An Integrated—Omics/Chemistry Approach Unravels Enzymatic and Spontaneous Steps to Form Flavoalkaloidal Nudicaulin Pigments in Flowers of Papaver nudicaule L.
Abstract
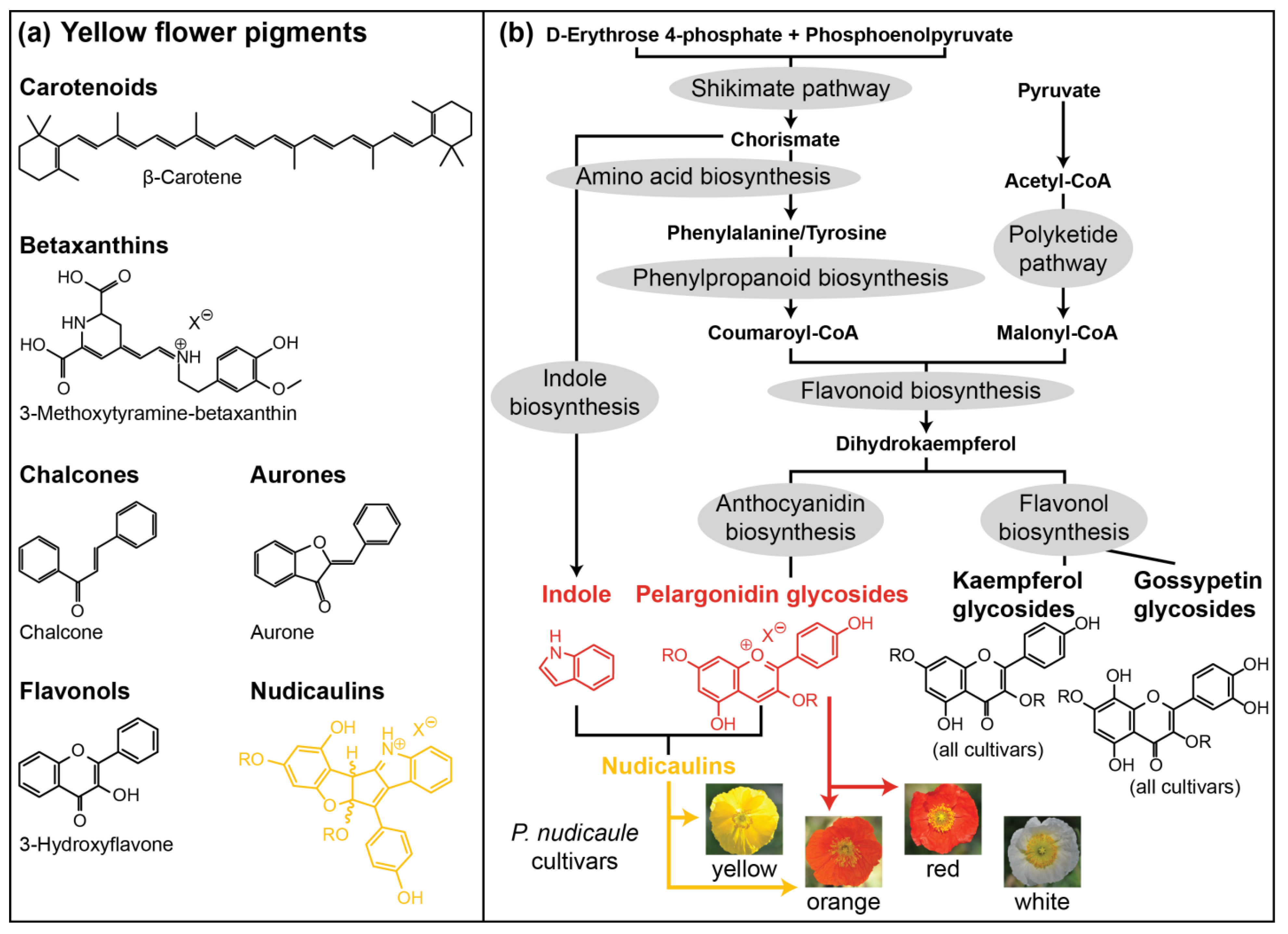
1. Introduction
2. Results and Discussion
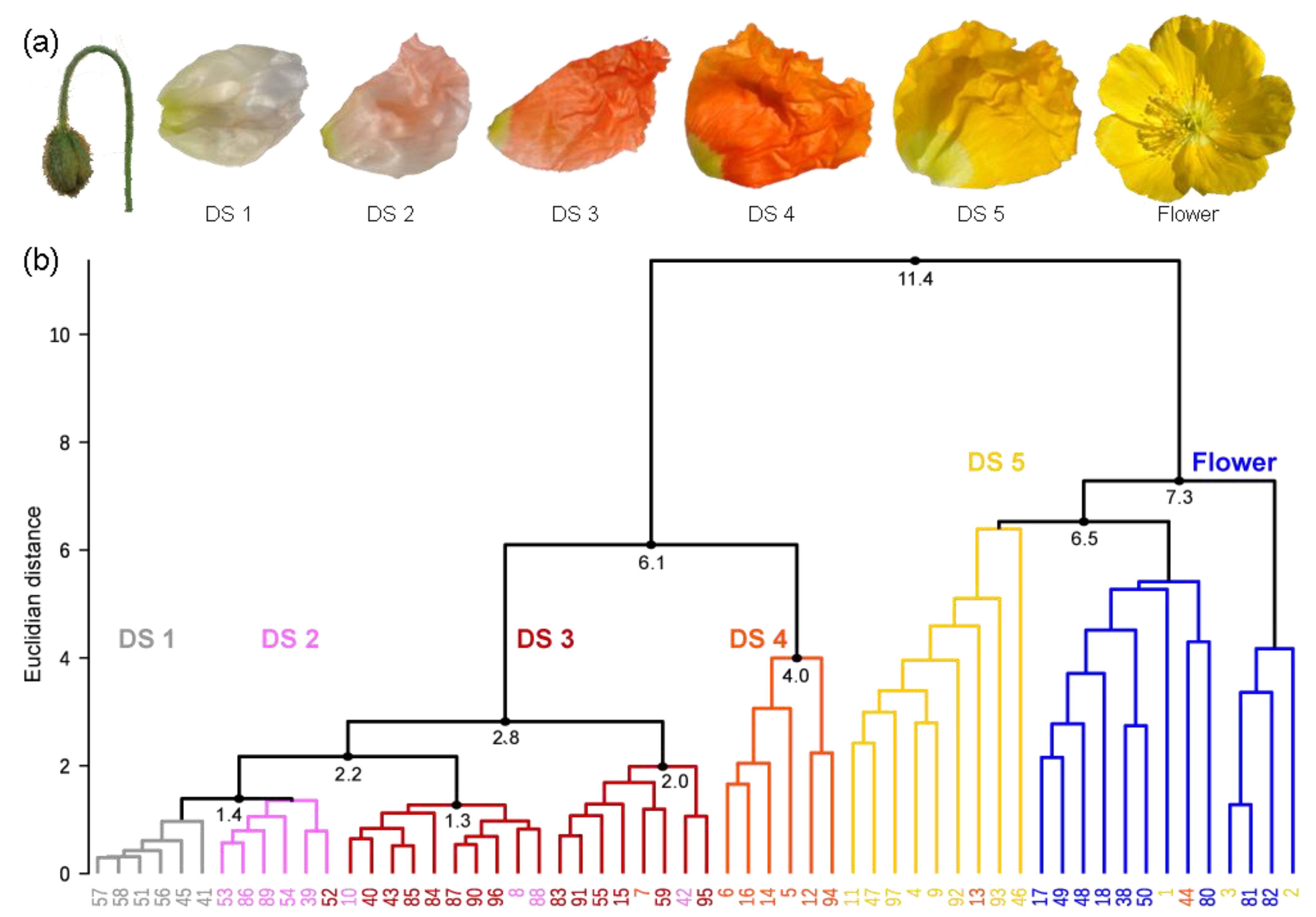
2.1. Definition of Stages in the Petal Development of the Yellow P. nudicaule Cultivar
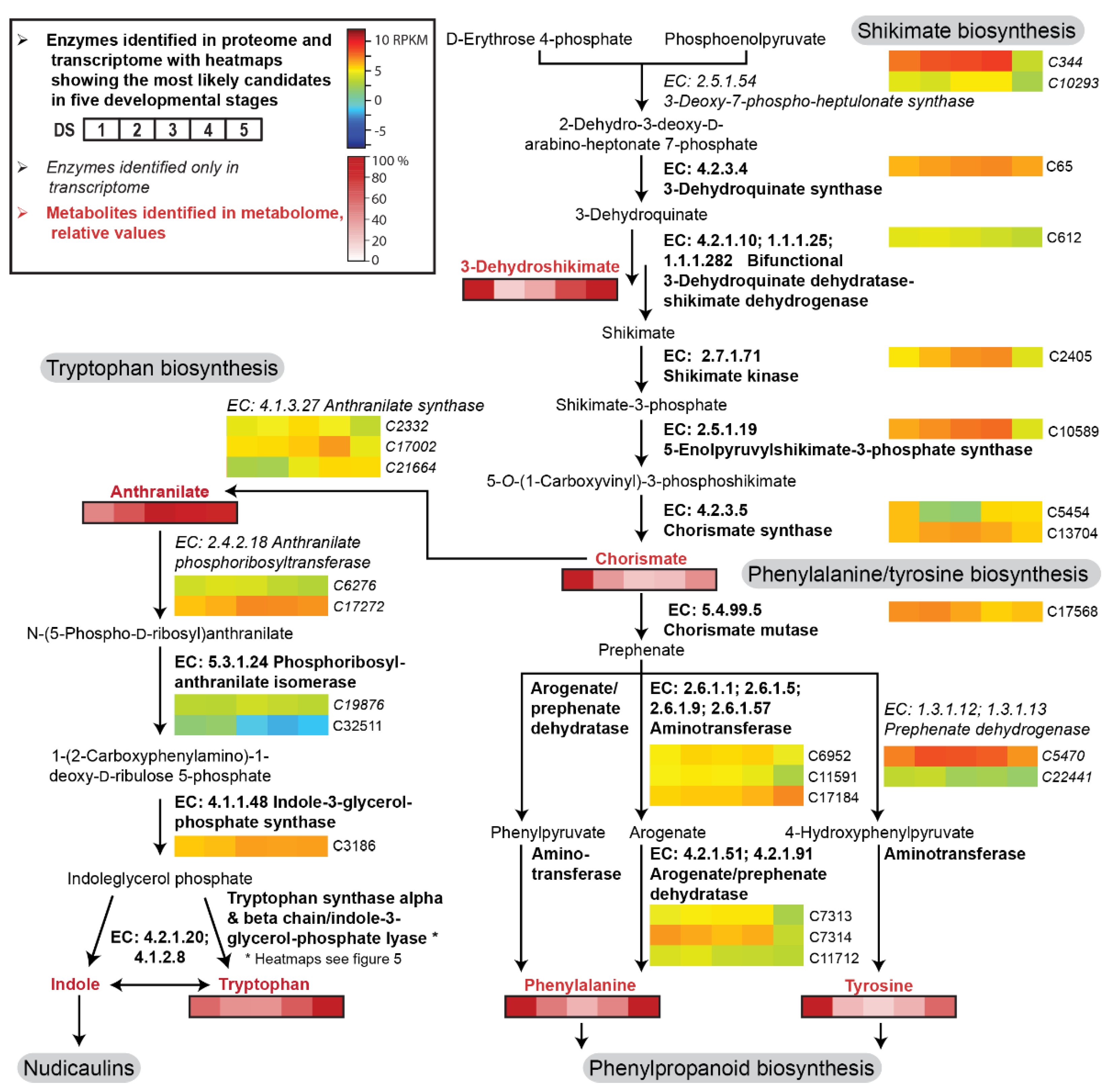
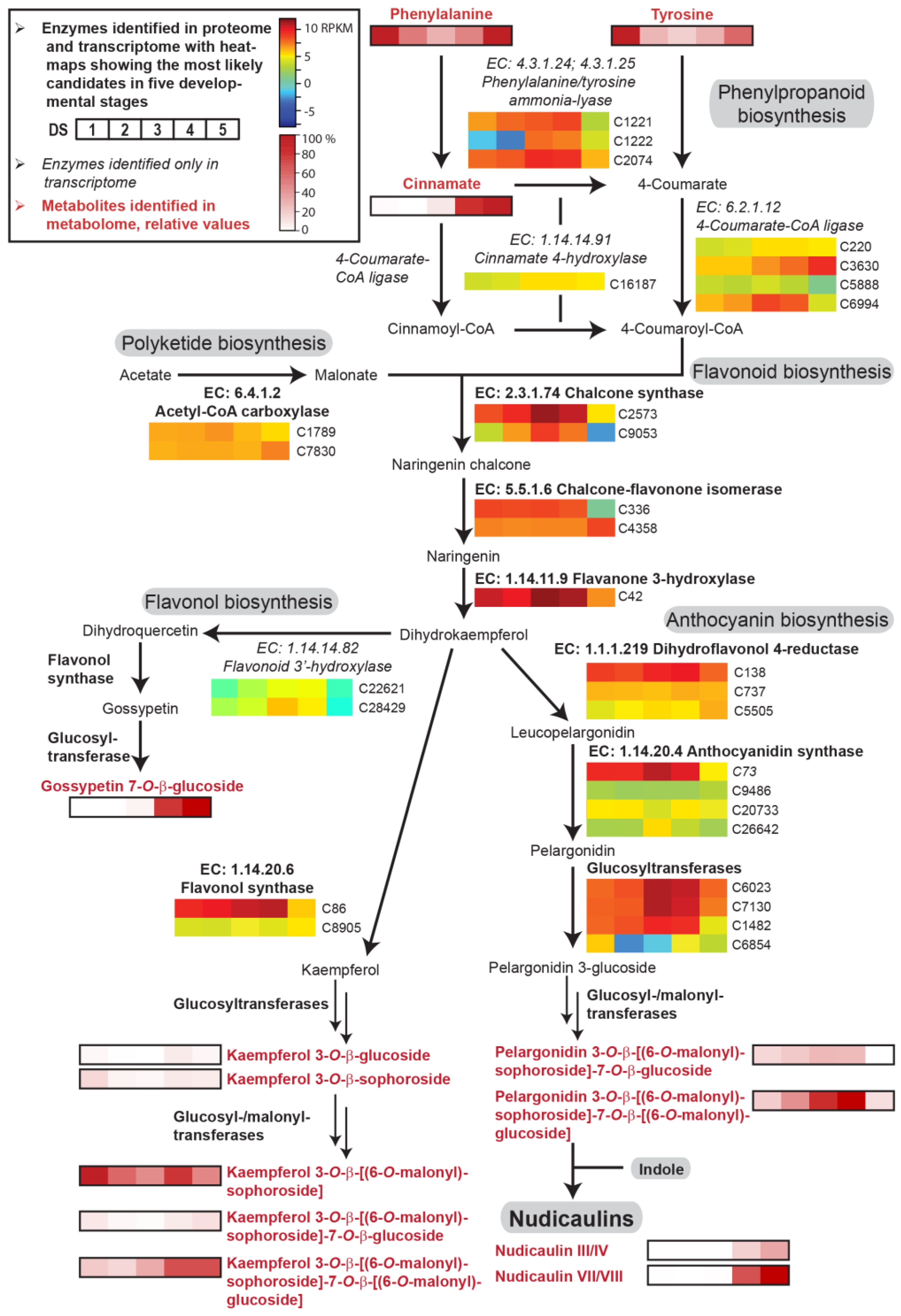
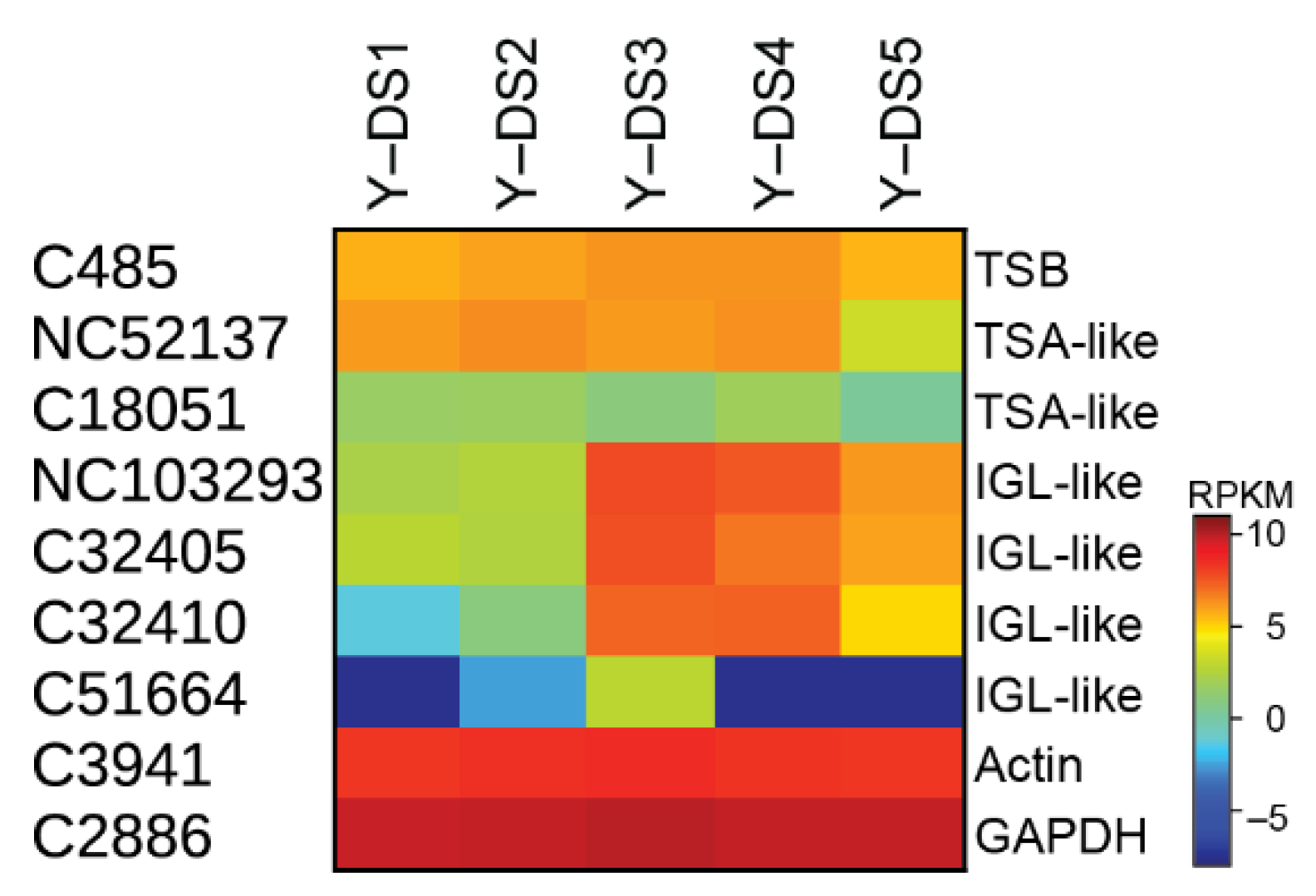
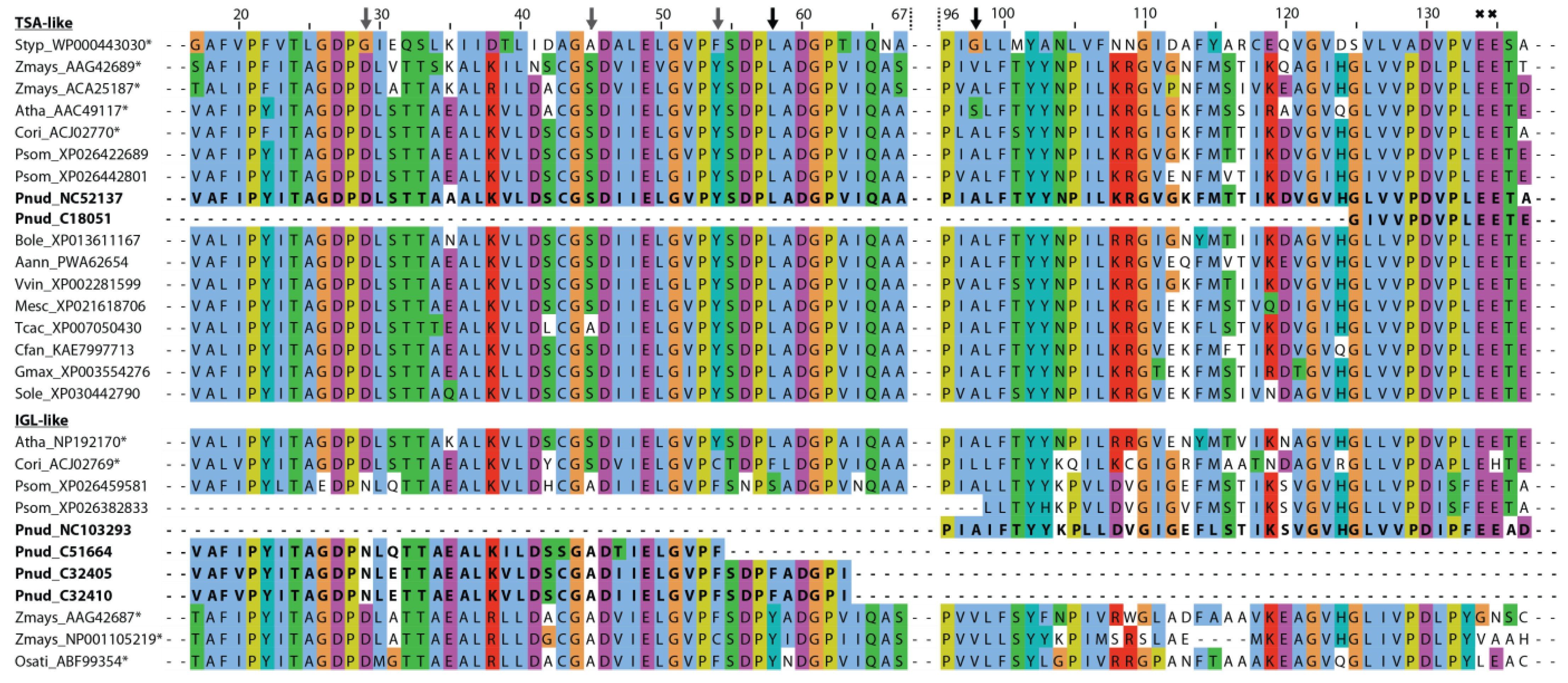
2.2. Transcriptome, Proteome, and Metabolome Analysis of the Nudicaulin Biosynthesis
2.3. Biosynthesis of Free Indole
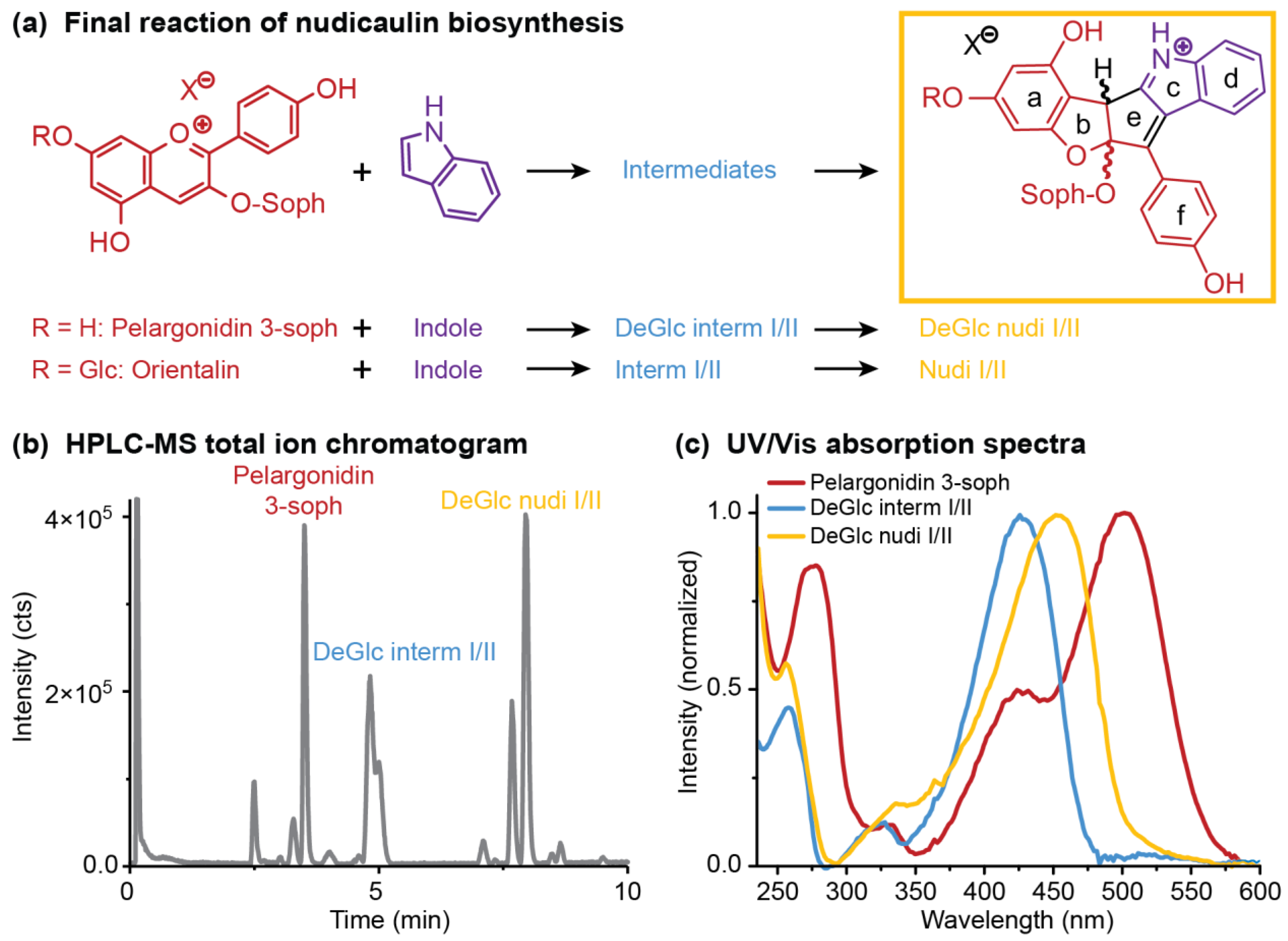
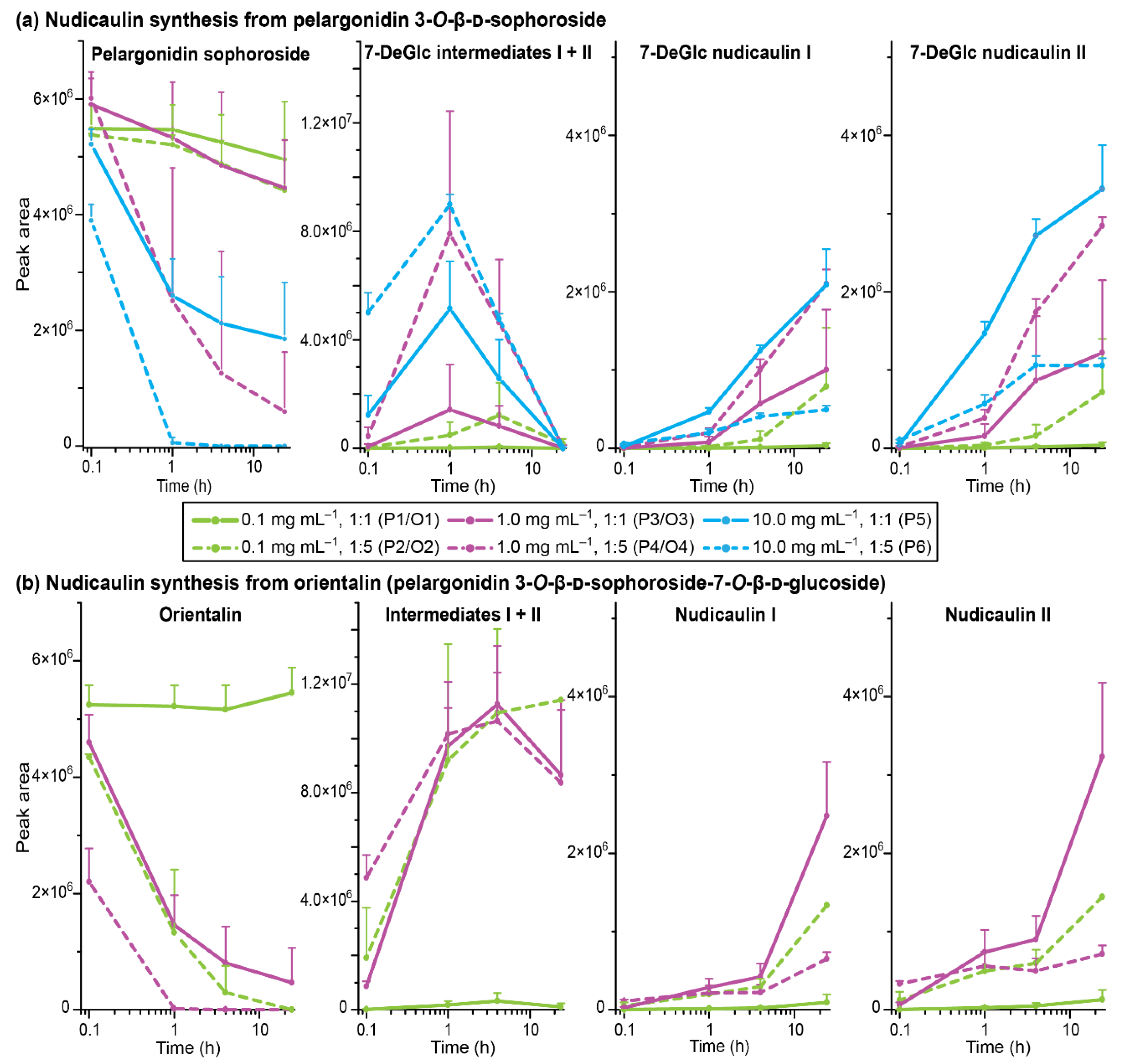
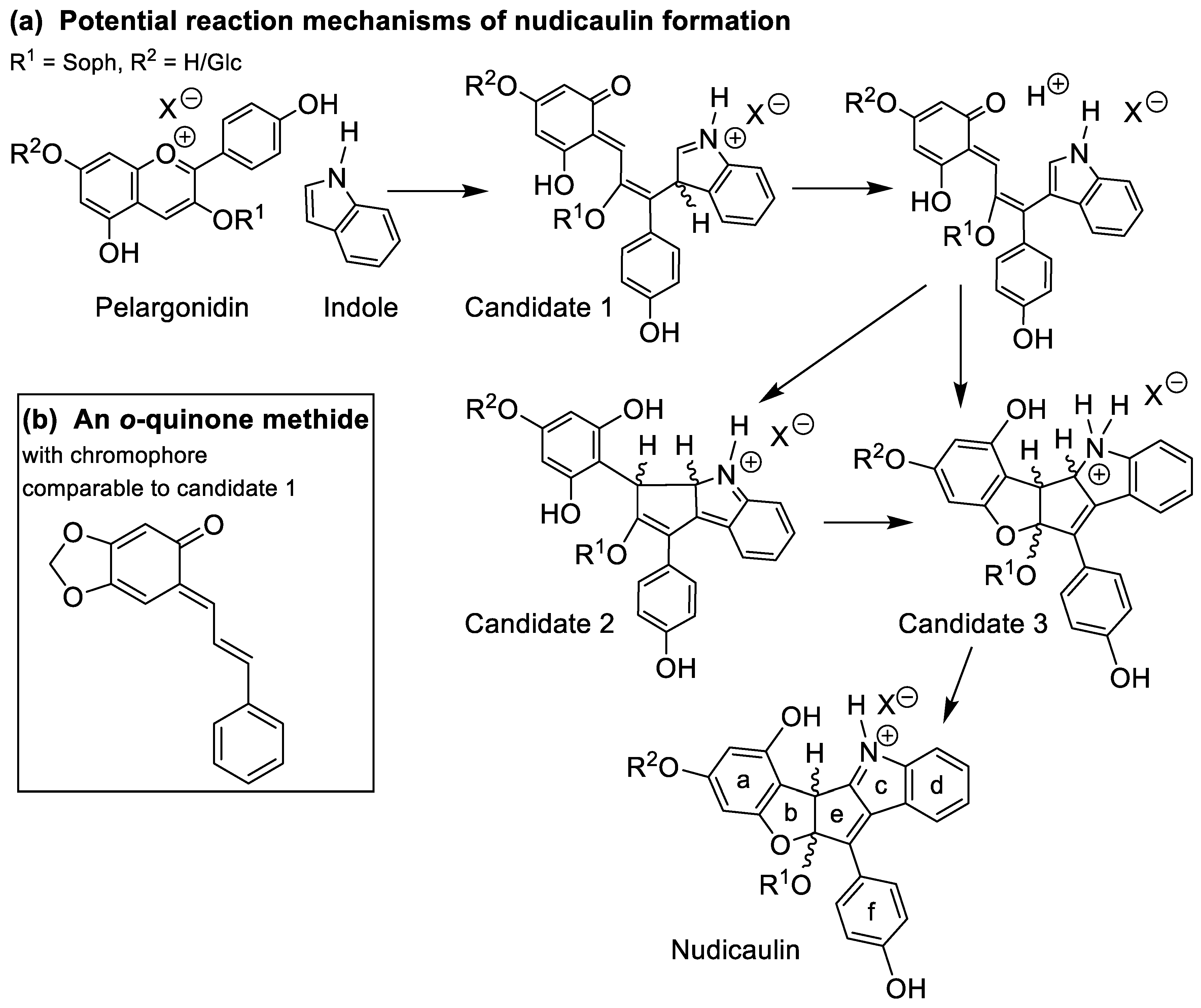
2.4. Concentration Dependency and Effect of the Glycosylation Pattern on the Final Step of Nudicaulin Biosynthesis
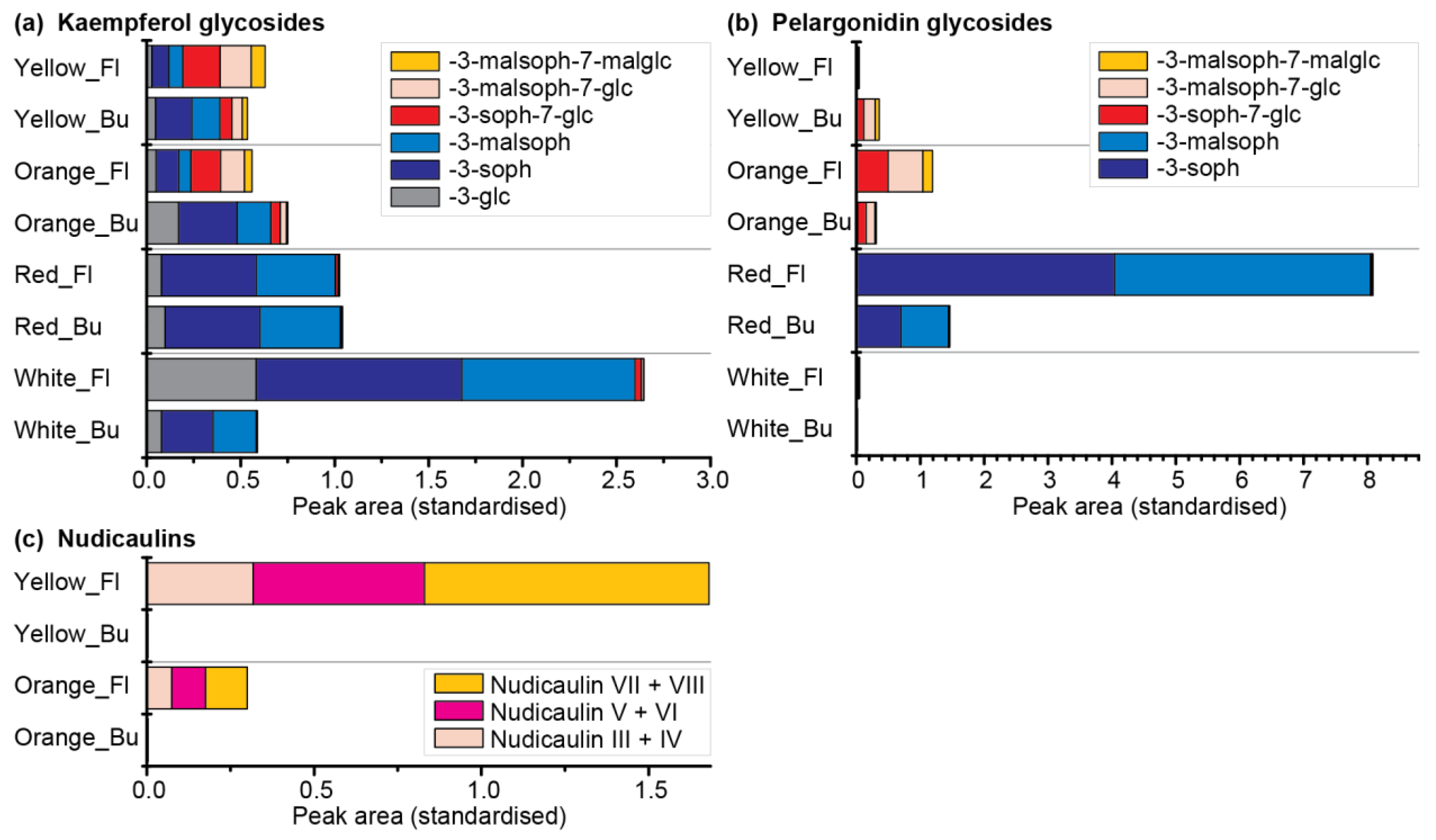
2.5. Comparative Metabolome and Transcriptome Analysis of Differently Coloured P. nudicaule Cultivars
3. Materials and Methods
3.1. Plant Material
3.2. Definition of Developmental Stages of Yellow P. nudicaule Petals
3.3. Transcriptomics
3.3.1. RNA-Seq and Differential Gene Expression Analysis
3.3.2. Phylogenetic Analysis of Plant TPS/IGL-like Protein Sequences
3.3.3. Data Accessibility
3.4. Proteomics
3.4.1. Protein Extraction and 2D Differential Gel Electrophoresis (2D-DIGE)
3.4.2. Protein Digestion and HPLC-MS Analysis
3.4.3. Data Processing and Protein Identification
3.5. Metabolomics
3.5.1. Sample Preparation and Metabolomics Analysis of Five Developmental Stages of Yellow P. nudicaule Flowers
3.5.2. Sample Preparation and Metabolomics Analysis of Developmental Stage 3 and Flower of Four P. nudicaule Cultivars
3.5.3. Data Analysis and Candidate Metabolite Identification
- Pnudicaule_YFP_DV1-5_Bud_neg_xcms-GNPS https://gnps.ucsd.edu/ProteoSAFe/status.jsp?task=c3f0d47205f449b6918b323816539307
- Pnudicaule_YFP_DV1-5_Bud_pos_xcms-GNPS https://gnps.ucsd.edu/ProteoSAFe/status.jsp?task=5489ef2fdc4743d093e25cb80700c39f
- Pnudicaule_DV3_Flower-Bud_pos_xcms-GNPS https://gnps.ucsd.edu/ProteoSAFe/status.jsp?task=28eee1f5df1547dbad02cc1693741b07
- Pnudicaule_DV3_Flower-Bud_neg_xcms-GNPS https://gnps.ucsd.edu/ProteoSAFe/status.jsp?task=fc1f75e783be4946827c83bd90f9e528
3.6. Isolation and Identification of Pelargonidin Glycosides
3.7. Concentration Dependency of the Final Step of Nudicaulin Biosynthesis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gronquist, M.; Bezzerides, A.; Attygalle, A.; Meinwald, J.; Eisner, M.; Eisner, T. Attractive and defensive functions of the ultraviolet pigments of a flower (Hypericum calycinum). Proc. Natl. Acad. Sci. USA 2001, 98, 13745–13750. [Google Scholar] [CrossRef] [PubMed]
- Harborne, J.B.; Williams, C.A. Advances in flavonoid research since 1992. Phytochemistry 2000, 55, 481–504. [Google Scholar] [CrossRef]
- Van der Kooi, C.J.; Elzenga, J.T.M.; Staal, M.; Stavenga, D.G. How to colour a flower: On the optical principles of flower coloration. Proc. R. Soc. B 2016, 283, 20160429:1–20160429:9. [Google Scholar] [CrossRef]
- Van der Kooi, C.J.; Dyer, A.G.; Kevan, P.G.; Lunau, K. Functional significance of the optical properties of flowers for visual signalling. Ann. Bot. 2019, 123, 263–276. [Google Scholar] [CrossRef]
- Kugler, H. UV-Musterungen auf Blüten und ihr Zustandekommen. Planta 1963, 59, 296–329. [Google Scholar] [CrossRef]
- Papiorek, S.; Junker, R.R.; Lunau, K. Gloss, colour and grip: Multifunctional epidermal cell shapes in bee and bird-pollinated flowers. PLoS ONE 2014, 9, e112013:1–e112013:7. [Google Scholar] [CrossRef]
- Whitney, H.M.; Kolle, M.; Andrew, P.; Chittka, L.; Steiner, U.; Glover, B.J. Floral iridescence, produced by diffractive optics, acts as a cue for animal pollinators. Science 2009, 323, 130–133. [Google Scholar] [CrossRef] [PubMed]
- Ben-Tal, Y.; King, R.W. Environmental factors involved in colouration of flowers of Kangaroo Paw. Sci. Hortic. 1997, 72, 35–48. [Google Scholar] [CrossRef]
- Albert, A.; Sareedenchai, V.; Heller, W.; Seidlitz, H.K.; Zidorn, C. Temperature is the key to altitudinal variation of phenolics in Arnica montana L. cv. ARBO. Oecologia 2009, 160, 1–8. [Google Scholar] [CrossRef]
- Chalker-Scott, L. Environmental significance of anthocyanins in plant stress responses. Photochem. Photobiol. 1999, 70, 1–9. [Google Scholar] [CrossRef]
- Llorens, L.; Badenes-Pérez, F.R.; Julkunen-Tiitto, R.; Zidorn, C.; Fereres, A.; Jansen, M.A. The role of UV-B radiation in plant sexual reproduction. Perspect. Plant. Ecol. Evol. Syst. 2015, 17, 243–254. [Google Scholar] [CrossRef]
- Mori, M.; Yoshida, K.; Ishigaki, Y.; Matsunaga, T.; Nikaido, O.; Kameda, K.; Kondo, T. UV-B protective effect of a polyacylated anthocyanin, HBA, in flower petals of the blue morning glory, Ipomoea tricolor. cv. Heavenly Blue. Bioorg. Med. Chem. 2005, 13, 2015–2020. [Google Scholar] [CrossRef] [PubMed]
- Coberly, L.C.; Rausher, M.D. Analysis of a chalcone synthase mutant in Ipomoea purpurea reveals a novel function for flavonoids: Amelioration of heat stress. Mol. Ecol. 2003, 12, 1113–1124. [Google Scholar] [CrossRef]
- Shvarts, M.; Borochov, A.; Weiss, D. Low temperature enhances petunia flower pigmentation and induces chalcone synthase gene expression. Physiol. Plant. 1997, 99, 67–72. [Google Scholar] [CrossRef]
- Grotewold, E. The genetics and biochemistry of floral pigments. Annu. Rev. Plant. Biol. 2006, 57, 761–780. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, W.J.C.; Price, J.R. The genetics and chemistry of flower colour variation. Biol. Rev. 1940, 15, 35–57. [Google Scholar] [CrossRef]
- Weevers, T. Flower colours and their frequency. Acta Bot. Neerl. 1952, 1, 81–92. [Google Scholar] [CrossRef]
- Lunau, K.; Maier, E.J. Innate colour preferences of flower visitors. Z. Vergl. Physiol. 1995, 177, 1–19. [Google Scholar] [CrossRef]
- Lunau, K. Visual ecology of flies with particular reference to colour vision and colour preferences. J. Comp. Physiol. A. 2014, 200, 497–512. [Google Scholar] [CrossRef]
- Goulson, D.; Cruise, J.L.; Sparrow, K.R.; Harris, A.J.; Park, K.J.; Tinsley, M.C.; Gilburn, A.S. Choosing rewarding flowers; perceptual limitations and innate preferences influence decision making in bumblebees and honeybees. Behav. Ecol. Sociobiol. 2007, 61, 1523–1529. [Google Scholar] [CrossRef]
- Iwashina, T. Contribution to flower colors of flavonoids including anthocyanins: A review. Nat. Prod. Commun. 2015, 10, 529–544. [Google Scholar] [CrossRef]
- Guldberg, L.D.; Atsatt, P.R. Frequency of reflection and absorption of ultraviolet light in flowering plants. Am. Midl. Nat. 1975, 93, 35–43. [Google Scholar] [CrossRef]
- Kevan, P.G.; Backhaus, W.G.K. Color vision: Ecology and evolution in making the best of the photic environment. In Color Vision; Backhaus, W.G.K., Kliegl, R., Werner, J.S., Eds.; De Gruyter: Berlin, Germany; De Gruyter: New York, NY, USA, 1998; pp. 163–184. [Google Scholar]
- Chittka, L.; Shmida, A.; Troje, N.; Menzel, R. Ultraviolet as a component of flower reflections, and the colour perception of Hymenoptera. Vis. Res. 1994, 34, 1489–1508. [Google Scholar] [CrossRef]
- Harborne, J.B.; Nash, R.J. Flavonoid pigments responsible for ultraviolet patterning in petals of the genus Potentilla. Biochem. Syst. Ecol. 1984, 12, 315–318. [Google Scholar] [CrossRef]
- Thompson, W.R.; Meinwald, J.; Aneshansley, D.; Eisner, T. Flavonols: Pigments responsible for ultraviolet absorption in nectar guide of flower. Science 1972, 177, 528–530. [Google Scholar] [CrossRef]
- Warskulat, A.-C.; Tatsis, E.C.; Dudek, B.; Kai, M.; Lorenz, S.; Schneider, B. Unprecedented utilization of pelargonidin and indole for the biosynthesis of plant indole alkaloids. ChemBioChem 2016, 17, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Dudek, B.; Warskulat, A.-C.; Schneider, B. The occurrence of flavonoids and related compounds in flower sections of Papaver nudicaule. Plants 2016, 5, 28. [Google Scholar] [CrossRef] [PubMed]
- Price, J.R.; Robinson, R.; Scott-Moncrieff, R. The yellow pigment of Papaver nudicaule. Part, I. J. Chem. Soc. 1939, 1465–1468. [Google Scholar] [CrossRef]
- Devlin, R.; Sperry, J. The curious yellow colouring matter of the Iceland poppy. Org. Biomol. Chem. 2020, 18, 5278–5286. [Google Scholar] [CrossRef]
- Tatsis, E.C.; Böhm, H.; Schneider, B. Occurrence of nudicaulin structural variants in flowers of papaveraceous species. Phytochemistry 2013, 92, 105–112. [Google Scholar] [CrossRef]
- Fabergé, A.C. Genetics of the Scapiflora section of Papaver. J. Genet. 1942, 44, 169–193. [Google Scholar] [CrossRef]
- Popov, M. Family Papaveraceae. In Flora of the U.S.S.R.: Volume VII. Ranales and Rhoeadales; Komarov, V.L., Ed.; Botanical Institute of the Academy of Sciences of the U.S.S.R.: Saint Petersburg, Russia, 1937; translated by Blake, Z., Israel Program for Scientific Translations Ltd.: Jerusalem, Israel; Keter Press: Jerusalem, Israel, 1970; pp. 437–549. [Google Scholar]
- Tolmatchew, A. Papaver nudicaule L. und einige verwandte asiatische Papaver-Formen. Sven. Bot. Tidskr. 1930, 24, 33–43. [Google Scholar]
- Tatsis, E.C.; Schaumlöffel, A.; Warskulat, A.-C.; Massiot, G.; Schneider, B.; Bringmann, G. Nudicaulins, yellow flower pigments of Papaver nudicaule: Revised constitution and assignment of absolute configuration. Org. Lett. 2013, 15, 156–159. [Google Scholar] [CrossRef] [PubMed]
- Dudek, B.; Schnurrer, F.; Dahse, H.-M.; Paetz, C.; Warskulat, A.-C.; Weigel, C.; Voigt, K.; Schneider, B. Formation of nudicaulins in vivo and in vitro and the biomimetic synthesis and bioactivity of O-methylated nudicaulin derivatives. Molecules 2018, 23, 3357. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, J.; Shao, X.; Li, J. Indole-3-glycerol phosphate, a branchpoint of indole-3-acetic acid biosynthesis from the tryptophan biosynthetic pathway in Arabidopsis thaliana. Plant. J. 2000, 24, 327–334. [Google Scholar] [CrossRef]
- Gierl, A.; Frey, M. Evolution of benzoxazinone biosynthesis and indole production in maize. Planta 2001, 213, 493–498. [Google Scholar] [CrossRef]
- Zhuang, X.; Fiesselmann, A.; Zhao, N.; Chen, H.; Frey, M.; Chen, F. Biosynthesis and emission of insect herbivory-induced volatile indole in rice. Phytochemistry 2012, 73, 15–22. [Google Scholar] [CrossRef]
- Schullehner, K.; Dick, R.; Vitzthum, F.; Schwab, W.; Brandt, W.; Frey, M.; Gierl, A. Benzoxazinoid biosynthesis in dicot plants. Phytochemistry 2008, 69, 2668–2677. [Google Scholar] [CrossRef]
- Tohge, T.; de Souza, L.P.; Fernie, A.R. Current understanding of the pathways of flavonoid biosynthesis in model and crop plants. J. Exp. Bot. 2017, 68, 4013–4028. [Google Scholar] [CrossRef]
- Saito, K.; Yonekura-Sakakibara, K.; Nakabayashi, R.; Higashi, Y.; Yamazaki, M.; Tohge, T.; Fernie, A.R. The flavonoid biosynthetic pathway in Arabidopsis: Structural and genetic diversity. Plant. Physiol. Biochem. 2013, 72, 21–34. [Google Scholar] [CrossRef]
- Schröder, J. A family of plant-specific polyketide synthases: Facts and predictions. Trends Plant. Sci. 1997, 2, 373–378. [Google Scholar] [CrossRef]
- Tatsis, E.C.; Eylert, E.; Maddula, R.K.; Ostrozhenkova, E.; Svatoš, A.; Eisenreich, W.; Schneider, B. Biosynthesis of nudicaulins: A 13CO2-pulse/chase labeling study with Papaver nudicaule. ChemBioChem 2014, 15, 1645–1650. [Google Scholar] [CrossRef] [PubMed]
- Devlin, R.; Sperry, J. Biomimetic synthesis of nudicaulins I and II, yellow pigments from the Iceland poppy. Papaver nudicaule. Chem. Commun. 2019, 55, 13594–13597. [Google Scholar] [CrossRef] [PubMed]
- Frey, M.; Chomet, P.; Glawischnig, E.; Stettner, C.; Grün, S.; Winklmair, A.; Eisenreich, W.; Bacher, A.; Meeley, R.B.; Briggs, S.P.; et al. Analysis of a chemical plant defense mechanism in grasses. Science 1997, 277, 696–699. [Google Scholar] [CrossRef]
- Zhang, R.; Wang, B.; Ouyang, J.; Li, J.; Wang, Y. Arabidopsis indole synthase, a homolog of tryptophan synthase alpha, is an enzyme involved in the Trp-independent indole-containing metabolite biosynthesis. J. Integr. Plant. Biol. 2008, 50, 1070–1077. [Google Scholar] [CrossRef]
- Martínez-Harms, J.; Warskulat, A.-C.; Dudek, B.; Kunert, G.; Lorenz, S.; Hansson, B.S.; Schneider, B. Biosynthetic and functional color-scent associations in flowers of Papaver nudicaule and its impact on pollinators. ChemBioChem 2018, 19, 1553–1562. [Google Scholar] [CrossRef]
- Frey, M.; Stettner, C.; Pare, P.W.; Schmelz, E.A.; Tumlinson, J.H.; Gierl, A. An herbivore elicitor activates the gene for indole emission in maize. Proc. Natl. Acad. Sci. USA 2000, 97, 14801–14806. [Google Scholar] [CrossRef]
- Cornuz, G.; Wyler, H.; Lauterwein, J. Pelargonidin 3-malonylsophoroside from the red Iceland poppy, Papaver nudicaule. Phytochemistry 1981, 20, 1461–1462. [Google Scholar] [CrossRef]
- Toki, K.; Saito, N.; Morita, Y. An acylated pelargonidin 3-sophoroside from the pale-brownish red flowers of Ipomoea nil. Heterocycles 2004, 63, 1449–1454. [Google Scholar] [CrossRef]
- Dudek, B. Untersuchungen zu Metabolitenprofil und Farbentstehung in gelben Papaver nudicaule-Blüten. Master’s Thesis, Friedrich-Schiller-Universität Jena, Jena, Germany, 1 June 2015. [Google Scholar]
- Jurd, L. Quinones and quinone-methides—I: Cyclization and dimerisation of crystalline ortho-quinone methides from phenol oxidation reactions. Tetrahedron 1977, 33, 163–168. [Google Scholar] [CrossRef]
- Schliemann, W.; Schneider, B.; Wray, V.; Schmidt, J.; Nimtz, M.; Porzel, A.; Böhm, H. Flavonols and an indole alkaloid skeleton bearing identical acylated glycosidic groups from yellow petals of Papaver nudicaule. Phytochemistry 2006, 67, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Virtanen, P.; Gommers, R.; Oliphant, T.E.; Haberland, M.; Reddy, T.; Cournapeau, D.; Burovski, E.; Peterson, P.; Weckesser, W.; Bright, J.; et al. SciPy 1.0-Fundamental algorithms for scientific computing in python. Nat. Methods 2020, 17, 261–272. [Google Scholar] [CrossRef]
- Hunter, J.D. Matplotlib: A 2D graphics environment. Comput. Sci. Eng. 2007, 9, 90–95. [Google Scholar] [CrossRef]
- Vogel, H.; Badapanda, C.; Knorr, E.; Vilcinskas, A. RNA-sequencing analysis reveals abundant developmental stage-specific and immunity-related genes in the pollen beetle Meligethes aeneus. Insect. Mol. Biol. 2014, 23, 98–112. [Google Scholar] [CrossRef] [PubMed]
- Götz, S.; García-Gómez, J.M.; Terol, J.; Williams, T.D.; Nagaraj, S.H.; Nueda, M.J.; Robles, M.; Talón, M.; Dopazo, J.; Conesa, A. High-throughput functional annotation and data mining with the Blast2GO suite. Nucleic. Acids. Res. 2008, 36, 3420–3435. [Google Scholar] [CrossRef]
- Waterhouse, R.M.; Seppey, M.; Simão, F.A.; Manni, M.; Ioannidis, P.; Klioutchnikov, G.; Kriventseva, E.V.; Zdobnov, E.M. BUSCO applications from quality assessments to gene prediction and phylogenomics. Mol. Biol. Evol. 2017, 35, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, C.G.C.; Steiger, S.; Heckel, D.G.; Wielsch, N.; Vilcinskas, A.; Vogel, H. Sex, offspring and carcass determine antimicrobial peptide expression in the burying beetle. Sci. Rep. 2016, 6, 25409:1–25409:8. [Google Scholar] [CrossRef]
- Trifinopoulos, J.; Nguyen, L.-T.; von Haeseler, A.; Minh, B.Q. W-IQ-TREE: A fast online phylogenetic tool for maximum likelihood analysis. Nucleic. Acids. Res. 2016, 44, W232–W235. [Google Scholar] [CrossRef]
- Faurobert, M.; Pelpoir, E.; Chaïb, J. Phenol extraction of proteins for proteomic studies of recalcitrant plant tissues. Methods. Mol. Biol. 2007, 355, 9–14. [Google Scholar] [CrossRef]
- Wartenberg, D.; Vödisch, M.; Kniemeyer, O.; Albrecht-Eckardt, D.; Scherlach, K.; Winkler, R.; Weide, M.; Brakhage, A.A. Proteome analysis of the farnesol-induced stress response in Aspergillus nidulans-The role of a putative dehydrin. J. Proteom. 2012, 75, 4038–4049. [Google Scholar] [CrossRef]
- Shevchenko, A.; Tomas, H.; Havlis, J.; Olsen, J.V.; Mann, M. In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nat. Protoc. 2006, 1, 2856–2860. [Google Scholar] [CrossRef] [PubMed]
- Shevchenko, A.; Sunyaev, S.; Loboda, A.; Bork, P.; Ens, W.; Standing, K.G. Charting the proteomes of organisms with unsequenced genomes by MALDI-quadrupole time-of-flight mass spectrometry and BLAST homology searching. Anal. Chem. 2001, 73, 1917–1926. [Google Scholar] [CrossRef]
- Wang, M.; Carver, J.J.; Phelan, V.V.; Sanchez, L.M.; Garg, N.; Peng, Y.; Nguyen, D.D.; Watrous, J.; Kapono, C.A.; Luzzatto-Knaan, T.; et al. Sharing and community curation of mass spectrometry data with Global Natural Products Social Molecular Networking. Nat. Biotechnol. 2016, 34, 828–837. [Google Scholar] [CrossRef] [PubMed]
- Kearns, C.A. Anthophilous fly distribution across an elevation gradient. Am. Midl. Nat. 1992, 127, 172–182. [Google Scholar] [CrossRef]
- Mølgaard, P. Temperature relations of yellow and white flowered Papaver radicatum in North Greenland. Arct. Alp. Res. 1989, 21, 83–90. [Google Scholar] [CrossRef]
- Dötterl, S.; Vereecken, N.J. The chemical ecology and evolution of bee–flower interactions: A review and perspectives. Can. J. Zool. 2010, 88, 668–697. [Google Scholar] [CrossRef]
- Knudsen, J.T.; Eriksson, R.; Gershenzon, J.; Ståhl, B. Diversity and distribution of floral scent. Bot. Rev. 2006, 72, 1–120. [Google Scholar] [CrossRef]
- Cna’ani, A.; Seifan, M.; Tzin, V. Indole is an essential molecule for plant interactions with herbivores and pollinators. J. Plant. Biol. Crop. Res. 2018, 1, 1003:1–1003:5. [Google Scholar] [CrossRef]
- Bischoff, M.; Jürgens, A.; Campbell, D.R. Floral scent in natural hybrids of Ipomopsis (Polemoniaceae) and their parental species. Ann. Bot. 2014, 113, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Raguso, R.A.; Light, D.M.; Pickersky, E. Electroantennogram responses of Hyles lineata (Sphingidae: Lepidoptera) to volatile compounds from Clarkia breweri (Onagraceae) and other moth-pollinated flowers. J. Chem. Ecol. 1996, 22, 1735–1766. [Google Scholar] [CrossRef]
- Zito, P.; Dötterl, S.; Sajeva, M. Floral volatiles in a sapromyiophilous plant and their importance in attracting house fly pollinators. J. Chem. Ecol. 2015, 41, 340–349. [Google Scholar] [CrossRef] [PubMed]
- Dötterl, S.; Füssel, U.; Jürgens, A.; Aas, G. 1,4-Dimethoxybenzene, a floral scent compound in willows that attracts an oligolectic bee. J. Chem. Ecol. 2005, 31, 2993–2998. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dudek, B.; Warskulat, A.-C.; Vogel, H.; Wielsch, N.; Menezes, R.C.; Hupfer, Y.; Paetz, C.; Gebauer-Jung, S.; Svatoš, A.; Schneider, B. An Integrated—Omics/Chemistry Approach Unravels Enzymatic and Spontaneous Steps to Form Flavoalkaloidal Nudicaulin Pigments in Flowers of Papaver nudicaule L. Int. J. Mol. Sci. 2021, 22, 4129. https://doi.org/10.3390/ijms22084129
Dudek B, Warskulat A-C, Vogel H, Wielsch N, Menezes RC, Hupfer Y, Paetz C, Gebauer-Jung S, Svatoš A, Schneider B. An Integrated—Omics/Chemistry Approach Unravels Enzymatic and Spontaneous Steps to Form Flavoalkaloidal Nudicaulin Pigments in Flowers of Papaver nudicaule L. International Journal of Molecular Sciences. 2021; 22(8):4129. https://doi.org/10.3390/ijms22084129
Chicago/Turabian StyleDudek, Bettina, Anne-Christin Warskulat, Heiko Vogel, Natalie Wielsch, Riya Christina Menezes, Yvonne Hupfer, Christian Paetz, Steffi Gebauer-Jung, Aleš Svatoš, and Bernd Schneider. 2021. "An Integrated—Omics/Chemistry Approach Unravels Enzymatic and Spontaneous Steps to Form Flavoalkaloidal Nudicaulin Pigments in Flowers of Papaver nudicaule L." International Journal of Molecular Sciences 22, no. 8: 4129. https://doi.org/10.3390/ijms22084129
APA StyleDudek, B., Warskulat, A.-C., Vogel, H., Wielsch, N., Menezes, R. C., Hupfer, Y., Paetz, C., Gebauer-Jung, S., Svatoš, A., & Schneider, B. (2021). An Integrated—Omics/Chemistry Approach Unravels Enzymatic and Spontaneous Steps to Form Flavoalkaloidal Nudicaulin Pigments in Flowers of Papaver nudicaule L. International Journal of Molecular Sciences, 22(8), 4129. https://doi.org/10.3390/ijms22084129





