C3H Expression Is Crucial for Methyl Jasmonate Induction of Chicoric Acid Production by Echinacea purpurea (L.) Moench Cell Suspension Cultures
Abstract
1. Introduction
2. Results
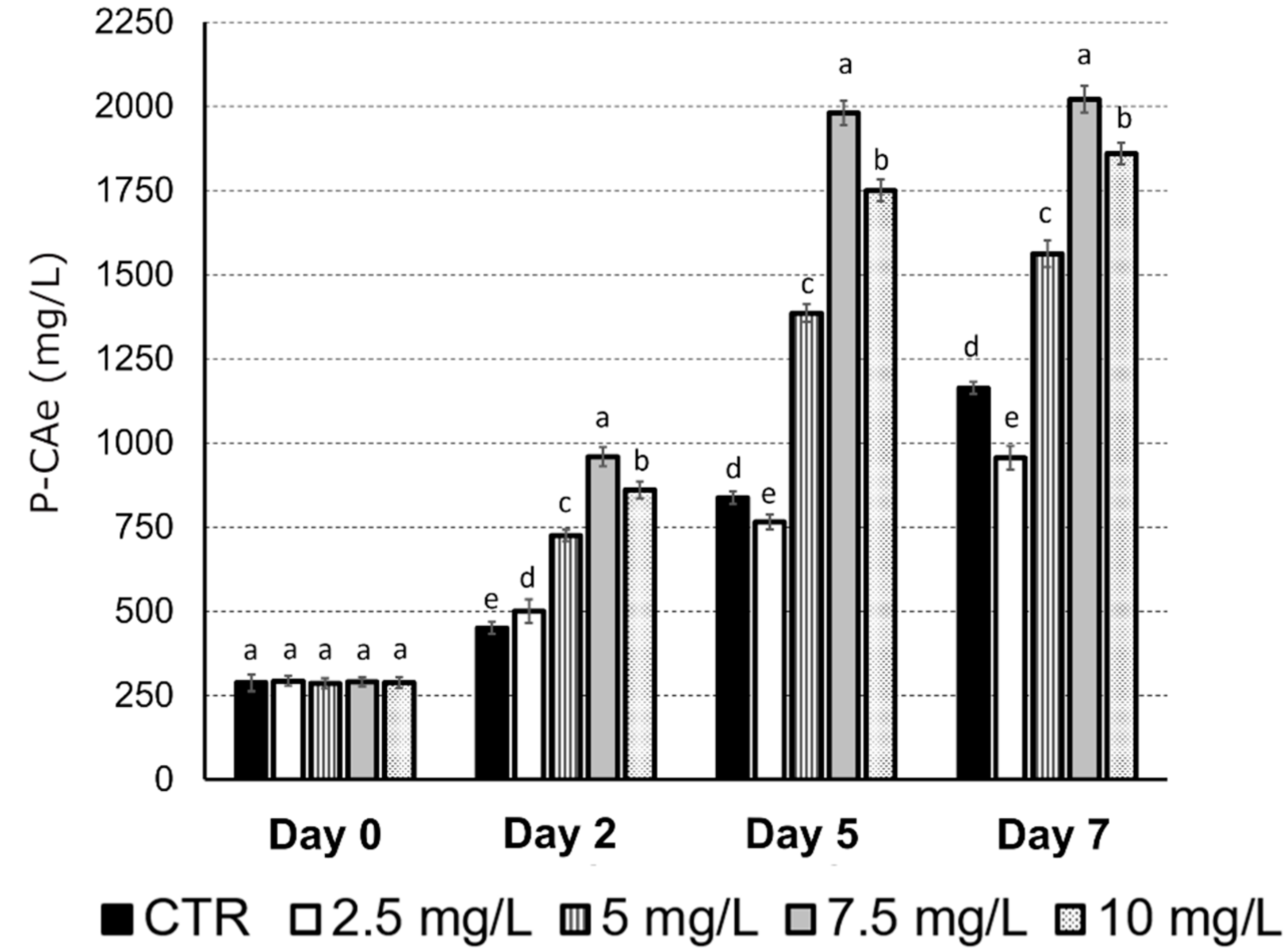
2.1. Evaluation of the Methyl Jasmonate (MeJa) Optimum Concentration to Stimulate Chicoric Acid Biosynthesis in Echinacea Purpurea Cell Suspensions
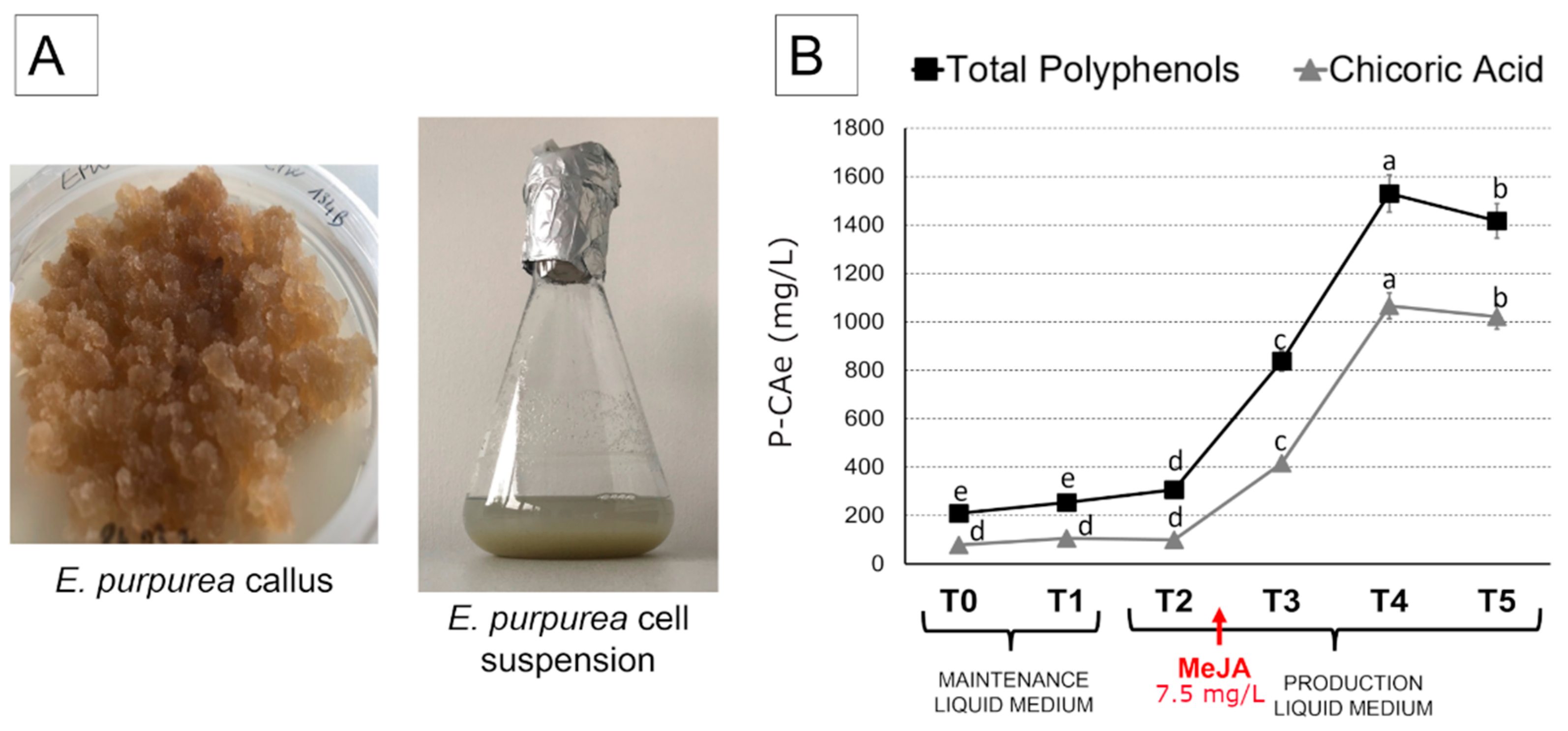
2.2. Total Polyphenols and Chicoric Acid Content in Echinacea Purpurea Cell Suspensions
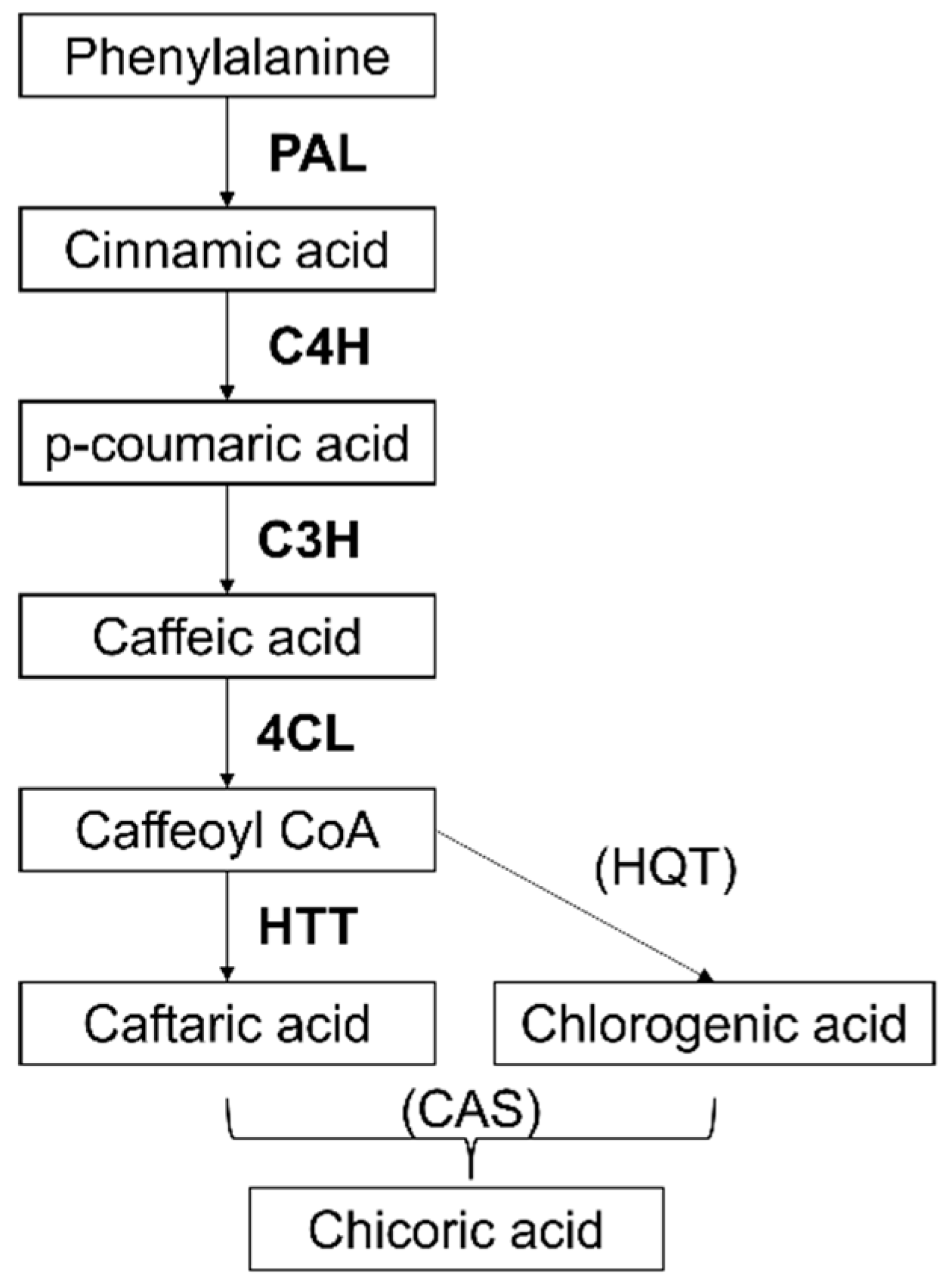
2.3. Identification of Gene Sequences Involved in Chicoric Acid Biosynthesis in Echinacea Purpurea and Expression Analysis on Cell Suspensions to Select Markers Elicited by Methyl Jasmonate (MeJa)
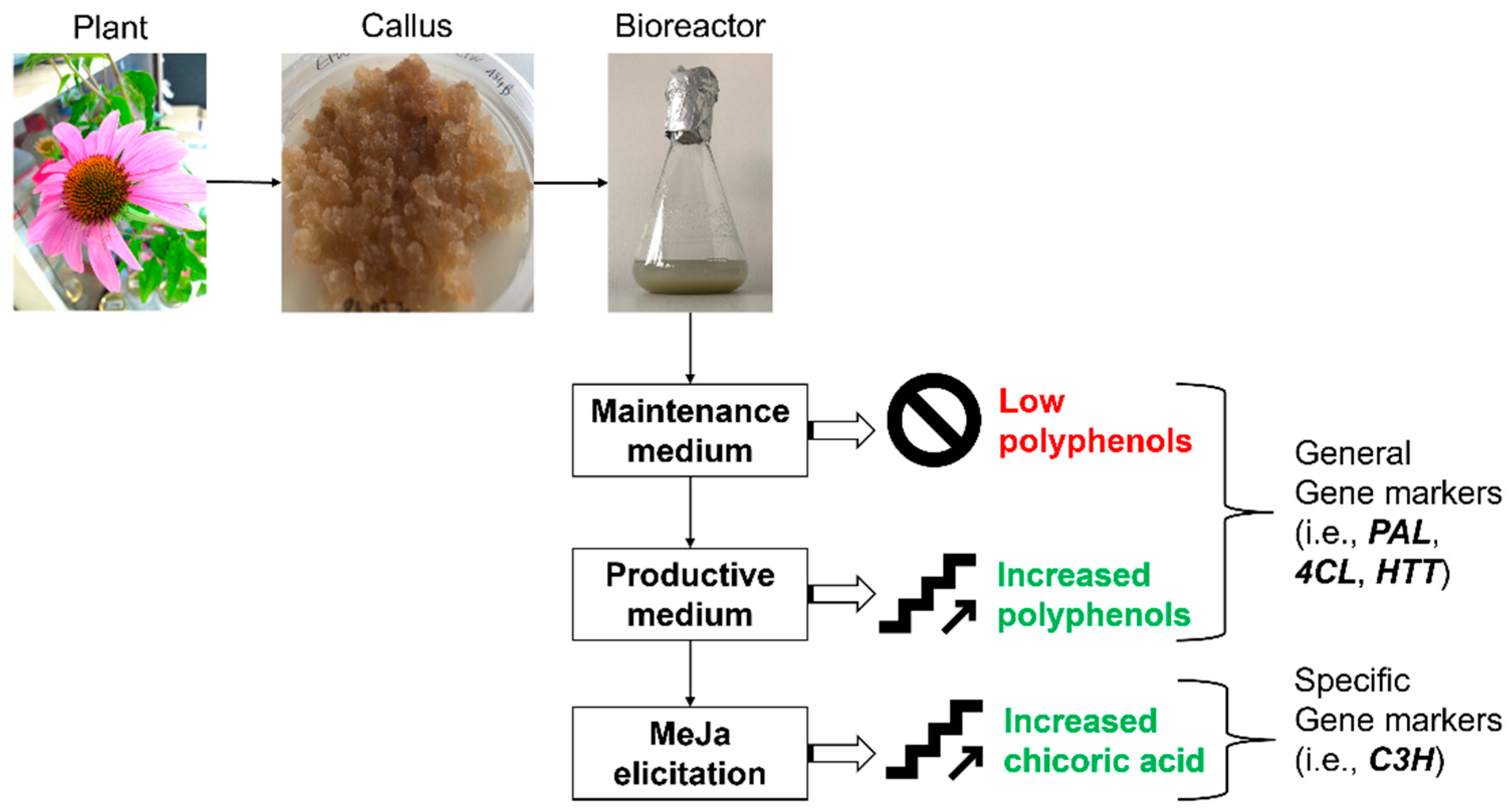
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Callus and Cell Suspension Culture Induction
4.3. Cell Growth Time-Points
4.4. Gene Bank Screening and Primer Design
4.5. RNA Extraction and cDNA Synthesis
4.6. Quantitative Real-Time PCR (RT-qPCR)
4.7. UPLC-DAD Analysis of Chicoric Acid and Total Polyphenol Content
5. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Barrett, B. Medicinal properties of Echinacea: A critical review. Phytomedicine 2003, 10, 66–86. [Google Scholar] [CrossRef] [PubMed]
- McKeown, K.A. A review of the taxonomy of the genus Echinacea. In Perspectives on New Crops and New Uses; Janick, J., Ed.; ASHS Press: Alexandria, VA, USA, 1999; pp. 482–489. [Google Scholar]
- Burlou-Nagy, C.; Bănică, F.; Jurca, T.; Vicaș, L.G.; Marian, E.; Muresan, M.E.; Bácskay, I.; Kiss, R.; Fehér, P.; Pallag, A. Echinacea purpurea (L.) Moench: Biological and Pharmacological Properties. A Review. Plants 2022, 11, 1244. [Google Scholar] [CrossRef]
- Tahmasebi, A.; Aram, F.; Pakniyat, H.; Niazi, A.; Tavakol, E.; Ebrahimie, E. Global analysis of gene expression and identification of modules in Echinacea purpurea using systems biology approach. J. Cell. Mol. Med. 2018, 10, 18–26. [Google Scholar] [CrossRef]
- Tsai, Y.L.; Chiu, C.C.; Chen, J.Y.F.; Chan, K.C.; Lin, S.D. Cytotoxic effects of Echinacea purpurea flower extracts and cichoric acid on human colon cancer cells through induction of apoptosis. J. Ethnopharmacol. 2012, 143, 914–919. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, M.; Mnayer, D.; Morais-Braga, M.; Carneiro, J.; Bezerra, C.F.; Coutinho, H.; Salehi, B.; Martorell, M.; Del Mar Contreras, M.; Soltani-Nejad, A.; et al. Echinacea plants as antioxidant and antibacterial agents: From traditional medicine to biotechnological applications. Phytother. Res. 2018, 32, 1653–1663. [Google Scholar] [CrossRef]
- Yang, M.; Wu, C.; Zhang, T.; Shi, L.; Li, J.; Liang, H.; Lv, X.; Jing, F.; Qin, L.; Zhao, T.; et al. Chicoric Acid: Natural Occurrence, Chemical Synthesis, Biosynthesis, and Their Bioactive Effects. Front. Chem. 2022, 10, 888673. [Google Scholar] [CrossRef]
- Qu, L.; Chen, Y.; Wang, X.; Scalzo, R.; Davis, J.M. Patterns of variation in alkamides and cichoric acid in roots and aboveground parts of Echinacea purpurea (L.) Moench. HortScience 2005, 40, 1239–1242. [Google Scholar] [CrossRef]
- Currier, N.L.; Miller, S.C. Echinacea purpurea and melatonin augment natural-killer cells in leukemic mice and prolong life span. J. Altern. Complement. Med. 2001, 7, 241–251. [Google Scholar] [CrossRef]
- Hudson, J.B. Applications of the phytomedicine Echinacea purpurea (purple coneflower) in infectious diseases. J. Biomed. Biotechnol. 2012, 2012, 769896. [Google Scholar] [CrossRef]
- Thygesen, L.; Thulin, J.; Mortensen, A.; Skibsted, L.H.; Molgaard, P. Antioxidant activity of cichoric acid and alkamides from Echinacea purpurea, alone and in combination. Food Chem. 2007, 101, 74–81. [Google Scholar] [CrossRef]
- Stanisavljević, I.; Stojičević, S.; Veličković, D.; Veljković, V.; Lazić, M. Antioxidant and antimicrobial activities of Echinacea (Echinacea purpurea L.) extracts obtained by classical and ultrasound extraction. Chin. J. Chem. Eng. 2009, 17, 478–483. [Google Scholar] [CrossRef]
- Langland, J.; Jacobs, B.; Wagner, C.E.; Ruiz, G.; Cahill, T.M. Antiviral activity of metal chelates of caffeic acid and similar compounds towards herpes simplex, VSV-Ebola pseudotyped and vaccinia viruses. Antivir. Res. 2018, 160, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Nagoor Meeran, M.F.; Javed, H.; Sharma, C.; Goyal, S.N.; Kumar, S.; Jha, N.K.; Ojha, S. Can Echinacea be a potential candidate to target immunity, inflammation, and infection—The trinity of coronavirus disease 2019. Heliyon 2021, 7, e05990. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Scagel, C.F. Chicoric acid: Chemistry, distribution, and production. Front. Chem. 2013, 1, 40. [Google Scholar] [CrossRef]
- Murthy, H.N.; Kim, Y.S.; Park, S.Y.; Paek, K.Y. Biotechnological production of caffeic acid derivatives from cell and organ cultures of Echinacea species. Appl. Microbiol. Biotechnol. 2014, 98, 7707–7717. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, M.L. Perennial peanut (Arachis glabrata Benth.) leaves contain hydroxycinnamoyl-CoA:tartaric acid hydroxycinnamoyl transferase activity and accumulate hydroxycinnamoyl-tartaric acid esters. Planta 2014, 239, 1091–1100. [Google Scholar] [CrossRef]
- Fu, R.; Zhang, P.; Jin, G.; Wang, L.; Qi, S.; Cao, Y.; Martin, C.; Zhang, Y. Versatility in acyltransferase activity completes chicoric acid biosynthesis in purple coneflower. Nat. Commun. 2021, 12, 1563. [Google Scholar] [CrossRef]
- Letchamo, W.; Polydeonny, L.V.; Gladisheva, N.O.; Arnason, T.J.; Livesey, J.; Awang, D.V.C. Factors affecting Echinacea quality. In Trends in New Crops and New Uses; Janick, J., Whipkey, A., Eds.; ASHS Press: Alexandria, VA, USA, 2002; pp. 514–521. [Google Scholar]
- Georgiev, M.; Eibl, R.; Zhong, J.J. Hosting the plant cells in vitro: Recent trends in bioreactors. Appl. Microbiol. Biotechnol. 2013, 97, 3787–3800. [Google Scholar] [CrossRef]
- Parsons, J.L.; Cameron, S.I.; Harris, C.S.; Smith, M.L. Echinacea biotechnology: Advances, commercialization and future considerations. Pharm. Biol. 2018, 56, 485–494. [Google Scholar] [CrossRef]
- Li, W.W.; Barz, W. Structure and accumulation of phenolics in elicited Echinacea purpurea cell cultures. Planta Med. 2006, 72, 248–254. [Google Scholar] [CrossRef]
- Patel, H.; Krishnamurthy, R. Elicitors in plant tissue culture. J. Pharmacogn. Phytochem. 2013, 2, 60–65. [Google Scholar]
- Tahmasebi, A.; Ebrahimie, E.; Pakniyat, H.; Ebrahimi, M.; Mohammadi-Dehcheshmeh, M. Insights from the Echinacea purpurea (L.) Moench transcriptome: Global reprogramming of gene expression patterns towards activation of secondary metabolism pathways. Ind. Crops Prod. 2019, 132, 365–376. [Google Scholar] [CrossRef]
- Salmanzadeh, M.; Sabet, M.S.; Moieni, A.; Homaee, M. Heterologous expression of an acid phosphatase gene and phosphate limitation leads to substantial production of chicoric acid in Echinacea purpurea transgenic hairy roots. Planta 2019, 251, 31. [Google Scholar] [CrossRef] [PubMed]
- Harborne, J.B.; Williams, C.A. Phytochemistry of the Genus Echinacea, Echinacea; CRC Press: Boca Raton, FL, USA, 2004; pp. 71–88. [Google Scholar]
- Rao, S.R.; Ravishankar, K.A. Plant cell cultures: Chemical factories of secondary metabolites. Biotechnol. Adv. 2002, 20, 101–153. [Google Scholar] [CrossRef]
- Wu, C.H.; Murthy, H.N.; Hahn, E.J.; Paek, K.Y. Enhanced production of caftaric acid, chlorogenic acid and cichoric acid in suspension cultures of Echinacea purpurea by the manipulation of incubation temperature and photoperiod. Biochem. Eng. J. 2007, 36, 301–303. [Google Scholar] [CrossRef]
- Chini, A.; Fonseca, S.; Fernández, G.; Adie, B.; Chico, J.M.; Lorenzo, O.; García-Casado, G.; López-Vidriero, I.; Lozano, F.M.; Ponce, M.R.; et al. The JAZ family of repressors is the missing link in jasmonate signalling. Nature 2007, 448, 666–671. [Google Scholar] [CrossRef]
- Wasternack, C.; Song, S. Jasmonates: Biosynthesis, metabolism, and signaling by proteins activating and repressing transcription. J. Exp. Bot. 2017, 68, 1303–1321. [Google Scholar] [CrossRef]
- Kriegshauser, L.; Knosp, S.; Grienenberger, E.; Tatsumi, K.; Gütle, D.D.; Sørensen, I.; Herrgott, L.; Zumsteg, J.; Rose, J.; Reski, R.; et al. Function of the HYDROXYCINNAMOYL-CoA:SHIKIMATE HYDROXYCINNAMOYL TRANSFERASE is evolutionarily conserved in embryophytes. Plant Cell 2021, 33, 1472–1491. [Google Scholar] [CrossRef]
- Mekapogu, M.; Vasamsetti, B.M.K.; Kwon, O.K.; Ahn, M.S.; Lim, S.H.; Jung, J.A. Anthocyanins in Floral Colors: Biosynthesis and Regulation in Chrysanthemum Flowers. Int. J. Mol. Sci. 2020, 21, 6537. [Google Scholar] [CrossRef]
- Barros, J.; Escamilla-Trevino, L.; Song, L.; Rao, X.; Serrani-Yarce, J.C.; Palacios, M.D.; Engle, N.; Choudhury, F.K.; Tschaplinski, T.J.; Venables, B.J.; et al. 4-Coumarate 3-hydroxylase in the lignin biosynthesis pathway is a cytosolic ascorbate peroxidase. Nat. Commun. 2019, 10, 1994. [Google Scholar] [CrossRef]
- Gao, W.; Li, H.Y.; Xiao, S.; Chye, M.L. Acyl-CoA-binding protein 2 binds lysophospholipase 2 and lysoPC to promote tolerance to cadmium-induced oxidative stress in transgenic Arabidopsis. Plant J. 2010, 62, 989–1003. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, T.; Koita, H.; Nakatsubo, T.; Hasegawa, K.; Wakabayashi, K.; Takahashi, H.; Umemura, K.; Umezawa, T.; Shimamoto, K. Cinnamoyl-CoA reductase, a key enzyme in lignin biosynthesis, is an effector of small GTPase Rac in defense signaling in rice. Proc. Natl. Acad. Sci. USA 2006, 103, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Gamborg, O.L.; Miller, R.A.; Ojima, K. Nutrient requirements of suspension cultures of soybean roots. Exp. Cell Res. 1968, 50, 151–158. [Google Scholar] [CrossRef]
- Rizhsky, L.; Jin, H.; Shepard, M.R.; Scott, H.W.; Teitgen, A.M.; Perera, M.A.; Mhaske, V.; Jose, A.; Zheng, X.; Crispin, M.; et al. Integrating metabolomics and transcriptomics data to discover a biocatalyst that can generate the amine precursors for alkamide biosynthesis. Plant J. 2016, 88, 775–793. [Google Scholar] [CrossRef]
- Manoli, A.; Sturaro, A.; Trevisan, S.; Quaggiotti, S.; Nonis, A. Evaluation of candidate reference genes for qPCR in maize. J. Plant Physiol. 2012, 169, 807–815. [Google Scholar] [CrossRef]
- Nonis, A.; Ruperti, B.; Falchi, R.; Casatta, E.; Thamashebi, S.E.; Vizzotto, G. Differential expression and regulation of a neutral invertase encoding gene from peach (Prunus persica): Evidence for a role in fruit development. Physiol. Plant 2007, 129, 436–446. [Google Scholar] [CrossRef]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative C(T) method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef]
- Rancurel, C.; Van Tran, T.; Elie, C.; Hilliou, F. SATQPCR: Website for statistical analysis of real-time quantitative PCR data. Mol. Cell Probes 2019, 46, 101418. [Google Scholar] [CrossRef]


| Time-Point | Growth Condition | Type of Nutrient Medium |
|---|---|---|
| T0 | 5 days in the maintenance medium (liquid EP-medium) | Maintenance medium (liquid EP-medium) |
| T1 | 7 days in the maintenance medium (liquid EP-medium) | Maintenance medium (liquid EP-medium) |
| T2 | 7 days in production medium (EP-F medium) | Production medium (EP-F medium) |
| T3 | 9 days in production medium (EP-F medium) | Production medium (EP-F medium) with MeJa for 2 d |
| T4 | 12 days in production medium (EP-F medium) | Production medium (EP-F medium) with MeJa for 5 d |
| T5 | 14 days in production medium (EP-F medium) | Production medium (EP-F medium) with MeJa for 7 d |
| Time from Start of the Analysis (Minutes) | Percentage of Solvent B | Slope |
|---|---|---|
| 0 | 1% | |
| 1 | 1% | Linear |
| 11 | 40% | Linear |
| 12 | 100% | Linear |
| 13 | 100% | Linear |
| 13.10 | 1% | Linear |
| 15 | 1% | Linear |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ravazzolo, L.; Ruperti, B.; Frigo, M.; Bertaiola, O.; Pressi, G.; Malagoli, M.; Quaggiotti, S. C3H Expression Is Crucial for Methyl Jasmonate Induction of Chicoric Acid Production by Echinacea purpurea (L.) Moench Cell Suspension Cultures. Int. J. Mol. Sci. 2022, 23, 11179. https://doi.org/10.3390/ijms231911179
Ravazzolo L, Ruperti B, Frigo M, Bertaiola O, Pressi G, Malagoli M, Quaggiotti S. C3H Expression Is Crucial for Methyl Jasmonate Induction of Chicoric Acid Production by Echinacea purpurea (L.) Moench Cell Suspension Cultures. International Journal of Molecular Sciences. 2022; 23(19):11179. https://doi.org/10.3390/ijms231911179
Chicago/Turabian StyleRavazzolo, Laura, Benedetto Ruperti, Marco Frigo, Oriana Bertaiola, Giovanna Pressi, Mario Malagoli, and Silvia Quaggiotti. 2022. "C3H Expression Is Crucial for Methyl Jasmonate Induction of Chicoric Acid Production by Echinacea purpurea (L.) Moench Cell Suspension Cultures" International Journal of Molecular Sciences 23, no. 19: 11179. https://doi.org/10.3390/ijms231911179
APA StyleRavazzolo, L., Ruperti, B., Frigo, M., Bertaiola, O., Pressi, G., Malagoli, M., & Quaggiotti, S. (2022). C3H Expression Is Crucial for Methyl Jasmonate Induction of Chicoric Acid Production by Echinacea purpurea (L.) Moench Cell Suspension Cultures. International Journal of Molecular Sciences, 23(19), 11179. https://doi.org/10.3390/ijms231911179









