Future Perspectives in Detecting EGFR and ALK Gene Alterations in Liquid Biopsies of Patients with NSCLC
Abstract
1. Introduction
2. Alterations in EGFR and ALK Genes as Predictive Biomarkers in NSCLC
2.1. Mutations in EGFR as Predictive Biomarkers and EGFR TKIs in NSCLC
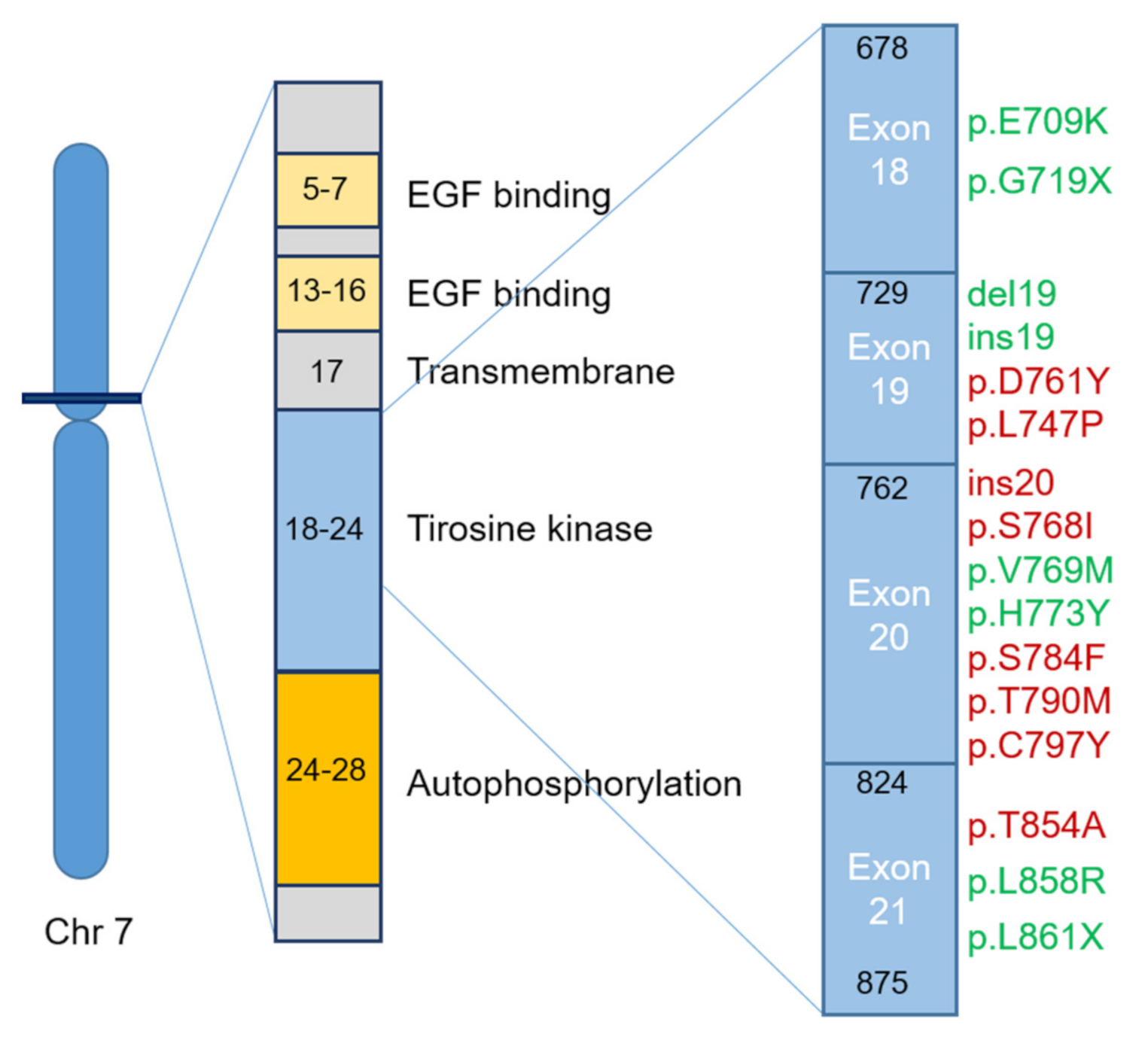
- Class I—in-frame deletions in exon 19. The most frequent mutations affect amino acids from L747 to E749 codons (Leu-Arg-Glu fragment, commonly known as LRE fragment)−p.delE746-A750 followed by p.delL747-P753insS, p.delL747-T751 and p.delL747-A750insP [15]. However, mutations can occur in all the amino acids encoded by exon 19, from E746 to D761. The different mutation types exhibit different therapeutic responses regarding the first-generation EGFR TKIs, being more effective in p.delE746 than in p.delL747 [15].
- Class II—single nucleotide substitutions can occur in exons 18, 20 and 21. The most frequent mutations in exon 18 of EGFR in NSCLC are p.E709X and p.G719X (X can be replaced by A, S, C and D) [16,17]. These mutations can be present in combination with other additional point mutations, such as p.S768I (exon 20) and p.L819Q (exon 21), reducing the sensitivity to the second-generation TKIs, specifically, afatinib and neratinib [18]. Frequently, in EGFR exon 20, a second mutation may occur—p.T790M, causing more than 50% of resistance acquisition to the first- and second-generation EGFR TKI drugs in NSCLC [19,20,21]. This last mutation increases the affinity to ATP and decreases the efficacy of ATP competitors, such as gefitinib and erlotinib [18,22]. In about 20–40% of NSCLCs, a third mutation, p.C797S, occurs in exon 20, resulting in resistance to the third-generation EGFR TKIs [23]. One of the most frequently observed EGFR mutation in NSCLC occurs on exon 21—p.L858R [24]. The p.L861Q mutation is also observed, but in a smaller number of cases, conferring sensitivity to EGFR TKIs [25,26].
2.2. Alterations in ALK as Predictive Biomarkers and ALK TKIs in NSCLC
3. State of the Art on the Analysis of EGFR and ALK Mutations in Liquid Biopsies—Can Liquid Biopsies Be Used as a Routine Clinical Practice in NSCLC Patients in the Near Future?
- Therapy monitoring—the use of liquid biopsies can be essential in the detection of early cancer recurrence (before radiographic or clinical detection), allowing an early treatment change. The detection of new mutations (not present in the primary tumor) is also possible, guiding the second-line therapy choice. Due to the non-invasive character of these biopsies, the patient follow-up along the treatment allows the detection of new mutations that can lead to resistance (e.g., EGFR p.T790M) [1,2]. Also, ctDNA presence during treatment revealed a significant increase in the progression-free survival from 55 to 295 days (non-ctDNA vs. ctDNA) [59];
- Cancer screening—the use of liquid biopsies has a potential to detect tumor presence before it can be clinically identifiable, reducing cancer morbidity and mortality. However, some problems related with its use in early cancer detection, such as overdiagnosis and high rate of false positives, have to be overcome [1,2].
EGFR and ALK Alteration Detection in Liquid Biopsies—The Input of NGS in ctDNA Analysis
- Foundation-One Liquid CDx (Foundation Medicine, Cambridge, MA, USA)—designed to detect alterations, such as point mutations, insertions and deletions, in 311 genes, rearrangements in four genes, and copy number variations in three genes using a targeted high-throughput hybridization-based capture technology [5];
- Guardant360 CDx (Guardant Health, Redwood City, CA, USA)—hybrid capture-based deep sequencing of defined regions in 74 genes reducing the number of false positives through the use of individually tagged cfDNA libraries [100].
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mattox, A.K.; Bettegowda, C.; Zhou, S.; Papadopoulos, N.; Kinzler, K.W.; Vogelstein, B. Applications of liquid biopsies for cancer. Sci. Transl. Med. 2019, 11, eaay1984. [Google Scholar] [CrossRef]
- Tie, J.; Wang, Y.; Tomasetti, C.; Li, L.; Springer, S.; Kinde, I.; Silliman, N.; Tacey, M.; Wong, H.-L.; Christie, M. Circulating tumor DNA analysis detects minimal residual disease and predicts recurrence in patients with stage II colon cancer. Sci. Transl. Med. 2016, 8, 346ra392. [Google Scholar] [CrossRef]
- Palmirotta, R.; Lovero, D.; Cafforio, P.; Felici, C.; Mannavola, F.; Pelle, E.; Quaresmini, D.; Tucci, M.; Silvestris, F. Liquid biopsy of cancer: A multimodal diagnostic tool in clinical oncology. Ther. Adv. Med. Oncol. 2018, 10. [Google Scholar] [CrossRef]
- Herbreteau, G.; Vallee, A.; Charpentier, S.; Normanno, N.; Hofman, P.; Denis, M.G. Circulating free tumor DNA in non-small cell lung cancer (NSCLC): Clinical application and future perspectives. J. Thorac. Dis. 2019, 11, S113–S126. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.; Hur, J.Y.; Choi, Y.L.; Lee, C.H.; Kim, W.S. Current status and future perspectives of liquid biopsy in non-small cell lung cancer. J. Pathol. Transl. Med. 2020, 54, 204–212. [Google Scholar] [CrossRef]
- Martin, P.; Kelly, C.M.; Carney, D. Epidermal growth factor receptor-targeted agents for lung cancer. Cancer Control 2006, 13, 129–140. [Google Scholar] [CrossRef]
- Bethune, G.; Bethune, D.; Ridgway, N.; Xu, Z. Epidermal growth factor receptor (EGFR) in lung cancer: An overview and update. J. Thorac. Dis. 2010, 2, 48. [Google Scholar] [PubMed]
- Leal, L.F.; de Paula, F.E.; De Marchi, P.; de Souza Viana, L.; Pinto, G.D.J.; Carlos, C.D.; Berardinelli, G.N.; Miziara, J.E.; da Silva, C.M.; Silva, E.C.A. Mutational profile of Brazilian lung adenocarcinoma unveils association of EGFR mutations with high Asian ancestry and independent prognostic role of KRAS mutations. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.V.; Bell, D.W.; Settleman, J.; Haber, D.A. Epidermal growth factor receptor mutations in lung cancer. Nat. Rev. Cancer 2007, 7, 169–181. [Google Scholar] [CrossRef]
- Mok, T.; Wu, Y.-L.; Lee, J.S.; Yu, C.-J.; Sriuranpong, V.; Sandoval-Tan, J.; Ladrera, G.; Thongprasert, S.; Srimuninnimit, V.; Liao, M. Detection and dynamic changes of EGFR mutations from circulating tumor DNA as a predictor of survival outcomes in NSCLC patients treated with first-line intercalated erlotinib and chemotherapy. Clin. Cancer Res. 2015, 21, 3196–3203. [Google Scholar] [CrossRef]
- Huang, L.; Fu, L. Mechanisms of resistance to EGFR tyrosine kinase inhibitors. Acta Pharm. Sin. B 2015, 5, 390–401. [Google Scholar] [CrossRef]
- Sharma, S.V.; Fischbach, M.A.; Haber, D.A.; Settleman, J. “Oncogenic shock”: Explaining oncogene addiction through differential signal attenuation. Clin. Cancer Res. 2006, 12, 4392s–4395s. [Google Scholar] [CrossRef]
- Mosesson, Y.; Yarden, Y. Oncogenic growth factor receptors: Implications for signal transduction therapy. Semin. Cancer Biol. 2004, 14, 262–270. [Google Scholar] [CrossRef]
- Gazdar, A. Activating and resistance mutations of EGFR in non-small-cell lung cancer: Role in clinical response to EGFR tyrosine kinase inhibitors. Oncogene 2009, 28, S24–S31. [Google Scholar] [CrossRef]
- Su, J.; Zhong, W.-Z.; Zhang, X.; Huang, Y.; Yan, H.; Yang, J.; Dong, Z.; Xie, Z.; Zhou, Q.; Huang, X.; et al. Molecular characteristics and clinical outcomes of EGFR exon 19 indel subtypes to EGFR TKIs in NSCLC patients. Oncotarget 2017, 8. [Google Scholar] [CrossRef]
- Harrison, P.T.; Vyse, S.; Huang, P.H. Rare epidermal growth factor receptor (EGFR) mutations in non-small cell lung cancer. Semin. Cancer Biol. 2020, 61, 167–179. [Google Scholar] [CrossRef]
- Haghgoo, S.M.; Allameh, A.; Mortaz, E.; Garssen, J.; Folkerts, G.; Barnes, P.J.; Adcock, I.M. Pharmacogenomics and targeted therapy of cancer: Focusing on non-small cell lung cancer. Eur. J. Pharmacol. 2015, 754, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Togashi, Y.; Yatabe, Y.; Mizuuchi, H.; Jangchul, P.; Kondo, C.; Shimoji, M.; Sato, K.; Suda, K.; Tomizawa, K. EGFR exon 18 mutations in lung cancer: Molecular predictors of augmented sensitivity to afatinib or neratinib as compared with first-or third-generation TKIs. Clin. Cancer Res. 2015, 21, 5305–5313. [Google Scholar] [CrossRef]
- Akula, S.; Kamasani, S.; Sivan, S.K.; Manga, V.; Vudem, D.R.; Kancha, R.K. Computational analysis of epidermal growth factor receptor mutations predicts differential drug sensitivity profiles toward kinase inhibitors. J. Thorac. Oncol. 2018, 13, 721–726. [Google Scholar] [CrossRef]
- Kosaka, T.; Yatabe, Y.; Endoh, H.; Yoshida, K.; Hida, T.; Tsuboi, M.; Tada, H.; Kuwano, H.; Mitsudomi, T. Analysis of epidermal growth factor receptor gene mutation in patients with non–small cell lung cancer and acquired resistance to gefitinib. Clin. Cancer Res. 2006, 12, 5764–5769. [Google Scholar] [CrossRef]
- Li, W.; Qiu, T.; Guo, L.; Ling, Y.; Gao, Y.; Ying, J.; He, J. Primary and acquired EGFR T790M-mutant NSCLC patients identified by routine mutation testing show different characteristics but may both respond to osimertinib treatment. Cancer Lett. 2018, 423, 9–15. [Google Scholar] [CrossRef]
- Yun, C.-H.; Mengwasser, K.E.; Toms, A.V.; Woo, M.S.; Greulich, H.; Wong, K.-K.; Meyerson, M.; Eck, M.J. The T790M mutation in EGFR kinase causes drug resistance by increasing the affinity for ATP. Proc. Natl. Acad. Sci. USA 2008, 105, 2070–2075. [Google Scholar] [CrossRef] [PubMed]
- Kohsaka, S.; Petronczki, M.; Solca, F.; Maemondo, M. Tumor clonality and resistance mechanisms in EGFR mutation-positive non-small-cell lung cancer: Implications for therapeutic sequencing. Future Oncol. 2019, 15, 637–652. [Google Scholar] [CrossRef] [PubMed]
- Dogan, S.; Shen, R.; Ang, D.C.; Johnson, M.L.; D’Angelo, S.P.; Paik, P.K.; Brzostowski, E.B.; Riely, G.J.; Kris, M.G.; Zakowski, M.F. Molecular epidemiology of EGFR and KRAS mutations in 3026 lung adenocarcinomas: Higher susceptibility of women to smoking-related KRAS-mutant cancers. Clin. Cancer Res. 2012, 18, 6169–6177. [Google Scholar] [CrossRef]
- da Cunha Santos, G.; Shepherd, F.A.; Tsao, M.S. EGFR mutations and lung cancer. Annu. Rev. Pathol. Mech. Dis. 2011, 6, 49–69. [Google Scholar] [CrossRef]
- Chou, T.-Y.; Chiu, C.-H.; Li, L.-H.; Hsiao, C.-Y.; Tzen, C.-Y.; Chang, K.-T.; Chen, Y.-M.; Perng, R.-P.; Tsai, S.-F.; Tsai, C.-M. Mutation in the tyrosine kinase domain of epidermal growth factor receptor is a predictive and prognostic factor for gefitinib treatment in patients with non–small cell lung cancer. Clin. Cancer Res. 2005, 11, 3750–3757. [Google Scholar] [CrossRef]
- Yasuda, H.; Park, E.; Yun, C.-H.; Sng, N.J.; Lucena-Araujo, A.R.; Yeo, W.-L.; Huberman, M.S.; Cohen, D.W.; Nakayama, S.; Ishioka, K. Structural, biochemical, and clinical characterization of epidermal growth factor receptor (EGFR) exon 20 insertion mutations in lung cancer. Sci. Transl. Med. 2013, 5, 216ra177. [Google Scholar] [CrossRef]
- Vyse, S.; Huang, P.H. Targeting EGFR exon 20 insertion mutations in non-small cell lung cancer. Signal Transduct. Target. Ther. 2019, 4, 5. [Google Scholar] [CrossRef]
- Boolell, V.; Alamgeer, M.; Watkins, D.N.; Ganju, V. The evolution of therapies in non-small cell lung cancer. Cancers 2015, 7, 1815–1846. [Google Scholar] [CrossRef]
- Ayati, A.; Moghimi, S.; Salarinejad, S.; Safavi, M.; Pouramiri, B.; Foroumadi, A. A review on progression of epidermal growth factor receptor (EGFR) inhibitors as an efficient approach in cancer targeted therapy. Bioorgan. Chem. 2020, 99, 103811. [Google Scholar] [CrossRef]
- Aran, V.; Omerovic, J. Current Approaches in NSCLC Targeting K-RAS and EGFR. Int. J. Mol. Sci. 2019, 20, 5701. [Google Scholar] [CrossRef] [PubMed]
- Sequist, L.V.; Soria, J.-C.; Goldman, J.W.; Wakelee, H.A.; Gadgeel, S.M.; Varga, A.; Papadimitrakopoulou, V.; Solomon, B.J.; Oxnard, G.R.; Dziadziuszko, R. Rociletinib in EGFR-mutated non–small-cell lung cancer. N. Engl. J. Med. 2015, 372, 1700–1709. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.-S.; Gilligan, D.; Pacey, S. Treatment approaches for EGFR-inhibitor-resistant patients with non-small-cell lung cancer. Lancet Oncol. 2015, 16, e447–e459. [Google Scholar] [CrossRef]
- Song, Z.; Ge, Y.; Wang, C.; Huang, S.; Shu, X.; Liu, K.; Zhou, Y.; Ma, X. Challenges and perspectives on the development of small-molecule EGFR inhibitors against T790M-mediated resistance in non-small-cell lung cancer: Miniperspective. J. Med. Chem. 2016, 59, 6580–6594. [Google Scholar] [CrossRef]
- Wang, S.; Song, Y.; Liu, D. EAI045: The fourth-generation EGFR inhibitor overcoming T790M and C797S resistance. Cancer Lett. 2017, 385, 51–54. [Google Scholar] [CrossRef]
- Hallberg, B.; Palmer, R.H. Mechanistic insight into ALK receptor tyrosine kinase in human cancer biology. Nat. Rev. Cancer 2013, 13, 685–700. [Google Scholar] [CrossRef]
- Wu, W.; Haderk, F.; Bivona, T.G. Non-Canonical Thinking for Targeting ALK-Fusion Onco-Proteins in Lung Cancer. Cancers 2017, 9, 164. [Google Scholar] [CrossRef]
- Bayliss, R.; Choi, J.; Fennell, D.A.; Fry, A.M.; Richards, M.W. Molecular mechanisms that underpin EML4-ALK driven cancers and their response to targeted drugs. Cell. Mol. Life Sci. 2016, 73, 1209–1224. [Google Scholar] [CrossRef]
- Childress, M.A.; Himmelberg, S.M.; Chen, H.; Deng, W.; Davies, M.A.; Lovly, C.M. ALK fusion partners impact response to ALK inhibition: Differential effects on sensitivity, cellular phenotypes, and biochemical properties. Mol. Cancer Res. 2018, 16, 1724–1736. [Google Scholar] [CrossRef]
- Soda, M.; Choi, Y.L.; Enomoto, M.; Takada, S.; Yamashita, Y.; Ishikawa, S.; Fujiwara, S.-I.; Watanabe, H.; Kurashina, K.; Hatanaka, H. Identification of the transforming EML4–ALK fusion gene in non-small-cell lung cancer. Nature 2007, 448, 561–566. [Google Scholar] [CrossRef]
- Lin, J.J.; Zhu, V.W.; Yoda, S.; Yeap, B.Y.; Schrock, A.B.; Dagogo-Jack, I.; Jessop, N.A.; Jiang, G.Y.; Le, L.P.; Gowen, K. Impact of EML4-ALK variant on resistance mechanisms and clinical outcomes in ALK-positive lung cancer. J. Clin. Oncol. 2018, 36, 1199. [Google Scholar] [CrossRef]
- Tuononen, K.; Sarhadi, V.K.; Wirtanen, A.; Rönty, M.; Salmenkivi, K.; Knuuttila, A.; Remes, S.; Telaranta-Keerie, A.I.; Bloor, S.; Ellonen, P. Targeted resequencing reveals ALK fusions in non-small cell lung carcinomas detected by FISH, immunohistochemistry, and real-time RT-PCR: A comparison of four methods. BioMed Res. Int. 2013, 2013, 757490. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Oya, Y.; Tanaka, K.; Shimizu, J.; Horio, Y.; Kuroda, H.; Sakao, Y.; Hida, T.; Yatabe, Y. Differential Crizotinib Response Duration Among ALK Fusion Variants in ALK-Positive Non-Small-Cell Lung Cancer. J. Clin. Oncol. 2016, 34, 3383–3389. [Google Scholar] [CrossRef] [PubMed]
- Cameron, L.; Solomon, B. New Treatment Options for ALK-Rearranged Non-Small Cell Lung Cancer. Curr. Treat. Options Oncol. 2015, 16, 49. [Google Scholar] [CrossRef]
- Sullivan, I.; Planchard, D. ALK inhibitors in non-small cell lung cancer: The latest evidence and developments. Ther. Adv. Med. Oncol. 2016, 8, 32–47. [Google Scholar] [CrossRef]
- El-Osta, H.; Shackelford, R. Personalized treatment options for ALK-positive metastatic non-small-cell lung cancer: Potential role for Ceritinib. Pharm. Pers. Med. 2015, 8, 145–154. [Google Scholar] [CrossRef]
- Zou, H.Y.; Li, Q.; Lee, J.H.; Arango, M.E.; McDonnell, S.R.; Yamazaki, S.; Koudriakova, T.B.; Alton, G.; Cui, J.J.; Kung, P.P.; et al. An orally available small-molecule inhibitor of c-Met, PF-2341066, exhibits cytoreductive antitumor efficacy through antiproliferative and antiangiogenic mechanisms. Cancer Res. 2007, 67, 4408–4417. [Google Scholar] [CrossRef]
- Malik, S.M.; Maher, V.E.; Bijwaard, K.E.; Becker, R.L.; Zhang, L.; Tang, S.W.; Song, P.; Liu, Q.; Marathe, A.; Gehrke, B. US Food and Drug Administration approval: Crizotinib for treatment of advanced or metastatic non–small cell lung cancer that is anaplastic lymphoma kinase positive. Clin. Cancer Res. 2014, 20, 2029–2034. [Google Scholar] [CrossRef]
- Friboulet, L.; Li, N.; Katayama, R.; Lee, C.C.; Gainor, J.F.; Crystal, A.S.; Michellys, P.Y.; Awad, M.M.; Yanagitani, N.; Kim, S.; et al. The ALK inhibitor ceritinib overcomes crizotinib resistance in non-small cell lung cancer. Cancer Discov. 2014, 4, 662–673. [Google Scholar] [CrossRef]
- Golding, B.; Luu, A.; Jones, R.; Viloria-Petit, A.M. The function and therapeutic targeting of anaplastic lymphoma kinase (ALK) in non-small cell lung cancer (NSCLC). Mol. Cancer 2018, 17, 1–15. [Google Scholar] [CrossRef]
- Shaw, A.T.; Solomon, B.J.; Besse, B.; Bauer, T.M.; Lin, C.-C.; Soo, R.A.; Riely, G.J.; Ou, S.-H.I.; Clancy, J.S.; Li, S. ALK resistance mutations and efficacy of lorlatinib in advanced anaplastic lymphoma kinase-positive non–small-cell lung cancer. J. Clin. Oncol. 2019, 37, 1370. [Google Scholar] [CrossRef] [PubMed]
- Santarpia, M.; Liguori, A.; D’Aveni, A.; Karachaliou, N.; Gonzalez-Cao, M.; Daffinà, M.G.; Lazzari, C.; Altavilla, G.; Rosell, R. Liquid biopsy for lung cancer early detection. J. Thorac. Dis. 2018, 10, S882–S897. [Google Scholar] [CrossRef] [PubMed]
- Alix-Panabières, C.; Pantel, K. Clinical applications of circulating tumor cells and circulating tumor DNA as liquid biopsy. Cancer Discov. 2016, 6, 479–491. [Google Scholar] [CrossRef] [PubMed]
- Goldman, J.W.; Noor, Z.S.; Remon, J.; Besse, B.; Rosenfeld, N. Are liquid biopsies a surrogate for tissue EGFR testing? Ann. Oncol. 2018, 29, i38–i46. [Google Scholar] [CrossRef]
- Barbany, G.; Arthur, C.; Lieden, A.; Nordenskjold, M.; Rosenquist, R.; Tesi, B.; Wallander, K.; Tham, E. Cell-free tumour DNA testing for early detection of cancer—A potential future tool. J. Intern. Med. 2019, 286, 118–136. [Google Scholar] [CrossRef]
- Grunt, M.; Hillebrand, T.; Schwarzenbach, H. Clinical relevance of size selection of circulating DNA. Transl. Cancer Res. 2017, 7, S171–S184. [Google Scholar] [CrossRef]
- Nishimoto, S.; Fukuda, D.; Higashikuni, Y.; Tanaka, K.; Hirata, Y.; Murata, C.; Kim-Kaneyama, J.-R.; Sato, F.; Bando, M.; Yagi, S. Obesity-induced DNA released from adipocytes stimulates chronic adipose tissue inflammation and insulin resistance. Sci. Adv. 2016, 2, e1501332. [Google Scholar] [CrossRef]
- Vora, N.L.; Johnson, K.L.; Subhabrata, B.; Catalano, P.M.; Hauguel-De Mouzon, S.; Bianchi, D.W. A multi-factorial relationship exists between total circulating cell-free DNA levels and maternal BMI. Prenat. Diagn. 2012, 32, 912. [Google Scholar] [CrossRef]
- Hellmann, M.D.; Ciuleanu, T.E.; Pluzanski, A.; Lee, J.S.; Otterson, G.A.; Audigier-Valette, C.; Minenza, E.; Linardou, H.; Burgers, S.; Salman, P.; et al. Nivolumab plus Ipilimumab in Lung Cancer with a High Tumor Mutational Burden. N. Engl. J. Med. 2018, 378, 2093–2104. [Google Scholar] [CrossRef]
- Malapelle, U.; Pisapia, P.; Rocco, D.; Smeraglio, R.; di Spirito, M.; Bellevicine, C.; Troncone, G. Next generation sequencing techniques in liquid biopsy: Focus on non-small cell lung cancer patients. Transl. Lung Cancer Res. 2016, 5, 505. [Google Scholar] [CrossRef]
- Thress, K.S.; Brant, R.; Carr, T.H.; Dearden, S.; Jenkins, S.; Brown, H.; Hammett, T.; Cantarini, M.; Barrett, J.C. EGFR mutation detection in ctDNA from NSCLC patient plasma: A cross-platform comparison of leading technologies to support the clinical development of AZD9291. Lung Cancer 2015, 90, 509–515. [Google Scholar] [CrossRef]
- Oxnard, G.R.; Thress, K.S.; Alden, R.S.; Lawrance, R.; Paweletz, C.P.; Cantarini, M.; Yang, J.C.; Barrett, J.C.; Janne, P.A. Association Between Plasma Genotyping and Outcomes of Treatment With Osimertinib (AZD9291) in Advanced Non-Small-Cell Lung Cancer. J. Clin. Oncol. 2016, 34, 3375–3382. [Google Scholar] [CrossRef]
- Jenkins, S.; Yang, J.C.; Ramalingam, S.S.; Yu, K.; Patel, S.; Weston, S.; Hodge, R.; Cantarini, M.; Janne, P.A.; Mitsudomi, T.; et al. Plasma ctDNA Analysis for Detection of the EGFR T790M Mutation in Patients with Advanced Non-Small Cell Lung Cancer. J. Thorac. Oncol. 2017, 12, 1061–1070. [Google Scholar] [CrossRef]
- Taus, A.; Camacho, L.; Rocha, P.; Hardy-Werbin, M.; Pijuan, L.; Piquer, G.; Lopez, E.; Dalmases, A.; Longaron, R.; Clave, S.; et al. Dynamics of EGFR Mutation Load in Plasma for Prediction of Treatment Response and Disease Progression in Patients With EGFR-Mutant Lung Adenocarcinoma. Clin. Lung Cancer 2018, 19, 387–394.e382. [Google Scholar] [CrossRef]
- Franovic, A.; Raymond, V.M.; Erlander, M.G.; Reckamp, K.L. Urine test for EGFR analysis in patients with non-small cell lung cancer. J. Thorac. Dis. 2017, 9, S1323–S1331. [Google Scholar] [CrossRef]
- Melkonyan, H.S.; Feaver, W.J.; Meyer, E.; Scheinker, V.; Shekhtman, E.M.; Xin, Z.; Umansky, S.R. Transrenal nucleic acids: From proof of principle to clinical tests. Ann. N. Y. Acad. Sci. 2008, 1137, 73–81. [Google Scholar] [CrossRef]
- Li, F.; Wei, F.; Huang, W.L.; Lin, C.C.; Li, L.; Shen, M.M.; Yan, Q.; Liao, W.; Chia, D.; Tu, M.; et al. Ultra-Short Circulating Tumor DNA (usctDNA) in Plasma and Saliva of Non-Small Cell Lung Cancer (NSCLC) Patients. Cancers 2020, 12, 2041. [Google Scholar] [CrossRef]
- Wu, Z.; Yang, Z.; Li, C.S.; Zhao, W.; Liang, Z.X.; Dai, Y.; Zhu, Q.; Miao, K.L.; Cui, D.H.; Chen, L.A. Differences in the genomic profiles of cell-free DNA between plasma, sputum, urine, and tumor tissue in advanced NSCLC. Cancer Med. 2019, 8, 910–919. [Google Scholar] [CrossRef]
- Husain, H.; Melnikova, V.O.; Kosco, K.; Woodward, B.; More, S.; Pingle, S.C.; Weihe, E.; Park, B.H.; Tewari, M.; Erlander, M.G.; et al. Monitoring Daily Dynamics of Early Tumor Response to Targeted Therapy by Detecting Circulating Tumor DNA in Urine. Clin. Cancer Res. 2017, 23, 4716–4723. [Google Scholar] [CrossRef]
- Reckamp, K.L.; Melnikova, V.O.; Karlovich, C.; Sequist, L.V.; Camidge, D.R.; Wakelee, H.; Perol, M.; Oxnard, G.R.; Kosco, K.; Croucher, P.; et al. A Highly Sensitive and Quantitative Test Platform for Detection of NSCLC EGFR Mutations in Urine and Plasma. J. Thorac. Oncol. 2016, 11, 1690–1700. [Google Scholar] [CrossRef]
- Berz, D.; Raymond, V.M.; Garst, J.H.; Erlander, M.G. Non-invasive urine testing of EGFR activating mutation and T790M resistance mutation in non-small cell lung cancer. Exp. Hematol. Oncol. 2015, 5, 24. [Google Scholar] [CrossRef]
- Wu, Z.; Yang, Z.; Li, C.S.; Zhao, W.; Liang, Z.X.; Dai, Y.; Zeng, J.; Zhu, Q.; Miao, K.L.; Cui, D.H.; et al. Non-invasive detection of EGFR and TP53 mutations through the combination of plasma, urine and sputum in advanced non-small cell lung cancer. Oncol. Lett. 2019, 18, 3581–3590. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, L.; Li, L.; Li, X.; Xu, Y.; Wang, M.; Liang, L.; Jiao, P.; Li, Y.; He, S.; et al. Sputum Cell-Free DNA: Valued Surrogate Sample for Detection of EGFR Mutation in Patients with Advanced Lung Adenocarcinoma. J. Mol. Diagn. 2020, 22, 934–942. [Google Scholar] [CrossRef]
- Kawahara, A.; Fukumitsu, C.; Azuma, K.; Taira, T.; Abe, H.; Takase, Y.; Murata, K.; Sadashima, E.; Hattori, S.; Naito, Y.; et al. A Combined test using both cell sediment and supernatant cell-free DNA in pleural effusion shows increased sensitivity in detecting activating EGFR mutation in lung cancer patients. Cytopathology 2018, 29, 150–155. [Google Scholar] [CrossRef]
- Lee, J.S.; Hur, J.Y.; Kim, I.A.; Kim, H.J.; Choi, C.M.; Lee, J.C.; Kim, W.S.; Lee, K.Y. Liquid biopsy using the supernatant of a pleural effusion for EGFR genotyping in pulmonary adenocarcinoma patients: A comparison between cell-free DNA and extracellular vesicle-derived DNA. BMC Cancer 2018, 18, 1236. [Google Scholar] [CrossRef]
- Song, Z.; Wang, W.; Li, M.; Liu, J.; Zhang, Y. Cytological-negative pleural effusion can be an alternative liquid biopsy media for detection of EGFR mutation in NSCLC patients. Lung Cancer 2019, 136, 23–29. [Google Scholar] [CrossRef]
- Tong, L.; Ding, N.; Tong, X.; Li, J.; Zhang, Y.; Wang, X.; Xu, X.; Ye, M.; Li, C.; Wu, X.; et al. Tumor-derived DNA from pleural effusion supernatant as a promising alternative to tumor tissue in genomic profiling of advanced lung cancer. Theranostics 2019, 9, 5532–5541. [Google Scholar] [CrossRef]
- Goodman, A.M.; Kato, S.; Bazhenova, L.; Patel, S.P.; Frampton, G.M.; Miller, V.; Stephens, P.J.; Daniels, G.A.; Kurzrock, R. Tumor Mutational Burden as an Independent Predictor of Response to Immunotherapy in Diverse Cancers. Mol. Cancer Ther. 2017, 16, 2598–2608. [Google Scholar] [CrossRef]
- Koessler, T.; Paradiso, V.; Piscuoglio, S.; Nienhold, R.; Ho, L.; Christinat, Y.; Terracciano, L.M.; Cathomas, G.; Wicki, A.; McKee, T.A.; et al. Reliability of liquid biopsy analysis: An inter-laboratory comparison of circulating tumor DNA extraction and sequencing with different platforms. Lab. Investig. 2020, 100, 1475–1484. [Google Scholar] [CrossRef]
- Gaur, P.; Bhattacharya, S.; Kant, S.; Kushwaha, R.; Singh, G.; Pandey, S. EGFR mutation detection and its association with clinicopathological characters of lung cancer patients. World J. Oncol. 2018, 9, 151. [Google Scholar] [CrossRef]
- Lin, C.; Shi, X.; Yang, S.; Zhao, J.; He, Q.; Jin, Y.; Yu, X. Comparison of ALK detection by FISH, IHC and NGS to predict benefit from crizotinib in advanced non-small-cell lung cancer. Lung Cancer 2019, 131, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Ohira, T.; Sakai, K.; Matsubayashi, J.; Kajiwara, N.; Kakihana, M.; Hagiwara, M.; Hibi, M.; Yoshida, K.; Maeda, J.; Ohtani, K. Tumor volume determines the feasibility of cell-free DNA sequencing for mutation detection in non-small cell lung cancer. Cancer Sci. 2016, 107, 1660–1666. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.-C.; Chang, I.-C.; Wang, C.-L.; Chen, T.-D.; Chen, Y.-T.; Liu, H.-P.; Chu, Y.; Chiu, Y.-T.; Wu, T.-H.; Chou, L.-H. Comparison of IHC, FISH and RT-PCR methods for detection of ALK rearrangements in 312 non-small cell lung cancer patients in Taiwan. PLoS ONE 2013, 8, e70839. [Google Scholar] [CrossRef] [PubMed]
- Hindson, B.J.; Ness, K.D.; Masquelier, D.A.; Belgrader, P.; Heredia, N.J.; Makarewicz, A.J.; Bright, I.J.; Lucero, M.Y.; Hiddessen, A.L.; Legler, T.C.; et al. High-throughput droplet digital PCR system for absolute quantitation of DNA copy number. Anal. Chem. 2011, 83, 8604–8610. [Google Scholar] [CrossRef] [PubMed]
- Forshew, T.; Murtaza, M.; Parkinson, C.; Gale, D.; Tsui, D.W.; Kaper, F.; Dawson, S.J.; Piskorz, A.M.; Jimenez-Linan, M.; Bentley, D.; et al. Noninvasive identification and monitoring of cancer mutations by targeted deep sequencing of plasma DNA. Sci. Transl. Med. 2012, 4, 136ra168. [Google Scholar] [CrossRef]
- Salk, J.J.; Schmitt, M.W.; Loeb, L.A. Enhancing the accuracy of next-generation sequencing for detecting rare and subclonal mutations. Nat. Rev. Genet. 2018, 19, 269–285. [Google Scholar] [CrossRef]
- Remon, J.; Garcia-Campelo, R.; de Alava, E.; Vera, R.; Rodriguez-Peralto, J.L.; Rodriguez-Lescure, A.; Bellosillo, B.; Garrido, P.; Rojo, F.; Alvarez-Alegret, R. Liquid biopsy in oncology: A consensus statement of the Spanish Society of Pathology and the Spanish Society of Medical Oncology. Clin. Transl. Oncol. 2020, 22, 823–834. [Google Scholar] [CrossRef]
- Siravegna, G.; Marsoni, S.; Siena, S.; Bardelli, A. Integrating liquid biopsies into the management of cancer. Nat. Rev. Clin. Oncol. 2017, 14, 531–548. [Google Scholar] [CrossRef]
- Chen, Y.L.; Lin, C.C.; Yang, S.C.; Chen, W.L.; Chen, J.R.; Hou, Y.H.; Lu, C.C.; Chow, N.H.; Su, W.C.; Ho, C.L. Five Technologies for Detecting the EGFR T790M Mutation in the Circulating Cell-Free DNA of Patients With Non-small Cell Lung Cancer: A Comparison. Front. Oncol. 2019, 9, 631. [Google Scholar] [CrossRef]
- Wu, Y.L.; Zhou, C.; Liam, C.K.; Wu, G.; Liu, X.; Zhong, Z.; Lu, S.; Cheng, Y.; Han, B.; Chen, L.; et al. First-line erlotinib versus gemcitabine/cisplatin in patients with advanced EGFR mutation-positive non-small-cell lung cancer: Analyses from the phase III, randomized, open-label, ENSURE study. Ann. Oncol. 2015, 26, 1883–1889. [Google Scholar] [CrossRef]
- Bordi, P.; Del Re, M.; Danesi, R.; Tiseo, M. Circulating DNA in diagnosis and monitoring EGFR gene mutations in advanced non-small cell lung cancer. Transl. Lung Cancer Res. 2015, 4, 584. [Google Scholar]
- Han, B.; Tjulandin, S.; Hagiwara, K.; Normanno, N.; Wulandari, L.; Konstantinovich, L.K.; Hudoyo, A.; Ratcliffe, M.; McCormack, R.; Reck, M. Determining the prevalence of EGFR mutations in Asian and Russian patients (pts) with advanced non-small-cell lung cancer (aNSCLC) of adenocarcinoma (ADC) and non-ADC histology: IGNITE study. Ann. Oncol. 2015, 26, i29. [Google Scholar] [CrossRef]
- Reck, M.; Hagiwara, K.; Han, B.; Tjulandin, S.; Grohe, C.; Yokoi, T.; Morabito, A.; Novello, S.; Arriola, E.; Molinier, O.; et al. ctDNA Determination of EGFR Mutation Status in European and Japanese Patients with Advanced NSCLC: The ASSESS Study. J. Thorac. Oncol. 2016, 11, 1682–1689. [Google Scholar] [CrossRef]
- Douillard, J.-Y.; Ostoros, G.; Cobo, M.; Ciuleanu, T.; Cole, R.; McWalter, G.; Walker, J.; Dearden, S.; Webster, A.; Milenkova, T. Gefitinib treatment in EGFR mutated caucasian NSCLC: Circulating-free tumor DNA as a surrogate for determination of EGFR status. J. Thorac. Oncol. 2014, 9, 1345–1353. [Google Scholar] [CrossRef]
- Zhou, C.; Wang, M.; Cheng, Y.; Chen, Y.; Ye, X.; Sun, Y.; Huang, X.; Patel, S.; Wu, Y. Detection of EGFR T790M in Asia-Pacific patients (pts) with EGFR mutation-positive advanced non-small cell lung cancer (NSCLC): Circulating tumour (ct) DNA analysis across 3 platforms. Ann. Oncol. 2017, 28, v474–v475. [Google Scholar] [CrossRef]
- Zhou, C.; Cheng, Y.; Lu, Y.; Shi, M.; Han, J.; Wang, M.; Kim, S.; Wang, J.; Chen, Y.; Wu, Y. CNS response to osimertinib in Asian-Pacific patients (pts) with T790M-positive advanced NSCLC: Data from an open-label Phase II trial (AURA17). Ann. Oncol. 2017, 28, v484. [Google Scholar] [CrossRef]
- Tong, Y.; Zhao, Z.; Liu, B.; Bao, A.; Zheng, H.; Gu, J.; McGrath, M.; Xia, Y.; Tan, B.; Song, C.; et al. 5’/3’ imbalance strategy to detect ALK fusion genes in circulating tumor RNA from patients with non-small cell lung cancer. J. Exp. Clin. Cancer Res. 2018, 37, 68. [Google Scholar] [CrossRef]
- McCoach, C.E.; Blakely, C.M.; Banks, K.C.; Levy, B.; Chue, B.M.; Raymond, V.M.; Le, A.T.; Lee, C.E.; Diaz, J.; Waqar, S.N.; et al. Clinical Utility of Cell-Free DNA for the Detection of ALK Fusions and Genomic Mechanisms of ALK Inhibitor Resistance in Non-Small Cell Lung Cancer. Clin. Cancer Res. 2018, 24, 2758–2770. [Google Scholar] [CrossRef]
- Bordi, P.; Tiseo, M.; Rofi, E.; Petrini, I.; Restante, G.; Danesi, R.; Del Re, M. Detection of ALK and KRAS Mutations in Circulating Tumor DNA of Patients With Advanced ALK-Positive NSCLC With Disease Progression During Crizotinib Treatment. Clin. Lung Cancer 2017, 18, 692–697. [Google Scholar] [CrossRef]
- Lanman, R.B.; Mortimer, S.A.; Zill, O.A.; Sebisanovic, D.; Lopez, R.; Blau, S.; Collisson, E.A.; Divers, S.G.; Hoon, D.S.; Kopetz, E.S.; et al. Analytical and Clinical Validation of a Digital Sequencing Panel for Quantitative, Highly Accurate Evaluation of Cell-Free Circulating Tumor DNA. PLoS ONE 2015, 10, e0140712. [Google Scholar] [CrossRef]

| Liquid Biopsies | Solid Biopsies | |
|---|---|---|
| Invasiveness | No | Yes |
| Continuous molecular assessment of the tumor | Biological evaluation of the tumor at any time allowing therapy monitoring | Difficulties in the follow-up of the tumor evolution |
| Cost | Low | High |
| Biopsy collection | Easy; simple blood (or urine, saliva) collection | Difficult; small tumors may require multiple attempts to retrieve enough tissue |
| Tumor heterogeneity | Allow examining the longitudinal evolution of the tumor; better reflect tumor heterogeneity | Only allow for a snapshot in time of the ever-evolving tumor biology; limited access to the intra- and intertumor heterogeneity |
| Specificity | High; overdiagnosis in early cancer detection and high rate of false positives are observed | Higher; they allow the application of several specific methods |
| Method | Advantage | Disadvantage | Clinical Application | Validated Assays for NSCLC Liquid Biopsies |
|---|---|---|---|---|
| PCR-based | Sensitivity (AF), 0.1–0.001%; straightforward data analysis; rapid; lower cost. | Need prior knowledge of the mutation; analysis limited to few targets; higher amount of sample. | Analysis of specific gene mutations (as in EGFR) in NSCLC patients’ liquid biopsies | Therascreen EGFR RGQ PCR Kit and Cobas EGFR Mutation Test v2 |
| NGS-based | No need in prior knowledge of the mutation; analysis of a high number of targets; smaller amount of sample. | Sensitivity (AF), 5–0.1%; needs extensive bioinformatics support for data analysis; slower; high cost. | Analysis of several gene mutations/alterations (as in EGFR and ALK) in NSCLC patients’ liquid biopsies | Foundation-One Liquid CDx and Guardant360 CDx |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferreira, D.; Miranda, J.; Martins-Lopes, P.; Adega, F.; Chaves, R. Future Perspectives in Detecting EGFR and ALK Gene Alterations in Liquid Biopsies of Patients with NSCLC. Int. J. Mol. Sci. 2021, 22, 3815. https://doi.org/10.3390/ijms22083815
Ferreira D, Miranda J, Martins-Lopes P, Adega F, Chaves R. Future Perspectives in Detecting EGFR and ALK Gene Alterations in Liquid Biopsies of Patients with NSCLC. International Journal of Molecular Sciences. 2021; 22(8):3815. https://doi.org/10.3390/ijms22083815
Chicago/Turabian StyleFerreira, Daniela, Juliana Miranda, Paula Martins-Lopes, Filomena Adega, and Raquel Chaves. 2021. "Future Perspectives in Detecting EGFR and ALK Gene Alterations in Liquid Biopsies of Patients with NSCLC" International Journal of Molecular Sciences 22, no. 8: 3815. https://doi.org/10.3390/ijms22083815
APA StyleFerreira, D., Miranda, J., Martins-Lopes, P., Adega, F., & Chaves, R. (2021). Future Perspectives in Detecting EGFR and ALK Gene Alterations in Liquid Biopsies of Patients with NSCLC. International Journal of Molecular Sciences, 22(8), 3815. https://doi.org/10.3390/ijms22083815






