Antioxidative, Anti-Inflammatory, Anti-Obesogenic, and Antidiabetic Properties of Tea Polyphenols—The Positive Impact of Regular Tea Consumption as an Element of Prophylaxis and Pharmacotherapy Support in Endometrial Cancer
Abstract
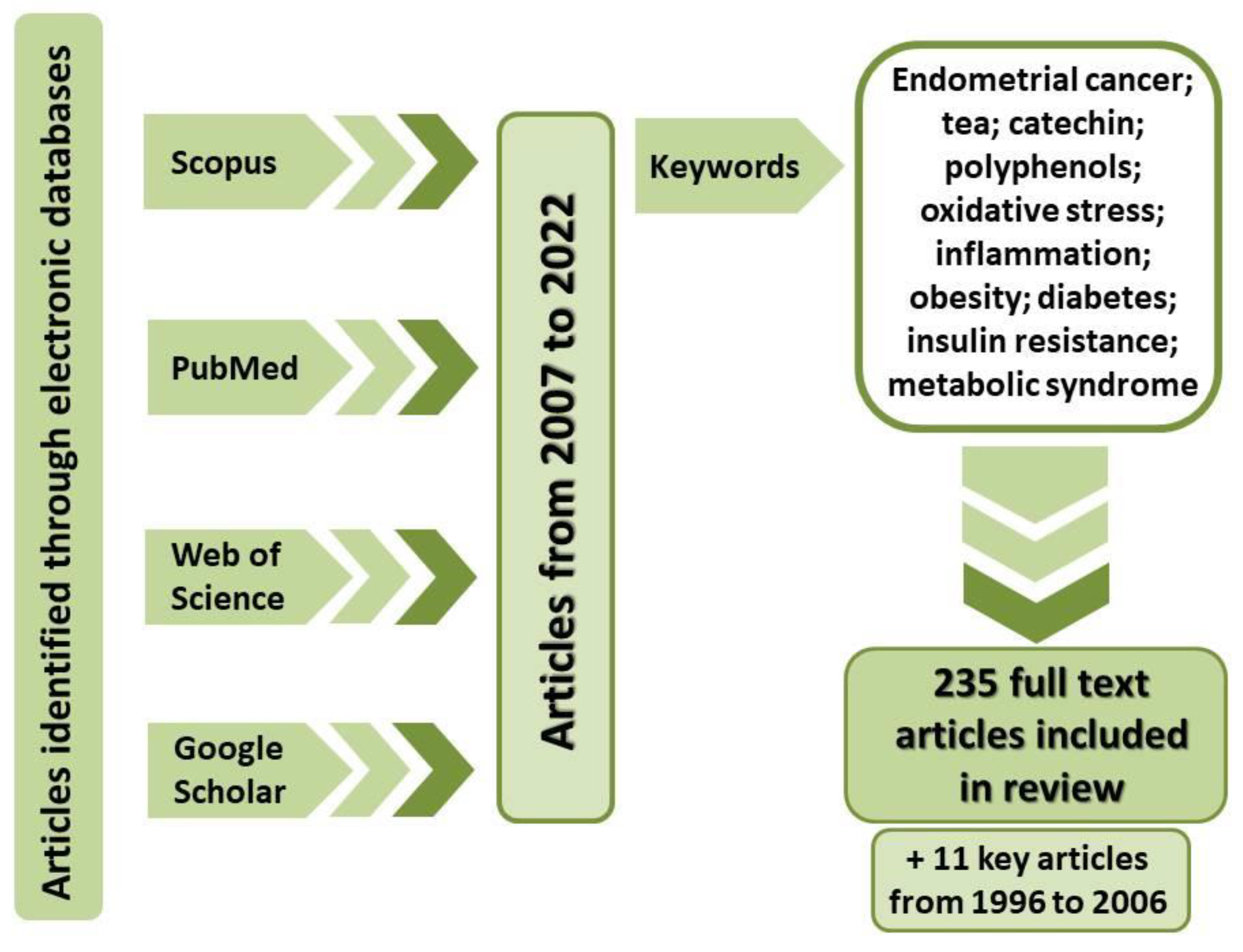
1. Introduction
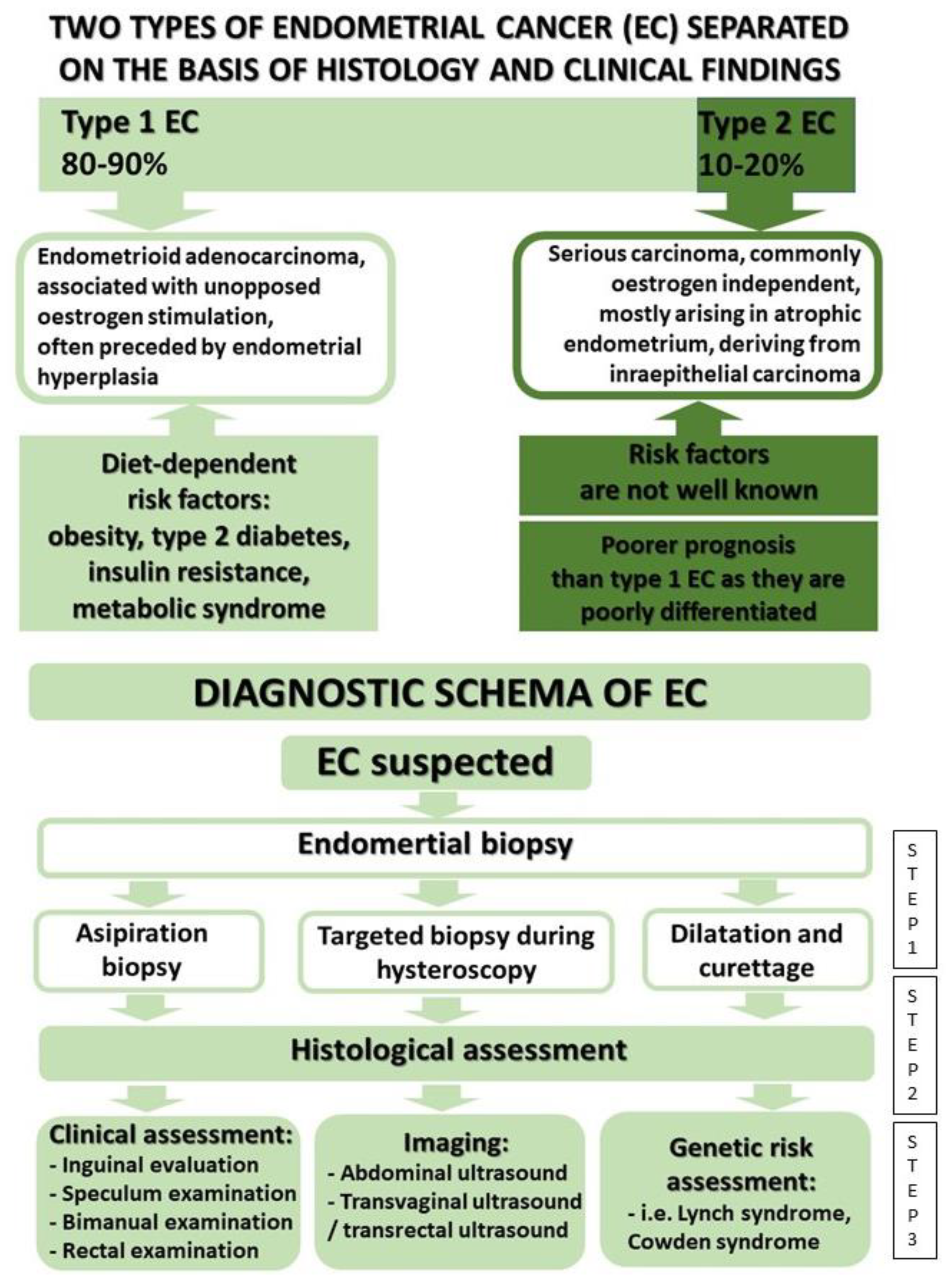
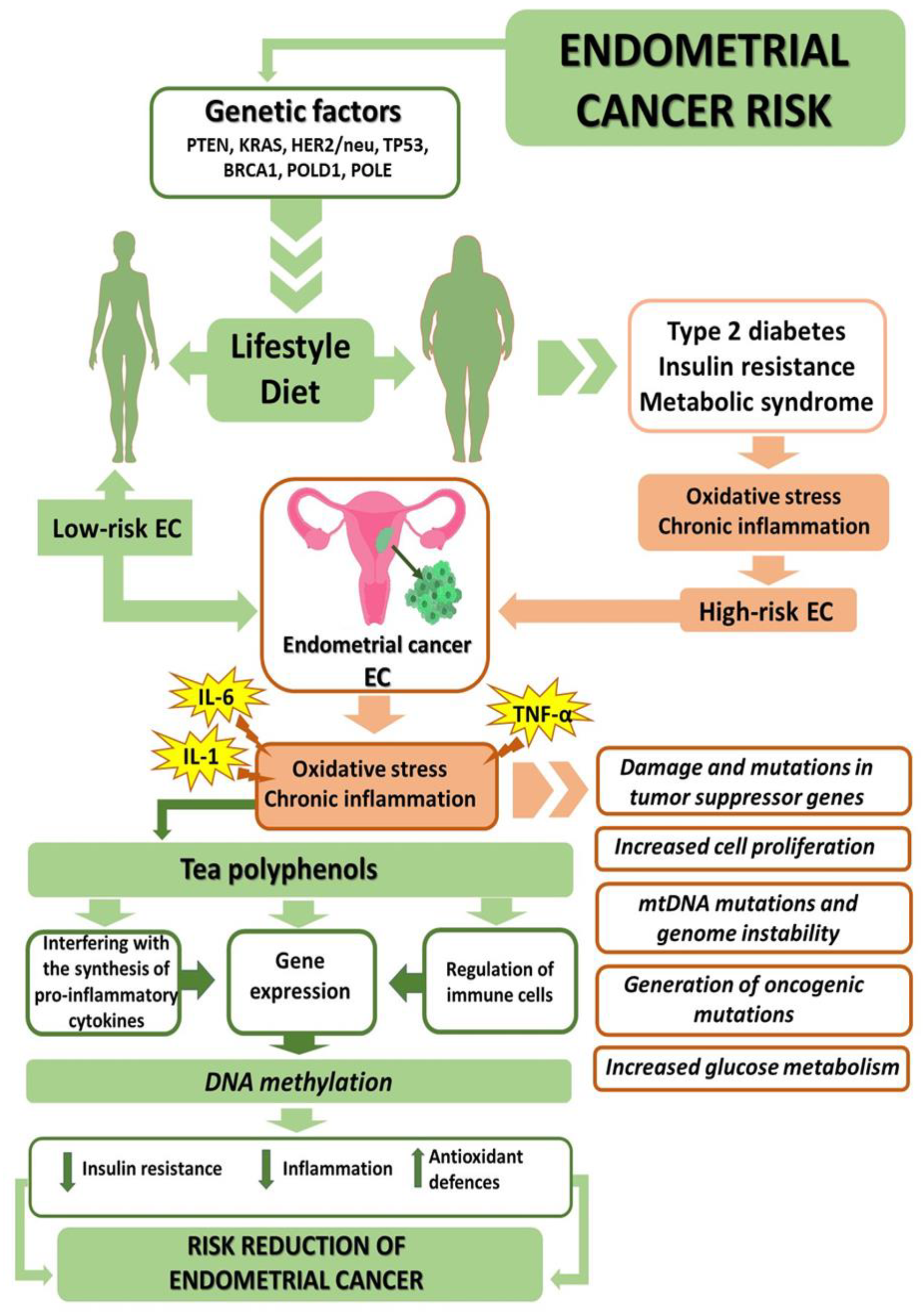
2. Genetic Factors
3. Treatment
4. Oxidative Stress in Endometrial Cancer
| Oxidative Stress Parameters | Target Sites | References |
|---|---|---|
| ↑ TOS; ↓ TAC | Serum | [13] |
| ↑ MDA, ↑ vit. E., ↑ TBARS | Serum | [54] |
| ↑ LOOHs, ↓ PON-1, ↑ Vit. E | Serum | [55] |
| ↑ Vit. E, ↓ SOD | Serum | [56] |
| Positive correlation between increasing BMI and markers of oxidative stress; positive correlation between cancer and the percent of GSH as GSSG in the samples run as cancer vs. non-cancer | Serum | [57] |
| No correlation between PON-1 and the stage of the disease | Serum | [58] |
| ↑ CAT, ↑ Cu, ↑ ceruloplasmin | Serum | [59] |
| ↓ TAS, ↓ native thiol | Serum | [60] |
| ↑ glutathionylated protein, ↓ GSH | Serum | [61] |
| ↑ TOS, ↑ OSI, ↑ TAS | Serum | [14] |
| ↓ SOD, ↑ NO, ↓ CAT | Serum | [48] |
| ↓ ORAC, ↓ Vit. C, ↓ Vit. E, ↓ Vit. A, ↑ GSH, ↑ SOD | Serum | [62] |
| ↑ Vit. A; ↓ Vit. E; ↓ Vit. C | Serum | [63] |
| ↓ GSH, ↑ MDA, ↑ SOD, ↑ 8-OHdG | Serum | [64] |
| ↑ SOD, ↓ GSH, ↓ GSH-Px | Serum | [65] |
| ↑ SOD | Serum | [66] |
| ↑ MDA, ↓ TAC | Serum | [67] |
| ↑ SOD, ↑ 8-OHdG, ↓ GSH-Px, ↑ 8-OHdG/GSH-Px | Serum | [68] |
| ↑ GSH-Px; ↑ SOD | Serum | [69] |
| ↑ AOPP, ↑ nitrates and nitrites; significant correlation between pelvic pain symptom scores and peritoneal protein oxidative stress markers | Peritoneal fluid | [70] |
| ↑ ox-LDL, ↑ 8-OHdG, ↑ 8-isoprostane, ↑ 8-isoPGF2α, ↑ 25-hydroxycholesterol | Peritoneal fluid | [71] |
| ↑ MDA, ↑ LOOHs | Peritoneal fluid | [72] |
| ↑ 8-OHdG, ↑ 8-isoprostane, ↑ 8-isoPGF2α, ↑ 25-hydroxycholesterol | Peritoneal fluid | [73] |
| ↑ 8-OHdG | Ovarian cortex tissue | [74] |
| ↑ LPO, ↓ TAC | Follicular fluid | [54] |
| ↑ ROS, ↑ MDA, ↑ NO, ↓ SOD, ↓ CAT, ↓ GSH-Px, ↓ Vit. A, ↓ Vit. C, ↓ vit. E | Follicular fluid | [75] |
| ↓ SOD, ↓ CAT, ↓ GSH-Px, ↓ GR/GSH, ↓ Vit. A, ↓ Vit. C, ↓ Vit. E | Follicular fluid | [56] |
| ↑ 8-isoPGF2α | Urine | [76] |
| ↓ 8-OHdG | Urine | [77] |
| ↓ GSH; ↓ SOD | Tissues gums | [78] |
5. Diet-Dependent EC Comorbidities
5.1. Obesity
5.2. Type 2 Diabetes and Insulin Resistance
5.3. Metabolic Syndrome
6. Anti-Obesogenic, Antidiabetic, and Antioxidative Properties of Tea
6.1. Anti-Obesogenic, Antidiabetic, and Antioxidative Substances Present in Tea
6.2. Impact of Tea on Body Weight—Research Overview
6.3. Antidiabetic Properties of Tea—Research Overview
6.4. Impact of Tea on the Organism’s Antioxidative Parameters—Research Overview
7. Tea as an Element of Prophylaxis and Pharmacotherapy Supplementation in Endometrial Cancer
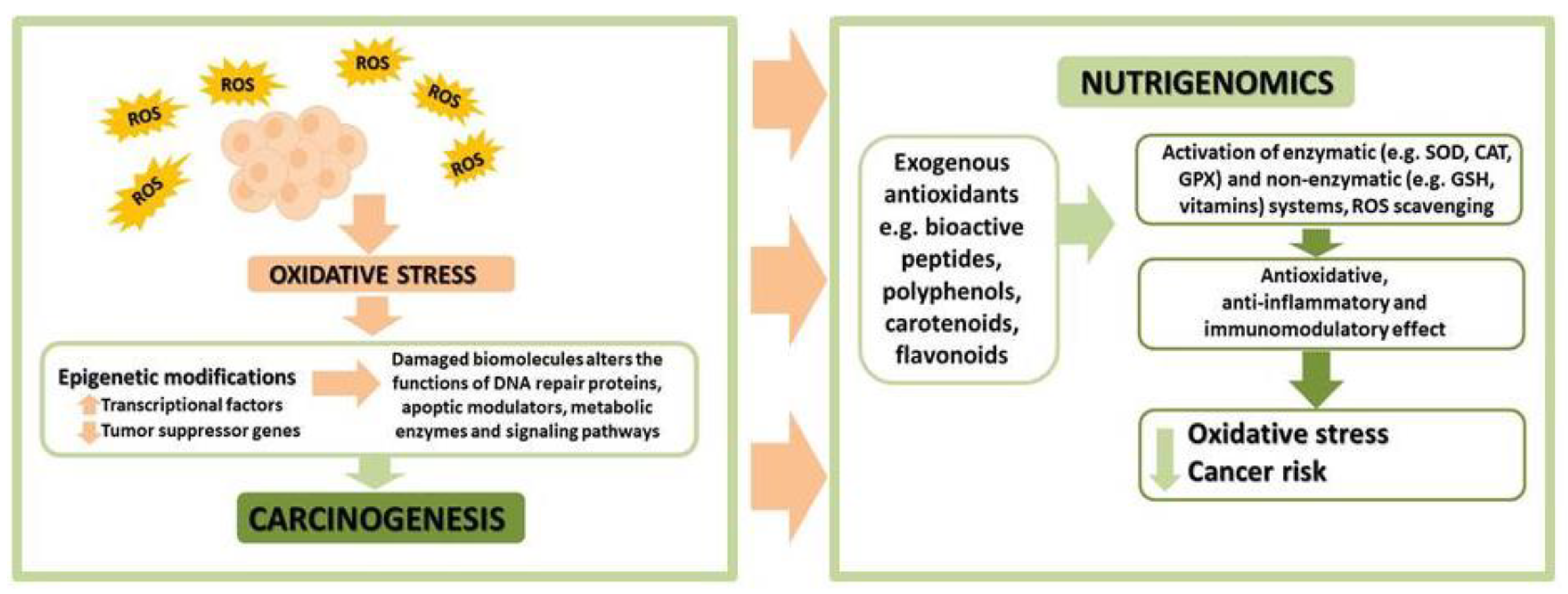
8. Oxidative Stress and Epigenetic Modifications in Cancer Cells
9. Perspectives—Tea as an Epigenetic Regulator in Endometrial Cancer Therapy
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- GLOBOCAN. 2020. Available online: https:gco.iarc.fr (accessed on 12 April 2022).
- Siegel, R.L.; Miller, K.D.; Jemaln, A. Cancer statistic, 2016. CA-Cancer J. Clin. 2016, 66, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Colombet, M.; Mery, L.; Piñeros, M.; Znaor, A.; Zanetti, R.; Ferlay, J. Cancer incidence in five Continents. In IARC Scientific Publication; No. 166; International Agency for Research on Cancer: Lyon, France, 2021; Volume XI. [Google Scholar]
- Jemal, A.; Siegel, R.; Xu, J.; Ward, E. Cancer statistics, 2010. CA-A Cancer J. Clin. 2010, 60, 277–300. [Google Scholar] [CrossRef] [PubMed]
- Oliver, K.E.; Enewold, L.R.; Zhu, K.; Conrads, T.P.; Rose, G.S.; Maxwell, G.L.; Farley, J.H. Racial disparities in histopathologic characteristics of uterine cancer are present in older, not younger blacks in an equal-access environment. Gynecol. Oncol. 2011, 123, 76–81. [Google Scholar] [CrossRef]
- Urick, M.E.; Bell, D.W. Clinical actionability of molecular targets in endometrial cancer. Nat. Rev. Cancer 2019, 19, 510–521. [Google Scholar] [CrossRef] [PubMed]
- Yasin, H.K.; Taylor, A.H.; Ayakannu, T. A narrative review of the role of diet and lifestyle factors in the development and prevention of endometrial cancer. Cancers 2021, 13, 2149. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Y.; Li, J.; Cragun, J.; Hatch, K.; Chambers, S.K.; Zheng, W. Lynch syndrome related endometrial cancer: Clinical significance beyond the endometrium. J. Hematol. Oncol. 2013, 6, 22. [Google Scholar] [CrossRef]
- Dörk, T.; Hillemanns, P.; Tempfer, C.; Breu, J.; Fleisch, M.C. Genetic susceptibility to endometrial cancer: Risk factors and clinical management. Cancers 2020, 12, 2407. [Google Scholar] [CrossRef]
- Noureen, F.; Khan, A.S. Analysis of Level of Antioxidants and Oxidative Stress in Diabetic Patients. Syst. Rev. Pharm. 2021, 12, 361–363. [Google Scholar]
- Pieme, C.A.; Tatangmo, J.A.; Simo, G.; Nya, P.C.B.; Moor, V.J.A.; Moukette, B.M.; Nzufo, F.T.; Nono, B.L.N.; Sobngwi, E. Relationship between hyperglycemia, antioxidant capacity and some enzymatic and non-enzymatic antioxidants in African patients with type 2 diabetes. BMC Res. Notes 2017, 10, 141. [Google Scholar] [CrossRef]
- Winiarska-Mieczan, A.; Tomaszewska, E.; Jachimowicz, K. Antioxidant, anti-inflammatory, and immunomodulatory properties of tea—The positive impact of tea consumption on patients with autoimmune diabetes. Nutrients 2021, 13, 3972. [Google Scholar] [CrossRef]
- Arioz, D.T.; Camuzcuoglu, H.; Toy, H.; Kurt, S.; Celik, H.; Aksoy, N. Serum prolidase activity and oxidative status in patients with stage I endometrial cancer. Int. J. Gynecol. Cancer 2009, 19, 1244–1247. [Google Scholar] [CrossRef] [PubMed]
- Camuzcuoğlu, A.; Sezgin, B.; Çelik, H.; Camuzcuoğlu, H. Evaluation of serum M30 and M65 activity in patients with stage-I endometrial cancer. J. Obst. Gynaecol. 2019, 39, 1112–1116. [Google Scholar] [CrossRef] [PubMed]
- Sezgin, B.; Pirinççi, F.; Camuzcuoğlu, A.; Erel, Ö.; Neşelioğlu, S.; Camuzcuoğlu, H. Assessment of thiol disulfide balance in early-stage endometrial cancer. J. Obstet. Gynaecol. Res. 2020, 46, 1140–1147. [Google Scholar] [CrossRef] [PubMed]
- Spurdle, A.; Bowman, M.; Shamsani, J.; Kirk, J. Endometrial cancer gene panels: Clinical diagnostic vs. research germline DNA testing. Modern Pathol. 2017, 30, 1048–1068. [Google Scholar] [CrossRef] [PubMed]
- Brooks, R.A.; Fleming, G.F.; Lastra, R.R.; Lee, N.K.; Moroney, J.W.; Son, C.H.; Tatebe, K.; Veneris, J.L. Current recommendations and recent progress in endometrial cancer. CA Cancer J. Clin. 2019, 69, 258–279. [Google Scholar] [CrossRef] [PubMed]
- Saule, C.; Mouret-Fourme, E.; Briaux, A.; Becette, V.; Rouzier, R.; Houdayer, C.; Stoppa-Lyonnet, D. Risk of serous endometrial carcinoma in women with pathogenic BRCA1/2 variant after risk-reducing salpingo-oophorectomy. J. Natl. Cancer Inst. 2018, 110, 213–215. [Google Scholar] [CrossRef]
- Church, D.N.; Stelloo, E.; Nout, R.A.; Valtcheva, N.; Depreeuw, J.; ter Haar, N.; Noske, A.; Amant, F.; Tomlinson, I.P.; Wild, P.J.; et al. Prognostic significance of POLE proofreading mutations in endometrial cancer. J. Natl. Cancer Inst. 2014, 107, 402. [Google Scholar] [CrossRef]
- Thompson, D.; Easton, D.F.; Breast Cancer Linkage Consortium. Cancer incidence in BRCA1 mutation carriers. J. Natl. Cancer Inst. 2002, 94, 1358–1365. [Google Scholar] [CrossRef]
- Beiner, M.E.; Finch, A.; Rosen, B.; Lubinski, J.; Møller, P.; Ghadirian, P.; Lynch, H.T.; Friedman, E.; Sun, P.; Narod, S.A. The risk of endometrial cancer in women with BRCA1 and BRCA2 mutations. A prospective study. Gynecol. Oncol. 2007, 104, 7–10. [Google Scholar] [CrossRef]
- Segev, Y.; Iqbal, J.; Lubinski, J.; Gronwald, J.; Lynch, H.T.; Moller, P.; Ghadirian, P.; Rosen, B.; Tung, N.; Kim-Sing, C.; et al. Hereditary Breast Cancer Study Group. The incidence of endometrial cancer in women with BRCA1 and BRCA2 mutations: An international prospective cohort study. Gynecol. Oncol. 2013, 130, 127–131. [Google Scholar] [CrossRef]
- Lucenteforte, E.; Talamini, R.; Montella, M.; Maso, L.D.; Pelucchi, C.; Franceschi, S.; La Vecchia, C.; Negri, E. Family history of cancer and the risk of endometrial cancer. Eur. J. Cancer Prev. 2009, 18, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Win, A.K.; Reece, J.C.; Ryan, S. Family history and risk of endometrial cancer: A systematic review and meta-analysis. Obstet. Gynecol. 2015, 125, 89–98. [Google Scholar] [CrossRef]
- Leclerc, J.; Vermaut, C.; Buisine, M.P. Diagnosis of Lynch syndrome and strategies to distinguish Lynch-related tumors from sporadic MSI/dMMR tumors. Cancers 2021, 13, 467. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Chen, L.; Zang, Y.; Liu, W.; Liu, S.; Teng, F.; Xue, F.; Wang, Y. Endometrial cancer in Lynch syndrome. Int. J. Cancer 2022, 150, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Bonadona, V.; Bonaiti, B.; Olschwang, S.; Grandjouan, S.; Huiart, L.; Longy, M.; Guimbaud, R.; Buecher, B.; Bignon, Y.J.; Caron, O.; et al. Cancer risks associated with germline mutations in MLH1, MSH2, and MSH6 genes in Lynch syndrome. JAMA 2011, 305, 2304–2310. [Google Scholar] [CrossRef]
- Oda, K.; Okada, J.; Timmerman, L.; Rodriguez-Viciana, P.; Stokoe, D.; Shoji, K.; Taketani, Y.; Kuramoto, H.; Knight, Z.A.; Shokat, K.M.; et al. PIK3CA cooperates with other phosphatidylinositol 3’-kinase pathway mutations to effect oncogenic transformation. Cancer Res. 2008, 68, 8127–8136. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.A.; Turner, E.H.; Beightol, M.B.; Jacobson, A.; Gooley, T.A.; Salipante, S.J.; Haraldsdottir, S.; Smith, C.; Scroggins, S.; Tait, J.F.; et al. Frequent PIK3CA mutations in colorectal and endometrial tumors with 2 or more somatic mutations in mismatch repair genes. Gastroenterology 2016, 151, 440–447. [Google Scholar] [CrossRef]
- Ryan, N.A.J.; Walker, T.D.J.; Bolton, J.; ter Haar, N.; Van Wezel, T.; Glaire, M.A.; Church, D.N.; Evans, D.G.; Bosse, T.; Crosbie, E.J. Histological and somatic mutational profiles of mismatch repair deficient endometrial tumours of different aetiologies. Cancers 2021, 13, 4538. [Google Scholar] [CrossRef]
- Meyer, L.A.; Broaddus, R.R.; Lu, K.H. Endometrial cancer and Lynch syndrome: Clinical and pathologic considerations. Cancer Control 2009, 16, 14–22. [Google Scholar] [CrossRef]
- Lawrence, J.; Richer, L.; Arseneau, J.; Zeng, X.; Chong, G.; Weber, E.; Foulkes, W.; Palma, L. Mismatch Repair Universal Screening of Endometrial Cancers (MUSE) in a Canadian Cohort. Curr. Oncol. 2021, 28, 509–522. [Google Scholar] [CrossRef]
- Pilarski, R.; Eng, C. Will the real Cowden syndrome please stand up (again)? Expanding mutational and clinical spectra of the PTEN hamartoma tumour syndrome. J. Med. Genet. 2004, 41, 323–326. [Google Scholar] [CrossRef] [PubMed]
- Orloff, M.S.; He, X.; Peterson, C.; Chen, F.; Chen, J.L.; Mester, J.L.; Eng, C. Germline PIK3CA and AKT1 mutations in Cowden and Cowden-like syndromes. Am. J. Hum. Genet. 2013, 92, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Yehia, L.; Ni, Y.; Sesock, K.; Niazi, F.; Fletcher, B.; Chen, H.; LaFramboise, T.; Eng, C. Unexpected cancer-predisposition gene variants in Cowden syndrome and Bannayan-Riley-Ruvalcaba syndrome patients without underlying germline PTEN mutations. PLoS Genet. 2018, 14, e1007352. [Google Scholar] [CrossRef] [PubMed]
- Sznurkowski, J.J.; Knapp, P.; Bodnar, L.; Bidziński, M.; Jach, R.; Misiek, M.; Bieńkiewicz, A.; Blecharz, P.; Kojs, Z.; Kotarski, J.; et al. Recommendations of the Polish Gynecological Oncology Society for the diagnosis and treatment of endometrial cancer. Curr. Gynecol. Oncol. 2017, 15, 34–44. [Google Scholar] [CrossRef]
- Gałczyński, K.; Chauvet, P.; Canis, M.; Bourdel, N. Laparoscopic extraperitoneal lumboaortic lymphadenectomy in 10 steps—Let’s make it easier! Gynecol. Oncol. 2016, 143, 448–449. [Google Scholar] [CrossRef]
- Boulay, E.; Gałczyński, K.; Canis, M.; Bourdel, N. Laparoscopic pelvic lymphadenectomy in 10 steps—Let’s make it easier! Gynecol. Oncol. 2017, 145, 217–218. [Google Scholar] [CrossRef]
- Kemp, M.M.; Gałczyński, K.; Botchorishvili, R.; Bourdel, N. How to facilitate laparoscopy in obese oncologic patients—10 steps towards successful surgery. Gynecol. Oncol. 2018, 149, 223–224. [Google Scholar] [CrossRef]
- Holloway, R.; Abu-Rustum, N.; Backes, F.; Boggess, J.; Gotlieb, W.; Lowery, W.; Rossi, E.; Tanner, E.; Wolsky, R. Sentinel lymph node mapping and staging in endometrial cancer: A society of gynecologic oncology literature review with consensus recommendations. Gynecol. Oncol. 2017, 146, 405–415. [Google Scholar] [CrossRef]
- Paracchini, S.; Chauvet, P.; Gałczyński, K.; Boulay, E.; Jaillet, L.; Canis, M.; Bourdel, N. Surgical technique for the sentinel lymph node (SLN) mapping in 10 steps. Gynecol. Oncol. 2020, 156, 746–747. [Google Scholar] [CrossRef]
- Gałczyński, K.; Nowakowski, Ł.; Rechberger, T.; Semczuk, A. Should we be more aware of endometrial cancer in adolescents? Dev. Period Med. 2016, 20, 169–173. [Google Scholar]
- Gałczyński, K.; Olcha, P.; Romanek-Piva, K.; Józwik, M.; Semczuk, A. Fertility-sparing methods in adolescents affected by endometrial cancer: A comprehensive review. J. Clin. Med. 2021, 10, 1020. [Google Scholar] [CrossRef]
- Lambert, J.D.; Elias, R.J. The antioxidant and pro-oxidant activities of green tea polyphenols, a role in cancer prevention. Arch. Biochem. Biophys. 2010, 501, 65–72. [Google Scholar] [CrossRef]
- Winiarska-Mieczan, A. Protective effect of tea against lead and cadmium-induced oxidative stress—A review. Biometals 2018, 31, 909–926. [Google Scholar] [CrossRef]
- Giridharan, S.; Srinivasan, M. Mechanisms of NF-κB p65 and strategies for therapeutic manipulation. J. Inflamm. Res. 2018, 11, 407–419. [Google Scholar] [CrossRef]
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef]
- Ścibior-Bentkowska, D.; Czeczot, H. Cancer cells and oxidative stress. Post. Hig. Med. Dosw. 2009, 63, 58–72. [Google Scholar]
- Fryczkowski, M.; Hejmo, T.; Bułdak, M.; Stachowska, M.; Rokicka, J.; Żwirska-Korczala, K. Impact of selected pro-inflammatory cytokines and oxidative stress on carcinogenesis and progression of prostate and colorectal adenocarcinomas. Ann. Acad. Med. Silesien. 2019, 73, 182–193. [Google Scholar] [CrossRef]
- Przybyszewski, W.; Rzeszowska-Wolny, J. Oxidative stress in prostate hypertrophy and carcinogenesis. Post. Hig. Med. Dośw. 2009, 63, 340–350. [Google Scholar]
- Reczek, C.R.; Chandel, N.S. The two faces of reactive oxygen species in cancer. Ann. Rev. Cancer Biol. 2017, 1, 79–98. [Google Scholar] [CrossRef]
- Winiarska-Mieczan, A.; Baranowska-Wójcik, E.; Kwiecień, M.; Grela, E.R.; Szwajgier, D.; Kwiatkowska, K.; Kiczorowska, B. The role of dietary antioxidants in the pathogenesis of neurodegenerative diseases and their impact on cerebral oxidoreductive balance. Nutrients 2020, 12, 435. [Google Scholar] [CrossRef]
- Winiarska-Mieczan, A. The potential protective effect of green, black, red and white tea infusions against adverse effect of cadmium and lead during chronic exposure—A rat model study. Regul. Toxicol. Pharmacol. 2015, 73, 521–529. [Google Scholar] [CrossRef]
- Nasiri, N.; Moini, A.; Eftekhari-Yazdi, P.; Karimian, L.; Salman-Yazdi, R.; Arabipoor, A. Oxidative stress statues in serum and follicular fluid of women with endometriosis. Cell J. 2017, 18, 582–587. [Google Scholar]
- Verit, F.; Erel, O.; Celik, N. Serum paraoxonase-1 activity in women with endometriosis and its relationship with the stage of the disease. Hum. Reprod. 2008, 23, 100–104. [Google Scholar] [CrossRef]
- Prieto, L.; Quesada, J.F.; Cambero, O.; Pacheco, A.; Pellicer, A.; Codoceo, R.; Garcia-Velasco, J.A. Analysis of follicular fluid and serum markers of oxidative stress in women with infertility related to endometriosis. Fertil. Steril. 2012, 98, 126–130. [Google Scholar] [CrossRef]
- Salinas, E.A.; Wernimont, S.A.; Mapuskar, K.A.; Szweda, L.I.; McCormick, M.L.; Spitz, D.; Goodheart, M.J. Oxidative stress biomarkers and risk of endometrial cancer. Gynecol. Oncol. 2015, 137, 160. [Google Scholar] [CrossRef]
- Bragatto, F.B.; Barbosa, C.P.; Christofolini, D.M.; Peluso, C.; dos Santos, A.A.; Mafra, F.A.; Cavalcanti, V.; Hix, S.; Bianco, B. There is no relationship between paraoxonase serum level activity in women with endometriosis and the stage of the disease: An observational study. Reprod. Health 2013, 10, 32. [Google Scholar] [CrossRef]
- Turkyilmaz, E.; Yildirim, M.; Cendek, B.D.; Baran, P.; Alisik, M.; Dalgaci, F.; Yavuz, A.F. Evaluation of oxidative stress markers and intra-extracellular antioxidant activities in patients with endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 199, 164–168. [Google Scholar] [CrossRef]
- Turgut, A.; Ozler, A.; Goruk, N.Y.; Tunc, S.Y.; Evliyaoglu, O.; Gul, T. Copper, ceruloplasmin and oxidative stress in patients with advanced-stage endometriosis. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 1472–1478. [Google Scholar]
- Andrisani, A.; Dona, G.; Brunatim, A.M.; Clari, G.; Armanini, D.; Ragazzi, E.; Ambrosinia, G.; Bordin, L. Increased oxidation-related glutathionylation and carbonic anhydrase activity in endometriosis. Reprod. Biomed. Online 2014, 28, 773–779. [Google Scholar] [CrossRef][Green Version]
- Badid, N.; Baba, F.Z.; Merzouk, H.; Belbraouet, S.; Mokharti, N.; Merzouk, S.A.; Benhabib, R.; Hamazaoui, D. Oxidant/antioxidant status, lipids and hormonal profile in overweight women with breast cancer. Pathol. Oncol. Res. 2010, 16, 159–167. [Google Scholar] [CrossRef]
- Shah, F.D.; Patel, J.B.; Shukla, S.N.; Shah, P.M.; Patel, P.S. Evaluation of plasma non- enzymatic antioxidants in breast cancer etiology. Asian Pac. J. Cancer Prev. 2009, 10, 91–96. [Google Scholar]
- Zahra, K.; Patel, S.; Dey, T.; Pandey, U.; Mishr, S.P. A study of oxidative stress in cervical cancer- an institutional study. Biochem. Biophys. Rep. 2021, 25, 100881. [Google Scholar] [CrossRef]
- Demirci, S.; Ozsaran, Z.; Celik, H.A.; Aras, A.B.; Aydin, H.H. The interaction between antioxidant status and cervical cancer: A case control study. Tumori J. 2011, 97, 290–295. [Google Scholar] [CrossRef]
- Nirmala, G.; Narendhirakannan, R.T. Detection and genotyping of high-risk HPV and evaluation of antioxidant status in cervical carcinoma patients in Tamil Nadu State, India-a case control study. Asian Pac. J. Cancer Prev. 2011, 12, 2689–2695. [Google Scholar]
- Shah, S.; Kalal, B.S. Oxidative stress in cervical cancer and its response to chemoradiation. Turk. J. Obstet. Gynecol. 2019, 16, 124–128. [Google Scholar] [CrossRef]
- Himmetoglu, S.; Dincer, Y.; Ersoy, Y.E.; Bayraktar, B.; CeliK, V.; Akcay, T. DNA oxidation and antioxidant status in breast cancer. J. Investig. Med. 2009, 57, 720–723. [Google Scholar] [CrossRef]
- Moradi, M.; Eftekhari, M.H.; Talei, A.; Fard, A.R. A comparative study of selenium concentration and glutathione peroxidase activity in normal and breast cancer patients. Public Health Nutr. 2009, 12, 59–63. [Google Scholar] [CrossRef]
- Santulli, P.; Chouzenoux, S.; Fiorese, M.; Marcellin, L.; Lemarechal, H.; Millischer, A.E.; Batteux, F.; Borderie, D.; Chapron, C. Protein oxidative stress markers in peritoneal fluids of women with deep infiltrating endometriosis are increased. Hum. Reprod. 2015, 30, 49–60. [Google Scholar] [CrossRef]
- Polak, G.; Barczyński, B.; Kwaśniewski, W.; Bednarek, W.; Wertel, I.; Drewnianka-Polak, M.; Kotarski, J. Low-density lipoproteins oxidation and endometriosis. Mediat. Inflamm. 2013, 2013, 624540. [Google Scholar] [CrossRef]
- Mier-Cabrera, J.; Jimenez-Zamudio, L.; Garcia-Latorre, E.; Cruz-Orozco, O.; Hernandez-Guerrero, C. Quantitative and qualitative peritoneal immune profiles, T-cell apoptosis and oxidative stress-associated characteristics in women with minimal and mild endometriosis. BJOG Int. J. Obstet. Gynaecol. 2011, 118, 6–16. [Google Scholar] [CrossRef]
- Sharma, I.; Dhaliwal, L.K.; Saha, S.C.; Sangwan, S.; Dhawan, V. Role of 8-iso-prostaglandin F2alpha and 25-hydroxycholesterol in the pathophysiology of endometriosis. Fertil. Steril. 2010, 94, 63–70. [Google Scholar] [CrossRef]
- Matsuzaki, S.; Schubert, B. Oxidative stress status in normal ovarian cortex surrounding ovarian endometriosis. Fertil. Steril. 2010, 93, 2431–2432. [Google Scholar] [CrossRef]
- Singh, A.K.; Chattopadhyay, R.; Chakravarty, B.; Chaudhury, K. Markers of oxidative stress in follicular fluid of women with endometriosis and tubal infertility undergoing IVF. Reprod. Toxicol. 2013, 42, 116–124. [Google Scholar] [CrossRef]
- Kędzierska, M.; Olas, B.; Wachowicz, B.; Jeziorsiki, A.; Piekarski, J. The lipid peroxidation in breast cancer patients. Gen. Physiol. Biophys. 2010, 29, 208–210. [Google Scholar]
- Kuo, H.W.; Chou, S.Y.; Hu, T.W.; Wu, F.Y.; Chen, D.J. Urinary 8-hydroxy-2′-deoxyguanosine (8-OHdG) and genetic polymorphisms in breast cancer patients. Mutat. Res. 2007, 631, 62–68. [Google Scholar] [CrossRef]
- Tao, L.; Park, J.Y.; Lambert, J.D. Differential prooxidative effects of the green tea polyphenol, (−)-epigallocatechin-3-gallate, in normal and oral cancer cells are related to differences in sirtuin 3 signaling. Mol. Nutr. Food Res. 2015, 59, 203–211. [Google Scholar] [CrossRef]
- Gałczyński, K.; Bełtowski, J.; Nowakowski, Ł.; Vasilevska, D.; Rechberger, T.; Semczuk, A. Serum paraoxonase 1 activity and protein N-homocysteinylation in primary human endometrial cancer. Tumor Biol. 2018, 40. [Google Scholar] [CrossRef]
- Gifkins, D.; Olson, S.H.; Demissie, K.; Lu, S.E.; Kong, A.N.; Bandera, E.V. Total and individual antioxidant intake and endometrial cancer risk: Results from a population-based case-control study in New Jersey. Cancer Causes Control 2012, 23, 887–895. [Google Scholar] [CrossRef][Green Version]
- Skotnicka, M.; Golan, M.; Szmukała, N. Role of natural antioxidants of plants in cancer prevention. Ann. Acad. Med. Gedan. 2017, 47, 119–127. [Google Scholar]
- Bhat, H.K.; Calaf, G.M.; Hei, T.K.; Loya, T.; Vadgama, J.V. Critical role of oxidative stress in estrogen-induced carcinogenesis. Proc. Natl. Acad. Sci. USA 2003, 100, 3913–3918. [Google Scholar] [CrossRef]
- MacKintosh, M.L.; Crosbie, E.J. Obesity-driven endometrial cancer: Is weight loss the answer? Int. J. Obstet. Gynaecol. 2013, 120, 791–794. [Google Scholar] [CrossRef]
- Zhao, H.; Zhou, L.; Shangguan, A.J.; Bulun, S.E. Aromatase expression and regulation in breast and endometrial cancer. J. Mol. Endocrinol. 2016, 57, 19–33. [Google Scholar] [CrossRef]
- Usluogullari, B.; Duvan, C.Z.; Usluogullari, C.A. Use of aromatase inhibitors in practice of gynecology. J. Ovarian Res. 2015, 8, 4. [Google Scholar] [CrossRef]
- Viola, A.S.; Gouveia, D.; Andrade, L.; Aldrighi, J.M.; Viola, C.F.; Bahamondes, L. Prevalence of endometrial cancer and hyperplasia in non-symptomatic overweight and obese women. Aust. N. Z. J. Obstet. Gynaecol. 2008, 48, 207–213. [Google Scholar] [CrossRef]
- Joehlin-Price, A.S.; Stephens, J.A.; Zhang, J.; Backes, F.J.; Cohn, D.E.; Suarez, A.A. Endometrial Cancer Insulin-Like Growth Factor 1 Receptor (IGF1R) Expression increases with body mass index and is associated with pathologic extent and prognosis. Cancer Epidem. Biomar. 2016, 25, 438–445. [Google Scholar] [CrossRef]
- Ma, Y.; Liu, Z.; Zhang, Y.; Lu, B. Serum leptin, adiponectin and endometrial cancer risk in Chinese women. J. Gynecol. Oncol. 2013, 24, 336–341. [Google Scholar] [CrossRef]
- Linkov, F.; Goughnour, S.L.; Ma, T.; Xu, Z.; Edwards, R.P.; Lokshin, A.E.; Ramanathan, R.C.; Hamad, G.G.; McCloskey, C.; Bovbjerg, D.H. Changes in inflammatory endometrial cancer risk biomarkers in individuals undergoing surgical weight loss. Gynecol. Oncol. 2017, 147, 133–138. [Google Scholar] [CrossRef]
- Bieńkiewicz, J.; Romanowicz-Makowska, H.; Malinowski, A. Endometrial cancer and adiponectin. Menop. Rev. 2012, 6, 490–494. [Google Scholar] [CrossRef]
- Weyer, C.; Funahashi, T.; Tanaka, S.; Hotta, K.; Matsuzawa, Y.; Pratley, R.E.; Tataranni, P.A. Hypoadiponectinemia in obesity and type 2 diabetes: Close association with insulin resistance and hyperinsulinemia. J. Clin. Endocrinol. Metab. 2001, 86, 1930–1935. [Google Scholar] [CrossRef]
- Uzunlulu, M.; Caklili, O.T.; Oguz, A. Association between Metabolic Syndrome and Cancer. Ann. Nutr. Metab. 2016, 68, 173–179. [Google Scholar] [CrossRef]
- Friberg, E.; Mantzoros, C.S.; Wolk, A. Physical activity and risk of endometrial cancer: A population-based prospective cohort study. Cancer Epidemiol. Biomarkers Prev. 2006, 15, 2136–2140. [Google Scholar] [CrossRef] [PubMed]
- McTiernan, A. Mechanisms linking physical activity with cancer. Nat. Rev. Cancer 2008, 8, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Sjöström, L.; Gummesson, A.; Sjöström, C.D.; Narbro, K.; Peltonen, M.; Wedel, H.; Bengtsson, C.; Bouchard, C.; Carlsson, B.; Dahlgren, S.; et al. Effects of bariatric surgery on cancer incidence in obese patients in Sweden (Swedish Obese Subjects Study): A prospective, controlled intervention trial. Lancet Oncol. 2009, 10, 653–662. [Google Scholar] [CrossRef]
- Anveden, Å.; Taube, M.; Peltonen, M.; Jacobson, P.; Andersson-Assarsson, J.C.; Sjöholm, K.; Svensson, P.A.; Carlsson, L.M.S. Long-term incidence of female-specific cancer after bariatric surgery or usual care in the Swedish Obese Subjects Study. Gynecol. Oncol. 2017, 145, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Adenan, D.M.; Jaafar, Z.; Jayapalan, J.J.; Abdul, A.A. Plasma antioxidants and oxidative stress status in obese women: Correlation with cardiopulmonary response. PeerJ 2020, 8, e9230. [Google Scholar] [CrossRef] [PubMed]
- Dludla, P.V.; Nkambule, B.B.; Jack, B.; Mkandla, Z.; Mutize, T.; Silvestri, S.; Orlando, P.; Tiano, L.; Louw, J.; Mazibuko-Mbeje, S.E. Inflammation and oxidative stress in an obese state and the protective effects of gallic acid. Nutrients 2018, 11, 23. [Google Scholar] [CrossRef] [PubMed]
- Otoda, T.; Takamura, T.; Misu, H.; Ota, T.; Murata, S.; Hayashi, H.; Takayama, H.; Kikuchi, A.; Kanamori, T.; Shima, K.R.; et al. Proteasome dysfunction mediates obesity-induced endoplasmic reticulum stress and insulin resistance in the liver. Diabetes 2013, 62, 811–824. [Google Scholar] [CrossRef]
- Wonisch, W.; Falk, A.; Sundl, I.; Winklhofer-Roob, B.M.; Lindschinger, M. Oxidative stress increases continuously with BMI and age with unfavourable profiles in males. Aging Male 2012, 15, 159–165. [Google Scholar] [CrossRef]
- Furukawa, S.; Fujita, T.; Shimabukuro, M.; Iwaki, M.; Yamada, Y.; Nakajima, Y.; Nakayama, O.; Makishima, M.; Matsuda, M.; Shimomura, I. Increased oxidative stress in obesity and its impact on metabolic syndrome. J. Clin. Investig. 2004, 114, 1752–1761. [Google Scholar] [CrossRef]
- Den Hartigh, L.J.; Omer, M.; Goodspeed, L.; Wang, S.; Wietecha, T.; O’Brien, K.D.; Han, C.Y. Adipocyte-specific deficiency of NADPH oxidase 4 delays the onset of insulin resistance and attenuates adipose tissue inflammation in obesity. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 466–475. [Google Scholar] [CrossRef]
- Bajaj, S.; Khan, A. Antioxidants and diabetes. Indian J. Endocrinol. Metab. 2012, 16, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Kacalska-Janssen, O.; Rajtar-Ciosek, A.; Zmaczyński, A.; Wyroba, J.; Milewicz, T.; Krzyczkowska-Sendrakowska, M.; Krzysiek, J. Markers of insulin resistance in perimenopausal women with endometrial pathology. Ginekol. Pol. 2013, 84, 922–929. [Google Scholar] [CrossRef] [PubMed]
- Mu, N.; Zhu, Y.; Wang, Y.; Zhang, H.; Xue, F. Insulin resistance: A significant risk factor of endometrial cancer. Gynecol. Oncol. 2012, 125, 751–757. [Google Scholar] [CrossRef]
- Stavropoulos, A.; Varras, M.; Philippou, A.; Vasilakaki, T.; Varra, V.K.; Varra, F.N.; Tsavari, A.; Lazaris, A.C.; Koutsilieris, M. Immunohistochemical expression of insulin-like growth factor-1Ec in primary endometrial carcinoma: Association with PTEN, p53 and survivin expression. Oncol. Lett. 2020, 20, 395. [Google Scholar] [CrossRef]
- Yasin, H.K.; Taylor, A.H.; Ayakannu, T. Evaluation of metformin performance on alloxan-induced diabetic rabbits. J. Med. Lifes 2022, 15, 405–407. [Google Scholar]
- Gadducci, A.; Biglia, N.; Tana, R.; Cosio, S.; Gallo, M. Metformin use and gynaecological cancers: A novel treatment option emerging from drug repositioning. Crit. Rev. Oncol. Hematol. 2016, 105, 73–83. [Google Scholar] [CrossRef]
- Yamana, H.; Kato, K.; Kobara, H.; Fujihara, S.; Fujita, K.; Namima, D.; Fujita, N.; Kobayashi, K.; Kamada, H.; Morishita, A.; et al. Metformin inhibits proliferation and tumor growth of QGP-1 pancreatic neuroendocrine tumor cells by inducing cell cycle arrest and apoptosis. Anticancer Res. 2020, 40, 121–132. [Google Scholar] [CrossRef]
- Liu, S.; Yue, C.; Chen, H.; Chen, Y.; Li, G. Metformin promotes beclin1-dependent autophagy to inhibit the progression of gastric cancer. Oncotargets Ther. 2020, 13, 4445–4455. [Google Scholar] [CrossRef]
- Mu, N.; Xu, T.; Gao, M.; Dong, M.; Tang, Q.; Hao, L.; Wang, G.; Li, Z.; Wang, W.; Yang, Y.; et al. Therapeutic effect of metformin in the treatment of endometrial cancer. Oncol. Lett. 2020, 20, 156. [Google Scholar] [CrossRef]
- Arima, R.; Marttila, M.; Hautakoski, A.; Arffman, M.; Sund, R.; Ilanne-Parikka, P.; Kangaskokko, J.; Läärä, E.; Puistola, U.; Hinkula, M. Antidiabetic medication, statins and the risk of endometrioid endometrial cancer in patients with type 2 diabetes. Gynecol. Oncol. 2017, 146, 636–641. [Google Scholar] [CrossRef]
- Mu, N.; Dong, M.; Liu, C.; Wang, X.; Cong, J.; Wang, L.; Wang, X.; Lakhani, I.; Liu, X.; Hou, J.; et al. Association between preoperative serum insulin levels and lymph node metastasis in endometrial cancer-a prospective cohort study. Cancer Med. 2018, 7, 1519–1527. [Google Scholar] [CrossRef] [PubMed]
- Barman, S.; Srinivasan, K. Attenuation of oxidative stress and cardioprotective effects of zinc supplementation in experimental diabetic rats. Br. J. Nutr. 2017, 117, 335–350. [Google Scholar] [CrossRef] [PubMed]
- Prabakaran, D.; Ashokkumar, N. Protective effect of esculetin on hyperglycemia-mediated oxidative damage in the hepatic and renal tissues of experimental diabetic rats. Biochimie 2013, 95, 366–373. [Google Scholar] [CrossRef]
- Koya, D.; Hayashi, K.; Kitada, M.; Kashiwagi, A.; Kikkawa, R.; Haneda, M. Effects of antioxidants in diabetes-induced oxidative stress in the glomeruli of diabetic rats. J. Am. Soc. Nephrol. 2003, 14, 250–253. [Google Scholar] [CrossRef]
- Oyenihi, O.R.; Brooks, N.L.; Oguntibeju, O.O. Effects of kolaviron on hepatic oxidative stress in streptozotocin induced diabetes. BMC Complement. Altern. Med. 2015, 15, 236. [Google Scholar] [CrossRef]
- Lee, T.Y.; Martinez-Outschoorn, U.E.; Schilder, R.J.; Kim, C.H.; Richard, S.D.; Rosenblum, N.G.; Johnson, J.M. Metformin as a Therapeutic Target in Endometrial Cancers. Front. Oncol. 2018, 8, 341. [Google Scholar] [CrossRef]
- Kyo, S.; Nakayama, K. Endometrial cancer as a metabolic disease with dysregulated PI3K signaling: Shedding light on novel therapeutic strategies. Int. J. Mol. Sci. 2020, 21, 6073. [Google Scholar] [CrossRef]
- Kokts-Porietis, R.; McNeil, J.; Nelson, G.S.; Courneya, K.S.; Cook, L.S.; Friedenreich, C.M. Prospective cohort study of metabolic syndrome and endometrial cancer survival. Gynecol. Oncol. 2020, 158, 727–733. [Google Scholar] [CrossRef]
- Rosato, V.; Zucchetto, A.; Bosetti, C.; Dal Maso, L.; Montella, M.; Pelucchi, C.; Negri, E.; Franceschi, S.; La Vecchia, C. Metabolic syndrome and endometrial cancer risk. Ann. Oncol. 2011, 22, 884–889. [Google Scholar] [CrossRef]
- Trabert, B.; Wentzensen, N.; Felix, A.S.; Yang, H.P.; Sherman, M.E.; Brinton, L.A. Metabolic syndrome and risk of endometrial cancer in the united states: A study in the SEER-medicare linked database. Cancer Epidemiol. Biomark. Prev. 2015, 24, 261–267. [Google Scholar] [CrossRef]
- Lin, Y.; Zhou, J.; Cao, L.; Xu, Q.; Hao, J.; Zhao, L.; Wang, J. Serum calcium is a novel parameter to assess metabolic syndrome in endometrial carcinoma. J. Gynecol. Oncol. 2019, 30, e12. [Google Scholar] [CrossRef] [PubMed]
- Martins, C.C.; Bagatini, M.D.; JSimões, L.B.; Cardoso, A.M.; Baldissarelli, J.; Dalenogare, D.P.; Dos Santos, D.L.; Schetinger, M.R.C.; Morsch, V.M. Increased oxidative stress and inflammatory markers contrasting with the activation of the cholinergic anti-inflammatory pathway in patients with metabolic syndrome. Clin. Biochem. 2021, 89, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Fernández-García, J.C.; Cardona, F.; Tinahones, F.J. Inflammation, oxidative stress and metabolic syndrome: Dietary modulation. Curr. Vasc. Pharmacol. 2013, 11, 906–919. [Google Scholar] [CrossRef] [PubMed]
- Maslov, L.N.; Naryzhnaya, N.V.; Boshchenko, A.A.; Popov, S.V.; Ivanov, V.V.; Oeltgen, P.R. Is oxidative stress of adipocytes a cause or a consequence of the metabolic syndrome? J. Clin. Translat. Endocrinol. 2018, 15, 1–5. [Google Scholar] [CrossRef]
- Aboonabi, A.; Meyer, R.R.; Singh, I. The association between metabolic syndrome components and the development of atherosclerosis. J. Hum. Hypertens. 2019, 33, 844–855. [Google Scholar] [CrossRef]
- Beydoun, M.A.; Canas, J.A.; Beydoun, H.A.; Chen, X.; Shroff, M.R.; Zonderman, A.B. Serum antioxidant concentrations and metabolic syndrome are associated among U.S. adolescents in recent national surveys. J. Nutr. 2012, 14, 1693–1704. [Google Scholar] [CrossRef]
- Kanagasabai, T.; Alkhalaqi, K.; Churilla, J.R.; Ardern, C.I. The association between metabolic syndrome and serum concentrations of micronutrients, inflammation, and oxidative stress outside of the clinical reference ranges: A cross-sectional study. Metab. Syndr. Relat. Disord. 2019, 17, 29–36. [Google Scholar] [CrossRef]
- Fader, A.N.; Arriba, L.N.; Frasure, H.E.; von Gruenigen, V.E. Endometrial cancer and obesity: Epidemiology, biomarkers, prevention and survivorship. Gynecol. Oncol. 2009, 114, 121–127. [Google Scholar] [CrossRef]
- Sun, N.N.; Wu, T.Y.; Chau, C.F. Natural dietary and herbal products in anti-obesity treatment. Molecules 2016, 21, 1351. [Google Scholar] [CrossRef]
- Wang, S.; Moustaid-Moussa, N.; Chen, L.; Mo, H.; Shastri, A.; Su, R.; Bapat, P.; Kwun, I.; Shen, C.L. Novel insights of dietary polyphenols and obesity. J. Nutr. Biochem. 2014, 25, 1–18. [Google Scholar] [CrossRef]
- Jakab, J.; Miškić, B.; Mikšić, S.; Juranić, B.; Ćosić, V.; Schwarz, D.; Včev, A. Adipogenesis as a potential anti-obesity target: A review of pharmacological treatment and natural products. Diabetes Metab. Syndr. Obes. 2021, 14, 67–83. [Google Scholar] [CrossRef] [PubMed]
- Vasileva, L.V.; Savova, M.S.; Amirova, K.M.; Balcheva-Sivenova, Z.; Ferrante, C.; Orlando, G.; Wabitsch, M.; Georgiev, M.I. Caffeic and chlorogenic acids synergistically activate browning program in human adipocytes: Implications of AMPK- and PPAR-mediated pathways. Int. J. Mol. Sci. 2020, 21, 9740. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Ding, Y.; Dai, X.; Wang, J.; Li, Y. Epigallocatechin-3-gallate protects pro-inflammatory cytokine induced injuries in insulin-producing cells through the mitochondrial pathway. Eur. J. Pharmacol. 2011, 670, 311–316. [Google Scholar] [CrossRef]
- Fu, Q.Y.; Li, Q.S.; Lin, X.M.; Qiao, R.Y.; Yang, R.; Li, X.M.; Dong, Z.B.; Xiang, L.P.; Zheng, X.Q.; Lu, J.L.; et al. Antidiabetic effects of tea. Molecules 2017, 22, 849. [Google Scholar] [CrossRef]
- Striegel, L.; Kang, B.; Pilkenton, S.J.; Rychlik, M.; Apostolidis, E. Effect of black tea and black tea pomace polyphenols on α-glucosidase and α-amylase inhibition, relevant to type 2 diabetes prevention. Front. Nutr. 2015, 2, 3. [Google Scholar] [CrossRef] [PubMed]
- Tan, A.A.; Ozdemir, D.; Bestepe, N.; Dellal, F.D.; Bilginer, M.C.; Faki, S.; Bicer, C.; Ersoy, R.; Cakir, B. Low rate of latent autoimmune diabetes in adults (LADA) in patients followed for type 2 diabetes: A single center’s experience in Turkey. Arch. Endocrinol. Metab. 2020, 64, 584–590. [Google Scholar]
- Simos, Y.V.; Verginadis, I.; Toliopoulos, I.K.; Velalopoulou, A.P.; Karagounis, I.V.; Karkabounas, S.C.; Evangelou, A.M. Effects of catechin and epicatechin on superoxide dismutase and glutathione peroxidase activity, in vivo. Redox Rep. 2012, 17, 181–186. [Google Scholar] [CrossRef]
- Shannon, E.; Jaiswal, A.K.; Abu-Ghannam, N. Polyphenolic content and antioxidant capacity of white, green, black, and herbal teas: A kinetic study. Food Res. 2018, 2, 1–11. [Google Scholar] [CrossRef]
- Xu, X.J.; Gauthier, M.S.; Hess, D.T.; Apovian, C.M.; Cacicedo, J.M.; Gokce, N.; Farb, M.; Valentine, R.J.; Ruderman, N.B. Insulin sensitive and resistant obesity in humans: AMPK activity, oxidative stress, and depot-specific changes in gene expression in adipose tissue. J. Lipid Res. 2012, 53, 792–801. [Google Scholar] [CrossRef]
- Noronha, N.Y.; Pinhel, M.A.S.; Nicoletti, C.F.; Quinhoneiro, D.C.G.; Pinhanelli, V.C.; Oliveira, B.A.P.; Cortes-Oliveira, C.; Delfino, H.B.P.; Wolf, L.S.; Frantz, F.G.; et al. Green tea supplementation improves oxidative stress biomarkers and modulates IL-6 circulating levels in obese women. Nutr. Hosp. 2019, 36, 583–588. [Google Scholar]
- Nieto-Vazquez, I.; Fernández-Veledo, S.; Krämer, D.K.; Vila-Bedmar, R.; Garcia-Guerra, L.; Lorenzo, M. Insulin resistance associated to obesity: The link TNF-alpha. Arch. Physiol. Biochem. 2008, 114, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.; Tsai, T.; Kao, Y.; Hwang, K.; Tseng, T.; Chou, P. Effect of green tea extract on obese women: A randomized, double-blind, placebo-controlled clinical trial. Clin. Nutr. 2008, 27, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Hursel, R.; Westerterp-Plantenga, M.S. Catechin—And caffeine-rich teas for control of body weight in humans. Am. J. Clin. Nutr. 2013, 98, 1682–1693. [Google Scholar] [CrossRef]
- Hsu, C.H.; Liao, Y.L.; Lin, S.C.; Tsai, T.H.; Huang, C.J.; Chou, P. Does supplementation with green tea extract improve insulin resistance in obese type 2 diabetics? A randomized, double-blind, and placebo-controlled clinical trial. Altern. Med. Rev. 2011, 16, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Pervin, M.; Goto, S.; Isemura, M.; Nakamura, Y. Beneficial effects of tea and the green tea catechin epigallocatechin-3-gallate on obesity. Molecules 2016, 21, 1305. [Google Scholar] [CrossRef]
- Hsu, T.F.; Kusumoto, A.; Abe, K.; Hosoda, K.; Kiso, Y.; Wang, M.F.; Yamamoto, S. Polyphenol-enriched oolong tea increases fecal lipid excretion. Europ. J. Clin. Nutr. 2006, 60, 133–136. [Google Scholar] [CrossRef]
- Huang, L.H.; Liu, C.Y.; Wang, L.Y.; Huang, C.J.; Hsu, C.A. Effects of green tea extract on overweight and obese women with high levels of low density-lipoprotein-cholesterol (LDL-C): A randomised, double-blind, and cross-over placebo-controlled clinical trial. BMC Complement. Altern. Med. 2018, 18, 294. [Google Scholar] [CrossRef]
- Chen, I.J.; Liu, C.Y.; Chiu, J.P.; Hsu, C.H. Therapeutic effect of high-dose green tea extract on weight reduction: A randomized, double-blind, placebo-controlled clinical trial. Clin. Nutr. 2016, 35, 592–599. [Google Scholar] [CrossRef]
- Dostal, A.M.; Samavat, H.; Espejo, L.; Arikawa, A.Y.; Stendell-Hollis, N.R.; Kurzer, M.S. Green tea extract and catechol-O-methyltransferase genotype modify fasting serum insulin and plasma adiponectin concentrations in a randomized controlled trial of overweight and obese postmenopausal women. J. Nutr. 2016, 146, 38–45. [Google Scholar] [CrossRef]
- Yang, C.S.; Zhang, J.; Zhang, L.; Huang, J.; Wang, Y. Mechanisms of body weight reduction and metabolic syndrome alleviation by tea. Mol. Nutr. Food Res. 2016, 60, 160–174. [Google Scholar] [CrossRef]
- Cardoso, G.A.; Salgado, J.M.; Cesar Mde, C.; Donado-Pestana, C.M. The effects of green tea consumption and resistance training on body composition and resting metabolic rate in overweight or obese women. J. Med. Food 2013, 16, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Stepaniak, U.; Micek, A.; Topor-Mądry, R.; Pikhart, H.; Szafraniec, K.; Pająk, A. Association of daily coffee and tea consumption and metabolic syndrome: Results from the Polish arm of the HAPIEE study. Eur. J. Nut. 2015, 54, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Gamal, A.M.; Sabrin, I.M.R.; Ehab, S.E.; Riham, S.E.D. Natural anti-obesity agents. Bull. Fac. Pharm. Cairo Univ. 2014, 52, 269–284. [Google Scholar]
- Yu, J.; Song, P.; Perry, R.; Penfold, C.; Cooper, A.R. The effectiveness of green tea or green tea extract on insulin resistance and glycemic control in type 2 diabetes mellitus: A meta-analysis. Diabetes Metab. J. 2017, 41, 251–262. [Google Scholar] [CrossRef] [PubMed]
- InterAct Consortium; van Woudenbergh, G.J.; Kuijsten, A.; Drogan, D.; van der A, D.L.; Romaguera, A.D.; Ardanaz, E.; Amiano, P.; Barricarte, A.; Beulens, J.W.; et al. Tea consumption and incidence of type 2 diabetes in Europe: The EPIC-InterAct case-cohort study. PLoS ONE 2012, 7, e36910. [Google Scholar]
- Yang, W.S.; Wang, W.Y.; Fan, W.Y.; Deng, Q.; Wang, X. Tea consumption and risk of type 2 diabetes: A dose-response meta-analysis of cohort studies. Brit. J. Nutr. 2014, 111, 1329–1339. [Google Scholar] [CrossRef]
- Iso, H.; Date, C.; Wakai, K.; Fukui, M.; Tamakoshi, A. JACC Study Group. The relationship between green tea and total caffeine intake and risk for self-reported type 2 diabetes among Japanese adults. Ann. Int. Med. 2006, 144, 554–562. [Google Scholar] [CrossRef]
- Nguyen, C.T.; Lee, A.H.; Pham, N.M.; Do, V.V.; Ngu, N.D.; Tran, B.Q.; Binns, C. Habitual tea drinking associated with a lower risk of type 2 diabetes in Vietnamese adults. Asia Pac. J. Clin. Nutr. 2018, 27, 701–706. [Google Scholar]
- Nie, J.; Yu, C.; Guo, Y.; Pei, P.; Chen, L.; Pang, Y.; Du, H.; Yang, L.; Chen, Y.; Yan, S.; et al. Tea consumption and long-term risk of type 2 diabetes and diabetic complications: A cohort study of 0.5 million Chinese adults. Am. J. Clin. Nutr. 2021, 114, 194–202. [Google Scholar] [CrossRef]
- Ni, D.; Ai, Z.; Munoz-Sandoval, D.; Suresh, R.; Ellis, P.R.; Yu-qiong, C.; Sharp, P.A.; Butterworth, P.J.; Yu, Z.; Corpe, C. Inhibition of the facilitative sugar transporters (GLUTs) by tea extracts and catechins. FASEB J. 2020, 34, 10010–19995. [Google Scholar] [CrossRef]
- Samarghandian, S.; Azimi-Nezhad, M.; Farkhondeh, T. Catechin treatment ameliorates diabetes and its complications in streptozotocin-induced diabetic rats. Dose Response 2017, 15. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.X.; Xu, Y.L.; Li, S.H.; Hui, R.; Wu, Y.J.; Huang, X.H. Effects of green tea catechins with or without caffeine on glycemic control in adults: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2013, 97, 750–762. [Google Scholar] [CrossRef] [PubMed]
- Toolsee, N.A.; Aruoma, O.I.; Gunness, T.K.; Kowlessur, S.; Dambala, V.; Murad, F.; Googoolye, K.; Daus, D.; Indelicato, J.; Rondeau, P.; et al. Effectiveness of green tea in a randomized human cohort: Relevance to diabetes and its complications. BioMed Res. Int. 2013, 2013, 412379. [Google Scholar] [CrossRef] [PubMed]
- Bazyar, H.; Hosseini, S.A.; Saradar, S.; Mombaini, D.; Allivand, M.; Labibzadeh, M.; Alipour, M. Effects of epigallocatechin-3-gallate of Camellia sinensis leaves on blood pressure, lipid profile, atherogenic index of plasma and some inflammatory and antioxidant markers in type 2 diabetes mellitus patients: A clinical trial. J. Complement. Integr. Med. 2020, 18, 405–411. [Google Scholar] [CrossRef]
- Li, W.; Zhu, C.; Liu, T.; Zhang, W.; Liu, X.; Li, P.; Zhu, T. Epigallocatechin-3-gallate ameliorates glucolipid metabolism and oxidative stress in type 2 diabetic rats. Diabetes Vasc. Dis. Res. 2020, 17. [Google Scholar] [CrossRef]
- Wu, Y.; Du, W.; Shi, L.; Liang, Q.; Zou, X. Vasculoprotective effects of water extracts of black, green and dark tea in vitro. Nat. Prod. Commun. 2017, 12, 387–390. [Google Scholar] [CrossRef]
- Han, M.; Zhao, G.; Wang, Y.; Wagg, D.; Sun, F.; Ning, J.; Wan, X.; Zhang, J. Safety and anti-hyperglycemic efficacy of various tea types in mice. Sci. Rep. 2016, 6, 31703. [Google Scholar] [CrossRef]
- Liu, J.; Lv, Y.-J.; Pan, J.X.; Jiang, J.L.; Zhu, Y.J.; Zhang, S.K. Effects of tea polyphenols and EGCG on glucose metabolism and intestinal flora in diabetic mice fed a cornstarch-based functional diet. Food Sci. Tech-Brasil. 2022, 42, e50821. [Google Scholar] [CrossRef]
- El-Ghazaly, M.N.; Mostafa, O.E.; Ali, Y.M.; Abdelrahman, A.M.; Fadal, N. Improvement of diabetic rats using green tea wealthy with catechin and inulin. Afr. J. Biol. Sci. 2020, 1, 63–77. [Google Scholar] [CrossRef]
- Renno, W.M.; Abdeen, S.; Alkhalaf, M.; Asfar, S. Effect of green tea on kidney tubules of diabetic rats. Brit. J. Nutr. 2008, 100, 652–659. [Google Scholar] [CrossRef]
- Hosseini, S.; Alipour, M.; Zakerkish, M.; Cheraghian, B.; Ghandil, P. Effects of epigallocatechin gallate on total antioxidant capacity, biomarkers of systemic low-grade inflammation and metabolic risk factors in patients with type 2 diabetes mellitus: The role of FTO-rs9939609 polymorphism. Arch. Med. Sci. 2020, 17, 1722–1729. [Google Scholar] [CrossRef]
- Zhang, L.; Zheng, Y.; Cheng, X.; Meng, M.; Luo, Y.; Li, B. The anti-photoaging effect of antioxidant collagen peptides from silver carp (Hypophthalmichthys molitrix) skin is preferable to tea polyphenols and casein peptides. Food Funct. 2017, 8, 1698–1707. [Google Scholar] [CrossRef] [PubMed]
- Hamadouche, N.A.; Lazeb, H.; Kaddouri, A.; Guellil, H.; Slimani, M.; Aoues, A. Ameliorated effects of green tea extract on lead induced kidney toxicity in rats. Algerian J. Nat. Prod. 2015, 3, 130–137. [Google Scholar]
- Mehana, E.; Meki, A.R.; Fazili, K.M. Ameliorated effects of green tea extract on lead induced liver toxicity in rats. Exp. Toxicol. Pathol. 2012, 64, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Szulińska, M.; Stępień, M.; Kręgielska-Narożna, M.; Suliburska, J.; Skrypnik, D.; Bąk-Sosnowska, M.; Kujawska-Łuczak, M.; Grzymisławska, M.; Bogdański, P. Effects of green tea supplementation on inflammation markers, antioxidant status and blood pressure in NaCl-induced hypertensive rat model. Food Nutr. Res. 2017, 61, 1295525. [Google Scholar] [CrossRef] [PubMed]
- Abolfathi, A.A.; Mohajeri, D.; Rezaie, A.; Nazeri, M. Protective effects of green tea extract against hepatic tissue injury in streptozotocin-induced diabetic rats. Evid.-Based Complement. Altern. 2012, 1, 740671. [Google Scholar] [CrossRef]
- Abdel-Maneim, H.A.M.; Meki, A.R.; Salem, A.M.A.; Mobasher, A.; Lutfi, M.F. The protective effect of green tea against lead toxicity in rats kidney. Asian J. Biomed. Pharm. Sci. 2014, 4, 30–34. [Google Scholar] [CrossRef]
- Bornhoeft, J.; Castaneda, D.; Nemoseck, T.; Wang, P.; Henning, S.M.; Hong, M.Y. The protective effects of green tea polyphenols: Lipid profile, inflammation, and antioxidant capacity in rats fed an atherogenic diet and dextran sodium sulfate. J. Med. Food 2012, 15, 726–732. [Google Scholar] [CrossRef]
- Khalaf, A.A.; Moselhy, W.A.; Abdel-Hamed, M.I. The protective effect of green tea extract on lead induced oxidative and DNA damage on rat brain. Neurotoxicology 2012, 33, 280–289. [Google Scholar] [CrossRef]
- Khan, G.; Haque, S.E.; Anwer, T.; Ahsan, M.N.; Safhi, M.M.; Alam, M.F. Cardioprotective effect of green tea extract on doxorubicin-induced cardiotoxicity in rats. Acta Pol. Pharm. 2014, 71, 861–868. [Google Scholar]
- Hegazi, M.M.; Mostafa, H.A.; Hegazi, M.A.M.; Elsaid, A.S.I. Effect of green tea and curcumin on the oxidative stress caused by gasoline in male mice brain. Egypt. J. Exp. Biol. 2009, 5, 473–480. [Google Scholar]
- Mosbah, R.; Yousef, M.I.; Mantovani, A. Nicotine-induced reproductive toxicity, oxidative damage, histological changes and haematotoxicity in male rats: The protective effects of green tea extract. Exp. Toxicol. Pathol. 2015, 67, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Meki, A.R.; Alghasham, A.; El-Deeb, E.S. Effect of green tea extract on lead toxicity in different organs of rats. Int. J. Health Sci. 2011, 5, 12–15. [Google Scholar]
- Hamed, E.A.; Meki, A.R.M.A.; Abd El-Mottaleb, N.A. Protective effect of green tea on lead-induced oxidative damage in rat’s blood and brain tissue homogenates. J. Physiol. Biochem. 2010, 66, 143–151. [Google Scholar] [CrossRef]
- Hong, I.S.; Lee, H.Y.; Kim, H.P. Anti-oxidative effects of rooibos tea (Aspalathus linearis) on immobilization-induced oxidative stress in rat brain. PLoS ONE 2014, 9, e87061. [Google Scholar] [CrossRef]
- Su, J.; Wang, X.; Song, W.; Bai, X.; Li, C. Reducing oxidative stress and hepatoprotective effect of water extracts from Pu-erh tea on rats with high-fat diet. Food Sci. Hum. Wellness 2016, 5, 199–206. [Google Scholar] [CrossRef]
- Pérez-Jiméneza, A.; Peresa, H.; Cruz, V.; AiresOliva-Telesm, R. The effect of hypoxia on intermediary metabolism and oxidative status in gilthead sea bream (Sparus aurata) fed on diets supplemented with methionine and white tea. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2012, 155, 506–516. [Google Scholar] [CrossRef]
- Silveira, A.C.; Rato, L.; Oliveira, P.F.; Alves, M.G.; Silva, B.M. White tea intake abrogates markers of streptozotocin-induced prediabetes oxidative stress in rat lungs’. Molecules 2021, 26, 3894. [Google Scholar] [CrossRef]
- Rai, A.; Gill, M.; Kinra, M.; Shetty, R.; Krishnadas, N.; Rao, C.M.; Sumalatha, S.; Kumar, N. Catechin ameliorates depressive symptoms in Sprague Dawley rats subjected to chronic unpredictable mild stress by decreasing oxidative stress. Biom. Rep. 2019, 11, 79–84. [Google Scholar] [CrossRef]
- Shanmugam, T.; Abdulla, S.; Yakulasamy, V.; Selvaraj, M.; Mathan, R. A mechanism underlying the neurotoxicity induced by sodium fluoride and its reversal by epigallocatechin gallate in the rat hippocampus: Involvement of NrF2/Keap-1 signaling pathway. J. Basic Appl. Zool. 2018, 79, 17. [Google Scholar] [CrossRef]
- Ahmed, N.A.; Radwan, N.M.; Aboul Ezz, H.S.; Salama, N.A. The antioxidant effect of green tea mega EGCG against electromagnetic radiation-induced oxidative stress in the hippocampus and striatum of rats. Electromagn. Biol. Med. 2017, 36, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Chiou, Y.S.; Pan, M.H.; Ho, C.T.; Shahidi, F. Protective effects of epigallocatechin gallate (EGCG) derivatives on azoxymethane-induced colonic carcinogenesis in mice. J. Funct. Foods 2012, 4, 323–330. [Google Scholar] [CrossRef]
- Chiou, Y.S.; Sang, S.; Cheng, K.H.; Ho, C.T.; Wang, Y.J.; Pan, M.H. Peracetylated (−)-epigallocatechin-3-gallate (AcEGCG) potently prevents skin carcinogenesis by suppressing the PKD1-dependent signaling pathway in CD34+ skin stem cells and skin tumors. Carcinogenesis 2013, 34, 1315–1322. [Google Scholar] [CrossRef]
- Chandronitha, C.; Ananthi, S.; Ramakrishnan, G.; Lakshmisundaram, R.; Gayathri, V.; Vasanthi, H.R. Protective role of tannin-rich fraction of Camellia sinensis in tissue arsenic burden in Sprague Dawley rats. Hum. Exp. Toxicol. 2010, 29, 705–719. [Google Scholar] [CrossRef]
- Winiarska-Mieczan, A. Protective effect of tannic acid on the brain of adult rats exposed to cadmium and lead. Environ. Toxicol. Phar. 2013, 36, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Ashafaq, M.; Tabassum, H.; Vishnoi, S.; Salman, M.; Raisuddin, S.; Parvez, S. Tannic acid alleviates lead acetate-induced neurochemical perturbations in rat brain. Neurosci. Lett. 2016, 617, 94–100. [Google Scholar] [CrossRef]
- Khan, A.; Ikram, M.; Muhammad, T.; Park, J.; Kim, M.O. Caffeine modulates cadmium-induced oxidative stress, neuroinflammation, an cognitive impairments by regulating Nrf-2/HO-1 in vivo and in vitro. J. Clin. Med. 2019, 8, 680. [Google Scholar] [CrossRef]
- El-Beshbishy, H.A.; Tork, O.M.; El-Bab, M.F.; Autifi, M.A. Antioxidant and antiapoptotic effects of green tea polyphenols against azathioprine-induced liver injury in rats. Pathophysiology 2011, 18, 125–135. [Google Scholar] [CrossRef]
- Yuan, C.F.; Li, Z.H.; Peng, F.; Xiao, F.X.; Ren, D.M.; Xue, H.; Chen, T.; Mushtaq, G.; Kamal, M.A. Combination of selenium-enriched green tea polysaccharides and Huo-ji polysaccharides synergistically enhances antioxidant and immune activity in mice. J. Sci. Food Agric. 2015, 95, 3211–3217. [Google Scholar] [CrossRef]
- Zhou, F.; Shen, T.; Duan, T.; Xu, Y.Y.; Khor, S.C.; Li, J.; Ge, J.; Zheng, Y.F.; Hsu, S.; De, S.J. Antioxidant effects of lipophilic tea polyphenols on diethylnitrosamine / phenobarbital-induced hepatocarcinogenesis in rats. In Vivo 2014, 28, 495–503. [Google Scholar]
- Yang, J.J.; Chen, B.; Gu, Y. Pharmacological evaluation of tea polysaccharides with antioxidant activity in gastric cancer mice. Carbohyd. Polym. 2012, 90, 943–947. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.M. Evaluation of antioxidant activity of polysaccharides isolated from Camellia sinensis (tea) in exhausting training mice. J. Med. Plants Res. 2011, 5, 791–795. [Google Scholar]
- Xu, R.; Ye, H.; Sun, Y.; Tu, Y.; Zeng, X. Preparation, preliminary characterization, antioxidant, hepatoprotective and antitumor activities of polysaccharides from the flower of tea plant (Camellia sinensis). Food Chem. Toxicol. 2012, 50, 2473–2480. [Google Scholar] [CrossRef]
- Marnewick, J.L.; Rautenbach, F.; Venter, I.; Neethling, H.; Blackhurst, D.M.; Wolmarans, P.; Macharia, M. Effects of rooibos (Aspalathus linearis) on oxidative stress and biochemical parameters in adults at risk for cardiovascular disease. J. Ethnopharmacol. 2011, 33, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Bertipaglia de Santana, M.; Mandarino, M.G.; Cardoso, J.R.; Dichi, I.; Dichi, J.B.; Camargo, A.E.; Fabris, B.A.; Rodrigues, R.J.; Fatel, E.C.S.; Nixdorf, S.L.; et al. Association between soy and green tea (Camellia sinensis) diminishes hypercholesterolemia and increases total plasma antioxidant potential in dyslipidemic subjects. Nutrition 2008, 24, 562–568. [Google Scholar] [CrossRef]
- Villaño, D.; Lettieri-Barbato, D.; Guadagni, F.; Schmid, M.; Serafini, M. Effect of acute consumption of oolong tea on antioxidant parameters in healthy individuals. Food Chem. 2012, 132, 2102–2106. [Google Scholar] [CrossRef]
- Choi, S.W.; Yeung, V.T.F.; Collins, A.R.; Benzie, I.F.F. Redox-linked effects of green tea on DNA damage and repair, and influence of microsatellite polymorphism in HMOX-1: Results of a human intervention trial. Mutagenesis 2015, 30, 129–137. [Google Scholar] [CrossRef]
- Basu, A.; Betts, N.M.; Mulugeta, A.; Tong, C.; Newman, E.; Lyons, T.J. Green tea supplementation increases glutathione and plasma antioxidant capacity in adults with the metabolic syndrome. Nutr. Res. 2013, 33, 180–187. [Google Scholar] [CrossRef]
- Zhang, J.; Cai, S.; Li, J.; Xiong, L.; Tian, L.; Liu, J.; Huang, J.; Liu, Z. Neuroprotective effects of theaflavins against oxidative stress-induced apoptosis in PC12 cells. Neurochem. Res. 2016, 41, 3364–3372. [Google Scholar] [CrossRef]
- Feng, B.; Fang, Y.; Wei, S.M. Effect and mechanism of epigallocatechin-3-gallate (EGCG) against the hydrogen peroxide-induced oxidative damage in human dermal fibroblasts. J. Cosmet. Sci. 2013, 64, 35–44. [Google Scholar]
- Fu, Y.; Zheng, S.; Lu, S.C.; Chen, A. Epigallocatechin-3-gallate inhibits growth of activated hepatic stellate cells by enhancing the capacity of glutathione synthesis. Mol. Pharmacol. 2008, 73, 1465–1473. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Cao, D.; Cui, W.; Ji, M.; Qian, X.; Zhong, L. Molecular bases of thioredoxin and thioredoxin reductase-mediated prooxidant actions of (−)-epigallocatechin-3-gallate. Free Radic Biol. Med. 2010, 49, 2010–2018. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S. Comparative efficacy of epigallocatechin-3-gallate against H2O2-induced ROS in cervical cancer biopsies and HeLa cell lines. Contemp. Oncol. 2017, 21, 209–212. [Google Scholar]
- Serafini, M.; Ghiselli, A.; Ferro-Luzzi, A. In vivo antioxidant effect of green and black tea in man. Eur. J. Clin. Nutr. 1996, 50, 28–32. [Google Scholar]
- Sung, H.; Nah, J.; Chun, S.; Park, H.; Yang, S.E.; Min, W.K. In vivo antioxidant effect of green tea. Eur. J. Clin. Nutr. 2000, 54, 527–529. [Google Scholar] [CrossRef]
- Leenen, R.; Roodenburg, A.J.; Tijburg, L.B.; Wiseman, S.A. A single dose of tea with or without milk increases plasma antioxidant activity in humans. Eur. J. Clin. Nutr. 2000, 54, 87–92. [Google Scholar] [CrossRef]
- Gawlik, M.; Czajka, A. The effect of green, black and white tea on the level of α and γ tocopherols in free radical-induced oxidative damage of human red blood cells. Acta Pol. Pharm. 2007, 64, 159–164. [Google Scholar]
- Mazzanti, G.; Di Sotto, A.; Vitalone, A. Hepatotoxicity of green tea: An update. Arch. Toxicol. 2015, 89, 1175–1191. [Google Scholar] [CrossRef]
- Sánchez, E.P.; Vargas, M.E.R.; Gutiérrez, J.C.R. Hepatotoxicity due to green tea consumption (Camellia Sinensis): A review. Rev. Colomb. Gastroenterol. 2013, 28, 46–52. [Google Scholar]
- Reddy, M.A.; Kumar, B.K.; Boobalan, G.; Reddy, M.K.; Kumar, C.S.V.S.; Reddy, G.A.; Lakshman, M. Hepatoprotective potential of green tea extract against experimental hepatotoxicity in rats. Indian J. Pharm. Sci. 2017, 79, 58–64. [Google Scholar] [CrossRef]
- Ali, A.H.A. Hepatoprotective effect of green tea extractagainst cyclophosphamide induced liver injury in albino rats. Forensic Med. Anat. Res. 2018, 6, 11–19. [Google Scholar] [CrossRef]
- Kakuta, Y.; Nakaya, N.; Nagase, S.; Fujita, M.; Koizumi, T.; Okamura, C.; Niikura, H.; Ohmori, K.; Kuriyama, S.; Tase, T.; et al. Case-control study of green tea consumption and the risk of endometrial endometrioid adenocarcinoma. Cancer Causes Control 2009, 20, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Manohar, M.; Fatima, I.; Saxena, R.; Chandra, V.; Sankhwar, P.L.; Dwivedi, A. (-)-Epigallocatechin-3-gallate induces apoptosis in human endometrial adenocarcinoma cells via ROS generation and p38 MAP kinase activation. J. Nutr. Biochem. 2013, 4, 940–947. [Google Scholar] [CrossRef] [PubMed]
- Park, S.B.; Bae, J.W.; Kim, J.M.; Lee, S.G.; Han, M. Antiproliferative and apoptotic effect of epigallocatechin-3-gallate on Ishikawa cells is accompanied by sex steroid receptor downregulation. Int. J. Mol. Med. 2012, 30, 1211–1218. [Google Scholar] [CrossRef] [PubMed]
- Laschke, M.W.; Schwender, C.; Scheuer, C.; Vollmar, B.; Menger, M.D. Epigallocatechin-3-gallate inhibits estrogen-induced activation of endometrial cells in vitro and causes regression of endometriotic lesions in vivo. Hum. Repr. 2008, 23, 2308–2318. [Google Scholar] [CrossRef]
- Xu, H.; Lui, W.T.; Chu, C.Y.; Ng, P.S.; Wang, C.C.; Rogers, M.S. Anti-angiogenic effects of green tea catechin on an experimental endometriosis mouse model. Hum. Reprod. 2009, 24, 608–618. [Google Scholar] [CrossRef]
- Man, G.C.W.; Wang, J.; Song, Y.; Wong, J.H.; Zhao, Y.; Lau, T.S.; Leung, K.T.; Chan, T.H.; Wang, H.; Kwong, J.; et al. Therapeutic potential of a novel prodrug of green tea extract in induction of apoptosis via ERK/JNK and Akt signaling pathway in human endometrial cancer. BMC Cancer 2020, 20, 964. [Google Scholar] [CrossRef]
- Qin, J.; Fu, M.; Wang, J.; Huang, F.; Liu, H.; Huangfu, M.; Yu, D.; Liu, H.; Li, X.; Guan, X.; et al. PTEN/AKT/mTOR signaling mediates anticancer effects of epigallocatechin-3-gallate in ovarian cancer. Oncol. Rep. 2020, 43, 1885–1896. [Google Scholar] [CrossRef]
- Tang, N.P.; Li, H.; Qiu, Y.L.; Zhou, G.M.; Ma, J. Tea consumption and risk of endometrial cancer: A metaanalysis. Am. J. Obstet. Gynecol. 2009, 201, 605. [Google Scholar] [CrossRef]
- Inoue, F.; Sone, K.; Toyohara, Y.; Takahashi, Y.; Kukita, A.; Hara, A.; Taguchi, A.; Tanikawa, M.; Tsuruga, T.; Osuga, Y. Targeting Epigenetic Regulators for Endometrial Cancer Therapy: Its Molecular Biology and Potential Clinical Applications. Int. J. Mol. Sci. 2021, 22, 2305. [Google Scholar] [CrossRef]
- Du, Q.; Luu, P.L.; Stirzaker, C.; Clark, S.J. Methyl-CpG-binding domain proteins: Readers of the epigenome. Epigenomics 2015, 7, 1051–1053. [Google Scholar] [CrossRef] [PubMed]
- Parry, L.; Clarke, A.R. The Roles of the Methyl-CpG Binding Proteins in Cancer. Genes Cancer 2011, 2, 618–630. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Yu, X.; Zhang, M.; Guo, W. Pan-cancer analysis of m5C regulator genes reveals consistent epigenetic landscape changes in multiple cancers. World J. Surg. Oncol. 2021, 19, 224. [Google Scholar] [CrossRef] [PubMed]
- Nombela, P.; Miguel-López, B.; Blanco, S. The role of m6A, m5C and Ψ RNA modifications in cancer: Novel therapeutic opportunities. Mol. Cancer 2021, 20, 18. [Google Scholar] [CrossRef] [PubMed]
- Barciszewska, A.M.; Giel-Pietraszuk, M.; Perrigue, P.M.; Naskręt-Barciszewska, M. Total DNA methylation changes reflect random oxidative DNA damage in gliomas. Cells 2019, 8, 1065. [Google Scholar] [CrossRef] [PubMed]
- Sabharwal, S.S.; Schumacker, P.T. Mitochondrial ROS in cancer: Initiators, amplifiers or an Achilles’ heel? Nat. Rev. Cancer 2014, 14, 709–721. [Google Scholar] [CrossRef]
- Perillo, B.; Di Donato, M.; Pezone, A.; Di Zazzo, E.; Giovannelli, P.; Galasso, G.; Castoria, G.; Migliaccio, A. ROS in cancer therapy: The bright side of the moon. Exp. Mol. Med. 2020, 52, 192–203. [Google Scholar] [CrossRef]
- Postawski, K.; Prządka-Rabaniuk, D.; Piersiak, T. 8-oxo-7,8-dihydroguanine level—The DNA oxidative stress marker—Recognized by fluorescence image analysis in sporadic uterine adenocarcinomas in women. Ginekol. Pol. 2013, 84, 44–50. [Google Scholar] [CrossRef]
- Romanek-Piva, K.; Gałczyński, K.; Adamiak-Godlewska, A.; Rechberger, T.; Postawski, K. DNA methylation and DNA methyltransferase (DNMT1) activity pattern in endometrial carcinoma. Ginekol. Pol. 2016, 87, 6–10. [Google Scholar] [CrossRef]
- Rodriguez, A.C.; Blanchard, Z.; Maurer, K.A.; Gertz, J. Estrogen signaling in endometrial cancer: A key oncogenic pathway with several open questions. Horm. Cancer 2019, 10, 51–63. [Google Scholar] [CrossRef]
- Tian, H.; Gao, Z.; Wang, G.; Li, H.; Zheng, J. Estrogen potentiates reactive oxygen species (ROS) tolerance to initiate carcinogenesis and promote cancer malignant transformation. Tumor Biol. 2016, 37, 141–150. [Google Scholar] [CrossRef]
- Chen, J.; Delannoy, M.; Odwin, S.; He, P.; Trush, M.A.; Yager, J.D. Enhanced mitochondrial gene transcript, ATP, Bcl-2 protein levels, and altered glutathione distribution in ethinyl estradiol-treated cultured female rat hepatocytes. Toxicol. Sci. 2003, 75, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Arita, A.; Costa, M. Oxidative stress and the epigenome in human disease. J. Genet. Genome Res. 2014, 1, 2. [Google Scholar] [CrossRef]
- You, J.S.; Jones, P.A. Cancer genetics and epigenetics: Two sides of the same coin? Cancer Cell 2012, 22, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Reen, J.K.; Yadav, A.K.; Singh, J. Nutrigenomics: Concept, advances and applications. Asian J. Dairy Food Res. 2015, 34, 205–212. [Google Scholar] [CrossRef]
- Almatroodi, S.A.; Almatroudi, A.; Khan, A.A.; Alhumaydhi, F.A.; Alsahli, M.A.; Rahmani, A.H. Potential therapeutic targets of epigallocatechin gallate (EGCG), the most abundant catechin in green tea, and its role in the therapy of various types of cancer. Molecules 2020, 25, 3146. [Google Scholar] [CrossRef]

| Type/Form of Tea | Antioxidant Factor | Oxidative Stress Parameters | Target Sites | Animal Species | References |
|---|---|---|---|---|---|
| Green tea | Photoaged skin | ↑ hydroxyproline; ↑ CAT; ↓ protein carbonyl | Serum, skin | Mice | [174] |
| Green tea | Lead acetate (0.4%) orally treated rats | ↓ LPO; ↑ GSH-Px; ↑ SOD; ↑ CAT | Kidney | Sprague Dawley rats | [175] |
| Green tea | Lead acetate (0.4%) orally treated rats | ↑ SOD; ↑ GST | Liver | Sprague Dawley rats | [176] |
| Green tea | High-sodium-diet (4 g NaCl/kg diet) induced hypertension | ↑ TAC | Serum | Wistar rats | [177] |
| Green tea | Streptozotocin-induced diabetic rats | ↓ MDA; ↑ GSH; ↑ SOD; ↑ CAT; ↑ GSH-Px | Liver, serum | Wistar rats | [178] |
| Green tea extract | Lead acetate (0.4%) orally treated rats | ↓ LPO; ↑ GSH; ↑ SOD; ↑ GST | Serum | Sprague Dawley rats | [179] |
| Green tea extract | Atherogenic diet consisting of 33% sugar, 21% fat, and 3% cholesterol | ↑ TAC; ↓ SOD; ↓ CAT; ↑ GSH; ↑ GSH-Px | Serum | Sprague Dawley rats | [180] |
| Green tea extract | Lead acetate (100 mg/kg b. w.) treated rats | ↑ SOD; ↑ GSH-Px | Total brain | Albino rats | [181] |
| Green tea extract | Doxorubicin-induced cardiotoxicity | ↑ GSH-Px; ↑ GR; ↑ GST; ↑ SOD; ↑ CAT | Heart | Wistar rats | [182] |
| Green tea extract | Chronic gasoline vapour inhalation | ↑ CAT; ↑ SOD; ↑ GSH-Px; ↑ GST; ↑ total thiol; ↓ TBARS; ↓ AOPP | Brain | Male mice CD1 strain | [183] |
| Green tea extract | Nicotine—injected intraperitoneally (1 mg/kg body weight/day) | ↑ SOD; ↑ CAT | Blood | Rats | [184] |
| Green tea infusion | 0.4% lead acetate in distilled H2O orally | ↓ LPO | Plasma, erythrocytes | Male rats | [184] |
| Green tea infusion | 0.4% lead acetate in distilled H2O orally | ↓ SOD; ↓ GST; ↓ GSH; ↓ NO; ↓ LPO | Liver, kidney, brain | Male rats | [185] |
| Green tea infusion | 0.4% lead acetate orally for 6 weeks | ↑ GST; ↑ GSH; ↑ SOD; ↑ TAC; ↓ LPO | Blood | Rats | [186] |
| Green tea infusion | 0.4% lead acetate orally for 6 weeks | ↑ GST; ↑ GSH; ↑ SOD; ↑ TAC; ↓ LPO | Brain | Rats | [186] |
| Aqueous extracts of rooibos tea | Chronic restraint or immobilization | ↓ LPO; ↑ GSH ↑ GSH/GSSG | Blood | Rats | [187] |
| Aqueous extracts of rooibos tea | Chronic restraint or immobilization | ↓ GR; ↑GSH-Px; ↓ SOD; ↓ CAT | Brain | Rats | [187] |
| Water extracts Pu-erh tea | High-fat diet | ↓ MDA; ↓ NOS; ↑ SOD; ↑ CAT ↑ GSH-Px | Serum | Rats | [188] |
| White tea | Acute hypoxia and subsequent recovery juveniles | ↓ SOD (especially Mn-SOD and CuZn-SOD isoforms); ↑ GSH-Px | Blood | Sparus aurata | [189] |
| White tea | Prediabetes rats | ↑ SOD; ↑ CAT; ↑ GSH-Px | Lungs | Wistar rats | [190] |
| Green, black, red and white tea | Cadmium chloride (7 mg/kg feed) and lead acetate (50 mg/kg feed) orally | ↑ SOD; ↑ CAT; ↑ GSH; ↑ GSH-Px | Lungs, brain, heart, liver, kidneys | Wistar rats | [53] |
| Catechin | Catechin—50 mg/kg or escitalopram—10 mg/kg orally | ↑ CAT; ↑ SOD; ↓ GSH; ↓ MDA | Brain | Rats | [191] |
| EGCG | Fluoride 25 mg/kg/bw orally | ↑ CAT; ↑ SOD; ↑ GSH-Px; ↑ GSH; ↑ GST; ↑ GR; ↓ ROS; ↓ TBARS; ↓ NO; ↑ Vit. C | Brain | Wistar rats | [192] |
| EGCG | Effect of electromagnetic radiation (frequency 900 MHz modulated at 217 Hz, power density 0.02 mW/cm2, SAR 1.245 W/kg) | ↑GSH, ↑CAT, ↑GSH-Px; ↑ SOD | Brain | Rats | [193] |
| EGCG | Azoxymethane—induced colon carcinogenesis (5 mg/kg body weight) | ↓ NOS 2, ↓ COX; ↓ NO | Colonic tissues | Mice | [194] |
| EGCG | 7,12-dimethylbenz[a]-anthracene and 12-O-tetradecanoylphorbol-13-acetate-promoted skin tumorigenesis | ↓ NOS; ↓ COX | Skin | Mice | [195] |
| Green tea tannins | Sodium arsenite (100 mg/kg/day) orally for 28 days | ↑ GSH; ↑ GSH-Px; ↑ SOD; ↑ ALAD; ↑ NOx; ↓ TBARS | Liver, kidneys | Sprague Dawley rats | [196] |
| Tannic acid | Cadmium chloride (7 mg/kg feed) and lead acetate (50 mg/kg feed) orally | ↑ SOD; ↑ CAT | Total brain | Wistar rats | [197] |
| Tannic acid | Lead acetate (50 mg/kg body weight) for three times a week for two weeks | ↓ LPO; ↑ GSH; ↑ GST; ↑ GSH-Px; ↑ SOD; ↑ CAT | Brain | Wistar rats | [198] |
| Caffeine | Cadmium chloride (5 mg/kg) | ↓ LPO; ↓ ROS, ↑ Nrf-2; ↑ HO-1 | Brain | C57BL/6N mice | [199] |
| Green tea | Azathioprine-induced liver damage | ↓ GSH; ↓ CAT; ↓ GSH-Px | Liver | Rats | [200] |
| Tea polyphenols | Gastric cancer | ↑ GSH-Px; ↑ SOD; ↓ MDA | Thymus, spleen | Mice | [201] |
| Tea polyphenols | Diethylnitrosamine/phenobarbital-induced hepatocarcinogenesis | ↑ TAC; ↑GSH-Px | Liver | Sprague Dawley rats | [202] |
| Tea polysaccharides | Gastric cancer | ↓ MDA; ↑ SOD; ↑ CAT; ↑ GSH-Px | Tissue | Mice | [203] |
| Tea polysaccharides | Exhausting training | ↑ SOD; ↑ CAT; ↑ GSH-Px; ↓ MDA | Serum, liver, kinder | Mice | [204] |
| Tea polysaccharides | Carbon tetrachloride-induced liver injury | ↑ SOD; ↑ GSH-Px; ↓ MDA | Liver | Mice | [205] |
| Type/Form of Tea | Antioxidant Factor | Oxidative Stress Parameters | Place of Collection | References |
|---|---|---|---|---|
| Rooibos | Risk for cardiovascular disease | ↓ CDs; ↓ TBARS, ↑ GSH, ↑ GSH/GSSG | Blood | [206] |
| Oolong tea | Hypercholesterolemia | ↑ NEAC | Serum, urine | [207] |
| Green tea | Healthy subjects | ↑ hOGG1; ↑ HMOX-1 | Blood | [208] |
| Green tea | Oxidation-induced DNA damage and redox-sensitive cytoprotective factors in type 2 diabetes patients | ↑ TAC; ↑ GSH | Plasma | [209] |
| Green tea | Metabolic syndrome | ↑ GSH | Blood | [210] |
| Black tea theaflavins | Neuronal apoptosis | ↑ SOD; ↑ CAT | PC12 cells | [211] |
| EGCG | Hydrogen peroxide-induced injury in human dermal fibroblasts | ↑ GSH-Px; ↑ SOD; ↓ MDA | Skin | [212] |
| EGCG | Hepatic stellate cell | ↑ GSH | Liver | [213] |
| EGCG | Induced cancer cell death | ↓ Trx/TrxR | Liver | [214] |
| EGCG | Cancer biopsies and HeLa cell line with hydrogen peroxide | ↓ GSH; ↓ SOD | Tissue | [215] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olcha, P.; Winiarska-Mieczan, A.; Kwiecień, M.; Nowakowski, Ł.; Miturski, A.; Semczuk, A.; Kiczorowska, B.; Gałczyński, K. Antioxidative, Anti-Inflammatory, Anti-Obesogenic, and Antidiabetic Properties of Tea Polyphenols—The Positive Impact of Regular Tea Consumption as an Element of Prophylaxis and Pharmacotherapy Support in Endometrial Cancer. Int. J. Mol. Sci. 2022, 23, 6703. https://doi.org/10.3390/ijms23126703
Olcha P, Winiarska-Mieczan A, Kwiecień M, Nowakowski Ł, Miturski A, Semczuk A, Kiczorowska B, Gałczyński K. Antioxidative, Anti-Inflammatory, Anti-Obesogenic, and Antidiabetic Properties of Tea Polyphenols—The Positive Impact of Regular Tea Consumption as an Element of Prophylaxis and Pharmacotherapy Support in Endometrial Cancer. International Journal of Molecular Sciences. 2022; 23(12):6703. https://doi.org/10.3390/ijms23126703
Chicago/Turabian StyleOlcha, Piotr, Anna Winiarska-Mieczan, Małgorzata Kwiecień, Łukasz Nowakowski, Andrzej Miturski, Andrzej Semczuk, Bożena Kiczorowska, and Krzysztof Gałczyński. 2022. "Antioxidative, Anti-Inflammatory, Anti-Obesogenic, and Antidiabetic Properties of Tea Polyphenols—The Positive Impact of Regular Tea Consumption as an Element of Prophylaxis and Pharmacotherapy Support in Endometrial Cancer" International Journal of Molecular Sciences 23, no. 12: 6703. https://doi.org/10.3390/ijms23126703
APA StyleOlcha, P., Winiarska-Mieczan, A., Kwiecień, M., Nowakowski, Ł., Miturski, A., Semczuk, A., Kiczorowska, B., & Gałczyński, K. (2022). Antioxidative, Anti-Inflammatory, Anti-Obesogenic, and Antidiabetic Properties of Tea Polyphenols—The Positive Impact of Regular Tea Consumption as an Element of Prophylaxis and Pharmacotherapy Support in Endometrial Cancer. International Journal of Molecular Sciences, 23(12), 6703. https://doi.org/10.3390/ijms23126703








