Surprising Solid-State ESIPT Emission from Apparently Ordinary Salicyliden Glycinates Schiff Bases
Abstract
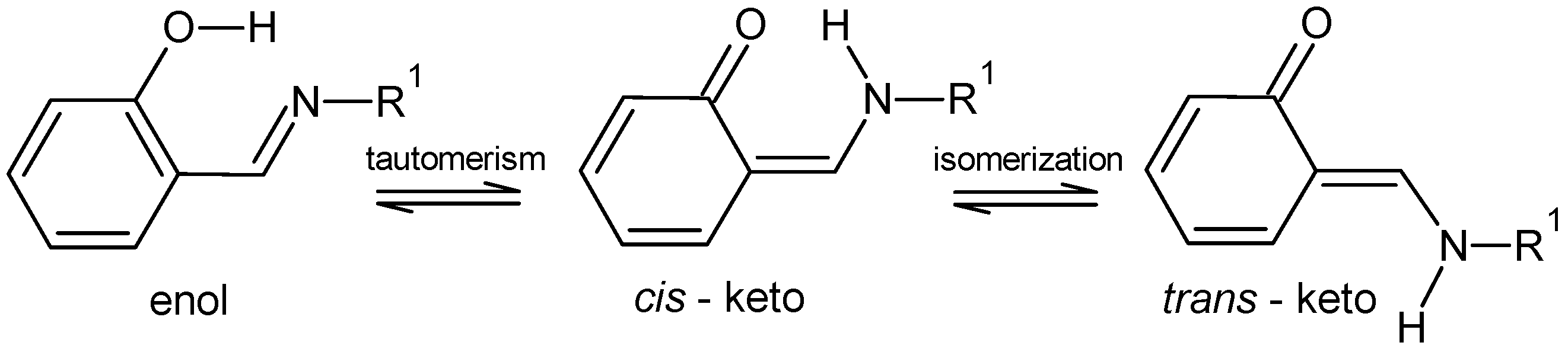
1. Introduction
2. Results
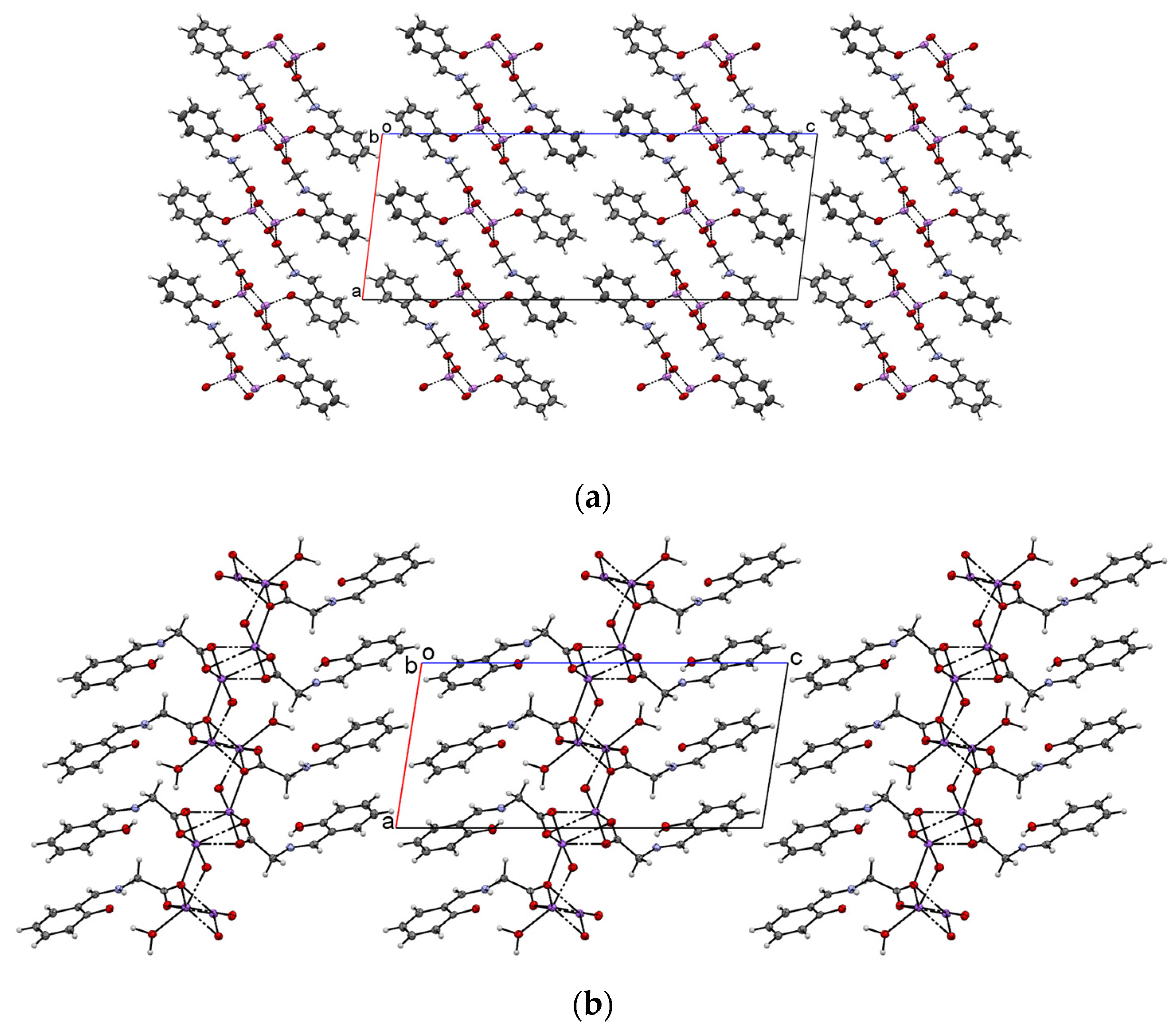
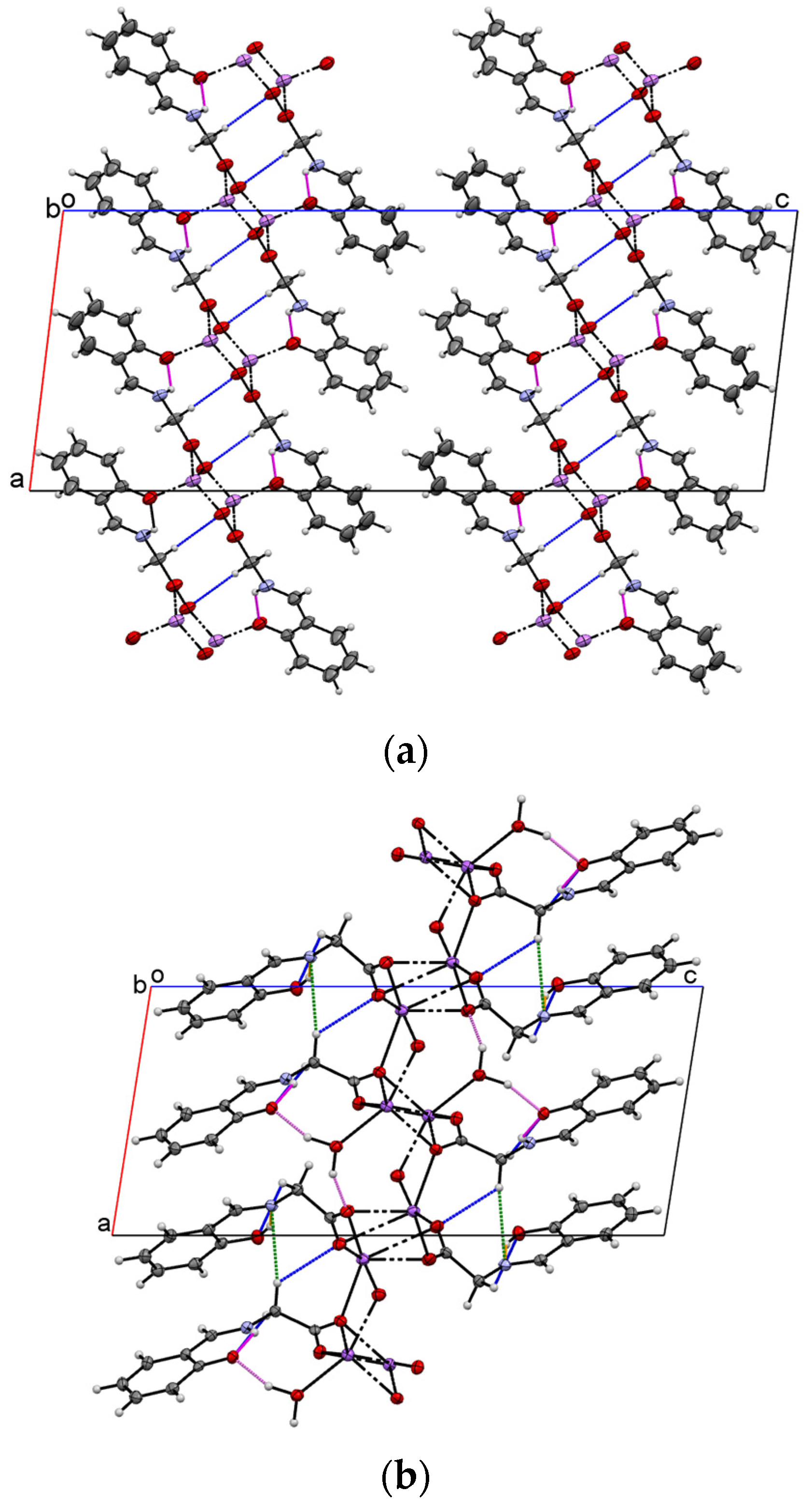
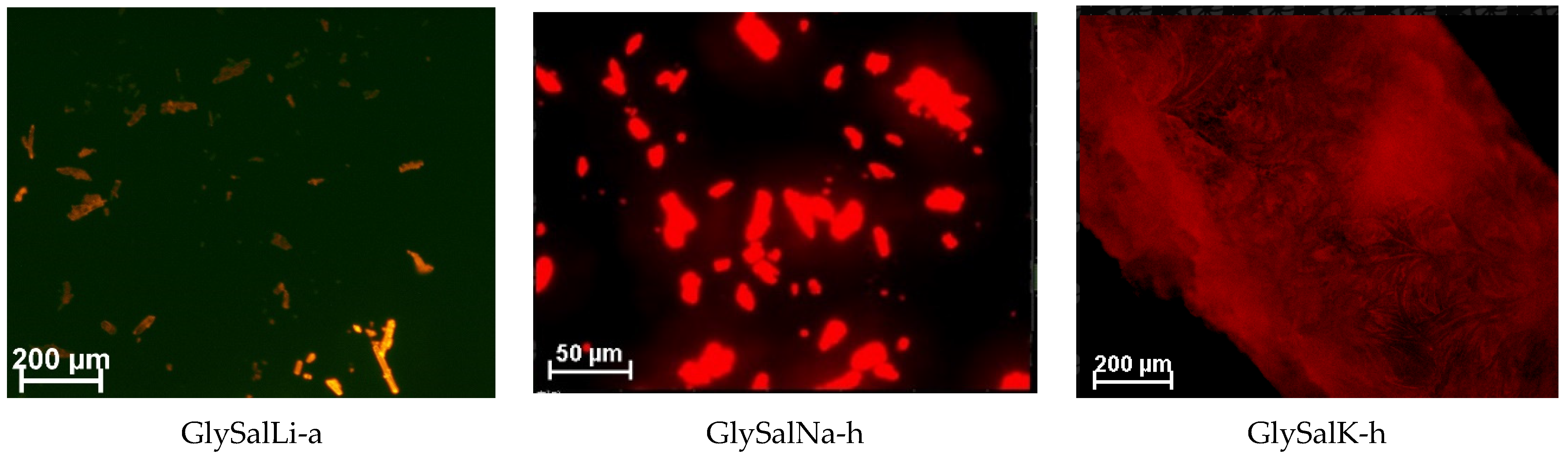
2.1. Crystal Structure
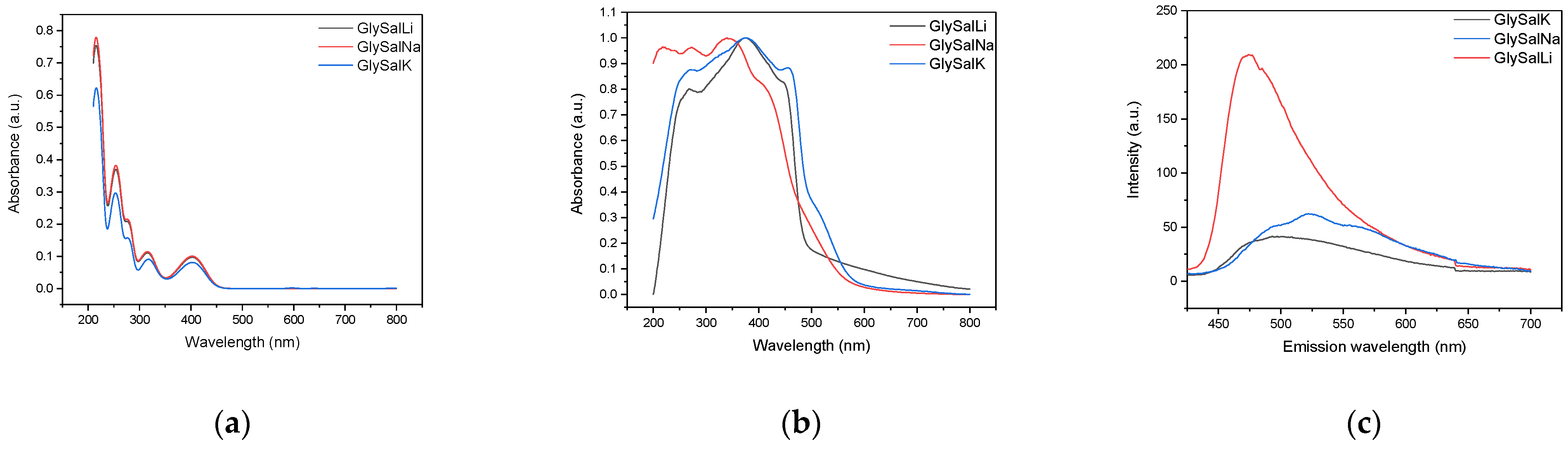
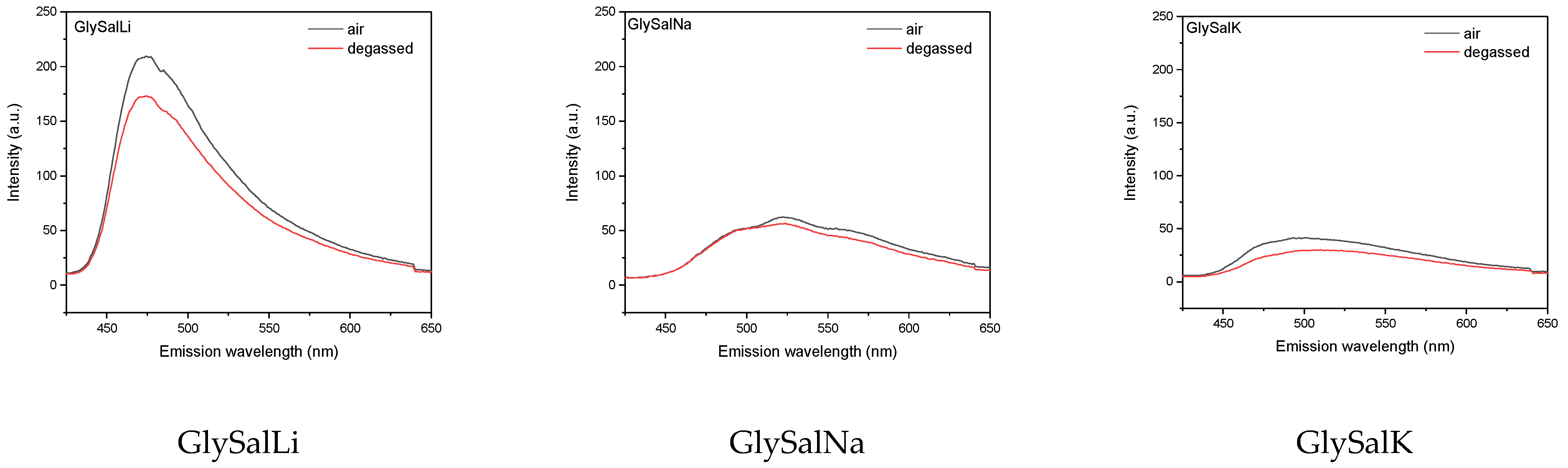
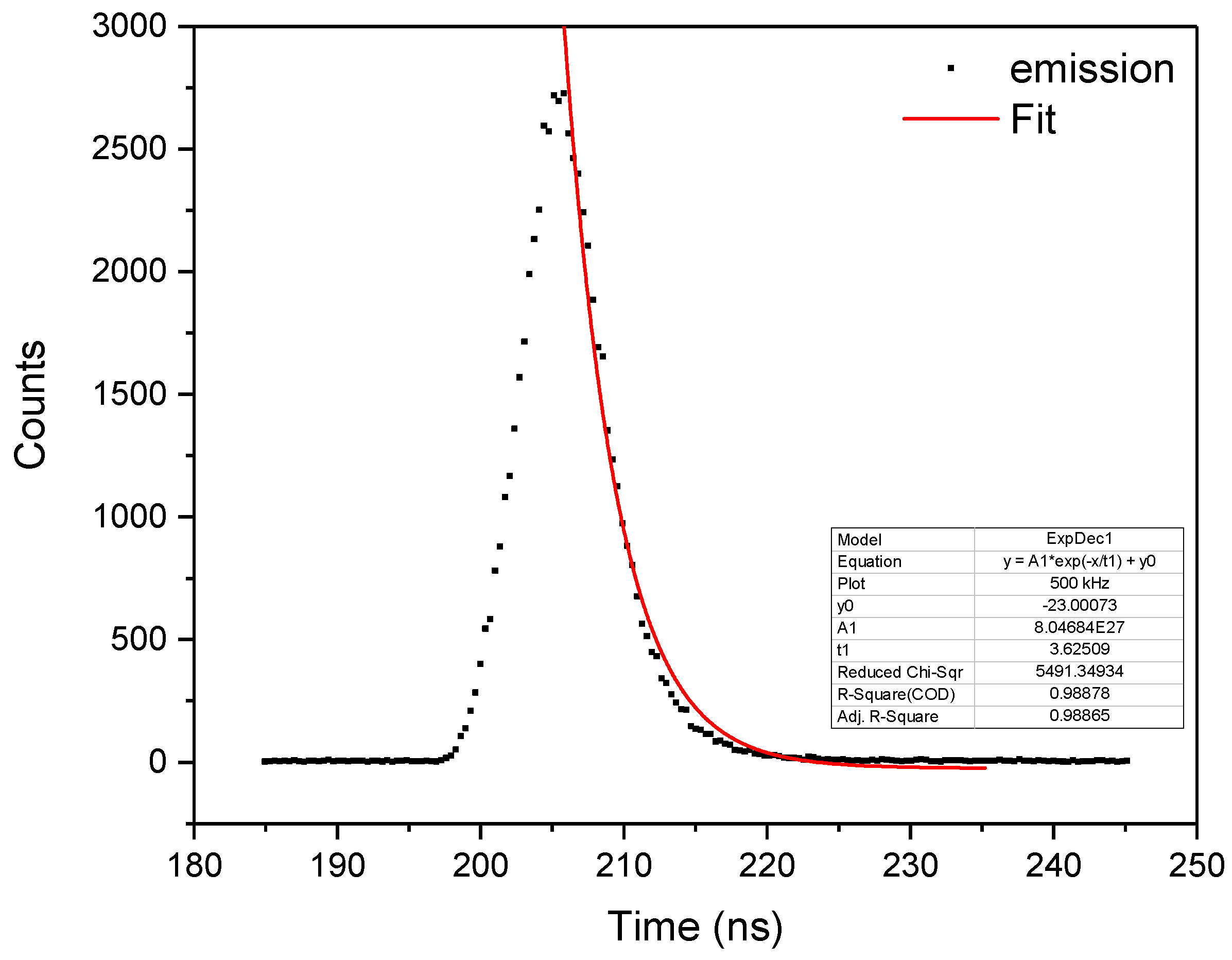
2.2. Absorption and Emission Properties
3. Materials and Methods
3.1. Synthesis of Salicyl-Glycine Schiff Bases Salts GlySalX (GlySalLi or GlySalNa)
3.2. Solid State and Solution Fluorescence Measurements
3.3. UV-Vis Absorption Spectroscopy
3.4. NMR Spectroscopy
3.5. X-Ray Crystallography
3.6. Confocal Microscopy
4. Summary and Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Padalkar, V.S.; Seki, S. Excited-State Intramolecular Proton-Transfer (ESIPT)-Inspired Solid State Emitters. Chem. Soc. Rev. 2016, 45, 169–202. [Google Scholar] [CrossRef] [PubMed]
- Hadjoudis, E. Photochromic and Thermochromic Anils. Mol. Eng. 1995, 5, 301–337. [Google Scholar] [CrossRef]
- Mitra, S.; Tamai, N. Dynamics of Photochromism in Salicylideneaniline: A Femtosecond Spectroscopic Study. Phys. Chem. Chem. Phys. 2003, 5. [Google Scholar] [CrossRef]
- Ganie, A.A.; Dar, A.A. Water Switched Reversible Thermochromism in Organic Salt of Sulfonated Anil. Cryst. Growth Des. 2021, 21, 3014–3023. [Google Scholar] [CrossRef]
- Cai, M.; Gao, Z.; Zhou, X.; Wang, X.; Chen, S.; Zhao, Y.; Qian, Y.; Shi, N.; Mi, B.; Xie, L.; et al. A Small Change in Molecular Structure, a Big Difference in the AIEE Mechanism. Phys. Chem. Chem. Phys. 2012, 14, 5289. [Google Scholar] [CrossRef] [PubMed]
- Hadjoudis, E.; Mavridis, I.M. Photochromism and Thermochromism of Schiff Bases in the Solid State: Structural Aspects. Chem. Soc. Rev. 2004, 33, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Weller, A. Über Die Fluoreszenz Der Salizylsäure Und Verwandter Verbindungen. Naturwissenschaften 1955, 42, 175–176. [Google Scholar] [CrossRef]
- Shida, T.; Mutai, T.; Araki, K. Sterically Induced Polymorphism: ON-OFF Control of Excited-State Intramolecular Proton Transfer (ESIPT) Luminescence of 1-Methyl-2-(2’-Hydroxyphenyl)Benzimidazole. CrystEngComm 2013, 15, 10179–10182. [Google Scholar] [CrossRef]
- Chou, P.-T.; Chen, Y.; Yu, W.; Chou, Y.; Wei, C.; Cheng, Y.-M. Excited-State Intramolecular Proton Transfer in 10-Hydroxybenzo[h]Quinoline. J. Phys. Chem. A 2001, 105, 1731–1740. [Google Scholar] [CrossRef]
- Oliveira, F.F.D.; Santos, D.C.B.D.; Lapis, A.A.M.; Corrêa, J.R.; Gomes, A.F.; Gozzo, F.C.; Moreira, P.F.; De Oliveira, V.C.; Quina, F.H.; Neto, B.A.D. On the Use of 2,1,3-Benzothiadiazole Derivatives as Selective Live Cell Fluorescence Imaging Probes. Bioorganic Med. Chem. Lett. 2010, 20, 6001–6007. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Kaneko, Y.; Arai, T. Photoinduced Intramolecular Hydrogen Atom Transfer of N-Salicylideneaniline in the Triplet State. Chem. Lett. 2000, 7, 756–757. [Google Scholar] [CrossRef]
- Mei, J.; Hong, Y.; Lam, J.W.Y.; Qin, A.; Tang, Y.; Tang, B.Z. Aggregation-Induced Emission: The Whole Is More Brilliant than the Parts. Adv. Mater. 2014, 26, 5429–5479. [Google Scholar] [CrossRef] [PubMed]
- Mei, J.; Leung, N.L.C.; Kwok, R.T.K.; Lam, J.W.Y.; Tang, B.Z. Aggregation-Induced Emission: Together We Shine, United We Soar! Chem. Rev. 2015, 115, 11718–11940. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Kato, T.; Huang, H.; Yoshikawa, I.; Mutai, T.; Houjou, H. Photochromism of Salicylideneanilines Bearing Super Bulky Substituents: Single-Crystal UV-Vis Spectroscopic Examination of Bleaching under Variable Temperature and Visible-Light Irradiation. J. Photochem. Photobiol. A Chem. 2019, 385, 112096. [Google Scholar] [CrossRef]
- Bakalorz, K.; Przypis, Ł.; Tomczyk, M.M.; Książek, M.; Grzesik, R.; Kuźnik, N. Unprecedented Water Effect as a Key Element in Salicyl-Glycine Schiff Base Synthesis. Molecules 2020, 25, 1257. [Google Scholar] [CrossRef] [PubMed]
- Desiraju, G.R.; Steiner, T. The Weak Hydrogen Bond in Structural Chemistry and Biology; Oxford University Press: Oxford, UK, 1999. [Google Scholar]
- Sakai, K.; Kawamura, H.; Kobayashi, N.; Ishikawa, T.; Ikeda, C.; Kikuchi, T.; Akutagawa, T. Highly Efficient Solid-State Red Fluorophores Using ESIPT: Crystal Packing and Fluorescence Properties of Alkoxy-Substituted Dibenzothiazolylphenols. CrystEngComm 2014, 16. [Google Scholar] [CrossRef]
- Rigaku Oxford Diffraction. Crysalispro Software System, version 1.171.38.41q; Rigaku Corporation: Wroclaw, Poland, 2015. [Google Scholar]
- Sheldrick, G.M. Crystal refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar] [CrossRef]

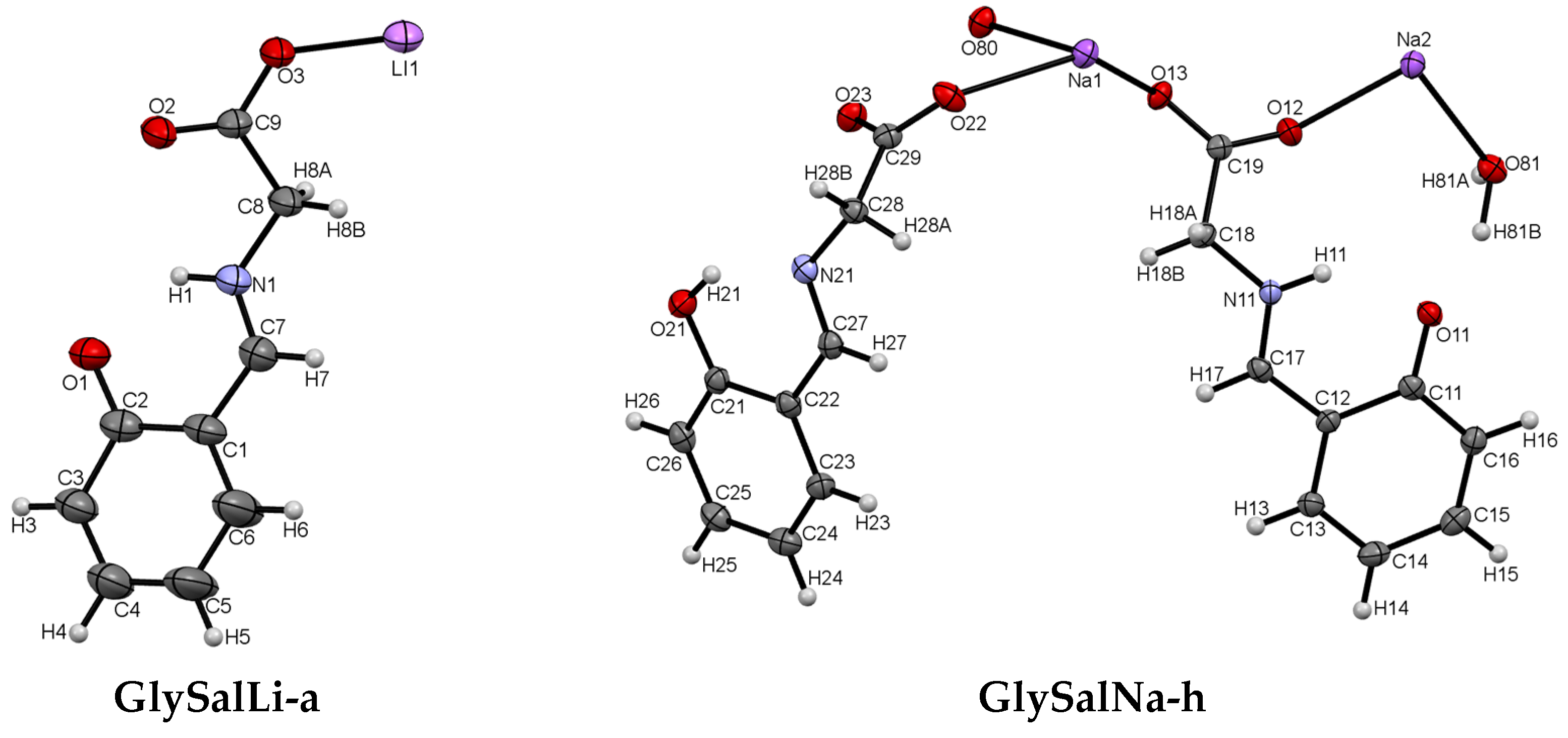
| GlySalLi-a | GlySalNa-h | |
|---|---|---|
| Empirical formula | C9H8LiNO3 | C18H18N2Na2O8 |
| Molecular weight | 185.10 | 435.32 |
| Temperature [K] | 100 | 100 |
| Crystal system | Monoclinic | Monoclinic |
| Space group | I2/a | P21 |
| Unit cell dimensions | ||
| a [Å] | 11.6482(9) | 8.6872(2) |
| b [Å] | 5.0003(4) | 5.8744(1) |
| c [Å] | 30.317(2) | 19.0522(6) |
| β [°] | 96.865(8) | 98.906(2) |
| V [Å3] | 1753.1(2) | 960.55(4) |
| Z | 8 | 2 |
| Density (calculated) [Mg/m3] | 1.403 | 1.509 |
| Absorption coefficient [mm−1] | 0.869 | 1.393 |
| F(000) | 768 | 452 |
| Theta range for data collection | 2.936 to 74.822 | 4.699 to 73.696 |
| Index ranges | −14 ≤ h ≤ 10 | −10 ≤ h ≤ 10 |
| −5 ≤ k ≤ 6 | −7 ≤ k ≤ 6 | |
| −37 ≤ l ≤ 36 | −23 ≤ l ≤ 22 | |
| Reflections collected | 4141 | 6598 |
| Independent reflections | 1685 | 3200 |
| R(int) | 0.0636 | 0.0258 |
| Completeness to theta = 67.684° [%] | 98.4 | 99.9 |
| Data/restraints/parameters | 1685/0/127 | 3200/0/273 |
| Goodness-of-fit on F2 | 1.031 | 1.071 |
| Final R indices [I > 2σ(I)] | R1 = 0.0714 wR2 = 0.1735 | R1 = 0.0361 wR2 = 0.0967 |
| R indices (all data) | R1 = 0.1245 wR2 = 0.2119 | R1 = 0.0381 wR2 = 0.0986 |
| Largest diff. peak and hole [eÅ−3] | 0.289 and −0.328 | 0.480 and −0.310 |
| Structure | Torsion Angle [°] | Conformation |
|---|---|---|
| GlySalLi-a | 135.4(4) | tilted anticlinal |
| GlySalNa-h | 118.3(3); 139.7(3) | anticlinal; tilted anticlinal |
| GlySalK-a * | 172.38(11) | antiperiplanar |
| GlySalK-h * | 124.4(4) | anticlinal |
| Compound | D-H···A | d(D-H) | d(H···A) | d(D···A) | <(DHA) |
|---|---|---|---|---|---|
| GlySalLi-a | N1-H1···O1 | 0.88 | 1.87 | 2.574(4) | 136.0 |
| C8-H8A···O3 (i) | 0.99 | 2.52 | 3.468(5) | 160.6 | |
| GlySalNa-h | N11-H11···O11 | 0.88 | 1.89 | 2.595(4) | 135.9 |
| C18-H18A···O23 | 0.99 | 2.61 | 3.309(3) | 127.3 | |
| C18-H18A···N21 (ii) | 0.99 | 2.67 | 3.651(3) | 168.6 | |
| C18-H18B···O11 (iii) | 0.99 | 2.50 | 3.389(4) | 149.1 | |
| C28-H28A···O21 (ii) | 0.99 | 2.71 | 3.351(4) | 122.8 | |
| O21-H21···N21 | 0.84 | 1.85 | 2.596(4) | 147.2 | |
| O81-H81A···O22 (iv) | 1.04 | 1.68 | 2.659(3) | 154.1 | |
| O81-H81B···O11 | 1.05 | 1.73 | 2.714(3) | 155.0 |
| Compound | Absorption (nm) | Emission (nm) | Stokes Shift (cm−1) | PLQY (%) |
|---|---|---|---|---|
| GlySalLi | 373 | 470 | 5533 | 12.1 |
| GlySalNa | 340 | 520 | 10,181 | 10.1 |
| GlySalK | 378 | 500 | 6455 | 8.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomczyk, M.M.; Przypis, Ł.; Shiraki, T.; Kusz, J.; Książek, M.; Janas, D.; Kuźnik, N. Surprising Solid-State ESIPT Emission from Apparently Ordinary Salicyliden Glycinates Schiff Bases. Int. J. Mol. Sci. 2022, 23, 14955. https://doi.org/10.3390/ijms232314955
Tomczyk MM, Przypis Ł, Shiraki T, Kusz J, Książek M, Janas D, Kuźnik N. Surprising Solid-State ESIPT Emission from Apparently Ordinary Salicyliden Glycinates Schiff Bases. International Journal of Molecular Sciences. 2022; 23(23):14955. https://doi.org/10.3390/ijms232314955
Chicago/Turabian StyleTomczyk, Mateusz M., Łukasz Przypis, Tomohiro Shiraki, Joachim Kusz, Maria Książek, Dawid Janas, and Nikodem Kuźnik. 2022. "Surprising Solid-State ESIPT Emission from Apparently Ordinary Salicyliden Glycinates Schiff Bases" International Journal of Molecular Sciences 23, no. 23: 14955. https://doi.org/10.3390/ijms232314955
APA StyleTomczyk, M. M., Przypis, Ł., Shiraki, T., Kusz, J., Książek, M., Janas, D., & Kuźnik, N. (2022). Surprising Solid-State ESIPT Emission from Apparently Ordinary Salicyliden Glycinates Schiff Bases. International Journal of Molecular Sciences, 23(23), 14955. https://doi.org/10.3390/ijms232314955








