In Silico Study of piRNA Interactions with the SARS-CoV-2 Genome
Abstract
:1. Introduction
2. Results
2.1. Thirteen piRNA Binding Cluster in SARS-CoV-2 gRNA
2.2. Thirty-Nine piRNA Binding Cluster in SARS-CoV-2 gRNA
2.3. Twenty-Four piRNA Binding Cluster in SARS-CoV-2 gRNA
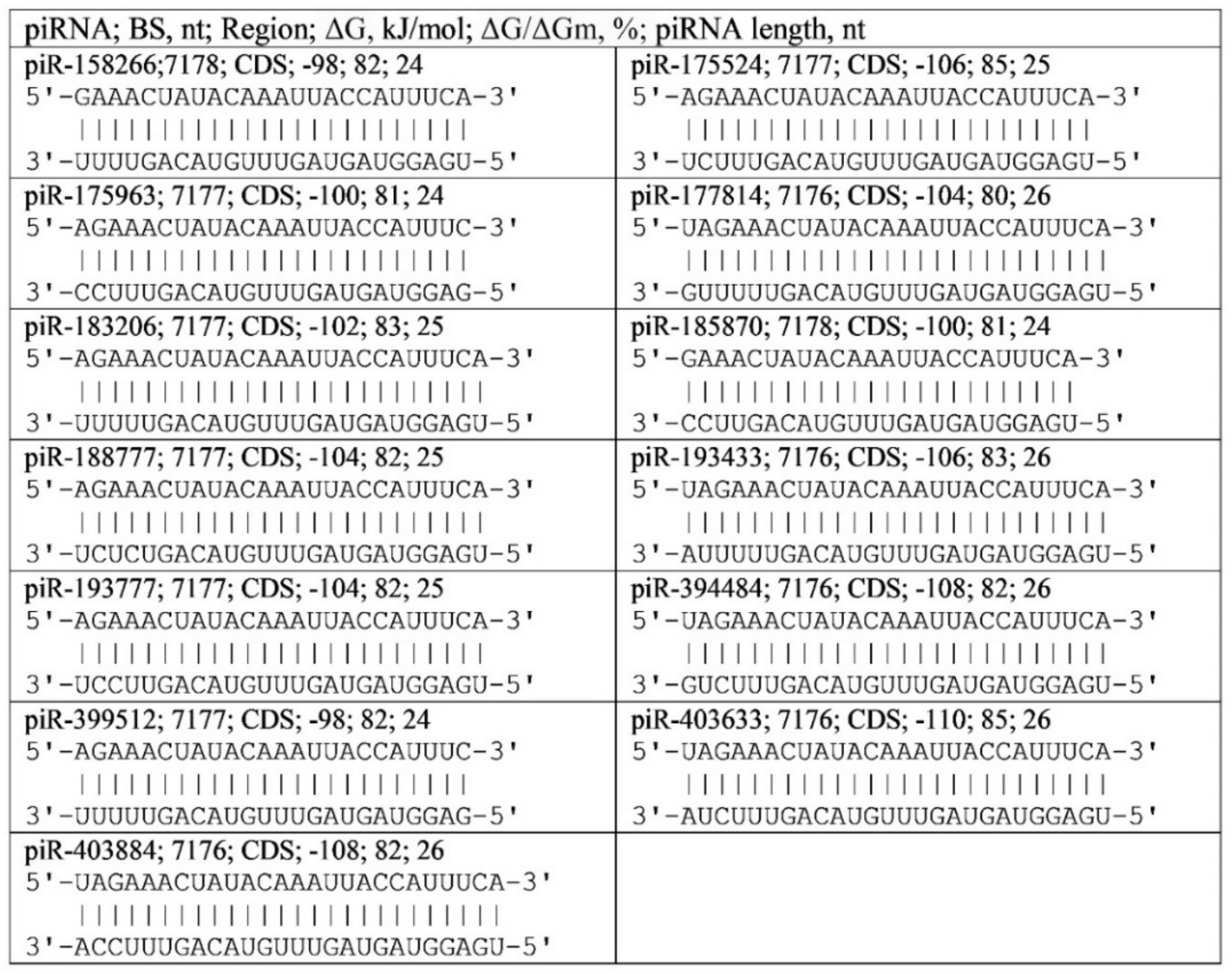
2.4. Twelve piRNA Binding Clusters in SARS-CoV-2 gRNA
3. Discussion
4. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ivashchenko, A.T.; Rakhmetullina, A.K.; Akimniyazova, A.N.; Aisina, D.E. How miRNAs Can Protect Humans from Coronaviruses COVID-19, SARS-CoV, and MERS-CoV. In Proceedings of the Bioinformatics of Genome Regulation and Structure/Systems Biology, (BGRS/SB-2020), Novosibirsk, Russia, 6–10 July 2020. [Google Scholar] [CrossRef]
- Ivashchenko, A.T.; Rakhmetullina, A.K.; Aisina, D.E. How miRNA can protect humans coronaviruses COVID-19, SARS-CoV, and MERS-CoV. Res. Sq. 2020; preprint. [Google Scholar] [CrossRef]
- Khaitov, M.; Nikonova, A.; Shilovskiy, I.; Kozhikhova, K.; Kofiadi, I.; Vishnyakova, L.; Nikolskii, A.; Gattinger, P.; Kovchina, V.; Barvinskaia, E.; et al. Silencing of SARS-CoV-2 with modified siRNA-peptide dendrimer formulation. Allergy 2021, 76, 2840–2854. [Google Scholar] [CrossRef] [PubMed]
- Ambike, S.; Cheng, C.C.; Feuerherd, M.; Velkov, S.; Baldassi, D.; Afridi, S.Q.; Porras-Gonzalez, D.; Wei, X.; Hagen, P.; Kneidinger, N.; et al. Targeting genomic SARS-CoV-2 RNA with siRNAs allows efficient inhibition of viral replication and spread. Nucleic Acids Res. 2022, 50, 333–349. [Google Scholar] [CrossRef] [PubMed]
- Shawan, M.M.A.K.; Sharma, A.R.; Bhattacharya, M.; Mallik, B.; Akhter, F.; Shakil, M.S.; Hossain, M.M.; Banik, S.; Lee, S.S.; Hasan, M.A.; et al. Designing an effective therapeutic siRNA to silence RdRp gene of SARS-CoV-2. Infect. Genet. Evol. 2021, 93, 104951. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, P.; Lu, Y.; Li, Y.; Zheng, Y.; Kan, Y.; Chen, R.; He, S. piRBase: A comprehensive database of piRNA sequences. Nucleic Acids Res. 2019, 47, 175–180. [Google Scholar] [CrossRef]
- Wang, J.; Shi, Y.; Zhou, H.; Zhang, P.; Song, T.; Ying, Z.; Yu, H.; Li, Y.; Zhao, Y.; Zeng, X.; et al. piRBase: Integrating piRNA annotation in all aspects. Nucleic Acids Res. 2021, 50, 265–272. [Google Scholar] [CrossRef]
- Kim, V.N. Small RNAs just got bigger: Piwi-interacting RNAs (piRNAs) in mammalian testes. Genes Dev. 2006, 20, 1993–1997. [Google Scholar] [CrossRef]
- Aravin, A.A.; Sachidanandam, R.; Girard, A.; Fejes-Toth, K.; Hannon, G.J. Developmentally regulated piRNA clusters implicate MILI in transposon control. Science 2007, 316, 744–747. [Google Scholar] [CrossRef]
- Chavda, V.; Madhwani, K.; Chaurasia, B. PiWi RNA in Neurodevelopment and Neurodegenerative disorders. Curr. Mol. Pharmacol. 2021, 15, 517–531. [Google Scholar] [CrossRef]
- Das, B.; Jain, N.; Mallick, B. piR-39980 mediates doxorubicin resistance in fibrosarcoma by regulating drug accumulation and DNA repair. Commun. Biol. 2021, 4, 1312. [Google Scholar] [CrossRef]
- Ding, L.; Wang, R.; Xu, W.; Shen, D.; Cheng, S.; Wang, H.; Lu, Z.; Zheng, Q.; Wang, L.; Xia, L.; et al. PIWI-interacting RNA 57125 restrains clear cell renal cell carcinoma metastasis by downregulating CCL3 expression. Cell Death Discov. 2021, 7, 333. [Google Scholar] [CrossRef] [PubMed]
- Qian, L.; Xie, H.; Zhang, L.; Zhao, Q.; Lü, J.; Yu, Z. Piwi-Interacting RNAs: A New Class of Regulator in Human Breast Cancer. Front. Oncol. 2021, 11, 695077. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.H.; Shiue, S.C.; Chen, K.Y.; Tseng, Y.Y.; Wu, W.S. Identifying piRNA targets on mRNAs in C. elegans using a deep multi-head attention network. BMC Bioinform. 2021, 22, 503. [Google Scholar] [CrossRef]
- Chu, H.; Chan, J.F.; Yuen, T.T.; Shuai, H.; Yuan, S.; Wang, Y.; Hu, B.; Yip, C.C.; Tsang, J.O.; Huang, X.; et al. Comparative tropism, replication kinetics, and cell damage profiling of SARS-CoV-2 and SARS-CoV with implications for clinical manifestations, transmissibility, and laboratory studies of COVID-19: An observational study. Lancet Microbe 2020, 1, 14–23. [Google Scholar] [CrossRef]
- Ikhlas, S.; Usman, A.; Kim, D.; Cai, D. Exosomes/microvesicles target SARS-CoV-2 via innate and RNA-induced immunity with PIWI-piRNA system. Life Sci. Alliance 2021, 5, e202101240. [Google Scholar] [CrossRef]
- Halajzadeh, J.; Dana, P.M.; Asemi, Z.; Mansournia, M.A.; Yousefi, B. An insight into the roles of piRNAs and PIWI proteins in the diagnosis and pathogenesis of oral, esophageal, and gastric cancer. Pathol. Res. Pract. 2020, 216, 153112. [Google Scholar] [CrossRef]
- Zhou, L.; Lim, M.Y.T.; Kaur, P.; Saj, A.; Bortolamiol-Beset, D.; Gopal, V.; Tolwinski, N.; Tucker-Kellogg, G.; Okamura, K. Importance of miRNA stability and alternative primary miRNA isoforms in gene regulation during Drosophila development. eLife 2018, 7, e3839. [Google Scholar] [CrossRef]
- Venter, J.C.; Adams, M.D.; Myers, E.W.; Li, P.W.; Mural, R.J.; Sutton, G.G.; Smith, H.O.; Yandell, M.; Evans, C.A.; Holt, R.A.; et al. The sequence of the human genome. Science 2001, 291, 1304–1351. [Google Scholar] [CrossRef]
- Huang, S.; Yoshitake, K.; Asakawa, S. A Review of Discovery Profiling of PIWI-Interacting RNAs and Their Diverse Functions in Metazoans. Int. J. Mol. Sci. 2021, 22, 11166. [Google Scholar] [CrossRef]
- Belkozhayev, A.; Niyazova, R.; Wilson, C.; Jainakbayev, N.; Pyrkova, A.; Ashirbekov, Y.; Akimniyazova, A.; Sharipov, K.; Ivashchenko, A. Bioinformatics analysis of the interaction of miRNAs, piRNAs with human mRNA genes having di- and trinucleotide repeats. Genes 2022, 13, 800. [Google Scholar] [CrossRef]
- Ivashchenko, A.; Berillo, O.; Pyrkova, A.; Niyazova, R.; Atambayeva, S. MiR-3960 Binding Sites with mRNA of Human Genes. Bioinformation 2014, 10, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Friedman, R.A.; Honig, B.A. Free Energy Analysis of Nucleic Acid Base Stacking in Aqueous Solution. Biophys. J. 1995, 69, 1528–1535. [Google Scholar] [CrossRef]
- Garg, A.; Heinemann, U. A Novel Form of RNA Double Helix Based on G·U and C·A+ Wobble Base Pairing. RNA 2018, 24, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Kool, E.T. Hydrogen Bonding, Base Stacking, and Steric Effects in DNA Replication. Annu. Rev. Biophys. Biomol. Struct. 2001, 30, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Leontis, N.B.; Stombaugh, J.; Westhof, E. The Non-watson-crick Base Pairs and Their Associated Isostericity Matrices. Nucleic Acids Res. 2002, 30, 3497–3531. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, Z.; Liu, B.; Chen, G.; Shao, N.; Ying, X.; Wang, Y. Systematic Study of Cis-Antisense miRNAs in Animal Species Reveals miR-3661 to Target PPP2CA in Human Cells. RNA 2016, 22, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Yurikova, O.Y.; Aisina, D.E.; Niyazova, R.E.; Atambayeva, S.A.; Labeit, S.; Ivashchenko, A.T. The Interactions of miRNA-5p and miRNA-3p with the mRNAs of Ortolologous Genes. Mol. Biol. 2019, 53, 692–704. [Google Scholar] [CrossRef]
| piRNA | Start of BSs, nt | ΔG, kJ/mol | ΔG/ΔGm, % | piRNA Length, nt | ΔG*, kJ/mol |
|---|---|---|---|---|---|
| piR-404056 | 12077 | −108 | 81 | 27 | −142 |
| piR-188123 | 12078 | −104 | 80 | 26 | −138 |
| piR-188808 | 12078 | −108 | 81 | 26 | −138 |
| piR-188962 | 12078 | −108 | 82 | 26 | −138 |
| piR-189542 | 12078 | −106 | 81 | 26 | −138 |
| piR-189637 | 12078 | −106 | 81 | 26 | −138 |
| piR-190555 | 12078 | −113 | 84 | 26 | −138 |
| piR-190706 | 12078 | −110 | 81 | 26 | −138 |
| piR-190772 | 12078 | −104 | 80 | 26 | −138 |
| piR-192160 | 12078 | −106 | 81 | 26 | −138 |
| piR-194135 | 12078 | −110 | 81 | 26 | −138 |
| piR-194397 | 12078 | −108 | 81 | 26 | −138 |
| piR-392668 | 12078 | −108 | 82 | 26 | −138 |
| piR-401969 | 12078 | −115 | 82 | 26 | −138 |
| piR-403862 | 12078 | −104 | 80 | 25 | −134 |
| piR-406209 | 12078 | −106 | 81 | 26 | −138 |
| piR-406508 | 12078 | −98 | 81 | 24 | −127 |
| piR-410103 | 12078 | −108 | 82 | 26 | −138 |
| piR-187101 | 12079 | −102 | 81 | 25 | −132 |
| piR-189493 | 12079 | −108 | 84 | 25 | −132 |
| piR-190719 | 12079 | −106 | 82 | 25 | −132 |
| piR-191124 | 12079 | −106 | 82 | 25 | −132 |
| piR-191185 | 12079 | −110 | 84 | 25 | −132 |
| piR-191492 | 12079 | −106 | 82 | 25 | −132 |
| piR-192078 | 12079 | −104 | 80 | 25 | −132 |
| piR-194007 | 12079 | −106 | 82 | 25 | −132 |
| piR-194172 | 12079 | −104 | 82 | 25 | −132 |
| piR-403725 | 12079 | −102 | 81 | 24 | −127 |
| piR-194781 | 12079 | −104 | 82 | 25 | −132 |
| piR-404008 | 12079 | −104 | 82 | 25 | −132 |
| piR-405339 | 12079 | −104 | 82 | 25 | −132 |
| piR-405683 | 12079 | −100 | 81 | 24 | −127 |
| piR-406684 | 12079 | −102 | 81 | 25 | −132 |
| piR-180819 | 12080 | −98 | 81 | 24 | −125 |
| piR-184604 | 12080 | −102 | 83 | 24 | −125 |
| piR-396601 | 12081 | −117 | 81 | 27 | −140 |
| piR-360432 | 12082 | −106 | 81 | 25 | −130 |
| piR-1177268 | 12082 | −102 | 81 | 25 | −130 |
| piR-345961 | 12084 | −104 | 82 | 24 | −125 |
| piRNA | Start of BSs, nt | ΔG, kJ/mol | ΔG/ΔGm, % | piRNA Length, nt | ΔG*, kJ/mol |
|---|---|---|---|---|---|
| piR-77587 | 20650 | −108 | 81 | 25 | −127 |
| piR-78640 | 20650 | −106 | 82 | 24 | −121 |
| piR-89731 | 20649 | −104 | 82 | 24 | −119 |
| piR-104672 | 20649 | −113 | 82 | 26 | −132 |
| piR-1874165 | 20649 | −115 | 84 | 26 | −132 |
| piR-1874441 | 20648 | −110 | 80 | 26 | −132 |
| piR-1877632 | 20646 | −127 | 82 | 29 | −151 |
| piR-1901265 | 20648 | −106 | 81 | 25 | −125 |
| piR-1905481 | 20650 | −110 | 84 | 25 | −127 |
| piR-1909341 | 20646 | −113 | 82 | 26 | −132 |
| piR-1916216 | 20649 | −110 | 81 | 26 | −132 |
| piR-1930602 | 20648 | −115 | 81 | 27 | −138 |
| piR-1950681 | 20645 | −119 | 81 | 27 | −136 |
| piR-1955234 | 20647 | −106 | 81 | 25 | −125 |
| piR-1957782 | 20646 | −123 | 81 | 29 | −151 |
| piR-1968262 | 20647 | −110 | 80 | 26 | −132 |
| piR-1970873 | 20646 | −108 | 85 | 24 | −121 |
| piR-1978038 | 20645 | −100 | 81 | 24 | −119 |
| piR-1981597 | 20645 | −108 | 82 | 25 | −125 |
| piR-2526803 | 20647 | −125 | 83 | 28 | −144 |
| piR-2540472 | 20647 | −104 | 83 | 24 | −121 |
| piR-2540716 | 20650 | −110 | 81 | 26 | −132 |
| piR-2557847 | 20645 | −113 | 82 | 26 | −132 |
| piR-2574698 | 20645 | −108 | 81 | 25 | −125 |
| piRNA | Start of BSs, nt | ΔG, kJ/mol | ΔG/ΔGm, % | Length, nt | ΔG*, kJ/mol |
|---|---|---|---|---|---|
| piR-2047904 | 4670 | −136 | 81 | 32 | −168 |
| piR-912075 | 9102 | −140 | 80 | 33 | −175 |
| piR-2352720 | 9123 | −138 | 81 | 34 | −170 |
| piR-2490582 | 10012 | −142 | 83 | 33 | −171 |
| piR-1491787 | 17115 | −138 | 80 | 32 | −173 |
| piR-3555322 | 21712 | −136 | 82 | 34 | −166 |
| piR-703629 | 25672 | −142 | 82 | 31 | −173 |
| piR-1525356 | 26106 | −136 | 80 | 32 | −170 |
| piR-2599982 | 27070 | −144 | 82 | 34 | −176 |
| piR-806264 | 28427 | −142 | 83 | 33 | −171 |
| piR-98504 | 29024 | −136 | 84 | 32 | −162 |
| piR-3218674 | 29475 | −138 | 81 | 34 | −170 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akimniyazova, A.; Yurikova, O.; Pyrkova, A.; Rakhmetullina, A.; Niyazova, T.; Ryskulova, A.-G.; Ivashchenko, A. In Silico Study of piRNA Interactions with the SARS-CoV-2 Genome. Int. J. Mol. Sci. 2022, 23, 9919. https://doi.org/10.3390/ijms23179919
Akimniyazova A, Yurikova O, Pyrkova A, Rakhmetullina A, Niyazova T, Ryskulova A-G, Ivashchenko A. In Silico Study of piRNA Interactions with the SARS-CoV-2 Genome. International Journal of Molecular Sciences. 2022; 23(17):9919. https://doi.org/10.3390/ijms23179919
Chicago/Turabian StyleAkimniyazova, Aigul, Oxana Yurikova, Anna Pyrkova, Aizhan Rakhmetullina, Togzhan Niyazova, Alma-Gul Ryskulova, and Anatoliy Ivashchenko. 2022. "In Silico Study of piRNA Interactions with the SARS-CoV-2 Genome" International Journal of Molecular Sciences 23, no. 17: 9919. https://doi.org/10.3390/ijms23179919
APA StyleAkimniyazova, A., Yurikova, O., Pyrkova, A., Rakhmetullina, A., Niyazova, T., Ryskulova, A.-G., & Ivashchenko, A. (2022). In Silico Study of piRNA Interactions with the SARS-CoV-2 Genome. International Journal of Molecular Sciences, 23(17), 9919. https://doi.org/10.3390/ijms23179919





