The Enhancement of Antimicrobial Photodynamic Therapy of Escherichia Coli by a Functionalized Combination of Photosensitizers: In Vitro Examination of Single Cells by Quantitative Phase Imaging
Abstract
:1. Introduction
- The photosensitizer is in close proximity to the bacteria but is not related in any way. In this way, damage to the structural elements of the bacteria is limited.
- The photosensitizer binds to the bacteria as a result of electrostatic, van der Waals, or hydrogen bonding interactions. Then the chance of damage to even the outer membrane increases.
- The photosensitizer penetrates the bacteria in several steps. The first is membrane binding, then active transport or diffusion into the cytoplasm. Such a location even allows DNA damage.
2. Results and Discussions
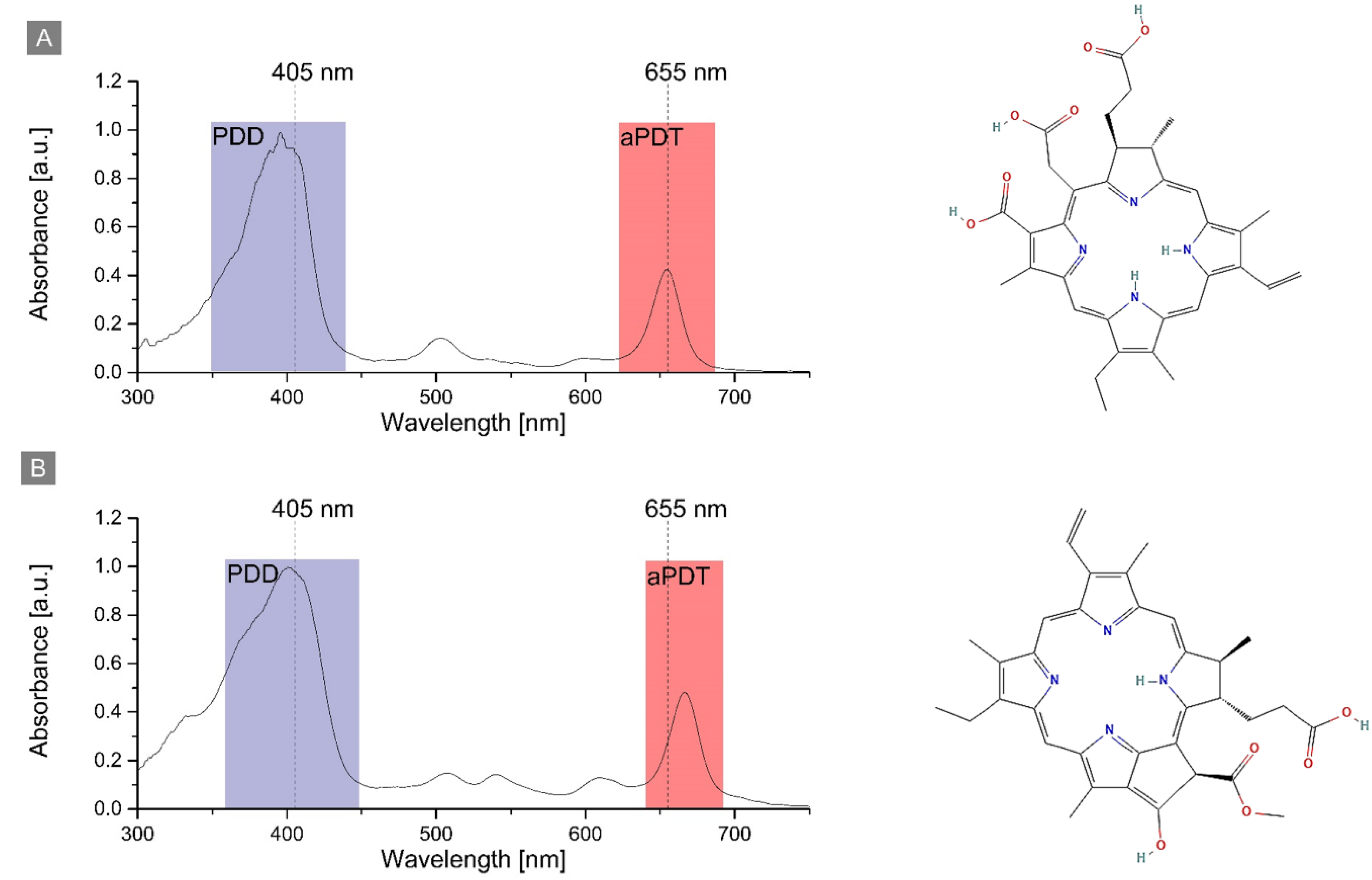
2.1. Photosensitizers’ Spectroscopic Properties
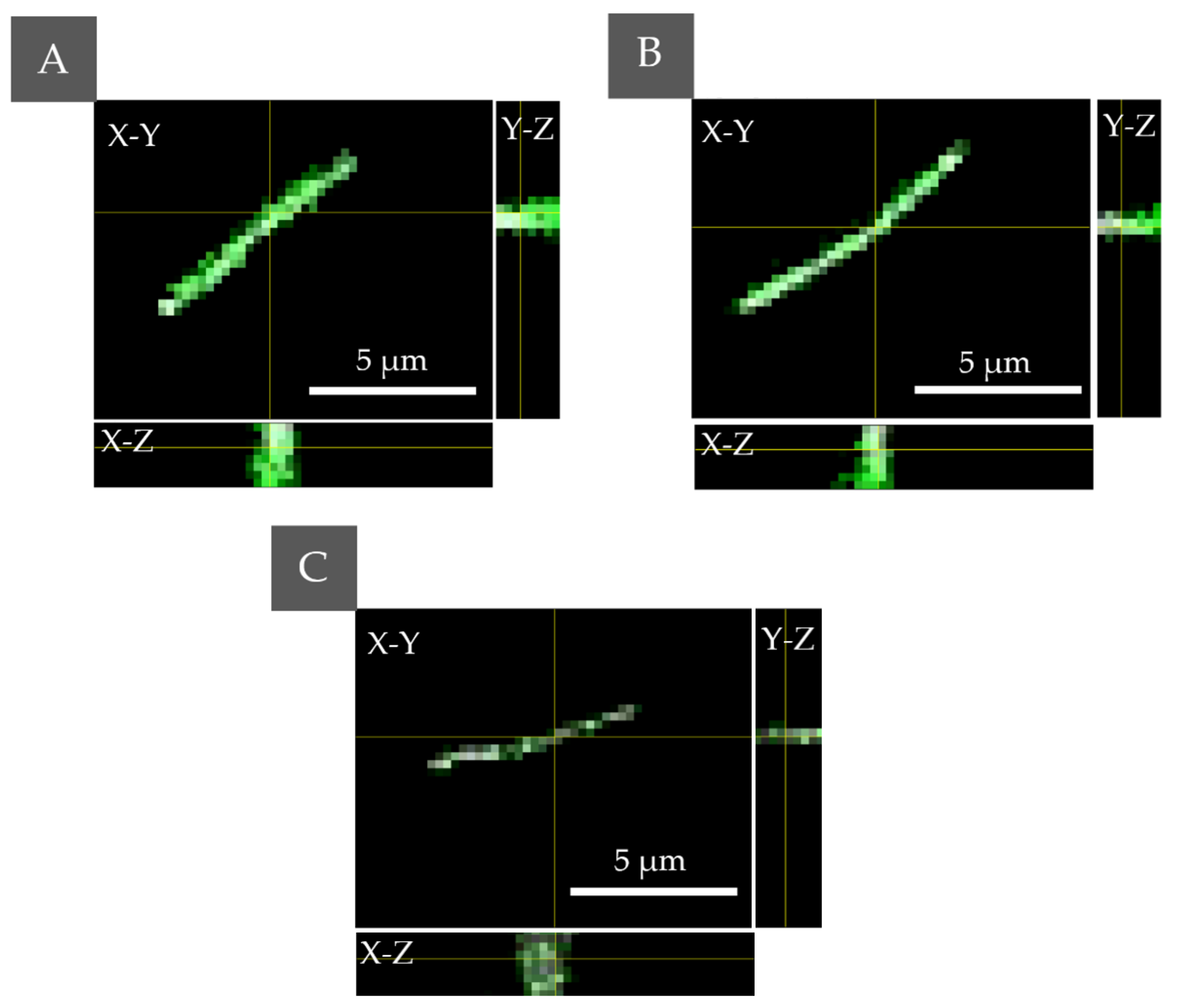
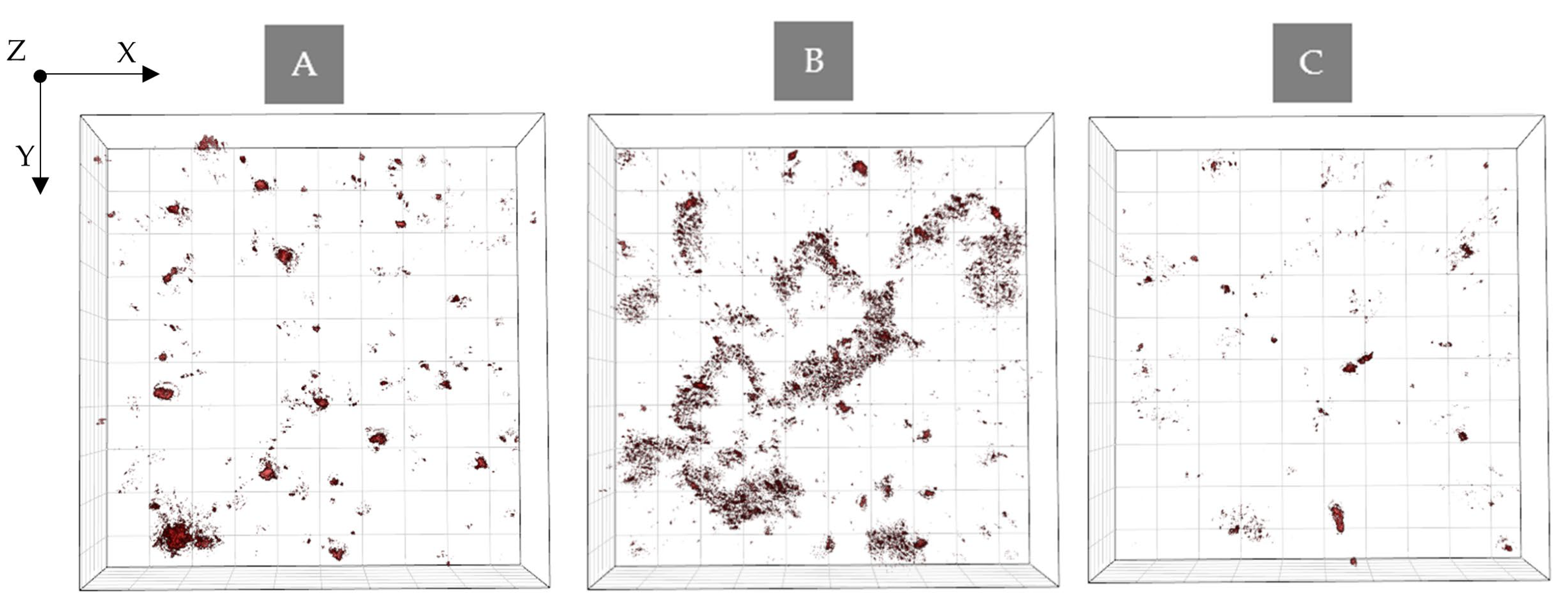
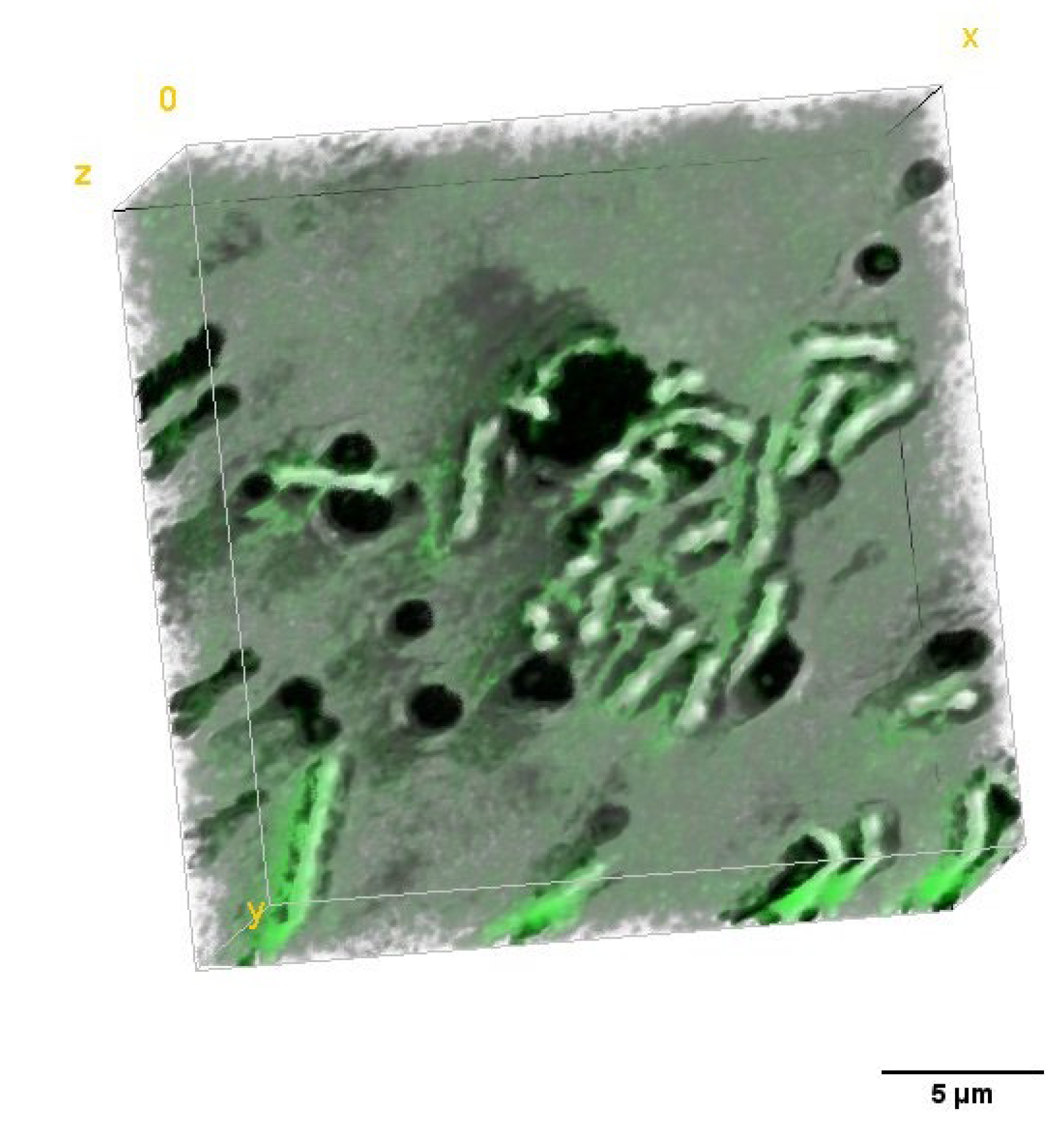
2.2. Study of the Photosensitizers Interaction with Bacteria by Confocal Microscopy
2.3. Bacterial Cell Penetration by Photosensitizers
2.3.1. The Examination of the Possible PSs Accumulation Inside the Bacteria Cells by DHT
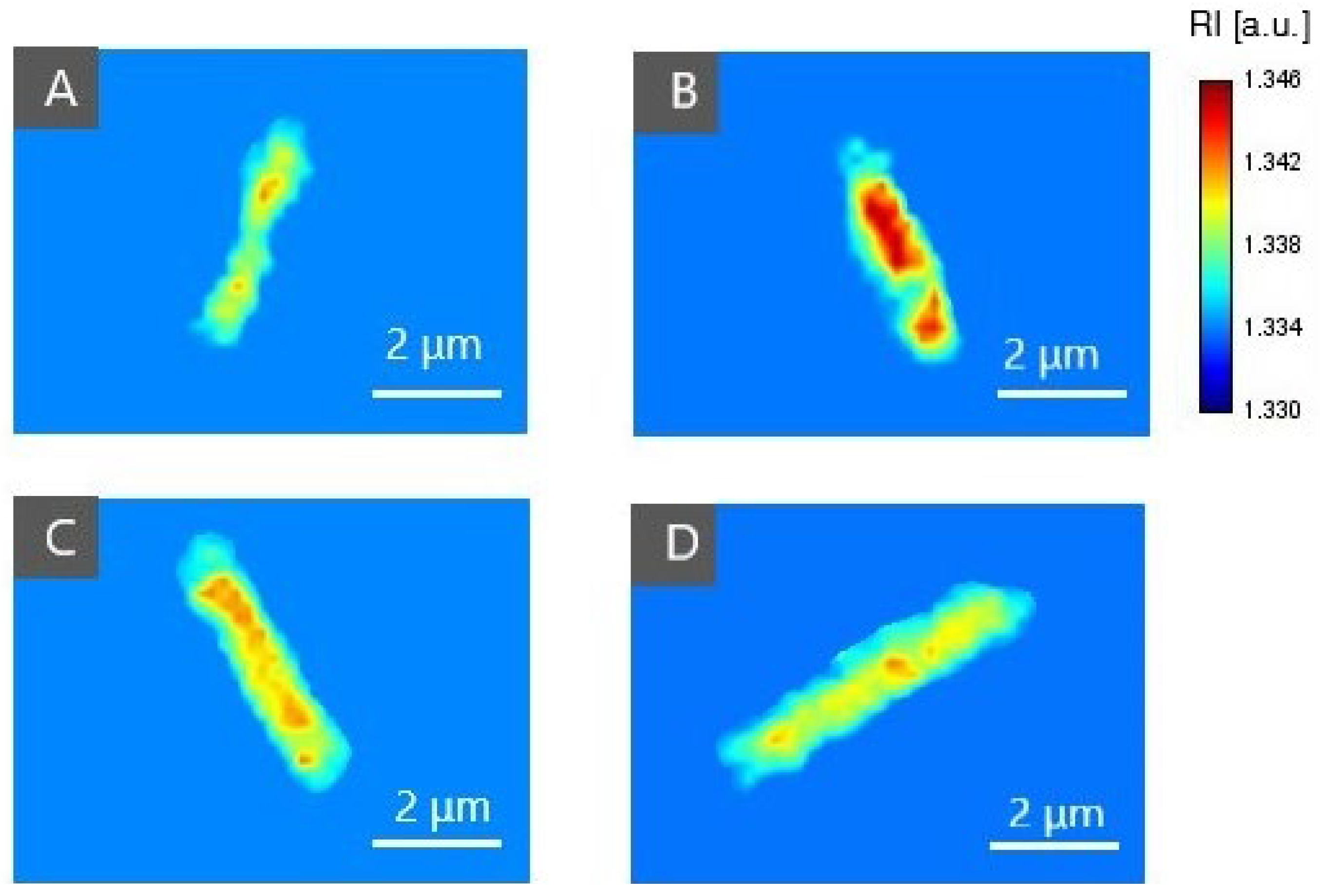
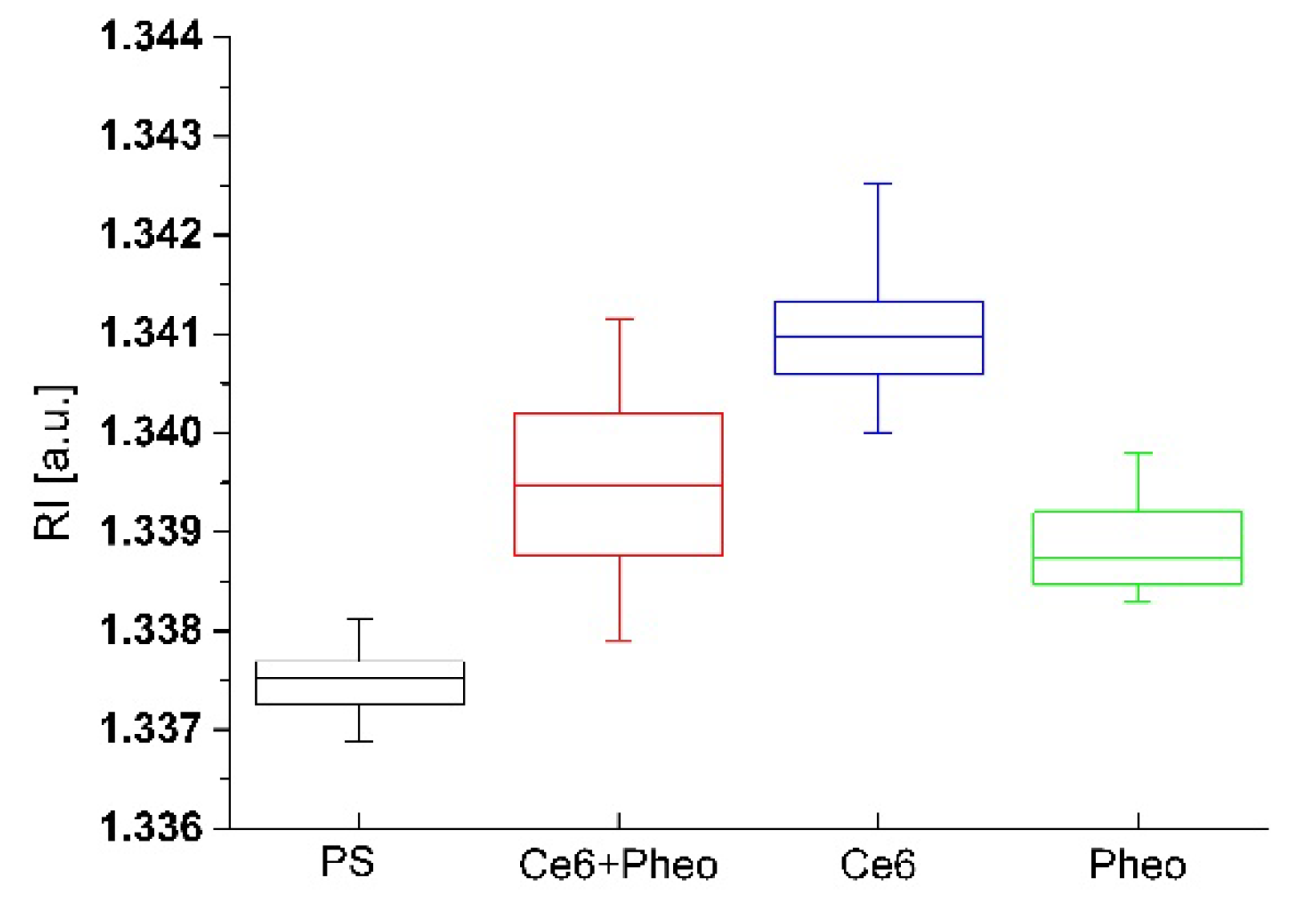
2.3.2. The Examination of Antibacterial Efficiency of PSs by DHT
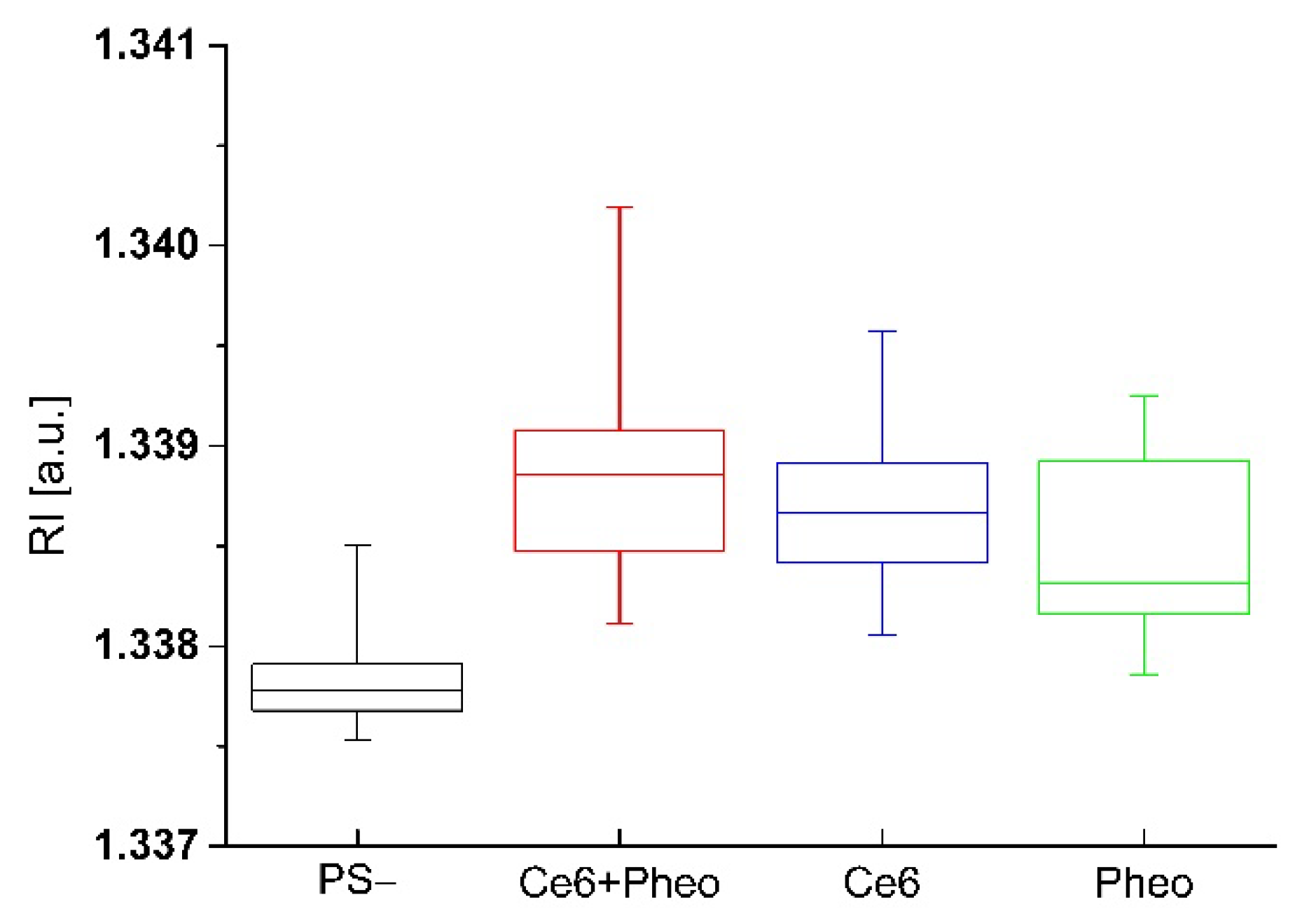
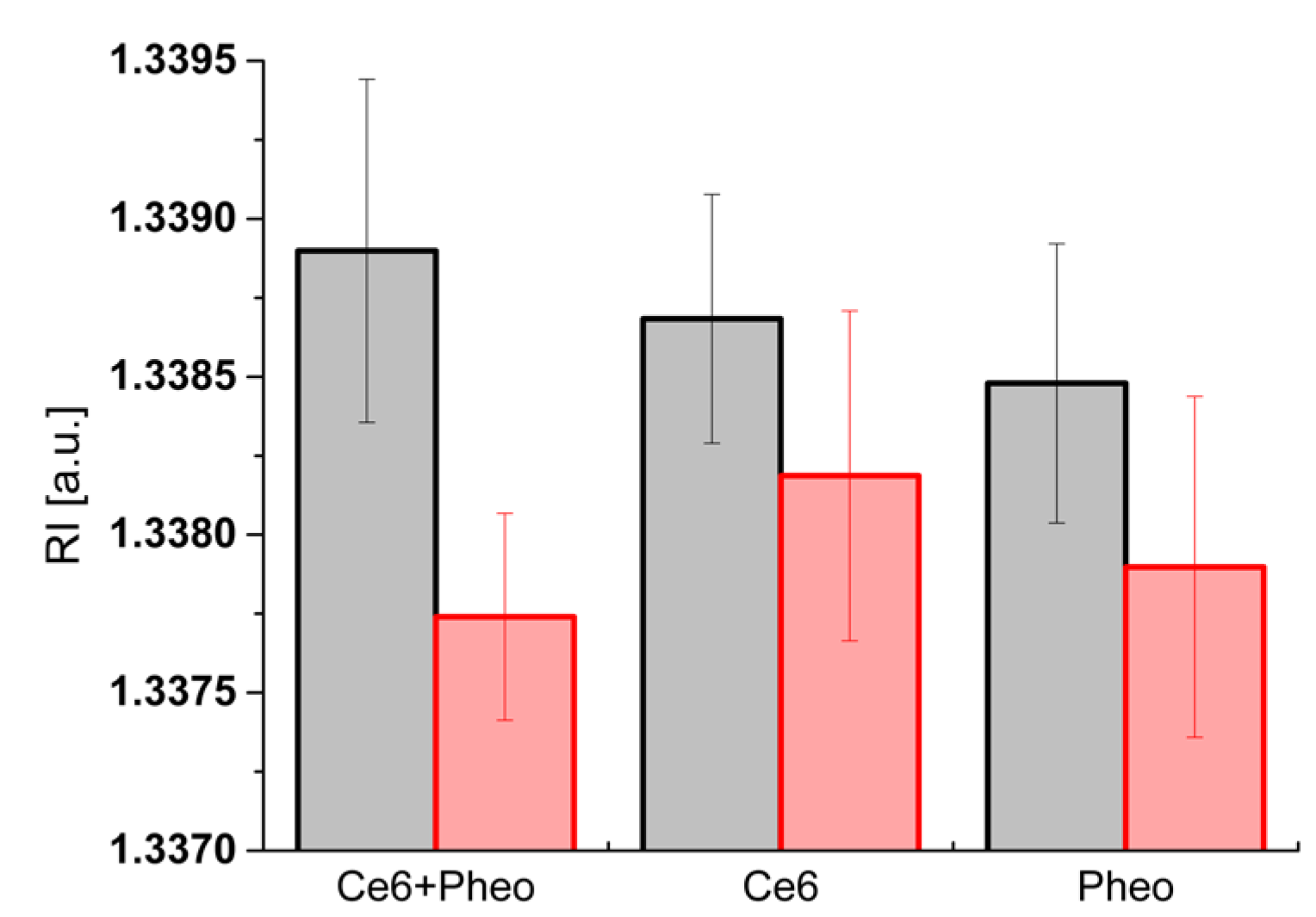
2.4. Analysis of the Dry Mass Density of Bacteria Cells
3. Materials and Methods
3.1. Bacterial Sample Preparation
3.2. Spectrophotometric Measurements
3.3. Irradiation Source for Bacteria Photoinactivation
3.4. Confocal Laser Microscopy
3.5. Digital Holographic Tomography and RI Data Processing
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sharma, D.; Misba, L.; Khan, A.U. Antibiotics versus Biofilm: An Emerging Battleground in Microbial Communities. Antimicrob. Resist. Infect. Control 2019, 8, 76. [Google Scholar] [CrossRef] [PubMed]
- Parvin, F.; Hu, H.; Whiteley, G.S.; Glasbey, T.; Vickery, K. Difficulty in Removing Biofilm from Dry Surfaces. J. Hosp. Infect. 2019, 103, 465–467. [Google Scholar] [CrossRef] [PubMed]
- Fotinos, N.; Convert, M.; Piffaretti, J.C.; Gurny, R.; Lange, N. Effects on Gram-Negative and Gram-Positive Bacteria Mediated by 5-Aminolevulinic Acid and 5-Aminolevulinic Acid Derivatives. Antimicrob. Agents Chemother. 2008, 52, 1366–1373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Otter, J.A.; Vickery, K.; Walker, J.T.; de Lancey Pulcini, E.; Stoodley, P.; Goldenberg, S.D.; Salkeld, J.A.G.; Chewins, J.; Yezli, S.; Edgeworth, J.D. Surface-Attached Cells, Biofilms and Biocide Susceptibility: Implications for Hospital Cleaning and Disinfection. J. Hosp. Infect. 2015, 89, 16–27. [Google Scholar] [CrossRef] [Green Version]
- Pajkos, A.; Vickery, K.; Cossart, Y. Is Biofilm Accumulation on Endoscope Tubing a Contributor to the Failure of Cleaning and Decontamination? J. Hosp. Infect. 2004, 58, 224–229. [Google Scholar] [CrossRef]
- Hrynyshyn, A.; Simões, M.; Borges, A. Biofilms in Surgical Site Infections: Recent Advances and Novel Prevention and Eradication Strategies. Antibiotics 2022, 11, 69. [Google Scholar] [CrossRef]
- Panawala, L. Difference between Gram Positive and Gram Negative Bacteria Stunning Images of Cells Discover How Scientists Use Main Difference—Gram Positive vs Gram Negative Bacteria. Pediaa 2017, 13, 1–3. [Google Scholar]
- Allison, R.R.; Moghissi, K. Photodynamic Therapy (PDT): PDT Mechanisms. Clin. Endosc. 2013, 46, 24–29. [Google Scholar] [CrossRef]
- Nordmann, P.; Poirel, L.; Mark, A.T.; Timothy, R.W. Does Broad-Spectrum β -Lactam Resistance Due to NDM-1 Herald the End of the Antibiotic Era for Treatment of Infections Caused by Gram-Negative Bacteria? J. Antimicrob. Chemother. 2011, 66, 689–692. [Google Scholar] [CrossRef]
- Youf, R.; Müller, M.; Balasini, A.; Thétiot, F.; Müller, M.; Hascoët, A.; Jonas, U.; Schönherr, H.; Lemercier, G.; Montier, T.; et al. Pharmaceutics Antimicrobial Photodynamic Therapy: Latest Developments with a Focus on Combinatory Strategies. Pharmaceutics 2021, 13, 1995. [Google Scholar] [CrossRef]
- Wainwright, M. Photodynamic Antimicrobial Chemotherapy (PACT). J. Antimicrob. Chemother. 1998, 42, 13–28. [Google Scholar] [CrossRef] [PubMed]
- Zolfaghari, P.S.; Packer, S.; Singer, M.; Nair, S.P.; Bennett, J.; Street, C.; Wilson, M. In Vivo Killing of Staphylococcus Aureus Using a Light-Activated Antimicrobial Agent. BMC Microbiol. 2009, 9, 27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeina, B.; Greenman, J.; Purcell, W.M.; Das, B. Killing of Cutaneous Microbial Species by Photodynamic Therapy. Br. J. Dermatol. 2001, 144, 274–278. [Google Scholar] [CrossRef] [PubMed]
- Alves, E.; Faustino, M.A.F.; Neves, M.G.P.M.S.; Cunha, A.; Tome, J.; Almeida, A. An Insight on Bacterial Cellular Targets of Photodynamic Inactivation. Future Med. Chem. 2014, 6, 141–164. [Google Scholar] [CrossRef]
- Nakonechny, F.; Nisnevitch, M. Aspects of Photodynamic Inactivation of Bacteria. In Microorganisms; IntechOpen: London, UK, 2020; ISBN 978-1-83880-188-5. [Google Scholar] [CrossRef] [Green Version]
- Peñ, Q.; Wang, A.; Zaremba, O.; Shi, Y.; Scheeren, H.W.; Metselaar, J.M.; Kiessling, F.; Pallares, R.M.; Wuttke, S.; Lammers, T. Metallodrugs in Cancer Nanomedicine. Chem. Soc. Rev. 2022, 51, 2544. [Google Scholar] [CrossRef]
- Jain, A.; Garrett, N.T.; Malone, Z.P. Special Issue Invited Review Ruthenium-Based Photoactive Metalloantibiotics. Photochem. Photobiol. 2022, 98, 6–16. [Google Scholar] [CrossRef]
- Jain, A.; Winkel, B.S.J.; Brewer, K.J. Photodynamic Antimicrobial Studies on a Ruthenium-Based Metal Complex. Inorg. Chim. Acta 2022, 538, 120996. [Google Scholar] [CrossRef]
- Hurst, A.N.; Scarbrough, B.; Saleh, R.; Hovey, J.; Ari, F.; Goyal, S.; Chi, R.J.; Troutman, J.M.; Vivero-Escoto, J.L. Influence of Cationic Meso-Substituted Porphyrins on the Antimicrobial Photodynamic Efficacy and Cell Membrane Interaction in Escherichia coli. Int. J. Mol. Sci. 2019, 20, 134. [Google Scholar] [CrossRef] [Green Version]
- Huang, L.; Dai, T.; Hamblin, M.R. Antimicrobial Photodynamic Inactivation and Photodynamic Therapy for Infections. Methods Mol. Biol. 2010, 635, 155–173. [Google Scholar] [CrossRef] [Green Version]
- Matsumoto, J.; Matsumoto, T.; Yasuda, K.; Yasuda, M. Photodynamic Inactivation of Escherichia Coli with Cationic Porphyrin Sensitizers. In The Universe of Escherichia coli; IntechOpen: London, UK, 2018; ISBN 978-1-83881-153-2. [Google Scholar] [CrossRef]
- Sharma, A.; Gupta, V.K.; Pathania, R. Efflux Pump Inhibitors for Bacterial Pathogens: From Bench to Bedside. Indian J. Med. Res. 2019, 149, 129–145. [Google Scholar] [CrossRef]
- Zechini, B.; Versace, I. Inhibitors of Multidrug Resistant Efflux Systems in Bacteria. Recent Pat. Anti-Infect. Drug Discov. 2009, 4, 37–50. [Google Scholar] [CrossRef] [PubMed]
- Malik, Z.; Ladan, H.; Nitzan, Y. Photodynamic Inactivation of Gram-Negative Bacteria: Problems and Possible Solutions. J. Photochem. Photobiol. B Biol. 1992, 14, 262–266. [Google Scholar] [CrossRef]
- Popescu, G.; Park, Y.K.; Lue, N.; Best-Popescu, C.; Deflores, L.; Dasari, R.R.; Feld, M.S.; Badizadegan, K. Optical Imaging of Cell Mass and Growth Dynamics. Am. J. Physiol.-Cell Physiol. 2008, 295, 538–544. [Google Scholar] [CrossRef] [Green Version]
- Buzalewicz, I.; Ulatowska-Jarża, A.; Kaczorowska, A.; Gąsior-Głogowska, M.; Podbielska, H.; Karwańska, M.; Wieliczko, A.; Matczuk, A.K.; Kowal, K.; Kopaczyńska, M. Bacteria Single-Cell and Photosensitizer Interaction Revealed by Quantitative Phase Imaging. Int. J. Mol. Sci. 2021, 22, 5068. [Google Scholar] [CrossRef]
- George, S.; Hamblin, M.R.; Kishen, A. Uptake Pathways of Anionic and Cationic Photosensitizers into Bacteria. Photochem. Photochem. Photobiol. Sci. Off. J. Eur. Photochem. Assoc. Eur. Soc. Photobiol. 2009, 8, 788. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, T.K.; Lee, B.W.; Fujii, F.; Lee, K.H.; Lee, S.; Park, Y.K.; Kim, J.K.; Lee, S.W.; Pack, C.G. Mitotic Chromosomes in Live Cells Characterized Using High-Speed and Label-Free Optical Diffraction Tomography. Cells 2019, 8, 1368. [Google Scholar] [CrossRef] [Green Version]
- Kim, T.K.; Lee, B.W.; Fujii, F.; Kim, J.K.; Pack, C.G. Physicochemical Properties of Nucleoli in Live Cells Analyzed by Label-Free Optical Diffraction Tomography. Cells 2019, 8, 699. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ni, J.; Wang, Y.; Zhang, H.; Sun, J.Z.; Tang, B.Z. Aggregation-Induced Generation of Reactive Oxygen Species: Mechanism and Photosensitizer Construction. Molecules 2021, 26, 268. [Google Scholar] [CrossRef]
- Lam, M.; Dimaano, M.L.; Oyetakin-White, P.; Retuerto, M.A.; Chandra, J.; Mukherjee, P.K.; Ghannoum, M.A.; Cooper, K.D.; Baron, E.D. Silicon Phthalocyanine 4 Phototoxicity in Trichophyton Rubrum. Antimicrob. Agents Chemother. 2014, 58, 3029. [Google Scholar] [CrossRef] [Green Version]
- Chlorin E6|CAS 19660-77-6|SCBT—Santa Cruz Biotechnology. Available online: https://www.scbt.com/p/chlorin-e6-19660-77-6 (accessed on 16 August 2021).
- Pheophorbide a|CAS 15664-29-6|SCBT—Santa Cruz Biotechnology. Available online: https://www.scbt.com/p/pheophorbide-a-15664-29-6 (accessed on 16 August 2021).
- Hanakova, A.; Bogdanova, K.; Tomankova, K.; Pizova, K.; Malohlava, J.; Binder, S.; Bajgar, R.; Langova, K.; Kolar, M.; Mosinger, J.; et al. The Application of Antimicrobial Photodynamic Therapy on S. Aureus and E. Coli Using Porphyrin Photosensitizers Bound to Cyclodextrin. Microbiol. Res. 2014, 169, 163–170. [Google Scholar] [CrossRef]
- Huang, L.; Xuan, Y.; Koide, Y.; Zhiyentayev, T.; Tanaka, M.; Hamblin, M.R. Type i and Type II Mechanisms of Antimicrobial Photodynamic Therapy: An in Vitro Study on Gram-Negative and Gram-Positive Bacteria. Lasers Surg. Med. 2012, 44, 490–499. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oh, J.; Ryu, J.S.; Lee, M.; Jung, J.; Han, S.; Chung, H.J.; Park, Y. Three-Dimensional Label-Free Observation of Individual Bacteria upon Antibiotic Treatment Using Optical Diffraction Tomography. Biomed. Opt. Express 2020, 11, 1257–1267. [Google Scholar] [CrossRef] [PubMed]
- Nanolive. Quantitative Analysis of 3D Refractive Index Maps. In Application Note by Nanolive SA; Nanolive SA: Geneva, Switzerland, 2018. [Google Scholar]
- Friebel, M.; Meinke, M. Model Function to Calculate the Refractive Index of Native Hemoglobin in the Wavelength Range of 250–1100 Nm Dependent on Concentration. Appl. Opt. 2006, 45, 2838–2842. [Google Scholar] [CrossRef] [PubMed]
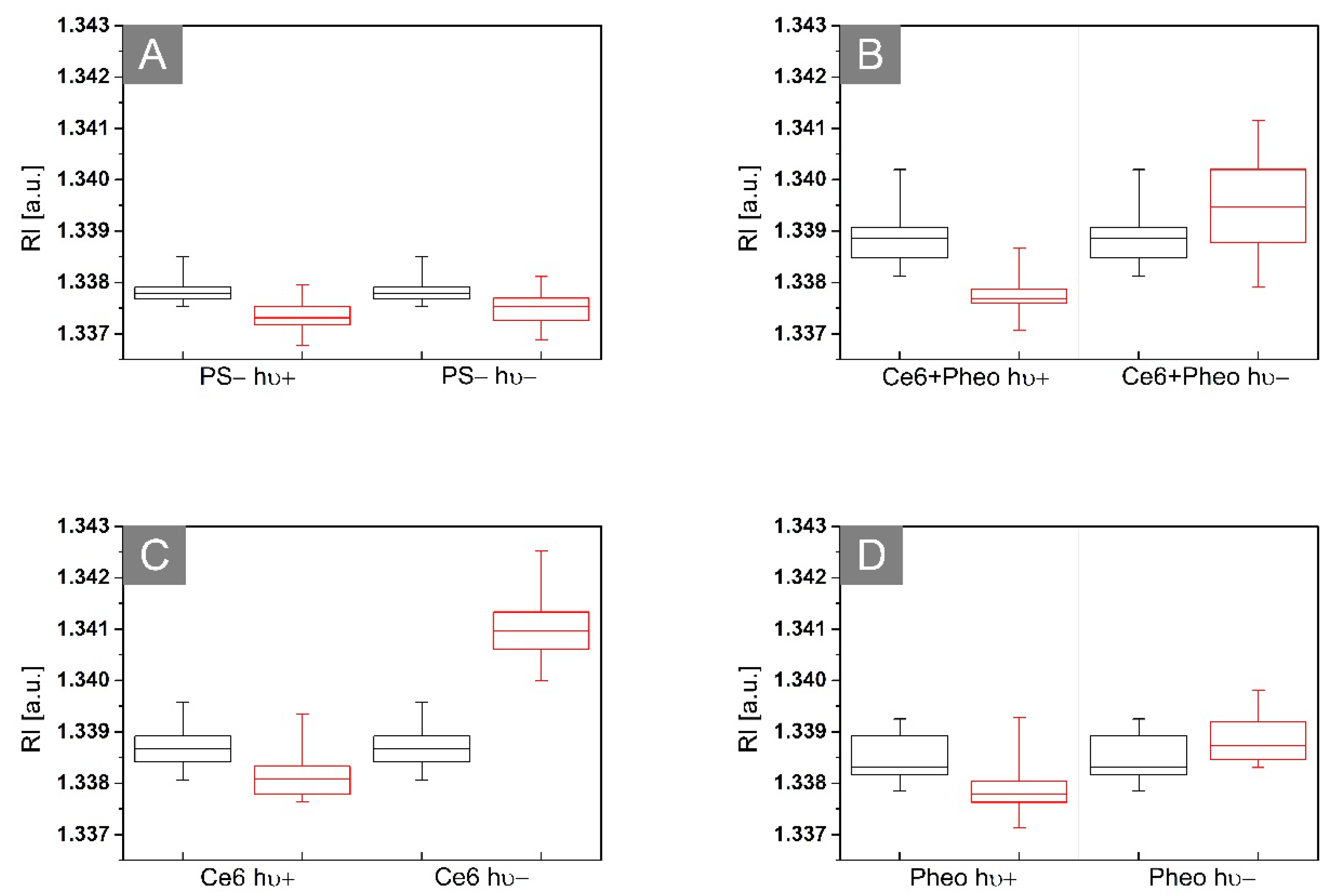


| Source of Variability | SS 1 | df 1 | MS 1 | F 1 | Prob > F |
|---|---|---|---|---|---|
| Group (between) | 1.254 × 10−5 | 3 | 4.180 × 10−6 | 23.586 | <<0.05 |
| Error (within) | 1.347 × 10−5 | 76 | 1.772 × 10−7 | ||
| Total | 2.601 × 10−5 | 79 |
| PS− | |||||
| Source of variability | SS 1 | df 1 | MS 1 | F 1 | Prob > F |
| Group (between) | 2.337 × 10−7 | 1 | 2.337 × 10−7 | 2.538 | 0.119 |
| Error (within) | 3.499 × 10−6 | 38 | 9.207 × 10−8 | ||
| Total | 3.732 × 10−6 | 39 | |||
| Ce6 + Pheo | |||||
| Source of variability | SS 1 | df 1 | MS 1 | F 1 | Prob > F |
| Group (between) | 2.903 × 10−5 | 1 | 2.903 × 10−5 | 57.539 | <<0.05 |
| Error (within) | 1.917 × 10−5 | 38 | 5.045 × 10−7 | ||
| Total | 4.820 × 10−5 | 39 | |||
| Ce6 | |||||
| Source of variability | SS 1 | df 1 | MS 1 | F 1 | Prob > F |
| Group (between) | 8.265 × 10−5 | 1 | 8.265 × 10−5 | 249.346 | <<0.05 |
| Error (within) | 1.260 × 10−5 | 38 | 3.315 × 10−7 | ||
| Total | 9.525 × 10−5 | 39 | |||
| Pheo | |||||
| Source of variability | SS 1 | df 1 | MS 1 | F 1 | Prob > F |
| Group (between) | 9.604 × 10−6 | 1 | 9.604 × 10−6 | 36.623 | <<0.05 |
| Error (within) | 9.965 × 10−6 | 38 | 2.622 × 10−7 | ||
| Total | 1.957 × 10−5 | 39 | |||
| Photosensitizers | Ce6 + Pheo | Ce6 | Pheo | |||
|---|---|---|---|---|---|---|
| Groups | Before irradiation | After irradiation | Before irradiation | After irradiation | Before irradiation | After irradiation |
| Mean dry mass density (g/dL) | 1.46 ± 0.20 | 1.03 ± 0.12 | 1.38 ± 0.15 | 1.19 ± 0.20 | 1.30 ± 0.17 | 1.09 ± 0.20 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pietrowska, A.; Hołowacz, I.; Ulatowska-Jarża, A.; Guźniczak, M.; Matczuk, A.K.; Wieliczko, A.; Wolf-Baca, M.; Buzalewicz, I. The Enhancement of Antimicrobial Photodynamic Therapy of Escherichia Coli by a Functionalized Combination of Photosensitizers: In Vitro Examination of Single Cells by Quantitative Phase Imaging. Int. J. Mol. Sci. 2022, 23, 6137. https://doi.org/10.3390/ijms23116137
Pietrowska A, Hołowacz I, Ulatowska-Jarża A, Guźniczak M, Matczuk AK, Wieliczko A, Wolf-Baca M, Buzalewicz I. The Enhancement of Antimicrobial Photodynamic Therapy of Escherichia Coli by a Functionalized Combination of Photosensitizers: In Vitro Examination of Single Cells by Quantitative Phase Imaging. International Journal of Molecular Sciences. 2022; 23(11):6137. https://doi.org/10.3390/ijms23116137
Chicago/Turabian StylePietrowska, Aleksandra, Iwona Hołowacz, Agnieszka Ulatowska-Jarża, Mateusz Guźniczak, Anna K. Matczuk, Alina Wieliczko, Mirela Wolf-Baca, and Igor Buzalewicz. 2022. "The Enhancement of Antimicrobial Photodynamic Therapy of Escherichia Coli by a Functionalized Combination of Photosensitizers: In Vitro Examination of Single Cells by Quantitative Phase Imaging" International Journal of Molecular Sciences 23, no. 11: 6137. https://doi.org/10.3390/ijms23116137
APA StylePietrowska, A., Hołowacz, I., Ulatowska-Jarża, A., Guźniczak, M., Matczuk, A. K., Wieliczko, A., Wolf-Baca, M., & Buzalewicz, I. (2022). The Enhancement of Antimicrobial Photodynamic Therapy of Escherichia Coli by a Functionalized Combination of Photosensitizers: In Vitro Examination of Single Cells by Quantitative Phase Imaging. International Journal of Molecular Sciences, 23(11), 6137. https://doi.org/10.3390/ijms23116137







