Development and Characterization of a Novel Soil Amendment Based on Biomass Fly Ash Encapsulated in Calcium Alginate Microspheres
Abstract
:1. Introduction
2. Results and Discussion
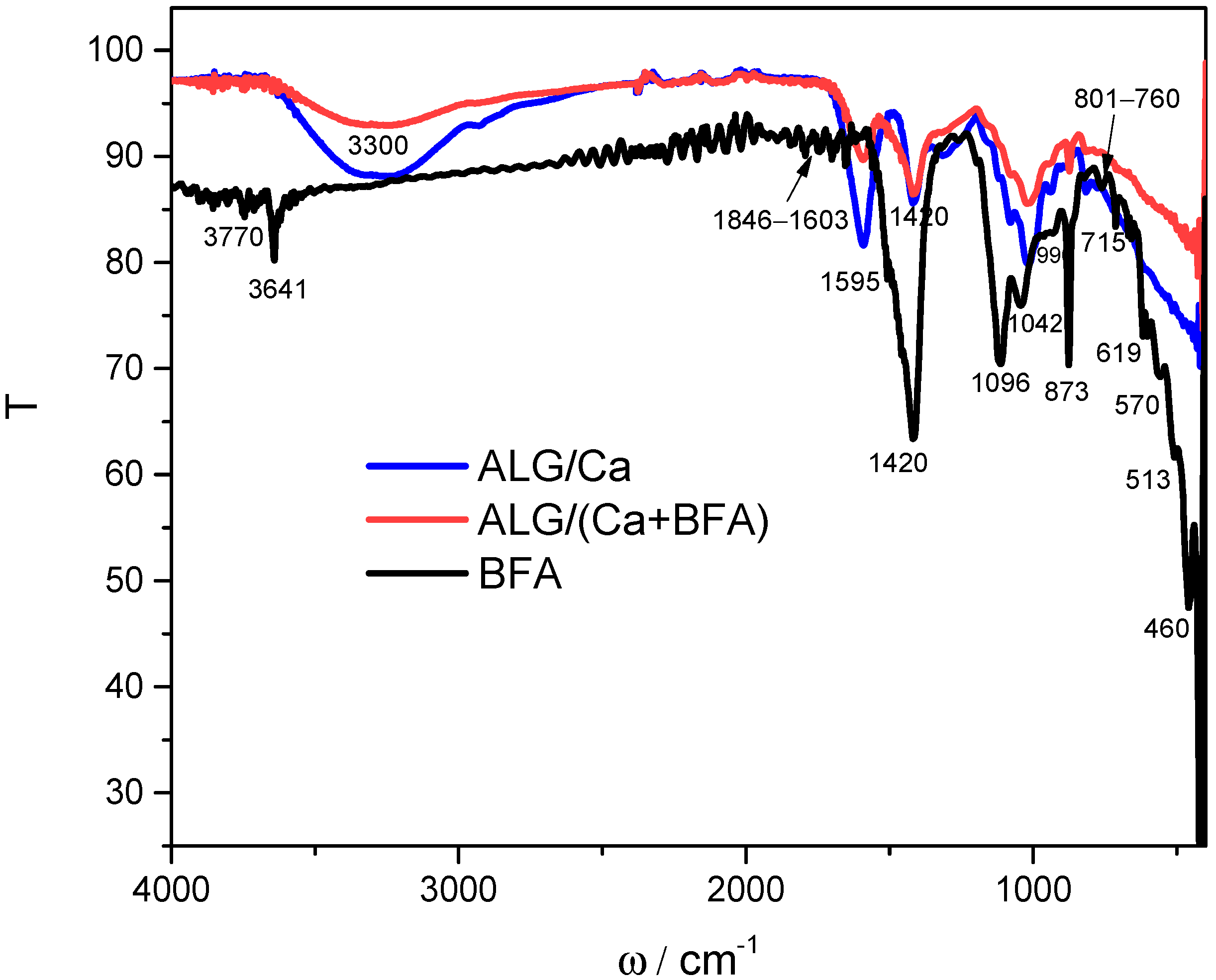
2.1. Molecular Interaction between Constituents in Microsphere Formulation
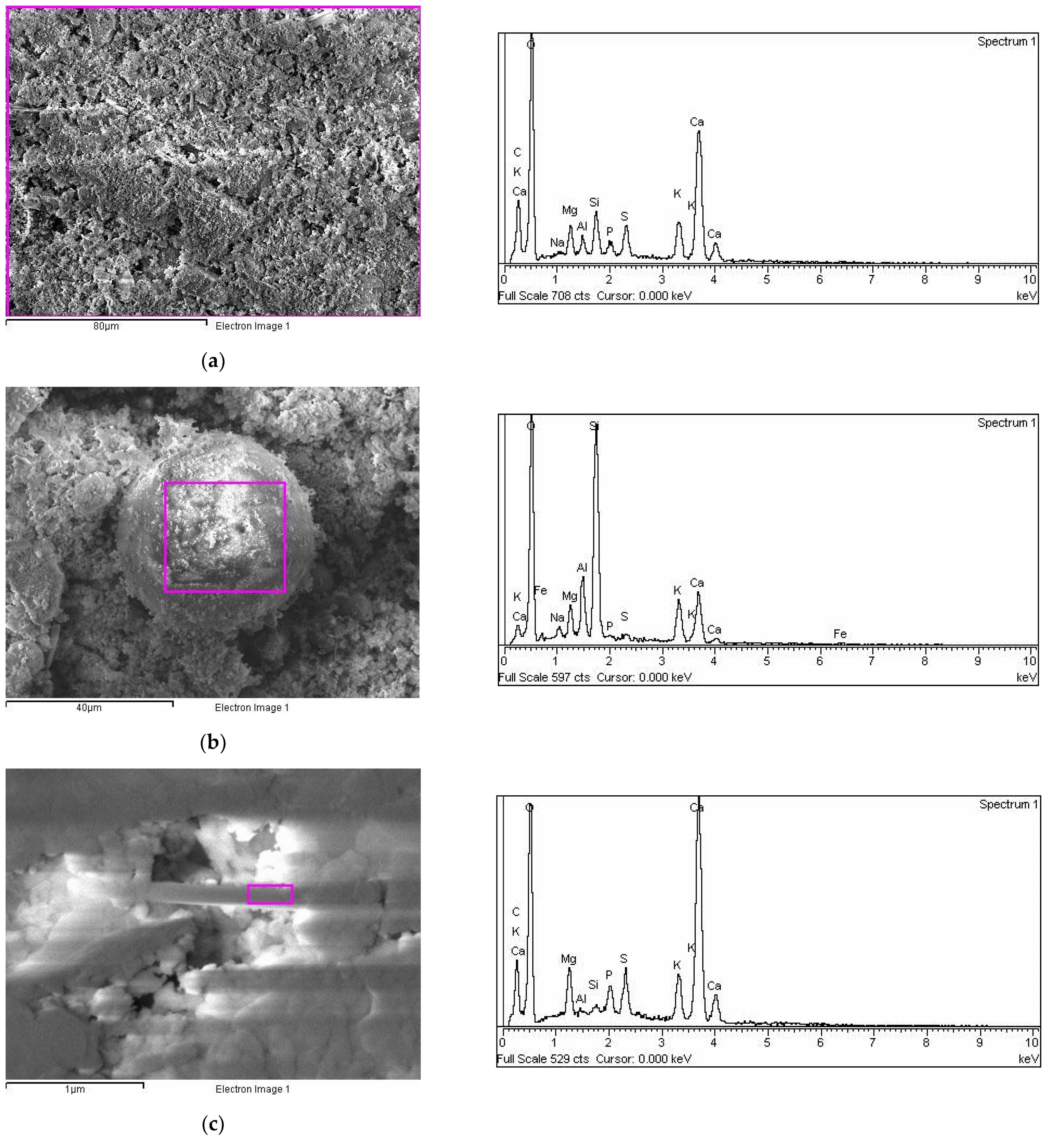
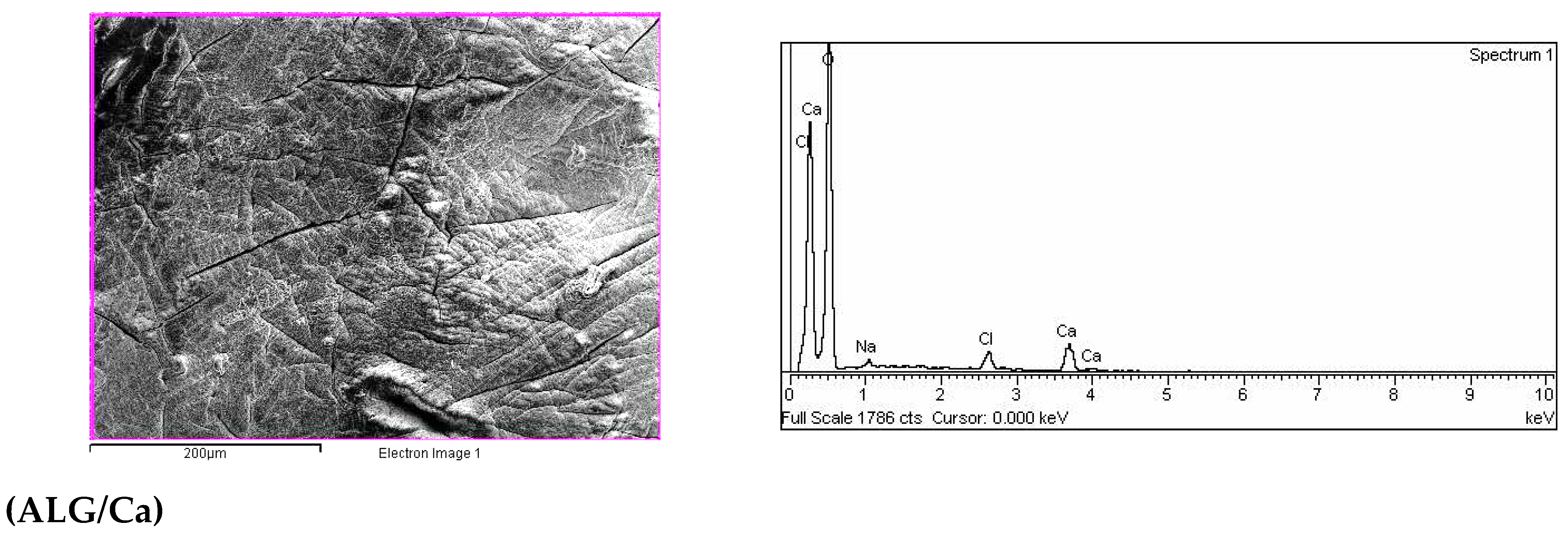
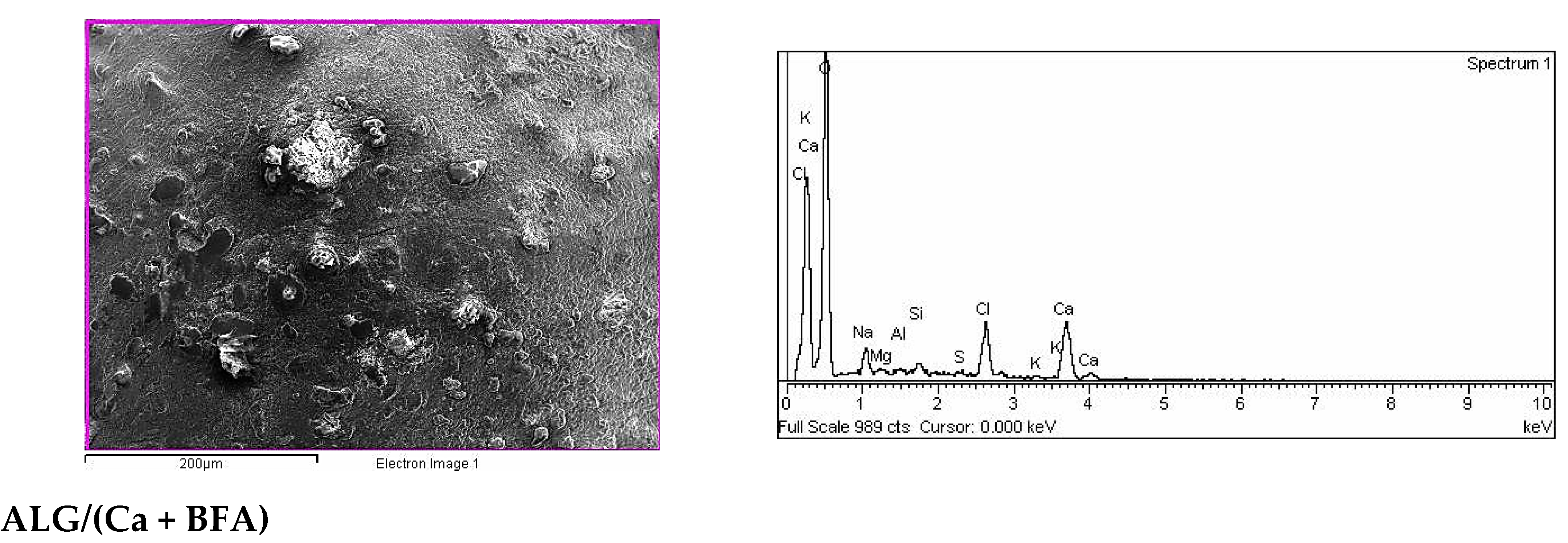
2.2. SEM-EDX Analysis
2.3. AFM Analysis
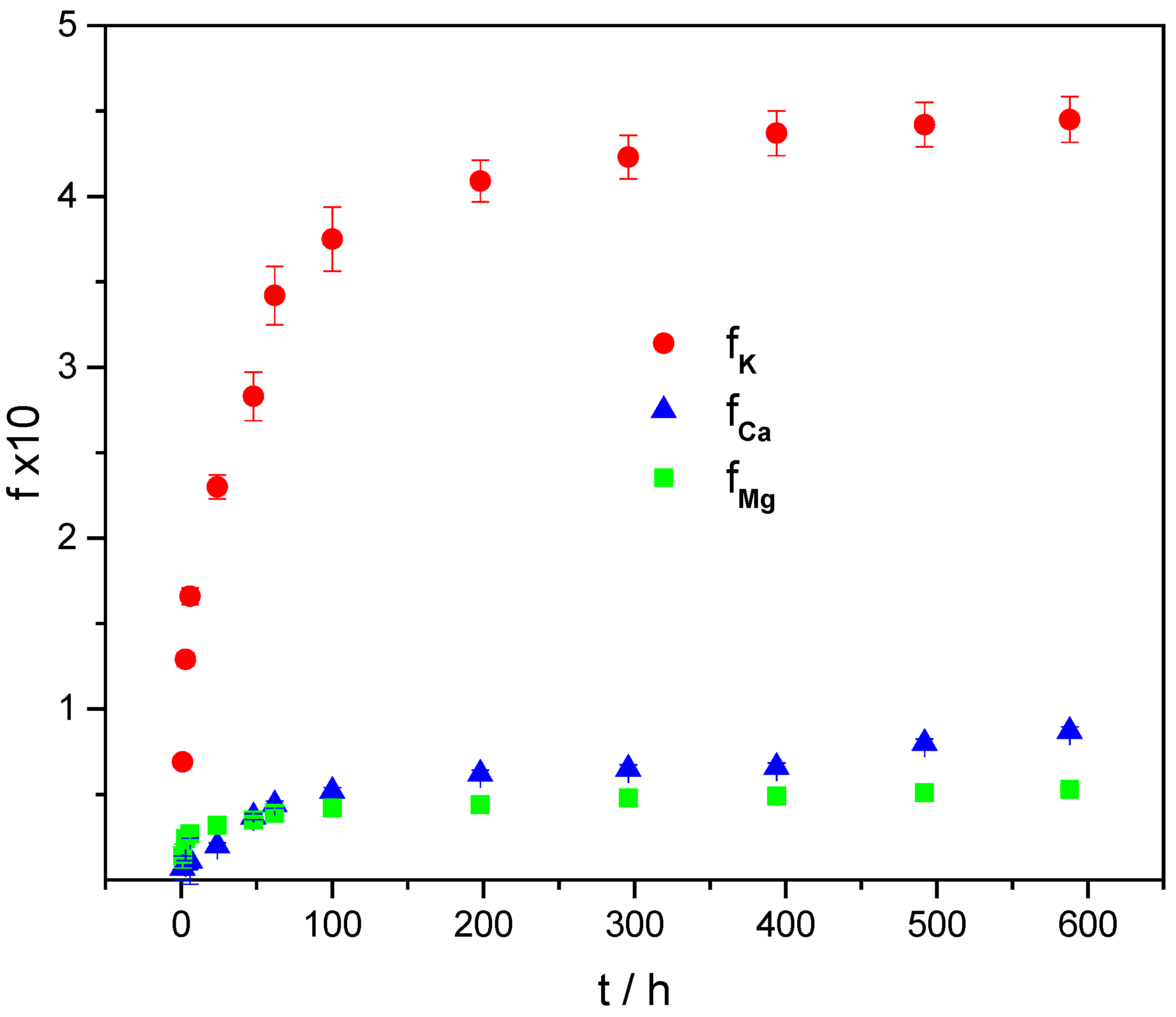
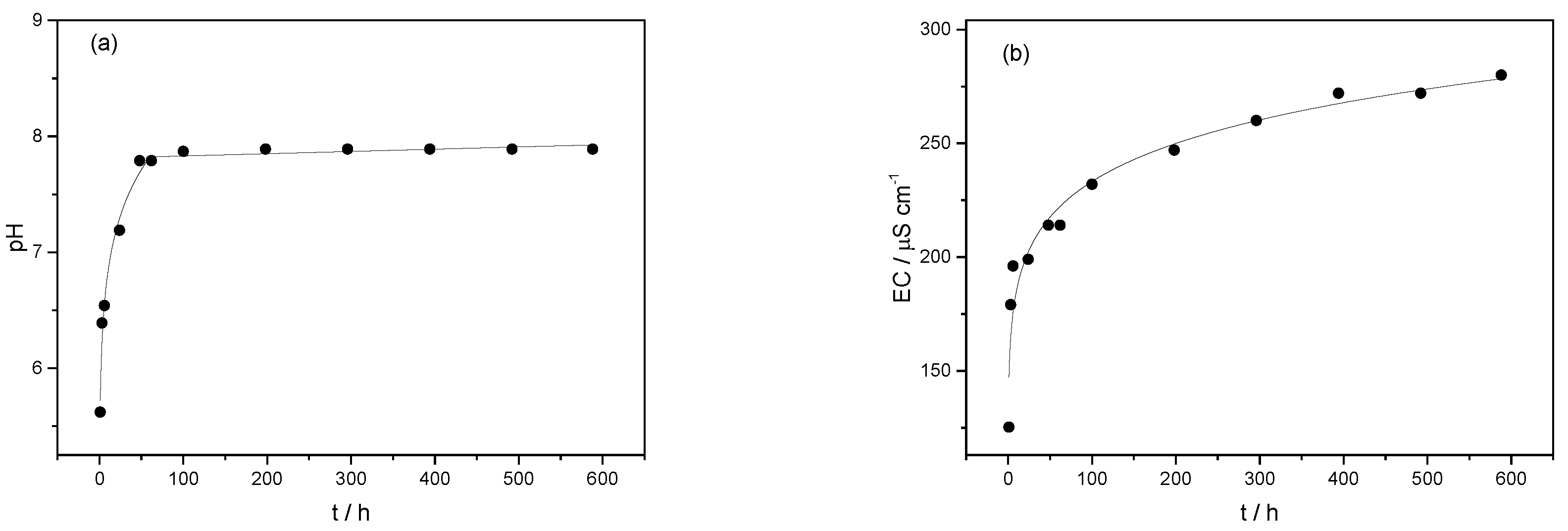
2.4. Swelling and In Vitro Release of K, Ca, and Mg from Microsphere Formulations
3. Materials and Methods
3.1. Materials
Preparation of Microsphere Formulations
3.2. Methods
Swelling Degree and Release of Potassium, Calcium, and Magnesium from Microsphere Formulation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Lamers, F.; Cremers, M.; Matschegg, D.; Schmidl, C.; Hannam, K.; Hazlett, P.; Madrali, S.; Primdal Dam, B.; Roberto, R.; Mager, R.; et al. Options for Increased Use of Ash from Biomass Combustion and Co-Firing. Biomass Combustion and Cofiring (2018). IEA Bioenergy: Task 32, Deliverable D7, 1-61. Available online: https://task32.ieabioenergy.com/publications/options-for-increased-use-of-ash-from-biomass-combustion-and-co-firing/ (accessed on 18 July 2022).
- Knapp, B.A.; Insam, H. Recycling of Biomass Ashes: Current Technologies and Future Research Needs. In Recycling of Biomass Ashes; Insam, H., Knapp, B., Eds.; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar] [CrossRef]
- Pitman, R.M. Wood ash use in forestry—A review of the environmental impacts. For. Int. J. For. Res. 2009, 79, 563–568. [Google Scholar] [CrossRef]
- Ondrašek, G.; Kranjčec, F.; Filipović, L.; Filipović, V.; Kovačić, M.B.; Badovinac, I.J.; Peter, R.; Petravić, M.; Macan, J.; Rengel, Z. Biomass bottom ash & dolomite similarly ameliorate an acidic low-nutrient soil, improve phytonutrition and growth, but increase Cd accumulation in radish. Sci. Total Environ. 2021, 753, 141902. [Google Scholar] [CrossRef]
- Regulation (EC) No. 2003/2003 of the European Parliament and of the Council of 13 October 2003 Relating to Fertilisers. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32003R2003&from=EN (accessed on 15 August 2022).
- Patterson, S.J.; Acharya, S.N.; Thomas, J.E.; Bertsch, A.I.B.; Rothwell, R.L. Barley biomass and grain yield and canola seed yield response to land application of wood ash. Agron. J. 2004, 96, 971–977. [Google Scholar] [CrossRef]
- Mbah, C.N.; Nwite, J.N.; Njoku, C.; Nwek, I.A. Response of maize (Zea mays L.) to different rates of wood-ash application in acid ultisol in Southeast Nigeria. Afric. J. Agric. Res. 2010, 5, 580–583. [Google Scholar]
- Wang, G.; Shen, L.; Sheng, C. Characterization of biomass ashes from power plants firing agricultural residues. Energy Fuels 2012, 26, 102–111. [Google Scholar] [CrossRef]
- Ou, Y.; Ma, S.; Zhou, X.; Wang, X.; Shi, J.; Zhang, Y. The Effect of a Fly Ash-Based Soil Conditioner on Corn and Wheat Yield and Risk Analysis of Heavy Metal Contamination. Sustainability 2020, 12, 7281. [Google Scholar] [CrossRef]
- Shi, R.; Li, J.; Jiang, J.; Mehmood, K.; Liu, J.; Xu, R. Characteristics of biomass ashes from different materials and their ameliorative effects on acid soils. J. Environ. Sci. 2016, 55, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Maschowski, C.; Kruspan, P.; Garra, P.; Arif, A.T.; Trouvé, G.; Gieré, R. Physicochemical and mineralogical characterization of biomass ash from different power plants in the Upper Rhine Region. Fuel 2019, 258, 116020. [Google Scholar] [CrossRef]
- Ondrašek, G.; Zovko, M.; Kranjčec, F.; Savić, R.; Romić, D.; Rengel, Z. Wood biomass fly ash ameliorates acidic, low- nutrient hydromorphic soil & reduces metal accumulation in maize. J. Clean Prod. 2021, 283, 124650. [Google Scholar] [CrossRef]
- Mahmoudkhani, M.; Richard, T. Sustainable use of biofuel by recycling ash to forests: Treatment of biofuel ash. Environ. Sci. Technol. 2007, 41, 4118–4123. [Google Scholar] [CrossRef]
- Agusta, H.; Nisya, F.N.; Iman, R.N.; Bilad, D.B.C. Granulation of coal fly ash by using different types of granule agents. IOP Conf. Ser. Earth Environ. Sci. 2017, 65, 12023. [Google Scholar] [CrossRef]
- Pesonen, J.; Kuokkanen, T.; Rautio, P.; Lassi, U. Bioavailability of nutrients and harmful elements in ash fertilizers: Effect of granulation. Biomass Bioenergy 2017, 100, 92. [Google Scholar] [CrossRef]
- Davidsson, D.; Gu, X. Materials for sustained and controlled release of nutrients and molecules to support plant growth. J. Agric. Food Chem. 2012, 60, 870. [Google Scholar] [CrossRef] [PubMed]
- Vinceković, M.; Jalšenjak, N.; Topolovec-Pintarić, S.; Đermić, E.; Bujan, M.; Jurić, S. Encapsulation of Biological and Chemical Agents for Plant Nutrition and Protection: Chitosan/Alginate Microcapsules Loaded with Copper Cations and Trichoderma viride. J. Agric. Food Chem. 2016, 64, 8073. [Google Scholar] [CrossRef]
- Jurić, S.; Jurić, M.; Režek-Jambrak, A.; Vinceković, M. Tailoring alginate/chitosan microparticles loaded with chemical and biological agents for agricultural application and production of value-added foods. Appl. Sci. 2021, 11, 4061. [Google Scholar] [CrossRef]
- Pereira, L.; Cotas, J. Alginate—Recenet uses of this natural polymer. In Recent Uses of Their Natural Polymer; Pereira, L., Cotas, J., Blumenberg, M., Eds.; New York University Langone Medical Center: New York, NY, USA, 2020; Chapter 1; pp. 1–16. [Google Scholar]
- Farmer, V.C. Vibrational Spectroscopy in Mineral Chemistry. In The Infrared Spectra of Minerals; Farmer, V.C., Ed.; Mineralogical Society of Great Britain and Ireland: London, UK, 1974; Chapter 1; pp. 1–36. [Google Scholar] [CrossRef]
- Hlavay, J.; Jonas, K.; Elek, S.; Inczedy, J. Characterization of the particle size and the crystallinity of certain minerals by IR spectrophotometry and other instrumental methods. II Investigations of quartz and feldspar. Clays Clay Miner. 1978, 26, 139. [Google Scholar] [CrossRef]
- Ennaciri, Y.; Alaoui-Belghiti, H.E.; Bettach, M. Comparative study of K2SO4 production by wet conversion from phosphogypsum and synthetic gypsum. J. Mater. Res. Technol. 2019, 8, 2586. [Google Scholar] [CrossRef]
- Selvam, N.C.S.; Kumar, R.T.; Kennedy, L.J.; Vijaya, J.J. Comparative study of microwave and conventional methods for the preparation and optical properties of novel MgO-micro and nano-structures. J. Alloys Comp. 2011, 509, 9809. [Google Scholar] [CrossRef]
- Mondal, B.K.; Islam, M.N.; Hossain, M.E.; Abser, M.N. Microstructural and mineralogical properties of acid and alkali activated coal fly ash. J. Adv. Chem. Sci. 2019, 5, 612. [Google Scholar] [CrossRef]
- Cruz-Yusta, M.; Marmol, I.; Morales, J.; Sanchez, L. Use of olive bimass fly ash in the preparation of environmentally friendly mortars. Environ. Sci. Technol. 2011, 45, 6991. [Google Scholar] [CrossRef]
- Mozgawa, W.; Król, M.; Dyczek, J.; Deja, J. Investigation of the coal fly ashes using IR spectroscopy. Spectrochim. Acta A Mol. Biomol. 2014, 132, 889. [Google Scholar] [CrossRef] [PubMed]
- Jurić, S.; Šegota, S.; Vinceković, M. Influence of surface morphology and structure of alginate microparticles on the bioactive agents release behavior. Carbohyd. Polym. 2019, 218, 234. [Google Scholar] [CrossRef] [PubMed]
- Saiki, K.; Ishikawa, T. Controlling factors of te particle size of spherical silica synthesized by a dry process. Int. J. Applied Ceram. Technol. 2022, 19, 1894. [Google Scholar] [CrossRef]
- Verrecchia, E.P.; Verecchia, K.E. Needle-fiber calcite: A Critical review and a proposed classification. J. Sed. Res. 1994, 64, 650. [Google Scholar] [CrossRef]
- Siepmann, J.; Siepmann, F. Modeling of diffusion controlled drug delivery. J. Control. Release 2012, 161, 351. [Google Scholar] [CrossRef]
- Briggs, F.; Browne, D.; Asuri, P. Role of polymer concentration and crosslinking density on release rates of small molecule drugs. Inter. J. Mol. Sci. 2022, 23, 4118. [Google Scholar] [CrossRef]
- Roger, S.; Talbot, D.; Bee, A. Preparation and effect of Ca2+ on water solubility, particle release and swelling properties of magnetic alginate films. J. Magn. Magn. Mater. 2006, 305, 221. [Google Scholar] [CrossRef]
- Korsmeyer, R.W.; Gurny, R.; Doelker, E.; Buri, P.; Peppas, N.A. Mechanisms of solute release from porous hydrophilic polymers. Int. J. Pharm. 1983, 15, 25. [Google Scholar] [CrossRef]
- Mellbo, P.; Sarenbo, S.; Stälnacke, O.; Claesson, T. Leaching of wood ash products aimed for spreading in forest floors—Influence of method and L/S ratio. Waste Manage. 2008, 28, 2235. [Google Scholar] [CrossRef]
- Haberl, J.; Schuster, M. Solubility of elements in waste incineration fly ash and bottom ash under various leaching conditions studied by a sequential extraction procedure. Waste Manage. 2019, 87, 268. [Google Scholar] [CrossRef]
- Iyer, R. The surface chemistry of leaching coal fly ash. J. Hazard Mater. B 2002, 93, 321. [Google Scholar] [CrossRef]
- Ugurlu, A. Leaching characteristics of fly ash. Environ. Geol. 2004, 46, 890. [Google Scholar] [CrossRef]
- Roy, A.; Bajapi, J.; Bajapi, A.K. Development of calcium alginate-gelatin based microspheres for controlled release of endosulfan as a model pesticide. Indian J. Chem. Technol. 2009, 16, 388. [Google Scholar]
- Al-Zahrani, S.M. Controlled-release of fertilizers: Modelling and simulation. Inter. J. Engin. Sci. 1999, 37, 1299–1307. [Google Scholar] [CrossRef]
- Acharya, G.; Park, K. Mechanisms of controlled drug release from drug-eluting stents. Adv. Drug Deliv. Rev. 2006, 58, 387. [Google Scholar] [CrossRef]
- Kim, A.G.; Kazonich, G.; Dahlberg, M. Relative solubility of cations in Class F fly ash. Environ. Sci. Technol. 2003, 37, 4507. [Google Scholar] [CrossRef] [PubMed]
- Du, C.-w.; Zhou, J.; Shaviv, A. Release characteristics of nutrients from polymer-coated compound controlled release fertilizers. J. Polym. Environ. 2006, 14, 223–230. [Google Scholar] [CrossRef]
- Marinina, O.; Nevskaya, M.; Jonek-Kowalska, I.; Wolniak, R.; Marinin, M. Recycling of Coal Fly Ash as an Example of an Efficient Circular Economy: A Stakeholder Approach. Energies 2021, 14, 3597. [Google Scholar] [CrossRef]
- Kudasova, D.; Mutaliyeva, B.; Vlahoviček-Kahlina, K.; Jurić, S.; Marijan, M.; Khalus, S.V.; Prosyanik, A.V.; Šegota, S.; Španić, N.; Vinceković, M. Encapsulation of Synthesized Plant Growth Regulator Based on Copper(II) Complex in Chitosan/Alginate Microcapsules. Int. J. Mol. Sci. 2021, 22, 2663. [Google Scholar] [CrossRef]
- Vlahoviček-Kahlina, K.; Jurić, S.; Marijan, M.; Mutaliyeva, B.; Khalus, S.V.; Prosyanik, A.V.; Vinceković, M. Synthesis, Characterization and Encapsulation of Novel Plant Growth Regulators (PGRs) in Biopolymer Matrices. Int. J. Mol. Sci. 2021, 22, 1847. [Google Scholar] [CrossRef]


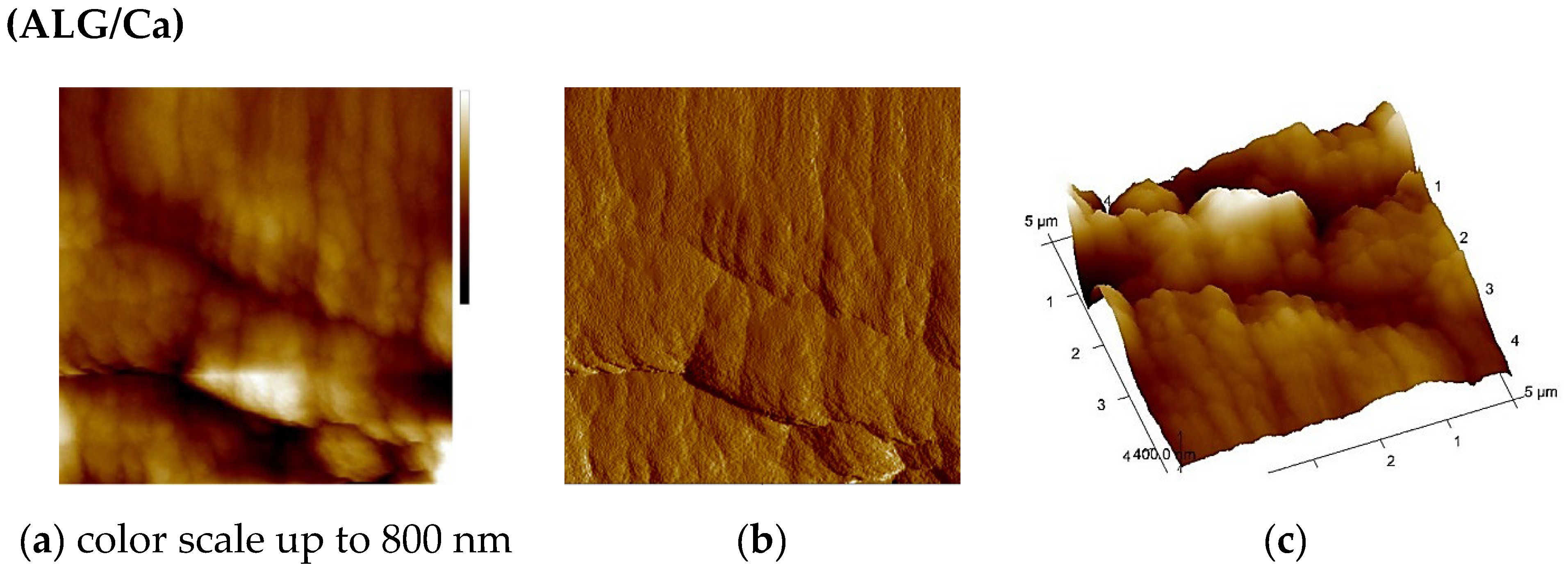
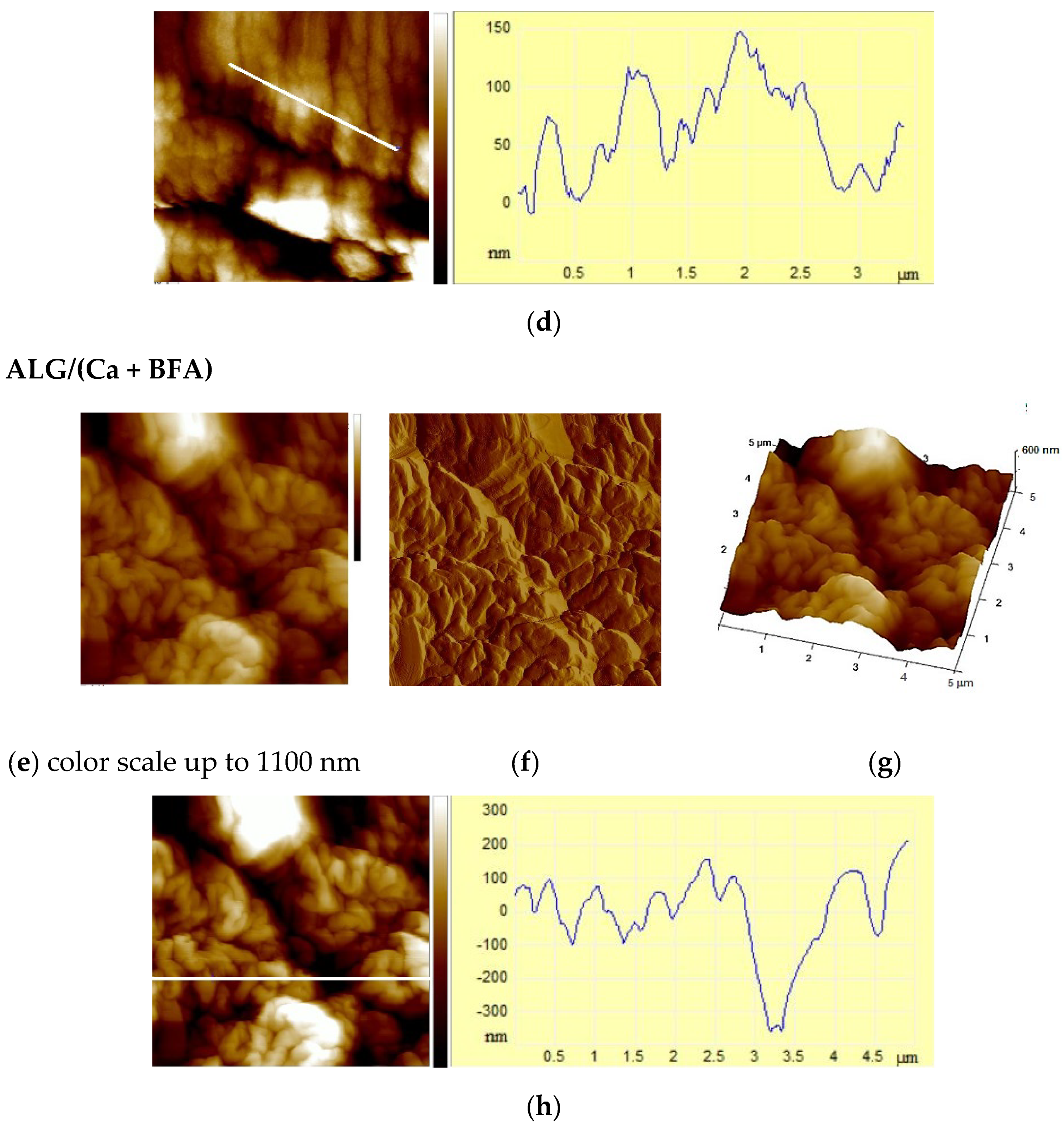
| Sample | Ra/nm | Rms/nm | Z/nm |
|---|---|---|---|
| ALG/Ca | 76 ± 1 | 106 ± 2 | 1127 ± 21 |
| ALG/(Ca + BFA) | 122 ± 2 | 159 ± 1 | 1054 ± 21 |
| ALG/(Ca + BFA) | k | n | R2 |
|---|---|---|---|
| K | 0.12 | 0.22 | 0.98 |
| Ca | 0.019 | 0.16 | 0.98 |
| Mg | 0.009 | 0.36 | 0.97 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vinceković, M.; Šegota, S.; Jurić, S.; Harja, M.; Ondrasek, G. Development and Characterization of a Novel Soil Amendment Based on Biomass Fly Ash Encapsulated in Calcium Alginate Microspheres. Int. J. Mol. Sci. 2022, 23, 9984. https://doi.org/10.3390/ijms23179984
Vinceković M, Šegota S, Jurić S, Harja M, Ondrasek G. Development and Characterization of a Novel Soil Amendment Based on Biomass Fly Ash Encapsulated in Calcium Alginate Microspheres. International Journal of Molecular Sciences. 2022; 23(17):9984. https://doi.org/10.3390/ijms23179984
Chicago/Turabian StyleVinceković, Marko, Suzana Šegota, Slaven Jurić, Maria Harja, and Gabrijel Ondrasek. 2022. "Development and Characterization of a Novel Soil Amendment Based on Biomass Fly Ash Encapsulated in Calcium Alginate Microspheres" International Journal of Molecular Sciences 23, no. 17: 9984. https://doi.org/10.3390/ijms23179984
APA StyleVinceković, M., Šegota, S., Jurić, S., Harja, M., & Ondrasek, G. (2022). Development and Characterization of a Novel Soil Amendment Based on Biomass Fly Ash Encapsulated in Calcium Alginate Microspheres. International Journal of Molecular Sciences, 23(17), 9984. https://doi.org/10.3390/ijms23179984










