Singularity and Commonality in Response to SARS-CoV-2 in Lung and Colon Cell Models
Abstract
:1. Introduction
2. Results
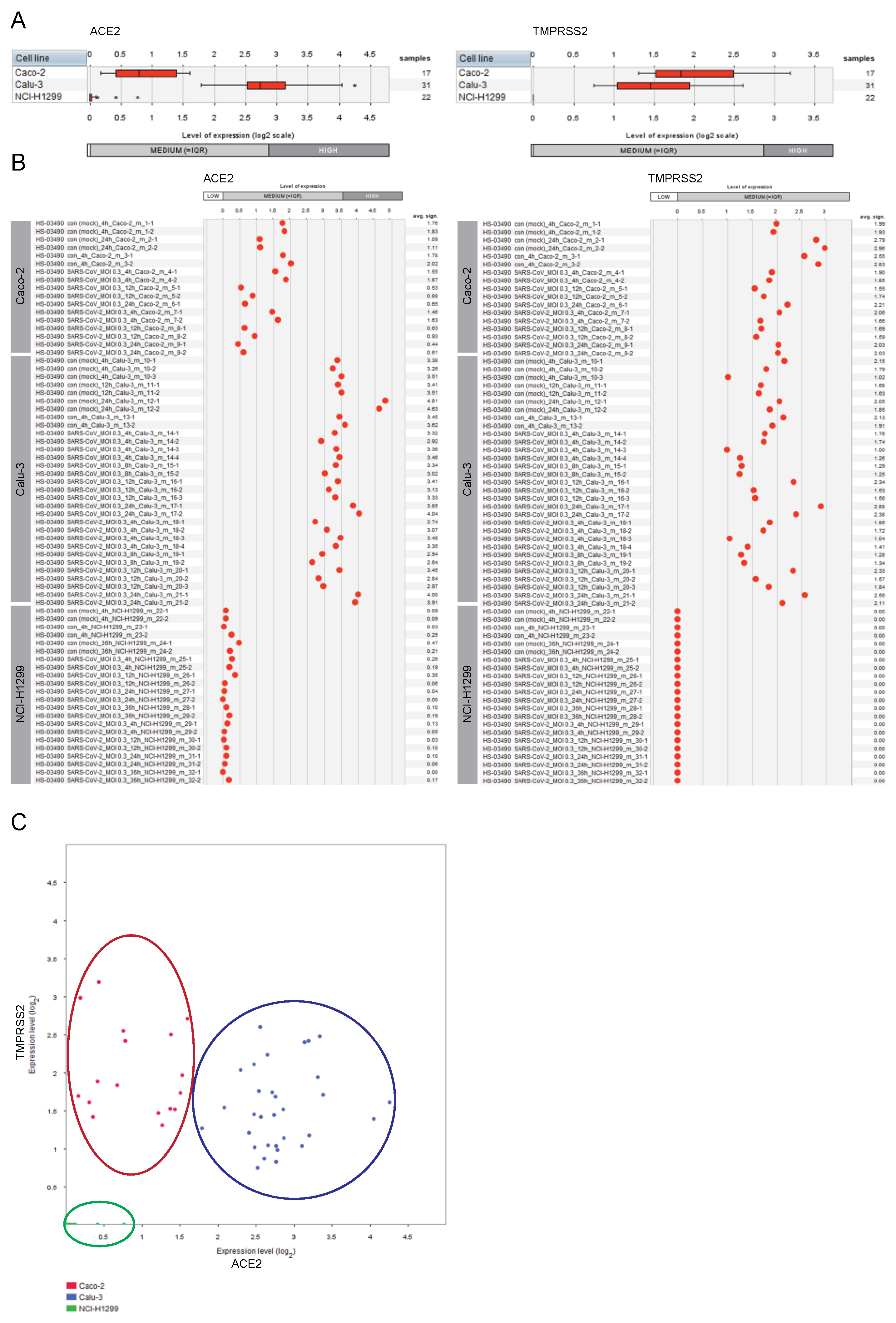
2.1. Expression of ACE2 and TMPRSS2 in Lung and Colon Cell Lines
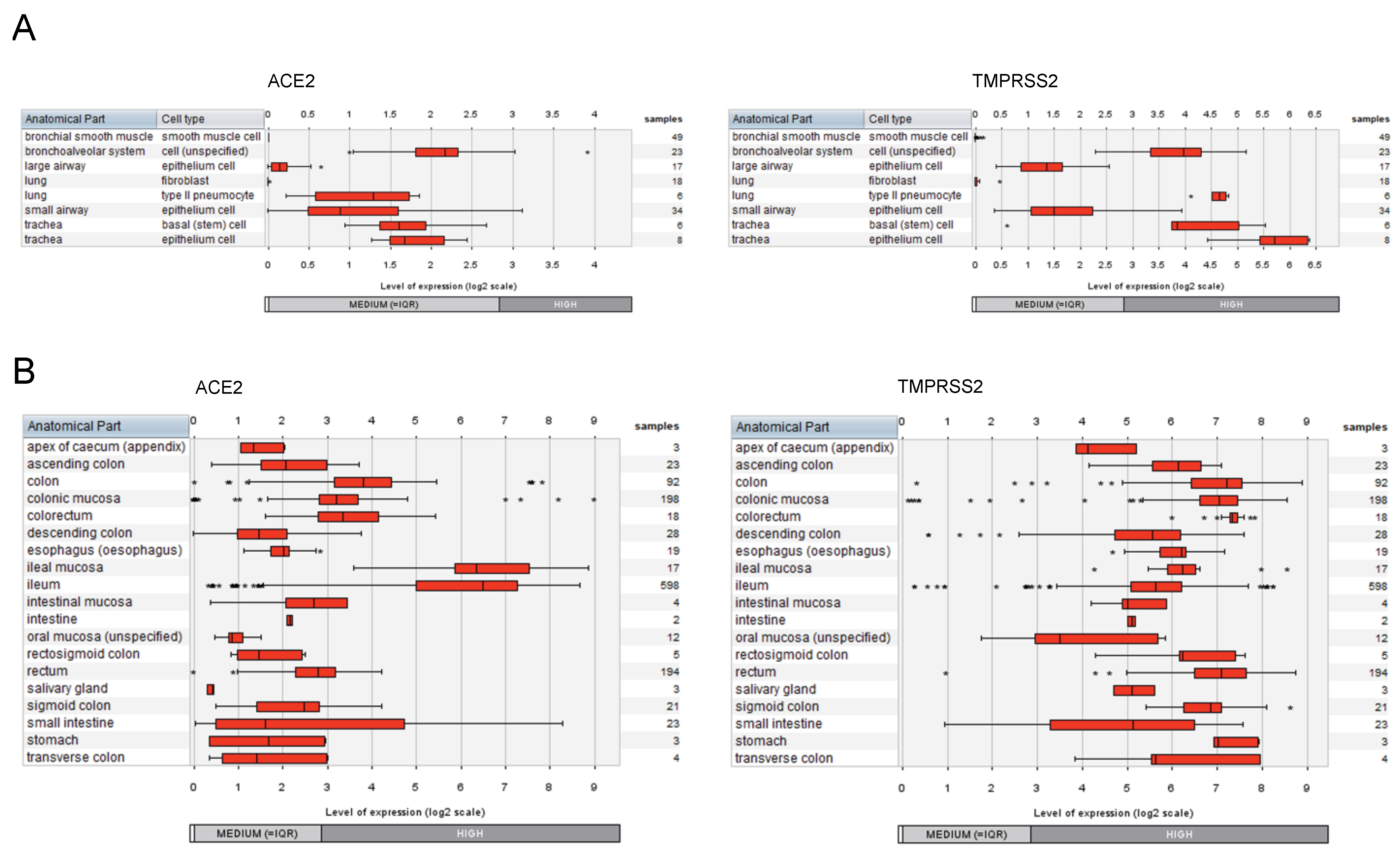
2.2. Expression of ACE2 and TMPRSS2 in Cell Types of the Respiratory System and the Gastrointestinal Tract
2.3. Understanding the Pathobiology of COVID-19 by Applying a Multi-Modular Integrative Approach
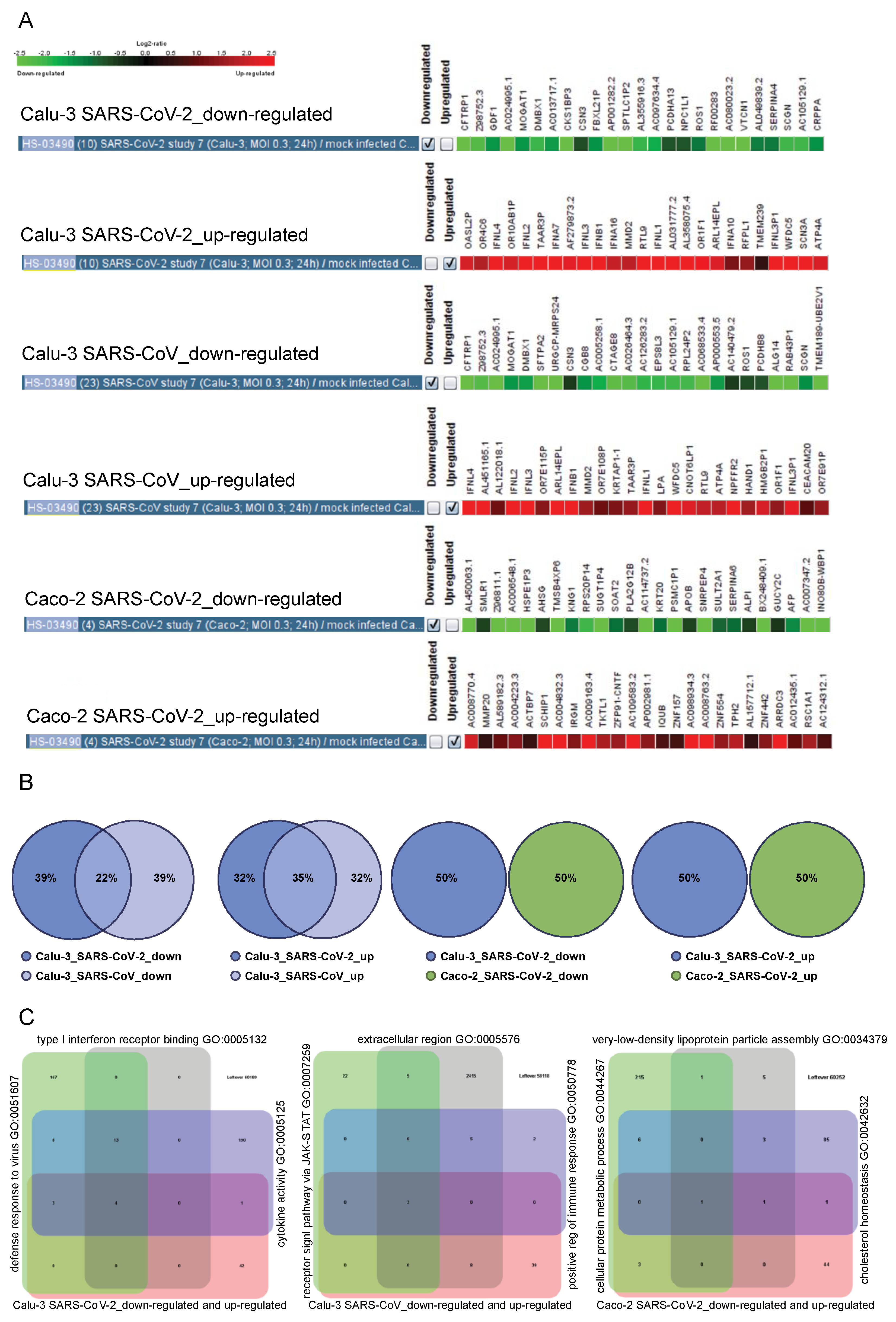
2.4. Tissue-Specific Response to SARS-CoV and SARS-CoV-2
2.5. Singularity and Commonality in Canonical Pathways and Upstream Regulators Activated in Response to SARS-CoV-2 and SARS-CoV in Lung and Colon Cell Models
2.6. The Cell Type-Specific Gene Signatures
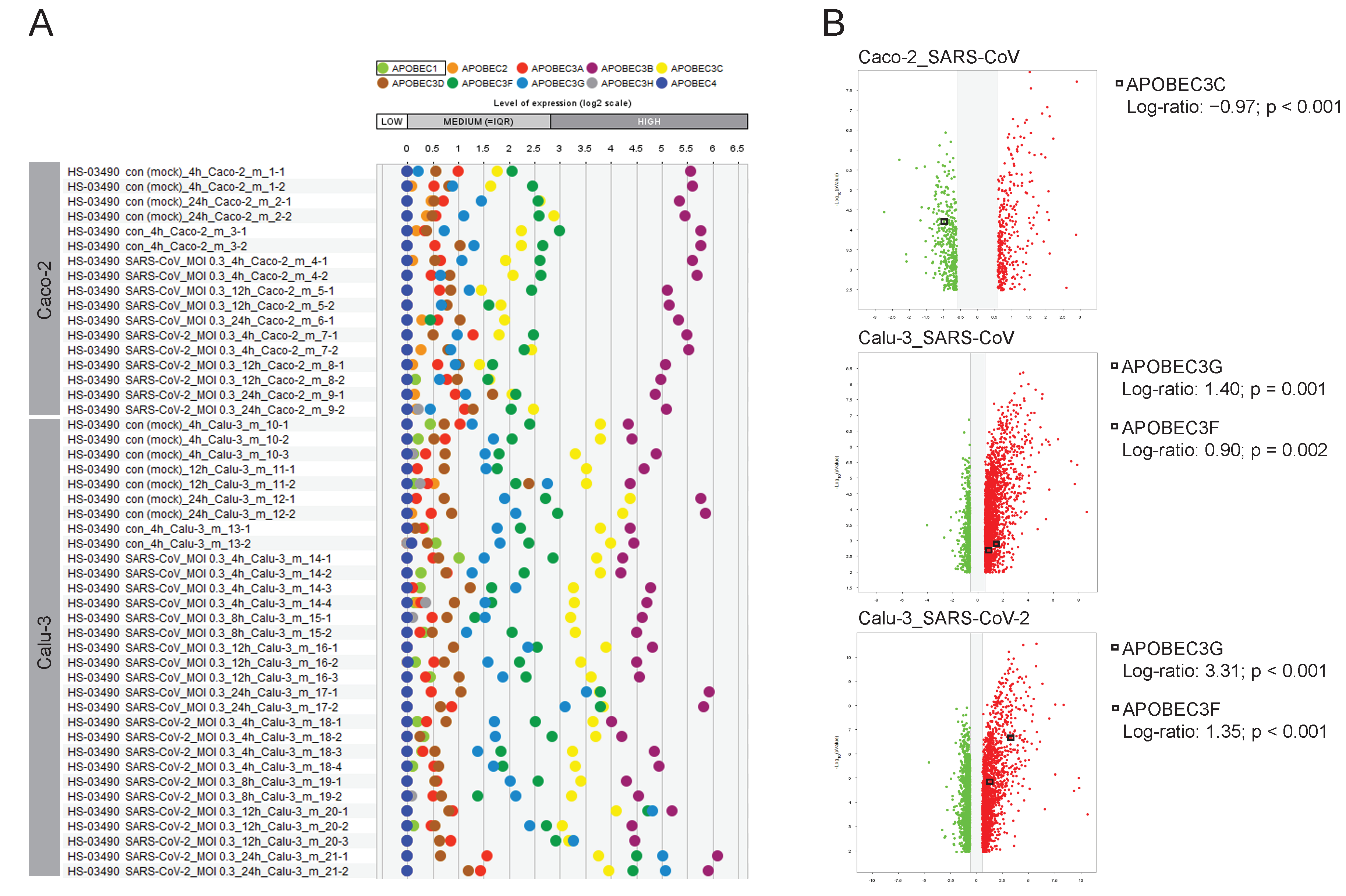
2.7. The Road Map of APOBECs in Caco-2 and Calu-3 Models and the Upregulation of APOBEC3G
3. Discussion
4. Materials and Methods
4.1. Comprehensive Analysis of Transcriptomic Data
4.2. Core Analysis for Canonical Pathways and Upstream Regulators
4.3. Comparative Analysis and Data Visualization
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACE2 | angiotensin-converting enzyme 2 |
| AID | activation-induced cytidine deaminase |
| APOBEC | apolipoprotein B mRNA editing enzyme catalytic subunit |
| C-to-U | cytosine-to-uracil |
| COVID-19 | Coronavirus disease 19 |
| CRISPR-Cas9 | clustered regularly interspaced short palindromic repeats-CRISPR-associated protein 9 |
| EGR | early growth response |
| ERK | extracellular signal-regulated kinase |
| GI | gastrointestinal |
| HDL | high-density lipoproteins |
| IFNG | interferon gamma |
| IPA | Ingenuity Pathway Analysis |
| JAK-STAT | janus kinases-signal transducer and activator of transcription proteins |
| LDL | low-density lipoproteins |
| MAPK | mitogen-activated protein kinase |
| MEK | also known as MAP2K, mitogen-activated protein kinase kinase |
| MERS-CoV | Middle East respiratory syndrome coronavirus |
| mRNASeq | mRNA sequencing |
| MSP | macrophage-stimulating protein |
| NFkappaB | nuclear factor kappa-light-chain-enhancer of activated B cells |
| PDGF | platelet derived growth factor |
| RON | recepteur d’origine nantais |
| SARS-CoV | severe acute respiratory syndrome coronavirus |
| S | spike |
| TMPRSS2 | transmembrane serine protease 2 |
| TNF | tumor necrosis factor |
| TNFR2 | tumor necrosis factor receptor 2 |
| TP63 | tumor protein p63 |
| UNCOVIDING | Understanding COVID-19 by Integrative Data Mining |
References
- Fung, T.S.; Liu, D.X. Human Coronavirus: Host-Pathogen Interaction. Annu. Rev. Microbiol. 2019, 73, 529–557. [Google Scholar] [CrossRef] [PubMed]
- Fehr, A.R.; Perlman, S. Coronaviruses: An overview of their replication and pathogenesis. Methods Mol. Biol. 2015, 1282, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Park, S.E. Epidemiology, virology, and clinical features of severe acute respiratory syndrome -coronavirus-2 (SARS-CoV-2; Coronavirus Disease-19). Clin. Exp. Pediatr. 2020, 63, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Cook, A.R.; Lim, J.T.; Sun, Y.; Dickens, B.L. A Systematic Review of COVID-19 Epidemiology Based on Current Evidence. J. Clin. Med. 2020, 9, 967. [Google Scholar] [CrossRef]
- Lu, R.; Zhao, X.; Li, J.; Niu, P.; Yang, B.; Wu, H.; Wang, W.; Song, H.; Huang, B.; Zhu, N.; et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding. Lancet 2020, 395, 565–574. [Google Scholar] [CrossRef]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Kruger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280.e278. [Google Scholar] [CrossRef]
- Ou, X.; Liu, Y.; Lei, X.; Li, P.; Mi, D.; Ren, L.; Guo, L.; Guo, R.; Chen, T.; Hu, J.; et al. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat. Commun. 2020, 11, 1620. [Google Scholar] [CrossRef]
- Singh, M.; Bansal, V.; Feschotte, C. A Single-Cell RNA Expression Map of Human Coronavirus Entry Factors. Cell Rep 2020, 32, 108175. [Google Scholar] [CrossRef]
- Li, M.Y.; Li, L.; Zhang, Y.; Wang, X.S. Expression of the SARS-CoV-2 cell receptor gene ACE2 in a wide variety of human tissues. Infect. Dis. Poverty 2020, 9, 45. [Google Scholar] [CrossRef]
- Hikmet, F.; Mear, L.; Edvinsson, A.; Micke, P.; Uhlen, M.; Lindskog, C. The protein expression profile of ACE2 in human tissues. Mol. Syst. Biol. 2020, 16, e9610. [Google Scholar] [CrossRef]
- Gupta, A.; Madhavan, M.V.; Sehgal, K.; Nair, N.; Mahajan, S.; Sehrawat, T.S.; Bikdeli, B.; Ahluwalia, N.; Ausiello, J.C.; Wan, E.Y.; et al. Extrapulmonary manifestations of COVID-19. Nat. Med. 2020, 26, 1017–1032. [Google Scholar] [CrossRef]
- Sarkesh, A.; Daei Sorkhabi, A.; Sheykhsaran, E.; Alinezhad, F.; Mohammadzadeh, N.; Hemmat, N.; Bannazadeh Baghi, H. Extrapulmonary Clinical Manifestations in COVID-19 Patients. Am. J. Trop. Med. Hyg. 2020, 103, 1783–1796. [Google Scholar] [CrossRef]
- Mao, R.; Qiu, Y.; He, J.S.; Tan, J.Y.; Li, X.H.; Liang, J.; Shen, J.; Zhu, L.R.; Chen, Y.; Iacucci, M.; et al. Manifestations and prognosis of gastrointestinal and liver involvement in patients with COVID-19: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2020, 5, 667–678. [Google Scholar] [CrossRef]
- Redd, W.D.; Zhou, J.C.; Hathorn, K.E.; McCarty, T.R.; Bazarbashi, A.N.; Thompson, C.C.; Shen, L.; Chan, W.W. Prevalence and Characteristics of Gastrointestinal Symptoms in Patients With Severe Acute Respiratory Syndrome Coronavirus 2 Infection in the United States: A Multicenter Cohort Study. Gastroenterology 2020, 159, 765–767.e762. [Google Scholar] [CrossRef]
- Mechtcheriakova, D.; Svoboda, M.; Meshcheryakova, A.; Jensen-Jarolim, E. Activation-induced cytidine deaminase (AID) linking immunity, chronic inflammation, and cancer. Cancer Immunol. Immunother. 2012, 61, 1591–1598. [Google Scholar] [CrossRef]
- Moris, A.; Murray, S.; Cardinaud, S. AID and APOBECs span the gap between innate and adaptive immunity. Front. Microbiol. 2014, 5, 534. [Google Scholar] [CrossRef]
- Cervantes-Gracia, K.; Gramalla-Schmitz, A.; Weischedel, J.; Chahwan, R. APOBECs orchestrate genomic and epigenomic editing across health and disease. Trends Genet. 2021, 37, 1028–1043. [Google Scholar] [CrossRef]
- Olson, M.E.; Harris, R.S.; Harki, D.A. APOBEC Enzymes as Targets for Virus and Cancer Therapy. Cell. Chem. Biol. 2018, 25, 36–49. [Google Scholar] [CrossRef]
- Vieira, V.C.; Soares, M.A. The role of cytidine deaminases on innate immune responses against human viral infections. Biomed. Res. Int. 2013, 2013, 683095. [Google Scholar] [CrossRef]
- Stavrou, S.; Ross, S.R. APOBEC3 Proteins in Viral Immunity. J. Immunol. 2015, 195, 4565–4570. [Google Scholar] [CrossRef] [Green Version]
- Meshcheryakova, A.; Pietschmann, P.; Zimmermann, P.; Rogozin, I.B.; Mechtcheriakova, D. AID and APOBECs as Multifaceted Intrinsic Virus-Restricting Factors: Emerging Concepts in the Light of COVID-19. Front. Immunol. 2021, 12, 690416. [Google Scholar] [CrossRef]
- Wyler, E.; Mosbauer, K.; Franke, V.; Diag, A.; Gottula, L.T.; Arsie, R.; Klironomos, F.; Koppstein, D.; Honzke, K.; Ayoub, S.; et al. Transcriptomic profiling of SARS-CoV-2 infected human cell lines identifies HSP90 as target for COVID-19 therapy. iScience 2021, 24, 102151. [Google Scholar] [CrossRef]
- Meshcheryakova, A.; Zimmermann, P.; Ecker, R.; Mungenast, F.; Heinze, G.; Mechtcheriakova, D. An Integrative MuSiCO Algorithm: From the Patient-Specific Transcriptional Profiles to Novel Checkpoints in Disease Pathobiology. In Systems Biology; Springer: Cham, Switzerland, 2018; pp. 351–372. [Google Scholar] [CrossRef]
- Meshcheryakova, A.; Svoboda, M.; Jaritz, M.; Mungenast, F.; Salzmann, M.; Pils, D.; Cacsire Castillo-Tong, D.; Hager, G.; Wolf, A.; Braicu, E.I.; et al. Interrelations of Sphingolipid and Lysophosphatidate Signaling with Immune System in Ovarian Cancer. Comput. Struct. Biotechnol. J. 2019, 17, 537–560. [Google Scholar] [CrossRef]
- Svoboda, M.; Meshcheryakova, A.; Heinze, G.; Jaritz, M.; Pils, D.; Castillo-Tong, D.C.; Hager, G.; Thalhammer, T.; Jensen-Jarolim, E.; Birner, P.; et al. AID/APOBEC-network reconstruction identifies pathways associated with survival in ovarian cancer. BMC Genomics 2016, 17, 643. [Google Scholar] [CrossRef] [PubMed]
- Meshcheryakova, A.; Mungenast, F.; Ecker, R.; Mechtcheriakova, D. Tissue image cytometry. In Imaging Modalities for Biological and Preclinical Research: A Compendium, Volume 1: Part I: Ex Vivo Biological Imaging; IOP Publishing: Bristol, UK, 2021. [Google Scholar] [CrossRef]
- Mungenast, F.; Meshcheryakova, A.; Beer, A.; Salzmann, M.; Tamandl, D.; Gruenberger, T.; Pietschmann, P.; Koperek, O.; Birner, P.; Kirsch, I.; et al. The Immune Phenotype of Isolated Lymphoid Structures in Non-Tumorous Colon Mucosa Encrypts the Information on Pathobiology of Metastatic Colorectal Cancer. Cancers 2020, 12, 3117. [Google Scholar] [CrossRef] [PubMed]
- Byrne, A.L.; Marais, B.J.; Mitnick, C.D.; Lecca, L.; Marks, G.B. Tuberculosis and chronic respiratory disease: A systematic review. Int. J. Infect. Dis. 2015, 32, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, C.; Crimi, C.; Vatrella, A.; Tinello, C.; Terracciano, R.; Pelaia, G. Molecular Targets for Biological Therapies of Severe Asthma. Front. Immunol. 2020, 11, 603312. [Google Scholar] [CrossRef] [PubMed]
- Cukic, V.; Lovre, V.; Dragisic, D.; Ustamujic, A. Asthma and Chronic Obstructive Pulmonary Disease (COPD)—Differences and Similarities. Mater. Sociomed. 2012, 24, 100–105. [Google Scholar] [CrossRef] [Green Version]
- Gajendran, M.; Loganathan, P.; Catinella, A.P.; Hashash, J.G. A comprehensive review and update on Crohn’s disease. Dis. Mon. 2018, 64, 20–57. [Google Scholar] [CrossRef]
- Gajendran, M.; Loganathan, P.; Jimenez, G.; Catinella, A.P.; Ng, N.; Umapathy, C.; Ziade, N.; Hashash, J.G. A comprehensive review and update on ulcerative colitis. Dis. Mon. 2019, 65, 100851. [Google Scholar] [CrossRef]
- Sheka, A.C.; Adeyi, O.; Thompson, J.; Hameed, B.; Crawford, P.A.; Ikramuddin, S. Nonalcoholic Steatohepatitis: A Review. JAMA 2020, 323, 1175–1183. [Google Scholar] [CrossRef]
- Wu, Y.; Guo, C.; Tang, L.; Hong, Z.; Zhou, J.; Dong, X.; Yin, H.; Xiao, Q.; Tang, Y.; Qu, X.; et al. Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. Lancet Gastroenterol. Hepatol. 2020, 5, 434–435. [Google Scholar] [CrossRef]
- Xiao, F.; Tang, M.; Zheng, X.; Liu, Y.; Li, X.; Shan, H. Evidence for Gastrointestinal Infection of SARS-CoV-2. Gastroenterology 2020, 158, 1831–1833.e3. [Google Scholar] [CrossRef]
- Qian, Q.; Fan, L.; Liu, W.; Li, J.; Yue, J.; Wang, M.; Ke, X.; Yin, Y.; Chen, Q.; Jiang, C. Direct Evidence of Active SARS-CoV-2 Replication in the Intestine. Clin. Infect. Dis. 2021, 73, 361–366. [Google Scholar] [CrossRef]
- Kocar, E.; Rezen, T.; Rozman, D. Cholesterol, lipoproteins, and COVID-19: Basic concepts and clinical applications. Biochim. Biophys. Acta. Mol. Cell Biol. Lipids. 2021, 1866, 158849. [Google Scholar] [CrossRef]
- Masana, L.; Correig, E.; Ibarretxe, D.; Anoro, E.; Arroyo, J.A.; Jerico, C.; Guerrero, C.; Miret, M.; Naf, S.; Pardo, A.; et al. Low HDL and high triglycerides predict COVID-19 severity. Sci. Rep. 2021, 11, 7217. [Google Scholar] [CrossRef]
- Heldin, C.H.; Westermark, B. Mechanism of action and in vivo role of platelet-derived growth factor. Physiol. Rev. 1999, 79, 1283–1316. [Google Scholar] [CrossRef] [PubMed]
- Hirota, J.A.; Ask, K.; Farkas, L.; Smith, J.A.; Ellis, R.; Rodriguez-Lecompte, J.C.; Kolb, M.; Inman, M.D. In vivo role of platelet-derived growth factor-BB in airway smooth muscle proliferation in mouse lung. Am. J. Respir. Cell Mol. Biol. 2011, 45, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Mechtcheriakova, D.; Schabbauer, G.; Lucerna, M.; Clauss, M.; De Martin, R.; Binder, B.R.; Hofer, E. Specificity, diversity, and convergence in VEGF and TNF-alpha signaling events leading to tissue factor up-regulation via EGR-1 in endothelial cells. FASEB J. 2001, 15, 230–242. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Simoneau, C.R.; Kulsuptrakul, J.; Bouhaddou, M.; Travisano, K.A.; Hayashi, J.M.; Carlson-Stevermer, J.; Zengel, J.R.; Richards, C.M.; Fozouni, P.; et al. Genetic Screens Identify Host Factors for SARS-CoV-2 and Common Cold Coronaviruses. Cell 2021, 184, 106–119.e114. [Google Scholar] [CrossRef] [PubMed]
- Fehrholz, M.; Kendl, S.; Prifert, C.; Weissbrich, B.; Lemon, K.; Rennick, L.; Duprex, P.W.; Rima, B.K.; Koning, F.A.; Holmes, R.K.; et al. The innate antiviral factor APOBEC3G targets replication of measles, mumps and respiratory syncytial viruses. J. Gen. Virol. 2012, 93, 565–576. [Google Scholar] [CrossRef]
- Tiwarekar, V.; Wohlfahrt, J.; Fehrholz, M.; Scholz, C.J.; Kneitz, S.; Schneider-Schaulies, J. APOBEC3G-Regulated Host Factors Interfere with Measles Virus Replication: Role of REDD1 and Mammalian TORC1 Inhibition. J. Virol. 2018, 92, e00835-18. [Google Scholar] [CrossRef]
- Yang, H.; Ito, F.; Wolfe, A.D.; Li, S.; Mohammadzadeh, N.; Love, R.P.; Yan, M.; Zirkle, B.; Gaba, A.; Chelico, L.; et al. Understanding the structural basis of HIV-1 restriction by the full length double-domain APOBEC3G. Nat. Commun. 2020, 11, 632. [Google Scholar] [CrossRef] [Green Version]
- Oliveros, J.C. Venny. An Interactive Tool for Comparing Lists with Venn’s Diagrams. Available online: https://bioinfogp.cnb.csic.es/tools/venny/ (accessed on 15 April 2021).



| Top Five Canonical Pathways for Caco-2_SARS-CoV-2 | ||||
|---|---|---|---|---|
| Canonical Pathway | Caco-2_SARS-CoV-2 | Calu-3_SARS-CoV-2 | Caco-2_SARS-CoV | Calu-3_SARS-CoV |
| TNFR2 Signaling | 1 | 36 | 37 | 2 |
| MSP-RON Signaling In Cancer Cells Pathway | 2 | 111 | 16 | 127 |
| Aryl Hydrocarbon Receptor Signaling | 3 | 29 | 5 | 98 |
| Acute Phase Response Signaling | 4 | 18 | 101 | 38 |
| Hepatic Fibrosis Signaling Pathway | 5 | 24 | 6 | 16 |
| Top Five Canonical Pathways for Calu-3_SARS-CoV-2 | ||||
| Canonical Pathway | Caco-2_SARS-CoV-2 | Calu-3_SARS-CoV-2 | Caco-2_SARS-CoV | Calu-3_SARS-CoV |
| Superpathway of Cholesterol Biosynthesis | - | 1 | 173 | 193 |
| Dendritic Cell Maturation | - | 2 | - | 5 |
| Neuroinflammation Signaling Pathway | 107 | 3 | 94 | 1 |
| Antigen Presentation Pathway | - | 4 | - | 25 |
| Role of PRRs in Recognition of Bacteria and Viruses | - | 5 | 148 | 10 |
| Top Five Canonical Pathways for Caco-2_SARS-CoV | ||||
| Canonical Pathway | Caco-2_SARS-CoV-2 | Calu-3_SARS-CoV-2 | Caco-2_SARS-CoV | Calu-3_SARS-CoV |
| Molecular Mechanisms of Cancer | 13 | 53 | 1 | 96 |
| Tec Kinase Signaling | 40 | 61 | 2 | 90 |
| ERK/MAPK Signaling | 8 | 161 | 3 | 128 |
| Tumor Microenvironment Pathway | 18 | 15 | 4 | 11 |
| Aryl Hydrocarbon Receptor Signaling | 3 | 29 | 5 | 98 |
| Top Five Canonical Pathways for Calu-3_SARS-CoV | ||||
| Canonical Pathway | Caco-2_SARS-CoV-2 | Calu-3_SARS-CoV-2 | Caco-2_SARS-CoV | Calu-3_SARS-CoV |
| Neuroinflammation Signaling Pathway | 107 | 3 | 94 | 1 |
| TNFR2 Signaling | 1 | 36 | 37 | 2 |
| Death Receptor Signaling | 34 | 13 | 20 | 3 |
| Activation of IRF by Cytosolic PRRs | 144 | 9 | - | 4 |
| Dendritic Cell Maturation | - | 2 | - | 5 |
| Top Five Upstream Regulators for Caco-2_SARS-CoV-2 | ||||
|---|---|---|---|---|
| Upstream Regulator | Caco-2_SARS-CoV-2 | Calu-3_SARS-CoV-2 | Caco-2_SARS-CoV | Calu-3_SARS-CoV |
| PDGF BB | 1 | 85 | 1 | 56 |
| TNF | 2 | 5 | 9 | 4 |
| PD98059 | 3 | 87 | 3 | 115 |
| U0126 | 4 | 55 | 2 | 62 |
| beta-estradiol | 5 | 10 | 4 | 26 |
| Top Five Upstream Regulators for Calu-3_SARS-CoV-2 | ||||
| Upstream Regulator | Caco-2_SARS-CoV-2 | Calu-3_SARS-CoV-2 | Caco-2_SARS-CoV | Calu-3_SARS-CoV |
| lipopolysaccharide | 43 | 1 | 60 | 2 |
| IFNG | 55 | 2 | 38 | 5 |
| poly rI:rC-RNA | 46 | 3 | 54 | 3 |
| Interferon alpha | 465 | 4 | 644 | 1 |
| TNF | 2 | 5 | 9 | 4 |
| Top Five Upstream Regulators for Caco-2_SARS-CoV | ||||
| Upstream Regulator | Caco-2_SARS-CoV-2 | Calu-3_SARS-CoV-2 | Caco-2_SARS-CoV | Calu-3_SARS-CoV |
| PDGF BB | 1 | 85 | 1 | 56 |
| U0126 | 4 | 55 | 2 | 62 |
| PD98059 | 3 | 87 | 3 | 115 |
| beta-estradiol | 5 | 10 | 4 | 26 |
| TP63 | 41 | 298 | 5 | 182 |
| Top Five Upstream Regulators for Calu-3_SARS-CoV | ||||
| Upstream Regulator | Caco-2_SARS-CoV-2 | Calu-3_SARS-CoV-2 | Caco-2_SARS-CoV | Calu-3_SARS-CoV |
| Interferon alpha | 465 | 4 | 644 | 1 |
| lipopolysaccharide | 43 | 1 | 60 | 2 |
| poly rI:rC-RNA | 46 | 3 | 54 | 3 |
| TNF | 2 | 5 | 9 | 4 |
| IFNG | 55 | 2 | 38 | 5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meshcheryakova, A.; Zimmermann, P.; Salzmann, M.; Pietschmann, P.; Mechtcheriakova, D. Singularity and Commonality in Response to SARS-CoV-2 in Lung and Colon Cell Models. Int. J. Mol. Sci. 2022, 23, 10451. https://doi.org/10.3390/ijms231810451
Meshcheryakova A, Zimmermann P, Salzmann M, Pietschmann P, Mechtcheriakova D. Singularity and Commonality in Response to SARS-CoV-2 in Lung and Colon Cell Models. International Journal of Molecular Sciences. 2022; 23(18):10451. https://doi.org/10.3390/ijms231810451
Chicago/Turabian StyleMeshcheryakova, Anastasia, Philip Zimmermann, Martina Salzmann, Peter Pietschmann, and Diana Mechtcheriakova. 2022. "Singularity and Commonality in Response to SARS-CoV-2 in Lung and Colon Cell Models" International Journal of Molecular Sciences 23, no. 18: 10451. https://doi.org/10.3390/ijms231810451
APA StyleMeshcheryakova, A., Zimmermann, P., Salzmann, M., Pietschmann, P., & Mechtcheriakova, D. (2022). Singularity and Commonality in Response to SARS-CoV-2 in Lung and Colon Cell Models. International Journal of Molecular Sciences, 23(18), 10451. https://doi.org/10.3390/ijms231810451







