Monooxygenase- and Dioxygenase-Catalyzed Oxidative Dearomatization of Thiophenes by Sulfoxidation, cis-Dihydroxylation and Epoxidation
Abstract
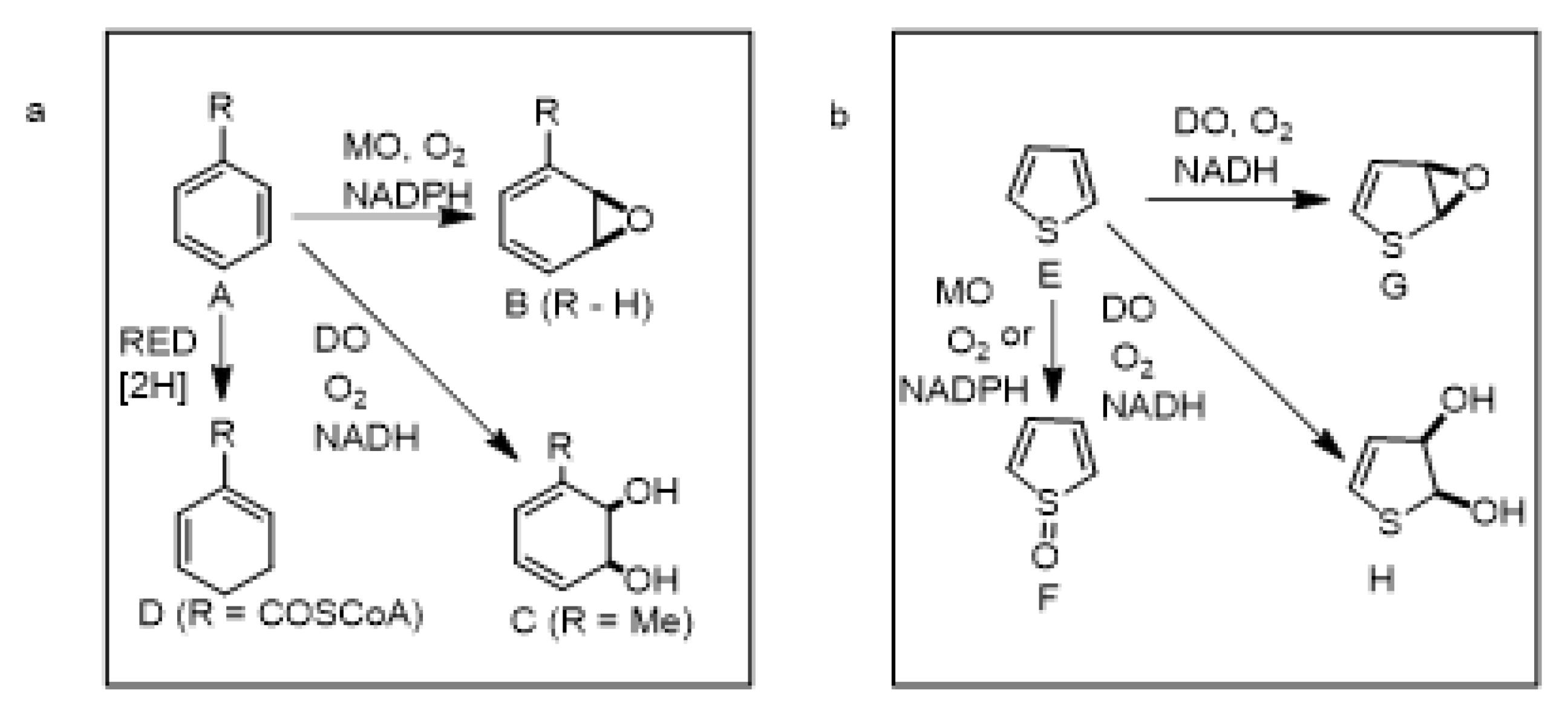
1. Introduction
2. Dioxygenase-Catalyzed Dearomatization of Thiophenes 1a–g
2.1. Enzymatic Oxidation of Thiophenes 1a–g to Yield cis-Dihydrodiols
2.2. Oxidations of Thiophenes 1a–g to Yield Sulfoxide Metabolites
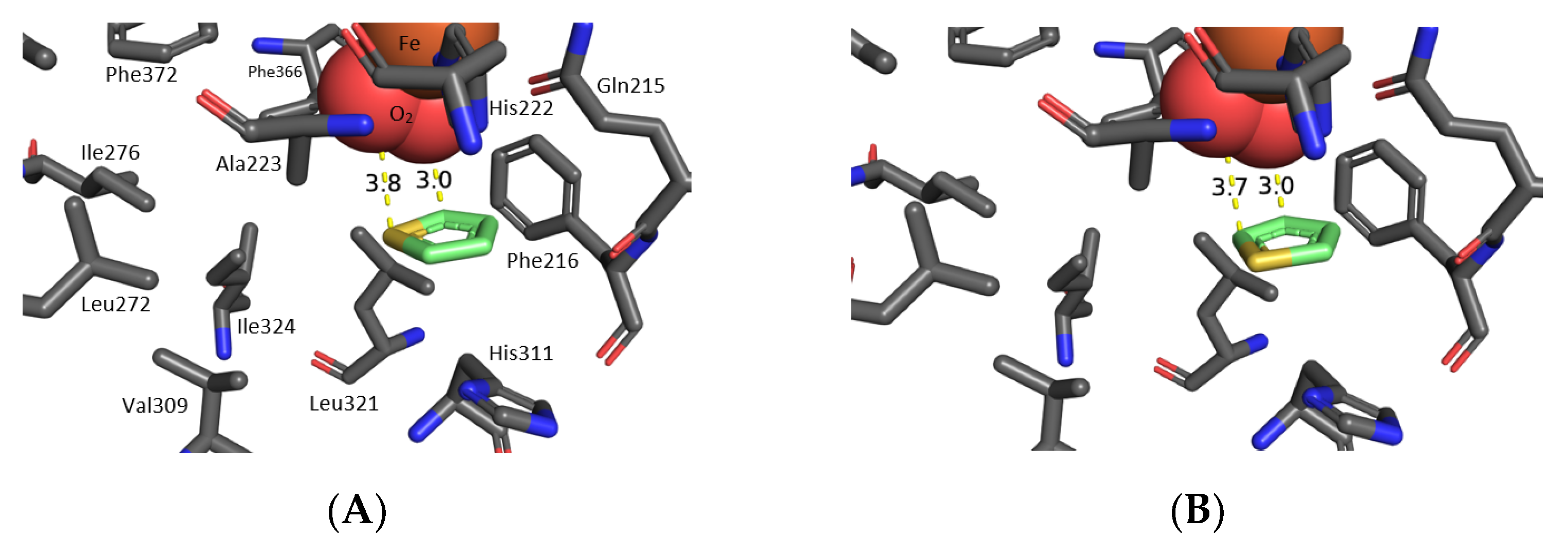
2.3. Molecular Docking of Thiophenes 1a and 1g at the TDO Active Site
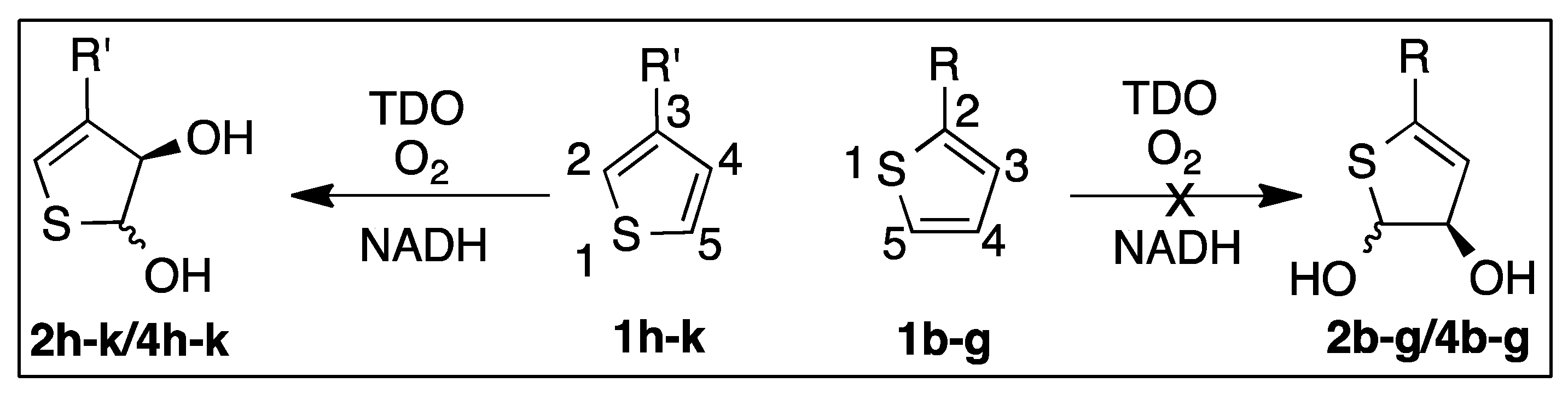
3. Dioxygenase-Catalyzed Dearomatization of 3-Substituted Thiophenes 1h–k
3.1. Oxidations of Thiophenes 1h–k to Yield cis-Dihydrodiol and Sulfoxide Metabolites
3.2. Molecular Docking of 3-Phenylthiophene 1k at the TDO Active Site
4. Dioxygenase-Catalyzed Dearomatization of Benzo[b]thiophenes 10a–d
4.1. Biotransformations of Benzo[b]thiophenes 10a–d to Yield cis-Dihydrodiols 11a–c
4.2. Biotransformations of Benzo[b]thiophenes 10a–d to Yield cis-Dihydrodiols 17a–d and trans-Dihydrodiols 19a, 19d
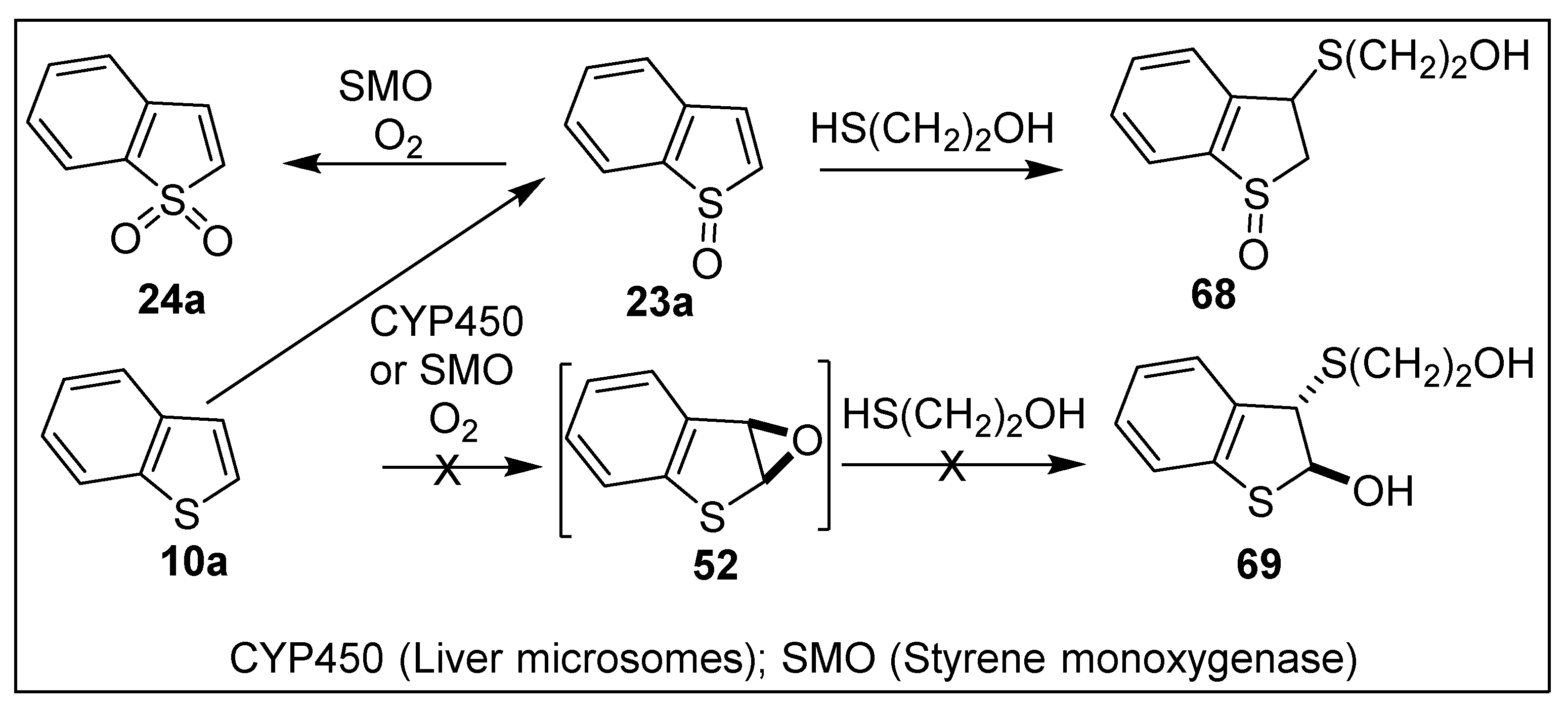
4.3. Biotransformations of Benzo[b]thiophenes 10a–d to Yield Sulfoxides 23a–d
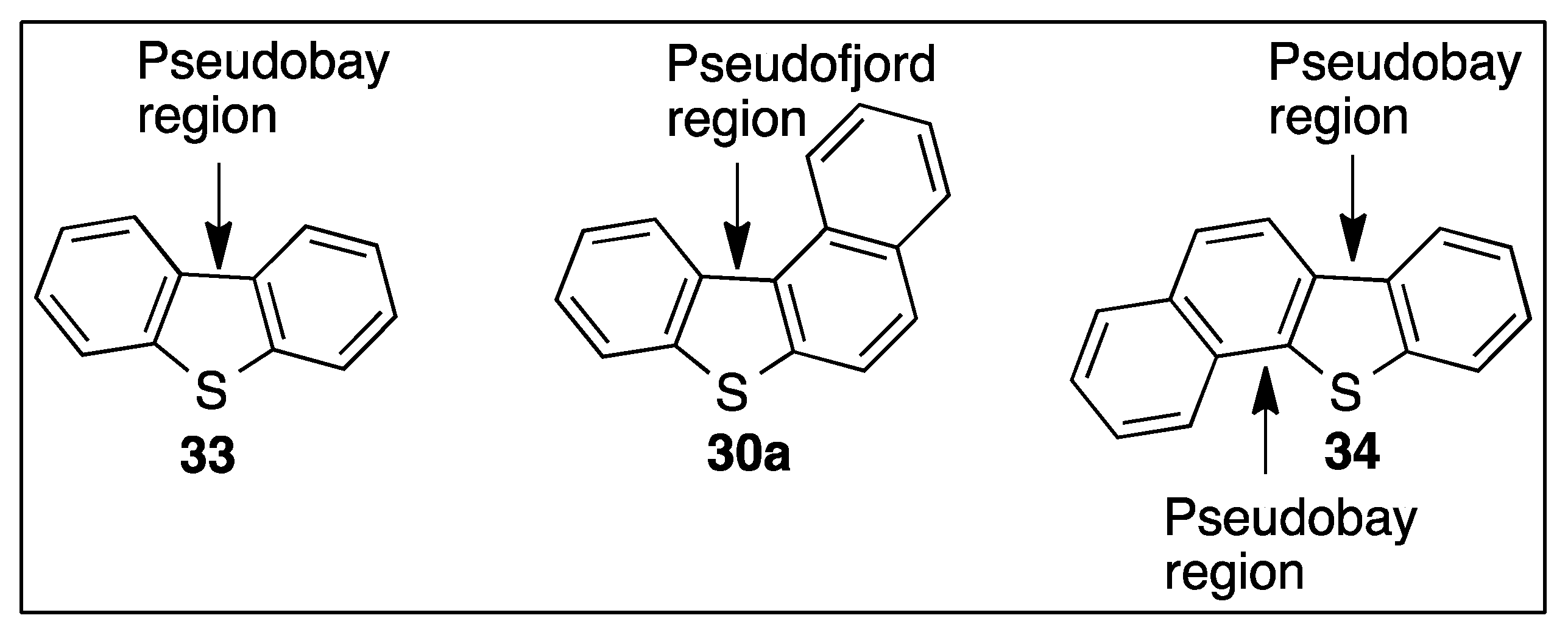
5. Dioxygenase-Catalyzed Dearomatization of Tri- and Tetra-Cyclic Thiaarenes
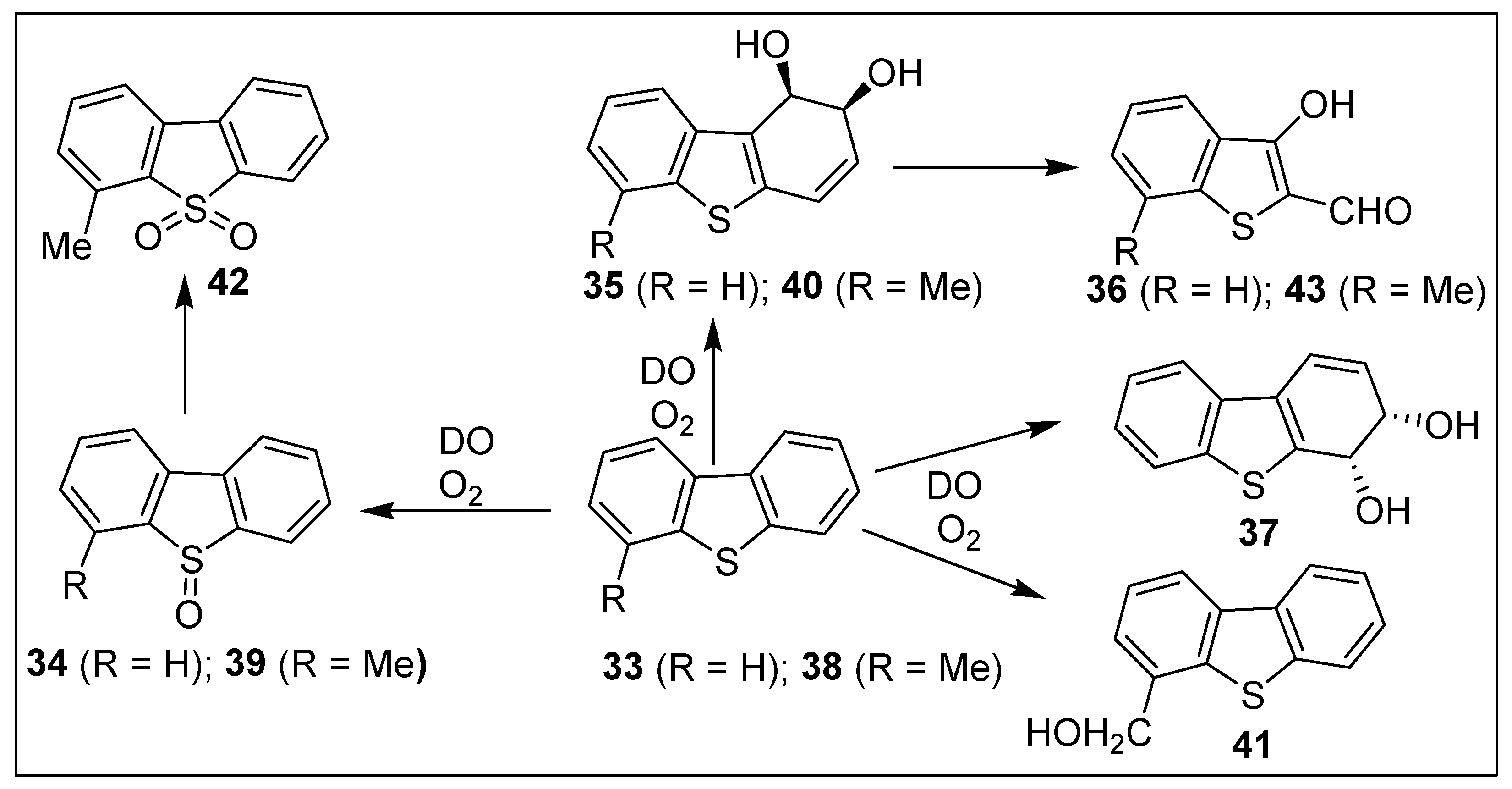
5.1. Dioxygenase-Catalyzed Biotransformations of Dibenzo[b,d]thiophenes 33 and 38
5.2. Dioxygenase-Catalyzed Biotransformation of Benzo[b]naphtho[1,2-d]thiophene 30a and Tetrahydrobenzo[b]naphtho[1,2-d]thiophene 45
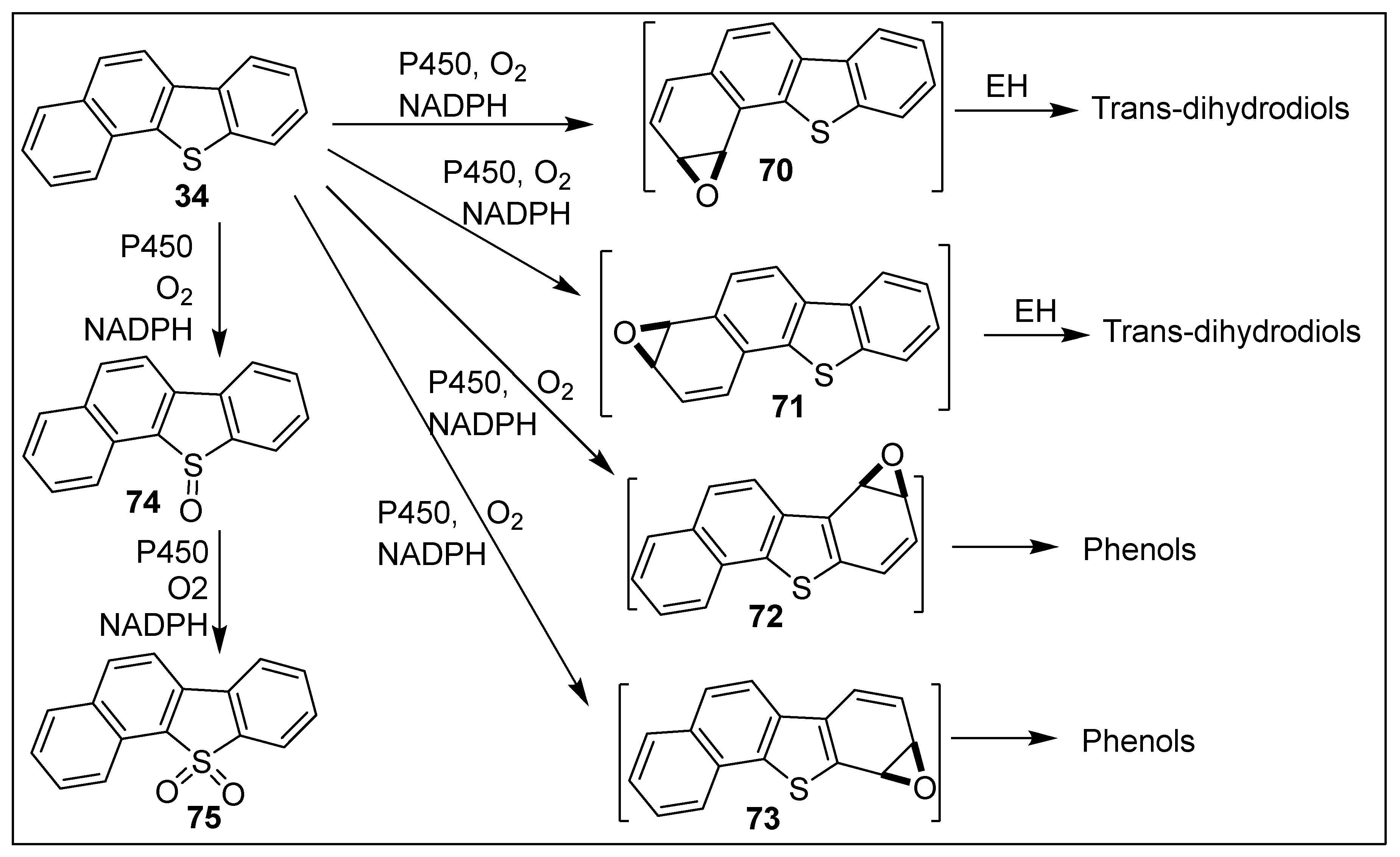
5.3. Dioxygenase-Catalyzed Biotransformation of Benzo[b]naphtho[2,1-d]thiophene 34
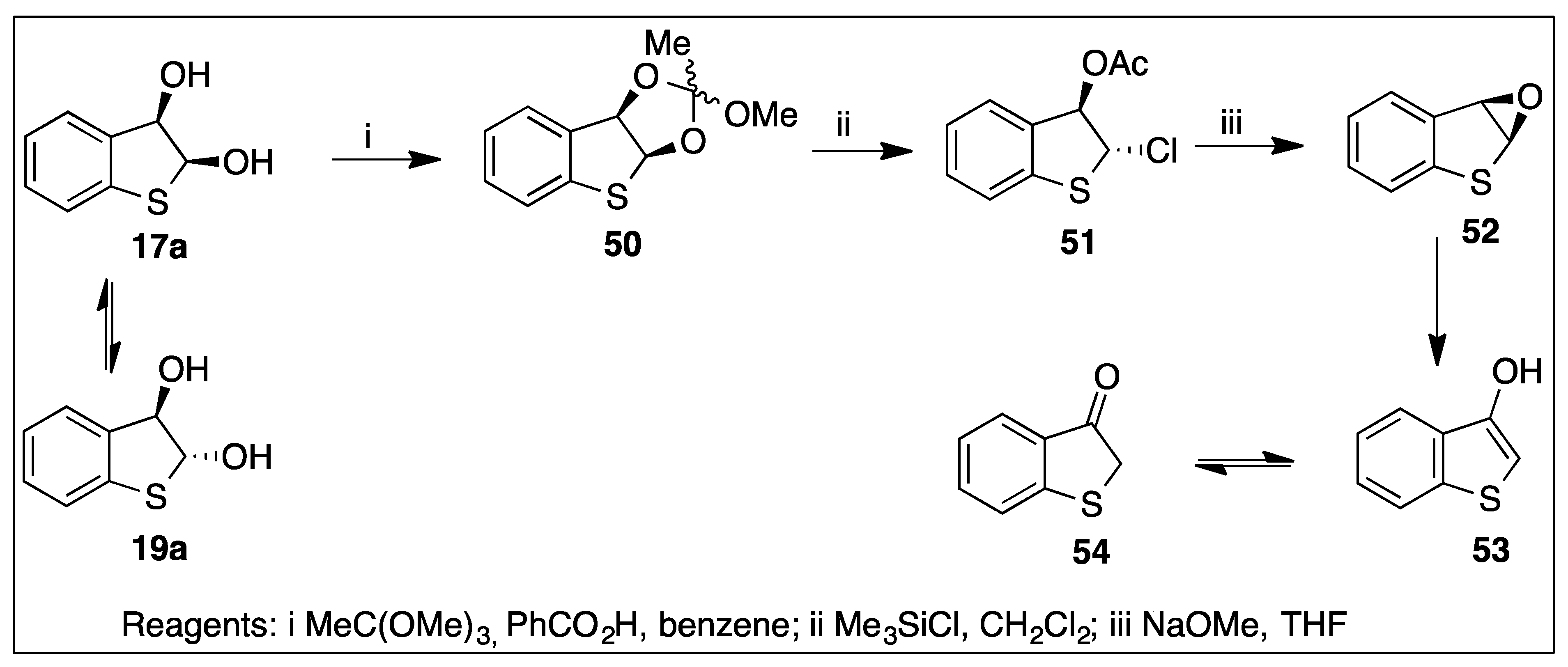
5.4. Application of Thiophene cis-Dihydrodiols in Thiophene Epoxide Synthesis
6. Monooxygenase-Catalyzed Sulfoxidation and Epoxidation of Thiophenes
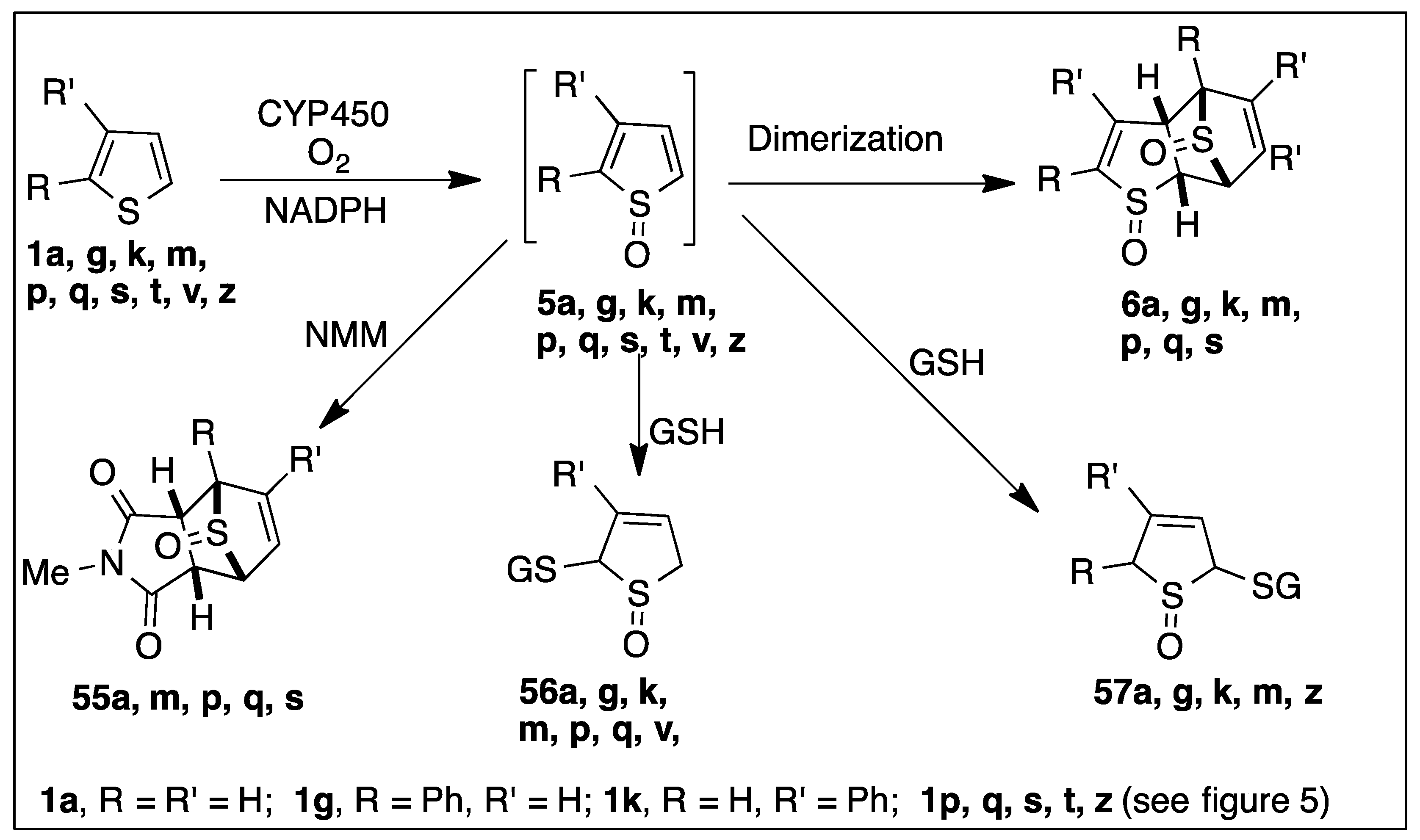
6.1. CYP450-Catalyzed Sulfoxidation of Monocyclic Thiophenes
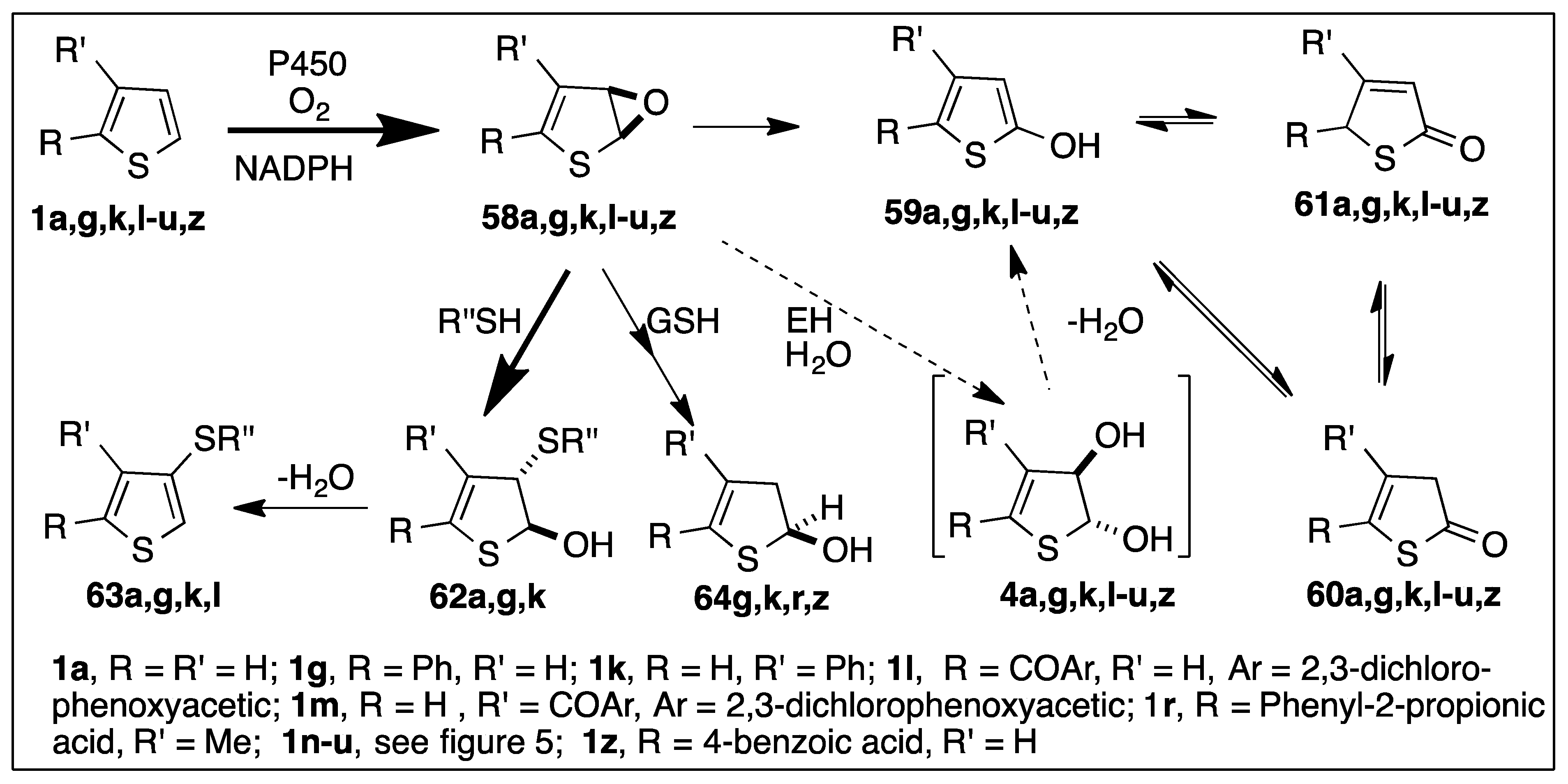
6.2. CYP450-Catalyzed Epoxidation of Monocyclic Thiophenes
6.3. Monooxygenase-Catalyzed Oxidation of Polycyclic Thiophenes
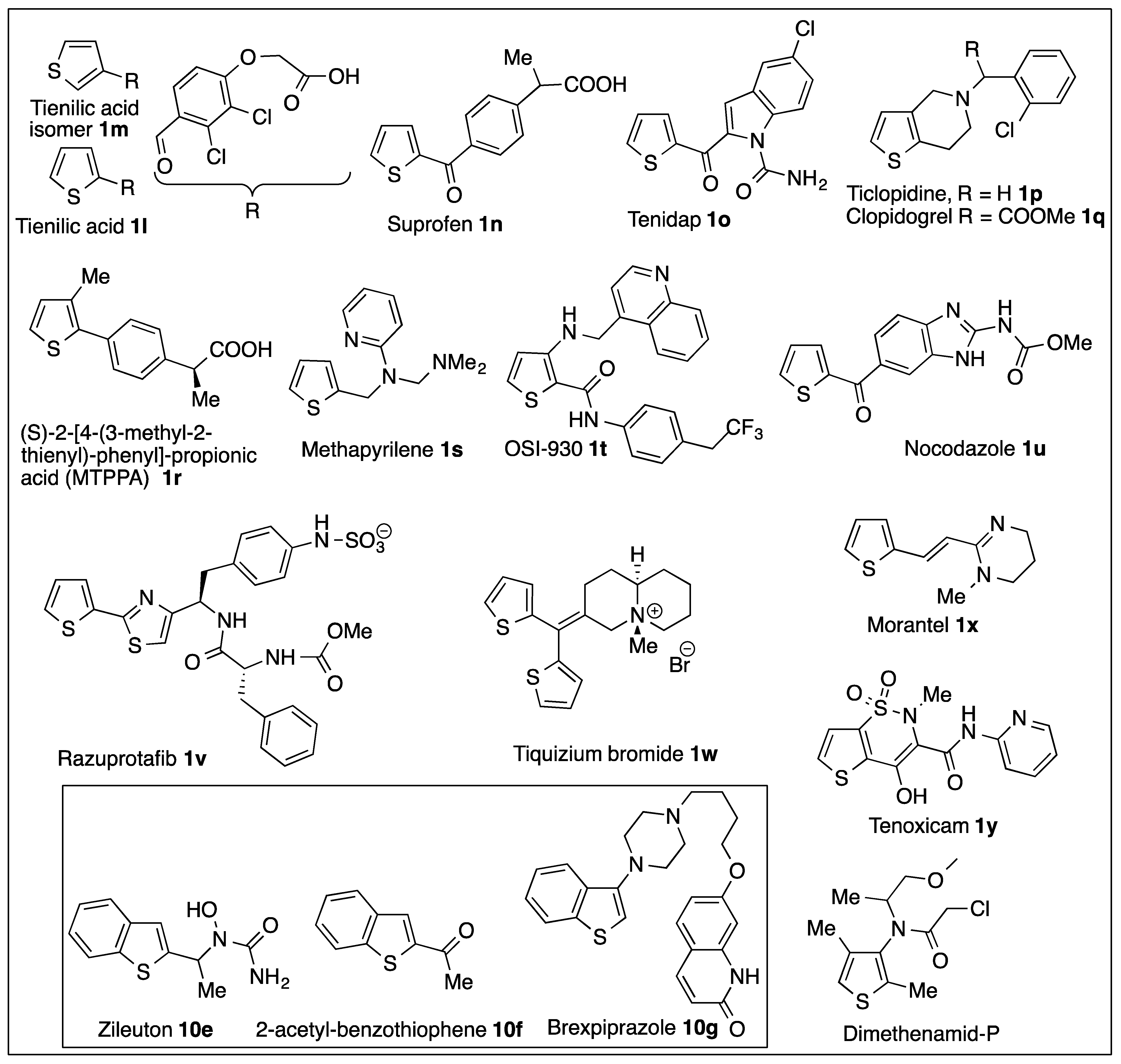
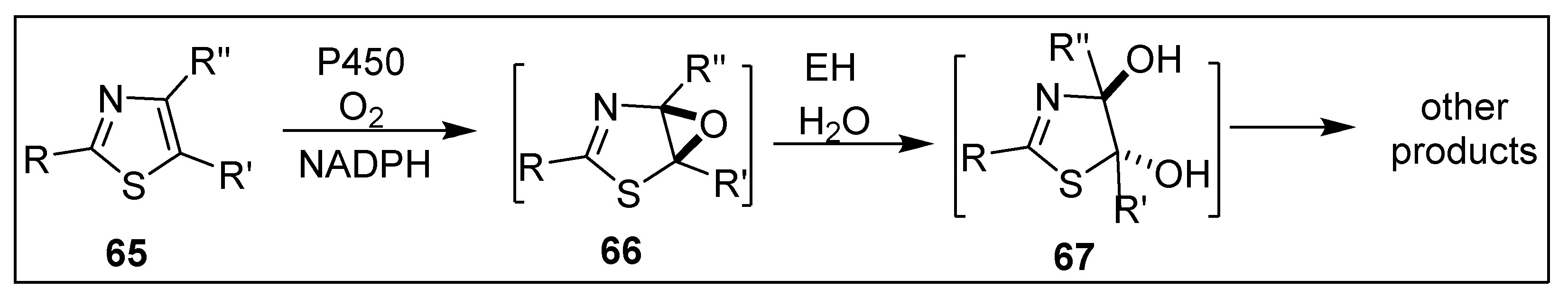
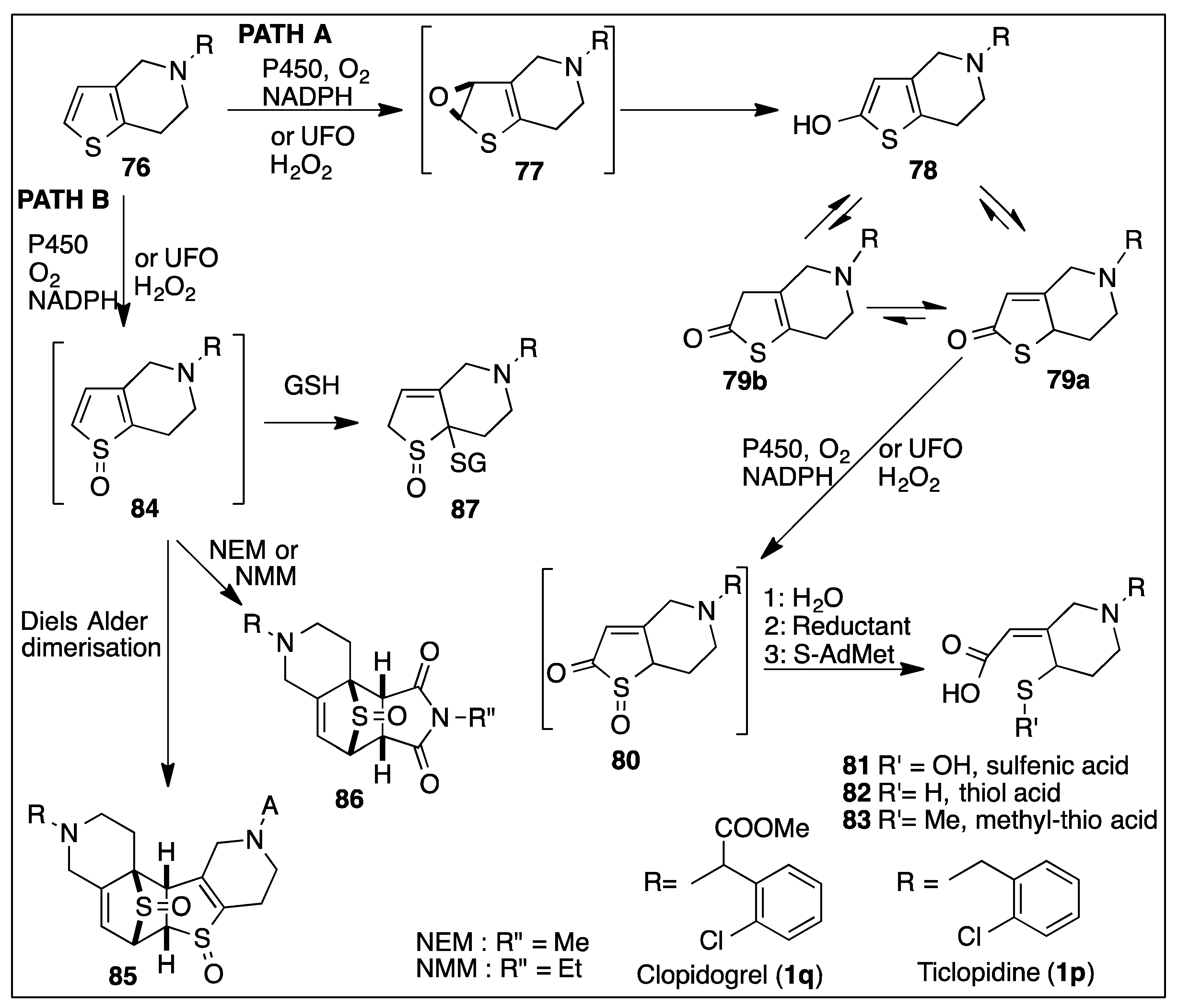
6.4. Monooxygenase-Catalyzed Thiophene Ring Oxidation of Thienopyridine Prodrugs
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Dedication
References
- Monks, T.J.; Butterworth, M.; Lau, S.S. The fate of benzene-oxide. Chem. Biol. Interact. 2010, 184, 201–206. [Google Scholar] [CrossRef]
- Ziffer, H.; Jerina, D.M.; Gibson, D.T.; Kobal, V.M. Absolute stereochemistry of the (+)-cis-1,2-dihydroxy-3-methylcyclohexa-3,5-diene produced from toluene by Pseudomonas putida. J. Am. Chem. Soc. 1973, 95, 4048–4049. [Google Scholar] [CrossRef]
- Heider, J.; Fuchs, G. Anaerobic Metabolism of Aromatic Compounds. Eur. J. Biochem. 1997, 243, 577–596. [Google Scholar] [CrossRef] [PubMed]
- Boyd, D.R.; Sheldrake, G.N. The dioxygenase-catalysed formation of vicinal cis-diols. Nat. Prod. Rep. 1998, 15, 309–324. [Google Scholar] [CrossRef]
- Hudlicky, T.; Gonzalez, D.; Gibson, D.T. Enzymatic dihydroxylation of aromatics in enantioselective synthesis: Expanding asymmetric methodology. Aldrichim. Acta 1999, 32, 35–62. [Google Scholar] [CrossRef]
- Johnson, R.A. Microbial Arene Oxidations. Org. React. 2004, 63, 117–264. [Google Scholar] [CrossRef]
- Lewis, S.E. Asymmetric Dearomatization Under Enzymatic Conditions. In Asymmetric Dearomatization Reactions; Wiley-VHF Verlag GMBH: Weinheim, Germany, 2016; pp. 279–346. [Google Scholar] [CrossRef]
- Hudlicky, T. Benefits of Unconventional Methods in the Total Synthesis of Natural Products. ACS Omega 2018, 3, 17326–17340. [Google Scholar] [CrossRef] [PubMed]
- Taher, E.S.; Banwell, M.; Buckler, J.; Yan, Q.; Lan, P. The Exploitation of Enzymatically-Derivedcis-1,2-Dihydrocatechols and Related Compounds in the Synthesis of Biologically Active Natural Products. Chem. Rec. 2017, 18, 239–264. [Google Scholar] [CrossRef]
- Solà, M. Why Aromaticity Is a Suspicious Concept? Why? Front. Chem. 2017, 5, 22. [Google Scholar] [CrossRef]
- Horner, K.E.; Karadakov, P.B. Chemical Bonding and Aromaticity in Furan, Pyrrole, and Thiophene: A Magnetic Shielding Study. J. Org. Chem. 2013, 78, 8037–8043. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Gunaratne, N.; Haughey, S.A.; Kennedy, M.A.; Malone, J.F.; Allen, C.C.R.; Dalton, H. Dioxygenase-catalysed oxidation of monosubstituted thiophenes: Sulfoxidation versus dihydrodiol formation. Org. Biomol. Chem. 2003, 1, 984–994. [Google Scholar] [CrossRef]
- Jacob, J. Sulfur Analogues of Polycyclic Aromatic Hydrocarbons (Thiaarenes); Cambridge University Press: Cambridge, UK, 1990; pp. 41–54. [Google Scholar]
- Boshagh, F.; Rahmani, M.; Rostami, K.; Yousefifar, M. Key Factors Affecting the Development of Oxidative Desulfurization of Liquid Fuels: A Critical Review. Energy Fuels 2021, 36, 98–132. [Google Scholar] [CrossRef]
- Fedorak, P. Microbial metabolism of organosulfur compounds in petroleum. In Geochemistry of Sulfur in Fossil Fuels; Orr, W., White, C.M., Eds.; American Chemical Society: Washington, DC, USA, 1990; Volume 429, pp. 93–112. [Google Scholar] [CrossRef]
- Fedorak, P.M.; Grbić-Galić, D. Aerobic Microbial Cometabolism of Benzothiophene and 3-Methylbenzothiophene. Appl. Environ. Microbiol. 1991, 57, 932–940. [Google Scholar] [CrossRef]
- Saftic, S.; Fedorak, P.M.; Andersson, J.T. Diones, sulfoxides, and sulfones from the aerobic cometabolism of methylbenzothiophenes by Pseudomonas strain BT1. Environ. Sci. Technol. 1992, 26, 1759–1764. [Google Scholar] [CrossRef]
- Fedorak, P.M.; Peakman, T.M. Aerobic microbial metabolism of some alkylthiophenes found in petroleum. Biodegradation 1992, 2, 223–236. [Google Scholar] [CrossRef]
- Kropp, K.G.; Gonçalves, J.A.; Andersson, J.T.; Fedorak, P.M. Microbially Mediated Formation of Benzonaphthothiophenes from Benzo[ b ]thiophenes. Appl. Environ. Microbiol. 1994, 60, 3624–3631. [Google Scholar] [CrossRef]
- Kropp, K.G.; Goncalves, J.A.; Andersson, J.T.; Fedorak, P.M. Bacterial Transformations of Benzothiophene and Methylbenzothiophenes. Environ. Sci. Technol. 1994, 28, 1348–1356. [Google Scholar] [CrossRef] [PubMed]
- Kropp, K.G.; Saftic, S.; Andersson, J.T.; Fedorak, P.M. Transformations of six isomers of dimethylbenzothiophene by three Pseudomonas strains. Biodegradation 1996, 7, 203–221. [Google Scholar] [CrossRef] [PubMed]
- Fedorak, P.M.; Coy, D.L.; Peakman, T.M. Microbial metabolism of some 2,5-substituted thiophenes. Biodegradation 1996, 7, 313–327. [Google Scholar] [CrossRef]
- Kropp, K.G.; Andersson, J.T.; Fedorak, P.M. Bacterial transformations of 1,2,3,4-tetrahydrodibenzothiophene and dibenzothiophene. Appl. Environ. Microbiol. 1997, 63, 3032–3042. [Google Scholar] [CrossRef]
- Kropp, K.G.; Andersson, J.T.; Fedorak, P.M. Bacterial transformations of naphthothiophenes. Appl. Environ. Microbiol. 1997, 63, 3463–3473. [Google Scholar] [CrossRef]
- Bressler, D.C.; Norman, J.A.; Fedorak, P.M. Ring cleavage of sulfur heterocycles: How does it happen? Biodegradation 1997, 8, 297–311. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Boyle, R.; McMurray, B.T.; Evans, T.A.; Malone, J.F.; Dalton, H.; Chima, J.; Sheldrake, G.N. Biotransformation of unsaturated heterocyclic rings by Pseudomonas putida to yield cis-diols. J. Chem. Soc. Chem. Commun. 1993, 49–51. [Google Scholar] [CrossRef]
- Eaton, R.W.; Nitterauer, J.D. Biotransformation of benzothiophene by isopropylbenzene-degrading bacteria. J. Bacteriol. 1994, 176, 3992–4002. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Brand, J.; Gibson, D. Stereospecific Sulfoxidation by Toluene and Naphthalene Dioxygenases. Biochem. Biophys. Res. Commun. 1995, 212, 9–15. [Google Scholar] [CrossRef]
- Bugg, T.D.; Ramaswamy, S. Non-heme iron-dependent dioxygenases: Unravelling catalytic mechanisms for complex enzymatic oxidations. Curr. Opin. Chem. Biol. 2008, 12, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Barry, S.M.; Challis, G.L. Mechanism and Catalytic Diversity of Rieske Non-Heme Iron-Dependent Oxygenases. ACS Catal. 2013, 3, 2362–2370. [Google Scholar] [CrossRef]
- Ibrahim, S.R.M.; Abdallah, H.M.; El-Halawany, A.M.; Mohamed, G.A. Naturally occurring thiophenes: Isolation, purification, structural elucidation, and evaluation of bioactivities. Phytochem. Rev. 2016, 15, 197–220. [Google Scholar] [CrossRef]
- Misawa, N.; Shindo, K.; Takahashi, H.; Suenaga, H.; Iguchi, K.; Okazaki, H.; Harayama, S.; Furukawa, K. Hydroxylation of various molecules including heterocyclic aromatics using recombinant Escherichia coli cells expressing modified biphenyl dioxygenase genes. Tetrahedron 2002, 58, 9605–9612. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Hand, M.V.; Groocock, M.R.; Kerley, N.A.; Dalton, H.; Chima, J.; Sheldrake, G.N. Stereodirecting substituent effects during enzyme-catalysed synthesis of cis-dihydrodiol metabolites of 1,4-disubstituted benzene substrates. J. Chem. Soc. Chem. Commun. 1993, 974–976. [Google Scholar] [CrossRef]
- Yildirim, S.; Franco, T.T.; Wohlgemuth, R.; Kohler, H.-P.E.; Witholt, B.; Schmid, A. Recombinant Chlorobenzene Dioxygenase fromPseudomonas sp. P51: A Biocatalyst for Regioselective Oxidation of Aromatic Nitriles. Adv. Synth. Catal. 2005, 347, 1060–1072. [Google Scholar] [CrossRef]
- Hoye, T.R.; Jeffrey, C.S.; Shao, F. Mosher ester analysis for the determination of absolute configuration of stereogenic (chiral) carbinol carbons. Nat. Protoc. 2007, 2, 2451–2458. [Google Scholar] [CrossRef] [PubMed]
- Boyd, D.R.; Dorrity, M.R.J.; Hand, M.V.; Malone, J.F.; Sharma, N.D.; Dalton, H.; Gray, D.J.; Sheldrake, G.N. Enantiomeric excess and absolute configuration determination of cis-dihydrodiols from bacterial metabolism of monocyclic arenes. J. Am. Chem. Soc. 1991, 113, 666–667. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Boyle, R.; McMordie, R.S.; Chima, J.; Dalton, H. A H-nmr method for the determination of enantiomeric excess and absolute configuration of cis-dihydrodiol metabolites of polycyclic arenes and heteroarenes. Tetrahedron Lett. 1992, 33, 1241–1244. [Google Scholar] [CrossRef]
- Burgess, K.; Porte, A.M. A Reagent for Determining Optical Purities of Diols by Formation of Diastereomeric Arylboronate Esters. Angew. Chem. Int. Ed. 1994, 33, 1182–1184. [Google Scholar] [CrossRef]
- Resnick, S.M.; Torok, D.S.; Lee, K.; Brand, J.M.; Gibson, D.T. Regiospecific and streoselective hydroxylation pf 1-indanone and 2-indanone by naphthalene dioxygenase and toluene dioxygenase. Appl. Environ. Microbiol. 1995, 61, 847. [Google Scholar] [CrossRef] [PubMed]
- Boyd, D.R.; Sharma, N.D.; Goodrich, P.A.; Malone, J.F.; McConville, G.; Harrison, J.S.; Stevenson, P.J.; Allen, C.C. Enantiopurity and absolute configuration determination of arenecis-dihydrodiol metabolites and derivatives using chiral boronic acids. Chirality 2017, 30, 5–18. [Google Scholar] [CrossRef]
- Gawronski, J.K.; Kwit, M.; Boyd, D.R.; Sharma, N.D.; Malone, A.J.F.; Drake, A.F. Absolute Configuration, Conformation, and Circular Dichroism of Monocyclic Arene Dihydrodiol Metabolites: It is All Due to the Heteroatom Substituents. J. Am. Chem. Soc. 2005, 127, 4308–4319. [Google Scholar] [CrossRef]
- Kwit, M.; Gawroński, J.; Boyd, D.R.; Sharma, N.D.; Kaik, M.; More O’Ferrall, R.A.; Kudavalli, J.S. Toluene Dioxygenase-Catalyzed Synthesis of cis-Dihydrodiol Metabolites from 2-Substituted Naphthalene Substrates: Assignments of Absolute Configurations and Conformations from Circular Dichroism and Optical Rotation Measurements. Chem.—Eur. J. 2008, 14, 11500–11511. [Google Scholar] [CrossRef]
- Kwit, M.; Gawronski, J.; Sbircea, L.; Sharma, N.D.; Kaik, M.; Boyd, D.R. Circular dichroism spectra, optical rotations and absolute configurations ofcis-dihydrodiol metabolites of quinoline and derivatives: The role of the nitrogen atom. Chirality 2009, 21, E37–E47. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Ljubez, V.; McGeehin, P.K.M.; Stevenson, P.J.; Blain, M.; Allen, C.C.R. Chemoenzymatic synthesis of monocyclic arene oxides and arene hydrates from substituted benzene substrates. Org. Biomol. Chem. 2013, 11, 3020–3029. [Google Scholar] [CrossRef] [PubMed]
- Jerina, D.M.; Selander, H.; Yagi, H.; Wells, M.C.; Davey, J.F.; Mahadevan, V.; Gibson, D.T. Dihydrodiols from anthracene and phenanthrene. J. Am. Chem. Soc. 1976, 98, 5988–5996. [Google Scholar] [CrossRef] [PubMed]
- Parales, R.E.; Resnick, S.M.; Yu, C.-L.; Boyd, D.R.; Sharma, N.D.; Gibson, D.T. Regioselectivity and Enantioselectivity of Naphthalene Dioxygenase during Arene cis -Dihydroxylation: Control by Phenylalanine 352 in the α Subunit. J. Bacteriol. 2000, 182, 5495–5504. [Google Scholar] [CrossRef] [PubMed]
- Boyd, D.R.; Sharma, N.D.; Coen, G.P.; Hempenstall, F.; Ljubez, V.; Malone, J.F.; Allen, C.C.R.; Hamilton, J.T.G. Regioselectivity and stereoselectivity of dioxygenase catalysed cis-dihydroxylation of mono- and tri-cyclic azaarene substrates. Org. Biomol. Chem. 2008, 6, 3957–3966. [Google Scholar] [CrossRef]
- Alien, C.C.R.; Boyd, D.R.; Dalton, H.; Sharma, N.D.; Haughey, S.A.; McMordie, R.A.S.; McMurray, B.T.; Sheldrake, G.N.; Sproule, K. Sulfoxides of high enantiopurity from bacterial dioxygenase-catalysed oxidation. J. Chem. Soc. Chem. Commun. 1995, 119–120. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Haughey, S.A.; Kennedy, M.A.; McMurray, B.T.; Sheldrake, G.N.; Allen, C.C.R.; Dalton, H.; Sproule, K. Toluene and naphthalene dioxygenase-catalysed sulfoxidation of alkyl aryl sulfides. J. Chem. Soc. Perkin Trans. 1998, 1929–1934. [Google Scholar] [CrossRef]
- Kerridge, A.; Willetts, A.; Holland, H. Stereoselective oxidation of sulfides by cloned naphthalene dioxygenase. J. Mol. Catal. B Enzym. 1999, 6, 59–65. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Byrne, B.E.; Haughey, S.A.; Kennedy, M.A.; Allen, C.C.R. Dioxygenase-catalysed oxidation of alkylaryl sulfides: Sulfoxidation versus cis-dihydrodiol formation. Org. Biomol. Chem. 2004, 2, 2530–2537. [Google Scholar] [CrossRef]
- Mansuy, D.; Valadon, P.; Erdelmeier, I.; Lopez-Garcia, P.; Amar, C.; Girault, J.P.; Dansette, P.M. Thiophene S-oxides as new reactive metabolites: Formation by cytochrome P-450 dependent oxidation and reaction with nucleophiles. J. Am. Chem. Soc. 1991, 113, 7825–7826. [Google Scholar] [CrossRef]
- Treiber, A.; Dansette, P.M.; El Amri, H.; Girault, J.-P.; Ginderow, D.; Mornon, A.J.-P.; Mansuy, D. Chemical and Biological Oxidation of Thiophene: Preparation and Complete Characterization of Thiophene S-Oxide Dimers and Evidence for Thiophene S-Oxide as an Intermediate in Thiophene Metabolism In Vivo and In Vitro. J. Am. Chem. Soc. 1997, 119, 1565–1571. [Google Scholar] [CrossRef]
- Dansette, P.M.; Bertho, G.; Mansuy, D. First evidence that cytochrome P450 may catalyze both S-oxidation and epoxidation of thiophene derivatives. Biochem. Biophys. Res. Commun. 2005, 338, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Medower, C.; Wen, L.; Johnson, W.W. Cytochrome P450 Oxidation of the Thiophene-Containing Anticancer Drug 3-[(Quinolin-4-ylmethyl)-amino]-thiophene-2-carboxylic Acid (4-Trifluoromethoxy-phenyl)-amide to an Electrophilic Intermediate. Chem. Res. Toxicol. 2008, 21, 1570–1577. [Google Scholar] [CrossRef]
- Rademacher, P.M.; Woods, C.M.; Huang, Q.; Szklarz, G.D.; Nelson, S.D. Differential Oxidation of Two Thiophene-Containing Regioisomers to Reactive Metabolites by Cytochrome P450 2C9. Chem. Res. Toxicol. 2012, 25, 895–903. [Google Scholar] [CrossRef] [PubMed]
- Gramec, D.; Mašič, L.P.; Dolenc, M.S. Bioactivation Potential of Thiophene-Containing Drugs. Chem. Res. Toxicol. 2014, 27, 1344–1358. [Google Scholar] [CrossRef] [PubMed]
- Heine, T.; Scholtissek, A.; Hofmann, S.; Koch, R.; Tischler, D. Accessing Enantiopure Epoxides and Sulfoxides: Related Flavin-Dependent Monooxygenases Provide Reversed Enantioselectivity. ChemCatChem 2019, 12, 199–209. [Google Scholar] [CrossRef]
- Yang, J.; Yuan, Z.; Zhou, Y.; Zhao, J.; Yang, M.; Cheng, X.; Ou, G.; Chen, Y. Asymmetric reductive resolution of racemic sulfoxides by recombinant methionine sulfoxide reductase from a pseudomonas monteilii strain. J. Mol. Catal. B Enzym. 2016, 133, S588–S592. [Google Scholar] [CrossRef]
- Yang, J.; Wen, Y.; Peng, L.; Chen, Y.; Cheng, X.; Chen, Y.-Z. Identification of MsrA homologues for the preparation of (R)-sulfoxides at high substrate concentrations. Org. Biomol. Chem. 2019, 17, 3381–3388. [Google Scholar] [CrossRef]
- Luckarift, H.R.; Dalton, H.; Sharma, N.D.; Boyd, D.R.; Holt, R.A. Isolation and characterisation of bacterial strains containing enantioselective DMSO reductase activity: Application to the kinetic resolution of racemic sulfoxides. Appl. Microbiol. Biotechnol. 2004, 65, 678–685. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; King, A.W.T.; Shepherd, S.D.; Allen, C.C.R.; Holt, R.A.; Luckarift, H.R.; Dalton, H. Stereoselective reductase-catalysed deoxygenation of sulfoxides in aerobic and anaerobic bacteria. Org. Biomol. Chem. 2004, 2, 554–561. [Google Scholar] [CrossRef]
- Anselmi, S.; Aggarwal, N.; Moody, T.S.; Castagnolo, D. Unconventional Biocatalytic Approaches to the Synthesis of Chiral Sulfoxides. ChemBioChem 2020, 22, 298–307. [Google Scholar] [CrossRef]
- Verlinden, S.; Woller, T.; De Proft, F.; Verniest, G.; Alonso, M. Towards the Design of Optically Active Thiophene S-Oxides using Quantum Chemistry. Chem.—Eur. J. 2019, 25, 2840–2851. [Google Scholar] [CrossRef]
- Jenks, W.S.; Matsunaga, N.; Gordon, M. Effects of Conjugation and Aromaticity on the Sulfoxide Bond. J. Org. Chem. 1996, 61, 1275–1283. [Google Scholar] [CrossRef]
- Bongini, A.; Arbizzani, C.; Barbarella, G.; Zambianchi, M.; Mastragostino, M. Thiophene S-oxides: Orbital energies and electrochemical properties. Chem. Commun. 2000, 439–440. [Google Scholar] [CrossRef]
- Pouzet, P.; Erdelmeier, I.; Ginderow, D.; Mornon, J.-P.; Dansette, P.; Mansuy, D. Thiophene S-oxides: Convenient preparation, first complete structural characterization and unexpected dimerization of one of them, 2,5-diphenylthiophene-1-oxide. J. Chem. Soc. Chem. Commun. 1995, 473–474. [Google Scholar] [CrossRef]
- Otani, T.; Miyoshi, M.; Shibata, T.; Matsuo, T.; Hashizume, D.; Tamao, K. Thermally Stable Monosubstituted Thiophene 1-Oxide and 1-Imides Stabilized by a Bulky Rind Group at Their 3-Position: Synthesis, Structure, and Inversion Barriers on the Sulfur Atom. Bull. Chem. Soc. Jpn. 2017, 90, 697–705. [Google Scholar] [CrossRef]
- Mock, W.L. Stable thiophene sulfoxides. J. Am. Chem. Soc. 1970, 92, 7610–7612. [Google Scholar] [CrossRef]
- Xiong, F.; Yang, B.-B.; Zhang, J.; Li, L. Enantioseparation, Stereochemical Assignment and Chiral Recognition Mechanism of Sulfoxide-Containing Drugs. Molecules 2018, 23, 2680. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Loke, P.L.; Carroll, J.G.; Stevenson, P.J.; Hoering, P.; Allen, C.C.R. Toluene Dioxygenase-Catalyzed cis-Dihydroxylation of Quinolines: A Molecular Docking Study and Chemoenzymatic Synthesis of Quinoline Arene Oxides. Front. Bioeng. Biotechnol. 2021, 8. [Google Scholar] [CrossRef]
- Höring, P.; Rothschild-Mancinelli, K.; Sharma, N.D.; Boyd, D.R.; Allen, C.C.R. Oxidative biotransformations of phenol substrates catalysed by toluene dioxygenase: A molecular docking study. J. Mol. Catal. B Enzym. 2016, 134, 396–406. [Google Scholar] [CrossRef][Green Version]
- Friemann, R.; Lee, K.; Brown, E.; Gibson, D.T.; Eklund, H.; Ramaswamy, S. Structures of the multicomponent Rieske non-heme iron toluene 2,3-dioxygenase enzyme system. Acta Crystallogr. Sect. D Biol. Crystallogr. 2008, 65, 24–33. [Google Scholar] [CrossRef]
- Karlsson, A.; Parales, J.V.; Parales, R.E.; Gibson, D.T.; Eklund, H.; Ramaswamy, S. Crystal Structure of Naphthalene Dioxygenase: Side-on Binding of Dioxygen to Iron. Science 2003, 299, 1039–1042. [Google Scholar] [CrossRef]
- Vila, M.A.; Pazos, M.; Iglesias, C.; Veiga, N.; Seoane, G.; Carrera, I. Toluene Dioxygenase-Catalysed Oxidation of Benzyl Azide to Benzonitrile: Mechanistic Insights for an Unprecedented Enzymatic Transformation. ChemBioChem 2016, 17, 291–295. [Google Scholar] [CrossRef]
- Vila, M.A.; Umpiérrez, D.; Seoane, G.; Rodríguez, S.; Carrera, I.; Veiga, N. Computational insights into the oxidation of mono- and 1,4 disubstituted arenes by the Toluene Dioxygenase enzymatic complex. J. Mol. Catal. B Enzym. 2016, 133, S410–S419. [Google Scholar] [CrossRef]
- Vila, M.A.; Umpiérrez, D.; Veiga, N.; Seoane, G.; Carrera, I.; Giordano, S.R. Site-Directed Mutagenesis Studies on the Toluene Dioxygenase Enzymatic System: Role of Phenylalanine 366, Threonine 365 and Isoleucine 324 in the Chemo-, Regio-, and Stereoselectivity. Adv. Synth. Catal. 2017, 359, 2149–2157. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; McIntyre, P.B.A.; Stevenson, P.J.; McRoberts, W.C.; Gohil, A.; Hoering, P.; Allen, C. Enzyme-Catalysed Synthesis of Cyclohex-2-en-1-one cis-Diols from Substituted Phenols, Anilines and Derived 4-Hydroxycyclohex-2-en-1-ones. Adv. Synth. Catal. 2017, 359, 4002–4014. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Brannigan, I.N.; McGivern, C.J.; Nockemann, P.; Stevenson, P.J.; McRoberts, C.; Hoering, P.; Allen, C.C.R. Cis-Dihydroxylation of Tricyclic Arenes and Heteroarenes Catalyzed by Toluene Dioxygenase: A Molecular Docking Study and Experimental Validation. Adv. Synth. Catal. 2019, 361, 2526–2537. [Google Scholar] [CrossRef]
- González-Rosende, M.E.; Castillo, E.; Jennings, W.B.; Malone, J.F. Stereodynamics and edge-to-face CH–π aromatic interactions in imino compounds containing heterocyclic rings. Org. Biomol. Chem. 2017, 15, 1484–1494. [Google Scholar] [CrossRef] [PubMed]
- Boyd, D.R.; Sharma, N.D.; Brannigan, I.N.; Haughey, S.A.; Malone, J.F.; Clarke, D.A.; Dalton, H. Dioxygenase-catalysed formation of cis/trans-dihydrodiol metabolites of mono- and bi-cyclic heteroarenes. Chem. Commun. 1996, 2361–2362. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Brannigan, I.N.; Evans, T.A.; Haughey, S.A.; McMurray, B.T.; Malone, J.F.; McIntyre, P.B.A.; Stevenson, P.J.; Allen, C.C.R. Toluene dioxygenase-catalyzed cis-dihydroxylation of benzo[b]thiophenes and benzo[b]furans: Synthesis of benzo[b]thiophene 2,3-oxide. Org. Biomol. Chem. 2012, 10, 7292–7304. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; McMurray, B.; Haughey, S.A.; Allen, C.C.R.; Hamilton, J.T.G.; McRoberts, W.C.; More O’Ferrall, R.A.; Nikodinovic-Runic, J.; Coulombel, L.A.; et al. Bacterial dioxygenase- and monooxygenase-catalysed sulfoxidation of benzo[b]thiophenes. Org. Biomol. Chem. 2011, 10, 782–790. [Google Scholar] [CrossRef]
- Gai, Z.H.; Yu, B.; Wang, X.Y.; Deng, Z.X.; Xu, P. Microbial transformation of benzothiophenes, with carbazole as the auxiliary substrate, by Sphingomonas sp strain XLDN2–5. Microbiology 2008, 154, 3804–3812. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Haughey, S.A.; Malone, J.F.; McMurray, B.T.; Sheldrake, G.N.; Allen, C.C.R.; Dalton, H. Enantioselective dioxygenase-catalysed formation and thermal racemisation of chiral thiophene sulfoxides. Chem. Commun. 1996, 2363–2364. [Google Scholar] [CrossRef]
- Thiemann, T.; Fujii, H.; Ohira, D.; Arima, K.; Li, Y.; Mataka, S. Cycloaddition of thiophene S-oxides to allenes, alkynes and to benzyne. New J. Chem. 2003, 27, 1377–1384. [Google Scholar] [CrossRef]
- Joost, M.; Nava, M.; Transue, W.J.; Martin-Drumel, M.-A.; McCarthy, M.C.; Patterson, D.; Cummins, C.C. Sulfur monoxide thermal release from an anthracene-based precursor, spectroscopic identification, and transfer reactivity. Proc. Natl. Acad. Sci. USA 2018, 115, 5866–5871. [Google Scholar] [CrossRef]
- Torok, D.S.; Resnick, S.M.; Brand, J.M.; Cruden, D.L.; Gibson, D.T. Desaturation and oxygenation of 1,2-dihydronaphthalene by toluene dioxygenase and naphthalene dioxygenase. J. Bacteriol. 1995, 177, 5799–5805. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Kerley, N.A.; McMordie, R.A.S.; Sheldrake, G.N.; Williams, P.; Dalton, H. Dioxygenase-catalysed oxidation of dihydronaphthalenes to yield arene hydrate and cis-dihydro naphthalenediols. J. Chem. Soc. Perkin Trans. 1996, 67–74. [Google Scholar] [CrossRef]
- Koreeda, M.; Akhtar, M.N.; Boyd, D.R.; Neill, J.D.; Gibson, D.T.; Jerina, D.M. Absolute stereochemistry of cis-1,2-, trans-1,2-, and cis-3,4-dihydrodiol metabolites of phenanthrene. J. Org. Chem. 1978, 43, 1023–1027. [Google Scholar] [CrossRef]
- Jerina, D.M.; Van Bladeren, P.J.; Yagi, H.; Gibson, D.T.; Mahadevan, V.; Neese, A.S.; Koreeda, M.; Sharma, N.D.; Boyd, D.R. Synthesis and absolute configuration of the bacterial cis-1,2-, cis-8,9-, and cis-10,11-dihydro diol metabolites of benz[a]anthracene formed by strain of Beijerinckia. J. Org. Chem. 1984, 49, 3621–3628. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Agarwal, R.; Resnick, S.M.; Schocken, M.J.; Gibson, D.T.; Sayer, J.M.; Yagi, H.; Jerina, D.M. Bacterial dioxygenase-catalysed dihydroxylation and chemical resolution routes to enantiopure cis-dihydrodiols of chrysene. J. Chem. Soc. Perkin Trans. 1997, 1715–1724. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Hempenstall, F.; Kennedy, M.A.; Malone, J.F.; Allen, C.C.R.; Resnick, S.M.; Gibson, D.T. bis-cis-Dihydrodiols: A New Class of Metabolites Resulting from Biphenyl Dioxygenase-Catalyzed Sequential Asymmetriccis-Dihydroxylation of Polycyclic Arenes and Heteroarenes. J. Org. Chem. 1999, 64, 4005–4011. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Harrison, J.S.; Kennedy, M.A.; Allen, C.C.R.; Gibson, D.T. Regio- and stereo-selective dioxygenase-catalysed cis-dihydroxylation of fjord-region polycyclic arenes. J. Chem. Soc. Perkin Trans. 2001, 1264–1269. [Google Scholar] [CrossRef]
- Ferraro, D.J.; Brown, E.N.; Yu, C.L.; Parales, R.E.; Gibson, D.T.; Ramaswamy, S. Structural investigations of the ferredoxin and terminal oxygenase components of the biphenyl 2,3-dioxygenase from Sphingobium yanoikuyae B1. BMC Struct. Biol. 2007, 7, 10. [Google Scholar] [CrossRef]
- Ferraro, D.J.; Okerlund, A.; Brown, E.; Ramaswamy, S. One enzyme, many reactions: Structural basis for the various reactions catalyzed by naphthalene 1,2-dioxygenase. IUCrJ 2017, 4, 648–656. [Google Scholar] [CrossRef] [PubMed]
- Gai, Z.; Yu, B.; Li, L.; Wang, Y.; Ma, C.; Feng, J.; Deng, Z.; Xu, P. Cometabolic Degradation of Dibenzofuran and Dibenzothiophene by a Newly Isolated Carbazole-Degrading Sphingomonas sp. Strain. Appl. Environ. Microbiol. 2007, 73, 2832–2838. [Google Scholar] [CrossRef] [PubMed]
- Kodama, K.; Umehara, K.; Shimizu, K.; Nakatani, S.; Minoda, Y.; Yamada, K. Identification of Microbial Products from Dibenzothiophene and Its Proposed Oxidation Pathway. Agric. Biol. Chem. 1973, 37, 45–50. [Google Scholar] [CrossRef]
- Kodama, K.; Nakatani, S.; Umehara, K.; Shimizu, K.; Minoda, Y.; Yamada, K. Microbial Conversion of Petro-sulfur Compounds. Agric. Biol. Chem. 1970, 34, 1320–1324. [Google Scholar] [CrossRef]
- Resnick, S.M.; Gibson, D.T. Regio- and stereospecific oxidation of fluorene, dibenzofuran, and dibenzothiophene by naphthalene dioxygenase from Pseudomonas sp strain NCIB 9816–4. Appl. Environ. Microbiol. 1996, 62, 4073–4080. [Google Scholar] [CrossRef]
- Laborde, A.L.; Gibson, D.T. Metabolism of dibenzothiophene by a Beijerinckia species. Appl. Environ. Microbiol. 1977, 34, 783–790. [Google Scholar] [CrossRef]
- Khan, A.A.; Wang, R.F.; Cao, W.W.; Franklin, W.; Cerniglia, C.E. Reclassification of a polycyclic aromatic hydrocarbon-metabolizing bacterium, Beijerinckia sp strain B1, as Sphingomonas yanoikuyae by fatty acid analysis, protein pattern analysis, DNA-DNA hybridization, and 16S ribosomal DNA sequencing. Int. J. System. Bacteriol. 1996, 46, 466–469. [Google Scholar] [CrossRef]
- Bianchi, D.; Bosetti, A.; Cidaria, D.; Bernardi, A.; Gagliardi, I.; D’Amico, P. Oxidation of polycyclic aromatic heterocycles by Pseudomonas fluorescens TTC1. Appl. Microbiol. Biotechnol. 1997, 47, 596–599. [Google Scholar] [CrossRef]
- Saftic, S.; Fedorak, P.M.; Andersson, J.T. Transformations of methyldibenzothiophenes by three Pseudomonas isolates. Environ. Sci. Technol. 1993, 27, 2577–2584. [Google Scholar] [CrossRef]
- Gibson, D.T.; Resnick, S.M.; Lee, K.; Brand, J.M.; Torok, D.S.; Wackett, L.P.; Schocken, M.J.; Haigler, B.E. Desaturation, dioxygenation, and monooxygenation reactions catalyzed by naphthalene dioxygenase from Pseudomonas SP strain-9816–4. J. Bacteriol. 1995, 177, 2615–2621. [Google Scholar] [CrossRef]
- Resnick, S.; Lee, K.; Gibson, D.T. Diverse reactions catalyzed by naphthalene dioxygenase fromPseudomonas sp strain NCIB 9816. J. Ind. Microbiol. Biotechnol. 1996, 17, 438–457. [Google Scholar] [CrossRef]
- Eaton, S.L.; Resnick, S.M.; Gibson, D.T. Initial reactions in the oxidation of 1,2-dihydronaphthalene by Sphingomonas yanoikuyae strains. Appl. Environ. Microbiol. 1996, 62, 4388–4394. [Google Scholar] [CrossRef] [PubMed]
- Misra, B.; Amin, S. Synthesis and mutagenicity of trans-dihydrodiol metabolites of benzo[b]naphtho[2,1-d]thiophene. Chem. Res. Toxicol. 1990, 3, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Murphy, S.E.; Amin, S.; Coletta, K.; Hoffmann, D. Rat liver metabolism of benzo[b]naphtho[2,1-d]thiophene. Chem. Res. Toxicol. 1992, 5, 491–495. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D.; Sbircea, L.; Murphy, D.; Belhocine, T.; Malone, J.F.; James, S.L.; Allen, C.C.R.; Hamilton, J.T.G. Azaarene cis-dihydrodiol-derived 2,2′-bipyridine ligands for asymmetric allylic oxidation and cyclopropanation. Chem. Commun. 2008, 5535–5537. [Google Scholar] [CrossRef] [PubMed]
- Boyd, D.R.; Sharma, N.D.; Sbircea, L.; Murphy, D.; Malone, J.F.; James, S.L.; Allen, C.C.R.; Hamilton, J.T.G. Chemoenzymatic synthesis of chiral 2,2′-bipyridine ligands and their N-oxide derivatives: Applications in the asymmetric aminolysis of epoxides and asymmetric allylation of aldehydes. Org. Biomol. Chem. 2010, 8, 1081–1090. [Google Scholar] [CrossRef]
- Sbircea, L.; Sharma, N.D.; Clegg, W.; Harrington, R.W.; Horton, P.N.; Hursthouse, M.B.; Apperley, D.C.; Boyd, D.R.; James, S.L. Chemoenzymatic synthesis of chiral 4,4′-bipyridyls and their metal–organic frameworks. Chem. Commun. 2008, 5538–5540. [Google Scholar] [CrossRef]
- Nakayama, J.; Tajima, Y.; Xue-Hua, P.; Sugihara, Y. [1 + 2] Cycloadditions of Sulfur Monoxide (SO) to Alkenes and Alkynes and [1 + 4] Cycloadditions to Dienes (Polyenes). Generation and Reactions of Singlet SO? J. Am. Chem. Soc. 2007, 129, 7250–7251. [Google Scholar] [CrossRef]
- Dansette, P.; Jerina, D.M. Facile synthesis of arene oxides at the K regions of polycyclic hydrocarbons. J. Am. Chem. Soc. 1974, 96, 1224–1225. [Google Scholar] [CrossRef]
- Mitchell, J.R.; Jollows, D.J. Metabolic Activation of Drugs to Toxic Substances. Gastroenterology 1975, 68, 392–410. [Google Scholar] [CrossRef]
- McMurtry, R.; Mitchell, J. Renal and hepatic necrosis after metabolic activation of 2-substituted furans and thiophenes, including furosemide and cephaloridine. Toxicol. Appl. Pharmacol. 1977, 42, 285–300. [Google Scholar] [CrossRef]
- Dansette, P.M.; Thang, D.C.; ElAmri, H.; Mansuy, D. Evidence for thiophene-S-oxide as primary reactive metabolite of thiophene in vivo- formation of a dihydrothiophene sulfoxide mercapturic acid. Biochem. Biophys. Res. Commun. 1992, 186, 1624–1630. [Google Scholar] [CrossRef]
- Li, Y.; Thiemann, T.; Sawada, T.; Mataka, S.; Tashiro, M. Lewis Acid Catalysis in the Oxidative Cycloaddition of Thiophenes1. J. Org. Chem. 1997, 62, 7926–7936. [Google Scholar] [CrossRef]
- Sodhi, J.K.; Delarosa, E.M.; Halladay, J.S.; Driscoll, J.P.; Mulder, T.; Dansette, P.M.; Khojasteh, S.C. Inhibitory Effects of Trapping Agents of Sulfur Drug Reactive Intermediates against Major Human Cytochrome P450 Isoforms. Int. J. Mol. Sci. 2017, 18, 1553. [Google Scholar] [CrossRef] [PubMed]
- Dansette, P.M.; Thebault, S.; Durand-Gasselin, L.; Bertho, G.; Mansuy, D. Reactive metabolites of thiophenic compounds: A new trapping method for thiophene sulfoxides. Drug Metab. Rev. 2010, 42, 233–234. [Google Scholar]
- Du, F.; Ruan, Q.; Zhu, M.; Xing, J. Detection and characterization of ticlopidine conjugates in rat bile using high-resolution mass spectrometry: Applications of various data acquisition and processing tools. J. Mass Spectrom. 2013, 48, 413–422. [Google Scholar] [CrossRef]
- Jaladanki, C.K.; Taxak, N.; Varikoti, R.A.; Bharatam, P.V. Toxicity Originating from Thiophene Containing Drugs: Exploring the Mechanism using Quantum Chemical Methods. Chem. Res. Toxicol. 2015, 28, 2364–2376. [Google Scholar] [CrossRef] [PubMed]
- Imami, A.; Herold, N.; Spielmeyer, A.; Hausmann, H.; Dötzer, R.; Behnken, H.N.; Leonhardt, S.; Weil, A.; Schoof, S.; Zorn, H. Biotransformation of Dimethenamid-P by the basidiomycete Irpex consors. Chemosphere 2016, 165, 59–66. [Google Scholar] [CrossRef]
- Jerina, D.M.; Daly, J.W.; Witkop, B.; Zaltzman-Nirenberg, P.; Udenfriend, S. The role of arene oxide-oxepin systems in the metabolism of aromatic substrates. III. Formation of 1,2-naphthalene oxide from naphthalene by liver microsomes. J. Am. Chem. Soc. 1968, 90, 6525–6527. [Google Scholar] [CrossRef]
- Jerina, D.M.; Daly, J.W. Arene Oxides: A New Aspect of Drug Metabolism. Science 1974, 185, 573–582. [Google Scholar] [CrossRef] [PubMed]
- Mansuy, D.; Dansette, P.M.; Foures, C.; Jaouen, M.; Moinet, G.; Bayer, N. Metabolic hydroxylation of the thiophene ring: Isolation of 5-hydroxy-tienilic acid as the major urinary metabolite of tienilic acid in man and rat. Biochem. Pharmacol. 1984, 33, 1429–1435. [Google Scholar] [CrossRef]
- Dalvie, D.K.; Kalgutkar, A.S.; Khojasteh-Bakht, S.C.; Obach, R.S.; O’Donnell, J.P. Biotransformation Reactions of Five-Membered Aromatic Heterocyclic Rings. Chem. Res. Toxicol. 2002, 15, 269–299. [Google Scholar] [CrossRef]
- Belghazi, M.; Jean, P.; Poli, S.; Schmitter, J.M.; Mansuy, D.; Dansette, P.M. Use of isotopes and LC-MS-ESI-TOF for mechanistic studies of Tienilic Acid metabolic activation. In Biological Reactive Intermediates VI; Dansette, P.M., Snyder, R., Delaforge, M., Gibson, G.G., Greim, H., Jollow, D.J., Monks, T.J., Sipes, I.G., Eds.; Kluwer Academic/Plenum Publishers: New York, NY, USA; Dordrecht, The Netherlands; London, UK; Moscow, Russia, 2001; Volume 500, pp. 139–144. [Google Scholar] [CrossRef]
- Mori, Y.; Sakai, Y.; Kuroda, N.; Yokoya, F.; Toyoshi, K.; Horie, M.; Baba, S. Further structural analysis of urinary metabolites of suprofen in the rat. Drug Metab. Dispos. 1984, 12, 767–771. [Google Scholar]
- Fouda, H.G.; Avery, M.J.; Dalvie, D.; Falkner, F.C.; Melvin, L.S.; Ronfeld, R.A. Disposition and metabolism of tenidap in the rat. Drug Metab. Dispos. 1997, 25, 140–148. [Google Scholar]
- Neau, E.; Dansette, P.; Andronik, V.; Mansuy, D. Hydroxylation of the thiophene ring by hepatic monooxygenases: Evidence for 5-hydroxylation of 2-aroylthiophenes as a general metabolic pathway using a simple UV-visible assay. Biochem. Pharmacol. 1990, 39, 1101–1107. [Google Scholar] [CrossRef]
- Nishikawa, T.; Nagata, O.; Tanbo, K.; Yamada, T.; Takahara, Y.; Kato, H.; Yamamoto, Y. Absorption, excretion and metabolism of tiquizium bromide in dogs, and relationship between pharmacological effect and plasma levels of unchanged drug. Xenobiotica 1985, 15, 1053–1060. [Google Scholar] [CrossRef]
- Lynch, M.J.; Mosher, F.R.; Levesque, W.R.; Newby, T.J. The In Vitro and In Vivo Metabolism of Morantel in Cattle and Toxicology Species. Drug Metab. Rev. 1987, 18, 253–288. [Google Scholar] [CrossRef] [PubMed]
- Ichihara, S.; Tsuyuki, Y.; Tomisawa, H.; Fukazawa, H.; Nakayama, N.; Tateishi, M.; Joly, R. Metabolism of tenoxicam in rats. Xenobiotica 1984, 14, 727–739. [Google Scholar] [CrossRef]
- Boyd, D.R.; Sharma, N.D. The changing face of arene oxide–oxepine chemistry. Chem. Soc. Rev. 1996, 25, 289–296. [Google Scholar] [CrossRef]
- Barr, S.A.; Bowers, N.; Boyd, D.R.; Sharma, N.D.; Hamilton, L.; Austin, R.; McMordie, R.A.S.; Dalton, H. The potential role of cis-dihydrodiol intermediates in bacterial aromatic hydroxylation and the NIH Shift. J. Chem. Soc. Perkin Trans. 1998, 3443–3452. [Google Scholar] [CrossRef]
- Stok, J.E.; Chow, S.; Krenske, E.H.; Soto, C.F.; Matyas, C.; Poirier, R.A.; Williams, C.M.; De Voss, J.J. Direct Observation of an Oxepin from a Bacterial Cytochrome P450-Catalyzed Oxidation. Chem.—Eur. J. 2016, 22, 4408–4412. [Google Scholar] [CrossRef] [PubMed]
- Boyd, D.R.; Sharma, N.D.; Harrison, J.S.; Malone, J.F.; McRoberts, W.C.; Hamilton, J.T.G.; Harper, D.B. Enzyme-catalysed synthesis and reactions of benzene oxide/oxepine derivatives of methyl benzoates. Org. Biomol. Chem. 2008, 6, 1251–1259. [Google Scholar] [CrossRef]
- Taguchi, K.; Konishi, T.; Nishikawa, H.; Kitamura, S. Identification of human cytochrome P450 isoforms involved in the metabolism ofS-2-[4-(3-methyl-2-thienyl)phenyl]propionic acid. Xenobiotica 1999, 29, 899–907. [Google Scholar] [CrossRef]
- Dansette, P.M.; Kitamura, S.; Bertho, G.; Mansuy, D. A new thiophene metabolite: NADPH, O2 and thiol dependent formation of a thiophene hydrate from (S) MTPPA (methyl-thienyl-phenyl-propionic acid) by CYP2C9. Drug Metab. Rev. 2005, 37, 34. [Google Scholar]
- Boyd, D.R.; Sharma, N.D.; Agarwal, R.; McMordie, R.A.S.; Bessems, J.; van Ommen, B.; van Bladeren, P.J. Biotransformation of 1,2-dihydronaphthalene and 1,2-dihydroanthracene by rat liver microsomes and purified cytochromes P-450. Formation of arene hydrates of naphthalene and anthracene. Chem. Res. Toxicol. 1993, 6, 808–812. [Google Scholar] [CrossRef]
- Lopez-Garcia, M.P.; Dansette, P.M.; Mansuy, D. Thiophene derivatives as new mechanism-based inhibitors of cytochromes P-450: Inactivation of yeast-expressed human liver cytochrome P-450 2C9 by tienilic acid. Biochemistry 1994, 33, 166–175. [Google Scholar] [CrossRef]
- Ha-Duong, N.-T.; Dijols, S.; Macherey, A.-C.; Goldstein, J.A.; Dansette, A.P.M.; Mansuy, D. Ticlopidine as a Selective Mechanism-Based Inhibitor of Human Cytochrome P450 2C19. Biochemistry 2001, 40, 12112–12122. [Google Scholar] [CrossRef] [PubMed]
- Richter, T.; Mürdter, T.E.; Heinkele, G.; Pleiss, J.; Tatzel, S.; Schwab, M.; Eichelbaum, M.; Zanger, U.M. Potent Mechanism-Based Inhibition of Human CYP2B6 by Clopidogrel and Ticlopidine. J. Pharmacol. Exp. Ther. 2003, 308, 189–197. [Google Scholar] [CrossRef]
- Hutzler, J.M.; Balogh, L.M.; Zientek, M.; Kumar, V.; Tracy, T.S. Mechanism-Based Inactivation of Cytochrome P450 2C9 by Tienilic Acid and (±)-Suprofen: A Comparison of Kinetics and Probe Substrate Selection. Drug Metab. Dispos. 2008, 37, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.-L.; Zhang, H.; Medower, C.; Hollenberg, P.F.; Johnson, W.W. Inactivation of Cytochrome P450 (P450) 3A4 but not P450 3A5 by OSI-930, a Thiophene-Containing Anticancer Drug. Drug Metab. Dispos. 2010, 39, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Kalgutkar, A.S.; Driscoll, J.; Zhao, S.X.; Walker, G.S.; Shepard, R.M.; Soglia, J.R.; Atherton, J.; Yu, L.; Mutlib, A.E.; Munchhof, M.J.; et al. A Rational Chemical Intervention Strategy To Circumvent Bioactivation Liabilities Associated with a Nonpeptidyl Thrombopoietin Receptor Agonist Containing a 2-Amino-4-arylthiazole Motif. Chem. Res. Toxicol. 2007, 20, 1954–1965. [Google Scholar] [CrossRef] [PubMed]
- Barnette, D.; Schleiff, M.; Osborn, L.R.; Flynn, N.; Matlock, M.; Swamidass, S.J.; Miller, G.P. Dual mechanisms suppress meloxicam bioactivation relative to sudoxicam. Toxicology 2020, 440, 152478. [Google Scholar] [CrossRef]
- Obach, R.S.; Kalgutkar, A.S.; Ryder, T.F.; Walker, G.S. In Vitro Metabolism and Covalent Binding of Enol-Carboxamide Derivatives and Anti-Inflammatory Agents Sudoxicam and Meloxicam: Insights into the Hepatotoxicity of Sudoxicam. Chem. Res. Toxicol. 2008, 21, 1890–1899. [Google Scholar] [CrossRef] [PubMed]
- Nikodinovic-Runic, J.; Coulombel, L.; Francuski, D.; Sharma, N.D.; Boyd, D.R.; More O’Ferrall, R.A.; O’Connor, K.E. The oxidation of alkylaryl sulfides and benzo[b]thiophenes by Escherichia coli cells expressing wild-type and engineered styrene monooxygenase from Pseudomonas putida CA-3. Appl. Microbiol. Biotechnol. 2013, 97, 4849–4858. [Google Scholar] [CrossRef]
- Gursky, L.J.; Nikodinović-Runić, J.; Feenstra, K.A.; O’Connor, K.E. In vitro evolution of styrene monooxygenase from Pseudomonas putida CA-3 for improved epoxide synthesis. Appl. Microbiol. Biotechnol. 2010, 85, 995–1004. [Google Scholar] [CrossRef]
- O’Connor, K.E.; Dobson, A.D.; Hartmans, S. Indigo formation by microorganisms expressing styrene monooxygenase activity. Appl. Environ. Microbiol. 1997, 63, 4287–4291. [Google Scholar] [CrossRef]
- Chen, B.; Zhang, X.-D.; Wen, J.; Zhang, B.; Chen, D.; Wang, S.; Cai, J.-P.; Hu, G.-X. Effects of 26 Recombinant CYP3A4 Variants on Brexpiprazole Metabolism. Chem. Res. Toxicol. 2020, 33, 172–180. [Google Scholar] [CrossRef]
- Joshi, E.M.; Heasley, B.H.; Chordia, M.; Macdonald, T.L. In Vitro Metabolism of 2-Acetylbenzothiophene: Relevance to Zileuton Hepatotoxicity. Chem. Res. Toxicol. 2004, 17, 137–143. [Google Scholar] [CrossRef]
- Parker, M.H.; Smith-Swintosky, V.L.; McComsey, D.F.; Huang, Y.; Brenneman, D.; Klein, B.; Malatynska, E.; White, H.S.; Milewski, M.E.; Herb, M.; et al. Novel, Broad-Spectrum Anticonvulsants Containing a Sulfamide Group: Advancement of N-((Benzo[b]thien-3-yl)methyl)sulfamide (JNJ-26990990) into Human Clinical Studies. J. Med. Chem. 2009, 52, 7528–7536. [Google Scholar] [CrossRef]
- Jacob, J.; Schmoldt, A.; Grimmer, G. The predominant role of S-oxidation in rat liver metabolism of thiaarenes. Cancer Lett. 1986, 32, 107–116. [Google Scholar] [CrossRef]
- Hollopeter, G.; Jantzen, H.-M.; Vincent, D.; Li, G.; England, L.; Ramakrishnan, V.; Yang, R.-B.; Nurden, P.; Nurden, A.; Julius, D.; et al. Identification of the platelet ADP receptor targeted by antithrombotic drugs. Nature 2001, 409, 202–207. [Google Scholar] [CrossRef]
- Maffrand, J.-P. The story of clopidogrel and its predecessor, ticlopidine: Could these major antiplatelet and antithrombotic drugs be discovered and developed today? Comptes Rendus Chim. 2012, 15, 737–743. [Google Scholar] [CrossRef]
- Kiebist, J.; Schmidtke, K.-U.; Schramm, M.; König, R.; Quint, S.; Kohlmann, J.; Zuhse, R.; Ullrich, R.; Hofrichter, M.; Scheibner, K. Biocatalytic Syntheses of Antiplatelet Metabolites of the Thienopyridines Clopidogrel and Prasugrel Using Fungal Peroxygenases. J. Fungi 2021, 7, 752. [Google Scholar] [CrossRef]
- Dansette, P.M.; Libraire, J.; Bertho, G.; Mansuy, D. Metabolic Oxidative Cleavage of Thioesters: Evidence for the Formation of Sulfenic Acid Intermediates in the Bioactivation of the Antithrombotic Prodrugs Ticlopidine and Clopidogrel. Chem. Res. Toxicol. 2009, 22, 369–373. [Google Scholar] [CrossRef]
- Dansette, P.M.; Thébault, S.; Bertho, G.; Mansuy, D. Formation and Fate of a Sulfenic Acid Intermediate in the Metabolic Activation of the Antithrombotic Prodrug Prasugrel. Chem. Res. Toxicol. 2010, 23, 1268–1274. [Google Scholar] [CrossRef] [PubMed]
- Dansette, P.M.; Rosi, J.; Bertho, G.; Mansuy, D. Paraoxonase-1 and clopidogrel efficacy. Nat. Med. 2011, 17, 1040–1041. [Google Scholar] [CrossRef] [PubMed]
- Dansette, P.M.; Rosi, J.; Bertho, G.; Mansuy, D. Cytochromes P450 Catalyze Both Steps of the Major Pathway of Clopidogrel Bioactivation, whereas Paraoxonase Catalyzes the Formation of a Minor Thiol Metabolite Isomer. Chem. Res. Toxicol. 2012, 25, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Dansette, P.M.; Levent, D.; Hessani, A.; Bertho, G.; Mansuy, D. Thiolactone Sulfoxides as New Reactive Metabolites Acting as Bis-Electrophiles: Implication in Clopidogrel and Prasugrel Bioactivation. Chem. Res. Toxicol. 2013, 26, 794–802. [Google Scholar] [CrossRef]
- Zhang, H.; Lau, W.C.; Hollenberg, P.F. Formation of the thiol conjugates and active metabolite of clopidogrel by human liver microsomes. Mol. Pharmacol. 2012, 82, 302–309. [Google Scholar] [CrossRef]
- Tuong, A.; Bouyssou, A.; Paret, J.; Cuong, T.G. Metabolism of triclopidine in rats: Identification and quantitative determination of some its metabolites in plasma, urine and bile. Eur. J. Drug Metab. Pharmacokinet. 1981, 6, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Farid, N.A.; Smith, R.L.; Gillespie, T.A.; Rash, T.J.; Blair, P.E.; Kurihara, A.; Goldberg, M.J. The Disposition of Prasugrel, a Novel Thienopyridine, in Humans. Drug Metab. Dispos. 2007, 35, 1096–1104. [Google Scholar] [CrossRef]
- Liu, C.; Chen, Z.; Zhong, K.; Li, L.; Zhu, W.; Chen, X.; Zhong, D. Human Liver Cytochrome P450 Enzymes and Microsomal Thiol Methyltransferase Are Involved in the Stereoselective Formation and Methylation of the Pharmacologically Active Metabolite of Clopidogrel. Drug Metab. Dispos. 2015, 43, 1632–1641. [Google Scholar] [CrossRef] [PubMed]
- Kazui, M.; Hagihara, K.; Izumi, T.; Ikeda, T.; Kurihara, A. Hepatic Microsomal Thiol Methyltransferase Is Involved in Stereoselective Methylation of Pharmacologically Active Metabolite of Prasugrel. Drug Metab. Dispos. 2014, 42, 1138–1145. [Google Scholar] [CrossRef]
- Camilleri, P.; Soldo, B.; Buch, A.; Janusz, J. Oxidative metabolism of razuprotafib (AKB-9778), a sulfamic acid phosphatase inhibitor, in human microsomes and recombinant human CYP2C8 enzyme. Xenobiotica 2021, 51, 1110–1121. [Google Scholar] [CrossRef]
- Soldo, B.L.; Camilleri, P.; Buch, A.; Janusz, J. The in vivo disposition of subcutaneous injected 14C-razuprotafib (14C-AKB-9778), a sulphamic acid phosphatase inhibitor, in nonclinical species and human. Xenobiotica 2021, 51, 1132–1145. [Google Scholar] [CrossRef] [PubMed]
- Guengerich, F.P. A history of the roles of cytochrome P450 enzymes in the toxicity of drugs. Toxicol. Res. 2020, 37, 1–23. [Google Scholar] [CrossRef]
- Shimizu, S.; Atsumi, R.; Nakazawa, T.; Fujimaki, Y.; Sudo, K.; Okazaki, O. Metabolism of Ticlopidine in Rats: Identification of the Main Biliary Metabolite as a Glutathione Conjugate of Ticlopidine S-Oxide. Drug Metab. Dispos. 2009, 37, 1904–1915. [Google Scholar] [CrossRef]
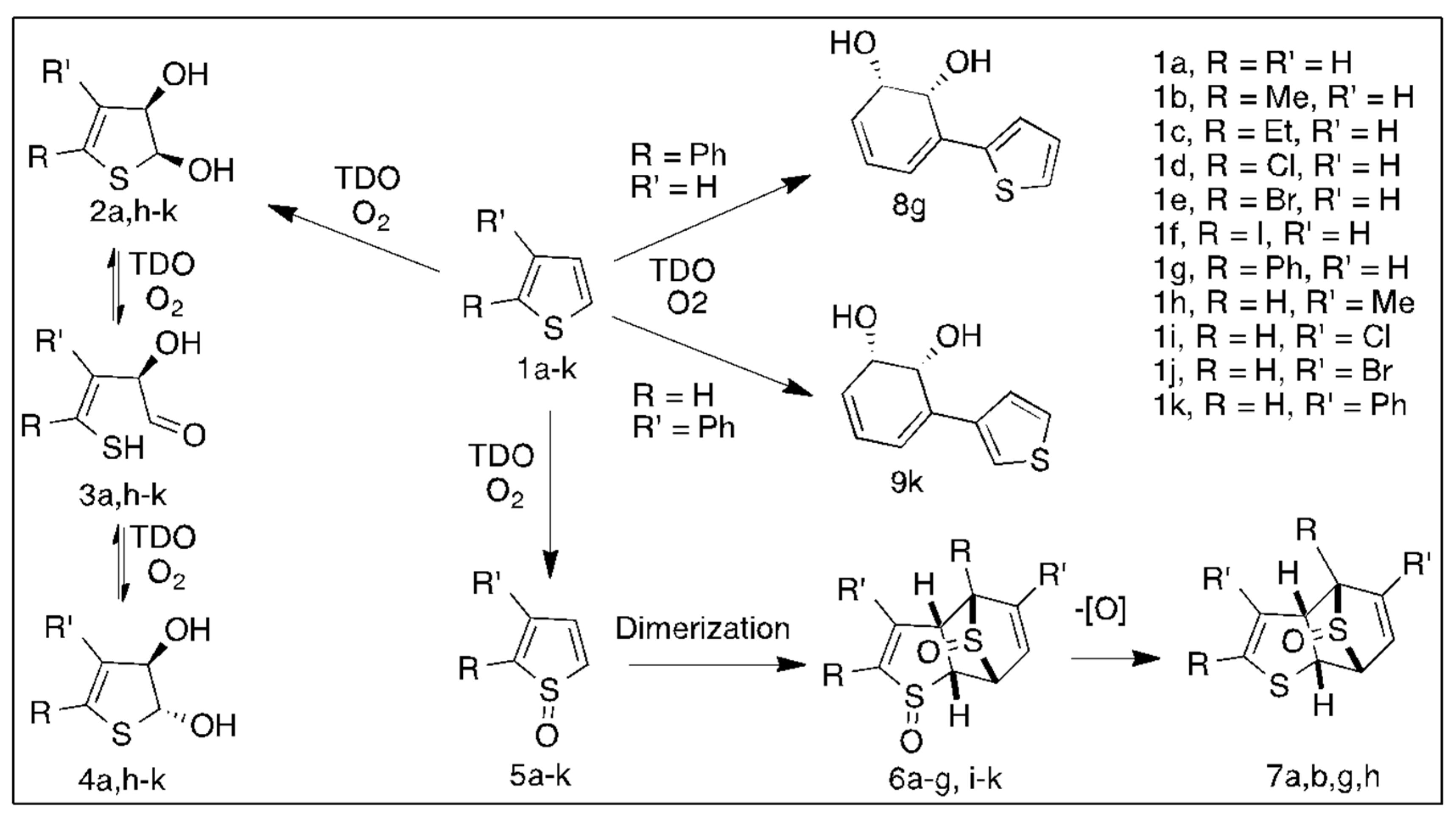

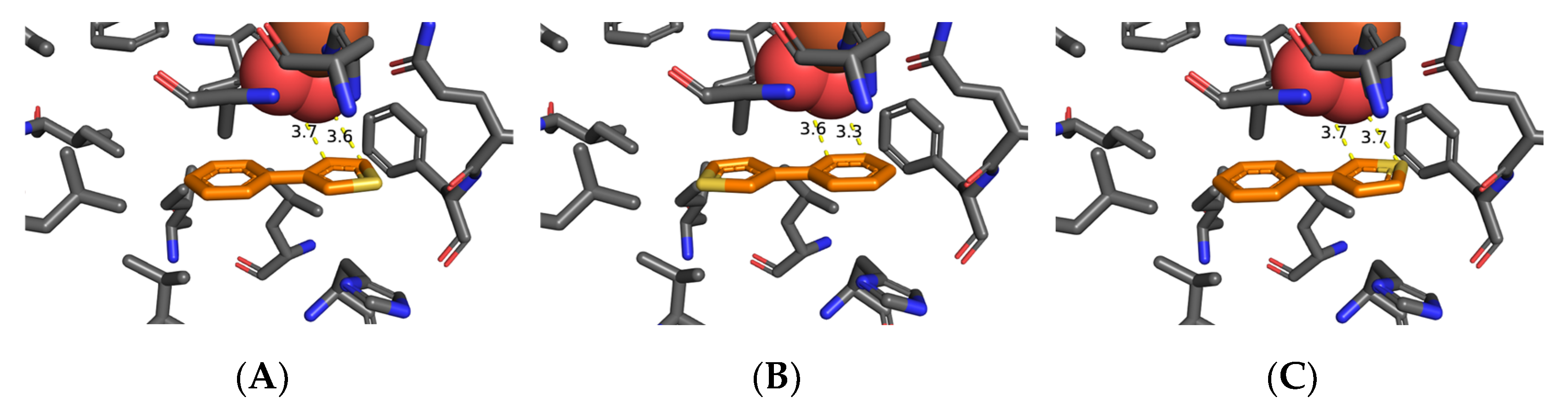




| Substrates 1a–g; Strain | 5a–g (%) | 6a–g (%) | 7a–g (%) | 2a–g/4a–g (%) | 8a–g (%) |
|---|---|---|---|---|---|
| 1a, R = R′ = H (TDO) a | 0 | 79 | 21 | <1 (0.2) | 0 |
| 1b, R = Me, R′ = H (TDO) a | 0 | 25 | 75 | 0 | 0 |
| 1c, R = Et, R′ = H (TDO) a | 0 | 100 | 0 | 0 | 0 |
| 1d, R = Cl, R′ = H (TDO) a | 0 | 100 | 0 | 0 | 0 |
| 1e, R = Br, R′ = H (TDO) a | 0 | 100 | 0 | 0 | 0 |
| 1f, R = I, R′ = H (TDO) a | 0 | 100 | 0 | 0 | 0 |
| 1g, R = Ph, R′ = H (TDO) a | 0 | 67 | 6 | 0 | 27 |
| 1g, R = Ph, R′= H (TDO) b | 0 | 40 | 0 | 0 | 60 |
| 1g, R = Ph, R′ = H (NDO) c | 0 | 0 | 0 | 0 | 100 |
| 1g, R= Ph, R′ = H (NDO) d | 0 | 0 | 0 | 100 | 0 |
| Substrates 1h–k; Strain | 5h–k (%) | 6h–k(%) | 7h–k (%) | 2h–k/4h–k (%) | 9k(%) |
|---|---|---|---|---|---|
| 1h, R = H, R′ = Me (TDO) a | 0 | 0 | 24 | 76 | 0 |
| 1i, R = H, R′ = Cl (TDO) a | 0 | 59 | 0 | 41 | 0 |
| 1j, R = H, R′ = Br (TDO) a | 0 | 46 | 0 | 54 | 0 |
| 1k, R = H, R′ = Ph (TDO) a | 0 | 35 | 0 | 48 | 17 |
| 1k, R = H, R′ = Ph (NDO) b | 0 | 2 | 0 | 0 | 98 |
| 1k, R = H, R′ = Ph (NDO) c | 0 | 80 | 0 | 20 | 0 |
| Substrates 10a–d (Dioxygenase) | 11a–c (%) | 17a–d (%) | 23a–c (%) |
|---|---|---|---|
| 10a, R = R′ = R″ = H (TDO) a | 10 e | 63 | 12 i |
| 10a, R = R′ = R″ = H (NDO) b | 100 | ||
| 10a, R = R′ = R″ = H (NDO) c | 0 | 85 | 15 |
| 10a, R = R′ = R″ = H (NDO) d | 0 | 86 | 14 |
| 10b, R = Me, R′ = R″ = H (TDO) a | 83 | 10 f | 7 |
| 10b, R = Me, R′ = R″ = H (NDO) b | 0 | 0 g | 90 |
| 10b, R = Me, R′ = R″ = H (NDO) c | 90 | 0 | 10 |
| 10b, R = Me, R′ = R″ = H (NDO) d | 0 | 60 | 42 |
| 10c, R′ = Me, R = R″ = H (TDO) a | 100 | 0 | 0 |
| 10c, R′ = Me, R = R″ = H (NDO) b | 0 | 0 h | 42 |
| 10c, R′ = Me, R = R″ = H (NDO) c | 0 | 10 f | 90 |
| 10c, R′ = Me, R = R″ = H (NDO) d | 0 | 0 | 100 |
| 10d, R″ = Me, R = R′ = H (TDO) a | 0 | 100 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boyd, D.R.; Sharma, N.D.; Stevenson, P.J.; Hoering, P.; Allen, C.C.R.; Dansette, P.M. Monooxygenase- and Dioxygenase-Catalyzed Oxidative Dearomatization of Thiophenes by Sulfoxidation, cis-Dihydroxylation and Epoxidation. Int. J. Mol. Sci. 2022, 23, 909. https://doi.org/10.3390/ijms23020909
Boyd DR, Sharma ND, Stevenson PJ, Hoering P, Allen CCR, Dansette PM. Monooxygenase- and Dioxygenase-Catalyzed Oxidative Dearomatization of Thiophenes by Sulfoxidation, cis-Dihydroxylation and Epoxidation. International Journal of Molecular Sciences. 2022; 23(2):909. https://doi.org/10.3390/ijms23020909
Chicago/Turabian StyleBoyd, Derek R., Narain D. Sharma, Paul J. Stevenson, Patrick Hoering, Christopher C. R. Allen, and Patrick M. Dansette. 2022. "Monooxygenase- and Dioxygenase-Catalyzed Oxidative Dearomatization of Thiophenes by Sulfoxidation, cis-Dihydroxylation and Epoxidation" International Journal of Molecular Sciences 23, no. 2: 909. https://doi.org/10.3390/ijms23020909
APA StyleBoyd, D. R., Sharma, N. D., Stevenson, P. J., Hoering, P., Allen, C. C. R., & Dansette, P. M. (2022). Monooxygenase- and Dioxygenase-Catalyzed Oxidative Dearomatization of Thiophenes by Sulfoxidation, cis-Dihydroxylation and Epoxidation. International Journal of Molecular Sciences, 23(2), 909. https://doi.org/10.3390/ijms23020909







