Investigation of the Associations of Novel Inflammatory Biomarkers—Systemic Inflammatory Index (SII) and Systemic Inflammatory Response Index (SIRI)—With the Severity of Coronary Artery Disease and Acute Coronary Syndrome Occurrence
Abstract
:1. Introduction
2. Results
2.1. Characteristics of Participants
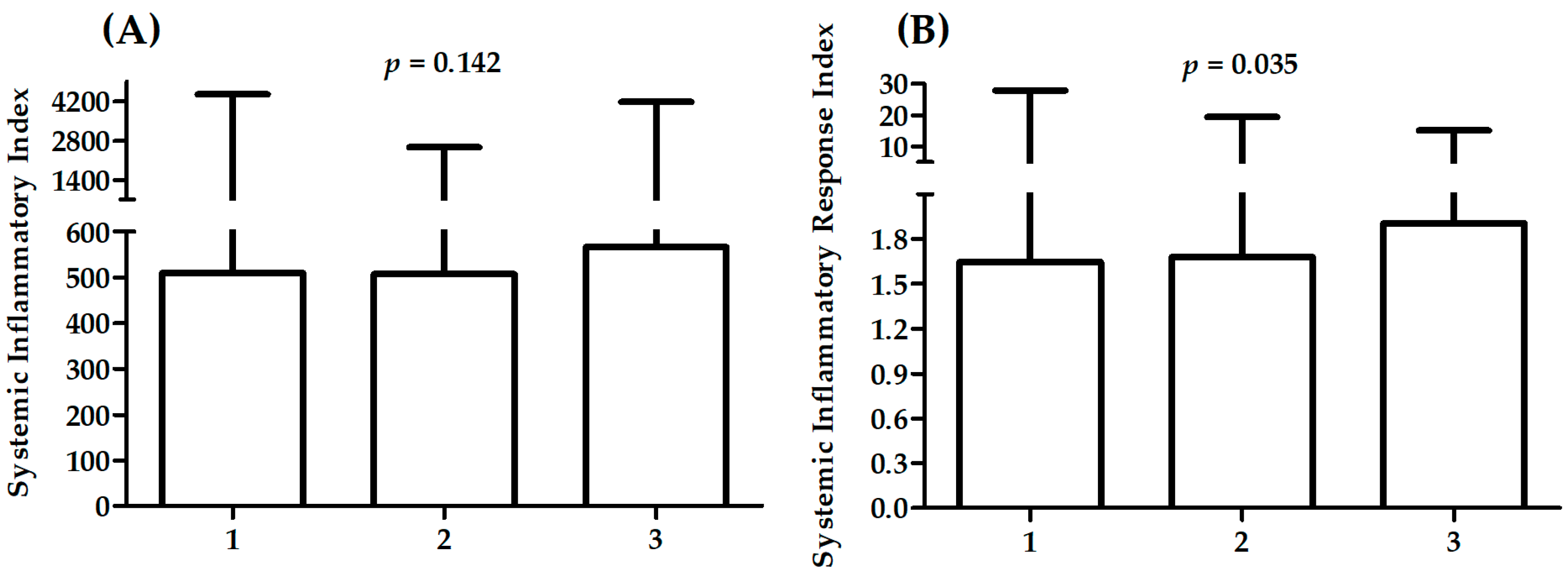
2.2. Association between Measured and Derived Parameters and Severity of Cad
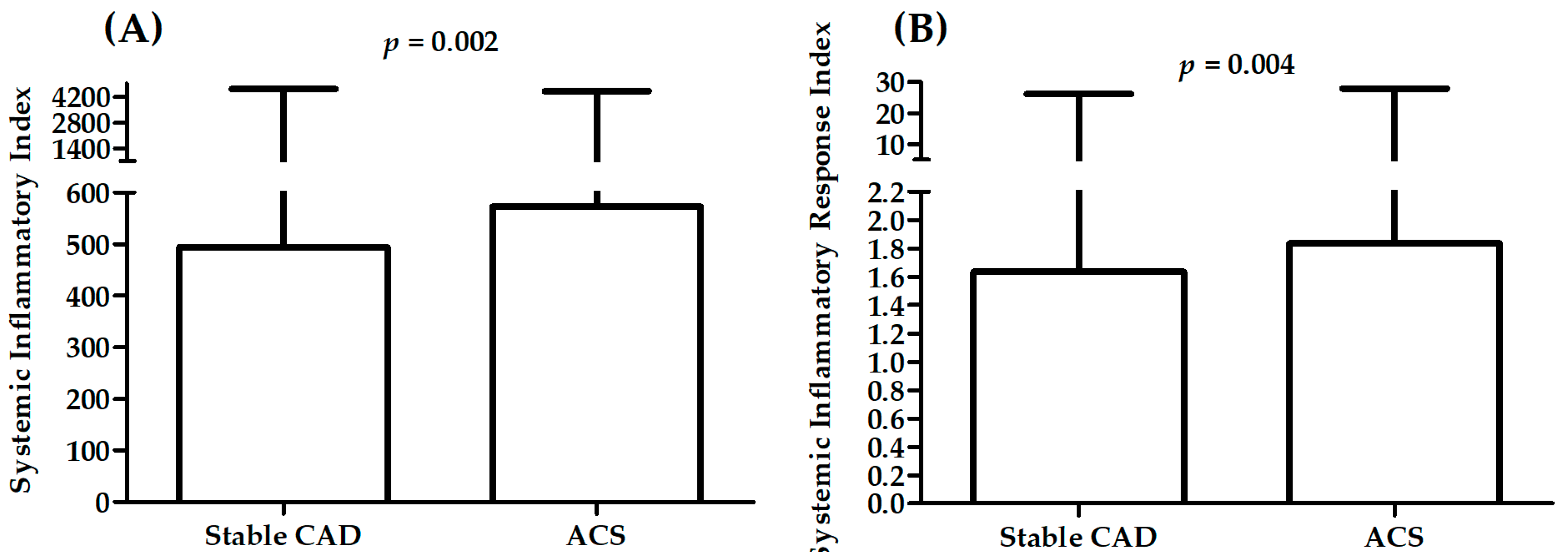
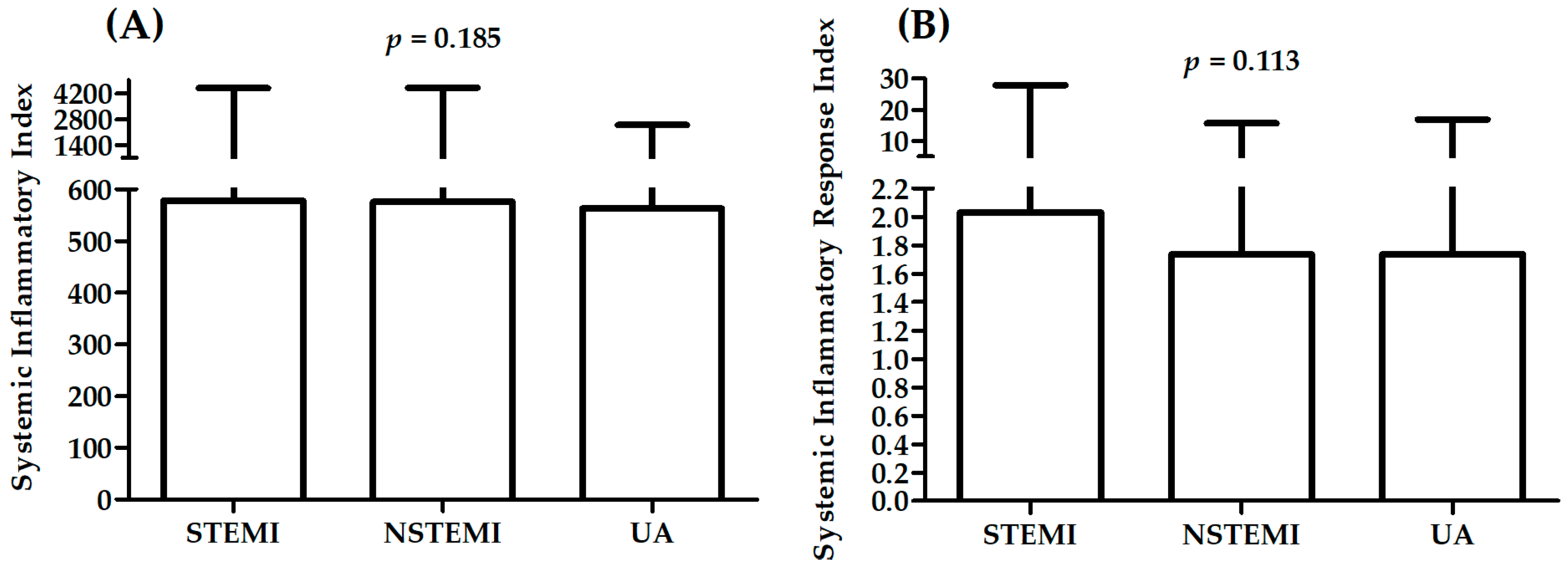
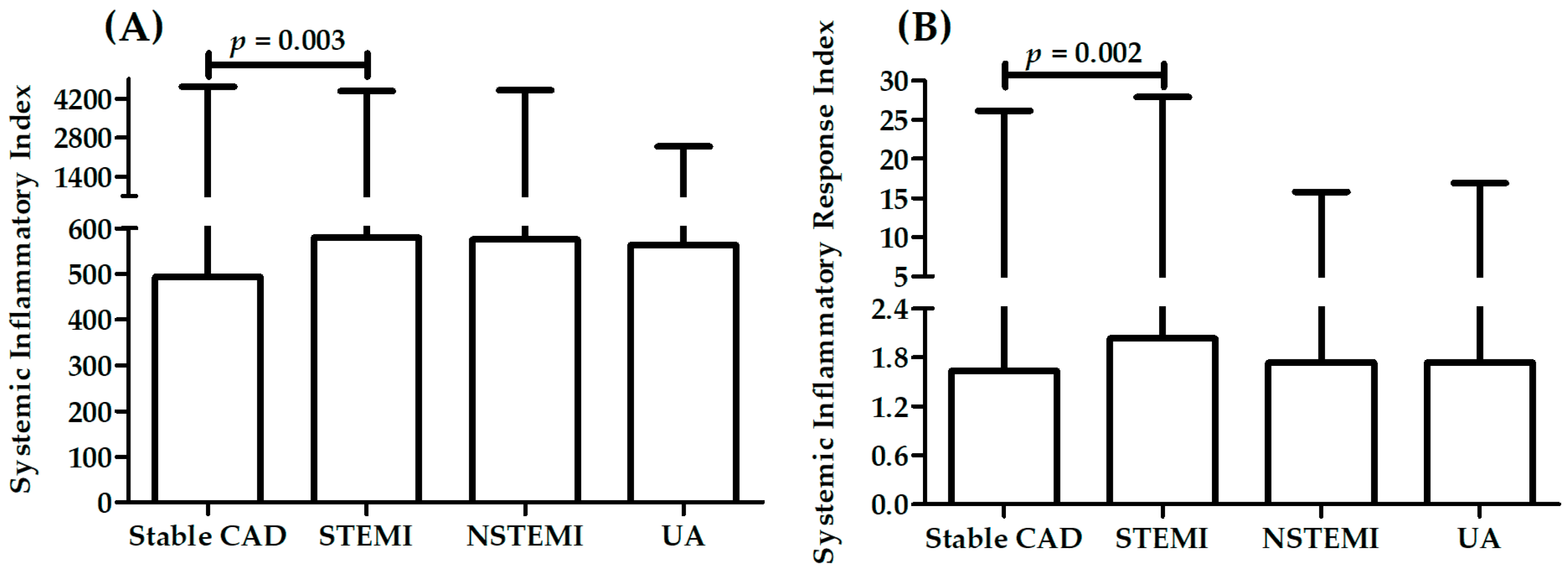
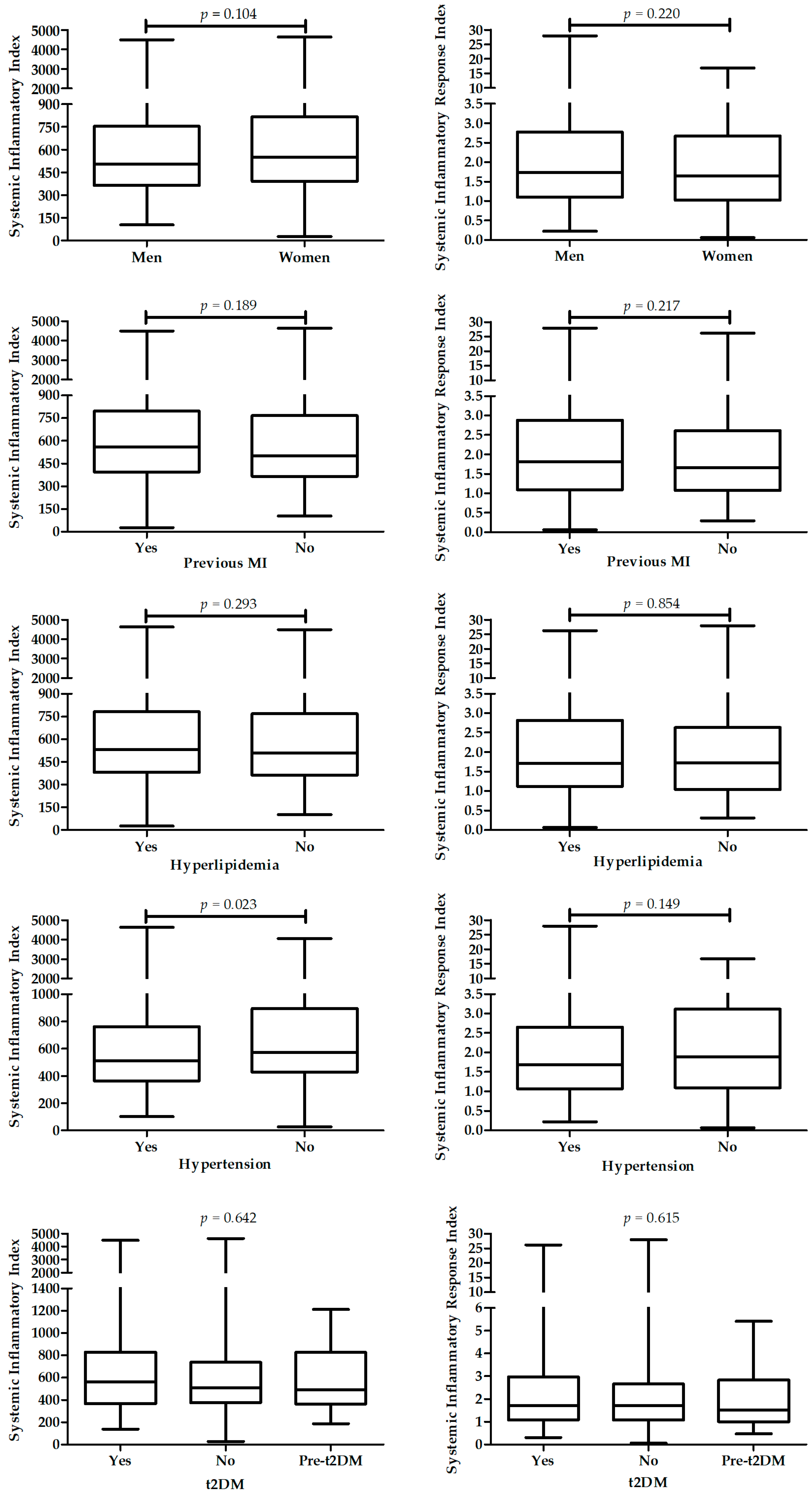
2.3. Association between Measured Parameters and Diagnosis
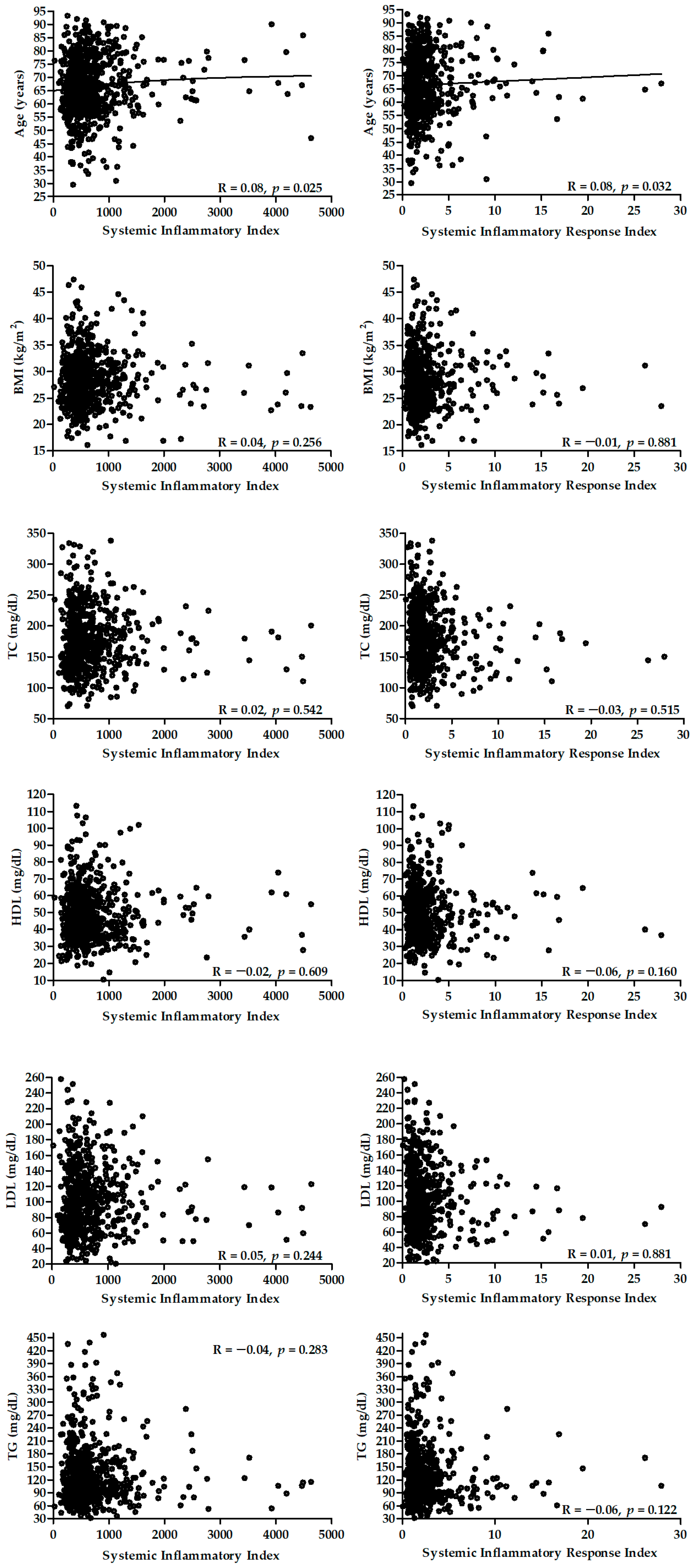
2.4. Association of Sii and Siri with Selected Parameters
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Clinical and Laboratory Data
4.3. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organisation. Global Health Estimates: Life Expectancy and Leading Causes of Death and Disability. Available online: https://www.who.int/data/gho/data/themes/mortality-and-global-health-estimates (accessed on 26 July 2022).
- Björkegren, J.L.M.; Lusis, A.J. Atherosclerosis: Recent developments. Cell 2022, 185, 1630–1645. [Google Scholar] [CrossRef] [PubMed]
- Crea, F.; Libby, P. Acute Coronary Syndromes: The Way Forward from Mechanisms to Precision Treatment. Circulation 2017, 136, 1155–1166. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Pasterkamp, G.; Crea, F.; Jang, I.-K. Reassessing the Mechanisms of Acute Coronary Syndromes. Circ. Res. 2019, 124, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Hansson, G.K. From Focal Lipid Storage to Systemic Inflammation: JACC Review Topic of the Week. J. Am. Coll. Cardiol. 2019, 74, 1594–1607. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Liu, Z.; Shao, J.; Lin, L.; Jiang, M.; Wang, L.; Lu, X.; Zhang, H.; Chen, Y.; Zhang, R. Immune and Inflammation in Acute Coronary Syndrome: Molecular Mechanisms and Therapeutic Implications. J. Immunol. Res. 2020, 2020, 4904217. [Google Scholar] [CrossRef] [PubMed]
- Spirig, R.; Tsui, J.; Shaw, S. The Emerging Role of TLR and Innate Immunity in Cardiovascular Disease. Cardiol. Res. Pract. 2012, 2012, 181394. [Google Scholar] [CrossRef] [Green Version]
- Pant, S.; Deshmukh, A.; Gurumurthy, G.S.; Pothineni, N.V.; Watts, T.E.; Romeo, F.; Mehta, J.L. Inflammation and atherosclerosis—Revisited. J. Cardiovasc. Pharmacol. Ther. 2014, 19, 170–178. [Google Scholar] [CrossRef]
- Buckley, D.I.; Fu, R.; Freeman, M.; Rogers, K.; Helfand, M. C-reactive protein as a risk factor for coronary heart disease: A systematic review and meta-analyses for the U.S. Preventive Services Task Force. Ann. Intern. Med. 2009, 151, 483–495. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.; Han, W.; Gong, D.; Man, C.; Fan, Y. Hs-CRP in stroke: A meta-analysis. Clin. Chim. Acta 2016, 453, 21–27. [Google Scholar] [CrossRef]
- Kim, J.H.; Lim, S.; Park, K.S.; Jang, H.C.; Choi, S.H. Total and differential WBC counts are related with coronary artery atherosclerosis and increase the risk for cardiovascular disease in Koreans. PLoS ONE 2017, 12, e0180332. [Google Scholar] [CrossRef] [Green Version]
- Wu, T.-H.; Chien, K.-L.; Lin, H.-J.; Hsu, H.-C.; Su, T.-C.; Chen, M.-F.; Lee, Y.-T. Total white blood cell count or neutrophil count predict ischemic stroke events among adult Taiwanese: Report from a community-based cohort study. BMC Neurol. 2013, 13, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zia, E.; Melander, O.; Björkbacka, H.; Hedblad, B.; Engström, G. Total and differential leucocyte counts in relation to incidence of stroke subtypes and mortality: A prospective cohort study. J. Intern. Med. 2012, 272, 298–304. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wheeler, J.G.; Mussolino, M.E.; Gillum, R.F.; Danesh, J. Associations between differential leucocyte count and incident coronary heart disease: 1764 incident cases from seven prospective studies of 30,374 individuals. Eur. Heart J. 2004, 25, 1287–1292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, T.; Jiang, C.Q.; Xu, L.; Zhang, W.S.; Zhu, F.; Jin, Y.L.; Thomas, G.N.; Cheng, K.K.; Lam, T.H. White blood cell count and all-cause and cause-specific mortality in the Guangzhou biobank cohort study. BMC Public Health 2018, 18, 1232. [Google Scholar] [CrossRef] [Green Version]
- Abete, I.; Lu, Y.; Lassale, C.; Verschuren, M.; van der Schouw, Y.; Bueno-de-Mesquita, B. White cell counts in relation to mortality in a general population of cohort study in the Netherlands: A mediating effect or not? BMJ Open 2019, 9, e030949. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fan, Z.; Ji, H.; Li, Y.; Jian, X.; Li, L.; Liu, T. Relationship between monocyte-to-lymphocyte ratio and coronary plaque vulnerability in patients with stable angina. Biomark. Med. 2017, 11, 979–990. [Google Scholar] [CrossRef] [PubMed]
- Ateş, A.H.; Aytemir, K.; Koçyiğit, D.; Yalcin, M.U.; Gürses, K.M.; Yorgun, H.; Canpolat, U.; Hazırolan, T.; Özer, N. Association of Neutrophil-to-Lymphocyte Ratio with the Severity and Morphology of Coronary Atherosclerotic Plaques Detected by Multidetector Computerized Tomography. Acta Cardiol. Sin. 2016, 32, 676–683. [Google Scholar] [CrossRef]
- Balta, S.; Ozturk, C. The platelet-lymphocyte ratio: A simple, inexpensive and rapid prognostic marker for cardiovascular events. Platelets 2015, 26, 680–681. [Google Scholar] [CrossRef]
- Geng, Y.; Zhu, D.; Wu, C.; Wu, J.; Wang, Q.; Li, R.; Jiang, J.; Wu, C. A novel systemic inflammation response index (SIRI) for predicting postoperative survival of patients with esophageal squamous cell carcinoma. Int. Immunopharmacol. 2018, 65, 503–510. [Google Scholar] [CrossRef]
- Xie, Q.-K.; Chen, P.; Hu, W.-M.; Sun, P.; He, W.-Z.; Jiang, C.; Kong, P.-F.; Liu, S.-S.; Chen, H.-T.; Yang, Y.-Z.; et al. The systemic immune-inflammation index is an independent predictor of survival for metastatic colorectal cancer and its association with the lymphocytic response to the tumor. J. Transl. Med. 2018, 16, 273. [Google Scholar] [CrossRef] [Green Version]
- Huang, J.; Zhang, Q.; Wang, R.; Ji, H.; Chen, Y.; Quan, X.; Zhang, C. Systemic Immune-Inflammatory Index Predicts Clinical Outcomes for Elderly Patients with Acute Myocardial Infarction Receiving Percutaneous Coronary Intervention. Med. Sci. Monit. 2019, 25, 9690–9701. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Yang, X.-R.; Xu, Y.; Sun, Y.-F.; Sun, C.; Guo, W.; Zhang, X.; Wang, W.-M.; Qiu, S.-J.; Zhou, J.; et al. Systemic immune-inflammation index predicts prognosis of patients after curative resection for hepatocellular carcinoma. Clin. Cancer Res. 2014, 20, 6212–6222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erdoğan, M.; Erdöl, M.A.; Öztürk, S.; Durmaz, T. Systemic immune-inflammation index is a novel marker to predict functionally significant coronary artery stenosis. Biomark. Med. 2020, 14, 1553–1561. [Google Scholar] [CrossRef]
- Liu, Y.; Ye, T.; Chen, L.; Jin, T.; Sheng, Y.; Wu, G.; Zong, G. Systemic immune-inflammation index predicts the severity of coronary stenosis in patients with coronary heart disease. Coron. Artery Dis. 2021, 32, 715–720. [Google Scholar] [CrossRef] [PubMed]
- Candemir, M.; Kiziltunç, E.; Nurkoç, S.; Şahinarslan, A. Relationship Between Systemic Immune-Inflammation Index (SII) and the Severity of Stable Coronary Artery Disease. Angiology 2021, 72, 575–581. [Google Scholar] [CrossRef]
- Kelesoglu, S.; Yilmaz, Y.; Elcık, D.; Kalay, N. Systemic immune inflammation index: A novel predictor for coronary collateral circulation. Perfusion 2021, 2676591211014822. [Google Scholar] [CrossRef]
- Seo, M.; Yamada, T.; Morita, T.; Furukawa, Y.; Tamaki, S.; Iwasaki, Y.; Kawasaki, M.; Kikuchi, A.; Kawai, T.; Ikeda, I.; et al. P589Prognostic value of systemic immune-inflammation index in patients with chronic heart failure. Eur. Heart J. 2018, 39. [Google Scholar] [CrossRef]
- Li, Q.; Ma, X.; Shao, Q.; Yang, Z.; Wang, Y.; Gao, F.; Zhou, Y.; Yang, L.; Wang, Z. Prognostic Impact of Multiple Lymphocyte-Based Inflammatory Indices in Acute Coronary Syndrome Patients. Front. Cardiovasc. Med. 2022, 9, 811790. [Google Scholar] [CrossRef]
- Yang, Y.-L.; Wu, C.-H.; Hsu, P.-F.; Chen, S.-C.; Huang, S.-S.; Chan, W.L.; Lin, S.-J.; Chou, C.-Y.; Chen, J.-W.; Pan, J.-P.; et al. Systemic immune-inflammation index (SII) predicted clinical outcome in patients with coronary artery disease. Eur. J. Clin. Investig. 2020, 50, e13230. [Google Scholar] [CrossRef]
- Urbanowicz, T.; Michalak, M.; Al-Imam, A.; Olasińska-Wiśniewska, A.; Rodzki, M.; Witkowska, A.; Haneya, A.; Buczkowski, P.; Perek, B.; Jemielity, M. The Significance of Systemic Immune-Inflammatory Index for Mortality Prediction in Diabetic Patients Treated with Off-Pump Coronary Artery Bypass Surgery. Diagnostics 2022, 12, 634. [Google Scholar] [CrossRef]
- Tosu, A.R.; Kalyoncuoglu, M.; Biter, H.İ.; Cakal, S.; Selcuk, M.; Çinar, T.; Belen, E.; Can, M.M. Prognostic Value of Systemic Immune-Inflammation Index for Major Adverse Cardiac Events and Mortality in Severe Aortic Stenosis Patients after TAVI. Medicina 2021, 57, 588. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.; Jung, J.; Ahn, Y.; Oh, J. Systemic Immune-Inflammation Index Predicted Short-Term Outcomes in Patients Undergoing Isolated Tricuspid Valve Surgery. J. Clin. Med. 2021, 10, 4147. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Zhang, J.; Liu, T.; Yin, Z.; Jin, Y.; Han, J.; Guo, Z.; Wang, H. The systemic-immune-inflammation index predicts the recurrence of atrial fibrillation after cryomaze concomitant with mitral valve surgery. BMC Cardiovasc. Disord. 2022, 22, 45. [Google Scholar] [CrossRef] [PubMed]
- Gok, M.; Kurtul, A. A novel marker for predicting severity of acute pulmonary embolism: Systemic immune-inflammation index. Scand. Cardiovasc. J. 2021, 55, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Li, D.; Xu, T.; Chen, Z.; Shan, Y.; Zhao, L.; Fu, G.; Luan, Y.; Xia, S.; Zhang, W. Systemic Immune-Inflammation Index Predicts Contrast-Induced Acute Kidney Injury in Patients Undergoing Coronary Angiography: A Cross-Sectional Study. Front. Med. 2022, 9, 841601. [Google Scholar] [CrossRef]
- Kelesoglu, S.; Yilmaz, Y.; Elcık, D.; Çetınkaya, Z.; Inanc, M.T.; Dogan, A.; Oguzhan, A.; Kalay, N. Systemic Immune Inflammation Index: A Novel Predictor of Contrast-Induced Nephropathy in Patients with Non-ST Segment Elevation Myocardial Infarction. Angiology 2021, 72, 889–895. [Google Scholar] [CrossRef]
- Selcuk, M.; Cinar, T.; Saylik, F.; Dogan, S.; Selcuk, I.; Orhan, A.L. Predictive Value of Systemic Immune Inflammation Index for Postoperative Atrial Fibrillation in Patients Undergoing Isolated Coronary Artery Bypass Grafting. Med. Med. J. 2021, 36, 318–324. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, W.; Yu, H.; Chen, Z.; Zhang, C.; Ti, Y.; Bu, P. Value of the Systemic Immune-Inflammatory Index (SII) in Predicting the Prognosis of Patients With Peripartum Cardiomyopathy. Front. Cardiovasc. Med. 2022, 9, 811079. [Google Scholar] [CrossRef]
- Chao, B.; Ju, X.; Zhang, L.; Xu, X.; Zhao, Y. A Novel Prognostic Marker Systemic Inflammation Response Index (SIRI) for Operable Cervical Cancer Patients. Front. Oncol. 2020, 10, 766. [Google Scholar] [CrossRef]
- Jin, Z.; Wu, Q.; Chen, S.; Gao, J.; Li, X.; Zhang, X.; Zhou, Y.; He, D.; Cheng, Z.; Zhu, Y.; et al. The Associations of Two Novel Inflammation Indexes, SII and SIRI with the Risks for Cardiovascular Diseases and All-Cause Mortality: A Ten-Year Follow-Up Study in 85,154 Individuals. J. Inflamm. Res. 2021, 14, 131–140. [Google Scholar] [CrossRef]
- Lin, K.-B.; Fan, F.-H.; Cai, M.-Q.; Yu, Y.; Fu, C.-L.; Ding, L.-Y.; Sun, Y.-D.; Sun, J.-W.; Shi, Y.-W.; Dong, Z.-F.; et al. Systemic immune inflammation index and system inflammation response index are potential biomarkers of atrial fibrillation among the patients presenting with ischemic stroke. Eur. J. Med. Res. 2022, 27, 106. [Google Scholar] [CrossRef] [PubMed]
- Han, K.; Shi, D.; Yang, L.; Wang, Z.; Li, Y.; Gao, F.; Liu, Y.; Ma, X.; Zhou, Y. Prognostic value of systemic inflammatory response index in patients with acute coronary syndrome undergoing percutaneous coronary intervention. Ann. Med. 2022, 54, 1667–1677. [Google Scholar] [CrossRef] [PubMed]
- Dziedzic, E.A.; Gąsior, J.S.; Tuzimek, A.; Dąbrowski, M.; Jankowski, P. Neutrophil-to-Lymphocyte Ratio Is Not Associated with Severity of Coronary Artery Disease and Is Not Correlated with Vitamin D Level in Patients with a History of an Acute Coronary Syndrome. Biology 2022, 11, 1001. [Google Scholar] [CrossRef]
- Dziedzic, E.A.; Gąsior, J.S.; Sowińska, I.; Dąbrowski, M.; Jankowski, P. Vitamin D Level in Patients with Consecutive Acute Coronary Syndrome Is Not Correlated with the Parameters of Platelet Activity. J. Clin. Med. 2022, 11, 707. [Google Scholar] [CrossRef] [PubMed]
- Chistiakov, D.A.; Kashirskikh, D.A.; Khotina, V.A.; Grechko, A.V.; Orekhov, A.N. Immune-Inflammatory Responses in Atherosclerosis: The Role of Myeloid Cells. J. Clin. Med. 2019, 8, 1798. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Libby, P.; Ridker, P.M.; Hansson, G.K. Inflammation in atherosclerosis: From pathophysiology to practice. J. Am. Coll. Cardiol. 2009, 54, 2129–2138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montecucco, F.; Liberale, L.; Bonaventura, A.; Vecchiè, A.; Dallegri, F.; Carbone, F. The Role of Inflammation in Cardiovascular Outcome. Curr. Atheroscler. Rep. 2017, 19, 11. [Google Scholar] [CrossRef]
- Möckel, M.; Danne, O.; Müller, R.; Vollert, J.O.; Müller, C.; Lueders, C.; Störk, T.; Frei, U.; Koenig, W.; Dietz, R.; et al. Development of an optimized multimarker strategy for early risk assessment of patients with acute coronary syndromes. Clin. Chim. Acta 2008, 393, 103–109. [Google Scholar] [CrossRef]
- Hatmi, Z.N.; Saeid, A.K.; Broumand, M.A.; Khoshkar, S.N.; Danesh, Z.F. Multiple inflammatory prognostic factors in acute coronary syndromes: A prospective inception cohort study. Acta Med. Iran. 2010, 48, 51–57. [Google Scholar]
- Ikonomidis, I.; Michalakeas, C.A.; Parissis, J.; Paraskevaidis, I.; Ntai, K.; Papadakis, I.; Anastasiou-Nana, M.; Lekakis, J. Inflammatory markers in coronary artery disease. Biofactors 2012, 38, 320–328. [Google Scholar] [CrossRef]
- Blaschke, F.; Bruemmer, D.; Yin, F.; Takata, Y.; Wang, W.; Fishbein, M.C.; Okura, T.; Higaki, J.; Graf, K.; Fleck, E.; et al. C-reactive protein induces apoptosis in human coronary vascular smooth muscle cells. Circulation 2004, 110, 579–587. [Google Scholar] [CrossRef] [PubMed]
- de Lemos, J.A.; Morrow, D.A.; Blazing, M.A.; Jarolim, P.; Wiviott, S.D.; Sabatine, M.S.; Califf, R.M.; Braunwald, E. Serial measurement of monocyte chemoattractant protein-1 after acute coronary syndromes: Results from the A to Z trial. J. Am. Coll. Cardiol. 2007, 50, 2117–2124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaptoge, S.; Di Angelantonio, E.; Pennells, L.; Wood, A.M.; White, I.R.; Gao, P.; Walker, M.; Thompson, A.; Sarwar, N.; Caslake, M.; et al. C-reactive protein, fibrinogen, and cardiovascular disease prediction. N. Engl. J. Med. 2012, 367, 1310–1320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eisen, A.; Giugliano, R.P.; Braunwald, E. Updates on Acute Coronary Syndrome: A Review. JAMA Cardiol. 2016, 1, 718–730. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Everett, B.M.; Thuren, T.; MacFadyen, J.G.; Chang, W.H.; Ballantyne, C.; Fonseca, F.; Nicolau, J.; Koenig, W.; Anker, S.D.; et al. Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N. Engl. J. Med. 2017, 377, 1119–1131. [Google Scholar] [CrossRef]
- Antoniades, C.; Antonopoulos, A.S.; Deanfield, J. Imaging residual inflammatory cardiovascular risk. Eur. Heart J. 2020, 41, 748–758. [Google Scholar] [CrossRef] [Green Version]
- Pradhan, A.D.; Aday, A.W.; Rose, L.M.; Ridker, P.M. Residual Inflammatory Risk on Treatment with PCSK9 Inhibition and Statin Therapy. Circulation 2018, 138, 141–149. [Google Scholar] [CrossRef]
- Ridker, P.M. Anticytokine Agents: Targeting Interleukin Signaling Pathways for the Treatment of Atherothrombosis. Circ. Res. 2019, 124, 437–450. [Google Scholar] [CrossRef]
- Libby, P.; Tabas, I.; Fredman, G.; Fisher, E.A. Inflammation and its resolution as determinants of acute coronary syndromes. Circ. Res. 2014, 114, 1867–1879. [Google Scholar] [CrossRef] [Green Version]
- Shah, A.D.; Denaxas, S.; Nicholas, O.; Hingorani, A.D.; Hemingway, H. Low eosinophil and low lymphocyte counts and the incidence of 12 cardiovascular diseases: A CALIBER cohort study. Open Heart 2016, 3, e000477. [Google Scholar] [CrossRef] [Green Version]
- Horne, B.D.; Anderson, J.L.; John, J.M.; Weaver, A.; Bair, T.L.; Jensen, K.R.; Renlund, D.G.; Muhlestein, J.B. Which white blood cell subtypes predict increased cardiovascular risk? J. Am. Coll. Cardiol. 2005, 45, 1638–1643. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gratchev, A.; Sobenin, I.; Orekhov, A.; Kzhyshkowska, J. Monocytes as a diagnostic marker of cardiovascular diseases. Immunobiology 2012, 217, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.-Y.; Li, C.-J.; Hou, M.-F.; Chu, P.-Y. New Insights into the Role of Inflammation in the Pathogenesis of Atherosclerosis. Int. J. Mol. Sci. 2017, 18, 2034. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berg, K.E.; Ljungcrantz, I.; Andersson, L.; Bryngelsson, C.; Hedblad, B.; Fredrikson, G.N.; Nilsson, J.; Björkbacka, H. Elevated CD14++CD16− monocytes predict cardiovascular events. Circ. Cardiovasc. Genet. 2012, 5, 122–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rogacev, K.S.; Seiler, S.; Zawada, A.M.; Reichart, B.; Herath, E.; Roth, D.; Ulrich, C.; Fliser, D.; Heine, G.H. CD14++CD16+ monocytes and cardiovascular outcome in patients with chronic kidney disease. Eur. Heart J. 2011, 32, 84–92. [Google Scholar] [CrossRef] [Green Version]
- Shah, A.D.; Denaxas, S.; Nicholas, O.; Hingorani, A.D.; Hemingway, H. Neutrophil Counts and Initial Presentation of 12 Cardiovascular Diseases: A CALIBER Cohort Study. J. Am. Coll. Cardiol. 2017, 69, 1160–1169. [Google Scholar] [CrossRef]
- Fernández-Ruiz, I. Neutrophil-driven SMC death destabilizes atherosclerotic plaques. Nat. Rev. Cardiol. 2019, 16, 455. [Google Scholar] [CrossRef]
- Sheridan, F.M.; Cole, P.G.; Ramage, D. Leukocyte adhesion to the coronary microvasculature during ischemia and reperfusion in an in vivo canine model. Circulation 1996, 93, 1784–1787. [Google Scholar] [CrossRef]
- Núñez, J.; Miñana, G.; Bodí, V.; Núñez, E.; Sanchis, J.; Husser, O.; Llàcer, A. Low lymphocyte count and cardiovascular diseases. Curr. Med. Chem. 2011, 18, 3226–3233. [Google Scholar] [CrossRef]
- Ommen, S.R.; Gibbons, R.J.; Hodge, D.O.; Thomson, S.P. Usefulness of the lymphocyte concentration as a prognostic marker in coronary artery disease. Am. J. Cardiol. 1997, 79, 812–814. [Google Scholar] [CrossRef]
- Levy, W.C.; Mozaffarian, D.; Linker, D.T.; Sutradhar, S.C.; Anker, S.D.; Cropp, A.B.; Anand, I.; Maggioni, A.; Burton, P.; Sullivan, M.D.; et al. The Seattle Heart Failure Model: Prediction of survival in heart failure. Circulation 2006, 113, 1424–1433. [Google Scholar] [CrossRef] [PubMed]
- Núñez, J.; Núñez, E.; Bodí, V.; Sanchis, J.; Mainar, L.; Miñana, G.; Fácila, L.; Bertomeu, V.; Merlos, P.; Darmofal, H.; et al. Low lymphocyte count in acute phase of ST-segment elevation myocardial infarction predicts long-term recurrent myocardial infarction. Coron. Artery Dis. 2010, 21, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Massberg, S.; Brand, K.; Grüner, S.; Page, S.; Müller, E.; Müller, I.; Bergmeier, W.; Richter, T.; Lorenz, M.; Konrad, I.; et al. A critical role of platelet adhesion in the initiation of atherosclerotic lesion formation. J. Exp. Med. 2002, 196, 887–896. [Google Scholar] [CrossRef]
- Gawaz, M.; Langer, H.; May, A.E. Platelets in inflammation and atherogenesis. J. Clin. Investig. 2005, 115, 3378–3384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pasalic, L.; Wang, S.S.Y.; Chen, V.M.Y. Platelets as Biomarkers of Coronary Artery Disease. Semin. Thromb. Hemost. 2016, 42, 223–233. [Google Scholar] [CrossRef]
- Yumuk, V.; Tsigos, C.; Fried, M.; Schindler, K.; Busetto, L.; Micic, D.; Toplak, H. European Guidelines for Obesity Management in Adults. Obes. Facts 2015, 8, 402–424. [Google Scholar] [CrossRef]
- Stergiou, G.S.; Palatini, P.; Parati, G.; O’Brien, E.; Januszewicz, A.; Lurbe, E.; Persu, A.; Mancia, G.; Kreutz, R. 2021 European Society of Hypertension practice guidelines for office and out-of-office blood pressure measurement. J. Hypertens. 2021, 39, 1293–1302. [Google Scholar] [CrossRef]
- Cosentino, F.; Grant, P.J.; Aboyans, V.; Bailey, C.J.; Ceriello, A.; Delgado, V.; Federici, M.; Filippatos, G.; Grobbee, D.E.; Hansen, T.B.; et al. 2019 ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD. Eur. Heart J. 2020, 41, 255–323. [Google Scholar] [CrossRef] [Green Version]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; de Backer, G.G.; Delgado, V.; Ference, B.A.; et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk. Eur. Heart J. 2020, 41, 111–188. [Google Scholar] [CrossRef]
- Lawton, J.S.; Tamis-Holland, J.E.; Bangalore, S.; Bates, E.R.; Beckie, T.M.; Bischoff, J.M.; Bittl, J.A.; Cohen, M.G.; DiMaio, J.M.; Don, C.W.; et al. 2021 ACC/AHA/SCAI Guideline for Coronary Artery Revascularization: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2022, 79, e21–e129. [Google Scholar] [CrossRef]
- Collet, J.-P.; Thiele, H.; Barbato, E.; Barthélémy, O.; Bauersachs, J.; Bhatt, D.L.; Dendale, P.; Dorobantu, M.; Edvardsen, T.; Folliguet, T.; et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur. Heart J. 2021, 42, 1289–1367. [Google Scholar] [CrossRef] [PubMed]

| Variable | Values |
|---|---|
| N of participants [♂/♀] | 699 (444/256) |
| Age [years] | 66.3 (29.5–93.3) |
| BMI [kg/m2] | 27.8 (16.1–47.4) |
| Cause of hospitalization [stable CAD/STEMI/NSTEMI/UA] | 366/147/108/78 |
| Previous MI [yes/no] | 269/430 |
| Total cholesterol (TC) [mg/dL] | 172.0 (70.3–338.3) |
| High-density lipoprotein (HDL) [mg/dL] | 47.2 (10.4–113.2) |
| Low-density lipoprotein (LDL) [mg/dL] | 95.7 (20.5–257.9) |
| Triglycerides (TG) [mg/dL] | 113.9 (31.3–456.7) |
| Hyperlipidemia [yes/no] (N = 644) | 377/267 |
| Hypertension [yes/no] | 577/122 |
| Smoking [active/former smoker/no] | 195/75/429 |
| Type 2 diabetes mellitus (t2DM) [yes/pre-diabetes/no] | 236/30/433 |
| Coronary Artery Surgery Study Class (CASSC) [0/1/2/3] | 177/193/174/155 |
| Leukocytes [thousand cells/µL] (N = 694) | 8.0 (3.0–26.0) |
| Platelet (PLT) [mcL] | 220 (150–445) |
| Neutrophils [thousand cells/µL] | 4.8 (1.4–44.7) |
| Monocytes [thousand cells/µL] | 0.7 (0.2–3.0) |
| Lymphocytes [thousand cells/µL] | 1.9 (0.4–41.8) |
| SII | 519 (26–4634) |
| SIRI | 1.7 (0.06–27.9) |
| Variable | CASSC 0 | CASSC 1 | CASSC 2 | CASSC 3 | p-Value |
|---|---|---|---|---|---|
| N of participants [♂/♀] | 85/92 | 124/69 | 127/47 | 107/48 | <0.001 |
| Age [years] | 65.9 (36.7–91.5) | 65.2 (34.7–93.3) | 64.0 (33.6–89.2) | 68.9 (29.4–90.8) | 0.019 |
| BMI [kg/m2] | 27.8 (17.3–47.4) | 27.1 (16.9–43.4) | 27.8 (17.4–44.6) | 28.3 (16.1–45.9) | 0.635 |
| Cause of hospitalization [stable CAD/STEMI/NSTEMI/UA] | 147/8/12/10 | 71/63/36/23 | 81/39/30/24 | 67/37/30/21 | <0.001 |
| Previous MI [yes/no] | 13/164 | 79/114 | 84/90 | 93/62 | <0.001 |
| TC [mg/dL] | 179.4 (73.3–328.7) | 178.9 (70.3–331.7) | 165.5 (70.9–338.3) | 161.7 (84.4–310.8) | 0.007 |
| HDL [mg/dL] | 53.8 (10.4–107.6) | 46.6 (20.5–97.4) | 46.7 (21.3–113.2) | 44.3 (14.6–89.2) | <0.001 |
| LDL [mg/dL] | 95.8 (20.5–230.7) | 105.0 (23.5–251.7) | 94.0 (24.4–258.0) | 87.0 (22.3–228.3) | 0.048 |
| TG [mg/dL] | 115.3 (31.3–340.0) | 110.6 (43.6–438.3) | 113.4 (38.0–456.7) | 115.4 (47.6–391.8) | 0.847 |
| Hyperlipidemia [yes/no] | 89/71 | 115/61 | 92/70 | 81/65 | 0.197 |
| Hypertension [yes/no] | 132/45 | 157/36 | 152/22 | 136/19 | 0.003 |
| Smoking [active/former smoker/no] | 30/13/134 | 69/17/107 | 57/24/93 | 39/21/95 | <0.001 |
| t2DM [yes/pre-diabetes/no] | 51/7/119 | 57/6/130 | 62/10/102 | 66/7/82 | 0.070 |
| Leukocytes [thousand cells/µL] | 7.7 (3.9–21.2) | 8.1 (3.9–26.0) | 8.0 (3.6–19.6) | 8.2 (3.0–18.1) | 0.085 |
| PLT [mcL] | 212 (150–445) | 224 (150–438) | 223 (151–439) | 218 (150–429) | 0.570 |
| Neutrophils [thousand cells/µL] | 4.6 (1.7–44.7) | 4.8 (1.7–23.8) | 4.8 (1.4–16.8) | 5.0 (1.4–14.2) | 0.260 |
| Monocytes [thousand cells/µL] | 0.7 (0.3–1.7) | 0.7 (0.3–2.4) | 0.7 (0.3–2.0) | 0.8 (0.2–3.0) | 0.046 |
| Lymphocytes [thousand cells/µL] | 1.9 (0.4–4.6) | 2.1 (0.4–4.9) | 2.0 (0.7–41.8) | 1.9 (0.6–5.0) | 0.162 |
| SII | 505 (103–4932) | 510 (142–4467) | 507 (26–2574) | 566 (136–4191) | 0.277 |
| SIRI | 1.6 (0.3–16.7) | 1.7 (0.3–27.9) | 1.7 (0.1–19.4) | 1.9 (0.5–15.2) | 0.066 |
| Variable | Stable CAD | STEMI | NSTEMI | UA | p-Value |
|---|---|---|---|---|---|
| Number of participants [♂/♀] | 225/141 | 110/37 | 67/41 | 41/37 | 0.005 |
| Age [years] | 67.1 (29.4–93.3) | 63.0 (36.3–89.1) | 65.1 (36.1–92.1) | 69.6 (33.6–91.5) | <0.001 |
| BMI [kg/m2] | 28.0 (16.1–47.4) | 26.9 (16.9–44.6) | 27.7 (17.8–42.4) | 28.6 (17.3–43.4) | 0.549 |
| Previous MI [yes/no] | 109/257 | 71/76 | 63/45 | 26/52 | <0.001 |
| TC [mg/dL] | 170.4 (84.8–328.7) | 180.6 (97.4–320.3) | 168.9 (70.3–338.3) | 159.2 (81.4–331.7) | 0.026 |
| HDL [mg/dL] | 50.2 (10.4–113.2) | 45.0 (20.5–92.9) | 42.6 (22.8–78.9) | 46.5 (19.5–72.9) | <0.001 |
| LDL [mg/dL] | 89.0 (20.5–258.0) | 108.2 (28.3–214.1) | 100.1 (23.5–244.3) | 86.8 (32.9–251.7) | <0.001 |
| TG [mg/dL] | 115.8 (35.7–438.3) | 107.6 (47.6–367.8) | 111.2 (43.6–456.7) | 114.7 (31.3–251.7) | 0.971 |
| Hyperlipidemia [yes/no] | 187/151 | 99/44 | 59/38 | 32/34 | 0.011 |
| Hypertension [yes/no] | 289/77 | 119/28 | 93/15 | 76/2 | <0.001 |
| Smoking [active/former smoker/no] | 70/56/240 | 67/8/72 | 45/3/60 | 13/8/57 | <0.001 |
| t2DM [yes/pre-diabetes/no] | 127/16/223 | 44/3/100 | 33/8/67 | 32/3/43 | 0.236 |
| Leukocytes [thousand cells/µL] | 7.8 (3.6–18.6) | 8.7 (3.0–26.0) | 7.9 (3.9–21.2) | 7.8 (4.7–19.6) | 0.0006 |
| PLT [mcL] | 214 (150–445) | 230 (150–428) | 224 (151–410) | 219 (150–432) | 0.099 |
| Neutrophils [thousand cells/µL] | 4.7 (1.4–44.7) | 5.3 (1.4–23.8) | 4.8 (1.7–19.3) | 4.8 (2.3–15.1) | 0.0008 |
| Monocytes [thousand cells/µL] | 0.7 (0.2–1.8) | 0.8 (0.3–2.0) | 0.7 (0.3–3.0) | 0.7 (0.3–2.4) | 0.030 |
| Lymphocytes [thousand cells/µL] | 1.9 (0.6–41.8) | 2.0 (0.6–5.3) | 1.9 (0.4–3.8) | 2.0 (0.4–4.6) | 0.756 |
| SII | 494 (26–4634) | 579 (142–4467) | 576 (103–4491) | 563 (192–2482) | 0.0052 |
| SIRI | 1.6 (0.1–26.2) | 2.0 (0.6–27.9) | 1.7 (0.3–15.8) | 1.7 (0.5–16.9) | 0.0053 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dziedzic, E.A.; Gąsior, J.S.; Tuzimek, A.; Paleczny, J.; Junka, A.; Dąbrowski, M.; Jankowski, P. Investigation of the Associations of Novel Inflammatory Biomarkers—Systemic Inflammatory Index (SII) and Systemic Inflammatory Response Index (SIRI)—With the Severity of Coronary Artery Disease and Acute Coronary Syndrome Occurrence. Int. J. Mol. Sci. 2022, 23, 9553. https://doi.org/10.3390/ijms23179553
Dziedzic EA, Gąsior JS, Tuzimek A, Paleczny J, Junka A, Dąbrowski M, Jankowski P. Investigation of the Associations of Novel Inflammatory Biomarkers—Systemic Inflammatory Index (SII) and Systemic Inflammatory Response Index (SIRI)—With the Severity of Coronary Artery Disease and Acute Coronary Syndrome Occurrence. International Journal of Molecular Sciences. 2022; 23(17):9553. https://doi.org/10.3390/ijms23179553
Chicago/Turabian StyleDziedzic, Ewelina A., Jakub S. Gąsior, Agnieszka Tuzimek, Justyna Paleczny, Adam Junka, Marek Dąbrowski, and Piotr Jankowski. 2022. "Investigation of the Associations of Novel Inflammatory Biomarkers—Systemic Inflammatory Index (SII) and Systemic Inflammatory Response Index (SIRI)—With the Severity of Coronary Artery Disease and Acute Coronary Syndrome Occurrence" International Journal of Molecular Sciences 23, no. 17: 9553. https://doi.org/10.3390/ijms23179553
APA StyleDziedzic, E. A., Gąsior, J. S., Tuzimek, A., Paleczny, J., Junka, A., Dąbrowski, M., & Jankowski, P. (2022). Investigation of the Associations of Novel Inflammatory Biomarkers—Systemic Inflammatory Index (SII) and Systemic Inflammatory Response Index (SIRI)—With the Severity of Coronary Artery Disease and Acute Coronary Syndrome Occurrence. International Journal of Molecular Sciences, 23(17), 9553. https://doi.org/10.3390/ijms23179553








