Apoptosis Disorder, a Key Pathogenesis of HCMV-Related Diseases
Abstract
:1. Introduction
2. The Prevalence and Perniciousness of HCMV
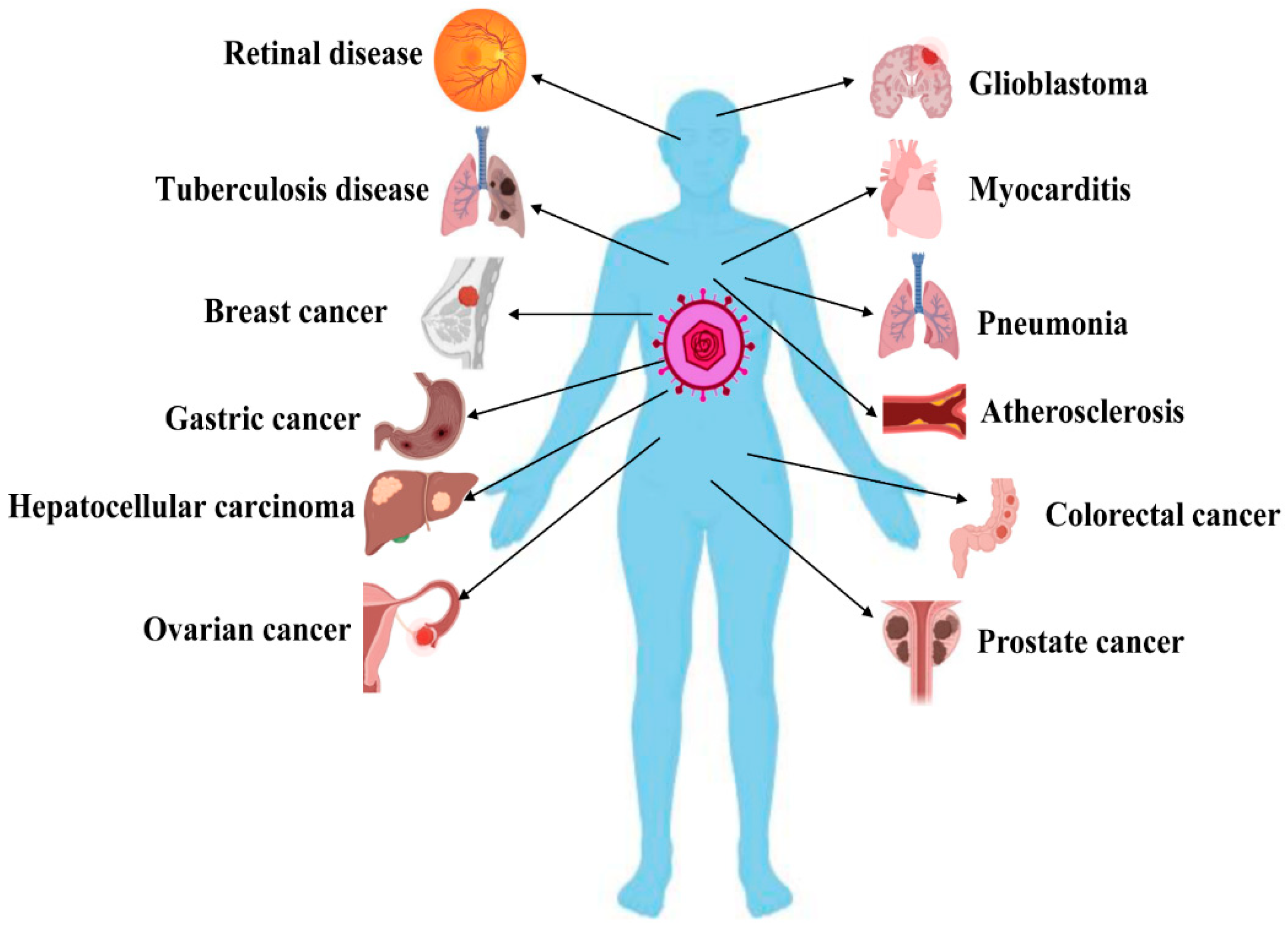
3. HCMV Causes Numerous Diseases by Inducing Apoptosis Disorder
3.1. Immune System Diseases
3.1.1. Systemic Lupus Erythematosus
3.1.2. Systemic Sclerosis
3.2. Pneumonia
3.3. Atherosclerosis
3.4. Cancers
3.4.1. Glioblastoma
3.4.2. Neuroblastoma
3.4.3. Breast Cancer
3.4.4. Acute Myeloid Leukemia
3.4.5. Hepatocellular Carcinoma
3.4.6. Gastric Cancer
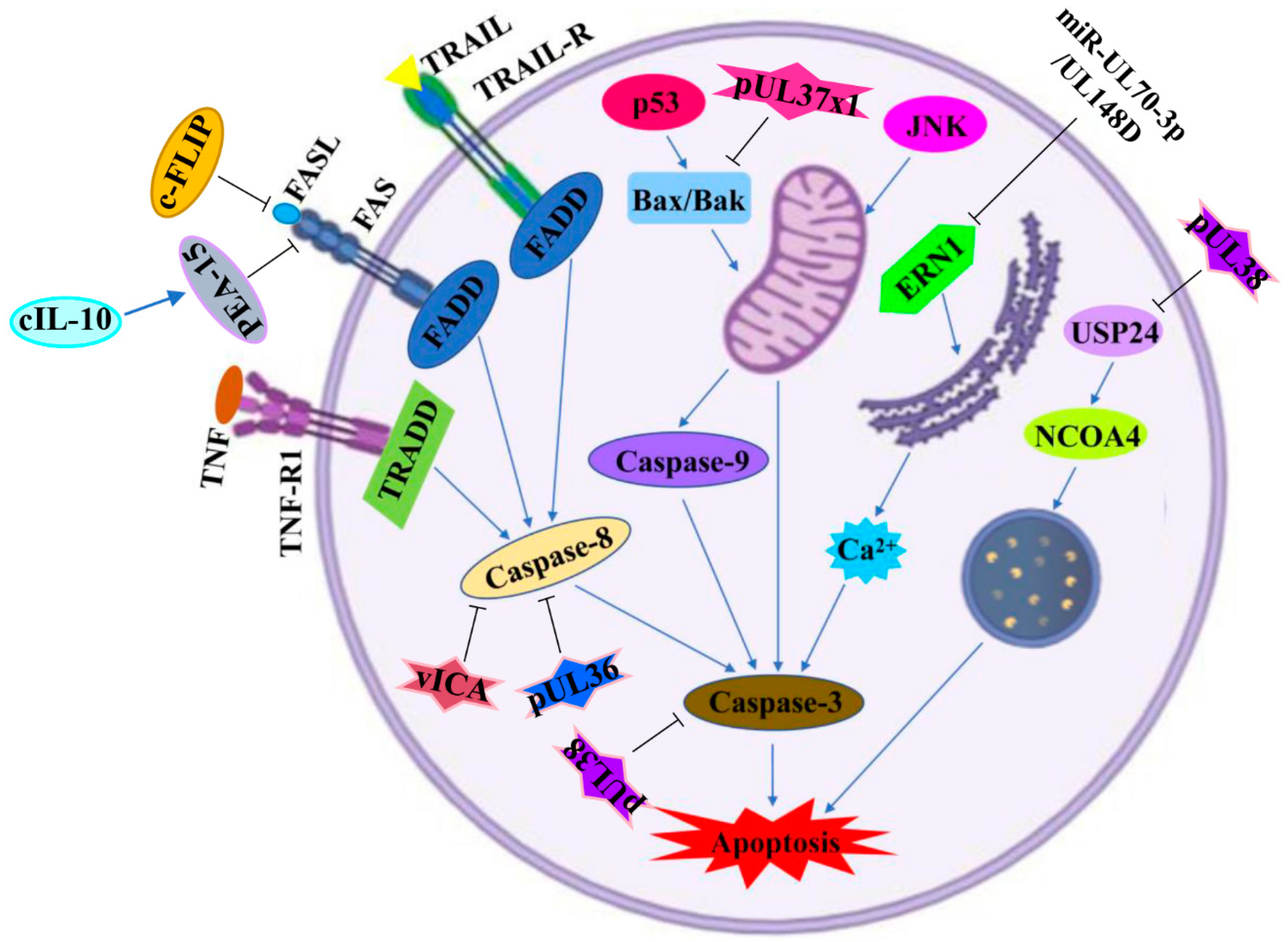
4. Molecular Mechanism of Apoptosis Mediated by HCMV Infection
4.1. Extrinsic Pathway
4.2. Intrinsic Pathway
4.2.1. Mitochondrial Pathway
4.2.2. Endoplasmic Reticulum Pathway
4.2.3. Lysosome Pathway
5. Novel Treatment Strategies for HCMV-Related Diseases
6. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HCMV | human cytomegalovirus |
| MCMV | murine CMV |
| AID | autoimmune disease |
| SLE | systemic lupus erythematosus |
| SSc | systemic sclerosis |
| AIP | acute interstitial pneumonia |
| IE2 | immediately-early 2 |
| STAT3 | signal transducer and activator of transcription 3 |
| hnRNP A2/B1 | heterogeneous ribonucleoprotein A2/B1 |
| c-FLIP | cellular FLIP |
| FAS | Fas cell surface death receptor |
| FASL | FAS ligand |
| FADD | FAS-associated death domain |
| TRAIL | TNF-related apoptosis-inducing ligand |
| TRAIL-R | TNF-related apoptosis-inducing ligand receptor |
| TNF | tumor necrosis factor |
| TNF-R1 | tumor necrosis factor receptor 1 |
| TRADD | TNF receptor-1-associated death domain |
| PEA-15 | phosphoprotein enriched in astrocytes-15 |
| cIL-10 | cellular interleukin 10 |
| vICA | viral inhibitor of caspase-8 activation |
| Bax | BCL2-associated X protein 2 |
| Bak | BCL2 homologous antagonist killer |
| JNK | c-Jun N-terminal kinase |
| ERN1 | reticulum-to-nucleus signaling 1 |
| USP24 | ubiquitin-specific protease 24 |
| NCOA4 | nuclear receptor coactivator 4 |
| JAK | Janus kinase |
| MAPK/ERK | mitogen-activated protein kinase/extracellular-regulated protein kinase |
| Bag-1 | BCL2-associated athanogene 1 |
| PARP | poly ADP-ribose polymerase |
| TGF | transforming growth factor |
References
- Clement, M.; Humphreys, I.R. Cytokine-Mediated Induction and Regulation of Tissue Damage During Cytomegalovirus Infection. Front Immunol. 2019, 10, 78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Geisler, J.; Touma, J.; Rahbar, A.; Söderberg-Nauclér, C.; Vetvik, K. A Review of the Potential Role of Human Cytomegalovirus (HCMV) Infections in Breast Cancer Carcinogenesis and Abnormal Immunity. Cancers 2019, 11, 1842. [Google Scholar] [CrossRef] [Green Version]
- Babu, S.G.; Pandeya, A.; Verma, N.; Shukla, N.; Kumar, R.V.; Saxena, S. Role of HCMV miR-UL70-3p and miR-UL148D in overcoming the cellular apoptosis. Mol. Cell. Biochem. 2014, 393, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Ma, Q.; Hu, M.; Qian, D.; Wang, B.; He, N. The Inhibition of miR-144-3p on Cell Proliferation and Metastasis by Targeting TOP2A in HCMV-Positive Glioblastoma Cells. Molecules 2018, 23, 3259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Joseph, G.P.; McDermott, R.; Baryshnikova, M.A.; Cobbs, C.S.; Ulasov, I.V. Cytomegalovirus as an oncomodulatory agent in the progression of glioma. Cancer Lett. 2017, 384, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Landolfo, S.; Gariglio, M.; Gribaudo, G.; Lembo, D. The human cytomegalovirus. Pharmacol. Ther. 2003, 98, 269–297. [Google Scholar] [CrossRef]
- Liao, X.H.; Dong, X.; Wu, C.; Wang, T.; Liu, F.; Zhou, J.; Zhang, T.C. Human cytomegalovirus immediate early protein 2 enhances myocardin-mediated survival of rat aortic smooth muscle cells. Virus Res. 2014, 192, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, A.; Forman, M.; Arav-Boger, R. Activation of nucleotide oligomerization domain 2 (NOD2) by human cytomegalovirus initiates innate immune responses and restricts virus replication. PLoS ONE 2014, 9, e92704. [Google Scholar] [CrossRef] [PubMed]
- Lepiller, Q.; Abbas, W.; Kumar, A.; Tripathy, M.K.; Herbein, G. HCMV activates the IL-6-JAK-STAT3 axis in HepG2 cells and primary human hepatocytes. PLoS ONE 2013, 8, e59591. [Google Scholar] [CrossRef]
- de Jong, M.D.; Galasso, G.J.; Gazzard, B.; Griffiths, P.D.; Jabs, D.A.; Kern, E.R.; Spector, S.A. Summary of the II International Symposium on Cytomegalovirus. Antivir. Res. 1998, 39, 141–162. [Google Scholar] [CrossRef]
- Lu, T.; Aron, L.; Zullo, J.; Pan, Y.; Kim, H.; Chen, Y.; Yang, T.H.; Kim, H.M.; Drake, D.; Liu, X.S.; et al. REST and stress resistance in ageing and Alzheimer’s disease. Nature 2014, 507, 448–454. [Google Scholar] [CrossRef] [Green Version]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Letai, A.; Sarosiek, K. Regulation of apoptosis in health and disease: The balancing act of BCL-2 family proteins. Nat. Rev. Mol. Cell Biol. 2019, 20, 175–193. [Google Scholar] [CrossRef] [PubMed]
- Galluzzi, L.; Vitale, I.; Aaronson, S.A.; Abrams, J.M.; Adam, D.; Agostinis, P.; Alnemri, E.S.; Altucci, L.; Amelio, I.; Andrews, D.W.; et al. Molecular mechanisms of cell death: Recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 2018, 25, 486–541. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Nicholson, D.W. Cross-talk in cell death signaling. J. Exp. Med. 2000, 192, 21–26. [Google Scholar] [CrossRef] [Green Version]
- Salaun, B.; Romero, P.; Lebecque, S. Toll-like receptors’ two-edged sword: When immunity meets apoptosis. Eur. J. Immunol. 2007, 37, 3311–3318. [Google Scholar] [CrossRef] [PubMed]
- Chaudhry, M.Z.; Kasmapour, B.; Plaza-Sirvent, C.; Bajagic, M.; Casalegno Garduño, R.; Borkner, L.; Lenac Roviš, T.; Scrima, A.; Jonjic, S.; Schmitz, I.; et al. UL36 Rescues Apoptosis Inhibition and In vivo Replication of a Chimeric MCMV Lacking the M36 Gene. Front. Cell. Infect. Microbiol. 2017, 7, 312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, M.G. Propagation in tissue cultures of a cytopathogenic virus from human salivary gland virus (SGV) disease. Proc. Soc. Exp. Biol. Med. Soc. Exp. Biol. Med. 1956, 92, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Rowe, W.P.; Hartley, J.W.; Waterman, S.; Turner, H.C.; Huebner, R.J. Cytopathogenic agent resembling human salivary gland virus recovered from tissue cultures of human adenoids. Proc. Soc. Exp. Biol. Med. Soc. Exp. Biol. Med. 1956, 92, 418–424. [Google Scholar]
- Chee, M.S.; Bankier, A.T.; Beck, S.; Bohni, R.; Brown, C.M.; Cerny, R.; Horsnell, T.; Hutchison, C.A., 3rd; Kouzarides, T.; Martignetti, J.A.; et al. Analysis of the protein-coding content of the sequence of human cytomegalovirus strain AD169. Curr. Top. Microbiol. Immunol. 1990, 154, 125–169. [Google Scholar] [CrossRef]
- Stern-Ginossar, N.; Weisburd, B.; Michalski, A.; Le, V.T.; Hein, M.Y.; Huang, S.X.; Ma, M.; Shen, B.; Qian, S.B.; Hengel, H.; et al. Decoding human cytomegalovirus. Science 2012, 338, 1088–1093. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cannon, M.J.; Schmid, D.S.; Hyde, T.B. Review of cytomegalovirus seroprevalence and demographic characteristics associated with infection. Rev. Med. Virol. 2010, 20, 202–213. [Google Scholar] [CrossRef]
- Pass, R.F. Epidemiology and transmission of cytomegalovirus. J. Infect. Dis. 1985, 152, 243–248. [Google Scholar] [CrossRef]
- Ho, M. Epidemiology of cytomegalovirus infections. Rev. Infect. Dis. 1990, 12 (Suppl. 7), S701–S710. [Google Scholar] [CrossRef]
- Griffiths, P.; Baraniak, I.; Reeves, M. The pathogenesis of human cytomegalovirus. J. Pathol. 2015, 235, 288–297. [Google Scholar] [CrossRef]
- Staras, S.A.; Dollard, S.C.; Radford, K.W.; Flanders, W.D.; Pass, R.F.; Cannon, M.J. Seroprevalence of cytomegalovirus infection in the United States, 1988-1994. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2006, 43, 1143–1151. [Google Scholar] [CrossRef] [PubMed]
- Wreghitt, T.G.; Teare, E.L.; Sule, O.; Devi, R.; Rice, P. Cytomegalovirus infection in immunocompetent patients. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2003, 37, 1603–1606. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abgueguen, P.; Delbos, V.; Ducancelle, A.; Jomaa, S.; Fanello, S.; Pichard, E. Venous thrombosis in immunocompetent patients with acute cytomegalovirus infection: A complication that may be underestimated. Clin. Microbiol. Infect. 2010, 16, 851–854. [Google Scholar] [CrossRef] [Green Version]
- Andrei, G.; De Clercq, E.; Snoeck, R. Novel inhibitors of human CMV. Curr. Opin. Investig. Drugs 2008, 9, 132–145. [Google Scholar]
- Britt, W. Manifestations of human cytomegalovirus infection: Proposed mechanisms of acute and chronic disease. Curr. Top. Microbiol. Immunol. 2008, 325, 417–470. [Google Scholar] [CrossRef] [PubMed]
- Boppana, S.B.; Fowler, K.B.; Pass, R.F.; Rivera, L.B.; Bradford, R.D.; Lakeman, F.D.; Britt, W.J. Congenital cytomegalovirus infection: Association between virus burden in infancy and hearing loss. J. Pediatrics 2005, 146, 817–823. [Google Scholar] [CrossRef] [PubMed]
- Burny, W.; Liesnard, C.; Donner, C.; Marchant, A. Epidemiology, pathogenesis and prevention of congenital cytomegalovirus infection. Expert Rev. Anti-Infect. Ther. 2004, 2, 881–894. [Google Scholar] [CrossRef] [PubMed]
- Rider, J.R.; Ollier, W.E.; Lock, R.J.; Brookes, S.T.; Pamphilon, D.H. Human cytomegalovirus infection and systemic lupus erythematosus. Clin. Exp. Rheumatol. 1997, 15, 405–409. [Google Scholar] [PubMed]
- Stockdale, L.; Nash, S.; Farmer, R.; Raynes, J.; Mallikaarjun, S.; Newton, R.; Fletcher, H.A. Cytomegalovirus Antibody Responses Associated With Increased Risk of Tuberculosis Disease in Ugandan Adults. J. Infect. Dis. 2020, 221, 1127–1134. [Google Scholar] [CrossRef] [PubMed]
- Lucchese, G.; Flöel, A.; Stahl, B. A Peptide Link Between Human Cytomegalovirus Infection, Neuronal Migration, and Psychosis. Front. Psychiatry 2020, 11, 349. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Liu, X.; Zhang, X.; Marshall, B.; Dong, Z.; Liu, Y.; Espinosa-Heidmann, D.G.; Zhang, M. Ocular cytomegalovirus latency exacerbates the development of choroidal neovascularization. J. Pathol. 2020, 251, 200–212. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, N.A.; Papadimitriou, J.M.; Shellam, G.R. Cytomegalovirus-induced pneumonitis and myocarditis in newborn mice. A model for perinatal human cytomegalovirus infection. Arch. Virol. 1990, 115, 75–88. [Google Scholar] [CrossRef]
- Marinho-Dias, J.; Sousa, H. Cytomegalovirus infection and cervical cancer: From past doubts to present questions. Acta Med. Port. 2013, 26, 154–160. [Google Scholar]
- Richardson, A.K.; Walker, L.C.; Cox, B.; Rollag, H.; Robinson, B.A.; Morrin, H.; Pearson, J.F.; Potter, J.D.; Paterson, M.; Surcel, H.M.; et al. Breast cancer and cytomegalovirus. Clin. Transl. Oncol. 2020, 22, 585–602. [Google Scholar] [CrossRef]
- Bai, B.; Wang, X.; Chen, E.; Zhu, H. Human cytomegalovirus infection and colorectal cancer risk: A meta-analysis. Oncotarget 2016, 7, 76735–76742. [Google Scholar] [CrossRef] [PubMed]
- Shanmughapriya, S.; Senthilkumar, G.; Vinodhini, K.; Das, B.C.; Vasanthi, N.; Natarajaseenivasan, K. Viral and bacterial aetiologies of epithelial ovarian cancer. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 2311–2317. [Google Scholar] [CrossRef] [PubMed]
- Samanta, M.; Harkins, L.; Klemm, K.; Britt, W.J.; Cobbs, C.S. High prevalence of human cytomegalovirus in prostatic intraepithelial neoplasia and prostatic carcinoma. J. Urol. 2003, 170, 998–1002. [Google Scholar] [CrossRef]
- Saravani, S.; Kadeh, H.; Miri-Moghaddam, E.; Zekri, A.; Sanadgol, N.; Gholami, A. Human Cytomegalovirus in Oral Squamous Cell Carcinoma in Southeast of Iran. Jundishapur J. Microbiol. 2015, 8, e21838. [Google Scholar] [CrossRef] [Green Version]
- Mehravaran, H.; Makvandi, M.; Samarbaf Zade, A.; Neisi, N.; Kiani, H.; Radmehr, H.; Shahani, T.; Hoseini, S.Z.; Ranjbari, N.; Nahid Samiei, R. Association of Human Cytomegalovirus with Hodgkin’s Disease and Non-Hodgkin’s lymphomas. Asian Pac. J. Cancer Prev. 2017, 18, 593–597. [Google Scholar] [CrossRef]
- Lisyany, N.I.; Klyuchnikova, A.A.; Belskaya, L.N.; Lisyany, A.A.; Gnedkova, I.A. Cytomegaloviruses and malignant brain tumors. Exp. Oncol. 2019, 41, 300–303. [Google Scholar] [CrossRef]
- Baryawno, N.; Rahbar, A.; Wolmer-Solberg, N.; Taher, C.; Odeberg, J.; Darabi, A.; Khan, Z.; Sveinbjörnsson, B.; FuskevÅg, O.M.; Segerström, L.; et al. Detection of human cytomegalovirus in medulloblastomas reveals a potential therapeutic target. J. Clin. Investig. 2011, 121, 4043–4055. [Google Scholar] [CrossRef] [Green Version]
- Maple, P.A.C. Cytomegalovirus and Epstein-Barr Virus Associations with Neurological Diseases and the Need for Vaccine Development. Vaccines 2020, 8, 35. [Google Scholar] [CrossRef] [Green Version]
- Dupont, L.; Reeves, M.B. Cytomegalovirus latency and reactivation: Recent insights into an age old problem. Rev. Med. Virol. 2016, 26, 75–89. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rabe, T.; Lazar, K.; Cambronero, C.; Goelz, R.; Hamprecht, K. Human Cytomegalovirus (HCMV) Reactivation in the Mammary Gland Induces a Proinflammatory Cytokine Shift in Breast Milk. Microorganisms 2020, 8, 289. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sison, S.L.; O’Brien, B.S.; Johnson, A.J.; Seminary, E.R.; Terhune, S.S.; Ebert, A.D. Human Cytomegalovirus Disruption of Calcium Signaling in Neural Progenitor Cells and Organoids. J. Virol. 2019, 93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- López-Botet, M.; Muntasell, A.; Vilches, C. The CD94/NKG2C+ NK-cell subset on the edge of innate and adaptive immunity to human cytomegalovirus infection. Semin. Immunol. 2014, 26, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Lin, K.; Huang, X.; Xie, W.; Xiang, D.; Ding, N.; Hu, C.; Shen, X.; Xue, X.; Huang, Y. Overexpression of the human cytomegalovirus UL111A is correlated with favorable survival of patients with gastric cancer and changes T-cell infiltration and suppresses carcinogenesis. J. Cancer Res. Clin. Oncol. 2020, 146, 555–568. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.; Lin, K.; Zhang, L.; Guo, G.; Sun, X.; Chen, J.; Ye, L.; Ye, S.; Mao, C.; Xu, J.; et al. The cytomegalovirus protein UL138 induces apoptosis of gastric cancer cells by binding to heat shock protein 70. Oncotarget 2016, 7, 5630–5645. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Bao, Q.; Xuan, B.; Xu, W.; Pan, D.; Li, Q.; Qian, Z. Human Cytomegalovirus Protein pUL38 Prevents Premature Cell Death by Binding to Ubiquitin-Specific Protease 24 and Regulating Iron Metabolism. J. Virol. 2018, 92. [Google Scholar] [CrossRef] [Green Version]
- Halenius, A.; Hengel, H. Human cytomegalovirus and autoimmune disease. BioMed Res. Int. 2014, 2014, 472978. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rees, F.; Doherty, M.; Grainge, M.J.; Lanyon, P.; Zhang, W. The worldwide incidence and prevalence of systemic lupus erythematosus: A systematic review of epidemiological studies. Rheumatology 2017, 56, 1945–1961. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, G.; Ye, S.; Xie, S.; Ye, L.; Lin, C.; Yang, M.; Shi, X.; Wang, F.; Li, B.; Li, M.; et al. The cytomegalovirus protein US31 induces inflammation through mono-macrophages in systemic lupus erythematosus by promoting NF-κB2 activation. Cell Death Dis. 2018, 9, 104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, J.; Zhang, H.; Chen, P.; Lin, Q.; Zhu, X.; Zhang, L.; Xue, X. Correlation between systemic lupus erythematosus and cytomegalovirus infection detected by different methods. Clin. Rheumatol. 2015, 34, 691–698. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, A.H.; Kuo, C.F.; Chou, I.J.; Tseng, W.Y.; Chen, Y.F.; Yu, K.H.; Luo, S.F. Human cytomegalovirus pp65 peptide-induced autoantibodies cross-reacts with TAF9 protein and induces lupus-like autoimmunity in BALB/c mice. Sci. Rep. 2020, 10, 9662. [Google Scholar] [CrossRef] [PubMed]
- Neo, J.Y.J.; Wee, S.Y.K.; Bonne, I.; Tay, S.H.; Raida, M.; Jovanovic, V.; Fairhurst, A.M.; Lu, J.; Hanson, B.J.; MacAry, P.A. Characterisation of a human antibody that potentially links cytomegalovirus infection with systemic lupus erythematosus. Sci. Rep. 2019, 9, 9998. [Google Scholar] [CrossRef]
- Attanasio, U.; Cuomo, A.; Pirozzi, F.; Loffredo, S.; Abete, P.; Petretta, M.; Marone, G.; Bonaduce, D.; De Paulis, A.; Rossi, F.W.; et al. Pulmonary Hypertension Phenotypes in Systemic Sclerosis: The Right Diagnosis for the Right Treatment. Int. J. Mol. Sci. 2020, 21, 4430. [Google Scholar] [CrossRef]
- Efthymiou, G.; Dardiotis, E.; Liaskos, C.; Marou, E.; Scheper, T.; Meyer, W.; Daponte, A.; Daoussis, D.; Hadjigeorgiou, G.; Bogdanos, D.P.; et al. A comprehensive analysis of antigen-specific antibody responses against human cytomegalovirus in patients with systemic sclerosis. Clin. Immunol. 2019, 207, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Marou, E.; Liaskos, C.; Efthymiou, G.; Dardiotis, E.; Daponte, A.; Scheper, T.; Meyer, W.; Hadjigeorgiou, G.; Bogdanos, D.P.; Sakkas, L.I. Increased immunoreactivity against human cytomegalovirus UL83 in systemic sclerosis. Clin. Exp. Rheumatol. 2017, 35 (Suppl. 106), 31–34. [Google Scholar]
- Namboodiri, A.M.; Rocca, K.M.; Pandey, J.P. IgG antibodies to human cytomegalovirus late protein UL94 in patients with systemic sclerosis. Autoimmunity 2004, 37, 241–244. [Google Scholar] [CrossRef] [PubMed]
- Arcangeletti, M.C.; Maccari, C.; Vescovini, R.; Volpi, R.; Giuggioli, D.; Sighinolfi, G.; De Conto, F.; Chezzi, C.; Calderaro, A.; Ferri, C. A Paradigmatic Interplay between Human Cytomegalovirus and Host Immune System: Possible Involvement of Viral Antigen-Driven CD8+ T Cell Responses in Systemic Sclerosis. Viruses 2018, 10, 508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pastano, R.; Dell’Agnola, C.; Bason, C.; Gigli, F.; Rabascio, C.; Puccetti, A.; Tinazzi, E.; Cetto, G.; Peccatori, F.; Martinelli, G.; et al. Antibodies against human cytomegalovirus late protein UL94 in the pathogenesis of scleroderma-like skin lesions in chronic graft-versus-host disease. Int. Immunol. 2012, 24, 583–591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, L.; Tang, R.Z.; Ruan, J.; Zhu, X.B.; Yang, Y. Up-regulation of THY1 attenuates interstitial pulmonary fibrosis and promotes lung fibroblast apoptosis during acute interstitial pneumonia by blockade of the WNT signaling pathway. Cell Cycle 2019, 18, 670–681. [Google Scholar] [CrossRef] [Green Version]
- Maidji, E.; Kosikova, G.; Joshi, P.; Stoddart, C.A. Impaired surfactant production by alveolar epithelial cells in a SCID-hu lung mouse model of congenital human cytomegalovirus infection. J. Virol. 2012, 86, 12795–12805. [Google Scholar] [CrossRef] [Green Version]
- Lusis, A.J. Atherosclerosis. Nature 2000, 407, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Prochnau, D.; Lehmann, M.; Straube, E.; Figulla, H.R.; Rödel, J. Human cytomegalovirus induces MMP-1 and MMP-3 expression in aortic smooth muscle cells. Acta Microbiol. Immunol. Hung. 2011, 58, 303–317. [Google Scholar] [CrossRef]
- Tanaka, K.; Zou, J.P.; Takeda, K.; Ferrans, V.J.; Sandford, G.R.; Johnson, T.M.; Finkel, T.; Epstein, S.E. Effects of human cytomegalovirus immediate-early proteins on p53-mediated apoptosis in coronary artery smooth muscle cells. Circulation 1999, 99, 1656–1659. [Google Scholar] [CrossRef] [Green Version]
- Fan, J.; Zhang, W.; Liu, Q. Human cytomegalovirus-encoded miR-US25-1 aggravates the oxidised low density lipoprotein-induced apoptosis of endothelial cells. BioMed Res. Int. 2014, 2014, 531979. [Google Scholar] [CrossRef] [Green Version]
- Lunardi, C.; Dolcino, M.; Peterlana, D.; Bason, C.; Navone, R.; Tamassia, N.; Tinazzi, E.; Beri, R.; Corrocher, R.; Puccetti, A. Endothelial cells’ activation and apoptosis induced by a subset of antibodies against human cytomegalovirus: Relevance to the pathogenesis of atherosclerosis. PLoS ONE 2007, 2, e473. [Google Scholar] [CrossRef] [Green Version]
- Priel, E.; Wohl, A.; Teperberg, M.; Nass, D.; Cohen, Z.R. Human cytomegalovirus viral load in tumor and peripheral blood samples of patients with malignant gliomas. J. Clin. Neurosci. 2015, 22, 326–330. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Liang, J.; Xu, G.X.; Ding, L.M.; Huang, H.M.; Su, Q.Z.; Yan, J.; Li, Y.C. Human cytomegalovirus glycoprotein B inhibits migration of breast cancer MDA-MB-231 cells and impairs TGF-β/Smad2/3 expression. Oncol. Lett. 2018, 15, 7730–7738. [Google Scholar] [CrossRef]
- Koldehoff, M.; Lindemann, M.; Ross, S.R.; Elmaagacli, A.H. Cytomegalovirus induces HLA-class-II-restricted alloreactivity in an acute myeloid leukemia cell line. PLoS ONE 2018, 13, e0191482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liang, Q.; Wang, K.; Wang, B.; Cai, Q. HCMV-encoded miR-UL112-3p promotes glioblastoma progression via tumour suppressor candidate 3. Sci. Rep. 2017, 7, 44705. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, T.; Qian, D.; Hu, M.; Li, L.; Zhang, L.; Chen, H.; Yang, R.; Wang, B. Human cytomegalovirus inhibits apoptosis by regulating the activating transcription factor 5 signaling pathway in human malignant glioma cells. Oncol. Lett. 2014, 8, 1051–1057. [Google Scholar] [CrossRef] [PubMed]
- Cinatl, J., Jr.; Cinatl, J.; Vogel, J.U.; Kotchetkov, R.; Driever, P.H.; Kabickova, H.; Kornhuber, B.; Schwabe, D.; Doerr, H.W. Persistent human cytomegalovirus infection induces drug resistance and alteration of programmed cell death in human neuroblastoma cells. Cancer Res. 1998, 58, 367–372. [Google Scholar] [PubMed]
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef] [Green Version]
- Valle Oseguera, C.A.; Spencer, J.V. cmvIL-10 stimulates the invasive potential of MDA-MB-231 breast cancer cells. PLoS ONE 2014, 9, e88708. [Google Scholar] [CrossRef]
- Döhner, H.; Weisdorf, D.J.; Bloomfield, C.D. Acute Myeloid Leukemia. N. Engl. J. Med. 2015, 373, 1136–1152. [Google Scholar] [CrossRef] [Green Version]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [Green Version]
- Kumar, A.; Coquard, L.; Pasquereau, S.; Russo, L.; Valmary-Degano, S.; Borg, C.; Pothier, P.; Herbein, G. Tumor control by human cytomegalovirus in a murine model of hepatocellular carcinoma. Mol. Ther. Oncolytics 2016, 3, 16012. [Google Scholar] [CrossRef]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Khanipouyani, F.; Akrami, H.; Fattahi, M.R. Circular RNAs as important players in human gastric cancer. Clin. Transl. Oncol. 2020, 23, 10–21. [Google Scholar] [CrossRef]
- Hu, S.J.; Jiang, S.S.; Zhang, J.; Luo, D.; Yu, B.; Yang, L.Y.; Zhong, H.H.; Yang, M.W.; Liu, L.Y.; Hong, F.F.; et al. Effects of apoptosis on liver aging. World J. Clin. Cases 2019, 7, 691–704. [Google Scholar] [CrossRef]
- Zhong, H.H.; Hu, S.J.; Yu, B.; Jiang, S.S.; Zhang, J.; Luo, D.; Yang, M.W.; Su, W.Y.; Shao, Y.L.; Deng, H.L.; et al. Apoptosis in the aging liver. Oncotarget 2017, 8, 102640–102652. [Google Scholar] [CrossRef] [Green Version]
- Chaudhry, M.Z.; Casalegno-Garduno, R.; Sitnik, K.M.; Kasmapour, B.; Pulm, A.K.; Brizic, I.; Eiz-Vesper, B.; Moosmann, A.; Jonjic, S.; Mocarski, E.S.; et al. Cytomegalovirus inhibition of extrinsic apoptosis determines fitness and resistance to cytotoxic CD8 T cells. Proc. Natl. Acad. Sci. USA 2020, 117, 12961–12968. [Google Scholar] [CrossRef]
- Fletcher-Etherington, A.; Nobre, L.; Nightingale, K.; Antrobus, R.; Nichols, J.; Davison, A.J.; Stanton, R.J.; Weekes, M.P. Human cytomegalovirus protein pUL36: A dual cell death pathway inhibitor. Proc. Natl. Acad. Sci. USA 2020, 117, 18771–18779. [Google Scholar] [CrossRef]
- Zhang, A.; Hildreth, R.L.; Colberg-Poley, A.M. Human cytomegalovirus inhibits apoptosis by proteasome-mediated degradation of Bax at endoplasmic reticulum-mitochondrion contacts. J. Virol. 2013, 87, 5657–5668. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, R.; Hu, M.; Liang, S.; Wang, B.; Yu, B.; Yang, G.; Qian, D. IE86 Inhibits the apoptosis and promotes the cell proliferation of glioma cells via the hnRNP A2/B1-mediated alternative splicing of Bcl-x. Int. J. Clin. Exp. Pathol. 2019, 12, 2775–2785. [Google Scholar] [PubMed]
- Luganini, A.; Di Nardo, G.; Munaron, L.; Gilardi, G.; Fiorio Pla, A.; Gribaudo, G. Human cytomegalovirus US21 protein is a viroporin that modulates calcium homeostasis and protects cells against apoptosis. Proc. Natl. Acad. Sci. USA 2018, 115, e12370–e12377. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.H.; Kim, J.; Roh, J.; Park, C.S.; Seoh, J.Y.; Hwang, E.S. Reactive oxygen species-induced parthanatos of immunocytes by human cytomegalovirus-associated substance. Microbiol. Immunol. 2018, 62, 229–242. [Google Scholar] [CrossRef] [Green Version]
- Shao, Y.; Qi, Y.; Huang, Y.; Liu, Z.; Ma, Y.; Guo, X.; Jiang, S.; Sun, Z.; Ruan, Q. Human cytomegalovirus miR-US4-5p promotes apoptosis via downregulation of p21-activated kinase 2 in cultured cells. Mol. Med. Rep. 2017, 16, 4171–4178. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Wang, B.; Qian, D.; Wang, M.; Huang, R.; Wei, L.; Li, L.; Zhang, L.; Liu, D.X. Human cytomegalovirus immediate-early protein promotes survival of glioma cells through interacting and acetylating ATF5. Oncotarget 2017, 8, 32157–32170. [Google Scholar] [CrossRef] [Green Version]
- Shao, Y.; Qi, Y.; Huang, Y.; Liu, Z.; Ma, Y.; Guo, X.; Jiang, S.; Sun, Z.; Ruan, Q. Human cytomegalovirus-encoded miR-US4-1 promotes cell apoptosis and benefits discharge of infectious virus particles by targeting QARS. J. Biosci. 2016, 41, 183–192. [Google Scholar] [CrossRef]
- Cojohari, O.; Peppenelli, M.A.; Chan, G.C. Human Cytomegalovirus Induces an Atypical Activation of Akt To Stimulate the Survival of Short-Lived Monocytes. J. Virol. 2016, 90, 6443–6452. [Google Scholar] [CrossRef] [Green Version]
- Guo, X.; Huang, Y.; Qi, Y.; Liu, Z.; Ma, Y.; Shao, Y.; Jiang, S.; Sun, Z.; Ruan, Q. Human cytomegalovirus miR-UL36-5p inhibits apoptosis via downregulation of adenine nucleotide translocator 3 in cultured cells. Arch. Virol. 2015, 160, 2483–2490. [Google Scholar] [CrossRef]
- Lares, A.P.; Tu, C.C.; Spencer, J.V. The human cytomegalovirus US27 gene product enhances cell proliferation and alters cellular gene expression. Virus Res. 2013, 176, 312–320. [Google Scholar] [CrossRef] [Green Version]
- Smith, W.; Tomasec, P.; Aicheler, R.; Loewendorf, A.; Nemčovičová, I.; Wang, E.C.; Stanton, R.J.; Macauley, M.; Norris, P.; Willen, L.; et al. Human cytomegalovirus glycoprotein UL141 targets the TRAIL death receptors to thwart host innate antiviral defenses. Cell Host. Microbe 2013, 13, 324–335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.P.; Qi, Y.; Huang, Y.J.; Qi, M.L.; Ma, Y.P.; He, R.; Ji, Y.H.; Sun, Z.R.; Ruan, Q. Identification of immediate early gene X-1 as a cellular target gene of hcmv-mir-UL148D. Int. J. Mol. Med. 2013, 31, 959–966. [Google Scholar] [CrossRef] [Green Version]
- Yin, X.M.; Ding, W.X. Death receptor activation-induced hepatocyte apoptosis and liver injury. Curr. Mol. Med. 2003, 3, 491–508. [Google Scholar] [CrossRef] [PubMed]
- Raducka-Jaszul, O.; Bogusławska, D.M.; Jędruchniewicz, N.; Sikorski, A.F. Role of Extrinsic Apoptotic Signaling Pathway during Definitive Erythropoiesis in Normal Patients and in Patients with β-Thalassemia. Int. J. Mol. Sci. 2020, 21, 3325. [Google Scholar] [CrossRef]
- Seirafian, S.; Prod’homme, V.; Sugrue, D.; Davies, J.; Fielding, C.; Tomasec, P.; Wilkinson, G.W.G. Human cytomegalovirus suppresses Fas expression and function. J. Gen. Virol. 2014, 95, 933–939. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Terrasson, J.; Allart, S.; Martin, H.; Lulé, J.; Haddada, H.; Caput, D.; Davrinche, C. p73-dependent apoptosis through death receptor: Impairment by human cytomegalovirus infection. Cancer Res. 2005, 65, 2787–2794. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poole, E.; Lau, J.C.H.; Sinclair, J. Latent infection of myeloid progenitors by human cytomegalovirus protects cells from FAS-mediated apoptosis through the cellular IL-10/PEA-15 pathway. J. Gen. Virol. 2015, 96, 2355–2359. [Google Scholar] [CrossRef]
- Chiou, S.H.; Yang, Y.P.; Lin, J.C.; Hsu, C.H.; Jhang, H.C.; Yang, Y.T.; Lee, C.H.; Ho, L.L.; Hsu, W.M.; Ku, H.H.; et al. The immediate early 2 protein of human cytomegalovirus (HCMV) mediates the apoptotic control in HCMV retinitis through up-regulation of the cellular FLICE-inhibitory protein expression. J. Immunol. 2006, 177, 6199–6206. [Google Scholar] [CrossRef]
- McCormick, A.L.; Roback, L.; Livingston-Rosanoff, D.; St Clair, C. The human cytomegalovirus UL36 gene controls caspase-dependent and -independent cell death programs activated by infection of monocytes differentiating to macrophages. J. Virol. 2010, 84, 5108–5123. [Google Scholar] [CrossRef] [Green Version]
- Chien, H.; Dix, R.D. Evidence for multiple cell death pathways during development of experimental cytomegalovirus retinitis in mice with retrovirus-induced immunosuppression: Apoptosis, necroptosis, and pyroptosis. J. Virol. 2012, 86, 10961–10978. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feldstein, A.E.; Gores, G.J. Apoptosis in alcoholic and nonalcoholic steatohepatitis. Front. Biosci. J. Virtual Libr. 2005, 10, 3093–3099. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, H.P.; Yuan, C.L.; Zho, Y.C. Human cytomegalovirus inhibits apoptosis involving upregulation of the antiapoptotic protein Bag-1. J. Med. Virol. 2015, 87, 1953–1959. [Google Scholar] [CrossRef] [PubMed]
- Chiou, S.H.; Liu, J.H.; Chen, S.S.; Liu, W.T.; Lin, J.C.; Wong, W.W.; Tseng, W.S.; Chou, C.K.; Liu, C.Y.; Ho, L.L.; et al. Apoptosis of human retina and retinal pigment cells induced by human cytomegalovirus infection. Ophthalmic Res. 2002, 34, 77–82. [Google Scholar] [CrossRef]
- Dou, J.; Li, X.; Cai, Y.; Chen, H.; Zhu, S.; Wang, Q.; Zou, X.; Mei, Y.; Yang, Q.; Li, W.; et al. Human cytomegalovirus induces caspase-dependent apoptosis of megakaryocytic CHRF-288-11 cells by activating the JNK pathway. Int. J. Hematol. 2010, 91, 620–629. [Google Scholar] [CrossRef]
- Shen, Y.H.; Utama, B.; Wang, J.; Raveendran, M.; Senthil, D.; Waldman, W.J.; Belcher, J.D.; Vercellotti, G.; Martin, D.; Mitchelle, B.M.; et al. Human cytomegalovirus causes endothelial injury through the ataxia telangiectasia mutant and p53 DNA damage signaling pathways. Circ. Res. 2004, 94, 1310–1317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Terhune, S.; Torigoi, E.; Moorman, N.; Silva, M.; Qian, Z.; Shenk, T.; Yu, D. Human cytomegalovirus UL38 protein blocks apoptosis. J. Virol. 2007, 81, 3109–3123. [Google Scholar] [CrossRef] [Green Version]
- Qi, R.; Hua-Song, Z.; Xiao-Feng, Z. Leflunomide inhibits the apoptosis of human embryonic lung fibroblasts infected by human cytomegalovirus. Eur. J. Med. Res. 2013, 18, 3. [Google Scholar] [CrossRef] [Green Version]
- Biolatti, M.; Blangetti, M.; D’Arrigo, G.; Spyrakis, F.; Cappello, P.; Albano, C.; Ravanini, P.; Landolfo, S.; De Andrea, M.; Prandi, C.; et al. Strigolactone Analogs Are Promising Antiviral Agents for the Treatment of Human Cytomegalovirus Infection. Microorganisms 2020, 8, 703. [Google Scholar] [CrossRef]
- Mo, J.; Zhang, M.; Marshall, B.; Smith, S.; Covar, J.; Atherton, S. Interplay of autophagy and apoptosis during murine cytomegalovirus infection of RPE cells. Mol. Vis. 2014, 20, 1161–1173. [Google Scholar]
| No. | Virus | Key Gene/Protein | Expression Phase | Expression Levels | Host Tissue/Cell Line | Effector Gene/Protein |
Results (Impact Apoptosis) | Reference |
|---|---|---|---|---|---|---|---|---|
| 1 | HCMV | miR-UL70-3p/ miR-UL148D | Latent phases | ↑ | / | MOAP1/PHAP/ERN1 | ↓ | [3] |
| 2 | HCMV | IE2 | Immediate early phases | ↑ | Rat aortic smooth muscle cell | Mcl-1/Bcl-2 | ↓ | [7] |
| 3 | HCMV | UL36 | Immediate early phases | ↑ | THP-1 cells | Caspase-8 | ↓ | [17] |
| 4 | HCMV | UL138 | Latent phases | ↓ | Gastric cancer cell | HSP70 | ↑ | [53] |
| 5 | HCMV | pUL38 | Immediate early phases | ↑ | Human embryonic lung fibroblasts | USP24/NCOA4 | ↓ | [54] |
| 6 | HCMV | miR-US25-1 | Late phase | ↑ | Endothelial cells | BRCC 3 | ↑ | [72] |
| 7 | HCMV | miR-UL112-3p | Immediate early phases | ↑ | Glioblastoma cell | TUSC3 | ↓ | [77] |
| 8 | HCMV | cmvIL-10 | Productive and latent phases | ↑ | Breast cancer cell | Stat3 | ↓ | [81] |
| 9 | HCMV/MCMV | vICA | Immediate early phases | ↑ | CD8 T cell | Caspase-8 | ↓ | [89] |
| 10 | HCMV | pUL36 | Immediate early phases | ↑ |
Mouse embryonic fibroblasts/ primary human fetal foreskin fibroblasts | MLKL | ↓ | [90] |
| 11 | HCMV | pUL37x1/vMIA | Immediate early phases | ↑ | Human fibroblasts | Bax | ↓ | [91] |
| 12 | HCMV | IE86 | Immediate early phases | ↑ | U251 cell | hnRNP A2/B1 | ↓ | [92] |
| 13 | HCMV | pUS21 | Late phase | ↑ | Human foreskin fibroblasts | Caspase-7/3 | ↓ | [93] |
| 14 | HCMV | HCMVAIS | / | ↑ | Human embryo lung fibroblasts | NOX-2/PARP-1 | ↑ | [94] |
| 15 | HCMV | hcmv-miR-US4-5p | Immediate early phases | ↑ |
Human embryonic kidney cell/ human embryonic lung fibroblast cell/ human monocytic cell | PAK2 | ↑ | [95] |
| 16 | HCMV | IE86 | Immediate early phases | ↑ | Glioma cell | ATF5 | ↓ | [96] |
| 17 | HCMV | hcmv-miR-US4-1 | / | ↑ | Human embryonic lung fibroblast | QARS | ↑ | [97] |
| 18 | HCMV | Glycoprotein gB | Immediate early phases | ↑ | Human peripheral blood monocytes | Akt | ↓ | [98] |
| 19 | HCMV | hcmv-miR-UL36-5p | Immediate early phases | ↑ | Human embryonic kidney cell/human embryonic lung fibroblasts/glioma cell | ANT3 | ↓ | [99] |
| 20 | HCMV | US27 | Late phase | ↑ | Human embryonic kidney cell | Bcl-x/AP-1 | ↓ | [100] |
| 21 | HCMV | UL141 | / | ↑ |
Human fibroblasts/ normal human dermal fibroblasts | TRAIL-R | ↓ | [101] |
| 22 | HCMV | hcmv-mir-UL148D | Immediate early phases | ↑ | Human embryonic kidney cell | IEX-1 | ↓ | [102] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, Z.; Wang, Y.; Liu, L.; Zhang, X.; Jiang, S.; Wang, B. Apoptosis Disorder, a Key Pathogenesis of HCMV-Related Diseases. Int. J. Mol. Sci. 2021, 22, 4106. https://doi.org/10.3390/ijms22084106
Yu Z, Wang Y, Liu L, Zhang X, Jiang S, Wang B. Apoptosis Disorder, a Key Pathogenesis of HCMV-Related Diseases. International Journal of Molecular Sciences. 2021; 22(8):4106. https://doi.org/10.3390/ijms22084106
Chicago/Turabian StyleYu, Zhongjie, Yashuo Wang, Lili Liu, Xianjuan Zhang, Shasha Jiang, and Bin Wang. 2021. "Apoptosis Disorder, a Key Pathogenesis of HCMV-Related Diseases" International Journal of Molecular Sciences 22, no. 8: 4106. https://doi.org/10.3390/ijms22084106
APA StyleYu, Z., Wang, Y., Liu, L., Zhang, X., Jiang, S., & Wang, B. (2021). Apoptosis Disorder, a Key Pathogenesis of HCMV-Related Diseases. International Journal of Molecular Sciences, 22(8), 4106. https://doi.org/10.3390/ijms22084106






