Genome-Wide Identification of C2H2 ZFPs and Functional Analysis of BRZAT12 under Low-Temperature Stress in Winter Rapeseed (Brassica rapa)
Abstract
1. Introduction
2. Results
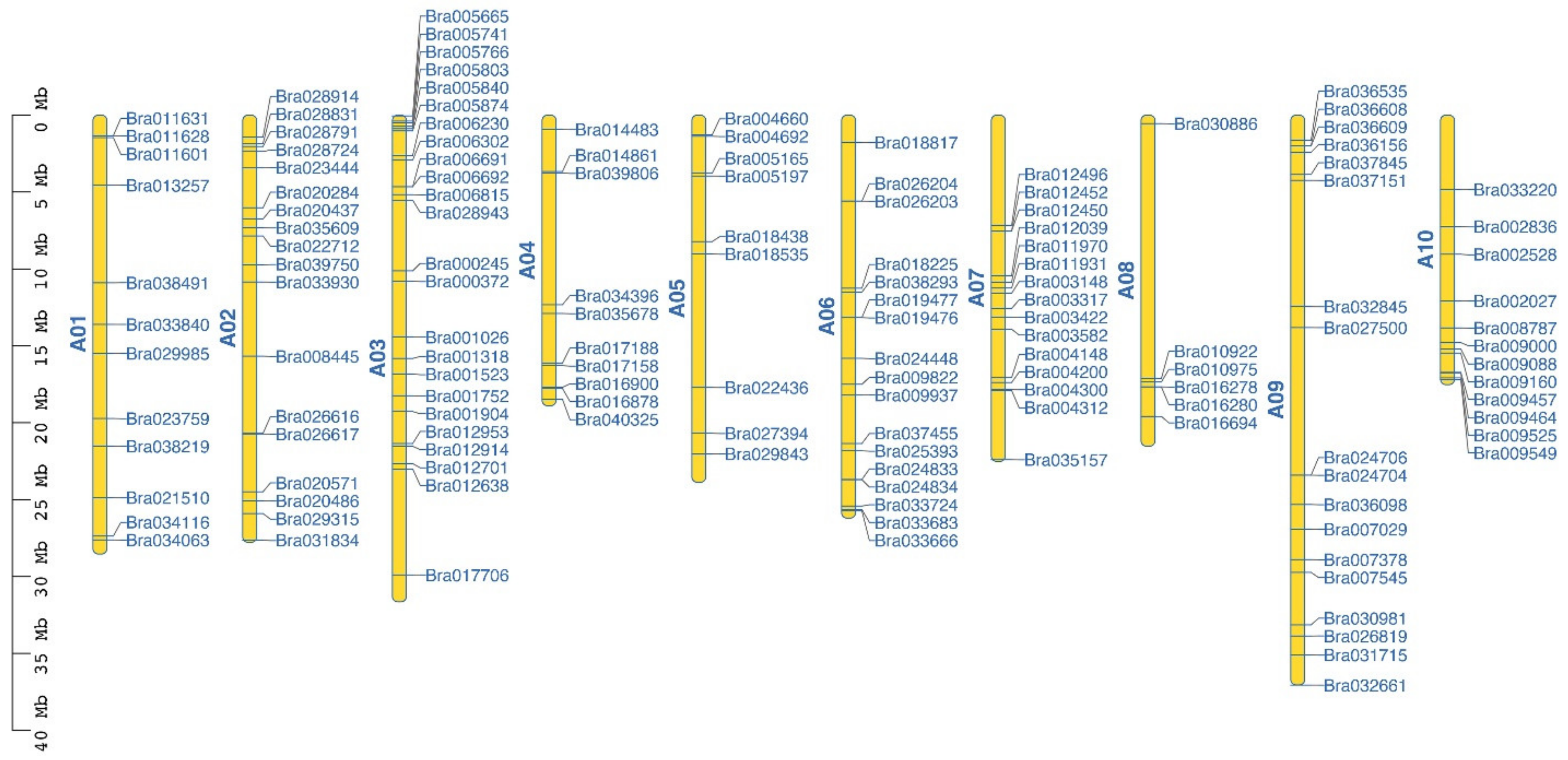
2.1. Chromosome Distribution of the C2H2 ZFP Gene Family
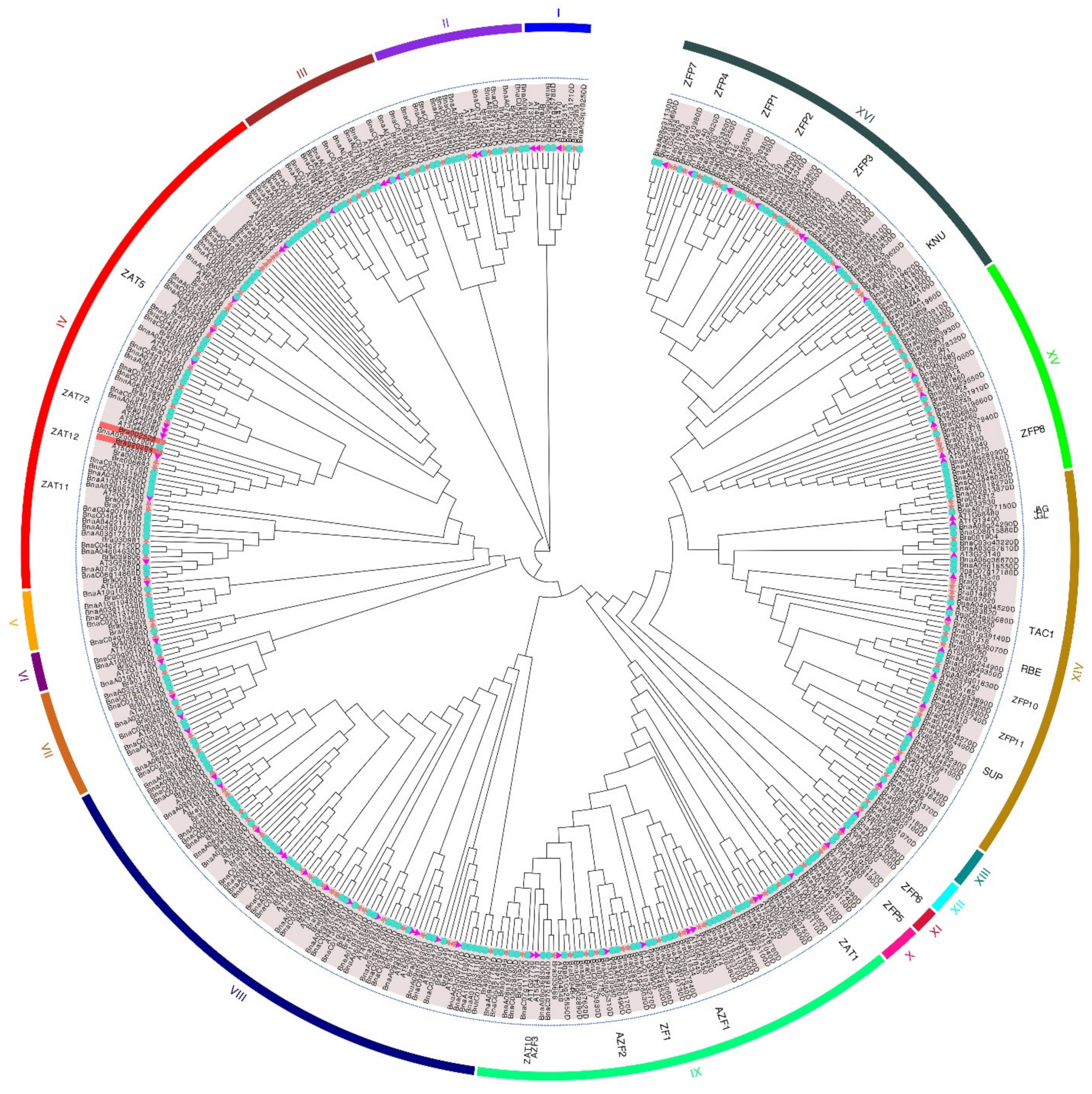
2.2. Phylogenetic Analysis of the C2H2 ZFP Gene Family
2.3. Gene Structure of the C2H2 ZFP Gene Family
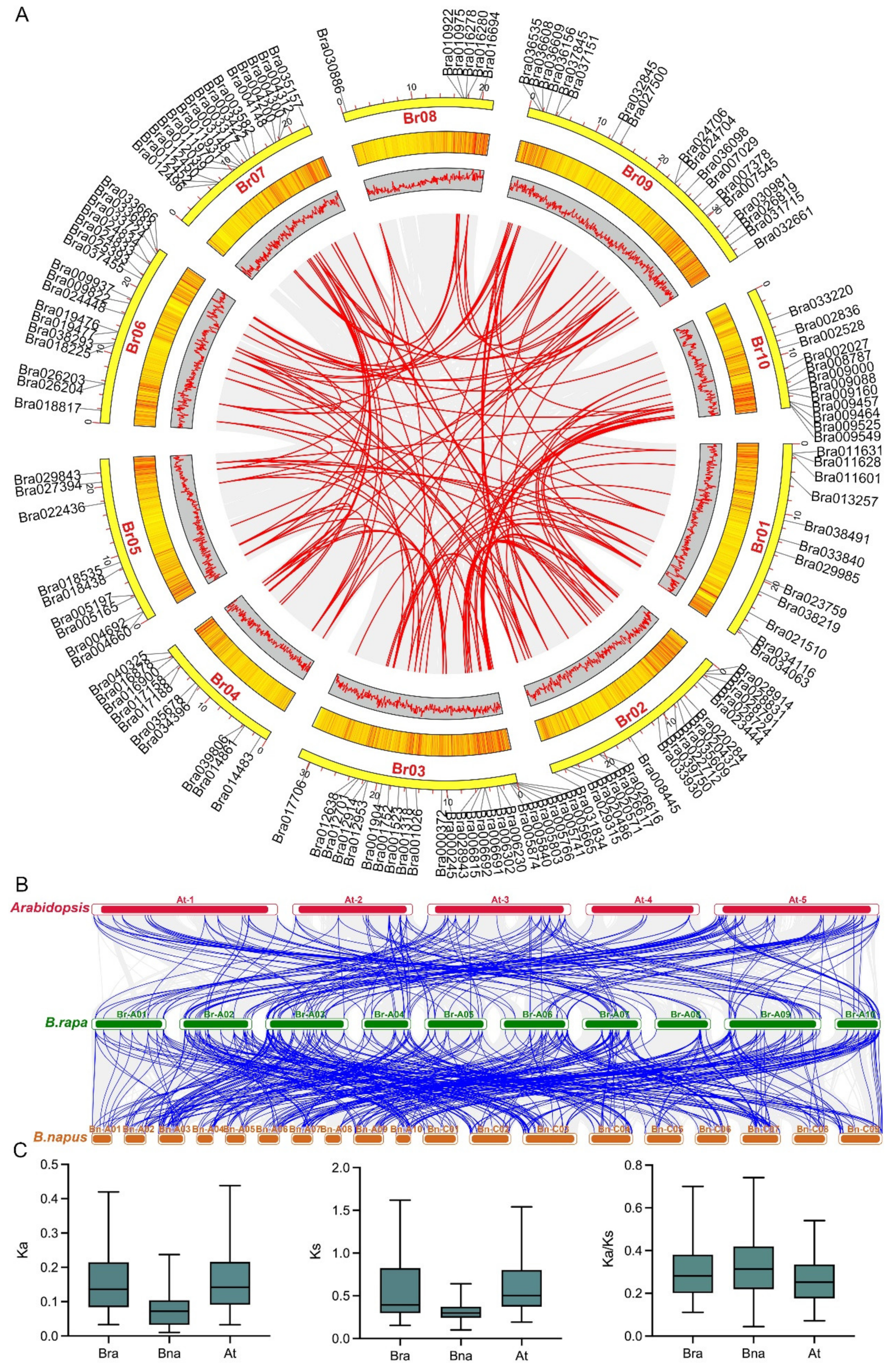
2.4. Genomic Collinearity Analysis of the C2H2 ZFP Gene Family
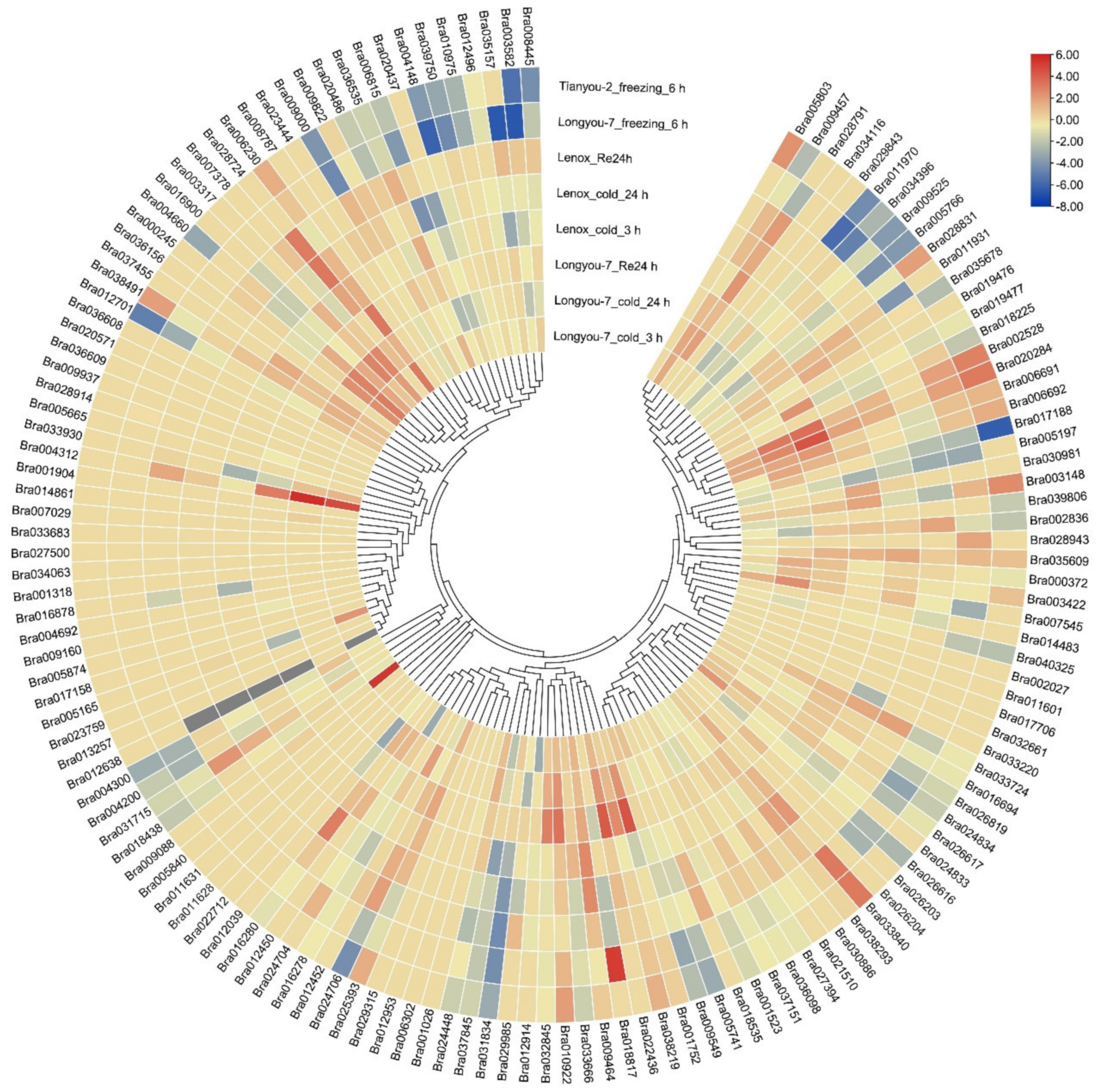
2.5. Analysis of the Gene Expression Characteristics of Members of the Gene Family
2.6. Analysis of the Expression Pattern of Gene Family Members
2.7. Screening and Identification of Transgenic Arabidopsis with the BrZAT12 Gene
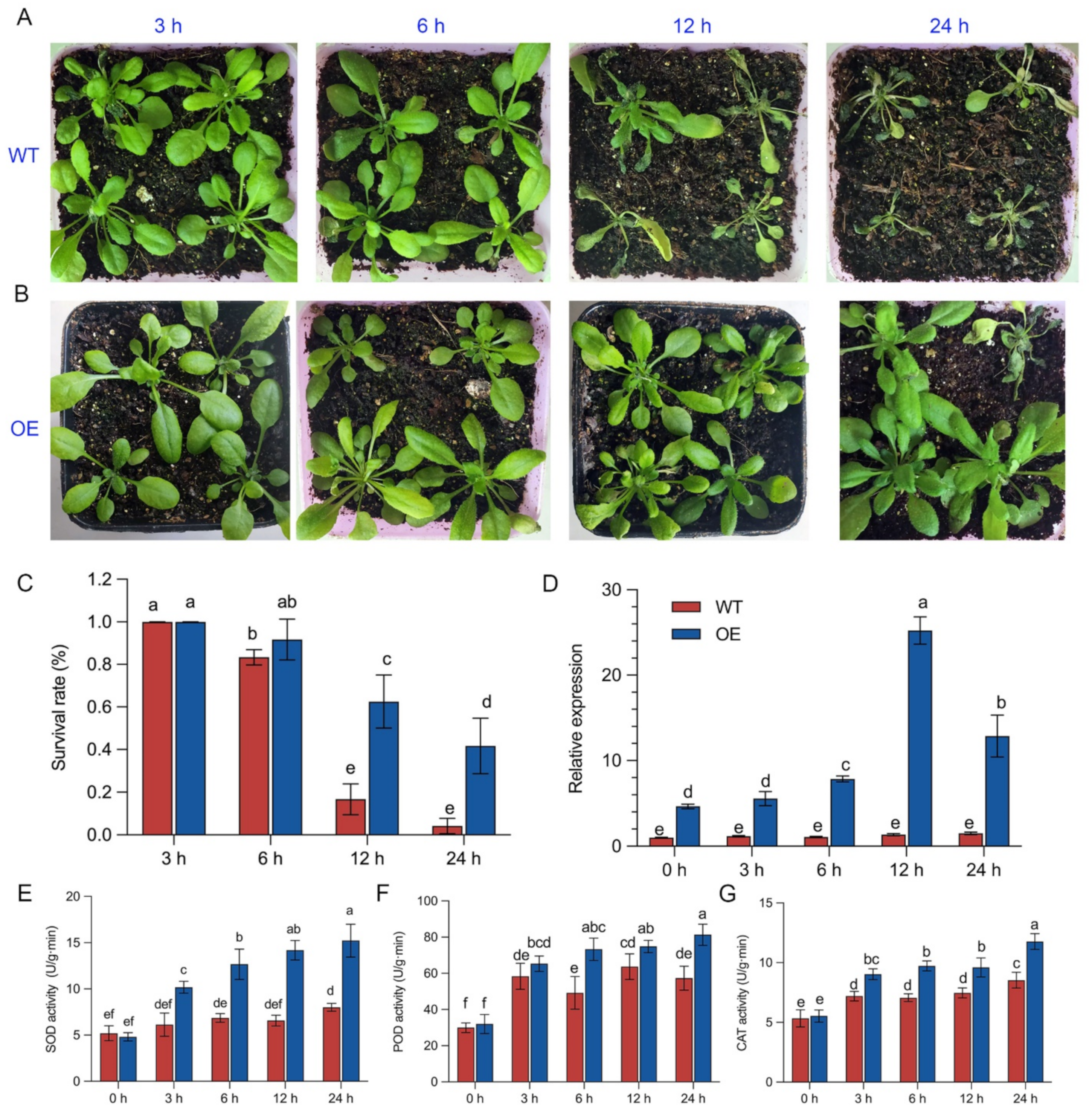
2.8. Phenotypic and Expression Analysis of BrZAT12-Transformed Plants under Low-Temperature Stress
2.9. Physiological-Activity Analysis of the BrZAT12 Gene in Transgenic Plants at Low Temperature
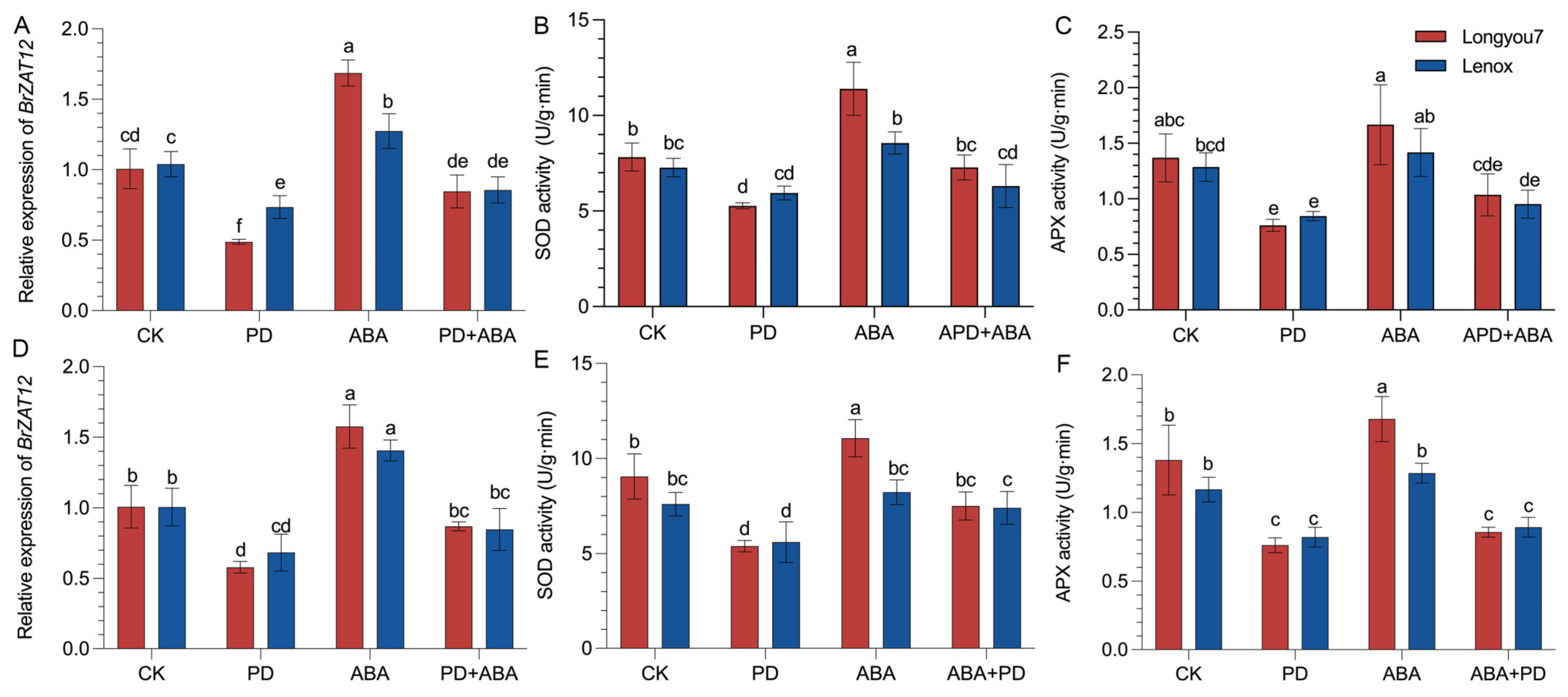
2.10. The BrZAT12 Gene Is Involved in the Regulation of ABA and MAPK Signaling Pathways
3. Discussion
4. Materials and Methods
4.1. Plant Growth and Treatments
4.2. Identification of Members of the C2H2 ZFP Gene Family
4.3. Analysis of Members of the C2H2 ZFP Gene Family
4.4. Collinear Analysis of Gene Family Members
4.5. RNA Isolation, Reverse Transcription, qRT-PCR, and Transcriptome Expression Analysis
4.6. Preliminary Verification of BrZAT12 Function
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| B. rapa | Brassica rapa |
| Arabidopsis | Arabidopsis thaliana |
| C2H2 ZFP | C2H2 zinc-finger protein |
| B. napus | Brassica napus |
| BrZAT12 | Brassica rapa zinc finger protein ZAT12-1 |
| qRT-PCR | Real-time fluorescence quantitative PCR |
| ABA | Abscisic acid |
| HMM | Hidden Markov model |
| Ks | Synonymous substitution rate |
| Ka | Non-synonymous substitution rate |
| POD | Peroxidase activity |
| SOD | Superoxide dismutase activity |
| CAT | Catalase activity |
References
- Wu, J.; Xu, X.; Liu, L.; Ma, L.; Pu, Y.; Wang, W.; Hua, X.; Song, J.; Liu, K.; Lu, G.; et al. Assembly of a Winter Turnip Rape (Brassica rapa L.) to Explore the Genetic Basis of Cold Tolerance. Front. Plant Sci. 2022, 13, 936958. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Qi, W.; Bai, J.; Li, H.; Fang, Y.; Xu, J.; Xu, Y.; Zeng, X.; Pu, Y.; Wang, W.; et al. Genome-Wide Identification and Analysis of the Ascorbate Peroxidase (APX) Gene Family of Winter Rapeseed (Brassica Rapa L.) Under Abiotic Stress. Front. Genet. 2022, 12, 753624. [Google Scholar] [CrossRef]
- Han, G.; Lu, C.; Guo, J.; Qiao, Z.; Sui, N.; Qiu, N.; Wang, B. C2H2 Zinc Finger Proteins: Master Regulators of Abiotic Stress Responses in Plants. Front. Plant Sci. 2020, 11, 115. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Shi, Y.; Yang, S. Advances and Challenges in Uncovering Cold Tolerance Regulatory Mechanisms in Plants. New Phytol. 2019, 222, 1690–1704. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhu, J.; Gong, Z.; Zhu, J.K. Abiotic Stress Responses in Plants. Nat. Rev. Genet. 2022, 23, 104–119. [Google Scholar] [CrossRef]
- Hassan, M.A.; Xiang, C.; Farooq, M.; Muhammad, N.; Yan, Z.; Hui, X.; Yuanyuan, K.; Bruno, A.K.; Lele, Z.; Jincai, L. Cold Stress in Wheat: Plant Acclimation Responses and Management Strategies. Front. Plant Sci. 2021, 12, 676884. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Ding, Y.; Yang, Y.; Song, C.; Wang, B.; Yang, S.; Guo, Y.; Gong, Z. Protein Kinases in Plant Responses to Drought, Salt, and Cold Stress. J. Integr. Plant Biol. 2021, 63, 53–78. [Google Scholar] [CrossRef]
- Hwang, I.; Jung, H.J.; Park, J.I.; Yang, T.J.; Nou, I.S. Transcriptome Analysis of Newly Classified BZIP Transcription Factors of Brassica Rapa in Cold Stress Response. Genomics 2014, 104, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Tian, Z.D.; Zhang, Y.; Liu, J.; Xie, C.H. Novel Potato C2H2-Type Zinc Finger Protein Gene, StZFP1, Which Responds to Biotic and Abiotic Stress, Plays a Role in Salt Tolerance. Plant Biol. 2010, 12, 689–697. [Google Scholar] [CrossRef]
- Xie, T.; Chen, C.; Li, C.; Liu, J.; Liu, C.; He, Y. Genome-Wide Investigation of WRKY Gene Family in Pineapple: Evolution and Expression Profiles during Development and Stress. BMC Genom. 2018, 19, 490. [Google Scholar] [CrossRef]
- Liu, Y.; Khan, A.R.; Gan, Y. C2H2 Zinc Finger Proteins Response to Abiotic Stress in Plants. Int. J. Mol. Sci. 2022, 23, 2730. [Google Scholar] [CrossRef]
- Selvaraj, M.G.; Jan, A.; Ishizaki, T.; Valencia, M.; Dedicova, B.; Maruyama, K.; Ogata, T.; Todaka, D.; Yamaguchi-Shinozaki, K.; Nakashima, K.; et al. Expression of the CCCH-Tandem Zinc Finger Protein Gene OsTZF5 under a Stress-Inducible Promoter Mitigates the Effect of Drought Stress on Rice Grain Yield under Field Conditions. Plant Biotechnol. J. 2020, 18, 1711–1721. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Ding, Y.; Cai, C.; Chen, Z.; Zhu, C. The Role of C2H2 Zinc Finger Proteins in Plant Responses to Abiotic Stresses. Physiol. Plant. 2019, 165, 690–700. [Google Scholar] [CrossRef] [PubMed]
- Wei, K.; Pan, S.; Li, Y. Functional Characterization of Maize C2H2 Zinc-Finger Gene Family. Plant Mol. Biol. Report. 2016, 34, 761–776. [Google Scholar] [CrossRef]
- Englbrecht, C.C.; Schoof, H.; Böhm, S. Conservation, Diversification and Expansion of C2H2 Zinc Finger Proteins in the Arabidopsis Thaliana Genome. BMC Genom. 2004, 5, 39. [Google Scholar] [CrossRef]
- Liu, Z.; Coulter, J.A.; Li, Y.; Zhang, X.; Meng, J.; Zhang, J.; Liu, Y. Genome-Wide Identification and Analysis of the Q-Type C2H2 Gene Family in Potato (Solanum tuberosum L.). Int. J. Biol. Macromol. 2020, 153, 327–340. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Chen, J.; Liu, M.; Zhang, H.; Zhang, S.; Liu, D.; Chen, S. Genome-Wide Analysis of C2H2 Zinc Finger Gene Family and Its Response to Cold and Drought Stress in Sorghum [Sorghum Bicolor (L.) Moench]. Int. J. Mol. Sci. 2022, 23, 5571. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Wu, T.; Zhang, J.; Wang, Z.; Pei, T.; Yang, H.; Li, J.; Xu, X. Genome-Wide Analyses of the Genetic Screening of C2H2-Type Zinc Finger Transcription Factors and Abiotic and Biotic Stress Responses in Tomato (Solanum Lycopersicum) Based on RNA-Seq Data. Front. Genet. 2020, 11, 540. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Wang, Z.; Xu, X.; Zhang, H.; Li, C. Genome-Wide Analysis of C2H2 Zinc-Finger Family Transcription Factors and Their Responses to Abiotic Stresses in Poplar (Populus Trichocarpa). PLoS ONE 2015, 10, e0134753. [Google Scholar] [CrossRef] [PubMed]
- Vogel, J.T.; Zarka, D.G.; Van Buskirk, H.A.; Fowler, S.G.; Thomashow, M.F. Roles of the CBF2 and ZAT12 Transcription Factors in Configuring the Low Temperature Transcriptome of Arabidopsis. Plant J. 2005, 41, 195–211. [Google Scholar] [CrossRef] [PubMed]
- Chandra Rai, A.; Singh, M.; Shah, K.; Rai, A.C.; Singh, I.; Singh, M.; Shah, K.; Chandra Rai, A.; Singh, M.; Shah, K. Effect of Water Withdrawal on Formation of Free Radical, Proline Accumulation and Activities of Antioxidant Enzymes in ZAT12-Transformed Transgenic Tomato Plants. Plant Physiol. Biochem. 2012, 61, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.H.; Jiang, L.L.; Ma, X.F.; Xu, Z.S.; Liu, M.M.; Shan, S.G.; Cheng, X.G. A Soybean C2H2-Type Zinc Finger Gene GmZF1 Enhanced Cold Tolerance in Transgenic Arabidopsis. PLoS ONE 2014, 9, e109399. [Google Scholar] [CrossRef] [PubMed]
- Chong, L.; Guo, P.; Zhu, Y. Mediator Complex: A Pivotal Regulator of Aba Signaling Pathway and Abiotic Stress Response in Plants. Int. J. Mol. Sci. 2020, 21, 7755. [Google Scholar] [CrossRef] [PubMed]
- Ku, Y.S.; Sintaha, M.; Cheung, M.Y.; Lam, H.M. Plant Hormone Signaling Crosstalks between Biotic and Abiotic Stress Responses. Int. J. Mol. Sci. 2018, 19, 3206. [Google Scholar] [CrossRef] [PubMed]
- Faraji, S.; Rasouli, S.H.; Kazemitabar, S.K. Genome-Wide Exploration of C2H2 Zinc Finger Family in Durum Wheat (Triticum Turgidum Ssp. Durum): Insights into the Roles in Biological Processes Especially Stress Response. BioMetals 2018, 31, 1019–1042. [Google Scholar] [CrossRef]
- Fadda, A.; Syed, N.; Mackeh, R.; Papadopoulou, A.; Suzuki, S.; Jithesh, P.V.; Kino, T. Genome-Wide Regulatory Roles of the C2H2-Type Zinc Finger Protein ZNF764 on the Glucocorticoid Receptor. Sci. Rep. 2017, 7, 41598. [Google Scholar] [CrossRef]
- Chalhoub, B.; Denoeud, F.; Liu, S.; Parkin, I.A.P.; Tang, H.; Wang, X.; Chiquet, J.; Belcram, H.; Tong, C.; Samans, B.; et al. Early Allopolyploid Evolution in the Post-Neolithic Brassica Napus Oilseed Genome. Science 2014, 345, 950–953. [Google Scholar] [CrossRef]
- Lawrence, S.D.; Novak, N.G. Comparative Analysis of the Genetic Variability within the Q-Type C2H2 Zinc-Finger Transcription Factors in the Economically Important Cabbage, Canola and Chinese Cabbage Genomes. Hereditas 2018, 155, 29. [Google Scholar] [CrossRef]
- Lawrence, S.D.; Novak, N.G. Mining the Brassica Oleracea Genome for Q-Type C2H2 Zinc Finger Transcription Factor Genes. Plant Mol. Biol. Report. 2015, 33, 1611–1617. [Google Scholar] [CrossRef]
- Schranz, M.E.; Lysak, M.A.; Mitchell-Olds, T. The ABC’s of Comparative Genomics in the Brassicaceae: Building Blocks of Crucifer Genomes. Trends Plant Sci. 2006, 11, 535–542. [Google Scholar] [CrossRef]
- Liu, S.; Liu, Y.; Yang, X.; Tong, C.; Edwards, D.; Parkin, I.A.P.; Zhao, M.; Ma, J.; Yu, J.; Huang, S.; et al. The Brassica Oleracea Genome Reveals the Asymmetrical Evolution of Polyploid Genomes. Nat. Commun. 2014, 5, 3930. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Zhao, Y.; Hou, X.; Tao, X.; Sun, B.; Zeng, R.; Zhu, M.; Sun, W.; Wu, J.; Liu, L.; et al. Cloning and Functional Analysis of Zinc Finger Protein Gene BrZFP1 in Brassica Campestris. J. Agric. Biotechnol. 2022, 30, 236–248. [Google Scholar] [CrossRef]
- Davletova, S.; Schlauch, K.; Coutu, J.; Mittler, R. The Zinc-Finger Protein Zat12 Plays a Central Role in Reactive Oxygen and Abiotic Stress Signaling in Arabidopsis. Plant Physiol. 2005, 139, 847–856. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Liang, W.; Gu, P.; Huang, Z. Salt Tolerance Function of the Novel C2H2-Type Zinc Finger Protein TaZNF in Wheat. Plant Physiol. Biochem. 2016, 106, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.C.; Fu, C.C. Cold-Inducible MaC2H2s Are Associated with Cold Stress Response of Banana Fruit via Regulating MaICE1. Plant Cell Rep. 2019, 38, 673–680. [Google Scholar] [CrossRef]
- Kim, J.C.; Lee, S.H.; Cheong, Y.H.; Yoo, C.M.; Lee, S.I.; Chun, H.J.; Yun, D.J.; Hong, J.C.; Lee, S.Y.; Lim, C.O.; et al. A Novel Cold-Inducible Zinc Finger Protein from Soybean, SCOF-1, Enhances Cold Tolerance in Transgenic Plants. Plant J. 2001, 25, 247–259. [Google Scholar] [CrossRef]
- Kim, Y.H.; Kim, M.D.; Park, S.C.; Yang, K.S.; Jeong, J.C.; Lee, H.S.; Kwak, S.S. SCOF-1-Expressing Transgenic Sweetpotato Plants Show Enhanced Tolerance to Low-Temperature Stress. Plant Physiol. Biochem. 2011, 49, 1436–1441. [Google Scholar] [CrossRef]
- Kim, Y.H.; Kim, M.D.; Park, S.C.; Jeong, J.C.; Kwak, S.S.; Lee, H.S. Transgenic Potato Plants Expressing the Cold-Inducible Transcription Factor SCOF-1 Display Enhanced Tolerance to Freezing Stress. Plant Breed. 2016, 135, 513–518. [Google Scholar] [CrossRef]
- Huang, J.; Sun, S.; Xu, D.; Lan, H.; Sun, H.; Wang, Z.; Bao, Y.; Wang, J.; Tang, H.; Zhang, H. A TFIIIA-Type Zinc Finger Protein Confers Multiple Abiotic Stress Tolerances in Transgenic Rice (Oryza sativa L.). Plant Mol. Biol. 2012, 80, 337–350. [Google Scholar] [CrossRef]
- Yilmaz, S.C.; Mittler, R. The Zinc Finger Network of Plants. Cell. Mol. Life Sci. 2008, 65, 1150–1160. [Google Scholar] [CrossRef]
- Kiełbowicz, M.A. Involvement of Plant C2H2-Type Zinc Finger Transcription Factors in Stress Responses. Plant Sci. 2012, 185–186, 78–85. [Google Scholar] [CrossRef]
- Huang, J.; Sun, S.J.; Xu, D.Q.; Yang, X.; Bao, Y.M.; Wang, Z.F.; Tang, H.J.; Zhang, H. Increased Tolerance of Rice to Cold, Drought and Oxidative Stresses Mediated by the Overexpression of a Gene That Encodes the Zinc Finger Protein ZFP245. Biochem. Biophys. Res. Commun. 2009, 389, 556–561. [Google Scholar] [CrossRef]
- Huang, J.; Wang, J.F.; Wang, Q.H.; Zhang, H.S. Identification of a Rice Zinc Finger Protein Whose Expression Is Transiently Induced by Drought, Cold but Not by Salinity and Abscisic Acid. DNA Seq.-J. DNA Seq. Mapp. 2005, 16, 130–136. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, Y.; Wen, F.; Yao, D.; Wang, L.; Guo, J.; Ni, L.; Zhang, A.; Tan, M.; Jiang, M. A Novel Rice C2H2-Type Zinc Finger Protein, ZFP36, Is a Key Player Involved in Abscisic Acid-Induced Antioxidant Defence and Oxidative Stress Tolerance in Rice. J. Exp. Bot. 2014, 65, 5795–5809. [Google Scholar] [CrossRef]
- Huang, L.; Zhang, M.; Jia, J.; Zhao, X.; Huang, X.; Ji, E.; Ni, L.; Jiang, M. An Atypical Late Embryogenesis Abundant Protein OsLEA5 Plays a Positive Role in ABA-Induced Antioxidant Defense in Oryza sativa L. Plant Cell Physiol. 2018, 59, 916–929. [Google Scholar] [CrossRef]
- Brumbarova, T.; Le, C.T.T.; Ivanov, R.; Bauer, P. Regulation of ZAT12 Protein Stability: The Role of Hydrogen Peroxide. Plant Signal. Behav. 2016, 11, e1137408. [Google Scholar] [CrossRef][Green Version]
- Ben Daniel, B.H.; Cattan, E.; Wachtel, C.; Avrahami, D.; Glick, Y.; Malichy, A.; Gerber, D.; Miller, G. Identification of Novel Transcriptional Regulators of Zat12 Using Comprehensive Yeast One-Hybrid Screens. Physiol. Plant. 2016, 157, 422–441. [Google Scholar] [CrossRef]
- Le, C.T.T.; Brumbarova, T.; Ivanov, R.; Stoof, C.; Weber, E.; Mohrbacher, J.; Fink-Straube, C.; Bauer, P. Zinc Finger of Arabidopsis Thaliana12 (Zat12) Interacts with Fer-like Iron Deficiency-Induced Transcription Factor (FIT) Linking Iron Deficiency and Oxidative Stress Responses. Plant Physiol. 2016, 170, 540–557. [Google Scholar] [CrossRef]
- Lingam, S.; Mohrbacher, J.; Brumbarova, T.; Potuschak, T.; Fink-Straube, C.; Blondet, E.; Genschik, P.; Bauer, P. Interaction between the BHLH Transcription Factor FIT and ETHYLENE INSENSITIVE3/ETHYLENE INSENSITIVE3-LIKE1 Reveals Molecular Linkage between the Regulation of Iron Acquisition and Ethylene Signaling in Arabidopsis. Plant Cell 2011, 23, 1815–1829. [Google Scholar] [CrossRef]
- Yoo, S.D.; Cho, Y.H.; Tena, G.; Xiong, Y.; Sheen, J. Dual Control of Nuclear EIN3 by Bifurcate MAPK Cascades in C2H4 Signalling. Nature 2008, 451, 789–795. [Google Scholar] [CrossRef]
- Peng, J.; Li, Z.; Wen, X.; Li, W.; Shi, H.; Yang, L.; Zhu, H.; Guo, H. Salt-Induced Stabilization of EIN3/EIL1 Confers Salinity Tolerance by Deterring ROS Accumulation in Arabidopsis. PLoS Genet. 2014, 10, e1004664. [Google Scholar] [CrossRef] [PubMed]
- Rizhsky, L.; Davletova, S.; Liang, H.; Mittler, R. The Zinc Finger Protein Zat12 Is Required for Cytosolic Ascorbate Peroxidase 1 Expression during Oxidative Stress in Arabidopsis. J. Biol. Chem. 2004, 279, 11736–11743. [Google Scholar] [CrossRef] [PubMed]
- Ciftci-Yilmaz, S.; Morsy, M.R.; Song, L.; Coutu, A.; Krizek, B.A.; Lewis, M.W.; Warren, D.; Cushman, J.; Connolly, E.L.; Mittler, R. The EAR-Motif of the Cys2/His2-Type Zinc Finger Protein Zat7 Plays a Key Role in the Defense Response of Arabidopsis to Salinity Stress. J. Biol. Chem. 2007, 282, 9260–9268. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.M.; Nguyen, X.C.; Kim, K.E.; Han, H.J.; Yoo, J.; Lee, K.; Kim, M.C.; Yun, D.J.; Chung, W.S. Phosphorylation of the Zinc Finger Transcriptional Regulator ZAT6 by MPK6 Regulates Arabidopsis Seed Germination under Salt and Osmotic Stress. Biochem. Biophys. Res. Commun. 2013, 430, 1054–1059. [Google Scholar] [CrossRef]
- Nguyen, X.C.; Kim, S.H.; Lee, K.; Kim, K.E.; Liu, X.M.; Han, H.J.; Hoang, M.H.T.; Lee, S.W.; Hong, J.C.; Moon, Y.H.; et al. Identification of a C2H2-Type Zinc Finger Transcription Factor (ZAT10) from Arabidopsis as a Substrate of MAP Kinase. Plant Cell Rep. 2012, 31, 737–745. [Google Scholar] [CrossRef]
- Finn, R.D.; Coggill, P.; Eberhardt, R.Y.; Eddy, S.R.; Mistry, J.; Mitchell, A.L.; Potter, S.C.; Punta, M.; Qureshi, M.; Sangrador-Vegas, A.; et al. The Pfam Protein Families Database: Towards a More Sustainable Future. Nucleic Acids Res. 2016, 44, D279–D285. [Google Scholar] [CrossRef]
- Mistry, J.; Finn, R.D.; Eddy, S.R.; Bateman, A.; Punta, M. Challenges in Homology Search: HMMER3 and Convergent Evolution of Coiled-Coil Regions. Nucleic Acids Res. 2013, 41, e121. [Google Scholar] [CrossRef]
- Marchler-Bauer, A.; Bo, Y.; Han, L.; He, J.; Lanczycki, C.J.; Lu, S.; Chitsaz, F.; Derbyshire, M.K.; Geer, R.C.; Gonzales, N.R.; et al. CDD/SPARCLE: Functional Classification of Proteins via Subfamily Domain Architectures. Nucleic Acids Res. 2017, 45, D200–D203. [Google Scholar] [CrossRef]
- Chen, C.; Chen, H.; Zhang, Y.; Thomas, H.R.; Frank, M.H.; He, Y.; Xia, R. TBtools: An Integrative Toolkit Developed for Interactive Analyses of Big Biological Data. Mol. Plant 2020, 13, 1194–1202. [Google Scholar] [CrossRef]
- Bailey, T.L.; Boden, M.; Buske, F.A.; Frith, M.; Grant, C.E.; Clementi, L.; Ren, J.; Li, W.W.; Noble, W.S. MEME Suite: Tools for Motif Discovery and Searching. Nucleic Acids Res. 2009, 37, W202–W208. [Google Scholar] [CrossRef]
- Lescot, M.; Déhais, P.; Thijs, G.; Marchal, K.; Moreau, Y.; Van De Peer, Y.; Rouzé, P.; Rombauts, S. PlantCARE, a Database of Plant Cis-Acting Regulatory Elements and a Portal to Tools for in Silico Analysis of Promoter Sequences. Nucleic Acids Res. 2002, 30, 325–327. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Tu, X.; Ding, Y.; Gao, G.; Deng, M. Expectation Pooling: An Effective and Interpretable Pooling Method for Predicting DNA-Protein Binding. Bioinformatics 2020, 36, 1405–1412. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Zhang, H.; Kong, L.; Gao, G.; Luo, J. PlantTFDB 3.0: A Portal for the Functional and Evolutionary Study of Plant Transcription Factors. Nucleic Acids Res. 2014, 42, 1182–1187. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, B.; Gao, S.; Lercher, M.J.; Hu, S.; Chen, W.H. Evolview v3: A Webserver for Visualization, Annotation, and Management of Phylogenetic Trees. Nucleic Acids Res. 2019, 47, W270–W275. [Google Scholar] [CrossRef] [PubMed]
- Voorrips, R.E. Mapchart: Software for the Graphical Presentation of Linkage Maps and QTLs. J. Hered. 2002, 93, 77–78. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2-ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Coulter, J.A.; Liu, L.; Zhao, Y.; Chang, Y.; Pu, Y.; Zeng, X.; Xu, Y.; Wu, J.; Fang, Y.; et al. Transcriptome Analysis Reveals Key Cold-Stress-Responsive Genes in Winter Rapeseed (Brassica Rapa L.). Int. J. Mol. Sci. 2019, 20, 1071. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Coulter, J.A.; Wu, J.; Liu, L.; Li, X.; Dong, Y.; Ma, L.; Pu, Y.; Sun, B.; Niu, Z.; et al. Identification of Differentially Expressed Genes Involved in Amino Acid and Lipid Accumulation of Winter Turnip Rape (Brassica Rapa L.) in Response to Cold Stress. PLoS ONE 2021, 16, e0245494. [Google Scholar] [CrossRef] [PubMed]
- Quiroga, M.; Guerrero, C.; Botella, M.A.; Barceló, A.; Amaya, I.; Medina, M.I.; Alonso, F.J.; De Forchetti, S.M.; Tigier, H.; Valpuesta, V. A Tomato Peroxidase Involved in the Synthesis of Lignin and Suberin. Plant Physiol. 2000, 122, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
- Gill, T.; Sreenivasulu, Y.; Kumar, S.; Singh Ahuja, P. Over-Expression of Superoxide Dismutase Exhibits Lignification of Vascular Structures in Arabidopsis Thaliana. J. Plant Physiol. 2010, 167, 757–760. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, L.; Xu, J.; Tao, X.; Wu, J.; Wang, W.; Pu, Y.; Yang, G.; Fang, Y.; Liu, L.; Li, X.; et al. Genome-Wide Identification of C2H2 ZFPs and Functional Analysis of BRZAT12 under Low-Temperature Stress in Winter Rapeseed (Brassica rapa). Int. J. Mol. Sci. 2022, 23, 12218. https://doi.org/10.3390/ijms232012218
Ma L, Xu J, Tao X, Wu J, Wang W, Pu Y, Yang G, Fang Y, Liu L, Li X, et al. Genome-Wide Identification of C2H2 ZFPs and Functional Analysis of BRZAT12 under Low-Temperature Stress in Winter Rapeseed (Brassica rapa). International Journal of Molecular Sciences. 2022; 23(20):12218. https://doi.org/10.3390/ijms232012218
Chicago/Turabian StyleMa, Li, Jia Xu, Xiaolei Tao, Junyan Wu, Wangtian Wang, Yuanyuan Pu, Gang Yang, Yan Fang, Lijun Liu, Xuecai Li, and et al. 2022. "Genome-Wide Identification of C2H2 ZFPs and Functional Analysis of BRZAT12 under Low-Temperature Stress in Winter Rapeseed (Brassica rapa)" International Journal of Molecular Sciences 23, no. 20: 12218. https://doi.org/10.3390/ijms232012218
APA StyleMa, L., Xu, J., Tao, X., Wu, J., Wang, W., Pu, Y., Yang, G., Fang, Y., Liu, L., Li, X., & Sun, W. (2022). Genome-Wide Identification of C2H2 ZFPs and Functional Analysis of BRZAT12 under Low-Temperature Stress in Winter Rapeseed (Brassica rapa). International Journal of Molecular Sciences, 23(20), 12218. https://doi.org/10.3390/ijms232012218






