Involvement of circRNAs in the Development of Heart Failure
Abstract
1. Introduction
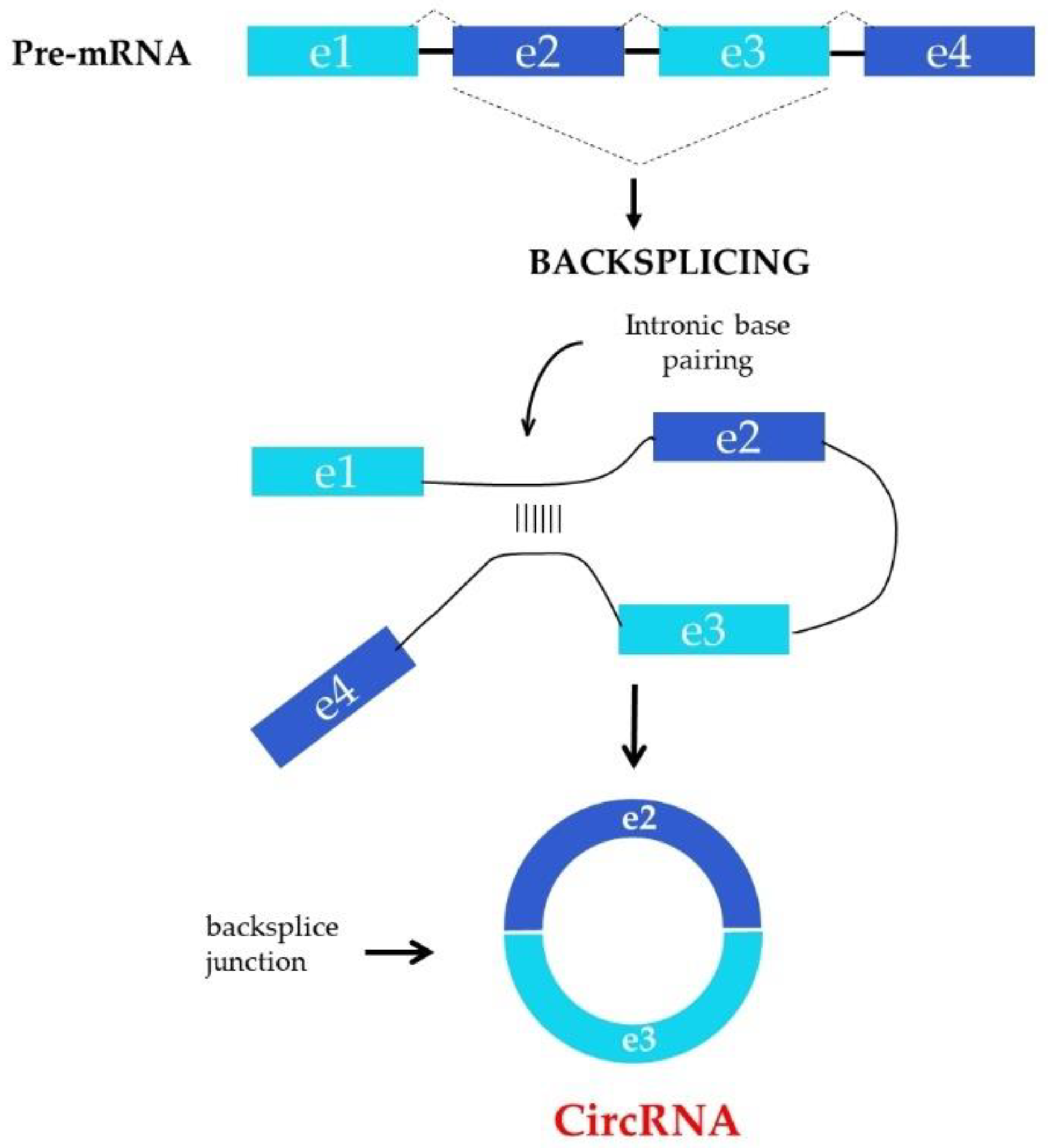
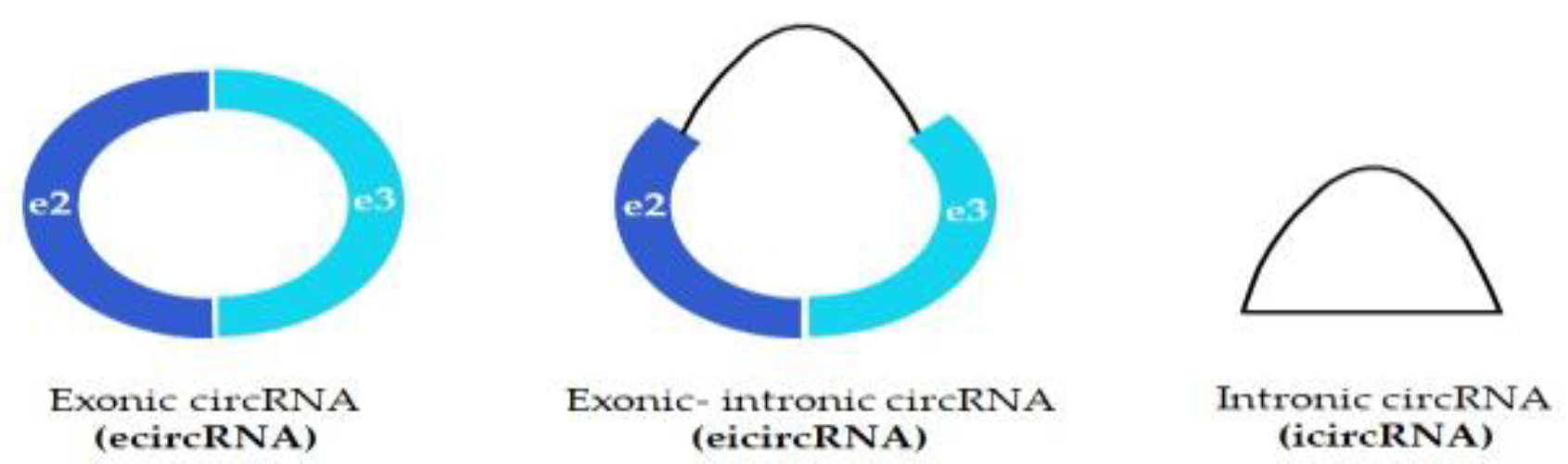
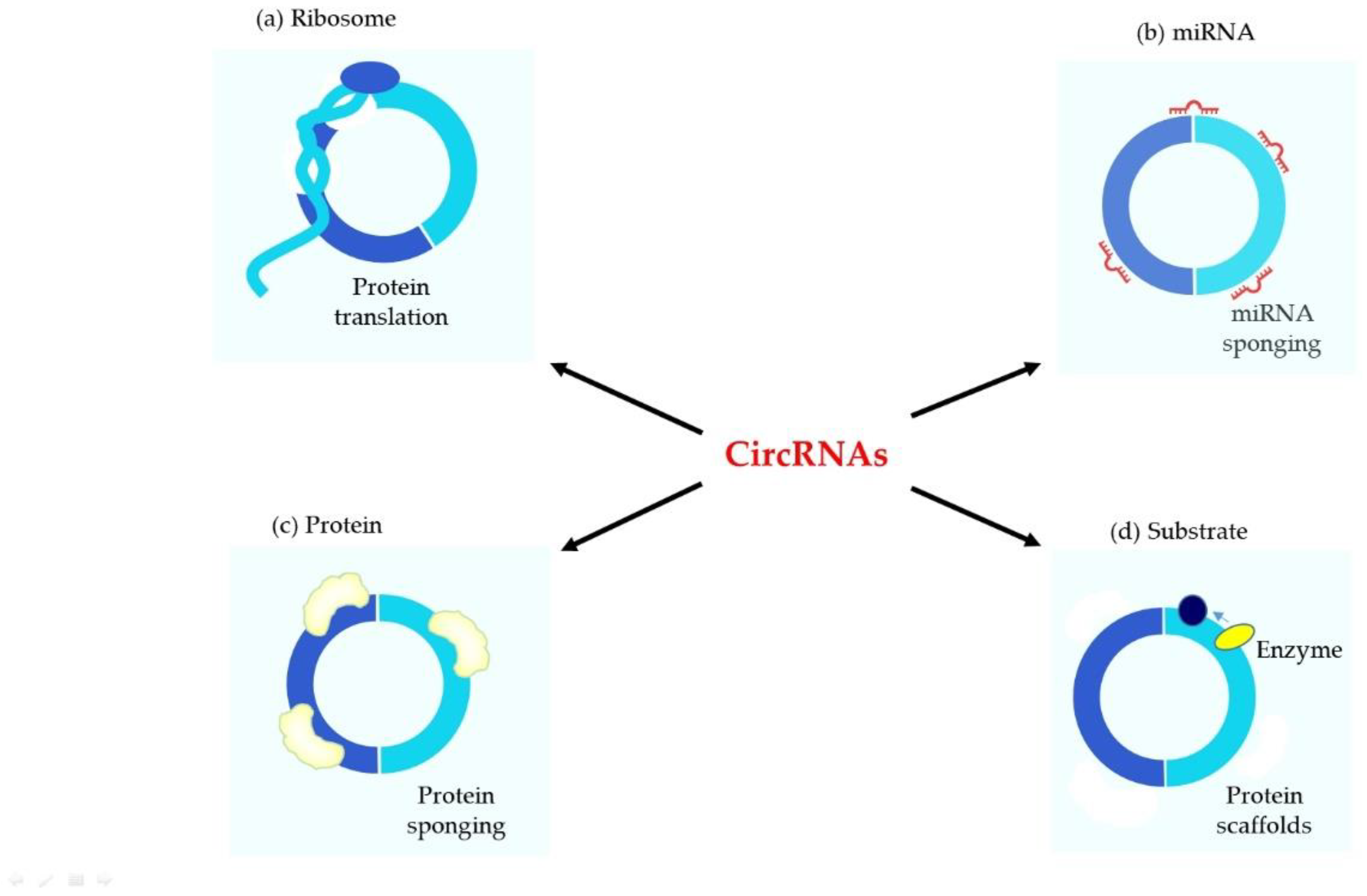
2. Biogenesis and Functions of circRNAs
3. CircRNAs in the Development of Heart Failure
3.1. Myocardial Infarction
3.2. Diabetic Cardiomyopathy
3.3. Cardiac Hypertrophy
3.4. Cardiac Fibrosis
3.5. Autophagy and Apoptosis
4. Conclusions
5. Future Directions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- ENCODE Project Consortium. An integrated encyclopedia of DNA elements in the human genome. Nature 2012, 489, 57–74. [Google Scholar] [CrossRef] [PubMed]
- Barrett, S.P.; Salzman, J. Circular RNAs: Analysis, expression and potential functions. Development 2016, 143, 1838–1847. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Han, P.; Zhou, T.; Guo, X.; Song, X.; Li, Y. circRNADb: A comprehensive database for human circular RNAs with protein-coding annotations. Sci. Rep. 2016, 6, 34985. [Google Scholar] [CrossRef] [PubMed]
- Townsend, N.; Wilson, L.; Bhatnagar, P.; Wickramasinghe, K.; Rayner, M.; Nichols, M. Cardiovascular disease in Europe: Epidemiological update. Eur. Heart J. 2016, 37, 3232–3245. [Google Scholar] [CrossRef]
- Beermann, J.; Piccoli, M.T.; Viereck, J.; Thum, T. Non-coding RNAs in development and disease: Background, mechanisms, and therapeutic approaches. Physiol. Rev. 2016, 96, 1297–1325. [Google Scholar] [CrossRef]
- Goretti, E.; Wagner, D.R.; Devaux, Y. miRNAs as biomarkers of myocardial infarction: A step forward towards personalized medicine? Trends Mol. Med. 2014, 20, 716–725. [Google Scholar] [CrossRef]
- Sygitowicz, G.; Maciejak-Jastrzębska, A.; Sitkiewicz, D. MicroRNAs in the development of left ventricular remodeling and postmyocardial infarction heart failure. Pol. Arch. Intern Med. 2020, 120, 59–65. [Google Scholar]
- Gomes, C.P.C.; Spencer, H.; Ford, K.L.; Michel, L.Y.M.; Baker, A.H.; Emanueli, C.; Balligand, J.L.; Devaux, Y. Cardiolinc network. The function and therapeutic potential of long non-coding RNAs in cardiovascular development and disease. Mol. Ther. Nucleic Acids 2017, 8, 494–507. [Google Scholar] [CrossRef]
- Greco, S.; Somoza, A.S.; Devaux, Y.; Martelli, F. Long noncoding RNAs and cardiac disease. Antioxid. Redox. Signal. 2018, 29, 880–901. [Google Scholar] [CrossRef]
- Zhang, X.O.; Wang, H.B.; Zhang, Y.; Lu, X.; Chen, L.L.; Yang, L. Complementary sequence-mediated exon circularization. Cell 2014, 159, 134–147. [Google Scholar] [CrossRef]
- Conn, S.J.; Pillman, K.A.; Toubia, J.; Conn, V.M.; Salmanidis, M.; Phillips, C.A.; Roslan, S.; Schreiber, A.W.; Gregory, P.A.; Goodall, G.J. The RNA binding protein quaking regulates formation of circRNAs. Cell 2015, 160, 1125–1134. [Google Scholar] [CrossRef] [PubMed]
- Barrett, S.P.; Wang, P.L.; Salzman, J. Circular RNA biogenesis can proceed through an exon-containing lariat precursor. eLife 2015, 4, e07540. [Google Scholar] [CrossRef] [PubMed]
- Kelly, S.; Greenman, C.; Cook, P.R. A Papantonis. Exon skipping is correlated with exon circularization. J. Mol. Biol. 2015, 427, 2414–2417. [Google Scholar] [CrossRef] [PubMed]
- Monat, C.; Quiroga, C.; Laroche-Johnston, F.; Cousineau, B. The Ll.LtrB intron from Lactococcus lactis excises as circles in vivo: Insights into the group II intron circularization pathway. RNA 2015, 21, 1286–1293. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Huang, C.; Bao, C.; Chen, L.; Lin, M.; Wang, X.; Zhong, G.; Yu, B.; Hu, W.; Dai, L. Exon-intron circular RNAs regulate transcription in the nucleus. Nat. Struct. Mol. Biol. 2015, 22, 256–264. [Google Scholar] [CrossRef]
- Werfel, S.; Nothjunge, S.; Schwarzmayr, T.; Strom, T.M.; Meitinger, T.; Engelhardt, S. Characterization of circular RNAs in human, mouse and rat hearts. J. Mol. Cell Cardiol. 2016, 98, 103–107. [Google Scholar] [CrossRef]
- Lu, Q.; Liu, T.; Feng, H.; Yang, R.; Zhao, X.; Chen, W.; Jiang, B.; Qin, H.; Guo, X.; Liu, M.; et al. Circular RNA circSLC8A1 acts as a sponge of miR-130b/miR-494 in suppressing bladder cancer progression via regulating PTEN. Mol. Cancer 2019, 18, 111. [Google Scholar] [CrossRef]
- Zhu, Y.J.; Zheng, B.; Luo, G.J.; Ma, X.K.; Lu, X.Y.; Lin, X.M.; Yang, S.; Zhao, Q.; Wu, T.; Li, Z.X. Circular RNAs negatively regulate cancer stem cells by physically binding FMRP against CCAR1 complex in hepatocellular carcinoma. Theranostics 2019, 9, 3526–3540. [Google Scholar] [CrossRef]
- Ashwal-Fluss, R.; Meyer, M.; Pamudurti, N.R.; Ivanov, A.; Bartok, O.; Hanan, M.; Evantal, N.; Memczak, S.; Rajewsky, N.; Kadener, S. CircRNA biogenesis competes with pre-mRNA splicing. Mol. Cell 2014, 56, 55–66. [Google Scholar] [CrossRef]
- Zhang, M.; Huang, N.; Yang, X.; Luo, J.; Yan, S.; Xiao, F.; Chen, W.; Gao, X.; Zhao, K.; Zhou, H.; et al. A novel protein encoded by the circular form of the SHPRH gene suppresses glioma tumorigenesis. Oncogene 2018, 37, 1805–1814. [Google Scholar] [CrossRef]
- Hansen, T.B.; Jensen, T.I.; Clausen, B.H.; Bramsen, J.B.; Finsen, B.; Damgaard, C.K.; Kjems, J. Natural RNA circles function as efficient microRNA sponges. Nature 2013, 495, 384–388. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Li, X.; Zheng, H.; Si, X.; Li, B.; Wei, G.; Li, C.; Chen, Y.; Chen, Y.; Liao, W.; et al. Loss of super-enhancer-regulated circRNA Nfix induces cardiac regeneration after myocardial infarction in adult mice. Circulation 2019, 139, 2857–2876. [Google Scholar] [CrossRef] [PubMed]
- Hall, L.F.; Climent, M.; Quintavalle, M.; Farina, F.M.; Schorn, T.; Zani, S.; Carullo, P.; Kunderfranco, P.; Civilini, E.; Condorelli, G.; et al. Circ_Lrp6, a circular RNA enriched in vascular smooth muscle cells, acts as a sponge regulating miRNA-145 function. Circ. Res. 2019, 124, 498–510. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhang, Y.; Wang, Y.; Zhao, Y.; Ding, H.; Li, P. Circular RNAs: Functions and clinical significance in cardiovascular disease. Front. Cell Dev. Biol. 2020, 8, 584051. [Google Scholar] [CrossRef]
- Memczak, S.; Jens, M.; Elefsinioti, A.; Torti, F.; Krueger, J.; Rybak, A.; Maier, L.; Mackowiak, S.D.; Gregersen, L.H.; Munschauer, M.; et al. Circular RNAs are a large class of animal RNAs with regulatory potency. Nature 2013, 495, 333–338. [Google Scholar] [CrossRef]
- Wong, C.H.; Lou, U.K.; Li, Y.; Chan, S.L.; Tong, J.H.; To, K.F.; Chen, Y. CircFOXK2 promotes growth and metastasis of pancreatic ductal adenocarcinoma by complexing with RNA-binding proteins and sponging miR-942. Cancer Res. 2020, 80, 2138–2149. [Google Scholar] [CrossRef] [PubMed]
- Dong, K.; He, X.; Su, H.; Fulton, D.J.R.; Zhou, J. Genomic analysis of circular RNAs in heart. BMC Med. Genom. 2020, 13, 167. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Shen, H.B. RNA-protein binding motifs mining with a new hybrid deep learning based cross-domain knowledge integration approach. BMC Bioinform. 2017, 18, 136. [Google Scholar] [CrossRef]
- Pan, X.; Rijnbeek, P.; Yan, J.; Shen, H.B. Prediction of RNA-protein sequence and structure binding preferences using deep convolutional and recurrent neural networks. BMC Gen. 2018, 19, 511. [Google Scholar] [CrossRef]
- Pan, X.; Shen, H.B. Predicting RNA-protein binding sites and motifs through combining local and global deep convolutional neural networks. Bioinformatics 2018, 34, 3427–3436. [Google Scholar] [CrossRef]
- Wang, Z.; Lei, X.; Wu, F.X. Identifying cancer-specific circRNA-RBP binding sites based on deep learning. Molecules 2019, 24, 4035. [Google Scholar] [CrossRef] [PubMed]
- Bhuyan, R.; Bagchi, A. Prediction of the differentially expressed circRNAs to decipher their roles in the onset of human colorectal cancers. Gene 2020, 762, 145035. [Google Scholar] [CrossRef] [PubMed]
- Wawrzyniak, O.; Zarebska, Z.; Kuczynski, K.; Gotz-Wieckowska, A.; Rolle, K. Protein-related circular RNAs in human pathologies. Cells 2020, 9, 1841. [Google Scholar] [CrossRef] [PubMed]
- Zang, J.; Lu, D.; Xu, A. The interaction of circRNAs and RNA binding proteins: An important part of circRNA maintenance and function. J. Neurosci. Res. 2020, 98, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Abdelmohsen, K.; Panda, A.C.; Munk, R.; Grammatikakis, I.; Dudekula, D.B.; De, S.; Kim, J.; Noh, J.H.; Kim, K.M.; Martindole, J.L.; et al. Identification of HuR target circular RNAs uncovers suppression of PABPN1 translation by CircPABPN1. RNA Biol. 2017, 14, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Holdt, L.M.; Stahringer, A.; Sass, K.; Pichler, G.; Kulak, N.A.; Wilfert, W.; Kohlmaier, A.; Herbst, A.; Northoff, B.H.; Nicolaon, A.; et al. Circular non-coding RNA ANRIL modulates ribosomal RNA maturation and atherosclerosis in humans. Nat. Commun. 2016, 7, 12429. [Google Scholar] [CrossRef] [PubMed]
- Du, W.W.; Yang, W.; Chen, Y.; Wu, Z.K.; Foster, F.S.; Yang, Z.; Li, X.; Yang, B.B. Foxo3 circular RNA promotes cardiac senescence by modulating multiple factors associated with stress and senescence responses. Eur. Heart J. 2017, 38, 1402–1412. [Google Scholar] [CrossRef]
- Yang, Z.G.; Awan, F.M.; Du, W.W.; Zeng, Y.; Lyu, J.; Wu, D.; Gupta, S.; Yang, W.; Yang, B.B. The circular RNA interacts with STAT3, increasing its nuclear translocation and wound repair by modulating Dnmt3a and miR-17 function. Mol. Ther. 2017, 25, 2062–2074. [Google Scholar] [CrossRef]
- Zeng, Y.; Du, W.W.; Wu, Y.; Yang, Z.; Awan, F.M.; Li, X.; Yang, W.; Zhang, C.; Yang, Q.; Yee, A.; et al. A circular RNA binds to and activates AKT phosphorylation and nuclear localization reducing apoptosis and enhancing cardiac repair. Theranostics 2017, 7, 3842–3855. [Google Scholar] [CrossRef]
- Hou, J.; Kang, Y.J. Regression of pathological cardiac hypertrophy: Signaling pathways and therapeutic targets. Pharmacol. Ther. 2012, 135, 337–354. [Google Scholar] [CrossRef]
- Ladage, D.; Tilemann, L.; Ishikawa, K.; Correll, R.N.; Kawase, Y.; Houser, S.R.; Molkentin, J.D.; Hajjar, R.J. Inhibition of PKCalpha/beta with ruboxistaurin antagonizes heart failure in pigs after myocardial infarction injury. Circ. Res. 2011, 109, 1396–1400. [Google Scholar] [CrossRef] [PubMed]
- Tham, Y.K.; Bernardo, B.C.; Ooi, J.Y.Y.; Weeks, K.L.; McMullen, J.R. Pathophysiology of cardiac hypertrophy and heart failure: Signaling pathways and novel therapeutic targets. Arch. Toxicol. 2015, 89, 1401–1438. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; de Castro Bras, L.E.; Toba, H.; Iyer, R.P.; Hall, M.E.; Winniford, M.D.; Lange, R.A.; Tyagi, S.C.; Lindsay, M.L. Myofibroblasts and the extracellular matrix network in postmyocardial infarction cardiac remodeling. Pflug. Arch.-Eur. J. Physiol. 2014, 466, 1113–1127. [Google Scholar]
- Takemura, G.; Kanamari, H.; Okada, H.; Miyazaki, N.; Watanabe, T.; Tsujimoto, A.; Goto, K.; Maruyama, R.; Fujiwara, T.; Fujiwara, H. Anti-apoptosis in nonmyocytes and pro-autophagy in cardiomyocytes: Two strategies against postinfarction heart failure through regulation of cell death/degeneration. Heart Fail. Rev. 2018, 23, 759–772. [Google Scholar] [CrossRef] [PubMed]
- Kroemer, G.; Galluzzi, L.; Vandenabeele, P.; Abrams, J.; Alnemri, E.S.; Baehrecke, E.H.; Blagosklonmy, M.V.; El-Deiry, W.S.; Golstein, P.; Green, D.R.; et al. Classification of cell death: Recommendations of the Nomenclature Committee on Cell Death 2009. Cell Death Differ. 2009, 16, 3–11. [Google Scholar] [CrossRef]
- Salgado-Somoza, A.; Zhang, L.; Vausort, M.; Devaux, Y. The circular RNA MICRA for risk stratification after myocardial infarction. IJC Heart Vasculat. 2017, 17, 33–36. [Google Scholar] [CrossRef]
- Vausort, M.; Salgado-Somoza, A.; Zhang, L.; Leszek, P.; Scholz, M.; Teren, A.; Burkhardt, R.; Thiery, J.; Wagner, D.R.; Devaux, Y. Myocardial infarction-associated circular RNA predicting left ventricular dysfunction. J. Am. Coll. Cardiol. 2016, 68, 1247–1248. [Google Scholar] [CrossRef]
- Garikipati, V.N.S.; Verma, S.K.; Cheng, Z.; Liang, D.; Truongcao, M.M.; Cimini, M.; Yue, Y.; Huang, G.; Wang, C.; Benedict, C. Circular RNA CircFndc3b modulates cardiac repair after myocardial infarction via FUS/VEGF-A axis. Nat. Commun. 2019, 10, 1–14. [Google Scholar] [CrossRef]
- Cai, L.; Qi, B.; Wu, X.; Peng, S.; Zhou, G.; Wei, Y.; Xu, J.; Chen, S.; Liu, S. Circular RNA Ttc3 regulates cardiac function after myocardial infarction by sponging miR-15b. J. Mol. Cell Cardiol. 2019, 130, 4317. [Google Scholar] [CrossRef]
- Si, X.; Zheng, H.; Wei, G.; Li, M.; Li, W.; Wang, H.; Guo, H.; Sun, J.; Li, C.; Zhong, S.; et al. circRNA Hipk3 induces cardiac regeneration after myocardial infarction in mice by binding to Notch1 and miR-133a. Mol. Ther. Nucleic Acids 2020, 21, 636–655. [Google Scholar] [CrossRef]
- Rai, A.K.; Lee, B.; Hebbard, C.; Uchida, S.; Garikipati, V.N.S. Decoding the complexicity of circular RNAs in cardiovascular disease. Pharmacol. Res. 2021, 171, 105766. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.L. The expanding regulatory mechanisms and cellular functions of circular RNAs. Nat. Rev. Mol. Cell Biol. 2020, 21, 475–490. [Google Scholar] [CrossRef] [PubMed]
- Geng, H.H.; Li, R.; Su, Y.M.; Xiao, J.; Pan, M.; Cai, X.X.; Ji, X.P. The circular RNA Cdr1as promotes myocardial infarction by mediating the regulation of miR-7a on its target genes expression. PLoS ONE 2016, 11, e0151753. [Google Scholar] [CrossRef] [PubMed]
- Jia, G.; Hill, M.A.; Sowers, J.R. Diabetic Cardiomyopathy: An update of mechanisms contributing to this clinical entity. Circ. Res. 2018, 122, 624–638. [Google Scholar] [CrossRef] [PubMed]
- Rubler, S.; Dlugash, J.; Yuceoglu, Y.Z.; Kumral, T.; Branwood, A.W.; Grishman, A. New type of cardiomyopathy associated with diabetic glomerulosclerosis. Am. J. Cardiol. 1972, 30, 595–602. [Google Scholar] [CrossRef]
- Chavali, V.; Tyagi, S.C.; Mishra, P.K. Predictors and prevention of diabetic cardiomyopathy. Diabetes Metab. Syndr. Obes. 2013, 6, 151–160. [Google Scholar] [PubMed]
- Falcao-Pires, I.; Leite-Moreira, A.F. Diabetic cardiomyopathy: Understanding the molecular and cellular basis to progress in diagnosis and treatment. Heart Fail Rev. 2012, 17, 325–344. [Google Scholar] [CrossRef] [PubMed]
- Kayama, Y.; Raaz, U.; Jagger, A.; Adam, M.; Schellinger, I.; Sakamoto, M.; Suzuki, H.; Toyama, K.; Spin, J.; Tsao, P. Diabetic cardiovascular disease induced by oxidative stress. Int. J. Mol. Sci. 2015, 16, 25234–25263. [Google Scholar] [CrossRef]
- Hu, X.; Bai, T.; Xu, Z.; Liu, Q.; Zheng, Y.; Cai, L. Pathophysiological fundamentals of diabetic cardiomyopathy. Compr. Physiol. 2017, 7, 693–711. [Google Scholar]
- Varga, Z.V.; Giricz, A.; Liaudet, L.; Hask´o, G.; Ferdinandy, P.; Pacher, P. Interplay of oxidative, nitrosative/nitrative stress, inflammation, cell death and autophagy in diabetic cardiomyopathy. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2015, 1852, 232–242. [Google Scholar] [CrossRef]
- Wan, H.; Zhao, S.; Zeng, Q.; Tan, Y.; Zhang, C.; Liu, L.; Qu, S. CircRNAs in diabetic cardiomyopathy. Clin. Chim. Acta 2021, 517, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Xia, F.; Sun, J.J.; Jiang, Y.Q.; Li, C.F. MicroRNA-384-3p inhibits retinal neovascularization through targeting hexokinase 2 in mice with diabetic retinopathy. J. Cell Physiol. 2019, 234, 721–730. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Zhao, Q.; Chen, W.; Zhang, Y. Oncogene Lin28B increases chemosensitivity of colon cancer cells in a let-7-independent manner. Oncol. Lett. 2018, 15, 6975–6981. [Google Scholar] [CrossRef]
- Brennan, E.; Wang, B.; McClelland, A.; Mohan, M.; Marai, M.; Beuscart, O.; Derouiche, S.; Gray, S.; Pickering, R.; Tikellis, C. Protective effect of let-7 miRNA family in regulating inflammation in diabetes-associated atherosclerosis. Diabetes 2017, 66, 2266–2277. [Google Scholar] [CrossRef] [PubMed]
- Jin, G.; Wang, Q.; Hu, X.; Li, X.; Pei, X.; Xu, E.; Li, M. Profiling and functional analysis of differentially expressed circular RNAs in high glucose-induced human umbilical vein endothelial cells. FEBS Open Biol. 2019, 9, 1640–1651. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Sui, Y. CircBPTF knockdown ameliorates high glucose-induced inflammatory injuries and oxidative stress by targeting the miR-384/LIN28B axis in human umbilical vein endothelial cells. Mol. Cell Biochem. 2020, 471, 101–111. [Google Scholar] [CrossRef]
- Tang, C.M.; Zhang, M.; Huang, L.; Hu, Z.Q.; Zhu, J.N.; Xiao, Z.; Zhang, Z.; Lin, Q.X.; Zheng, X.L.; Yang, M.; et al. CircRNA_000203 enhances the expression of fibrosis-associated genes by derepressing targets of miR-26b-5p, Col1a2 and CTGF, in cardiac fibroblasts. Sci. Rep. 2017, 7, 40342. [Google Scholar] [CrossRef]
- Zhou, B.; Yu, J.W. A novel identified circular RNA, circRNA_010567, promotes myocardial fibrosis via suppressing miR-141 by targeting TGF-β1. Biochem. Biophys. Res. Commun. 2017, 487, 769–775. [Google Scholar] [CrossRef]
- Yang, F.; Li, A.; Qin, Y.; Che, H.; Wang, Y.; Lv, J.; Li, Y.; Li, H.; Yue, E.; Ding, X.; et al. A novel circular RNA mediates pyroptosis of diabetic cardiomyopathy by functioning as a competing endogenous RNA. Mol. Ther. Nucleic Acids. 2019, 17, 636–643. [Google Scholar] [CrossRef]
- Nakamura, M.; Sadoshima, J. Mechanisms of physiological and pathological cardiac hypertrophy. Nat. Rev. Cardiol. 2018, 15, 387–407. [Google Scholar] [CrossRef]
- Wang, K.; Long, B.; Liu, F.; Wang, J.X.; Liu, C.Y.; Zhao, B.; Zhou, L.Y.; Sun, T.; Wang, M.; Yu, T.; et al. A circular RNA protects the heart from pathological hypertrophy and heart failure by targeting miR-223. Eur. Heart J. 2016, 37, 2602–2611. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Xu, J.D.; Fang, X.H.; Zhu, J.N.; Yang, J.; Pan, R.; Yuan, S.J.; Zeng, N.; Yang, Z.Z.; Yang, H.; et al. Circular RNA circRNA_000203 aggravates cardiac hypertrophy via suppressing miR-26b-5p and miR-140-3p binding to Gata4. Cardiovasc. Res. 2020, 116, 1323–1334. [Google Scholar] [CrossRef] [PubMed]
- Lim, T.B.; Aliwarga, E.; Luu, T.D.A.; Li, Y.P.; Ng, S.L.; Annadoray, L.; Sian, S.; Ackers-Johnson, M.A.; Foo, R.S.Y. Targeting the highly abundant circular RNA circSlc8a1 in cardiomyocytes attenuates pressure overload induced hypertrophy. Cardiovasc. Res. 2019, 115, 1998–2007. [Google Scholar] [CrossRef] [PubMed]
- Lavenniah, A.; Luu, T.D.A.; Li, Y.P.; Lim, T.B.; Jiang, J.; Ackers-Johnson, M.; Foo, R.S.Y. Engineered circular RNA sponges act as miRNA inhibitors to attenuate pressure overload induced cardiac hypertrophy. Mol. Ther. 2020, 28, 1506–1517. [Google Scholar] [CrossRef] [PubMed]
- Kong, P.; Christia, P.; Frangogiannis, N.G. The pathogenesis of cardiac fibrosis. Cell Mol. Life Sci. 2014, 71, 549–574. [Google Scholar] [CrossRef]
- Yong, K.W.; Li, Y.; Huang, G.; Lu, T.J.; WKZWSafwani Pingguan-Murphy, B.; Xu, F. Mechanoregulation of cardiac myofibroblast differentiation: Implications for cardiac fibrosis and therapy. Am. J. Physiol. Heart Circ. Physiol. 2015, 309, H532–H542. [Google Scholar] [CrossRef]
- Kuwahara, F.; Kai, H.; Tokuda, K.; Takeya, M.; Takeshita, A.; Egashira, K.; Imaizumi, T. Hypertensive myocardial fibrosis and diastolic dysfunction: Another model of inflammation? Hypertension 2004, 43, 739–745. [Google Scholar] [CrossRef]
- Hulsmans, M.; Sam, F.; Nahrendorf, M. Monocyte and macrophage contributions to cardiac remodeling. J. Mol. Cell Cardiol. 2016, 93, 149–155. [Google Scholar] [CrossRef]
- Zhu, Y.; Pan, W.; Yang, T.; Meng, X.; Jiang, Z.; Tao, L.; Wang, L. Upregulation of circular RNA CircNFIB attenuates cardiac fibrosis by sponging miR-433. Front Genet. 2019, 10, 564. [Google Scholar] [CrossRef]
- Ni, H.; Li, W.F.; Zhuge, Y.; Xu, S.; Wang, Y.; Chen, Y.; Shen, G.; Wang, F. Inhibition of circHIPK3 prevents angiotensin II-induced cardiac fibrosis by sponging miR-29b-3p. Int. J. Cardiol. 2019, 292, 188–196. [Google Scholar] [CrossRef]
- Xu, M.; Xie, F.; Tang, X.; Wang, T.; Wang, S. Insights into the role of circular RNA in macrophage activation and fibrosis disease. Pharmacol. Res. 2020, 156, 104777. [Google Scholar] [CrossRef]
- Mizushima, N. Autophagy: Process and function. Genes Dev. 2007, 21, 2861–2873. [Google Scholar] [CrossRef] [PubMed]
- Galluzzi, L.; Bravo-San Pedro, J.M.; Demaria, S.; Farmenti, S.C.; Kroemer, G. Activating autophagy to potentiate immunogenic chemotherapy and radiation therapy. Nat. Rev. Clin. Oncol. 2017, 14, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Maejima, Y.; Kyoi, S.; Zhai, P.; Liu, T.; Li, H.; Ivessa, A.; Sciaretta, S.; Del Re, D.P.; Zabłocki, D.K.; Hsu, C.P.; et al. Mst1 inhibits autophagy by promoting the interaction between Beclin1 and Bcl-2. Nat. Med. 2013, 19, 1478–1488. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.Y.; Zhai, M.; Huang, Y.; Xu, S.; An, T.; Wang, Y.H.; Zhang, R.C.; Liu, C.Y.; Dong, Y.H.; Wang, M.; et al. The circular RNA ACR attenuates myocardial ischemia/reperfusion injury by suppressing autophagy via modulation of the Pink1/FAM65B pathway. Cell Death Differ. 2019, 26, 1299–1315. [Google Scholar] [CrossRef] [PubMed]
- Savitskaya, M.A.; Onishchenko, G.E. Mechanisms of apoptosis. Biochemistry 2015, 80, 1393–1405. [Google Scholar] [CrossRef]
- Wencker, D.; Chandra, M.; Nguyen, K.; Miao, W.; Garantziotis, S.; Factor, S.M.; Shirani, J.; Amstrong, R.C.; Kitsis, R.N. A mechanistic role for cardiac myocyte apoptosis in heart failure. J. Clin. Investig. 2003, 111, 1497–1504. [Google Scholar] [CrossRef]
- Wang, K.; Gan, T.Y.; Li, N.; Liu, C.Y.; Zhou, L.Y.; Gao, J.N.; Chen, C.; Yan, K.W.; Ponnusamy, M.; Zhang, Y.H.; et al. Circular RNA mediates cardiomyocyte death via miRNA dependent upregulation of MTP18 expression. Cell Death Differ. 2017, 24, 1111–1120. [Google Scholar] [CrossRef]
- Sygitowicz, G.; Sitkiewicz, D. Mitochondrial quality control: The role in cardiac injury. Front Biosci. 2022, 27, 96. [Google Scholar] [CrossRef]
- Tondera, D.; Czauderna, F.; Paulick, K.; Schwarzer, R.; Kaufmann, J.; Santel, A. The mitochondrial protein MTP18 contributes to mitochondrial fission in mammalian cells. J. Cell Sci. 2005, 118, 3049–3059. [Google Scholar] [CrossRef]
- Deng, Y.; Wang, J.; Xie, G.; Zeng, X.; Li, H. Circ-HIPK3 strengthens the effects of adrenaline in heart failure by miR-17-3p-ADCY6 axis. Int. J. Biol. Sci. 2019, 15, 2484–2496. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.Y.; Zhao, J.C.; Ge, X.M.; Zhang, H.; Wang, C.M.; Bie, Z.D. Circ_LAS1L regulates cardiac fibroblast activation, growth, and migration through miR-125b/SFRP5pathway. Cell Biochem. Funct. 2020, 38, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Long, T.Y.; Bi, S.S.; Sheikh, S.A.; Zhang, C.L. circPAN3 exerts a profibrotic role via sponging miR-221 through FoxO3/ATG7-activated autophagy in a rat model of myocardial infarction. Life Sci. 2020, 257, 118015. [Google Scholar] [CrossRef]
- Ouyang, Q.; Huang, Q.; Jiang, Z.; Zhao, J.; Shi, G.P.; Yang, M. Using plasma circRNA_002453 as a novel biomarker in the diagnosis of lupus nephritis. Mol. Immunol. 2018, 101, 531–538. [Google Scholar] [CrossRef]
- Wu, J.; Zhou, Q.; Niu, Y.; Chen, J.; Zhu, Y.; Ye, S.; Xi, Y.; Wang, F.; Qiu, H.; Bu, S. Aberrant expression of serum circANRIL and hsa_circ_0123996 in children with Kawasaki disease. J. Clin. Lab. Anal. 2019, 33, e22874. [Google Scholar] [CrossRef] [PubMed]
- Bahn, J.H.; Zhang, Q.; Li, F.; Chan, T.M.; Lin, X.; Kim, Y.; Wong, D.T.; Xiao, X. The landscape of microRNA, piwi-interacting RNA, and circular RNA in human saliva. Clin. Chem. 2015, 61, 221–230. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, R.; Liu, W.; Wang, Z.; Rong, J.; Long, X.; Liu, Z.; Ge, J.; Shi, B. Exosomal circHIPK3 released from hypoxia-pretreated cardiomyocytes regulates oxidative damage in cardiac microvascular endothelial cells via the miR-29a/IGF-1 pathway. Oxid. Med. Cell Longev. 2019, 2019, 7954657. [Google Scholar] [CrossRef]
- Tuo, B.; Chen, Z.; Dong, Q.; Chen, C.; Zhang, H.; Hu, S.; Sun, Z. Role of exosomal circRNAs in tumor immunity and cancer progression. Cell Death Dis. 2022, 13, 539. [Google Scholar] [CrossRef]
- Lu, P.; Ding, F.; Xiang, Y.K.; Hao, L.; Zhao, M. Noncoding RNAs in cardiac hypertrophy and heart failure. Cells 2022, 11, 777. [Google Scholar] [CrossRef]
- Zhu, L.; Li, N.; Sun, L.; Zheng, D.; Shao, G. Non-coding RNAs: The key detectors and regulators in cardiovascular disease. Genomics 2021, 113, 1233–1246. [Google Scholar] [CrossRef]
- Azuaje, F.J.; Dewey, F.E.; Brutsaert, D.L.; Devaux, Y.; Ashley, E.A.; Wagner, D.R. Systems-based approaches to cardiovascular biomarker discovery. Circ. Cardiovasc. Genet. 2012, 5, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Devaux, Y.; Vausort, M.; McCann, G.P.; Zangrando, J.; Kelly, D.; Razvi, N.; Zhang, L.; Ng, L.L.; Wagner, D.R.; Squire, L.B. Micro-RNA-150: A novel marker of left ventricular remodeling after acute myocardial infarction. Circ. Cardiovasc. Genet. 2013, 6, 290–298. [Google Scholar] [CrossRef] [PubMed]
- Devaux, Y.; Zangrando, J.; Schroen, B.; Creemers, E.E.; Pedrazzini, T.; Chang, C.P.; Dorn, G.W.; Thum, T.; Heymans, S. Long noncoding RNAs in cardiac development and ageing. Nat. Rev. Cardiol. 2015, 12, 415–425. [Google Scholar] [PubMed]
- Yuan, T.; Krishnan, J. Non-coding RNAs in cardiac regeneration. Front. Physiol. 2021, 12, 650566. [Google Scholar] [CrossRef] [PubMed]
- Devaux, Y.; Creemers, E.E.; Boon, R.A.; Werfel, S.; Thum, T.; Engelhardt, S.; Dimmeler, S.; Squire, I. Circular RNAs in heart failure. Eur. J. Heart Fail. 2017, 19, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Long, T.; Du, T.; Chen, Y.; Dong, Y.; Huang, Z.P. Circle the cardiac remodeling with circRNAs. Front. Cardiovasc. Med. 2021, 8, 702586. [Google Scholar] [CrossRef]
| Diabetic Cardiomyopathy | |||||
|---|---|---|---|---|---|
| CircRNAs | Expression | Targets | Place of Action | Effect | References |
| CircBPTF | ↑ | miR-384 ↓ | Endothelial cells | Endothelial dysfunction | [65,66] |
| CircRNA_000203 | ↑ | miR-26b-5p ↓ | Myocardial fibroblasts | Fibrosis | [67] |
| CircRNA_010567 | ↑ | miR-141 ↓ | Myocardial fibroblasts | Fibrosis | [68] |
| CACR | ↑ | miR-214-3p ↓ | Cardiomiocytes | Inflammation Pyroptosis Apoptosis | [69] |
| CircRNAs | Expression | Targets | Subcellular Localization | Affected Pathway | Effect | References |
|---|---|---|---|---|---|---|
| Cardiac hypertrophy | ||||||
| Circ-HRCR | ↓ | miR-223 | Cytoplasm | ARC axis | Reduces hypertrophy and heart failure | [70] |
| CircRNA_000203 | ↑ | miR-140-3p or miR-26b-5p | Cytoplasm | GATA4 axis | Exacerbates cardiac hypertrophy Excludes the anti-fibrotic effect of miR-26b in mouse CFs | [72] [67] |
| CiRS-7 | ↑ | miR-7 | Cytoplasm | PARP/SP1 | Worsens cardiac function | [53] |
| Cardiac fibrosis | ||||||
| CircNF1B | ↓ | miR-433 | Cytoplasm | Fibroblast proliferation | Supports protection against cardiac fibrosis, attenuates fibrosis of cardiac | [79] |
| CircHIPK3 | ↑ | miR-29b-3p or miR-17-3p | Cytoplasm | ADCY6 axis | Advances fibrosis of cardiac by up-regulating fibrosis-associated genes | [80,91] |
| CircRNA_010567 | ↑ | miR-141/TGF | Cytoplasm | β1 axis | Reduces fibrosis-associated Col I, Col III and α-SMA protein expression | [68] |
| CircRNA_005647 | ↓ | miR-27b-3p | Cytoplasm | DAPK | Reduces expressions of fibrosis-related genes | [81] |
| Circ_LAS1L | ↓ | miR-125b | Cytoplasm | SFRP5 axis | Reduces the activation, proliferation and migration of cardiac fibroblasts | [92] |
| CircPAN3 | ↑ | miR-221 | Cytoplasm | FoxO3/TG7 | Advances cardiac fibrosis post-MI | [93] |
| Autophagy and apoptosis | ||||||
| CircACR | ↓ | Pink1 | Cytoplasm | FAM65B axis | Extinguish autophagy to attenuate myocardial ischemia/reperfusion injury | [85] |
| Circ-MFACR | ↑ | miR-652-3p | Cytoplasm | MTP18 axis | Advances mitochondrial fission and cardiomyocyte apoptosis | [71] |
| Circ_Ttc3 | ↓ | miR-15b | Cytoplasm | Arl2 axis | Causes cardioprotection by reducing cardiomyocyte apoptosis | [49] |
| CircFndc3b | ↑ | RNA binding protein | Nucleus | FUS/ VEGF-A axis | Extinguish cardiomyocyte apoptosis, improves neovascularization and pick up left ventricular function in post-MI events | [48] |
| CircNfix | ↑ | miR-214 | Cytoplasm | YBX1/NEDD4L | Extinguish cardiomyocyte proliferation, angiogenesis and increases CM apoptosis | [22] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sygitowicz, G.; Sitkiewicz, D. Involvement of circRNAs in the Development of Heart Failure. Int. J. Mol. Sci. 2022, 23, 14129. https://doi.org/10.3390/ijms232214129
Sygitowicz G, Sitkiewicz D. Involvement of circRNAs in the Development of Heart Failure. International Journal of Molecular Sciences. 2022; 23(22):14129. https://doi.org/10.3390/ijms232214129
Chicago/Turabian StyleSygitowicz, Grażyna, and Dariusz Sitkiewicz. 2022. "Involvement of circRNAs in the Development of Heart Failure" International Journal of Molecular Sciences 23, no. 22: 14129. https://doi.org/10.3390/ijms232214129
APA StyleSygitowicz, G., & Sitkiewicz, D. (2022). Involvement of circRNAs in the Development of Heart Failure. International Journal of Molecular Sciences, 23(22), 14129. https://doi.org/10.3390/ijms232214129






