Mesenchymal Stromal Cell Therapy in Novel Porcine Model of Diffuse Liver Damage Induced by Repeated Biliary Obstruction
Abstract
1. Introduction
2. Results
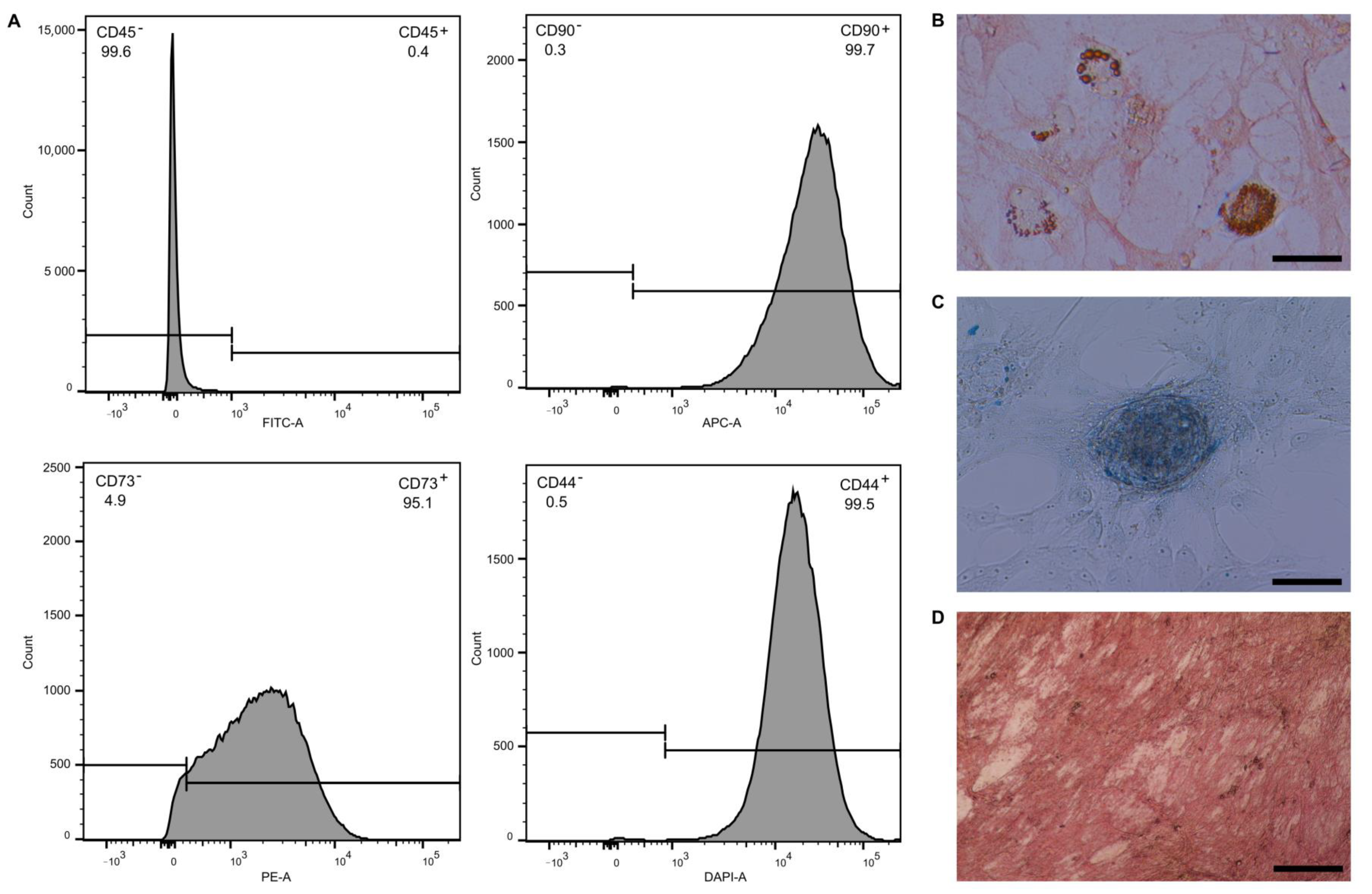
2.1. Evaluation of MSC Phenotype and Differentiation Ability
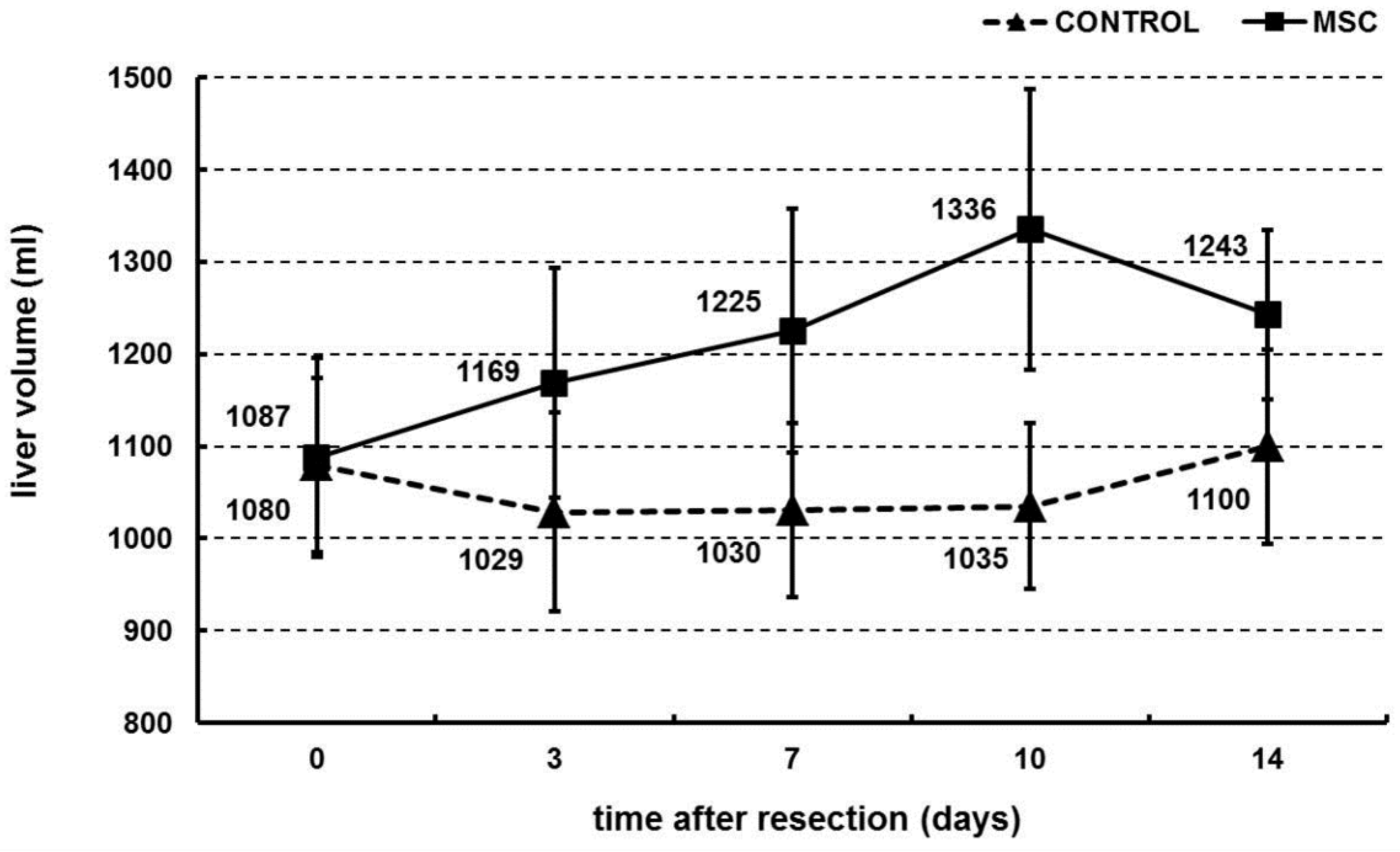
2.2. Portal Vein Transplantation of MSC Stimulated Liver Regeneration in a Porcine Model of Biliary Obstruction
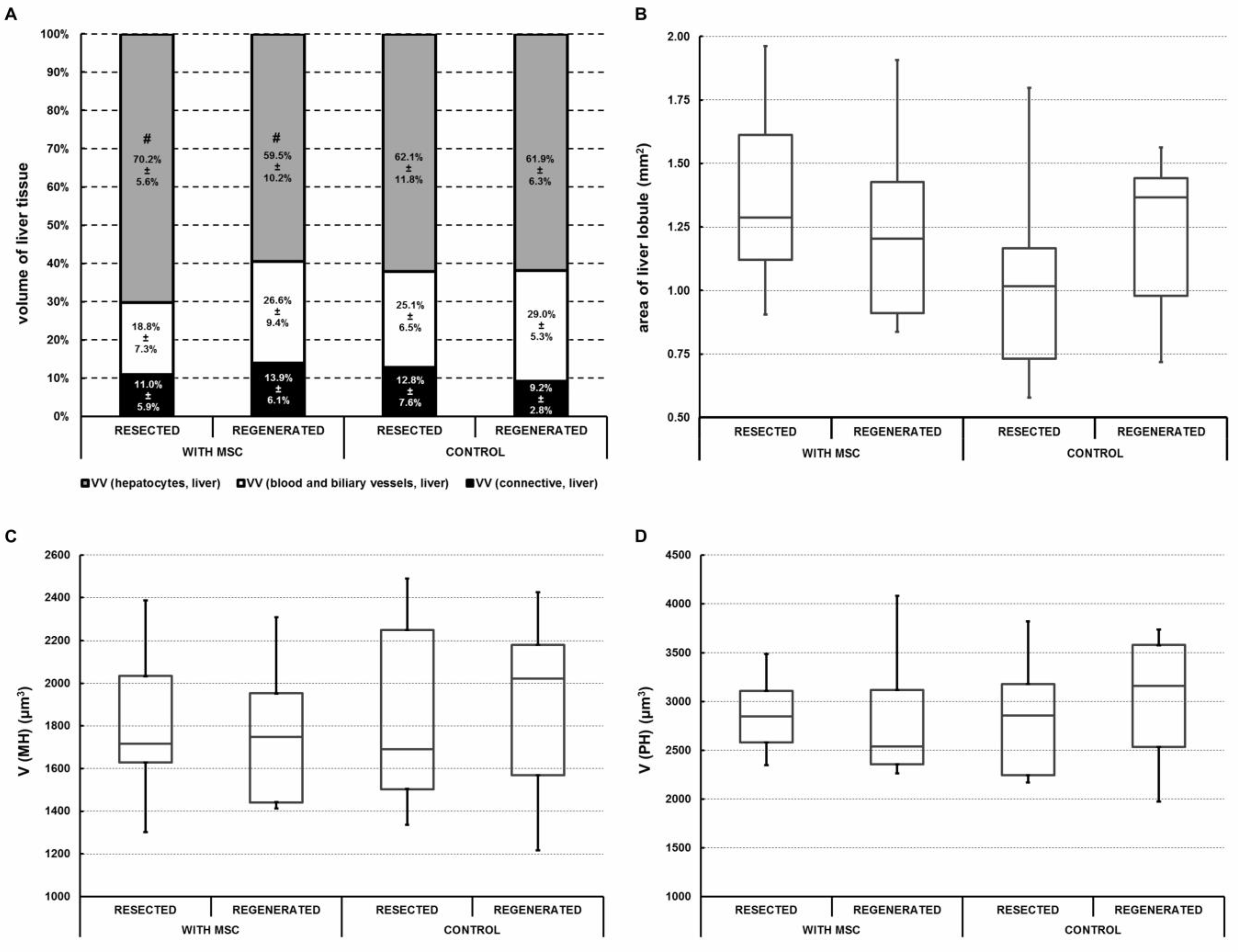
2.3. Portal Vein Transplantation of MSC Slightly Modulated Morphometric Parameters of the Regenerated Liver in a Porcine Model of Biliary Obstruction
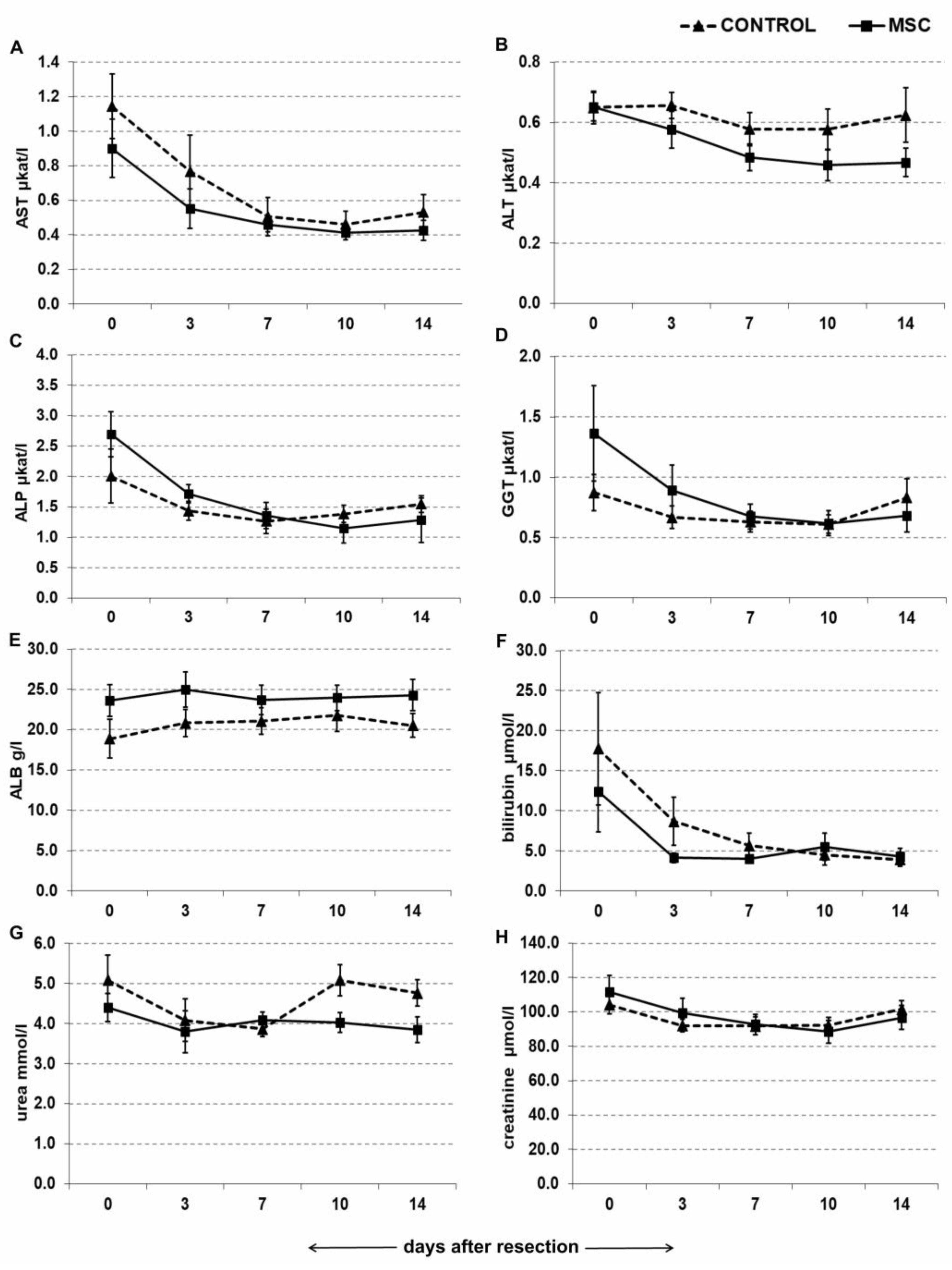
2.4. Portal Vein Transplantation of MSC Did Not Affect Biochemical Parameters in a Porcine Model of Biliary Obstruction
2.5. Portal Vein Transplantation of MSC Did Not Modulate Pro-Inflammatory Cytokines and TGF-β in a Porcine Model of Biliary Obstruction
3. Discussion
4. Materials and Methods
4.1. MSC Isolation and Culture
4.2. Identification of MSC by Flow Cytometry
4.3. Adipogenic, Osteogenic and Chondrogenic Differentiation Ability of MSC
4.4. Surgical Procedure and Development of Biliary Obstruction
4.4.1. Animal Preparation
4.4.2. Surgical Procedure
4.4.3. Postoperative Treatment
4.4.4. Liver Resection and MSC Transplantation
4.4.5. Postoperative Observation
4.5. Volumetry
4.6. Biochemical Analysis of Liver Enzymes
4.7. Quantification of Cytokines and Growth Factors in Plasma
4.8. Sampling and Histological Staining of Liver Tissue
4.9. Quantitative Morphometric Analysis of Liver Tissue
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Johnson, S.R.; Koehler, A.; Pennington, L.K.; Hanto, D.W. Long-term results of surgical repair of bile duct injuries following laparoscopic cholecystectomy. Surgery 2000, 128, 668–677. [Google Scholar] [CrossRef]
- Ardiles, V.; McCormack, L.; Quinonez, E.; Goldaracena, N.; Mattera, J.; Pekolj, J.; Ciardullo, M.; de Santibanes, E. Experience using liver transplantation for the treatment of severe bile duct injuries over 20 years in Argentina: Results from a National Survey. HPB 2011, 13, 544–550. [Google Scholar] [CrossRef] [PubMed]
- Barbier, L.; Souche, R.; Slim, K.; Ah-Soune, P. Long-term consequences of bile duct injury after cholecystectomy. J. Visc. Surg. 2014, 151, 269–279. [Google Scholar] [CrossRef] [PubMed]
- De Santibanes, E.; Ardiles, V.; Gadano, A.; Palavecino, M.; Pekolj, J.; Ciardullo, M. Liver transplantation: The last measure in the treatment of bile duct injuries. World J. Surg. 2008, 32, 1714–1721. [Google Scholar] [CrossRef]
- Lubikowski, J.; Chmurowicz, T.; Post, M.; Jarosz, K.; Bialek, A.; Milkiewicz, P.; Wojcicki, M. Liver transplantation as an ultimate step in the management of iatrogenic bile duct injury complicated by secondary biliary cirrhosis. Ann. Transpl. 2012, 17, 38–44. [Google Scholar]
- Ruemmele, P.; Hofstaedter, F.; Gelbmann, C.M. Secondary sclerosing cholangitis. Nat. Rev. Gastroenterol. Hepatol. 2009, 6, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Negi, S.S.; Sakhuja, P.; Malhotra, V.; Chaudhary, A. Factors predicting advanced hepatic fibrosis in patients with postcholecystectomy bile duct strictures. Arch. Surg. 2004, 139, 299–303. [Google Scholar] [CrossRef]
- Lazaridis, K.N.; Gores, G.J.; Lindor, K.D. Ursodeoxycholic acid “mechanisms of action and clinical use in hepatobiliary disorders”. J. Hepatol. 2001, 35, 134–146. [Google Scholar] [CrossRef]
- Nordin, A.; Halme, L.; Makisalo, H.; Isoniemi, H.; Hockerstedt, K. Management and outcome of major bile duct injuries after laparoscopic cholecystectomy: From therapeutic endoscopy to liver transplantation. Liver Transpl. 2002, 8, 1036–1043. [Google Scholar] [CrossRef]
- Patkowski, W.; Skalski, M.; Zieniewicz, K.; Nyckowski, P.; Smoter, P.; Krawczyk, M. Orthotopic liver transplantation for cholestatic diseases. Hepatogastroenterology 2010, 57, 605–610. [Google Scholar]
- Pottakkat, B.; Vijayahari, R.; Prakash, A.; Singh, R.K.; Behari, A.; Kumar, A.; Kapoor, V.K.; Saxena, R. Factors predicting failure following high bilio-enteric anastomosis for post-cholecystectomy benign biliary strictures. J. Gastrointest Surg. 2010, 14, 1389–1394. [Google Scholar] [CrossRef]
- Loinaz, C.; Gonzalez, E.M.; Jimenez, C.; Garcia, I.; Gomez, R.; Gonzalez-Pinto, I.; Colina, F.; Gimeno, A. Long-term biliary complications after liver surgery leading to liver transplantation. World J. Surg. 2001, 25, 1260–1263. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, S.I. Biliary tract surgery and cirrhosis: A critical combination. Surgery 1981, 90, 577–583. [Google Scholar] [PubMed]
- Chapman, W.C.; Halevy, A.; Blumgart, L.H.; Benjamin, I.S. Postcholecystectomy bile duct strictures. Management and outcome in 130 patients. Arch. Surg. 1995, 130, 594–597. [Google Scholar] [CrossRef]
- Fang, B.; Shi, M.; Liao, L.; Yang, S.; Liu, Y.; Zhao, R.C. Systemic infusion of FLK1(+) mesenchymal stem cells ameliorate carbon tetrachloride-induced liver fibrosis in mice. Transplantation 2004, 78, 83–88. [Google Scholar] [CrossRef]
- Li, T.; Zhu, J.; Ma, K.; Liu, N.; Feng, K.; Li, X.; Wang, S.; Bie, P. Autologous bone marrow-derived mesenchymal stem cell transplantation promotes liver regeneration after portal vein embolization in cirrhotic rats. J. Surg. Res. 2013, 184, 1161–1173. [Google Scholar] [CrossRef] [PubMed]
- Higashiyama, R.; Inagaki, Y.; Hong, Y.Y.; Kushida, M.; Nakao, S.; Niioka, M.; Watanabe, T.; Okano, H.; Matsuzaki, Y.; Shiota, G.; et al. Bone marrow-derived cells express matrix metalloproteinases and contribute to regression of liver fibrosis in mice. Hepatology 2007, 45, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Oyagi, S.; Hirose, M.; Kojima, M.; Okuyama, M.; Kawase, M.; Nakamura, T.; Ohgushi, H.; Yagi, K. Therapeutic effect of transplanting HGF-treated bone marrow mesenchymal cells into CCl4-injured rats. J. Hepatol. 2006, 44, 742–748. [Google Scholar] [CrossRef]
- Amin, M.A.; Sabry, D.; Rashed, L.A.; Aref, W.M.; el-Ghobary, M.A.; Farhan, M.S.; Fouad, H.A.; Youssef, Y.A. Short-term evaluation of autologous transplantation of bone marrow-derived mesenchymal stem cells in patients with cirrhosis: Egyptian study. Clin. Transpl. 2013, 27, 607–612. [Google Scholar] [CrossRef]
- Jang, Y.O.; Kim, Y.J.; Baik, S.K.; Kim, M.Y.; Eom, Y.W.; Cho, M.Y.; Park, H.J.; Park, S.Y.; Kim, B.R.; Kim, J.W.; et al. Histological improvement following administration of autologous bone marrow-derived mesenchymal stem cells for alcoholic cirrhosis: A pilot study. Liver Int. 2014, 34, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Gerling, B.; Becker, M.; Waldschmidt, J.; Rehmann, M.; Schuppan, D. Elevated serum aminoterminal procollagen type-III-peptide parallels collagen accumulation in rats with secondary biliary fibrosis. J. Hepatol. 1996, 25, 79–84. [Google Scholar] [CrossRef]
- Heller, J.; Trebicka, J.; Shiozawa, T.; Schepke, M.; Neef, M.; Hennenberg, M.; Sauerbruch, T. Vascular, hemodynamic and renal effects of low-dose losartan in rats with secondary biliary cirrhosis. Liver Int. 2005, 25, 657–666. [Google Scholar] [CrossRef] [PubMed]
- Raetsch, C.; Jia, J.D.; Boigk, G.; Bauer, M.; Hahn, E.G.; Riecken, E.O.; Schuppan, D. Pentoxifylline downregulates profibrogenic cytokines and procollagen I expression in rat secondary biliary fibrosis. Gut 2002, 50, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Shiesh, S.C.; Wu, M.C.; Lin, X.Z. The effects of bile duct obstruction on the biliary secretion of ciprofloxacin in piglets. Am. J. Gastroenterol. 1999, 94, 2408–2411. [Google Scholar] [CrossRef] [PubMed]
- Daneze, E.R.; Terra, G.A.; Terra, J.A., Jr.; Campos, A.G.; Silva, A.A.; Terra, S.A. Comparative study between ligature with thread or metallic clamping by means of laparoscopy with the purpose of experimental biliary obstruction in swines. Acta Cir. Bras. 2011, 26 (Suppl. S2), 31–37. [Google Scholar] [CrossRef] [PubMed]
- Shamiyeh, A.; Vattay, P.; Tulipan, L.; Schrenk, P.; Bogner, S.; Danis, J.; Wayand, W. Closure of the cystic duct during laparoscopic cholecystectomy with a new feedback-controlled bipolar sealing system in case of biliary obstruction—An experimental study in pigs. Hepatogastroenterology 2004, 51, 931–933. [Google Scholar]
- Adas, G.; Koc, B.; Adas, M.; Duruksu, G.; Subasi, C.; Kemik, O.; Kemik, A.; Sakiz, D.; Kalayci, M.; Purisa, S.; et al. Effects of mesenchymal stem cells and VEGF on liver regeneration following major resection. Langenbecks Arch. Surg. 2016, 401, 725–740. [Google Scholar] [CrossRef] [PubMed]
- Mohamadnejad, M.; Alimoghaddam, K.; Mohyeddin-Bonab, M.; Bagheri, M.; Bashtar, M.; Ghanaati, H.; Baharvand, H.; Ghavamzadeh, A.; Malekzadeh, R. Phase 1 trial of autologous bone marrow mesenchymal stem cell transplantation in patients with decompensated liver cirrhosis. Arch. Iran Med. 2007, 10, 459–466. [Google Scholar]
- Carvalho, A.B.; Quintanilha, L.F.; Dias, J.V.; Paredes, B.D.; Mannheimer, E.G.; Carvalho, F.G.; Asensi, K.D.; Gutfilen, B.; Fonseca, L.M.; Resende, C.M.; et al. Bone marrow multipotent mesenchymal stromal cells do not reduce fibrosis or improve function in a rat model of severe chronic liver injury. Stem. Cells 2008, 26, 1307–1314. [Google Scholar] [CrossRef]
- Suk, K.T.; Yoon, J.H.; Kim, M.Y.; Kim, C.W.; Kim, J.K.; Park, H.; Hwang, S.G.; Kim, D.J.; Lee, B.S.; Lee, S.H.; et al. Transplantation with autologous bone marrow-derived mesenchymal stem cells for alcoholic cirrhosis: Phase 2 trial. Hepatology 2016, 64, 2185–2197. [Google Scholar] [CrossRef] [PubMed]
- Verstegen, M.M.A.; Mezzanotte, L.; Yanto Ridwan, R.; Wang, K.; De Haan, J.; Schurink, I.J.; Sierra Parraga, J.M.; Hoogduijn, M.; Kessler, B.M.; Huang, H.; et al. First report on ex vivo delivery of paracrine active human mesenchymal stromal cells to liver grafts during machine perfusion. Transplantation 2020, 104, e5–e7. [Google Scholar] [CrossRef] [PubMed]
- De Witte, S.F.H.; Luk, F.; Sierra Parraga, J.M.; Gargesha, M.; Merino, A.; Korevaar, S.S.; Shankar, A.S.; O’Flynn, L.; Elliman, S.J.; Roy, D.; et al. Immunomodulation by Therapeutic Mesenchymal Stromal Cells (MSC) Is Triggered Through Phagocytosis of MSC By Monocytic Cells. Stem. Cells 2018, 36, 602–615. [Google Scholar] [CrossRef] [PubMed]
- You, Y.; Zhang, J.; Gong, J.; Chen, Y.; Li, Y.; Yang, K.; Liu, Z. Mesenchymal stromal cell-dependent reprogramming of Kupffer cells is mediated by TNF-alpha and PGE2 and is crucial for liver transplant tolerance. Immunol. Res. 2015, 62, 292–305. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Cheng, X.; Wang, H.; Huang, W.; la Ga Hu, Z.; Wang, D.; Zhang, K.; Zhang, H.; Xue, Z.; Da, Y.; et al. Mesenchymal stem cells and their secreted molecules predominantly ameliorate fulminant hepatic failure and chronic liver fibrosis in mice respectively. J. Transl. Med. 2016, 14, 45. [Google Scholar] [CrossRef] [PubMed]
- Jang, Y.O.; Kim, M.Y.; Cho, M.Y.; Baik, S.K.; Cho, Y.Z.; Kwon, S.O. Effect of bone marrow-derived mesenchymal stem cells on hepatic fibrosis in a thioacetamide-induced cirrhotic rat model. BMC Gastroenterol. 2014, 14, 198. [Google Scholar] [CrossRef]
- Forbes, S.J.; Russo, F.P.; Rey, V.; Burra, P.; Rugge, M.; Wright, N.A.; Alison, M.R. A significant proportion of myofibroblasts are of bone marrow origin in human liver fibrosis. Gastroenterology 2004, 126, 955–963. [Google Scholar] [CrossRef]
- Russo, F.P.; Alison, M.R.; Bigger, B.W.; Amofah, E.; Florou, A.; Amin, F.; Bou-Gharios, G.; Jeffery, R.; Iredale, J.P.; Forbes, S.J. The bone marrow functionally contributes to liver fibrosis. Gastroenterology 2006, 130, 1807–1821. [Google Scholar] [CrossRef]
- Junatas, K.L.; Tonar, Z.; Kubikova, T.; Liska, V.; Palek, R.; Mik, P.; Kralickova, M.; Witter, K. Stereological analysis of size and density of hepatocytes in the porcine liver. J. Anat. 2017, 230, 575–588. [Google Scholar] [CrossRef] [PubMed]
- Cupertino, M.C.; Costa, K.L.; Santos, D.C.; Novaes, R.D.; Condessa, S.S.; Neves, A.C.; Oliveira, J.A.; Matta, S.L. Long-lasting morphofunctional remodelling of liver parenchyma and stroma after a single exposure to low and moderate doses of cadmium in rats. Int. J. Exp. Pathol. 2013, 94, 343–351. [Google Scholar] [CrossRef]
- De Freitas, K.M.; Almeida, J.M.; Monteiro, J.C.; Diamante, M.A.; Vale, J.S.; Camargo, C.; Jorge, M.H.; Dolder, H. The effects of cyclosporin A and Heteropterys tomentosa on the rat liver. An. Acad. Bras. Cienc. 2015, 87, 369–379. [Google Scholar] [CrossRef]
- Gorla, G.R.; Malhi, H.; Gupta, S. Polyploidy associated with oxidative injury attenuates proliferative potential of cells. J. Cell Sci. 2001, 114, 2943–2951. [Google Scholar]
- Dai, L.J.; Li, H.Y.; Guan, L.X.; Ritchie, G.; Zhou, J.X. The therapeutic potential of bone marrow-derived mesenchymal stem cells on hepatic cirrhosis. Stem Cell Res. 2009, 2, 16–25. [Google Scholar] [CrossRef]
- Volarevic, V.; Nurkovic, J.; Arsenijevic, N.; Stojkovic, M. Concise review: Therapeutic potential of mesenchymal stem cells for the treatment of acute liver failure and cirrhosis. Stem Cells 2014, 32, 2818–2823. [Google Scholar] [CrossRef]
- Mattfeldt, T.; Mall, G.; Gharehbaghi, H.; Moller, P. Estimation of surface area and length with the orientator. J. Microsc. 1990, 159, 301–317. [Google Scholar] [CrossRef]
- Nyengaard, J.R.; Gundersen, H.J.G. The Isector—A Simple and Direct Method for Generating Isotropic, Uniform Random Sections from Small Specimens. J. Microsc. 1992, 165, 427–431. [Google Scholar] [CrossRef]
- Marcos, R.; Monteiro, R.A.; Rocha, E. The use of design-based stereology to evaluate volumes and numbers in the liver: A review with practical guidelines. J. Anat. 2012, 220, 303–317. [Google Scholar] [CrossRef] [PubMed]
- Mouton, P.R. Unbiased Stereology: A Concise Guide; Johns Hopkins University Press: Baltimore, MD, USA, 2011; ISBN 9780801899850. [Google Scholar]
- Nyengaard, J.R.; Gundersen, H.J.G. Sampling for stereology in lungs. Eur. Respir. Rev. 2006, 15, 107–114. [Google Scholar] [CrossRef]
- Bruha, J.; Vycital, O.; Tonar, Z.; Mirka, H.; Haidingerova, L.; Benes, J.; Palek, R.; Skala, M.; Treska, V.; Liska, V. Monoclonal antibody against transforming growth factor Beta 1 does not influence liver regeneration after resection in large animal experiments. In Vivo 2015, 29, 327–340. [Google Scholar] [PubMed]
- Gundersen, H.J.; Jensen, E.B. Stereological estimation of the volume-weighted mean volume of arbitrary particles observed on random sections. J. Microsc. 1985, 138, 127–142. [Google Scholar] [CrossRef] [PubMed]
- Mik, P.; Tonar, Z.; Malečková, A.; Eberlová, L.; Liška, V.; Pálek, R.; Rosendorf, J.; Jiřík, M.; Mírka, H.; Králíčková, M.; et al. Distribution of Connective Tissue in the Male and Female Porcine Liver: Histological Mapping and Recommendations for Sampling. J. Comp. Pathol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Palek, R.; Rosendorf, J.; Maleckova, A.; Vistejnova, L.; Bajcurova, K.; Mirka, H.; Tegl, V.; Brzon, O.; Kumar, A.; Bednar, L.; et al. Influence of Mesenchymal Stem Cell Administration on the Outcome of Partial Liver Resection in a Porcine Model of Sinusoidal Obstruction Syndrome. Anticancer Res. 2020. [Google Scholar] [CrossRef] [PubMed]


| Parameter 1 | Parameter 2 | WITH MSC | CONTROL | ||
|---|---|---|---|---|---|
| RESECTED | REGENERATED | RESECTED | REGENERATED | ||
| VV (hepatocytes,liver) | VV (BB,liver) | −0.76 | −0.71 | −0.82 | −0.93 |
| VV (MH,liver) | - | 0.69 | 0.66 | 0.23 | |
| VV (PH,liver) | - | −0.69 | −0.66 | −0.23 | |
| V (MH) | V (PH) | 0.65 | 0.72 | 0.78 | 0.61 |
| VV (MH,liver) | VV (PH,liver) | −0.96 | - | - | - |
| VV (connective,liver) | V (MH) | - | −0.78 | - | - |
| V (PH) | - | −0.70 | - | - | |
| Abbreviation | Parameter (Unit) | Stereological Principle Used for Quantification | Section Staining and Objective Magnification |
|---|---|---|---|
| A(lobule) | Mean cross-sectional area of classical morphological lobules (mm2) | Step 1. Systematic uniform random sampling of one lobule per tissue section for quantification. Step 2. Nucleator probe in isotropic uniform random (IUR) sections. | Anilin blue and nuclear red objective 2× |
| VV(connective, liver) | Volume fraction of connective tissue in the liver (%) | Step 1. Systematic uniform random sampling of microscopic image fields selected for quantification from multiple physical sections. Step 2. Point grid and Cavalieri of Delesse principle. | Anilin blue and nuclear red objective 2× |
| VV(hepatocytes, liver) | Volume fraction of hepatocytes in the liver (%) | Step 1. Systematic uniform random sampling of microscopic image fields selected for quantification from multiple physical sections. Step 2. Point grid and Cavalieri of Delesse principle. | PAS objective 40× |
| VV(MH,liver) | Volume fraction of mononuclear hepatocytes in the liver (%) | Step 1. Systematic uniform random sampling of microscopic image fields selected for quantification from multiple physical sections. Step 2. Point grid and Cavalieri of Delesse principle. | PAS objective 40× |
| VV(PH,liver) | Volume fraction of polynuclear hepatocytes in the liver (%) | Step 1. Systematic uniform random sampling of microscopic image fields selected for quantification from multiple physical sections. Step 2. Point grid and Cavalieri of Delesse principle. | PAS objective 40× |
| VV(BB,liver) | Volume fraction of blood and biliary vessels (%) | The parameter was calculated by subtracting the total liver volume and the fractions of connective tissue and hepatocytes from 1. | - |
| V(MH) | Mean volume of mononuclear hepatocytes (µm3) | Step 1. Systematic uniform random sampling of the lobules (at least 30 per tissue section) selected for quantification. Step 2. Nucleator probe in isotropic uniform random (IUR) sections. | PAS objective 40× |
| V(PH) | Mean volume of polynuclear hepatocytes (µm3) | Step 1. Systematic uniform random sampling of the lobules (at least 30 per tissue section) selected for quantification. Step 2. Nucleator probe in isotropic uniform random (IUR) sections. | PAS objective 40× |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vištejnová, L.; Liška, V.; Kumar, A.; Křečková, J.; Vyčítal, O.; Brůha, J.; Beneš, J.; Kolinko, Y.; Blassová, T.; Tonar, Z.; et al. Mesenchymal Stromal Cell Therapy in Novel Porcine Model of Diffuse Liver Damage Induced by Repeated Biliary Obstruction. Int. J. Mol. Sci. 2021, 22, 4304. https://doi.org/10.3390/ijms22094304
Vištejnová L, Liška V, Kumar A, Křečková J, Vyčítal O, Brůha J, Beneš J, Kolinko Y, Blassová T, Tonar Z, et al. Mesenchymal Stromal Cell Therapy in Novel Porcine Model of Diffuse Liver Damage Induced by Repeated Biliary Obstruction. International Journal of Molecular Sciences. 2021; 22(9):4304. https://doi.org/10.3390/ijms22094304
Chicago/Turabian StyleVištejnová, Lucie, Václav Liška, Arvind Kumar, Jana Křečková, Ondřej Vyčítal, Jan Brůha, Jan Beneš, Yaroslav Kolinko, Tereza Blassová, Zbyněk Tonar, and et al. 2021. "Mesenchymal Stromal Cell Therapy in Novel Porcine Model of Diffuse Liver Damage Induced by Repeated Biliary Obstruction" International Journal of Molecular Sciences 22, no. 9: 4304. https://doi.org/10.3390/ijms22094304
APA StyleVištejnová, L., Liška, V., Kumar, A., Křečková, J., Vyčítal, O., Brůha, J., Beneš, J., Kolinko, Y., Blassová, T., Tonar, Z., Brychtová, M., Karlíková, M., Racek, J., Mírka, H., Hošek, P., Lysák, D., & Králíčková, M. (2021). Mesenchymal Stromal Cell Therapy in Novel Porcine Model of Diffuse Liver Damage Induced by Repeated Biliary Obstruction. International Journal of Molecular Sciences, 22(9), 4304. https://doi.org/10.3390/ijms22094304






