Ubiquitin Specific Protease USP48 Destabilizes NF-κB/p65 in Retinal Pigment Epithelium Cells
Abstract
1. Introduction
2. Results
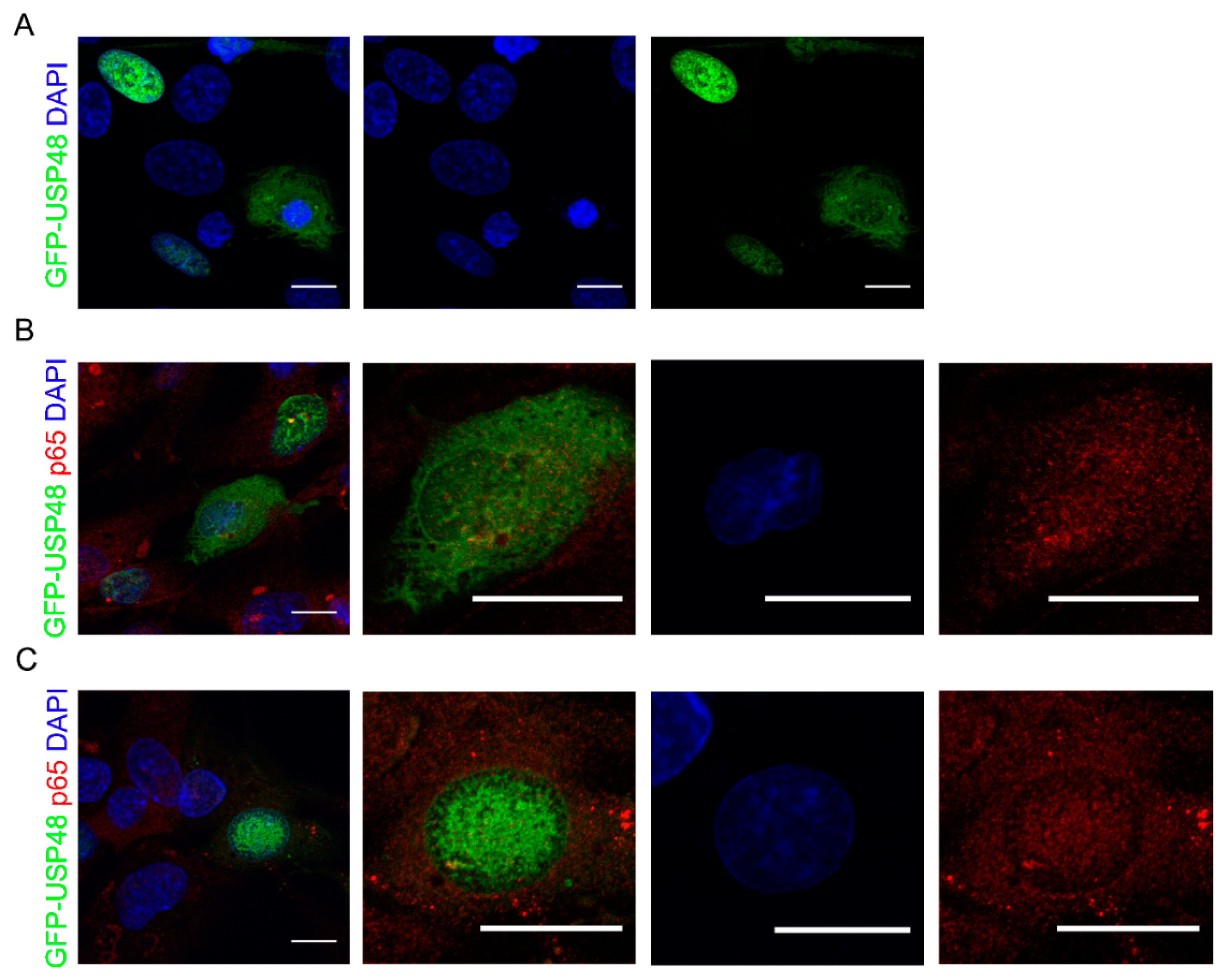
2.1. USP48 Overexpression Promotes p65 Nuclear Localization
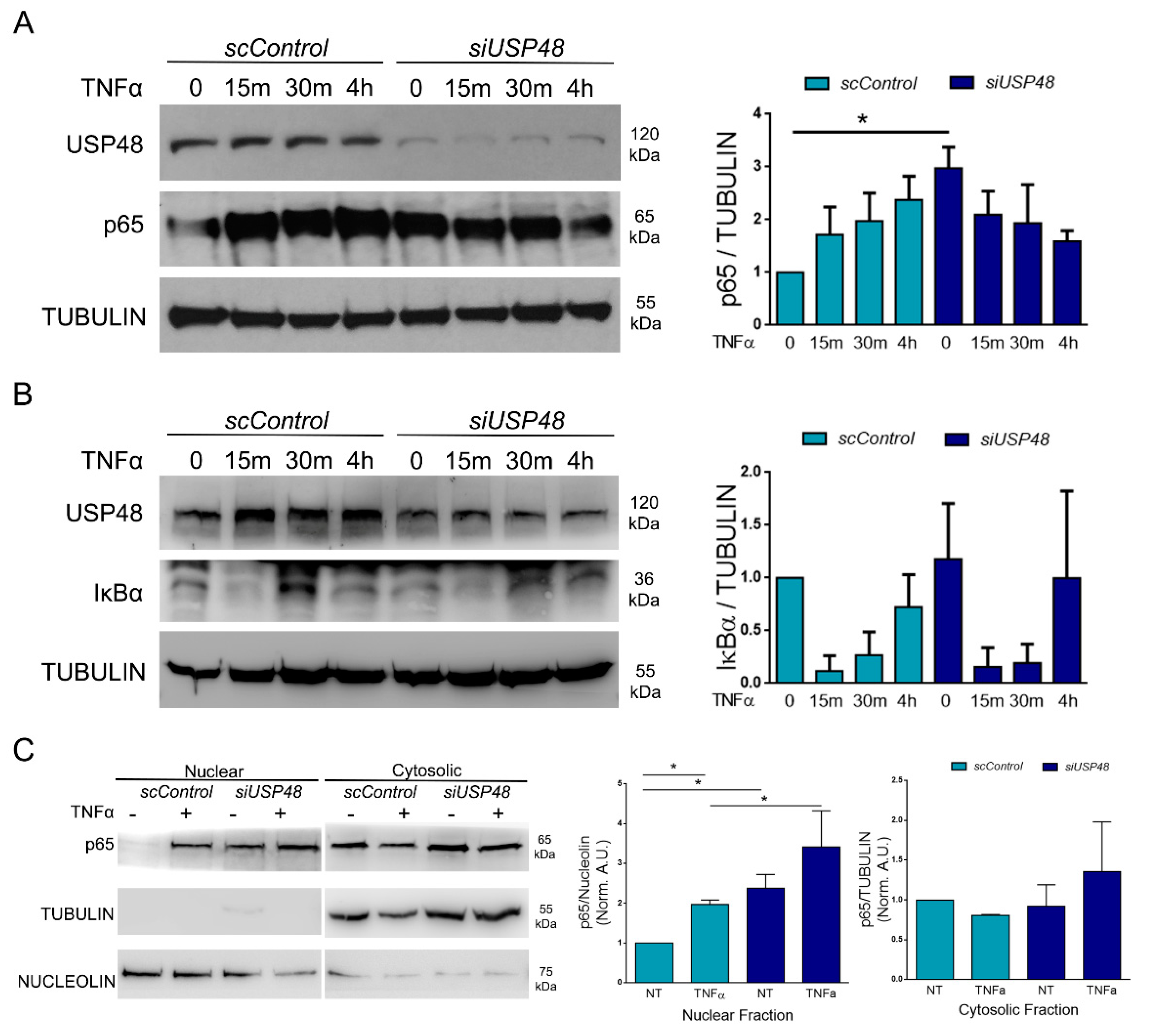
2.2. USP48 Downregulation Induces Basal p65 Stabilization
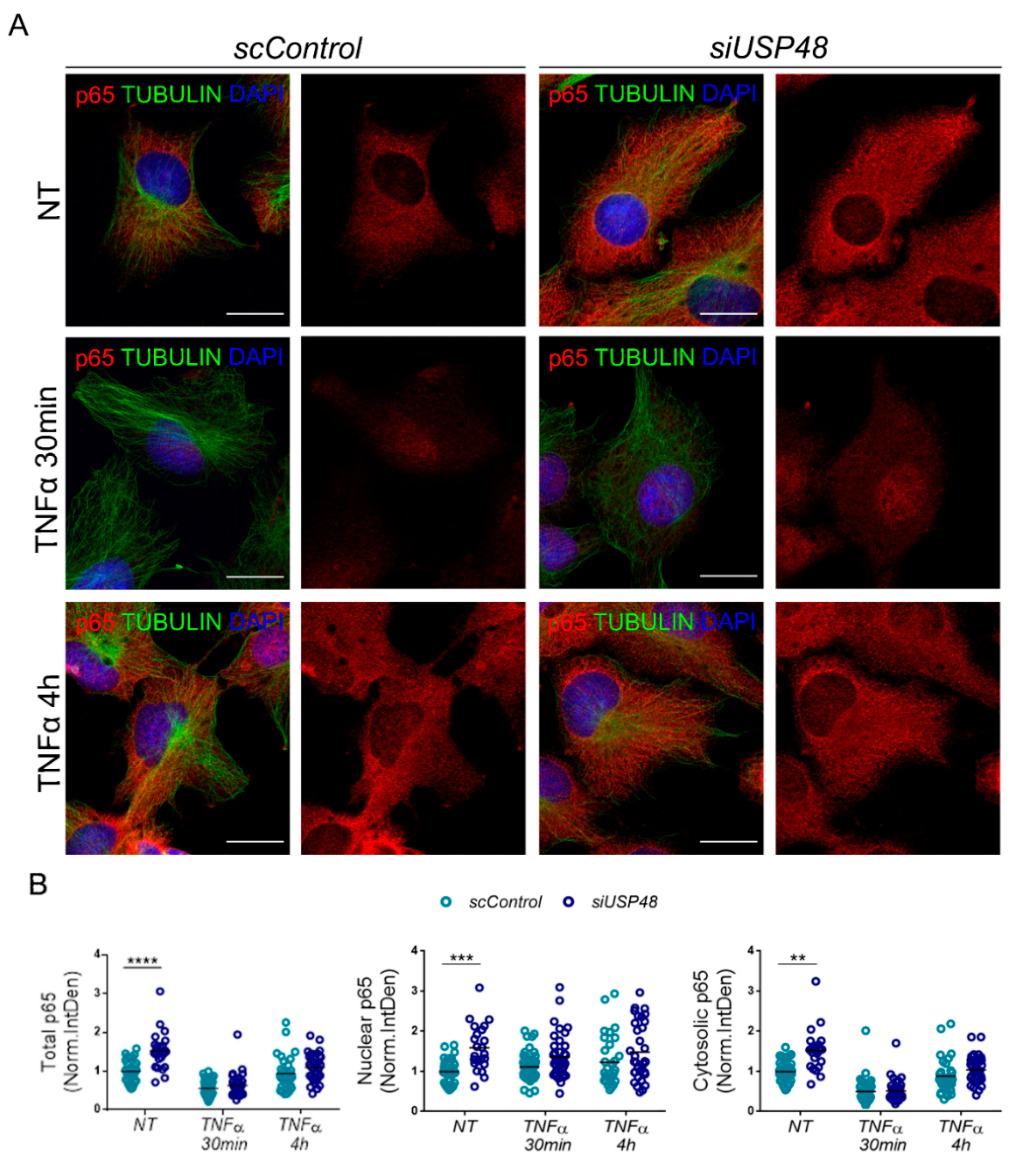
2.3. USP48 Downregulation Increases p65 Nuclear Accumulation
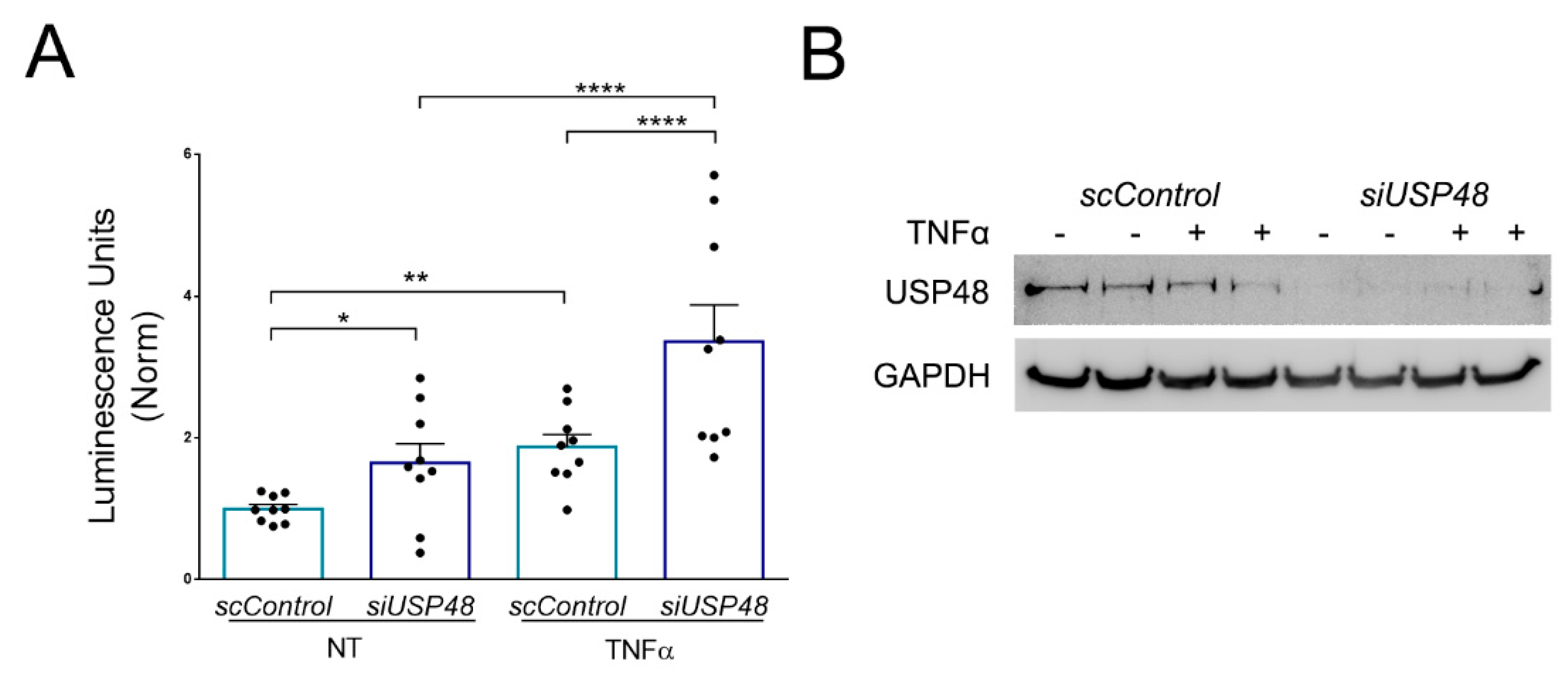
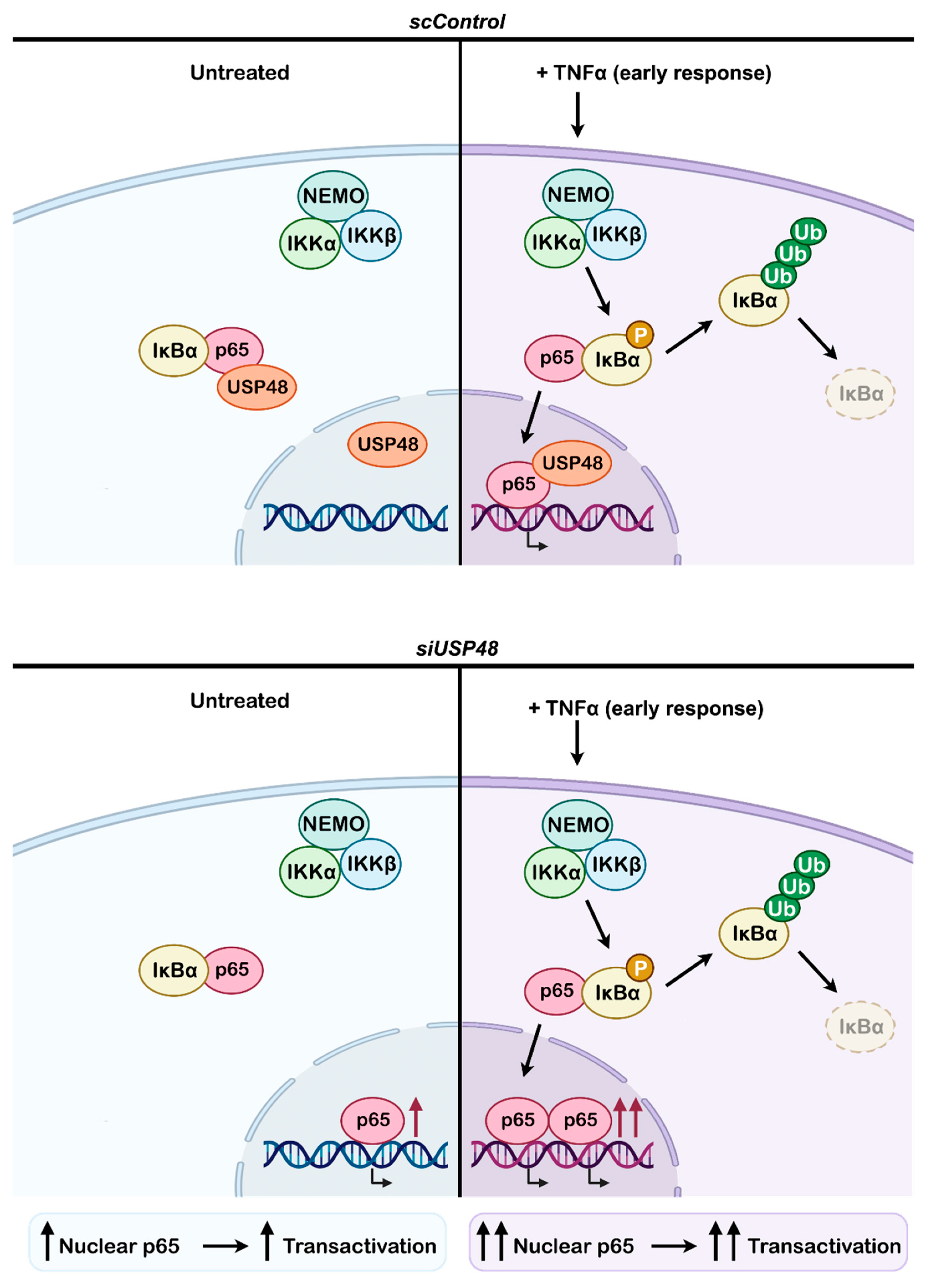
2.4. USP48 Downregulation Enhances TNFα-Induced NF-κB Activity
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Transfections
4.2. Plasmid Vectors
4.3. Immunofluorescence
4.4. Microscope Image Acquisition
4.5. Western Blot
4.6. Nuclear/Cytosol Subcellular Fractionation
4.7. Luciferase Assay
4.8. Real-Time-PCR
4.9. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mirra, S.; Marfany, G. Mitochondrial Gymnastics in Retinal Cells: A Resilience Mechanism Against Oxidative Stress and Neurodegeneration. In Advances in Experimental Medicine and Biology; Springer: Berlin/Heidelberg, Germany, 2019; Volume 1185, pp. 513–517. [Google Scholar]
- Olivares-González, L.; Velasco, S.; Campillo, I.; Rodrigo, R. Retinal Inflammation, Cell Death and Inherited Retinal Dystrophies. Int. J. Mol. Sci. 2021, 22, 2096. [Google Scholar] [CrossRef] [PubMed]
- De Dias, J.R.O.; Rodrigues, E.B.; Maia, M.; Magalhaƭes, O.; Penha, F.M.; Farah, M.E. Cytokines in neovascular age-related macular degeneration: Fundamentals of targeted combination therapy. Br. J. Ophthalmol. 2011, 95, 1631–1637. [Google Scholar] [CrossRef] [PubMed]
- Kinuthia, U.M.; Wolf, A.; Langmann, T. Microglia and Inflammatory Responses in Diabetic Retinopathy. Front. Immunol. 2020, 11, 564077. [Google Scholar] [CrossRef] [PubMed]
- Akhtar-Schäfer, I.; Wang, L.; Krohne, T.U.; Xu, H.; Langmann, T. Modulation of three key innate immune pathways for the most common retinal degenerative diseases. EMBO Mol. Med. 2018, 10, e8259. [Google Scholar] [CrossRef] [PubMed]
- Varfolomeev, E.; Vucic, D. Intracellular regulation of TNF activity in health and disease. Cytokine 2018, 101, 26–32. [Google Scholar] [CrossRef]
- Baeuerle, P.A.; Baltimore, D. Activation of DNA-binding activity in an apparently cytoplasmic precursor of the NF-kappa B transcription factor. Cell 1988, 53, 211–217. [Google Scholar] [CrossRef]
- Oeckinghaus, A.; Ghosh, S. The NF-kappaB family of transcription factors and its regulation. Cold Spring Harb. Perspect. Biol. 2009, 1, a000034. [Google Scholar] [CrossRef]
- Palombella, V.J.; Rando, O.J.; Goldberg, A.L.; Maniatis, T. The ubiquitin-proteasome pathway is required for processing the NF-kappa B1 precursor protein and the activation of NF-kappa B. Cell 1994, 78, 773–785. [Google Scholar] [CrossRef]
- Verzella, D.; Pescatore, A.; Capece, D.; Vecchiotti, D.; Ursini, M.V.; Franzoso, G.; Alesse, E.; Zazzeroni, F. Life, death, and autophagy in cancer: NF-κB turns up everywhere. Cell Death Dis. 2020, 11, 210. [Google Scholar] [CrossRef]
- Velimezi, G.; Robinson-Garcia, L.; Muñoz-Martínez, F.; Wiegant, W.W.; Da Silva, J.F.; Owusu, M.; Moder, M.; Wiedner, M.; Rosenthal, S.B.; Fisch, K.M.; et al. Map of synthetic rescue interactions for the Fanconi anemia DNA repair pathway identifies USP48. Nat. Commun. 2018, 9, 2280. [Google Scholar] [CrossRef]
- Ghanem, A.; Schweitzer, K.; Naumann, M. Catalytic domain of deubiquitinylase USP48 directs interaction with Rel homology domain of nuclear factor kappaB transcription factor RelA. Mol. Biol. Rep. 2019, 46, 1369–1375. [Google Scholar] [CrossRef] [PubMed]
- Zhou, A.; Lin, K.; Zhang, S.; Ma, L.; Xue, J.; Morris, S.; Aldape, K.D.; Huang, S. Gli1-induced deubiquitinase USP48 aids glioblastoma tumorigenesis by stabilizing Gli1. EMBO Rep. 2017, 18, 1318–1330. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Li, Y.; Kang, M.; Feng, M.; Ren, Y.; Dai, H.; Wang, Y.; Wang, Y.; Tang, B. USP48 Is Upregulated by Mettl14 to Attenuate Hepatocellular Carcinoma via Regulating SIRT6 Stabilization. Cancer Res. 2021, 81, 3822–3834. [Google Scholar] [CrossRef]
- Zhang, P.; Li, L.; Wang, B.; Ran, X.; Yang, S.; Luo, Y.; Li, Y.; Wang, Z.; Liu, Y.; Zhu, B. miR-489-3p promotes malignant progression of non-small cell lung cancer through the inactivation of Wnt/β-catenin signaling pathway via regulating USP48. Respir. Res. 2022, 23, 93. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.L.; Yan, J.; Zheng, J.; Tian, Q.B. 938 RHRK 941 is responsible for Ubiquitin specific protease 48 nuclear translocation which can stabilize NF-κB (p65) in the nucleus. Gene 2018, 669, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Schweitzer, K.; Naumann, M. CSN-associated USP48 confers stability to nuclear NF-κB/RelA by trimming K48-linked Ub-chains. Biochim. Biophys. Acta 2015, 1853, 453–469. [Google Scholar] [CrossRef] [PubMed]
- Uckelmann, M.; Densham, R.M.; Baas, R.; Winterwerp, H.H.K.; Fish, A.; Sixma, T.K.; Morris, J.R. USP48 restrains resection by site-specific cleavage of the BRCA1 ubiquitin mark from H2A. Nat. Commun. 2018, 9, 229. [Google Scholar] [CrossRef] [PubMed]
- Pahl, H.L. Activators and target genes of Rel/NF-kappaB transcription factors. Oncogene 1999, 18, 6853–6866. [Google Scholar] [CrossRef] [PubMed]
- Stein, B.; Cogswell, P.C.; Baldwin, A.S. Functional and physical associations between NF-kappa B and C/EBP family members: A Rel domain-bZIP interaction. Mol. Cell. Biol. 1993, 13, 3964–3974. [Google Scholar] [CrossRef]
- Fagerlund, R.; Melén, K.; Cao, X.; Julkunen, I. NF-κB p52, RelB and c-Rel are transported into the nucleus via a subset of importin α molecules. Cell. Signal. 2008, 20, 1442–1451. [Google Scholar] [CrossRef]
- Fagerlund, R.; Kinnunen, L.; Köhler, M.; Julkunen, I.; Melén, K. NF-κB is transported into the nucleus by importin α3 and importin α4. J. Biol. Chem. 2005, 280, 15942–15951. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Yang, X.D.; Lamb, A.; Chen, L.F. Posttranslational modifications of NF-kappaB: Another layer of regulation for NF-kappaB signaling pathway. Cell. Signal. 2010, 22, 1282–1290. [Google Scholar] [CrossRef] [PubMed]
- Zerfaoui, M.; Errami, Y.; Naura, A.S.; Suzuki, Y.; Kim, H.; Ju, J.; Liu, T.; Hans, C.P.; Kim, J.G.; Abd Elmageed, Z.Y.; et al. Poly(ADP-ribose) polymerase-1 is a determining factor in Crm1-mediated nuclear export and retention of p65 NF-kappa B upon TLR4 stimulation. J. Immunol. 2010, 185, 1894–1902. [Google Scholar] [CrossRef] [PubMed]
- Ren, R.; Wang, N.; Zhang, X.; Tian, G.; Jonas, J.B. Cerebrospinal fluid pressure correlated with body mass index. Graefes Arch. Clin. Exp. Ophthalmol. 2012, 250, 445–446. [Google Scholar] [CrossRef]
- Strebovsky, J.; Walker, P.; Lang, R.; Dalpke, A.H. Suppressor of cytokine signaling 1 (SOCS1) limits NFkappaB signaling by decreasing p65 stability within the cell nucleus. FASEB J. 2011, 25, 863–874. [Google Scholar] [CrossRef]
- Ryo, A.; Suizu, F.; Yoshida, Y.; Perrem, K.; Liou, Y.C.; Wulf, G.; Rottapel, R.; Yamaoka, S.; Lu, K.P. Regulation of NF-kappaB signaling by Pin1-dependent prolyl isomerization and ubiquitin-mediated proteolysis of p65/RelA. Mol. Cell 2003, 12, 1413–1426. [Google Scholar] [CrossRef]
- Dubiel, W.; Chaithongyot, S.; Dubiel, D.; Naumann, M. The COP9 Signalosome: A Multi-DUB Complex. Biomolecules 2020, 10, 1082. [Google Scholar] [CrossRef]
- Medzhitov, R.; Horng, T. Transcriptional control of the inflammatory response. Nat. Rev. Immunol. 2009, 9, 692–703. [Google Scholar] [CrossRef]
- Lee, R.E.C.; Walker, S.R.; Savery, K.; Frank, D.A.; Gaudet, S. Fold change of nuclear NF-κB determines TNF-induced transcription in single cells. Mol. Cell 2014, 53, 867–879. [Google Scholar] [CrossRef]
- Janbandhu, V.C.; Singh, A.K.; Mukherji, A.; Kumar, V. p65 Negatively regulates transcription of the cyclin E gene. J. Biol. Chem. 2010, 285, 17453–17464. [Google Scholar] [CrossRef]
- Detrick, B.; Hooks, J.J. The RPE Cell and the Immune System. Retin. Pigment Ep. Health Dis. 2020, 101–114. [Google Scholar] [CrossRef]
- Collins, P.E.; Mitxitorena, I.; Carmody, R.J. The Ubiquitination of NF-κB Subunits in the Control of Transcription. Cells 2016, 5, 23. [Google Scholar] [CrossRef]
- Hanpude, P.; Bhattacharya, S.; Dey, A.K.; Maiti, T.K. Deubiquitinating enzymes in cellular signaling and disease regulation. IUBMB Life 2015, 67, 544–555. [Google Scholar] [CrossRef] [PubMed]
- Harhaj, E.W.; Dixit, V.M. Regulation of NF-κB by deubiquitinases. Immunol. Rev. 2012, 246, 107–124. [Google Scholar] [CrossRef] [PubMed]
- Colleran, A.; Collins, P.E.; O’Carroll, C.; Ahmed, A.; Mao, X.; McManus, B.; Kiely, P.A.; Burstein, E.; Carmody, R.J. Deubiquitination of NF-κB by Ubiquitin-Specific Protease-7 promotes transcription. Proc. Natl. Acad. Sci. USA 2013, 110, 618–623. [Google Scholar] [CrossRef]
- Tse, W.K.F.; Eisenhaber, B.; Ho, S.H.K.; Ng, Q.; Eisenhaber, F.; Jiang, Y.J. Genome-wide loss-of-function analysis of deubiquitylating enzymes for zebrafish development. BMC Genom. 2009, 10, 637. [Google Scholar] [CrossRef]
- Esquerdo-Barragán, M.; Brooks, M.J.; Toulis, V.; Swaroop, A.; Marfany, G. Expression of deubiquitinating enzyme genes in the developing mammal retina. Mol. Vis. 2019, 25, 800. [Google Scholar]
- Alzahrani, A.B. Gene Expression in Proliferative Diabetic Retinopathy Using RNA-Seq Data: A Computational Study on Saudi Patients. Biosci. Biotechnol. Res. Commun. 2021, 14, 1760–1763. [Google Scholar] [CrossRef]
- Tzimas, C.; Michailidou, G.; Arsenakis, M.; Kieff, E.; Mosialos, G.; Hatzivassiliou, E.G. Human ubiquitin specific protease 31 is a deubiquitinating enzyme implicated in activation of nuclear factor-kappaB. Cell. Signal. 2006, 18, 83–92. [Google Scholar] [CrossRef]
- Zhang, C.; Peng, Z.; Zhu, M.; Wang, P.; Du, X.; Li, X.; Liu, Y.; Jin, Y.; McNutt, M.A.; Yin, Y. USP9X destabilizes pVHL and promotes cell proliferation. Oncotarget 2016, 7, 60519–60534. [Google Scholar] [CrossRef][Green Version]
- Saccani, S.; Marazzi, I.; Beg, A.A.; Natoli, G. Degradation of promoter-bound p65/RelA is essential for the prompt termination of the nuclear factor kappaB response. J. Exp. Med. 2004, 200, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, T.; Bebien, M.; Liu, G.Y.; Nizet, V.; Karin, M. IKKalpha limits macrophage NF-kappaB activation and contributes to the resolution of inflammation. Nature 2005, 434, 1138–1143. [Google Scholar] [CrossRef] [PubMed]
- Ankers, J.M.; Awais, R.; Jones, N.A.; Boyd, J.; Ryan, S.; Adamson, A.D.; Harper, C.V.; Bridge, L.; Spiller, D.G.; Jackson, D.A.; et al. Dynamic NF-κB and E2F interactions control the priority and timing of inflammatory signalling and cell proliferation. eLife 2016, 5, e10473. [Google Scholar] [CrossRef]
- Li, L.; Wang, Y.; Zhang, X.; Song, G.; Guo, Q.; Zhang, Z.; Diao, Y.; Yin, H.; Liu, H.; Jiang, G. Deubiquitinase USP48 promotes ATRA-induced granulocytic differentiation of acute promyelocytic leukemia cells. Int. J. Oncol. 2018, 53, 895–903. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.; Yang, C.; Bao, X.; Wang, R. Genetic and Epigenetic Causes of Pituitary Adenomas. Front. Endocrinol. (Lausanne) 2021, 11, 596554. [Google Scholar] [CrossRef] [PubMed]
- Treppiedi, D.; Barbieri, A.M.; Di Muro, G.; Marra, G.; Mangili, F.; Catalano, R.; Esposito, E.; Ferrante, E.; Serban, A.L.; Locatelli, M.; et al. Genetic Profiling of a Cohort of Italian Patients with ACTH-Secreting Pituitary Tumors and Characterization of a Novel USP8 Gene Variant. Cancers 2021, 13, 4022. [Google Scholar] [CrossRef] [PubMed]
- Lichtlen, P.; Lam, T.T.; Nork, T.M.; Streit, T.; Urech, D.M. Relative contribution of VEGF and TNF-alpha in the cynomolgus laser-induced CNV model: Comparing the efficacy of bevacizumab, adalimumab, and ESBA105. Investig. Ophthalmol. Vis. Sci. 2010, 51, 4738–4745. [Google Scholar] [CrossRef]
- Al-Gayyar, M.M.; Elsherbiny, N.M. Contribution of TNF-α to the development of retinal neurodegenerative disorders. Eur. Cytokine Netw. 2013, 24, 27–36. [Google Scholar] [CrossRef]
- Regatieri, C.V.; Dreyfuss, J.L.; Melo, G.B.; Lavinsky, D.; Farah, M.E.; Nader, H.B. Dual role of intravitreous infliximab in experimental choroidal neovascularization: Effect on the expression of sulfated glycosaminoglycans. Investig. Ophthalmol. Vis. Sci. 2009, 50, 5487–5494. [Google Scholar] [CrossRef]
- Shi, X.; Semkova, I.; Müther, P.S.; Dell, S.; Kociok, N.; Joussen, A.M. Inhibition of TNF-alpha reduces laser-induced choroidal neovascularization. Exp. Eye Res. 2006, 83, 1325–1334. [Google Scholar] [CrossRef]
- Esposito, E.; Napolitano, G.; Pescatore, A.; Calculli, G.; Incoronato, M.R.; Leonardi, A.; Ursini, M.V. COMMD7 as a novel NEMO interacting protein involved in the termination of NF-κB signaling. J. Cell. Physiol. 2016, 231, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Denuc, A.; Bosch-Comas, A.; Gonzàlez-Duarte, R.; Marfany, G. The UBA-UIM domains of the USP25 regulate the enzyme ubiquitination state and modulate substrate recognition. PLoS ONE 2009, 4, e5571. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mirra, S.; Sánchez-Bellver, L.; Casale, C.; Pescatore, A.; Marfany, G. Ubiquitin Specific Protease USP48 Destabilizes NF-κB/p65 in Retinal Pigment Epithelium Cells. Int. J. Mol. Sci. 2022, 23, 9682. https://doi.org/10.3390/ijms23179682
Mirra S, Sánchez-Bellver L, Casale C, Pescatore A, Marfany G. Ubiquitin Specific Protease USP48 Destabilizes NF-κB/p65 in Retinal Pigment Epithelium Cells. International Journal of Molecular Sciences. 2022; 23(17):9682. https://doi.org/10.3390/ijms23179682
Chicago/Turabian StyleMirra, Serena, Laura Sánchez-Bellver, Carmela Casale, Alessandra Pescatore, and Gemma Marfany. 2022. "Ubiquitin Specific Protease USP48 Destabilizes NF-κB/p65 in Retinal Pigment Epithelium Cells" International Journal of Molecular Sciences 23, no. 17: 9682. https://doi.org/10.3390/ijms23179682
APA StyleMirra, S., Sánchez-Bellver, L., Casale, C., Pescatore, A., & Marfany, G. (2022). Ubiquitin Specific Protease USP48 Destabilizes NF-κB/p65 in Retinal Pigment Epithelium Cells. International Journal of Molecular Sciences, 23(17), 9682. https://doi.org/10.3390/ijms23179682





