Drusenoid Pigment Epithelial Detachment: Genetic and Clinical Characteristics
Abstract
1. Introduction
2. Results
3. Discussion
4. Methods
4.1. Patients
4.2. Diagnosis
4.3. Genotyping
4.4. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Casswell, A.G.; Kohen, D.; Bird, A.C. Retinal pigment epithelial detachments in the elderly: Classification and outcome. Br. J. Ophthalmol. 1985, 69, 397–403. [Google Scholar] [CrossRef]
- Balaratnasingam, C.; Yannuzzi, L.A.; Curcio, C.A.; Morgan, W.H.; Querques, G.; Capuano, V.; Souied, E.; Jung, J.; Freund, K.B. Associations Between Retinal Pigment Epithelium and Drusen Volume Changes During the Lifecycle of Large Drusenoid Pigment Epithelial Detachments. Investig. Opthalmol. Vis. Sci. 2016, 57, 5479–5489. [Google Scholar] [CrossRef]
- Roquet, W.; Roudot-Thoraval, F.; Coscas, G.; Soubrane, G. Clinical features of drusenoid pigment epithelial detachment in age related macular degeneration. Br. J. Ophthalmol. 2004, 88, 638–642. [Google Scholar] [CrossRef]
- Cukras, C.; Agrón, E.; Klein, M.L.; Ferris, F.L., 3rd; Chew, E.Y.; Gensler, G.; Wong, W.T. Natural History of Drusenoid Pigment Epithelial Detachment in Age-Related Macular Degeneration: Age-Related Eye Disease Study Report No. 28. Ophthalmology 2010, 117, 489–499. [Google Scholar] [CrossRef]
- Yu, J.J.; Agrón, E.; Clemons, T.E.; Domalpally, A.; Van Asten, F.; Keenan, T.D.; Cukras, C.; Chew, E.Y.; Ferris, F.L.; SanGiovanni, J.P.; et al. Natural History of Drusenoid Pigment Epithelial Detachment Associated with Age-Related Macular Degeneration. Ophthalmology 2019, 126, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.-Y.; Yamashiro, K.; Chen, L.J.; Ahn, J.; Huang, L.; Huang, L.; Cheung, C.M.G.; Miyake, M.; Cackett, P.D.; Yeo, I.Y.; et al. New loci and coding variants confer risk for age-related macular degeneration in East Asians. Nat. Commun. 2015, 6, 6063. [Google Scholar] [CrossRef] [PubMed]
- Fritsche, L.G.; Igl, W.; Bailey, J.N.C.; Grassmann, F.; Sengupta, S.; Bragg-Gresham, J.L.; Burdon, K.P.; Hebbring, S.J.; Wen, C.; Gorski, M.; et al. A large genome-wide association study of age-related macular degeneration highlights contributions of rare and common variants. Nat. Genet. 2016, 48, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Nakayama, T.; Yuzawa, M.; Wang, Z.; Kawamura, A.; Mori, R.; Nakashizuka, H.; Sato, N.; Mizutani, Y. Analysis of candidate genes for age-related macular degeneration subtypes in the Japanese population. Mol. Vis. 2011, 17, 2751–2758. [Google Scholar] [PubMed]
- Yoneyama, S.; Sakurada, Y.; Mabuchi, F.; Sugiyama, A.; Kubota, T.; Iijima, H. Genetic Variants in the SKIV2L Gene in Exudative Age-related Macular Degeneration in the Japanese Population. Ophthalmic Genet. 2014, 35, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Seddon, J.M.; Francis, P.J.; George, S.; Schultz, D.W.; Rosner, B.; Klein, M.L. Association of CFH Y402H and LOC387715 A69S With Progression of Age-Related Macular Degeneration. JAMA 2007, 297, 1793–1800. [Google Scholar] [CrossRef]
- Kim, J.H.; Kang, S.W.; Kim, S.J.; Ha, H.S.; Kim, J.R. Thinner Choroid and Greater Drusen Extent in Retinal Angiomatous Proliferation Than in Typical Exudative Age-Related Macular Degeneration. Am. J. Ophthalmol. 2013, 155, 743–749.e2. [Google Scholar] [CrossRef]
- Mimoun, G.; Soubrane, G.; Coscas, G. [Macular drusen]. J. Fr. Ophtalmol. 1990, 13, 511–530. [Google Scholar] [PubMed]
- Alten, F.; Clemens, C.R.; Milojcic, C.; Eter, N. Subretinal drusenoid deposits associated with pigment epithelium detachment in age-related macular degeneration. Retina 2012, 32, 1727–1732. [Google Scholar] [CrossRef]
- Donato, L.; Scimone, C.; Alibrandi, S.; Pitruzzella, A.; Scalia, F.; D’Angelo, R.; Sidoti, A. Possible A2E Mutagenic Effects on RPE Mitochondrial DNA from Innovative RNA-Seq Bioinformatics Pipeline. Antioxidants 2020, 9, 1158. [Google Scholar] [CrossRef] [PubMed]
- Scimone, C.; Alibrandi, S.; Scalinci, S.Z.; Battagliola, E.T.; D’Angelo, R.; Sidoti, A.; Donato, L. Expression of Pro-Angiogenic Markers Is Enhanced by Blue Light in Human RPE Cells. Antioxidants 2020, 9, 1154. [Google Scholar] [CrossRef] [PubMed]
- Donato, L.; Scimone, C.; Alibrandi, S.; Abdalla, E.M.; Nabil, K.M.; D’Angelo, R.; Sidoti, A. New Omics–Derived Perspectives on Retinal Dystrophies: Could Ion Channels-Encoding or Related Genes Act as Modifier of Pathological Phenotype? Int. J. Mol. Sci. 2020, 22, 70. [Google Scholar] [CrossRef] [PubMed]
- Tan, A.C.; Simhaee, D.; Balaratnasingam, C.; Dansingani, K.K.; Yannuzzi, L.A. A Perspective on the Nature and Frequency of Pigment Epithelial Detachments. Am. J. Ophthalmol. 2016, 172, 13–27. [Google Scholar] [CrossRef]
- Sakurada, Y.; Yoneyama, S.; Sugiyama, A.; Tanabe, N.; Kikushima, W.; Mabuchi, F.; Kume, A.; Kubota, T.; Iijima, H. Prevalence and Genetic Characteristics of Geographic Atrophy among Elderly Japanese with Age-Related Macular Degeneration. PLoS ONE 2016, 11, e0149978. [Google Scholar] [CrossRef] [PubMed]
- Shijo, T.; Sakurada, Y.; Yoneyama, S.; Sugiyama, A.; Kikushima, W.; Tanabe, N.; Iijima, H. Prevalence and characteristics of pseudodrusen subtypes in advanced age-related macular degeneration. Graefe’s Arch. Clin. Exp. Ophthalmol. 2017, 255, 1125–1131. [Google Scholar] [CrossRef]
- Tanaka, K.; Nakayama, T.; Mori, R.; Sato, N.; Kawamura, A.; Mizutani, Y.; Yuzawa, M. Associations of Complement Factor H (CFH) and Age-Related Maculopathy Susceptibility 2 (ARMS2) Genotypes with Subtypes of Polypoidal Choroidal Vasculopathy. Investig. Opthalmol. Vis. Sci. 2011, 52, 7441–7444. [Google Scholar] [CrossRef]
- Miki, A.; Honda, S.; Kondo, N.; Negi, A. The Association ofAge-related Maculopathy Susceptibility 2 (ARMS2)andComplement Factor H (CFH)Variants with Two Angiographic Subtypes of Polypoidal Choroidal Vasculopathy. Ophthalmic Genet. 2013, 34, 146–150. [Google Scholar] [CrossRef] [PubMed]
- Shijo, T.; Sakurada, Y.; Yoneyama, S.; Kikushima, W.; Sugiyama, A.; Matsubara, M.; Fukuda, Y.; Mabuchi, F.; Kashiwagi, K. Association between Polygenic Risk Score and One-Year Outcomes Following As-Needed Aflibercept Therapy for Exudative Age-Related Macular Degeneration. Pharmaceuticals 2020, 13, 257. [Google Scholar] [CrossRef] [PubMed]
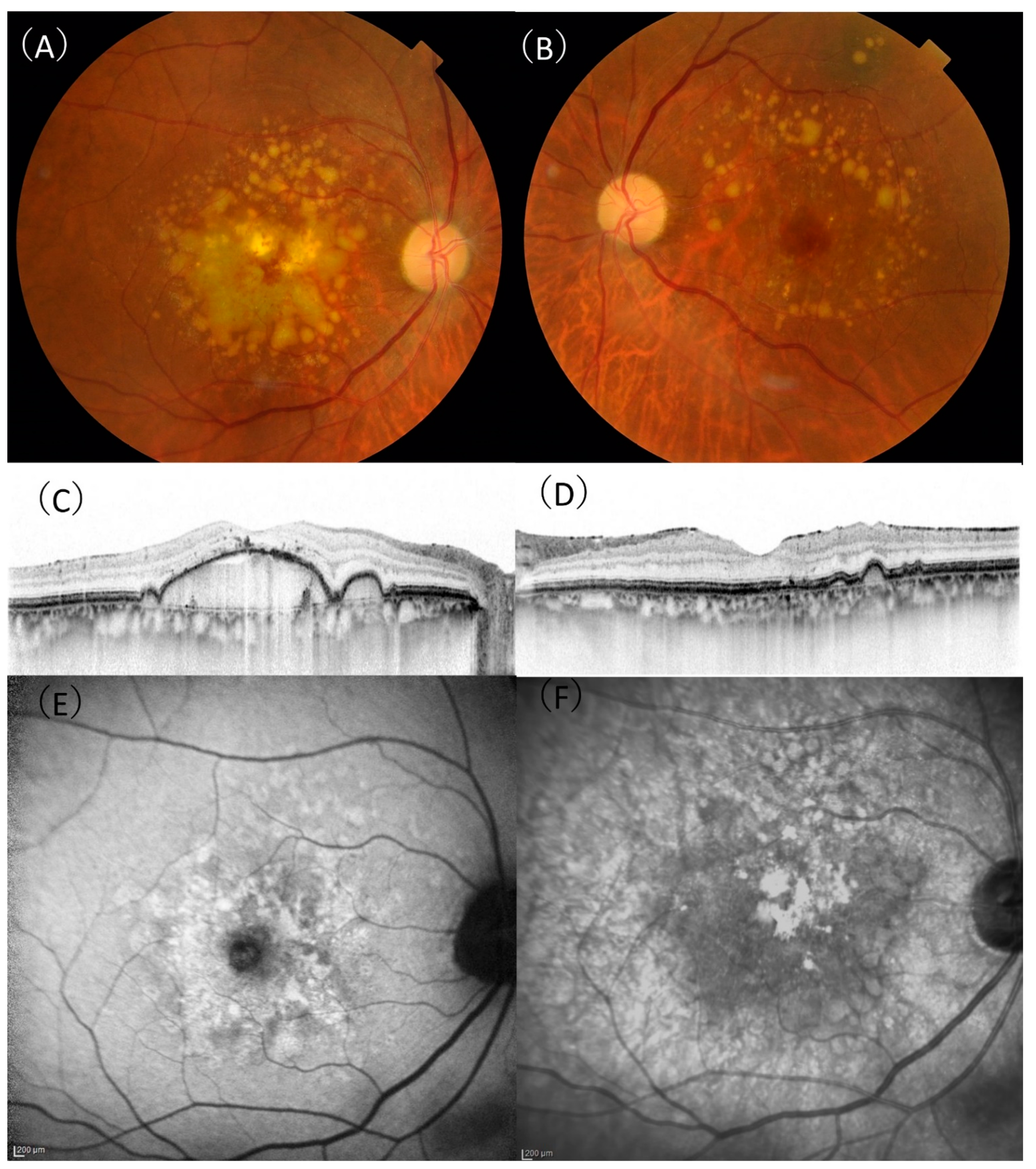
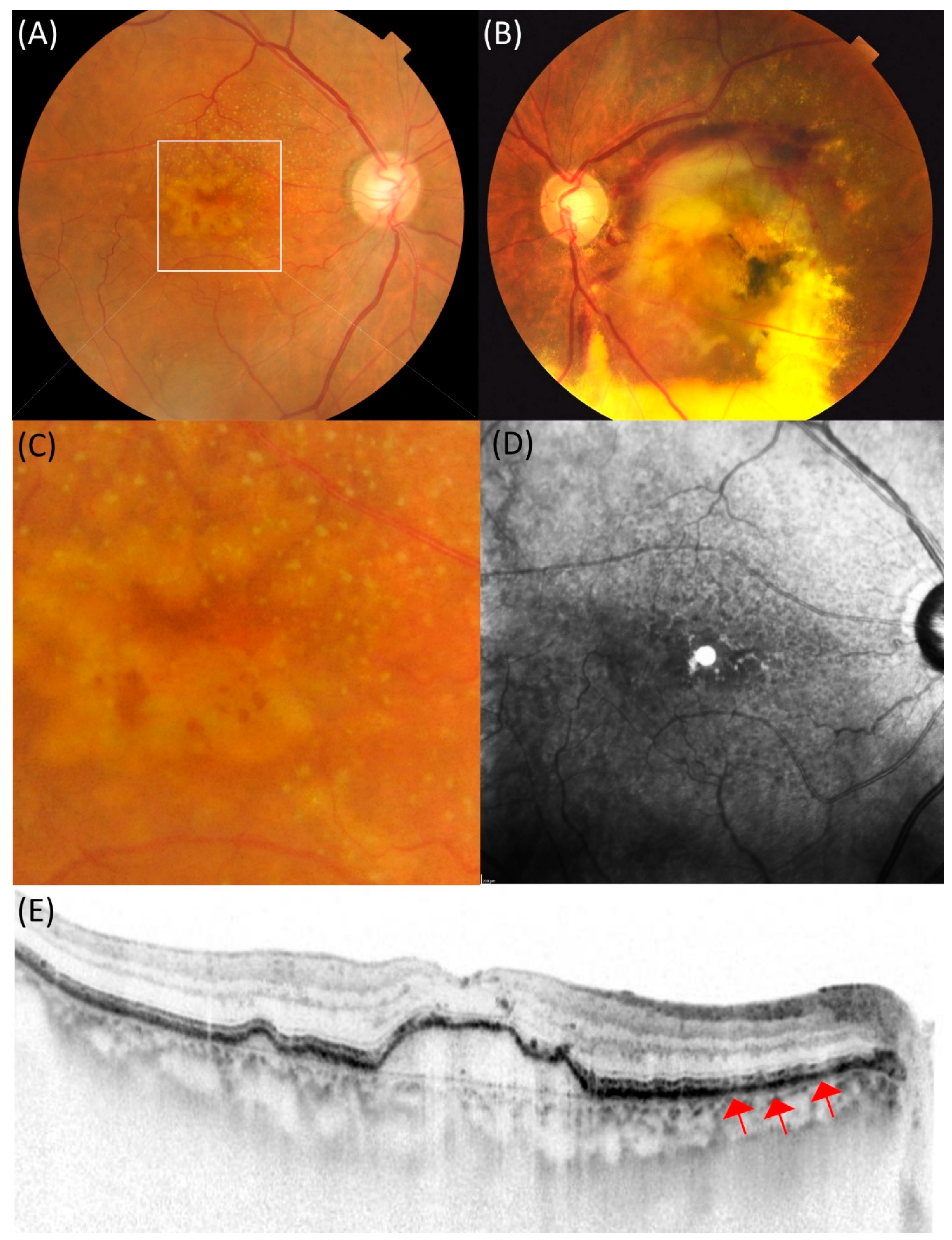
| DPED (n = 76) | tAMD (n = 328) | PCV (n = 476) | RAP (n = 57) | |
|---|---|---|---|---|
| Mean age (years) | 77.2 ± 7.0 | 76.6 ± 8.7 | 73.3 ± 8.2 | 82.5 ± 6.5 |
| p-value (vs. DPED) | 0.97 | 4.3 × 10−4 | 2.3 × 10−5 | |
| Gender: Male | 42 (55.3%) | 227 (69.2%) | 359 (75.4%) | 23 (40.4%) |
| p-value (vs. DPED) | 0.02 | 0.0003 | 0.09 | |
| Presence of RPD | 17 (22.4%) | 65 (19.8%) | 9 (1.9%) | 34 (60.0%) |
| p-value (vs. DPED) | 0.62 | 5.1 × 10−15 | 1.2 × 10−5 | |
| DPED (n = 64) | tAMD (n = 328) | PCV (n = 476) | RAP (n = 57) | |
| ARMS2 A69S(c.205G > T) (rs10490924)) | ||||
| TT | 38 (59.4%) | 152 (46.3%) | 160 (33.6%) | 39 (68.4%) |
| TG | 23 (36.0%) | 126 (38.4%) | 223 (46.8%) | 16 (28.1%) |
| GG | 3 (4.6%) | 50 (15.3%) | 93 (19.6%) | 2 (3.5%) |
| T-allele frequency | 0.77 | 0.66 | 0.57 | 0.82 |
| p-value (vs. DPED) | 9.2 × 10−3 | 7.2 × 10−5 | 0.32 | |
| CFH I62V(c.184G > A) (rs800292) | ||||
| GG | 48 (75.0%) | 179 (54.6%) | 259 (54.4%) | 35 (61.4%) |
| GA | 15 (23.4%) | 122 (37.2%) | 176 (37.0%) | 20 (35.1%) |
| AA | 1 (1.6%) | 27 (8.2%) | 41 (8.6%) | 2 (3.5%) |
| G-allele frequency | 0.87 | 0.73 | 0.73 | 0.79 |
| p-value (vs. DPED) | 0.0011 | 0.008 | 0.11 |
| RPD(+) (n = 17) | RPD(−) (n = 59) | p-Value | |
|---|---|---|---|
| Mean age (years) | 77.65 ± 7.47 | 77.19 ± 6.96 | 0.78 |
| Gender: Male | 8 (47.1%) | 34 (57.6%) | 0.44 |
| Baseline VA (LogMAR) | 0.11 ± 0.22 | 0.082 ± 0.16 | 0.80 |
| Presence of MNV in the contralateral eye | 14 (82.4%) | 37 (62.7%) | 0.11 |
| DPED height (µm) | 151.12 ± 52.44 | 227.24 ± 142.49 | 0.028 |
| DPED width (µm) | 1339.77 ± 1182.09 | 1718.05 ± 1094.18 | 0.12 |
| RPD(+) (n = 16) | RPD(−) (n = 48) | p-Value | |
| ARMS2 A69S(c.205G > T) (rs10490924) | |||
| TT | 12 (75.0%) | 26 (54.2%) | |
| TG | 4 (25.0%) | 19 (39.6%) | |
| GG | 0 (0.0%) | 3 (6.2%) | |
| T-allele frequency | 0.88 | 0.74 | 0.11 |
| CFH I62V (c.184G > A) (rs800292) | |||
| GG | 12 (75.0%) | 36 (75.0%) | |
| GA | 3 (18.8%) | 12 (25.0%) | |
| AA | 1 (6.2%) | 0 (0.0%) | |
| G-allele frequency | 0.84 | 0.88 | 0.65 |
| MNV(+) (n = 51) | MNV(−) (n = 25) | p-Value | |
|---|---|---|---|
| Mean age (years) | 77.43 ± 6.96 | 76.84 ± 7.30 | 0.74 |
| Gender: Male | 27 (53.0%) | 15 (60.0%) | 0.56 |
| Baseline VA (LogMAR) | 0.083 ± 0.18 | 0.095 ± 0.17 | 0.56 |
| Presence of RPD | 14 (27.5%) | 3 (12%) | 0.13 |
| DPED height (µm) | 183.50 ± 112.43 | 264.72 ± 152.12 | 0.0018 |
| DPED width (µm) | 1520.63 ± 1138.50 | 1863.56 ± 1058.77 | 0.086 |
| MNV(+) (n = 48) | MNV(−) (n = 16) | p-Value | |
| ARMS2 A69S(c.205G > T) (rs10490924)) | |||
| TT | 31 (64.6%) | 7 (43.8%) | |
| TG | 14 (29.2%) | 9 (56.2%) | |
| GG | 3 (6.2%) | 0 (0.0%) | |
| T-allele frequency | 0.79 | 0.72 | 0.39 |
| CFH I62V(c.184G > A) (rs800292) | |||
| GG | 37 (77.1%) | 11 (68.8%) | |
| GA | 10 (20.8%) | 5 (31.2%) | |
| AA | 1 (2.1%) | 0 (0.0%) | |
| G-allele frequency | 0.88 | 0.84 | 0.65 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shijo, T.; Sakurada, Y.; Tanaka, K.; Miki, A.; Yoneyama, S.; Machida, Y.; Chubachi, A.; Wakatsuki, Y.; Sugiyama, A.; Onoe, H.; et al. Drusenoid Pigment Epithelial Detachment: Genetic and Clinical Characteristics. Int. J. Mol. Sci. 2021, 22, 4074. https://doi.org/10.3390/ijms22084074
Shijo T, Sakurada Y, Tanaka K, Miki A, Yoneyama S, Machida Y, Chubachi A, Wakatsuki Y, Sugiyama A, Onoe H, et al. Drusenoid Pigment Epithelial Detachment: Genetic and Clinical Characteristics. International Journal of Molecular Sciences. 2021; 22(8):4074. https://doi.org/10.3390/ijms22084074
Chicago/Turabian StyleShijo, Taiyo, Yoichi Sakurada, Koji Tanaka, Akiko Miki, Seigo Yoneyama, Yumiko Machida, Aya Chubachi, Yu Wakatsuki, Atsushi Sugiyama, Hajime Onoe, and et al. 2021. "Drusenoid Pigment Epithelial Detachment: Genetic and Clinical Characteristics" International Journal of Molecular Sciences 22, no. 8: 4074. https://doi.org/10.3390/ijms22084074
APA StyleShijo, T., Sakurada, Y., Tanaka, K., Miki, A., Yoneyama, S., Machida, Y., Chubachi, A., Wakatsuki, Y., Sugiyama, A., Onoe, H., Kikushima, W., Mori, R., & Kashiwagi, K. (2021). Drusenoid Pigment Epithelial Detachment: Genetic and Clinical Characteristics. International Journal of Molecular Sciences, 22(8), 4074. https://doi.org/10.3390/ijms22084074







