Iron Oxide Powder as Responsible for the Generation of Industrial Polypropylene Waste and as a Co-Catalyst for the Pyrolysis of Non-Additive Resins
Abstract
1. Introduction
2. Results and Discussion
2.1. Elemental Analysis of PP Samples
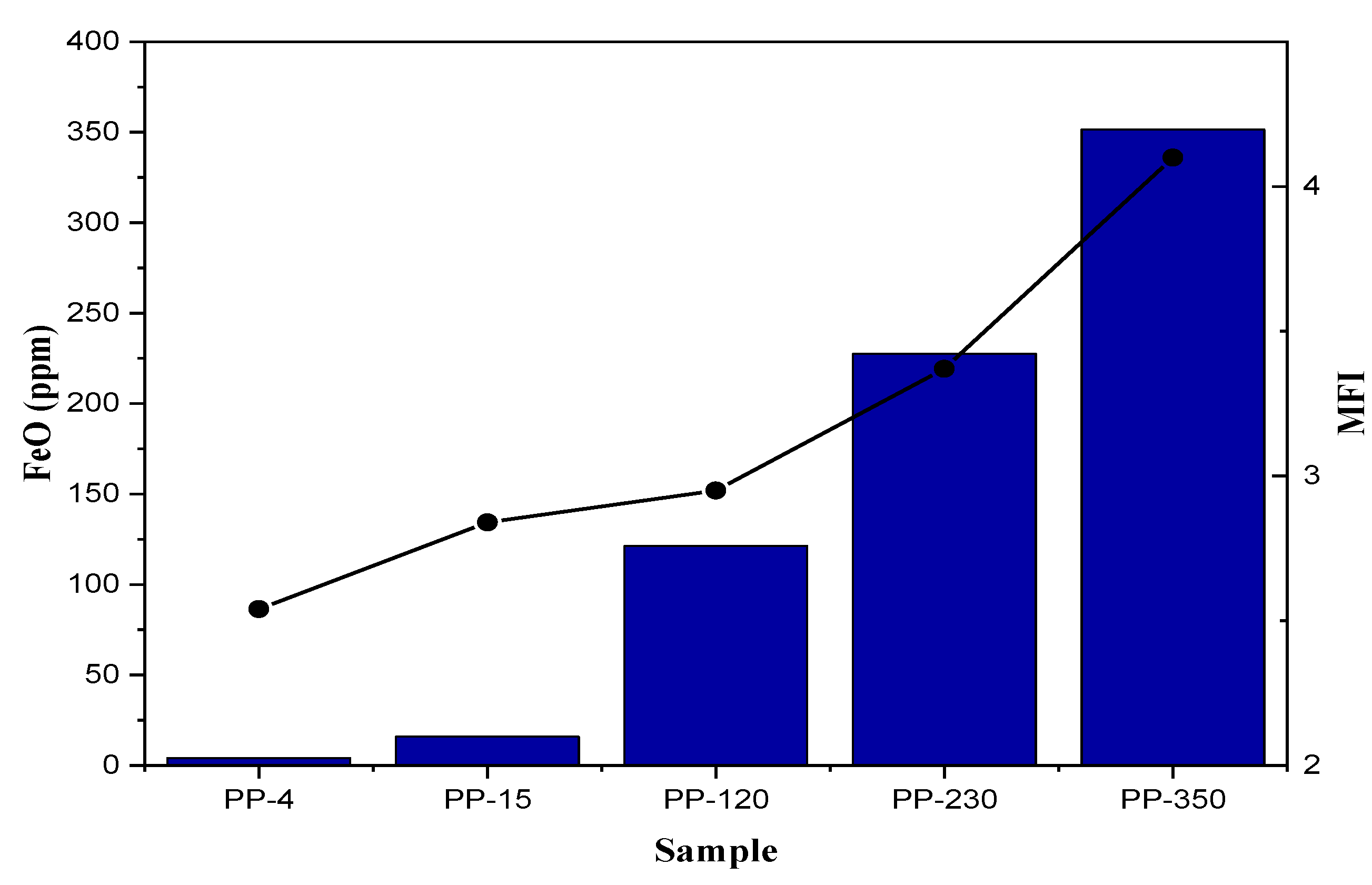
2.2. MFI
2.3. TGA
2.4. Characterization of Pyrolysis Gaseous and Liquid Products
3. Materials and Methods
3.1. Standards
3.2. Preparation of FeO and Ziegler Natta Catalyst Mixtures
3.3. Site of Sampling and Collection of Samples
3.4. XRF
3.5. MFI
3.6. TGA
3.7. Pyrolysis
3.8. GC-MS/PDHID/FIDA Analysis of Pyrolysis Gases and Condensates
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Experimental Study of Catalytic Pyrolysis of Polyethylene and Polypropylene over USY Zeolite and Separation to Gasoline and Diesel-like Fuels—ScienceDirect. Available online: https://www.sciencedirect.com/science/article/abs/pii/S0165237017301183 (accessed on 22 August 2022).
- Marcilla, A.; Beltrán, M.I.; Navarro, R. Thermal and Catalytic Pyrolysis of Polyethylene over HZSM5 and HUSY Zeolites in a Batch Reactor under Dynamic Conditions. Appl. Catal. B Environ. 2009, 86, 78–86. [Google Scholar] [CrossRef]
- Hernández-Fernandez, J.; Rodríguez, E. Determination of Phenolic Antioxidants Additives in Industrial Wastewater from Polypropylene Production Using Solid Phase Extraction with High-Performance Liquid Chromatography. J. Chromatogr. A 2019, 1607, 460442. [Google Scholar] [CrossRef] [PubMed]
- Martínez, M.C.; Benavente, R.; Gómez-Elvira, J.M. Molecular Weight Dependence and Stereoselective Chain Cleavage during the Early Stages of the Isotactic Polypropylene Pyrolysis. Polym. Degrad. Stab. 2017, 143, 26–34. [Google Scholar] [CrossRef]
- Onu, P.; Vasile, C.; Ciocílteu, S.; Iojoiu, E.; Darie, H. Thermal and Catalytic Decomposition of Polyethylene and Polypropylene. J. Anal. Appl. Pyrolysis 1999, 49, 145–153. [Google Scholar] [CrossRef]
- Sakata, Y.; Azhar Uddin, M.; Muto, A.; Kanada, Y.; Koizumi, K.; Murata, K. Catalytic Degradation of Polyethylene into Fuel Oil over Mesoporous Silica (KFS-16) Catalyst. J. Anal. Appl. Pyrolysis 1997, 43, 15–25. [Google Scholar] [CrossRef]
- Sakata, Y.; Uddin, M.A.; Muto, A. Degradation of Polyethylene and Polypropylene into Fuel Oil by Using Solid Acid and Non-Acid Catalysts. J. Anal. Appl. Pyrolysis 1999, 51, 135–155. [Google Scholar] [CrossRef]
- Abbas-Abadi, M.S.; Haghighi, M.N.; Yeganeh, H.; McDonald, A.G. Evaluation of Pyrolysis Process Parameters on Polypropylene Degradation Products. J. Anal. Appl. Pyrolysis 2014, 109, 272–277. [Google Scholar] [CrossRef]
- Bockhorn, H.; Hornung, A.; Hornung, U.; Schawaller, D. Kinetic Study on the Thermal Degradation of Polypropylene and Polyethylene. J. Anal. Appl. Pyrolysis 1999, 48, 93–109. [Google Scholar] [CrossRef]
- Albizzati, E.; Galimberti, M. Catalysts for Olefins Polymerization. Catal. Today 1998, 41, 159–168. [Google Scholar] [CrossRef]
- Celina, M.; George, G.A.; Billingham, N.C. Physical Spreading of Oxidation in Solid Polypropylene as Studied by Chemiluminescence. Polym. Degrad. Stab. 1993, 42, 335–344. [Google Scholar] [CrossRef]
- Thermo Gravimetric Kinetics of Polypropylene Degradation on ZSM-12 and ZSM-5 Catalysts—ScienceDirect. Available online: https://www.sciencedirect.com/science/article/abs/pii/S0920586105003743 (accessed on 22 August 2022).
- Goss, B.G.S.; Nakatani, H.; George, G.A.; Terano, M. Catalyst Residue Effects on the Heterogeneous Oxidation of Polypropylene. Polym. Degrad. Stab. 2003, 82, 119–126. [Google Scholar] [CrossRef]
- Analysis of the Nonvolatile Oxidation Products of Polypropylene I. Thermal Oxidation—Adams—1970—Journal of Polymer Science Part A-1: Polymer Chemistry—Wiley Online Library. Available online: https://onlinelibrary.wiley.com/doi/abs/10.1002/pol.1970.150080505 (accessed on 22 August 2022).
- Aging and Degradation of Polyolefins. II. Γ-Initiated Oxidations of Atactic Polypropylene—Decker—1973—Journal of Polymer Science: Polymer Chemistry Edition—Wiley Online Library. Available online: https://onlinelibrary.wiley.com/doi/abs/10.1002/pol.1973.170111109 (accessed on 22 August 2022).
- Photo-Oxidation of Polymers—Part V: A New Chain Scission Mechanism in Polyolefins—ScienceDirect. Available online: https://www.sciencedirect.com/science/article/pii/0141391082900076 (accessed on 22 August 2022).
- Wu, X.; Wu, Y.; Wu, K.; Chen, Y.; Hu, H.; Yang, M. Study on Pyrolytic Kinetics and Behavior: The Co-Pyrolysis of Microalgae and Polypropylene. Bioresour. Technol. 2015, 192, 522–528. [Google Scholar] [CrossRef] [PubMed]
- Fowell, P. The Heat of Formation of Some Metal Alkyls and of Some Phosphine Imines. Ph.D. Thesis, University of Manchester, Manchester, UK, 1961. [Google Scholar]
- Reaction Hazards of Triethylaluminum under Closed Conditions—ScienceDirect. Available online: https://www.sciencedirect.com/science/article/abs/pii/S0950423011000787 (accessed on 22 August 2022).
- Kunwar, B.; Cheng, H.N.; Chandrashekaran, S.R.; Sharma, B.K. Plastics to Fuel: A Review. Renew. Sustain. Energy Rev. 2016, 54, 421–428. [Google Scholar] [CrossRef]
- Joaquin, H.-F.; Juan, L.-M. Autocatalytic Influence of Different Levels of Arsine on the Thermal Stability and Pyrolysis of Polypropylene. J. Anal. Appl. Pyrolysis 2022, 161, 105385. [Google Scholar] [CrossRef]
- Hernández-Fernández, J.; Rayón, E.; López, J.; Arrieta, M.P. Enhancing the Thermal Stability of Polypropylene by Blending with Low Amounts of Natural Antioxidants. Macromol. Mater. Eng. 2019, 304, 1900379. [Google Scholar] [CrossRef]
- Pavon, C.; Aldas, M.; López-Martínez, J.; Hernández-Fernández, J.; Arrieta, M.P. Films Based on Thermoplastic Starch Blended with Pine Resin Derivatives for Food Packaging. Foods 2021, 10, 1171. [Google Scholar] [CrossRef]
- Gómez-Contreras, P.; Figueroa-Lopez, K.J.; Hernández-Fernández, J.; Cortés Rodríguez, M.; Ortega-Toro, R. Effect of Different Essential Oils on the Properties of Edible Coatings Based on Yam (Dioscorea rotundata L.) Starch and Its Application in Strawberry (Fragaria vesca L.) Preservation. Appl. Sci. 2021, 11, 11057. [Google Scholar] [CrossRef]
- Hernández-Fernández, J.; Guerra, Y.; Puello-Polo, E.; Marquez, E. Effects of Different Concentrations of Arsine on the Synthesis and Final Properties of Polypropylene. Polymers 2022, 14, 3123. [Google Scholar] [CrossRef]
- FBR Sustainable Chemistry & Technology; Alvarado Chacon, F.; Brouwer, M.T.; Thoden van Velzen, E.U.; Smeding, I.W. A First Assessment of the Impact Ofimpurities in PP and PE Recycled Plastics; Wageningen Food & Biobased Research: Wageningen, The Netherlands, 2020. [Google Scholar]
- Aurrekoetxea, J.; Sarrionandia, M.A.; Urrutibeascoa, I.; Maspoch, M.L. Effects of Recycling on the Microstructure and the Mechanical Properties of Isotactic Polypropylene. J. Mater. Sci. 2001, 36, 2607–2613. [Google Scholar] [CrossRef]
- Bahlouli, N.; Pessey, D.; Raveyre, C.; Guillet, J.; Ahzi, S.; Dahoun, A.; Hiver, J.M. Recycling Effects on the Rheological and Thermomechanical Properties of Polypropylene-Based Composites. Mater. Des. 2012, 33, 451–458. [Google Scholar] [CrossRef]
- Salakhov, I.I.; Bukatov, G.D.; Batyrshin, A.Z.; Matsko, M.A.; Barabanov, A.A.; Tavtorkin, A.N.; Temnikova, E.V.; Sakhabutdinov, A.G. Synthesis of Polypropylene in the Liquid Monomer in the Presence of a Titanium—Magnesium Catalyst: Effect of Various Internal Donors. Russ. J. Appl. Chem. 2019, 92, 796–808. [Google Scholar] [CrossRef]
- Forte, M.C.; Coutinho, F.M.B. The Influence of Catalyst System and Polymerization Conditions on Polypropylene Properties. Eur. Polym. J. 1996, 32, 605–611. [Google Scholar] [CrossRef]
- Arjmand, S.; Shakeri, A.; Arabi, H. Effect of Water and Carbonyl Sulfide Toxins in Gas Propylene Feed in Polymerization Process on Physical Properties of Polypropylene. J. Polym. Res. 2019, 26, 195. [Google Scholar] [CrossRef]
- Bremner, T.; Rudin, A.; Cook, D.G. Melt Flow Index Values and Molecular Weight Distributions of Commercial Thermoplastics. J. Appl. Polym. Sci. 1990, 41, 1617–1627. [Google Scholar] [CrossRef]
- Bremner, T.; Cook, D.G.; Rudin, A. Further Comments on the Relations between Melt Flow Index Values and Molecular Weight Distributions of Commercial Plastics. J. Appl. Polym. Sci. 1991, 43, 1773. [Google Scholar] [CrossRef]
- He, Q.; Yuan, T.; Zhu, J.; Luo, Z.; Haldolaarachchige, N.; Sun, L.; Khasanov, A.; Li, Y.; Young, D.P.; Wei, S.; et al. Magnetic High Density Polyethylene Nanocomposites Reinforced with In-Situ Synthesized Fe@FeO Core-Shell Nanoparticles. Polymer 2012, 53, 3642–3652. [Google Scholar] [CrossRef]
- Chancharoenrith, S.; Kamonsatikul, C.; Namkajorn, M.; Kiatisevi, S.; Somsook, E. Iron Oxide/Cassava Starch-Supported Ziegler–Natta Catalysts for in Situ Ethylene Polymerization. Carbohydr. Polym. 2015, 117, 319–323. [Google Scholar] [CrossRef]
- Bahri-Laleh, N. Interaction of Different Poisons with MgCl2/TiCl4 Based Ziegler-Natta Catalysts. Appl. Surf. Sci. 2016, 379, 395–401. [Google Scholar] [CrossRef]
- Detailed Kinetic Modeling of the Formation of Toxic Polycyclic Aromatic Hydrocarbons (PAHs) Coming from Pyrolysis in Low-Pressure Gas Carburizing Conditions—ScienceDirect. Available online: https://www.sciencedirect.com/science/article/abs/pii/S0165237016301577 (accessed on 22 August 2022).
- Feng, J.; Hao, J.; Du, J.; Yang, R. Using TGA/FTIR TGA/MS and Cone Calorimetry to Understand Thermal Degradation and Flame Retardancy Mechanism of Polycarbonate Filled with Solid Bisphenol A Bis(Diphenyl Phosphate) and Montmorillonite. Polym. Degrad. Stab. 2012, 97, 605–614. [Google Scholar] [CrossRef]
- Park, Y.-K.; Jung, J.; Ryu, S.; Lee, H.W.; Siddiqui, M.Z.; Jae, J.; Watanabe, A.; Kim, Y.-M. Catalytic Co-Pyrolysis of Yellow Poplar Wood and Polyethylene Terephthalate over Two Stage Calcium Oxide-ZSM-5. Appl. Energy 2019, 250, 1706–1718. [Google Scholar] [CrossRef]
- Kumagai, S.; Yamasaki, R.; Kameda, T.; Saito, Y.; Watanabe, A.; Watanabe, C.; Teramae, N.; Yoshioka, T. Catalytic Pyrolysis of Poly(Ethylene Terephthalate) in the Presence of Metal Oxides for Aromatic Hydrocarbon Recovery Using Tandem μ-Reactor-GC/MS. Energy Fuels 2020, 34, 2492–2500. [Google Scholar] [CrossRef]
- Lin, X.; Zhang, Z.; Zhang, Z.; Sun, J.; Wang, Q.; Pittman, C.U. Catalytic Fast Pyrolysis of a Wood-Plastic Composite with Metal Oxides as Catalysts. Waste Manag. 2018, 79, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Lei, J.; Yuan, G.; Weerachanchai, P.; Wang, J.-Y.; Zhao, J.; Yang, Y. Fe-, Ti-, Zr- and Al-Pillared Clays for Efficient Catalytic Pyrolysis of Mixed Plastics. Chem. Eng. J. 2017, 317, 800–809. [Google Scholar] [CrossRef]
- Wang, J.; Jiang, J.; Meng, X.; Li, M.; Wang, X.; Pang, S.; Wang, K.; Sun, Y.; Zhong, Z.; Ruan, R.; et al. Promoting Aromatic Hydrocarbon Formation via Catalytic Pyrolysis of Polycarbonate Wastes over Fe- and Ce-Loaded Aluminum Oxide Catalysts. Environ. Sci. Technol. 2020, 54, 8390–8400. [Google Scholar] [CrossRef] [PubMed]
- Joaquin, H.-F.; Juan, L. Quantification of Poisons for Ziegler Natta Catalysts and Effects on the Production of Polypropylene by Gas Chromatographic with Simultaneous Detection: Pulsed Discharge Helium Ionization, Mass Spectrometry and Flame Ionization. J. Chromatogr. A 2020, 1614, 460736. [Google Scholar] [CrossRef] [PubMed]

| Feed | PP-4 | PP-15 | PP-120 | PP-230 | PP-350 |
|---|---|---|---|---|---|
| Proximate analysis (as received) | |||||
| Moisture, wt.% | 0.21 | 0.37 | 0.41 | 0.34 | 0.42 |
| Volatile matter, wt.% | 99.58 | 99.43 | 99.42 | 99.39 | 99.45 |
| Fixed carbon, wt.% | 0.11 | 0.13 | 0.13 | 0.18 | 0.09 |
| Ash, wt.%) | 0.05 | 0.06 | 0.04 | 0.09 | 0.04 |
| Ultimate analysis (as received, wt.%) | |||||
| C, wt.% | 83.79 | 83.79 | 84.15 | 84.37 | 83.84 |
| H, wt.% | 14.75 | 14.82 | 14.18 | 14.05 | 14.58 |
| N, wt.% | 0.07 | 0.05 | 0.09 | 0.07 | 0.05 |
| S, wt.% | 0.32 | 0.39 | 0.41 | 0.47 | 0.41 |
| O, wt.% | 1.06 | 0.95 | 1.18 | 1.04 | 1.11 |
| Ti, mg Kg−1 | 0.87 | 0.87 | 0.87 | 0.87 | 0.87 |
| Al, mg Kg−1 | 9.16 | 10.11 | 8.53 | 9.27 | 8.45 |
| Cl, mg Kg−1 | 13.43 | 13.82 | 13.91 | 14.17 | 13.95 |
| FeO, mg Kg−1 | 4.13 | 15 | 121 | 227 | 351 |
| Compounds | SAMPLES | ||||
|---|---|---|---|---|---|
| PP-4 | PP-15 | PP-120 | PP-230 | PP-350 | |
| ALKANS | |||||
| Methane, % mol | 5.84 ± 0.0529 | 4.13 ± 0.1155 | 4.57 ± 0.0577 | 3.38 ± 0.0764 | 1.37 ± 0.0577 |
| Ethane, % mol | 12.33 ± 0.1155 | 11.80 ± 0.1000 | 9.38 ± 0.0764 | 4.67 ± 0.0577 | 1.44 ± 0.0693 |
| Propane, % mol | 3.10 ± 0.10 | 3.38 ± 0.0681 | 4.27 ± 0.1155 | 2.85 ± 0.050 | 1.17 ± 0.1155 |
| Cyclopropane, % mol | 0.05 ± 0.0058 | 0.57 ± 0.0577 | 0.24 ± 0.0058 | 0.21 ± 0.0173 | 0.07 ± 0.0058 |
| Isobutane, % mol | 0.48 ± 0.0764 | 0.70 ± 0.0058 | 0.22 ± 0.0289 | 0.32 ± 0.0289 | 0.12 ± 0.0289 |
| N-Butane, % mol | 1.40 ± 0.10 | 1.00 ± 0.1732 | 1.40 ± 0.0 | 2.18 ± 0.1041 | 1.33 ± 0.0577 |
| Isopentane, % mol | 12.03 ± 0.0577 | 8.79 ± 0.0503 | 6.52 ± 0.0289 | 11.81 ± 0.0173 | 5.37 ± 0.0577 |
| Total Amount | 35.24 ± 0.2873 | 30.37 ± 0.1102 | 26.59 ± 0.0551 | 25.42 ± 0.1825 | 10.86 ± 0.1137 |
| ALKENES | |||||
| Ethylene, % mol | 1.71 ± 0.0231 | 0.89 ± 0.0173 | 1.47 ± 0.1155 | 1.50 ± 0.0058 | 1.47 ± 0.1155 |
| Propylene, % mol | 57.15 ± 0.05 | 54.10 ± 0.10 | 49.60 ± 0.1732 | 37.81 ± 0.0173 | 25.76 ± 0.0231 |
| Propadyene, % mol | 0.82 ± 0.0153 | 0.11 ± 0.0115 | 0.41 ± 0.0115 | 0.71 ± 0.0115 | 1.13 ± 0.1155 |
| Trans-2-Butene, % mol | 0.21 ± 0.0173 | 0.22 ± 0.0289 | 0.40 ± 0.0 | 0.51 ± 0.0173 | 1.07 ± 0.0577 |
| 1-Butene, % mol | 0.91 ± 0.0173 | 0.23 ± 0.0306 | 0.61 ± 0.0231 | 0.71 ± 0.0115 | 2.09 ± 0.0808 |
| Cis-2-Butene, % mol | 0.31 ± 0.0173 | 0.44 ± 0.0551 | 0.71 ± 0.0115 | 0.30 ± 0.0058 | 0.82 ± 0.0289 |
| 1,3-Butadiene, % mol | 0.70 ± 0.0058 | 9.12 ± 0.1258 | 5.38 ± 0.0289 | 0.10 ± 0.0012 | 1.32 ± 0.0346 |
| 1-Pentene, % mol | 0.91 ± 0.0173 | 0.81 ± 0.0115 | 0.49 ± 0.0115 | 1.80 ± 0.0058 | 0.90 ± 0.0058 |
| Total Amount | 62.64 ± 0.0635 | 65.85 ± 0.1848 | 59.00 ± 0.1674 | 43.41 ± 0.0115 | 34.58 ± 0.0520 |
| ALKYNES | |||||
| Acetylene, % mol | 0.21 ± 0.0115 | 0.23 ± 0.1155 | 0.80 ± 0.0058 | 0.50 ± 0.0 | 1.41 ± 0.0115 |
| Methyl acetylene, % mol | 0.42 ± 0.0153 | 0.71 ± 0.0173 | 0.52 ± 0.0289 | 0.31 ± 0.0231 | 1.60 ± 0.0 |
| Total Amount | 0.61 ± 0.0115 | 0.94 ± 0.0981 | 1.30 ± 0.0058 | 0.81 ± 0.0231 | 3.01 ± 0.0115 |
| ALCOHOL | |||||
| Methanol, % mol | 0.01 ± 0.0 | 0.05 ± 0.0058 | 3.71 ± 0.0231 | 5.43 ± 0.0577 | 8.40 ± 0.0 |
| Ethanol, % mol | 0.11 ± 0.0115 | 0.21 ± 0.0058 | 0.86 ± 0.0200 | 1.61 ± 0.0115 | 3.73 ± 0.0577 |
| Isopropyl Alcohol, % mol | 0.04 ± 0.0100 | 0.01 ± 0.0 | 1.03 ± 0.0577 | 2.43 ± 0.0577 | 4.20 ± 0.0 |
| N-Propanol, % mol | 0.03 ± 0.0 | 0.09 ± 0.0058 | 0.83 ± 0.0173 | 3.07 ± 0.0577 | 5.83 ± 0.0577 |
| N-Butyl Alcohol, % mol | 0 | 0.05 ± 0.0058 | 1.43 ± 0.0577 | 2.65 ± 0.1286 | 3.31 ± 0.0173 |
| 1,2- Isobutenediol, % mol | 0 | 0.02 ± 0.0 | 0.98 ± 0.0115 | 1.80 ± 0.0 | 3.87 ± 0.0608 |
| 3-Methyl-2-Pentanol, % mol | 0 | 0 | 0.22 ± 0.0289 | 1.62 ± 0.0252 | 3.17 ± 0.0577 |
| Total Amount | 0.18 ± 0.0058 | 0.44 ± 0.0173 | 9.08 ± 0.0058 | 18.61 ± 0.0115 | 32.53 ± 0.0577 |
| KETONE | |||||
| Acetone, % mol | 0.08 ± 0.0 | 0.05 ± 0.0058 | 0.20 ± 0.0 | 0.80 ± 0.0058 | 1.83 ± 0.0577 |
| 1-Hydroxy-2-Propanone, % mol | 0.01 ± 0.0058 | 0.04 ± 0.0 | 0.30 ± 0.0 | 1.20 ± 0.0 | 2.87 ± 0.0643 |
| 2,4-Pentadione, % mol | 0.02 ± 0.0058 | 0.05 ± 0.0058 | 0.23 ± 0.0577 | 1.60 ± 0.0 | 3.08 ± 0.0764 |
| 2-Pentanone, % mol | 0.13 ± 0.0153 | 0.11 ± 0.0058 | 0.70 ± 0.0000 | 2.43 ± 0.0577 | 2.80 ± 0.0058 |
| Total Amount | 0.25 ± 0.0115 | 0.24 ± 0.0115 | 1.43 ± 0.0577 | 6.03 ± 0.0577 | 10.57 ± 0.0577 |
| ACIDS | |||||
| Formic Acid, % mol | 0.20 ± 0.0 | 0.72 ± 0.0058 | 1.20 ± 0.0 | 2.43 ± 0.0577 | 4.57 ± 0.0577 |
| Acetic Acid, % mol | 0.10 ± 0.0 | 0.05 ± 0.0 | 1.27 ± 0.1155 | 3.40 ± 0.0 | 3.83 ± 0.0577 |
| Total Amount | 0.30 ± 0.0 | 0.77 ± 0.0058 | 2.47 ± 0.1155 | 5.83 ± 0.0577 | 8.40 ± 0.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández-Fernández, J.; Castro-Suarez, J.R.; Toloza, C.A.T. Iron Oxide Powder as Responsible for the Generation of Industrial Polypropylene Waste and as a Co-Catalyst for the Pyrolysis of Non-Additive Resins. Int. J. Mol. Sci. 2022, 23, 11708. https://doi.org/10.3390/ijms231911708
Hernández-Fernández J, Castro-Suarez JR, Toloza CAT. Iron Oxide Powder as Responsible for the Generation of Industrial Polypropylene Waste and as a Co-Catalyst for the Pyrolysis of Non-Additive Resins. International Journal of Molecular Sciences. 2022; 23(19):11708. https://doi.org/10.3390/ijms231911708
Chicago/Turabian StyleHernández-Fernández, Joaquín, John R. Castro-Suarez, and Carlos A. T. Toloza. 2022. "Iron Oxide Powder as Responsible for the Generation of Industrial Polypropylene Waste and as a Co-Catalyst for the Pyrolysis of Non-Additive Resins" International Journal of Molecular Sciences 23, no. 19: 11708. https://doi.org/10.3390/ijms231911708
APA StyleHernández-Fernández, J., Castro-Suarez, J. R., & Toloza, C. A. T. (2022). Iron Oxide Powder as Responsible for the Generation of Industrial Polypropylene Waste and as a Co-Catalyst for the Pyrolysis of Non-Additive Resins. International Journal of Molecular Sciences, 23(19), 11708. https://doi.org/10.3390/ijms231911708







