Vitamin D Deficiency and COVID-19: A Biological Database Study on Pathways and Gene-Disease Associations
Abstract
1. Introduction
2. Results
2.1. Description of Genetic Variants of a VD Deficiency and COVID-19
2.2. Functional Analyses and Enrichment Analyses
2.3. Gene-Disease Associations
3. Discussion
4. Materials and Methods
4.1. Selection of Genetic Variants
4.2. Annotation of Genes
4.3. Linkage Disequilibrium Analysis
4.4. Identification of the Overlapping Genes
4.5. Biological Database Studies
4.5.1. In Silico Functional Analyses and Enrichment Analyses
4.5.2. SNP-Disease and Gene-Disease Associations, including Enrichment Analyses
4.6. Workflow of the Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- DeLuca, H.F. Metabolism of Vitamin D: Current Status. Am. J. Clin. Nutr. 1976, 29, 1258–1270. [Google Scholar] [CrossRef] [PubMed]
- Pike, J.W.; Christakos, S. Biology and Mechanisms of Action of the Vitamin D Hormone. Endocrinol. Metab. Clin. N. Am. 2017, 46, 815–843. [Google Scholar] [CrossRef] [PubMed]
- Kumar Maurya, V.; Aggarwal, M. Factors Influencing the Absorption of Vitamin D in GIT: An Overview. J. Food Sci. Technol. 2017, 54, 3753–3765. [Google Scholar] [CrossRef]
- Weber, F. Absorption Mechanisms for Fat-Soluble Vitamins and the Effect of Other Food Constituents. Prog. Clin. Biol. Res. 1981, 77, 119–135. [Google Scholar] [PubMed]
- Niramitmahapanya, S.; Harris, S.S.; Dawson-Hughes, B. Type of Dietary Fat Is Associated with the 25-Hydroxyvitamin D3 Increment in Response to Vitamin D Supplementation. J. Clin. Endocrinol. Metab. 2011, 96, 3170–3174. [Google Scholar] [CrossRef] [PubMed]
- Natri, A.-M.; Salo, P.; Vikstedt, T.; Palssa, A.; Huttunen, M.; Kärkkäinen, M.U.M.; Salovaara, H.; Piironen, V.; Jakobsen, J.; Lamberg-Allardt, C.J. Bread Fortified with Cholecalciferol Increases the Serum 25-Hydroxyvitamin D Concentration in Women as Effectively as a Cholecalciferol Supplement. J. Nutr. 2006, 136, 123–127. [Google Scholar] [CrossRef]
- Clemens, T.L.; Zhouf, X.-Y.; Myles, M.; Endres, D.; Lindsay, R. Serum Vitamin D2 and Vitamin D3 Metabolite Concentrations and Absorption of Vitamin D2 in Elderly Subjects. J. Clin. Endocrinol. Metab. 1986, 63, 656–660. [Google Scholar] [CrossRef]
- Wortsman, J.; Matsuoka, L.Y.; Chen, T.C.; Lu, Z.; Holick, M.F. Decreased Bioavailability of Vitamin D in Obesity. Am. J. Clin. Nutr. 2000, 72, 690–693. [Google Scholar] [CrossRef]
- Farraye, F.A.; Nimitphong, H.; Stucchi, A.; Dendrinos, K.; Boulanger, A.B.; Vijjeswarapu, A.; Tanennbaum, A.; Biancuzzo, R.; Chen, T.C.; Holick, M.F. Use of a Novel Vitamin D Bioavailability Test Demonstrates That Vitamin D Absorption Is Decreased in Patients with Quiescent Crohnʼs Disease. Inflamm. Bowel Dis. 2011, 17, 2116–2121. [Google Scholar] [CrossRef]
- Manousaki, D.; Mitchell, R.; Dudding, T.; Haworth, S.; Harroud, A.; Forgetta, V.; Shah, R.L.; Luan, J.; Langenberg, C.; Timpson, N.J.; et al. Genome-Wide Association Study for Vitamin D Levels Reveals 69 Independent Loci. Am. J. Hum. Genet. 2020, 106, 327–337. [Google Scholar] [CrossRef]
- Fu, L.; Yun, F.; Oczak, M.; Wong, B.Y.L.; Vieth, R.; Cole, D.E.C. Common Genetic Variants of the Vitamin D Binding Protein (DBP) Predict Differences in Response of Serum 25-Hydroxyvitamin D [25(OH)D] to Vitamin D Supplementation. Clin. Biochem. 2009, 42, 1174–1177. [Google Scholar] [CrossRef] [PubMed]
- Sepulveda-Villegas, M.; Elizondo-Montemayor, L.; Trevino, V. Identification and Analysis of 35 Genes Associated with Vitamin D Deficiency: A Systematic Review to Identify Genetic Variants. J. Steroid Biochem. Mol. Biol. 2020, 196, 105516. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Chen, T.C. Vitamin D Deficiency: A Worldwide Problem with Health Consequences. Am. J. Clin. Nutr. 2008, 87, 1080S–1086S. [Google Scholar] [CrossRef] [PubMed]
- Peppelenbosch, M.P.; Fuhler, G.M.; Verhaar, A.P.; van der Laan, L.J.W. Action and Function of Vitamin D in Digestive Tract Physiology and Pathology. Curr. Med. Chem. 2017, 24, 928–936. [Google Scholar] [CrossRef]
- IOM (Institute of Medicine). Dietary Reference Intakes for Calcium and Vitamin D; Ross, A.C., Taylor, C.L., Yaktine, A.L., del Valle, H.B., Eds.; National Academies Press: Washington, DC, USA, 2011; ISBN 978-0-309-16394-1. [Google Scholar]
- Newberry, S.J.; Chung, M.; Shekelle, P.G.; Suttorp Booth, M.; Liu, J.L.; Ruelaz Maher, A.; Motala, A.; Cui, M.; Perry, T.; Shanman, R.; et al. Vitamin D and Calcium: A Systematic Review of Health Outcomes (Update). Evid. Rep. Technol. Assess. (Full Rep.) 2014, 217, 1–929. [Google Scholar] [CrossRef]
- Zhang, R.; Zhang, Y.; Liu, Z.; Pei, Y.; Xu, P.; Chong, W.; Hai, Y.; He, L.; He, Y.; Yu, J.; et al. Association between Vitamin D Supplementation and Cancer Mortality: A Systematic Review and Meta-Analysis. Cancers 2022, 14, 3717. [Google Scholar] [CrossRef]
- Pei, Y.-Y.; Zhang, Y.; Peng, X.-C.; Liu, Z.-R.; Xu, P.; Fang, F. Association of Vitamin D Supplementation with Cardiovascular Events: A Systematic Review and Meta-Analysis. Nutrients 2022, 14, 3158. [Google Scholar] [CrossRef]
- Lo, A.C.Q.; Lo, C.C.W. The Effect of Vitamin D Supplementation on Glycemic Control/Glucose Metabolism and Maternal-Neonatal Outcomes in Women with Established Gestational Diabetes Mellitus: An Updated Meta-Analysis. Clin. Nutr. 2022, 41, 2420–2423. [Google Scholar] [CrossRef]
- Dewansingh, P.; Reckman, G.A.R.; Mijlius, C.F.; Krijnen, W.P.; van der Schans, C.P.; Jager-Wittenaar, H.; van den Heuvel, E.G.H.M. Protein, Calcium, Vitamin D Intake and 25(OH)D Status in Normal Weight, Overweight, and Obese Older Adults: A Systematic Review and Meta-Analysis. Front. Nutr. 2021, 8, 718658. [Google Scholar] [CrossRef]
- Furuie, I.N.; Mauro, M.J.J.; Petruzziello, S.; Riechi, S.C.; Petterle, R.R.; Boguszewski, C.L.; Borba, V.Z.C. Two Threshold Levels of Vitamin D and the Prevalence of Comorbidities in Outpatients of a Tertiary Hospital. Osteoporos. Int. 2018, 29, 433–440. [Google Scholar] [CrossRef]
- Gil, A.; Plaza-Diaz, J.; Mesa, M.D. Vitamin D: Classic and Novel Actions. Ann. Nutr. Metab. 2018, 72, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Huang, X.; Wu, Y.; Li, A.; Tian, Y.; Ren, M.; Li, Z.; Zhang, T.; Wu, H.; Wang, W. Increased Risk of Vitamin D Deficiency Among HIV-Infected Individuals: A Systematic Review and Meta-Analysis. Front. Nutr. 2021, 8, 722032. [Google Scholar] [CrossRef] [PubMed]
- Ghasemian, R.; Shamshirian, A.; Heydari, K.; Malekan, M.; Alizadeh-Navaei, R.; Ebrahimzadeh, M.A.; Ebrahimi Warkiani, M.; Jafarpour, H.; Razavi Bazaz, S.; Rezaei Shahmirzadi, A.; et al. The Role of Vitamin D in the Age of COVID-19: A Systematic Review and Meta-analysis. Int. J. Clin. Pract. 2021, 75, e14675. [Google Scholar] [CrossRef]
- Cucinotta, D.; Vanelli, M. WHO Declares COVID-19 a Pandemic. Acta Biomed. 2020, 91, 157–160. [Google Scholar] [CrossRef]
- Tossetta, G.; Fantone, S.; delli Muti, N.; Balercia, G.; Ciavattini, A.; Giannubilo, S.R.; Marzioni, D. Preeclampsia and Severe Acute Respiratory Syndrome Coronavirus 2 Infection: A Systematic Review. J. Hypertens. 2022, 40, 1629–1638. [Google Scholar] [CrossRef]
- Delli Muti, N.; Finocchi, F.; Tossetta, G.; Salvio, G.; Cutini, M.; Marzioni, D.; Balercia, G. Could SARS-CoV-2 Infection Affect Male Fertility and Sexuality? APMIS 2022, 130, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Dale, L. Neurological Complications of COVID-19: A Review of the Literature. Cureus 2022, 14, e27633. [Google Scholar] [CrossRef]
- Ragab, D.; Salah Eldin, H.; Taeimah, M.; Khattab, R.; Salem, R. The COVID-19 Cytokine Storm; What We Know So Far. Front. Immunol. 2020, 11, 1446. [Google Scholar] [CrossRef]
- Clerbaux, L.-A.; Albertini, M.C.; Amigó, N.; Beronius, A.; Bezemer, G.F.G.; Coecke, S.; Daskalopoulos, E.P.; del Giudice, G.; Greco, D.; Grenga, L.; et al. Factors Modulating COVID-19: A Mechanistic Understanding Based on the Adverse Outcome Pathway Framework. J. Clin. Med. 2022, 11, 4464. [Google Scholar] [CrossRef]
- Grimaudo, S.; Amodio, E.; Pipitone, R.M.; Maida, C.M.; Pizzo, S.; Prestileo, T.; Tramuto, F.; Sardina, D.; Vitale, F.; Casuccio, A.; et al. PNPLA3 and TLL-1 Polymorphisms as Potential Predictors of Disease Severity in Patients With COVID-19. Front. Cell Dev. Biol. 2021, 9, 627914. [Google Scholar] [CrossRef]
- Gupta, K.; Kaur, G.; Pathak, T.; Banerjee, I. Systematic Review and Meta-Analysis of Human Genetic Variants Contributing to COVID-19 Susceptibility and Severity. Gene 2022, 844, 146790. [Google Scholar] [CrossRef]
- van der Made, C.I.; Netea, M.G.; van der Veerdonk, F.L.; Hoischen, A. Clinical Implications of Host Genetic Variation and Susceptibility to Severe or Critical COVID-19. Genome Med. 2022, 14, 96. [Google Scholar] [CrossRef] [PubMed]
- Ejaz, H.; Alsrhani, A.; Zafar, A.; Javed, H.; Junaid, K.; Abdalla, A.E.; Abosalif, K.O.A.; Ahmed, Z.; Younas, S. COVID-19 and Comorbidities: Deleterious Impact on Infected Patients. J. Infect. Public Health 2020, 13, 1833–1839. [Google Scholar] [CrossRef]
- Bigdelou, B.; Sepand, M.R.; Najafikhoshnoo, S.; Negrete, J.A.T.; Sharaf, M.; Ho, J.Q.; Sullivan, I.; Chauhan, P.; Etter, M.; Shekarian, T.; et al. COVID-19 and Preexisting Comorbidities: Risks, Synergies, and Clinical Outcomes. Front. Immunol. 2022, 13, 890517. [Google Scholar] [CrossRef]
- Tian, Y.; Qiu, X.; Wang, C.; Zhao, J.; Jiang, X.; Niu, W.; Huang, J.; Zhang, F. Cancer Associates with Risk and Severe Events of COVID-19: A Systematic Review and Meta-Analysis. Int. J. Cancer 2021, 148, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Ticinesi, A.; Parise, A.; Cerundolo, N.; Nouvenne, A.; Prati, B.; Chiussi, G.; Guerra, A.; Meschi, T. Multimorbidity and Frailty Are the Key Characteristics of Patients Hospitalized with COVID-19 Breakthrough Infection during Delta Variant Predominance in Italy: A Retrospective Study. J. Clin. Med. 2022, 11, 5442. [Google Scholar] [CrossRef] [PubMed]
- Ahlström, B.; Frithiof, R.; Larsson, I.-M.; Strandberg, G.; Lipcsey, M.; Hultström, M. A Comparison of Impact of Comorbidities and Demographics on 60-Day Mortality in ICU Patients with COVID-19, Sepsis and Acute Respiratory Distress Syndrome. Sci. Rep. 2022, 12, 15703. [Google Scholar] [CrossRef]
- Mishra, P.; Parveen, R.; Bajpai, R.; Agarwal, N. Vitamin D Deficiency and Comorbidities as Risk Factors of COVID-19 Infection: A Systematic Review and Meta-Analysis. J. Prev. Med. Public Health 2022, 55, 321–333. [Google Scholar] [CrossRef]
- Cereda, E.; Bogliolo, L.; Klersy, C.; Lobascio, F.; Masi, S.; Crotti, S.; de Stefano, L.; Bruno, R.; Corsico, A.G.; di Sabatino, A.; et al. Vitamin D 25OH Deficiency in COVID-19 Patients Admitted to a Tertiary Referral Hospital. Clin. Nutr. 2021, 40, 2469–2472. [Google Scholar] [CrossRef]
- Jude, E.B.; Ling, S.F.; Allcock, R.; Yeap, B.X.Y.; Pappachan, J.M. Vitamin D Deficiency Is Associated With Higher Hospitalization Risk From COVID-19: A Retrospective Case-Control Study. J. Clin. Endocrinol. Metab. 2021, 106, e4708–e4715. [Google Scholar] [CrossRef]
- Feng, S.; Song, F.; Guo, W.; Tan, J.; Zhang, X.; Qiao, F.; Guo, J.; Zhang, L.; Jia, X. Potential Genes Associated with COVID-19 and Comorbidity. Int. J. Med. Sci. 2022, 19, 402–415. [Google Scholar] [CrossRef] [PubMed]
- Pe’er, I.; Yelensky, R.; Altshuler, D.; Daly, M.J. Estimation of the Multiple Testing Burden for Genomewide Association Studies of Nearly All Common Variants. Genet. Epidemiol. 2008, 32, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, N.; Nishida, N.; Yamamoto, R.; Gojobori, T.; Shimotohno, K.; Mizokami, M.; Ariumi, Y. Angiotensin-Converting Enzyme (ACE) 1 Gene Polymorphism and Phenotypic Expression of COVID-19 Symptoms. Genes 2021, 12, 1572. [Google Scholar] [CrossRef] [PubMed]
- Arnett, D.K.; Davis, B.R.; Ford, C.E.; Boerwinkle, E.; Leiendecker-Foster, C.; Miller, M.B.; Black, H.; Eckfeldt, J.H. Pharmacogenetic Association of the Angiotensin-Converting Enzyme Insertion/Deletion Polymorphism on Blood Pressure and Cardiovascular Risk in Relation to Antihypertensive Treatment. Circulation 2005, 111, 3374–3383. [Google Scholar] [CrossRef] [PubMed]
- Saengsiwaritt, W.; Jittikoon, J.; Chaikledkaew, U.; Udomsinprasert, W. Genetic Polymorphisms of ACE1, ACE2, and TMPRSS2 Associated with COVID-19 Severity: A Systematic Review with Meta-Analysis. Rev. Med. Virol. 2022, 32, e2323. [Google Scholar] [CrossRef]
- Yamamoto, N.; Yamamoto, R.; Ariumi, Y.; Mizokami, M.; Shimotohno, K.; Yoshikura, H. Does Genetic Predisposition Contribute to the Exacerbation of COVID-19 Symptoms in Individuals with Comorbidities and Explain the Huge Mortality Disparity between the East and the West? Int. J. Mol. Sci. 2021, 22, 5000. [Google Scholar] [CrossRef]
- Khoo, S.K.; Hurst, T.; Webb, M.J.; Dickie, G.; Kearsley, J.; Parsons, P.G.; Mackay, E.V. Cisplatin Chemotherapy of Ovarian Cancer: Is Short-Term In Vitro Chemosensitivity Predictive of Long-Term Patient Survival? Aust. N. Z. J. Obstet. Gynaecol. 1988, 28, 313–317. [Google Scholar] [CrossRef]
- Li, Y.C. Vitamin D Regulation of the Renin–Angiotensin System. J. Cell. Biochem. 2003, 88, 327–331. [Google Scholar] [CrossRef]
- Hashemi, S.M.A.; Thijssen, M.; Hosseini, S.Y.; Tabarraei, A.; Pourkarim, M.R.; Sarvari, J. Human Gene Polymorphisms and Their Possible Impact on the Clinical Outcome of SARS-CoV-2 Infection. Arch. Virol. 2021, 166, 2089–2108. [Google Scholar] [CrossRef]
- Aranow, C. Vitamin D and the Immune System. J. Investig. Med. 2011, 59, 881–886. [Google Scholar] [CrossRef]
- Malek Mahdavi, A. A Brief Review of Interplay between Vitamin D and Angiotensin-Converting Enzyme 2: Implications for a Potential Treatment for COVID-19. Rev. Med. Virol. 2020, 30, e2119. [Google Scholar] [CrossRef] [PubMed]
- Datta, P.K.; Liu, F.; Fischer, T.; Rappaport, J.; Qin, X. SARS-CoV-2 Pandemic and Research Gaps: Understanding SARS-CoV-2 Interaction with the ACE2 Receptor and Implications for Therapy. Theranostics 2020, 10, 7448–7464. [Google Scholar] [CrossRef] [PubMed]
- Migliorini, F.; Vaishya, R.; Eschweiler, J.; Oliva, F.; Hildebrand, F.; Maffulli, N. Vitamins C and D and COVID-19 Susceptibility, Severity and Progression: An Evidence Based Systematic Review. Medicina 2022, 58, 941. [Google Scholar] [CrossRef]
- D’Ecclesiis, O.; Gavioli, C.; Martinoli, C.; Raimondi, S.; Chiocca, S.; Miccolo, C.; Bossi, P.; Cortinovis, D.; Chiaradonna, F.; Palorini, R.; et al. Vitamin D and SARS-CoV2 Infection, Severity and Mortality: A Systematic Review and Meta-Analysis. PLoS ONE 2022, 17, e0268396. [Google Scholar] [CrossRef]
- Bassatne, A.; Basbous, M.; Chakhtoura, M.; el Zein, O.; Rahme, M.; El-Hajj Fuleihan, G. The Link between COVID-19 and VItamin D (VIVID): A Systematic Review and Meta-Analysis. Metabolism 2021, 119, 154753. [Google Scholar] [CrossRef] [PubMed]
- Sharif-Askari, F.S.; Hafezi, S.; Sharif-Askari, N.S.; Alsayed, H.A.H.; Mdkhana, B.; Selvakumar, B.; Temsah, M.-H.; Saddik, B.; al Anouti, F.; Halwani, R. Vitamin D Modulates Systemic Inflammation in Patients with Severe COVID-19. Life Sci. 2022, 307, 120909. [Google Scholar] [CrossRef]
- Kraja, A.T.; Vaidya, D.; Pankow, J.S.; Goodarzi, M.O.; Assimes, T.L.; Kullo, I.J.; Sovio, U.; Mathias, R.A.; Sun, Y.V.; Franceschini, N.; et al. A Bivariate Genome-Wide Approach to Metabolic Syndrome: STAMPEED Consortium. Diabetes 2011, 60, 1329–1339. [Google Scholar] [CrossRef]
- Hidalgo-Bravo, A.; Rivera-Paredez, B.; León-Reyes, G.; Patiño, N.; Castillejos-López, M.; Salmerón, J.; Velázquez-Cruz, R. Unravelling the Contribution of the Rs7041 and Rs4588 Polymorphisms of the GC Gene and Serum VDBP Levels for Developing Metabolic Syndrome in the Mexican Population. Int. J. Mol. Sci. 2022, 23, 10581. [Google Scholar] [CrossRef]
- Zhao, T.; Zhang, D.; Liu, Y.; Feng, M.; Xu, Z.; Huang, H.; Zhang, L.; Li, W.; Li, X. The Association Between GC Gene Polymorphisms and Metabolic Syndrome in Chinese Rural Population: A Case-Control Study. Diabetes Metab. Syndr. Obes. 2022, 15, 165–174. [Google Scholar] [CrossRef]
- Ruiz-Ojeda, F.J.; Anguita-Ruiz, A.; Leis, R.; Aguilera, C.M. Genetic Factors and Molecular Mechanisms of Vitamin D and Obesity Relationship. Ann. Nutr. Metab. 2018, 73, 89–99. [Google Scholar] [CrossRef]
- Hsieh, M.-C.; Hsiao, P.-J.; Liao, M.-T.; Hou, Y.-C.; Chang, Y.-C.; Chiang, W.-F.; Wu, K.-L.; Chan, J.-S.; Lu, K.-C. The Role of Vitamin D in SARS-CoV-2 Infection and Acute Kidney Injury. Int. J. Mol. Sci. 2022, 23, 7368. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, T.; Gupta, G.K.; Agrawal, D.K. Vitamin D Deficiency Decreases the Expression of VDR and Prohibitin in the Lungs of Mice with Allergic Airway Inflammation. Exp. Mol. Pathol. 2012, 93, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Kere, J. Genetics of Complex Disorders. Biochem. Biophys. Res. Commun. 2010, 396, 143–146. [Google Scholar] [CrossRef] [PubMed]
- Buniello, A.; MacArthur, J.A.L.; Cerezo, M.; Harris, L.W.; Hayhurst, J.; Malangone, C.; McMahon, A.; Morales, J.; Mountjoy, E.; Sollis, E.; et al. The NHGRI-EBI GWAS Catalog of Published Genome-Wide Association Studies, Targeted Arrays and Summary Statistics 2019. Nucleic Acids Res. 2019, 47, D1005–D1012. [Google Scholar] [CrossRef] [PubMed]
- Kamat, M.A.; Blackshaw, J.A.; Young, R.; Surendran, P.; Burgess, S.; Danesh, J.; Butterworth, A.S.; Staley, J.R. PhenoScanner V2: An Expanded Tool for Searching Human Genotype-Phenotype Associations. Bioinformatics 2019, 35, 4851–4853. [Google Scholar] [CrossRef]
- Staley, J.R.; Blackshaw, J.; Kamat, M.A.; Ellis, S.; Surendran, P.; Sun, B.B.; Paul, D.S.; Freitag, D.; Burgess, S.; Danesh, J.; et al. PhenoScanner: A Database of Human Genotype-Phenotype Associations. Bioinformatics 2016, 32, 3207–3209. [Google Scholar] [CrossRef]
- Cunningham, F.; Allen, J.E.; Allen, J.; Alvarez-Jarreta, J.; Amode, M.R.; Armean, I.M.; Austine-Orimoloye, O.; Azov, A.G.; Barnes, I.; Bennett, R.; et al. Ensembl 2022. Nucleic Acids Res. 2022, 50, D988–D995. [Google Scholar] [CrossRef]
- Howe, K.L.; Achuthan, P.; Allen, J.; Allen, J.; Alvarez-Jarreta, J.; Amode, M.R.; Armean, I.M.; Azov, A.G.; Bennett, R.; Bhai, J.; et al. Ensembl 2021. Nucleic Acids Res. 2021, 49, D884–D891. [Google Scholar] [CrossRef]
- Borg, J.; Georgitsi, M.; Aleporou-Marinou, V.; Kollia, P.; Patrinos, G.P. Genetic Recombination as a Major Cause of Mutagenesis in the Human Globin Gene Clusters. Clin. Biochem. 2009, 42, 1839–1850. [Google Scholar] [CrossRef]
- Machiela, M.J.; Chanock, S.J. LDlink: A Web-Based Application for Exploring Population-Specific Haplotype Structure and Linking Correlated Alleles of Possible Functional Variants. Bioinformatics 2015, 31, 3555–3557. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. 2020. Available online: https://www.scirp.org/%28S%28351jmbntvnsjt1aadkposzje%29%29/reference/referencespapers.aspx?referenceid=2903185 (accessed on 3 November 2022).
- Sherman, B.T.; Hao, M.; Qiu, J.; Jiao, X.; Baseler, M.W.; Lane, H.C.; Imamichi, T.; Chang, W. DAVID: A Web Server for Functional Enrichment Analysis and Functional Annotation of Gene Lists (2021 Update). Nucleic Acids Res. 2022, 50, W216–W221. [Google Scholar] [CrossRef] [PubMed]
- GTEx Consortium. Genetic Effects on Gene Expression across Human Tissues. Nature 2017, 550, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.; Taskesen, E.; van Bochoven, A.; Posthuma, D. Functional Mapping and Annotation of Genetic Associations with FUMA. Nat. Commun. 2017, 8, 1826. [Google Scholar] [CrossRef] [PubMed]
- Supek, F.; Bošnjak, M.; Škunca, N.; Šmuc, T. REVIGO Summarizes and Visualizes Long Lists of Gene Ontology Terms. PLoS ONE 2011, 6, e21800. [Google Scholar] [CrossRef]
- Yu, G.; Wang, L.-G.; Han, Y.; He, Q.-Y. ClusterProfiler: An R Package for Comparing Biological Themes Among Gene Clusters. OMICS 2012, 16, 284–287. [Google Scholar] [CrossRef]
- Wu, T.; Hu, E.; Xu, S.; Chen, M.; Guo, P.; Dai, Z.; Feng, T.; Zhou, L.; Tang, W.; Zhan, L.; et al. ClusterProfiler 4.0: A Universal Enrichment Tool for Interpreting Omics Data. Innovation 2021, 2, 100141. [Google Scholar] [CrossRef]
- Piñero, J.; Ramírez-Anguita, J.M.; Saüch-Pitarch, J.; Ronzano, F.; Centeno, E.; Sanz, F.; Furlong, L.I. The DisGeNET Knowledge Platform for Disease Genomics: 2019 Update. Nucleic Acids Res. 2020, 48, D845–D855. [Google Scholar] [CrossRef] [PubMed]
- Piñero, J.; Bravo, À.; Queralt-Rosinach, N.; Gutiérrez-Sacristán, A.; Deu-Pons, J.; Centeno, E.; García-García, J.; Sanz, F.; Furlong, L.I. DisGeNET: A Comprehensive Platform Integrating Information on Human Disease-Associated Genes and Variants. Nucleic Acids Res. 2017, 45, D833–D839. [Google Scholar] [CrossRef]
- Piñero, J.; Queralt-Rosinach, N.; Bravo, À.; Deu-Pons, J.; Bauer-Mehren, A.; Baron, M.; Sanz, F.; Furlong, L.I. DisGeNET: A Discovery Platform for the Dynamical Exploration of Human Diseases and Their Genes. Database 2015, 2015, bav028. [Google Scholar] [CrossRef]
- Glueck, D.H.; Mandel, J.; Karimpour-Fard, A.; Hunter, L.; Muller, K.E. Exact Calculations of Average Power for the Benjamini-Hochberg Procedure. Int. J. Biostat. 2008, 4, 11. [Google Scholar] [CrossRef] [PubMed]
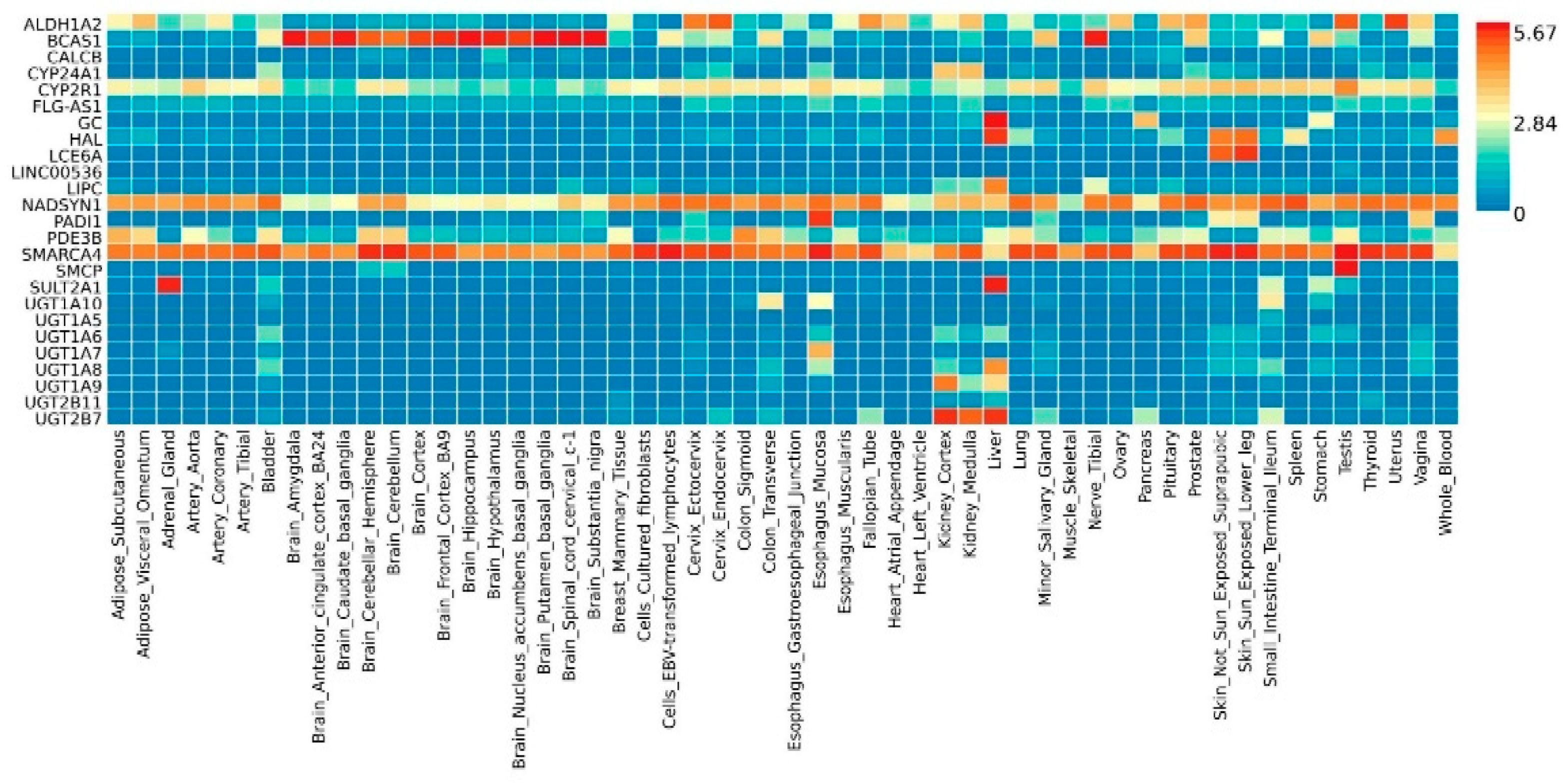
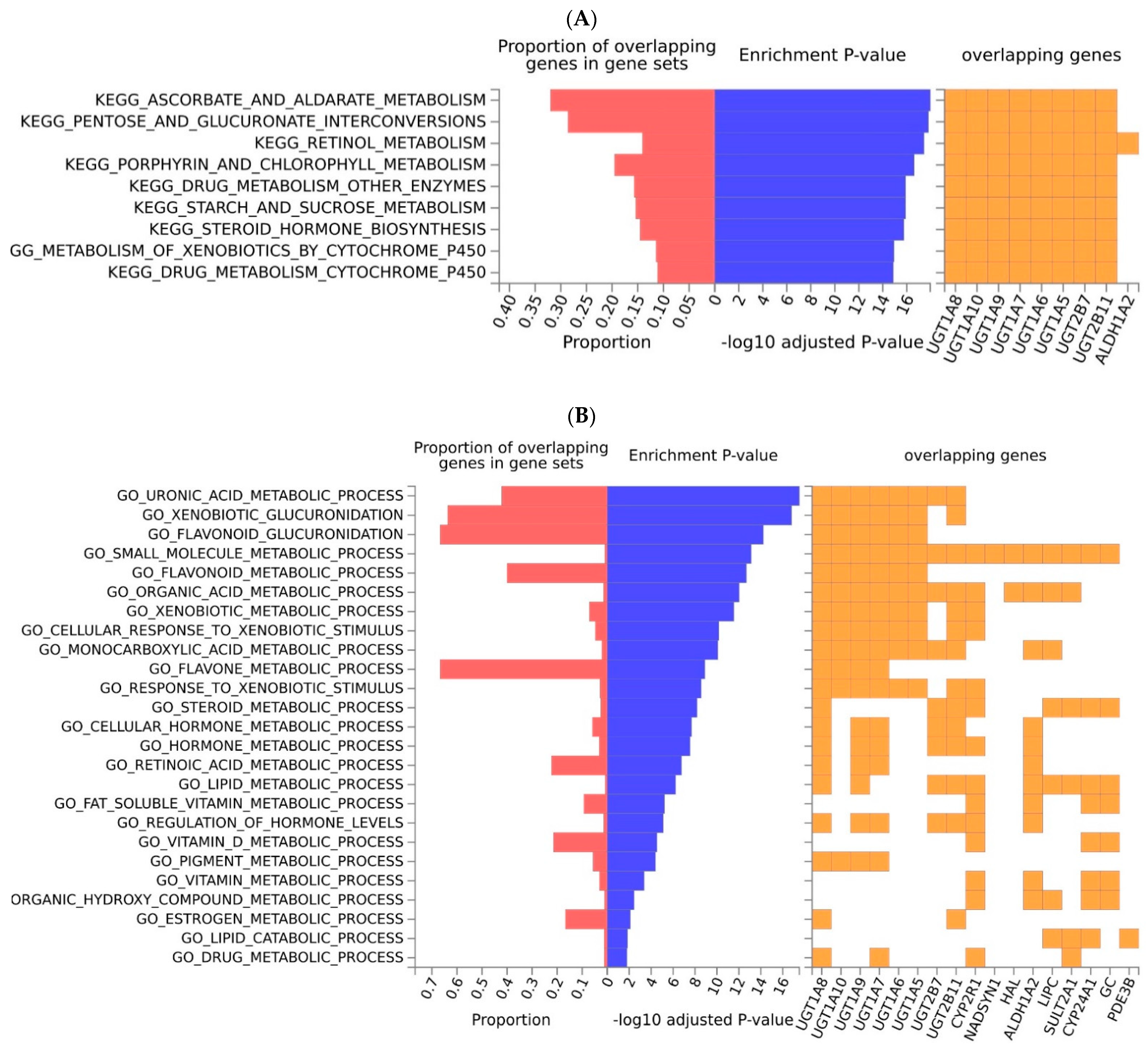


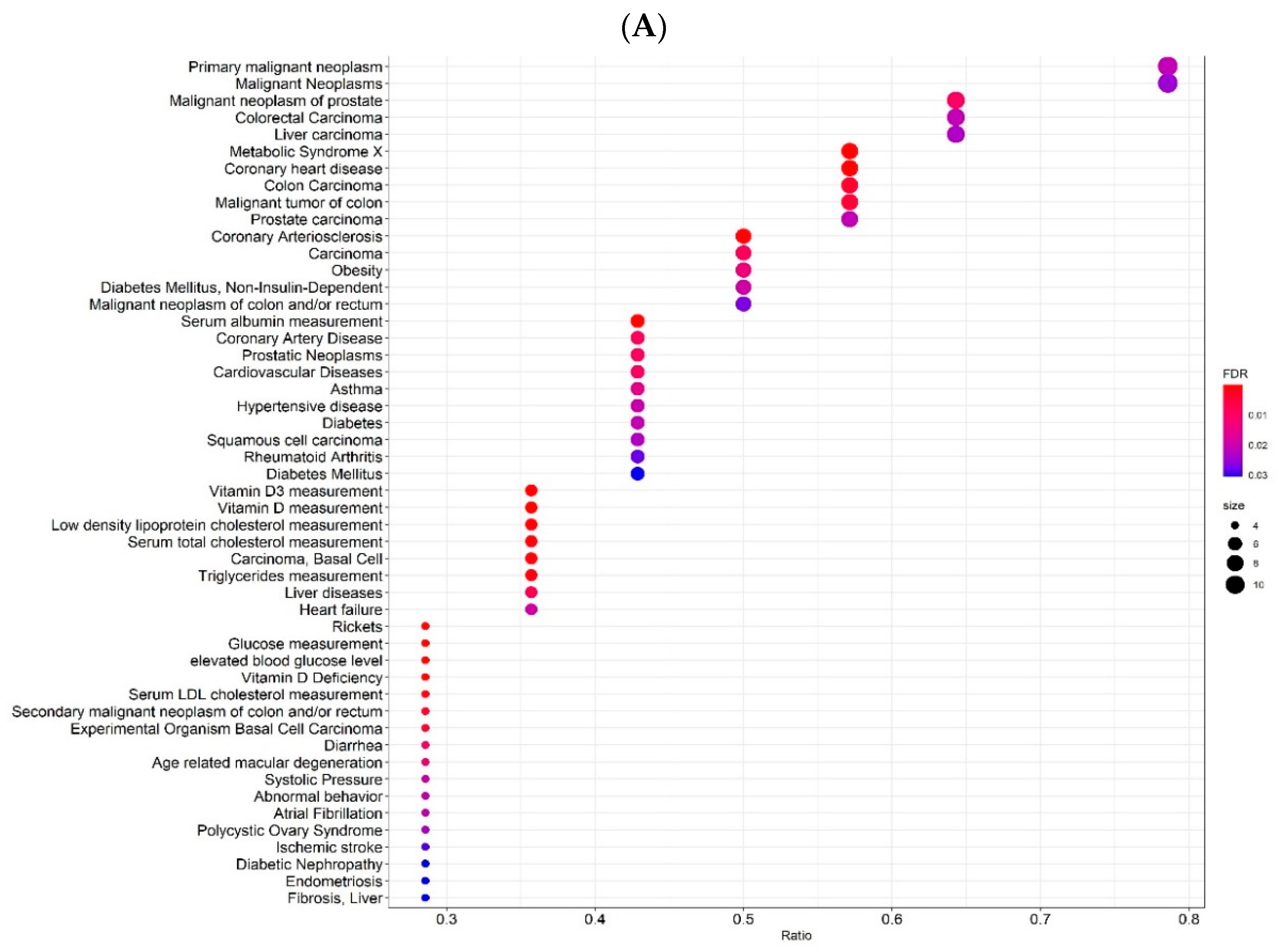
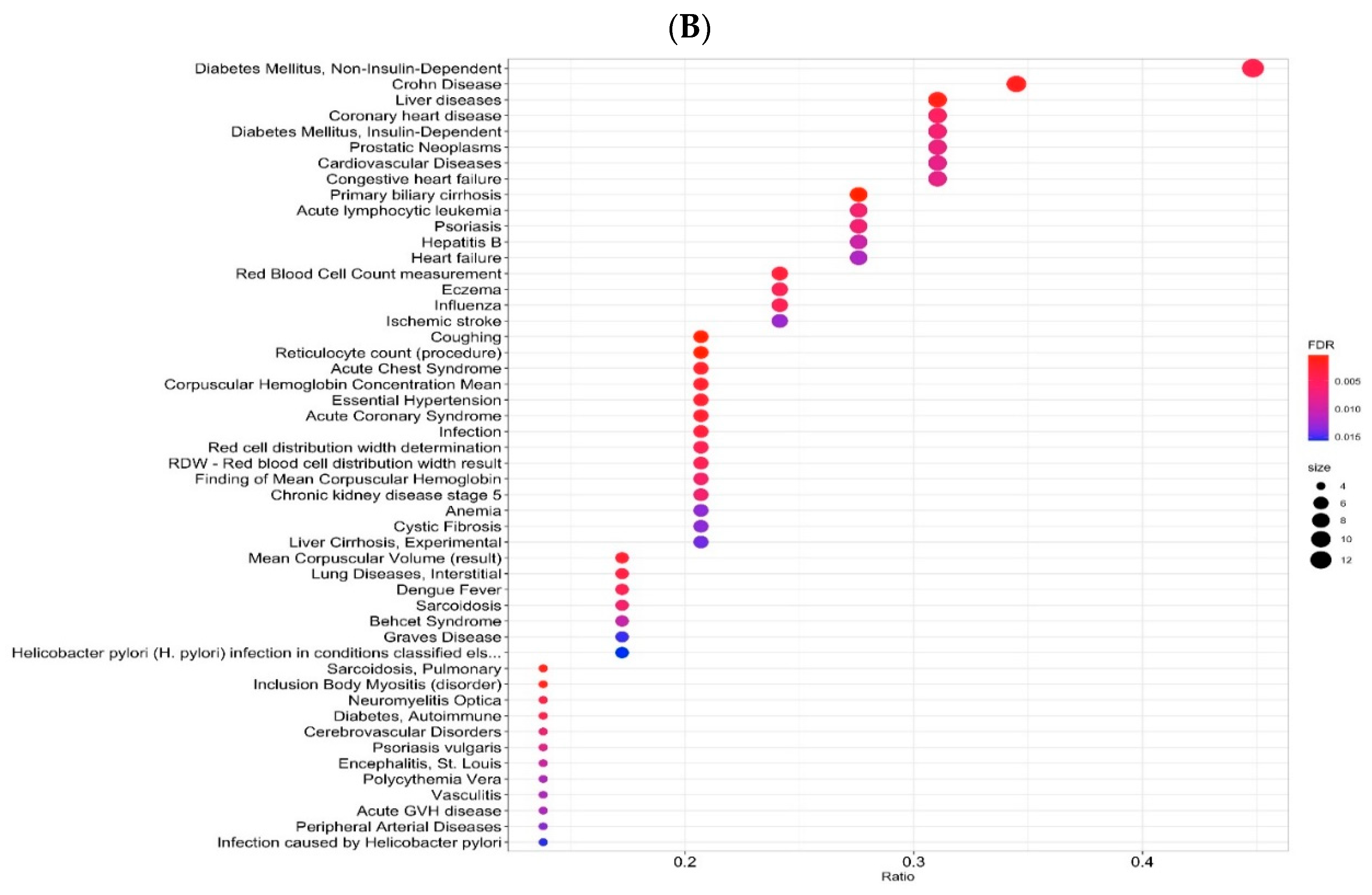

| Variant | Risk Allele | Gene | Location (Chr: Position) | p-Value | Study Accession | PubMed ID |
|---|---|---|---|---|---|---|
| rs3750297 | A | PADI1 | 1:17233181 | 3 × 10−10 | GCST90020244 | 34308111 |
| rs12123821 | T | FLG-AS1 | 1:152206676 | 6 × 10−26 | ||
| rs4845491 | C | SMCP, LCE6A | 1:152877093 | 7 × 10−10 | ||
| rs3755322 | G | UGT1A9, UGT1A5, UGT1A10, UGT1A7, UGT1A8, UGT1A6 | 2:233713141 | 6 × 10−12 | ||
| rs6600893 | C | UGT2B11, UGT2B7 | 4:69113183 | 5 × 10−15 | ||
| rs2282679 | G | GC | 4:71742666 | 1 × 10−200 | ||
| rs2205262 | C | LINC00536 | 8:115999659 | 5 × 10−11 | ||
| rs7129781 | C | CYP2R1 | 11:14890871 | 4 × 10−33 | ||
| rs4944958 | A | NADSYN1 | 11:71457027 | 9 × 10−143 | ||
| rs964184 | G | ZPR1 | 11:116778201 | 3 × 10−14 | ||
| rs10859995 | T | HAL | 12:95981904 | 3 × 10−33 | ||
| rs1800588 | T | LIPC, ALDH1A2 | 15:58431476 | 2 × 10−10 | ||
| rs55791371 | C | SMARCA4 | 19:11077477 | 1 × 10−9 | ||
| rs10426201 | G | SULT2A1 | 19:47881492 | 8 × 10−20 | ||
| rs17217119 | G | BCAS1, CYP24A1 | 20:54126051 | 5 × 10−16 | ||
| rs11723621 | G | GC | 4:71749645 | 2 × 10−24 | GCST90101732 | 34852423 |
| rs7041 | G | GC | 4:71752617 | 2 × 10−9 | ||
| rs11023332 | G | PDE3B | 11:14762564 | 3 × 10−11 | ||
| rs12785878 | G | NADSYN1 | 11:71456403 | 2 × 10−27 | GCST000697 | 20541252 |
| rs10741657 | G | CALCB, CYP2R1 | 11:14893332 | 3 × 10−20 |
| Description | FDR | Ratio | BgRatio |
|---|---|---|---|
| Vitamin D3 measurement | 5.029 × 10−11 | 5/14 | 14/21,666 |
| Vitamin D measurement | 5.029 × 10−11 | 5/14 | 14/21,666 |
| Rickets | 2.933 × 10−5 | 4/14 | 72/21,666 |
| Metabolic Syndrome X | 2.933 × 10−5 | 8/14 | 1125/21,666 |
| Serum albumin measurement | 3.233 × 10−5 | 6/14 | 433/21,666 |
| Glucose measurement | 3.689 × 10−5 | 4/14 | 89/21,666 |
| Elevated blood glucose level | 3.689 × 10−5 | 4/14 | 89/21,666 |
| High density lipoprotein cholesterol level quantitative trait locus 12 | 4.847 × 10−5 | 2/14 | 2/21,666 |
| Coronary heart disease | 1.732 × 10−4 | 8/14 | 1576/21,666 |
| Vitamin D Deficiency | 2.267 × 10−4 | 4/14 | 153/21,666 |
| Description | FDR | Ratio | BgRatio |
|---|---|---|---|
| Primary biliary cirrhosis | 3.305 × 10−4 | 8/29 | 478/21,666 |
| Coughing | 4.286 × 10−4 | 6/29 | 235/21,666 |
| Reticulocyte count (procedure) | 4.286 × 10−4 | 6/29 | 234/21,666 |
| Eosinophilia-Myalgia Syndrome, L-Tryptophan-Related | 9.429 × 10−4 | 2/29 | 2/21,666 |
| Liver diseases | 1.163 × 10−3 | 9/29 | 1019/21,666 |
| Rales | 1.163 × 10−3 | 3/29 | 23/21,666 |
| Sarcoidosis, Pulmonary | 1.163 × 10−3 | 4/29 | 81/21,666 |
| Inclusion Body Myositis (disorder) | 1.163 × 10−3 | 4/29 | 87/21,666 |
| Juvenile Graves disease | 1.163 × 10−3 | 2/29 | 3/21,666 |
| Podoconiosis | 1.163 × 10−3 | 2/29 | 3/21,666 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alcalá-Santiago, Á.; Rodríguez-Barranco, M.; Rava, M.; Jiménez-Sousa, M.Á.; Gil, Á.; Sánchez, M.J.; Molina-Montes, E. Vitamin D Deficiency and COVID-19: A Biological Database Study on Pathways and Gene-Disease Associations. Int. J. Mol. Sci. 2022, 23, 14256. https://doi.org/10.3390/ijms232214256
Alcalá-Santiago Á, Rodríguez-Barranco M, Rava M, Jiménez-Sousa MÁ, Gil Á, Sánchez MJ, Molina-Montes E. Vitamin D Deficiency and COVID-19: A Biological Database Study on Pathways and Gene-Disease Associations. International Journal of Molecular Sciences. 2022; 23(22):14256. https://doi.org/10.3390/ijms232214256
Chicago/Turabian StyleAlcalá-Santiago, Ángela, Miguel Rodríguez-Barranco, Marta Rava, María Ángeles Jiménez-Sousa, Ángel Gil, María José Sánchez, and Esther Molina-Montes. 2022. "Vitamin D Deficiency and COVID-19: A Biological Database Study on Pathways and Gene-Disease Associations" International Journal of Molecular Sciences 23, no. 22: 14256. https://doi.org/10.3390/ijms232214256
APA StyleAlcalá-Santiago, Á., Rodríguez-Barranco, M., Rava, M., Jiménez-Sousa, M. Á., Gil, Á., Sánchez, M. J., & Molina-Montes, E. (2022). Vitamin D Deficiency and COVID-19: A Biological Database Study on Pathways and Gene-Disease Associations. International Journal of Molecular Sciences, 23(22), 14256. https://doi.org/10.3390/ijms232214256







