Methionine γ-Lyase-Daidzein in Combination with S-Propyl-L-cysteine Sulfoxide as a Targeted Prodrug Enzyme System for Malignant Solid Tumor Xenografts
Abstract
1. Introduction
2. Results
2.1. Steady-State Kinetic Parameters of the β-Elimination Reaction Catalyzed by the Enzyme and Characterization of the Conjugated C115H MGL-Dz
2.2. C115H MGL-Dz Cytotoxicity on Different Cancer Cells Viability in the Presence of Propiin In Vitro
2.3. Cytotoxicity Effect of Dipropyl Thiosulfinate on Various Cancer Cells Viability
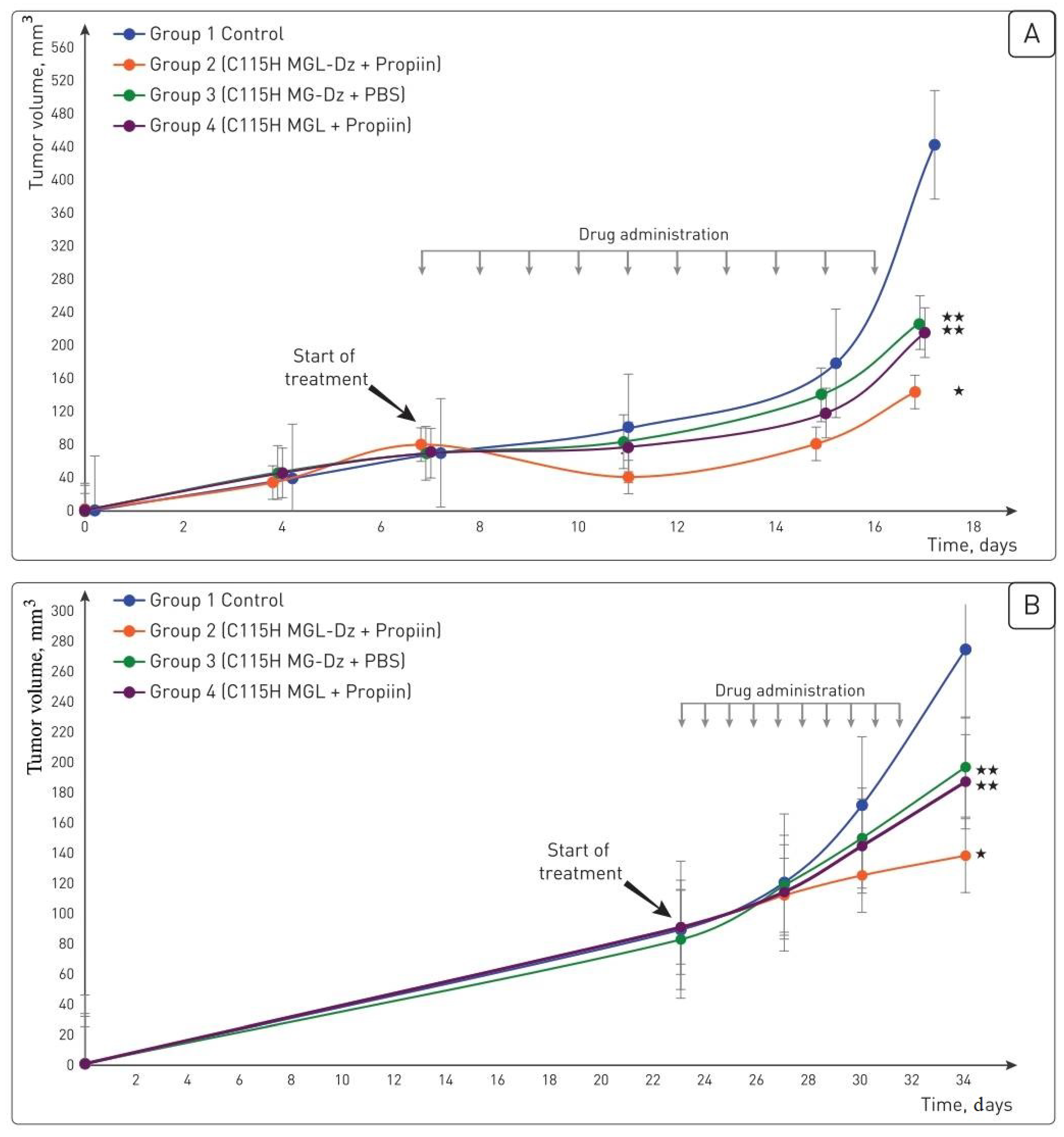
2.4. SW620 and HT29 Xenografts Showed Different Responses to the Pharmacological Pair In Situ
2.5. The Pharmacological Pair Inhibits Growth of Pancreatic Adenocarcinoma Panc1 and MIA-PaCa2 Xenografts
2.6. The Pharmacological Pair Enhances the Suppression of Prostate Cancer 22Rv1 Xenograft Growth
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Preparation of C115H MGL and Enzymes Assays
4.3. Steady-State Kinetics
4.4. Preparation of C115H MGL/Propiin Mixture and Quantitative Determination of Dipropyl Thiosulfinate
4.5. Synthesis of C115H MGL-Dz
4.6. Cell Lines and Cytotoxicity Evaluation
4.6.1. The Cytotoxicity of the C115H MGL-Dz in the Presence of Propiin
4.6.2. The Cytotoxicity of Dipropyl Thiosulfinate Generated by the Pharmacological Pair C115H MGL/Propiin
4.7. Animals and Evaluation of Antitumor Activity
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Padma, V.V. An overview of targeted cancer therapy. BioMedicine 2015, 5, 1–6. [Google Scholar] [CrossRef]
- Pokrovsky, V.S.; Chepikova, O.E.; Davydov, D.Z.; Zamyatnin, A.A., Jr.; Lukashev, A.N.; Lukasheva, E.V. Amino Acid Degrading Enzymes and their Application in Cancer Therapy. Curr. Med. Chem. 2017, 26, 446–464. [Google Scholar] [CrossRef]
- Xu, G.; Mcleod, H.L. Strategies for Enzyme / Prodrug Cancer Therapy. Clin. Cancer Res. 2001, 7, 3314–3324. [Google Scholar]
- Torchilin, V.P. Drug Targeting. Eur. J. Pharm. Sci. 2000, 11, 581–591. [Google Scholar] [CrossRef]
- Springer, C.J.; Niculescu-Duvaz, I. Prodrug-activating systems in suicide gene therapy. J. Clin. Investig. 2000, 105, 1161–1167. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, S.; Gustafsson, J.-Å. Estrogen receptors: Therapies targeted to receptor subtypes. Clin. Pharmacol. Ther. 2011, 89, 44–55. [Google Scholar] [CrossRef]
- di Gioia, F.; Petropoulos, S.A. Phytoestrogens, phytosteroids and saponins in vegetables: Biosynthesis, functions, health effects and practical applications. Adv. Food Nutr. Res. 2019, 90, 351–421. [Google Scholar] [CrossRef] [PubMed]
- Molina, L.; Bustamante, F.A.; Bhoola, K.D.; Figueroa, C.D.; Ehrenfeld, P. Possible role of phytoestrogens in breast cancer via GPER-1/GPR30 signaling. Clin. Sci. 2018, 132, 2583–2598. [Google Scholar] [CrossRef]
- Somjen, D.; Katzburg, S.; Nevo, N.; Gayer, B.; Hodge, R.P.; Renevey, M.D.; Kalchenko, V.; Meshorer, A.; Stern, N.; Kohen, F. A daidzein–daunomycin conjugate improves the therapeutic response in an animal model of ovarian carcinoma. J. Steroid Biochem. Mol. Biol. 2008, 110, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Appel, E.; Rabinkov, A.; Neeman, M.; Kohen, F.; Mirelman, D. Conjugates of daidzein-alliinase as a targeted pro-drug enzyme system against ovarian carcinoma. J. Drug Target. 2011, 19, 326–335. [Google Scholar] [CrossRef] [PubMed]
- Morozova, E.; Anufrieva, N.; Koval, V.; Lesnova, E.; Kushch, A.; Timofeeva, V.; Solovieva, A.; Kulikova, V.; Revtovich, S.; Demidkina, T. Conjugates of methionine γ-lyase with polysialic acid: Two approaches to antitumor therapy. Int. J. Biol. Macromol. 2021, 182, 394–401. [Google Scholar] [CrossRef]
- Morozova, E.A.; Kulikova, V.V.; Rodionov, A.N.; Revtovich, S.V.; Anufrieva, N.V.; Demidkina, T.V. Engineered Citrobacter freundii methionine γ-lyase effectively produces antimicrobial thiosulfinates. Biochimie 2016, 128–129, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Walther, R.; Rautio, J.; Zelikin, A.N. Prodrugs in medicinal chemistry and enzyme prodrug therapies. Adv. Drug Deliv. Rev. 2017, 118, 65–77. [Google Scholar] [CrossRef]
- Pokrovsky, V.S.; Anisimova, N.Y.; Davydov, D.Z.; Bazhenov, S.V.; Bulushova, N.V.; Zavilgelsky, G.B.; Kotova, V.Y.; Manukhov, I.V. Methionine gamma lyase from clostridium sporogenes increases the anticancer efficacy of doxorubicin on A549 cancer cells in vitro and human cancer xenografts. Methods Mol. Biol. 2019, 1866, 243–261. [Google Scholar] [CrossRef]
- Hoffman, R.M. Development of recombinant methioninase to target the general cancer-specific metabolic defect of methionine dependence: A 40-year odyssey. Expert Opin. Biol. Ther. 2015, 15, 21–31. [Google Scholar] [CrossRef]
- Filardo, E.J.; Quinn, J.A.; Frackelton, A.R.J.; Bland, K.I. Estrogen action via the G protein-coupled receptor, GPR30: Stimulation of adenylyl cyclase and cAMP-mediated attenuation of the epidermal growth factor receptor-to-MAPK signaling axis. Mol. Endocrinol. 2002, 16, 70–84. [Google Scholar] [CrossRef] [PubMed]
- Ariyani, W.; Miyazaki, W.; Amano, I.; Hanamura, K.; Shirao, T.; Koibuchi, N. Soy Isoflavones Accelerate Glial Cell Migration via GPER-Mediated Signal Transduction Pathway. Front. Endocrinol. 2020, 11, 854. [Google Scholar] [CrossRef] [PubMed]
- Gruhlke, M.C.H.; Nicco, C.; Batteux, F.; Slusarenko, A.J. The effects of allicin, a reactive sulfur species from garlic, on a selection of mammalian cell lines. Antioxidants 2017, 6, 1. [Google Scholar] [CrossRef]
- Sarvizadeh, M.; Hasanpour, O.; Ghale-noie, Z.N. Allicin and Digestive System Cancers: From Chemical Structure to Its Therapeutic Opportunities. Front. Oncol. 2021, 11, 1–18. [Google Scholar] [CrossRef]
- Bat-chen, W.; Golan, T.; Peri, I.; Ludmer, Z.; Schwartz, B. Allicin Purified From Fresh Garlic Cloves Induces Apoptosis in Colon Cancer Cells Via Nrf2 Allicin Purified From Fresh Garlic Cloves Induces Apoptosis in Colon Cancer Cells Via Nrf2. Nutr. Cancer 2010, 62, 947–957. [Google Scholar] [CrossRef] [PubMed]
- Roseblade, A.; Ung, A.; Bebawy, M. Synthesis and in vitro biological evaluation of thiosulfinate derivatives for the treatment of human multidrug-resistant breast cancer. Acta Pharmacol. Sin. 2017, 38, 1353–1368. [Google Scholar] [CrossRef] [PubMed]
- Morozova, E.; Abo Qoura, L.; Anufrieva, N.; Koval, V.; Lesnova, E.; Kushch, A.; Kulikova, V.; Revtovich, S.; Pokrovsky, V.; Demidkina, T. Daidzein-directed methionine γ-lyase in enzyme prodrug therapy against breast cancer. Biochimie 2022, 201, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Jacenik, D.; Beswick, E.J.; Krajewska, W.M.; Prossnitz, E.R. G protein-coupled estrogen receptor in colon function, immune regulation and carcinogenesis. World J. Gastroenterol. 2019, 25, 4092–4104. [Google Scholar] [CrossRef] [PubMed]
- Mesmar, F.; Dai, B.; Ibrahim, A.; Hases, L.; Jafferali, M.H.; Augustine, J.J.; DiLorenzo, S.; Kang, Y.; Zhao, Y.; Wang, J.; et al. Clinical candidate and genistein analogue AXP107-11 has chemoenhancing functions in pancreatic adenocarcinoma through G protein-coupled estrogen receptor signaling. Cancer Med. 2019, 8, 7705–7719. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.M.; Xiao, B.X.; Dai, D.J.; Liu, Q.; Ma, H.H. Effects of daidzein on estrogen-receptor-positive and negative pancreatic cancer cells in vitro. World J. Gastroenterol. 2004, 10, 860–863. [Google Scholar] [CrossRef]
- Chan, Q.K.Y.; Lam, H.-M.; Ng, C.-F.; Lee, A.Y.Y.; Chan, E.S.; Ng, H.-K.; Ho, S.-M.; Lau, K.-M. Activation of GPR30 inhibits the growth of prostate cancer cells through sustained activation of Erk1/2, c-jun/c-fos-dependent upregulation of p21, and induction of G2 cell-cycle arrest. Cell Death Differ. 2010, 17, 1511–1523. [Google Scholar] [CrossRef]
- Kulikova, V.; Morozova, E.; Rodionov, A.; Koval, V.; Anufrieva, N.; Revtovich, S.; Demidkina, T. Non-stereoselective decomposition of (±)-S-alk(en)yl-L-cysteine sulfoxides to antibacterial thiosulfinates catalyzed by C115H mutant methionine γ-lyase from Citrobacter freundii. Biochimie 2018, 151, 42–44. [Google Scholar] [CrossRef]
- Miron, T.; Rabinkov, A.; Mirelman, D.; Weiner, L.; Wilchek, M. A spectrophotometric assay for allicin and alliinase (Alliin lyase) activity: Reaction of 2-nitro-5-thiobenzoate with thiosulfinates. Anal. Biochem. 1998, 265, 317–325. [Google Scholar] [CrossRef]
- Kulikova, V.V.; Morozova, E.A.; Revtovich, S.V.; Kotlov, M.I.; Anufrieva, N.V.; Bazhulina, N.P.; Raboni, S.; Faggiano, S.; Gabellieri, E.; Cioni, P.; et al. Gene cloning, characterization, and cytotoxic activity of methionine γ-lyase from Clostridium novyi. IUBMB Life 2017, 69, 668–676. [Google Scholar] [CrossRef]
- Schwimmer, M.M.S. Characterization of Alliinase of Allium cepa (Onion). Arch. Biochem. Biophys. 1963, 100, 66–73. [Google Scholar] [CrossRef]
- Kohen, F.; Gayer, B.; Kulik, T.; Frydman, V.; Nevo, N.; Katzburg, S.; Limor, R.; Sharon, O.; Stern, N.; Somjen, D. Synthesis and evaluation of the antiproliferative activities of derivatives of carboxyalkyl isoflavones linked to N-t-Boc-hexylenediamine. J. Med. Chem. 2007, 50, 6405–6410. [Google Scholar] [CrossRef] [PubMed]
- Erlanger, B.F.; Borek, F.; Beiser, S.M.; Lieberman, S. Steroid-Protein Conjugates. J. Biol. Chem. 1957, 228, 713–727. [Google Scholar] [CrossRef]
- Bank, U.; Reinhold, D.; Ansorge, S. Measurement of cellular activity with the MTT test. Optimization of the method. Allerg. Immunol. 1991, 37, 119–123. [Google Scholar]
- Tomayko, M.M.; Reynolds, C.P. Determination of subcutaneous tumor size in athymic (nude) mice. Cancer Chemother. Pharmacol. 1989, 24, 148–154. [Google Scholar] [CrossRef] [PubMed]



| Substrate | C115H MGL * | C115H MGL-Dz | ||||
|---|---|---|---|---|---|---|
| kcat, s−1 | Km, mM | kcat/Km, (M−1 s−1) | kcat, s−1 | Km, mM | kcat/Km, (M−1 s−1) | |
| S-Met-L-Cys | 29.6 | 7.8 | 3.8 × 103 | 20.8 ± 2.0 | 7.1 ± 0.52 | 2.9 × 103 |
| Alliin | 26.8 | 1.0 | 2.6 × 104 | 17.9 ± 1.4 | 0.6 ± 0.07 | 2.5 × 104 |
| Methiin | 6.1 | 1.1 | 5.5 × 103 | 1.6 ± 0.2 | 0.3 ± 0.05 | 4.6 × 103 |
| Propiin | 28 | 1.6 | 1.7 × 104 | 16.9 ± 1.8 | 1.0 ± 0.14 | 1.6 × 104 |
| Tissue Sources | Cell Lines | C115H MGL-Dz |
|---|---|---|
| IC50 (µM) | ||
| Colon cancer | HT29 | 6.9 ± 0.4 |
| COLO205 | ND | |
| SW620 * | ND | |
| Pancreatic cancer | Panc1 | 3.8 ± 0.1 |
| MIA-PaCa-2 | 6.4 ± 0.5 | |
| Prostate adenocarcinoma | 22Rv1 | 5.4 ± 0.2 |
| DU-145 | 17.4 ± 0.9 | |
| PC3 | 16.9 ± 0.5 |
| Tissue Sources | Cell Lines | Dipropyl Thiosulfinate |
|---|---|---|
| IC50 (µM) | ||
| Colon cancer | HT-29 | 43.1 ± 3.4 |
| COLO205 | 48.6 ± 4.3 | |
| HCT116 | 43.4 ± 4.3 | |
| SW620 * | 19.2 ± 0.9 | |
| Pancreatic cancer | Panc1 | 18.6 ± 0.5 |
| MIA-PaCa-2 | 62.6 ± 1.8 | |
| Prostate adenocarcinoma | 22Rv1 | 66.3 ± 2.6 |
| DU-145 | 60.9 ± 0.6 | |
| PC3 | 51.2 ± 3.0 |
| HT-29 | SW 620 | |||||||
|---|---|---|---|---|---|---|---|---|
| Specimen | Initial Tumor Volume, mm3 | Final Tumor Volume, mm3 | TGI, % | Body Weight, g | Initial Tumor Volume, mm3 | Final Tumor Volume, mm3 | TGI, % | Body Weight, g |
| Control | 104 ± 44 | 571 ± 114 | - | 22.2 ± 1 | 82 ± 13 | 219 ± 27 | - | 25.8 ± 0.4 |
| C115H MGL-Dz + propiin | 102 ± 20 | 275 ± 48 | 52 (p = 0.04) | 21.4 ± 1 | 82 ± 12 | 170.9 ± 9 | 22 (p = 0.12) | 22.1 ± 0.4 |
| C115H MGL-Dz + PBS | 98 ± 26 | 361 ± 69 | 37 (p = 0.1) | 22.7 ± 1 | 85 ± 11 | 185 ± 12 | 15 (p = 0.258) | 24.8 ± 1.9 |
| C115H MGL + propiin | 100 ± 22 | 320 ± 69 | 44 (p = 0.09) | 21.1 ± 1 | 83 ± 9.3 | 167 ± 36 | 24 (p = 0.155) | 24.6 ± 1.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abo Qoura, L.; Morozova, E.; Kulikova, V.; Karshieva, S.; Sokolova, D.; Koval, V.; Revtovich, S.; Demidkina, T.; Pokrovsky, V.S. Methionine γ-Lyase-Daidzein in Combination with S-Propyl-L-cysteine Sulfoxide as a Targeted Prodrug Enzyme System for Malignant Solid Tumor Xenografts. Int. J. Mol. Sci. 2022, 23, 12048. https://doi.org/10.3390/ijms231912048
Abo Qoura L, Morozova E, Kulikova V, Karshieva S, Sokolova D, Koval V, Revtovich S, Demidkina T, Pokrovsky VS. Methionine γ-Lyase-Daidzein in Combination with S-Propyl-L-cysteine Sulfoxide as a Targeted Prodrug Enzyme System for Malignant Solid Tumor Xenografts. International Journal of Molecular Sciences. 2022; 23(19):12048. https://doi.org/10.3390/ijms231912048
Chicago/Turabian StyleAbo Qoura, Louay, Elena Morozova, Vitalia Kulikova, Saida Karshieva, Darina Sokolova, Vasiliy Koval, Svetlana Revtovich, Tatyana Demidkina, and Vadim S. Pokrovsky. 2022. "Methionine γ-Lyase-Daidzein in Combination with S-Propyl-L-cysteine Sulfoxide as a Targeted Prodrug Enzyme System for Malignant Solid Tumor Xenografts" International Journal of Molecular Sciences 23, no. 19: 12048. https://doi.org/10.3390/ijms231912048
APA StyleAbo Qoura, L., Morozova, E., Kulikova, V., Karshieva, S., Sokolova, D., Koval, V., Revtovich, S., Demidkina, T., & Pokrovsky, V. S. (2022). Methionine γ-Lyase-Daidzein in Combination with S-Propyl-L-cysteine Sulfoxide as a Targeted Prodrug Enzyme System for Malignant Solid Tumor Xenografts. International Journal of Molecular Sciences, 23(19), 12048. https://doi.org/10.3390/ijms231912048







