillus casei (L. casei)
Lactobacillus helveticus R0052
Lactobacillus plantarum
Lactobacillus rhamnosus zz-1
3. Discussion
4. Materials and Methods
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mayer, E.A.; Nance, K.; Chen, S. The Gut–Brain Axis. Annu. Rev. Med. 2022, 73, 439–453. [Google Scholar] [CrossRef]
- Morais, L.H.; Schreiber, H.L.; Mazmanian, S.K. The Gut Microbiota–Brain Axis in Behaviour and Brain Disorders. Nat. Rev. Microbiol. 2021, 19, 241–255. [Google Scholar] [CrossRef]
- Jiang, H.; Chen, S.; Li, C.; Lu, N.; Yue, Y.; Yin, Y.; Zhang, Y.; Zhi, X.; Zhang, D.; Yuan, Y. The Serum Protein Levels of the TPA–BDNF Pathway Are Implicated in Depression and Antidepressant Treatment. Transl. Psychiatry 2017, 7, e1079. [Google Scholar] [CrossRef] [PubMed]
- Mathers, C.D.; Loncar, D. Projections of Global Mortality and Burden of Disease from 2002 to 2030. PLoS Med. 2006, 3, e442. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, A.J.; Charlson, F.J.; Norman, R.E.; Patten, S.B.; Freedman, G.; Murray, C.J.L.; Vos, T.; Whiteford, H.A. Burden of Depressive Disorders by Country, Sex, Age, and Year: Findings from the Global Burden of Disease Study 2010. PLoS Med. 2013, 10, e1001547. [Google Scholar] [CrossRef] [PubMed]
- Bawari, S.; Tewari, D.; Argüelles, S.; Sah, A.N.; Nabavi, S.F.; Xu, S.; Vacca, R.A.; Nabavi, S.M.; Shirooie, S. Targeting BDNF Signaling by Natural Products: Novel Synaptic Repair Therapeutics for Neurodegeneration and Behavior Disorders. Pharmacol. Res. 2019, 148, 104458. [Google Scholar] [CrossRef]
- Lang, U.E.; Borgwardt, S. Molecular Mechanisms of Depression: Perspectives on New Treatment Strategies. Cell. Physiol. Biochem. 2013, 31, 761–777. [Google Scholar] [CrossRef]
- Evrensel, A.; Tarhan, K.N. Emerging Role of Gut-Microbiota-Brain Axis in Depression and Therapeutic Implication. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2021, 106, 110138. [Google Scholar] [CrossRef]
- Travaglia, A.; La Mendola, D. Chapter Three–Zinc Interactions with Brain-Derived Neurotrophic Factor and Related Peptide Fragments. Vitam. Horm. 2017, 104, 29–56. [Google Scholar]
- Miranda, M.; Morici, J.F.; Zanoni, M.B.; Bekinschtein, P. Brain-Derived Neurotrophic Factor: A Key Molecule for Memory in the Healthy and the Pathological Brain. Front. Cell. Neurosci. 2019, 13, 1–25. [Google Scholar] [CrossRef]
- Mitre, M.; Mariga, A.; Chao, M. V Neurotrophin Signalling: Novel Insights into Mechanisms and Pathophysiology. Clin. Sci. 2017, 131, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Amen, A.M.; Pham, D.L.; Meffert, M.K. Post-Transcriptional Regulation by Brain-Derived Neurotrophic Factor in the Nervous System. In Post-Transcriptional Mechanisms in Endocrine Regulation; Springer: Berlin/Heidelberg, Germany, 2016; pp. 315–337. [Google Scholar]
- Yang, B.; Ren, Q.; Zhang, J.C.; Chen, Q.X.; Hashimoto, K. Altered Expression of BDNF, BDNF pro-Peptide and Their Precursor ProBDNF in Brain and Liver Tissues from Psychiatric Disorders: Rethinking the Brain–Liver Axis. Transl. Psychiatry 2017, 7, e1128. [Google Scholar] [CrossRef] [PubMed]
- Duman, R.S.; Monteggia, L.M. A Neurotrophic Model for Stress-Related Mood Disorders. Biol. Psychiatry 2006, 59, 1116–1127. [Google Scholar] [CrossRef] [PubMed]
- Miyanishi, H.; Nitta, A. A Role of BDNF in the Depression Pathogenesis and a Potential Target as Antidepressant: The Modulator of Stress Sensitivity “Shati/Nat8l-BDNF System” in the Dorsal Striatum. Pharmaceuticals 2021, 14, 889. [Google Scholar] [CrossRef] [PubMed]
- Lu, B.; Nagappan, G.; Guan, X.; Nathan, P.J.; Wren, P. BDNF-Based Synaptic Repair as a Disease-Modifying Strategy for Neurodegenerative Diseases. Nat. Rev. Neurosci. 2013, 14, 401–416. [Google Scholar] [CrossRef] [PubMed]
- Mora, E.; Portella, M.J.; Piñol-Ripoll, G.; López, R.; Cuadras, D.; Forcada, I.; Teres, M.; Vieta, E.; Mur, M. High BDNF Serum Levels Are Associated to Good Cognitive Functioning in Bipolar Disorder. Eur. Psychiatry 2019, 60, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, K.A.; O’Neill, L.A.J.; Gearing, A.J.H.; Callard, R.E. The Cytokine Factsbook and Webfacts; Second, W.E., Ed.; Academic Press: London, UK, 2001; pp. 160–165. ISBN 978–0-12–155142–1. [Google Scholar]
- Binder, D.K.; Scharfman, H.E. Brain-Derived Neurotrophic Factor. Growth Factors 2004, 22, 123. [Google Scholar] [CrossRef]
- Duan, W.; Lee, J.; Guo, Z.; Mattson, M.P. Dietary Restriction Stimulates BDNF Production in the Brain and Thereby Protects Neurons against Excitotoxic Injury. J. Mol. Neurosci. 2001, 16, 1–12. [Google Scholar] [CrossRef]
- Gerhard, D.M.; Wohleb, E.S.; Duman, R.S. Emerging Treatment Mechanisms for Depression: Focus on Glutamate and Synaptic Plasticity. Drug Discov. Today 2016, 21, 454–464. [Google Scholar] [CrossRef]
- Karege, F.; Perret, G.; Bondolfi, G.; Schwald, M.; Bertschy, G.; Aubry, J.M. Decreased Serum Brain-Derived Neurotrophic Factor Levels in Major Depressed Patients. Psychiatry Res. 2002, 109, 143–148. [Google Scholar] [CrossRef]
- Meeker, R.B.; Williams, K.S. The P75 Neurotrophin Receptor: At the Crossroad of Neural Repair and Death. Neural Regen. Res. 2015, 10, 721. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Hong, E.J.; Cohen, S.; Zhao, W.; Ho, H.H.; Schmidt, L.; Chen, W.G.; Lin, Y.; Savner, E.; Griffith, E.C. Brain-Specific Phosphorylation of MeCP2 Regulates Activity-Dependent Bdnf Transcription, Dendritic Growth, and Spine Maturation. Neuron 2006, 52, 255–269. [Google Scholar] [CrossRef]
- Ghosh, A.; Carnahan, J.; Greenberg, M.E. Requirement for BDNF in Activity-Dependent Survival of Cortical Neurons. Science 1994, 263, 1618–1623. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Poo, M. Neurotrophin Regulation of Neural Circuit Development and Function. Nat. Rev. Neurosci. 2013, 14, 7–23. [Google Scholar] [CrossRef] [PubMed]
- Noebels, J.L.; Avoli, M.; Rogawski, M.A.; Olsen, R.W.; Delgado-Escueta, A.V. Jasper’s Basic Mechanisms of the Epilepsies [Internet]. 2012. Available online: https://www.ncbi.nlm.nih.gov/books/NBK50785/ (accessed on 24 November 2022).
- Harward, S.C.; Hedrick, N.G.; Hall, C.E.; Parra-Bueno, P.; Milner, T.A.; Pan, E.; Laviv, T.; Hempstead, B.L.; Yasuda, R.; McNamara, J.O. Autocrine BDNF–TrkB Signalling within a Single Dendritic Spine. Nature 2016, 538, 99–103. [Google Scholar] [CrossRef]
- Antal, A.; Chaieb, L.; Moliadze, V.; Monte-Silva, K.; Poreisz, C.; Thirugnanasambandam, N.; Nitsche, M.A.; Shoukier, M.; Ludwig, H.; Paulus, W. Brain-Derived Neurotrophic Factor (BDNF) Gene Polymorphisms Shape Cortical Plasticity in Humans. Brain Stimul. 2010, 3, 230–237. [Google Scholar] [CrossRef]
- McAllister, A.K.; Katz, L.C.; Lo, D.C. Neurotrophins and Synaptic Plasticity. Annu. Rev. Neurosci. 1999, 22, 295–318. [Google Scholar] [CrossRef]
- Marini, A.M.; Rabin, S.J.; Lipsky, R.H.; Mocchetti, I. Activity-Dependent Release of Brain-Derived Neurotrophic Factor Underlies the Neuroprotective Effect of N-Methyl-D-Aspartate. J. Biol. Chem. 1998, 273, 29394–29399. [Google Scholar] [CrossRef]
- Duman, R.S.; Aghajanian, G.K. Synaptic Dysfunction in Depression: Potential Therapeutic Targets. Science 2012, 338, 68–72. [Google Scholar] [CrossRef]
- Duman, R.S.; Voleti, B. Signaling Pathways Underlying the Pathophysiology and Treatment of Depression: Novel Mechanisms for Rapid-Acting Agents. Trends Neurosci. 2012, 35, 47–56. [Google Scholar] [CrossRef]
- Hoeffer, C.A.; Klann, E. MTOR Signaling: At the Crossroads of Plasticity, Memory and Disease. Trends Neurosci. 2010, 33, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Hazra, S.; Kumar, S.; Saha, G.K.; Mondal, A.C. Reversion of BDNF, Akt and CREB in Hippocampus of Chronic Unpredictable Stress Induced Rats: Effects of Phytochemical, Bacopa Monnieri. Psychiatry Investig. 2017, 14, 74. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, V.; Han, M.-H.; Mazei-Robison, M.; Iñiguez, S.D.; Ables, J.L.; Vialou, V.; Berton, O.; Ghose, S.; Covington III, H.E.; Wiley, M.D. AKT Signaling within the Ventral Tegmental Area Regulates Cellular and Behavioral Responses to Stressful Stimuli. Biol. Psychiatry 2008, 64, 691–700. [Google Scholar] [CrossRef]
- Murawska-Ciałowicz, E.; Wiatr, M.; Ciałowicz, M.; Gomes de Assis, G.; Borowicz, W.; Rocha-Rodrigues, S.; Paprocka-Borowicz, M.; Marques, A. BDNF Impact on Biological Markers of Depression-Role of Physical Exercise and Training. Int. J. Environ. Res. Public Health 2021, 18, 7553. [Google Scholar] [CrossRef] [PubMed]
- Macedo, D.; Chaves Filho, A.J.M.; de Sousa, C.N.S.; Quevedo, J.; Barichello, T.; Júnior, H.V.N.; de Lucena, D.F. Antidepressants, Antimicrobials or Both? Gut Microbiota Dysbiosis in Depression and Possible Implications of the Antimicrobial Effects of Antidepressant Drugs for Antidepressant Effectiveness. J. Affect. Disord. 2017, 208, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Simpson, C.A.; Diaz-Arteche, C.; Eliby, D.; Schwartz, O.S.; Simmons, J.G.; Cowan, C.S.M. The Gut Microbiota in Anxiety and Depression–A Systematic Review. Clin. Psychol. Rev. 2021, 83, 101943. [Google Scholar] [CrossRef]
- Chang, L.; Wei, Y.; Hashimoto, K. Brain–Gut–Microbiota Axis in Depression: A Historical Overview and Future Directions. Brain Res. Bull. 2022, 182, 44–56. [Google Scholar] [CrossRef]
- Mohajeri, M.H.; La Fata, G.; Steinert, R.E.; Weber, P. Relationship between the Gut Microbiome and Brain Function. Nutr. Rev. 2018, 76, 481–496. [Google Scholar] [CrossRef]
- Cryan, J.F.; O’Riordan, K.J.; Cowan, C.S.M.; Sandhu, K.V.; Bastiaanssen, T.F.S.; Boehme, M.; Codagnone, M.G.; Cussotto, S.; Fulling, C.; Golubeva, A.V.; et al. The Microbiota-Gut-Brain Axis. Physiol. Rev. 2019, 99, 1877–2013. [Google Scholar] [CrossRef]
- Luna, R.A.; Foster, J.A. Gut Brain Axis: Diet Microbiota Interactions and Implications for Modulation of Anxiety and Depression. Curr. Opin. Biotechnol. 2015, 32, 35–41. [Google Scholar] [CrossRef]
- Bercik, P.; Denou, E.; Collins, J.; Jackson, W.; Lu, J.; Jury, J.; Deng, Y.; Blennerhassett, P.; Macri, J.; McCoy, K.D. The Intestinal Microbiota Affect Central Levels of Brain-Derived Neurotropic Factor and Behavior in Mice. Gastroenterology 2011, 141, 599–609. [Google Scholar] [CrossRef] [PubMed]
- Ait-Belgnaoui, A.; Colom, A.; Braniste, V.; Ramalho, L.; Marrot, A.; Cartier, C.; Houdeau, E.; Theodorou, V.; Tompkins, T. Probiotic Gut Effect Prevents the Chronic Psychological Stress-induced Brain Activity Abnormality in Mice. Neurogastroenterol. Motil. 2014, 26, 510–520. [Google Scholar] [CrossRef] [PubMed]
- Sherwin, E.; Rea, K.; Dinan, T.G.; Cryan, J.F. A Gut (Microbiome) Feeling about the Brain. Curr. Opin. Gastroenterol. 2016, 32, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Ling, Z.; Zhang, Y.; Mao, H.; Ma, Z.; Yin, Y.; Wang, W.; Tang, W.; Tan, Z.; Shi, J. Altered Fecal Microbiota Composition in Patients with Major Depressive Disorder. Brain. Behav. Immun. 2015, 48, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Petra, A.I.; Panagiotidou, S.; Hatziagelaki, E.; Stewart, J.M.; Conti, P.; Theoharides, T.C. Gut-Microbiota-Brain Axis and Its Effect on Neuropsychiatric Disorders with Suspected Immune Dysregulation. Clin. Ther. 2015, 37, 984–995. [Google Scholar] [CrossRef] [PubMed]
- Desbonnet, L.; Garrett, L.; Clarke, G.; Kiely, B.; Cryan, J.F.; Dinan, T. Effects of the Probiotic Bifidobacterium Infantis in the Maternal Separation Model of Depression. Neuroscience 2010, 170, 1179–1188. [Google Scholar] [CrossRef]
- Bravo, J.A.; Forsythe, P.; Chew, M.V.; Escaravage, E.; Savignac, H.M.; Dinan, T.G.; Bienenstock, J.; Cryan, J.F. Ingestion of Lactobacillus Strain Regulates Emotional Behavior and Central GABA Receptor Expression in a Mouse via the Vagus Nerve. Proc. Natl. Acad. Sci. USA 2011, 108, 16050–16055. [Google Scholar] [CrossRef]
- Kelly, J.R.; Borre, Y.; O’Brien, C.; Patterson, E.; El Aidy, S.; Deane, J.; Kennedy, P.J.; Beers, S.; Scott, K.; Moloney, G. Transferring the Blues: Depression-Associated Gut Microbiota Induces Neurobehavioural Changes in the Rat. J. Psychiatr. Res. 2016, 82, 109–118. [Google Scholar] [CrossRef]
- Maes, M. Evidence for an Immune Response in Major Depression: A Review and Hypothesis. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 1995, 19, 11–38. [Google Scholar] [CrossRef]
- Maes, M.; Berk, M.; Goehler, L.; Song, C.; Anderson, G.; Gałecki, P.; Leonard, B. Depression and Sickness Behavior Are Janus-Faced Responses to Shared Inflammatory Pathways. BMC Med. 2012, 10, 1–19. [Google Scholar] [CrossRef]
- Kiecolt-Glaser, J.K.; Derry, H.M.; Fagundes, C.P. Inflammation: Depression Fans the Flames and Feasts on the Heat. Am. J. Psychiatry 2015, 172, 1075–1091. [Google Scholar] [CrossRef] [PubMed]
- Köhler, C.A.; Freitas, T.H.; Maes, M.; De Andrade, N.Q.; Liu, C.S.; Fernandes, B.S.; Stubbs, B.; Solmi, M.; Veronese, N.; Herrmann, N. Peripheral Cytokine and Chemokine Alterations in Depression: A Meta-analysis of 82 Studies. Acta Psychiatr. Scand. 2017, 135, 373–387. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C. Thinking from the Gut. Nature 2015, 518, S12–S14. [Google Scholar] [CrossRef] [PubMed]
- Simkin, D.R. Microbiome and Mental Health, Specifically as It Relates to Adolescents. Curr. Psychiatry Rep. 2019, 21, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Qin, G.; Liu, D.-R.; Wang, Y.; Yao, S.K. Increased Expression of Brain-Derived Neurotrophic Factor Is Correlated with Visceral Hypersensitivity in Patients with Diarrhea-Predominant Irritable Bowel Syndrome. World J. Gastroenterol. 2019, 25, 269. [Google Scholar] [CrossRef]
- Konturek, T.J.; Martinez, C.; Niesler, B.; van der Voort, I.; Mönnikes, H.; Stengel, A.; Goebel-Stengel, M. The Role of Brain-Derived Neurotrophic Factor in Irritable Bowel Syndrome. Front. Psychiatry 2021, 11, 531385. [Google Scholar] [CrossRef]
- Orlando, A.; Chimienti, G.; Notarnicola, M.; Russo, F. The Ketogenic Diet Improves Gut–Brain Axis in a Rat Model of Irritable Bowel Syndrome: Impact on 5-HT and BDNF Systems. Int. J. Mol. Sci. 2022, 23, 1098. [Google Scholar] [CrossRef]
- Naseribafrouei, A.; Hestad, K.; Avershina, E.; Sekelja, M.; Linløkken, A.; Wilson, R.; Rudi, K. Correlation between the Human Fecal Microbiota and Depression. Neurogastroenterol. Motil. 2014, 26, 1155–1162. [Google Scholar] [CrossRef]
- Park, A.J.; Collins, J.; Blennerhassett, P.A.; Ghia, J.E.; Verdu, E.F.; Bercik, P.; Collins, S. Altered Colonic Function and Microbiota Profile in a Mouse Model of Chronic Depression. Neurogastroenterol. Motil. 2013, 25, 857. [Google Scholar] [CrossRef]
- Tillmann, S.; Abildgaard, A.; Winther, G.; Wegener, G. Altered Fecal Microbiota Composition in the Flinders Sensitive Line Rat Model of Depression. Psychopharmacology 2019, 236, 1445–1457. [Google Scholar] [CrossRef]
- Zheng, P.; Zeng, B.; Zhou, C.; Liu, M.; Fang, Z.; Xu, X.; Zeng, L.; Chen, J.; Fan, S.; Du, X. Gut Microbiome Remodeling Induces Depressive-like Behaviors through a Pathway Mediated by the Host’s Metabolism. Mol. Psychiatry 2016, 21, 786–796. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zheng, P.; Li, Y.; Wu, J.; Tan, X.; Zhou, J.; Sun, Z.; Chen, X.; Zhang, G.; Zhang, H. Landscapes of Bacterial and Metabolic Signatures and Their Interaction in Major Depressive Disorders. Sci. Adv. 2020, 6, eaba8555. [Google Scholar] [CrossRef] [PubMed]
- Stevens, B.R.; Roesch, L.; Thiago, P.; Russell, J.T.; Pepine, C.J.; Holbert, R.C.; Raizada, M.K.; Triplett, E.W. Depression Phenotype Identified by Using Single Nucleotide Exact Amplicon Sequence Variants of the Human Gut Microbiome. Mol. Psychiatry 2021, 26, 4277–4287. [Google Scholar] [CrossRef] [PubMed]
- Zheng, P.; Yang, J.; Li, Y.; Wu, J.; Liang, W.; Yin, B.; Tan, X.; Huang, Y.; Chai, T.; Zhang, H. Gut Microbial Signatures Can Discriminate Unipolar from Bipolar Depression. Adv. Sci. 2020, 7, 1902862. [Google Scholar] [CrossRef]
- Sanada, K.; Nakajima, S.; Kurokawa, S.; Barceló-Soler, A.; Ikuse, D.; Hirata, A.; Yoshizawa, A.; Tomizawa, Y.; Salas-Valero, M.; Noda, Y. Gut Microbiota and Major Depressive Disorder: A Systematic Review and Meta-Analysis. J. Affect. Disord. 2020, 266, 1–13. [Google Scholar] [CrossRef]
- Nikolova, V.L.; Hall, M.R.B.; Hall, L.J.; Cleare, A.J.; Stone, J.M.; Young, A.H. Perturbations in Gut Microbiota Composition in Psychiatric Disorders: A Review and Meta-Analysis. JAMA Psychiatry 2021, 78, 1343–1354. [Google Scholar] [CrossRef]
- An, X.; Yao, X.; Li, B.; Yang, W.; Cui, R.; Zhao, G.; Jin, Y. Role of BDNF-MTORC1 Signaling Pathway in Female Depression. Neural Plast. 2021, 2021, 6619515. [Google Scholar] [CrossRef]
- Widman, A.J.; McMahon, L.L. Disinhibition of CA1 Pyramidal Cells by Low-Dose Ketamine and Other Antagonists with Rapid Antidepressant Efficacy. Proc. Natl. Acad. Sci. USA 2018, 115, E3007–E3016. [Google Scholar] [CrossRef]
- Wohleb, S.E.; Gerhard, D.; Thomas, A.; Duman, S.R. Molecular and Cellular Mechanisms of Rapid-Acting Antidepressants Ketamine and Scopolamine. Curr. Neuropharmacol. 2017, 15, 11–20. [Google Scholar] [CrossRef]
- Sun, L.; Zhang, L.; Liu, D.; Xue, R.; Liu, Y.; Li, L.; Guo, Y.; Shang, C.; Yao, J.; Zhang, Y. The Faster-Onset Antidepressant Effects of Hypidone Hydrochloride (YL-0919). Metab. Brain Dis. 2019, 34, 1375–1384. [Google Scholar] [CrossRef]
- Yin, Y.Y.; Tian, C.Y.; Fang, X.X.; Shang, C.; Zhang, L.M.; Xu, Q.; Li, Y.F. The Faster-Onset Antidepressant Effects of Hypidone Hydrochloride (YL-0919) in Monkeys Subjected to Chronic Unpredictable Stress. Front. Pharmacol. 2020, 1816, 586879. [Google Scholar] [CrossRef]
- Ran, Y.; Jin, Z.; Chen, X.; Zhao, N.; Fang, X.; Zhang, L.; Zhang, Y.; Li, Y. Hypidone Hydrochloride (YL-0919) Produces a Fast-Onset Reversal of the Behavioral and Synaptic Deficits Caused by Chronic Stress Exposure. Front. Cell. Neurosci. 2018, 395, 395. [Google Scholar] [CrossRef] [PubMed]
- Cavalleri, L.; Merlo Pich, E.; Millan, M.J.; Chiamulera, C.; Kunath, T.; Spano, P.F.; Collo, G. Ketamine Enhances Structural Plasticity in Mouse Mesencephalic and Human IPSC-Derived Dopaminergic Neurons via AMPAR-Driven BDNF and MTOR Signaling. Mol. Psychiatry 2018, 23, 812–823. [Google Scholar] [CrossRef] [PubMed]
- Fukumoto, K.; Fogaça, M.V.; Liu, R.J.; Duman, C.; Kato, T.; Li, X.Y.; Duman, R.S. Activity-Dependent Brain-Derived Neurotrophic Factor Signaling Is Required for the Antidepressant Actions of (2R, 6R)-Hydroxynorketamine. Proc. Natl. Acad. Sci. USA 2019, 116, 297–302. [Google Scholar] [CrossRef]
- Sharma, P.; Sharma, S.; Singh, D. Apigenin Reverses Behavioural Impairments and Cognitive Decline in Kindled Mice via CREB-BDNF Upregulation in the Hippocampus. Nutr. Neurosci. 2020, 23, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wei, N.; Li, X. Preclinical Evidence and Possible Mechanisms of Baicalein for Rats and Mice with Parkinson’s Disease: A Systematic Review and Meta-Analysis. Front. Aging Neurosci. 2020, 12, 277. [Google Scholar] [CrossRef]
- Cleary, C.; Linde, J.A.S.; Hiscock, K.M.; Hadas, I.; Belmaker, R.H.; Agam, G.; Flaisher-Grinberg, S.; Einat, H. Antidepressive-like Effects of Rapamycin in Animal Models: Implications for MTOR Inhibition as a New Target for Treatment of Affective Disorders. Brain Res. Bull. 2008, 76, 469–473. [Google Scholar] [CrossRef]
- Sengupta, S.; Giaime, E.; Narayan, S.; Hahm, S.; Howell, J.; O’Neill, D.; Vlasuk, G.P.; Saiah, E. Discovery of NV-5138, the First Selective Brain MTORC1 Activator. Sci. Rep. 2019, 9, 4107. [Google Scholar] [CrossRef]
- Duman, R.S.; Aghajanian, G.K.; Sanacora, G.; Krystal, J.H. Synaptic Plasticity and Depression: New Insights from Stress and Rapid-Acting Antidepressants. Nat. Med. 2016, 22, 238–249. [Google Scholar] [CrossRef]
- Krystal, J.H.; Sanacora, G.; Duman, R.S. Rapid-Acting Glutamatergic Antidepressants: The Path to Ketamine and Beyond. Biol. Psychiatry 2013, 73, 1133–1141. [Google Scholar] [CrossRef]
- Mattson, M.P.; Maudsley, S.; Martin, B. BDNF and 5-HT: A Dynamic Duo in Age-Related Neuronal Plasticity and Neurodegenerative Disorders. Trends Neurosci. 2004, 27, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Homberg, J.R.; Molteni, R.; Calabrese, F.; Riva, M.A. The Serotonin–BDNF Duo: Developmental Implications for the Vulnerability to Psychopathology. Neurosci. Biobehav. Rev. 2014, 43, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Casarotto, P.C.; Girych, M.; Fred, S.M.; Kovaleva, V.; Moliner, R.; Enkavi, G.; Biojone, C.; Cannarozzo, C.; Sahu, M.P.; Kaurinkoski, K. Antidepressant Drugs Act by Directly Binding to TRKB Neurotrophin Receptors. Cell 2021, 184, 1299–1313. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Wang, J.L.; Liu, R.; Li, X.X.; Li, J.F.; Zhang, L. Neuroprotective, Anti-Amyloidogenic and Neurotrophic Effects of Apigenin in an Alzheimer’s Disease Mouse Model. Molecules 2013, 18, 9949–9965. [Google Scholar] [CrossRef] [PubMed]
- Weng, L.; Guo, X.; Li, Y.; Yang, X.; Han, Y. Apigenin Reverses Depression-like Behavior Induced by Chronic Corticosterone Treatment in Mice. Eur. J. Pharmacol. 2016, 774, 50–54. [Google Scholar] [CrossRef]
- Zhao, X.; Kong, D.; Zhou, Q.; Wei, G.; Song, J.; Liang, Y.; Du, G. Baicalein Alleviates Depression-like Behavior in Rotenone-Induced Parkinson’s Disease Model in Mice through Activating the BDNF/TrkB/CREB Pathway. Biomed. Pharmacother. 2021, 140, 111556. [Google Scholar] [CrossRef]
- Liu, H.T.; Lin, Y.N.; Tsai, M.C.; Wu, Y.C.; Lee, M.C. Baicalein Exerts Therapeutic Effects against Endotoxin-Induced Depression-like Behavior in Mice by Decreasing Inflammatory Cytokines and Increasing Brain-Derived Neurotrophic Factor Levels. Antioxidants 2022, 11, 947. [Google Scholar] [CrossRef]
- Kosari-Nasab, M.; Shokouhi, G.; Ghorbanihaghjo, A.; Abbasi, M.M.; Salari, A.A. Hesperidin Attenuates Depression-Related Symptoms in Mice with Mild Traumatic Brain Injury. Life Sci. 2018, 213, 198–205. [Google Scholar] [CrossRef]
- Fu, H.; Liu, L.; Tong, Y.; Li, Y.; Zhang, X.; Gao, X.; Yong, J.; Zhao, J.; Xiao, D.; Wen, K.; et al. The Antidepressant Effects of Hesperidin on Chronic Unpredictable Mild Stress-Induced Mice. Eur. J. Pharmacol. 2019, 853, 236–246. [Google Scholar] [CrossRef]
- Xie, L.; Gu, Z.; Liu, H.; Jia, B.; Wang, Y.; Cao, M.; Song, R.; Zhang, Z.; Bian, Y. The Anti-Depressive Effects of Hesperidin and the Relative Mechanisms Based on the NLRP3 Inflammatory Signaling Pathway. Front. Pharmacol. 2020, 11, 1251. [Google Scholar] [CrossRef]
- Ryu, D.; Jee, H.J.; Kim, S.Y.; Hwang, S.H.; Pil, G.B.; Jung, Y.S. Luteolin-7-O-Glucuronide Improves Depression-like and Stress Coping Behaviors in Sleep Deprivation Stress Model by Activation of the BDNF Signaling. Nutr. 2022, 3314. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.A.; Subhan, N.; Rahman, M.M.; Uddin, S.J.; Reza, H.M.; Sarker, S.D. Effect of Citrus Flavonoids, Naringin and Naringenin, on Metabolic Syndrome and Their Mechanisms of Action. Adv. Nutr. 2014, 5, 404–417. [Google Scholar] [CrossRef] [PubMed]
- Yi, L.T.; Liu, B.B.; Li, J.; Luo, L.; Liu, Q.; Geng, D.; Tang, Y.; Xia, Y.; Wu, D. BDNF Signaling Is Necessary for the Antidepressant-like Effect of Naringenin. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2014, 48, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Olugbemide, A.S.; Ben-Azu, B.; Bakre, A.G.; Ajayi, A.M.; Femi-Akinlosotu, O.; Umukoro, S. Naringenin Improves Depressive- and Anxiety-like Behaviors in Mice Exposed to Repeated Hypoxic Stress through Modulation of Oxido-Inflammatory Mediators and NF-KB/BDNF Expressions. Brain Res. Bull. 2021, 169, 214–227. [Google Scholar] [CrossRef]
- Fang, K.; Li, H.R.; Chen, X.X.; Gao, X.R.; Huang, L.L.; Du, A.Q.; Jiang, C.; Li, H.; Ge, J.F. Quercetin Alleviates LPS-Induced Depression-Like Behavior in Rats via Regulating BDNF-Related Imbalance of Copine 6 and TREM1/2 in the Hippocampus and PFC. Front. Pharmacol. 2020, 10, 1544. [Google Scholar] [CrossRef]
- Ma, Z.X.; Zhang, R.Y.; Rui, W.J.; Wang, Z.Q.; Feng, X. Quercetin Alleviates Chronic Unpredictable Mild Stress-Induced Depressive-like Behaviors by Promoting Adult Hippocampal Neurogenesis via FoxG1/CREB/ BDNF Signaling Pathway. Behav. Brain Res. 2021, 406, 113245. [Google Scholar] [CrossRef]
- Shi, T.; Bian, X.; Yao, Z.; Wang, Y.; Gao, W.; Guo, C. Quercetin Improves Gut Dysbiosis in Antibiotic-Treated Mice. Food Funct. 2020, 11, 8003–8013. [Google Scholar] [CrossRef]
- Ye, M.; Ke, Y.; Liu, B.; Yuan, Y.; Wang, F.; Bu, S.; Zhang, Y. Root Bark of Morus Alba Ameliorates the Depressive-like Behaviors in Diabetic Rats. Neurosci. Lett. 2017, 637, 136–141. [Google Scholar] [CrossRef]
- Song, X.; Zhou, B.; Zhang, P.; Lei, D.; Wang, Y.; Yao, G.; Hayashi, T.; Xia, M.; Tashiro, S.; Onodera, S. Protective Effect of Silibinin on Learning and Memory Impairment in LPS-Treated Rats via ROS–BDNF–TrkB Pathway. Neurochem. Res. 2016, 41, 1662–1672. [Google Scholar] [CrossRef]
- Li, Y.J.; Li, Y.J.; Yang, L.D.; Zhang, K.; Zheng, K.Y.; Wei, X.M.; Yang, Q.; Niu, W.M.; Zhao, M.G.; Wu, Y.M. Silibinin Exerts Antidepressant Effects by Improving Neurogenesis through BDNF/TrkB Pathway. Behav. Brain Res. 2018, 348, 184–191. [Google Scholar] [CrossRef]
- Song, X.; Liu, B.; Cui, L.; Zhou, B.; Liu, W.; Xu, F.; Hayashi, T.; Hattori, S.; Ushiki-Kaku, Y.; Tashiro, S. Silibinin Ameliorates Anxiety/Depression-like Behaviors in Amyloid β-Treated Rats by Upregulating BDNF/TrkB Pathway and Attenuating Autophagy in Hippocampus. Physiol. Behav. 2017, 179, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Liaqat, H.; Parveen, A.; Kim, S.Y. Neuroprotective Natural Products’ Regulatory Effects on Depression via Gut–Brain Axis Targeting Tryptophan. Nutrients 2022, 14, 3270. [Google Scholar] [CrossRef] [PubMed]
- Pontifex, M.G.; Malik, M.M.A.H.; Connell, E.; Müller, M.; Vauzour, D. Citrus Polyphenols in Brain Health and Disease: Current Perspectives. Front. Neurosci. 2021, 15, 640648. [Google Scholar] [CrossRef]
- Park, M.; Choi, J.; Lee, H.J. Flavonoid-Rich Orange Juice Intake and Altered Gut Microbiome in Young Adults with Depressive Symptom: A Randomized Controlled Study. Nutrients 2020, 12, 1815. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, S.; Fujita, Y.; Oikawa, H.; Takekoshi, H.; Soya, H.; Ogata, M.; Fujikawa, T. Combination of Syringaresinol–Di–O–β-d-Glucoside and Chlorogenic Acid Shows Behavioral Pharmacological Anxiolytic Activity and Activation of Hippocampal BDNF–TrkB Signaling. Sci. Rep. 2020, 10, 18177. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.Q.; Tian, D.; Hu, C.Y.; Meng, Y.H. Chlorogenic Acid Ameliorates High-Fat and High-Fructose Diet-Induced Cognitive Impairment via Mediating the Microbiota–Gut–Brain Axis. J. Agric. Food Chem. 2022, 70, 2600–2615. [Google Scholar] [CrossRef]
- Afzal, A.; Batool, Z.; Sadir, S.; Liaquat, L.; Shahzad, S.; Tabassum, S.; Ahmad, S.; Kamil, N.; Perveen, T.; Haider, S. Therapeutic Potential of Curcumin in Reversing the Depression and Associated Pseudodementia via Modulating Stress Hormone, Hippocampal Neurotransmitters, and BDNF Levels in Rats. Neurochem. Res. 2021, 46, 3273–3285. [Google Scholar] [CrossRef]
- Liu, D.; Wang, Z.; Gao, Z.; Xie, K.; Zhang, Q.; Jiang, H.; Pang, Q. Effects of Curcumin on Learning and Memory Deficits, BDNF, and ERK Protein Expression in Rats Exposed to Chronic Unpredictable Stress. Behav. Brain Res. 2014, 271, 116–121. [Google Scholar] [CrossRef]
- Hurley, L.L.; Akinfiresoye, L.; Nwulia, E.; Kamiya, A.; Kulkarni, A.A.; Tizabi, Y. Antidepressant-like Effects of Curcumin in WKY Rat Model of Depression Is Associated with an Increase in Hippocampal BDNF. Behav. Brain Res. 2013, 239, 27–30. [Google Scholar] [CrossRef]
- Farbood, Y.; Rashno, M.; Ghaderi, S.; Khoshnam, S.E.; Sarkaki, A.; Rashidi, K.; Rashno, M.; Badavi, M. Ellagic Acid Protects against Diabetes-Associated Behavioral Deficits in Rats: Possible Involved Mechanisms. Life Sci. 2019, 225, 8–19. [Google Scholar] [CrossRef]
- Bedel, H.A.; Kencebay Manas, C.; Özbey, G.; Usta, C. The Antidepressant-like Activity of Ellagic Acid and Its Effect on Hippocampal Brain Derived Neurotrophic Factor Levels in Mouse Depression Models. Nat. Prod. Res. 2018, 32, 2932–2935. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Li, W.; You, B.; Tang, W.; Gan, T.; Feng, C.; Li, C.; Yang, R. Serum Metabonomic Study on the Antidepressant-like Effects of Ellagic Acid in a Chronic Unpredictable Mild Stress-Induced Mouse Model. J. Agric. Food Chem. 2020, 68, 9546–9556. [Google Scholar] [CrossRef] [PubMed]
- Stringer, T.P.; Guerrieri, D.; Vivar, C.; van Praag, H. Plant-Derived Flavanol (-)Epicatechin Mitigates Anxiety in Association with Elevated Hippocampal Monoamine and BDNF Levels, but Does Not Influence Pattern Separation in Mice. Transl. Psychiatry 2015, 5, e493. [Google Scholar] [CrossRef] [PubMed]
- Ko, S.; Jang, W.S.; Jeong, J.H.; Ahn, J.W.; Kim, Y.H.; Kim, S.; Chae, H.K.; Chung, S. (−)-Gallocatechin Gallate from Green Tea Rescues Cognitive Impairment through Restoring Hippocampal Silent Synapses in Post-Menopausal Depression. Sci. Rep. 2021, 11, 910. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, J.; Xiong, Y.; Zhang, X.; Lin, Y.; Liu, Z. Jasmine Tea Attenuates Chronic Unpredictable Mild Stress-Induced Depressive-like Behavior in Rats via the Gut-Brain Axis. Nutrients 2022, 14, 99. [Google Scholar] [CrossRef]
- Miyazaki, S.; Oikawa, H.; Takekoshi, H.; Hoshizaki, M.; Ogata, M.; Fujikawa, T. Anxiolytic Effects of Acanthopanax Senticosus HARMS Occur via Regulation of Autonomic Function and Activate Hippocampal BDNF–TrkB Signaling. Molecules 2018, 24, 132. [Google Scholar] [CrossRef]
- Song, C.; Duan, F.; Ju, T.; Qin, Y.; Zeng, D.; Shan, S.; Shi, Y.; Zhang, Y.; Lu, W. Eleutheroside E Supplementation Prevents Radiation-Induced Cognitive Impairment and Activates PKA Signaling via Gut Microbiota. Commun. Biol. 2022, 5, 1–14. [Google Scholar] [CrossRef]
- Tian, Y.; Deng, F. Phytochemistry and Biological Activity of Mustard (Brassica Juncea): A Review. CyTA-J. Food 2020, 18, 704–718. [Google Scholar] [CrossRef]
- Hwang, K.A.; Hwang, H.J.; Hwang, Y.J.; Kim, Y.J. Mustard Leaf Extract Suppresses Psychological Stress in Chronic Restraint Stress-Subjected Mice by Regulation of Stress Hormone, Neurotransmitters, and Apoptosis. Nutrients 2020, 12, 3640. [Google Scholar] [CrossRef]
- Li, M.; Zhao, Y.; Wang, Y.; Geng, R.; Fang, J.; Kang, S.; Huang, K.; Tong, T. Eugenol, A Major Component of Clove Oil, Attenuates Adiposity and Modulates Gut Microbiota in High-Fat Diet-fed Mice. Mol. Nutr. Food Res. 2022, 66, 2200387. [Google Scholar] [CrossRef]
- Yang, X.H.; Song, S.Q.; Xu, Y. Resveratrol Ameliorates Chronic Unpredictable Mild Stress-Induced Depression-like Behavior: Involvement of the HPA Axis, Inflammatory Markers, BDNF, and Wnt/β-Catenin Pathway in Rats. Neuropsychiatr. Dis. Treat. 2017, 13, 2727–2736. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.C.; Li, J.; Zhang, M.; Pan, J.C.; Yu, Y.; Zhang, J.B.; Zheng, L.; Si, J.M.; Xu, Y. Resveratrol Improves Brain-Gut Axis by Regulation of 5-HT-Dependent Signaling in the Rat Model of Irritable Bowel Syndrome. Front. Cell. Neurosci. 2019, 13, 30. [Google Scholar] [CrossRef] [PubMed]
- Ogaly, H.A.; Abdel-Rahman, R.F.; Mohamed, M.A.E.; Ahmed-Farid, O.A.; Khattab, M.S.; Abd-Elsalam, R.M. Thymol Ameliorated Neurotoxicity and Cognitive Deterioration in a Thioacetamide-Induced Hepatic Encephalopathy Rat Model; Involvement of the BDNF/CREB Signaling Pathway. Food Funct. 2022, 13, 6180–6194. [Google Scholar] [CrossRef] [PubMed]
- Capibaribe, V.C.C.; Vasconcelos Mallmann, A.S.; Lopes, I.S.; Oliveira, I.C.M.; de Oliveira, N.F.; Chaves, R.d.C.; Fernandes, M.L.; de Araujo, M.A.; da Silva, D.M.A.; Valentim, J.T. Thymol Reverses Depression-like Behaviour and Upregulates Hippocampal BDNF Levels in Chronic Corticosterone-Induced Depression Model in Female Mice. J. Pharm. Pharmacol. 2019, 71, 1774–1783. [Google Scholar] [CrossRef] [PubMed]
- Li, X.X.; Yu, Y.; Lang, X.Y.; Jiang, C.Y.; Lan, R.; Qin, X.Y. 2, 3, 5, 4′-Tetrahydroxystilbene-2-O-β-d-Glucoside Restores BDNF-TrkB and FGF2-Akt Signaling Axis to Attenuate Stress-Induced Depression. Neuroscience 2020, 430, 25–33. [Google Scholar] [CrossRef]
- Ren, Y.; Wang, J.L.; Zhang, X.; Wang, H.; Ye, Y.; Song, L.; Wang, Y.J.; Tu, M.J.; Wang, W.W.; Yang, L. Antidepressant-like Effects of Ginsenoside Rg2 in a Chronic Mild Stress Model of Depression. Brain Res. Bull. 2017, 134, 211–219. [Google Scholar] [CrossRef]
- Han, S.K.; Joo, M.K.; Kim, J.K.; Jeung, W.; Kang, H.; Kim, D.H. Bifidobacteria-Fermented Red Ginseng and Its Constituents Ginsenoside Rd and Protopanaxatriol Alleviate Anxiety/Depression in Mice by the Amelioration of Gut Dysbiosis. Nutrients 2020, 12, 901. [Google Scholar] [CrossRef]
- Zhang, L.L.; Yang, Z.Y.; Fan, G.; Ren, J.N.; Yin, K.J.; Pan, S.Y. Antidepressant-like Effect of Citrus Sinensis (L.) Osbeck Essential Oil and Its Main Component Limonene on Mice. J. Agric. Food Chem. 2019, 67, 13817–13828. [Google Scholar] [CrossRef]
- Tang, M.; Ai, Y.; Zhu, S.; Song, N.; Xu, X.; Liang, L.; Rong, B.; Zheng, X.; Zhang, L.; He, T. Antidepressant-Like Effect of Essential Oils from Citrus Reticulata in Reserpine-Induced Depressive Mouse. Nat. Prod. Commun. 2022, 17, 1934578X221093916. [Google Scholar] [CrossRef]
- Logan, A.C. Neurobehavioral Aspects of Omega-3 Fatty Acids: Possible Mechanisms and Therapeutic Value in Major Depression. Altern. Med. Rev. 2003, 8, 410–425. [Google Scholar]
- Wu, A.; Ying, Z.; Gomez-Pinilla, F. Dietary Omega-3 Fatty Acids Normalize BDNF Levels, Reduce Oxidative Damage, and Counteract Learning Disability after Traumatic Brain Injury in Rats. J. Neurotrauma 2004, 21, 1457–1467. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.J.; Lee, H.J.; Lee, H.E.; Park, S.J.; Gwon, Y.; Kim, H.; Zhang, J.; Shin, C.Y.; Kim, D.H.; Ryu, J.H. Oleanolic Acid Ameliorates Cognitive Dysfunction Caused by Cholinergic Blockade via TrkB-Dependent BDNF Signaling. Neuropharmacology 2017, 113, 100–109. [Google Scholar] [CrossRef] [PubMed]
- Xue, C.; Lv, H.; Li, Y.; Dong, N.; Wang, Y.; Zhou, J.; Shi, B.; Shan, A. Oleanolic Acid Reshapes the Gut Microbiota and Alters Immune-related Gene Expression of Intestinal Epithelial Cells. J. Sci. Food Agric. 2022, 102, 764–773. [Google Scholar] [CrossRef]
- Huang, H.S.; Wu, H.Y.; Chang, W.T.; Lin, Y.E.; Huang, Y.J.; Lu, K.H.; Lu, Y.S.; Chen, M.H.; Sheen, L.Y. The Antidepressive and Anxiolytic Effects of Formula Consisted of Poria Cocos and Cordyceps Militaris Waster Medium Extract in Unpredictable Chronic Mild Stress Animal Model. Curr. Dev. Nutr. 2020, 4, 1212. [Google Scholar] [CrossRef]
- Gao, X.; Feng, Y.; Xue, H.; Meng, M.; Qin, X. Antidepressant-like Effect of Triterpenoids Extracts from Poria Cocos on the CUMS Rats by 16S RRNA Gene Sequencing and LC–MS Metabolomics. J. Liq. Chromatogr. Relat. Technol. 2020, 43, 494–507. [Google Scholar] [CrossRef]
- Zhang, D.; Li, H.; Zhang, H.; Ye, X. Poria Cocos Water-Soluble Polysaccharide Modulates Anxiety-like Behavior Induced by Sleep Deprivation by Regulating the Gut Dysbiosis, Metabolic Disorders and TNF-α/NF-ΚB Signaling Pathway. Food Funct. 2022, 13, 6648–6664. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Qiao, Q.; Sun, Y.; Chen, Y.; Ren, B.; Liu, X. Sesamol Ameliorates Diet-induced Obesity in C57BL/6J Mice and Suppresses Adipogenesis in 3T3-L1 Cells via Regulating Mitochondria-lipid Metabolism. Mol. Nutr. Food Res. 2017, 61, 1600717. [Google Scholar] [CrossRef]
- Liu, Z.; Liu, X.; Luo, S.; Chu, C.; Wu, D.; Liu, R.; Wang, L.; Wang, J.; Liu, X. Extract of Sesame Cake and Sesamol Alleviate Chronic Unpredictable Mild Stress-Induced Depressive-like Behaviors and Memory Deficits. J. Funct. Foods 2018, 42, 237–247. [Google Scholar] [CrossRef]
- Xia, B.; Liu, X.; Li, X.; Wang, Y.; Wang, D.; Kou, R.; Zhang, L.; Shi, R.; Ye, J.; Bo, X. Sesamol Ameliorates Dextran Sulfate Sodium-Induced Depression-like and Anxiety-like Behaviors in Colitis Mice: The Potential Involvement of the Gut–Brain Axis. Food Funct. 2022, 13, 2865–2883. [Google Scholar] [CrossRef]
- Jiang, Y.; Liu, Y.; Gao, M.; Xue, M.; Wang, Z.; Liang, H. Nicotinamide Riboside Alleviates Alcohol-Induced Depression-like Behaviours in C57BL/6J Mice by Altering the Intestinal Microbiota Associated with Microglial Activation and BDNF Expression. Food Funct. 2020, 11, 378–391. [Google Scholar] [CrossRef] [PubMed]
- Alzoubi, K.H.; Abdel-Hafiz, L.; Khabour, O.F.; El-Elimat, T.; Alzubi, M.A.; Alali, F.Q. Evaluation of the Effect of Hypericum Triquetrifolium Turra on Memory Impairment Induced by Chronic Psychosocial Stress in Rats: Role of BDNF. Drug Des. Devel. Ther. 2020, 14, 5299–5314. [Google Scholar] [CrossRef] [PubMed]
- Sajjadi, S.E.; Mehregan, I.; Taheri, M. Essential Oil Composition of Hypericum Triquetrifolium Turra Growing Wild in Iran. Res. Pharm. Sci. 2015, 10, 90–94. [Google Scholar] [PubMed]
- Zhai, X.; Chen, Y.; Han, X.; Zhu, Y.; Li, X.; Zhang, Y.; Lu, Y. The Protective Effect of Hypericin on Postpartum Depression Rat Model by Inhibiting the NLRP3 Inflammasome Activation and Regulating Glucocorticoid Metabolism. Int. Immunopharmacol. 2022, 105, 108560. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Mou, C.; Bao, Y.; Xie, Y.; Jin, H.; Shen, H.; Zhou, W.; Zhang, J.; He, S.; Chen, B. Fucoxanthin Alleviates Methamphetamine-Induced Neurotoxicity Possibly via the Inhibition of Interaction between Keap1 and Nrf2. J. Funct. Foods 2021, 86, 104713. [Google Scholar] [CrossRef]
- Paudel, P.; Seong, S.H.; Jung, H.A.; Choi, J.S. Characterizing Fucoxanthin as a Selective Dopamine D3/D4 Receptor Agonist: Relevance to Parkinson’s Disease. Chem. Biol. Interact. 2019, 310, 108757. [Google Scholar] [CrossRef]
- Zhao, D.; Kwon, S.H.; Chun, Y.S.; Gu, M.Y.; Yang, H.O. Anti-Neuroinflammatory Effects of Fucoxanthin via Inhibition of Akt/NF-ΚB and MAPKs/AP-1 Pathways and Activation of PKA/CREB Pathway in Lipopolysaccharide-Activated BV-2 Microglial Cells. Neurochem. Res. 2017, 42, 667–677. [Google Scholar] [CrossRef]
- Jiang, X.; Wang, G.; Lin, Q.; Tang, Z.; Yan, Q.; Yu, X. Fucoxanthin Prevents Lipopolysaccharide-Induced Depressive-like Behavior in Mice via AMPK-NF-ΚB Pathway. Metab. Brain Dis. 2019, 34, 431–442. [Google Scholar] [CrossRef]
- Reboul, E. Absorption of Vitamin A and Carotenoids by the Enterocyte: Focus on Transport Proteins. Nutrients 2013, 5, 3563–3581. [Google Scholar] [CrossRef]
- Li, F.; Xiang, H.; Lu, J.; Chen, Z.; Huang, C.; Yuan, X. Lycopene Ameliorates PTSD-like Behaviors in Mice and Rebalances the Neuroinflammatory Response and Oxidative Stress in the Brain. Physiol. Behav. 2020, 224, 113026. [Google Scholar] [CrossRef]
- Zhao, B.; Wu, J.; Li, J.; Bai, Y.; Luo, Y.; Ji, B.; Xia, B.; Liu, Z.; Tan, X.; Lv, J. Lycopene Alleviates DSS-Induced Colitis and Behavioral Disorders via Mediating Microbes-Gut–Brain Axis Balance. J. Agric. Food Chem. 2020, 68, 3963–3975. [Google Scholar] [CrossRef]
- Al-Rahbi, B.; Zakaria, R.; Othman, Z.; Hassan, A.; Ahmad, A.H. Enhancement of BDNF Concentration and Restoration of the Hypothalamic-Pituitary-Adrenal Axis Accompany Reduced Depressive-like Behaviour in Stressed Ovariectomised Rats Treated with Either Tualang Honey or Estrogen. Sci. World J. 2014, 2014, 310821. [Google Scholar] [CrossRef] [PubMed]
- Azman, K.F.; Zakaria, R.; Abdul Aziz, C.B.; Othman, Z. Tualang Honey Attenuates Noise Stress-Induced Memory Deficits in Aged Rats. Oxid. Med. Cell. Longev. 2016, 2016, 1549158. [Google Scholar] [CrossRef] [PubMed]
- Sihoglu Tepe, A.; Ozaslan, M. Anti-Alzheimer, Anti-Diabetic, Skin-Whitening, and Antioxidant Activities of the Essential Oil of Cinnamomum Zeylanicum. Ind. Crops Prod. 2020, 145, 112069. [Google Scholar] [CrossRef]
- Aryanezhad, M.; Abdi, M.; Amini, S.; Hassanzadeh, K.; Valadbeigi, E.; Rahimi, K.; Izadpanah, E.; Moloudi, M.R. Cinnamomum Zeylanicum Extract Has Antidepressant-like Effects by Increasing Brain-Derived Neurotrophic Factor (BDNF) and Its Receptor in Prefrontal Cortex of Rats. Avicenna J. Phytomedicine 2021, 11, 302. [Google Scholar]
- Chen, W.; Lu, J.; Zhang, J.; Wu, J.; Yu, L.; Qin, L.; Zhu, B. Traditional Uses, Phytochemistry, Pharmacology, and Quality Control of Dendrobium Officinale Kimura et. Migo. Front. Pharmacol. 2021, 2026, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Fan, L.; Peng, Y.; Peng, C.; Li, X. Alcohol–Soluble Polysaccharides from Dendrobium Officinale Flowers as an Antidepressant by Regulating the Gut–Brain Axis. Int. J. Biol. Macromol. 2022, 216, 836–849. [Google Scholar] [CrossRef]
- Ye, Y.L.; Zhong, K.; Liu, D.D.; Xu, J.; Pan, B.B.; Li, X.; Yu, Y.P.; Zhang, Q. Huanglian-Jie-Du-Tang Extract Ameliorates Depression-like Behaviors through BDNF-TrkB-CREB Pathway in Rats with Chronic Unpredictable Stress. Evid. Based Complement. Altern. Med. 2017, 2017, 7903918. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.S.; Najam, R. Young Coconut Water Ameliorates Depression via Modulation of Neurotransmitters: Possible Mechanism of Action. Metab. Brain Dis. 2016, 31, 1165–1170. [Google Scholar] [CrossRef] [PubMed]
- Lima, E.B.C.; de Sousa, C.N.S.; Vasconcelos, G.S.; Meneses, L.N.; Ximenes, N.C.; Santos Júnior, M.A.; Matos, N.C.B.; Brito, R.; Miron, D.; Leal, L.K.A.M. Antidepressant, Antioxidant and Neurotrophic Properties of the Standardized Extract of Cocos Nucifera Husk Fiber in Mice. J. Nat. Med. 2016, 70, 510–521. [Google Scholar] [CrossRef]
- Chen, Y.; Xiao, N.; Chen, Y.; Chen, X.; Zhong, C.; Cheng, Y.; Du, B.; Li, P. Semen Sojae Praeparatum Alters Depression-like Behaviors in Chronic Unpredictable Mild Stress Rats via Intestinal Microbiota. Food Res. Int. 2021, 150, 110808. [Google Scholar] [CrossRef]
- Ma, S.; Xu, S.; Liu, B.; Li, J.; Feng, N.; Wang, L.; Wang, X. Long-Term Treatment of l-3-n-Butylphthalide Attenuated Neurodegenerative Changes in Aged Rats. Naunyn. Schmiedebergs. Arch. Pharmacol. 2009, 379, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Ma, H.; Fu, Z. Antidepressant-like Effect of 3-n-Butylphthalide in Rats Exposed to Chronic Unpredictable Mild Stress: Modulation of Brain-Derived Neurotrophic Factor Level and MTOR Activation in Cortex. Neurochem. Res. 2021, 46, 3075–3084. [Google Scholar] [CrossRef] [PubMed]
- Miyaoka, T.; Kanayama, M.; Wake, R.; Hashioka, S.; Hayashida, M.; Nagahama, M.; Okazaki, S.; Yamashita, S.; Miura, S.; Miki, H.; et al. Clostridium Butyricum MIYAIRI 588 as Adjunctive Therapy for Treatment-Resistant Major Depressive Disorder: A Prospective Open-Label Trial. Clin. Neuropharmacol. 2018, 41, 151–155. [Google Scholar] [CrossRef]
- Sun, J.; Wang, F.; Hu, X.; Yang, C.; Xu, H.; Yao, Y.; Liu, J. Clostridium Butyricum Attenuates Chronic Unpredictable Mild Stress-Induced Depressive-like Behavior in Mice via the Gut-Brain Axis. J. Agric. Food Chem. 2018, 66, 8415–8421. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.X.; Kang, X.N.; Cao, Y.; Zheng, D.X.; Lu, Y.M.; Pang, C.F.; Wang, Z.; Cheng, B.; Peng, Y. Porphyromonas Gingivalis Induces Depression via Downregulating P75NTR-Mediated BDNF Maturation in Astrocytes. Brain. Behav. Immun. 2019, 81, 523–534. [Google Scholar] [CrossRef]
- Bercik, P.; Park, A.J.; Sinclair, D.; Khoshdel, A.; Lu, J.; Huang, X.; Deng, Y.; Blennerhassett, P.A.; Fahnestock, M.; Moine, D. The Anxiolytic Effect of Bifidobacterium Longum NCC3001 Involves Vagal Pathways for Gut–Brain Communication. Neurogastroenterol. Motil. 2011, 23, 1132–1139. [Google Scholar] [CrossRef]
- Pinto-Sanchez, M.I.; Hall, G.B.; Ghajar, K.; Nardelli, A.; Bolino, C.; Lau, J.T.; Martin, F.P.; Cominetti, O.; Welsh, C.; Rieder, A. Probiotic Bifidobacterium Longum NCC3001 Reduces Depression Scores and Alters Brain Activity: A Pilot Study in Patients with Irritable Bowel Syndrome. Gastroenterology 2017, 153, 448–459. [Google Scholar] [CrossRef]
- Gu, F.; Wu, Y.; Liu, Y.; Dou, M.; Jiang, Y.; Liang, H. Lactobacillus Casei Improves Depression-like Behavior in Chronic Unpredictable Mild Stress-Induced Rats by the BDNF-TrkB Signal Pathway and the Intestinal Microbiota. Food Funct. 2020, 11, 6148–6157. [Google Scholar] [CrossRef]
- Yang, Y.; Zhao, S.; Yang, X.; Li, W.; Si, J.; Yang, X. The Antidepressant Potential of Lactobacillus Casei in the Postpartum Depression Rat Model Mediated by the Microbiota-Gut-Brain Axis. Neurosci. Lett. 2022, 774, 136474. [Google Scholar] [CrossRef]
- Bermúdez-Humarán, L.G.; Salinas, E.; Ortiz, G.G.; Ramirez-Jirano, L.J.; Morales, J.A.; Bitzer-Quintero, O.K. From Probiotics to Psychobiotics: Live Beneficial Bacteria Which Act on the Brain-Gut Axis. Nutrients 2019, 11, 890. [Google Scholar] [CrossRef]
- Heidarzadeh-Rad, N.; Gökmen-Özel, H.; Kazemi, A.; Almasi, N.; Djafarian, K. Effects of a Psychobiotic Supplement on Serum Brain-Derived Neurotrophic Factor Levels in Depressive Patients: A Post Hoc Analysis of a Randomized Clinical Trial. J. Neurogastroenterol. Motil. 2020, 26, 486. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.W.; Liong, M.T.; Tsai, Y.C. New Perspectives of Lactobacillus Plantarum as a Probiotic: The Gut-Heart-Brain Axis. J. Microbiol. 2018, 56, 601–613. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Kim, Y.K.; Han, P.L. Extracellular Vesicles Derived from Lactobacillus Plantarum Increase BDNF Expression in Cultured Hippocampal Neurons and Produce Antidepressant-like Effects in Mice. Exp. Neurobiol. 2019, 28, 158–171. [Google Scholar] [CrossRef]
- Cheon, M.J.; Lee, N.K.; Paik, H.D. Neuroprotective Effects of Heat-Killed Lactobacillus Plantarum 200655 Isolated from Kimchi against Oxidative Stress. Probiotics Antimicrob. Proteins 2021, 13, 788–795. [Google Scholar] [CrossRef]
- Ranuh, R.; Athiyyah, A.F.; Darma, A.; Risky, V.P.; Riawan, W.; Surono, I.S.; Sudarmo, S.M. Effect of the Probiotic Lactobacillus Plantarum IS-10506 on BDNF and 5HT Stimulation: Role of Intestinal Microbiota on the Gut-Brain Axis. Iran. J. Microbiol. 2019, 11, 145. [Google Scholar] [CrossRef]
- Schifano, E.; Zinno, P.; Guantario, B.; Roselli, M.; Marcoccia, S.; Devirgiliis, C.; Uccelletti, D. The Foodborne Strain Lactobacillus Fermentum MBC2 Triggers Pept-1-Dependent pro-Longevity Effects in Caenorhabditis Elegans. Microorganisms 2019, 7, 45. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Tang, M.; Wu, X.; Kong, X.; Liu, Y.; Xu, X. Lactobacillus Rhamnosus Zz-1 Exerts Preventive Effects on Chronic Unpredictable Mild Stress-Induced Depression in Mice via Regulating the Intestinal Microenvironment. Food Funct. 2022, 13, 4331–4343. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.L.; Luo, Y.; Jin, S.H.; Yuan, K.; Guo, Y. Ameliorative Effect of Anthocyanin on Depression Mice by Increasing Monoamine Neurotransmitter and Up-Regulating BDNF Expression. J. Funct. Foods 2020, 66, 103757. [Google Scholar] [CrossRef]
- Irie, Y.; Itokazu, N.; Anjiki, N.; Ishige, A.; Watanabe, K.; Keung, W.M. Eugenol Exhibits Antidepressant-like Activity in Mice and Induces Expression of Metallothionein-III in the Hippocampus. Brain Res. 2004, 1011, 243–246. [Google Scholar] [CrossRef]
- Donato, F.; de Gomes, M.G.; Goes, A.T.R.; Filho, C.B.; Del Fabbro, L.; Antunes, M.S.; Souza, L.C.; Boeira, S.P.; Jesse, C.R. Hesperidin Exerts Antidepressant-like Effects in Acute and Chronic Treatments in Mice: Possible Role of l-Arginine-NO-CGMP Pathway and BDNF Levels. Brain Res. Bull. 2014, 104, 19–26. [Google Scholar] [CrossRef]
- Witkamp, R.F.; van Norren, K. Let Thy Food Be Thy Medicine when Possible. Eur. J. Pharmacol. 2018, 836, 102–114. [Google Scholar] [CrossRef] [PubMed]
- Cohen-Cory, S.; Kidane, A.H.; Shirkey, N.J.; Marshak, S. Brain-derived Neurotrophic Factor and the Development of Structural Neuronal Connectivity. Dev. Neurobiol. 2010, 70, 271–288. [Google Scholar] [CrossRef] [PubMed]
- Cartwright, C.; Gibson, K.; Read, J.; Cowan, O.; Dehar, T. Long-Term Antidepressant Use: Patient Perspectives of Benefits and Adverse Effects. Patient Prefer. Adherence 2016, 10, 1401. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Kim, M.H.; Kawachi, I.; Cho, Y. Comparative Epidemiology of Suicide in South Korea and Japan: Effects of Age, Gender and Suicide Methods. Cris. J. Cris. Interv. Suicide Prev. 2011, 32, 5. [Google Scholar] [CrossRef]
- Zadeh, A.R.; Eghbal, A.F.; Mirghazanfari, S.M.; Ghasemzadeh, M.R.; Nassireslami, E.; Donyavi, V. Nigella Sativa Extract in the Treatment of Depression and Serum Brain-Derived Neurotrophic Factor (BDNF) Levels. J. Res. Med. Sci. Off. J. Isfahan Univ. Med. Sci. 2022, 27, 1–19. [Google Scholar]
- Sangiovanni, E.; Brivio, P.; Dell’Agli, M.; Calabrese, F. Botanicals as Modulators of Neuroplasticity: Focus on BDNF. Neural Plast. 2017, 2017, 5965371. [Google Scholar] [CrossRef]
- Dean, J.; Keshavan, M. The Neurobiology of Depression: An Integrated View. Asian J. Psychiatr. 2017, 27, 101–111. [Google Scholar] [CrossRef]
- Zhang, J.; Onakpoya, I.J.; Posadzki, P.; Eddouks, M. The Safety of Herbal Medicine: From Prejudice to Evidence. Evid. Based Complement. Altern. Med. 2015, 2015, 316706. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, T.; Wang, Y.; Si, C.; Wang, X.; Wang, R.T.; Lv, Z. Baicalin Ameliorates Neuropathology in Repeated Cerebral Ischemia-Reperfusion Injury Model Mice by Remodeling the Gut Microbiota. Aging 2020, 12, 3791. [Google Scholar] [CrossRef]
- McCain, J.A. Antidepressants and Suicide in Adolescents and Adults: A Public Health Experiment with Unintended Consequences? Pharm. Ther. 2009, 34, 355. [Google Scholar]
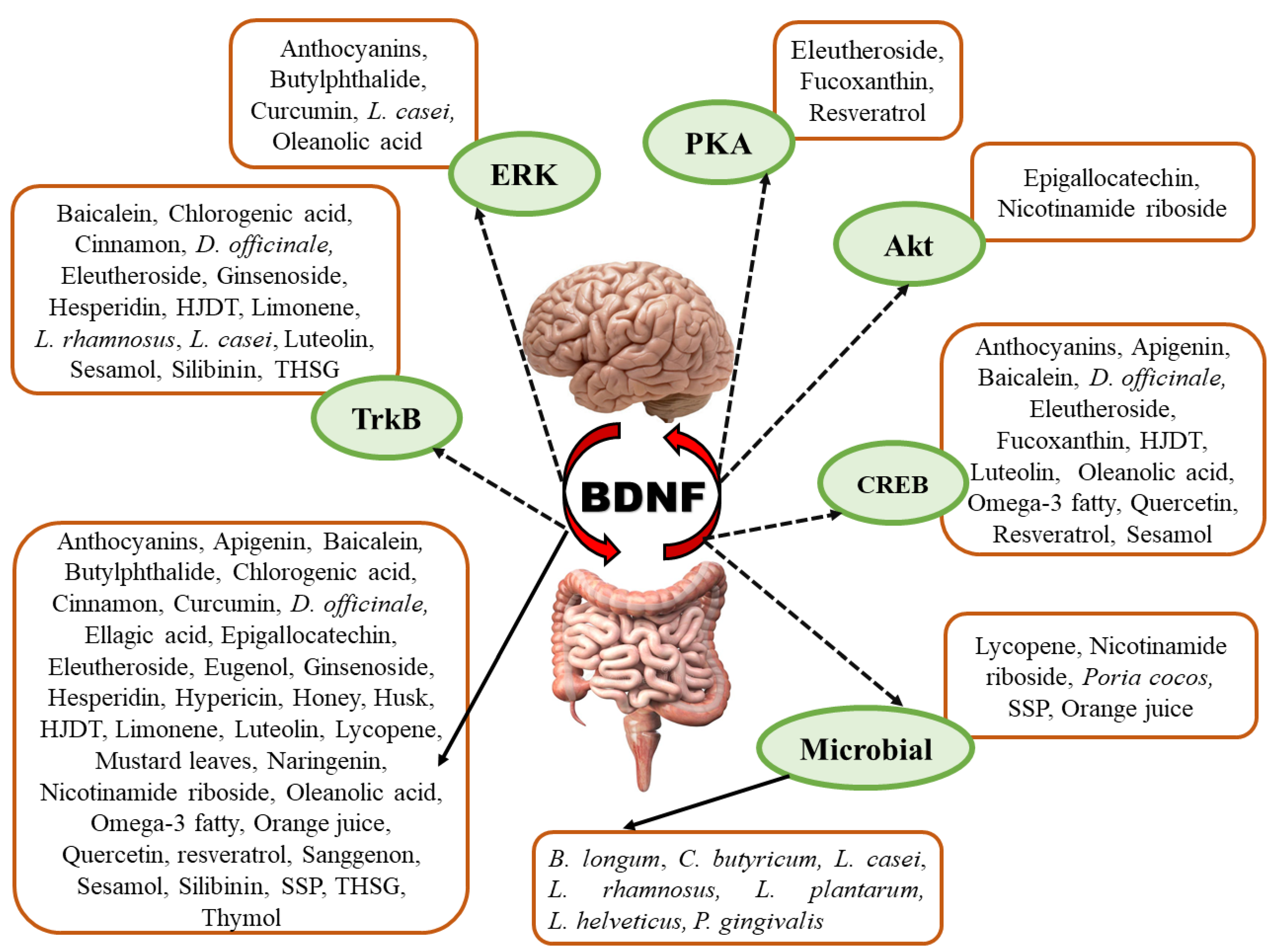
| Natural Products | Sources | Targeted Pathway | Model | Pharmacological Action | References |
|---|---|---|---|---|---|
| Cyandin-3-glucoside and cyanidin-3-sophoroside-5-glucoside | Cauliflower (Brassica oleraceavar. Botrytis L.) | ERK/CREB/ BDNF | Pathogen-free female mice (4 weeks old) | Upregulates BDNF, which also promotes neurogenesis and dendrite development in hippocampus | [181] |
| Apigenin | Fruits and vegetables | CREB/BDNF | Male Swiss albino mice (12 weeks old) | Prevents cognitive deficit and CREB-BDNF upregulation in the hippocampus | [78] |
| Baicalein | Scutellaria baicalensis Georgi | BDNF/TrkB/ CREB | Male C57BL/6J mice (8 weeks old) | Synaptic function protection and neuroprotection against inflammation and depression | [89,90] |
| 3-n-Butylphthalide | Chinese celery | BDNF/ERK/ mTOR and serotonergic | Male Sprague–Dawley (SD; 6 weeks old) | Restores the levels of BDNF, p-ERK, and mTOR | [165] |
| Chlorogenic acid | Coffee beans | BDNF/TrkB | Male SD rats (6 weeks old) | Induces anxiolytic behavior, modulates autonomic regulation, and activates hippocampal BDNF signaling | [108] |
| Cinnamon | Cinnamomum zeylanicum | BDNF/TrkB | Thirty-two male Wistar rats | Antidepressant | [157] |
| Curcumin | Curcuma longa | BDNF/ERK | Adult male Wistar (6 weeks old) | Antidepressant | [110,111,112] |
| Alcohol-soluble polysaccharides | Dendrobium officinale | BDNF/TrkB/ CREB | Male ICR mice (6 weeks old) and SD rats (6 weeks old) | Antidepressant-like effect by regulating the gut–brain axis. | [159] |
| Ellagic acid | Punica granatum | BDNF and serotoninergic | Male C57BL/6 mice (6 weeks old) | Antidepressant | [114,115] |
| Epigallocatechin-3-gallate | Green tea (Camellia sinensis) | BDNF, Akt, monoaminergic | Female SD rats (6 weeks old), male C57BL/6J mice | Modulates mood, mitigates anxiety, and improves postmenopausal depression | [116,117] |
| Eleutheroside E | Acanthopanax senticosus HARM | PKA/CREB/ BDNF, CREB/BDNF | Male SD rats (6 weeks old) | Antidepressant and regulates autonomic function | [119,120] |
| Eugenol | Rhizoma acori graminei | BDNF | Fifty male ddy mice (6 weeks old) | Antidepressant | [182] |
| Fucoxanthin | PKA/cAMP, CREB | BV-2 cells | Ameliorates neurodegenerative diseases | [149] | |
| Ginsenoside Rg2 | Panax ginseng | BDNF/TrkB | Adult male C57BL/6J mice (8 weeks old) | Mitigates anxiety/depression and colitis | [129,130] |
| Hesperidin | Citrus fruits | BDNF/TrkB | ICR male mice (6–8 weeks old), male NMRI mice (10–11 weeks old) | Reduces CUMS and depression caused by mild traumatic brain injury | [91,92,183] |
| Hypericin | Hypericum triquetrifolium | BDNF | Adult male Wistar rats | Neuroprotection against depression and inflammation | [144,146] |
| Limonene | Citrus Sinensis, C. Reticulata | BDNF/TrkB | 155 male Kunming mice at 5 weeks | Antidepressant | [131,132] |
| Luteolin-7-O-glucuronide | Perilla frutescens | BDNF/TrkB/ CREB | C57BL/6 male mice (7 weeks old) | Improves depression-like and stress-coping behaviors in sleep deprivation | [94] |
| Lycopene | Solanum lycopersicum, Citrullus lanatus | BDNF, gut microbiota | Male C57BL/6 mice (8 weeks old) | Improves DSS-induced depression and anxiety-like behavior | [152,153] |
| Mustard leaves | Brassica juncea | BNDF and apoptosis | Male C57BL/6 mice (6 weeks old) | Reduces stress and depression | [122] |
| Naringenin | Grapes (Vitis vinifera) | BDNF | Thirty-five male Swiss mice | Reduces stress and depression | [96,97] |
| Nicotinamide riboside | Milk and yeast | gut microbial pathway, BDNF/AKT | Male C57BL/6J mice (7 weeks old) | Alters intestinal microbiota with microglial activation and exhibits antidepressant activity | [143] |
| Oleanolic acid | Olea europaea | ERK1/CREB/ BDNF | Male ICR mice (6 weeks old) | Antidepressant | [135,136] |
| Omega-3 fatty acids | Fish oil | BDNF/CREB | Male SD rats | Maintains neuronal function and plasticity, improves impaired learning ability after traumatic brain injury, and exhibits antidepressant activity | [134] |
| Quercetin | Fruits and vegetables | CREB/BDNF | Male SD rats (8 weeks old) | Antidepressant | [98,99] |
| Resveratrol | Polygonum cuspidatum | PKA/CREB/ BDNF | Male adult SD | Exhibits anti-IBS-like effects on depression, anxiety, visceral hypersensitivity, and intestinal motility | [124,125] |
| Sanggenon G | Morus alba | BDNF | Male SD rats (8 weeks old) | Antidepressant | [101] |
| Sesamol | Sesame oil and seeds (Sesamum indicum L.) | BDNF/TrkB/ CREB | C57BL/6J male mice (8 weeks old) | Prevents IBD and associated symptoms of anxiety and depression | [142] |
| Silibinin | Silybum marianum | BDNF/TrkB | Male SD rats (6–8 weeks old), male C57BL/6 mice | Attenuated autophagy and neuroprotection against anxiety and depression | [103,104] |
| Triterpenoids | Poria cocos | microbial | Male SD rats | Antidepressant | [138] |
| 2,3,5,4′-Tetrahydroxystilbene-2-O-β-d-glucoside | Polygonum multiflorum | BDNF/TrkB | C57BL/6J mice (6–8 weeks old) | Antidepressant | [128] |
| Thymol | T. vulgaris | BDNF | Adult female Swiss mice (8–10 weeks old) | Restores short-term memory and improves depression | [127] |
| Orange juice | Gut microbiota and BDNF | Young adults | Reduces depression through gut microbiota modulation | [107] | |
| Honey | Tualang honey | BDNF | Female SD rats (8 weeks old) | Exhibits anxiolytic and antidepressant effects | [155] |
| Huanglian-Jie-Du-Tang | Coptis chinensis Franch | BDNF/TrkB/ CREB | Male SD rats | Exhibits antidepressant-like effect | [160] |
| Husk fiber | Cocos nucifera | BDNF | Male Swiss mice (8 weeks old) | Neuroprotection against oxidative stress and depression | [162] |
| Semen Sojae Praeparatum | Mixture of mulberry leaves, lentils, black beans fermented with bacillus sp. DU-106, R. Chinensis 12 | microbial and BDNF | Adult male SD rats | Antidepressant and modulates gut microbiota | [163] |
| Probiotic | Bifidobacterium longum NCC3001 | BDNF | Adult patients with a diagnosis of IBS with diarrhea and mild to moderate anxiety and/or depression | Antidepressant and adequate relief of IBS symptoms | [170] |
| Probiotic | Clostridium butyricum | BDNF and serotonin | Adult (age 6–8 weeks, 18–22 g) male C57BL/6J mice | Regulates gut microbiota and acts as antidepressant | [166,167] |
| Probiotic | Lactobacillus casei | BDNF/ERK, BDNF/TrkB | Pregnant SD female rats | Antidepressant altering gut microbiota composition, brain monoamines, and oxidative stress | [171,172] |
| Probiotic | Lactobacillus helveticus R0052 | BDNF | Patients with mild to moderate melancholic depression | Antidepressant | [174] |
| Probiotic | Lactobacillus plantarum | BDNF | Male C57BL/6J mice (7 weeks old), male Wistar rats (8–12 weeks old) | Antidepressant, stimulates the gut–brain axis, and can potentially promote brain development and function. | [176,177,178] |
| Probiotic | Lactobacillus rhamnosus zz-1 | BDNF/TrkB | Male C57BL/6 mice (4 weeks old) | Antidepressant via regulating the intestinal microenvironment | [180] |
| Probiotic | Porphyromonas gingivalis | BDNF | Female C57Bl/6J wild-type mice | Antidepressant | [168] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liaqat, H.; Parveen, A.; Kim, S.-Y. Antidepressive Effect of Natural Products and Their Derivatives Targeting BDNF-TrkB in Gut–Brain Axis. Int. J. Mol. Sci. 2022, 23, 14968. https://doi.org/10.3390/ijms232314968
Liaqat H, Parveen A, Kim S-Y. Antidepressive Effect of Natural Products and Their Derivatives Targeting BDNF-TrkB in Gut–Brain Axis. International Journal of Molecular Sciences. 2022; 23(23):14968. https://doi.org/10.3390/ijms232314968
Chicago/Turabian StyleLiaqat, Humna, Amna Parveen, and Sun-Yeou Kim. 2022. "Antidepressive Effect of Natural Products and Their Derivatives Targeting BDNF-TrkB in Gut–Brain Axis" International Journal of Molecular Sciences 23, no. 23: 14968. https://doi.org/10.3390/ijms232314968
APA StyleLiaqat, H., Parveen, A., & Kim, S.-Y. (2022). Antidepressive Effect of Natural Products and Their Derivatives Targeting BDNF-TrkB in Gut–Brain Axis. International Journal of Molecular Sciences, 23(23), 14968. https://doi.org/10.3390/ijms232314968






