Effect of Autolyzed Yarrowia lipolytica on the Growth Performance, Antioxidant Capacity, Intestinal Histology, Microbiota, and Transcriptome Profile of Juvenile Largemouth Bass (Micropterus salmoides)
Abstract
:1. Introduction
2. Results
2.1. Growth Performance
2.2. Flesh Composition
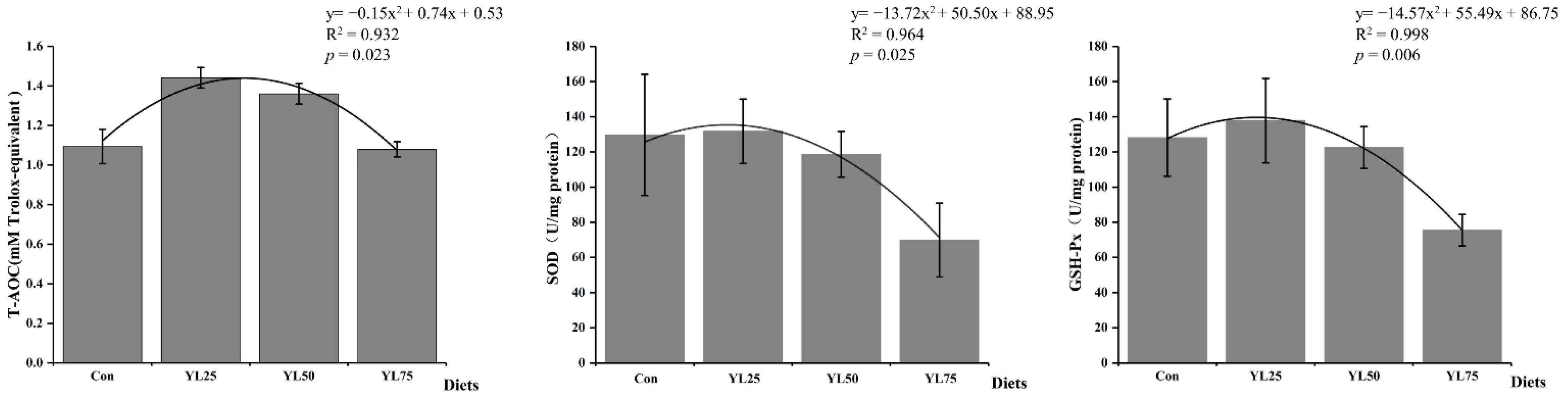
2.3. Antioxidant-Related Enzyme Activities
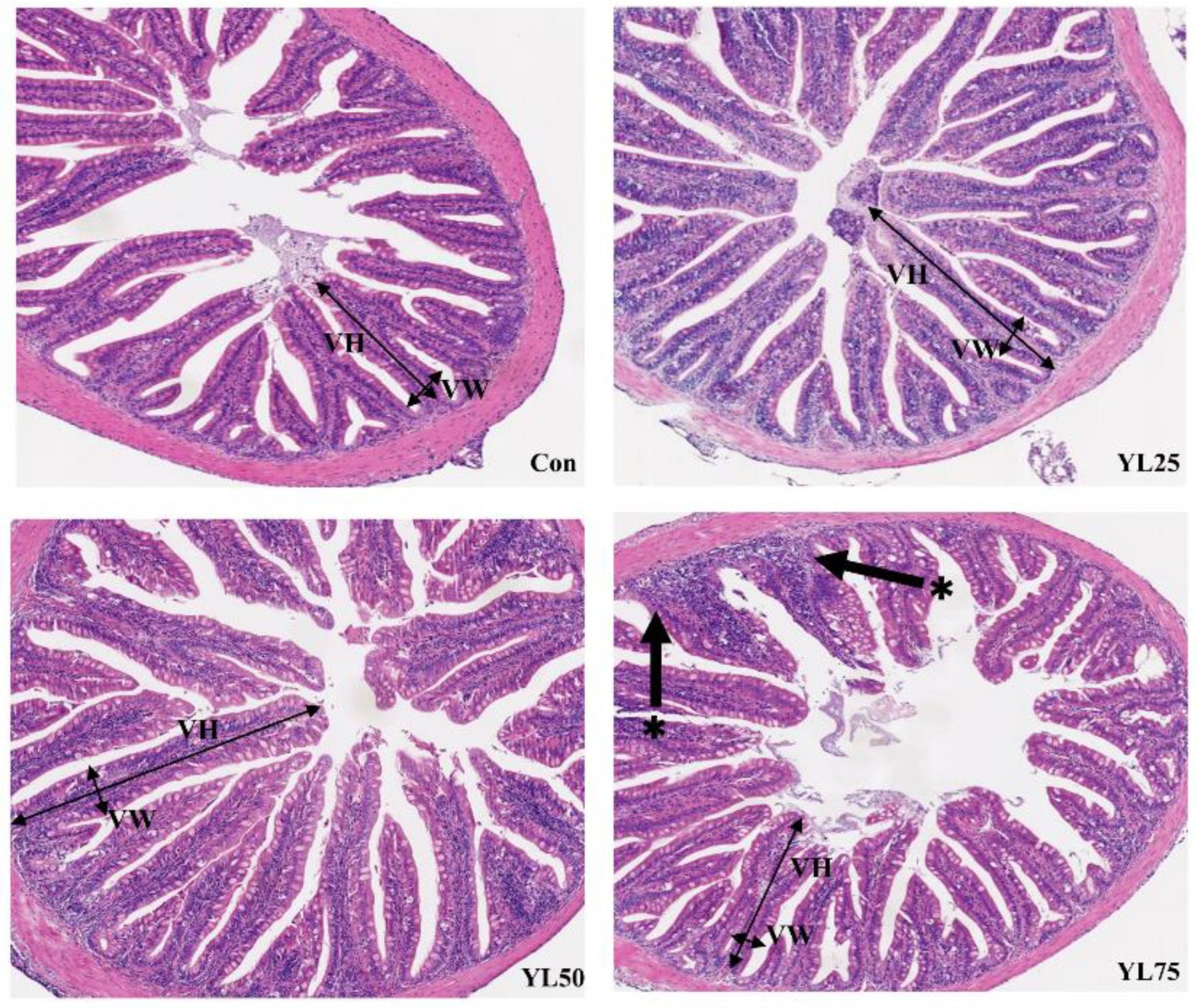
2.4. Intestinal Histomorphology
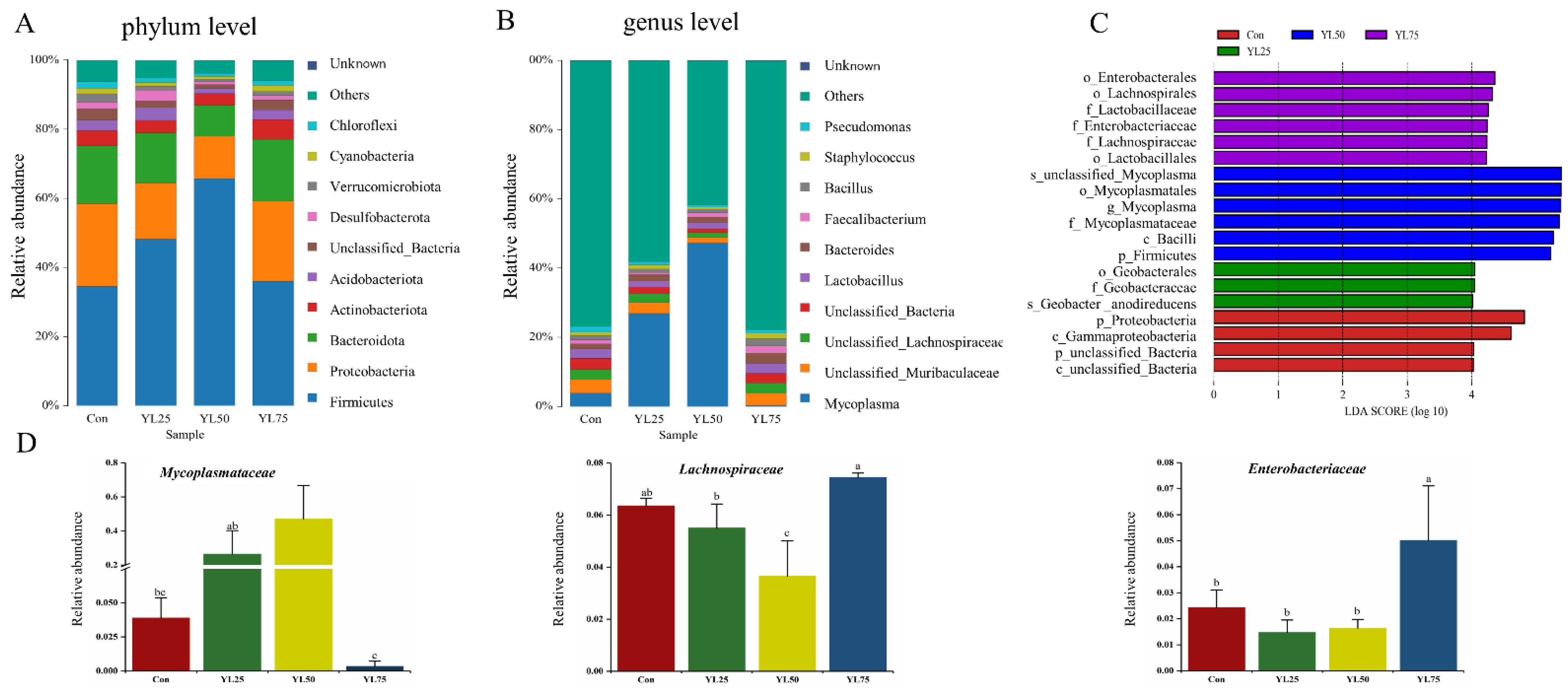
2.5. Analysis of Intestinal Microbiota between Control and YL Treatments
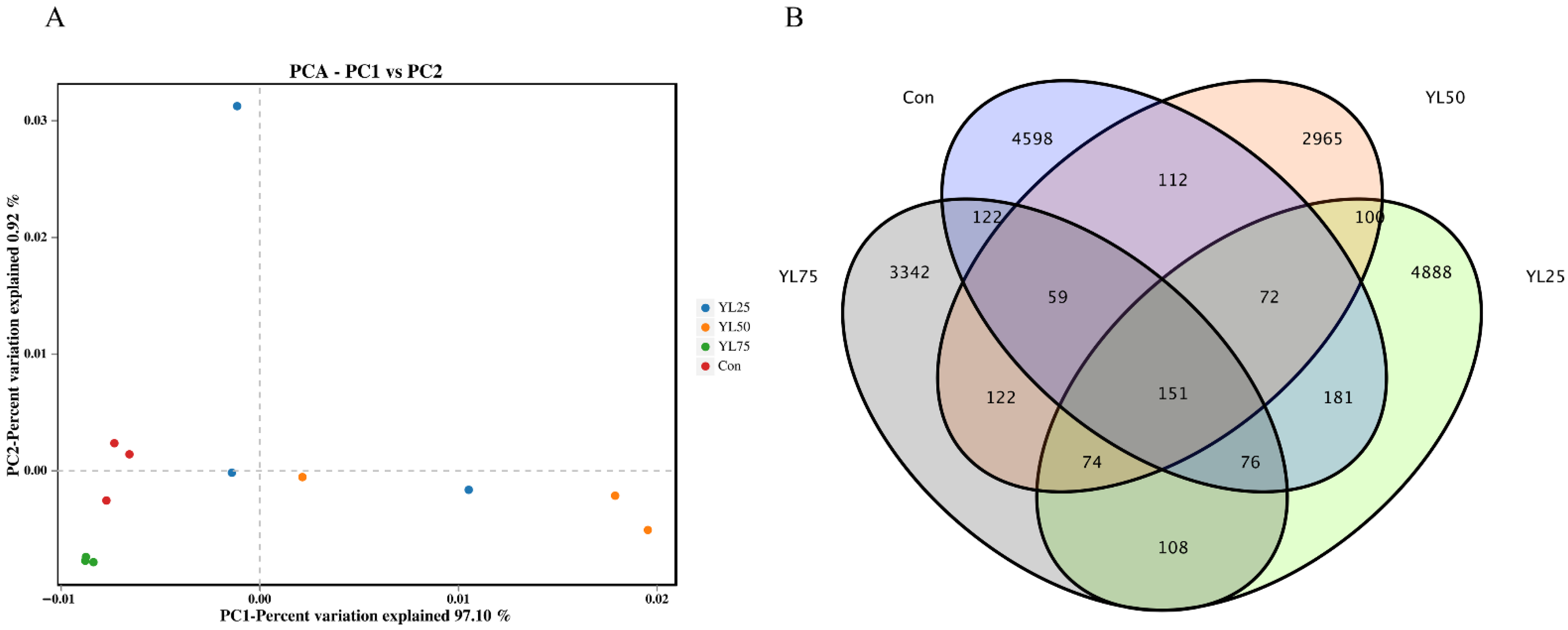
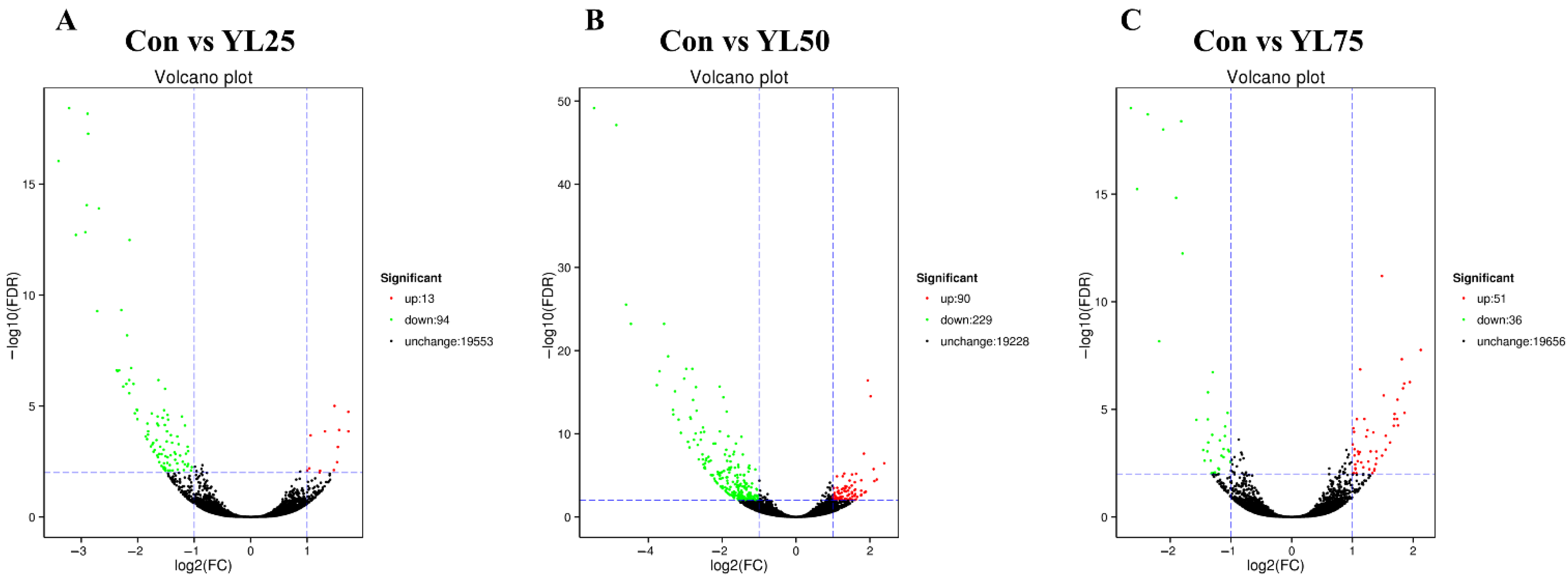
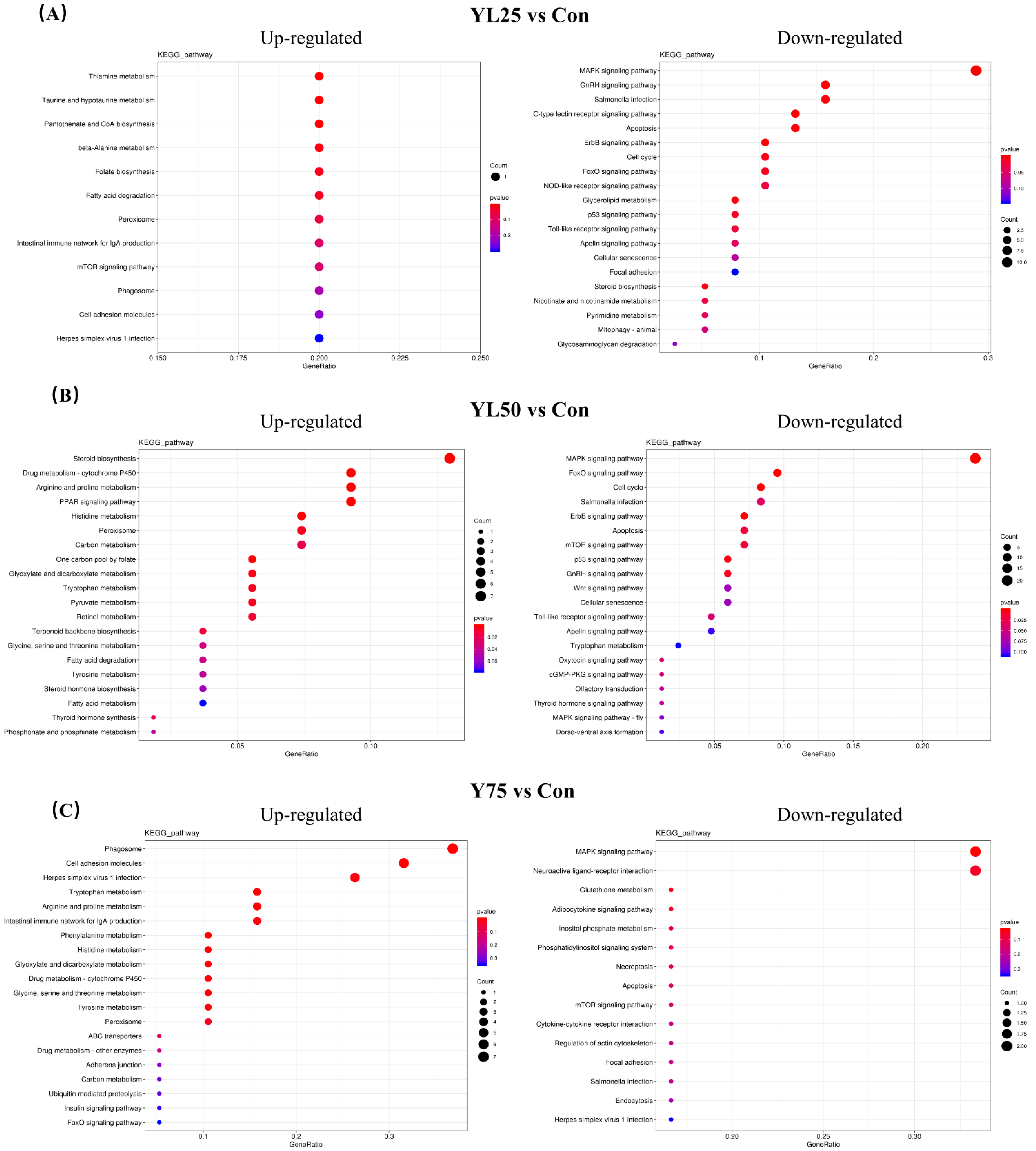
2.6. Differential Gene Expression between the Control and YL Treatments
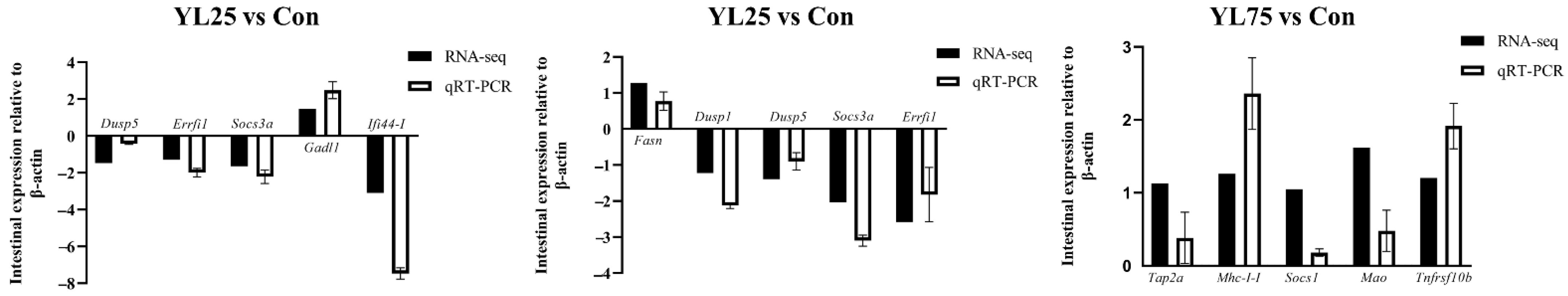
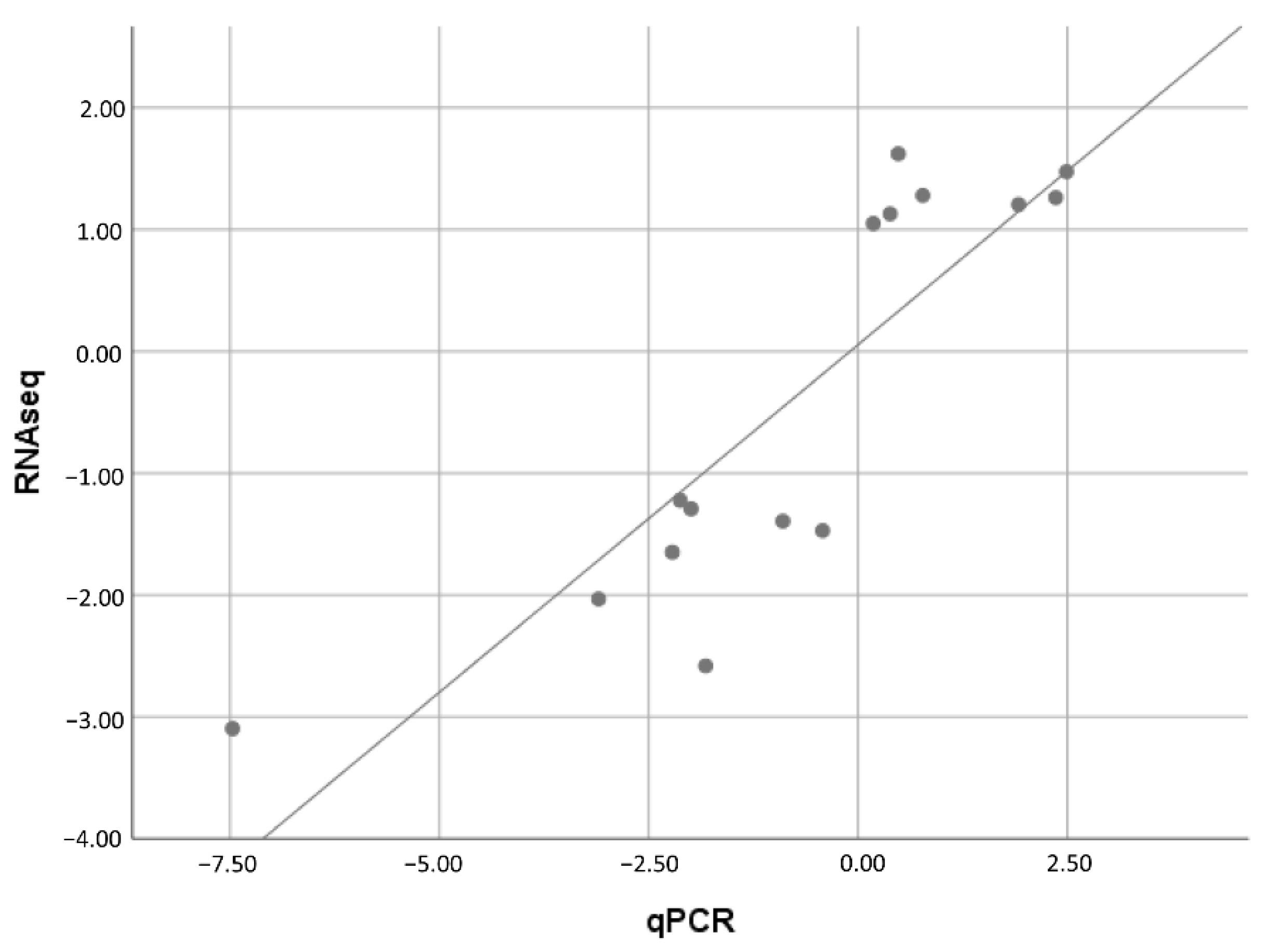
2.7. Validation of Selected DEGs by qRT-PCR
3. Discussion
4. Materials and Methods
4.1. Autolyzed Yarrowia lipolytica Preparation
4.2. Experimental Diets
4.3. Experimental Design
4.4. Sampling
4.5. Analysis of Flesh Composition
4.6. Analysis of Antioxidant-Related Enzyme Activities in Intestine
4.7. Analysis of Intestinal Histology
4.8. RNA Extraction, cDNA Library Construction, Sequencing, De Novo Assembly and Annotation
4.9. Differential Expression Analysis and Functional Enrichment
4.10. Quantitative Real-Time PCR
4.11. High Throughput Sequencing of the 16S rRNA Gene
4.12. Statistics Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Coyle, S.D.; Patton, S.; Schneider, K.; Tidwell, J.H. The Effect of Water Temperature on Growth and Survival of Largemouth Bass during Feed Training. N. Am. J. Aquac. 2016, 71, 256–259. [Google Scholar] [CrossRef]
- Zhong, Y.-F.; Shi, C.-M.; Zhou, Y.-L.; Chen, Y.-J.; Lin, S.-M.; Tang, R.-J. Optimum dietary fiber level could improve growth, plasma biochemical indexes and liver function of largemouth bass, Micropterus salmoides. Aquaculture 2020, 518, 734661. [Google Scholar] [CrossRef]
- Coursey, A.R.; Trushenski, J.T.; Kohler, C.C. Alternative Feeding Ftrategies to Maximize Fish Oil and Fish Meal Sparing in Largemouth Bass Culture while Maintaining Production Performance and Product Value. N. Am. J. Aquacult. 2013, 75, 266–276. [Google Scholar] [CrossRef]
- Yuan, X.Y.; Zhang, X.T.; Xia, Y.T.; Zhang, Y.Q.; Wang, B.; Ye, W.W.; Ye, Z.F.; Qian, S.C.; Huang, M.M.; Yang, S.; et al. Transcriptome and 16S rRNA analyses revealed differences in the responses of largemouth bass (Micropterus salmoides) to early Aeromonas hydrophila infection and immunization. Aquaculture 2021, 541, 736759. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, Y.; Ren, X.; Huang, D.; Si, G.; Chen, J. Replacement of fish meal with gamma-ray irradiated soybean meal in the diets of largemouth bass Micropterus salmoides. Aquac. Nutr. 2021, 27, 977–985. [Google Scholar] [CrossRef]
- Romano, N.; Fischer, H.; Rossi, W.; Quintero, H.; Limbaugh, N.; Sinha, A.K. Effects of Bioprocessed Soybean Meal and Nu-cleotide Supplementation on Growth, Physiology and Histomorphology in Largemouth Bass, Micropterus Salmoides, Juveniles. Com. Biochem. Phys. A 2021, 260, 111038. [Google Scholar] [CrossRef]
- Liu, Y.; Lu, Q.; Xi, L.; Gong, Y.; Su, J.; Han, D.; Zhang, Z.; Liu, H.; Jin, J.; Yang, Y.; et al. Effects of Replacement of Dietary Fishmeal by Cottonseed Protein Concentrate on Growth Performance, Liver Health, and Intestinal Histology of Largemouth Bass (Micropterus salmoides). Front. Physiol. 2021, 12, 764987. [Google Scholar] [CrossRef]
- Xie, X.; Wang, J.; Guan, Y.; Xing, S.; Liang, X.; Xue, M.; Wang, J.; Chang, Y.; Leclercq, E. Cottonseed protein concentrate as fishmeal alternative for largemouth bass (Micropterus salmoides) supplemented a yeast-based paraprobiotic: Effects on growth performance, gut health and microbiome. Aquaculture 2022, 551, 737898. [Google Scholar] [CrossRef]
- Yang, H.; Bian, Y.; Huang, L.; Lan, Q.; Ma, L.; Li, X.; Leng, X. Effects of replacing fish meal with fermented soybean meal on the growth performance, intestinal microbiota, morphology and disease resistance of largemouth bass (Micropterus salmoides). Aquac. Rep. 2022, 22, 100954. [Google Scholar] [CrossRef]
- Caruffo, M.; Navarrete, N.C.; Salgado, O.A.; Faúndez, N.B.; Gajardo, M.C.; Feijóo, C.G. Protective Yeasts Control V. Anguil-larum Pathogenicity and Modulate the Innate Immune Response of Challenged Zebrafish (Danio rerio) Larvae. Front. Cell. Infect. Microbiol. 2016, 6, 127. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.; Dong, X.; Tan, B.; Du, T.; Zhang, S.; Yang, Y.; Chi, S.; Yang, Q.; Liu, H. Effects of fish meal replacement by low-gossypol cottonseed meal on growth performance, digestive enzyme activity, intestine histology and inflammatory gene expression of silver sillago (Sillago sihama Forsskál) (1775). Aquac. Nutr. 2020, 26, 1724–1735. [Google Scholar] [CrossRef]
- Wan, M.; Yin, P.; Fang, W.; Xie, S.; Chen, S.; Tian, L.; Niu, J. The effect of replacement of fishmeal by concentrated de-phenolization cottonseed protein on the growth, body composition, haemolymph indexes and haematological enzyme activities of the Pacific white shrimp (Litopenaeus vannamei). Aquac. Nutr. 2018, 24, 1845–1854. [Google Scholar] [CrossRef]
- Gatlin, D.M., III; Barrows, F.T.; Brown, P.; Dabrowski, K.; Gaylord, T.G.; Hardy, R.W.; Herman, E.; Hu, G.; Krogdahl, Å.; Nelson, R.; et al. Expanding the utilization of sustainable plant products in aquafeeds: A review. Aquac. Res. 2007, 38, 551–579. [Google Scholar] [CrossRef]
- Worland, A.M.; Czajka, J.J.; Xing, Y.; Harper, W.F., Jr.; Moore, A.; Xiao, Z.; Han, Z.; Wang, Y.; Su, W.W.; Tang, Y.J. Analysis of Yarrowia lipolytica growth, catabolism, and terpenoid biosynthesis during utilization of lipid-derived feedstock. Metab. Eng. Commun. 2020, 11, e00130. [Google Scholar] [CrossRef] [PubMed]
- Poli, J.S.; da Silva, M.A.; Siqueira, E.P.; Pasa, V.M.; Rosa, C.A.; Valente, P. Microbial Lipid Produced by Yarrowia Lipolytica Qu21 Using Industrial Easte: A Potential Feedstock for Biodiesel Production. Bioresour. Technol. 2014, 161, 320–326. [Google Scholar] [CrossRef]
- Beopoulos, A.; Cescut, J.; Haddouche, R.; Uribelarrea, J.-L.; Molina-Jouve, C.; Nicaud, J.-M. Yarrowia lipolytica as a model for bio-oil production. Prog. Lipid Res. 2009, 48, 375–387. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, L.; Chen, H.; Chen, Y.Q.; Chen, W.; Song, Y.; Ratledge, C. Enhanced lipid accumulation in the yeast Yarrowia lipolytica by over-expression of ATP:citrate lyase from Mus musculus. J. Biotechnol. 2014, 192, 78–84. [Google Scholar] [CrossRef]
- Hatlen, B.; Berge, G.M.; Odom, J.M.; Mundheim, H.; Ruyter, B. Growth Performance, Reed Utilisation and Ratty Acid Depo-sition in Atlantic Salmon, Salmo Salar, L. Fed Graded Levels of High-Lipid/High-EPA Yarrowia lipolytica Biomass. Aquaculture 2012, 364, 39–47. [Google Scholar] [CrossRef]
- Berge, G.M.; Hatlen, B.; Odom, J.M.; Ruyter, B. Physical Treatment of High EPA Yarrowia lipolytica Biomass Increases the Availability of n-3 Highly Unsaturated Fatty Acids when Fed to Atlantic Salmon. Aquacult. Nutr. 2013, 19, 110–121. [Google Scholar] [CrossRef]
- Reyes-Becerril, M.; Alamillo, E.; Angulo, C. Probiotic and Immunomodulatory Activity of Marine Yeast Yarrowia lipolytica Strains and Response Against Vibrioparahaemolyticus in Fish. Probiotics Antimicrob. 2021, 13, 1292–1305. [Google Scholar] [CrossRef]
- Licona-Jain, A.; Campa-Córdova, Á.; Luna-González, A.; Racotta, I.S.; Tello, M.; Angulo, C. Dietary Supplementation of Marine Yeast Yarrowia Lipolytica Modulates Immune Response in Litopenaeus vannamei. Fish Shellfish Immun. 2020, 105, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.-M.; Hou, S.-W.; Wang, G.-X.; Gong, J.-Y.; Zhou, L.; Huang, Y.-H.; Huang, X.-D.; Liu, L. Effects of zymolytic black soldier fly (Hermetia illucens) pulp as dietary supplementation in largemouth bass (Micropterus salmoides). Aquac. Rep. 2021, 21, 100823. [Google Scholar] [CrossRef]
- Baldridge, M.T.; Nice, T.J.; McCune, B.T.; Yokoyama, C.C.; Kambal, A.; Wheadon, M.; Diamond, M.S.; Ivanova, Y.; Artyomov, M.; Virgin, H.W. Commensal microbes and interferon-λ determine persistence of enteric murine norovirus infection. Science 2015, 347, 266–269. [Google Scholar] [CrossRef]
- Su, S.; Jing, X.; Zhang, C.; Hou, Y.; Li, Z.; Yang, X.; Zhou, X.; Xu, P.; Tang, Y.; Zhu, J. Interaction Between the Intestinal Microbial Community and Transcriptome Profile in Common Carp (Cyprinus carpio L.). Front. Microbiol. 2021, 12, 659602. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Wei, H.; Yang, C.; Li, Q.; Li, P.; Chen, J.; Sun, Y.; Wen, H.; Jiang, M.; Wang, G. Effects of dietary protein levels on growth performance and liver transcriptome changes in juvenile top-mouth culter Erythroculter ilishaeformis. Aquac. Rep. 2021, 21, 100964. [Google Scholar] [CrossRef]
- Yu, G.; Liu, Y.; Dai, J.; Ou, W.; Ai, Q.; Zhang, W.; Mai, K.; Zhang, Y. Dietary daidzein improves the development of intestine subjected to soybean meal in juvenile turbot (Scophthalmus maximus L.). Aquac. Nutr. 2020, 27, 17–27. [Google Scholar] [CrossRef]
- Czech, A.; Smolczyk, A.; Ognik, K.; Kiesz, M. Nutritional Value of Yarrowia lipolytica Yeast and Its Effect on Growth Per-formance Indicators n Piglets. Ann. Anim. Sci. 2016, 16, 1091–1100. [Google Scholar] [CrossRef]
- Stefańska, B.; Komisarek, J.; Stanisławski, D.; Gąsiorek, M.; Kasprowicz-Potocka, M.; Frankiewicz, A.; Nowak, W. The effect of Yarrowia lipolytica culture on growth performance, ruminal fermentation and blood parameters of dairy calves. Anim. Feed Sci. Technol. 2018, 243, 72–79. [Google Scholar] [CrossRef]
- Czech, A.; Merska, M.; Ognik, K.; Ziȩba, G. Effect of the Use of Yarrowia lipolytica or Saccharomyces cerevisiae Yeast with a Probiotic in the Diet of Turkey Hens on Growth Performance and Gut Histology. Ann. Anim. Sci. 2020, 20, 1047–1063. [Google Scholar] [CrossRef]
- Verland, M.; Skrede, A. Yeast Derived from Lignocellulosic Biomass as a Sustainable Feed Resource for Use in Aquaculture. J. Sci. Food Agr. 2017, 97, 733–742. [Google Scholar] [CrossRef]
- Kubitza, F.; Lovshin, L.L.; Lovell, R.T. Identification of feed enhancers for juvenile largemouth bass Micropterus salmoides. Aquaculture 1997, 148, 191–200. [Google Scholar] [CrossRef]
- Burrells, C.; Williams, P.; Southgate, P.; Wadsworth, S. Dietary nucleotides: A novel supplement in fish feeds: Effects on vaccination, salt water transfer, growth rates and physiology of Atlantic salmon (Salmo salar L.). Aquaculture 2001, 199, 171–184. [Google Scholar] [CrossRef]
- Aas, T.S.; Grisdale-Helland, B.; Terjesen, B.F.; Helland, S.J. Improved Growth and Nutrient utilisation in Atlantic Salmon (Salmo Salar) Fed Diets Containing a Bacterialprotein Meal. Aquaculture 2006, 259, 365–376. [Google Scholar] [CrossRef]
- Gause, B.; Trushenski, J. Replacement of Fish Meal with Ethanol Yeast in the Diets of Sunshine Bass. N. Am. J. Aquac. 2011, 73, 97–103. [Google Scholar] [CrossRef]
- Wu, F.; Huang, F.; Wen, H.; Jiang, M.; Liu, W.; Tian, J.; Yang, C.G. Vitamin C requirement of adult genetically improved farmed tilapia, Oreochromis niloticus. Aquac. Int. 2015, 23, 1203–1215. [Google Scholar] [CrossRef]
- Oliveira, M.S.B.; Silva, L.M.A.; Prestes, L.; Tavares-Dias, M. Length-Weight Relationship and Condition Factor for Twelve Fish Species from the Igarapé Fortaleza Basin, a Small Tributary of the Amazonas River Estuary. Acta Amaz. 2020, 50, 8–11. [Google Scholar] [CrossRef]
- Ahn, S.E.; Jeong, W.; Kim, J.-H.; Lim, W.; Kim, J.; Bazer, F.W.; Han, J.Y.; Song, G. ERBB receptor feedback inhibitor 1: Identification and regulation by estrogen in chickens. Gen. Comp. Endocrinol. 2011, 175, 194–205. [Google Scholar] [CrossRef]
- Ram, P.A.; Waxman, D.J. SOCS/CIS Protein Inhibition of Growth Hormone-stimulated STAT5 Signaling by Multiple Mechanisms. J. Biol. Chem. 1999, 274, 35553–35561. [Google Scholar] [CrossRef]
- Ueki, I.; Giesy, S.L.; Harvatine, K.J.; Kim, J.W.; Boisclair, Y.R. The Acid-Labile Subunit Is Required for Full Effects of Exogenous Growth Hormone on Growth and Carbohydrate Metabolism. Endocrinology 2009, 150, 3145–3152. [Google Scholar] [CrossRef]
- Zhao, X.Y.; Zhao, S.S.; Zheng, G.D.; Zhou, J.G.; Zhou, S.M. Functional Conservation and Divergence of Duplicated the Sup-pressor of Cytokine Signaling 1 in Blunt Snout Bream (Megalobrama amblycephala). Gen. Comp. Endocr. 2019, 284, 113243. [Google Scholar] [CrossRef]
- Menendez, J.; Lupu, R. Fatty acid synthase and the lipogenic phenotype in cancer pathogenesis. Nat. Cancer 2007, 7, 763–777. [Google Scholar] [CrossRef]
- Ye, B.; Li, J.; Xu, L.; Liu, H.; Yang, M. Metabolomic Effects of the Dietary Inclusion of Hermetia illucens Larva Meal in Tilapia. Metabolites 2022, 12, 286. [Google Scholar] [CrossRef] [PubMed]
- Lei, C.; Li, M.; Zhang, M.; Wang, S.; Tian, J.; Wen, J.; Li, Y. Cloning, molecular characterization, and nutritional regulation of fatty acid-binding protein family genes in gold pompanos (Trachinotus ovatus). Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2020, 246–247, 110463. [Google Scholar] [CrossRef]
- Fawole, F.J.; Labh, S.N.; Hossain, S.; Overturf, K.; Small, B.C.; Welker, T.L.; Hardy, R.W.; Kumar, V. Insect (black soldier fly larvae) oil as a potential substitute for fish or soy oil in the fish meal-based diet of juvenile rainbow trout (Oncorhynchus mykiss). Anim. Nutr. 2021, 7, 1360–1370. [Google Scholar] [CrossRef] [PubMed]
- Douce, R.; Bourguignon, J.; Neuburger, M.; Rébeillé, F. The glycine decarboxylase system: A fascinating complex. Trends Plant Sci. 2001, 6, 167–176. [Google Scholar] [CrossRef]
- Benedetti, M.S.; Tipton, K.F.; Whomsley, R. Amine oxidases and monooxygenases in the in vivo metabolism of xenobiotic amines in humans: Has the involvement of amine oxidases been neglected. Fundam. Clin. Pharmacol. 2007, 21, 467–480. [Google Scholar] [CrossRef]
- Viala, J.P.M.; Méresse, S.; Pocachard, B.; Guilhon, A.-A.; Aussel, L.; Barras, F. Sensing and Adaptation to Low pH Mediated by Inducible Amino Acid Decarboxylases in Salmonella. PLoS ONE 2011, 6, e22397. [Google Scholar] [CrossRef]
- Solans, A.; Estivill, X.; de la Luna, S. Cloning and characterization of human FTCD on 21q22.3, a candidate gene for glutamate formiminotransferase deficiency. Cytogenet. Genome Res. 2000, 88, 43–49. [Google Scholar] [CrossRef]
- Stojanov, S.; Berlec, A.; Štrukelj, B. The Influence of Probiotics on the Firmicutes/Bacteroidetes Ratio in the Treatment of Obesity and Inflammatory Bowel disease. Microorganisms 2020, 8, 1715. [Google Scholar] [CrossRef]
- Feng, Z.; Zhong, Y.; He, G.; Sun, H.; Chen, Y.; Zhou, W.; Lin, S. Yeast culture improved the growth performance, liver function, intestinal barrier and microbiota of juvenile largemouth bass (Micropterus salmoides) fed high-starch diet. Fish Shellfish Immunol. 2022, 120, 706–715. [Google Scholar] [CrossRef]
- Razin, S.; Yogev, D.; Naot, Y. Molecular Biology and Pathogenicity of Mycoplasmas. Microbiol. Mol. Biol. Rev. 1998, 62, 1094–1156. [Google Scholar] [CrossRef] [PubMed]
- Gnanadurai, R.; Fifer, H. Mycoplasma genitalium: A Review. Microbiology 2020, 166, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Liang, R.; Mo, J.; Yang, S.; Gu, N.; Wu, Z.; Babu, V.S.; Li, J.; Huang, Y.; Lin, L. Effects of brewer’s yeast hydrolysate on the growth performance and the intestinal bacterial diversity of largemouth bass (Micropterus salmoides). Aquaculture 2018, 484, 139–144. [Google Scholar] [CrossRef]
- Chen, W.; Chang, K.; Chen, J.; Zhao, X.; Gao, S. Dietary sodium butyrate supplementation attenuates intestinal inflammatory response and improves gut microbiota composition in largemouth bass (Micropterus salmoides) fed with a high soybean meal diet. Fish Physiol. Biochem. 2021, 47, 1805–1819. [Google Scholar] [CrossRef]
- Lian, C.-A.; Yan, G.-Y.; Huang, J.-M.; Danchin, A.; Wang, Y.; He, L.-S. Genomic Characterization of a Novel Gut Symbiont from the Hadal Snailfish. Front. Microbiol. 2020, 10, 2978. [Google Scholar] [CrossRef]
- Cheaib, B.; Yang, P.; Kazlauskaite, R.; Lindsay, E.; Heys, C.; Dwyer, T.; De Noia, M.; Schaal, P.; Sloan, W.; Ijaz, U.; et al. Genome erosion and evidence for an intracellular niche—Exploring the biology of mycoplasmas in Atlantic salmon. Aquaculture 2021, 541, 736772. [Google Scholar] [CrossRef]
- Cotta, M.; Forster, R. The Family Lachnospiraceae, Including the Genera Butyrivibrio, Lachnospira and Roseburia. Prokaryotes 2006, 4, 1002–1021. [Google Scholar] [CrossRef]
- Sheridan, P.; Martin, J.C.; Lawley, T.D.; Browne, H.; Harris, H.M.B.; Bernalier-Donadille, A.; Duncan, S.; O’Toole, P.W.; Scott, K.P.; Flint, H.J. Polysaccharide utilization loci and nutritional specialization in a dominant group of butyrate-producing human colonic Firmicutes. Microb. Genom. 2016, 2, e000043. [Google Scholar] [CrossRef]
- Jiang, Z.; Zhou, Y.; Lu, F.; Han, Z.; Wang, T. Effects of Different Levels of Supplementary Alpha-amylase on Digestive Enzyme Activities and Pancreatic Amylase mRNA Expression of Young Broilers. Asian-Australas. J. Anim. Sci. 2008, 21, 97–102. [Google Scholar] [CrossRef]
- Xu, W.-N.; Liu, W.-B.; Shen, M.-F.; Li, G.-F.; Wang, Y.; Zhang, W.-W. Effect of different dietary protein and lipid levels on growth performance, body composition of juvenile red swamp crayfish (Procambarus clarkii). Aquac. Int. 2013, 21, 687–697. [Google Scholar] [CrossRef]
- Iliev, D.B.; Hansen, T.; Jørgensen, S.M.; Krasnov, A.; Jørgensen, J.B. Cpg- and Lps-Activated Mapk Signaling in n Vitro Cul-tured Salmon (Salmo Salar) Mononuclear Phagocytes. Fish Shellfish Immun. 2013, 35, 1079–1085. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Hao, G.; Li, J.; Peng, W.; Geng, X.; Sun, J. Comparative analysis of dual specificity protein phosphatase genes 1, 2 and 5 in response to immune challenges in Japanese flounder Paralichthys olivaceus. Fish Shellfish Immunol. 2017, 68, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Chuang, H.C.; Tan, T.H. Map4k Family Kinases and Dusp Family Phosphatases in T-cell Signaling and Systemic Lupus Ery-thematosus. Cells 2019, 8, 1433. [Google Scholar] [CrossRef] [PubMed]
- Montserrat, C.; Anton, N.; Steve, A. Dual Specificity Phosphatases: A Gene Family for Control of Map Kinase Function. FASEB J. 2000, 14, 6–16. [Google Scholar]
- Abraham, S.; Clark, A. Dual-specificity phosphatase 1: A critical regulator of innate immune responses. Biochem. Soc. Trans. 2006, 34, 1018–1023. [Google Scholar] [CrossRef]
- Salojin, K.V.; Owusu, I.B.; Millerchip, K.A.; Potter, M.; Platt, K.A.; Oravecz, T. Essential Role of MAPK Phosphatase-1 in the Negative Control of Innate Immune Responses. J. Immunol. 2006, 176, 1899–1907. [Google Scholar] [CrossRef]
- Moon, S.J.; Lim, M.A.; Park, J.S.; Byun, J.K.; Kim, S.M.; Park, M.K.; Kim, E.K.; Moon, Y.M.; Min, J.K.; Ahn, S.M.; et al. Dual-specificity phosphatase 5 attenuates autoimmune arthritis in mice via reciprocal regulation of the th17/treg cell balance and inhibition of osteoclastogenesis. Arthritis Rheumatol. 2014, 66, 3083–3095. [Google Scholar] [CrossRef]
- Holmes, D.A.; Yeh, J.H.; Yan, D.; Xu, M.; Chan, A.C. Dusp5 negatively regulates il-33-mediated eosinophil survival and function. EMBO J. 2015, 4, 218–235. [Google Scholar] [CrossRef]
- Luo, J.; Xue, D.; Song, F.; Liu, X.; Li, W.; Wang, Y. Dusp5 (Dual-Specificity Protein Phosphatase 5) Suppresses Bcg-Induced Autophagy via Erk 1/2 Signaling Pathway. Mol. Immunol. 2020, 126, 101–109. [Google Scholar] [CrossRef]
- Jeffrey, K.; Brummer, T.; Rolph, M.S.; Liu, S.M.; Callejas, N.A.; Grumont, R.J.; Gillieron, C.; Mackay, F.; Grey, S.; Camps, M.; et al. Positive regulation of immune cell function and inflammatory responses by phosphatase PAC-1. Nat. Immunol. 2006, 7, 274–283. [Google Scholar] [CrossRef]
- Giuliani, M.E.; Regoli, F. Identification of the Nrf2-Keap1 Pathway in the European Eel Anguilla Anguilla: Role for a Tran-scriptional Regulation of Antioxidant Genes in Aquatic Organisms. Aquat. Toxicol. 2014, 150, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Alamillo, E.; Reyes-Becerril, M.; Cuesta, A.; Angulo, C. Marine yeast Yarrowia lipolytica improves the immune responses in Pacific red snapper (Lutjanus peru) leukocytes. Fish Shellfish Immunol. 2017, 70, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Lupp, C.; Robertson, M.L.; Wickham, M.E.; Sekirov, I.; Champion, O.L.; Gaynor, E.C.; Finlay, B.B. Host-Mediated Inflammation Disrupts the Intestinal Microbiota and Promotes the Overgrowth of Enterobacteriaceae. Cell Host Microbe 2007, 2, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Garrett, W.S.; Gallini, C.A.; Yatsunenko, T.; Michaud, M.; DuBois, A.; Delaney, M.L.; Punit, S.; Karlsson, M.; Bry, L.; Glickman, J.N.; et al. Enterobacteriaceae Act in Concert with the Gut Microbiota to Induce Spontaneous and Maternally Transmitted Colitis. Cell Host Microbe 2010, 8, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Abele, R.; Tampé, R. Peptides Induce ATP Hydrolysis at Both Subunits of the Transporter Associated with Antigen Processing. J. Biol. Chem. 2003, 278, 29686–29692. [Google Scholar] [CrossRef]
- Gherardin, N.A.; Keller, A.N.; Woolley, R.E.; Le Nours, J.; Ritchie, D.S.; Neeson, P.J.; Birkinshaw, R.W.; Eckle, S.B.; Waddington, J.N.; Liu, L.; et al. Diversity of T Cells Restricted by the MHC Class I-Related Molecule MR1 Facilitates Differential Antigen Recognition. Immunity 2016, 44, 32–45. [Google Scholar] [CrossRef]
- Wang, B.; Du, H.H.; Huang, H.Q.; Xian, J.A.; Xia, Z.H.; Hu, Y.H. Major Histocompatibility Complex Class I (MHC I Alpha) of Japanese flounder (Paralichthys Olivaceus) Plays a Critical Role in Defense Against Intracellular Pathogen Infection. Fish Shellfish Immun. 2019, 94, 122–131. [Google Scholar]
- Luo, W.; Zhang, J.; Wen, J.F.; Liu, H.; Wang, W.M.; Gao, Z.X. Molecular Cloning and Expression Analysis of Major Histo-compatibility Complex Class I, IIA and IIB Genes of Blunt Snout Bream (Megalobrama Amblycephala). Dev. Comp. Immunol. 2014, 42, 169–173. [Google Scholar] [CrossRef]
- Nurkkala, M.; Nordström, I.; Telemo, E.; Eriksson, K. Mhc Expression and Chemokine Production in the Murine Vagina Fol-lowing Intra-Vaginal Administration of Ligands to Toll-Like Receptors 3, 7 and 9. J. Reprod. Immunol. 2007, 73, 148–157. [Google Scholar]
- Cao, Z.; He, M.; Chen, X.; Wang, S.; Cai, Y.; Xie, Z.; Sun, Y.; Zhou, Y. Identification, polymorphism and expression of MHC class Iα in golden pompano, Trachinotus ovatus. Fish Shellfish Immunol. 2017, 67, 55–65. [Google Scholar] [CrossRef]
- Yan, J.; Han, B.; Gui, X.; Wang, G.; Xu, L.; Yan, Y.; Madzak, C.; Pan, D.; Wang, Y.; Zha, G.; et al. Engineering Yarrowia Lipolytica to Simultaneously Produce Lipase and Single Cell Protein from Agro-industrial Wastes for Feed. Sci. Rep. 2018, 8, 758. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Gao, W.; Wen, Z.; Chi, S.; Shi, Y.; Hu, W.; Tan, B. Partial substitution of fish meal by Clostridium autoethanogenum protein in the diets of juvenile largemouth bass (Micropterus salmoides). Aquac. Rep. 2022, 22, 100938. [Google Scholar] [CrossRef]
- Yang, L.; Wang, C.; Shu, J.; Feng, H.; He, Y.; Chen, J.; Shu, J. Porcine Epidemic Diarrhea Virus Induces Vero Cell Apoptosis via the p53-PUMA Signaling Pathway. Viruses 2021, 13, 1218. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Wang, R.; Jiang, Q.; Zhang, D.; Mu, R.; Xu, Y.; Nnaemeka, V.E.; Mei, J.; Zhao, Y.; Cai, F.; et al. Identification and functional analysis of a pollen fertility-associated gene GhGLP4 of Gossypium hirsutum L. Theor. Appl. Genet. 2021, 134, 3237–3247. [Google Scholar] [CrossRef]
- Feng, T.; Jiang, Y.; Jia, Q.; Han, R.; Wang, D.; Zhang, X.; Liang, Z. Transcriptome Analysis of Different Sections of Rhizome in Polygonatum sibiricum Red. and Mining Putative Genes Participate in Polysaccharide Biosynthesis. Biochem. Genet. 2022, 60, 1547–1566. [Google Scholar] [CrossRef] [PubMed]
- Ashburner, M.; Ball, C.A.; Blake, J.A.; Botstein, D.; Butler, H.; Cherry, J.M.; Davis, A.P.; Dolinski, K.; Dwight, S.S.; Eppig, J.T.; et al. Gene ontology: Tool for the unification of biology. Nat. Genet. 2000, 25, 25–29. [Google Scholar] [CrossRef]
- Ogata, H.; Goto, S.; Sato, K.; Fujibuchi, W.; Bono, H.; Kanehisa, M. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 2000, 28, 29–34. [Google Scholar] [CrossRef]
- Wu, F.; Xu, J.; Gao, T.; Huang, D.; Jin, W. Molecular mechanism of modulating miR482b level in tomato with botrytis cinerea infection. BMC Plant Biol. 2021, 21, 49. [Google Scholar] [CrossRef]
- Ye, T.; Huang, X.; Wang, X.-W.; Shi, Y.-R.; Hui, K.-M.; Ren, Q. Characterization of a gC1qR from the giant freshwater prawn, Macrobrachium rosenbergii. Fish Shellfish Immunol. 2015, 43, 200–208. [Google Scholar] [CrossRef]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Quince, C.; Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Diets | Pooled SEM | Regression (P/R2) | ||||
|---|---|---|---|---|---|---|
| Con | YL25 | YL50 | YL75 | Quadratic | ||
| IBW (g) | 1.07 | 1.07 | 1.06 | 1.11 | n.a. | n.a. |
| FBW (g) | 4.52 | 4.77 | 4.85 | 4.13 | 0.09 | 0.006/0.678 |
| WGR (%) | 322.43 | 345.79 | 357.55 | 272.07 | 8.55 | <0.001/0.817 |
| SGR (%/day) | 5.15 | 5.34 | 5.43 | 4.69 | 0.08 | <0.001/0.818 |
| FCR | 1.06 | 1.05 | 1.02 | 1.31 | 0.03 | 0.001/0.778 |
| CF (g/cm3) | 1.40 | 1.43 | 1.49 | 1.32 | 0.04 | 0.007/0.668 |
| HSI (%) | 1.79 | 1.82 | 1.90 | 1.59 | 0.12 | n.s. |
| Diets | Pooled SEM | Regression (P/R2) | ||||
|---|---|---|---|---|---|---|
| Con | YL25 | YL50 | YL75 | Quadratic | ||
| Moisture (%) | 74.86 | 74.91 | 74.55 | 74.87 | 0.24 | n.s. |
| Crude protein (%) | 17.89 | 17.95 | 18.02 | 17.81 | 0.17 | n.s. |
| Crude lipid (%) | 3.47 | 3.97 | 4.33 | 3.19 | 0.06 | <0.001/0.949 |
| Diets | Pooled SEM | Regression (P/R2) | ||||
|---|---|---|---|---|---|---|
| Con | YL25 | YL50 | YL75 | Quadratic | ||
| Villus height (μm) | 386.38 | 434.22 | 449.18 | 312.86 | 15.92 | 0.002/0.760 |
| Villus width (μm) | 59.83 | 57.12 | 62.03 | 49.65 | 4.70 | n.s |
| Ingredients (g/kg) | Diets | |||
|---|---|---|---|---|
| Con | YL25 | YL50 | YL75 | |
| Fish meal | 600 | 450 | 300 | 150 |
| Fermented soybean meal | 120 | 120 | 120 | 120 |
| Shrimp meal | 50 | 50 | 50 | 50 |
| YL | 0 | 150 | 300 | 450 |
| Fish oil | 50 | 50 | 50 | 50 |
| Wheat flour | 50 | 50 | 50 | 50 |
| Gluten | 40 | 40 | 40 | 40 |
| Soluble starch | 40 | 40 | 40 | 40 |
| Microcrystalline fiber | 10 | 10 | 10 | 10 |
| Ca(H2PO4)2 | 10 | 10 | 10 | 10 |
| Mineral premix 2 | 12 | 12 | 12 | 12 |
| Vitamin premix 3 | 10 | 10 | 10 | 10 |
| Choline chloride | 3 | 3 | 3 | 3 |
| L-lysine | 3 | 3 | 3 | 3 |
| L-methionine | 2 | 2 | 2 | 2 |
| Proximate composition (%) | ||||
| Dry matter (%) | 93.42 | 93.75 | 93.93 | 94.06 |
| Crude protein (%) | 50.34 | 50.07 | 49.83 | 49.75 |
| Crude lipid (%) | 9.42 | 9.51 | 9.56 | 9.63 |
| Ash (%) | 10.45 | 10.57 | 10.61 | 10.65 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fei, H.; Cheng, Y.; Zhang, H.; Yu, X.; Yi, S.; Huang, M.; Yang, S. Effect of Autolyzed Yarrowia lipolytica on the Growth Performance, Antioxidant Capacity, Intestinal Histology, Microbiota, and Transcriptome Profile of Juvenile Largemouth Bass (Micropterus salmoides). Int. J. Mol. Sci. 2022, 23, 10780. https://doi.org/10.3390/ijms231810780
Fei H, Cheng Y, Zhang H, Yu X, Yi S, Huang M, Yang S. Effect of Autolyzed Yarrowia lipolytica on the Growth Performance, Antioxidant Capacity, Intestinal Histology, Microbiota, and Transcriptome Profile of Juvenile Largemouth Bass (Micropterus salmoides). International Journal of Molecular Sciences. 2022; 23(18):10780. https://doi.org/10.3390/ijms231810780
Chicago/Turabian StyleFei, Hui, Yan Cheng, Huimin Zhang, Xiang Yu, Shunfa Yi, Mengmeng Huang, and Shun Yang. 2022. "Effect of Autolyzed Yarrowia lipolytica on the Growth Performance, Antioxidant Capacity, Intestinal Histology, Microbiota, and Transcriptome Profile of Juvenile Largemouth Bass (Micropterus salmoides)" International Journal of Molecular Sciences 23, no. 18: 10780. https://doi.org/10.3390/ijms231810780
APA StyleFei, H., Cheng, Y., Zhang, H., Yu, X., Yi, S., Huang, M., & Yang, S. (2022). Effect of Autolyzed Yarrowia lipolytica on the Growth Performance, Antioxidant Capacity, Intestinal Histology, Microbiota, and Transcriptome Profile of Juvenile Largemouth Bass (Micropterus salmoides). International Journal of Molecular Sciences, 23(18), 10780. https://doi.org/10.3390/ijms231810780





