Lipid-Based Nanoparticulate Systems for the Ocular Delivery of Bioactives with Anti-Inflammatory Properties
Abstract
1. Introduction
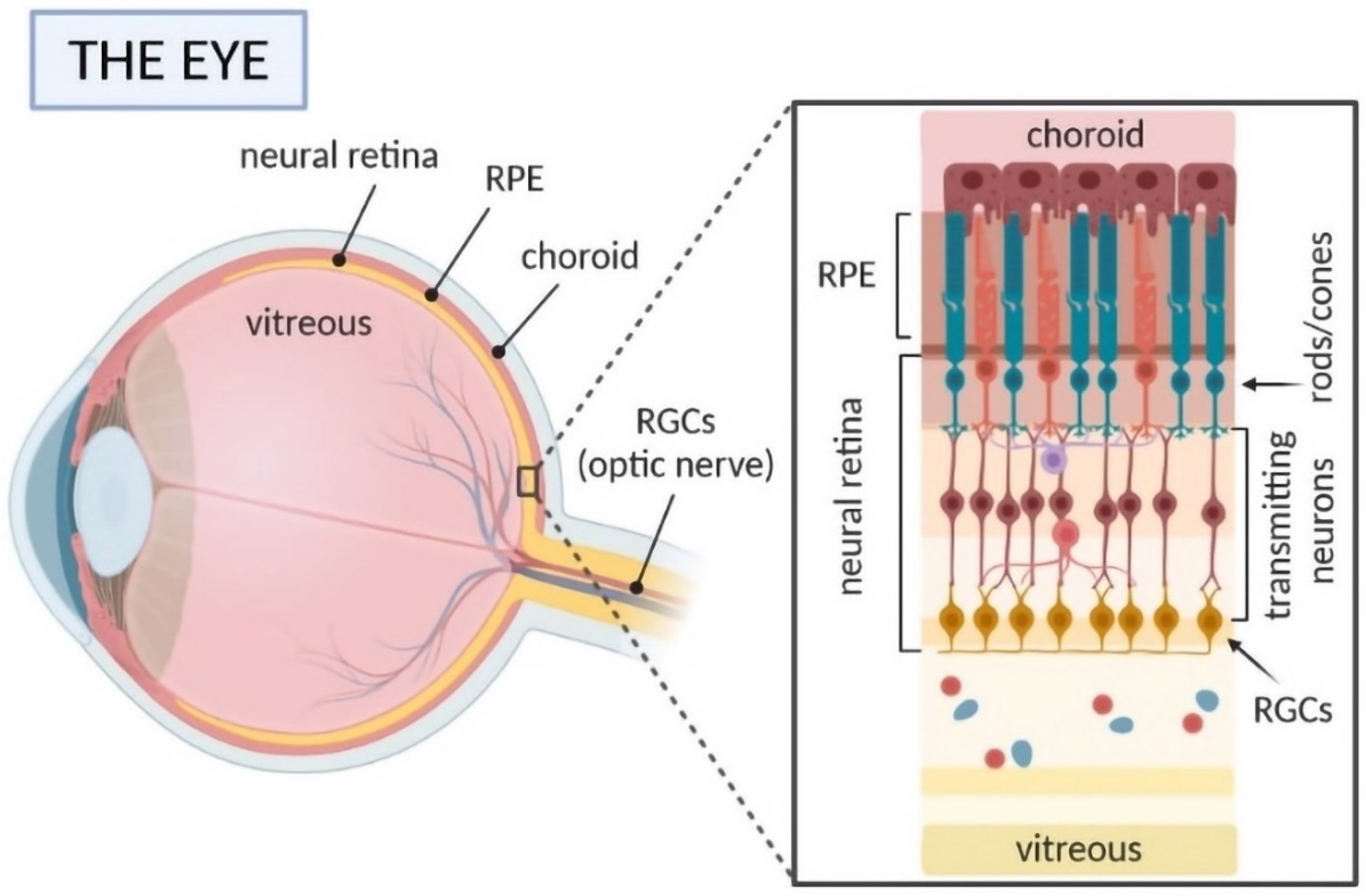
2. Overcoming Obstacles in Ocular Drug Delivery—Ocular Anatomy and Challenges in Drug Delivery
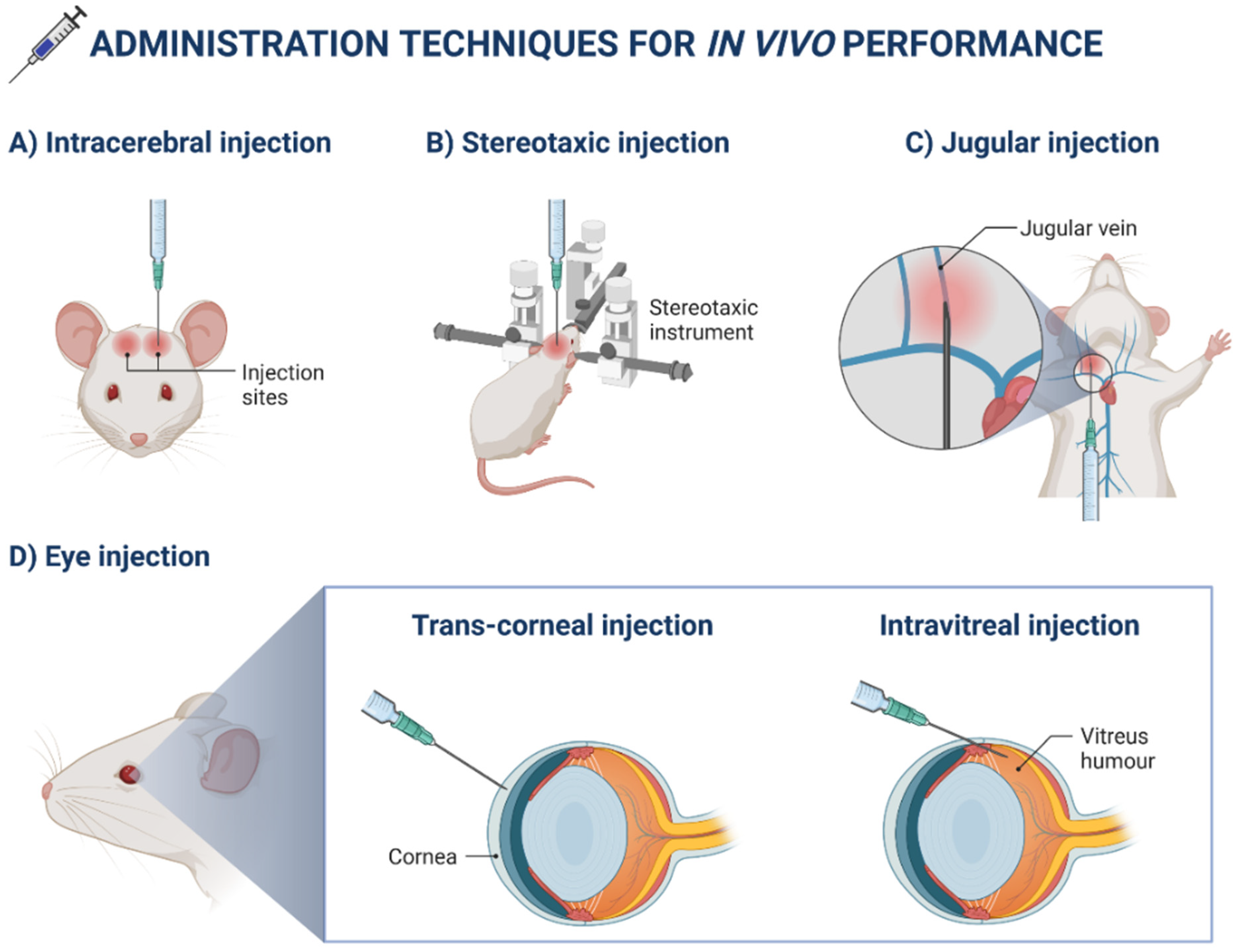
3. Application of Nanotechnology in Ocular Drug Delivery
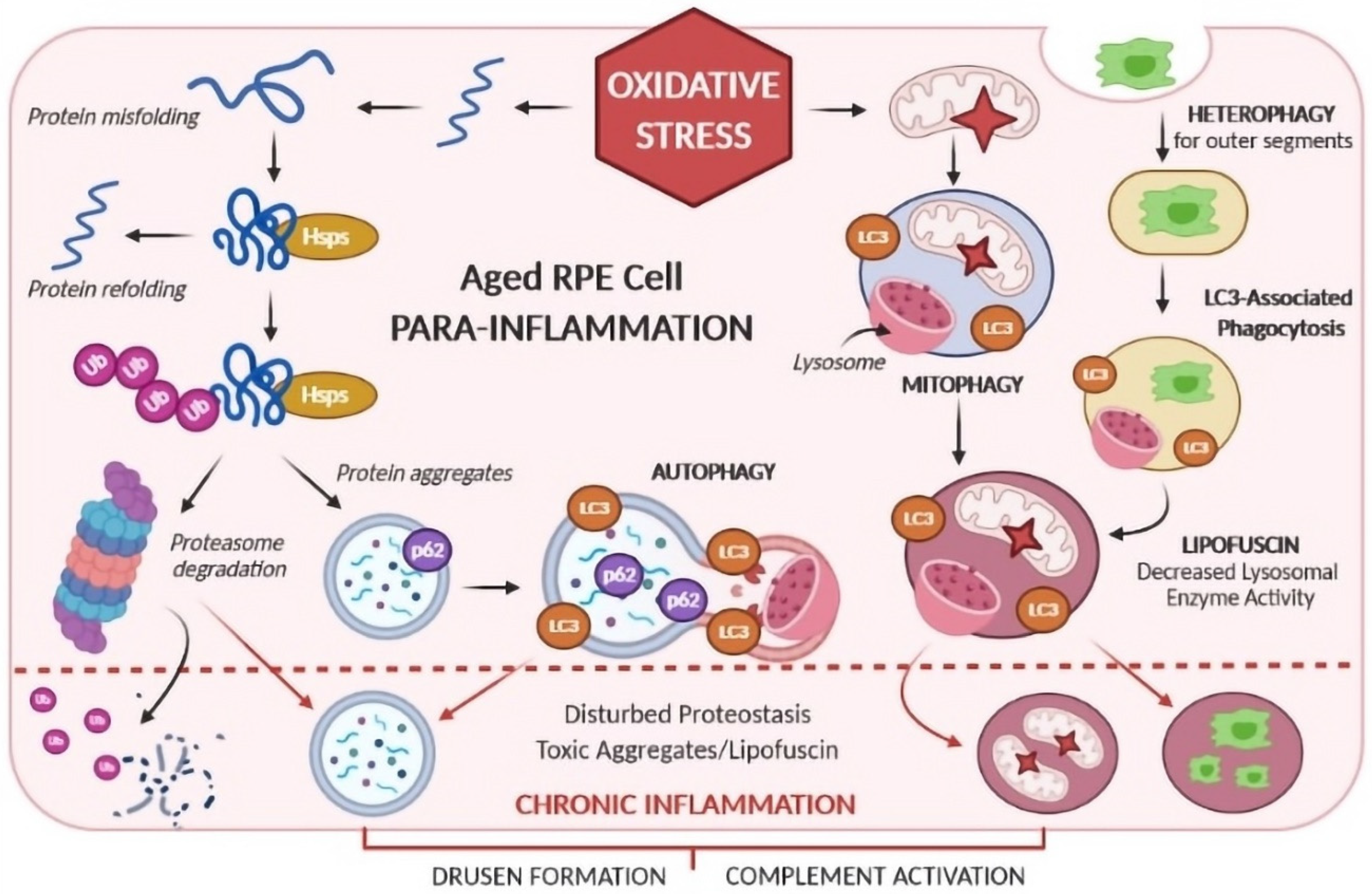
4. Ocular Inflammation
4.1. Corticosteroids
4.2. Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)
4.3. Immunosuppressive Agents
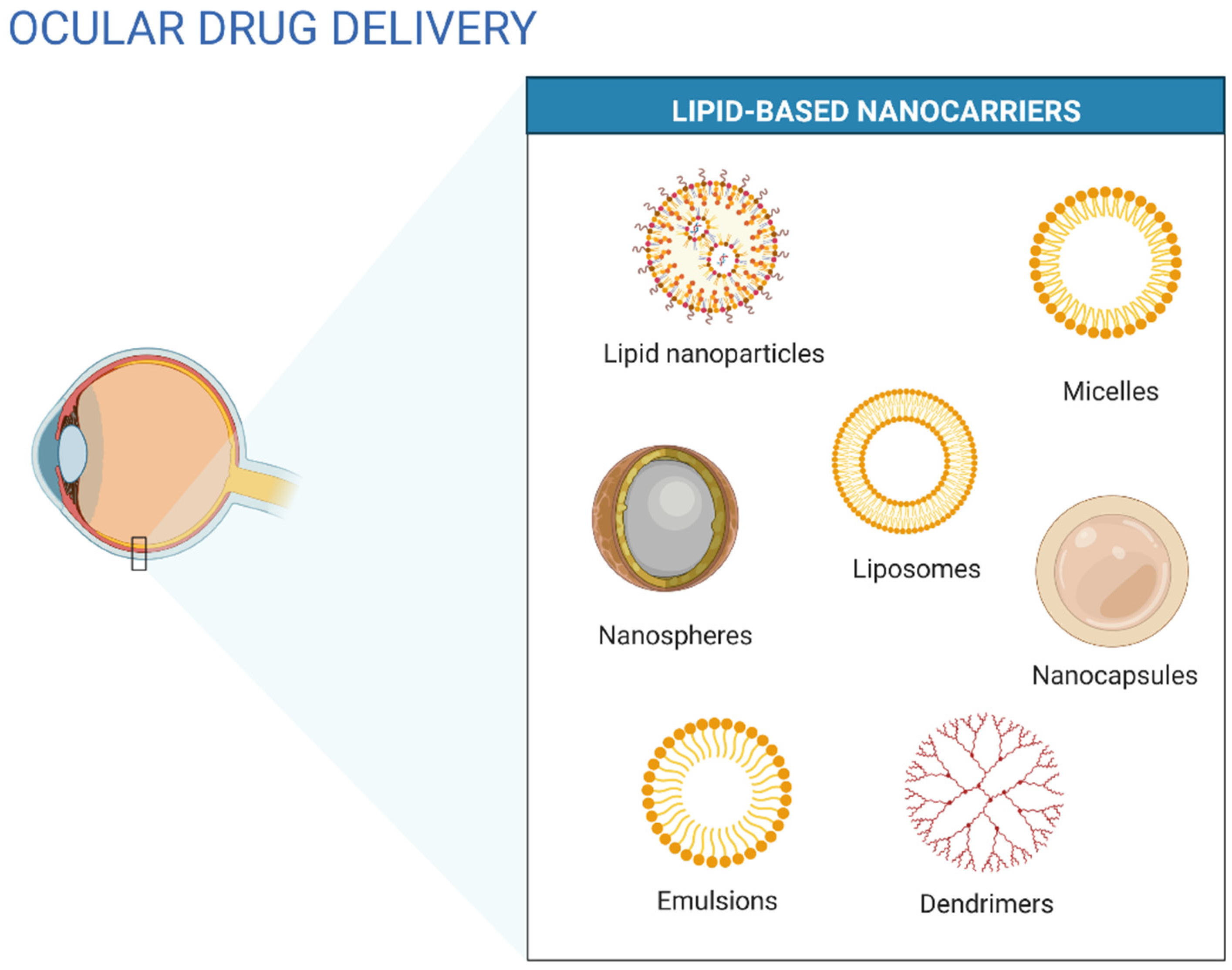
5. Lipid-Based Nanosystems Applied in Ocular Delivery
6. Natural Compounds with Anti-Inflammatory Action
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Seyfoddin, A.; Shaw, J.; Al-Kassas, R. Solid lipid nanoparticles for ocular drug delivery. Drug Deliv. 2010, 17, 467–489. [Google Scholar] [CrossRef]
- Souto, E.B.; Doktorovova, S.; Gonzalez-Mira, E.; Egea, M.A.; Garcia, M.L. Feasibility of Lipid Nanoparticles for Ocular Delivery of Anti-Inflammatory Drugs. Curr. Eye Res. 2010, 35, 537–552. [Google Scholar] [CrossRef] [PubMed]
- Battaglia, L.; Serpe, L.; Foglietta, F.; Muntoni, E.; Gallarate, M.; Del Pozo Rodriguez, A.; Solinis, M.A. Application of lipid nanoparticles to ocular drug delivery. Expert Opin. Drug Deliv. 2016, 13, 1743–1757. [Google Scholar] [CrossRef]
- Sánchez-López, E.; Espina, M.; Doktorovova, S.; Souto, E.B.; García, M.L. Lipid nanoparticles (SLN, NLC): Overcoming the anatomical and physiological barriers of the eye—Part II—Ocular drug-loaded lipid nanoparticles. Eur. J. Pharm. Biopharm. 2017, 110, 58–69. [Google Scholar] [CrossRef]
- Dhiman, N.; Awasthi, R.; Sharma, B.; Kharkwal, H.; Kulkarni, G.T. Lipid Nanoparticles as Carriers for Bioactive Delivery. Front. Chem. 2021, 9, 580118. [Google Scholar] [CrossRef]
- del Pozo-Rodríguez, A.; Delgado, D.; Gascón, A.R.; Solinís, M.Á. Lipid nanoparticles as drug/gene delivery systems to the retina. J. Ocul. Pharmacol. Ther. 2013, 29, 173–188. [Google Scholar] [CrossRef]
- Ghasemiyeh, P.; Mohammadi-Samani, S. Solid lipid nanoparticles and nanostructured lipid carriers as novel drug delivery systems: Applications, advantages and disadvantages. Res. Pharm. Sci. 2018, 13, 288. [Google Scholar]
- Wang, R.; Gao, Y.; Liu, A.; Zhai, G. A review of nanocarrier-mediated drug delivery systems for posterior segment eye disease: Challenges analysis and recent advances. J. Drug Target. 2021, 29, 687–702. [Google Scholar] [CrossRef]
- Khiev, D.; Mohamed, Z.A.; Vichare, R.; Paulson, R.; Bhatia, S.; Mohapatra, S.; Lobo, G.P.; Valapala, M.; Kerur, N.; Passaglia, C.L.; et al. Emerging Nano-Formulations and Nanomedicines Applications for Ocular Drug Delivery. Nanomaterials 2021, 11, 173. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, I.F.; Barbosa, E.J.; Peters, M.C.C.; Henostroza, M.A.B.; Yukuyama, M.N.; dos Santos Neto, E.; Löbenberg, R.; Bou-Chacra, N. Cutting-edge advances in therapy for the posterior segment of the eye: Solid lipid nanoparticles and nanostructured lipid carriers. Int. J. Pharm. 2020, 589, 119831. [Google Scholar] [CrossRef]
- González-Fernández, F.M.; Bianchera, A.; Gasco, P.; Nicoli, S.; Pescina, S. Lipid-Based Nanocarriers for Ophthalmic Administration: Towards Experimental Design Implementation. Pharmaceutics 2021, 13, 447. [Google Scholar] [CrossRef]
- Gorantla, S.; Rapalli, V.K.; Waghule, T.; Singh, P.P.; Dubey, S.K.; Saha, R.N.; Singhvi, G. Nanocarriers for ocular drug delivery: Current status and translational opportunity. RSC Adv. 2020, 10, 27835–27855. [Google Scholar] [CrossRef]
- Venkata Lokesh, G.; Madhuvani, M.; Chatla, C.; Sarad Pawar Naik, B.; Bhargavi, S.; Manasa, A. Conventional Opthalamic Drug Delivery System: An Outlook. Future J. Pharm. Health Sci. 2021, 1, 7–12. [Google Scholar]
- Alvarez-Trabado, J.; Diebold, Y.; Sanchez, A. Designing lipid nanoparticles for topical ocular drug delivery. Int. J. Pharm. 2017, 532, 204–217. [Google Scholar] [CrossRef]
- Wang, H.; Zhou, Y.; Sun, Q.; Zhou, C.; Hu, S.; Lenahan, C.; Xu, W.; Deng, Y.; Li, G.; Tao, S. Update on Nanoparticle-Based Drug Delivery System for Anti-inflammatory Treatment. Front. Bioeng. Biotechnol. 2021, 9, 630352. [Google Scholar] [CrossRef]
- Araújo, J.; Gonzalez, E.; Egea, M.A.; Garcia, M.L.; Souto, E.B. Nanomedicines for ocular NSAIDs: Safety on drug delivery. Nanomed. Nanotechnol. Biol. Med. 2009, 5, 394–401. [Google Scholar] [CrossRef]
- Texeira, M.C.; Carbone, C.; Souto, E.B. Beyond liposomes: Recent advances on lipid based nanostructures for poorly soluble/poorly permeable drug delivery. Prog. Lipid Res. 2017, 68, 1–11. [Google Scholar] [CrossRef]
- Wang, Y.; Rajala, A.; Rajala, R.V.S. Lipid Nanoparticles for Ocular Gene Delivery. J. Funct. Biomater. 2015, 6, 379–394. [Google Scholar] [CrossRef]
- Sánchez-López, E.; Espina, M.; Doktorovova, S.; Souto, E.B.; García, M.L. Lipid nanoparticles (SLN, NLC): Overcoming the anatomical and physiological barriers of the eye—Part I—Barriers and determining factors in ocular delivery. Eur. J. Pharm. Biopharm. 2017, 110, 70–75. [Google Scholar] [CrossRef]
- Jacob, S.; Nair, A.B.; Shah, J.; Gupta, S.; Boddu, S.H.S.; Sreeharsha, N.; Joseph, A.; Shinu, P.; Morsy, M.A. Lipid Nanoparticles as a Promising Drug Delivery Carrier for Topical Ocular Therapy—An Overview on Recent Advances. Pharmaceutics 2022, 14, 533. [Google Scholar] [CrossRef] [PubMed]
- Dubashynskaya, N.V.; Bokatyi, A.N.; Golovkin, A.S.; Kudryavtsev, I.V.; Serebryakova, M.K.; Trulioff, A.S.; Dubrovskii, Y.A.; Skorik, Y.A. Synthesis and Characterization of Novel Succinyl Chitosan-Dexamethasone Conjugates for Potential Intravitreal Dexamethasone Delivery. Int. J. Mol. Sci. 2021, 22, 10960. [Google Scholar] [CrossRef]
- Dubashynskaya, N.V.; Bokatyi, A.N.; Skorik, Y.A. Dexamethasone Conjugates: Synthetic Approaches and Medical Prospects. Biomedicines 2021, 9, 341. [Google Scholar] [CrossRef]
- Kicková, E.; Salmaso, S.; Mastrotto, F.; Caliceti, P.; Urtti, A. Pullulan Based Bioconjugates for Ocular Dexamethasone Delivery. Pharmaceutics 2021, 13, 791. [Google Scholar] [CrossRef]
- Ioele, G.; Grande, F.; De Luca, M.; Occhiuzzi, M.A.; Garofalo, A.; Ragno, G. Photodegradation of Anti-Inflammatory Drugs: Stability Tests and Lipid Nanocarriers for Their Photoprotection. Molecules 2021, 26, 5989. [Google Scholar] [CrossRef]
- Pasadhika, S.; Kempen, J.H.; Newcomb, C.W.; Liesegang, T.L.; Pujari, S.S.; Rosenbaum, J.T.; Thorne, J.E.; Foster, C.S.; Jabs, D.A.; Levy-Clarke, G.A.; et al. Azathioprine for Ocular Inflammatory Diseases. Am. J. Ophthalmol. 2009, 148, 500–509.e502. [Google Scholar] [CrossRef]
- Gangaputra, S.; Newcomb, C.W.; Liesegang, T.L.; Kaçmaz, R.O.; Jabs, D.A.; Levy-Clarke, G.A.; Nussenblatt, R.B.; Rosenbaum, J.T.; Suhler, E.B.; Thorne, J.E.; et al. Methotrexate for Ocular Inflammatory Diseases. Ophthalmology 2009, 116, 2188–2198.e2181. [Google Scholar] [CrossRef]
- Jap, A.; Chee, S.-P. Immunosuppressive therapy for ocular diseases. Curr. Opin. Ophthalmol. 2008, 19, 535–540. [Google Scholar] [CrossRef]
- Dubashynskaya, N.; Poshina, D.; Raik, S.; Urtti, A.; Skorik, Y.A. Polysaccharides in Ocular Drug Delivery. Pharmaceutics 2020, 12, 22. [Google Scholar] [CrossRef]
- Jurišić Dukovski, B.; Juretić, M.; Bračko, D.; Randjelović, D.; Savić, S.; Crespo Moral, M.; Diebold, Y.; Filipović-Grčić, J.; Pepić, I.; Lovrić, J. Functional ibuprofen-loaded cationic nanoemulsion: Development and optimization for dry eye disease treatment. Int. J. Pharm. 2020, 576, 118979. [Google Scholar] [CrossRef]
- Saha, M.; Saha, D.R.; Ulhosna, T.; Sharker, S.M.; Shohag, M.H.; Islam, M.S.; Ray, S.K.; Rahman, G.M.S.; Reza, H.M. QbD based development of resveratrol-loaded mucoadhesive lecithin/chitosan nanoparticles for prolonged ocular drug delivery. J. Drug Deliv. Sci. Technol. 2021, 63, 102480. [Google Scholar] [CrossRef]
- Ghafoorianfar, S.; Ghorani-Azam, A.; Mohajeri, S.A.; Farzin, D. Efficiency of nanoparticles for treatment of ocular infections: Systematic literature review. J. Drug Deliv. Sci. Technol. 2020, 57, 101765. [Google Scholar] [CrossRef]
- Tatke, A.; Dudhipala, N.; Janga, K.Y.; Balguri, S.P.; Avula, B.; Jablonski, M.M.; Majumdar, S. In Situ Gel of Triamcinolone Acetonide-Loaded Solid Lipid Nanoparticles for Improved Topical Ocular Delivery: Tear Kinetics and Ocular Disposition Studies. Nanomaterials 2019, 9, 33. [Google Scholar] [CrossRef]
- Nirbhavane, P.; Sharma, G.; Singh, B.; Begum, G.; Jones, M.-C.; Rauz, S.; Vincent, R.; Denniston, A.K.; Hill, L.J.; Katare, O.P. Triamcinolone acetonide loaded-cationic nano-lipoidal formulation for uveitis: Evidences of improved biopharmaceutical performance and anti-inflammatory activity. Colloids Surf. B Biointerfaces 2020, 190, 110902. [Google Scholar] [CrossRef]
- Desoqi, M.H.; El-Sawy, H.S.; Kafagy, E.; Ghorab, M.; Gad, S. Fluticasone propionate–loaded solid lipid nanoparticles with augmented anti-inflammatory activity: Optimisation, characterisation and pharmacodynamic evaluation on rats. J. Microencapsul. 2021, 38, 177–191. [Google Scholar] [CrossRef]
- Alami-Milani, M.; Zakeri-Milani, P.; Valizadeh, H.; Sattari, S.; Salatin, S.; Jelvehgari, M. Evaluation of anti-inflammatory impact of dexamethasone-loaded PCL-PEG-PCL micelles on endotoxin-induced uveitis in rabbits. Pharm. Dev. Technol. 2019, 24, 680–688. [Google Scholar] [CrossRef]
- Salimi, A. Preparation and evaluation of celecoxib nanoemulsion for ocular drug delivery. Asian J. Pharm. (AJP) 2017, 11, 543–550. [Google Scholar]
- Zhang, J.; Liu, Z.; Tao, C.; Lin, X.; Zhang, M.; Zeng, L.; Chen, X.; Song, H. Cationic nanoemulsions with prolonged retention time as promising carriers for ophthalmic delivery of tacrolimus. Eur. J. Pharm. Sci. 2020, 144, 105229. [Google Scholar] [CrossRef]
- Blazaki, S.; Pachis, K.; Tzatzarakis, M.; Tsilimbaris, M.; Antimisiaris, S.G. Novel Liposome Aggregate Platform (LAP) system for sustained retention of drugs in the posterior ocular segment following intravitreal injection. Int. J. Pharm. 2020, 576, 118987. [Google Scholar] [CrossRef]
- Mu, H.; Wang, Y.; Chu, Y.; Jiang, Y.; Hua, H.; Chu, L.; Wang, K.; Wang, A.; Liu, W.; Li, Y.; et al. Multivesicular liposomes for sustained release of bevacizumab in treating laser-induced choroidal neovascularization. Drug Deliv. 2018, 25, 1372–1383. [Google Scholar] [CrossRef]
- Lançon, A.; Frazzi, R.; Latruffe, N. Anti-oxidant, anti-inflammatory and anti-angiogenic properties of resveratrol in ocular diseases. Molecules 2016, 21, 304. [Google Scholar] [CrossRef]
- Li, M.; Xin, M.; Guo, C.; Lin, G.; Wu, X. New nanomicelle curcumin formulation for ocular delivery: Improved stability, solubility, and ocular anti-inflammatory treatment. Drug Dev. Ind. Pharm. 2017, 43, 1846–1857. [Google Scholar] [CrossRef] [PubMed]
- Sun, F.; Zheng, Z.; Lan, J.; Li, X.; Li, M.; Song, K.; Wu, X. New micelle myricetin formulation for ocular delivery: Improved stability, solubility, and ocular anti-inflammatory treatment. Drug Deliv. 2019, 26, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Giannaccare, G.; Pellegrini, M.; Senni, C.; Bernabei, F.; Scorcia, V.; Cicero, A.F.G. Clinical applications of astaxanthin in the treatment of ocular diseases: Emerging insights. Mar. Drugs 2020, 18, 239. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhang, L.; Li, R.; Yan, M. New resveratrol micelle formulation for ocular delivery: Characterization and in vitro/in vivo evaluation. Drug Dev. Ind. Pharm. 2020, 46, 1960–1970. [Google Scholar] [CrossRef]
| Route of Administration | Main Routes | Advantages | Disadvantages |
|---|---|---|---|
| Topical | Corneal and conjunctival-scleral pathway |
|
|
| Intravitreal | Direct injection into the vitreous humour |
|
|
| Periocular | Primarily via the trans-scleral pathway |
|
|
| Suprachoroidal | Hollow microneedle injection targeting the choroidal layer | Drug effects at sites maximized by sclera bypassing |
|
| Systemic | Reach of the choroid through the systemic circulation | Better patient compliance |
|
| Advantages | Disadvantages/Limitations |
|---|---|
| High encapsulation efficiency | Initial burst release from SLNs |
| High ocular permeation | Low drug loading capacity |
| Appropriated pharmacokinetic properties | Lack of recent extended clinical trials since most of the studies are just in vivo assessment |
| Sustained and controlled release | The toxicity of lipid nanoparticles on retinal cells is not entirely studied |
| Enhancing drug pre-corneal retention time and drug corneal permeability | |
| Increase ocular bioavailability and distribution | |
| Prevent ocular toxicity | |
| Good stability and biocompatibility |
| Lipid-Based Nanosystems | Encapsulated Drug | Disease | Administration Route | Main Insights | Ref. |
|---|---|---|---|---|---|
| Chitosan-coated liposomes | Triamcinolone Acetonide | Macular Edema | Topical | Improved permeability compared to control suspensions. Prolonged drug residence time on the ocular surface and conjunctival sac by sustained release from the delivery system and reduced pre-corneal drug loss. Good biocompatibility. | [32] |
| NLC | Triamcinolone Acetonide | Uveitis | Topical | cTA-NLC exhibited slow and sustained in vitro release, good transcorneal permeation ex vivo, and good biocompatibility. The formulation also showed enhanced ocular bioavailability and anti-inflammatory response. | [33] |
| SLN | Fluticasone | - | Topical | Optimized FP-loaded SLNs displayed an efficient entrapment capacity, small particle size, good stability, and Higuchi release pattern. They also exhibited augmented anti-inflammatory effects when compared with pure and marketed drugs. | [34] |
| PCL-PEG-PCL micelles | Dexametasone | Uveitis | Injection | DEX-loaded PCL-PEG-PCL micelles showed an improved anti-inflammatory response when compared to the marketed drug once they could reduce the clinical symptoms of uveitis after a lag time. They demonstrate some potential as carriers for DEX in treating anterior uveitis. | [35] |
| Nanoemulsion | Celecoxib | - | - | NEs increased drug flux through rabbit cornea. They also significantly increased rabbit cornea partitioning, flux, and permeability coefficient. CXB NE formulations can act as permeation enhancers to improve corneal drug delivery. | [36] |
| Nanoemulsion | Tacrolimus | - | Topical | Optimized CNE formulation exhibited prolonged retention at the corneal surface. In vivo ocular pharmacokinetic studies revealed an increased AUC of the formulation compared to the marketed drug. In vitro cytotoxicity study confirmed the safety of the CNE formulation. | [37] |
| Chitosan-coated nanoemulsions | Ibuprofen | Dry eye | - | The optimized formulation showed appropriated physicochemical properties for ophthalmic application, good stability, and can be easily sterilized after preparation. The formulation exhibited mucoadhesive properties and excellent biocompatibility. It also provided a prolonged residence time at the ocular surface. | [29] |
| Liposomes | Flurbiprofen | - | Intravitreous administration | LAP system increased the drug retention time in ocular tissues and high ocular bioavailability. The system showed the capability to decrease inflammatory reactions. It offers good potential for intravitreal drug-sustained delivery. | [38] |
| Liposomes | Bevacizumab | Choroidal Neovascularization | Intravitreous administration | Bev-MVLs exhibited high encapsulation efficiency and sustained drug release effects in vitro and in vivo. The structural stability of bevacizumab was maintained. The formulation also significantly inhibited the thickness of CNV lesions in in vivo studies. | [39] |
| Lipid Nanoparticles Type | Natural Compound | Disease | Administration Route | Main Insights | Ref. |
|---|---|---|---|---|---|
| PVCL-PVA-PEG nanomicelles | Curcumin | Corneal and choroid neovascularization | Topical | Curcumin PVCL-PVA-PEG nanomicelles had narrow size distribution, high drug encapsulation, and increased storage stability. It enhanced cell uptake, in vivo corneal permeation, and improved anti-inflammatory activity. | [41] |
| PVCL-PVA-PEG micelles | Myricetin | Dry eye and chronic anterior uveitis | Topical | PVCL-PVA-PEG micelle formulation showed high encapsulation of myricetin, no significant cytotoxicity, and good in vivo ocular tolerance. They significantly enhanced the aqueous solubility and stability of the compound and improved in vitro antioxidant and in vivo anti-inflammatory activity. | [42] |
| Lecithin/chitosan nanoparticles | Resveratrol | Age-related macular degeneration, diabetic retinopathy, glaucoma, cataracts. | Topical | RMLCN optimized formulation was retained on the eye surface, ensuring a sustained drug delivery. | [30] |
| Micelles | Resveratrol | - | Topical | Res was highly loaded into micelles. They provided more chemical stability in an aqueous solution, good short-term storage and exhibited good tolerance. Cornea permeation was also greatly improved. | [44] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ana, R.d.; Fonseca, J.; Karczewski, J.; Silva, A.M.; Zielińska, A.; Souto, E.B. Lipid-Based Nanoparticulate Systems for the Ocular Delivery of Bioactives with Anti-Inflammatory Properties. Int. J. Mol. Sci. 2022, 23, 12102. https://doi.org/10.3390/ijms232012102
Ana Rd, Fonseca J, Karczewski J, Silva AM, Zielińska A, Souto EB. Lipid-Based Nanoparticulate Systems for the Ocular Delivery of Bioactives with Anti-Inflammatory Properties. International Journal of Molecular Sciences. 2022; 23(20):12102. https://doi.org/10.3390/ijms232012102
Chicago/Turabian StyleAna, Raquel da, Joel Fonseca, Jacek Karczewski, Amélia M. Silva, Aleksandra Zielińska, and Eliana B. Souto. 2022. "Lipid-Based Nanoparticulate Systems for the Ocular Delivery of Bioactives with Anti-Inflammatory Properties" International Journal of Molecular Sciences 23, no. 20: 12102. https://doi.org/10.3390/ijms232012102
APA StyleAna, R. d., Fonseca, J., Karczewski, J., Silva, A. M., Zielińska, A., & Souto, E. B. (2022). Lipid-Based Nanoparticulate Systems for the Ocular Delivery of Bioactives with Anti-Inflammatory Properties. International Journal of Molecular Sciences, 23(20), 12102. https://doi.org/10.3390/ijms232012102







