Neurobiological Mechanisms Modulating Emotionality, Cognition and Reward-Related Behaviour in High-Fat Diet-Fed Rodents
Abstract
1. Introduction
2. Aim of the Present Review
3. Important Considerations on the Use of High-Fat Diet Treatments in Rodents
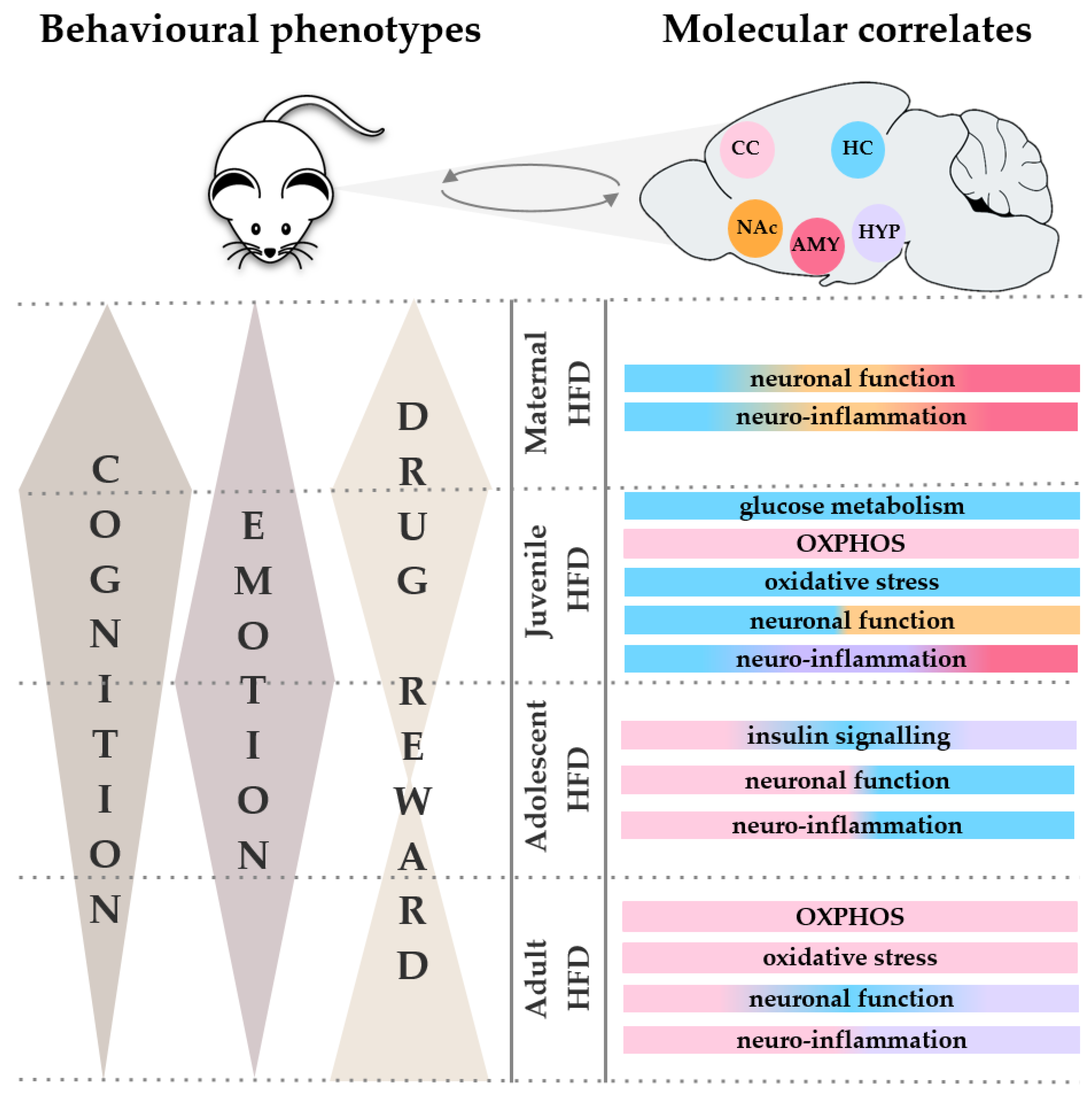
4. Impact of a High-Fat Diet Treatment on Emotionality, Cognition and Reward-Related Behaviour
4.1. Impact of a Maternal High-Fat Diet Treatment in Dams
4.2. Impact of a Maternal High-Fat Diet Treatment in Offsspring
4.3. Impact of a High-Fat Diet Treatment during the Onset of Puberty and Early Adolescence
4.3.1. Cognition and Emotionality
4.3.2. Psychostimulant-Related Behaviour
4.4. Impact of a High-Fat Diet Treatment during Late Adolescence and Young Adulthood
4.5. Impact of a High-Fat Diet Treatment in Adulhood
4.6. Conclusions
5. Impact of a High-Fat Diet Treatment on Molecular Correlates of Energy Metabolism and Plasticity
5.1. Impact of a High-Fat Diet Treatment on the Cerebral Cortex
5.1.1. Energy Metabolism
5.1.2. Mitochondria-Related Functions and Oxidative Stress
5.1.3. Neuroplasticity and Neuroinflammation
5.2. Impact of a High-Fat Diet Treatment on the Hippocampus
5.2.1. Energy Metabolism
5.2.2. Mitochondria-Related Functions and Oxidative Stress
5.2.3. Neuroplasticity and Neuroinflammation
5.3. Impact of a High-Fat Diet Treatment on the Hypothalamus
5.3.1. Energy Metabolism
| Brain Area & Ref. | Overall Function | Outcomes | ||
|---|---|---|---|---|
| ⬆ (Increased) | ⬇ (Decreased) | ⬌ (Not Changed) | ||
| Maternal HFD treatment Testing in offspring | ||||
| PFC [87] | Neuronal func. | Level of DA, DOPAC | ||
| HC [89,145] | Neuronal func. | Dendritic spine length | Dendritic spine density DCX-positive cells | |
| Neuroinflam. | Astrocyte process number and total length | |||
| AMY [145] | Neuronal func. | Dendritic spine length | Dendritic spine density | |
| NAc [84,87] | Neuronal func. | Level of DA, DOPAC Ddr2 mRNA Htr1a mRNA | TH density fibres Expression of TH, DAT, D1/2R | |
| STR [85,87] | Neuronal func. | Expression of TH, DAT, D1/2R | ||
| VTA [84,87] | Neuronal func. | Htr1a mRNA | TH positive neurons § Th mRNA § | TH positive neurons § Th mRNA § |
| Juvenile HFD treatment | ||||
| CC [105] | Glucose metab. | Glucose transport, glycolysis | ||
| Insulin signaling | Insulin level | Insulin sensitivity | ||
| OXPHOS | Expression of ETC CIV-V | Spare respiratory capacity | Expression of ETC CI-III, basal OCR, State 3 OCR, level of ATP | |
| HC [103,104,105] | Glucose metab. | Glycolysis | Glucose transport | |
| Insulin signaling | Insulin level and sensitivity | |||
| OXPHOS | Expression of ETC CI-II | Expression of ETC CI-V, level of ATP | ||
| MT biogenesis | Expression of PGC1α, PPARγ | |||
| Oxidative stress | Lipid peroxidation | Anti-oxidant defenses (SOD, GPX) | ||
| Neuronal func. | Dendritic spines, expression of SYP | Expression of BDNF | ||
| Neuroinflam. | Level of IL10, micro/astrogliosis | Level of IL6 | Level of IL6, IL1β, TNFα, microgliosis | |
| HYP [104,147,148] | Insulin signaling | Ins mRNA | Mtor, Irs1 mRNA | |
| OXPHOS | Expression of ETC CI-V, level of ATP | |||
| Oxidative stress | Anti-oxidant defenses (SOD, CAT), lipid peroxidation | |||
| Neuroinflam. | Microgliosis | Tgfβ mRNA, microgliosis | ||
| AMY [103,104] | Neuroinflam. | Level of TNFα, microgliosis | Level of IL6 | |
| NAc [107,108,113] | Insulin signaling | Mtor mRNA | ||
| Neuronal func. | Expression of D1R | Expression of DAT | Basal DA release; level of DA, DOPAC; expression of TH, DAT, D1/2R | |
| VTA [113] | Neuronal func. | Spontaneous/bursting DA activity Expression of TH, DAT | ||
| HFD treatment in late adolescence | ||||
| CC [115,117,119,121] | Insulin signaling | Insulin sensitivity | ||
| Oxidative stress | Anti-oxidant defenses (GSH, GSSG), lipid peroxidation | |||
| Neuronal func. | Ddr2 mRNA | Gabbr1/2 mRNA | Expression of BDNF, SYP Drd1 mRNA | |
| Neuroinflam. | Il1β, Il2 mRNA | Il10, Il4, Il6, Tnfα, Tgfβ, Ifnγ mRNA | ||
| HC [117,118,119,121] | Insulin signaling | Insulin sensitivity | ||
| Oxidative stress | Anti-oxidant defenses (GSH, GSSG), lipid peroxidation | |||
| Neuronal func. | Nissl staining, Bdnf mRNA, level of 5-HT; Htr1a, Slc6a4, Ido2 mRNA | CA1 pyramidal layer thickness, CA1 LTP, expression of PSD95, BDNF, SYP | ||
| Neuroinflam. | Il1β, Il2, Il6, Il17 mRNA | Il10 mRNA | Il4, Tnfα, Tgfβ, Ifnγ mRNA | |
| HYP [149] | Neuroinflam. | IL1β mRNA, astrogliosis | ||
| Adult HFD treatment | ||||
| CC [127,144] | Glucose metab. | Level of glucose, lactate | ||
| AA/PL metab. | Level of PEA | Level of Gln, Glu, GABA | ||
| OXPHOS | State 3 OCR, Spare respiratory capacity | |||
| Oxidative stress | Lipid peroxidation | Anti-oxidant defenses (GSH, GSSG, SOD) | Anti-oxidant defenses (GSH, As) | |
| Neuronal func. | Expression of BDNF | Level of NAA, expression of PSD95, SYP, SYN, VGLUT1/2, VGAT | ||
| Neuroinflam. | Level of TNFα, IL1β | Micro/astrogliosis | ||
| HC [126,127] | Glucose metab. | Level of glucose, lactate | ||
| AA/PL metab. | Level of Gln, Glu, GABA, PEA | |||
| Oxidative stress | Anti-oxidant defenses (GSH, As) | |||
| Neuronal func. | Expression of SYN, VGLUT1, VGAT | Level of NAA, expression of PSD95, SYP, VGLUT2 | ||
| Neuroinflam. | Tnfα, mRNA, microgliosis | |||
| HYP [127,146] | Glucose metab. | Level of glucose, lactate | ||
| Insulin signaling | Ins mRNA | |||
| AA/PL metab. | Level of Gln, Glu, GABA, PEA | |||
| Oxidative stress | Anti-oxidant defenses (GSH, As) | |||
| Neuronal func. | Expression of VGLUT1/2, VGAT | Level of NAA, expression of PSD95, SYP, SYN | ||
| Neuroinflam. | Level of TNFα, IL1β, IL6, astrogliosis | Microgliosis | ||
5.3.2. Mitochondria-Related Functions and Oxidative Stress
5.3.3. Neuroplasticity and Neuroinflammation
5.4. Impact of a High-Fat Diet Treatment on the VTA-NAc DA System
5.4.1. Impact of a High-Fat Diet Treatment
5.4.2. Combined Effecs of a High-Fat Diet Treatment with Psychostimulant Drugs
5.5. Conclusions
6. Combined Impact of High-Fat Diet and Stress Exposure in Rodents
7. Discussion
7.1. Overview of the Impact of a High-Fat Diet Treatment on Behaviour and Brain in Rodents
7.2. Limitations
7.2.1. Lack of Systematic Assessment of the Metabolic Status
7.2.2. Lack of Evaluation of the Neuroendocrine and Immune Systems
7.2.3. Lack of Studies on the Sensitivity to Drugs of Abuse and Dopaminergic Neurotransmission
7.2.4. Lack of Studies on Sex Differences
7.2.5. Remaining Challenges in Translating Rodent High-Fat Diet Treatments to Human Obesity
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ng, M.; Fleming, T.; Robinson, M.; Thomson, B.; Graetz, N.; Margono, C.; Mullany, E.C.; Biryukov, S.; Abbafati, C.; Abera, S.F.; et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2014, 384, 766–781. [Google Scholar] [CrossRef]
- Drewnowski, A.; Kurth, C.; Holden-Wiltse, J.; Saari, J. Food preferences in human obesity: Carbohydrates versus fats. Appetite 1992, 18, 207–221. [Google Scholar] [CrossRef]
- Hebebrand, J.; Albayrak, Ö.; Adan, R.; Antel, J.; Dieguez, C.; de Jong, J.; Leng, G.; Menzies, J.; Mercer, J.G.; Murphy, M.; et al. “Eating addiction”, rather than “food addiction”, better captures addictive-like eating behavior. Neurosci. Biobehav. Rev. 2014, 47, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Corsica, J.A.; Pelchat, M.L. Food addiction: True or false? Curr. Opin. Gastroenterol. 2010, 26, 165–169. [Google Scholar] [CrossRef]
- Pi-Sunyer, X. The Medical Risks of Obesity. Postgrad. Med. 2009, 121, 21–33. [Google Scholar] [CrossRef]
- Bray, G.A.; Frühbeck, G.; Ryan, D.H.; Wilding, J. Management of obesity. Lancet 2016, 387, 1947–1956. [Google Scholar] [CrossRef]
- Kim, G.W.; Lin, J.E.; Blomain, E.S.; Waldman, S.A. Antiobesity Pharmacotherapy: New Drugs and Emerging Targets. Clin. Pharmacol. Ther. 2014, 95, 53–66. [Google Scholar] [CrossRef]
- GBD 2015 Obesity Collaborators; Afshin, A.; Forouzanfar, M.H.; Reitsma, M.B.; Sur, P.; Estep, K.; Lee, A.; Marczak, L.; Mokdad, A.H.; Moradi-Lakeh, M.; et al. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef]
- Di Angelantonio, E.; Bhupathiraju, S.N.; Wormser, D.; Gao, P.; Kaptoge, S.; de Gonzalez, A.B.; Cairns, B.J.; Huxley, R.; Jackson, C.L.; Joshy, G.; et al. Body-mass index and all-cause mortality: Individual-participant-data meta-analysis of 239 prospective studies in four continents. Lancet 2016, 388, 776–786. [Google Scholar] [CrossRef]
- Citrome, L. Binge eating disorder revisited: What’s new, what’s different, what’s next. CNS Spectr. 2019, 24, 4–13. [Google Scholar] [CrossRef]
- Gariepy, G.; Nitka, D.; Schmitz, N. The association between obesity and anxiety disorders in the population: A systematic review and meta-analysis. Int. J. Obes. 2010, 34, 407–419. [Google Scholar] [CrossRef] [PubMed]
- Petry, N.M.; Barry, D.; Pietrzak, R.H.; Wagner, J.A. Overweight and Obesity are Associated with Psychiatric Disorders: Results From the National Epidemiologic Survey on Alcohol and Related Conditions. Psychosom. Med. 2008, 70, 288–297. [Google Scholar] [CrossRef]
- Preiss, K.; Brennan, L.; Clarke, D. A systematic review of variables associated with the relationship between obesity and depression. Obes. Rev. 2013, 14, 906–918. [Google Scholar] [CrossRef] [PubMed]
- Simon, G.E.; Von Korff, M.; Saunders, K.; Miglioretti, D.L.; Crane, P.; Van Belle, G.; Kessler, R.C. Association Between Obesity and Psychiatric Disorders in the US Adult Population. Arch. Gen. Psychiatry 2006, 63, 824–830. [Google Scholar] [CrossRef] [PubMed]
- Jane-Llopis, E.; Matytsina, I. Mental health and alcohol, drugs and tobacco: A review of the comorbidity between mental disorders and the use of alcohol, tobacco and illicit drugs. Drug Alcohol Rev. 2006, 25, 515–536. [Google Scholar] [CrossRef] [PubMed]
- Onaemo, V.N.; Fawehinmi, T.O.; D’Arcy, C. Comorbid Cannabis Use Disorder with Major Depression and Generalized Anxiety Disorder: A Systematic Review with Meta-analysis of Nationally Representative Epidemiological Surveys. J. Affect. Disord. 2021, 281, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Rappeneau, V.; Bérod, A. Reconsidering depression as a risk factor for substance use disorder: Insights from rodent models. Neurosci. Biobehav. Rev. 2017, 77, 303–316. [Google Scholar] [CrossRef]
- Rapaka, R.; Schnur, P.; Shurtleff, D. Obesity and addiction: Common neurological mechanisms and drug development. Physiol. Behav. 2008, 95, 2–9. [Google Scholar] [CrossRef]
- Volkow, N.D.; Wang, G.-J.; Tomasi, D.; Baler, R.D. Obesity and addiction: Neurobiological overlaps. Obes. Rev. 2013, 14, 2–18. [Google Scholar] [CrossRef]
- Miñarro, J.; Rodríguez-Arias, M. Common Neural Mechanisms of Palatable Food Intake and Drug Abuse: Knowledge Obtained with Animal Models. Curr. Pharm. Des. 2020, 26, 2372–2384. [Google Scholar] [CrossRef]
- DiLeone, R.J.; Taylor, J.R.; Picciotto, M.R. The drive to eat: Comparisons and distinctions between mechanisms of food reward and drug addiction. Nat. Neurosci. 2012, 15, 1330–1335. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Wang, G.J.; Fowler, J.S.; Tomasi, D.; Baler, R. Food and Drug Reward: Overlapping Circuits in Human Obesity and Addiction. Brain Imaging Behav. Neurosci. 2012, 11, 1–24. [Google Scholar] [CrossRef]
- McTeague, L.M.; Rosenberg, B.M.; Lopez, J.W.; Carreon, D.M.; Huemer, J.; Jiang, Y.; Chick, C.F.; Eickhoff, S.B.; Etkin, A. Identification of Common Neural Circuit Disruptions in Emotional Processing Across Psychiatric Disorders. Am. J. Psychiatry 2020, 177, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, P.; Yang, Y. Neural Circuit Mechanisms Underlying Emotional Regulation of Homeostatic Feeding. Trends Endocrinol. Metab. 2017, 28, 437–448. [Google Scholar] [CrossRef] [PubMed]
- Trinko, R.; Sears, R.M.; Guarnieri, D.J.; DiLeone, R.J. Neural mechanisms underlying obesity and drug addiction. Physiol. Behav. 2007, 91, 499–505. [Google Scholar] [CrossRef]
- Hyman, S.E.; Malenka, R.C.; Nestler, E.J. Neural mechanisms of addiction: The Role of Reward-Related Learning and Memory. Annu. Rev. Neurosci. 2006, 29, 565–598. [Google Scholar] [CrossRef]
- Nestler, E.J. The Neurobiology of Cocaine Addiction. Sci. Pr. Perspect. 2005, 3, 4–10. [Google Scholar] [CrossRef]
- Macedo, I.C.; de Freitas, J.S.; Torres, I.L.D.S. The Influence of Palatable Diets in Reward System Activation: A Mini Review. Adv. Pharmacol. Sci. 2016, 2016, 7238679. [Google Scholar] [CrossRef]
- Carr, K.D. Augmentation of drug reward by chronic food restriction: Behavioral evidence and underlying mechanisms. Physiol. Behav. 2002, 76, 353–364. [Google Scholar] [CrossRef]
- Deroche-Gamonet, V.; Piazza, P.V.; Casolini, P.; Le Moal, M.; Simon, H. Sensitization to the psychomotor effects of amphetamine and morphine induced by food restriction depends on corticosterone secretion. Brain Res. 1993, 611, 352–356. [Google Scholar] [CrossRef]
- Stamp, J.A.; Mashoodh, R.; van Kampen, J.M.; Robertson, H.A. Food restriction enhances peak corticosterone levels, cocaine-induced locomotor activity, and ΔFosB expression in the nucleus accumbens of the rat. Brain Res. 2008, 1204, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Sulzer, D.; Cragg, S.J.; Rice, M.E. Striatal dopamine neurotransmission: Regulation of release and uptake. Basal Ganglia 2016, 6, 123–148. [Google Scholar] [CrossRef] [PubMed]
- Wanat, M.J.; Willuhn, I.; Clark, J.J.; Phillips, P. Phasic Dopamine Release in Appetitive Behaviors and Drug Addiction. Curr. Drug Abus. Rev. 2009, 2, 195–213. [Google Scholar] [CrossRef]
- Wang, G.-J.; Volkow, N.D.; Logan, J.; Pappas, N.R.; Wong, C.T.; Zhu, W.; Netusll, N.; Fowler, J.S. Brain dopamine and obesity. Lancet 2001, 357, 354–357. [Google Scholar] [CrossRef]
- Volkow, N.D.; Wang, G.-J.; Telang, F.; Fowler, J.S.; Thanos, P.K.; Logan, J.; Alexoff, D.; Ding, Y.-S.; Wong, C.; Ma, Y.; et al. Low dopamine striatal D2 receptors are associated with prefrontal metabolism in obese subjects: Possible contributing factors. NeuroImage 2008, 42, 1537–1543. [Google Scholar] [CrossRef]
- Volkow, N.D.; Wang, G.-J.; Fowler, J.S.; Telang, F. Overlapping neuronal circuits in addiction and obesity: Evidence of systems pathology. Philos. Trans. R. Soc. B Biol. Sci. 2008, 363, 3191–3200. [Google Scholar] [CrossRef] [PubMed]
- Stice, E.; Spoor, S.; Bohon, C.; Veldhuizen, M.G.; Small, D.M. Relation of reward from food intake and anticipated food intake to obesity: A functional magnetic resonance imaging study. J. Abnorm. Psychol. 2008, 117, 924–935. [Google Scholar] [CrossRef]
- Nummenmaa, L.; Hirvonen, J.; Hannukainen, J.; Immonen, H.; Lindroos, M.M.; Salminen, P.; Nuutila, P. Dorsal Striatum and Its Limbic Connectivity Mediate Abnormal Anticipatory Reward Processing in Obesity. PLoS ONE 2012, 7, e31089. [Google Scholar] [CrossRef]
- Stice, E.; Figlewicz, D.P.; Gosnell, B.A.; Levine, A.S.; Pratt, W.E. The contribution of brain reward circuits to the obesity epidemic. Neurosci. Biobehav. Rev. 2013, 37, 2047–2058. [Google Scholar] [CrossRef]
- Colon-Perez, L.; Montesinos, J.; Monsivais, M. The future of neuroimaging and gut-brain axis research for substance use disorders. Brain Res. 2022, 1781, 147835. [Google Scholar] [CrossRef]
- Gupta, A.; Osadchiy, V.; Mayer, E.A. Brain–gut–microbiome interactions in obesity and food addiction. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 655–672. [Google Scholar] [CrossRef] [PubMed]
- de Araujo, I.E.; Schatzker, M.; Small, D.M. Rethinking Food Reward. Annu. Rev. Psychol. 2020, 71, 139–164. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, C.J.; Haberman, E.R.; Domingos, A.I. A Tale of Three Systems: Toward a Neuroimmunoendocrine Model of Obesity. Annu. Rev. Cell Dev. Biol. 2021, 37, 549–573. [Google Scholar] [CrossRef]
- Tobore, T.O. Towards a comprehensive theory of obesity and a healthy diet: The causal role of oxidative stress in food addiction and obesity. Behav. Brain Res. 2020, 384, 112560. [Google Scholar] [CrossRef] [PubMed]
- Bournat, J.C.; Brown, C.W. Mitochondrial dysfunction in obesity. Curr. Opin. Endocrinol. Diabetes Obes. 2010, 17, 446–452. [Google Scholar] [CrossRef]
- Fernández-Sánchez, A.; Madrigal-Santillán, E.; Bautista, M.; Esquivel-Soto, J.; Morales-González, Á.; Esquivel-Chirino, C.; Durante-Montiel, I.; Sánchez-Rivera, G.; Valadez-Vega, C.; Morales-González, J.A. Inflammation, Oxidative Stress, and Obesity. Int. J. Mol. Sci. 2011, 12, 3117–3132. [Google Scholar] [CrossRef]
- Furukawa, S.; Fujita, T.; Shimabukuro, M.; Iwaki, M.; Yamada, Y.; Nakajima, Y.; Nakayama, O.; Makishima, M.; Matsuda, M.; Shimomura, I. Increased oxidative stress in obesity and its impact on metabolic syndrome. J. Clin. Investig. 2004, 114, 1752–1761. [Google Scholar] [CrossRef]
- Savini, I.; Gasperi, V.; Catani, M.V. Oxidative Stress and Obesity. In Obesity: A Practical Guide; Springer International Publishing: Cham, Switzerland, 2016; pp. 65–86. [Google Scholar]
- Beiser, T.; Yaka, R. The Role of Oxidative Stress in Cocaine Addiction. J. Neurol. Neuromed. 2019, 4, 17–21. [Google Scholar] [CrossRef]
- Jîtcă, G.; Ősz, B.; Tero-Vescan, A.; Vari, C. Psychoactive Drugs—From Chemical Structure to Oxidative Stress Related to Dopaminergic Neurotransmission. A Review. Antioxidants 2020, 10, 381. [Google Scholar] [CrossRef]
- Olguín, H.J.; Guzmán, D.C.; García, E.H.; Mejía, G.B. The Role of Dopamine and Its Dysfunction as a Consequence of Oxidative Stress. Oxidative Med. Cell. Longev. 2016, 2016, 9730467. [Google Scholar] [CrossRef]
- Kovacic, P. Unifying mechanism for addiction and toxicity of abused drugs with application to dopamine and glutamate mediators: Electron transfer and reactive oxygen species. Med. Hypotheses 2005, 65, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Thornton, C.; Grad, E.; Yaka, R. The role of mitochondria in cocaine addiction. Biochem. J. 2021, 478, 749–764. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Xu, X.; Yan, Y. Estimated global overweight and obesity burden in pregnant women based on panel data model. PLoS ONE 2018, 13, e0202183. [Google Scholar] [CrossRef]
- Gawlińska, K.; Gawliński, D.; Filip, M.; Przegaliński, E. Relationship of maternal high-fat diet during pregnancy and lactation to offspring health. Nutr. Rev. 2020, 79, 709–725. [Google Scholar] [CrossRef] [PubMed]
- Beam, A.; Clinger, E.; Hao, L. Effect of Diet and Dietary Components on the Composition of the Gut Microbiota. Nutrients 2021, 13, 2795. [Google Scholar] [CrossRef] [PubMed]
- Leigh, S.-J.; Morris, M.J. Diet, inflammation and the gut microbiome: Mechanisms for obesity-associated cognitive impairment. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2020, 1866, 165767. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Zorita, S.; Aguirre, L.; Milton-Laskibar, I.; Fernández-Quintela, A.; Trepiana, J.; Kajarabille, N.; Mosqueda-Solís, A.; González, M.; Portillo, M.P. Relationship between Changes in Microbiota and Liver Steatosis Induced by High-Fat Feeding—A Review of Rodent Models. Nutrients 2019, 11, 2156. [Google Scholar] [CrossRef]
- Mokkala, K.; Houttu, N.; Cansev, T.; Laitinen, K. Interactions of dietary fat with the gut microbiota: Evaluation of mechanisms and metabolic consequences. Clin. Nutr. 2020, 39, 994–1018. [Google Scholar] [CrossRef]
- Ingle, D.J. A Simple Means of Producing Obesity in the Rat. Exp. Biol. Med. 1949, 72, 604–605. [Google Scholar] [CrossRef]
- Buettner, R.; Schölmerich, J.; Bollheimer, L.C. High-fat Diets: Modeling the Metabolic Disorders of Human Obesity in Rodents. Obesity 2007, 15, 798–808. [Google Scholar] [CrossRef]
- Hariri, N.; Thibault, L. High-fat diet-induced obesity in animal models. Nutr. Res. Rev. 2010, 23, 270–299. [Google Scholar] [CrossRef] [PubMed]
- Pandit, R.; Mercer, J.G.; Overduin, J.; la Fleur, S.E.; Adan, R.A. Dietary Factors Affect Food Reward and Motivation to Eat. Obes. Facts 2012, 5, 221–242. [Google Scholar] [CrossRef] [PubMed]
- Preguiça, I.; Alves, A.; Nunes, S.; Fernandes, R.; Gomes, P.; Viana, S.D.; Reis, F. Diet-induced rodent models of obesity-related metabolic disorders—A guide to a translational perspective. Obes. Rev. 2020, 21, e13081. [Google Scholar] [CrossRef] [PubMed]
- Rosini, T.C.; da Silva, A.S.R.; de Moraes, C. Diet-Induced Obesity: Rodent Model for the Study of Obesity-Related Disorders. Rev. Assoc. Med. Bras. 2012, 58, 383–387. [Google Scholar]
- Abbott, K.N.; Arnott, C.K.; Westbrook, R.F.; Tran, D.M. The effect of high fat, high sugar, and combined high fat-high sugar diets on spatial learning and memory in rodents: A meta-analysis. Neurosci. Biobehav. Rev. 2019, 107, 399–421. [Google Scholar] [CrossRef]
- Cordner, Z.A.; Tamashiro, K.L. Effects of high-fat diet exposure on learning & memory. Physiol. Behav. 2015, 152, 363–371. [Google Scholar] [CrossRef]
- Freeman, L.R.; Haley-Zitlin, V.; Rosenberger, D.S.; Granholm, A.-C. Damaging effects of a high-fat diet to the brain and cognition: A review of proposed mechanisms. Nutr. Neurosci. 2014, 17, 241–251. [Google Scholar] [CrossRef]
- Liang, Y.; Zou, L.; Tian, Y.; Zhou, S.; Chen, X.; Lin, C. Dietary and metabolic risk of neuropsychiatric disorders: Insights from animal models. Br. J. Nutr. 2021, 126, 1771–1787. [Google Scholar] [CrossRef]
- Morris, M.J.; Beilharz, J.E.; Maniam, J.; Reichelt, A.C.; Westbrook, R.F. Why is obesity such a problem in the 21st century? The intersection of palatable food, cues and reward pathways, stress, and cognition. Neurosci. Biobehav. Rev. 2015, 58, 36–45. [Google Scholar] [CrossRef]
- Murphy, M.; Mercer, J.G. Diet-Regulated Anxiety. Int. J. Endocrinol. 2013, 2013, 701967. [Google Scholar] [CrossRef]
- Sarangi, M.; Dus, M. Crème de la Créature: Dietary Influences on Behavior in Animal Models. Front. Behav. Neurosci. 2021, 15. [Google Scholar] [CrossRef]
- Archer, J. Tests for emotionality in rats and mice: A review. Anim. Behav. 1973, 21, 205–235. [Google Scholar] [CrossRef]
- Speakman, J.R. Use of high-fat diets to study rodent obesity as a model of human obesity. Int. J. Obes. 2019, 43, 1491–1492. [Google Scholar] [CrossRef] [PubMed]
- Fats and Fatty Acids in Human Nutrition. Report of an Expert Consultation. FAO Food Nutr. Pap. 2010, 91, 1–166. [Google Scholar]
- Trumbo, P.; Schlicker, S.; Yates, A.A.; Poos, M.; Food and Nutrition Board of the Institute of Medicine, The National Academies. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein and Amino Acids. J. Am. Diet. Assoc. 2002, 102, 1621–1630. [Google Scholar] [CrossRef]
- Ritchie, H.; Roser, M. Diet Compositions. Available online: https://ourworldindata.org/diet-compositions?fbclid=IwAR3k-_TpvIw2zZUdZiBB3xZJHsgjfJTM0rtIrptoMSwxcTNtoajIaMnJkmw (accessed on 15 July 2022).
- Eilander, A.; Harika, R.K.; Zock, P.L. Intake and sources of dietary fatty acids in Europe: Are current population intakes of fats aligned with dietary recommendations? Eur. J. Lipid Sci. Technol. 2015, 117, 1370–1377. [Google Scholar] [CrossRef] [PubMed]
- Bellisario, V.; Panetta, P.; Balsevich, G.; Baumann, V.; Noble, J.; Raggi, C.; Nathan, O.; Berry, A.; Seckl, J.; Schmidt, M.; et al. Maternal high-fat diet acts as a stressor increasing maternal glucocorticoids’ signaling to the fetus and disrupting maternal behavior and brain activation in C57BL/6J mice. Psychoneuroendocrinology 2015, 60, 138–150. [Google Scholar] [CrossRef] [PubMed]
- Baptissart, M.; Lamb, H.E.; To, K.; Bradish, C.; Tehrani, J.; Reif, D.; Cowley, M. Neonatal mice exposed to a high-fat diet in utero influence the behaviour of their nursing dam. Proc. R. Soc. B Boil. Sci. 2018, 285, 20181237. [Google Scholar] [CrossRef]
- Connor, K.L.; Vickers, M.H.; Beltrand, J.; Meaney, M.J.; Sloboda, D.M. Nature, nurture or nutrition? Impact of maternal nutrition on maternal care, offspring development and reproductive function. J. Physiol. 2012, 590, 2167–2180. [Google Scholar] [CrossRef]
- Leuthardt, A.S.; Bayer, J.; Rodríguez, J.M.M.; Boyle, C.N. Influence of High Energy Diet and Polygenic Predisposition for Obesity on Postpartum Health in Rat Dams. Front. Physiol. 2021, 12, 772707. [Google Scholar] [CrossRef]
- Rodriguez, J.; Rodríguez-González, G.; Reyes-Castro, L.; Ibáñez, C.; Ramírez, A.; Chavira, R.; Larrea, F.; Nathanielsz, P.; Zambrano, E. Maternal obesity in the rat programs male offspring exploratory, learning and motivation behavior: Prevention by dietary intervention pre-gestation or in gestation. Int. J. Dev. Neurosci. 2012, 30, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Paradis, J.; Boureau, P.; Moyon, T.; Nicklaus, S.; Parnet, P.; Paillé, V. Perinatal Western Diet Consumption Leads to Profound Plasticity and GABAergic Phenotype Changes within Hypothalamus and Reward Pathway from Birth to Sexual Maturity in Rat. Front. Endocrinol. 2017, 8, 216. [Google Scholar] [CrossRef]
- Cunha, F.D.S.; Molle, R.D.; Portella, A.K.; Benetti, C.D.S.; Noschang, C.; Goldani, M.Z.; Silveira, P.P. Both Food Restriction and High-Fat Diet during Gestation Induce Low Birth Weight and Altered Physical Activity in Adult Rat Offspring: The “Similarities in the Inequalities” Model. PLoS ONE 2015, 10, e0118586. [Google Scholar] [CrossRef] [PubMed]
- Gawlińska, K.; Gawliński, D.; Korostyński, M.; Borczyk, M.; Frankowska, M.; Piechota, M.; Filip, M.; Przegaliński, E. Maternal dietary patterns are associated with susceptibility to a depressive-like phenotype in rat offspring. Dev. Cogn. Neurosci. 2021, 47, 100879. [Google Scholar] [CrossRef] [PubMed]
- Naef, L.; Srivastava, L.; Gratton, A.; Hendrickson, H.; Owens, S.M.; Walker, C.-D. Maternal high fat diet during the perinatal period alters mesocorticolimbic dopamine in the adult rat offspring: Reduction in the behavioral responses to repeated amphetamine administration. Psychopharmacology 2008, 197, 83–94. [Google Scholar] [CrossRef]
- Gawliński, D.; Gawlińska, K.; Frankowska, M.; Filip, M. Maternal Diet Influences the Reinstatement of Cocaine-Seeking Behavior and the Expression of Melanocortin-4 Receptors in Female Offspring of Rats. Nutrients 2020, 12, 1462. [Google Scholar] [CrossRef]
- Lépinay, A.L.; Larrieu, T.; Joffre, C.; Acar, N.; Gárate, I.; Castanon, N.; Ferreira, G.; Langelier, B.; Guesnet, P.; Brétillon, L.; et al. Perinatal high-fat diet increases hippocampal vulnerability to the adverse effects of subsequent high-fat feeding. Psychoneuroendocrinology 2015, 53, 82–93. [Google Scholar] [CrossRef]
- Niu, X.; Wu, X.; Ying, A.; Shao, B.; Li, X.; Zhang, W.; Lin, C.; Lin, Y. Maternal high fat diet programs hypothalamic-pituitary-adrenal function in adult rat offspring. Psychoneuroendocrinology 2019, 102, 128–138. [Google Scholar] [CrossRef]
- Schneider, M. Adolescence as a vulnerable period to alter rodent behavior. Cell Tissue Res. 2013, 354, 99–106. [Google Scholar] [CrossRef]
- Aslani, S.; Vieira, N.; Marques, F.; Costa, P.; Sousa, N.; Palha, J.A. The effect of high-fat diet on rat’s mood, feeding behavior and response to stress. Transl. Psychiatry 2015, 5, e684. [Google Scholar] [CrossRef]
- Blanco-Gandía, M.C.; Miñarro, J.; Rodríguez-Arias, M. Behavioral profile of intermittent vs. continuous access to a high fat diet during adolescence. Behav. Brain Res. 2019, 368, 111891. [Google Scholar] [CrossRef] [PubMed]
- Busquets, O.; Ettcheto, M.; Eritja, À.; Espinosa-Jiménez, T.; Verdaguer, E.; Olloquequi, J.; Beas-Zarate, C.; Castro-Torres, R.D.; Casadesús, G.; Auladell, C.; et al. c-Jun N-terminal Kinase 1 ablation protects against metabolic-induced hippocampal cognitive impairments. J. Mol. Med. 2019, 97, 1723–1733. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Morales, L.; Ruiz-Gayo, M.; Del Olmo, N. Effect of high-fat diets on mood and learning performance in adolescent mice. Behav. Brain Res. 2016, 311, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Valladolid-Acebes, I.; Stucchi, P.; Cano, V.; Fernández-Alfonso, M.; Merino, B.; Gil-Ortega, M.; Fole, A.; Morales, L.; Ruiz-Gayo, M.; Del Olmo, N. High-fat diets impair spatial learning in the radial-arm maze in mice. Neurobiol. Learn. Mem. 2011, 95, 80–85. [Google Scholar] [CrossRef]
- Isgor, C.; Kabbaj, M.; Akil, H.; Watson, S.J. Delayed effects of chronic variable stress during peripubertal-juvenile period on hippocampal morphology and on cognitive and stress axis functions in rats. Hippocampus 2004, 14, 636–648. [Google Scholar] [CrossRef]
- Tzanoulinou, S.; Gantelet, E.; Sandi, C.; Márquez, C. Programming effects of peripubertal stress on spatial learning. Neurobiol. Stress 2020, 13, 100282. [Google Scholar] [CrossRef]
- Blanco-Gandia, M.; Aracil-Fernández, A.; Romero, S.M.; Aguilar, M.; Manzanares, J.; Miñarro, J.; Rodríguez-Arias, M. Changes in gene expression and sensitivity of cocaine reward produced by a continuous fat diet. Psychopharmacology 2017, 234, 2337–2352. [Google Scholar] [CrossRef]
- de Noronha, S.R.; Campos, G.V.; Abreu, A.R.; de Souza, A.A.; Chianca, D.A.; de Menezes, R.C. High fat diet induced-obesity facilitates anxiety-like behaviors due to GABAergic impairment within the dorsomedial hypothalamus in rats. Behav. Brain Res. 2017, 316, 38–46. [Google Scholar] [CrossRef]
- Del Rosario, A.; McDermott, M.M.; Panee, J. Effects of a high-fat diet and bamboo extract supplement on anxiety- and depression-like neurobehaviours in mice. Br. J. Nutr. 2012, 108, 1143–1149. [Google Scholar] [CrossRef]
- Finger, B.; Dinan, T.; Cryan, J. High-fat diet selectively protects against the effects of chronic social stress in the mouse. Neuroscience 2011, 192, 351–360. [Google Scholar] [CrossRef]
- Noronha, S.; Lima, P.; Campos, G.; Chírico, M.; Abreu, A.; Figueiredo, A.; Silva, F.; Chianca, D.; Lowry, C.; De Menezes, R. Association of high-fat diet with neuroinflammation, anxiety-like defensive behavioral responses, and altered thermoregulatory responses in male rats. Brain Behav. Immun. 2019, 80, 500–511. [Google Scholar] [CrossRef] [PubMed]
- White, K.A.; Hutton, S.R.; Weimer, J.M.; Sheridan, P.A. Diet-induced obesity prolongs neuroinflammation and recruits CCR2 + monocytes to the brain following herpes simplex virus (HSV)-1 latency in mice. Brain Behav. Immun. 2016, 57, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Głombik, K.; Detka, J.; Góralska, J.; Kurek, A.; Solnica, B.; Budziszewska, B. Brain Metabolic Alterations in Rats Showing Depression-Like and Obesity Phenotypes. Neurotox. Res. 2020, 37, 406–424. [Google Scholar] [CrossRef] [PubMed]
- Loebens, M.; Barros, H. Diet influences cocaine withdrawal behaviors in the forced swimming test. Pharmacol. Biochem. Behav. 2003, 74, 259–267. [Google Scholar] [CrossRef]
- Blanco-Gandia, M.; Cantacorps, L.; Aracil-Fernández, A.; Romero, S.M.; Aguilar, M.; Manzanares, J.; Valverde, O.; Miñarro, J.; Rodríguez-Arias, M. Effects of bingeing on fat during adolescence on the reinforcing effects of cocaine in adult male mice. Neuropharmacology 2017, 113, 31–44. [Google Scholar] [CrossRef]
- Hryhorczuk, C.; Florea, M.; Rodaros, D.; Poirier, I.; Daneault, C.; Rosiers, C.D.; Arvanitogiannis, A.; Alquier, T.; Fulton, S. Dampened Mesolimbic Dopamine Function and Signaling by Saturated but not Monounsaturated Dietary Lipids. Neuropsychopharmacology 2016, 41, 811–821. [Google Scholar] [CrossRef]
- Morales, L.; Del Olmo, N.; Valladolid-Acebes, I.; Fole, A.; Cano, V.; Merino, B.; Stucchi, P.; Ruggieri, D.; López, L.; Alguacil, L.F.; et al. Shift of Circadian Feeding Pattern by High-Fat Diets Is Coincident with Reward Deficits in Obese Mice. PLoS ONE 2012, 7, e36139. [Google Scholar] [CrossRef]
- Ródenas-González, F.; Blanco-Gandía, M.D.C.; Pascual, M.; Molari, I.; Guerri, C.; López, J.M.; Rodríguez-Arias, M. A limited and intermittent access to a high-fat diet modulates the effects of cocaine-induced reinstatement in the conditioned place preference in male and female mice. Psychopharmacology 2021, 238, 2091–2103. [Google Scholar] [CrossRef]
- Blanco-Gandia, M.C.; Montagud-Romero, S.; Rodríguez-Arias, M. Binge eating and psychostimulant addiction. World J. Psychiatry 2021, 11, 517–529. [Google Scholar] [CrossRef]
- Blanco-Gandía, M.C.; Montagud-Romero, S.; Aguilar, M.A.; Miñarro, J.; Rodríguez-Arias, M. Housing conditions modulate the reinforcing properties of cocaine in adolescent mice that binge on fat. Physiol. Behav. 2018, 183, 18–26. [Google Scholar] [CrossRef]
- Naneix, F.; Tantot, F.; Glangetas, C.; Kaufling, J.; Janthakhin, Y.; Boitard, C.; De Smedt-Peyrusse, V.; Pape, J.R.; Vancassel, S.; Trifilieff, P.; et al. Impact of Early Consumption of High-Fat Diet on the Mesolimbic Dopaminergic System. ENeuro 2017, 4, 4. [Google Scholar] [CrossRef] [PubMed]
- Ganji, A.; Salehi, I.; Sarihi, A.; Shahidi, S.; Komaki, A. Effects of Hypericum Scabrum extract on anxiety and oxidative stress biomarkers in rats fed a long-term high-fat diet. Metab. Brain Dis. 2017, 32, 503–511. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, L.M.; Gonçalves, J.L.; Pedersen, A.S.B.; Damasceno, S.; Júnior, R.E.M.; Maioli, T.U.; de Faria, A.M.C.; Godard, A.L.B. High-fat diet withdrawal modifies alcohol preference and transcription of dopaminergic and GABAergic receptors. J. Neurogenet. 2019, 33, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Takase, K.; Tsuneoka, Y.; Oda, S.; Kuroda, M.; Funato, H. High-fat diet feeding alters olfactory-, social-, and reward-related behaviors of mice independent of obesity. Obesity 2016, 24, 886–894. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Lv, W.; Pan, Q.; Kalavagunta, P.K.; Liu, Q.; Qin, G.; Cai, M.; Zhou, L.; Wang, T.; Xia, Z.; et al. Simvastatin therapy in adolescent mice attenuates HFD-induced depression-like behavior by reducing hippocampal neuroinflammation. J. Affect. Disord. 2019, 243, 83–95. [Google Scholar] [CrossRef] [PubMed]
- Zemdegs, J.; Quesseveur, G.; Jarriault, D.; Pénicaud, L.; Fioramonti, X.; Guiard, B.P. High-fat diet-induced metabolic disorders impairs 5-HT function and anxiety-like behavior in mice. J. Cereb. Blood Flow Metab. 2016, 173, 2095–2110. [Google Scholar] [CrossRef]
- Arnold, S.E.; Lucki, I.; Brookshire, B.R.; Carlson, G.C.; Browne, C.A.; Kazi, H.; Bang, S.; Choi, B.-R.; Chen, Y.; McMullen, M.F.; et al. High fat diet produces brain insulin resistance, synaptodendritic abnormalities and altered behavior in mice. Neurobiol. Dis. 2014, 67, 79–87. [Google Scholar] [CrossRef]
- Finger, B.C.; Dinan, T.G.; Cryan, J.F. The temporal impact of chronic intermittent psychosocial stress on high-fat diet-induced alterations in body weight. Psychoneuroendocrinology 2012, 37, 729–741. [Google Scholar] [CrossRef]
- Li, L.; Wang, Z.; Zuo, Z. Chronic Intermittent Fasting Improves Cognitive Functions and Brain Structures in Mice. PLoS ONE 2013, 8, e66069. [Google Scholar] [CrossRef]
- Mielke, J.G.; Nicolitch, K.; Avellaneda, V.; Earlam, K.; Ahuja, T.; Mealing, G.; Messier, C. Longitudinal study of the effects of a high-fat diet on glucose regulation, hippocampal function, and cerebral insulin sensitivity in C57BL/6 mice. Behav. Brain Res. 2006, 175, 374–382. [Google Scholar] [CrossRef]
- Novick, A.M.; Levandowski, M.L.; Laumann, L.E.; Philip, N.S.; Price, L.H.; Tyrka, A.R. The effects of early life stress on reward processing. J. Psychiatr. Res. 2018, 101, 80–103. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, A.; Notaras, M.; Du, X.; Hill, R.A. On the Developmental Timing of Stress: Delineating Sex-Specific Effects of Stress across Development on Adult Behavior. Brain Sci. 2018, 8, 121. [Google Scholar] [CrossRef] [PubMed]
- Hersey, M.; Woodruff, J.L.; Maxwell, N.; Sadek, A.T.; Bykalo, M.K.; Bain, I.; Grillo, C.A.; Piroli, G.G.; Hashemi, P.; Reagan, L.P. High-fat diet induces neuroinflammation and reduces the serotonergic response to escitalopram in the hippocampus of obese rats. Brain Behav. Immun. 2021, 96, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Duffy, C.; Hofmeister, J.; Nixon, J.; Butterick, T. High fat diet increases cognitive decline and neuroinflammation in a model of orexin loss. Neurobiol. Learn. Mem. 2019, 157, 41–47. [Google Scholar] [CrossRef]
- Lizarbe, B.; Soares, A.F.; Larsson, S.; Duarte, J.M.N.; Lizarbe, B.; Soares, A.F.; Larsson, S.; Duarte, J.M.N. Neurochemical Modifications in the Hippocampus, Cortex and Hypothalamus of Mice Exposed to Long-Term High-Fat Diet. Front. Neurosci. 2018, 12, 985. [Google Scholar] [CrossRef]
- Erhardt, E.; Zibetti, L.; Godinho, J.; Bacchieri, B.; Barros, H. Behavioral changes induced by cocaine in mice are modified by a hyperlipidic diet or recombinant leptin. Braz. J. Med Biol. Res. 2006, 39, 1625–1635. [Google Scholar] [CrossRef]
- Orsini, C.A.; Ginton, G.; Shimp, K.G.; Avena, N.M.; Gold, M.S.; Setlow, B. Food consumption and weight gain after cessation of chronic amphetamine administration. Appetite 2014, 78, 76–80. [Google Scholar] [CrossRef]
- Casey, B.; Jones, R.M.; Levita, L.; Libby, V.; Pattwell, S.S.; Ruberry, E.J.; Soliman, F.; Somerville, L.H. The storm and stress of adolescence: Insights from human imaging and mouse genetics. Dev. Psychobiol. 2010, 52, 225–235. [Google Scholar] [CrossRef]
- Romeo, R.D.; Patel, R.; Pham, L.; So, V.M. Adolescence and the ontogeny of the hormonal stress response in male and female rats and mice. Neurosci. Biobehav. Rev. 2016, 70, 206–216. [Google Scholar] [CrossRef]
- Spear, L. The adolescent brain and age-related behavioral manifestations. Neurosci. Biobehav. Rev. 2000, 24, 417–463. [Google Scholar] [CrossRef]
- Brydges, N.M. Pre-pubertal stress and brain development in rodents. Curr. Opin. Behav. Sci. 2016, 7, 8–14. [Google Scholar] [CrossRef]
- Burke, A.R.; McCormick, C.M.; Pellis, S.M.; Lukkes, J.L. Impact of adolescent social experiences on behavior and neural circuits implicated in mental illnesses. Neurosci. Biobehav. Rev. 2017, 76, 280–300. [Google Scholar] [CrossRef] [PubMed]
- McCormick, C.; Green, M. From the stressed adolescent to the anxious and depressed adult: Investigations in rodent models. Neuroscience 2013, 249, 242–257. [Google Scholar] [CrossRef] [PubMed]
- Green, M.R.; McCormick, C.M. Effects of stressors in adolescence on learning and memory in rodent models. Horm. Behav. 2013, 64, 364–379. [Google Scholar] [CrossRef] [PubMed]
- McCormick, C.M.; Mathews, I.Z.; Thomas, C.; Waters, P. Investigations of HPA function and the enduring consequences of stressors in adolescence in animal models. Brain Cogn. 2010, 72, 73–85. [Google Scholar] [CrossRef]
- Eiland, L.; Romeo, R. Stress and the developing adolescent brain. Neuroscience 2013, 249, 162–171. [Google Scholar] [CrossRef]
- Romeo, R.D. The impact of stress on the structure of the adolescent brain: Implications for adolescent mental health. Brain Res. 2017, 1654, 185–191. [Google Scholar] [CrossRef]
- Tottenham, N.; Galván, A. Stress and the Adolescent Brain: Amygdala-Prefrontal Cortex Circuitry and Ventral Striatum as Developmental Targets. Neurosci. Biobehav. Rev. 2016, 70, 217–227. [Google Scholar] [CrossRef]
- Andersen, S.L. Trajectories of brain development: Point of vulnerability or window of opportunity? Neurosci. Biobehav. Rev. 2003, 27, 3–18. [Google Scholar] [CrossRef]
- Paus, T.; Keshavan, M.; Giedd, J.N. Why do many psychiatric disorders emerge during adolescence? Nat. Rev. Neurosci. 2008, 9, 947–957. [Google Scholar] [CrossRef]
- Crews, F.; He, J.; Hodge, C. Adolescent cortical development: A critical period of vulnerability for addiction. Pharmacol. Biochem. Behav. 2007, 86, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Cavaliere, G.; Trinchese, G.; Penna, E.; Cimmino, F.; Pirozzi, C.; Lama, A.; Annunziata, C.; Catapano, A.; Mattace Raso, G.; Meli, R.; et al. High-Fat Diet Induces Neuroinflammation and Mitochondrial Impairment in Mice Cerebral Cortex and Synaptic Fraction. Front. Cell. Neurosci. 2019, 13, 509. [Google Scholar] [CrossRef] [PubMed]
- Janthakhin, Y.; Rincel, M.; Costa, A.M.; Darnaudéry, M.; Ferreira, G. Maternal high-fat diet leads to hippocampal and amygdala dendritic remodeling in adult male offspring. Psychoneuroendocrinology 2017, 83, 49–57. [Google Scholar] [CrossRef]
- De Souza, C.T.; Araujo, E.P.; Bordin, S.; Ashimine, R.; Zollner, R.D.L.; Boschero, A.C.; Saad, M.J.A.; Velloso, L.A. Consumption of a Fat-Rich Diet Activates a Proinflammatory Response and Induces Insulin Resistance in the Hypothalamus. Endocrinology 2005, 146, 4192–4199. [Google Scholar] [CrossRef] [PubMed]
- Terrien, J.; Seugnet, I.; Seffou, B.; Herrero, M.J.; Bowers, J.; Chamas, L.; Decherf, S.; Duvernois-Berthet, E.; Djediat, C.; Ducos, B.; et al. Reduced central and peripheral inflammatory responses and increased mitochondrial activity contribute to diet-induced obesity resistance in WSB/EiJ mice. Sci. Rep. 2019, 9, 19696. [Google Scholar] [CrossRef] [PubMed]
- Toniazzo, A.P.; Arcego, D.M.; Lazzaretti, C.; Mota, C.; Schnorr, C.E.; Pettenuzzo, L.F.; Krolow, R.; Moreira, J.C.F.; Dalmaz, C. Sex-dependent effect on mitochondrial and oxidative stress parameters in the hypothalamus induced by prepubertal stress and access to high fat diet. Neurochem. Int. 2019, 124, 114–122. [Google Scholar] [CrossRef]
- Lorenzo, P.I.; Vazquez, E.M.; López-Noriega, L.; Fuente-Martín, E.; Mellado-Gil, J.M.; Franco, J.M.; Cobo-Vuilleumier, N.; Martínez, J.A.G.; Romero-Zerbo, S.Y.; Perez-Cabello, J.A.; et al. The metabesity factor HMG20A potentiates astrocyte survival and reactive astrogliosis preserving neuronal integrity. Theranostics 2021, 11, 6983–7004. [Google Scholar] [CrossRef]
- Thaler, J.P.; Yi, C.-X.; Schur, E.A.; Guyenet, S.J.; Hwang, B.H.; Dietrich, M.; Zhao, X.; Sarruf, D.A.; Izgur, V.; Maravilla, K.R.; et al. Obesity is associated with hypothalamic injury in rodents and humans. J. Clin. Investig. 2012, 122, 153–162. [Google Scholar] [CrossRef]
- Koob, G.F.; Volkow, N.D. Neurocircuitry of Addiction. Neuropsychopharmacology 2010, 35, 217–238. [Google Scholar] [CrossRef]
- Kalyan-Masih, P.; Vega-Torres, J.D.; Miles, C.; Haddad, E.; Rainsbury, S.; Baghchechi, M.; Obenaus, A.; Figueroa, J.D. Western High-Fat Diet Consumption during Adolescence Increases Susceptibility to Traumatic Stress while Selectively Disrupting Hippocampal and Ventricular Volumes. Eneuro 2016, 3. [Google Scholar] [CrossRef]
- Sial, O.K.; Gnecco, T.; Cardona-Acosta, A.M.; Vieregg, E.; Cardoso, E.A.; Parise, L.F.; Bolaños-Guzmán, C.A. Exposure to Vicarious Social Defeat Stress and Western-Style Diets during Adolescence Leads to Physiological Dysregulation, Decreases in Reward Sensitivity, and Reduced Antidepressant Efficacy in Adulthood. Front. Neurosci. 2021, 15, 701919. [Google Scholar] [CrossRef] [PubMed]
- Coccurello, R.; Romano, A.; Giacovazzo, G.; Tempesta, B.; Fiore, M.; Giudetti, A.M.; Marrocco, I.; Altieri, F.; Moles, A.; Gaetani, S. Increased intake of energy-dense diet and negative energy balance in a mouse model of chronic psychosocial defeat. Eur. J. Nutr. 2018, 57, 1485–1498. [Google Scholar] [CrossRef]
- MacKay, J.; Kent, P.; James, J.; Cayer, C.; Merali, Z. Ability of palatable food consumption to buffer against the short- and long-term behavioral consequences of social defeat exposure during juvenility in rats. Physiol. Behav. 2017, 177, 113–121. [Google Scholar] [CrossRef]
- Lutter, M.; Nestler, E.J. Homeostatic and Hedonic Signals Interact in the Regulation of Food Intake. J. Nutr. 2009, 139, 629–632. [Google Scholar] [CrossRef] [PubMed]
- Eller, O.C.; Morris, E.M.; Thyfault, J.P.; Christianson, J.A. Early life stress reduces voluntary exercise and its prevention of diet-induced obesity and metabolic dysfunction in mice. Physiol. Behav. 2020, 223, 113000. [Google Scholar] [CrossRef] [PubMed]
- Isingrini, E.; Camus, V.; Le Guisquet, A.-M.; Pingaud, M.; Devers, S.; Belzung, C. Association between Repeated Unpredictable Chronic Mild Stress (UCMS) Procedures with a High Fat Diet: A Model of Fluoxetine Resistance in Mice. PLoS ONE 2010, 5, e10404. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, R.S.; Park, K.Y.; Law, C.W.Y.; Sultan, F.; Adams, A.; Lourenço, M.T.; Lo, A.K.S.; Soczynska, J.K.; Woldeyohannes, H.; Alsuwaidan, M.; et al. The Association between Conventional Antidepressants and the Metabolic Syndrome: A Review of the Evidence and Clinical Implications. CNS Drugs 2010, 24, 741–753. [Google Scholar] [CrossRef]
- Woo, Y.S.; Seo, H.-J.; McIntyre, R.S.; Bahk, W.-M. Obesity and Its Potential Effects on Antidepressant Treatment Outcomes in Patients with Depressive Disorders: A Literature Review. Int. J. Mol. Sci. 2016, 17, 80. [Google Scholar] [CrossRef]
- Ditzen, C.; Jastorff, A.M.; Kessler, M.S.; Bunck, M.; Teplytska, L.; Erhardt, A.; Krömer, S.A.; Varadarajulu, J.; Targosz, B.-S.; Sayan-Ayata, E.F.; et al. Protein Biomarkers in a Mouse Model of Extremes in Trait Anxiety. Mol. Cell. Proteom. 2006, 5, 1914–1920. [Google Scholar] [CrossRef]
- Kullmann, S.; Kleinridders, A.; Small, D.M.; Fritsche, A.; Häring, H.-U.; Preissl, H.; Heni, M. Central nervous pathways of insulin action in the control of metabolism and food intake. Lancet Diabetes Endocrinol. 2020, 8, 524–534. [Google Scholar] [CrossRef]
- Morrison, C.D. Leptin signaling in brain: A link between nutrition and cognition? Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2009, 1792, 401–408. [Google Scholar] [CrossRef] [PubMed]
- D’Asti, E.; Long, H.; Tremblay-Mercier, J.; Grajzer, M.; Cunnane, S.C.; Di Marzo, V.; Walker, C.-D. Maternal Dietary Fat Determines Metabolic Profile and the Magnitude of Endocannabinoid Inhibition of the Stress Response in Neonatal Rat Offspring. Endocrinology 2010, 151, 1685–1694. [Google Scholar] [CrossRef] [PubMed]
- Schipper, L.; van Heijningen, S.; Karapetsas, G.; van der Beek, E.M.; van Dijk, G. Individual housing of male C57BL/6J mice after weaning impairs growth and predisposes for obesity. PLoS ONE 2020, 15, e0225488. [Google Scholar] [CrossRef]
- Chandra, R.; Engeln, M.; Schiefer, C.; Patton, M.H.; Martin, J.A.; Werner, C.T.; Riggs, L.M.; Francis, T.C.; McGlincy, M.; Evans, B.; et al. Drp1 Mitochondrial Fission in D1 Neurons Mediates Behavioral and Cellular Plasticity during Early Cocaine Abstinence. Neuron 2017, 96, 1327–1341.e6. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yan, G.-Y.; Zhou, J.-Q.; Bu, Q.; Deng, P.-C.; Yang, Y.-Z.; Lv, L.; Deng, Y.; Zhao, J.-X.; Shao, X.; et al. 1H NMR-based metabonomics in brain nucleus accumbens and striatum following repeated cocaine treatment in rats. Neuroscience 2012, 218, 196–205. [Google Scholar] [CrossRef]
- Vitcheva, V.; Simeonova, R.; Kondeva-Burdina, M.; Mitcheva, M. Selective Nitric Oxide Synthase Inhibitor 7-Nitroindazole Protects against Cocaine-Induced Oxidative Stress in Rat Brain. Oxidative Med. Cell. Longev. 2015, 2015, 157876. [Google Scholar] [CrossRef]
- Dietrich, J.-B.; Poirier, R.; Aunis, M.; Zwiller, J. Cocaine Downregulates the Expression of the Mitochondrial Genome in Rat Brain. Ann. N. Y. Acad. Sci. 2004, 1025, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Sadakierska-Chudy, A.; Kotarska, A.; Frankowska, M.; Jastrzębska, J.; Wydra, K.; Miszkiel, J.; Przegaliński, E.; Filip, M. The Alterations in Mitochondrial DNA Copy Number and Nuclear-Encoded Mitochondrial Genes in Rat Brain Structures after Cocaine Self-Administration. Mol. Neurobiol. 2017, 54, 7460–7470. [Google Scholar] [CrossRef][Green Version]
- Castellani, C.A.; Longchamps, R.J.; Sun, J.; Guallar, E.; Arking, D.E. Thinking outside the nucleus: Mitochondrial DNA copy number in health and disease. Mitochondrion 2020, 53, 214–223. [Google Scholar] [CrossRef]
- Chandra, R.; Engeln, M.; Francis, T.C.; Konkalmatt, P.; Patel, D.; Lobo, M.K. A Role for Peroxisome Proliferator-Activated Receptor Gamma Coactivator-1α in Nucleus Accumbens Neuron Subtypes in Cocaine Action. Biol. Psychiatry 2017, 81, 564–572. [Google Scholar] [CrossRef] [PubMed]
- Cole, S.L.; Chandra, R.; Harris, M.; Patel, I.; Wang, T.; Kim, H.; Jensen, L.; Russo, S.J.; Turecki, G.; Gancarz-Kausch, A.M.; et al. Cocaine-induced neuron subtype mitochondrial dynamics through Egr3 transcriptional regulation. Mol. Brain 2021, 14, 101. [Google Scholar] [CrossRef] [PubMed]
- Bobadilla, A.-C.; Heinsbroek, J.A.; Gipson, C.D.; Griffin, W.C.; Fowler, C.D.; Kenny, P.J.; Kalivas, P.W. Corticostriatal plasticity, neuronal ensembles, and regulation of drug-seeking behavior. Prog. Brain. Res. 2017, 235, 93–112. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Park, B.-H.; Lee, J.H.; Park, S.K.; Kim, J.-H. Cell Type-Specific Alterations in the Nucleus Accumbens by Repeated Exposures to Cocaine. Biol. Psychiatry 2011, 69, 1026–1034. [Google Scholar] [CrossRef] [PubMed]
- Griffin, T.M.; Fermor, B.; Huebner, J.L.; Kraus, V.B.; Rodriguiz, R.M.; Wetsel, W.C.; Cao, L.; Setton, L.A.; Guilak, F. Diet-induced obesity differentially regulates behavioral, biomechanical, and molecular risk factors for osteoarthritis in mice. Arthritis Res. Ther. 2010, 12, R130. [Google Scholar] [CrossRef]
- Bangasser, D.A.; Valentino, R.J. Sex differences in stress-related psychiatric disorders: Neurobiological perspectives. Front. Neuroendocr. 2014, 35, 303–319. [Google Scholar] [CrossRef]
- Kanter, R.; Caballero, B. Global Gender Disparities in Obesity: A Review. Adv. Nutr. Int. Rev. J. 2012, 3, 491–498. [Google Scholar] [CrossRef]
- Mauvais-Jarvis, F.; Merz, N.B.; Barnes, P.J.; Brinton, R.D.; Carrero, J.-J.; DeMeo, D.L.; De Vries, G.J.; Epperson, C.N.; Govindan, R.; Klein, S.L.; et al. Sex and gender: Modifiers of health, disease, and medicine. Lancet 2020, 396, 565–582. [Google Scholar] [CrossRef]
- Murphy, M.O.; Loria, A.S. Sex-specific effects of stress on metabolic and cardiovascular disease: Are women at higher risk? Am. J. Physiol. Integr. Comp. Physiol. 2017, 313, R1–R9. [Google Scholar] [CrossRef]
- Ahmed, H.; Hannan, J.L.; Apolzan, J.; Osikoya, O.; Cushen, S.; Romero, S.A.; Goulopoulou, S. A free-choice high-fat, high-sucrose diet induces hyperphagia, obesity, and cardiovascular dysfunction in female cycling and pregnant rats. Am. J. Physiol. Integr. Comp. Physiol. 2019, 316, R472–R485. [Google Scholar] [CrossRef]
- Apolzan, J.W.; Harris, R.B. Differential effects of chow and purified diet on the consumption of sucrose solution and lard and the development of obesity. Physiol. Behav. 2012, 105, 325–331. [Google Scholar] [CrossRef][Green Version]
- Harris, R.B.S.; Apolzan, J.W. Changes in glucose tolerance and leptin responsiveness of rats offered a choice of lard, sucrose, and chow. Am. J. Physiol. Integr. Comp. Physiol. 2012, 302, R1327–R1339. [Google Scholar] [CrossRef] [PubMed][Green Version]
- La Fleur, S.E.; Luijendijk, M.C.M.; Van Rozen, A.J.; Kalsbeek, A.; Adan, R.A.H. A free-choice high-fat high-sugar diet induces glucose intolerance and insulin unresponsiveness to a glucose load not explained by obesity. Int. J. Obes. 2011, 35, 595–604. [Google Scholar] [CrossRef] [PubMed][Green Version]
- La Fleur, S.E.; Vanderschuren, L.; Luijendijk, M.C.; Kloeze, B.M.; Tiesjema, B.; Adan, R.A.H. A reciprocal interaction between food-motivated behavior and diet-induced obesity. Int. J. Obes. 2007, 31, 1286–1294. [Google Scholar] [CrossRef] [PubMed]
- Rolls, B.J.; Van Duijvenvoorde, P.; Rowe, E.A. Variety in the diet enhances intake in a meal and contributes to the development of obesity in the rat. Physiol. Behav. 1983, 31, 21–27. [Google Scholar] [CrossRef]
- Slomp, M.; Belegri, E.; Blancas-Velazquez, A.S.; Diepenbroek, C.; Eggels, L.; Gumbs, M.C.; Joshi, A.; Koekkoek, L.L.; Lamuadni, K.; Ugur, M.; et al. Stressing the importance of choice: Validity of a preclinical free-choice high-caloric diet paradigm to model behavioural, physiological and molecular adaptations during human diet-induced obesity and metabolic dysfunction. J. Neuroendocr. 2019, 31, e12718. [Google Scholar] [CrossRef]
- Ruigrok, S.; Kotah, J.; Kuindersma, J.; Speijer, E.; van Irsen, A.; la Fleur, S.; Korosi, A. Adult food choices depend on sex and exposure to early-life stress: Underlying brain circuitry, adipose tissue adaptations and metabolic responses. Neurobiol. Stress 2021, 15, 100360. [Google Scholar] [CrossRef] [PubMed]
| Behavioural Test | Assessement | Outcomes & Ref. | |||
|---|---|---|---|---|---|
| ⬆ (Increased) | ⬇ (Decreased) | ⬌ (Not Changed) | Conclusion | ||
| Maternal HFD treatment (pre-partum) Testing in adult offspring | |||||
| EPM | Anxiety | ♂♀ (pooled) [83r] | ⬌ (not changed) | ||
| OFT | Locomotion/exploration | ♂♀ (pooled) [83r] | ⬌ (not changed) | ||
| OBT | Learning & memory | ♂♀ (pooled) [83r] | ⬇ (decreased) | ||
| Maternal HFD treatment (pre- and post-partum) Testing in juvenile offspring | |||||
| EZM | Anxiety | ♂♀ [86r] | ⬌ (not changed) | ||
| FST | Passive stress-coping | ♂♀ [86r] | ⬆ (increased) | ||
| AC | Locomotion/exploration | ♂♀ [86r, 87r] | ⬌ (not changed) | ||
| RW | Volontary exercise | ♀ [85r] | ♂ [85r] | Δ (sign. changes) | |
| SPT | Anhedonia | ♂♀ [86r] | ⬌ (not changed) | ||
| NORT | Learning & memory | ♂♀ [86r] | ⬌ (not changed) | ||
| AC | AMPH locomotion | [87r] | ⬇ (decreased) | ||
| AC | AMPH sensitization | [87r] | |||
| Maternal HFD treatment (pre- and post-partum) Testing in adolescent offspring | |||||
| AC | Locomotion/exploration | ♀ [88r] | ⬌ (not changed) | ||
| ACQ | COC- self-administration | ♀ [88r] | ⬌ (not changed) | ||
| EXT | ♀ [88r] | ||||
| RST | ♀ [88r] | ||||
| Maternal HFD treatment (pre- and post-partum) Testing in adult offspring | |||||
| EZM | Anxiety | ♂♀ (pooled) [83r] | ⬌ (not changed) | ||
| AC/OFT | Locomotion/exploration | ♂♀ (pooled) [83r, 145r] | ⬌ (not changed) | ||
| OBT | Learning & memory | ♂♀ (pooled) [83r] | ⛒ (inconcl.) | ||
| MWM | [89r] | ||||
| Juvenile HFD treatment | |||||
| EPM/ETM/MBT/ OFT/DaLi | Anxiety | [92r, 100r, 101m, 103r, 104m] | [104m] | [93m, 94m, 95m, 99m, 102m, 104m] | ⛒ (inconcl.) |
| FST | Passive stress-coping | [92r] | [95m, 105r] | [101m, 102m, 106r] | ⛒ (inconcl.) |
| OFT/CA | Locomotion/exploration | [93m, 94m, 95m, 100r, 101m, 102m,103r] | ⬌ (not changed) | ||
| HBT | [101m] | ||||
| FUST/SIT | Anhedonia | [102m] | ⬌ (not changed) | ||
| NORT/RAM/HWM/ | Learning & memory | [93m, 94m, 95m, 96m] | ⬇ (decreased) | ||
| MWM/PAT | [92r, 109m] | ||||
| EXP | COC/AMPH-ind. CPP | [112m *$] | [108r *, 109m, 112m *$] | ♂♀ [107m *, 108r, 109m, 110m *] | Δ (sign. changes) |
| EXT | ♂♀ [107m *, 110m *, 112m *] | ||||
| RST | [99m] | [110m *] | [107m *, 112m *] | ||
| ACQ/RST | COC self-administration | [107m *] | ⬆ (increased) | ||
| OFT/AC | COC/AMPH locomotion | [99m, 113r] | Δ (sign. changes) | ||
| AC | AMPH sensitization | [113 r] | [108r] | ||
| EPM | COC-ind. anxiety | [107m *] | |||
| FST | COC-ind. immobility | [106r] | |||
| HFD treatment in late adolescence | |||||
| EPM/EZM/MBT/OFT/ NSF/DaLi | Anxiety | [114r, 115m, 118m, 177m] | [116m, 120m] | ⬆ (increased) | |
| FST/TST | Passive stress-coping | [117m] | [102m, 116m, 118m] | ⛒ (inconcl.) | |
| OFT/TM | Locomotion/exploration | [117m, 119m] | ⬇ (decreased) | ||
| SPT/SxB/FUST | Anhedonia | [116m] | [102m] | ⛒ (inconcl.) | |
| SIT | [116m] | [120m] | |||
| SRT | [116m] | ||||
| NORT/MWM/BM | Learning & memory | [116m, 121m, 122m] | ⬌ (not changed) | ||
| OBT | [122m] | ||||
| Adult HFD treatment | |||||
| OFT | Anxiety | [126m] | ⬌ (not changed) | ||
| OFT/YM | Locomotion/exploration | [127m] | [126m] | ⛒ (inconcl.) | |
| SPT | Anhedonia | [125r] | ⬌ (not changed) | ||
| TWAT | Learning & memory | [126m] | ⬌ (not changed) | ||
| OFT/AC | COC/AMPH locomotion | [129r] | [128r] | Δ (sign. changes) | |
| EPM | COC-ind. anxiolysis | [128r] | |||
| FST | COC-ind. active coping | [128r] | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ziemens, D.; Touma, C.; Rappeneau, V. Neurobiological Mechanisms Modulating Emotionality, Cognition and Reward-Related Behaviour in High-Fat Diet-Fed Rodents. Int. J. Mol. Sci. 2022, 23, 7952. https://doi.org/10.3390/ijms23147952
Ziemens D, Touma C, Rappeneau V. Neurobiological Mechanisms Modulating Emotionality, Cognition and Reward-Related Behaviour in High-Fat Diet-Fed Rodents. International Journal of Molecular Sciences. 2022; 23(14):7952. https://doi.org/10.3390/ijms23147952
Chicago/Turabian StyleZiemens, Dorothea, Chadi Touma, and Virginie Rappeneau. 2022. "Neurobiological Mechanisms Modulating Emotionality, Cognition and Reward-Related Behaviour in High-Fat Diet-Fed Rodents" International Journal of Molecular Sciences 23, no. 14: 7952. https://doi.org/10.3390/ijms23147952
APA StyleZiemens, D., Touma, C., & Rappeneau, V. (2022). Neurobiological Mechanisms Modulating Emotionality, Cognition and Reward-Related Behaviour in High-Fat Diet-Fed Rodents. International Journal of Molecular Sciences, 23(14), 7952. https://doi.org/10.3390/ijms23147952






