Diffuse Idiopathic Skeletal Hyperostosis of Cervical Spine with Dysphagia—Molecular and Clinical Aspects
Abstract
1. Introduction
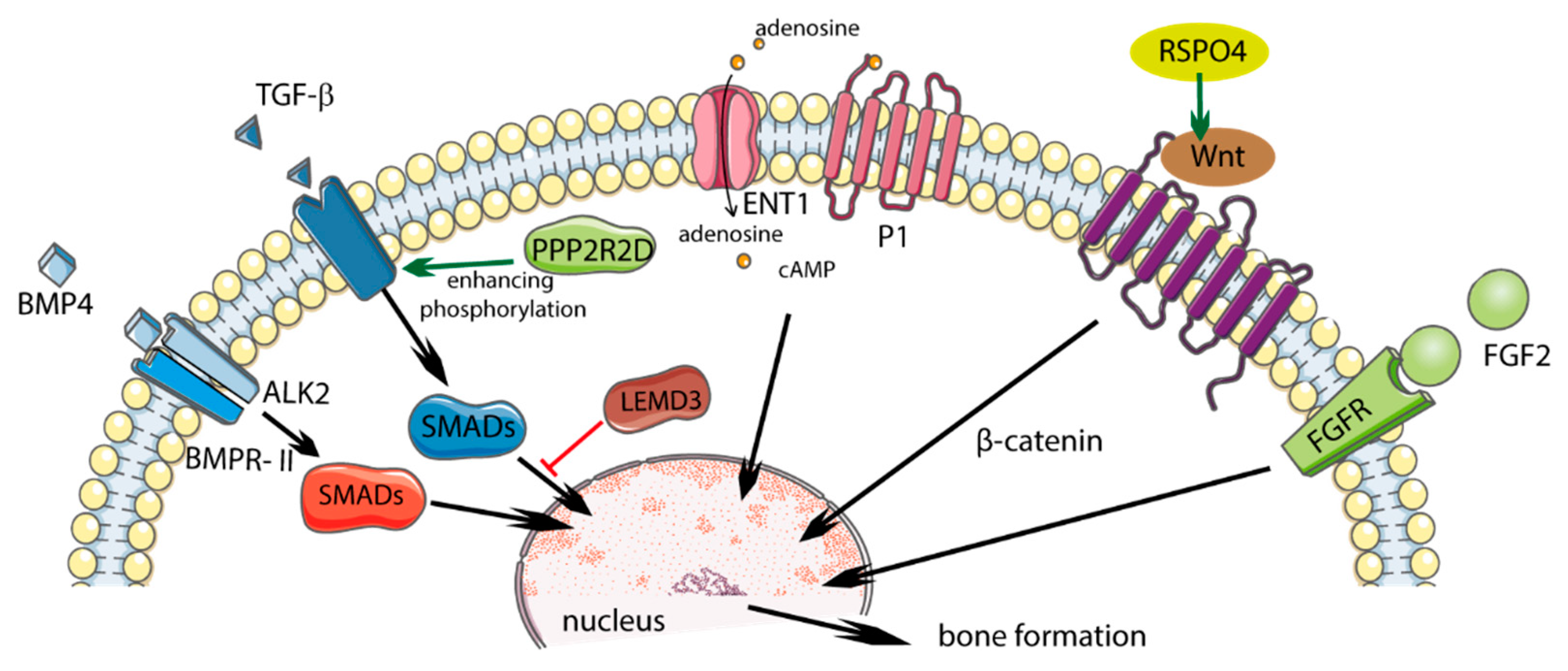
2. Etiopathogenesis—Molecular Aspects
2.1. Genetic Factors
2.1.1. COL6A1
2.1.2. FGF2
2.1.3. RSPO4
2.1.4. LEMD3
2.1.5. PPP2R2D
2.1.6. BMP4
2.1.7. ALK2
2.1.8. ENT1
2.1.9. Epigenetics in DISH
2.2. Metabolic Factors
2.2.1. Metabolic Syndrome and Obesity
2.2.2. Diabetes
2.2.3. Gout
2.3. Vascular Factors
2.4. Inflammatory Factors
3. Clinical Aspects
3.1. Symptoms
3.2. Diagnostics
3.3. Treatment
|
4. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Resnick, D.; Niwayama, G. Radiographic and pathologic features of spinal involvement in diffuse idiopathic skeletal hyperostosis (DISH). Radiology 1976, 119, 559–568. [Google Scholar] [CrossRef]
- Nascimento, F.A.; Gatto, L.A.; Lages, R.O.; Neto, H.M.; Demartini, Z.; Koppe, G.L. Diffuse idiopathic skeletal hyperostosis: A review. Surg. Neurol. Int. 2014, 5, 122–125. [Google Scholar] [CrossRef]
- Vaishya, R.; Vijay, V.; Nwagbara, I.C.; Agarwal, A.K. Diffuse idiopathic skeletal hyperostosis (DISH)—A common but less known cause of back pain. J. Clin. Orthop. Trauma 2017, 8, 191–196. [Google Scholar] [CrossRef][Green Version]
- Kuperus, J.S.; Mohamed Hoesein, F.A.A.; de Jong, P.A.; Verlaan, J.J. Diffuse idiopathic skeletal hyperostosis: Etiology and clinical relevance. Best Pract. Res. Clin. Rheumatol. 2020, 34, 101527. [Google Scholar] [CrossRef] [PubMed]
- Mader, R.; Verlaan, J.J.; Buskila, D. Diffuse idiopathic skeletal hyperostosis: Clinical features and pathogenic mechanisms. Nat. Rev. Rheumatol. 2013, 9, 741–750. [Google Scholar] [CrossRef] [PubMed]
- Mader, R.; Pappone, N.; Baraliakos, X.; Eshed, I.; Sarzi-Puttini, P.; Atzeni, F.; Bieber, A.; Novofastovski, I.; Kiefer, D.; Verlaan, J.J.; et al. Diffuse Idiopathic Skeletal Hyperostosis (DISH) and a Possible Inflammatory Component. Curr. Rheumatol. Rep. 2021, 23, 6. [Google Scholar] [CrossRef] [PubMed]
- Parreira, B. Genetic Variants Associated with Ectopic Calcifications. Ph.D. Thesis, University of Algarve, Faro, Portugal, 2018. [Google Scholar]
- Gregson, C.L.; Duncan, E.L. The Genetic Architecture of High Bone Mass. Front. Endocrinol. 2020, 11, 595653. [Google Scholar] [CrossRef]
- Frost, M.; Rahbek, E.T.; Ejersted, C.; Hoilund-Carlsen, P.F.; Bygum, A.; Thomsen, J.S.; Andreasen, C.M.; Andersen, T.L.; Frederiksen, A.L. Modeling-based bone formation transforms trabeculae to cortical bone in the sclerotic areas in Buschke-Ollendorff syndrome. A case study of two females with LEMD3 variants. Bone 2020, 135, 115313. [Google Scholar] [CrossRef]
- Gorman, C.; Jawad, A.S.; Chikanza, I. A family with diffuse idiopathic skeletal hyperostosis. Ann. Rheum. Dis. 2005, 64, 1794–1795. [Google Scholar] [CrossRef] [PubMed]
- Kranenburg, H.C.; Westerveld, L.A.; Verlaan, J.J.; Oner, F.C.; Dhert, W.J.; Voorhout, G.; Hazewinkel, H.A.; Meij, B.P. The dog as an animal model for DISH? Eur. Spine J. 2010, 19, 1325–1329. [Google Scholar] [CrossRef][Green Version]
- Warraich, S.; Bone, D.B.; Quinonez, D.; Ii, H.; Choi, D.S.; Holdsworth, D.W.; Drangova, M.; Dixon, S.J.; Seguin, C.A.; Hammond, J.R. Loss of equilibrative nucleoside transporter 1 in mice leads to progressive ectopic mineralization of spinal tissues resembling diffuse idiopathic skeletal hyperostosis in humans. J. Bone Miner. Res. 2013, 28, 1135–1149. [Google Scholar] [CrossRef]
- Couto, A.R.; Parreira, B.; Thomson, R.; Soares, M.; Power, D.M.; Stankovich, J.; Armas, J.B.; Brown, M.A. Combined approach for finding susceptibility genes in DISH/chondrocalcinosis families: Whole-genome-wide linkage and IBS/IBD studies. Hum. Genome Var. 2017, 4, 17041. [Google Scholar] [CrossRef]
- Parreira, B.; Couto, A.R.; Rocha, F.; Sousa, M.; Faustino, V.; Power, D.M.; Bruges-Armas, J. Whole exome sequencing of patients with diffuse idiopathic skeletal hyperostosis and calcium pyrophosphate crystal chondrocalcinosis. Acta Reumatol. Port. 2020, 45, 116–126. [Google Scholar] [PubMed]
- Jun, J.K.; Kim, S.M. Association study of fibroblast growth factor 2 and fibroblast growth factor receptors gene polymorphism in korean ossification of the posterior longitudinal ligament patients. J. Korean Neurosurg. Soc. 2012, 52, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Tsukahara, S.; Miyazawa, N.; Akagawa, H.; Forejtova, S.; Pavelka, K.; Tanaka, T.; Toh, S.; Tajima, A.; Akiyama, I.; Inoue, I. COL6A1, the candidate gene for ossification of the posterior longitudinal ligament, is associated with diffuse idiopathic skeletal hyperostosis in Japanese. Spine 2005, 30, 2321–2324. [Google Scholar] [CrossRef] [PubMed]
- Servier Medical Art. Available online: https://smart.servier.com (accessed on 4 February 2021).
- Roberts, S.J.; Ke, H.Z. Anabolic Strategies to Augment Bone Fracture Healing. Curr. Osteoporos. Rep. 2018, 16, 289–298. [Google Scholar] [CrossRef]
- Moosa, S.; Wollnik, B. Altered FGF signalling in congenital craniofacial and skeletal disorders. Semin. Cell Dev. Biol. 2016, 53, 115–125. [Google Scholar] [CrossRef]
- Jann, J.; Gascon, S.; Roux, S.; Faucheux, N. Influence of the TGF-beta Superfamily on Osteoclasts/Osteoblasts Balance in Physiological and Pathological Bone Conditions. Int. J. Mol. Sci. 2020, 21, 7597. [Google Scholar] [CrossRef]
- Gupta, A.; Zimmermann, M.T.; Wang, H.; Broski, S.M.; Sigafoos, A.N.; Macklin, S.K.; Urrutia, R.A.; Clark, K.J.; Atwal, P.S.; Pignolo, R.J.; et al. Molecular characterization of known and novel ACVR1 variants in phenotypes of aberrant ossification. Am. J. Med. Genet. A 2019, 179, 1764–1777. [Google Scholar] [CrossRef]
- Tsukamoto, S.; Kuratani, M.; Katagiri, T. Functional characterization of a unique mutant of ALK2, p.K400E, that is associated with a skeletal disorder, diffuse idiopathic skeletal hyperostosis. Bone 2020, 137, 115410. [Google Scholar] [CrossRef]
- Lopez, C.D.; Bekisz, J.M.; Corciulo, C.; Mediero, A.; Coelho, P.G.; Witek, L.; Flores, R.L.; Cronstein, B.N. Local delivery of adenosine receptor agonists to promote bone regeneration and defect healing. Adv. Drug Deliv. Rev. 2019, 146, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Corciulo, C.; Cronstein, B.N. Signaling of the Purinergic System in the Joint. Front. Pharmacol. 2019, 10, 1591. [Google Scholar] [CrossRef] [PubMed]
- Daniels, G.; Ballif, B.A.; Helias, V.; Saison, C.; Grimsley, S.; Mannessier, L.; Hustinx, H.; Lee, E.; Cartron, J.P.; Peyrard, T.; et al. Lack of the nucleoside transporter ENT1 results in the Augustine-null blood type and ectopic mineralization. Blood 2015, 125, 3651–3654. [Google Scholar] [CrossRef]
- Dixon, J.; Beaucage, K.; Nagao, M.; Lajoie, G.; Veras, M.; Fournier, D.; Holdsworth, D.; Bailey, C.; Hammond, J.; Séguin, C. Mice lacking the nucleoside transporter ENT1: A model of diffuse idiopathic skeletal hyperostosis in humans. In Proceedings of the Orthopaedic Proceedings, Montreal, QC, Canada, 17 July 2020; p. 123. [Google Scholar]
- Strazzulla, L.C.; Cronstein, B.N. Regulation of bone and cartilage by adenosine signaling. Purinergic Signal. 2016, 12, 583–593. [Google Scholar] [CrossRef] [PubMed]
- Husain, A.; Jeffries, M.A. Epigenetics and Bone Remodeling. Curr. Osteoporos. Rep. 2017, 15, 450–458. [Google Scholar] [CrossRef] [PubMed]
- Chiba, N.; Furukawa, K.; Takayama, S.; Asari, T.; Chin, S.; Harada, Y.; Kumagai, G.; Wada, K.; Tanaka, T.; Ono, A.; et al. Decreased DNA methylation in the promoter region of the WNT5A and GDNF genes may promote the osteogenicity of mesenchymal stem cells from patients with ossified spinal ligaments. J. Pharmacol. Sci. 2015, 127, 467–473. [Google Scholar] [CrossRef]
- Yuan, X.; Shi, L.; Chen, Y. Non-coding RNAs in ossification of spinal ligament. Eur. Spine J. 2021, 30, 801–808. [Google Scholar] [CrossRef] [PubMed]
- Hong, L.; Sun, H.; Amendt, B.A. MicroRNA function in craniofacial bone formation, regeneration and repair. Bone 2021, 144, 115789. [Google Scholar] [CrossRef]
- Zhang, H.; Xu, C.; Liu, Y.; Yuan, W. MicroRNA-563 promotes the osteogenic differentiation of posterior longitudinal ligament cells by inhibiting SMURF1. Zhonghua Wai Ke Za Zhi 2017, 55, 203–207. [Google Scholar] [CrossRef]
- Esser, J.S.; Saretzki, E.; Pankratz, F.; Engert, B.; Grundmann, S.; Bode, C.; Moser, M.; Zhou, Q. Bone morphogenetic protein 4 regulates microRNAs miR-494 and miR-126-5p in control of endothelial cell function in angiogenesis. Thromb. Haemost. 2017, 117, 734–749. [Google Scholar] [CrossRef]
- Pillai, S.; Littlejohn, G. Metabolic factors in diffuse idiopathic skeletal hyperostosis—A review of clinical data. Open Rheumatol. J. 2014, 8, 116–128. [Google Scholar] [CrossRef] [PubMed]
- Chaput, C.D.; Siddiqui, M.; Rahm, M.D. Obesity and calcification of the ligaments of the spine: A comprehensive CT analysis of the entire spine in a random trauma population. Spine J. 2019, 19, 1346–1353. [Google Scholar] [CrossRef]
- Sohn, S.; Chung, C.K.; Han, I.; Park, S.B.; Kim, H. Increased Bone Mineral Density in Cervical or Thoracic Diffuse Idiopathic Skeletal Hyperostosis (DISH): A Case-Control Study. J. Clin. Densitom. 2018, 21, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Mader, R.; Novofestovski, I.; Adawi, M.; Lavi, I. Metabolic syndrome and cardiovascular risk in patients with diffuse idiopathic skeletal hyperostosis. Semin. Arthritis Rheum. 2009, 38, 361–365. [Google Scholar] [CrossRef]
- Glick, K.; Novofastovski, I.; Schwartz, N.; Mader, R. Cardiovascular disease in diffuse idiopathic skeletal hyperostosis (DISH): From theory to reality-a 10-year follow-up study. Arthritis Res. Ther. 2020, 22, 190. [Google Scholar] [CrossRef] [PubMed]
- Fassio, A.; Adami, G.; Idolazzi, L.; Giollo, A.; Viapiana, O.; Bosco, E.; Negrelli, R.; Sani, E.; Sandri, D.; Mantovani, A.; et al. Diffuse Idiopathic Skeletal Hyperostosis (DISH) in Type 2 Diabetes: A New Imaging Possibility and a New Biomarker. Calcif. Tissue Int. 2021, 108, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Mader, R.; Lavi, I. Diabetes mellitus and hypertension as risk factors for early diffuse idiopathic skeletal hyperostosis (DISH). Osteoarthr. Cartilage 2009, 17, 825–828. [Google Scholar] [CrossRef]
- Corkill, M.; Topless, R.; Worthington, A.; Mitchell, R.; Gregory, K.; Stamp, L.K.; Brown, M.; Merriman, T.R.; Dalbeth, N. Exploring the Relationship between Gout and Diffuse Idiopathic Skeletal Hyperostosis (DISH): An Epidemiologic and Genetic Study. Arthritis Rheumatol. 2018, 70 (Suppl. 10). Available online: https://acrabstracts.org/abstract/exploring-the-relationship-between-gout-and-diffuse-idiopathic-skeletal-hyperostosis-dish-an-epidemiologic-and-genetic-study/ (accessed on 4 February 2021).
- El Miedany, Y.M.; Wassif, G.; el Baddini, M. Diffuse idiopathic skeletal hyperostosis (DISH): Is it of vascular aetiology? Clin. Exp. Rheumatol. 2000, 18, 193–200. [Google Scholar]
- Pappone, N.; Ambrosino, P.; di Minno, M.N.D.; Iervolino, S. Is diffuse idiopathic skeletal hyperostosis a disease or a syndrome? Rheumatology 2017, 56, 1635–1636. [Google Scholar] [CrossRef][Green Version]
- Bakker, J.T.; Kuperus, J.S.; Kuijf, H.J.; Oner, F.C.; de Jong, P.A.; Verlaan, J.J. Morphological characteristics of diffuse idiopathic skeletal hyperostosis in the cervical spine. PLoS ONE 2017, 12, e0188414. [Google Scholar] [CrossRef]
- Xu, M.; Liu, Y.; Yang, J.; Liu, H.; Ding, C. Ossification of the cervical anterior longitudinal ligament is an underdiagnosed cause of difficult airway: A case report and review of the literature. BMC Anesthesiol. 2020, 20, 161. [Google Scholar] [CrossRef] [PubMed]
- Dell’Era, V.; Garzaro, M.; Farri, F.; Gorris, C.; Rosa, M.S.; Toso, A.; Aluffi Valletti, P. Respiratory presentation of diffuse idiopathic skeletal hyperostosis (DISH): Management and review of the literature. Cranio 2019, 1–4. [Google Scholar] [CrossRef]
- Souza, S.; Raggio, B.; Bareiss, A.; Friedlander, P. Diffuse Idiopathic Skeletal Hyperostosis of the Cervical Spine: A Risk for Acute Airway Obstruction. Ear Nose Throat J. 2020. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, H.; Endo, K.; Aihara, T.; Murata, K.; Suzuki, H.; Matsuoka, Y.; Takamatsu, T.; Maekawa, A.; Sawaji, Y.; Tsuji, H.; et al. Risk factors of dysphagia in patients with ossification of the anterior longitudinal ligament. J. Orthop. Surg. 2020, 28. [Google Scholar] [CrossRef]
- Garcia Callejo, F.J.; Oishi, N.; Lopez Sanchez, I.; Pallares Marti, B.; Rubio Fernandez, A.; Gomez Gomez, M.J. Incidence of diffuse idiopathic skeletal hyperostosis from a model of dysphagia. Acta Otorrinolaringol. Esp. 2020, 71, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Paeng, S.H.; Pyo, S.Y.; Kim, S.T.; Lee, W.H. Swallowing Difficulty in Diffuse Idiopathic Skeletal Hyperostosis with Metabolic Syndrome. Korean J. Neurotrauma 2020, 16, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Bartalena, T.; Buia, F.; Borgonovi, A.; Rinaldi, M.F.; Modolon, C.; Bassi, F. DISH of the cervical spine causing epiglottis impingement. Indian J. Radiol. Imaging 2009, 19, 132–134. [Google Scholar] [CrossRef]
- Candelario, N.; Lo, K.B.; Naranjo, M. Cervical diffuse idiopathic skeletal hyperostosis (DISH) causing oropharyngeal dysphagia. BMJ Case Rep. 2017, 2017. [Google Scholar] [CrossRef]
- Dabrowski, M.; Sulewski, A.; Kaczmarczyk, J.; Kubaszewski, L. Surgical treatment of diffuse idiopathic skeletal hyperostosis of cervical spine with dysphagia—Case report. Ann. Med. Surg. 2020, 57, 37–40. [Google Scholar] [CrossRef]
- Weglowski, R.; Piech, P. Dysphagia as a symptom of anterior cervical hyperostosis—Case report. Ann. Agric. Environ. Med. 2020, 27, 314–316. [Google Scholar] [CrossRef]
- Mader, R.; Baraliakos, X.; Eshed, I.; Novofastovski, I.; Bieber, A.; Verlaan, J.J.; Kiefer, D.; Pappone, N.; Atzeni, F. Imaging of diffuse idiopathic skeletal hyperostosis (DISH). RMD Open 2020, 6. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Shahi, P.B.; Adsul, N.; Acharya, S.; Kalra, K.L.; Chahal, R.S. Progressive dysphagia and dysphonia secondary to DISH-related anterior cervical osteophytes: A case report. Surg. Neurol. Int. 2020, 11, 69. [Google Scholar] [CrossRef] [PubMed]
- Butler, A.J.; Ghasem, A.; Al Maaieh, M. Dysphagia following lumbar spine surgery in the setting of undiagnosed DISH of the cervical spine: A case report. AME Case Rep. 2019, 3, 13. [Google Scholar] [CrossRef]
- Kuperus, J.S.; Oudkerk, S.F.; Foppen, W.; Mohamed Hoesein, F.A.; Gielis, W.P.; Waalwijk, J.; Regan, E.A.; Lynch, D.A.; Oner, F.C.; de Jong, P.A.; et al. Criteria for Early-Phase Diffuse Idiopathic Skeletal Hyperostosis: Development and Validation. Radiology 2019, 291, 420–426. [Google Scholar] [CrossRef] [PubMed]
- Soejima, Y.; Arima, J.; Doi, T. Diffuse Idiopathic Skeletal Hyperostosis: A Case with Dysphonia, Dysphagia and Myelopathy. Am. J. Case Rep. 2019, 20, 349–353. [Google Scholar] [CrossRef]
- Zarei, M.; Golbakhsh, M.; Rostami, M.; Moosavi, M. Dysphonia, Stridor, and Dysphagia Caused by Diffuse Idiopathic Skeletal Hyperostosis: Case Report and Review of Literature. Adv. Biomed. Res. 2020, 9, 47. [Google Scholar] [CrossRef]
- Kolz, J.M.; Alvi, M.A.; Bhatti, A.R.; Tomov, M.N.; Bydon, M.; Sebastian, A.S.; Elder, B.D.; Nassr, A.N.; Fogelson, J.L.; Currier, B.L.; et al. Anterior Cervical Osteophyte Resection for Treatment of Dysphagia. Glob. Spine J. 2020. [Google Scholar] [CrossRef]
- Von der Hoeh, N.H.; Voelker, A.; Jarvers, J.S.; Gulow, J.; Heyde, C.E. Results after the surgical treatment of anterior cervical hyperostosis causing dysphagia. Eur. Spine J. 2015, 24, 489–493. [Google Scholar] [CrossRef]
- Ruetten, S.; Baraliakos, X.; Godolias, G.; Komp, M. Surgical treatment of anterior cervical osteophytes causing dysphagia. J. Orthop. Surg. 2019, 27. [Google Scholar] [CrossRef]
- Von Glinski, A.; Takayanagi, A.; Elia, C.; Ishak, B.; Listmann, M.; Pierre, C.A.; Blecher, R.; Hayman, E.; Chapman, J.R.; Oskouian, R.J. Surgical Treatment of Ossifications of the Cervical Anterior Longitudinal Ligament: A Retrospective Cohort Study. Glob. Spine J. 2020. [Google Scholar] [CrossRef] [PubMed]
- Damade, C.; Masse, R.; Ghailane, S.; Petit, M.; Castelain, J.E.; Gille, O.; Mazas, S.; Bouyer, B.; Kieser, D.C. Anterior Cervical Idiopathic Hyperostosis and Dysphagia: The Impact of Surgical Management—Study of a Series of 11 Cases. World Neurosurg. 2020, 138, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, K.; Sugiyama, S.; Hosoe, H.; Iinuma, N.; Suzuki, Y.; Shimizu, K. Postsurgical recurrence of osteophytes causing dysphagia in patients with diffuse idiopathic skeletal hyperostosis. Eur. Spine J. 2009, 18, 1652–1658. [Google Scholar] [CrossRef] [PubMed]
| Gene | SNP | Population | Disorder Associated with DISH | References |
|---|---|---|---|---|
| FGF2 | rs1476217/rs3747676 | Korean | OPLL | Jun et al. [15] |
| COL6A1 | intron 32 (−29) | Japanese | OPLL | Tsukahara et al. [16] |
| PPP2R2D | rs34473884 | Azorean | CC | Parreira et al. [14] |
| RSPO4 | rs146447064, rs14915407 | Azorean | CC | Couto er al. [13] |
| BMP4 | rs17563 | Azorean | CC | Couto er al. [13] |
| LEMD3 | rs201930700 | Azorean | CC | Couto er al. [13] |
| Symptom | Characteristics and Frequency | Mechanism |
|---|---|---|
| Neck pain, radicular pain, and stiffness. | Frequently, motor segments are C5/C6 (40%) followed by C4/C5. | Increasing degeneration of intervertebral discs and ossification of posterior longitudinal ligament may lead to narrowing of spinal canal. |
| Dysphagia (DISHphagia) | Present with solid foods, improved by neck flexion, and aggravated by neck straightening [48]. Incidence in patients with DISH is in the range of 0.2–28% [49]. Combined symptoms: foreign-body sensation, odynophagia, saliva stagnation, dysphonia, dyspnea [50]. Incidence of dysphagia in patients with cervical-spine DISH was influenced by osteophyte thickness, cervical-spine mobility, and craniocervical position [48]. | Lower cervical-spine osteophytes on levels C4–C6 can cause esophageal stricture and mechanical trouble, causing varying degrees of esophageal obstruction, impaired motility of the epiglottis, and larynx-cartilage deformity [51]. Large osteophytes can lead to dysphagia through direct mechanical obstruction; smaller osteophytes can act at the sites accompanying immobilization of the esophagus because the esophagus is anatomically anchored on the cricoid-cartilage and diaphragm level. Chronic pressure may cause inflammatory reaction in the wall of the esophagus and adjacent soft tissue, leading to fibrosis and adhesions, and painful reflex esophageal-spasm-induced irritant osteophyte [52]. Recurrent nerve paralysis caused by hyperostosis may aggravate narrowing [50]. |
| Breathing disturbances | Concomitant symptoms: dyspnea (14%), cough (3%), dysphonia (2.5%), and stridor [3,47]. | Upper cervical-spine osteophytes are responsible for the oropharynx, causing respiratory distress and stridor. Respiratory symptoms related to DISH are associated with osteophytes inflammation and fibrosis of the esophageal wall and adjacent tissues causing obstruction of the upper respiratory tract, and direct lesion of the recurrent laryngeal nerve [3,47] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dąbrowski, M.; Kubaszewski, Ł. Diffuse Idiopathic Skeletal Hyperostosis of Cervical Spine with Dysphagia—Molecular and Clinical Aspects. Int. J. Mol. Sci. 2021, 22, 4255. https://doi.org/10.3390/ijms22084255
Dąbrowski M, Kubaszewski Ł. Diffuse Idiopathic Skeletal Hyperostosis of Cervical Spine with Dysphagia—Molecular and Clinical Aspects. International Journal of Molecular Sciences. 2021; 22(8):4255. https://doi.org/10.3390/ijms22084255
Chicago/Turabian StyleDąbrowski, Mikołaj, and Łukasz Kubaszewski. 2021. "Diffuse Idiopathic Skeletal Hyperostosis of Cervical Spine with Dysphagia—Molecular and Clinical Aspects" International Journal of Molecular Sciences 22, no. 8: 4255. https://doi.org/10.3390/ijms22084255
APA StyleDąbrowski, M., & Kubaszewski, Ł. (2021). Diffuse Idiopathic Skeletal Hyperostosis of Cervical Spine with Dysphagia—Molecular and Clinical Aspects. International Journal of Molecular Sciences, 22(8), 4255. https://doi.org/10.3390/ijms22084255







