Molecular and Cellular Mechanisms of Propolis and Its Polyphenolic Compounds against Cancer
Abstract
1. Introduction
2. Propolis
3. Molecular and Cellular Targets of Propolis and Its Flavonoids in Cancer Chemoprevention
3.1. Oxidative Stress and Tumor Development: Role of Propolis and Its Polyphenolic/Flavonoid Components
3.1.1. Antioxidative Activity of Propolis and Its Polyphenolic Compounds
3.1.2. Prooxidative Activity of Propolis and Its Polyphenolic Compounds
3.2. Regulation of Cell Proliferation by Propolis and Its Polyphenolic/Flavonoid Compounds
Key Mechanisms of Propolis and Its Polyphenolic/Flavonoid Compounds Involved in Inhibition of Cell Proliferation
3.3. Epigenetic Regulation by Propolis and Its Polyphenolic/Flavonoid Compounds
3.3.1. Propolis and Its Flavonoids as Inhibitors of Methyltransferase and Histone Deacetylase
3.3.2. Propolis and Its Flavonoids Modulate miRNAs
3.4. Regulation of Cell Death by Propolis and Its Polyphenolic/Flavonoid Compounds
3.4.1. Modulation of Apoptosis Pathway by Propolis and Its Polyphenolic/Flavonoid Compounds
3.4.2. Propolis and Its Polyphenolic/Flavonoid Compounds Reduce Expression of Autophagy-Related Proteins in Cancer
3.5. Regulation of Inflammatory Pathways by Propolis and Its Polyphenolic/Flavonoid Compounds
Propolis and Functional Activation of Macrophages in Cancer
4. Antitumor Activity of Propolis and Its Polyphenolic/Flavonoid Compounds In Vivo
4.1. Immunoregulation by Propolis and Its Polyphenolic/Flavonoid Compounds as Important Effector Mechanism of Antitumor Activity In Vivo
4.1.1. Role of Propolis and Its Flavonoids in Macrophage Activation and Polarization
4.1.2. Propolis, TLRs, and Immune System
4.1.3. The Dual Role of TLRs Activation with Propolis and Its Components in Tumor Inhibition and Growth
4.1.4. Role of Propolis and Its Polyphenolic/Flavonoid Compounds on Chronic Viral or Bacterial Infections
4.1.5. Associations between Flavonoid Intake, Gut Microbiota, and Cancer
4.2. Antiangiogenic Effect of Propolis and Its Polyphenolic/Flavonoid Compounds
4.3. Antimetastatic Effect of Propolis and Its Polyphenolic/Flavonoid Compounds
5. Propolis and Its Polyphenolic/Flavonoid Compounds in the Regulation of Glucose Metabolism in Tumor
6. Telomerase-Related Anticancer Strategies by Propolis and Its Polyphenolic/Flavonoid Components
7. Present Status and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Kunnumakkara, A.B.; Bordoloi, D.; Harsha, C.; Banik, K.; Gupta, S.C.; Aggarwal, B.B. Curcumin mediates anticancer effects by modulating multiple cell signaling pathways. Clin. Sci. 2017, 131, 1781–1799. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Oršolić, N.; Bašić, I. Cancer chemoprevention by propolis and its polyphenolic compounds in experimental animals. In Recent Progress in Medicinal Plants; Singh, V.K., Govil, J.N., Arunachalam, C., Eds.; Studium Press LLC.: Houston, TX, USA, 2007; pp. 55–113. [Google Scholar]
- Aggarwal, B.B.; Van Kuiken, M.E.; Iyer, L.H.; Harikumar, K.B.; Sung, B. Molecular targets of nutraceuticals derived from dietary spices: Potential role in suppression of inflammation and tumorigenesis. Exp. Bio. Med. 2009, 234, 825–849. [Google Scholar] [CrossRef] [PubMed]
- Kasote, D.; Bankova, V.; Viljoen, A.M. Propolis: Chemical diversity and challenges in quality control. Phytochem. Rev. 2022, 24, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Sena-Lopes, Â.; Bezerra, F.S.B.; das Neves, R.N.; de Pinho, R.B.; Silva, M.T.O.; Savegnago, L.; Collares, T.; Seixas, F.; Begnini, K.; Henriques, J.A.P.; et al. Chemical composition, immunostimulatory, cytotoxic and antiparasitic activities of the essential oil from Brazilian red propolis. PLoS ONE 2018, 13, e0191797. [Google Scholar] [CrossRef]
- Forma, E.; Bryś, M. Anticancer Activity of Propolis and Its Compounds. Nutrients 2021, 13, 2594. [Google Scholar] [CrossRef]
- Popova, M.; Giannopoulou, E.; Skalicka-Woźniak, K.; Graikou, K.; Widelski, J.; Bankova, V.; Kalofonos, H.; Sivolapenko, G.; Gaweł-Bęben, K.; Antosiewicz, B.; et al. Characterization and Biological Evaluation of Propolis from Poland. Molecules 2017, 22, 1159. [Google Scholar] [CrossRef]
- Chiu, H.F.; Han, Y.C.; Shen, Y.C.; Golovinskaia, O.; Venkatakrishnan, K.; Wang, C.K. Chemopreventive and Chemotherapeutic Effect of Propolis and Its Constituents: A Mini-review. J. Cancer Prev. 2020, 25, 70–78. [Google Scholar] [CrossRef]
- Zabaiou, N.; Fouache, A.; Trousson, A.; Baron, S.; Zellagui, A.; Lahouel, M.; Lobaccaro, J.A. Biological properties of propolis extracts: Something new from an ancient product. Chem. Phys. Lipids 2017, 207 Pt B, 214–222. [Google Scholar] [CrossRef]
- Kuo, Y.-Y.; Jim, W.-T.; Su, L.-C.; Chung, C.-J.; Lin, C.-Y.; Huo, C.; Tseng, J.-C.; Huang, S.-H.; Lai, C.-J.; Chen, B.-C.; et al. Caffeic Acid Phenethyl Ester Is a Potential Therapeutic Agent for Oral Cancer. Int. J. Mol. Sci. 2015, 16, 10748–10766. [Google Scholar] [CrossRef]
- Patel, S. Emerging Adjuvant Therapy for Cancer: Propolis and its Constituents. J. Diet. Suppl. 2016, 13, 245–268. [Google Scholar] [CrossRef] [PubMed]
- Oršolić, N. A review of propolis antitumor action in vivo and in vitro. JAAS 2010, 2, 1–20. [Google Scholar]
- Oršolić, N.; Karač, I.; Sirovina, D.; Kukolj, M.; Kunštić, M.; Gajski, G.; Garaj-Vrhovac, V.; Štajcar, D. Chemotherapeutic potential of quercetin on human bladder cancer cells. J. Environ. Sci. Health A 2016, 51, 776–781. [Google Scholar] [CrossRef] [PubMed]
- Oršolić, N.; Brbot Šaranović, A.; Bašić, I. Direct and indirect mechanism(s) of antitumor activity of propolis and its polyphenolic compounds. Planta Med. 2006, 72, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Oršolić, N.; Horvat Knežević, A.; Šver, L.; Terzić, S.; Bašić, I. Immunomodulatory and antimetastatic action of propolis and related poyphenolic compounds. J. Ethnopharmacol. 2004, 94, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Chan, G.C.; Cheung, K.W.; Sze, D.M. The immunomodulatory and anticancer properties of propolis. Clin. Rev. Allergy Immunol. 2013, 44, 262–273. [Google Scholar] [CrossRef]
- Botteon, C.E.A.; Silva, L.B.; Ccana-Ccapatinta, G.V.; Silva, T.S.; Ambrosio, S.R.; Veneziani, R.C.S.; Bastos, J.K.; Marcato, P.D. Biosynthesis and characterization of gold nanoparticles using Brazilian red propolis and evaluation of its antimicrobial and anticancer activities. Sci. Rep. 2021, 11, 1974. [Google Scholar] [CrossRef]
- El-Seedi, H.R.; Eid, N.; Abd El-Wahed, A.A.; Rateb, M.E.; Afifi, H.S.; Algethami, A.F.; Zhao, C.; Al Naggar, Y.; Alsharif, S.M.; Tahir, H.E.; et al. Honey Bee Products: Preclinical and Clinical Studies of Their Anti-inflammatory and Immunomodulatory Properties. Front. Nutr. 2022, 8, 761267. [Google Scholar] [CrossRef]
- Nani, B.D.; Franchin, M.; Lazarini, J.G.; Freires, I.A.; da Cunha, M.G.; Bueno-Silva, B.; de Alencar, S.M.; Murata, R.M.; Rosalen, P.L. Isoflavonoids from Brazilian red propolis down-regulate the expression of cancer-related target proteins: A pharmacogenomic analysis. Phytother. Res. 2018, 32, 750–754. [Google Scholar] [CrossRef]
- Oršolić, N.; Bašić, I. Immunomodulation by water-soluble derivative of propolis (WSDP) a factor of antitumor reactivity. J. Ethnopharmacol. 2003, 84, 265–273. [Google Scholar] [CrossRef]
- Orsi, R.O.; Funari, S.R.C.; Soares, A.M.V.C.; Calvi, S.A.; Oliveira, S.L.; Sforcin, J.M.; Bankova, V. Immunomodulatory action of propolis on macrophage activation. J. Venom. Anim. Toxins 2000, 6, 205–219. [Google Scholar] [CrossRef]
- Oršolić, N.; Bašić, I. Water soluble derivative of propolis and its polyphenolic compounds enhance tumoricidal activity of macrophages. J. Ethnopharmacol. 2005, 102, 37–45. [Google Scholar] [CrossRef]
- Sforcin, J.M. Propolis and the immune system: A review. J. Ethnopharmacol. 2007, 113, 1–14. [Google Scholar] [CrossRef]
- Cardoso, E.O.; Conti, B.J.; Santiago, K.B.; Conte, F.L.; Oliveira, L.P.; Hernandes, R.T.; Golim, M.A.; Sforcin, J.M. Phenolic compounds alone or in combination may be involved in propolis effects on human monocytes. J. Pharm. Pharmacol. 2017, 69, 99–108. [Google Scholar] [CrossRef]
- Oršolić, N.; Car, N.; Lisičić, D.; Benković, V.; Knežević, A.H.; Dikić, D.; Petrik, J. Synergism between propolis and hyperthermal intraperitoneal chemotherapy with cisplatin on Ehrlich ascites tumor in mice. J. Pharm. Sci. 2013, 102, 4395–4405. [Google Scholar] [CrossRef]
- Conti, B.J.; Santiago, K.B.; Cardoso, E.O.; Freire, P.P.; Carvalho, R.F.; Golim, M.A.; Sforcin, J.M. Propolis modulates miRNAs involved in TLR-4 pathway, NF-κB activation, cytokine production and in the bactericidal activity of human dendritic cells. J. Pharm. Pharmacol. 2016, 68, 1604–1612. [Google Scholar] [CrossRef]
- Oršolić, N.; Bašić, I. Antitumor, hematostimulative and radioprotective action of water-soluble derivative of propolis (WSDP). Biomed. Pharmacother. 2005, 59, 561–570. [Google Scholar] [CrossRef]
- Montoro, A.; Barquinero, J.F.; Almonacid, M.; Montoro, A.; Sebastià, N.; Verdú, G.; Sahuquillo, V.; Serrano, J.; Saiz, M.; Villaescusa, J.I.; et al. Concentration-Dependent Protection by Ethanol Extract of Propolis against γ-Ray-Induced Chromosome Damage in Human Blood Lymphocytes. Evid. Based Complement. Alternat. Med. 2011, 2011, 174853. [Google Scholar] [CrossRef]
- Patil, S.L.; Rao, N.B.; Somashekarappa, H.M.; Rajashekhar, K.P. Antigenotoxic potential of rutin and quercetin in Swiss mice exposed to gamma radiation. Biomed. J. 2014, 37, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Benkovic, V.; Knezevic, A.H.; Dikic, D.; Lisicic, D.; Orsolic, N.; Basic, I.; Kosalec, I.; Kopjar, N. Radioprotective effects of propolis and quercetin in gamma-irradiated mice evaluated by the alkaline comet assay. Phytomedicine 2008, 15, 851–858. [Google Scholar] [CrossRef]
- Orsolić, N.; Benković, V.; Horvat-Knezević, A.; Kopjar, N.; Kosalec, I.; Bakmaz, M.; Mihaljević, Z.; Bendelja, K.; Basić, I. Assessment by survival analysis of the radioprotective properties of propolis and its polyphenolic compounds. Biol. Pharm. Bull. 2007, 30, 946–951. [Google Scholar] [CrossRef] [PubMed]
- Altay, H.; Demir, E.; Binici, H.; Aytac, İ.; Taysi, M.E.; Taysi, S. Radioprotective Effects of Propolis and Caffeic acid Phenethyl Ester on the Tongue-Tissues of Total-Head Irradiated Rats. Eur. J. Ther. 2020, 26, 202–207. [Google Scholar] [CrossRef]
- Anjaly, K.; Tiku, A.B. Caffeic acid phenethyl ester induces radiosensitization via inhibition of DNA damage repair in androgen-independent prostate cancer cells. Environ. Toxicol. 2022, 37, 995–1006. [Google Scholar] [CrossRef]
- Oršolić, N.; Benković, V.; Horvat-Knežević, A.; Bašić, I. Propolis and related flavonoids as radioprotective agents. In Herbal Radiomodulators: Applications in Medicine, Homeland Defence and Space; Sharma, R.K., Arora, R., Eds.; CABI Publishing: Wallingford, UK, 2007; pp. 175–194. [Google Scholar]
- Oryan, A.; Alemzadeh, E.; Moshiri, A. Potential role of propolis in wound healing: Biological properties and therapeutic activities. Biomed. Pharmacother. 2018, 98, 469–483. [Google Scholar] [CrossRef]
- Pasupuleti, V.R.; Sammugam, L.; Ramesh, N.; Gan, S.H. Honey, Propolis, and Royal Jelly: A Comprehensive Review of Their Biological Actions and Health Benefits. Oxid Med. Cell Longev. 2017, 2017, 1259510. [Google Scholar] [CrossRef]
- Oršolić, N.; Bašić, I. Polyphenols from propolis and plants in control of allergy and inflammation. In Scientific Evidence of the Use of Propolis in Ethnomedicine; Oršolić, N., Bašić, I., Eds.; Ethnopharmacology-Review Book; Transworld Research Network: Trivandrum, India, 2008; pp. 311–336. [Google Scholar]
- Kosalec, I.; Sanković, K.; Zovko, M.; Oršolić, N.; Bakmaz, M.; Kalođera, Z.; Pepeljnjak, S. Antimicrobial and Antioxidant Activity of Propolis from Croatia and Brazil: A Comparative Study. In Scientific Evidence of the Use of Propolis in Ethnomedicine; Oršolić, N., Bašić, I., Eds.; Ethnopharmacology-Review Book; Transworld Research Network: Trivandrum, India, 2008; pp. 175–194. [Google Scholar]
- Sirovina, D.; Oršolić, N.; Zovko Končić, M.; Kovačević, G.; Benković, V.; Gregorović, G. Quercetin vs chrysin: Effect on liver histopathology in diabetic mice. Hum. Exp. Toxicol. 2013, 2, 1058–1066. [Google Scholar] [CrossRef]
- Oršolić, N.; Sirovina, D.; Gajski, G.; Garaj-Vrhovac, V.; Jazvinšćak Jembrek, M.; Kosalec, I. Assessment of DNA damage and lipid peroxidation in diabetic mice: Effects of propolis and epigallocatechin gallate (EGCG). Mutat. Res. 2013, 757, 36–44. [Google Scholar] [CrossRef]
- Cao, X.P.; Chen, Y.F.; Zhang, J.L.; You, M.M.; Wang, K.; Hu, F.L. Mechanisms underlying the wound healing potential of propolis based on its in vitro antioxidant activity. Phytomedicine 2017, 34, 76–84. [Google Scholar] [CrossRef]
- Oršolić, N.; Skurić, J.; Đikić, Đ.; Stanić, G. Inhibitory effect of a propolis on Di-n-Propyl Disulfide or n-Hexyl salicilate-induced skin irritation, oxidative stress and inflammatory responses in mice. Fitoterapia 2014, 93, 18–30. [Google Scholar] [CrossRef]
- Oršolić, N. Bee venom in cancer therapy. Cancer Metastasis Rev. 2012, 31, 173–194. [Google Scholar] [CrossRef]
- Jazvinšćak Jembrek, M.; Vuković, L.; Puhović, J.; Erhardt, J.; Oršolić, N. Neuroprotective Effect of Quercetin against Hydrogen Peroxide-induced Oxidative Injury in P19 Neurons. J. Mol. Neurosci. 2012, 47, 286–299. [Google Scholar] [CrossRef] [PubMed]
- Oršolić, N.; Terzić, S.; Šver, L.; Bašić, I. Honey-bee products in prevention and/or terapy of murine translantable tumors. J. Sci. Food Agric. 2005, 85, 363–370. [Google Scholar] [CrossRef]
- Celińska-Janowicz, K.; Zaręba, I.; Lazarek, U.; Teul, J.; Tomczyk, M.; Pałka, J.; Miltyk, W. Constituents of Propolis: Chrysin, Caffeic Acid, p-Coumaric Acid, and Ferulic Acid Induce PRODH/POX-Dependent Apoptosis in Human Tongue Squamous Cell Carcinoma Cell (CAL-27). Front. Pharmacol. 2018, 9, 336. [Google Scholar] [CrossRef]
- Galati, G.; O’Brien, P.J. Potential toxicity of flavonoids and other dietary phenolics: Significance for their chemopreventive and anticancer properties. Serial Review: Flavonoids and Isoflavones (Phytoestrogens): Absorbtion, Metabolism, and Bioactivity. Free Radic. Biol. Med. 2004, 37, 287–303. [Google Scholar] [CrossRef]
- Kabała-Dzik, A.; Rzepecka-Stojko, A.; Kubina, R.; Jastrzębska-Stojko, Ż.; Stojko, R.; Wojtyczka, R.D.; Stojko, J. Comparison of Two Components of Propolis: Caffeic Acid (CA) and Caffeic Acid Phenethyl Ester (CAPE) Induce Apoptosis and Cell Cycle Arrest of Breast Cancer Cells MDA-MB-231. Molecules 2017, 22, 1554. [Google Scholar] [CrossRef]
- Tao, L.; Chen, X.; Zheng, Y.; Wu, Y.; Jiang, X.; You, M.; Li, S.; Hu, F. Chinese Propolis Suppressed Pancreatic Cancer Panc-1 Cells Proliferation and Migration via Hippo-YAP Pathway. Molecules 2021, 26, 2803. [Google Scholar] [CrossRef]
- Huang, S.; Zhang, C.P.; Wang, K.; Li, G.Q.; Hu, F.L. Recent advances in the chemical composition of propolis. Molecules 2014, 19, 19610–19632. [Google Scholar] [CrossRef]
- Salatino, A.; Fernandes-Silva, C.C.; Righi, A.A.; Salatino, M.L.F. Propolis research and the chemistry of plant products. Nat. Prod. Rep. 2011, 28, 925–936. [Google Scholar] [CrossRef]
- Toreti, V.C.; Sato, H.H.; Pastore, G.M.; Park, Y.K. Recent progress of propolis for its biological and chemical compositions and its botanical origin. Evid. Based Complement. Alternat. Med. 2013, 2013, 697390. [Google Scholar] [CrossRef]
- Wang, K.; Ping, S.; Huang, S.; Hu, L.; Xuan, H.Z.; Zhang, C.P.; Hu, F.L. Molecular mechanisms underlying the in vitro anti-inflammatory effects of a favonoid-rich ethanol extract from Chinese propolis (poplar type). Evid. Based Complement. Alternat. Med. 2013, 2013, 127672. [Google Scholar] [CrossRef]
- Ribeiro, V.P.; Ccana-Ccapatinta, G.V.; Aldana-Mejía, J.A.; Berretta, A.A.; Moraes, L.A.; Bastos, J.K. Chemical characterization of Brazilian propolis using automated direct thermal desorption-gas chromatography-mass spectrometry. J Sci. Food Agric. 2022, 102, 4345–4354. [Google Scholar] [CrossRef]
- Conti, B.J.; Santiago, K.B.; Búfalo, M.C.; Herrera, Y.F.; Alday, E.; Velazquez, C.; Hernandez, J.; Sforcin, J.M. Modulatory effects of propolis samples from Latin America (Brazil, Cuba and Mexico) on cytokine production by human monocytes. J. Pharm. Pharmacol. 2015, 67, 1431–1438. [Google Scholar] [CrossRef] [PubMed]
- Martinotti, S.; Ranzato, E. Propolis: A new frontier for wound healing? Burns Trauma 2015, 3, 9. [Google Scholar] [CrossRef]
- Oršolić, N.; Bendelja, K.; Brbot–Šaranović, A.; Bašić, I. Effects of caffeic acid phenethyl ester and caffeic acid, antioxidants from propolis, on inducing apoptosis in HeLa human cervical carcinoma and Chinese hamster lung V79 fibroblast cells. Period. Biol. 2004, 106, 367–372. [Google Scholar]
- Oršolić, N.; Šver, L.; Terzić, S.; Tadić, Z.; Bašić, I. Inhibitory effect of water-soluble derivative of propolis and its polyphenolic compounds on tumor growth and metastasing ability: A possible mode of antitumor action. Nutr. Cancer 2003, 47, 156–163. [Google Scholar] [CrossRef]
- Oršolić, N.; Šver, L.; Terzić, S.; Bašić, I. Peroral aplication of water-soluble derivative of propolis (WSDP) and its related polyphenolic compounds and their influence on immunological and antitumor activity. Vet. Res. Commun. 2005, 29, 575–593. [Google Scholar] [CrossRef]
- Oršolić, N.; Terzić, S.; Mihaljević, Ž.; Šver, L.; Bašić, I. Effect of local administration of propolis and its polyphenolic compounds on the tumour formation and growth. Biol. Pharm. Bull. 2005, 28, 1928–1933. [Google Scholar] [CrossRef]
- Oršolić, N.; Terzić, S.; Šver, L.; Bašić, I. Polyphenolic compounds from propolis modulate immune responses and increase host resistance to tumor cells. Food Agric. Immunol. 2005, 16, 165–179. [Google Scholar] [CrossRef]
- Oršolić, N.; Jazvinšćak Jembrek, M. Multimodal approach to tumor therapy with quercetin, chemotherapy, radiotherapy and hyperthermia. In Quercetin: Food Sources, Antioxidant Properties and Health Effects; Gregory Malone, G., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2015; pp. 43–84. [Google Scholar]
- Cogulu, O.; Biray, C.; Gunduz, C.; Karaca, E.; Aksoylar, S.; Sorkun, K.; Salih, B.; Ozkinay, F. Effects of Manisa propolis on telomerase activity in leukemia cells obtained from the bone marrow of leukemia patients. Int. J. Food Sci. Nutr. 2009, 60, 601–605. [Google Scholar] [CrossRef]
- Avci, C.B.; Sahin, F.; Gunduz, C.; Selvi, N.; Aydin, H.H.; Oktem, G.; Topcuoglu, N.; Saydam, G. Protein phosphatase 2A (PP2A) has a potential role in CAPE-induced apoptosis of CCRF-CEM cells via effecting human telomerase reverse transcriptase activity. Hematology 2007, 126, 519–525. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, S.; Wang, K.; Lu, J.; Bao, X.; Wang, R.; Qiu, Y.; Wang, T.; Yu, H. Cellular senescence and cancer: Focusing on traditional Chinese medicine and natural products. Cell Prolif. 2020, 53, e12894. [Google Scholar] [CrossRef]
- Kashafi, E.; Moradzadeh, M.; Mohamadkhani, A.; Erfanian, S. Kaempferol increases apoptosis in human cervical cancer HeLa cells via PI3K/AKT and telomerase pathways. Biomed. Pharmacother. 2017, 89, 573–577. [Google Scholar] [CrossRef]
- Platella, C.; Guida, S.; Bonmassar, L.; Aquino, A.; Bonmassar, E.; Ravagnan, G.; Montesarchio, D.; Roviello, G.N.; Musumeci, D.; Fuggetta, M.P. Antitumour activity of resveratrol on human melanoma cells: A possible mechanism related to its interaction with malignant cell telomerase. Biochim. Biophys. Acta 2017, 1861 Pt A, 2843–2851. [Google Scholar] [CrossRef]
- Ganesan, K.; Xu, B. Telomerase Inhibitors from Natural Products and Their Anticancer Potential. Int. J. Mol. Sci. 2018, 19, 13. [Google Scholar] [CrossRef]
- Radovanović, V.; Vlainić, J.; Hanžić, N.; Ukić, P.; Oršolić, N.; Baranović, G.; Jazvinšćak Jembrek, M. Neurotoxic Effect of Ethanolic Extract of Propolis in the Presence of Copper Ions is Mediated through Enhanced Production of ROS and Stimulation of Caspase-3/7 Activity. Toxins 2019, 11, 273. [Google Scholar] [CrossRef]
- Sadžak, A.; Vlašić, I.; Kiralj, Z.; Batarelo, M.; Oršolić, N.; Jazvinšćak Jembrek, M.; Kušen, I.; Šegota, S. Neurotoxic Effect of Flavonol Myricetin in the Presence of Excess Copper. Molecules 2021, 26, 845. [Google Scholar] [CrossRef]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef]
- Oršolić, N.; Kunštić, M.; Kukolj, M.; Gračan, R.; Nemrava, J. Oxidative stress, polarization of macrophages and tumour angiogenesis: Efficacy of caffeic acid. Chem. Biol. Interact. 2016, 256, 111–124. [Google Scholar] [CrossRef]
- Actis-Goretta, L.; Ottaviani, J.I.; Keen, C.L.; Fraga, C.G. Inhibition of angiotensin converting enzyme (ACE) activity by flavan-3-ols and procyanidins. FEBS Lett. 2003, 555, 597–600. [Google Scholar] [CrossRef]
- Marchetti, F.; De Santi, C.; Vietri, M.; Pietrabissa, A.; Spisni, R.; Mosca, F.; Pacifici, G.M. Differential inhibition of human liver and duodenum sulphotransferase activities by quercetin, a flavonoid present in vegetables, fruit and wine. Xenobiotica 2001, 31, 841–847. [Google Scholar] [CrossRef]
- Giovannini, L.; Bianchi, S. Role of nutraceutical SIRT1 modulators in AMPK and mTOR pathway: Evidence of a synergistic effect. Nutrition 2017, 34, 82–96. [Google Scholar] [CrossRef] [PubMed]
- Havsteen, B.H. The biochemistry and medical significance of the flavonoids. Pharmacol. Ther. 2002, 96, 67–202. [Google Scholar] [CrossRef]
- Oršolić, N.; Bevanda, M.; Bendelja, K.; Horvat-Knežević, A.; Benković, V.; Bašić, I. Propolis and related polyphenolic compounds; their relevance on host resistance and interaction with chemotherapy. In Scientific Evidence of the Use of Propolis in Ethnomedicine; Oršolić, N., Bašić, I., Eds.; Ethnopharmacology-Review Book; Transworld Research Network: Trivandrum, India, 2008; pp. 223–250. [Google Scholar]
- Albassam, A.A.; Markowitz, J.S. An Appraisal of Drug-Drug Interactions with Green Tea (Camellia sinensis). Planta Med. 2017, 83, 496–508. [Google Scholar] [CrossRef]
- Wang, G.; Wang, J.J.; Guan, R.; Du, L.; Gao, J.; Fu, X.L. Strategies to Target Glucose Metabolism in Tumor Microenvironment on Cancer by Flavonoids. Nutr. Cancer 2017, 69, 534–554. [Google Scholar] [CrossRef]
- Blaschek, W. Natural Products as Lead Compounds for Sodium Glucose Cotransporter (SGLT) Inhibitors. Planta Med. 2017, 83, 985–993. [Google Scholar] [CrossRef]
- León, D.; Uribe, E.; Zambrano, A.; Salas, M. Implications of Resveratrol on Glucose Uptake and Metabolism. Molecules 2017, 22, 398. [Google Scholar] [CrossRef]
- Faggio, C.; Sureda, A.; Morabito, S.; Sanches-Silva, A.; Mocan, A.; Nabavi, S.F.; Nabavi, S.M. Flavonoids and platelet aggregation: A brief review. Eur. J. Pharmacol. 2017, 807, 91–101. [Google Scholar] [CrossRef]
- Ziaei, S.; Halaby, R. Dietary Isoflavones and Breast Cancer Risk. Medicines 2017, 4, 18. [Google Scholar] [CrossRef]
- Qadir, M.I.; Cheema, B.N. Phytoestrogens and Related Food Components in the Prevention of Cancer. Crit. Rev. Eukaryot. Gene Expr. 2017, 27, 99–112. [Google Scholar] [CrossRef]
- Alamolhodaei, N.S.; Tsatsakis, A.M.; Ramezani, M.; Hayes, A.W.; Karimi, G. Resveratrol as MDR reversion molecule in breast cancer: An overview. Food Chem. Toxicol. 2017, 103, 223–232. [Google Scholar] [CrossRef]
- Goszcz, K.; Duthie, G.G.; Stewart, D.; Leslie, S.J.; Megson, I.L. Bioactive polyphenols and cardiovascular disease: Chemical antagonists, pharmacological agents or xenobiotics that drive an adaptive response? Br. J. Pharmacol. 2017, 174, 1209–1225. [Google Scholar] [CrossRef]
- Xue, Z.; Li, D.; Yu, W.; Zhang, Q.; Hou, X.; He, Y.; Kou, X. Mechanisms and therapeutic prospects of polyphenols as modulators of the aryl hydrocarbon receptor. Food Funct. 2017, 8, 1414–1437. [Google Scholar] [CrossRef] [PubMed]
- Balica, G.; Vostinaru, O.; Stefanescu, C.; Mogosan, C.; Iaru, I.; Cristina, A.; Pop, C.E. Potential Role of Propolis in the Prevention and Treatment of Metabolic Diseases. Plants 2021, 10, 883. [Google Scholar] [CrossRef] [PubMed]
- Sies, H.; Belousov, V.V.; Chandel, N.S.; Davies, M.J.; Jones, D.P.; Mann, G.E.; Murphy, M.P.; Yamamoto, M.; Winterbourn, C. Defining roles of specific reactive oxygen species (ROS) in cell biology and physiology. Nat. Rev. Mol. Cell Biol. 2022, 23, 499–515. [Google Scholar] [CrossRef]
- Bekhet, O.H.; Eid, M.E. The interplay between reactive oxygen species and antioxidants in cancer progression and therapy: A narrative review. Transl. Cancer Res. 2021, 10, 4196–4206. [Google Scholar] [CrossRef] [PubMed]
- Soh, R.; Hardy, A.; Zur Nieden, N.I. The FOXO signaling axis displays conjoined functions in redox homeostasis and stemness. Free Radic. Biol. Med. 2021, 169, 224–237. [Google Scholar] [CrossRef]
- Han, Y.; Liu, D.; Li, L. PD-1/PD-L1 pathway: Current researches in cancer. Am. J. Cancer Res. 2020, 10, 727–742. [Google Scholar]
- Jia, Y.; Jiang, S.; Chen, C.; Lu, G.; Xie, Y.; Sun, X.; Huang, L. Caffeic acid phenethyl ester attenuates nuclear factor κB mediated inflammatory responses in Müller cells and protects against retinal ganglion cell death. Mol. Med. Rep. 2019, 19, 4863–4871. [Google Scholar] [CrossRef]
- Li, X.; Hao, S.; Han, A.; Yang, Y.; Fang, G.; Liu, J.; Wang, S. Intracellular Fenton Reaction based on Mitochondria-Targeted Copper(ΙΙ)-Peptide Complex for Induced Apoptosis. J. Mater. Chem. B 2019, 7, 4008–4016. [Google Scholar] [CrossRef]
- da Silva, W.M.B.; de Oliveira Pinheiro, S.; Alves, D.R.; de Menezes, J.E.S.A.; Magalhães, F.E.A.; Silva, F.C.O.; Silva, J.; Marinho, E.S.; de Morais, S.M. Synthesis of Quercetin-Metal Complexes, In Vitro and In Silico Anticholinesterase and Antioxidant Evaluation, and In Vivo Toxicological and Anxiolitic Activities. Neurotox. Res. 2020, 37, 893–903. [Google Scholar] [CrossRef]
- Liu, Y.; Guo, M. Studies on transition metal-quercetin complexes using electrospray ionization tandem mass spectrometry. Molecules 2015, 20, 8583–8594. [Google Scholar] [CrossRef]
- Ančić, D.; Oršolić, N.; Odeh, D.; Tomašević, M.; Pepić, I.; Ramić, S. Resveratrol and its nanocrystals: A promising approach for cancer therapy? Toxicol. Appl. Pharmacol. 2022, 435, 115851. [Google Scholar] [CrossRef] [PubMed]
- Duca, A.; Sturza, A.; Moacă, E.-A.; Negrea, M.; Lalescu, V.-D.; Lungeanu, D.; Dehelean, C.-A.; Muntean, D.-M.; Alexa, E. Identification of Resveratrol as Bioactive Compound of Propolis from Western Romania and Characterization of Phenolic Profile and Antioxidant Activity of Ethanolic Extracts. Molecules 2019, 24, 3368. [Google Scholar] [CrossRef] [PubMed]
- Shaito, A.; Posadino, A.M.; Younes, N.; Hasan, H.; Halabi, S.; Alhababi, D.; Al-Mohannadi, A.; Abdel-Rahman, W.M.; Eid, A.H.; Nasrallah, G.K.; et al. Potential Adverse Effects of Resveratrol: A Literature Review. Int. J. Mol. Sci. 2020, 21, 2084. [Google Scholar] [CrossRef]
- Plauth, A.; Geikowski, A.; Cichon, S.; Wowro, S.J.; Liedgens, L.; Rousseau, M.; Weidner, C.; Fuhr, L.; Kliem, M.; Jenkins, G.; et al. Hormetic shifting of redox environment by pro-oxidative resveratrol protects cells against stress. Free Radic. Biol. Med. 2016, 99, 608–622. [Google Scholar] [CrossRef]
- Agarwal, K.; Sharma, A.; Talukder, G. Effects of copper on mammalian cell components. Chem. Biol. Interact. 1989, 69, 1–16. [Google Scholar] [CrossRef]
- Aso, K.; Kanno, S.; Tadano, T.; Satoh, S.; Ishikawa, M. Inhibitory effect of propolis on the growth of human leukemia U937. Biol. Pharm. Bull. 2004, 27, 727–730. [Google Scholar] [CrossRef]
- Awale, S.; Li, F.; Onozuka, H.; Esumi, H.; Tezuka, Y.; Kadota, S. Constituents of Brazilian red propolis and their preferential cytotoxic activity against human pancreatic PANC-1 cancer cell line in nutrient-deprived condition. Bioorg. Med. Chem. 2008, 16, 181–189. [Google Scholar] [CrossRef]
- Messerli, S.M.; Ahn, M.R.; Kunimasa, K.; Yanagihara, M.; Tatefuji, T.; Hashimoto, K.; Mautner, V.; Uto, Y.; Hori, H.; Kumazawa, S.; et al. Artepillin C (ARC) in Brazilian green propolis selectively blocks oncogenic PAK1 signaling and suppresses the growth of NF tumors in mice. Phytother. Res. 2009, 23, 423–427. [Google Scholar] [CrossRef]
- Chen, C.N.; Hsiao, C.J.; Lee, S.S.; Guh, J.H.; Chiang, P.C.; Huang, C.C.; Huang, W.J. Chemical modification and anticancer effect of prenylated flavanones from Taiwanese propolis. Nat. Prod. Res. 2012, 26, 116–124. [Google Scholar] [CrossRef]
- Li, J.; Liu, H.; Liu, X.; Hao, S.; Zhang, Z.; Xuan, H. Chinese Poplar Propolis Inhibits MDA-MB-231 Cell Proliferation in an Inflammatory Microenvironment by Targeting Enzymes of the Glycolytic Pathway. J. Immunol. Res. 2021, 2021, 6641341. [Google Scholar] [CrossRef]
- Ha, T.K.; Kim, M.E.; Yoon, J.H.; Bae, S.J.; Yeom, J.; Lee, J.S. Galangin induces human colon cancer cell death via the mitochondrial dysfunction and caspase-dependent pathway. Exp. Biol. Med. 2013, 238, 1047–1054. [Google Scholar] [CrossRef]
- Markiewicz-Żukowska, R.; Borawska, M.H.; Fiedorowicz, A.; Naliwajko, S.K.; Sawicka, D.; Car, H. Propolis changes the anticancer activity of temozolomide in U87MG human glioblastoma cell line. BMC Complement. Altern. Med. 2013, 13, 50. [Google Scholar] [CrossRef]
- Yang, F.; Jin, H.; Pi, J.; Jiang, J.H.; Liu, L.; Bai, H.H.; Yang, P.H.; Cai, J.Y. Anti-tumor activity evaluation of novel chrysin-organogermanium(IV) complex in MCF-7 cells. Bioorg. Med. Chem. Lett. 2013, 23, 5544–5551. [Google Scholar] [CrossRef]
- Abubakar, M.B.; Abdullah, W.Z.; Sulaiman, S.A.; Ang, B.S. Polyphenols as Key Players for the Antileukaemic Effects of Propolis. Evid. Based Complement. Alternat. Med. 2014, 2014, 371730. [Google Scholar] [CrossRef]
- Oršolić, N.; Štajcar, D.; Bašić, I. Propolis and its flavonoid compounds cause cytotoxicity on human urinary bladder transitional cell carcinoma in primary culture. Period. Biol. 2009, 111, 113–121. [Google Scholar]
- Hazafa, A.; Iqbal, M.O.; Javaid, U.; Tareen, M.B.K.; Amna, D.; Ramzan, A.; Piracha, S.; Naeem, M. Inhibitory effect of polyphenols (phenolic acids, lignans, and stilbenes) on cancer by regulating signal transduction pathways: A review. Clin. Transl. Oncol. 2022, 24, 432–445. [Google Scholar] [CrossRef]
- Sherif, M.S.; Rehab, T.A.; Hamdia, Z.A.; Torchilin, V.P. Cytotoxicity of propolis nanopreparations in cancer cell monolayers: Multimode of action including apoptotsis and nitric oxide production. Gen. Physiol. Biophys. 2018, 37, 101–110. [Google Scholar] [CrossRef]
- Chang, H.; Wang, Y.; Yin, X.; Liu, X.; Xuan, H. Ethanol extract of propolis and its constituent caffeic acid phenethyl ester inhibit breast cancer cells proliferation in inflammatory microenvironment by inhibiting TLR4 signal pathway and inducing apoptosis and autophagy. BMC Complement. Altern. Med. 2017, 17, 471. [Google Scholar] [CrossRef]
- Franchi, G.C., Jr.; Moraes, C.S.; Toreti, V.C.; Daugsch, A.; Nowill, A.E.; Park, Y.K. Comparison of effects of the ethanolic extracts of brazilian propolis on human leukemic cells as assessed with the MTT assay. Evid. Based Complement. Alternat. Med. 2012, 2012, 918956. [Google Scholar] [CrossRef]
- Yilmaz, U.C.; Bagca, B.G.; Karaca, E.; Durmaz, A.; Durmaz, B.; Aykut, A.; Kayalar, H.; Avci, C.B.; Susluer, S.Y.; Gunduz, C.; et al. Evaluation of the miRNA profiling and effectiveness of the propolis on B-cell acute lymphoblastic leukemia cell line. Biomed. Pharmacother. 2016, 84, 1266–1273. [Google Scholar] [CrossRef] [PubMed]
- Cotoraci, C.; Ciceu, A.; Sasu, A.; Miutescu, E.; Hermenean, A. The Anti-Leukemic Activity of Natural Compounds. Molecules 2021, 26, 2709. [Google Scholar] [CrossRef] [PubMed]
- Raynal, N.J.; Momparler, L.; Charbonneau, M.; Momparler, R.L. Antileukemic activity of genistein, a major isoflavone present in soy products. J. Nat. Prod. 2008, 71, 3–7. [Google Scholar] [CrossRef]
- Hewitt, A.L.; Singletary, K.W. Soy extract inhibits mammary adenocarcinoma growth in a syngeneic mouse model. Cancer Lett. 2003, 192, 133–143. [Google Scholar] [CrossRef]
- Moiseeva, E.P.; Almeida, G.M.; Jones, G.D.; Manson, M.M. Extended treatment with physiologic concentrations of dietary phytochemicals results in altered gene expression, reduced growth, and apoptosis of cancer cells. Mol. Cancer Ther. 2007, 6, 3071–3079. [Google Scholar] [CrossRef]
- Santell, R.C.; Kieu, N.; Helferich, W.G. Genistein inhibits growth of estrogen-independent human breast cancer cells in culture but not in athymic mice. J. Nutrit. 2000, 130, 1665–1669. [Google Scholar] [CrossRef] [PubMed]
- Linseisen, J.; Piller, R.; Hermann, S.; Chang-Claude, J. Dietary phytoestrogen intake and premenopausal breast cancer risk in a German case-control study. Int. J. Cancer 2004, 110, 284–290. [Google Scholar] [CrossRef]
- He, Y.J.; Li, W.L.; Liu, B.H.; Dong, H.; Mou, Z.R.; Wu, Y.Z. Identification of differential proteins in colorectal cancer cells treated with caffeic acid phenethyl ester. World J. Gastroenterol. 2014, 20, 11840–11849. [Google Scholar] [CrossRef]
- Chuu, C.P.; Lin, H.P.; Ciaccio, M.F.; Kokontis, J.M.; Hause, R.J., Jr.; Hiipakka, R.A.; Liao, S.; Jones, R.B. Caffeic acid phenethyl ester suppresses the proliferation of human prostate cancer cells through inhibition of p70S6K and Akt signaling networks. Cancer Prev. Res. 2012, 5, 788–797. [Google Scholar] [CrossRef]
- Silva-Carvalho, R.; Baltazar, F.; Almeida-Aguiar, C. Propolis: A Complex Natural Product with a Plethora of Biological Activities That Can Be Explored for Drug Development. Evid. Based Complement. Alternat. Med. 2015, 2015, 206439. [Google Scholar] [CrossRef]
- Xiong, H.; Chen, Z.; Lin, B.; Xie, B.; Liu, X.; Chen, C.; Li, Z.; Jia, Y.; Wu, Z.; Yang, M.; et al. Naringenin Regulates FKBP4/NR3C1/NRF2 Axis in Autophagy and Proliferation of Breast Cancer and Differentiation and Maturation of Dendritic Cell. Front. Immunol. 2022, 12, 745111. [Google Scholar] [CrossRef]
- Sánchez, Y.; Amrán, D.; Fernández, C.; De Blas, E.; Aller, P. Genistein selectively potentiates arsenic trioxide-induced apoptosis in human leukemia cells via reactive oxygen species generation and activation of reactive oxygen species-inducible protein kinases (p38-MAPK, AMPK). Int. J. Cancer 2008, 123, 1205–1214. [Google Scholar] [CrossRef]
- Xiang, X.; Hoang, H.D.; Gilchrist, V.H.; Langlois, S.; Alain, T.; Cowan, K.N. Quercetin induces pannexin 1 expression via an alternative transcript with a translationally active 5′ leader in rhabdomyosarcoma. Oncogenesis 2022, 11, 9. [Google Scholar] [CrossRef]
- Ke, D.Y.J.; El-Sahli, S.; Wang, L. The Potential of Natural Products in the Treatment of Triple-Negative Breast Cancer. Curr. Cancer Drug Targets 2022, 22, 388–403. [Google Scholar] [CrossRef]
- Qin, W.; Zhu, W.; Shi, H.; Hewett, J.E.; RuhleN, R.L.; Macdonald, R.S.; Rottinghaus, G.E.; Chen, Y.C.; Sauter, E.R. Soy Isoflavones Have an Antiestrogenic Effect and Alter Mammary Promoter Hypermethylation in Healthy Premenopausal Women. Nutr. Cancer 2009, 61, 238–244. [Google Scholar] [CrossRef]
- Ren, M.; Zhou, X.; Gu, M.; Jiao, W.; Yu, M.; Wang, Y.; Liu, S.; Yang, J.; Ji, F. Resveratrol synergizes with cisplatin in antineoplastic effects against AGS gastric cancer cells by inducing endoplasmic reticulum stress-mediated apoptosis and G2/M phase arrest. Oncol. Rep. 2020, 44, 1605–1615. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhao, X.H.; Wang, Z.J. Cytotoxicity of flavones and flavonols to a human esophageal squamous cell carcinoma cell line (KYSE-510) by induction of G2/M arrest and apoptosis. Toxicol. Vitr. 2009, 23, 797–807. [Google Scholar] [CrossRef]
- Ren, X.; Liu, J.; Hu, L.; Liu, Q.; Wang, D.; Ning, X. Caffeic Acid Phenethyl Ester Inhibits the Proliferation of HEp2 Cells by Regulating Stat3/Plk1 Pathway and Inducing S Phase Arrest. Biol. Pharm. Bull. 2019, 42, 1689–1693. [Google Scholar] [CrossRef]
- Davis, C.D.; Milner, J.A. Biomarkers for diet and cancer prevention research: Potentials and challenges. Acta Pharmacol. Sin. 2007, 28, 1262–1273. [Google Scholar] [CrossRef]
- Rouibah, H.; Kebsa, W.; Lahouel, M.; Zihlif, M.; Ahram, M.; Aburmaileh, B.; Mansour Al Shhab, M.A.; Al-Ameer, H.J.; Mustafa, E. Algerian propolis: Between protection of normal cells and potentialisation of the anticancer effects of doxorubicin against breast cancer cells via P-glycoprotein inhibition and cell cycle arrest in the S phase. J. Physiol. Pharmacol. 2021, 72, 239–248. [Google Scholar] [CrossRef]
- Jiang, X.S.; Xie, H.Q.; Li, C.G.; You, M.M.; Zheng, Y.F.; Li, G.Q.; Chen, X.; Zhang, C.P.; Hu, F.L. Chinese Propolis Inhibits the Proliferation of Human Gastric Cancer Cells by Inducing Apoptosis and Cell Cycle Arrest. Evid. Based Complement. Alternat. Med. 2020, 2020, 2743058. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhao, X.H.; Wang, Z.J. Flavones and flavonols exert cytotoxic effects on a human oesophageal adenocarcinoma cell line (OE33) by causing G2/M arrest and inducing apoptosis. Food Chem. Toxicol. 2008, 46, 2042–2053. [Google Scholar] [CrossRef] [PubMed]
- Ding, M.; Zhao, J.; Bowman, L.; Lu, Y.; Shi, X. Inhibition of AP-1 and MAPK signaling and activation of Nrf2/ARE pathway by quercitrin. Int. J. Oncol. 2010, 36, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Tanigawa, S.; Fujii, M.; Hou, D.X. Action of Nrf2 and Keap1 in ARE-mediated NQO1 expression by quercetin. Free Radic. Biol. Med. 2007, 42, 1690–1703. [Google Scholar] [CrossRef]
- Vargas, A.J.; Burd, R. Hormesis and synergy: Pathways and mechanisms of quercetin in cancer prevention and management. Nutr. Rev. 2010, 68, 418–428. [Google Scholar] [CrossRef]
- Shan, B.E.; Wang, M.X.; Li, R.Q. Quercetin inhibit human SW480 colon cancer growth in association with inhibition of cyclin D1 and survivin expression through Wnt/beta-catenin signaling pathway. Cancer Inv. 2009, 27, 604–612. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef]
- Hwang, K.A.; Choi, K.C. Anticarcinogenic Effects of Dietary Phytoestrogens and Their Chemopreventive Mechanisms. Nutr. Cancer 2015, 67, 796–803. [Google Scholar] [CrossRef]
- Song, Y.S.; Jin, C.; Jung, K.J.; Park, E.H. Estrogenic effects of ethanol and ether extract of propolis. J. Ethnopharmacol. 2002, 82, 89–95. [Google Scholar] [CrossRef]
- Jung, B.; Kim, M.; Kim, H.A.; Kim, D.; Yang, J.; Her, S.; Son, Y.S. Caffeic acid phenethyl ester, a component of beehive propolis is a novel selective estrogen receptor modulator. Phytother. Res. 2010, 24, 295–300. [Google Scholar] [CrossRef]
- Brueggemeier, R.W.; Hackett, J.C.; Diaz-Cruz, E.S. Aromatase inhibitors in the treatment of breast cancer. Endocr. Rev. 2005, 26, 331–345. [Google Scholar] [CrossRef] [PubMed]
- Mocanu, M.M.; Nagy, P.; Szöllősi, J. Chemoprevention of Breast Cancer by Dietary Polyphenols. Molecules 2015, 20, 22578–22620. [Google Scholar] [CrossRef] [PubMed]
- Kao, Y.C.; Zhou, C.; Sherman, M.; Laughton, C.A.; Chen, S. Molecular basis of the inhibition of human aromatase (estrogen synthetase) by flavone and isoflavone phytoestrogens: A site-directed mutagenesis study. Environ. Health Perspect. 1998, 106, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Lephart, E.D. Modulation of Aromatase by Phytoestrogens. Enzyme Res. 2015, 2015, 594656. [Google Scholar] [CrossRef]
- Iannone, M.; Botrè, F.; Cardillo, N.; de la Torre, X. Synthetic isoflavones and doping: A novel class of aromatase inhibitors? Drug Test. Anal. 2019, 11, 208–214. [Google Scholar] [CrossRef]
- Wang, S.D.; Chen, B.C.; Kao, S.T.; Liu, C.J.; Yeh, C.C. Genistein inhibits tumor invasion by suppressing multiple signal transduction pathways in human hepatocellular carcinoma cells. BMC Complement. Altern. Med. 2014, 14, 26. [Google Scholar] [CrossRef]
- Kaushik, S.; Shyam, H.; Agarwal, S.; Sharma, R.; Nag, T.C.; Dwivedi, A.K.; Balapure, A.K. Genistein potentiates Centchroman induced antineoplasticity in breast cancer via PI3K/Akt deactivation and ROS dependent induction of apoptosis. Life Sci. 2019, 239, 117073. [Google Scholar] [CrossRef]
- Hillman, G.G.; Wang, Y.; Kucuk, O.; Che, M.; Doerge, D.R.; Yudelev, M.; Joiner, M.C.; Marples, B.; Forman, J.D.; Sarkar, F.H. Genistein potentiates inhibition of tumor growth by radiation in a prostate cancer orthotopic model. Mol. Cancer Ther. 2004, 3, 1271–1279. [Google Scholar] [CrossRef]
- Eitsuka, T.; Nakagawa, K.; Kato, S.; Ito, J.; Otoki, Y.; Takasu, S.; Shimizu, N.; Takahashi, T.; Miyazawa, T. Modulation of Telomerase Activity in Cancer Cells by Dietary Compounds: A Review. Int. J. Mol. Sci. 2018, 19, 478. [Google Scholar] [CrossRef]
- Priyadarsini, R.V.; Vinothini, G.; Murugan, R.S.; Manikandan, P.; Nagini, S. The flavonoid quercetin modulates the hallmark capabilities of hamster buccal pouch tumors. Nutr. Cancer 2011, 63, 218–226. [Google Scholar] [CrossRef]
- Yang, P.; He, X.; Malhotra, A. Epigenetic targets of polyphenols in cancer. J. Environ. Pathol. Toxicol. Oncol. 2014, 33, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Abdulla, A.; Zhao, X.; Yang, F. Natural polyphenols inhibit lysine-specific demethylase-1 in vitro. J. Biochem. Pharmacol. Res. 2013, 1, 56–63. [Google Scholar] [PubMed]
- Chen, Y.; Jie, W.; Yan, W.; Zhou, K.; Xiao, Y. Lysine-specific histone demethylase 1 (LSD1): A potential molecular target for tumor therapy. Crit. Rev. Eukaryot. Gene Expr. 2012, 22, 53–59. [Google Scholar] [CrossRef]
- Fatima, N.; Baqri, S.S.R.; Bhattacharya, A.; Koney, N.K.; Husain, K.; Abbas, A.; Ansari, R.A. Role of Flavonoids as Epigenetic Modulators in Cancer Prevention and Therapy. Front. Genet. 2021, 12, 758733. [Google Scholar] [CrossRef] [PubMed]
- Tuli, H.S.; Sak, K.; Adhikary, S.; Kaur, G.; Aggarwal, D.; Kaur, J.; Kumar, M.; Parashar, N.C.; Parashar, G.; Sharma, U.; et al. Galangin: A metabolite that suppresses anti-neoplastic activities through modulation of oncogenic targets. Exp. Biol. Med. 2022, 247, 345–359. [Google Scholar] [CrossRef]
- Tan, S.; Wang, C.; Lu, C.; Zhao, B.; Cui, Y.; Shi, X.; Ma, X. Quercetin is able to demethylate the p16INK4a gene promoter. Chemotherapy 2009, 55, 6–10. [Google Scholar] [CrossRef]
- Raynal, N.J.; Charbonneau, M.; Momparler, L.F.; Momparler, R.L. Synergistic effect of 5-Aza-2′-deoxycytidine and genistein in combination against leukemia. Oncol. Res. 2008, 17, 223–230. [Google Scholar] [CrossRef]
- Fang, M.Z.; Chen, D.; Sun, Y.; Jin, Z.; Christman, J.K.; Yang, C.S. Reversal of hypermethylation and reactivation of p16INK4a, RARbeta, and MGMT genes by genistein and other isoflavones from soy. Clin. Cancer Res. 2005, 11 Pt 1, 7033–7041. [Google Scholar] [CrossRef]
- Omene, C.; Kalac, M.; Wu, J.; Marchi, E.; Frenkel, K.; O’Connor, O.A. Propolis and its Active Component, Caffeic Acid Phenethyl Ester (CAPE), Modulate Breast Cancer Therapeutic Targets via an Epigenetically Mediated Mechanism of Action. J. Cancer Sci. Ther. 2013, 5, 334–342. [Google Scholar]
- Assumpção, J.H.M.; Takeda, A.A.S.; Sforcin, J.M.; Rainho, C.A. Effects of Propolis and Phenolic Acids on Triple-Negative Breast Cancer Cell Lines: Potential Involvement of Epigenetic Mechanisms. Molecules 2020, 25, 1289. [Google Scholar] [CrossRef]
- Morel, D.; Jeffery, D.; Aspeslagh, S.; Almouzni, G.; Postel-Vinay, S. Combining epigenetic drugs with other therapies for solid tumours-past lessons and future promise. Nat. Rev. Clin. Oncol. 2020, 17, 91–107. [Google Scholar] [CrossRef] [PubMed]
- Shin, E.J.; Jo, S.; Choi, H.K.; Choi, S.; Byun, S.; Lim, T.G. Caffeic Acid Phenethyl Ester Inhibits UV-Induced MMP-1 Expression by Targeting Histone Acetyltransferases in Human Skin. Int. J. Mol. Sci. 2019, 20, 3055. [Google Scholar] [CrossRef] [PubMed]
- Zheng, N.G.; Wang, J.L.; Yang, S.L.; Wu, J.L. Aberrant Epigenetic Alteration in Eca9706 Cells Modulated by Nanoliposomal Quercetin Combined with Butyrate Mediated via Epigenetic-NF-κB Signaling. Asian Pac. J. Cancer Prev. 2014, 5, 4539–4543. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ma, L.; Feugang, J.M.; Konarski, P.; Wang, J.; Lu, J.; Fu, S.; Ma, B.; Tian, B.; Zou, C.; Wang, Z. Growth inhibitory effects of quercetin on bladder cancer cell. Front. Biosci. 2006, 11, 2275–2285. [Google Scholar] [CrossRef]
- Hao, Y.; Wei, Z.; Wang, Z.; Li, G.; Yao, Y.; Dun, B. Biotransformation of Flavonoids Improves Antimicrobial and Anti-Breast Cancer Activities In Vitro. Foods 2021, 10, 2367. [Google Scholar] [CrossRef]
- Kim, Y.H.; Lee, D.H.; Jeong, J.H.; Guo, Z.S.; Lee, Y.J. Quercetin augments TRAIL-induced apoptotic death: Involvement of the ERK signal transduction pathway. Biochem. Pharmacol. 2008, 75, 1946–1958. [Google Scholar] [CrossRef]
- Lee, W.J.; Chen, Y.R.; Tseng, T.H. Quercetin induces FasL-related apoptosis, in part, through promotion of histone H3 acetylation in human leukemia HL-60 cells. Oncol. Rep. 2011, 25, 583–591. [Google Scholar] [CrossRef]
- Xiao, X.; Shi, D.; Liu, L.; Wang, J.; Xie, X.; Kang, T.; Deng, W. Quercetin suppresses cyclooxygenase-2 expression and angiogenesis through inactivation of p300 signaling. PLoS ONE 2011, 6, e22934. [Google Scholar] [CrossRef]
- Mutoh, M.; Takahashi, M.; Fukuda, K.; Matsushima-Hibiya, Y.; Mutoh, H.; Sugimura, T.; Wakabayashi, K. Suppression of cyclooxygenase-2 promoter-dependent transcriptional activity in colon cancer cells by chemopreventive agents with a resorcin-type structure. Carcinogenesis 2000, 21, 959–963. [Google Scholar] [CrossRef]
- Lai, W.W.; Hsu, S.C.; Chueh, F.S.; Chen, Y.Y.; Yang, J.S.; Lin, J.P.; Lien, J.C.; Tsai, C.H.; Chung, J.G. Quercetin inhibits migration and invasion of SAS human oral cancer cells through inhibition of NF-κB and matrix metalloproteinase-2/-9 signaling pathways. Anticancer Res. 2013, 33, 1941–1950. [Google Scholar]
- Alvarez, M.C.; Maso, V.; Torello, C.O.; Ferro, K.P.; Saad, S. The polyphenol quercetin induces cell death in leukemia by targeting epigenetic regulators of pro-apoptotic genes. Clin. Epigenet. 2018, 10, 139. [Google Scholar] [CrossRef] [PubMed]
- Malireddy, S.; Kotha, S.R.; Secor, J.D.; Gurney, T.O.; Abbott, J.L.; Maulik, G.; Maddipati, K.R.; Parinandi, N.L. Phytochemical antioxidants modulate mammalian cellular epigenome: Implications in health and disease. Antioxid. Redox Signal. 2012, 17, 327–339. [Google Scholar] [CrossRef]
- Huang, W.J.; Lin, C.W.; Lee, C.Y.; Chi, L.L.; Chao, Y.C.; Wang, H.N.; Chiou, B.L.; Chen, T.J.; Huang, C.Y.; Chen, C.N. NBM-HD-3, a novel histone deacetylase inhibitor with anticancer activity through modulation of PTEN and AKT in brain cancer cells. J. Ethnopharmacol. 2011, 136, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Oršolić, N.; Jazvinšćak Jembrek, M.; Terzić, S. Honey and quercetin reduces ochratoxin A-induced DNA damage in the liver and the kidney through the modulation of intestinal microflora. Food Agric. Immunol. 2017, 28, 812–833. [Google Scholar] [CrossRef]
- Xu, W.; Xie, S.; Chen, X.; Pan, S.; Qian, H.; Zhu, X. Effects of Quercetin on the Efficacy of Various Chemotherapeutic Drugs in Cervical Cancer Cells. Drug Des. Devel. Ther. 2021, 15, 577–588. [Google Scholar] [CrossRef]
- Selvakumar, P.; Badgeley, A.; Murphy, P.; Anwar, H.; Sharma, U.; Lawrence, K.; Lakshmikuttyamma, A. Flavonoids and Other Polyphenols Act as Epigenetic Modifiers in Breast Cancer. Nutrients 2020, 12, 761. [Google Scholar] [CrossRef]
- Zaccaria, V.; Curti, V.; Di Lorenzo, A.; Baldi, A.; Maccario, C.; Sommatis, S.; Mocchi, R.; Daglia, M. Effect of Green and Brown Propolis Extracts on the Expression Levels of microRNAs, mRNAs and Proteins, Related to Oxidative Stress and Inflammation. Nutrients 2017, 9, 1090. [Google Scholar] [CrossRef]
- Cuevas, A.; Saavedra, N.; Cavalcante, M.F.; Salazar, L.A.; Abdalla, D.S. Identification of microRNAs involved in the modulation of pro-angiogenic factors in atherosclerosis by a polyphenol-rich extract from propolis. Arch. Biochem. Biophys. 2014, 557, 28–35. [Google Scholar] [CrossRef]
- Debnath, T.; Deb Nath, N.C.; Kim, E.K.; Lee, K.G. Role of phytochemicals in the modulation of miRNA expression in cancer. Food Funct. 2017, 8, 3432–3442. [Google Scholar] [CrossRef]
- Sayeed, M.A.; Bracci, M.; Lucarini, G.; Lazzarini, R.; Di Primio, R.; Santarelli, L. Regulation of microRNA using promising dietary phytochemicals: Possible preventive and treatment option of malignant mesothelioma. Biomed. Pharmacother. 2017, 94, 1197–1224. [Google Scholar] [CrossRef]
- Lee, P.S.; Chiou, Y.S.; Ho, C.T.; Pan, M.H. Chemoprevention by resveratrol and pterostilbene: Targeting on epigenetic regulation. Biofactors 2018, 44, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Penta, D.; Somashekar, B.S.; Meeran, S.M. Epigenetics of skin cancer: Interventions by selected bioactive phytochemicals. Photodermatol. Photoimmunol. Photomed. 2018, 34, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, H.; Masoudifar, A.; Sahebkar, A.; Zare, N.; Sadri Nahand, J.; Rashidi, B.; Mehrabian, E.; Mohammadi, M.; Mirzaei, H.R.; Jaafari, M.R. MicroRNA: A novel target of curcumin in cancer therapy. J. Cell Physiol. 2018, 233, 3004–3015. [Google Scholar] [CrossRef] [PubMed]
- Saini, S.; Arora, S.; Majid, S.; Shahryari, V.; Chen, Y.; Deng, G.; Yamamura, S.; Ueno, K.; Dahiya, R. Curcumin modulates microRNA-203-mediated regulation of the Src-Akt axis in bladder cancer. Cancer Prev. Res. 2011, 4, 1698–1709. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Li, J.; Yuan, W.; Hao, S.; Wang, M.; Wang, F.; Xuan, H. Bioactive components and mechanisms of poplar propolis in inhibiting proliferation of human hepatocellular carcinoma HepG2 cells. Biomed. Pharmacother. 2021, 144, 112364. [Google Scholar] [CrossRef] [PubMed]
- Niyomtham, N.; Koontongkaew, S.; Yingyongnarongkul, B.E.; Utispan, K. Apis mellifera propolis enhances apoptosis and invasion inhibition in head and neck cancer cells. Peer J. 2021, 9, e12139. [Google Scholar] [CrossRef]
- Ramos, S. Effects of dietary flavonoids on apoptotic pathways related to cancer chemoprevention. J. Nutr. Biochem. 2007, 18, 427–442. [Google Scholar] [CrossRef]
- Pereira, F.A.N.; Barboza, J.R.; Vasconcelos, C.C.; Lopes, A.J.O.; Ribeiro, M.N.S. Use of Stingless Bee Propolis and Geopropolis against Cancer—A Literature Review of Preclinical Studies. Pharmaceuticals 2021, 14, 1161. [Google Scholar] [CrossRef]
- Campoccia, D.; Ravaioli, S.; Santi, S.; Mariani, V.; Santarcangelo, C.; De Filippis, A.; Montanaro, L.; Arciola, C.R.; Daglia, M. Exploring the anticancer effects of standardized extracts of poplar-type propolis: In vitro cytotoxicity toward cancer and normal cell lines. Biomed. Pharmacother. 2021, 141, 111895. [Google Scholar] [CrossRef]
- Ferraresi, R.; Troiano, L.; Roat, E.; Lugli, E.; Nemes, E.; Nasi, M.; Pinti, M.; Fernandez, M.I.; Cooper, E.L.; Cossarizza, A. Essential requirement of reduced glutathione (GSH) for the anti-oxidant effect of the flavonoid quercetin. Free. Radic. Res. 2005, 39, 1249–1258. [Google Scholar] [CrossRef]
- Zubčić, K.; Radovanović, V.; Vlainić, J.; Hof, P.R.; Oršolić, N.; Šimić, G.; Jazvinšćak Jembrek, M. PI3K/Akt and ERK1/2 signalling are involved in quercetin-mediated neuroprotection against copper-induced injury. Oxid. Med. Cell. Longev. 2020, 2020, 9834742. [Google Scholar] [CrossRef] [PubMed]
- Jazvinšćak Jembrek, M.; Vlainić, J.; Čadež, V.; Šegota, S. Atomic force microscopy reveals new biophysical markers for monitoring subcellular changes in oxidative injury: Neuroprotective effects of quercetin at the nanoscale. PLoS ONE 2018, 13, e0200119. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Wang, X.; Zhuang, J.; Zhang, L.; Lin, Y. Induction of death receptor 5 and suppression of survivin contribute to sensitization of TRAIL-induced cytotoxicity by quercetin in non-small cell lung cancer cells. Carcinogenesis 2007, 28, 2114–2121. [Google Scholar] [CrossRef] [PubMed]
- Naimi, A.; Entezari, A.; Hagh, M.F.; Hassanzadeh, A.; Saraei, R.; Solali, S. Quercetin sensitizes human myeloid leukemia KG-1 cells against TRAIL-induced apoptosis. J. Cell Physiol. 2019, 234, 13233–13241. [Google Scholar] [CrossRef]
- Szliszka, E.; Czuba, Z.P.; Domino, M.; Mazur, B.; Zydowicz, G.; Krol, W. Ethanolic extract of propolis (EEP) enhances the apoptosis-inducing potential of TRAIL in cancer cells. Molecules 2009, 14, 738–754. [Google Scholar] [CrossRef]
- Szliszka, E.; Zydowicz, G.; Janoszka, B.; Dobosz, C.; Kowalczyk-Ziomek, G.; Krol, W. Ethanolic extract of Brazilian green propolis sensitizes prostate cancer cells to TRAIL-induced apoptosis. Int. J. Oncol. 2011, 38, 941–953. [Google Scholar] [CrossRef]
- Szliszka, E.; Zydowicz, G.; Mizgala, E.; Krol, W. Artepillin C (3,5-diprenyl-4-hydroxycinnamic acid) sensitizes LNCaP prostate cancer cells to TRAIL-induced apoptosis. Int. J. Oncol. 2012, 41, 818–828. [Google Scholar] [CrossRef]
- Wu, J.; Omene, C.; Karkoszka, J.; Bosland, M.; Eckard, J.; Klein, C.B.; Frenkel, K. Caffeic acid phenethyl ester (CAPE), derived from a honeybee product propolis, exhibits a diversity of anti-tumor effects in pre-clinical models of human breast cancer. Cancer Lett. 2011, 308, 43–53. [Google Scholar] [CrossRef]
- McEleny, K.; Coffey, R.; Morrissey, C.; Fitzpatrick, J.M.; Watson, R.W. Caffeic acid phenethyl ester-induced PC-3 cell apoptosis is caspase-dependent and mediated through the loss of inhibitors of apoptosis proteins. BJU Int. 2004, 94, 402–406. [Google Scholar] [CrossRef]
- Cavaliere, V.; Papademetrio, D.L.; Lombardo, T.; Costantino, S.N.; Blanco, G.A.; Alvarez, E.M. Caffeic acid phenylethyl ester and MG132, two novel nonconventional chemotherapeutic agents, induce apoptosis of human leukemic cells by disrupting mitochondrial function. Target. Oncol. 2014, 9, 25–42. [Google Scholar] [CrossRef]
- Byun, S.Y.; Kim, D.B.; Kim, E. Curcumin ameliorates the tumor-enhancing effects of a high-protein diet in an azoxymethane-induced mouse model of colon carcinogenesis. Nutr. Res. 2015, 35, 726–735. [Google Scholar] [CrossRef]
- Wu, X.; Pfalzer, A.C.; Koh, G.Y.; Tang, S.; Crott, J.W.; Thomas, M.J.; Meydani, M.; Mason, J.B. Curcumin and Salsalate Suppresses Colonic Inflammation and Procarcinogenic Signaling in High-Fat-Fed, Azoxymethane-Treated Mice. J. Agric. Food Chem. 2017, 65, 7200–7209. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, A.M.; Khaniki, M.; Azizian, S.; Mohaghgheghi, M.A.; Sadeghizadeh, M.; Najafi, F. Chemoprevention of azoxymethane-initiated colon cancer in rat by using a novel polymeric nanocarrier—Curcumin. Eur. J. Pharmacol. 2012, 689, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Chuang, S.E.; Cheng, A.L.; Lin, J.K.; Kuo, M.L. Inhibition by curcumin of diethylnitrosamine-induced hepatic hyperplasia, inflammation, cellular gene products and cell-cycle-related proteins in rats. Food Chem. Toxicol. 2000, 38, 991–995. [Google Scholar] [CrossRef]
- Tessitore, L.; Davit, A.; Sarotto, I.; Caderni, G. Resveratrol depresses the growth of colorectal aberrant crypt foci by affecting bax and p21(CIP) expression. Carcinogenesis 2000, 21, 1619–1622. [Google Scholar] [CrossRef]
- Sengottuvelan, M.; Viswanathan, P.; Nalini, N. Chemopreventive effect of trans-resveratrol a phytoalexin against colonic aberrant crypt foci and cell proliferation in 1,2-dimethylhydrazine induced colon carcinogenesis. Carcinogenesis 2006, 27, 1038–1046. [Google Scholar] [CrossRef]
- Kucinska, M.; Piotrowska, H.; Lucza, M.W.; Mikula-Pietrasik, J.; Ksiazek, K.; Wozniak, M.; Wierzchowski, M.; Dudka, J.; Jäger, W.; Murias, M. Effects of hydroxylated resveratrol analogs on oxidative stress and cancer cells death in human acute T cell leukemia cell line: Prooxidative potential of hydroxylated resveratrol analogs. Chem. Biol. Interact. 2014, 209, 96–110. [Google Scholar] [CrossRef]
- Asensi, M.; Medina, I.; Ortega, A.; Carretero, J.; Bano, M.C.; Obrador, E.; Estrela, J.M. Inhibition of cancer growth by resveratrol is related to its low bioavailability. Free. Radic. Biol. Med. 2002, 33, 387–398. [Google Scholar] [CrossRef]
- Wang, W.; Zhou, M.; Xu, Y.; Peng, W.; Zhang, S.; Li, R.; Zhang, H.; Zhang, H.; Cheng, S.; Wang, Y.; et al. Resveratrol-Loaded TPGS-Resveratrol-Solid Lipid Nanoparticles for Multidrug-Resistant Therapy of Breast Cancer: In Vivo and In Vitro Study. Front. Bioeng. Biotechnol. 2021, 9, 762489. [Google Scholar] [CrossRef]
- Inbaraj, B.S.; Hua, L.H.; Chen, B.H. Comparative Study on Inhibition of Pancreatic Cancer Cells by Resveratrol Gold Nanoparticles and a Resveratrol Nanoemulsion Prepared from Grape Skin. Pharmaceutics 2021, 13, 1871. [Google Scholar] [CrossRef] [PubMed]
- González-Sarrías, A.; Iglesias-Aguirre, C.E.; Cortés-Martín, A.; Vallejo, F.; Cattivelli, A.; Del Pozo-Acebo, L.; Del Saz, A.; López de Las Hazas, M.C.; Dávalos, A.; Espín, J.C. Milk-Derived Exosomes as Nanocarriers to Deliver Curcumin and Resveratrol in Breast Tissue and Enhance Their Anticancer Activity. Int. J. Mol. Sci. 2022, 23, 2860. [Google Scholar] [CrossRef] [PubMed]
- Rysz, J.; Franczyk, B.; Rysz-Górzyńska, M.; Gluba-Brzózka, A. Ageing, Age-Related Cardiovascular Risk and the Beneficial Role of Natural Components Intake. Int. J. Mol. Sci. 2021, 23, 183. [Google Scholar] [CrossRef]
- D’Arcy, M.S. Cell death: A review of the major forms of apoptosis, necrosis and autophagy. Cell Biol. Int. 2019, 43, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Abotaleb, M.; Samuel, S.M.; Varghese, E.; Varghese, S.; Kubatka, P.; Liskova, A.; Büsselberg, D. Flavonoids in Cancer and Apoptosis. Cancers 2019, 11, 28. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.M.; Deng, X.T.; Zhou, J.; Li, Q.P.; Ge, X.X.; Miao, L. Pharmacological basis and new insights of quercetin action in respect to its anti-cancer effects. Biomed. Pharmacother. 2020, 121, 109604. [Google Scholar] [CrossRef]
- Oršolić, N.; Odeh, D.; Jembrek, M.J.; Knežević, J.; Kučan, D. Interactions between Cisplatin and Quercetin at Physiological and Hyperthermic Conditions on Cancer Cells In Vitro and In Vivo. Molecules 2020, 25, 3271. [Google Scholar] [CrossRef]
- Maleki, M.; Khelghati, N.; Alemi, F.; Bazdar, M.; Asemi, Z.; Majidinia, M.; Sadeghpoor, A.; Mahmoodpoor, A.; Jadidi-Niaragh, F.; Targhazeh, N.; et al. Stabilization of telomere by the antioxidant property of polyphenols: Anti-aging potential. Life Sci. 2020, 259, 118341. [Google Scholar] [CrossRef]
- Colhado Rodrigues, B.L.; Lallo, M.A.; Perez, E.C. The Controversial Role of Autophagy in Tumor Development: A Systematic Review. Immunol. Investig. 2020, 49, 386–396. [Google Scholar] [CrossRef]
- Endo, S.; Hoshi, M.; Matsunaga, T.; Inoue, T.; Ichihara, K.; Ikari, A. Autophagy inhibition enhances anticancer efficacy of artepillin C, a cinnamic acid derivative in Brazilian green propolis. Biochem. Biophys. Res. Commun. 2018, 497, 437–443. [Google Scholar] [CrossRef]
- Zhang, Z.; Shi, J.; Nice, E.C.; Huang, C.; Shi, Z. The Multifaceted Role of Flavonoids in Cancer Therapy: Leveraging Autophagy with a Double-Edged Sword. Antioxidants 2021, 10, 1138. [Google Scholar] [CrossRef]
- Ashrafizadeh, M.; Ahmadi, Z.; Farkhondeh, T.; Samarghandian, S. Autophagy regulation using luteolin: New insight into its anti-tumor activity. Cancer Cell Int. 2020, 20, 537. [Google Scholar] [CrossRef] [PubMed]
- Benvenuto, M.; Albonici, L.; Focaccetti, C.; Ciuffa, S.; Fazi, S.; Cifaldi, L.; Miele, M.T.; De Maio, F.; Tresoldi, I.; Manzari, V.; et al. Polyphenol-Mediated Autophagy in Cancer: Evidence of In Vitro and In Vivo Studies. Int. J. Mol. Sci. 2020, 21, 6635. [Google Scholar] [CrossRef] [PubMed]
- Musial, C.; Siedlecka-Kroplewska, K.; Kmiec, Z.; Gorska-Ponikowska, M. Modulation of Autophagy in Cancer Cells by Dietary Polyphenols. Antioxidants 2021, 10, 123. [Google Scholar] [CrossRef]
- Yang, R.; Dong, H.; Jia, S.; Yang, Z. Resveratrol as a modulatory of apoptosis and autophagy in cancer therapy. Clin. Transl. Oncol. 2022, 24, 1219–1230. [Google Scholar] [CrossRef] [PubMed]
- Kiruthiga, C.; Devi, K.P.; Nabavi, S.M.; Bishayee, A. Autophagy: A Potential Therapeutic Target of Polyphenols in Hepatocellular Carcinoma. Cancers 2020, 12, 562. [Google Scholar] [CrossRef]
- Saha, S.; Panigrahi, D.P.; Patil, S.; Bhutia, S.K. Autophagy in health and disease: A comprehensive review. Biomed. Pharmacother. 2018, 104, 485–495. [Google Scholar] [CrossRef]
- Lin, W.W.; Karin, M.A. Cytokine-mediated link between innate immunity, inflammation, and cancer. J. Clin. Investig. 2007, 117, 1175–1183. [Google Scholar] [CrossRef]
- Hagerling, C.; Casbon, A.J.; Werb, Z. Balancing the innate immune system in tumor development. Trends Cell Biol. 2015, 25, 214–220. [Google Scholar] [CrossRef]
- Landskron, G.; De la Fuente, M.; Thuwajit, P.; Thuwajit, C.; Hermoso, M.A. Chronic inflammation and cytokines in the tumor microenvironment. J. Immunol. Res. 2014, 2014, 149185. [Google Scholar] [CrossRef]
- Qu, X.; Tang, Y.; Hua, S. Immunological Approaches Towards Cancer and Inflammation: A Cross Talk. Front. Immunol. 2018, 9, 563. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, M.K.; Lee, J.H.; Chai, E.Z.; Kanchi, M.M.; Kar, S.; Arfuso, F.; Dharmarajan, A.; Kumar, A.P.; Ramar, P.S.; Looi, C.Y.; et al. Cancer prevention and therapy through the modulation of transcription factors by bioactive natural compounds. Semin. Cancer Biol. 2016, 40–41, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Ginwala, R.; Bhavsar, R.; Chigbu, D.G.I.; Jain, P.; Khan, Z.K. Potential Role of Flavonoids in Treating Chronic Inflammatory Diseases with a Special Focus on the Anti-Inflammatory Activity of Apigenin. Antioxidants 2019, 8, 35. [Google Scholar] [CrossRef] [PubMed]
- Maleki, S.J.; Crespo, J.F.; Cabanillas, B. Anti-inflammatory effects of flavonoids. Food Chem. 2019, 299, 125124. [Google Scholar] [CrossRef] [PubMed]
- Al-Khayri, J.M.; Sahana, G.R.; Nagella, P.; Joseph, B.V.; Alessa, F.M.; Al-Mssallem, M.Q. Flavonoids as Potential Anti-Inflammatory Molecules: A Review. Molecules 2022, 27, 2901. [Google Scholar] [CrossRef]
- Kim, H.; Kim, W.; Yum, S.; Hong, S.; Oh, J.E.; Lee, J.W.; Kwak, M.K.; Park, E.J.; Na, D.H.; Jung, Y. Caffeic acid phenethyl ester activation of Nrf2 pathway is enhanced under oxidative state: Structural analysis and potential as a pathologically targeted therapeutic agent in treatment of colonic inflammation. Free Radic. Biol. Med. 2013, 65, 552–562. [Google Scholar] [CrossRef]
- Khan, M.N.; Lane, M.E.; McCarron, P.A.; Tambuwala, M.M. Caffeic acid phenethyl ester is protective in experimental ulcerative colitis via reduction in levels of pro-inflammatory mediators and enhancement of epithelial barrier function. Inflammopharmacology 2018, 26, 561–569. [Google Scholar] [CrossRef]
- Pahlavani, N.; Malekahmadi, M.; Firouzi, S.; Rostami, D.; Sedaghat, A.; Moghaddam, A.B.; Ferns, G.A.; Navashenaq, J.G.; Reazvani, R.; Safarian, M. Molecular and cellular mechanisms of the effects of propolis in inflammation, oxidative stress and glycemic control in chronic diseases. Nutr. Metab. 2020, 17, 65. [Google Scholar] [CrossRef]
- Khayyal, M.T.; el-Ghazaly, M.A.; el-Khatib, A.S. Mechanisms involved in the antiinflammatory effect of propolis extract. Drugs Exp. Clin. Res. 1993, 19, 197–203. [Google Scholar]
- Alanazi, S.; Alenzi, N.; Fearnley, J.; Harnett, W.; Watson, D.G. Temperate Propolis Has Anti-Inflammatory Effects and Is a Potent Inhibitor of Nitric Oxide Formation in Macrophages. Metabolites 2020, 10, 413. [Google Scholar] [CrossRef]
- Franchin, M.; Colón, D.F.; da Cunha, M.G.; Castanheira, F.V.; Saraiva, A.L.; Bueno-Silva, B.; Alencar, S.M.; Cunha, T.M.; Rosalen, P.L. Neovestitol, an isoflavonoid isolated from Brazilian red propolis, reduces acute and chronic inflammation: Involvement of nitric oxide and IL-6. Sci. Rep. 2016, 6, 36401. [Google Scholar] [CrossRef]
- Lim, K.M.; Bae, S.; Koo, J.E.; Kim, E.S.; Bae, O.N.; Lee, J.Y. Suppression of skin inflammation in keratinocytes and acute/chronic disease models by caffeic acid phenethyl ester. Arch. Dermatol. Res. 2015, 307, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Machado, J.L.; Assunção, A.K.; da Silva, M.C.; Dos Reis, A.S.; Costa, G.C.; Arruda, D.; Rocha, B.A.; Vaz, M.M.; Paes, A.M.; Guerra, R.N.; et al. Brazilian green propolis: Anti-inflammatory property by an immunomodulatory activity. Evid. Based Complement. Alternat. Med. 2012, 2012, 157652. [Google Scholar] [CrossRef]
- Liang, X.; Wang, P.; Yang, C.; Huang, F.; Wu, H.; Shi, H.; Wu, X. Galangin Inhibits Gastric Cancer Growth Through Enhancing STAT3 Mediated ROS Production. Front Pharmacol. 2021, 12, 646628. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-J.; Ng, S.-C.; Hsu, J.-Y.; Liu, H.; Chen, C.-J.; Huang, C.-Y.; Kuo, W.-W. Galangin Reverses H2O2-Induced Dermal Fibroblast Senescence via SIRT1-PGC-1α/Nrf2 Signaling. Int. J. Mol. Sci. 2022, 23, 1387. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Zhao, H.; Zhao, Y.; Sui, M.; Liu, J.; Li, P.; Liu, N.; Zhang, K. Role of Ginseng, Quercetin, and Tea in Enhancing Chemotherapeutic Efficacy of Colorectal Cancer. Front. Med. 2022, 9, 939424. [Google Scholar] [CrossRef]
- Yang, P.; Li, X.; Wen, Q.; Zhao, X. Quercetin attenuates the proliferation of arsenic-related lung cancer cells via a caspase-dependent DNA damage signaling. Mol. Carcinog. 2022, 61, 655–663. [Google Scholar] [CrossRef]
- Leyva-López, N.; Gutierrez-Grijalva, E.P.; Ambriz-Perez, D.L.; Heredia, J.B. Flavonoids as Cytokine Modulators: A Possible Therapy for Inflammation-Related Diseases. Int. J. Mol. Sci. 2016, 17, 921. [Google Scholar] [CrossRef]
- Li, X.; Jin, Q.; Yao, Q.; Xu, B.; Li, L.; Zhang, S.; Tu, C. The Flavonoid Quercetin Ameliorates Liver Inflammation and Fibrosis by Regulating Hepatic Macrophages Activation and Polarization in Mice. Front. Pharmacol. 2018, 9, 72. [Google Scholar] [CrossRef]
- Wadsworth, T.L.; McDonald, T.L.; Koop, D.R. Effects of Ginkgo biloba extract (EGb 761) and quercetin on lipopolysaccharide-induced signaling pathways involved in the release of tumor necrosis factor-alpha. Biochem. Pharmacol. 2001, 62, 963–974. [Google Scholar] [CrossRef]
- Través, P.G.; Luque, A.; Hortelano, S. Macrophages, inflammation, and tumor suppressors: ARF, a new player in the game. Mediat. Inflamm. 2012, 2012, 568783. [Google Scholar] [CrossRef]
- Shiri, S.; Alizadeh, A.M.; Baradaran, B.; Farhanghi, B.; Shanehbandi, D.; Khodayari, S.; Khodayari, H.; Tavassoli, A. Dendrosomal curcumin suppresses metastatic breast cancer in mice by changing M1/M2 macrophage balance in the tumor microenvironment. Asian Pac. J. Cancer Prev. 2015, 16, 3917–3922. [Google Scholar] [CrossRef] [PubMed]
- Boutilier, A.J.; Elsawa, S.F. Macrophage Polarization States in the Tumor Microenvironment. Int. J. Mol. Sci. 2021, 22, 6995. [Google Scholar] [CrossRef] [PubMed]
- Brauchli, Y.B.; Jick, S.S.; Miret, M.; Meier, C.R. Psoriasis and risk of incident cancer: An inception cohort study with a nested case-control analysis. J. Investig. Dermatol. 2009, 129, 2604–2612. [Google Scholar] [CrossRef] [PubMed]
- Martinez, C.; Vicente, V.; Yanez, J.; Alcaraz, M.; Castells, M.T.; Canteras, M.; Benavente-García, O.; Castillo, J. The effect of the flavonoid diosmin, grape seed extract and red wine on the pulmonary metastatic B16F10 melanoma. Histol. Histopathol. 2005, 20, 1121–1129. [Google Scholar] [CrossRef]
- Collard, M.; Austin, N.; Tallant, A.; Gallagher, P. Muscadine Grape Extract Reduces Lung and Liver Metastasis in Mice with Triple Negative Breast Cancer in Association with Changes in the Gut Microbiome (P05-017-19). Curr. Dev. Nutr. 2019, 3, nzz030-P05. [Google Scholar] [CrossRef]
- Ferris, R.L. Immunology and Immunotherapy of Head and Neck Cancer. J. Clin. Oncol. 2015, 33, 3293–3304. [Google Scholar] [CrossRef]
- Hayashi, A.; Gillen, A.C.; Lott, J.R. Effects of daily oral administration of quercetin chalcone and modified citrus pectin on implanted colon-25 tumor growth in Balb-c mice. Altern. Med. Rev. 2000, 5, 546–552. [Google Scholar]
- Xue, B.; Lu, Q.Y.; Massie, L.; Qualls, C.; Mao, J.T. Grape seed procyanidin extract against lung cancer: The role of microrna-106b, bioavailability, and bioactivity. Oncotarget 2018, 9, 15579–15590. [Google Scholar] [CrossRef]
- Rao, C.V.; Rivenson, A.; Simi, B.; Reddy, B.S. Chemoprevention of colon carcinogenesis by dietary curcumin, a naturally occurring plant phenolic compound. Cancer Res. 1995, 55, 259–266. [Google Scholar]
- Elbassiouni, F.E.; El-Kholy, W.M.; Elhabibi, E.M.; Albogami, S.; Fayad, E. Comparative Study between Curcumin and Nanocurcumin Loaded PLGA on Colon Carcinogenesis Induced Mice. Nanomaterials 2022, 12, 324. [Google Scholar] [CrossRef]
- Murray-Stewart, T.; Dunworth, M.; Lui, Y.; Giardiello, F.M.; Woster, P.M.; Casero, R.A., Jr. Curcumin mediates polyamine metabolism and sensitizes gastrointestinal cancer cells to antitumor polyamine-targeted therapies. PLoS ONE 2018, 13, e0202677. [Google Scholar] [CrossRef]
- Reddivari, L.; Charepalli, V.; Radhakrishnan, S.; Vadde, R.; Elias, R.J.; Lambert, J.D.; Vanamala, J.K. Grape compounds suppress colon cancer stem cells in vitro and in a rodent model of colon carcinogenesis. BMC Complement. Altern Med. 2016, 16, 278. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Yu, W.B.; Gao, Y.; Zhang, M.J.; Malhotra, A.; Yu, W.H. Modulatory Potential of Curcumin and Resveratrol on p53 Post-Translational Modifications during Gastric Cancer. J. Environ. Pathol. Toxicol. Oncol. 2018, 37, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Kweon, S.; Kim, Y.; Choi, H. Grape extracts suppress the formation of preneoplastic foci and activity of fatty acid synthase in rat liver. Exp. Mol. Med. 2003, 35, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Garvin, S.; Ollinger, K.; Dabrosin, C. Resveratrol induces apoptosis and inhibits angiogenesis in human breast cancer xenografts in vivo. Cancer Lett. 2006, 231, 113–122. [Google Scholar] [CrossRef]
- Wu, H.; He, L.; Shi, J.; Hou, X.; Zhang, H.; Zhang, X.; An, Q.; Fan, F. Resveratrol inhibits VEGF-induced angiogenesis in human endothelial cells associated with suppression of aerobic glycolysis via modulation of PKM2 nuclear translocation. Clin. Exp. Pharmacol. Physiol. 2018, 45, 1265–1273. [Google Scholar] [CrossRef]
- Sun, Y.; Zhou, Q.-M.; Lu, Y.-Y.; Zhang, H.; Chen, Q.-L.; Zhao, M.; Su, S.-B. Resveratrol Inhibits the Migration and Metastasis of MDA-MB-231 Human Breast Cancer by Reversing TGF-β1-Induced Epithelial-Mesenchymal Transition. Molecules 2019, 24, 1131. [Google Scholar] [CrossRef]
- Osmond, G.W.; Masko, E.M.; Tyler, D.S.; Freedland, S.J.; Pizzo, S. In vitro and in vivo evaluation of resveratrol and 3,5-dihydroxy-4′-acetoxy-trans-stilbene in the treatment of human prostate carcinoma and melanoma. J. Surg. Res. 2013, 179, e141–e148. [Google Scholar] [CrossRef] [PubMed]
- Park, E.-J.; Pezzuto, J.M. The pharmacology of resveratrol in animals and humans. Biochim. Biophys. Acta 2015, 1852, 1071–1113. [Google Scholar] [CrossRef]
- Kimoto, T.; Arai, S.; Kohguchi, M.; Aga, M.; Nomura, Y.; Micallef, M.J.; Kurimoto, M.; Mito, K. Apoptosis and suppression of tumor growth by art.epillin C extracted from Brazilian propolis. Cancer Detect. Prev. 1998, 22, 506–515. [Google Scholar] [CrossRef]
- Bhargava, P.; Grover, A.; Nigam, N.; Kaul, A.; Doi, M.; Ishida, Y.; Kakuta, H.; Kaul, S.C.; Terao, K.; Wadhwa, R. Anticancer activity of the supercritical extract of Brazilian green propolis and its active component, artepillin C: Bioinformatics and experimental analyses of its mechanisms of action. Int. J. Oncol. 2018, 52, 925–932. [Google Scholar] [CrossRef] [PubMed]
- Beserra, F.P.; Gushiken, L.F.S.; Hussni, M.F.; Ribeiro, V.P.; Bonamin, F.; Jackson, C.J.; Pellizzon, C.H.; Bastos, J.K. Artepillin C as an outstanding phenolic compound of Brazilian green propolis for disease treatment: A review on pharmacological aspects. Phytother. Res. 2020, 35, 2274–2286. [Google Scholar] [CrossRef] [PubMed]
- Oršolić, N.; Horvat Knežević, A.; Benković, V.; Bašić, I. Benefits of use of propolis and related flavonoids against the toxicity of chemotherapeutic agents. In Scientific Evidence of the Use of Propolis in Ethnomedicine; Oršolić, N., Bašić, I., Eds.; Transworld Research Network: Trivandrum, India, 2008; pp. 195–222. [Google Scholar]
- Bueno-Silva, B.; Kawamoto, D.; Ando-Suguimoto, E.S.; Casarin, R.C.V.; Alencar, S.M.; Rosalen, P.L.; Mayer, M.P.A. Brazilian red propolis effects on peritoneal macrophage activity: Nitric oxide, cell viability, pro-inflammatory cytokines and gene expression. J. Ethnopharmacol. 2017, 207, 100–107. [Google Scholar] [CrossRef]
- Han, S.; Sung, K.H.; Yim, D.; Lee, S.; Cho, K.; Lee, C.K.; Ha, N.J.; Kim, K. Activation of murine macrophage cell line RAW 264.7 by Korean propolis. Arch. Pharm. Res. 2002, 25, 895–902. [Google Scholar] [CrossRef] [PubMed]
- Missima, F.; Pagliarone, A.C.; Orsatti, C.L.; Araújo, J.P., Jr.; Sforcin, J.M. The effect of propolis on Th1/Th2 cytokine expression and production by melanoma-bearing mice submitted to stress. Phytother. Res. 2010, 24, 1501–1507. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.I.; Liao, J.C.; Kuo, L. Macrophage arginase promotes tumor cell growth and suppresses nitric oxide-mediated tumor cytotoxicity. Cancer Res. 2001, 61, 1100–1106. [Google Scholar] [PubMed]
- Bak, S.P.; Alonso, A.; Turk, M.J.; Berwin, B. Murine ovarian cancer vascular leukocytes require arginase-1 activity for T cell suppression. Mol. Immunol. 2008, 46, 258–268. [Google Scholar] [CrossRef]
- Rodríguez, P.C.; Ochoa, A.C. Arginine regulation by myeloid derived suppressor cells and tolerance in cancer: Mechanisms and therapeutic perspectives. Immunol. Rev. 2008, 222, 180–191. [Google Scholar] [CrossRef]
- Zhong, X.; Chen, B.; Yang, Z. The Role of Tumor-Associated Macrophages in Colorectal Carcinoma Progression. Cell Physiol. Biochem. 2018, 45, 356–365. [Google Scholar] [CrossRef]
- Zhao, H.; Zhang, X.; Chen, X.; Li, Y.; Ke, Z.; Tang, T.; Chai, H.; Guo, A.M.; Chen, H.; Yang, J. Isoliquiritigenin, a flavonoid from licorice, blocks M2 macrophage polarization in colitis-associated tumorigenesis through downregulating PGE2 and IL-6. J. Toxicol. Appl. Pharmacol. 2014, 279, 311–321. [Google Scholar] [CrossRef]
- Urban-Wojciuk, Z.; Khan, M.M.; Oyler, B.L.; Fåhraeus, R.; Marek-Trzonkowska, N.; Nita-Lazar, A.; Hupp, T.R.; Goodlett, D.R. The Role of TLRs in Anti-cancer Immunity and Tumor Rejection. Front. Immunol. 2019, 10, 2388. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Yunxiao, Z.; Fu, Y. The critical role of Toll-like receptor-mediated signaling in cancer immunotherapy. Med. Drug Discov. 2022, 14, 100122. [Google Scholar] [CrossRef]
- Sato, Y.; Goto, Y.; Narita, N.; Hoon, D.S. Cancer Cells Expressing Toll-like Receptors and the Tumor Microenvironment. Cancer Microenviron. 2009, 2 (Suppl. S1), 205–214. [Google Scholar] [CrossRef] [PubMed]
- Cen, X.; Liu, S.; Cheng, K. The Role of Toll-Like Receptor in Inflammation and Tumor Immunity. Front. Pharmacol. 2018, 9, 878. [Google Scholar] [CrossRef] [PubMed]
- Pitt, J.M.; Marabelle, A.; Eggermont, A.; Soria, J.C.; Kroemer, G.; Zitvogel, L. Targeting the tumor microenvironment: Removing obstruction to anticancer immune responses and immunotherapy. Ann. Oncol. 2016, 27, 1482–1492. [Google Scholar] [CrossRef] [PubMed]
- Alqarni, A.M.; Niwasabutra, K.; Sahlan, M.; Fearnley, H.; Fearnley, J.; Ferro, V.A.; Watson, D.G. Propolis Exerts an Anti-Inflammatory Effect on PMA-Differentiated THP-1 Cells via Inhibition of Purine Nucleoside Phosphorylase. Metabolites 2019, 9, 75. [Google Scholar] [CrossRef] [PubMed]
- Grudzien, M.; Rapak, A. Effect of Natural Compounds on NK Cell Activation. J. Immunol. Res. 2018, 2018, 4868417. [Google Scholar] [CrossRef]
- Giurini, E.F.; Madonna, M.B.; Zloza, A.; Gupta, K.H. Microbial-Derived Toll-like Receptor Agonism in Cancer Treatment and Progression. Cancers 2022, 14, 2923. [Google Scholar] [CrossRef]
- Khan, N.; Pahari, S.; Vidyarthi, A.; Aqdas, M.; Agrewala, J.N. Stimulation through CD40 and TLR-4 Is an Effective Host Directed Therapy against Mycobacterium tuberculosis. Front. Immunol. 2016, 7, 386. [Google Scholar] [CrossRef]
- Fang, H.; Ang, B.; Xu, X.; Huang, X.; Wu, Y.; Sun, Y.; Wang, W.; Li, N.; Cao, X.; Wan, T. TLR4 is essential for dendritic cell activation and anti-tumor T-cell response enhancement by DAMPs released from chemically stressed cancer cells. Cell. Mol. Immunol. 2014, 11, 150–159. [Google Scholar] [CrossRef]
- Henry, C.J.; Ornelles, D.A.; Mitchell, L.M.; Brzoza-Lewis, K.L.; Hiltbold, E.M. IL-12 produced by dendritic cells augments CD8+ T cell activation through the production of the chemokines CCL1 and CCL17. J. Immunol. 2008, 181, 8576–8584. [Google Scholar] [CrossRef] [PubMed]
- Orsatti, C.L.; Sforcin, J.M. Propolis immunomodulatory activity on TLR-2 and TLR-4 expression by chronically stressed mice. Nat. Prod. Res. 2012, 26, 446–453. [Google Scholar] [CrossRef]
- Kawai, T.; Akira, S. Pathogen recognition with Toll-like receptors. Curr. Opin. Immunol. 2005, 17, 338–344. [Google Scholar] [CrossRef]
- Murtaza, G.; Karim, S.; Akram, M.R.; Khan, S.A.; Azhar, S.; Mumtaz, A.; Bin Asad, M.H. Caffeic acid phenethyl ester and therapeutic potentials. Biomed. Res. Int. 2014, 2014, 145342. [Google Scholar] [CrossRef] [PubMed]
- Wolska, K.; Górska, A.; Antosik, K.; Ługowska, K. Immunomodulatory Effects of Propolis and its Components on Basic Immune Cell Functions. Indian, J. Pharm. Sci. 2019, 81, 575–588. [Google Scholar] [CrossRef]
- Armutcu, F.; Akyol, S.; Ustunsoy, S.; Turan, F.F. Therapeutic potential of caffeic acid phenethyl ester and its anti-inflammatory and immunomodulatory effects (Review). Exp. Ther. Med. 2015, 9, 1582–1588. [Google Scholar] [CrossRef] [PubMed]
- Nam, J.H.; Shin, D.H.; Zheng, H.; Kang, J.S.; Kim, W.K.; Kim, S.J. Inhibition of store-operated Ca2+ entry channels and K+ channels by caffeic acid phenethylester in T lymphocytes. Eur. J. Pharmacol. 2009, 612, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Papadaki, M.A.; Monastirioti, A.; Apostolopoulou, C.A.; Aggouraki, D.; Papadaki, C.; Michaelidou, K.; Vassilakopoulou, M.; Alexakou, K.; Mavroudis, D.; Agelaki, S. TLR4 and pSTAT3 Expression on Circulating Tumor Cells (CTCs) and Immune Cells in the Peripheral Blood of Breast Cancer Patients: Prognostic Implications. Cancers 2022, 14, 1053. [Google Scholar] [CrossRef]
- Khademalhosseini, M.; Arababadi, M.K. Toll-like receptor 4 and breast cancer: An updated systematic review. Breast Cancer 2019, 26, 265–271. [Google Scholar] [CrossRef]
- Mai, C.W.; Kang, Y.B.; Pichika, M.R. Should a Toll-like receptor 4 (TLR-4) agonist or antagonist be designed to treat cancer? TLR-4: Its expression and effects in the ten most common cancers. OncoTargets Ther. 2013, 6, 1573–1587. [Google Scholar] [CrossRef]
- Li, T.T.; Ogino, S.; Qian, Z.R. Toll-like receptor signaling in colorectal cancer: Carcinogenesis to cancer therapy. World J. Gastroenterol. 2014, 20, 17699–17708. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Wang, X.; Guo, Y.; Yan, J.; Abuduwaili, A.; Aximujiang, K.; Yan, J.; Wu, M. Gut microbiota influence tumor development and alter interactions with the human immune system. J. Exp. Clin. Cancer Res. 2021, 40, 42. [Google Scholar] [CrossRef] [PubMed]
- van Elsland, D.; Neefjes, J. Bacterial infections and cancer. EMBO Rep. 2018, 19, e46632. [Google Scholar] [CrossRef] [PubMed]
- Echeverría, J.; Opazo, J.; Mendoza, L.; Urzúa, A.; Wilkens, M. Structure-Activity and Lipophilicity Relationships of Selected Antibacterial Natural Flavones and Flavanones of Chilean Flora. Molecules 2017, 22, 608. [Google Scholar] [CrossRef] [PubMed]
- Ripari, N.; Sartori, A.A.; da Silva Honorio, M.; Conte, F.L.; Tasca, K.I.; Santiago, K.B.; Sforcin, J.M. Propolis antiviral and immunomodulatory activity: A review and perspectives for COVID-19 treatment. J. Pharm. Pharmacol. 2021, 73, 281–299. [Google Scholar] [CrossRef]
- Wang, L.; Song, J.; Liu, A.; Xiao, B.; Li, S.; Wen, Z.; Lu, Y.; Du, G. Research Progress of the Antiviral Bioactivities of Natural Flavonoids. Nat. Prod. Bioprospect. 2020, 10, 271–283. [Google Scholar] [CrossRef]
- Barchitta, M.; Maugeri, A.; La Mastra, C.; Rosa, M.C.; Favara, G.; Lio, R.M.S.; Agodi, A. Dietary antioxidant Intake and Human Papillomavirus Infection: Evidence from a Cross-Sectional Study in Italy. Nutrients 2020, 12, 1384. [Google Scholar] [CrossRef]
- Teresa, F.; Serra, N.; Capra, G.; Mascarella, C.; Gagliardi, C.; Di Carlo, P.; Cannella, S.; Simonte, M.R.; Lipari, D.; Sciortino, M.; et al. Helicobacter pylori and Epstein-Barr Virus Infection in Gastric Diseases: Correlation with IL-10 and IL1RN Polymorphism. J. Oncol. 2019, 2019, 1785132. [Google Scholar] [CrossRef]
- Ishaq, S.; Nunn, L. Helicobacter pylori and gastric cancer: A state of the art review. Gastroenterol. Hepatol. Bed Bench 2015, 8 (Suppl. S1), S6–S14. [Google Scholar]
- Farzaei, M.H.; Abdollahi, M.; Rahimi, R. Role of dietary polyphenols in the management of peptic ulcer. World J. Gastroenterol. 2015, 21, 6499–64517. [Google Scholar] [CrossRef]
- Li, L.; Somerset, S. Associations between Flavonoid Intakes and Gut Microbiota in a Group of Adults with Cystic Fibrosis. Nutrients 2018, 10, 1264. [Google Scholar] [CrossRef] [PubMed]
- Aghamajidi, A.; Maleki Vareki, S. The Effect of the Gut Microbiota on Systemic and Anti-Tumor Immunity and Response to Systemic Therapy against Cancer. Cancers 2022, 14, 3563. [Google Scholar] [CrossRef] [PubMed]
- Bagheri, Z.; Moeinzadeh, L.; Razmkhah, M. Roles of Microbiota in Cancer: From Tumor Development to Treatment. J. Oncol. 2022, 2022, 3845104. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liao, Y.; Tang, D. Intratumoral microbiota: New Frontiers in Tumor Immunity. Carcinogenesis 2022, bgac063. [Google Scholar] [CrossRef] [PubMed]
- Nowak, A.; Paliwoda, A.; Błasiak, J. Anti-proliferative, pro-apoptotic and anti-oxidative activity of Lactobacillus and Bifidobacterium strains: A review of mechanisms and therapeutic perspectives. Crit. Rev. Food Sci. Nutr. 2019, 59, 3456–3467. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.; Lagoudas, G.K.; Zhao, C.; Bullman, S.; Bhutkar, A.; Hu, B.; Ameh, S.; Sandel, D.; Liang, X.S.; Mazzilli, S.; et al. Commensal Microbiota Promote Lung Cancer Development via γδ T Cells. Cell 2019, 176, 998–1013.e16. [Google Scholar] [CrossRef] [PubMed]
- Helmink, B.A.; Khan, M.A.W.; Hermann, A.; Gopalakrishnan, V.; Wargo, J.A. The microbiome, cancer, and cancer therapy. Nat. Med. 2019, 25, 377–388. [Google Scholar] [CrossRef]
- Alfarrayeh, I.; Fekete, C.; Gazdag, Z.; Papp, G. Propolis ethanolic extract has double-face in vitro effect on the planktonic growth and biofilm formation of some commercial probiotics. Saudi J. Biol. Sci. 2021, 28, 1033–1039. [Google Scholar] [CrossRef]
- Okamoto, Y.; Tobe, T.; Ueda, K.; Takada, T.; Kojima, N. Oral administration of Brazilian propolis exerts estrogenic effect in ovariectomized rats. J. Toxicol. Sci. 2015, 40, 235–242. [Google Scholar] [CrossRef]
- Miryan, M.; Alavinejad, P.; Abbaspour, M.; Soleimani, D.; Ostadrahimi, A. Does propolis affect the quality of life and complications in subjects with irritable bowel syndrome (diagnosed with Rome IV criteria)? A study protocol of the randomized, double-blinded, placebo-controlled clinical trial. Trials 2020, 21, 698. [Google Scholar] [CrossRef]
- Onur, E.; Gökmen, G.G.; Nalbantsoy, A.; Kışla, D. Investigation of the supportive therapy potential of propolis extract and Lactobacillus acidophilus LA-5 milk combination against breast cancer in mice. Cytokine 2022, 149, 155743. [Google Scholar] [CrossRef]
- Soda, K. The mechanisms by which polyamines accelerate tumor spread. J. Exp. Clin. Cancer Res. 2011, 30, 95. [Google Scholar] [CrossRef] [PubMed]
- Mithul, A.S.; Wichienchot, S.; Tsao, R.; Ramakrishnan, S.; Chakkaravarthi, S. Role of dietary polyphenols on gut microbiota, their metabolites and health benefits. Food Res. Int. 2021, 142, 110189. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Kang, C.H.; Kim, G.H.; Cho, H. Anti-Tumor Effects of Heat-Killed, L. reuteri MG5346 and L. casei MG4584 against Human Colorectal Carcinoma through Caspase-9-Dependent Apoptosis in Xenograft Model. Microorganisms 2022, 10, 533. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.; Kleniewska, P.; Pawliczak, R. Antioxidative activity of probiotics. Arch. Med. Sci. 2019, 17, 792–804. [Google Scholar] [CrossRef]
- Kim, H.; Kim, J.S.; Kim, Y.G.; Jeong, Y.; Kim, J.-E.; Paek, N.-S.; Kang, C.-H. Antioxidant and Probiotic Properties of Lactobacilli and Bifidobacteria of Human Origins. Biotechnol. Bioproc. E 2020, 25, 421–430. [Google Scholar] [CrossRef]
- Ayyanna, R.; Ankaiah, D.; Arul, V. Anti-inflammatory and Antioxidant Properties of Probiotic Bacterium Lactobacillus mucosae AN1 and Lactobacillus fermentum SNR1 in Wistar Albino Rats. Front. Microbiol. 2018, 9, 3063. [Google Scholar] [CrossRef]
- Kodali, V.P.; Sen, R. Antioxidant and free radical scavenging activities of an exopolysaccharide from a probiotic bacterium. Biotechnol. J. 2008, 3, 245–251. [Google Scholar] [CrossRef]
- Petruzzi, L.; Rosaria Corbo, M.; Campaniello, D.; Speranza, B.; Sinigaglia, M.; Bevilacqua, A. Antifungal and Antibacterial Effect of Propolis: A Comparative Hit for Food-Borne Pseudomonas, Enterobacteriaceae and Fungi. Foods 2020, 9, 559. [Google Scholar] [CrossRef]
- Almuhayawi, M.S. Propolis as a novel antibacterial agent. Saudi J. Biol. Sci. 2020, 27, 3079–3086. [Google Scholar] [CrossRef]
- Berendika, M.; Domjanić Drozdek, S.; Odeh, D.; Oršolić, N.; Dragičević, P.; Sokolović, M.; Garofulić, I.E.; Đikić, D.; Jurčević, I.L. Beneficial Effects of Laurel (Laurus nobilis L.) and Myrtle (Myrtus communis L.) Extract on Rat Health. Molecules 2022, 27, 581. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Jin, X.; Li, Q.; Sawaya, A.C.H.F.; Leu, R.K.L.; Conlon, M.A.; Wu, L.; Hu, F. Propolis from Different Geographic Origins Decreases Intestinal Inflammation and Bacteroides spp. Populations in a Model of DSS-Induced Colitis. Mol. Nutr. Food Res. 2018, 62, e1800080. [Google Scholar] [CrossRef] [PubMed]
- Ono, M. Molecular links between tumour angiogenesis and inflammation: Inflammatory stimuli of macrophages and cancer cells as targets for therapeutic strategy. Cancer Sci. 2008, 99, 1501–1506. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yan, J.; Liu, B. Targeting VEGF/VEGFR to Modulate Antitumor Immunity. Front. Immunol. 2018, 9, 978. [Google Scholar] [CrossRef]
- Wang, S.; Liu, G.; Li, Y.; Pan, Y. Metabolic Reprogramming Induces Macrophage Polarization in the Tumor Microenvironment. Front. Immunol. 2022, 13, 840029. [Google Scholar] [CrossRef]
- Lee, K.W.; Kang, N.J.; Kim, J.H.; Lee, K.M.; Lee, D.E.; Hur, H.J.; Lee, H.J. Caffeic acid phenethyl ester inhibits invasion and expression of matrix metalloproteinase in SK-Hep1 human hepatocellular carcinoma cells by targeting nuclear factor kappa B. Genes Nutr. 2008, 2, 319–322. [Google Scholar] [CrossRef]
- Song, Y.S.; Park, E.H.; Jung, K.J.; Jin, C. Inhibition of angiogenesis by propolis. Arch. Pharm. Res. 2002, 25, 500–504. [Google Scholar] [CrossRef]
- Selmin, O.I.; Donovan, M.G.; Stillwater, B.J.; Neumayer, L.; Romagnolo, D.F. Epigenetic Regulation and Dietary Control of Triple Negative Breast Cancer. Front Nutr. 2020, 7, 159. [Google Scholar] [CrossRef]
- Huang, Q.; Li, S.; Zhang, L.; Qiao, X.; Zhang, Y.; Zhao, X.; Xiao, G.; Li, Z. CAPE-pNO2 Inhibited the Growth and Metastasis of Triple-Negative Breast Cancer via the EGFR/STAT3/Akt/E-Cadherin Signaling Pathway. Front. Oncol. 2019, 9, 461. [Google Scholar] [CrossRef]
- Izuta, H.; Shimazawa, M.; Tsuruma, K.; Araki, Y.; Mishima, S.; Hara, H. Bee products prevent VEGF-induced angiogenesis in human umbilical vein endothelial cells. BMC Complement. Altern. Med. 2009, 9, 45. [Google Scholar] [CrossRef]
- Ahn, M.R.; Kunimasa, K.; Ohta, T.; Kumazawa, S.; Kamihira, M.; Kaji, K.; Uto, Y.; Hori, H.; Nagasawa, H.; Nakayama, T. Suppression of tumor-induced angiogenesis by Brazilian propolis: Major component artepillin C inhibits in vitro tube formation and endothelial cell proliferation. Cancer Lett. 2007, 252, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Kunimasa, K.; Ahn, M.R.; Kobayashi, T.; Eguchi, R.; Kumazawa, S.; Fujimori, Y.; Nakano, T.; Nakayama, T.; Kaji, K.; Ohta, T. Brazilian Propolis Suppresses Angiogenesis by Inducing Apoptosis in Tube-Forming Endothelial Cells through Inactivation of Survival Signal ERK1/2. Evid. Based Complement. Alternat. Med. 2011, 2011, 870753. [Google Scholar] [CrossRef] [PubMed]
- Hattori, H.; Okuda, K.; Murase, T.; Shigetsura, Y.; Narise, K.; Semenza, G.L.; Nagasawa, H. Isolation, identification, and biological evaluation of HIF-1-modulating compounds from Brazilian green propolis. Bioorg. Med. Chem. 2011, 19, 5392–5401. [Google Scholar] [CrossRef]
- Masoud, G.N.; Li, W. HIF-1α pathway: Role, regulation and intervention for cancer therapy. Acta Pharm. Sin. 2015, 5, 378–389. [Google Scholar] [CrossRef] [PubMed]
- Valente, M.J.; Baltazar, A.F.; Henrique, R.; Estevinho, L.; Carvalho, M. Biological activities of Portuguese propolis: Protection against free radical-induced erythrocyte damage and inhibition of human renal cancer cell growth in vitro. Food Chem. Toxicol. 2011, 49, 86–92. [Google Scholar] [CrossRef]
- Arablou, T.; Aryaeian, N.; Khodaverdi, S.; Kolahdouz-Mohammadi, R.; Moradi, Z.; Rashidi, N.; Delbandi, A.A. The effects of resveratrol on the expression of VEGF, TGF-β, and MMP-9 in endometrial stromal cells of women with endometriosis. Sci. Rep. 2021, 11, 6054. [Google Scholar] [CrossRef]
- Rauf, A.; Imran, M.; Butt, M.S.; Nadeem, M.; Peters, D.G.; Mubarak, M.S. Resveratrol as an anti-cancer agent: A review. Crit. Rev. Food Sci. Nutr. 2018, 58, 1428–1447. [Google Scholar] [CrossRef]
- Shanmugam, M.K.; Warrier, S.; Kumar, A.P.; Sethi, G.; Arfuso, F. Potential Role of Natural Compounds as Anti-Angiogenic Agents in Cancer. Curr. Vasc. Pharmacol. 2017, 15, 503–519. [Google Scholar] [CrossRef]
- Hu, Y.; Wang, S.; Wu, X.; Zhang, J.; Chen, R.; Chen, M.; Wang, Y. Chinese herbal medicine-derived compounds for cancer therapy: A focus on hepatocellular carcinoma. J. Ethnopharmacol. 2013, 149, 601–612. [Google Scholar] [CrossRef]
- Kim, D.H.; Sung, B.; Kim, J.A.; Kang, Y.J.; Hwang, S.Y.; Hwang, N.L.; Suh, H.; Choi, Y.H.; Im, E.; Chung, H.Y.; et al. HS-1793, a resveratrol analogue, downregulates the expression of hypoxia-induced HIF-1 and VEGF and inhibits tumor growth of human breast cancer cells in a nude mouse xenograft model. Int. J. Oncol. 2017, 51, 715–723. [Google Scholar] [CrossRef]
- Uvez, A.; Aydinlik, S.; Esener, O.B.B.; Erkisa, M.; Karakus, D.; Armutak, E.I. Synergistic interactions between resveratrol and doxorubicin inhibit angiogenesis both in vitro and in vivo. Pol. J. Vet. Sci. 2020, 23, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, R.; Chatterjee, S.; Hembram, K.C.; Sethy, C.; Mandal, M.; Kundu, C.N. Nano formulated resveratrol inhibits metastasis and angiogenesis by reducing inflammatory cytokines in oral cancer cells by targeting tumor associated macrophages. J. Nutr. Biochem. 2021, 92, 108624. [Google Scholar] [CrossRef] [PubMed]
- Liskova, A.; Koklesova, L.; Samec, M.; Smejkal, K.; Samuel, S.M.; Varghese, E.; Abotaleb, M.; Biringer, K.; Kudela, E.; Danko, J.; et al. Flavonoids in Cancer Metastasis. Cancers 2020, 12, 1498. [Google Scholar] [CrossRef] [PubMed]
- Cayetano-Salazar, L.; Nava-Tapia, D.A.; Astudillo-Justo, K.D.; Arizmendi-Izazaga, A.; Sotelo-Leyva, C.; Herrera-Martinez, M.; Villegas-Comonfort, S.; Navarro-Tito, N. Flavonoids as regulators of TIMPs expression in cancer: Consequences, opportunities, and challenges. Life Sci. 2022; 120932, advance online publication. [Google Scholar] [CrossRef] [PubMed]
- Sari, A.N.; Dhanjal, J.K.; Elwakeel, A.; Kumar, V.; Meidinna, H.N.; Zhang, H.; Ishida, Y.; Terao, K.; Sundar, D.; Kaul, S.C.; et al. A Low Dose Combination of Withaferin A and Caffeic Acid Phenethyl Ester Possesses Anti-Metastatic Potential In Vitro: Molecular Targets and Mechanisms. Cancers 2022, 14, 787. [Google Scholar] [CrossRef]
- Masadah, R.; Ikram, D.; Rauf, S. Effects of propolis and its bioactive components on breast cancer cell pathways and the molecular mechanisms involved. Breast Dis. 2021, 40, S15–S25. [Google Scholar] [CrossRef]
- Fraser, S.P.; Hemsley, F.; Djamgoz, M.B.A. Caffeic acid phenethyl ester: Inhibition of metastatic cell behaviours via voltage-gated sodium channel in human breast cancer in vitro. Int. J. Biochem. Cell Biol. 2016, 71, 111–118. [Google Scholar] [CrossRef]
- Messeha, S.S.; Zarmouh, N.O.; Soliman, K.F.A. Polyphenols Modulating Effects of PD-L1/PD-1 Checkpoint and EMT-Mediated PD-L1 Overexpression in Breast Cancer. Nutrients 2021, 13, 1718. [Google Scholar] [CrossRef] [PubMed]
- Ruidas, B.; Sur, T.K.; Das Mukhopadhyay, C.; Sinha, K.; Som Chaudhury, S.; Sharma, P.; Bhowmick, S.; Majumder, R.; Saha, A. Quercetin: A silent retarder of fatty acid oxidation in breast cancer metastasis through steering of mitochondrial CPT1. Breast Cancer 2022, 29, 748–760. [Google Scholar] [CrossRef] [PubMed]
- Trinh, N.-T.; Nguyen, T.M.N.; Yook, J.-I.; Ahn, S.-G.; Kim, S.-A. Quercetin and Quercitrin from Agrimonia pilosa Ledeb Inhibit the Migration and Invasion of Colon Cancer Cells through the JNK Signaling Pathway. Pharmaceuticals 2022, 15, 364. [Google Scholar] [CrossRef]
- Dhanaraj, T.; Mohan, M.; Arunakaran, J. Quercetin attenuates metastatic ability of human metastatic ovarian cancer cells via modulating multiple signaling molecules involved in cell survival, proliferation, migration and adhesion. Arch. Biochem. Biophys. 2021, 701, 108795. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Park, S.U. Quercetin and its role in biological functions: An updated review. EXCLI J. 2018, 17, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.-H.; Lai, S.-L.; Chen, W.-S.; Hung, W.-Y.; Chow, J.-M.; Hsiao, M.; Lee, W.-J.; Chien, M.-H. Quercetin suppresses the metastatic ability of lung cancer through inhibiting Snail-dependent Akt activation and Snail-independent ADAM9 expression pathways. Biochim. Biophys. Acta Mol. Cell Res. 2017, 1864, 1746–1758. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Song, D.; Jiang, S.; Yang, X.; Ding, T.; Zhang, H.; Luo, J.; Liao, J.; Yin, Q. Quercetin restrains TGF-β1-induced epithelial-mesenchymal transition by inhibiting Twist1 and regulating E-cadherin expression. Biochem. Biophys. Res. Commun. 2018, 498, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.-H.; Cheng, C.-Y.; Su, T.; Fu, X.-Q.; Guo, H.; Li, T.; Tse, A.K.-W.; Kwan, H.-Y.; Yu, H.; Yu, Z.-L. Quercetin inhibits HGF/c-Met signaling and HGF-stimulated melanoma cell migration and invasion. Mol. Cancer 2015, 14, 103. [Google Scholar] [CrossRef]
- Cao, H.-H.; Tse, A.K.-W.; Kwan, H.-Y.; Yu, H.; Cheng, C.-Y.; Su, T.; Fong, W.-F.; Yu, Z.-L. Quercetin exerts anti-melanoma activities and inhibits STAT3 signaling. Biochem. Pharmacol. 2014, 87, 424–434. [Google Scholar] [CrossRef]
- Erdogan, S.; Turkekul, K.; Dibirdik, I.; Doganlar, O.; Doganlar, Z.B.; Bilir, A.; Oktem, G. Midkine downregulation increases the efficacy of quercetin on prostate cancer stem cell survival and migration through PI3K/AKT and MAPK/ERK pathway. Biomed. Pharmacother. 2018, 107, 793–805. [Google Scholar] [CrossRef]
- Zhang, X.M.; Huang, S.P.; Xu, Q. Quercetin inhibits the invasion of murine melanoma B16-BL6 cells by decreasing pro-MMP-9 via the PKC pathway. Cancer Chemother. Pharmacol. 2004, 53, 82–88. [Google Scholar] [CrossRef]
- Castillo-Pichardo, L.; Martínez-Montemayor, M.M.; Martínez, J.E.; Wall, K.M.; Cubano, L.A.; Dharmawardhane, S. Inhibition of mammary tumor growth and metastases to bone and liver by dietary grape polyphenols. Clin. Exp. Metastasis 2009, 26, 505–516. [Google Scholar] [CrossRef]
- Lu, J.; Wang, Z.; Li, S.; Xin, Q.; Yuan, M.; Li, H.; Song, X.; Gao, H.; Pervaiz, N.; Sun, X.; et al. Quercetin Inhibits the Migration and Invasion of HCCLM3 Cells by Suppressing the Expression of p-Akt1, Matrix Metalloproteinase (MMP) MMP-2, and MMP-9. Med. Sci. Monit. 2018, 24, 2583–2589. [Google Scholar] [CrossRef]
- Lin, C.W.; Hou, W.C.; Shen, S.C.; Juan, S.H.; Ko, C.H.; Wang, L.M.; Chen, Y.C. Quercetin inhibition of tumor invasion via suppressing PKC delta/ERK/AP-1-dependent matrix metalloproteinase-9 activation in breast carcinoma cells. Carcinogenesis 2008, 29, 1807–1815. [Google Scholar] [CrossRef] [PubMed]
- Lupo, G.; Cambria, M.T.; Olivieri, M.; Rocco, C.; Caporarello, N.; Longo, A.; Zanghì, G.; Salmeri, M.; Foti, M.C.; Anfuso, C.D. Anti-angiogenic effect of quercetin and its 8-methyl pentamethyl ether derivative in human microvascular endothelial cells. J. Cell. Mol. Med. 2019, 23, 6565–6577. [Google Scholar] [CrossRef] [PubMed]
- Lirdprapamongkol, K.; Sakurai, H.; Abdelhamed, S.; Yokoyama, S.; Maruyama, T.; Athikomkulchai, S.; Viriyaroj, A.; Awale, S.; Yagita, H.; Ruchirawat, S.; et al. A flavonoid chrysin suppresses hypoxic survival and metastatic growth of mouse breast cancer cells. Oncol. Rep. 2013, 30, 2357–2364. [Google Scholar] [CrossRef] [PubMed]
- Yufei, Z.; Yuqi, W.; Binyue, H.; Lingchen, T.; Xi, C.; Hoffelt, D.; Fuliang, H. Chrysin Inhibits Melanoma Tumor Metastasis via Interfering with the FOXM1/β-Catenin Signaling. J. Agric. Food Chem. 2020, 68, 9358–9367. [Google Scholar] [CrossRef]
- Zhang, W.H.; Chen, S.; Liu, X.L.; Bing-Lin; Liu, X.W.; Zhou, Y. Study on antitumor activities of the chrysin-chromene-spirooxindole on Lewis lung carcinoma C57BL/6 mice in vivo. Bioorg. Med. Chem. Lett. 2020, 30, 127410. [Google Scholar] [CrossRef]
- Prangsaengtong, O.; Athikomkulchai, S.; Xu, J.; Koizumi, K.; Inujima, A.; Shibahara, N.; Shimada, Y.; Tadtong, S.; Awale, S. Chrysin Inhibits Lymphangiogenesis In Vitro. Biol. Pharm. Bull. 2016, 39, 466–472. [Google Scholar] [CrossRef][Green Version]
- Chen, K.S.; Shi, M.D.; Chien, C.S.; Shih, Y.W. Pinocembrin suppresses TGF-β1-induced epithelial-mesenchymal transition and metastasis of human Y-79 retinoblastoma cells through inactivating αvβ3 integrin/FAK/p38α signaling pathway. Cell Biosci. 2014, 4, 41. [Google Scholar] [CrossRef]
- Zhu, X.; Li, R.; Wang, C.; Zhou, S.; Fan, Y.; Ma, S.; Gao, D.; Gai, N.; Yang, J. Pinocembrin Inhibits the Proliferation and Metastasis of Breast Cancer via Suppression of the PI3K/AKT Signaling Pathway. Front. Oncol. 2021, 11, 661184. [Google Scholar] [CrossRef]
- Guo, T.L.; Zhang, X.L.; Bartolucci, E.; McCay, J.A.; White, K.L., Jr.; You, L. Genistein and methoxychlor modulate the activity of natural killer cells and the expression of phenotypic markers by thymocytes and splenocytes in F0 and F1 generations of Sprague-Dawley rats. Toxicology 2002, 172, 205–215. [Google Scholar] [CrossRef]
- Liang, Y.S.; Qi, W.T.; Guo, W.; Wang, C.L.; Hu, Z.B.; Li, A.K. Genistein and daidzein induce apoptosis of colon cancer cells by inhibiting the accumulation of lipid droplets. Food Nutr. Res. 2018, 62. [Google Scholar] [CrossRef]
- Tanjak, P.; Thiantanawat, A.; Watcharasit, P.; Satayavivad, J. Genistein reduces the activation of AKT and EGFR, and the production of IL6 in cholangiocarcinoma cells involving estrogen and estrogen receptors. Int. J. Oncol. 2018, 53, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Ren, J.; Tang, L. Genistein inhibits invasion and migration of colon cancer cells by recovering WIF1 expression. Mol. Med. Rep. 2018, 17, 7265–7273. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Zhang, D.; Yang, S.; Wang, Y.; Tang, Z.; Fu, X. Co-administration of genistein with doxorubicin-loaded polypeptide nanoparticles weakens the metastasis of malignant prostate cancer by amplifying oxidative damage. Biomater. Sci. 2018, 6, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Koltai, T. Cancer: Fundamentals behind pH targeting and the double-edged approach. OncoTargets Ther. 2016, 9, 6343–6360. [Google Scholar] [CrossRef]
- Ocaña, M.C.; Martínez-Poveda, B.; Quesada, A.R.; Medina, M.Á. Metabolism within the tumor microenvironment and its implication on cancer progression: An ongoing therapeutic target. Med. Res. Rev. 2019, 39, 70–113. [Google Scholar] [CrossRef]
- De Santis, M.C.; Porporato, P.E.; Martini, M.; Morandi, A. Signaling Pathways Regulating Redox Balance in Cancer Metabolism. Front. Oncol. 2018, 8, 126. [Google Scholar] [CrossRef]
- Goetzman, E.S.; Prochownik, E.V. The Role for Myc in Coordinating Glycolysis, Oxidative Phosphorylation, Glutaminolysis, and Fatty Acid Metabolism in Normal and Neoplastic Tissues. Front. Endocrinol. 2018, 9, 129. [Google Scholar] [CrossRef]
- Gouirand, V.; Guillaumond, F.; Vasseur, S. Influence of the Tumor microenvironment on Cancer Cells Metabolic Reprogramming. Front. Oncol. 2018, 8, 117. [Google Scholar] [CrossRef]
- Valença, I.; Morais-Santos, F.; Miranda-Gonçalves, V.; Ferreira, A.M.; Almeida-Aguiar, C.; Baltazar, F. Portuguese propolis disturbs glycolytic metabolism of human colorectal cancer in vitro. BMC Complement. Altern. Med. 2013, 13, 184. [Google Scholar] [CrossRef]
- de la Cruz-López, K.G.; Castro-Muñoz, L.J.; Reyes-Hernández, D.O.; García-Carrancá, A.; Manzo-Merino, J. Lactate in the Regulation of Tumor Microenvironment and Therapeutic Approaches. Front Oncol. 2019, 9, 1143. [Google Scholar] [CrossRef]
- Cerella, C.; Radogna, F.; Dicato, M.; Diederich, M. Natural compounds as regulators of the cancer cell metabolism. Int. J. Cell Biol. 2013, 2013, 639401. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.L.; Chen, Y.G. Natural compounds regulate glycolysis in hypoxic tumor microenvironment. Biomed. Res. Int. 2015, 2015, 354143. [Google Scholar] [CrossRef] [PubMed]
- Shim, C.K.; Cheon, E.P.; Kang, K.W.; Cheon, K.S.; Han, H.K. Inhibition effect of flavonoids on monocarboxylate transporter 1 (MCT1) in Caco-2 cells. J. Pharm. Pharmacol. 2007, 59, 1515–1519. [Google Scholar] [CrossRef]
- Jia, L.; Huang, S.; Yin, X.; Zan, Y.; Guo, Y.; Han, L. Quercetin suppresses the mobility of breast cancer by suppressing glycolysis through Akt-mTOR pathway mediated autophagy induction. Life Sci. 2018, 208, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Keating, E.; Martel, F. Antimetabolic Effects of Polyphenols in Breast Cancer Cells: Focus on Glucose Uptake and Metabolism. Front. Nutr. 2018, 5, 25. [Google Scholar] [CrossRef]
- Colen, C.B.; Shen, Y.; Ghoddoussi, F.; Yu, P.; Francis, T.B.; Koch, B.J.; Monterey, M.D.; Galloway, M.P.; Sloan, A.E.; Mathupala, S.P. Metabolic targeting of lactate efflux by malignant glioma inhibits invasiveness and induces necrosis: An in vivo study. Neoplasia 2011, 13, 620–632. [Google Scholar] [CrossRef]
- Coss, R.A.; Storck, C.W.; Daskalakis, C.; Berd, D.; Wahl, M.L. Intracellular acidification abrogates the heat shock response and compromises survival of human melanoma cells. Mol. Cancer Ther. 2003, 2, 383–388. [Google Scholar]
- Fang, J.; Quinones, Q.J.; Holman, T.L.; Morowitz, M.J.; Wang, Q.; Zhao, H.; Sivo, F.; Maris, J.M.; Wahl, M.L. The H+ −linked monocarboxylate transporter (MCT1/SLC16A1): A potential therapeutic target for high-risk neuroblastoma. Mol. Pharmacol. 2006, 70, 2108–2115. [Google Scholar] [CrossRef]
- Gonzalez-Menendez, P.; Hevia, D.; Rodriguez-Garcia, A.; Mayo, J.C.; Sainz, R.M. Regulation of GLUT transporters by flavonoids in androgen-sensitive and -insensitive prostate cancer cells. Endocrinology 2014, 155, 3238–3250. [Google Scholar] [CrossRef]
- Pérez, A.; Ojeda, P.; Ojeda, L.; Salas, M.; Rivas, C.I.; Vera, J.C.; Reyes, A.M. Hexose transporter GLUT1 harbors several distinct regulatory binding sites for flavones and tyrphostins. Biochemistry 2011, 50, 8834–8845. [Google Scholar] [CrossRef]
- Park, J.B. Flavonoids are potential inhibitors of glucose uptake in U937 cells. Biochem. Biophys. Res. Commun. 1999, 260, 568–574. [Google Scholar] [CrossRef] [PubMed]
- Kueck, A.; Opipari, A.W., Jr.; Griffith, K.A.; Tan, L.; Choi, M.; Huang, J.; Wahl, H.; Liu, J.R. Resveratrol inhibits glucose metabolism in human ovarian cancer cells. Gynecol. Oncol. 2007, 107, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.; Wang, W.; He, G.; Kuick, R.D.; Gossner, G.; Kueck, A.S.; Wahl, H.; Opipari, A.W.; Liu, J.R. Resveratrol inhibits ovarian tumor growth in an in vivo mouse model. Cancer 2016, 122, 722–729. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.H.; Lee, J.H.; Thien Quach, C.H.; Paik, J.Y.; Oh, H.; Park, J.W.; Lee, E.J.; Moon, S.H.; Lee, K.H. Resveratrol suppresses cancer cell glucose uptake by targeting reactive oxygen species-mediated hypoxia-inducible factor-1α activation. J. Nucl. Med. 2013, 54, 2161–2167. [Google Scholar] [CrossRef]
- Salas, M.; Obando, P.; Ojeda, L.; Ojeda, P.; Perez, A.; Vargas-Uribe, M.; Rivas, C.I.; Vera, J.C.; Reyes, A.M. Resolution of the direct interaction with and inhibition of the human GLUT1 hexose transporter by resveratrol from its effect on glucose accumulation. Am. J. Physiol. Cell Physiol. 2013, 305, C90–C99. [Google Scholar] [CrossRef]
- Sohel, M.; Sultana, H.; Sultana, T.; Al Amin, M.; Aktar, S.; Ali, M.C.; Rahim, Z.B.; Hossain, M.A.; Al Mamun, A.; Amin, M.N.; et al. Chemotherapeutic potential of hesperetin for cancer treatment, with mechanistic insights: A comprehensive review. Heliyon 2022, 8, e08815. [Google Scholar] [CrossRef]
- Qian, Y.; Wang, X.; Chen, X. Inhibitors of glucose transport and glycolysis as novel anticancer therapeutics. World J. Transl. Med. 2014, 3, 37–57. [Google Scholar] [CrossRef]
- Sarvesvaran, J.; Going, J.J.; Milroy, R.; Kaye, S.B.; Keith, W.N. Is small cell lung cancer the perfect target for anti-telomerase treatment? Carcinogenesis 1999, 20, 1649–1651. [Google Scholar] [CrossRef]
- Kumaki, F.; Kawai, T.; Hiroi, S.; Shinomiya, N.; Ozeki, Y.; Ferrans, V.J.; Torikata, C. Telomerase activity and expression of human telomerase RNA component and human telomerase reverse transcriptase in lung carcinomas. Hum. Pathol. 2001, 32, 188–195. [Google Scholar] [CrossRef]
- Oztas, E.; Kara, H.; Kara, Z.P.; Aydogan, M.U.; Uras, C.; Ozhan, G. Association Between Human Telomerase Reverse Transcriptase Gene Variations and Risk of Developing Breast Cancer. Genet. Test. Mol. Biomark. 2016, 20, 459–464. [Google Scholar] [CrossRef]
- Shi, M.; Zheng, J.; Liu, C.; Tan, G.; Qing, Z.; Yang, S.; Yang, J.; Tan, Y.; Yang, R. SERS assay of telomerase activity at single-cell level and colon cancer tissues via quadratic signal amplification. Biosens. Bioelectron. 2016, 77, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, Y.; Sakamoto, H.; Satoh, K.; Yamamoto, T. Tamoxifen and gonadal steroids inhibit colon cancer growth in association with inhibition of thymidylate synthase, survivin and telomerase expression through estrogen receptor beta mediated system. Cancer Lett. 2000, 161, 63–71. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, Y. Functional and mechanistic analysis of telomerase: An antitumor drug target. Pharmacol. Ther. 2016, 163, 24–47. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Li, J.; Yu, P.; Du, G. Research Status and Hotspots of Anticancer Natural Products Based on the Patent Literature and Scientific Articles. Front. Pharmacol. 2022, 13, 903239. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.L.Y.; Sperry, J.; Ip, N.Y.; Brimble, M.A. Natural products targeting telomere maintenance. Med. Chem. Commun. 2011, 2, 229–245. [Google Scholar] [CrossRef]
- Rahmati-Yamchi, M.; Ghareghomi, S.; Haddadchi, G.; Milani, M.; Aghazadeh, M.; Daroushnejad, H. Fenugreek extract diosgenin and pure diosgenin inhibit the hTERT gene expression in A549 lung cancer cell line. Mol. Biol. Rep. 2014, 41, 6247–6252. [Google Scholar] [CrossRef]
- Noureini, S.K.; Wink, M. Antiproliferative Effect of the Isoquinoline Alkaloid Papaverine in Hepatocarcinoma HepG-2 Cells—Inhibition of Telomerase and Induction of Senescence. Molecules 2014, 19, 11846–11859. [Google Scholar] [CrossRef]
- Chung, S.S.; Dutta, P.; Austin, D.; Wang, P.; Awad, A.; Vadgama, J.V. Combination of resveratrol and 5-flurouracil enhanced anti-telomerase activity and apoptosis by inhibiting STAT3 and Akt signaling pathways in human colorectal cancer cells. Oncotarget 2018, 9, 32943–32957. [Google Scholar] [CrossRef]
- Chen, R.J.; Wu, P.H.; Ho, C.T.; Way, T.D.; Pan, M.H.; Chen, H.M.; Ho, Y.S.; Wang, Y.J. P53-dependent downregulation of hTERT protein expression and telomerase activity induces senescence in lung cancer cells as a result of pterostilbene treatment. Cell Death Dis. 2017, 8, e2985. [Google Scholar] [CrossRef]
- Cosan, D.T.; Soyocak, A.; Basaran, A.; Degirmenci, I.; Gunes, H.V.; Sahin, F.M. Effects of various agents on DNA fragmentation and telomerase enzyme activities in adenocarcinoma cell lines. Mol. Biol. Rep. 2011, 38, 2463–2469. [Google Scholar] [CrossRef]
- Savelyev, N.; Baykuzina, P.; Dokudovskaya, S.; Lavrik, O.; Rubtsova, M.; Dontsova, O. Comprehensive analysis of telomerase inhibition by gallotannin. Oncotarget 2018, 9, 18712–18719. [Google Scholar] [CrossRef] [PubMed]
- Avci, C.B.; Yilmaz, S.; Dogan, Z.O.; Saydam, G.; Dodurga, Y.; Ekiz, H.A.; Kartal, M.; Sahin, F.; Baran, Y.; Gunduz, C. Quercetin-induced apoptosis involves increased hTERT enzyme activity of leukemic cells. Hematology 2011, 16, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Salatino, A. Perspectives for Uses of Propolis in Therapy against Infectious Diseases. Molecules 2022, 27, 4594. [Google Scholar] [CrossRef] [PubMed]
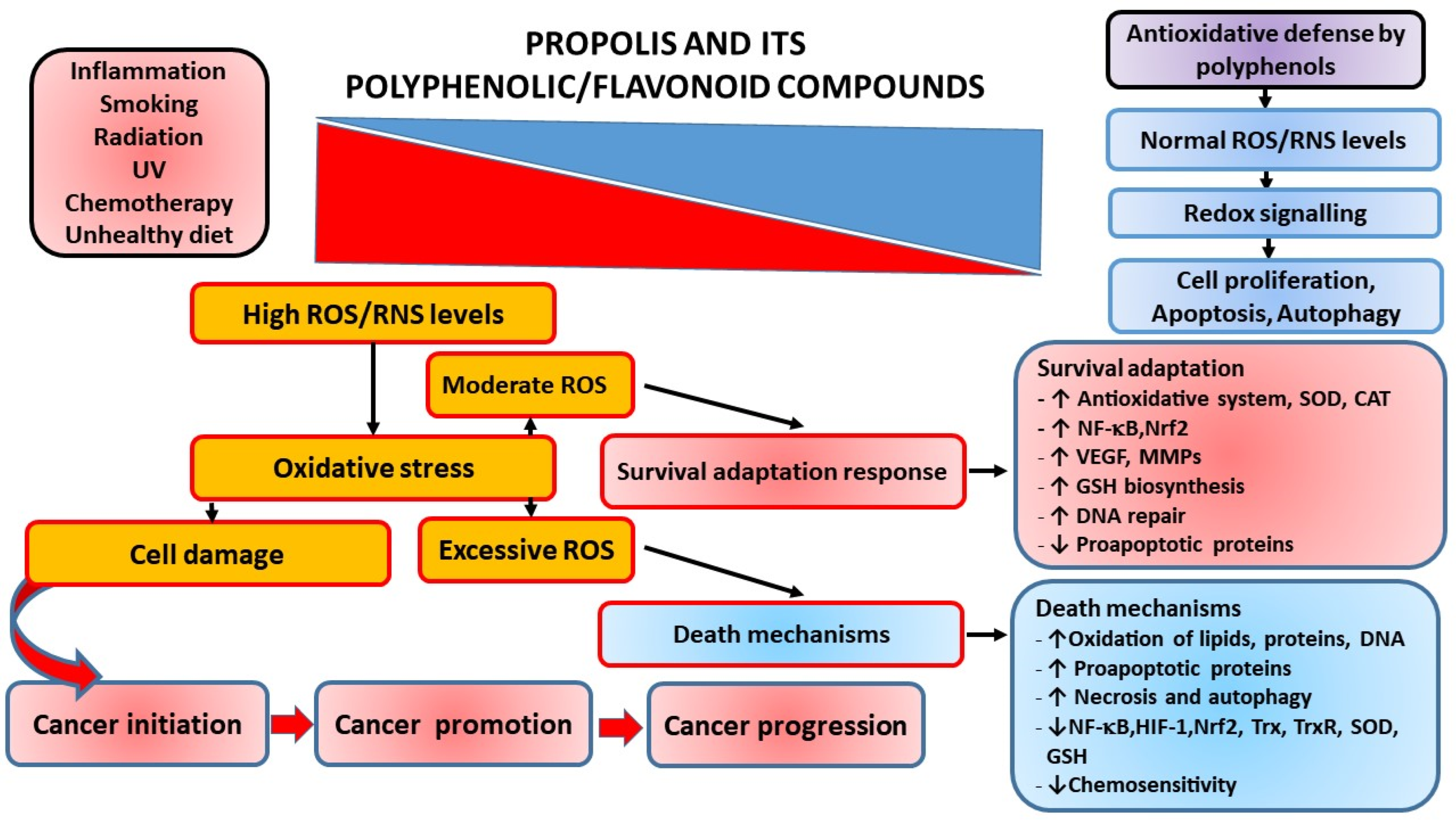

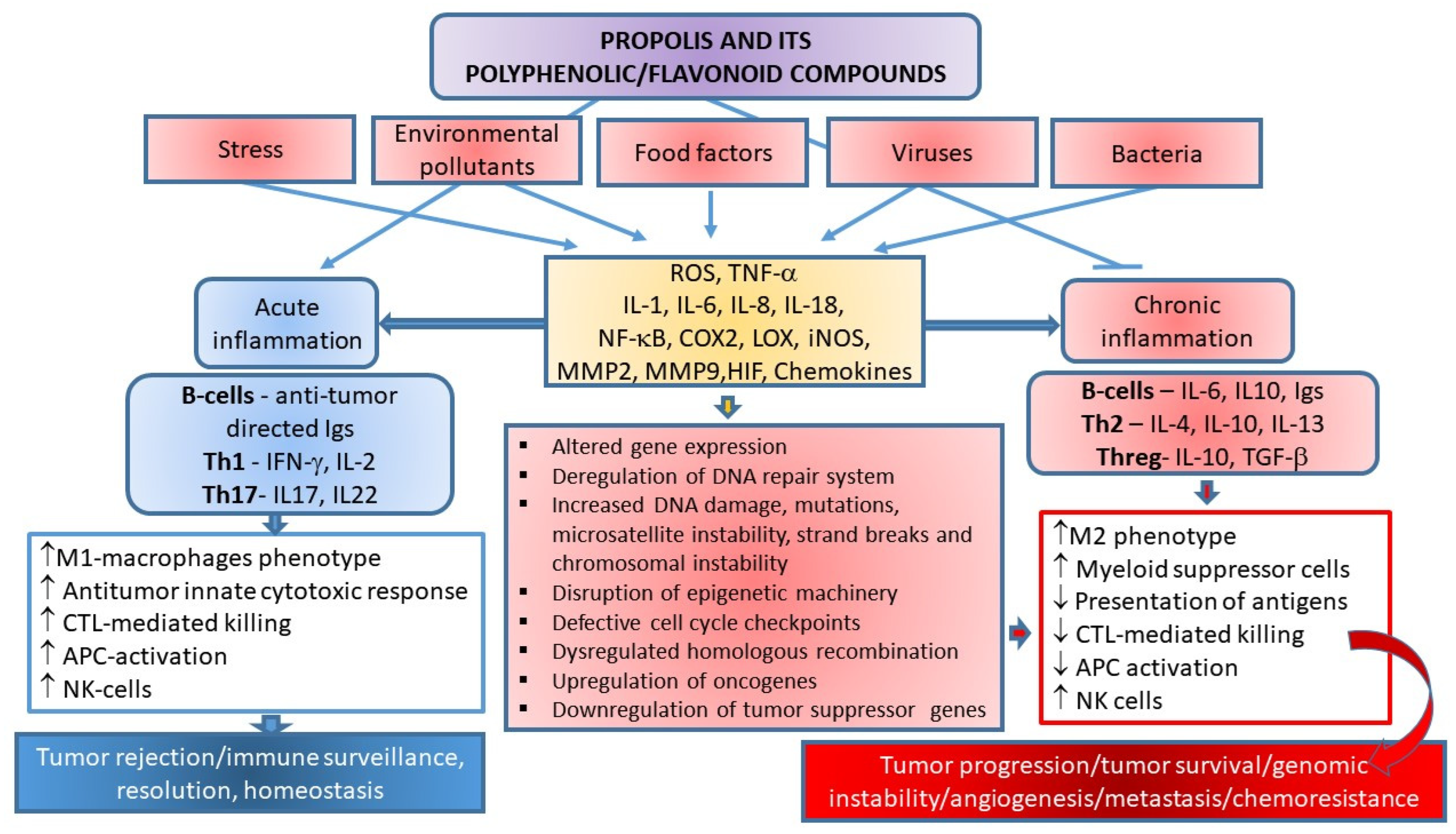
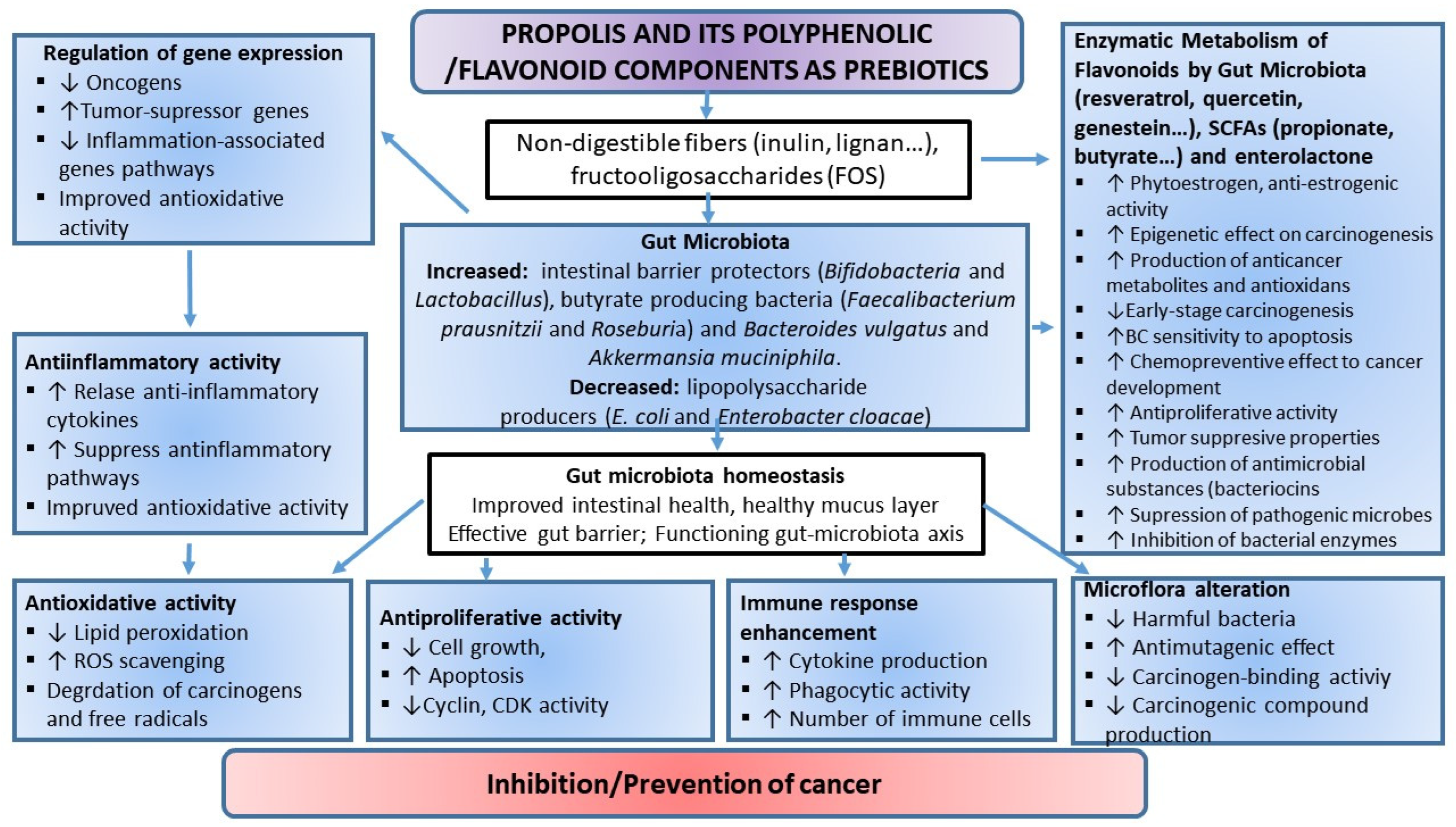
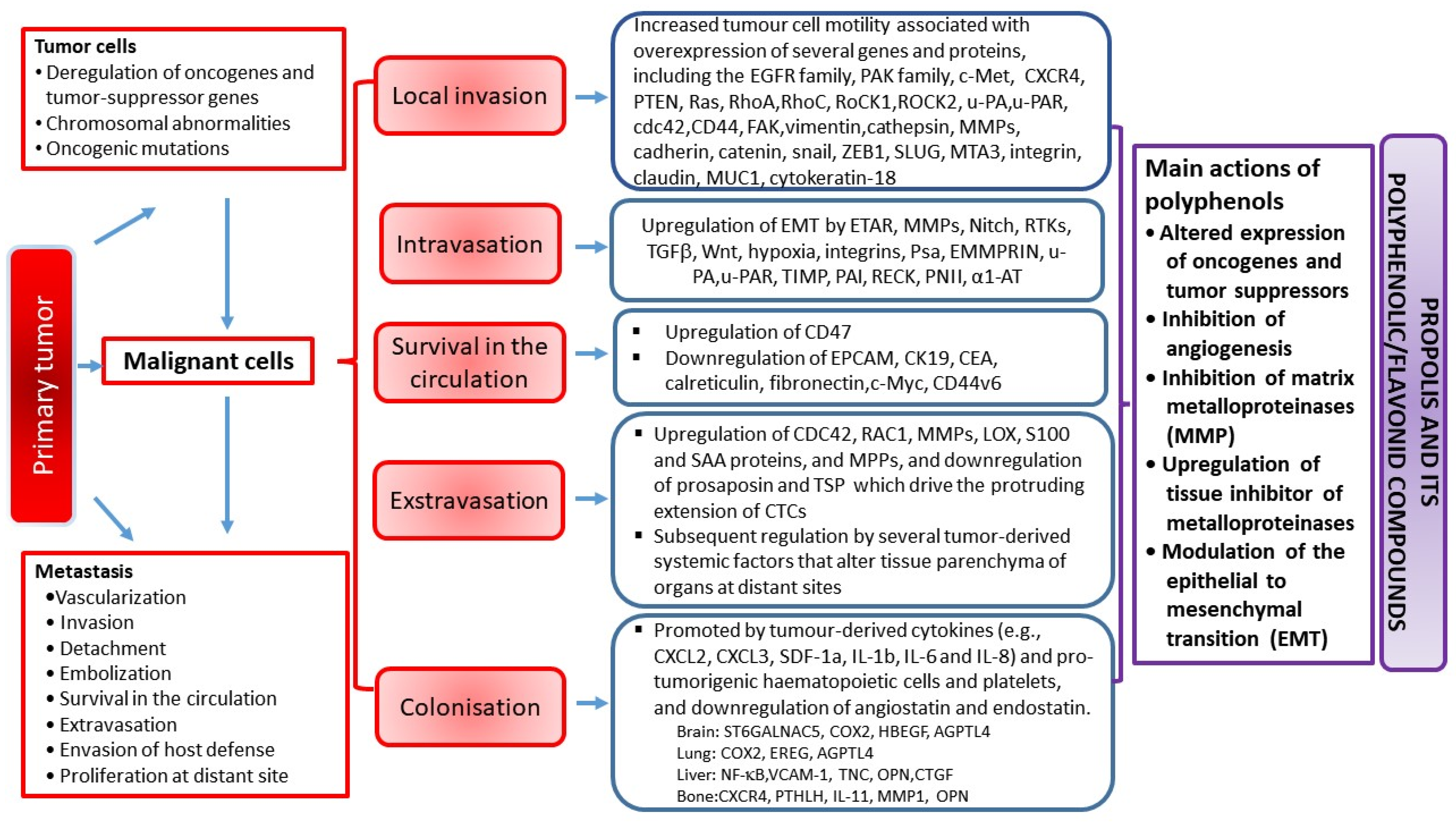
| Propolis Type | Geographic Origin | Plant Source | Typical Chemical Constituents |
|---|---|---|---|
| Poplar propolis | Europe, North America, non-tropical regions of Asia | Populus spp. (most often P. nigra L.) | pinocembrin, pinobanksin, pinobanksin-3-O-acetate, chrysin, galangin, phenolic acids, and their esters |
| Birch propolis | Russia | Betula verucosa Ehrh. | acacetin, apigenin, ermanin, rhamnocitrin, kaempferid, α-acetoxybetulenol, cinnamic acids, phenylpropanoid sesquiterpenols, |
| Green (Alecrim) propolis | Brazil | Baccharis ssp. (most often B. dracunculifolia DC.) | prenylated p-coumaric acids and o-hydroxy-acetophenone, labdane, diterpenic acids |
| Red propolis | Cuba, Mexico, Brazil, Venezuela, Amazon | Clussia ssp.(?) Clusia flower Dalbergia ecastaphyllum | phenylpropene derivative elemicin, triterpenic alcohol β-amyrin, prenylated benzophenones, polyprenylated benzophenones, formononetin, isoliquiritigenin, liquiritigenin, medicarpin, and biochanin A |
| Mediterranean propolis | Greece, Malta, Crete, Cyprus, Turkey, Algeria, Southern Italy | Cupressaceae/Juniperus/Pinus family), Conifer spp., Ferula communis, Castanea sativa, Cistus spp., Quercus ilex L., Fraxinus ornus L., and Olea europaea L. | diterpenes, communic, cupressic and isocupressic acids, totarol, labdane, abietane diterpenes, clerodane, pinobanksin esters, anthraquinones, esters of caffeic and ferulic acids, |
| “Canarian” propolis | Canarian Islands | unknown | furoruran lignans (sesamin, episesamin, methyl xanthoxylol, aschantin, sesartenin, and yangambin), sesquiterpenoids, spatulenol, nerolidol |
| “Pacific” propolis | Okinawa, Taiwan, Japan | Macaranga plants, Macaranga tanarius | C-prenyl-flavanones prenylflavonoids, more specifically isonymphaeol-B, nymphaeol-A, nymphaeol-B, nymphaeol-C, propolins, 3′-geranyl-naringenin |
| Stages of Carcinogenesis | Mechanisms of Carcinogenesis | Action of Dietary Polyphenolics/Flavonoid Components from Propolis: |
|---|---|---|
| 1. Initiation stage | Mutagens, activation of carcinogens⇒ DNA damage⇒ mutations | Antimutagenic effect Carcinogen metabolism ↓ Phase I metabolizing enzymes (CYP1A activity)
|
| Epigenetic modifications | Demethylation, re-expression of TSGs, HDAC inhibition, regulation of expression of non-coding miRNA ↓ DNMT, HDAC ↑ HAT
| |
| 2. Promotion stage | Tumor promotors
| Antioxidative mechanisms ↑ Scavenging of OH●, hydrogen peroxide (H2O2), superoxide anions (O2−●), perhydroxyl radical (HO2●), ROO● ↑ Gap-junctional intracellular communication Anti-inflammatory mechanisms ↓ Prooxidant enzymes (xanthine oxidase (XO), cyclooxygenase (COX), lipoxygenase (LOX)). ↓ NF-κB |
| 3. Tumor progression stage | Uncontrolled cell growth | Signal transduction pathways ↓EGF/EGFR signaling ↓GSK3β (Wnt-signaling) ↓Polyamine metabolism (PKC, ODC) Inhibition of cell growth ↓Notch, Wnt, PI3K/Akt, NF-κB, AP-1 ↑p53, p21Cip1/waf1, p27 ↑cyclins-d1, E, B
Induction of apoptosis
|
| Angiogenesis and Metastasis | ↑Innate immunity ↑Cell-cell attachment ↓Cell migration ↓Cell-matrix adhesion ↓Angiogenesis
|
| Mechanisms | Effectiveness Based On: |
|---|---|
| Antioxidant activity | Antioxidative effect of flavonoids in biological system is related to:
|
| Inhibition of nitrosation and nitration |
|
| Reduction in iron ions | Iron reduction has multiple anticancer actions, including:
|
| Anti-inflammatory effects | Anti-inflammatory effect of flavonoids is related to: Antioxidant activity
Regulation of inflammatory cells
Regulation of proinflammatory enzymes
Modulation of proinflammatory mediators
Modulation of pro-inflammatory gene expression
Inhibition of the Toll-like receptor 4 (TLR-4) signaling pathway |
| Antimutagenic mechanisms | Anti-mutagenic effect of flavonoids is related to: Extracellular mechanisms
|
| Process of detoxification by fibers |
|
| Enzyme inhibition | Enzyme inhibition effect of flavonoids is related to:
|
| Enzyme induction and enhanced detoxification | Enzymes included in this reaction are:
|
| Inhibition of signal transduction pathways | Regulate the signal transduction pathways including:
|
| Inhibition of cell proliferation | Suppression of cancer stem cells self-renewal, progenitor formation, and clonal growth Telomerase inhibition
The disruption of microtubules in mitosis
Inhibition of topoisomerase I and II Proteasome inhibition Cell cycle inhibition
Inactivation of prooxidative enzymes
Inhibition of ornithine decarboxylase and polyamines synthesis Modulation of signal transduction pathways Inhibition of various protein kinases:
Inhibition of DNA synthesis enzymes
|
| Induction of cell differentiation |
|
| Inhibition of oncogene expression |
|
| Induction of tumor suppressor gene expression |
|
| Induction of cell-cycle arrest |
|
| Induction of apoptosis | By inhibition of heat shock proteins Topoisomerase-mediated apoptosis
Mitochondrial toxin-mediated apoptosis Oxidative stress-induced apoptosis
Other mechanisms for inducing apoptosis
|
| Enhancement of immune functions and surveillance |
|
| Antiangiogenesis |
|
| Overcoming resistance to cancer therapy |
|
| Interaction with cellular drug transport systems |
|
| Inhibition of NF-κB | Inhibition of NF-κB leads to negative effects:
|
| Inhibition of cell adhesion and invasion |
|
| Inhibitors of metastasis |
|
| Disruption of tumor cell glycolytic metabolism |
|
| Receptor binding | High concentrations could modulate receptor or enzyme activity in vivo:
|
| Prevention of DNA adduct formation or DNA intercalation |
|
| Regulation of steroid hormone metabolism |
|
| Effects on biomarkers in tumor promotion |
|
| Prooxidative effect | Prooxidative effect of flavonoids is related to:
|
| Antibacterial, antiviral, and antiparasitic activity |
|
| The main mechanisms of antiviral, antibacterial, and antiparasitic action |
|
| Beneficial effect on microbiota activity | Direct effects of flavonoids are related to:
Indirect effects of microbiota on cancer are related to:
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oršolić, N.; Jazvinšćak Jembrek, M. Molecular and Cellular Mechanisms of Propolis and Its Polyphenolic Compounds against Cancer. Int. J. Mol. Sci. 2022, 23, 10479. https://doi.org/10.3390/ijms231810479
Oršolić N, Jazvinšćak Jembrek M. Molecular and Cellular Mechanisms of Propolis and Its Polyphenolic Compounds against Cancer. International Journal of Molecular Sciences. 2022; 23(18):10479. https://doi.org/10.3390/ijms231810479
Chicago/Turabian StyleOršolić, Nada, and Maja Jazvinšćak Jembrek. 2022. "Molecular and Cellular Mechanisms of Propolis and Its Polyphenolic Compounds against Cancer" International Journal of Molecular Sciences 23, no. 18: 10479. https://doi.org/10.3390/ijms231810479
APA StyleOršolić, N., & Jazvinšćak Jembrek, M. (2022). Molecular and Cellular Mechanisms of Propolis and Its Polyphenolic Compounds against Cancer. International Journal of Molecular Sciences, 23(18), 10479. https://doi.org/10.3390/ijms231810479







