Proteomes of Residual Tumors in Curcumin-Treated Rats Reveal Changes in Microenvironment/Malignant Cell Crosstalk in a Highly Invasive Model of Mesothelioma
Abstract
1. Introduction
2. Results
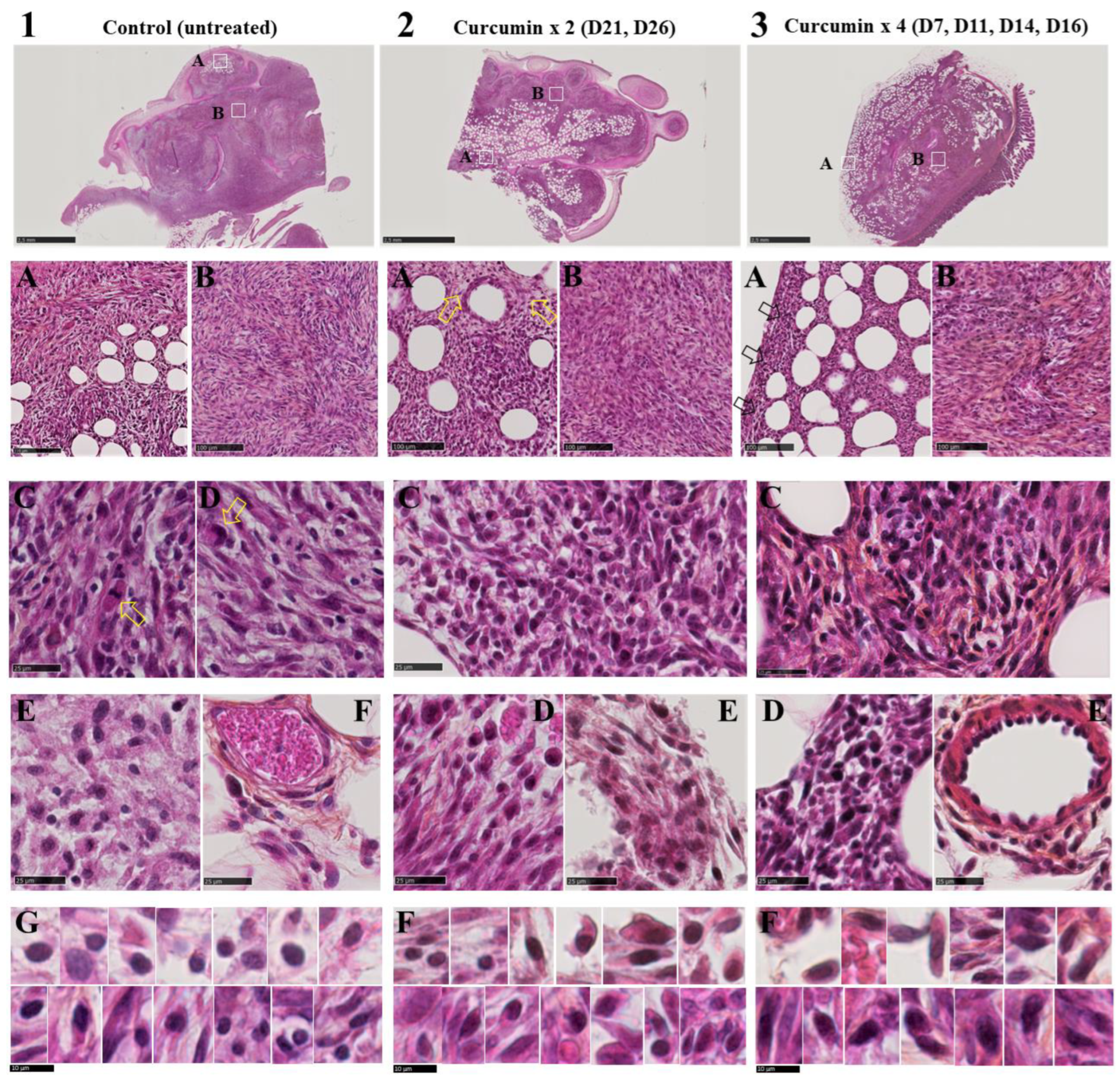
2.1. Tumors from Curcumin-Treated Rats Were Infiltrated with Numerous Activated Lymphocytes
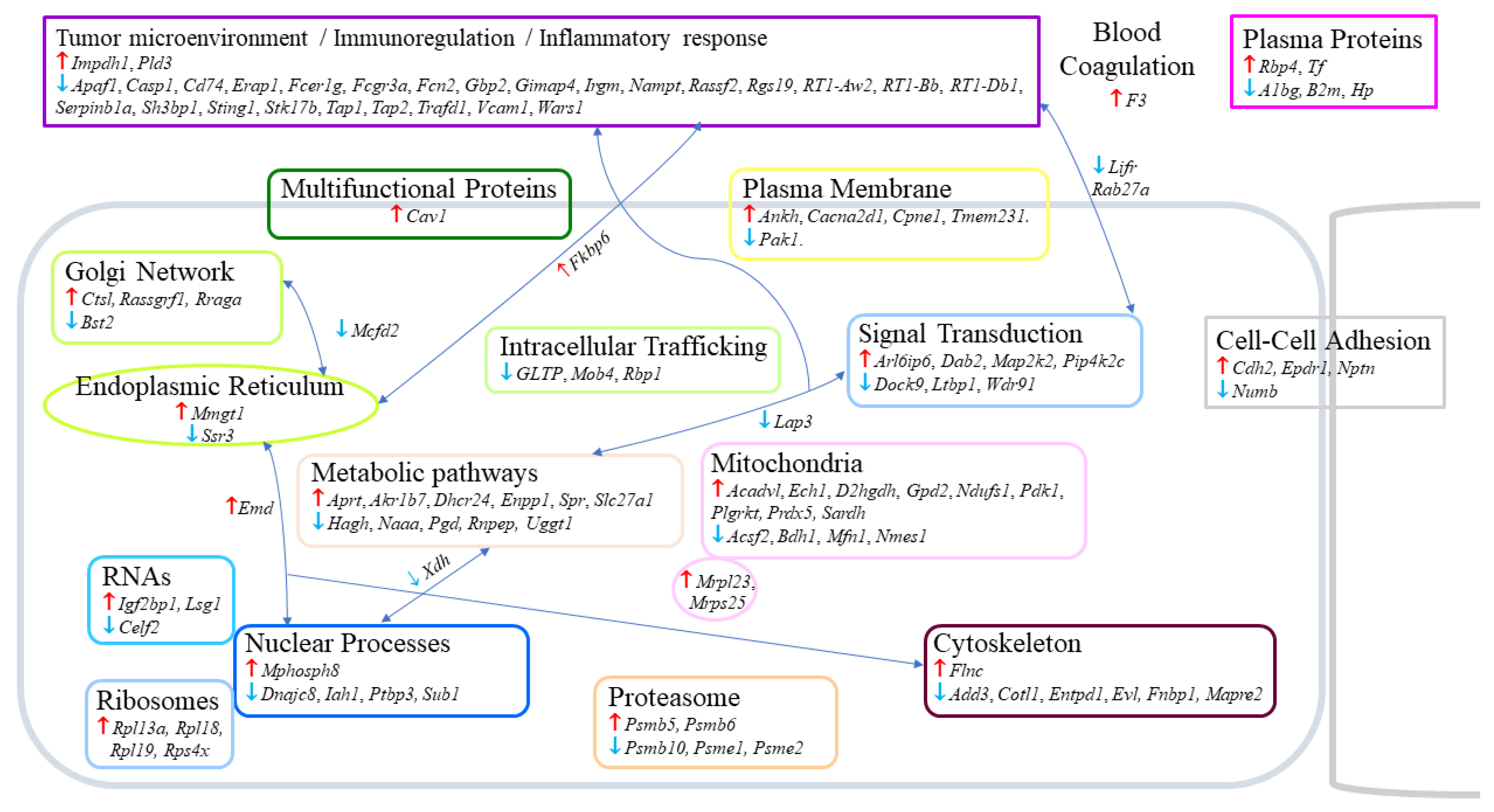
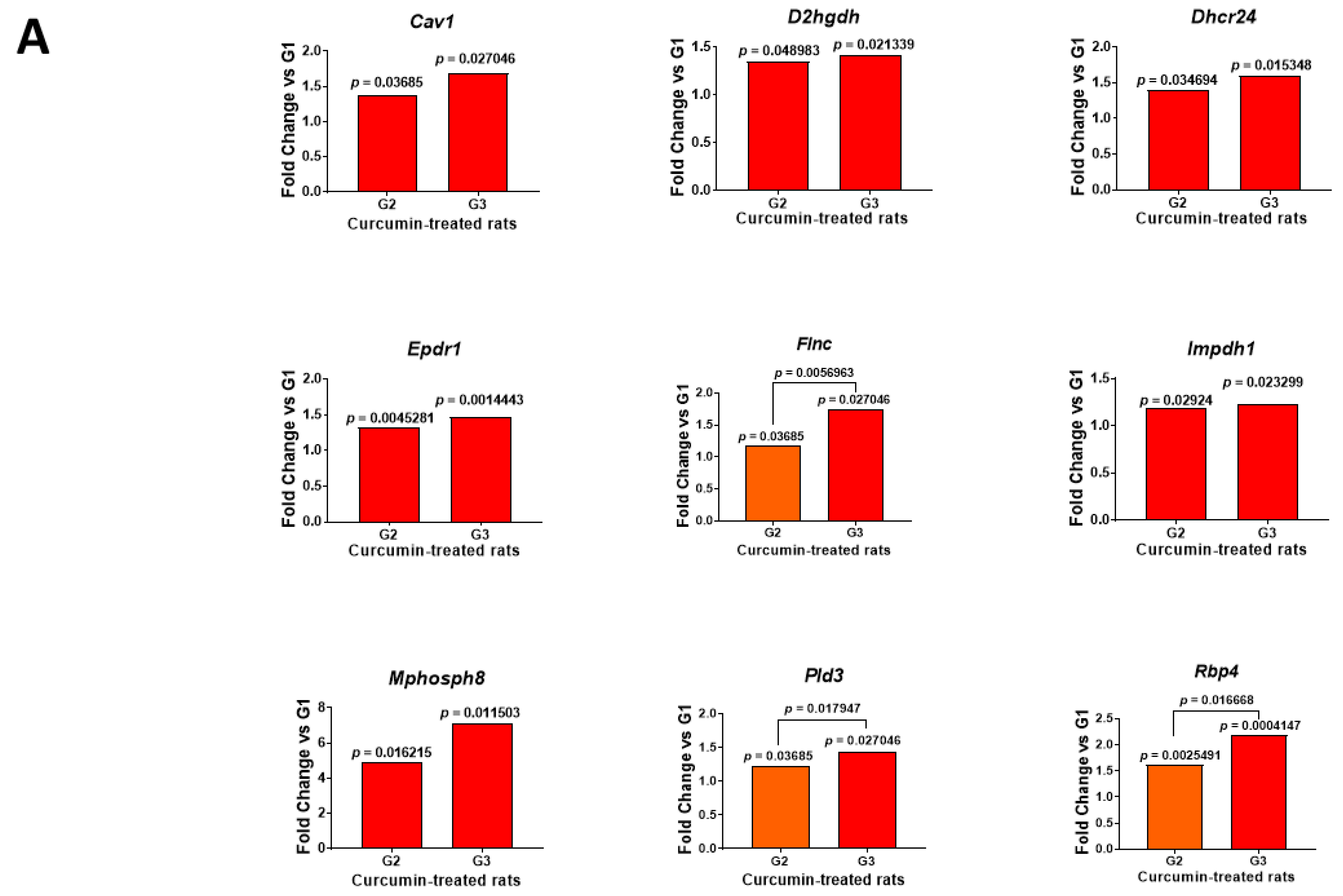
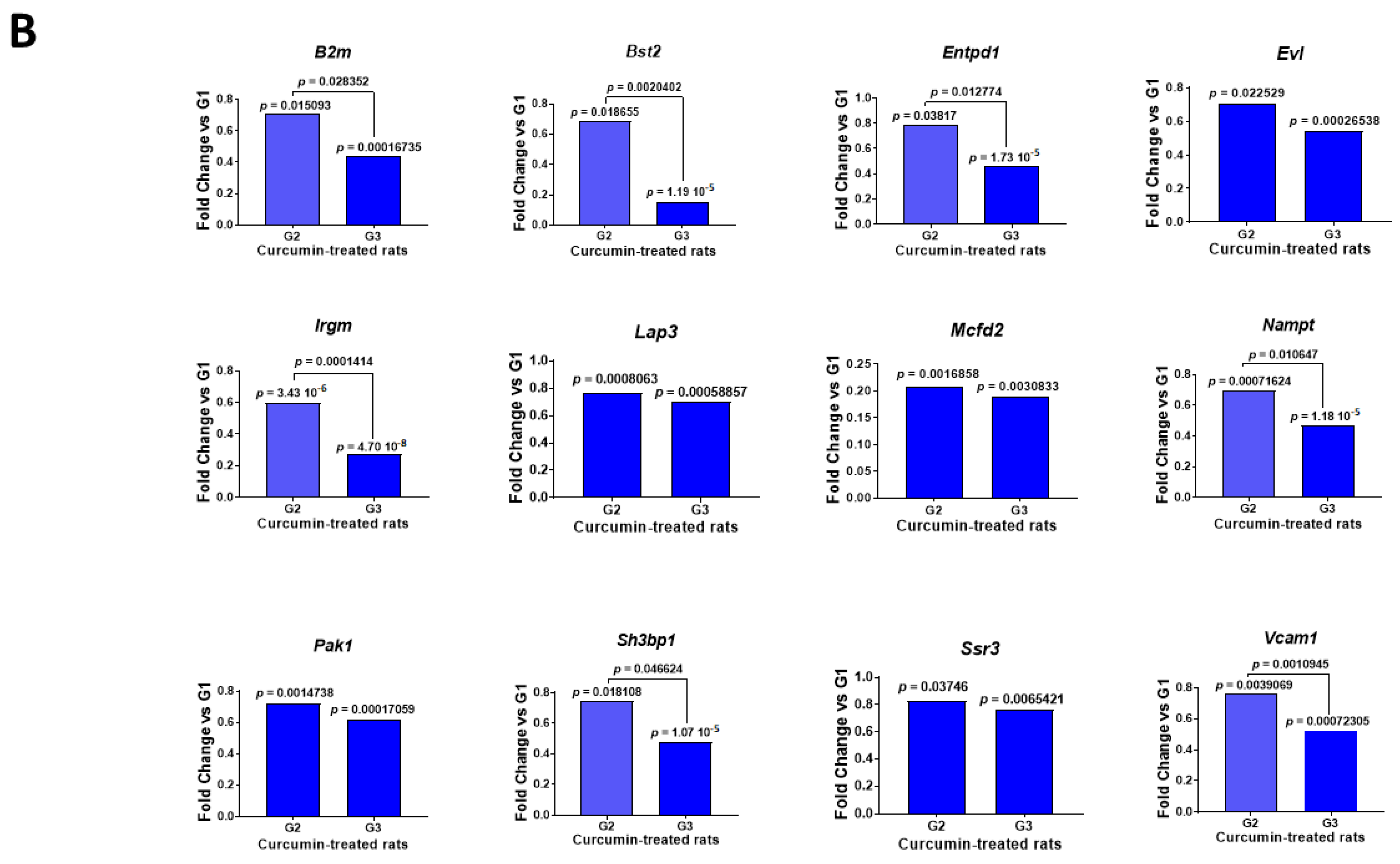
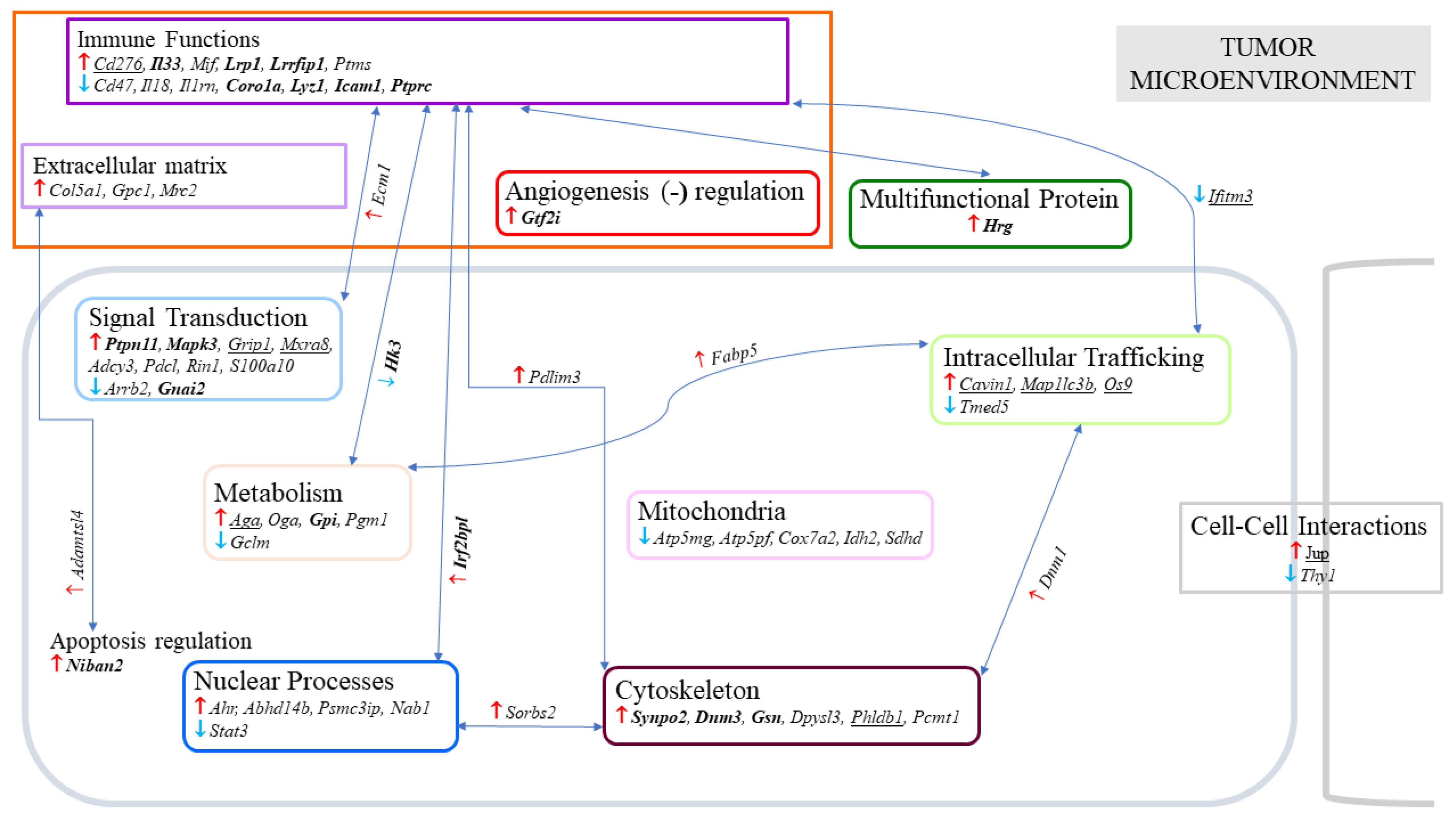
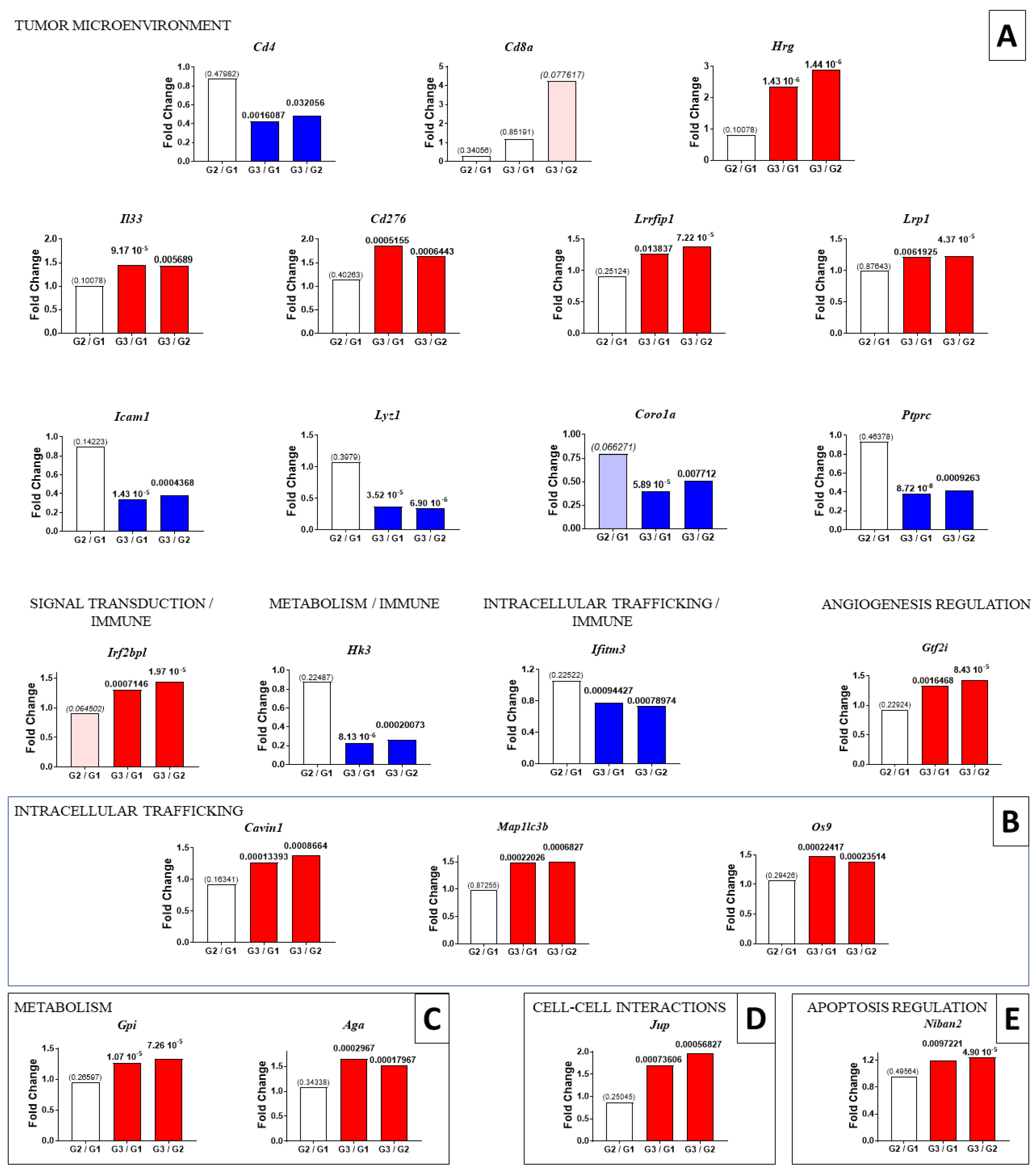
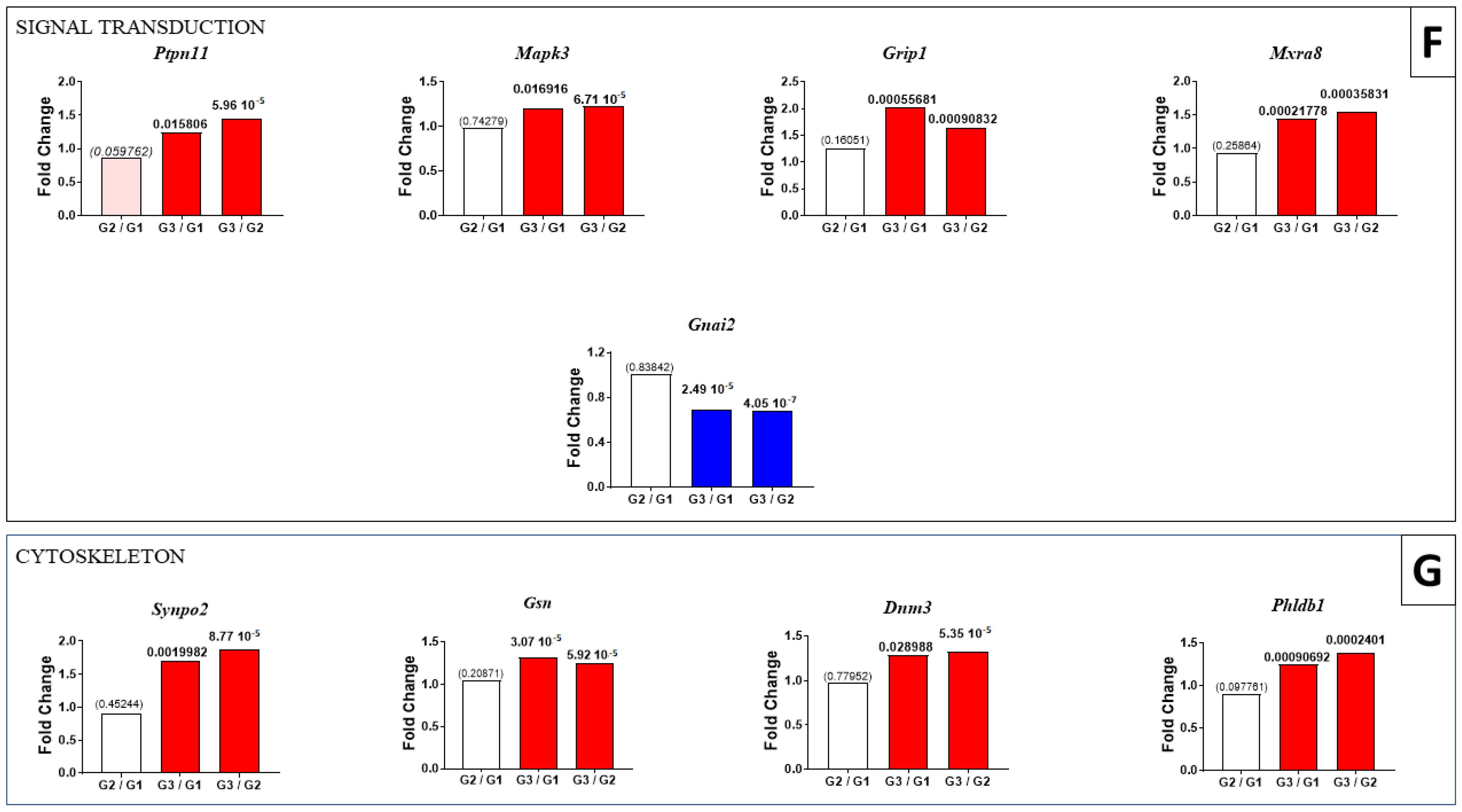
2.2. Residual Tumors from G2–G3 Curcumin-Treated Rats Exhibited Common Changes in 120 Proteins
2.3. Multiple and Early Curcumin Treatments Induced Specific Additional Events
3. Discussion
4. Materials and Methods
4.1. Experimental In Vivo Experiments
4.2. Histological Analyses
4.3. Proteomic Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Nagaraju, G.P.; Aliya, S.; Zafar, S.F.; Basha, R.; Diaz, R.; El-Rayes, B.F. The impact of curcumin on breast cancer. Integr. Biol. 2012, 4, 996–1007. [Google Scholar] [CrossRef] [PubMed]
- Gall Trošelj, K.; Samaržija, I.; Tomljanović, M.; Kujundžić, R.N.; Đaković, N.; Mojzeš, A. Implementing curcumin in translational oncology research. Molecules 2020, 25, 5240. [Google Scholar] [CrossRef] [PubMed]
- Fabianowska-Majewska, K.; Kaufman-Szymczyk, A.; Szymanska-Kolba, A.; Jakubik, J.; Majewski, G.; Lubecka, K. Curcumin from turmeric rhizome: A potential modulator of DNA methylation machinery in breast cancer inhibition. Nutrients 2021, 13, 332. [Google Scholar] [CrossRef] [PubMed]
- Da, W.; Zhu, J.; Wang, L.; Sun, Q. Curcumin suppresses lymphatic vessel density in an in vivo human gastric cancer model. Tumor Biol. 2015, 36, 5215–5223. [Google Scholar] [CrossRef]
- Zhao, G.; Han, X.; Zheng, S.; Li, Z.; Sha, Y.; Ni, J.; Sun, Z.; Qiao, S.; Song, Z. Curcumin induces autophagy, inhibits proliferation and invasion by downregulating AKT/mTOR signaling pathway in human melanoma cells. Oncol. Rep. 2016, 35, 1065–1074. [Google Scholar] [CrossRef]
- Pouliquen, D.L.; Nawrocki-Raby, B.; Nader, J.; Blandin, S.; Robard, M.; Birembaut, P.; Grégoire, M. Evaluation of intracavitary administration of curcumin for the treatment of sarcomatoid mesothelioma. Oncotarget 2017, 8, 57552–57573. [Google Scholar] [CrossRef][Green Version]
- Bose, S.; Panda, A.K.; Mukherjee, S.; Sa, G. Curcumin and tumor immune-editing: Resurrecting the immune system. Cell Div. 2015, 10, 6. [Google Scholar] [CrossRef]
- Xu, B.; Yu, L.; Zhao, L.-Z. Curcumin up regulates T helper 1 cells in patients with colon cancer. Am. J. Transl. Res. 2017, 9, 1866–1875. [Google Scholar]
- Zou, J.Y.; Su, C.H.; Luo, H.H.; Lei, Y.Y.; Zeng, B.; Zhu, H.S.; Chen, Z.G. Curcumin converts Foxp3+ regulatory T cells to helper 1 cells in patients with lung cancer. J. Cell. Biochem. 2018, 119, 1420–1428. [Google Scholar] [CrossRef]
- Liao, F.; Liu, L.; Luo, E.; Hu, J. Curcumin enhances anti-tumor immune response in tongue squamous cell carcinoma. Arch. Oral Biol. 2018, 92, 32–37. [Google Scholar] [CrossRef]
- Liu, L.; Lim, M.A.; Jung, S.-N.; Oh, C.; Won, H.-R.; Jin, Y.L.; Piao, Y.; Kim, H.J.; Chang, J.W.; Koo, B.S. The effect of curcumin on multi-level immune checkpoint blockade and T cell dysfunction in head and neck cancer. Phytomedicine 2021, 92, 153758. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, T.; Yaguchi, T.; Kawakami, Y. Enhanced anti-tumor effects of the PD-1 blockade combined with a highly absorptive form of curcumin targeting STAT3. Cancer Sci. 2020, 111, 4626–4635. [Google Scholar] [CrossRef] [PubMed]
- Pouliquen, D.L.; Boissard, A.; Henry, C.; Blandin, S.; Richomme, P.; Coqueret, O.; Guette, C. Curcumin treatment identifies therapeutic targets within biomarkers of liver colonization by highly invasive mesothelioma cells—potential links with sarcomas. Cancers 2020, 12, 3384. [Google Scholar] [CrossRef]
- Madden, K.; Flowers, L.; Salani, R.; Horowitz, I.; Logan, S.; Kowalski, K.; Xie, J.; Mohammed, S.I. Proteomics-based approach to elucidate the mechanism of antitumor effect of curcumin in cervical cancer. Prostaglandins Leukot. Essent. Fatty Acids 2009, 80, 9–18. [Google Scholar] [CrossRef]
- D’Aguanno, S.; D’Agnano, I.; De Canio, M.; Rossi, C.; Bernardini, S.; Federici, G.; Urbani, A. Shotgun proteomics and network analysis of neuroblastoma cell lines treated with curcumin. Mol. Biosyst. 2012, 8, 1068–1077. [Google Scholar] [CrossRef]
- Cai, X.Z.; Huang, W.Y.; Qiao, Y.; Du, S.Y.; Chen, Y.; Chen, D.; Yu, S.; Che, R.C.; Liu, N.; Jiang, Y. Inhibitory effects of curcumin on gastric cells: A proteomic study of molecular targets. Phytomedicine 2013, 20, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Ohnuma, K.; Uchiyama, M.; Yamochi, T.; Nishibashi, K.; Hosono, O.; Takahashi, N.; Kina, S.; Tanaka, H.; Lin, X.; Dang, N.H.; et al. Caveolin-1 triggers T-cell activation via CD26 in association with CARMA1. J. Biol. Chem. 2007, 282, 10117–10131. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Outschoorn, U.E.; Whitaker-Menezes, D.; Lin, Z.; Flomenberg, N.; Howell, A.; Pestell, R.G.; Lisanti, M.P.; Sotgia, F. Cytokine production and inflammation drive autophagy in the tumor microenvironment role of stromal caveolin-1 as a key regulator. Cell Cycle 2011, 10, 1784–1793. [Google Scholar] [CrossRef]
- Oyarce, C.; Cruz-Gomez, S.; Galvez-Cancino, F.; Vargas, P.; Moreau, H.D.; Diaz-Valdivia, N.; Diaz, J.; Salazar-Onfray, F.A.; Pacheco, R.; Lennon-Dumenil, A.M.; et al. Caveolin-1 expression increases upon maturation in dendritic cells and promotes their migration to lymph nodes thereby favoring the induction of CD8+ T cell responses. Front. Immunol. 2017, 8, 1794. [Google Scholar] [CrossRef]
- Schaffer, A.-M.; Minguet, S. Caveolin-1, tetraspanin CD81 and flotillins in lymphocyte cell membrane organization, signaling and immunopathology. Biochem. Soc. Trans. 2020, 48, 2387–2397. [Google Scholar] [CrossRef]
- Song, Y.; Du, Y.; Qin, C.; Liang, H.; Yang, W.; Lin, J.; Ding, M.; Han, J.; Xu, T. Gemcitabine-resistant biomarkers in bladder cancer are associated with tumor-immune microenvironment. Front. Cell Develop. Biol. 2022, 9, 809620. [Google Scholar] [CrossRef] [PubMed]
- Pavlides, S.; Whitaker-Menezes, D.; Castello-Cros, R.; Flomengerg, N.; Witkiewicz, A.K.; Frank, P.G.; Casimiro, M.C.; Wang, C.; Fortina, P.; Addya, S.; et al. The reverse Warburg effect: Aerobic glycolysis in cancer associated fibroblasts and the tumor stroma. Cell Cycle 2009, 8, 3984–4001. [Google Scholar] [CrossRef] [PubMed]
- Saeed-Vafa, D.; Marchion, D.C.; McCarthy, S.M.; Hakam, A.; Lopez, A.; Wenham, R.M.; Apte, S.M.; Chen, D.-T.; Magliocco, A.M.; Lancaster, J.M.; et al. Utilizing digital pathology to quantify stromal caveolin-1 expression in malignant and benign ovarian tumors: Associations with clinicopathological parameters and clinical outcomes. PLoS ONE 2021, 16, e0256615. [Google Scholar] [CrossRef]
- Nwosu, Z.C.; Ebert, M.P.; Dooley, S.; Meyer, C. Caveolin-1 in the regulation of cell metabolism: A cancer perspective. Mol. Cancer 2016, 15, 71. [Google Scholar] [CrossRef]
- Wang, S.; Wang, N.; Zheng, Y.; Yang, B.; Liu, P.; Zhang, F.; Li, M.; Song, J.; Chang, X.; Wang, Z. Caveolin-1 inhibits breast cancer stem cells via c-Myc-mediated metabolic reprogramming. Cell Death Dis. 2020, 11, 450. [Google Scholar] [CrossRef] [PubMed]
- Simón, L.; Campos, A.; Leyton, L.; Quest, A.F.G. Caveolin-1 function at the plasma membrane and in intracellular compartments in cancer. Cancer Metast. Rev. 2020, 39, 435–453. [Google Scholar] [CrossRef]
- Díaz, M.I.; Díaz, P.; Castillo Bennett, J.; Urra, H.; Ortiz, R.; Contreras Orellana, P.; Hetz, C.; Quest, A.F.G. Caveolin-1 suppresses tumor formation through the inhibition of the unfolded protein response. Cell Death Dis. 2020, 11, 648. [Google Scholar] [CrossRef]
- Yang, L.; Wu, H.; Zhu, Y.; Chen, X.; Chen, Y. Plasma exosomal caveolin-1 predicts poor prognosis in ovarian cancer. J. Cancer 2021, 12, 5005–5012. [Google Scholar] [CrossRef]
- Yin, D.; Qiu, J.; Hu, S.; Cheng, L.; Li, H.; Cheng, X.; Wang, S.; Lu, J. CAV1 is a prognostic predictor for patients with idiopathic pulmonary fibrosis and lung cancer. J. Biosci. 2022, 47, 13. [Google Scholar] [CrossRef]
- Yang, Q.; Hao, J.; Chi, M.; Wang, Y.; Li, J.; Huang, J.; Zhang, J.; Zhang, M.; Lu, J.; Zhou, S.; et al. D2HGDH-mediated D2HG catabolism enhances the anti-tumor activities of CAR-T cells in an immunosuppressive microenvironment. Mol. Ther. 2022, 30, 1188–1200. [Google Scholar] [CrossRef]
- Zerenturk, E.J.; Sharpe, L.J.; Ikonen, E.; Brown, A.J. Desmosterol and DHCR24: Unexpected new directions for a terminal step in cholesterol synthesis. Progr. Lipid Res. 2013, 52, 666–680. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Bai, J.; Chen, B. Overexpression of EPDR1 has an antitumorigenic effect on breast cancer in vitro. Int. J. Clin. Exp. Pathol. 2020, 13, 2628–2636. Available online: http://www-ncbi-nlm-nih-gov.proxy.insermbiblio.inist.fr/pmc/articles/pmc7642719/ (accessed on 4 May 2022). [PubMed]
- Zhao, Z.; Wang, Z.; Wang, P.; Liu, S.; Li, Y.; Yang, X. EPDR1, which is negatively regulated by miR-429, suppresses epithelial ovarian cancer progression via PI3K/AKT signaling pathway. Front. Oncol. 2021, 11, 751567. [Google Scholar] [CrossRef] [PubMed]
- Aakula, A.; Kohonen, P.; Leivonen, S.-K.; Mäkelä, R.; Hintsanen, P.; Mpindi, J.P.; Martens-Uzunova, E.; Aittokallio, T.; Jenster, G.; Perälä, M.; et al. Systematic identification of microRNAs that impact on proliferation of prostate cancer cells and display changed expression in tumor tissue. Eur. Urol. 2016, 69, 1120–1128. [Google Scholar] [CrossRef] [PubMed]
- Kokate, S.B.; Dixit, P.; Poirah, I.; Roy, A.D.; Chakraborty, D.; Rout, N.; Singh, S.P.; Ashktorab, H.; Smoot, D.T.; Bhattacharyya, A. Testin and filamin-C downregulation by acetylated Siah2 increases invasiveness of Helicobacter pylori-infected gastric cancer cells. Int. J. Biochem. Cell Biol. 2018, 103, 14–24. [Google Scholar] [CrossRef]
- Duong-Ly, K.C.; Kuo, Y.-M.; Johnson, M.C.; Cote, J.M.; Kollman, J.M.; Soboloff, J.; Rall, G.F.; Andrews, A.J.; Peterson, J.R. T cell activation triggers reversible inosine-5′-monophosphate dehydrogenase assembly. J. Cell Sci. 2018, 131, jcs223289. [Google Scholar] [CrossRef] [PubMed]
- Tunbak, H.; Enriquez-Gasca, R.; Tie, C.H.C.; Gould, P.A.; Mlcochova, P.; Gupta, R.K.; Fernandes, L.; Holt, J.; van der Veen, A.G.; Giampazolias, E.; et al. The HUSH complex is a gatekeeper of type I interferon through epigenetic regulation of LINE-1s. Nat. Commun. 2020, 11, 5387. [Google Scholar] [CrossRef]
- Valli, A.; Rodriguez, M.; Moutsianas, L.; Fischer, R.; Fedele, V.; Huang, H.-L.; van Stiphout, R.; Jones, D.; McCarthy, M.; Vinaxia, M.; et al. Hypoxia induces a lipogenic cancer cell phenotype via HIF1α-dependent and -independent pathways. Oncotarget 2015, 6, 1920–1941. [Google Scholar] [CrossRef]
- Guo, J.; Li, X.; Shen, S.; Wu, X. Expression of immune-related genes as prognostic biomarkers for the assessment of osteosarcoma clinical outcomes. Sci. Rep. 2021, 11, 24123. [Google Scholar] [CrossRef]
- Butera, A.; Roy, M.; Zampieri, C.; Mammarella, E.; Panatta, E.; Melino, G.; D’Alessandro, A.; Amelio, I. p53-driven lipidome influences non-cell-autonomous lysophospholipids in pancreatic cancer. Biol. Direct 2022, 17, 6. [Google Scholar] [CrossRef]
- Sobotka, R.; Čapoun, O.; Kalousová, M.; Hanuš, T.; Zima, T.; Koštířová, M.; Soukup, V. Prognostic importance of vitamins A, E and retinol-binding protein 4 in renal cell carcinoma patients. Anticancer Res. 2017, 37, 3801–3806. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Li, M.; Wang, Z.; Zhu, L.; Shui, Y.; Zhang, S.; Guo, W. Down-regulation of RBP4 indicates a poor prognosis and correlates with immune cell infiltration in hepatocellular carcinoma. Biosci. Rep. 2021, 41, BSR20210328. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Li, Q.; Wang, H. Identification of novel biomarkers associated with the prognosis and potential pathogenesis of breast cancer via integrated bioinformatics analysis. Technol. Cancer Res. Treat. 2021, 20, 1–16. [Google Scholar] [CrossRef]
- Guo, W.; Zhang, C.; Wang, X.; Dou, D.; Chen, D.; Li, J. Resolving the difference between left-sided and right-sided colorectal cancer by single-cell sequencing. JCI Insight 2022, 7, e152616. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Liu, B.; Wei, J. Beta2-microglobulin (B2M) in cancer immunotherapies: Biological function, resistance and remedy. Cancer Lett. 2021, 517, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Mahauad-Fernandez, W.D.; Naushad, W.; Panzner, T.D.; Bashir, A.; Lal, G.; Okeoma, C.M. BST-2 promotes survival in circulation and pulmonary metastatic seeding of breast cancer cells. Sci. Rep. 2018, 8, 17608. [Google Scholar] [CrossRef]
- Balança, C.-C.; Salvioni, A.; Scarlata, C.-M.; Michelas, M.; Martinez-Gomez, C.; Gomez-Roca, C.; Sarradin, V.; Tosolini, M.; Valle, C.; Pont, F.; et al. PD-1 blockade restores helper activity of tumor-infiltrating, exhausted PD-1hiCD39+CD4 T cells. JCI Insight 2021, 6, e142513. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Min, Y.K.; Jang, J.; Park, H.; Lee, S.; Lee, C.H. Single-cell RNA sequencing reveals distinct cellular factors for response to immunotherapy targeting CD73 and PD-1 in colorectal cancer. J. Immunother. Cancer 2021, 9, e002503. [Google Scholar] [CrossRef]
- Pang, L.; Ng, K.T.-P.; Liu, J.; Yeung, W.-H.O.; Zhu, J.; Chiu, T.-L.S.; Liu, H.; Chen, Z.; Lo, C.-M.; Man, K. Plasmacytoid dendritic cells recruited by HIF-1α/eADO/ADORA1 signaling induce immunosuppression in hepatocellular carcinoma. Cancer Lett. 2021, 522, 80–92. [Google Scholar] [CrossRef]
- Whitley, M.J.; Suwanpradid, J.; Lai, C.; Jiang, S.W.; Cook, J.L.; Zelac, D.E.; Rudolph, R.; Corcoran, D.L.; Degan, S.; Spasojevic, I.; et al. ENTPD1 (CD39) expression inhibits UVR-induced DNA damage repair through purinergic signaling and is associated with metastasis in human cutaneous squamous cell carcinoma. J. Investig. Dermatol. 2021, 141, 2509–2520. [Google Scholar] [CrossRef]
- Zhan, Y.; Zheng, L.; Liu, J.; Hu, D.; Wang, J.; Liu, K.; Guo, J.; Zhang, T.; Kong, D. PLA2G4A promotes right-sided colorectal cancer progression by inducing CD39+γδ Treg polarization. JCI Insight 2021, 6, e148028. [Google Scholar] [CrossRef] [PubMed]
- Tavares, S.; Vieira, A.F.; Taubenberger, A.V.; Araújo, M.; Martins, N.P.; Brás-Pereira, C.; Polónia, A.; Herbig, M.; Barreto, C.; Otto, O.; et al. Actin stress fiber organization promotes cell stiffening and proliferation of pre-invasive breast cancer cells. Nat. Commun. 2017, 8, 15237. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Xu, L.; Li, X.; Chu, Y.; Jiang, M.; Xu, B.; Zhao, M.; Wang, W.; Wang, H.; Kang, H.; et al. Enah overexpression is correlated with poor survival and aggressive phenotype in gastric cancer. Cell Death Dis. 2018, 9, 998. [Google Scholar] [CrossRef]
- Xu, Y.; Liu, R.; Liao, C.; Liu, J.; Zhao, H.; Li, Z.; Liu, W.; Chen, L.; Wu, C.; Tan, H.; et al. High expression of immunity-related GTPase family M protein in glioma promotes cell proliferation and autophagy protein expression. Pathol. Res. Pract. 2019, 215, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Meng, H.; Dong, X.; Li, X.; Shi, Z.; Li, H.; Zhang, L.; Yang, Y.; Liu, R.; Pei, C.; et al. IRGM promotes melanoma cell survival through autophagy and is a promising prognostic biomarker for clinical application. Mol. Ther. Oncolytics 2021, 20, 187–198. [Google Scholar] [CrossRef]
- He, X.; Huang, Q.; Qiu, X.; Liu, X.; Sun, G.; Guo, J.; Ding, Z.; Yang, L.; Ban, N.; Tao, T.; et al. LAP3 promotes glioma progression by regulating proliferation, migration and invasion of glioma cells. Int. J. Biol. Macromol. 2015, 72, 1081–1089. [Google Scholar] [CrossRef]
- Fukamachi, M.; Kasamatsu, A.; Endo-Sakamoto, Y.; Fushimi, K.; Kasama, H.; Iyoda, M.; Minakawa, Y.; Shiiba, M.; Tanzawa, H.; Uzawa, K. Multiple coagulation factor deficiency protein 2 as a crucial component in metastasis of human oral cancer. Exp. Cell Res. 2018, 368, 119–125. [Google Scholar] [CrossRef]
- Navas, L.E.; Carnero, A. NAD+ metabolism, stemness, the immune response, and cancer. Signal Transduct. Target. Ther. 2021, 6, 2. [Google Scholar] [CrossRef]
- Nacarelli, T.; Fukumoto, T.; Zundell, J.A.; Fatkhutdinov, N.; Jean, S.; Cadungog, M.G.; Borowsky, M.E.; Zhang, R. NAMPT inhibition suppresses cancer stem-like cells associated with therapy-induced senescence in ovarian cancer. Cancer Res. 2020, 80, 890–900. [Google Scholar] [CrossRef]
- Kumar, R.; Li, D.-Q. PAKs in human cancer progression: From inception to cancer therapeutic to future oncobiology. Adv. Cancer Res. 2016, 130, 137–209. [Google Scholar] [CrossRef]
- Tao, Y.; Hu, K.; Tan, F.; Zhang, S.; Zhou, M.; Luo, J.; Wang, Z. SH3-domain binding protein 1 in the tumor microenvironment promotes hepatocellular carcinoma metastasis through WAVE2 pathway. Oncotarget 2016, 7, 18356–18370. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Zhong, W.; Shi, Z.; Wang, K.; Jin, H.; Zhang, Z.; Wang, H.; Wei, Y.; Chen, S.; Zhou, Q.; et al. Overexpression of signal sequence receptor predicts poor survival in patients with hepatocellular carcinoma. Hum. Pathol. 2018, 81, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Zhai, J.; You, Q.; Zhang, G.; He, M.; Yao, X.; Shen, L. Cancer-associated fibroblasts-derived VCAM1 induced by H. pylori infection facilitates tumor invasion in gastric cancer. Oncogene 2020, 39, 2961–2974. [Google Scholar] [CrossRef]
- Kabir, A.; Muth, A. Polypharmacology: The science of multi-targeting molecules. Pharmacol. Res. 2022, 176, 106055. [Google Scholar] [CrossRef]
- Azeloglu, E.U.; Iyengar, R. Signaling networks: Information flow, computation, and decision making. Cold Spring Harb. Perspect. Biol. 2015, 7, a005934. [Google Scholar] [CrossRef] [PubMed]
- Pouliquen, D.L.; Boissard, A.; Henry, C.; Blandin, S.; Coqueret, O.; Guette, C. Lymphoid organ proteomes identify therapeutic efficacy biomarkers following the intracavitary administration of curcumin in a highly invasive rat model of peritoneal mesothelioma. Int. J. Mol. Sci. 2021, 22, 8566. [Google Scholar] [CrossRef] [PubMed]
- Pouliquen, D.L.; Boissard, A.; Henry, C.; Coqueret, O.; Guette, C. Curcuminoids as modulators of EMT in invasive cancers: A review of molecular targets with the contribution of malignant mesothelioma studies. Front. Pharmacol. 2022, 13, 934534. [Google Scholar] [CrossRef]
- Waki, K.; Kawano, K.; Tsuda, N.; Komatsu, N.; Yamada, A. CD4/CD8 ratio is a prognostic factor in IgG nonresponders among peptide vaccine-treated ovarian cancer patients. Cancer Sci. 2020, 111, 1124–1131. [Google Scholar] [CrossRef]
- Zurlo, I.V.; Schino, A.; Strippoli, A.; Calegari, M.A.; Cocomazzi, A.; Cassano, A.; Pozzo, C.; Di Salvatore, M.; Ricci, R.; Barone, C.; et al. Predictive value of NLR, TILs (CD4+/CD8+) and PD-L1 expression for prognosis and response to preoperative chemotherapy in gastric cancer. Cancer Immunol. Immunother. 2022, 71, 45–55. [Google Scholar] [CrossRef]
- Brog, R.A.; Ferry, S.L.; Schiebout, C.T.; Messier, C.M.; Cook, W.J.; Abdullah, L.; Zou, J.; Kumar, P.; Sentman, C.L.; Frost, H.R.; et al. Superkine IL-2 and IL-33 armored CAR T cells reshape the tumor microenvironment and reduce growth of multiple solid tumors. Cancer Immunol. Res. 2022, 10, 962–977. [Google Scholar] [CrossRef]
- Wang, H.; Zhou, J.; Li, J.; Geng, Y.; Meng, P.; Ma, C.; Zhu, Z.; Zhang, W.; Hong, L.; Quan, Y.; et al. A study of multinucleated giant cells in esophageal cancer. Clin. Immunol. 2021, 222, 108600. [Google Scholar] [CrossRef] [PubMed]
- Sedlacek, A.L.; Younker, T.P.; Zhou, Y.J.; Borghesi, L.; Shcheglova, T.; Mandoiu, I.I.; Binder, R.J. CD91 on dendritic cells governs immunosurveillance of nascent, emerging tumors. JCI Insight 2019, 4, e127239. [Google Scholar] [CrossRef] [PubMed]
- Lim, E.-J.; Kang, J.-H.; Kim, Y.-J.; Kim, S.; Lee, S.-J. ICAM-1 promotes cancer progression by regulating SRC activity as an adapter protein in colorectal cancer. Cell Death Dis. 2022, 13, 417. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Fang, D.; Zhou, W. CCR2 and PTPRC are regulators of tumor microenvironment and potential prognostic biomarkers of lung adenocarcinoma. Ann. Transl. Med. 2021, 9, 1419. [Google Scholar] [CrossRef] [PubMed]
- Rolny, C.; Mazzone, M.; Tugues, S.; Laoui, D.; Johansson, I.; Coulon, C.; Squadrito, M.L.; Segura, I.; Li, X.; Knevels, E.; et al. HRG inhibits tumor growth and metastasis by inducing macrophage polarization and vessel normalization through downregulation of PIGF. Cancer Cell 2011, 19, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Roche, F.P.; Pietilä, I.; Kaito, H.; Sjöström, E.O.; Sobotski, N.; Noguer, O.; Skare, T.P.; Essand, M.; Wollscheid, B.; Welsh, M.; et al. Leukocyte differentiation by histidine-rich glycoprotein/stanniocalcin-2 complex regulates murine glioma growth through modulation of antitumor immunity. Mol. Cancer Ther. 2018, 17, 1961–1972. [Google Scholar] [CrossRef]
- Nishimura, Y.; Wake, H.; Teshigawara, K.; Wang, D.; Sakaguchi, M.; Otsuka, F.; Nishibori, M. Histidine-rich glycoprotein augments natural killer cell function by modulating PD-1 expression via CLEC-1B. Pharmacol. Res. Perspect. 2019, 7, e00481. [Google Scholar] [CrossRef]
- Roche, F.; Sipilä, K.; Honjo, S.; Johansson, S.; Tugues, S.; Heino, J.; Claesson-Welsh, L. Histidine-rich glycoprotein blocks collagen-binding integrins and adhesion of endothelial cells through low-affinity interaction with α2 integrin. Matrix Biol. 2015, 48, 89–99. [Google Scholar] [CrossRef]
- Pastor, T.P.; Peixoto, B.C.; Viola, J.P.B. The transcriptional co-factor IRF2BP2: A new player in tumor development and microenvironment. Front. Cell Develop. Biol. 2021, 9, 655307. [Google Scholar] [CrossRef]
- Marcogliese, P.C.; Dutta, D.; Ray, S.S.; Dang, N.D.P.; Zuo, Z.; Wang, Y.; Lu, D.; Fazal, F.; Ravenscroft, T.A.; Chung, H.; et al. Loss of IRF2BPL impairs neuronal maintenance through excess Wnt signaling. Sci. Adv. 2022, 8, eabl5613. [Google Scholar] [CrossRef]
- Xu, W.; Liu, W.-R.; Xu, Y.; Tian, X.; Anwaier, A.; Su, J.-Q.; Zhu, W.-K.; Shi, G.-H.; Wei, G.-M.; Huang, Y.-P.; et al. Hexokinase 3 dysfunction promotes tumorigenesis and immune escape by upregulating monocyte/macrophage infiltration into the clear cell renal cell carcinoma microenvironment. Int. J. Biol. Sci. 2021, 17, 2205–2222. [Google Scholar] [CrossRef] [PubMed]
- Xia, E.; Zhou, X.; Bhandari, A.; Zhang, X.; Wang, O. Synaptopodin-2 plays an important role in the metastasis of breast cancer via PI3K/Akt/mTOR pathway. Cancer Manag. Res. 2018, 10, 1575–1583. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ye, L.; Li, Q.; Wu, X.; Wang, B.; Ouyang, Y.; Yuan, Z.; Li, J.; Lin, C. Synaptopodin-2 suppresses metastasis of triple-negative breast cancer via inhibition of YAP/TAZ activity. J. Pathol. 2018, 244, 71–83. [Google Scholar] [CrossRef] [PubMed]
- OuYang, C.; Xie, Y.; Fu, Q.; Xu, G. SYNPO2 suppresses hypoxia-induced proliferation and migration of colorectal cancer cells by regulating YAP-KLF5 axis. Tissue Cell 2021, 73, 101598. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Yang, L.; Qin, Y.; Liu, S.; Qiao, Y.; Wan, X.; Zeng, H.; Tang, X.; Liu, M.; Hou, Y. Effects of differential distributed-JUP on the malignancy of gastric cancer. J. Adv. Cancer 2020, 28, 195–208. [Google Scholar] [CrossRef]
- Chen, K.; Zeng, J.; Sun, Y.; Ouyang, W.; Yu, G.; Zhou, H.; Zhang, Y.; Yao, W.; Xiao, W.; Hu, J.; et al. Junction plakoglobin regulates and destabilizes HIF2a to inhibit tumorigenesis of renal cell carcinoma. Cancer Commun. 2021, 41, 316–332. [Google Scholar] [CrossRef]
- Ha, J.H.; Jayaraman, M.; Yan, M.; Dhanasekaran, P.; Isidoro, C.; Song, Y.S.; Dhanasekaran, D.N. GNAi2/gip2-regulated transcriptome and its therapeutic significance in ovarian cancer. Biomolecules 2021, 11, 1211. [Google Scholar] [CrossRef]
- Mammoto, A.; Connor, K.M.; Mammoto, T.; Yung, C.W.; Huh, D.; Aderman, C.M.; Mostoslavsky, G.; Smith, L.E.H.; Ingber, D.E. A mechanosensitive transcriptional mechanism that controls angiogenesis. Nature 2009, 457, 1103–1108. [Google Scholar] [CrossRef]
- Low, J.-H.; Brennen, W.N.; Meeker, A.K.; Ikonen, E.; Simons, B.W.; Laiho, M. Stromal CAVIN1 controls prostate cancer microenvironment and metastasis by modulating lipid distribution and inflammatory signaling. Mol. Cancer Res. 2020, 18, 1414–1426. [Google Scholar] [CrossRef]
- Nader, J.S.; Guillon, J.; Petit, C.; Boissard, A.; Franconi, F.; Blandin, S.; Lambot, S.; Grégoire, M.; Verrièle, V.; Nawrocki-Raby, B.; et al. S100A4 is a biomarker of tumorigenesis, EMT, invasion, and colonization of host organs in experimental malignant mesothelioma. Cancers 2020, 12, 939. [Google Scholar] [CrossRef]
| Gene | Authors (Year) | Main Findings |
|---|---|---|
| Cav1 | Ohnuma et al., 2007 [17] | Triggers T cell activation |
| Martinez-Outschoorn et al., 2011 [18] | Loss of stromal Cav1 predictor of poor outcome | |
| Oyarce et al., 2017 [19] | Increases maturation of dendritic cells | |
| Schaffer and Minguet, 2020 [20] | Lymphocyte cell membrane organization | |
| Song et al., 2022 [21] | Regulates immune cell infiltration | |
| Pavlides et al., 2009 [22] | Vimentin overexpression in tumor lacking Cav1 | |
| Saeed-Vafa et al., 2021 [23] | Loss of Cav1 in malignant ovarian carcinomas | |
| Nwosu et al., 2016 [24] | Cav1 modulates cell metabolism | |
| Wang S. et al., 2020 [25] | Inhibits breast cancer stem cells | |
| Simon et al., 2020 [26] | Regulates intracellular organelle communication | |
| Diaz et al., 2020 [27] | Inhibition of the unfolded protein response | |
| Yang et al., 2021 [28] | Exosomal Cav1 downregulated in ovarian cancer | |
| Yin et al., 2022 [29] | Worse survival in low Cav1 lung cancer patients | |
| D2hgdh | Yang et al., 2022 [30] | Overexpression increases CAR-T cell killing efficacy |
| Dhcr24 | Zerenturk et al., 2013 [31] | Involved in cholesterol biosynthesis |
| Epdr1 | Liang et al., 2020 [32] | Increases p53, p21 and Bcl-2 expression |
| Zhao et al., 2021 [33] | Tumor suppressor (PI3K/Akt signaling pathway) | |
| Flnc | Aakula et al., 2016 [34] | Increases free survival in prostate cancer patients |
| Kokate et al., 2018 [35] | Downregulation increases gastric cancer invasiveness | |
| Impdh1 | Duong-Ly et al., 2018 [36] | Upregulated in T cell activation |
| Mphosph8 | Tunbak et al., 2020 [37] | Frequently downregulated in diverse cancers |
| Pld3 | Valli et al., 2014 [38] | Reduced PLD3 levels in hypoxia |
| Guo et al., 2021 [39] | Better prognosis in high PLD3 osteosarcoma | |
| Butera et al., 2022 [40] | Correlates with p53 status and prognosis | |
| Rbp4 | Sobotka et al., 2017 [41] | Poorer prognosis of renal carcinoma, low RBP4 |
| Li M. et al., 2021 [42] | Poor prognosis of liver cancer with low RBP4 | |
| Wu et al., 2021 [43] | Low expression level in breast cancer tissues | |
| Guo et al. 2022 [44] | Link with responsiveness to immunotherapy | |
| B2m | Wang H. et al., 2021 [45] | B2M-deficient tumors express activating NK ligands |
| Bst2 | Mahauad-Fernandez et al., 2018 [46] | Promotes survival in metastatic breast cancer |
| Entpd1 | Balança et al., 2021 [47] | Involved in tumor-infiltrating exhausted T cells |
| Kim et al., 2021 [48] | Entpd1-T cells have increased TCR diversities | |
| Pang et al., 2021 [49] | HIF-1 upregulates Entpd1 in liver cancer | |
| Whitley et al., 2021 [50] | Entpd1 expression linked with metastasis | |
| Zhan et al., 2021 [51] | Entpd1+ Tregs correlates with poor prognosis | |
| Evl | Tavares et al., 2017 [52] | Regulates F-actin and promotes cell stiffening |
| Chen et al., 2018 [53] | Upregulated in gastric cancer, link with stage | |
| Irgm | Xu et al., 2019 [54] | Upregulated in glioma, link with autophagy |
| Tian et al., 2020 [55] | Promotes melanoma cell survival via autophagy | |
| Lap3 | He et al., 2015 [56] | Promotes glioma progression and invasion |
| Mcfd2 | Fukamachi et al., 2018 [57] | Crucial component in oral cancer metastasis |
| Nampt | Navas and Carnero, 2021 [58] | Activates stemness and dedifferentiation |
| Nacarelli et al., 2022 [59] | NAMPT inhibition suppresses cancer stem-like cells | |
| Pak1 | Kumar and Li, 2016 [60] | Converging nexus for cancer-promoting signals |
| Sh3bp1 | Tao et al., 2016 [61] | Promotes tumor invasion/microvessel formation |
| Ssr3 | Huang et al., 2018 [62] | Predicts poor survival in patients with liver cancer |
| Vcam1 | Shen et al., 2020 [63] | Facilitates tumor invasion in gastric cancer |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pouliquen, D.L.; Malloci, M.; Boissard, A.; Henry, C.; Guette, C. Proteomes of Residual Tumors in Curcumin-Treated Rats Reveal Changes in Microenvironment/Malignant Cell Crosstalk in a Highly Invasive Model of Mesothelioma. Int. J. Mol. Sci. 2022, 23, 13732. https://doi.org/10.3390/ijms232213732
Pouliquen DL, Malloci M, Boissard A, Henry C, Guette C. Proteomes of Residual Tumors in Curcumin-Treated Rats Reveal Changes in Microenvironment/Malignant Cell Crosstalk in a Highly Invasive Model of Mesothelioma. International Journal of Molecular Sciences. 2022; 23(22):13732. https://doi.org/10.3390/ijms232213732
Chicago/Turabian StylePouliquen, Daniel L., Marine Malloci, Alice Boissard, Cécile Henry, and Catherine Guette. 2022. "Proteomes of Residual Tumors in Curcumin-Treated Rats Reveal Changes in Microenvironment/Malignant Cell Crosstalk in a Highly Invasive Model of Mesothelioma" International Journal of Molecular Sciences 23, no. 22: 13732. https://doi.org/10.3390/ijms232213732
APA StylePouliquen, D. L., Malloci, M., Boissard, A., Henry, C., & Guette, C. (2022). Proteomes of Residual Tumors in Curcumin-Treated Rats Reveal Changes in Microenvironment/Malignant Cell Crosstalk in a Highly Invasive Model of Mesothelioma. International Journal of Molecular Sciences, 23(22), 13732. https://doi.org/10.3390/ijms232213732







