New, Fully Implantable Device for Selective Clearance of CSF-Target Molecules: Proof of Concept in a Murine Model of Alzheimer’s Disease
Abstract
:1. Introduction
2. Results
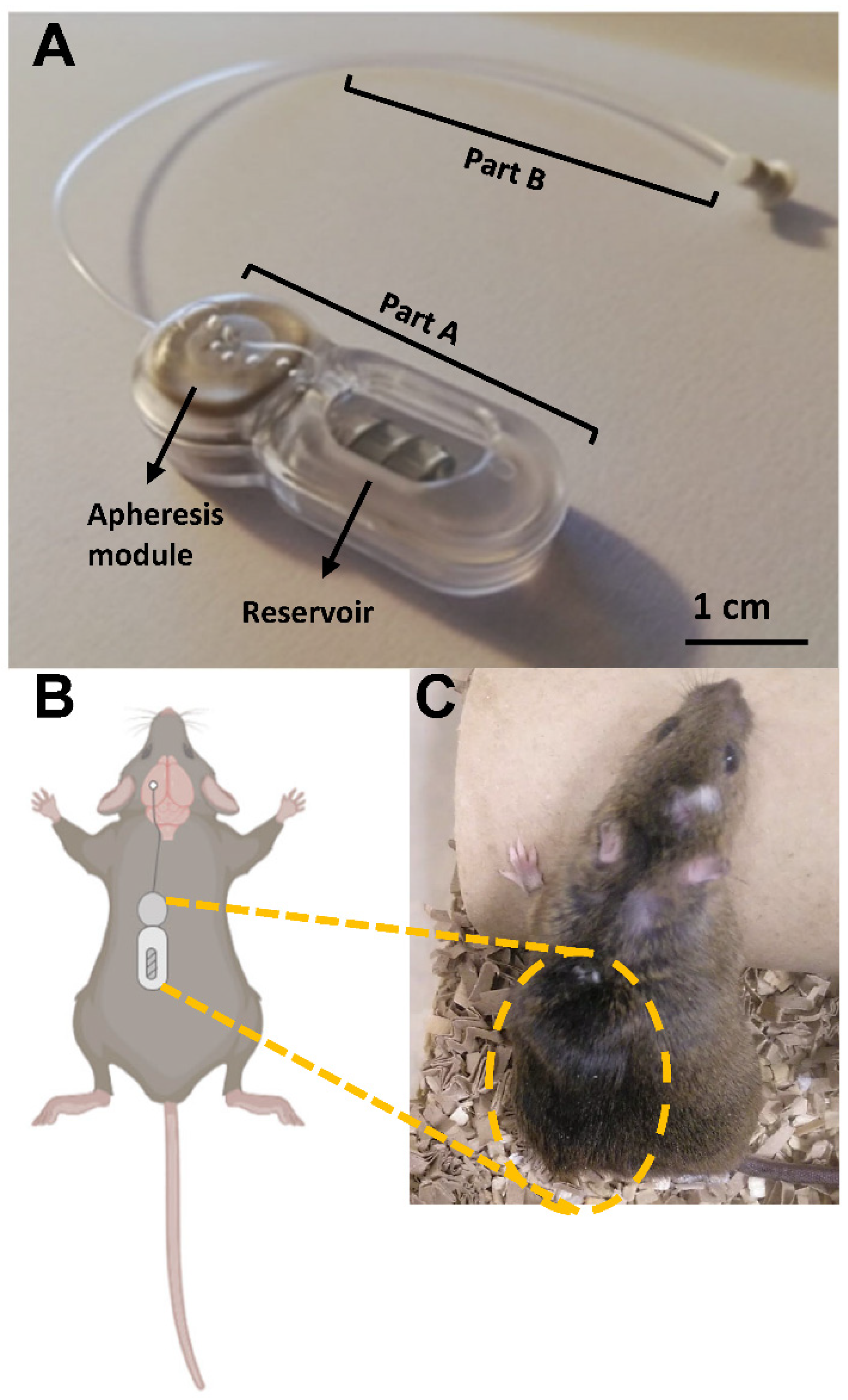
2.1. Implantation of the Miniaturized Prototype Was Feasible in a Murine Model of Alzheimer’s Disease
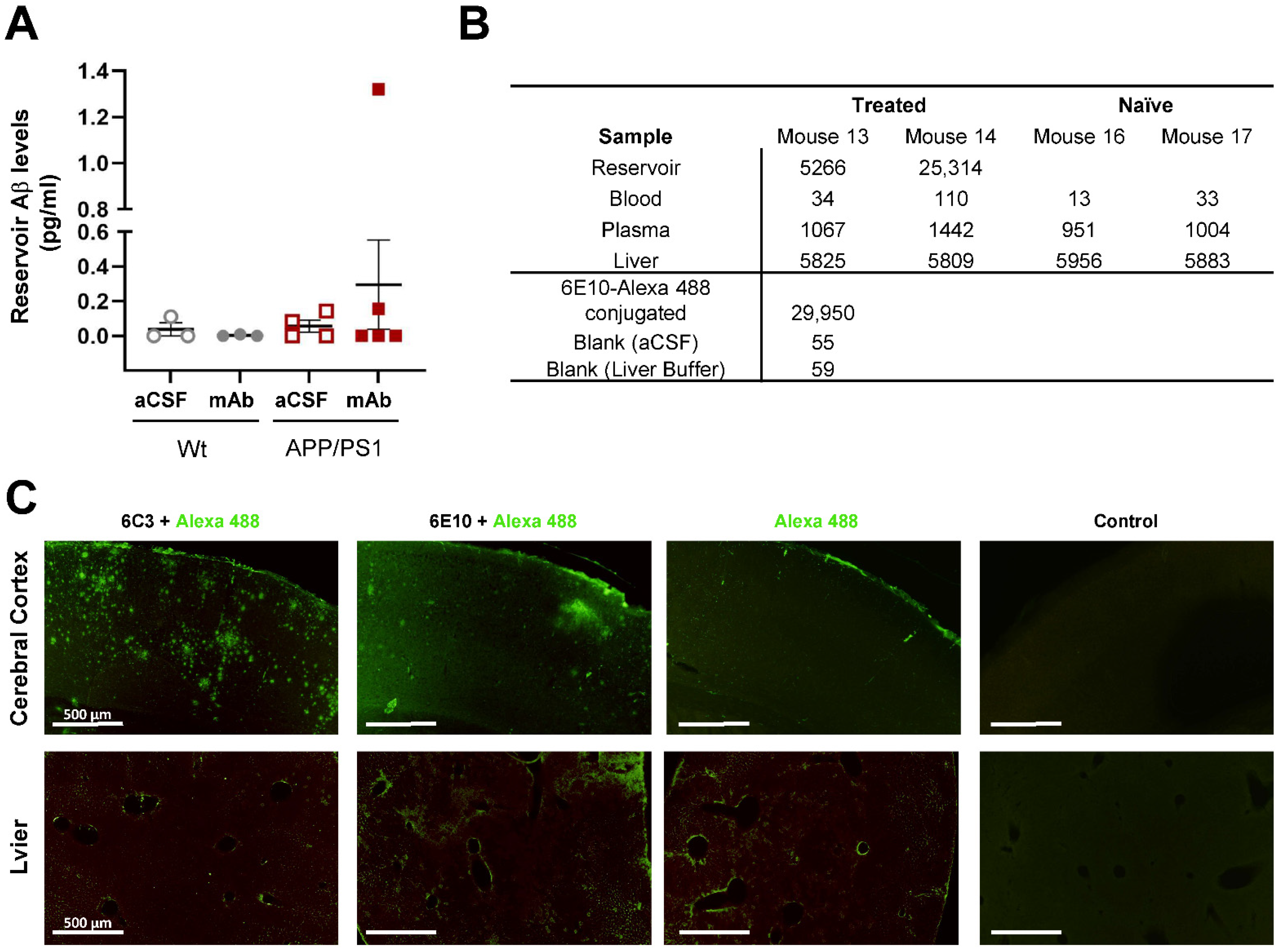
2.2. The Apheresis Module Is Permeable to Aβ and Impermeable to Anti-Aβ Antibody In Vivo
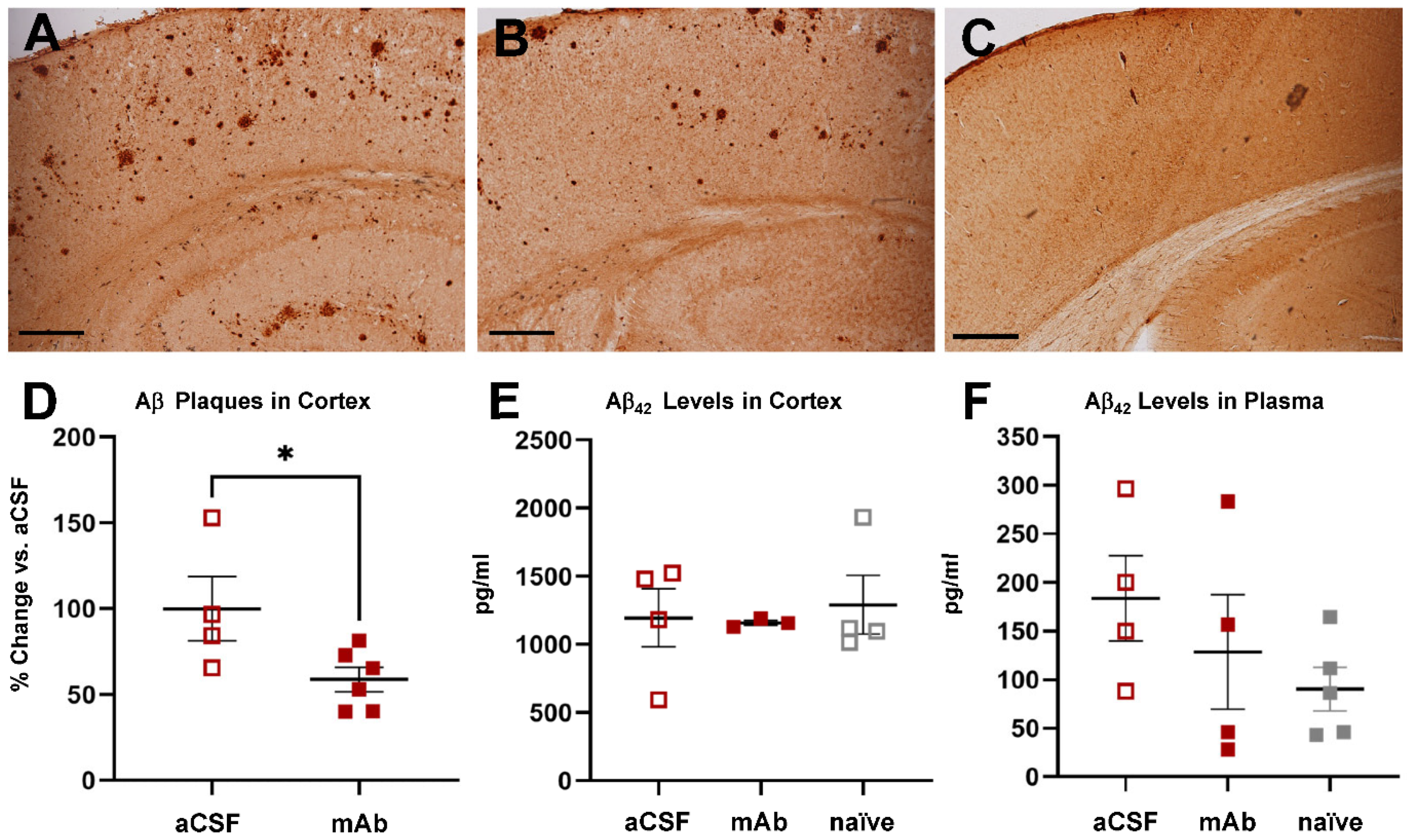
2.3. Continuous CSF Apheresis for Rapidly Decreased Area Covered by Aβ Plaques in the Cerebral Cortex
3. Discussion
4. Materials and Methods
4.1. Mice
4.2. Implantable Device for Mice
4.3. Filling the Reservoir with Anti-Aβ mAb or Vehicle
4.4. Surgery and Mice Welfare
4.5. Tissue and Fluids Collection and Processing
4.5.1. Tissue Processing for Biochemical Analysis
4.5.2. Tissue Processing for Histological Analysis
4.6. Quantification of Cerebral Cortex Aβ Plaques
4.7. Fluorescence Analysis
4.7.1. Immunofluorescence
4.7.2. Fluorescence Levels
4.8. Aβ1–42 Immunoassay
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yamamoto, A.; Lucas, J.J.; Hen, R. Reversal of neuropathology and motor dysfunction in a conditional model of Huntington’s disease. Cell 2000, 101, 57–66. [Google Scholar] [CrossRef]
- Martinez-Vicente, M.; Cuervo, A.M. Autophagy and neurodegeneration: When the cleaning crew goes on strike. Lancet Neurol. 2007, 6, 352–361. [Google Scholar] [CrossRef]
- Nixon, R.A. The role of autophagy in neurodegenerative disease. Nat. Med. 2013, 19, 983–997. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Happonen, K.E.; Burrola, P.G.; O’Connor, C.; Hah, N.; Huang, L.; Nimmerjahn, A.; Lemke, G. Microglia use TAM receptors to detect and engulf amyloid beta plaques. Nat. Immunol. 2021, 22, 586–594. [Google Scholar] [CrossRef]
- Sikanyika, N.L.; Parkington, H.C.; Smith, A.I.; Kuruppu, S. Powering Amyloid Beta Degrading Enzymes: A Possible Therapy for Alzheimer’s Disease. Neurochem. Res. 2019, 44, 1289–1296. [Google Scholar] [CrossRef] [PubMed]
- Elbert, D.L.; Patterson, B.W.; Lucey, B.P.; Benzinger, T.L.S.; Bateman, R.J. Importance of CSF-based Abeta clearance with age in humans increases with declining efficacy of blood-brain barrier/proteolytic pathways. Commun. Biol. 2022, 5, 98. [Google Scholar] [CrossRef] [PubMed]
- Mestre, H.; Verma, N.; Greene, T.D.; Lin, L.A.; Ladron-de-Guevara, A.; Sweeney, A.M.; Liu, G.; Thomas, V.K.; Galloway, C.A.; de Mesy Bentley, K.L.; et al. Periarteriolar spaces modulate cerebrospinal fluid transport into brain and demonstrate altered morphology in aging and Alzheimer’s disease. Nat. Commun. 2022, 13, 3897. [Google Scholar] [CrossRef]
- Peng, W.; Achariyar, T.M.; Li, B.; Liao, Y.; Mestre, H.; Hitomi, E.; Regan, S.; Kasper, T.; Peng, S.; Ding, F.; et al. Suppression of glymphatic fluid transport in a mouse model of Alzheimer’s disease. Neurobiol. Dis. 2016, 93, 215–225. [Google Scholar] [CrossRef]
- Da Mesquita, S.; Papadopoulos, Z.; Dykstra, T.; Brase, L.; Farias, F.G.; Wall, M.; Jiang, H.; Kodira, C.D.; de Lima, K.A.; Herz, J.; et al. Meningeal lymphatics affect microglia responses and anti-Abeta immunotherapy. Nature 2021, 593, 255–260. [Google Scholar] [CrossRef]
- Li, Y.; Rusinek, H.; Butler, T.; Glodzik, L.; Pirraglia, E.; Babich, J.; Mozley, P.D.; Nehmeh, S.; Pahlajani, S.; Wang, X.; et al. Decreased CSF clearance and increased brain amyloid in Alzheimer’s disease. Fluids Barriers CNS 2022, 19, 21. [Google Scholar] [CrossRef]
- Hladky, S.B.; Barrand, M.A. Elimination of substances from the brain parenchyma: Efflux via perivascular pathways and via the blood-brain barrier. Fluids Barriers CNS 2018, 15, 30. [Google Scholar] [CrossRef] [PubMed]
- Ryan, D.A.; Mastrangelo, M.A.; Narrow, W.C.; Sullivan, M.A.; Federoff, H.J.; Bowers, W.J. Aβ-directed Single-chain Antibody Delivery Via a Serotype-1 AAV Vector Improves Learning Behavior and Pathology in Alzheimer’s Disease Mice. Mol. Ther. 2010, 18, 1471–1481. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Chu, F.; Zhu, F.; Zhu, J. Impact of Anti-amyloid-beta Monoclonal Antibodies on the Pathology and Clinical Profile of Alzheimer’s Disease: A Focus on Aducanumab and Lecanemab. Front. Aging Neurosci. 2022, 14, 870517. [Google Scholar] [CrossRef]
- Lacorte, E.; Ancidoni, A.; Zaccaria, V.; Remoli, G.; Tariciotti, L.; Bellomo, G.; Sciancalepore, F.; Corbo, M.; Lombardo, F.L.; Bacigalupo, I.; et al. Safety and Efficacy of Monoclonal Antibodies for Alzheimer’s Disease: A Systematic Review and Meta-Analysis of Published and Unpublished Clinical Trials. J. Alzheimer’s Dis. 2022, 87, 101–129. [Google Scholar] [CrossRef]
- Tholen, S.; Schmaderer, C.; Chmielewski, S.; Forstl, H.; Heemann, U.; Baumann, M.; Steubl, D.; Grimmer, T. Reduction of Amyloid-beta Plasma Levels by Hemodialysis: An Anti-Amyloid Treatment Strategy? J. Alzheimer’s Dis. 2016, 50, 791–796. [Google Scholar] [CrossRef] [PubMed]
- Sakai, K.; Senda, T.; Hata, R.; Kuroda, M.; Hasegawa, M.; Kato, M.; Abe, M.; Kawaguchi, K.; Nakai, S.; Hiki, Y.; et al. Patients that have Undergone Hemodialysis Exhibit Lower Amyloid Deposition in the Brain: Evidence Supporting a Therapeutic Strategy for Alzheimer’s Disease by Removal of Blood Amyloid. J. Alzheimer’s Dis. 2016, 51, 997–1002. [Google Scholar] [CrossRef]
- Liu, Y.H.; Xiang, Y.; Wang, Y.R.; Jiao, S.S.; Wang, Q.H.; Bu, X.L.; Zhu, C.; Yao, X.Q.; Giunta, B.; Tan, J.; et al. Association Between Serum Amyloid-Beta and Renal Functions: Implications for Roles of Kidney in Amyloid-Beta Clearance. Mol. Neurobiol. 2015, 52, 115–119. [Google Scholar] [CrossRef]
- Kitaguchi, N.; Hasegawa, M.; Ito, S.; Kawaguchi, K.; Hiki, Y.; Nakai, S.; Suzuki, N.; Shimano, Y.; Ishida, O.; Kushimoto, H.; et al. A prospective study on blood Abeta levels and the cognitive function of patients with hemodialysis: A potential therapeutic strategy for Alzheimer’s disease. J. Neural. Transm. 2015, 122, 1593–1607. [Google Scholar] [CrossRef]
- Xiang, Y.; Bu, X.L.; Liu, Y.H.; Zhu, C.; Shen, L.L.; Jiao, S.S.; Zhu, X.Y.; Giunta, B.; Tan, J.; Song, W.H.; et al. Physiological amyloid-beta clearance in the periphery and its therapeutic potential for Alzheimer’s disease. Acta Neuropathol. 2015, 130, 487–499. [Google Scholar] [CrossRef]
- Jin, W.S.; Shen, L.L.; Bu, X.L.; Zhang, W.W.; Chen, S.H.; Huang, Z.L.; Xiong, J.X.; Gao, C.Y.; Dong, Z.; He, Y.N.; et al. Peritoneal dialysis reduces amyloid-beta plasma levels in humans and attenuates Alzheimer-associated phenotypes in an APP/PS1 mouse model. Acta Neuropathol. 2017, 134, 207–220. [Google Scholar] [CrossRef]
- Boada, M.; Lopez, O.; Nunez, L.; Szczepiorkowski, Z.M.; Torres, M.; Grifols, C.; Paez, A. Plasma exchange for Alzheimer’s disease Management by Albumin Replacement (AMBAR) trial: Study design and progress. Alzheimer’s Dement. Transl. Res. Clin. Interv. 2019, 5, 61–69. [Google Scholar] [CrossRef] [PubMed]
- DeMattos, R.B.; Bales, K.R.; Parsadanian, M.; O’Dell, M.A.; Foss, E.M.; Paul, S.M.; Holtzman, D.M. Plaque-associated disruption of CSF and plasma amyloid-beta (Abeta) equilibrium in a mouse model of Alzheimer’s disease. J. Neurochem. 2002, 81, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Menendez Gonzalez, M. Implantable Systems for Continuous Liquorpheresis and CSF Replacement. Cureus 2017, 9, e1022. [Google Scholar] [CrossRef] [PubMed]
- Menendez Gonzalez, M. Continuous Clearance of Beta-Amyloid from the CSF: A Hypothesis for a New Therapeutic Strategy. Alzheimer’s Dement. 2017, 13, P1578. [Google Scholar] [CrossRef]
- Menendez-Gonzalez, M.; Padilla-Zambrano, H.S.; Alvarez, G.; Capetillo-Zarate, E.; Tomas-Zapico, C.; Costa, A. Targeting Beta-Amyloid at the CSF: A New Therapeutic Strategy in Alzheimer’s Disease. Front. Aging Neurosci. 2018, 10, 100. [Google Scholar] [CrossRef]
- Schreiner, T.G.; Menéndez-González, M.; Popescu, B.O. The “Cerebrospinal Fluid Sink Therapeutic Strategy” in Alzheimer´s Disease: From Theory to Design of Applied Systems. Biomedicines 2022, 10, 1509. [Google Scholar] [CrossRef]
- Jankowsky, J.L.; Fadale, D.J.; Anderson, J.; Xu, G.M.; Gonzales, V.; Jenkins, N.A.; Copeland, N.G.; Lee, M.K.; Younkin, L.H.; Wagner, S.L.; et al. Mutant presenilins specifically elevate the levels of the 42 residue beta-amyloid peptide in vivo: Evidence for augmentation of a 42-specific gamma secretase. Hum. Mol. Genet. 2004, 13, 159–170. [Google Scholar] [CrossRef]
- Billings, L.M.; Oddo, S.; Green, K.N.; McGaugh, J.L.; LaFerla, F.M. Intraneuronal Abeta causes the onset of early Alzheimer’s disease-related cognitive deficits in transgenic mice. Neuron 2005, 45, 675–688. [Google Scholar] [CrossRef]
- Adiga, S.P.; Jin, C.; Curtiss, L.A.; Monteiro-Riviere, N.A.; Narayan, R.J. Nanoporous membranes for medical and biological applications. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2009, 1, 568–581. [Google Scholar] [CrossRef]
- Oddo, S.; Billings, L.; Kesslak, J.P.; Cribbs, D.H.; LaFerla, F.M. Abeta immunotherapy leads to clearance of early, but not late, hyperphosphorylated tau aggregates via the proteasome. Neuron 2004, 43, 321–332. [Google Scholar] [CrossRef]
- Shea, D.; Daggett, V. Amyloid-β Oligomers: Multiple Moving Targets. Biophysica 2022, 2, 91–110. [Google Scholar] [CrossRef]
- Chen, J.F.; Liu, K.; Hu, B.; Li, R.R.; Xin, W.; Chen, H.; Wang, F.; Chen, L.; Li, R.X.; Ren, S.Y.; et al. Enhancing myelin renewal reverses cognitive dysfunction in a murine model of Alzheimer’s disease. Neuron 2021, 109, 2292–2307.e5. [Google Scholar] [CrossRef]
- Cuevas, A.L.; González, A.S.; Vega, V.; Prida, V.M.; Benavente, J. Optical and Electrochemical Characterization of Nanoporous Alumina Structures: Pore Size, Porosity, and Structure Effect. Appl. Sci. 2020, 10, 4864. [Google Scholar] [CrossRef]
- González, A.S.; Vega, V.; Cuevas, A.L.; Yuso, M.d.V.M.d.; Prida, V.M.; Benavente, J. Surface Modification of Nanoporous Anodic Alumina during Self-Catalytic Atomic Layer Deposition of Silicon Dioxide from (3-Aminopropyl)Triethoxysilane. Materials 2021, 14, 5052. [Google Scholar] [CrossRef] [PubMed]
- DeVos, S.L.; Miller, T.M. Direct intraventricular delivery of drugs to the rodent central nervous system. J. Vis. Exp. 2013, 75, e50326. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Krishtal, J.; Metsla, K.; Bragina, O.; Tougu, V.; Palumaa, P. Toxicity of Amyloid-beta Peptides Varies Depending on Differentiation Route of SH-SY5Y Cells. J. Alzheimers Dis. 2019, 71, 879–887. [Google Scholar] [CrossRef]
| Mice ID | Genotype | Content Infused | Time Implanted | Complications | Aβ Permeability | mAb Impermeability | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Aβ Reservoir | Aβ Plasma | Aβ Cortex | IHC | IF | Fluorescence | |||||
| 7 | APP/PS1 | mAb | 48 h | Lethargy | + | + | ||||
| 20 | Wt | mAb | 48 h | Lethargy | ||||||
| 21 | Wt | aCSF | 72 h | Lethargy | + | |||||
| 5 | APP/PS1 | mAb | 1 week | Lethargy | + | |||||
| 11 | APP/PS1 | mAb | 2 weeks | Weight loss | + | + | + | |||
| 26 | APP/PS1 | mAb | 2 weeks | Lethargy | + | |||||
| 27 | APP/PS1 | aCSF | 2 weeks | Lethargy | + | |||||
| 28 | APP/PS1 | aCSF | 2 weeks | Lethargy | + | |||||
| 2 | APP/PS1 | aCSF | 3 weeks | None | + | + | + | |||
| 8 | APP/PS1 | aCSF | 3 weeks | None | + | + | + | + | ||
| 9 | APP/PS1 | aCSF | 3 weeks | None | + | + | + | |||
| 12 | APP/PS1 | aCSF | 3 weeks | None | + | + | + | + | ||
| 4 | APP/PS1 | aCSF | 3 weeks | None | + | + | ||||
| 17 | Wt | aCSF | 3 weeks | None | + | + | ||||
| 19 | Wt | aCSF | 3 weeks | None | + | + | ||||
| 3 | APP/PS1 | mAb | 3 weeks | None | + | + | + | |||
| 6 | APP/PS1 | mAb | 3 weeks | None | + | + | + | |||
| 10 | APP/PS1 | mAb | 3 weeks | None | + | + | + | + | ||
| 1 | APP/PS1 | mAb | 3 weeks | None | + | + | ||||
| 18 | Wt | mAb | 3 weeks | None | + | |||||
| 13 | APP/PS1 | mAb 488 | 3 weeks | None | + | + | ||||
| 14 | APP/PS1 | mAb 488 | 3 weeks | None | + | + | ||||
| 15 | Wt | mAb 488 | 3 weeks | None | + | + | ||||
| 16 | Wt | mAb 488 | 3 weeks | None | + | + | + | |||
| 29 | APP/PS1 | naïve | - | None | + | |||||
| 30 | APP/PS1 | naïve | - | None | + | + | ||||
| 31 | APP/PS1 | naïve | - | None | + | + | ||||
| 32 | APP/PS1 | naïve | - | None | + | + | ||||
| 33 | APP/PS1 | naïve | - | None | + | + | ||||
| 34 | APP/PS1 | naïve | - | None | + | |||||
| 35 | APP/PS1 | naïve | - | None | + | + | ||||
| 36 | Wt | naïve | - | None | + | |||||
| 37 | APP/PS1 | naïve | - | None | + | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coto-Vilcapoma, M.A.; Castilla-Silgado, J.; Fernández-García, B.; Pinto-Hernández, P.; Cipriani, R.; Capetillo-Zarate, E.; Menéndez-González, M.; Álvarez-Vega, M.; Tomás-Zapico, C. New, Fully Implantable Device for Selective Clearance of CSF-Target Molecules: Proof of Concept in a Murine Model of Alzheimer’s Disease. Int. J. Mol. Sci. 2022, 23, 9256. https://doi.org/10.3390/ijms23169256
Coto-Vilcapoma MA, Castilla-Silgado J, Fernández-García B, Pinto-Hernández P, Cipriani R, Capetillo-Zarate E, Menéndez-González M, Álvarez-Vega M, Tomás-Zapico C. New, Fully Implantable Device for Selective Clearance of CSF-Target Molecules: Proof of Concept in a Murine Model of Alzheimer’s Disease. International Journal of Molecular Sciences. 2022; 23(16):9256. https://doi.org/10.3390/ijms23169256
Chicago/Turabian StyleCoto-Vilcapoma, María Almudena, Juan Castilla-Silgado, Benjamín Fernández-García, Paola Pinto-Hernández, Raffaela Cipriani, Estibaliz Capetillo-Zarate, Manuel Menéndez-González, Marco Álvarez-Vega, and Cristina Tomás-Zapico. 2022. "New, Fully Implantable Device for Selective Clearance of CSF-Target Molecules: Proof of Concept in a Murine Model of Alzheimer’s Disease" International Journal of Molecular Sciences 23, no. 16: 9256. https://doi.org/10.3390/ijms23169256
APA StyleCoto-Vilcapoma, M. A., Castilla-Silgado, J., Fernández-García, B., Pinto-Hernández, P., Cipriani, R., Capetillo-Zarate, E., Menéndez-González, M., Álvarez-Vega, M., & Tomás-Zapico, C. (2022). New, Fully Implantable Device for Selective Clearance of CSF-Target Molecules: Proof of Concept in a Murine Model of Alzheimer’s Disease. International Journal of Molecular Sciences, 23(16), 9256. https://doi.org/10.3390/ijms23169256







