Recovery of Hydrochloric Acid from Industrial Wastewater by Diffusion Dialysis Using a Spiral-Wound Module
Abstract
:1. Introduction
2. Results
2.1. Effect of Volumetric Flow Ratio
2.2. Membrane Properties
2.3. Economic Analysis of Acid Recovery System
3. Discussion
4. Material and Methods
4.1. Feed Solution
4.2. Analytical Methods
4.3. Equipment
4.4. Membrane Characterization
4.5. Diffusion Dialysis Tests
4.6. Calculations
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AC | Activated carbon |
| AEMs | Anion-exchange membranes |
| ATR | Attenuated total reflection |
| BPPO | Bromomethylated poly (phenylene oxide) |
| DD | Diffusion dialysis |
| DIA | Dialysate |
| DIF | Diffusate |
| FF | Filtered feed |
| PET | Polyethylene terephthalate |
| RF | Raw feed |
| R.H. | Relative humidity |
| SEM TDS | Scanning electron microscope Total dissolved solids |
| WT | Water |
References
- Hughes, T.A.; Gray, N.F. Removal of metals and acidity from acid mine drainage using municipal wastewater and activated sludge. Mine Water Environ. 2013, 32, 170–184. [Google Scholar] [CrossRef]
- López, J.; Gibert, O.; Cortina, J. Integration of membrane technologies to enhance the sustainability in the treatment of metal-containing acidic liquid wastes. An overview. Sep. Purif. Technol. 2021, 265, 118485. [Google Scholar] [CrossRef]
- Luo, J.; Wu, C.; Xu, T.; Wu, Y. Diffusion dialysis-concept, principle and applications. J. Membr. Sci. 2011, 366, 1–16. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, W.; Wang, Y. Diffusion dialysis for acid recovery from acidic waste solutions: Anion exchange membranes and technology integration. Membranes 2020, 10, 169. [Google Scholar] [CrossRef]
- Wang, G.; Xu, J.; Sun, P.; Zhao, X.; Zhang, W.; Lv, L.; Pan, B.; Guo, Q.; Bin, Y.; Wang, J. Principle of diffusion dialysis method and its application in the treatment of wastewater of electroplating industry. Ion Exch. Adsorpt. 2015, 31, 569–576. [Google Scholar] [CrossRef]
- Zhang, P.; Wu, Y.; Liu, W.; Cui, P.; Huang, Q.; Ran, J. Construction of two dimensional anion exchange membranes to boost acid recovery performances. J. Membr. Sci. 2021, 618, 118692. [Google Scholar] [CrossRef]
- Chen, Q.; Luo, J.; Liao, J.; Zhu, C.; Li, J.; Xu, J.; Xu, Y.; Ruan, H.; Shen, J. Tuning the length of aliphatic chain segments in aromatic poly(arylene ether sulfone) to tailor the micro-structure of anion-exchange membrane for improved proton blocking performance. J. Memb. Sci. 2022, 641, 119860. [Google Scholar] [CrossRef]
- Sharma, J.; Misra, S.; Kulshrestha, V. Internally cross-linked poly (2,6-dimethyl-1,4-phenylene ether) based anion exchange membrane for recovery of different acids by diffusion dialysis. Chem. Eng. J. 2021, 414, 128776. [Google Scholar] [CrossRef]
- Lin, J.; Huang, J.; Wang, J.; Yu, J.; You, X.; Lin, X.; Van Der Bruggen, B.; Zhao, S. High-performance porous anion exchange membranes for efficient acid recovery from acidic wastewater by diffusion dialysis. J. Membr. Sci. 2021, 624, 119116. [Google Scholar] [CrossRef]
- Ye, H.; Zou, L.; Wu, C.; Wu, Y. Tubular membrane used in continuous and semi-continuous diffusion dialysis. Sep. Purif. Technol. 2020, 235, 116147. [Google Scholar] [CrossRef]
- Luo, F.; Zhang, X.; Pan, J.; Mondal, A.N.; Feng, H.; Xu, T. Diffusion dialysis of sulfuric acid in spiral wound membrane modules: Effect of module number and connection mode. Sep. Purif. Technol. 2015, 148, 25–31. [Google Scholar] [CrossRef]
- Gueccia, R.; Aguirre, A.R.; Randazzo, S.; Cipollina, A.; Micale, G. Diffusion dialysis for separation of hydrochloric acid, iron and zinc ions from highly concentrated pickling solutions. Membranes 2020, 10, 129. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Aguirre, A.; Lopez, J.; Gueccia, R.; Randazzo, S.; Cipollina, A.; Cortina, J.; Micale, G. Diffusion dialysis for the treatment of H2SO4-CuSO4 solutions from electroplating plants: Ions membrane transport characterization and modelling. Sep. Purif. Technol. 2021, 266, 118215. [Google Scholar] [CrossRef]
- Gueccia, R.; Winter, D.; Randazzo, S.; Cipollina, A.; Koschikowski, J.; Micale, G.D. An integrated approach for the HCl and metals recovery from waste pickling solutions: Pilot plant and design operations. Chem. Eng. Res. Des. 2021, 168, 383–396. [Google Scholar] [CrossRef]
- Deng, T.; Zeng, X.; Zhang, C.; Wang, Y.; Zhang, W. Constructing proton selective pathways using MOFs to enhance acid recovery efficiency of anion exchange membranes. Chem. Eng. J. 2022, 445, 136752. [Google Scholar] [CrossRef]
- Zhang, X.; Li, C.; Wang, H.; Xu, T. Recovery of hydrochloric acid from simulated chemosynthesis aluminum foil wastewater by spiral wound diffusion dialysis (SWDD) membrane module. J. Membr. Sci. 2011, 384, 219–225. [Google Scholar] [CrossRef]
- Merkel, A.; Čopák, L.; Dvořák, L.; Golubenko, D.; Šeda, L. Recovery of Spent Sulphuric Acid by Diffusion Dialysis Using a Spiral Wound Module. Int. J. Mol. Sci. 2021, 22, 11819. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Lee, Y.M. Anion exchange polyelectrolytes for membranes and ionomers. Prog. Polym. Sci. 2021, 113, 101345. [Google Scholar] [CrossRef]
- Wang, L.; Peng, X.; Mustain, W.E.; Varcoe, J.R. Radiation-grafted anion-exchange membranes: The switch from low- to high-density polyethylene leads to remarkably enhanced fuel cell performance. Energy Environ. Sci. 2019, 12, 1575–1579. [Google Scholar] [CrossRef] [Green Version]
- Biancolli, A.L.G.; Bsoul-Haj, S.; Douglin, J.C.; Barbosa, A.S.; de Sousa, R.R.; Rodrigues, O.; Lanfredi, A.J.C.; Dekel, D.R.; Santiago, E.I. High-performance radiation grafted anion-exchange membranes for fuel cell applications: Effects of irradiation conditions on ETFE-based membranes properties. J. Memb. Sci. 2022, 641, 119879. [Google Scholar] [CrossRef]
- Pal, S.; Mondal, R.; Guha, S.; Chatterjee, U.; Jewrajka, S.K. Crosslinked terpolymer anion exchange membranes for selective ion separation and acid recovery. J. Memb. Sci. 2020, 612, 118459. [Google Scholar] [CrossRef]
- Ji, W.; Wu, B.; Zhu, Y.; Irfan, M.; Afsar, N.U.; Ge, L.; Xu, T. Self-organized nanostructured anion exchange membranes for acid recovery. Chem. Eng. J. 2020, 382, 122838. [Google Scholar] [CrossRef]
- Dean, J.A. Lange’s Handbook of Chemistry; McGraw-Hill, Inc.: New York City, NY, USA, 2017; ISBN 0-07-016384-7. [Google Scholar]
- Safronova, E.Y.; Stenina, I.A.; Yaroslavtsev, A.B. The possibility of changing the transport properties of ion-exchange membranes by their treatment. Pet. Chem. 2017, 57, 299–305. [Google Scholar] [CrossRef]
- Stenina, I.; Yaroslavtsev, A. Ionic mobility in ion-exchange membranes. Membranes 2021, 11, 198. [Google Scholar] [CrossRef]
- Stenina, I.; Golubenko, D.; Nikonenko, V.; Yaroslavtsev, A. Selectivity of Transport Processes in Ion-Exchange Membranes: Relationship with the Structure and Methods for Its Improvement. Int. J. Mol. Sci. 2020, 21, 5517. [Google Scholar] [CrossRef] [PubMed]
- Park, H.B.; Kamcev, J.; Robeson, L.M.; Elimelech, M.; Freeman, B.D. Maximizing the right stuff: The trade-off between membrane permeability and selectivity. Science 2017, 356, eaab0530. [Google Scholar] [CrossRef] [Green Version]
- Russell, S.T.; Pereira, R.; Vardner, J.T.; Jones, G.N.; Dimarco, C.; West, A.C.; Kumar, S.K. Hydration Effects on the Permselectivity-Conductivity Trade-Off in Polymer Electrolytes. Macromolecules 2020, 53, 1014–1023. [Google Scholar] [CrossRef]
- Cukierman, S. Et tu, Grotthuss and other unfinished stories. Biochim. Biophys. Acta BBA Bioenerg. 2006, 1757, 876–885. [Google Scholar] [CrossRef] [Green Version]
- Morris, D.F.C.; Reed, G.L.; Short, E.L.; Slater, D.N.; Waters, D.N. Nickel (II) chloride complexes in aqueous solution. J. Inorg. Nucl. Chem. 1965, 27, 377–382. [Google Scholar] [CrossRef]
- Persson, I. Ferric Chloride Complexes in Aqueous Solution: An EXAFS Study. J. Solut. Chem. 2018, 47, 797–805. [Google Scholar] [CrossRef] [Green Version]
- Golubenko, D.; Pourcelly, G.; Yaroslavtsev, A. Permselectivity and ion-conductivity of grafted cation-exchange membranes based on UV-oxidized polymethylpenten and sulfonated polystyrene. Sep. Purif. Technol. 2018, 207, 329–335. [Google Scholar] [CrossRef]
- Safronova, E.; Golubenko, D.; Pourcelly, G.; Yaroslavtsev, A. Mechanical properties and influence of straining on ion conduc- tivity of perfluorosulfonic acid Nafion®-type membranes depending on water uptake. J. Membr. Sci. 2015, 473, 218–225. [Google Scholar] [CrossRef]
- Golubenko, D.V.; Van der Bruggen, B.; Yaroslavtsev, A.B. Novel anion exchange membrane with low ionic resistance based on chloromethylated/quaternized-grafted polystyrene for energy efficient electromembrane processes. J. Appl. Polym. Sci. 2020, 137, 48656. [Google Scholar] [CrossRef]




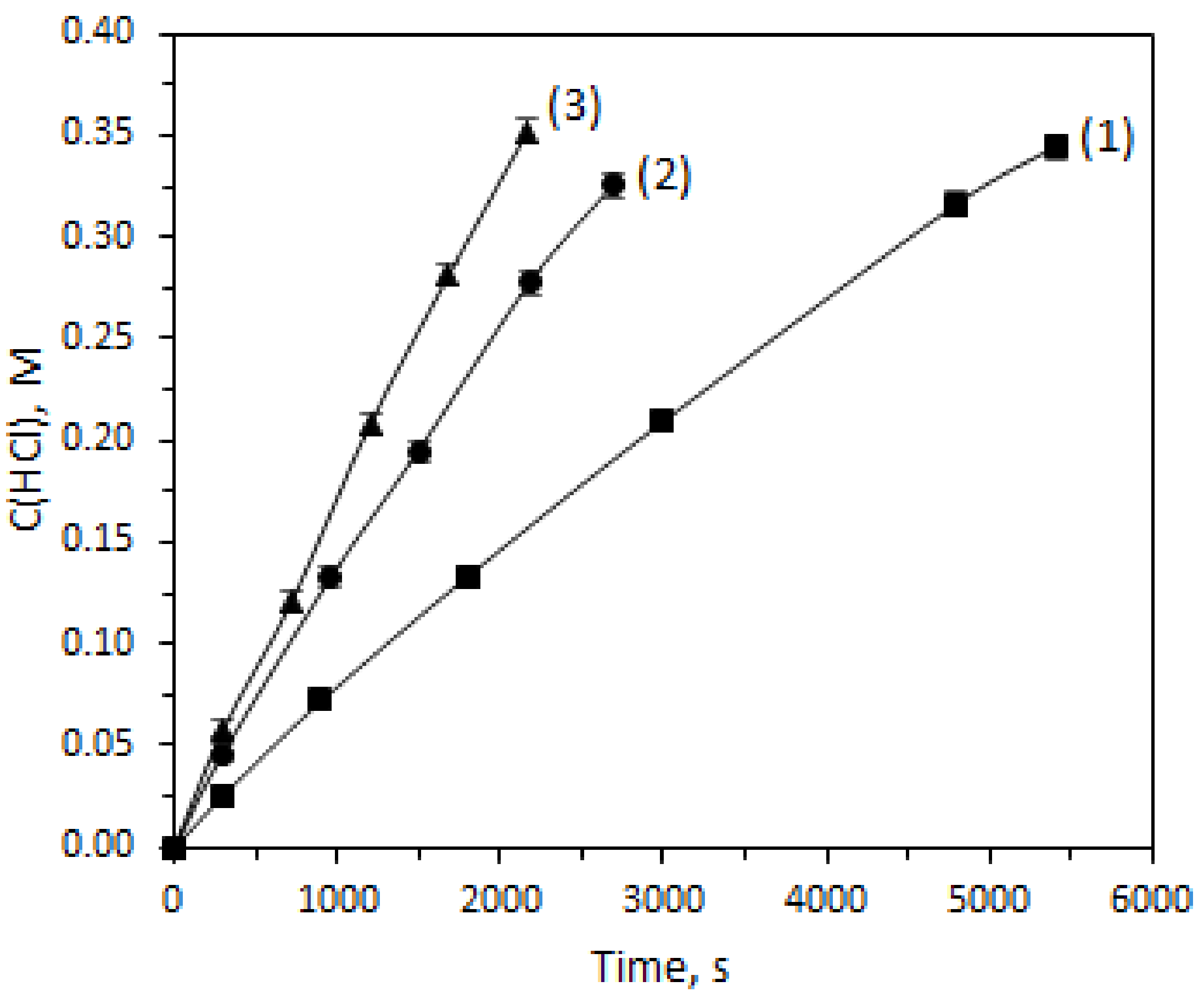
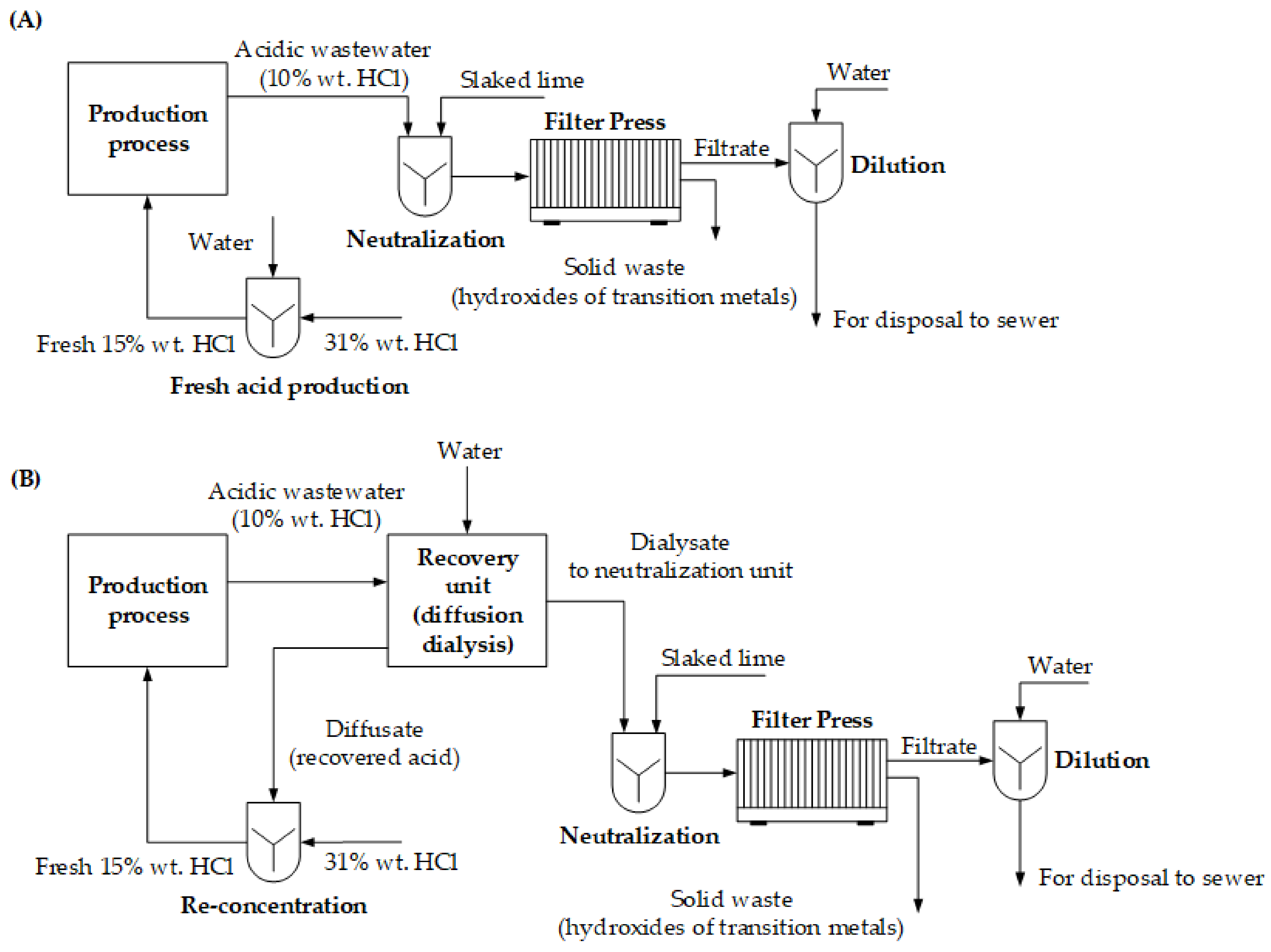

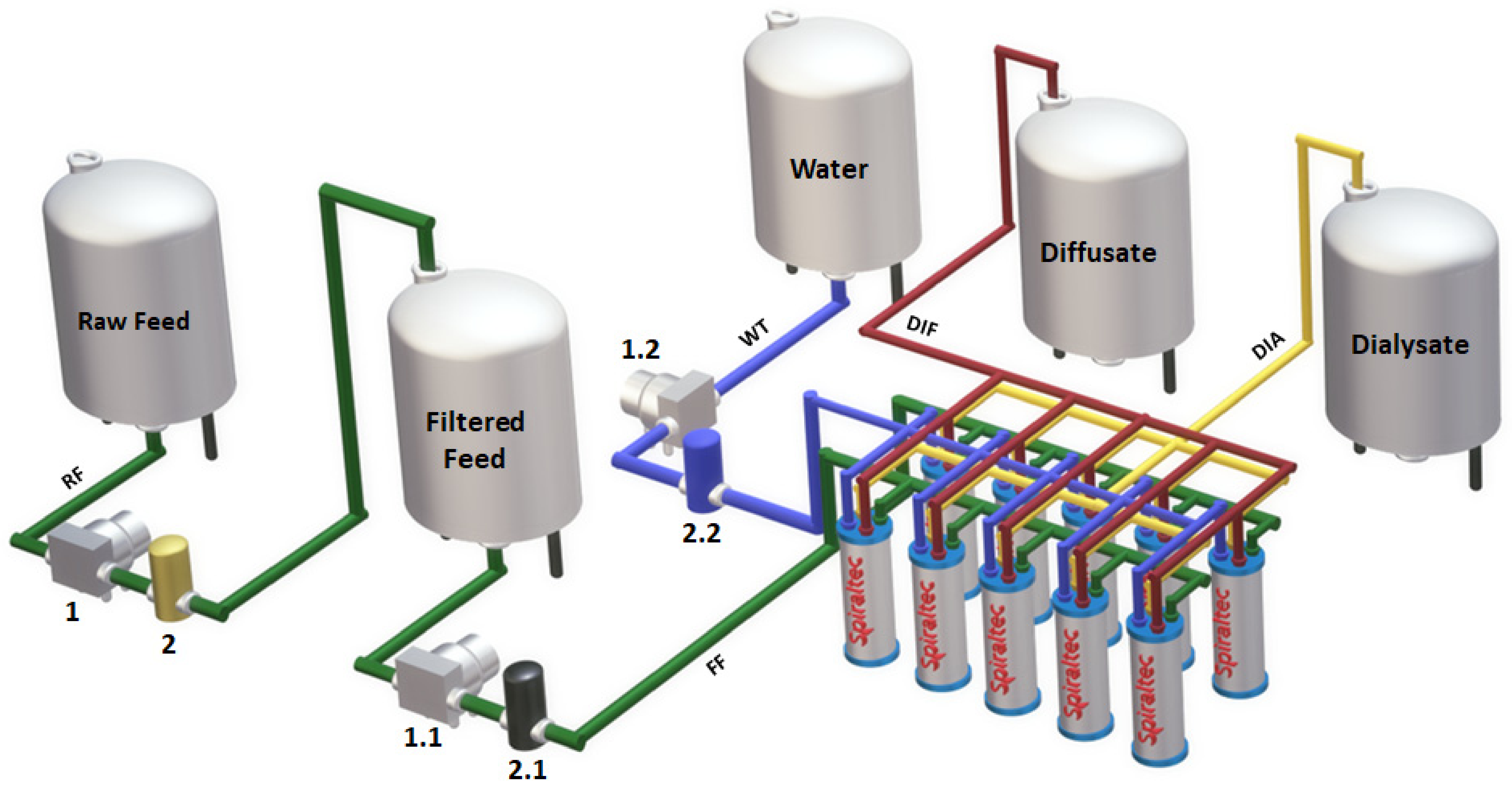
| Parameter | Unit | Value |
|---|---|---|
| HCl yield | % | 68 ± 5 |
| Cr3+ rejection | % | 95 ± 2 |
| Fe2+ rejection | % | 84 ± 3 |
| Ni2+ rejection | % | 85 ± 3 |
| Zn2+ rejection | % | 39 ± 15 |
| Parameter | Unit | Value |
|---|---|---|
| HCl yield | % | 77 ± 6 |
| Cr3+ rejection | % | 95 ± 1 |
| Fe2+ rejection | % | 92 ± 2 |
| Ni2+ rejection | % | 94 ± 2 |
| Zn2+ rejection | % | 35 ± 17 |
| Composition | Unit | Feed | Diffusate | Dialysate | Diffusate | Dialysate |
|---|---|---|---|---|---|---|
| Test 1 | Test 2 | |||||
| HCl | g∙L−1 | 114 ± 2 | 103 ± 2 | 27.4 ± 0.5 | 81 ± 1 | 25.3 ± 0.5 |
| % wt. | 10.3 ± 0.2 | 9.5 ± 0.2 | 2.6 ± 0.1 | 7.6 ± 0.2 | 2.4 ± 0.1 | |
| Fe2+ | ppm | 5500 ± 500 | 810 ± 80 | 216 ± 20 | 350 ± 30 | 3500 ± 300 |
| Zn2+ | ppm | 22,600 ± 2300 | 17,900 ± 1800 | 6500 ± 600 | 14,500 ± 1500 | 6200 ± 600 |
| Ni2+ | ppm | 1440 ± 100 | 216 ± 20 | 900 ± 90 | 94 ± 4 | 950 ± 90 |
| Cr3+ | ppm | 37 ± 4 | 3.6 ± 0.1 | 28 ± 3 | 1.6 ± 0.2 | 27 ± 3 |
| Density | kg∙L−1 | 1.108 ± 0.001 | 1.083 ± 0.001 | 1.039 ± 0.001 | 1.064 ± 0.001 | 1.037 ± 0.001 |
| Parameter | Unit | Value (Initial) | Value (25 °C) | Value (60 °C) |
|---|---|---|---|---|
| Thickness (wet) | μm | 78 ± 1 | 88 ± 1 | 93 ± 1 |
| Ion exchange capacity | meq·g−1 | 1.55 ± 0.02 | 1.50 ± 0.02 | 1.51 ± 0.02 |
| Specific conductivity in 0.5 M NaCl | mS·cm−1 | 11.9 ± 0.6 | 18 ± 1 | 24.5 ± 2 |
| Permselectivity (at 0.1/0.5 mol·kg−1 NaCl) | % | 95.2 ± 0.4 | 93.0 ± 0.4 | 91.3 ± 0.4 |
| Water uptake | % wt. | 23.5 ± 0.5 | 36.2 ± 1.0 | 37.0 ± 1.0 |
| Young’s modulus | MPa | 1160 ± 70 | 780 ± 34 | 890 ± 27 |
| Yield strength | MPa | 13.1 ± 1.0 | 11.4 ± 0.4 | 13.7 ± 1.5 |
| Tensile strength | MPa | 68.5 ± 0.7 | 64.6 ± 1.4 | 21.4 ± 2 |
| Elongation at break | % | 20.0 ± 1.0 | 28.5 ± 1.6 | 8.5 ± 1.2 |
| Membrane | UH | UFe | UZn | UNi | UCr | SH/Fe | SH/Zn | SH/Ni | SH/Cr |
|---|---|---|---|---|---|---|---|---|---|
| Original | 4.4 ± 0.1·10−3 | 1.1 ± 0.1·10−4 | 4.7 ± 0.2·10−3 | 1.2 ± 0.1·10−4 | 6.0 ± 0.8·10−5 | 39 ± 3 | 0.93 ± 0.04 | 36 ± 3 | 74 ± 9 |
| Stability test at 25 °C | 8.3 ± 0.2 ·10−3 | 1.6 ± 0.1·10−4 | 8.2 ± 0.3·10−3 | 0.8 ± 1·10−4 | 4.6 ± 0.1·10−5 | 52 ± 3 | 1.02 ± 0.03 | 101 ± 13 | 181 ± 6 |
| Stability test at 60 °C | 1.13 ± 0.02·10−2 | 3.4 ± 0.4·10−4 | 10.0 ± 0.1·10−3 | 2.7 ± 0.4·10−4 | 7.9 ± 0.4·10−5 | 34 ± 4 | 1.13 ± 0.07 | 42 ± 1 | 144 ± 7 |
| Item | Unit | Unit Cost |
|---|---|---|
| Electricity | EUR/kWh | 0.1 |
| Tap water | EUR/m3 | 2.8 |
| 31% wt. HCl | EUR/metric ton | 140.0 |
| 90% Ca(OH)2 | EUR/metric ton | 138.8 |
| Solid waste disposal Cartridge 10 μm | EUR/metric ton EUR/pc | 83.2 6.4 |
| Case | Unit | A | B | Savings |
|---|---|---|---|---|
| Water for DD | EUR | 0 | 1996 | −1996 |
| Water for TDS dilution | EUR | 141,176 | 51,085 | +90,091 |
| Water for bath preparation | EUR | 1033 | 0 | +1033 |
| Solid waste disposal | EUR | 6303 | 3155 | +3148 |
| Electricity for DD unit (including pumps for filtration) | EUR | 0 | 1584 | −1584 |
| Pre-treatment (cartridges) | EUR | 0 | 2128 | −2128 |
| 31% wt. HCl | EUR | 48,438 | 23,997 | +24,441 |
| Technical Ca(OH)2 | EUR | 15,593 | 5663 | +9930 |
| Total | EUR | 212,543 | 89,608 | +122,935 |
| Composition | Unit | Feed Stream |
|---|---|---|
| Electrical conductivity | mS·cm−1 | 658 ± 3 |
| Density | kg·L−1 | 1.108 ± 0.001 |
| HCl | g·L−1 | 114 ± 2 |
| Cr3+ | ppm | 37 ± 4 |
| Fe2+ | ppm | 5470 ± 500 |
| Ni2+ | ppm | 1440 ± 100 |
| Zn2+ | ppm | 22,600 ± 2300 |
| Cl− | ppm | 128,200 ± 6400 |
| Parameter | Unit | Value |
|---|---|---|
| Electrical conductivity | mS·cm−1 | 0.632 ± 0.003 |
| pH | - | 7.58 ± 0.01 |
| Hardness (Ca2+ + Mg2+) | mmol·L−1 | 2.6 ± 0.1 |
| Alkalinity (pH 4.5) | mmol·L−1 | 3.75 ± 0.08 |
| SO42− | ppm | 111 ± 5 |
| Cl− | ppm | 25 ± 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Merkel, A.; Čopák, L.; Golubenko, D.; Dvořák, L.; Vavro, M.; Yaroslavtsev, A.; Šeda, L. Recovery of Hydrochloric Acid from Industrial Wastewater by Diffusion Dialysis Using a Spiral-Wound Module. Int. J. Mol. Sci. 2022, 23, 6212. https://doi.org/10.3390/ijms23116212
Merkel A, Čopák L, Golubenko D, Dvořák L, Vavro M, Yaroslavtsev A, Šeda L. Recovery of Hydrochloric Acid from Industrial Wastewater by Diffusion Dialysis Using a Spiral-Wound Module. International Journal of Molecular Sciences. 2022; 23(11):6212. https://doi.org/10.3390/ijms23116212
Chicago/Turabian StyleMerkel, Arthur, Ladislav Čopák, Daniil Golubenko, Lukáš Dvořák, Matej Vavro, Andrey Yaroslavtsev, and Libor Šeda. 2022. "Recovery of Hydrochloric Acid from Industrial Wastewater by Diffusion Dialysis Using a Spiral-Wound Module" International Journal of Molecular Sciences 23, no. 11: 6212. https://doi.org/10.3390/ijms23116212
APA StyleMerkel, A., Čopák, L., Golubenko, D., Dvořák, L., Vavro, M., Yaroslavtsev, A., & Šeda, L. (2022). Recovery of Hydrochloric Acid from Industrial Wastewater by Diffusion Dialysis Using a Spiral-Wound Module. International Journal of Molecular Sciences, 23(11), 6212. https://doi.org/10.3390/ijms23116212








