Pan-Cancer Analysis Identifies CHD5 as a Potential Biomarker for Glioma
Abstract
1. Introduction
2. Results
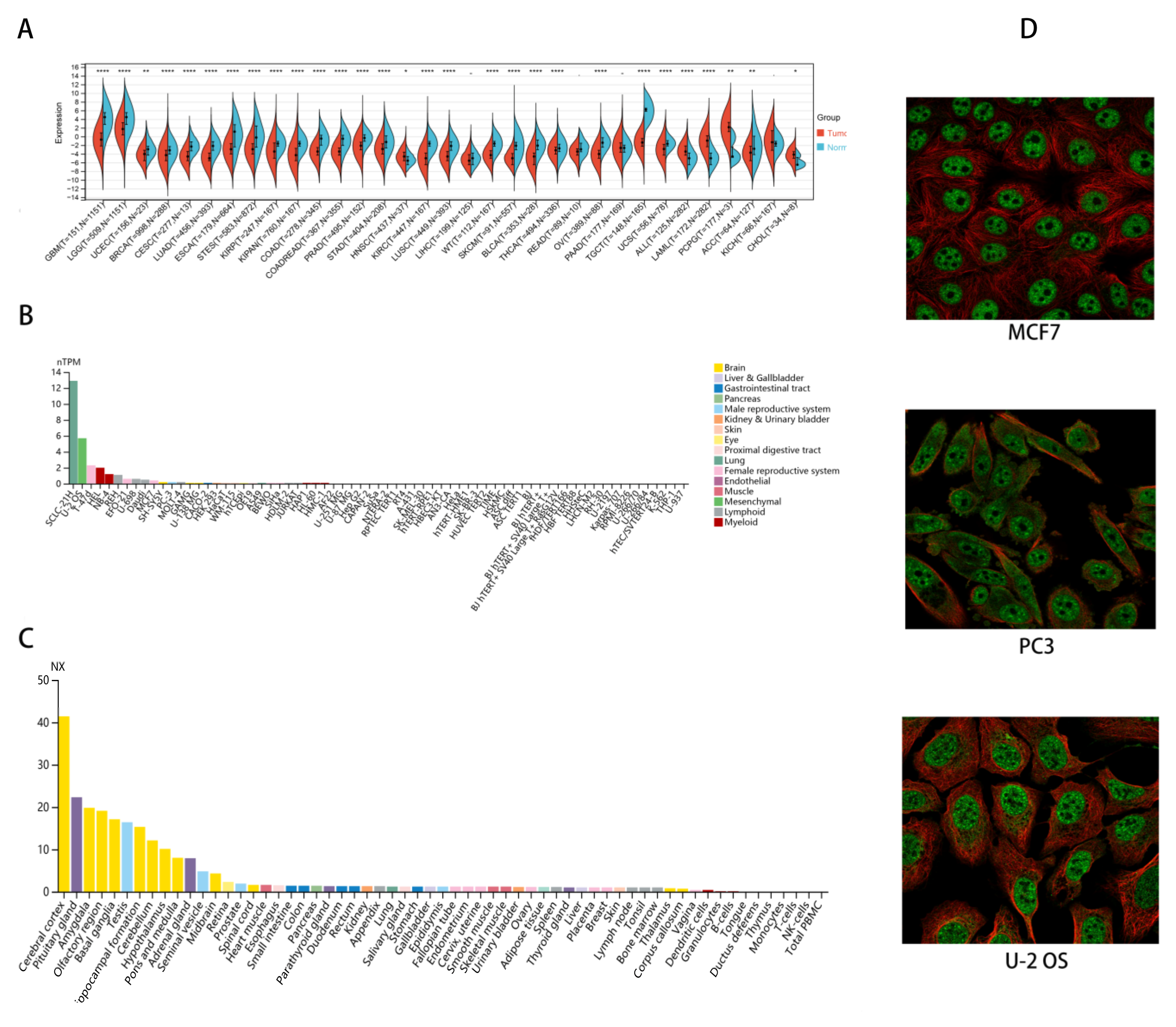
2.1. CHD5 Is Differentially Expressed between Tumor and Normal Tissues
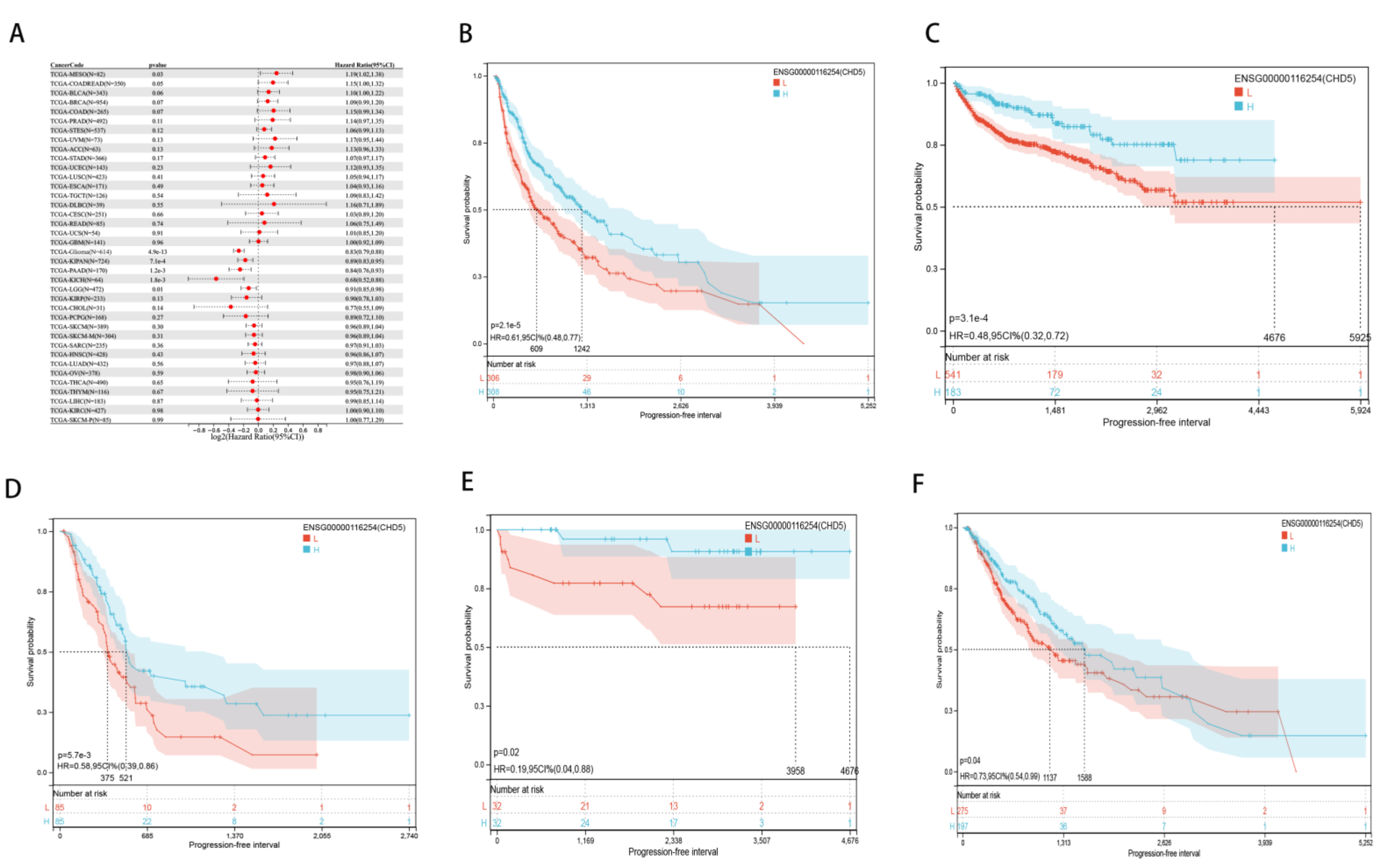
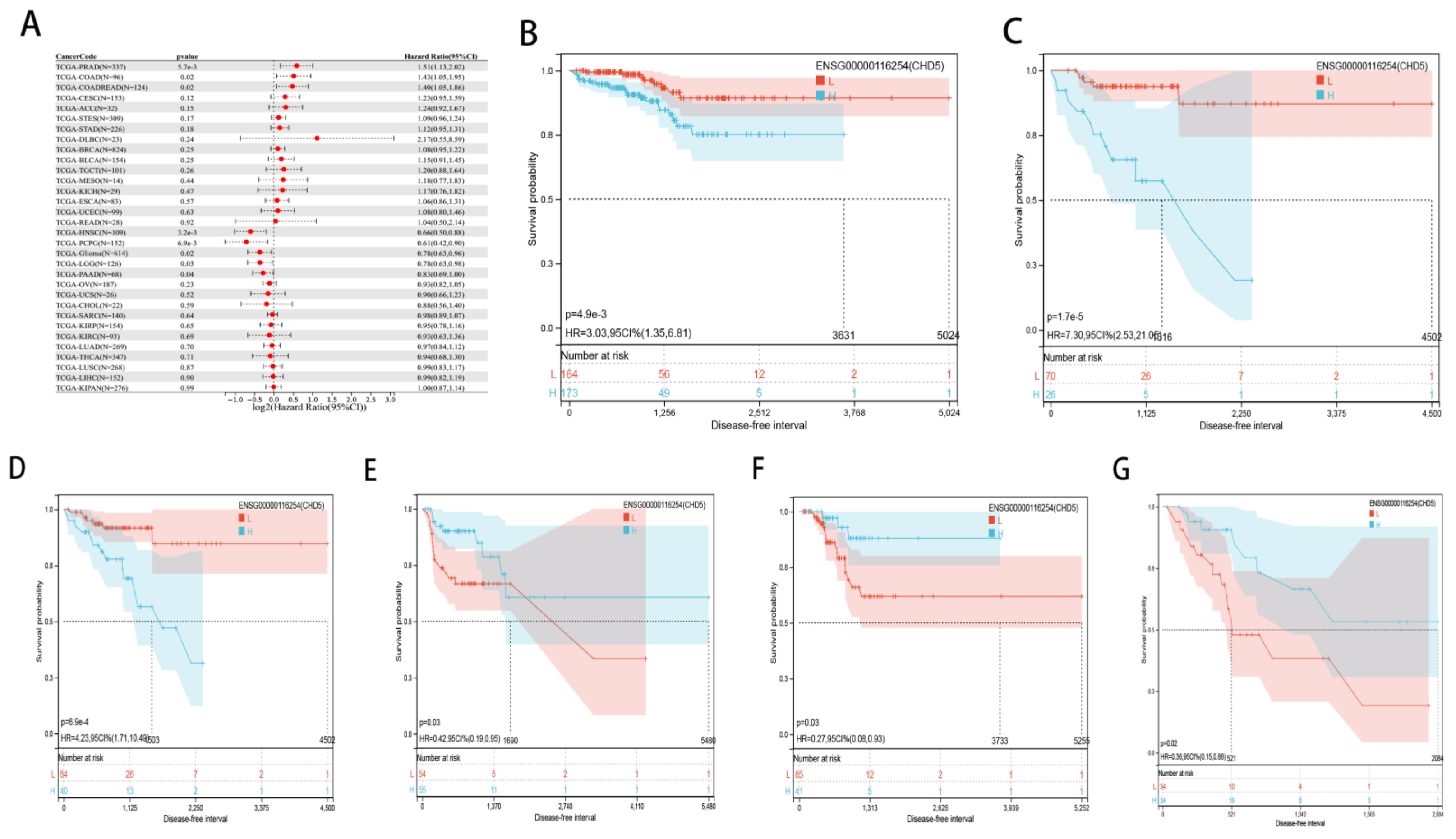
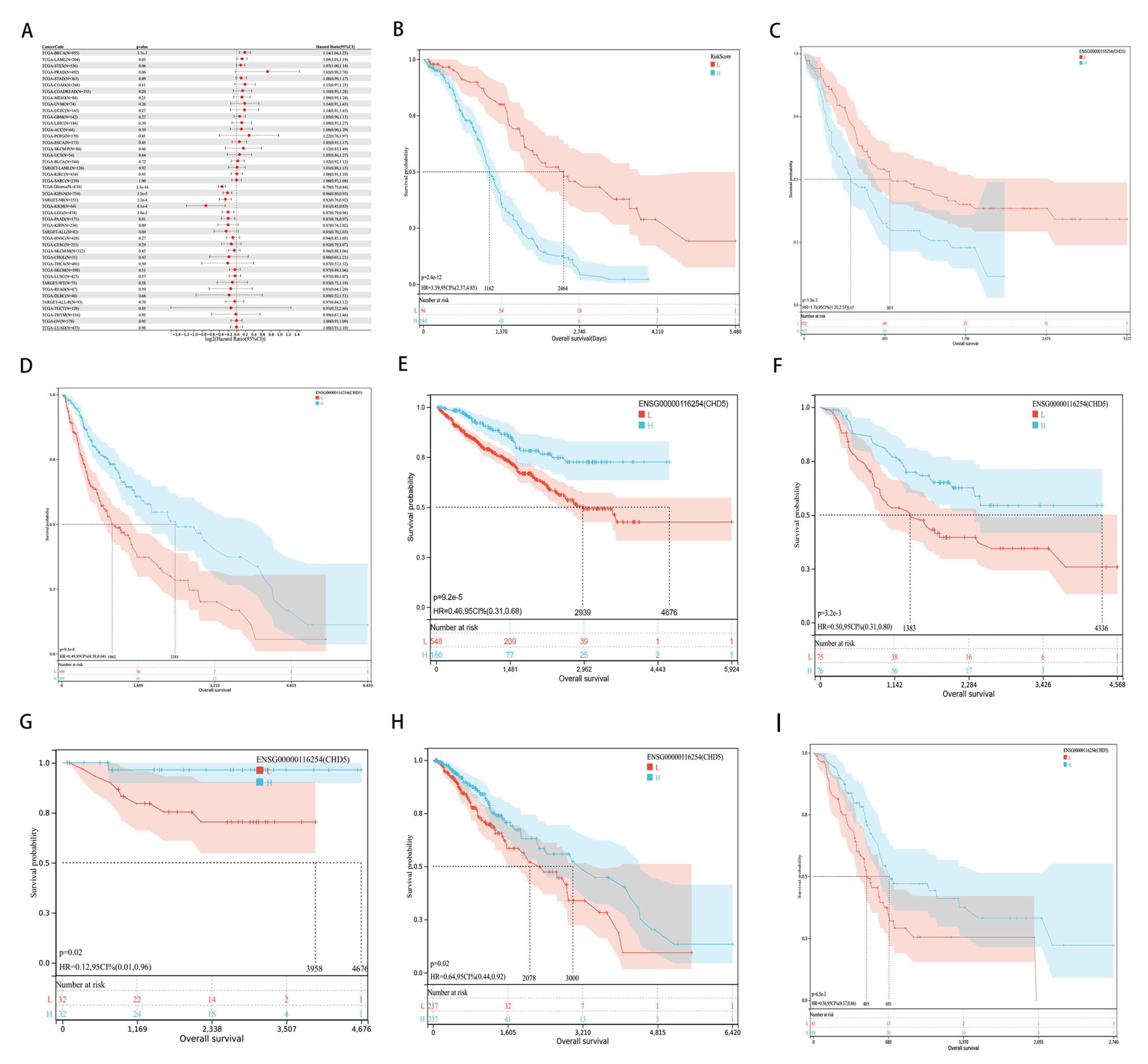
2.2. Pan-Cancer Analysis of the Prognostic Value of CHD5
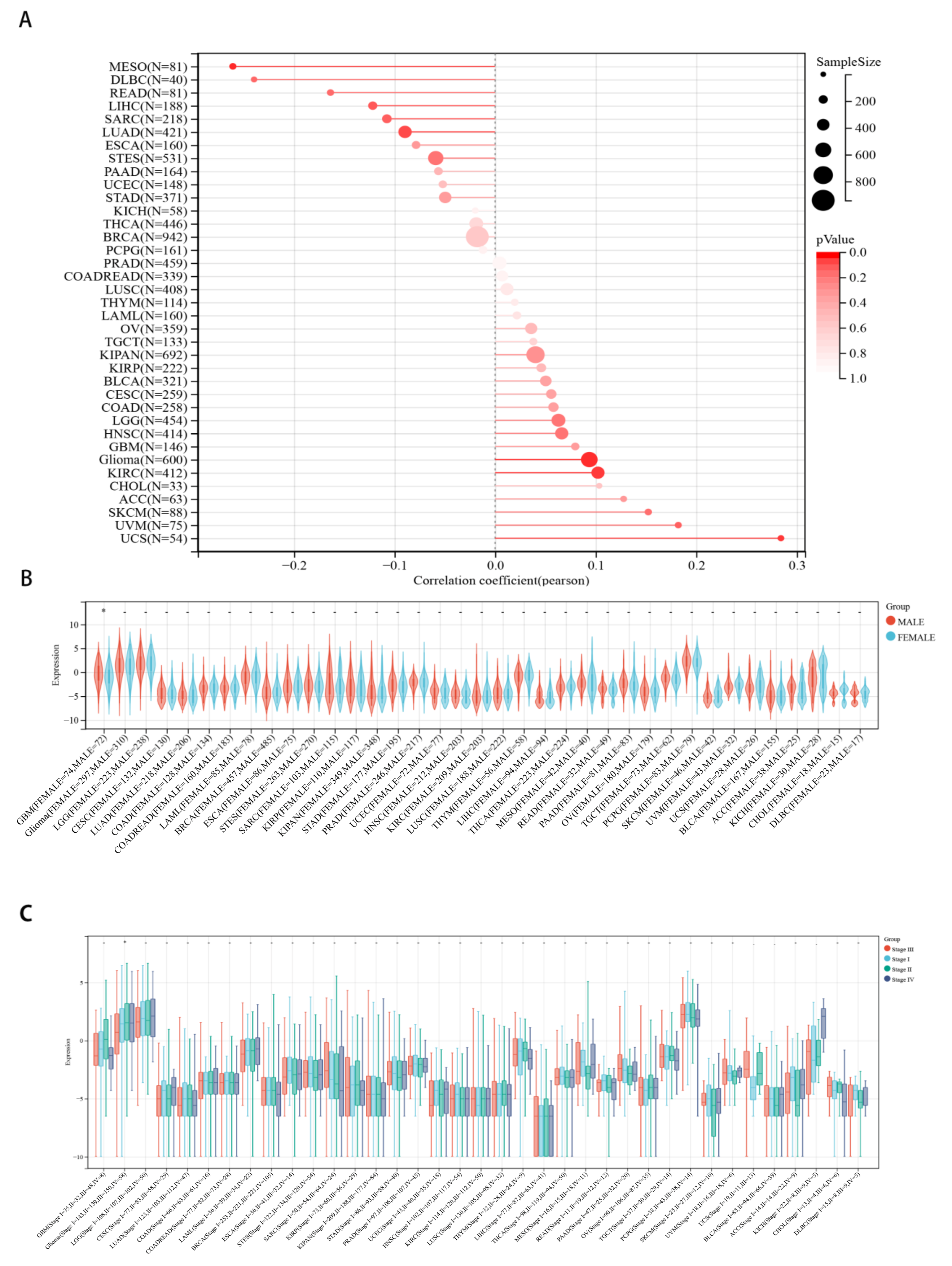
2.3. Correlations between CHD5 Expression and Pan-Cancer Clinicopathological Characteristics
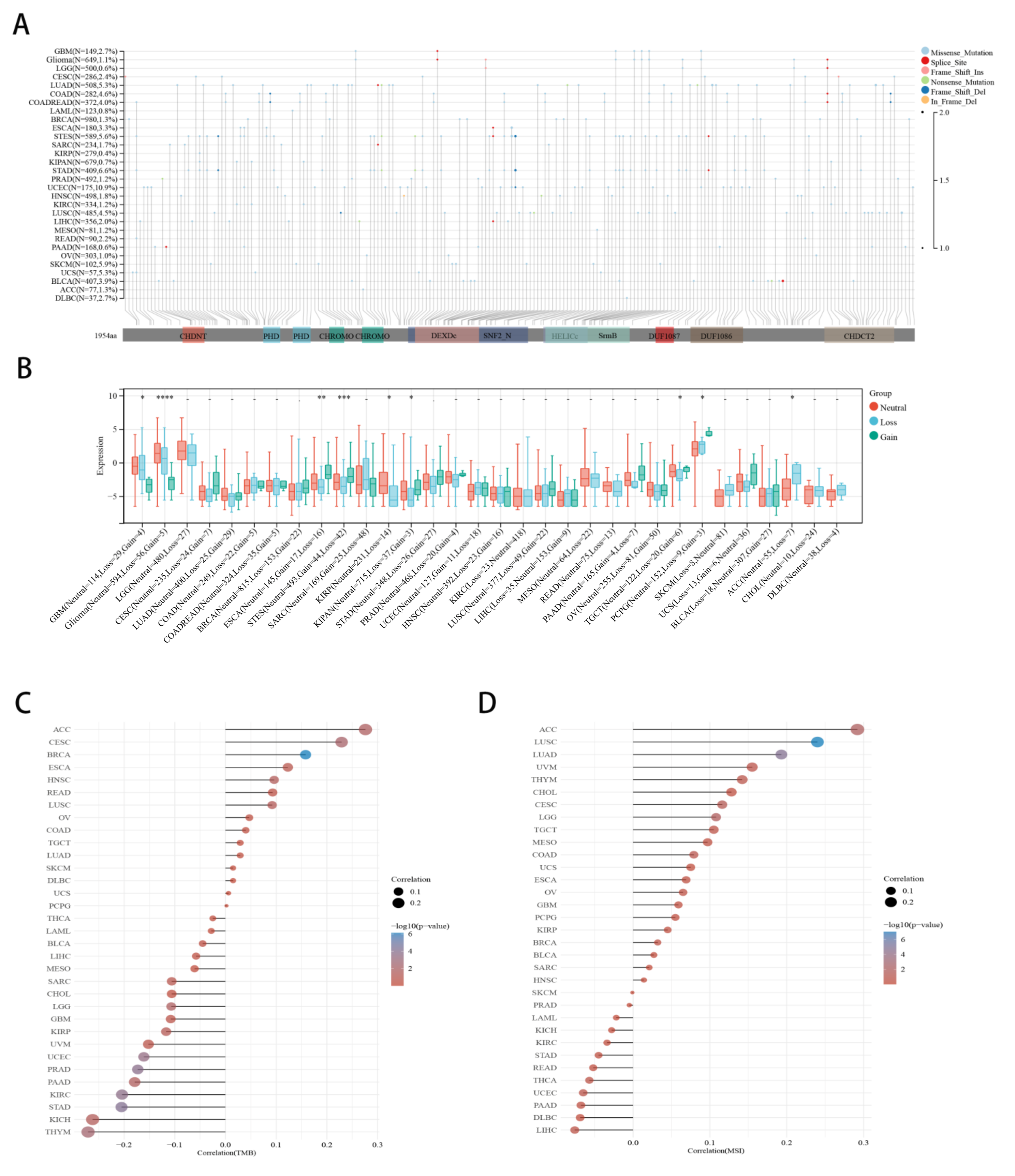
2.4. Correlation between CHD5 Expression and CNV, TMB, MSI in Various Cancer Types
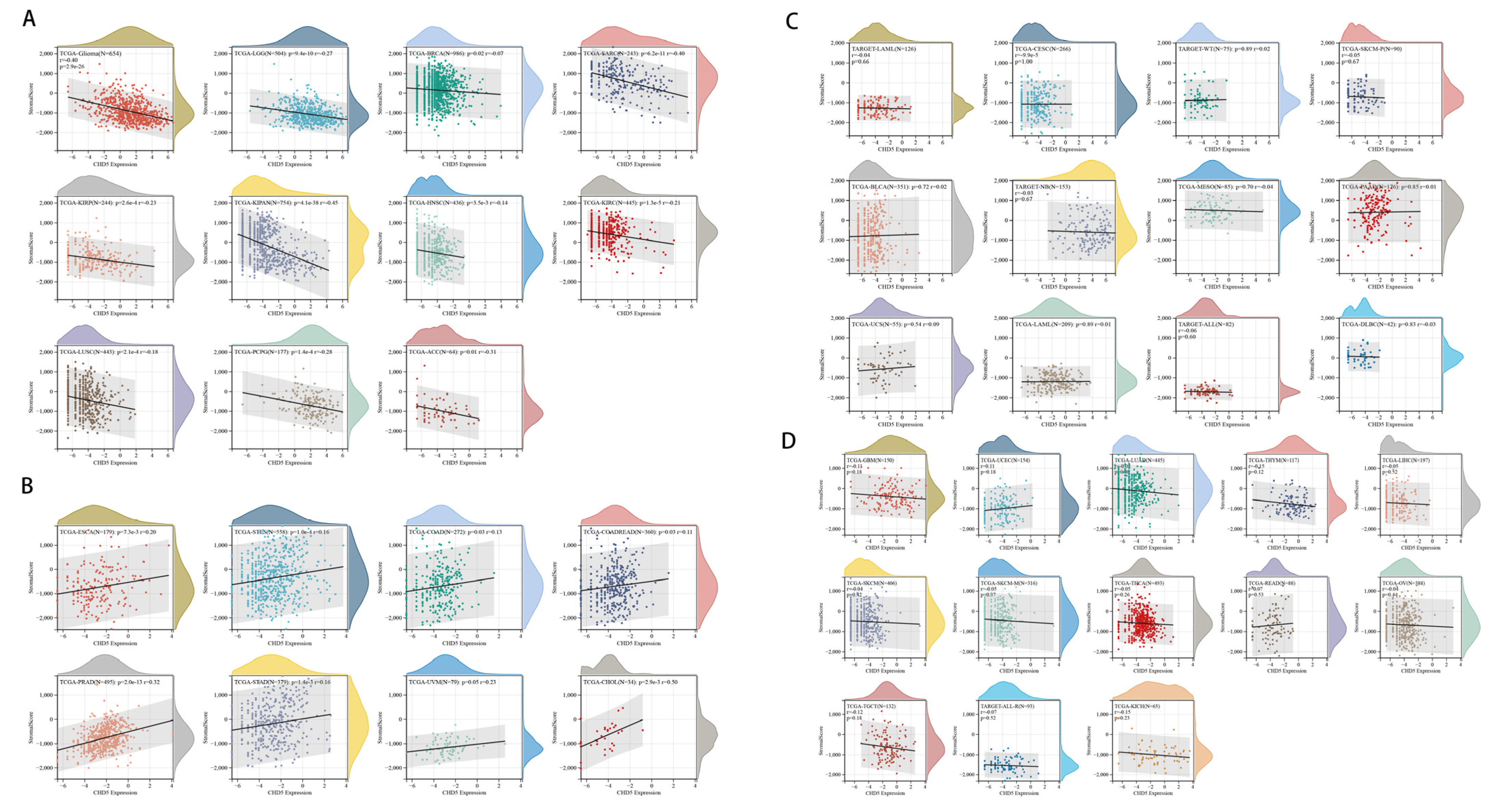
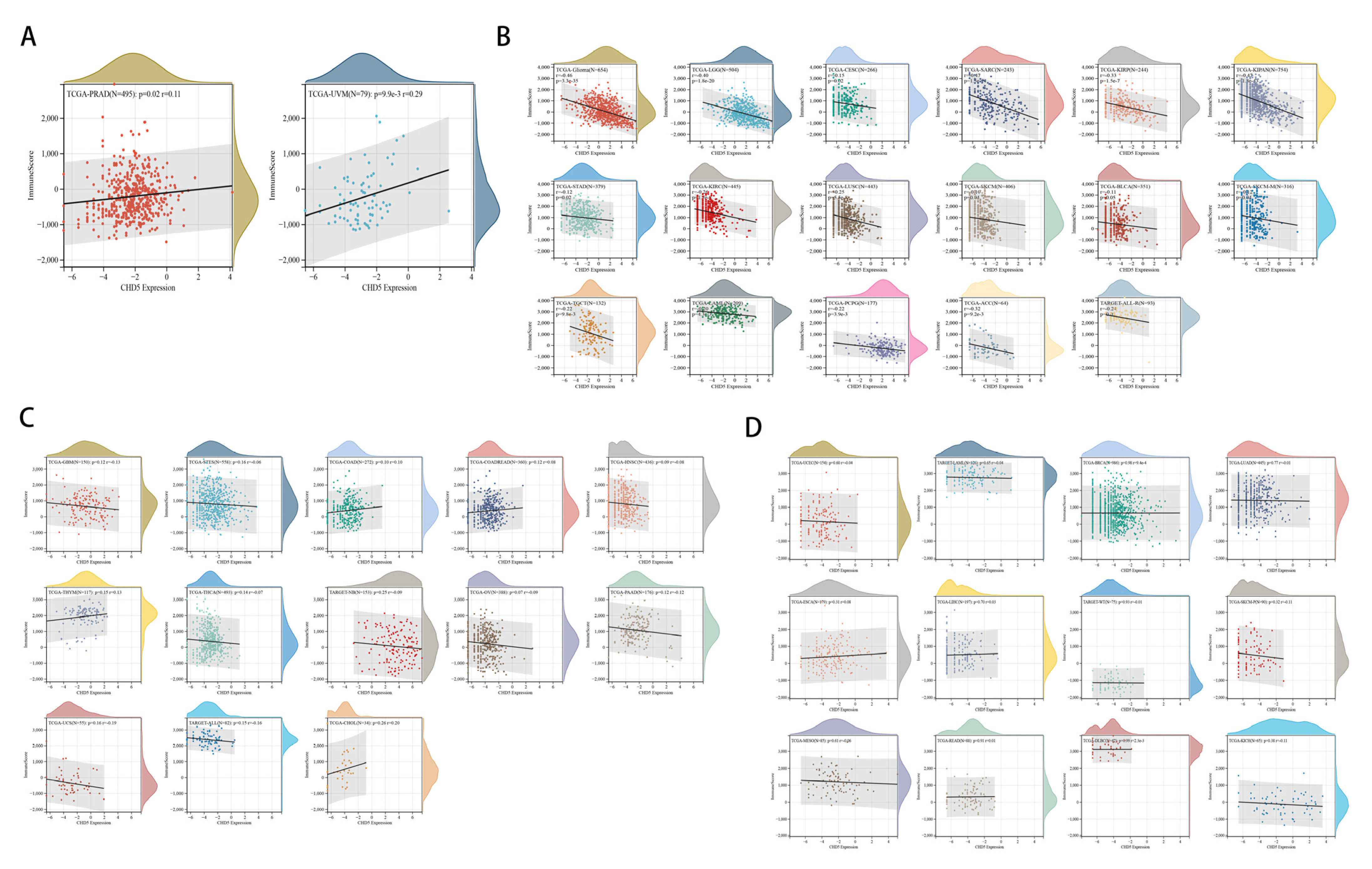
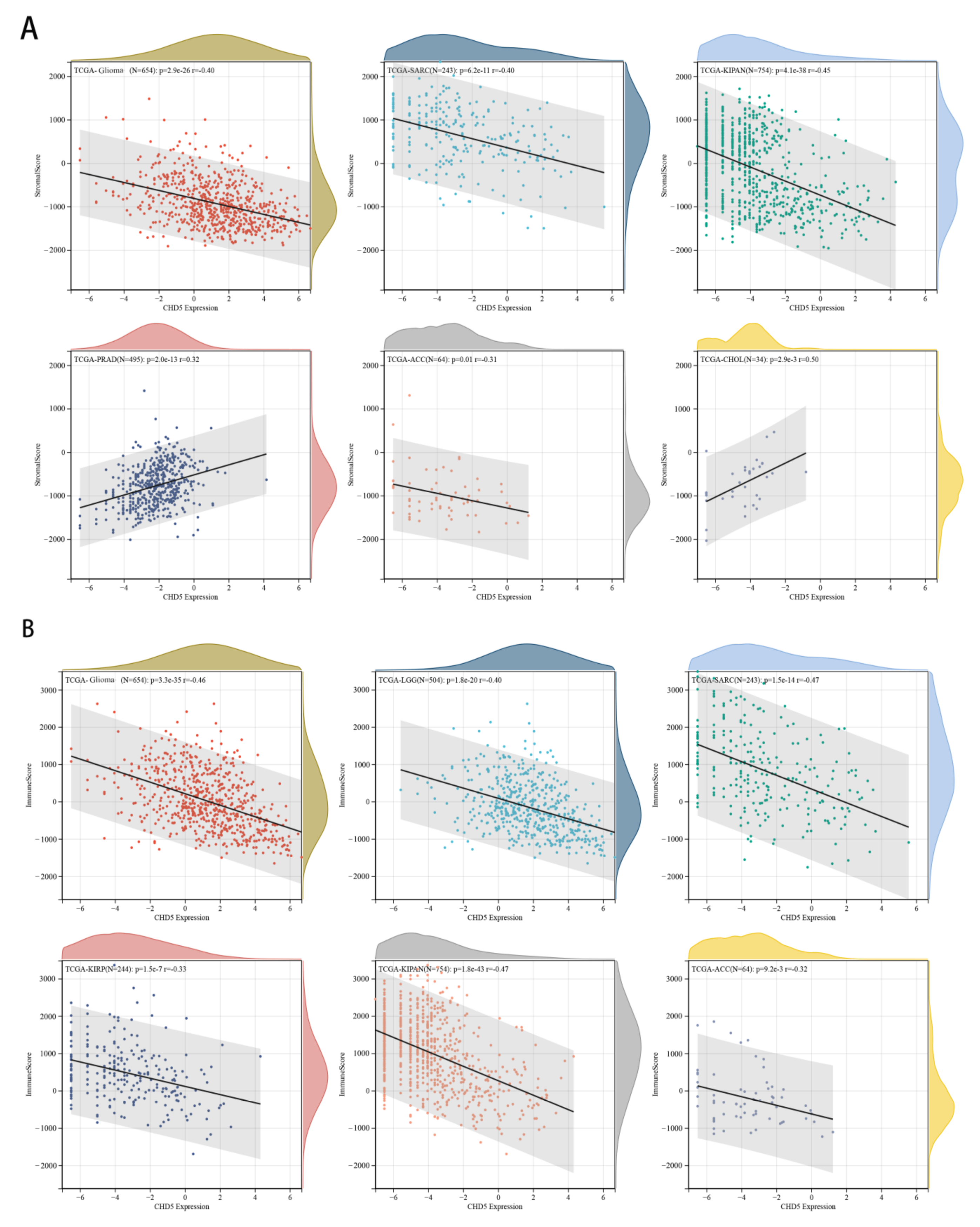
2.5. Correlation between CHD5 Expression and the TME in Different Types of Cancers
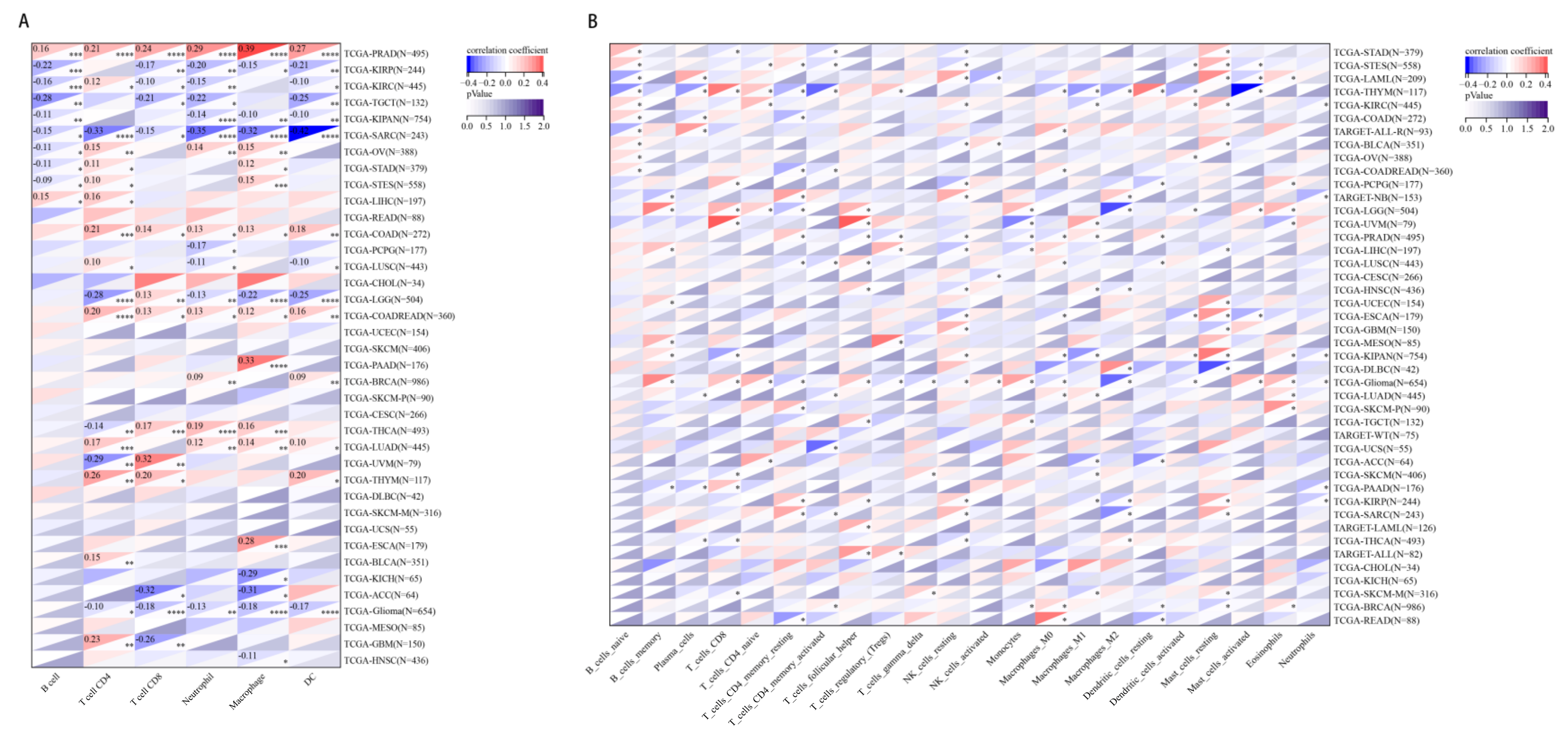
2.6. The Relationship between CHD5 Expression and Immune Cell Infiltration in Various Cancer Types
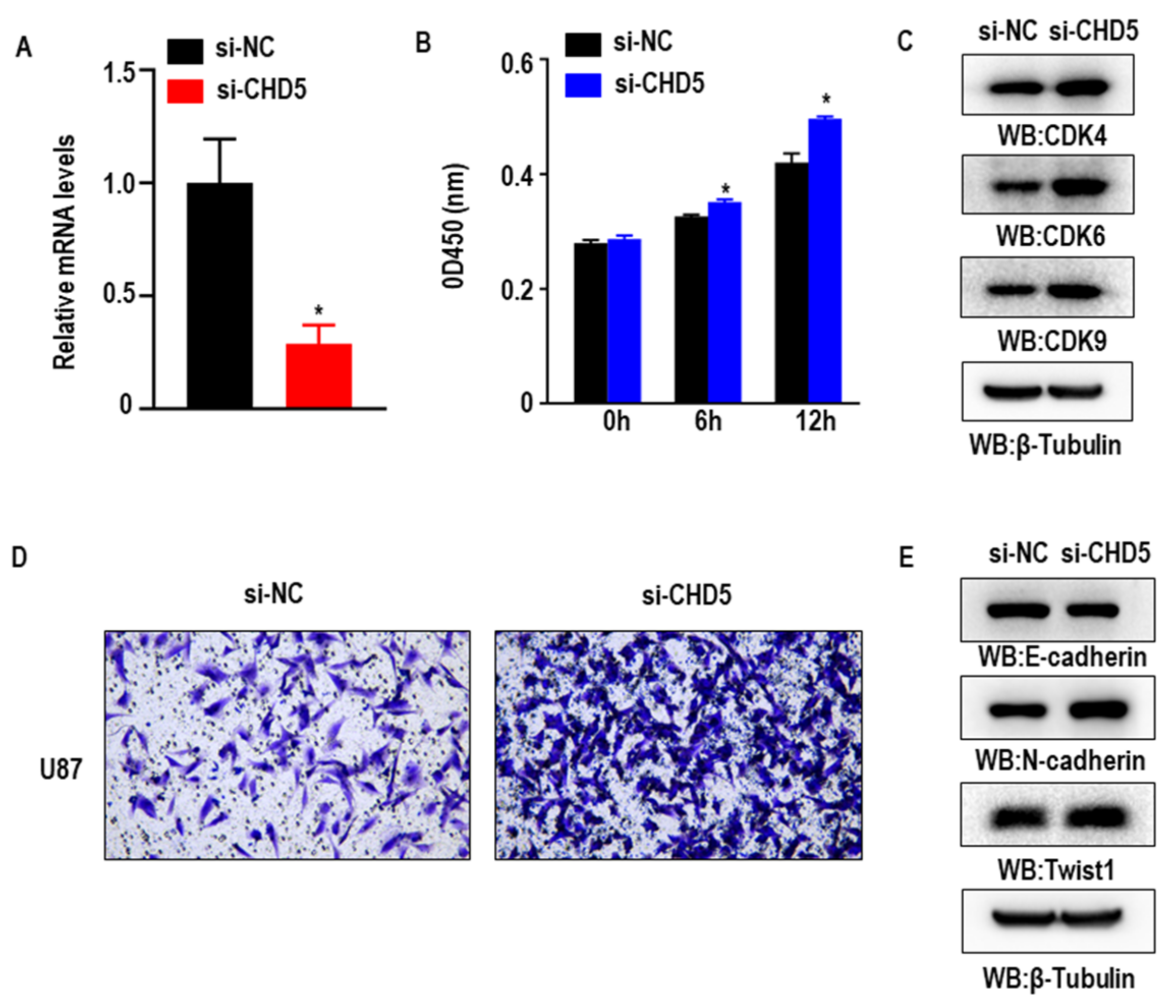
2.7. Knockdown of CHD5 Promotes the Proliferation, Migration and EMT in GBM Cells In Vitro
3. Discussion
4. Materials and Methods
4.1. Analysis of the Expression Level of CHD5 in the Pan-Cancer Datasets
4.2. Identification of the Correlation between CHD5 Expression Levels and Clinicopathological Characteristics and Survival in Human Cancers
4.3. Association between CHD5 Expression and Tumor Mutational Burden (TMB) or Microsatellite Instability (MSI) across Different Types of Cancers
4.4. Correlation between CHD5 Expression and the TME across Different Types of Cancers
4.5. Cell Culture and CHD5 Targeted siRNA Transfection
4.6. RNA Isolation and qPCR
4.7. Cell Viability Assay
4.8. Transwell Assay
4.9. Western Blot Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACC | Adrenocortical carcinoma |
| BLCA | Bladder urothelial carcinoma |
| BRCA | Breast invasive carcinoma |
| CESC | Cervical squamous cell carcinoma and endocervical adenocarcinoma |
| CHOL | Cholangiocarcinoma |
| COAD | Colon adenocarcinoma |
| COADREAD | Colon adenocarcinoma/Rectum adenocarcinoma esophageal carcinoma |
| DLBC | Lymphoid neoplasm diffuse large B-cell lymphoma |
| ESCA | Esophageal carcinoma |
| FPPP | FFPE pilot phase II |
| GBM | Glioblastoma |
| HNSC | Head and neck squamous cell carcinoma |
| KICH | Kidney chromophobe |
| KIPAN | Pan-kidney cohort (KICH+KIRC+KIRP) |
| KIRC | Kidney renal clear cell carcinoma |
| KIRP | Kidney renal papillary cell carcinoma |
| LAML | Acute myeloid leukemia |
| LGG | Low grade glioma |
| LIHC | Liver hepatocellular carcinoma |
| LUAD | Lung adenocarcinoma |
| LUSC | Lung squamous cell carcinoma |
| MESO | Mesothelioma |
| OV | Ovarian serous cystadenocarcinoma |
| PAAD | Pancreatic adenocarcinoma |
| PCPG | Pheochromocytoma and paraganglioma |
| PRAD | Prostate adenocarcinoma |
| READ | Rectum adenocarcinoma |
| SARC | Sarcoma |
| STAD | Stomach adenocarcinoma |
| SKCM | Skin cutaneous melanoma |
| STES | Stomach and esophageal carcinoma |
| TGCT | Testicular germ cell tumors |
| THCA | Thyroid carcinoma |
| THYM | Thymoma |
| UCEC | Uterine corpus endometrial carcinoma |
| UCS | Uterine carcinosarcoma |
| UVM | Uveal melanoma |
| OS | Osteosarcoma |
| ALL | Acute lymphoblastic leukemia |
| NB | Neuroblastoma |
| WT | High-risk wilms tumor |
| TCGA | The Cancer Genome Atlas |
| GEO | Gene Expression Omnibus |
| OS | Overall survival |
| DFI | Disease free interval |
| DFS | Disease free survival |
Appendix A

References
- Paul, S.; Kuo, A.; Schalch, T.; Vogel, H.; Joshua-Tor, L.; McCombie, W.; Gozani, O.; Hammell, M.; Mills, A. Chd5 requires PHD-mediated histone 3 binding for tumor suppression. Cell Rep. 2013, 3, 92–102. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Godfried, M.; Veenstra, M.; Valent, A.; Sluis, P.; Voûte, P.; Versteeg, R.; Caron, H. Lack of interstitial chromosome 1p deletions in clinically-detected neuroblastoma. Eur. J. Cancer 2002, 38, 1513–1519. [Google Scholar] [CrossRef]
- Mansfield, R.; Musselman, C.; Kwan, A.; Oliver, S.; Garske, A.; Davrazou, F.; Denu, J.; Kutateladze, T.; Mackay, J. Plant homeodomain (PHD) fingers of CHD4 are histone H3-binding modules with preference for unmodified H3K4 and methylated H3K9. J. Biol. Chem. 2011, 286, 11779–11791. [Google Scholar] [CrossRef]
- Rajakumara, E.; Wang, Z.; Ma, H.; Hu, L.; Chen, H.; Lin, Y.; Guo, R.; Wu, F.; Li, H.; Lan, F.; et al. PHD finger recognition of unmodified histone H3R2 links UHRF1 to regulation of euchromatic gene expression. Mol. Cell 2011, 43, 275–284. [Google Scholar] [CrossRef]
- Wysocka, J.; Swigut, T.; Xiao, H.; Milne, T.; Kwon, S.; Landry, J.; Kauer, M.; Tackett, A.; Chait, B.; Badenhorst, P.; et al. A PHD finger of NURF couples histone H3 lysine 4 trimethylation with chromatin remodelling. Nature 2006, 442, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Hall, W.; Petrova, A.; Colbert, L.; Hardy, C.; Fisher, S.; Saka, B.; Shelton, J.; Warren, M.; Pantazides, B.; Gandhi, K.; et al. Low CHD5 expression activates the DNA damage response and predicts poor outcome in patients undergoing adjuvant therapy for resected pancreatic cancer. Oncogene 2014, 33, 5450–5456. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Lau, K.; So, L.; Lam, Y. CHD5 is down-regulated through promoter hypermethylation in gastric cancer. J. Biomed. Sci. 2009, 16, 95. [Google Scholar] [CrossRef]
- Zhao, R.; Meng, F.; Wang, N.; Ma, W.; Yan, Q. Silencing of CHD5 gene by promoter methylation in leukemia. PLoS ONE 2014, 9, e85172. [Google Scholar] [CrossRef]
- Baykara, O.; Tansarikaya, M.; Bulut, P.; Demirkaya, A.; Buyru, N. CHD5 is a potential tumor suppressor in non small cell lung cancer (NSCLC). Gene 2017, 618, 65–68. [Google Scholar] [CrossRef]
- Fatemi, M.; Paul, T.; Brodeur, G.; Shokrani, B.; Brim, H.; Ashktorab, H. Epigenetic silencing of CHD5, a novel tumor-suppressor gene, occurs in early colorectal cancer stages. Cancer 2014, 120, 172–180. [Google Scholar] [CrossRef]
- Du, X.; Wu, T.; Lu, J.; Zang, L.; Song, N.; Yang, T.; Zhao, H.; Wang, S. Decreased expression of chromodomain helicase DNA-binding protein 5 is an unfavorable prognostic marker in patients with primary gallbladder carcinoma. Clin. Transl. Oncol. Off. Publ. Fed. Span. Oncol. Soc. Natl. Cancer Inst. Mex. 2013, 15, 198–204. [Google Scholar] [CrossRef]
- Wong, R.; Chan, L.; Tsang, T.; Lee, C.; Cheung, T.; Yim, S.; Siu, N.; Lee, S.; Yu, M.; Chim, S.; et al. CHD5 Downregulation Associated with Poor Prognosis in Epithelial Ovarian Cancer. Gynecol. Obstet. Investig. 2011, 72, 203–207. [Google Scholar] [CrossRef]
- Greten, F.; Grivennikov, S. Inflammation and Cancer: Triggers, Mechanisms, and Consequences. Immunity 2019, 51, 27–41. [Google Scholar] [CrossRef]
- Petitprez, F.; Meylan, M.; de Reyniès, A.; Sautès-Fridman, C.; Fridman, W. The Tumor Microenvironment in the Response to Immune Checkpoint Blockade Therapies. Front. Immunol. 2020, 11, 784. [Google Scholar] [CrossRef]
- Liu, Q.; Aminu, B.; Roscow, O.; Zhang, W. Targeting the Ubiquitin Signaling Cascade in Tumor Microenvironment for Cancer Therapy. Int. J. Mol. Sci. 2021, 22, 791. [Google Scholar] [CrossRef]
- Turley, S.; Cremasco, V.; Astarita, J. Immunological hallmarks of stromal cells in the tumour microenvironment. Nat. Rev. Immunol. 2015, 15, 669–682. [Google Scholar] [CrossRef]
- Galon, J.; Pagès, F.; Marincola, F.; Angell, H.; Thurin, M.; Lugli, A.; Zlobec, I.; Berger, A.; Bifulco, C.; Botti, G.; et al. Cancer classification using the Immunoscore: A worldwide task force. J. Transl. Med. 2012, 10, 205. [Google Scholar] [CrossRef]
- Angell, H.; Bruni, D.; Barrett, J.; Herbst, R.; Galon, J. The Immunoscore: Colon Cancer and Beyond. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2020, 26, 332–339. [Google Scholar] [CrossRef]
- Kolla, V.; Zhuang, T.; Higashi, M.; Naraparaju, K.; Brodeur, G. Role of CHD5 in human cancers: 10 years later. Cancer Res. 2014, 74, 652–658. [Google Scholar] [CrossRef]
- Laut, A.; Dorneburg, C.; Fürstberger, A.; Barth, T.; Kestler, H.; Debatin, K.; Beltinger, C. CHD5 inhibits metastasis of neuroblastoma. Oncogene 2022, 41, 622–633. [Google Scholar] [CrossRef]
- Lang, J.; Tobias, E.; Mackie, R. Preliminary evidence for involvement of the tumour suppressor gene CHD5 in a family with cutaneous melanoma. Br. J. Dermatol. 2011, 164, 1010–1016. [Google Scholar] [CrossRef] [PubMed]
- Pei, S.; Chen, Z.; Tan, H.; Fan, L.; Zhang, B.; Zhao, C. SLC16A1-AS1 enhances radiosensitivity and represses cell proliferation and invasion by regulating the miR-301b-3p/CHD5 axis in hepatocellular carcinoma. Environ. Sci. Pollut. Res. Int. 2020, 27, 42778–42790. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; Chen, L.; Luo, H.; Li, H.; Kong, Q.; Jiao, F.; Pang, S.; Zhang, M.; Lan, F.; Fan, W.; et al. A rare CHD5 haplotype and its interactions with environmental factors predicting hepatocellular carcinoma risk. BMC Cancer 2018, 18, 658. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Xiao, D.; Xu, T.; Yuan, Y. miRNA-24-3p promotes cell proliferation and regulates chemosensitivity in head and neck squamous cell carcinoma by targeting CHD5. Future Oncol. 2016, 12, 2701–2712. [Google Scholar] [CrossRef]
- Thompson, P.; Gotoh, T.; Kok, M.; White, P.; Brodeur, G. CHD5, a new member of the chromodomain gene family, is preferentially expressed in the nervous system. Oncogene 2003, 22, 1002–1011. [Google Scholar] [CrossRef]
- White, P.; Thompson, P.; Gotoh, T.; Okawa, E.; Igarashi, J.; Kok, M.; Winter, C.; Gregory, S.; Hogarty, M.; Maris, J.; et al. Definition and characterization of a region of 1p36.3 consistently deleted in neuroblastoma. Oncogene 2005, 24, 2684–2694. [Google Scholar] [CrossRef][Green Version]
- Fujita, T.; Igarashi, J.; Okawa, E.; Gotoh, T.; Manne, J.; Kolla, V.; Kim, J.; Zhao, H.; Pawel, B.; London, W.; et al. CHD5, a tumor suppressor gene deleted from 1p36.31 in neuroblastomas. J. Natl. Cancer Inst. 2008, 100, 940–949. [Google Scholar] [CrossRef]
- Okawa, E.; Gotoh, T.; Manne, J.; Igarashi, J.; Fujita, T.; Silverman, K.; Xhao, H.; Mosse, Y.; White, P.; Brodeur, G. Expression and sequence analysis of candidates for the 1p36.31 tumor suppressor gene deleted in neuroblastomas. Oncogene 2008, 27, 803–810. [Google Scholar] [CrossRef]
- Garcia, I.; Mayol, G.; Rodríguez, E.; Suñol, M.; Gershon, T.; Ríos, J.; Cheung, N.; Kieran, M.; George, R.; Perez-Atayde, A.; et al. Expression of the neuron-specific protein CHD5 is an independent marker of outcome in neuroblastoma. Mol. Cancer 2010, 9, 277. [Google Scholar] [CrossRef]
- Fancello, L.; Gandini, S.; Pelicci, P.; Mazzarella, L. Tumor mutational burden quantification from targeted gene panels: Major advancements and challenges. J. Immunother. Cancer 2019, 7, 183. [Google Scholar] [CrossRef]
- Goodman, A.; Kato, S.; Bazhenova, L.; Patel, S.; Frampton, G.; Miller, V.; Stephens, P.; Daniels, G.; Kurzrock, R. Tumor Mutational Burden as an Independent Predictor of Response to Immunotherapy in Diverse Cancers. Mol. Cancer Ther. 2017, 16, 2598–2608. [Google Scholar] [CrossRef]
- Janjigian, Y.; Bendell, J.; Calvo, E.; Kim, J.; Ascierto, P.; Sharma, P.; Ott, P.; Peltola, K.; Jaeger, D.; Evans, J.; et al. CheckMate-032 Study: Efficacy and Safety of Nivolumab and Nivolumab Plus Ipilimumab in Patients with Metastatic Esophagogastric Cancer. J. Clin. Oncol.: Off. J. Am. Soc. Clin. Oncol. 2018, 36, 2836–2844. [Google Scholar] [CrossRef]
- Klempner, S.; Fabrizio, D.; Bane, S.; Reinhart, M.; Peoples, T.; Ali, S.; Sokol, E.; Frampton, G.; Schrock, A.; Anhorn, R.; et al. Tumor Mutational Burden as a Predictive Biomarker for Response to Immune Checkpoint Inhibitors: A Review of Current Evidence. Oncol. 2020, 25, e147–e159. [Google Scholar] [CrossRef]
- Kim, T.; Laird, P.; Park, P. The landscape of microsatellite instability in colorectal and endometrial cancer genomes. Cell 2013, 155, 858–868. [Google Scholar] [CrossRef]
- Picard, E.; Verschoor, C.; Ma, G.; Pawelec, G. Relationships Between Immune Landscapes, Genetic Subtypes and Responses to Immunotherapy in Colorectal Cancer. Front. Immunol. 2020, 11, 369. [Google Scholar] [CrossRef]
- Stanley, F.; Moore, S.; Goodarzi, A. CHD chromatin remodelling enzymes and the DNA damage response. Mutat. Res. 2013, 750, 31–44. [Google Scholar] [CrossRef]
- Bejarano, L.; Jordāo, M.; Joyce, J. Therapeutic Targeting of the Tumor Microenvironment. Cancer Discov. 2021, 11, 933–959. [Google Scholar] [CrossRef]
- Petitprez, F.; de Reyniès, A.; Keung, E.; Chen, T.; Sun, C.; Calderaro, J.; Jeng, Y.; Hsiao, L.; Lacroix, L.; Bougoüin, A.; et al. B cells are associated with survival and immunotherapy response in sarcoma. Nature 2020, 577, 556–560. [Google Scholar] [CrossRef]
- Klaver, Y.; Rijnders, M.; Oostvogels, A.; Wijers, R.; Smid, M.; Grünhagen, D.; Verhoef, C.; Sleijfer, S.; Lamers, C.; Debets, R. Differential quantities of immune checkpoint-expressing CD8 T cells in soft tissue sarcoma subtypes. J. Immunother. Cancer 2020, 8. [Google Scholar] [CrossRef]
- Lee-Chang, C.; Rashidi, A.; Miska, J.; Zhang, P.; Pituch, K.; Hou, D.; Xiao, T.; Fischietti, M.; Kang, S.; Appin, C.; et al. Myeloid-Derived Suppressive Cells Promote B cell-Mediated Immunosuppression via Transfer of PD-L1 in Glioblastoma. Cancer Immunol. Res. 2019, 7, 1928–1943. [Google Scholar] [CrossRef]
- Altieri, R.; Barbagallo, D.; Certo, F.; Broggi, G.; Ragusa, M.; Di Pietro, C.; Caltabiano, R.; Magro, G.; Peschillo, S.; Purrello, M.; et al. Peritumoral Microenvironment in High-Grade Gliomas: From FLAIRectomy to Microglia-Glioma Cross-Talk. Brain Sci. 2021, 11, 200. [Google Scholar] [CrossRef]
- Goldman, M.; Craft, B.; Hastie, M.; Repečka, K.; McDade, F.; Kamath, A.; Banerjee, A.; Luo, Y.; Rogers, D.; Brooks, A.; et al. Visualizing and interpreting cancer genomics data via the Xena platform. Nat. Biotechnol. 2020, 38, 675–678. [Google Scholar] [CrossRef]
- Blum, A.; Wang, P.; Zenklusen, J. SnapShot: TCGA-Analyzed Tumors. Cell 2018, 173, 530. [Google Scholar] [CrossRef]
- The Genotype-Tissue Expression (GTEx) project. Nat. Genet. 2013, 45, 580–585. [CrossRef]
- Nusinow, D.; Szpyt, J.; Ghandi, M.; Rose, C.; McDonald, E.; Kalocsay, M.; Jané-Valbuena, J.; Gelfand, E.; Schweppe, D.; Jedrychowski, M.; et al. Quantitative Proteomics of the Cancer Cell Line Encyclopedia. Cell 2020, 180, 387–402.e16. [Google Scholar] [CrossRef]
- Beroukhim, R.; Mermel, C.; Porter, D.; Wei, G.; Raychaudhuri, S.; Donovan, J.; Barretina, J.; Boehm, J.; Dobson, J.; Urashima, M.; et al. The landscape of somatic copy-number alteration across human cancers. Nature 2010, 463, 899–905. [Google Scholar] [CrossRef]
- Mermel, C.; Schumacher, S.; Hill, B.; Meyerson, M.; Beroukhim, R.; Getz, G. GISTIC2.0 facilitates sensitive and confident localization of the targets of focal somatic copy-number alteration in human cancers. Genome Biol. 2011, 12, R41. [Google Scholar] [CrossRef]
- Thorsson, V.; Gibbs, D.; Brown, S.; Wolf, D.; Bortone, D.; Ou Yang, T.; Porta-Pardo, E.; Gao, G.; Plaisier, C.; Eddy, J.; et al. The Immune Landscape of Cancer. Immunity 2018, 48, 812–830.e14. [Google Scholar] [CrossRef]
- Bonneville, R.; Krook, M.; Kautto, E.; Miya, J.; Wing, M.; Chen, H.; Reeser, J.; Yu, L.; Roychowdhury, S. Landscape of Microsatellite Instability Across 39 Cancer Types. JCO Precis. Oncol. 2017, 2017, 1–15. [Google Scholar] [CrossRef]
- Yoshihara, K.; Shahmoradgoli, M.; Martínez, E.; Vegesna, R.; Kim, H.; Torres-Garcia, W.; Treviño, V.; Shen, H.; Laird, P.; Levine, D.; et al. Inferring tumour purity and stromal and immune cell admixture from expression data. Nat. Commun. 2013, 4, 2612. [Google Scholar] [CrossRef]
- Li, T.; Fan, J.; Wang, B.; Traugh, N.; Chen, Q.; Liu, J.; Li, B.; Liu, X. TIMER: A Web Server for Comprehensive Analysis of Tumor-Infiltrating Immune Cells. Cancer Res. 2017, 77, e108–e110. [Google Scholar] [CrossRef] [PubMed]
- Newman, A.; Liu, C.; Green, M.; Gentles, A.; Feng, W.; Xu, Y.; Hoang, C.; Diehn, M.; Alizadeh, A. Robust enumeration of cell subsets from tissue expression profiles. Nat. Methods 2015, 12, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Xia, H.; Ni, D.; Hu, Y.; Liu, J.; Qin, Y.; Zhou, Q.; Yi, Q.; Xie, Y. High-Dose Dexamethasone Manipulates the Tumor Microenvironment and Internal Metabolic Pathways in Anti-Tumor Progression. Int. J. Mol. Sci. 2020, 21, 1846. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, L.; Shao, F.; Luo, T.; Li, Q.; Tan, D.; Tan, Y. Pan-Cancer Analysis Identifies CHD5 as a Potential Biomarker for Glioma. Int. J. Mol. Sci. 2022, 23, 8489. https://doi.org/10.3390/ijms23158489
Xu L, Shao F, Luo T, Li Q, Tan D, Tan Y. Pan-Cancer Analysis Identifies CHD5 as a Potential Biomarker for Glioma. International Journal of Molecular Sciences. 2022; 23(15):8489. https://doi.org/10.3390/ijms23158489
Chicago/Turabian StyleXu, Lei, Fengling Shao, Tengling Luo, Qijun Li, Dongmei Tan, and Yi Tan. 2022. "Pan-Cancer Analysis Identifies CHD5 as a Potential Biomarker for Glioma" International Journal of Molecular Sciences 23, no. 15: 8489. https://doi.org/10.3390/ijms23158489
APA StyleXu, L., Shao, F., Luo, T., Li, Q., Tan, D., & Tan, Y. (2022). Pan-Cancer Analysis Identifies CHD5 as a Potential Biomarker for Glioma. International Journal of Molecular Sciences, 23(15), 8489. https://doi.org/10.3390/ijms23158489




