Mitochondrial Dysfunction and Chronic Inflammation in Polycystic Ovary Syndrome
Abstract
1. Introduction
PCOS as a Low-Grade Chronic Inflammation Disease
2. Role of Mitochondria in PCOS
Identification and Analysis of Pathogenic mtDNA Mutations in PCOS Patients
3. Novel Markers for PCOS Diagnosis
4. PCOS Treatments: Classical and Modern Mitochondria–Oriented Approaches
4.1. Mouse Studies
Mitochondria-Targeted Therapy
4.2. Human Therapy
5. Long Noncoding RNAs—New Regulators of PCOS Inflammation
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| αLA | α-linolenic acid |
| ALA | α lipoic acid |
| AMH | anti-Müllerian hormone |
| AMPK | 5’ AMP-activated protein kinase; 5’ adenosine monophosphate-activated protein kinase |
| ApoB | apolipoprotein B |
| ASP | acylation stimulating protein |
| BAX | B-cell lymphoma-2 (Bcl-2)-associated X protein |
| Bcl-xL | B-cell lymphoma-extra large |
| BMI | body mass index |
| CRP | C reactive protein |
| CVD | cardiovascular disease |
| DHEA | dehydroepiandrosterone |
| DHT | dihydrotestosterone |
| DM | diabetes mellitus |
| DPP4 | dipeptidyl peptidase-4 |
| Er | estrogen receptor |
| EV | estradiol valerate |
| FSH | follicle-stimulating hormone |
| GAS5 | growth-arrest specific transcript 5 |
| GH | growth hormone |
| GnRH | gonadotropin-releasing hormone |
| GPx | glutathione peroxidase |
| HbA1c | glycosylated haemoglobin |
| hCG | human chorionic gonadotropin |
| HDL-c | high-density lipoprotein cholesterol |
| HG | hyperglycemia |
| 3β-HSD | 3β-Hydroxysteroid dehydrogenase/Δ5–4 isomerase |
| IKK | IκB kinase |
| IL | interleukin |
| IR | insulin resistance |
| Ki-67 | MKI67 (Marker Of Proliferation Ki-67) |
| LDHA | lactate dehydrogenase A |
| LDL-c | low-density lipoprotein-cholesterol |
| LH | luteinizing hormone |
| lncRNA-SRLR | (sorafenib resistance-associated lncRNA in renal cell carcinoma) |
| MCP-1 | monocyte-chemoattractant-protein-1 |
| MDA | malondialdehyde |
| MetS | metabolic syndrome |
| MMP | mitochondrial membrane potential |
| mtDNA | mitochondrial DNA |
| NAFLD | nonalcoholic fatty liver disease |
| NK | natural killer cells; or large granular lymphocytes (LGL) |
| NO | nitrogen oxide |
| OS | oxidative stress |
| OSI | oxidative stress index |
| OXPHOS | oxidative phosphorylation |
| PCOS | polycystic ovary syndrome |
| PEBP1 | phosphatidylethanolamine-binding protein 1 |
| PFK | phosphofructokinase |
| PI3K | phosphatidylinositol 3-kinase |
| PKM2 | pyruvate kinase isozyme M2 isoform |
| PSME1 | proteasome activator complex subunit 1 |
| rRNA | ribosomal RNAs |
| SIRT1 | silent information regulator 1; sirtuin 1 |
| SOD | superoxide dismutase |
| SRA | steroid receptor RNA activator |
| STAT3 | signal transducer and activator of transcription 3 |
| T2DM | type 2 diabetes mellitus |
| TCA cycle | (tricarboxylic acid cycle); the Krebs cycle |
| TFAM | mitochondrial transcription factor A |
| Th | The T helper cells; CD4+ cells; CD4-positive cells |
| TNF-α | tumour necrosis factor-alpha |
| TOS | total oxidant status |
| TP | testosterone propionate |
| TPI1 | triosephosphate isomerase |
| tRNA | transport RNAs |
| WBC | white blood cell |
References
- Burt Solorzano, C.M.; McCartney, C.R. Polycystic Ovary Syndrome. Endocrinol. Metab. Clin. N. Am. 2021, 50, 25–42. [Google Scholar] [CrossRef]
- NICHD. About Polycystic Ovary Syndrome (PCOS). 2021. Available online: https://www.nichd.nih.gov/health/topics/pcos/conditioninfo (accessed on 20 February 2021).
- Yumiceba, V.; López-Cortés, A.; Pérez-Villa, A.; Yumiseba, I.; Guerrero, S.; García-Cárdenas, J.M.; Armendáriz-Castillo, I.; Guevara-Ramírez, P.; Leone, P.E.; Zambrano, A.K.; et al. Oncology and Pharmacogenomics Insights in Polycystic Ovary Syndrome: An Integrative Analysis. Front. Endocrinol. 2020, 11, 585130. [Google Scholar] [CrossRef]
- Combs, J.C.; Hill, M.J.; Decherney, A.H. Polycystic Ovarian Syndrome Genetics and Epigenetics. Clin. Obstet. Gynecol. 2021, 64, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S. Pathomechanisms of Polycystic Ovary Syndrome Multidimensional Approaches. Front. Biosci. 2018, 10, 384–422. [Google Scholar] [CrossRef] [PubMed]
- The Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 Consensus on Diagnostic Criteria and Long-Term Health Risks Related to Polycystic Ovary Syndrome (PCOS). Hum. Reprod. 2004, 19, 41–47. [CrossRef] [PubMed]
- Chang, S.; Dunaif, A. Diagnosis of Polycystic Ovary Syndrome. Endocrinol. Metab. Clin. N. Am. 2021, 50, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Teede, H.J.; Misso, M.L.; Costello, M.F.; Dokras, A.; Laven, J.; Moran, L.; Piltonen, T.; Norman, R.J.; Andersen, M.; Azziz, R.; et al. Recommendations from the International Evidence-Based Guideline for the Assessment and Management of Polycystic Ovary Syndrome. Fertil. Steril. 2018, 110, 364–379. [Google Scholar] [CrossRef]
- Belenkaia, L.V.; Lazareva, L.M.; Walker, W.; Lizneva, D.V.; Suturina, L.V. Criteria, Phenotypes and Prevalence of Polycystic Ovary Syndrome. Minerva Ginecol. 2019, 71. [Google Scholar] [CrossRef]
- Barber, T.M.; Hanson, P.; Weickert, M.O.; Franks, S. Obesity and Polycystic Ovary Syndrome: Implications for Pathogenesis and Novel Management Strategies. Clin. Med. Insights Reprod. Health 2019, 13, 1179558119874042. [Google Scholar] [CrossRef]
- Dadachanji, R.; Patil, A.; Joshi, B.; Mukherjee, S. Elucidating the Impact of Obesity on Hormonal and Metabolic Perturbations in Polycystic Ovary Syndrome Phenotypes in Indian Women. PLoS ONE 2021, 16, e0246862. [Google Scholar] [CrossRef]
- Cao, N.T.; Le, M.T.; Nguyen, V.Q.H.; Pilgrim, J.; Le, V.N.S.; Le, D.D.; Pham, C.K.; Aharon, D.; Hill, M.J. Defining Polycystic Ovary Syndrome Phenotype in Vietnamese Women. J. Obstet. Gynaecol. Res. 2019, 45, 2209–2219. [Google Scholar] [CrossRef]
- Bhatti, J.S.; Bhatti, G.K.; Reddy, P.H. Mitochondrial Dysfunction and Oxidative Stress in Metabolic DisordersA Step towards Mitochondria Based Therapeutic Strategies. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 1066–1077. [Google Scholar] [CrossRef] [PubMed]
- González, F.; Considine, R.V.; Abdelhadi, O.A.; Acton, A.J. Oxidative Stress in Response to Saturated Fat Ingestion Is Linked to Insulin Resistance and Hyperandrogenism in Polycystic Ovary Syndrome. J. Clin. Endocrinol. Metab. 2019, 104, 5360–5371. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, M. Oxidative Stress and Polycystic Ovary Syndrome: A Brief Review. Int. J. Prev. Med. 2019, 10, 86. [Google Scholar] [CrossRef] [PubMed]
- Cavalcante, M.B.; Sarno, M.; de Melo Bezerra Cavalcante, C.T.; Júnior, E.A.; Barini, R. Coagulation Biomarkers in Women with Recurrent Miscarriage and Polycystic Ovarian Syndrome: Systematic Review and Meta-Analysis. Geburtshilfe Frauenheilkd 2019, 79, 697–704. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Xie, Y.; Qu, L.; Zhang, M.; Mo, Z. Dyslipidemia Involvement in the Development of Polycystic Ovary Syndrome. Taiwan J. Obstet. Gynecol. 2019, 58, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Yao, X.-Y.; Shi, R.-X.; Liu, S.-F.; Wang, X.-Y. A Potential Link between Polycystic Ovary Syndrome and Non-Alcoholic Fatty Liver Disease: An Update Meta-Analysis. Reprod. Health 2018, 15, 77. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Cui, C.; Han, X.; Wang, Q.; Zhang, C. The Role of MiRNAs in Polycystic Ovary Syndrome with Insulin Resistance. J. Assist. Reprod. Genet. 2021, 38, 289–304. [Google Scholar] [CrossRef]
- Castillo-Higuera, T.; Alarcón-Granados, M.C.; Marin-Suarez, J.; Moreno-Ortiz, H.; Esteban-Pérez, C.I.; Ferrebuz-Cardozo, A.J.; Forero-Castro, M.; Camargo-Vill alba, G. A Comprehensive Overview of Common Polymorphic Variants in Genes Related to Polycystic Ovary Syndrome. Reprod. Sci. 2020. [Google Scholar] [CrossRef]
- Welt, C.K. Genetics of Polycystic Ovary Syndrome. Endocrinol. Metab. Clin. N. Am. 2021, 50, 71–82. [Google Scholar] [CrossRef]
- Gupta, M.; Yadav, R.; Mahey, R.; Agrawal, A.; Upadhyay, A.; Malhotra, N.; Bhatla, N. Correlation of Body Mass Index (BMI), Anti-Mullerian Hormone (AMH), and Insulin Resistance among Different Polycystic Ovary Syndrome (PCOS) Phenotypes—A Cross-Sectional Study. Gynecol. Endocrinol. 2019, 35, 970–973. [Google Scholar] [CrossRef] [PubMed]
- Nehir Aytan, A.; Bastu, E.; Demiral, I.; Bulut, H.; Dogan, M.; Buyru, F. Relationship between Hyperandrogenism, Obesity, Inflammation and Polycystic Ovary Syndrome. Gynecol. Endocrinol. 2016, 32, 709–713. [Google Scholar] [CrossRef]
- Patel, S. Polycystic Ovary Syndrome (PCOS), an Inflammatory, Systemic, Lifestyle Endocrinopathy. J. Steroid Biochem. Mol. Biol. 2018, 182, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Abraham Gnanadass, S.; Divakar Prabhu, Y.; Valsala Gopalakrishnan, A. Association of Metabolic and Inflammatory Markers with Polycystic Ovarian Syndrome (PCOS): An Update. Arch. Gynecol. Obstet. 2021. [Google Scholar] [CrossRef] [PubMed]
- Alissa, E.M.; Algarni, S.A.; Khaffji, A.J.; Al Mansouri, N.M. Role of Inflammatory Markers in Polycystic Ovaries Syndrome: In Relation to Insulin Resistance. J. Obstet. Gynaecol. Res. 2021. [Google Scholar] [CrossRef] [PubMed]
- Artimani, T.; Karimi, J.; Mehdizadeh, M.; Yavangi, M.; Khanlarzadeh, E.; Ghorbani, M.; Asadi, S.; Kheiripour, N. Evaluation of Pro-Oxidant-Antioxidant Balance (PAB) and Its Association with Inflammatory Cytokines in Polycystic Ovary Syndrome (PCOS). Gynecol. Endocrinol. 2018, 34, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Khashchenko, E.; Vysokikh, M.; Uvarova, E.; Krechetova, L.; Vtorushina, V.; Ivanets, T.; Volodina, M.; Tarasova, N.; Sukhanova, I.; Sukhikh, G. Activation of Systemic Inflammation and Oxidative Stress in Adolescent Girls with Polycystic Ovary Syndrome in Combination with Metabolic Disorders and Excessive Body Weight. J. Clin. Med. 2020, 9, 1399. [Google Scholar] [CrossRef]
- Rudnicka, E.; Kunicki, M.; Suchta, K.; Machura, P.; Grymowicz, M.; Smolarczyk, R. Inflammatory Markers in Women with Polycystic Ovary Syndrome. BioMed Res. Int. 2020. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.-Z.; Lee, M.-J.; Hu, H.; Pray, J.; Wu, H.-B.; Hansen, B.C.; Shuldiner, A.R.; Fried, S.K.; McLenithan, J.C.; Gong, D.-W. Identification of Omentin as a Novel Depot-Specific Adipokine in Human Adipose Tissue: Possible Role in Modulating Insulin Action. Am. J. Physiol. Endocrinol. Metab. 2006, 290, E1253–E1261. [Google Scholar] [CrossRef]
- Franik, G.; Sadlocha, M.; Madej, P.; Owczarek, A.; Skrzypulec-Plinta, V.; Plinta, R.; Chudek, J.; Olszanecka-Glinianowicz, M. Circulating Omentin-1 Levels and Inflammation in Polycystic Ovary Syndrome. Ginekol. Pol. 2020, 91, 308–312. [Google Scholar] [CrossRef]
- Yang, H.-Y.; Ma, Y.; Lu, X.-H.; Liang, X.-H.; Suo, Y.-J.; Huang, Z.-X.; Lu, D.-C.; Qin, Y.-F.; Luo, Z.-J. The Correlation of Plasma Omentin-1 with Insulin Resistance in Non-Obese Polycystic Ovary Syndrome. Ann. D’endocrinol. 2015, 76, 620–627. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Yu, K.; Yang, Z. Associations between TNF-α and Interleukin Gene Polymorphisms with Polycystic Ovary Syndrome Risk: A Systematic Review and Meta-Analysis. J. Assist. Reprod. Genet. 2015, 32, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Talaat, R.M.; Mohamed, Y.A.; Mohamad, E.H.; Elsharkawy, M.; Guirgis, A.A. Interleukin 10 (−1082 G/A) and (−819 C/T) Gene Polymorphisms in Egyptian Women with Polycystic Ovary Syndrome (PCOS). Meta Gene 2016, 9, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Hesampour, F.; Namavar Jahromi, B.; Tahmasebi, F.; Gharesi-Fard, B. Association between Interleukin-32 and Interleukin-17A Single Nucleotide Polymorphisms and Serum Levels with Polycystic Ovary Syndrome. Iran J. Allergy Asthma Immunol. 2019, 18, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Gong, P.; Li, C.; Pan, M.; Ding, Z.; Ge, X.; Zhu, W.; Shi, B. Correlation between Leptin and IFN-γ Involved in Granulosa Cell Apoptosis in PCOS. Gynecol. Endocrinol. 2020, 36, 1051–1056. [Google Scholar] [CrossRef] [PubMed]
- Hazafa, A.; Batool, A.; Ahmad, S.; Amjad, M.; Chaudhry, S.N.; Asad, J.; Ghuman, H.F.; Khan, H.M.; Naeem, M.; Ghani, U. Humanin: A Mitochondrial-Derived Peptide in the Treatment of Apoptosis-Related Diseases. Life Sci. 2021, 264, 118679. [Google Scholar] [CrossRef]
- Basu, U.; Bostwick, A.M.; Das, K.; Dittenhafer-Reed, K.E.; Patel, S.S. Structure, Mechanism, and Regulation of Mitochondrial DNA Transcription Initiation. J. Biol. Chem. 2020, 295, 18406–18425. [Google Scholar] [CrossRef] [PubMed]
- Montano, V.; Gruosso, F.; Simoncini, C.; Siciliano, G.; Mancuso, M. Clinical Features of MtDNA-Related Syndromes in Adulthood. Arch. Biochem. Biophys. 2021, 697, 108689. [Google Scholar] [CrossRef]
- Nielson, J.R.; Rutter, J.P. Lipid-Mediated Signals That Regulate Mitochondrial Biology. J. Biol. Chem. 2018, 293, 7517–7521. [Google Scholar] [CrossRef]
- Tan, D.Q.; Suda, T. Reactive Oxygen Species and Mitochondrial Homeostasis as Regulators of Stem Cell Fate and Function. Antioxid. Redox Signal. 2018, 29, 149–168. [Google Scholar] [CrossRef]
- Granatiero, V.; De Stefani, D.; Rizzuto, R. Mitochondrial Calcium Handling in Physiology and Disease. In Mitochondrial Dynamics in Cardiovascular Medicine; Advances in Experimental Medicine and Biology; Santulli, G., Ed.; Springer International Publishing: Cham, Germany, 2017; Volume 982, pp. 25–47. ISBN 978-3-319-55329-0. [Google Scholar]
- Vallese, F.; Barazzuol, L.; Maso, L.; Brini, M.; Calì, T. ER-Mitochondria Calcium Transfer, Organelle Contacts and Neurodegenerative Diseases. In Calcium Signaling; Advances in Experimental Medicine and Biology; Islam, M.S., Ed.; Springer International Publishing: Cham, Germany, 2020; Volume 1131, pp. 719–746. ISBN 978-3-030-12456-4. [Google Scholar]
- Mustafa, M.F.; Fakurazi, S.; Abdullah, M.A.; Maniam, S. Pathogenic Mitochondria DNA Mutations: Current Detection Tools and Interventions. Genes 2020, 11, 192. [Google Scholar] [CrossRef]
- Castellani, C.A.; Longchamps, R.J.; Sun, J.; Guallar, E.; Arking, D.E. Thinking Outside the Nucleus: Mitochondrial DNA Copy Number in Health and Disease. Mitochondrion 2020, 53, 214–223. [Google Scholar] [CrossRef]
- Eriksen, M.B.; Minet, A.D.; Glintborg, D.; Gaster, M. Intact Primary Mitochondrial Function in Myotubes Established from Women with PCOS. J. Clin. Endocrinol. Metab. 2011, 96, E1298–E1302. [Google Scholar] [CrossRef]
- Lee, S.-H.; Chung, D.-J.; Lee, H.-S.; Kim, T.-J.; Kim, M.-H.; Jeong, H.J.; Im, J.-A.; Lee, D.-C.; Lee, J.-W. Mitochondrial DNA Copy Number in Peripheral Blood in Polycystic Ovary Syndrome. Metabolism 2011, 60, 1677–1682. [Google Scholar] [CrossRef]
- Rabøl, R.; Svendsen, P.F.; Skovbro, M.; Boushel, R.; Schjerling, P.; Nilas, L.; Madsbad, S.; Dela, F. Skeletal Muscle Mitochondrial Function in Polycystic Ovarian Syndrome. Eur. J. Endocrinol. 2011, 165, 631–637. [Google Scholar] [CrossRef]
- Sies, H. Oxidative Stress. In Stress: Physiology, Biochemistry, and Pathology; Elsevier: Amsterdam, The Netherlands, 2019; pp. 153–163. ISBN 978-0-12-813146-6. [Google Scholar]
- Tauffenberger, A.; Magistretti, P.J. Reactive Oxygen Species: Beyond Their Reactive Behavior. Neurochem. Res. 2021, 46, 77–87. [Google Scholar] [CrossRef]
- Yildirim, E.; Derici, M.K. A Case-Control Study on the Oxidative Status in Women with Polycystic Ovary Syndrome Treated with Clomiphene Citrate. Med. Sci. Monit. 2019, 25, 3910–3917. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Liu, H.; Bai, H.; Zhang, Y.; Liu, Q.; Guan, L.; Fan, P. Oxidative Stress Status in Chinese Women with Different Clinical Phenotypes of Polycystic Ovary Syndrome. Clin. Endocrinol. 2017, 86, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Ruan, X.; Li, Y.; Cheng, J.; Mueck, A.O. Oxidative Stress Indicators in Chinese Women with PCOS and Correlation with Features of Metabolic Syndrome and Dependency on Lipid Patterns. Arch. Gynecol. Obstet. 2019, 300, 1413–1421. [Google Scholar] [CrossRef] [PubMed]
- Rottenberg, H.; Hoek, J.B. The Mitochondrial Permeability Transition: Nexus of Aging, Disease and Longevity. Cells 2021, 10, 79. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, G.; Feng, G.; Leng, J.; Yu, L.; Jiang, Y. A 9-Bp Deletion Homoplasmy in Women with Polycystic Ovary Syndrome Revealed by Mitochondrial Genome-Mutation Screen. Biochem. Genet. 2010, 48, 157–163. [Google Scholar] [CrossRef]
- Zhuo, G.; Ding, Y.; Feng, G.; Yu, L.; Jiang, Y. Analysis of Mitochondrial DNA Sequence Variants in Patients with Polycystic Ovary Syndrome. Arch. Gynecol. Obstet. 2012, 286, 653–659. [Google Scholar] [CrossRef]
- Ding, Y.; Zhuo, G.; Zhang, C. The Mitochondrial TRNA Leu(UUR) A3302G Mutation May Be Associated With Insulin Resistance in Woman With Polycystic Ovary Syndrome. Reprod. Sci. 2016, 23, 228–233. [Google Scholar] [CrossRef]
- Ding, Y.; Zhuo, G.; Zhang, C.; Leng, J. Point Mutation in Mitochondrial TRNA Gene Is Associated with Polycystic Ovary Syndrome and Insulin Resistance. Mol. Med. Rep. 2016, 13, 3169–3172. [Google Scholar] [CrossRef]
- Ding, Y.; Xia, B.-H.; Zhang, C.-J.; Zhuo, G.-C. Mitochondrial TRNALeu(UUR) C3275T, TRNAGln T4363C and TRNALys A8343G Mutations May Be Associated with PCOS and Metabolic Syndrome. Gene 2018, 642, 299–306. [Google Scholar] [CrossRef]
- Ding, Y.; Xia, B.-H.; Zhang, C.-J.; Zhuo, G.-C. Mutations in Mitochondrial TRNA Genes May Be Related to Insulin Resistance in Women with Polycystic Ovary Syndrome. Am. J. Transl. Res. 2017, 9, 2984–2996. [Google Scholar] [PubMed]
- Reddy, T.V.; Govatati, S.; Deenadayal, M.; Sisinthy, S.; Bhanoori, M. Impact of Mitochondrial DNA Copy Number and Displacement Loop Alterations on Polycystic Ovary Syndrome Risk in South Indian Women. Mitochondrion 2019, 44, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Shukla, P.; Mukherjee, S.; Patil, A. Identification of Variants in Mitochondrial D-Loop and OriL Region and Analysis of Mitochondrial DNA Copy Number in Women with Polycystic Ovary Syndrome. DNA Cell Biol. 2020, 39, 1458–1466. [Google Scholar] [CrossRef] [PubMed]
- Finsterer, J.; Zarrouk-Mahjoub, S. Polycystic Ovary Syndrome in Mitochondrial Disorders Due MtDNA or NDNA Variants. Am. J. Transl. Res. 2018, 10, 13–15. [Google Scholar] [PubMed]
- Zhu, Y.; You, J.; Xu, C.; Gu, X. Pathogenicity of the Homoplasmic C3275T, T4363C and A8343G Variant Requires Confirmation. Gene 2019, 680, 97–98. [Google Scholar] [CrossRef]
- Bozdag, G.; Mumusoglu, S.; Zengin, D.; Karabulut, E.; Yildiz, B.O. The Prevalence and Phenotypic Features of Polycystic Ovary Syndrome: A Systematic Review and Meta-Analysis. Hum. Reprod. 2016, 31, 2841–2855. [Google Scholar] [CrossRef]
- Zhao, H.; Zhao, Y.; Li, T.; Li, M.; Li, J.; Li, R.; Liu, P.; Yu, Y.; Qiao, J. Metabolism Alteration in Follicular Niche: The Nexus among Intermediary Metabolism, Mitochondrial Function, and Classic Polycystic Ovary Syndrome. Free Radic. Biol. Med. 2015, 86, 295–307. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yu, Z.; Zhao, S.; Cheng, L.; Man, Y.; Gao, X.; Zhao, H. Oxidative Stress Markers in the Follicular Fluid of Patients with Polycystic Ovary Syndrome Correlate with a Decrease in Embryo Quality. J. Assist. Reprod. Genet. 2021, 38, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Masjedi, F.; Keshtgar, S.; Agah, F.; Karbalaei, N. Association between Sex Steroids and Oxidative Status with Vitamin D Levels in Follicular Fluid of Non-Obese PCOS and Healthy Women. J. Reprod. Infertil. 2019, 20, 132–142. [Google Scholar] [PubMed]
- Ahmed, A.A.; Moselhy, S.S.; Kumosani, T.A.; Huwait, E.A.; Al-Ghamdi, M.A.; Al-Madani, K.A.; AlToukhi, M.H.; Kumosani, A.T. Ultrasonographic and Biochemical Assessments as Early Prediction of Polycystic Ovarian Syndrome in Obese Women. Afr. Health Sci. 2020, 20, 676–681. [Google Scholar] [CrossRef] [PubMed]
- Maryanovich, A.T.; Kormilets, D.Y.; Polyanovsky, A.D. Xenin: The Oldest after Insulin? Mol. Biol. Rep. 2018, 45, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Craig, S.L.; Gault, V.A.; Irwin, N. Emerging Therapeutic Potential for Xenin and Related Peptides in Obesity and Diabetes. Diabetes Metab. Res. Rev. 2018, 34, e3006. [Google Scholar] [CrossRef]
- Guclu, Y.A.; Sahin, E.; Aksit, M. The Relationship between Elevated Serum Xenin and Insulin Resistance in Women with Polycystic Ovary Syndrome: A Case-Control Study. Gynecol. Endocrinol. 2019, 35, 960–964. [Google Scholar] [CrossRef]
- Nasri, F.; Doroudchi, M.; Namavar Jahromi, B.; Gharesi-Fard, B. T Helper Cells Profile and CD4+CD25+Foxp3+Regulatory T Cells in Polycystic Ovary Syndrome. Iran. J. Immunol. 2018, 15. [Google Scholar] [CrossRef]
- Gong, P.; Shi, B.; Wang, J.; Cao, P.; Diao, Z.; Wang, Y.; Hu, Y.; Li, S. Association between Th1/Th2 Immune Imbalance and Obesity in Women with or without Polycystic Ovary Syndrome. Gynecol. Endocrinol. 2018, 34, 709–714. [Google Scholar] [CrossRef]
- Nasri, F.; Zare, M.; Doroudchi, M.; Gharesi-Fard, B. Proteome Analysis of CD4+ T Cells Reveals Differentially Expressed Proteins in Infertile Polycystic Ovary Syndrome Patients. EMIDDT 2020, 20. [Google Scholar] [CrossRef]
- He, S.; Mao, X.; Lei, H.; Dong, B.; Guo, D.; Zheng, B.; Sun, P. Peripheral Blood Inflammatory-Immune Cells as a Predictor of Infertility in Women with Polycystic Ovary Syndrome. JIR 2020, 13, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Fuertes-Martín, R.; Correig, X.; Vallvé, J.-C.; Amigó, N. Title: Human Serum/Plasma Glycoprotein Analysis by 1H-NMR, an Emerging Method of Inflammatory Assessment. J. Clin. Med. 2020, 9, 354. [Google Scholar] [CrossRef] [PubMed]
- Fizelova, M.; Jauhiainen, R.; Kangas, A.J.; Soininen, P.; Ala-Korpela, M.; Kuusisto, J.; Laakso, M.; Stančáková, A. Differential Associations of Inflammatory Markers With Insulin Sensitivity and Secretion: The Prospective METSIM Study. J. Clin. Endocrinol. Metab. 2017, 102, 3600–3609. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, C.; Festa, A.; Hanley, A.J.; Rewers, M.J.; Escalante, A.; Haffner, S.M. Novel Protein Glycan–Derived Markers of Systemic Inflammation and C-Reactive Protein in Relation to Glycemia, Insulin Resistance, and Insulin Secretion. Diabetes Care 2017, 40, 375–382. [Google Scholar] [CrossRef]
- Fuertes-Martín, R.; Moncayo, S.; Insenser, M.; Martínez-García, M.Á.; Luque-Ramírez, M.; Grau, N.A.; Blanchar, X.C.; Escobar-Morreale, H.F. Glycoprotein A and B Height-to-Width Ratios as Obesity-Independent Novel Biomarkers of Low-Grade Chronic Inflammation in Women with Polycystic Ovary Syndrome (PCOS). J. Proteome Res. 2019, 18, 4038–4045. [Google Scholar] [CrossRef]
- Martínez-García, M.Á.; Moncayo, S.; Insenser, M.; Montes-Nieto, R.; Fernández-Durán, E.; Álvarez-Blasco, F.; Luque-Ramírez, M.; Escobar-Morreale, H.F. Postprandial Inflammatory Responses after Oral Glucose, Lipid and Protein Challenges: Influence of Obesity, Sex and Polycystic Ovary Syndrome. Clin. Nutr. 2020, 39, 876–885. [Google Scholar] [CrossRef]
- Ryu, Y.; Kim, S.W.; Kim, Y.Y.; Ku, S.-Y. Animal Models for Human Polycystic Ovary Syndrome (PCOS) Focused on the Use of Indirect Hormonal Perturbations: A Review of the Literature. Int. J. Mol. Sci. 2019, 20, 2720. [Google Scholar] [CrossRef]
- Madiraju, A.K.; Erion, D.M.; Rahimi, Y.; Zhang, X.-M.; Braddock, D.T.; Albright, R.A.; Prigaro, B.J.; Wood, J.L.; Bhanot, S.; MacDonald, M.J.; et al. Metformin Suppresses Gluconeogenesis by Inhibiting Mitochondrial Glycerophosphate Dehydrogenase. Nature 2014, 510, 542–546. [Google Scholar] [CrossRef]
- Di Pietro, M.; Velazquez, C.; Matzkin, M.E.; Frungieri, M.B.; Peña, M.G.; de Zúñiga, I.; Pascuali, N.; Irusta, G.; Bianchi, M.S.; Parborell, F.; et al. Metformin Has a Direct Effect on Ovarian Cells That Is Dependent on Organic Cation Transporters. Mol. Cell. Endocrinol. 2020, 499, 110591. [Google Scholar] [CrossRef]
- Lord, J.M. Metformin in Polycystic Ovary Syndrome: Systematic Review and Meta-Analysis. BMJ 2003, 327, 951. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Li, X.; Liu, P.; Li, K.; Sha, L.; Yang, X.; Zhu, L.; Wang, Z.; Dong, Y.; Zhang, L.; et al. Inulin and Metformin Ameliorate Polycystic Ovary Syndrome via Anti-Inflammation and Modulating Gut Microbiota in Mice. Endocr. J. 2019, 66, 859–870. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Tu, H.; Yao, J.; Le, J.; Jiang, Z.; Tang, Q.; Zhang, R.; Huo, P.; Lei, X. Combined Use of Diane-35 and Metformin Improves the Ovulation in the PCOS Rat Model Possibly via Regulating Glycolysis Pathway. Reprod. Biol. Endocrinol. 2020, 18, 58. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Jiang, X.; Ma, H.; Wang, Y.; Xue, P.; Liu, Y. SIRT1 and Insulin Resistance. J. Diabetes Complicat. 2016, 30, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Tao, X.; Cai, L.; Chen, L.; Ge, S.; Deng, X. Effects of Metformin and Exenatide on Insulin Resistance and AMPKα-SIRT1 Molecular Pathway in PCOS Rats. J. Ovarian Res. 2019, 12, 86. [Google Scholar] [CrossRef] [PubMed]
- Naseri, L.; Khazaei, M.R.; Khazaei, M. Synergic Effect of Bee Pollen and Metformin on Proliferation and Apoptosis of Granulosa Cells: Rat Model of Polycystic Ovary Syndrome. J. Food Biochem. 2021. [Google Scholar] [CrossRef] [PubMed]
- Kupreeva, M.; Diane, A.; Lehner, R.; Watts, R.; Ghosh, M.; Proctor, S.; Vine, D. Effect of Metformin and Flutamide on Insulin, Lipogenic and Androgen-Estrogen Signaling, and Cardiometabolic Risk in a PCOS-Prone Metabolic Syndrome Rodent Model. Am. J. Physiol. Endocrinol. Metab. 2019, 316, E16–E33. [Google Scholar] [CrossRef]
- Valdivielso, P.; Rioja, J.; García-Arias, C.; Sánchez-Chaparro, M.; González-Santos, P. Omega 3 Fatty Acids Induce a Marked Reduction of Apolipoprotein B48 When Added to Fluvastatin in Patients with Type 2 Diabetes and Mixed Hyperlipidemia: A Preliminary Report. Cardiovasc. Diabetol. 2009, 8, 1. [Google Scholar] [CrossRef]
- Furat Rencber, S.; Kurnaz Ozbek, S.; Eraldemır, C.; Sezer, Z.; Kum, T.; Ceylan, S.; Guzel, E. Effect of Resveratrol and Metformin on Ovarian Reserve and Ultrastructure in PCOS: An Experimental Study. J. Ovarian Res. 2018, 11, 55. [Google Scholar] [CrossRef]
- Mojaverrostami, S.; Asghari, N.; Khamisabadi, M.; Heidari Khoei, H. The Role of Melatonin in Polycystic Ovary Syndrome: A Review. IJRM 2019. [Google Scholar] [CrossRef]
- Basheer, M.; Rai, S.; Ghosh, H.; Ahmad Haja, Y. Therapeutic Efficacy of Melatonin against Polycystic Ovary Syndrome (PCOS) Induced by Letrozole in Wistar Rat. Pak. J. Biol. Sci. 2018, 21, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zheng, J.; Cui, N.; Jiang, L.; Zhou, H.; Zhang, D.; Hao, G. Baicalin Ameliorates Polycystic Ovary Syndrome through AMP-Activated Protein Kinase. J. Ovarian Res. 2019, 12, 109. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Lv, L.; Liu, Q.; Song, J. Total Flavonoids Extracted from Nervilia Fordii Function in Polycystic Ovary Syndrome through IL-6 Mediated JAK2/STAT3 Signaling Pathway. Biosci. Rep. 2019, 39, BSR20181380. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.-R.; Xu, X.; Li, X.-L. Berberine Exerts a Protective Effect on Rats with Polycystic Ovary Syndrome by Inhibiting the Inflammatory Response and Cell Apoptosis. Reprod. Biol. Endocrinol. 2021, 19, 3. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Zhu, H.; Zhu, Y.; Hu, X. Guizhi Fuling Wan Reduces Autophagy of Granulosa Cell in Rats with Polycystic Ovary Syndrome via Restoring the PI3K/AKT/MTOR Signaling Pathway. J. Ethnopharmacol. 2021, 270, 113821. [Google Scholar] [CrossRef]
- Wang, C.; Ding, C.; Hua, Z.; Chen, C.; Yu, J. Cangfudaotan Decoction Alleviates Insulin Resistance and Improves Follicular Development in Rats with Polycystic Ovary Syndrome via IGF-1-PI3K/Akt-Bax/Bcl-2 Pathway. Mediat. Inflamm. 2020, 2020, 1–16. [Google Scholar] [CrossRef]
- Zeng, X.; Huang, Q.; Long, S.L.; Zhong, Q.; Mo, Z. Mitochondrial Dysfunction in Polycystic Ovary Syndrome. DNA Cell Biol. 2020, 39, 1401–1409. [Google Scholar] [CrossRef]
- Thomson, R.L.; Spedding, S.; Buckley, J.D. Vitamin D in the Aetiology and Management of Polycystic Ovary Syndrome. Clin. Endocrinol. 2012, 77, 343–350. [Google Scholar] [CrossRef]
- Gao, H.; Li, Y.; Yan, W.; Gao, F. The Effect of Vitamin D Supplementation on Blood Lipids in Patients with Polycystic Ovary Syndrome: A Meta-Analysis of Randomized Controlled Trials. Int. J. Endocrinol. 2021, 2021, 8849688. [Google Scholar] [CrossRef]
- He, C.; Lin, Z.; Robb, S.; Ezeamama, A. Serum Vitamin D Levels and Polycystic Ovary Syndrome: A Systematic Review and Meta-Analysis. Nutrients 2015, 7, 4555–4577. [Google Scholar] [CrossRef]
- Grzesiak, M.; Burzawa, G.; Kurowska, P.; Blaszczyk, K.; Szlaga, A.; Blasiak, A.; Sechman, A.; Rak, A. Altered Vitamin D3 Metabolism in the Ovary and Periovarian Adipose Tissue of Rats with Letrozole-Induced PCOS. Histochem. Cell Biol. 2021, 155, 101–116. [Google Scholar] [CrossRef]
- Safaei, Z.; Bakhshalizadeh, S.; Nasr-Esfahani, M.H.; Akbari Sene, A.; Najafzadeh, V.; Soleimani, M.; Shirazi, R. Vitamin D3 Affects Mitochondrial Biogenesis through Mitogen-activated Protein Kinase in Polycystic Ovary Syndrome Mouse Model. J. Cell. Physiol. 2020, 235, 6113–6126. [Google Scholar] [CrossRef] [PubMed]
- Safaei, Z.; Nasr Esfahani, M.H.; Akbari Sene, A.; Najafzadeh, V.; Soleimani, M.; Shirazi, R.; Bakhshalizadeh, S. Effect of Vitamin D3 on Mitochondrial Biogenesis in Granulosa Cells Derived from Polycystic Ovary Syndrome. Int. J. Fertil. Steril. 2020, 14. [Google Scholar] [CrossRef]
- Azhar, A.; Haider, G.; Naseem, Z.; Farooqui, N.; Farooqui, M.U.; Rehman, R. Morphological Changes in the Experimental Model of Polycystic Ovary Syndrome and Effects of Vitamin D Treatment. J. Obstet. Gynaecol. Res. 2021, jog.14671. [Google Scholar] [CrossRef]
- Bakhshalizadeh, S.; Amidi, F.; Shirazi, R.; Shabani Nashtaei, M. Vitamin D3 Regulates Steroidogenesis in Granulosa Cells through AMP-Activated Protein Kinase (AMPK) Activation in a Mouse Model of Polycystic Ovary Syndrome: Vitamin D3 Regulates Steroidogenesis in PCOS Granulosa Cells via AMPK. Cell Biochem. Funct. 2018, 36, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Kyei, G.; Sobhani, A.; Nekonam, S.; Shabani, M.; Ebrahimi, F.; Qasemi, M.; Salahi, E.; Fardin, A. Assessing the Effect of MitoQ10 and Vitamin D3 on Ovarian Oxidative Stress, Steroidogenesis and Histomorphology in DHEA Induced PCOS Mouse Model. Heliyon 2020, 6, e04279. [Google Scholar] [CrossRef]
- Ding, Y.; Jiang, Z.; Xia, B.; Zhang, L.; Zhang, C.; Leng, J. Mitochondria-Targeted Antioxidant Therapy for an Animal Model of PCOS-IR. Int. J. Mol. Med. 2018. [Google Scholar] [CrossRef]
- Atef, M.M.; Abd-Ellatif, R.N.; Emam, M.N.; Abo El Gheit, R.E.; Amer, A.I.; Hafez, Y.M. Therapeutic Potential of Sodium Selenite in Letrozole Induced Polycystic Ovary Syndrome Rat Model: Targeting Mitochondrial Approach (Selenium in PCOS). Arch. Biochem. Biophys. 2019, 671, 245–254. [Google Scholar] [CrossRef]
- Dantas, W.S.; das Neves, W.; Gil, S.; Barcellos, C.R.G.; Rocha, M.P.; de Sá-Pinto, A.L.; Roschel, H.; Gualano, B. Exercise-Induced Anti-Inflammatory Effects in Overweight/Obese Women with Polycystic Ovary Syndrome. Cytokine 2019, 120, 66–70. [Google Scholar] [CrossRef]
- Samadi, Z.; Bambaeichi, E.; Valiani, M.; Shahshahan, Z. Evaluation of Changes in Levels of Hyperandrogenism, Hirsutism and Menstrual Regulation after a Period of Aquatic High Intensity Interval Training in Women with Polycystic Ovary Syndrome. Int. J. Prev. Med. 2019, 10, 187. [Google Scholar] [CrossRef]
- Shele, G.; Genkil, J.; Speelman, D. A Systematic Review of the Effects of Exercise on Hormones in Women with Polycystic Ovary Syndrome. J. Funct. Morphol. Kinesiol. 2020, 5, 35. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Jiang, F.; Wei, K.; Jiang, Z. Exercise Activates the PI3K-AKT Signal Pathway by Decreasing the Expression of 5α-Reductase Type 1 in PCOS Rats. Sci. Rep. 2018, 8, 7982. [Google Scholar] [CrossRef]
- Stepto, N.K.; Moreno-Asso, A.; McIlvenna, L.C.; Walters, K.A.; Rodgers, R.J. Molecular Mechanisms of Insulin Resistance in Polycystic Ovary Syndrome: Unraveling the Conundrum in Skeletal Muscle? J. Clin. Endocrinol. Metab. 2019, 104, 5372–5381. [Google Scholar] [CrossRef] [PubMed]
- Stepto, N.K.; Hiam, D.; Gibson-Helm, M.; Cassar, S.; Harrison, C.L.; Hutchison, S.K.; Joham, A.E.; Canny, B.J.; Moreno-Asso, A.; Strauss, B.J.; et al. Exercise and Insulin Resistance in PCOS: Muscle Insulin Signalling and Fibrosis. Endocr. Connect. 2020, 9, 346–359. [Google Scholar] [CrossRef]
- Garruti, G.; Depalo, R.; De Angelis, M. Weighing the Impact of Diet and Lifestyle on Female Reproductive Function. CMC 2019, 26, 3584–3592. [Google Scholar] [CrossRef]
- Amirjani, S.; Asemi, Z.; Bazarganipour, F.; Aramesh, S.; Allan, H.; Sayadi, M.; Tabatabaei, M.-S.; Mohamadian, Z.; Zabti, F.; Iranpak, N.; et al. Dietary Intake and Lifestyle Behaviour in Different Phenotypes of Polycystic Ovarian Syndrome: A Case–Control Study. J. Hum. Nutr. Diet. 2019, 32, 413–421. [Google Scholar] [CrossRef]
- Li, J.; Bai, W.; Jiang, B.; Bai, L.; Gu, B.; Yan, S.; Li, F.; Huang, B. Ketogenic Diet in Women with Polycystic Ovary Syndrome and Liver Dysfunction Who Are Obese: A Randomized, Open-label, Parallel-group, Controlled Pilot Trial. J. Obstet. Gynaecol. Res. 2021, 47, 1145–1152. [Google Scholar] [CrossRef]
- Morin-Papunen, L.C.; Koivunen, R.M.; Tomás, C.; Ruokonen, A.; Martikainen, H.K. Decreased Serum Leptin Concentrations during Metformin Therapy in Obese Women with Polycystic Ovary Syndrome. J. Clin. Endocrinol. Metab. 1998, 83, 2566–2568. [Google Scholar] [CrossRef]
- Naderpoor, N.; Shorakae, S.; de Courten, B.; Misso, M.L.; Moran, L.J.; Teede, H.J. Metformin and Lifestyle Modification in Polycystic Ovary Syndrome: Systematic Review and Meta-Analysis. Hum. Reprod. Update 2015, 21, 560–574. [Google Scholar] [CrossRef]
- Pasquali, R.; Gambineri, A.; Biscotti, D.; Vicennati, V.; Gagliardi, L.; Colitta, D.; Fiorini, S.; Cognigni, G.E.; Filicori, M.; Morselli-Labate, A.M. Effect of Long-Term Treatment with Metformin Added to Hypocaloric Diet on Body Composition, Fat Distribution, and Androgen and Insulin Levels in Abdominally Obese Women with and without the Polycystic Ovary Syndrome. J. Clin. Endocrinol. Metab. 2000, 85, 2767–2774. [Google Scholar] [CrossRef]
- Wang, J.; Zhu, L.; Hu, K.; Tang, Y.; Zeng, X.; Liu, J.; Xu, J. Effects of Metformin Treatment on Serum Levels of C-Reactive Protein and Interleukin-6 in Women with Polycystic Ovary Syndrome: A Meta-Analysis: A PRISMA-Compliant Article. Medicine 2017, 96, e8183. [Google Scholar] [CrossRef]
- Chen, Y.; Li, M.; Deng, H.; Wang, S.; Chen, L.; Li, N.; Xu, D.; Wang, Q. Impact of Metformin on C-Reactive Protein Levels in Women with Polycystic Ovary Syndrome: A Meta-Analysis. Oncotarget 2017, 8, 35425–35434. [Google Scholar] [CrossRef]
- Kong, W.; Niu, X.; Zeng, T.; Lu, M.; Chen, L. Impact of Treatment with Metformin on Adipocytokines in Patients with Polycystic Ovary Syndrome: A Meta-Analysis. PLoS ONE 2015, 10, e0140565. [Google Scholar] [CrossRef]
- Gunturiz Albarracín, M.L.; Forero Torres, A.Y. Adiponectin and Leptin Adipocytokines in Metabolic Syndrome: What Is Its Importance? Dubai Diabetes Endocrinol. J. 2020, 26, 93–102. [Google Scholar] [CrossRef]
- Sathyapalan, T.; Hobkirk, J.P.; Javed, Z.; Carroll, S.; Coady, A.-M.; Pemberton, P.; Smith, A.; Cianflone, K.; Atkin, S.L. The Effect of Atorvastatin (and Subsequent Metformin) on Adipose Tissue Acylation-Stimulatory-Protein Concentration and Inflammatory Biomarkers in Overweight/Obese Women with Polycystic Ovary Syndrome. Front. Endocrinol. 2019, 10, 394. [Google Scholar] [CrossRef]
- Wang, T.; Zhang, J.; Hu, M.; Zhang, Y.; Cui, P.; Li, X.; Li, J.; Vestin, E.; Brännström, M.; Shao, L.R.; et al. Differential Expression Patterns of Glycolytic Enzymes and Mitochondria-Dependent Apoptosis in PCOS Patients with Endometrial Hyperplasia, an Early Hallmark of Endometrial Cancer, In Vivo and the Impact of Metformin In Vitro. Int. J. Biol. Sci. 2019, 15, 714–725. [Google Scholar] [CrossRef]
- Pradas, I.; Rovira-Llopis, S.; Naudí, A.; Bañuls, C.; Rocha, M.; Hernandez-Mijares, A.; Pamplona, R.; Victor, V.M.; Jové, M. Metformin Induces Lipid Changes on Sphingolipid Species and Oxidized Lipids in Polycystic Ovary Syndrome Women. Sci. Rep. 2019, 9, 16033. [Google Scholar] [CrossRef]
- Gancheva, S.; Jelenik, T.; Álvarez-Hernández, E.; Roden, M. Interorgan Metabolic Crosstalk in Human Insulin Resistance. Physiol. Rev. 2018, 98, 1371–1415. [Google Scholar] [CrossRef]
- Blauschmidt, S.; Greither, T.; Lampe, K.; Köller, S.; Kaltwaßer, P.; Behre, H.M. Dipeptidyl Peptidase 4 Serum Activity and Concentration Are Increased in Women with Polycystic Ovary Syndrome. Clin. Endocrinol. 2017, 87, 741–747. [Google Scholar] [CrossRef]
- Elmansi, A.M.; Awad, M.E.; Eisa, N.H.; Kondrikov, D.; Hussein, K.A.; Aguilar-Pérez, A.; Herberg, S.; Periyasamy-Thandavan, S.; Fulzele, S.; Hamrick, M.W.; et al. What Doesn’t Kill You Makes You Stranger: Dipeptidyl Peptidase-4 (CD26) Proteolysis Differentially Modulates the Activity of Many Peptide Hormones and Cytokines Generating Novel Cryptic Bioactive Ligands. Pharmacol. Ther. 2019, 198, 90–108. [Google Scholar] [CrossRef]
- Daneshjou, D.; Soleimani Mehranjani, M.; Zadeh Modarres, S.; Shariatzadeh, M.A. Sitagliptin/Metformin: A New Medical Treatment in Polycystic Ovary Syndrome. Trends Endocrinol. Metab. 2020, 31, 890–892. [Google Scholar] [CrossRef]
- Eggleton, J.S.; Jialal, I. Thiazolidinediones. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Xing, C.; Li, C.; He, B. Insulin Sensitizers for Improving the Endocrine and Metabolic Profile in Overweight Women with PCOS. J. Clin. Endocrinol. Metab. 2020, 105, 2950–2963. [Google Scholar] [CrossRef]
- Street, M.E.; Cirillo, F.; Catellani, C.; Dauriz, M.; Lazzeroni, P.; Sartori, C.; Moghetti, P. Current Treatment for Polycystic Ovary Syndrome: Focus on Adolescence. Minerva Pediatr. 2020, 72. [Google Scholar] [CrossRef]
- Tay, C.T.; Joham, A.E.; Hiam, D.S.; Gadalla, M.A.; Pundir, J.; Thangaratinam, S.; Teede, H.J.; Moran, L.J. Pharmacological and Surgical Treatment of Nonreproductive Outcomes in Polycystic Ovary Syndrome: An Overview of Systematic Reviews. Clin. Endocrinol. 2018, 89, 535–553. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.-K.; Wang, L.; Giampietro, A.; Lai, B.; Lee, J.-E.; Ge, K. Distinct Roles of Transcription Factors KLF4, Krox20, and Peroxisome Proliferator-Activated Receptor γ in Adipogenesis. Mol. Cell. Biol. 2017, 37, e00554-16. [Google Scholar] [CrossRef] [PubMed]
- Hafiane, A.; Gasbarrino, K.; Daskalopoulou, S.S. The Role of Adiponectin in Cholesterol Efflux and HDL Biogenesis and Metabolism. Metabolism 2019, 100, 153953. [Google Scholar] [CrossRef]
- Song, E.-K.; Lee, Y.-R.; Kim, Y.-R.; Yeom, J.-H.; Yoo, C.-H.; Kim, H.-K.; Park, H.-M.; Kang, H.-S.; Kim, J.-S.; Kim, U.-H.; et al. NAADP Mediates Insulin-Stimulated Glucose Uptake and Insulin Sensitization by PPARγ in Adipocytes. Cell Rep. 2012, 2, 1607–1619. [Google Scholar] [CrossRef]
- Jamilian, M.; Shojaei, A.; Samimi, M.; Afshar Ebrahimi, F.; Aghadavod, E.; Karamali, M.; Taghizadeh, M.; Jamilian, H.; Alaeinasab, S.; Jafarnejad, S.; et al. The Effects of Omega-3 and Vitamin E Co-Supplementation on Parameters of Mental Health and Gene Expression Related to Insulin and Inflammation in Subjects with Polycystic Ovary Syndrome. J. Affect. Disord. 2018, 229, 41–47. [Google Scholar] [CrossRef]
- Jamilian, M.; Foroozanfard, F.; Kavossian, E.; Kia, M.; Aghadavod, E.; Amirani, E.; Asemi, Z. Effects of Chromium and Carnitine Co-Supplementation on Body Weight and Metabolic Profiles in Overweight and Obese Women with Polycystic Ovary Syndrome: A Randomized, Double-Blind, Placebo-Controlled Trial. Biol. Trace Elem. Res. 2020, 193, 334–341. [Google Scholar] [CrossRef]
- Shabani, A.; Foroozanfard, F.; Kavossian, E.; Aghadavod, E.; Ostadmohammadi, V.; Reiter, R.J.; Eftekhar, T.; Asemi, Z. Effects of Melatonin Administration on Mental Health Parameters, Metabolic and Genetic Profiles in Women with Polycystic Ovary Syndrome: A Randomized, Double-Blind, Placebo-Controlled Trial. J. Affect. Disord. 2019, 250, 51–56. [Google Scholar] [CrossRef]
- Jamilian, M.; Foroozanfard, F.; Kavossian, E.; Aghadavod, E.; Shafabakhsh, R.; Hoseini, A.; Asemi, Z. Effects of Curcumin on Body Weight, Glycemic Control and Serum Lipids in Women with Polycystic Ovary Syndrome: A Randomized, Double-Blind, Placebo-Controlled Trial. Clin. Nutr. ESPEN 2020, 36, 128–133. [Google Scholar] [CrossRef]
- Olcese, J.M. Melatonin and Female Reproduction: An Expanding Universe. Front. Endocrinol. 2020, 11, 85. [Google Scholar] [CrossRef] [PubMed]
- Yi, S.; Zheng, B.; Zhu, Y.; Cai, Y.; Sun, H.; Zhou, J. Melatonin Ameliorates Excessive PINK1/Parkin-Mediated Mitophagy by Enhancing SIRT1 Expression in Granulosa Cells of PCOS. Am. J. Physiol. Endocrinol. Metab. 2020, 319, E91–E101. [Google Scholar] [CrossRef]
- Homburg, R.; Eshel, A.; Abdalla, H.I.; Jacobs, H.S. Growth Hormone Facilitates Ovulation Induction by Gonadotrophins. Clin. Endocrinol. 1988, 29, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Luo, S.; Fan, P.; Jin, S.; Zhu, H.; Deng, T.; Quan, Y.; Huang, W. Growth Hormone Alleviates Oxidative Stress and Improves Oocyte Quality in Chinese Women with Polycystic Ovary Syndrome: A Randomized Controlled Trial. Sci. Rep. 2020, 10, 18769. [Google Scholar] [CrossRef]
- Li, T.; Mo, H.; Chen, W.; Li, L.; Xiao, Y.; Zhang, J.; Li, X.; Lu, Y. Role of the PI3K-Akt Signaling Pathway in the Pathogenesis of Polycystic Ovary Syndrome. Reprod. Sci. 2017, 24, 646–655. [Google Scholar] [CrossRef]
- Gong, Y.; Luo, S.; Fan, P.; Zhu, H.; Li, Y.; Huang, W. Growth Hormone Activates PI3K/Akt Signaling and Inhibits ROS Accumulation and Apoptosis in Granulosa Cells of Patients with Polycystic Ovary Syndrome. Reprod. Biol. Endocrinol. 2020, 18, 121. [Google Scholar] [CrossRef] [PubMed]
- Monastra, G.; Unfer, V.; Harrath, A.H.; Bizzarri, M. Combining Treatment with Myo-Inositol and D -Chiro-Inositol (40:1) Is Effective in Restoring Ovary Function and Metabolic Balance in PCOS Patients. Gynecol. Endocrinol. 2017, 33, 1–9. [Google Scholar] [CrossRef]
- Lei, W.; Gao, Y.; Hu, S.; Liu, D.; Chen, Q. Effects of Inositol and Alpha Lipoic Acid Combination for Polycystic Ovary Syndrome: A Protocol for Systematic Review and Meta-Analysis. Medicine 2020, 99, e20696. [Google Scholar] [CrossRef]
- Wang, T.; Sha, L.; Li, Y.; Zhu, L.; Wang, Z.; Li, K.; Lu, H.; Bao, T.; Guo, L.; Zhang, X.; et al. Dietary α-Linolenic Acid-Rich Flaxseed Oil Exerts Beneficial Effects on Polycystic Ovary Syndrome Through Sex Steroid Hormones—Microbiota—Inflammation Axis in Rats. Front. Endocrinol. 2020, 11, 284. [Google Scholar] [CrossRef]
- Cabrera-Cruz, H.; Oróstica, L.; Plaza-Parrochia, F.; Torres-Pinto, I.; Romero, C.; Vega, M. The Insulin-Sensitizing Mechanism of Myo-Inositol Is Associated with AMPK Activation and GLUT-4 Expression in Human Endometrial Cells Exposed to a PCOS Environment. Am. J. Physiol. Endocrinol. Metab. 2020, 318, E237–E248. [Google Scholar] [CrossRef]
- Canosa, S.; Paschero, C.; Carosso, A.; Leoncini, S.; Mercaldo, N.; Gennarelli, G.; Benedetto, C.; Revelli, A. Effect of a Combination of Myo-Inositol, Alpha-Lipoic Acid, and Folic Acid on Oocyte Morphology and Embryo Morphokinetics in Non-PCOS Overweight/Obese Patients Undergoing IVF: A Pilot, Prospective, Randomized Study. J. Clin. Med. 2020, 9, 2949. [Google Scholar] [CrossRef] [PubMed]
- Colak, E.; Ozcimen, E.E.; Tohma, Y.A.; Ceran, M.U. May Myo-inositol and d -chiro-inositol (40:1) Treatment Be a Good Option on Normal-weighted Polycystic Ovary Syndrome Patients without Insulin Resistance? J. Obstet. Gynaecol. Res. 2020, 46, 2605–2611. [Google Scholar] [CrossRef] [PubMed]
- Artini, P.G.; Obino, M.E.R.; Micelli, E.; Malacarne, E.; Vacca, C.; Papini, F.; Cela, V. Effect of D-Chiro-Inositol and Alpha-Lipoic Acid Combination on COH Outcomes in Overweight/Obese PCOS Women. Gynecol. Endocrinol. 2020, 36, 755–759. [Google Scholar] [CrossRef] [PubMed]
- Fruzzetti, F.; Benelli, E.; Fidecicchi, T.; Tonacchera, M. Clinical and Metabolic Effects of Alpha-Lipoic Acid Associated with Two Different Doses of Myo-Inositol in Women with Polycystic Ovary Syndrome. Int. J. Endocrinol. 2020, 2020, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Merviel, P.; James, P.; Bouée, S.; Le Guillou, M.; Rince, C.; Nachtergaele, C.; Kerlan, V. Impact of Myo-Inositol Treatment in Women with Polycystic Ovary Syndrome in Assisted Reproductive Technologies. Reprod. Health 2021, 18, 13. [Google Scholar] [CrossRef] [PubMed]
- Genazzani, A. Inositols: Reflections on How to Choose the Appropriate One for PCOS. Gynecol. Endocrinol. 2020, 36, 1045–1046. [Google Scholar] [CrossRef]
- Kamenov, Z.; Gateva, A. Inositols in PCOS. Molecules 2020, 25, 5566. [Google Scholar] [CrossRef]
- Kuryłowicz, A.; Koźniewski, K. Anti-Inflammatory Strategies Targeting Metaflammation in Type 2 Diabetes. Molecules 2020, 25, 2224. [Google Scholar] [CrossRef]
- González, F.; Mather, K.J.; Considine, R.V.; Abdelhadi, O.A.; Acton, A.J. Salicylate Administration Suppresses the Inflammatory Response to Nutrients and Improves Ovarian Function in Polycystic Ovary Syndrome. Am. J. Physiol. Endocrinol. Metab. 2020, 319, E744–E752. [Google Scholar] [CrossRef]
- Salastekar, N.; Desai, T.; Hauser, T.; Schaefer, E.J.; Fowler, K.; Joseph, S.; Shoelson, S.E.; Goldfine, A.B. TINSAL-CVD Study Team. Salsalate Improves Glycaemia in Overweight Persons with Diabetes Risk Factors of Stable Statin-Treated Cardiovascular Disease: A 30-Month Randomized Placebo-Controlled Trial. Diabetes Obes. Metab. 2017, 19, 1458–1462. [Google Scholar] [CrossRef]
- Masjedi, F.; Keshtgar, S.; Zal, F.; Talaei-Khozani, T.; Sameti, S.; Fallahi, S.; Kazeroni, M. Effects of Vitamin D on Steroidogenesis, Reactive Oxygen Species Production, and Enzymatic Antioxidant Defense in Human Granulosa Cells of Normal and Polycystic Ovaries. J. Steroid Biochem. Mol. Biol. 2020, 197, 105521. [Google Scholar] [CrossRef]
- Uyanikoglu, H.; Sabuncu, T.; Dursun, H.; Sezen, H.; Aksoy, N. Circulating Levels of Apoptotic Markers and Oxidative Stress Parameters in Women with Polycystic Ovary Syndrome: A Case-Controlled Descriptive Study. Biomarkers 2017, 22, 643–647. [Google Scholar] [CrossRef] [PubMed]
- Brannan, C.I.; Dees, E.C.; Ingram, R.S.; Tilghman, S.M. The Product of the H19 Gene May Function as an RNA. Mol. Cell. Biol. 1990, 10, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Hüttenhofer, A.; Schattner, P.; Polacek, N. Non-Coding RNAs: Hope or Hype? Trends Genet. 2005, 21, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Flippot, R.; Beinse, G.; Boilève, A.; Vibert, J.; Malouf, G.G. Long Non-Coding RNAs in Genitourinary Malignancies: A Whole New World. Nat. Rev. Urol. 2019, 16, 484–504. [Google Scholar] [CrossRef] [PubMed]
- Statello, L.; Guo, C.-J.; Chen, L.-L.; Huarte, M. Gene Regulation by Long Non-Coding RNAs and Its Biological Functions. Nat. Rev. Mol. Cell Biol. 2021, 22, 96–118. [Google Scholar] [CrossRef] [PubMed]
- Xing, J.; Liu, H.; Jiang, W.; Wang, L. LncRNA-Encoded Peptide: Functions and Predicting Methods. Front. Oncol. 2020, 10, 622294. [Google Scholar] [CrossRef] [PubMed]
- Zeni, P.F.; Mraz, M. LncRNAs in Adaptive Immunity: Role in Physiological and Pathological Conditions. RNA Biol. 2020, 1–14. [Google Scholar] [CrossRef]
- Jiao, J.; Shi, B.; Wang, T.; Fang, Y.; Cao, T.; Zhou, Y.; Wang, X.; Li, D. Characterization of Long Non-Coding RNA and Messenger RNA Profiles in Follicular Fluid from Mature and Immature Ovarian Follicles of Healthy Women and Women with Polycystic Ovary Syndrome. Hum. Reprod. 2018, 33, 1735–1748. [Google Scholar] [CrossRef]
- Yang, X.; Xie, Z.; Lei, X.; Gan, R. Long Non-coding RNA GAS5 in Human Cancer (Review). Oncol. Lett. 2020, 20, 2587–2594. [Google Scholar] [CrossRef] [PubMed]
- Fawzy, M.S.; Abdelghany, A.A.; Toraih, E.A.; Mohamed, A.M. Circulating Long Noncoding RNAs H19 and GAS5 Are Associated with Type 2 Diabetes but Not with Diabetic Retinopathy: A Preliminary Study. Bosn. J. Basic Med. Sci. 2020, 20, 365–371. [Google Scholar] [CrossRef]
- Lin, H.; Xing, W.; Li, Y.; Xie, Y.; Tang, X.; Zhang, Q. Downregulation of Serum Long Noncoding RNA GAS5 May Contribute to Insulin Resistance in PCOS Patients. Gynecol. Endocrinol. 2018, 34, 784–788. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Yue, S.; Jiang, Y.; Mao, Y.; Zhao, Z.; Liu, X.; Zhang, X.; Pei, D.; Li, Y. LncRNA GAS5 Is Upregulated in Polycystic Ovary Syndrome and Regulates Cell Apoptosis and the Expression of IL-6. J. Ovarian Res. 2020, 13, 145. [Google Scholar] [CrossRef] [PubMed]
- Borthakur, A.; Prabhu, Y.D.; Valsala Gopalakrishnan, A. Role of IL-6 Signalling in Polycystic Ovarian Syndrome Associated Inflammation. J. Reprod. Immunol. 2020, 141, 103155. [Google Scholar] [CrossRef]
- Li, L.; Zhu, J.; Ye, F.; Duan, Z.; Zhou, J.; Huang, Z.; Wang, L. Upregulation of the LncRNA SRLR in Polycystic Ovary Syndrome Regulates Cell Apoptosis and IL-6 Expression. Cell Biochem. Funct. 2020, 38, 880–885. [Google Scholar] [CrossRef]
- Xu, Z.; Yang, F.; Wei, D.; Liu, B.; Chen, C.; Bao, Y.; Wu, Z.; Wu, D.; Tan, H.; Li, J.; et al. Long Noncoding RNA-SRLR Elicits Intrinsic Sorafenib Resistance via Evoking IL-6/STAT3 Axis in Renal Cell Carcinoma. Oncogene 2017, 36, 1965–1977. [Google Scholar] [CrossRef]
- Ma, J.; Qin, L.; Li, X. Role of STAT3 Signaling Pathway in Breast Cancer. Cell Commun. Signal. 2020, 18, 33. [Google Scholar] [CrossRef]
- Zhuang, Z.; Pan, X.; Zhao, K.; Gao, W.; Liu, J.; Deng, T.; Qin, W. The Effect of Interleukin-6 (IL-6), Interleukin-11 (IL-11), Signal Transducer and Activator of Transcription 3 (STAT3), and AKT Signaling on Adipocyte Proliferation in a Rat Model of Polycystic Ovary Syndrome. Med. Sci. Monit. 2019, 25, 7218–7227. [Google Scholar] [CrossRef]
- Liu, Y.; He, A.; Liu, B.; Huang, Z.; Mei, H. Potential Role of LncRNA H19 as a Cancer Biomarker in Human Cancers Detection and Diagnosis: A Pooled Analysis Based on 1585 Subjects. BioMed Res. Int. 2019, 2019, 9056458. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wei, J.; Hei, J.; Ren, Y.; Li, H. Long Non-Coding RNA H19 Regulates Proliferation of Ovarian Granulosa Cells via STAT3 in Polycystic Ovarian Syndrome. Arch. Med. Sci. 2019, 17. [Google Scholar] [CrossRef]
- Sheng, L.; Ye, L.; Zhang, D.; Cawthorn, W.P.; Xu, B. New Insights into the Long Non-Coding RNA SRA: Physiological Functions and Mechanisms of Action. Front. Med. 2018, 5, 244. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhao, W.; Wang, H.; Chen, C.; Zhou, D.; Li, S.; Zhang, X.; Zhao, H.; Zhou, D.; Chen, B. Silencing of LncRNA Steroid Receptor RNA Activator Attenuates Polycystic Ovary Syndrome in Mice. Biochimie 2019, 157, 48–56. [Google Scholar] [CrossRef] [PubMed]
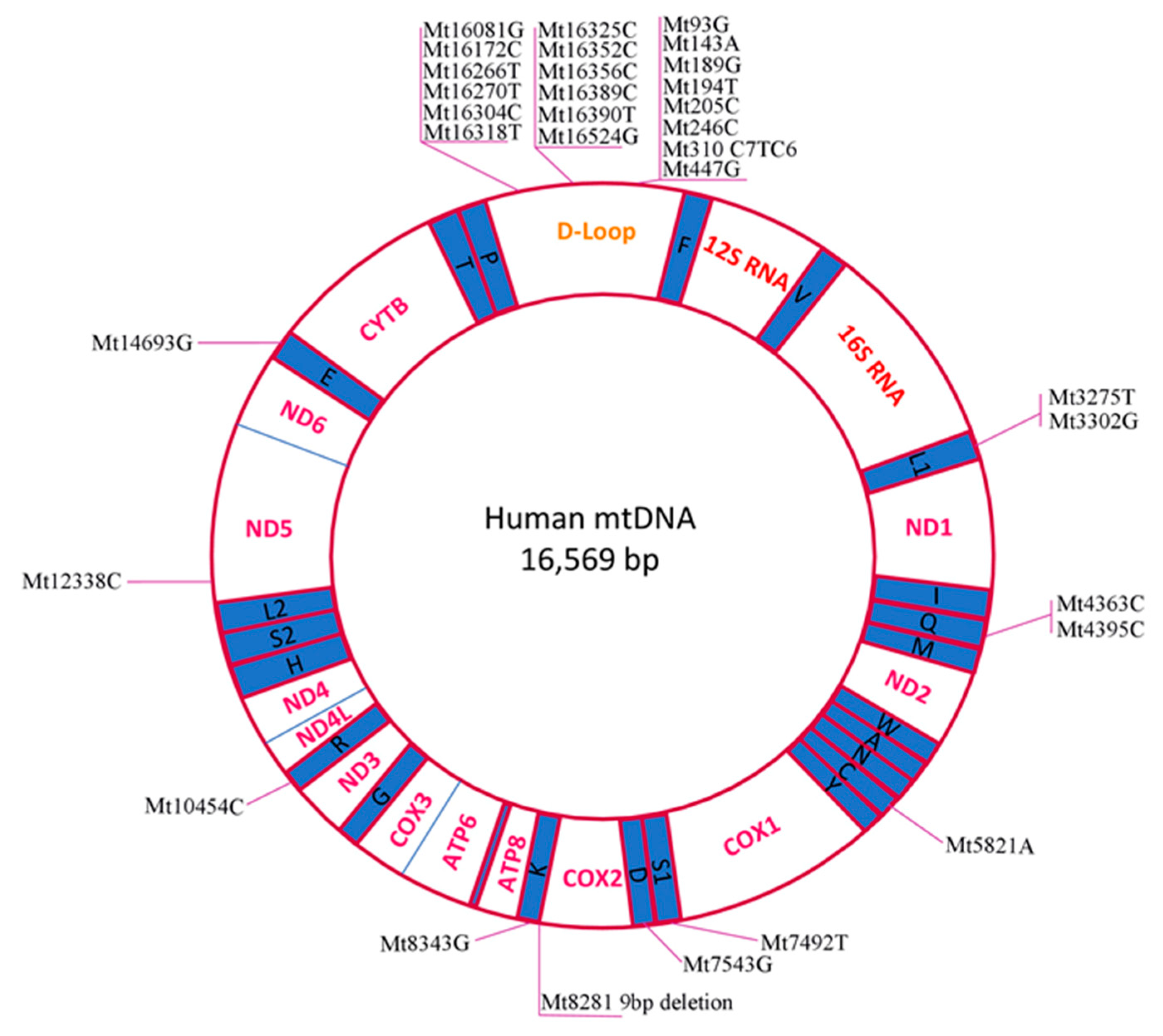
| Mutation | Gene | Other Notes | Year | |
|---|---|---|---|---|
| Mt8281 9 bp deletion | CO2/tRNALys | Chinese PCOS patients | 2010 | [55] |
| 2012 | [56] | |||
| Mt3302G (A-to-G) | tRNALeu(UUR) | One Chinese Han family case; T2DM and IR in family history, confirmed PCOS-IR in the proband | 2016 | [57] |
| Mt12338C (T-to-C)(M→T) | ND5 | One Chinese Han patient, PCOS-IR; double mutation | 2016 | [58] |
| Mt7492T (C-to-T) | tRNASer(UCN) | |||
| Mt3275T (C-to-T) | tRNALeu(UUR) | One Chinese family, PCOS-MetS diagnosed; hypertension and T2DM in family history; triple mutation | 2018 | [59] |
| Mt4363C (T-to-C) | tRNAGln | |||
| Mt8343G (A-to-G) | tRNALys | |||
| Mt3302G (A-to-G) | tRNALeu(UUR) | Chinese PCOS patients with IR | 2017 | [60] |
| Mt3275A (C-to-A) | ||||
| Mt4363C (T-to-C) | tRNAGln | |||
| Mt4395C (T-to-C) | ||||
| Mt5821A (G-to-A) | tRNACys | |||
| Mt7492T (C-to-T) | tRNASer(UCN) | |||
| Mt7543G (A-to-G) | tRNAAsp | |||
| Mt8343G (A-to-G) | tRNALys | |||
| Mt10454C (T-to-C) | tRNAArg | |||
| Mt14693G (A-to-G) | tRNAGlu | |||
| Mt189G (A-to-G) | H-origin | South Indian PCOS patients; only D-loop was analysed | 2017 | [61] |
| Mt310 (C7TC6-to-C8/C9/T/C6) | D-loop poly-C tract; TFAM binding site | |||
| Mt93G (A-to-G) | D-loop | Indian PCOS patients; only D-loop was analysed * | 2020 | [62] |
| Mt143A (G-to-A) | ||||
| Mt194T (C-to-T) | ||||
| Mt205C (G-to-C) | ||||
| Mt246C (T-to-C) | ||||
| Mt447G (C-to-G) | ||||
| Mt16081G (A-to-G) | ||||
| Mt16172C (T-to-C) | ||||
| Mt16266T (C-to-T) | ||||
| Mt16270T (C-to-T) | ||||
| Mt16304C (T-to-C) | ||||
| Mt16318T (A-to-T) | ||||
| Mt16325C (T-to-C) | ||||
| Mt16352C (T-to-C) | ||||
| Mt16356C (T-to-C) | ||||
| Mt16389C (G-to-C) | ||||
| Mt16390T (G-to-T) | ||||
| Mt16524G (A-to-G) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dabravolski, S.A.; Nikiforov, N.G.; Eid, A.H.; Nedosugova, L.V.; Starodubova, A.V.; Popkova, T.V.; Bezsonov, E.E.; Orekhov, A.N. Mitochondrial Dysfunction and Chronic Inflammation in Polycystic Ovary Syndrome. Int. J. Mol. Sci. 2021, 22, 3923. https://doi.org/10.3390/ijms22083923
Dabravolski SA, Nikiforov NG, Eid AH, Nedosugova LV, Starodubova AV, Popkova TV, Bezsonov EE, Orekhov AN. Mitochondrial Dysfunction and Chronic Inflammation in Polycystic Ovary Syndrome. International Journal of Molecular Sciences. 2021; 22(8):3923. https://doi.org/10.3390/ijms22083923
Chicago/Turabian StyleDabravolski, Siarhei A., Nikita G. Nikiforov, Ali H. Eid, Ludmila V. Nedosugova, Antonina V. Starodubova, Tatyana V. Popkova, Evgeny E. Bezsonov, and Alexander N. Orekhov. 2021. "Mitochondrial Dysfunction and Chronic Inflammation in Polycystic Ovary Syndrome" International Journal of Molecular Sciences 22, no. 8: 3923. https://doi.org/10.3390/ijms22083923
APA StyleDabravolski, S. A., Nikiforov, N. G., Eid, A. H., Nedosugova, L. V., Starodubova, A. V., Popkova, T. V., Bezsonov, E. E., & Orekhov, A. N. (2021). Mitochondrial Dysfunction and Chronic Inflammation in Polycystic Ovary Syndrome. International Journal of Molecular Sciences, 22(8), 3923. https://doi.org/10.3390/ijms22083923








