The Impact of Spaceflight and Microgravity on the Human Islet-1+ Cardiovascular Progenitor Cell Transcriptome
Abstract
1. Introduction
2. Results
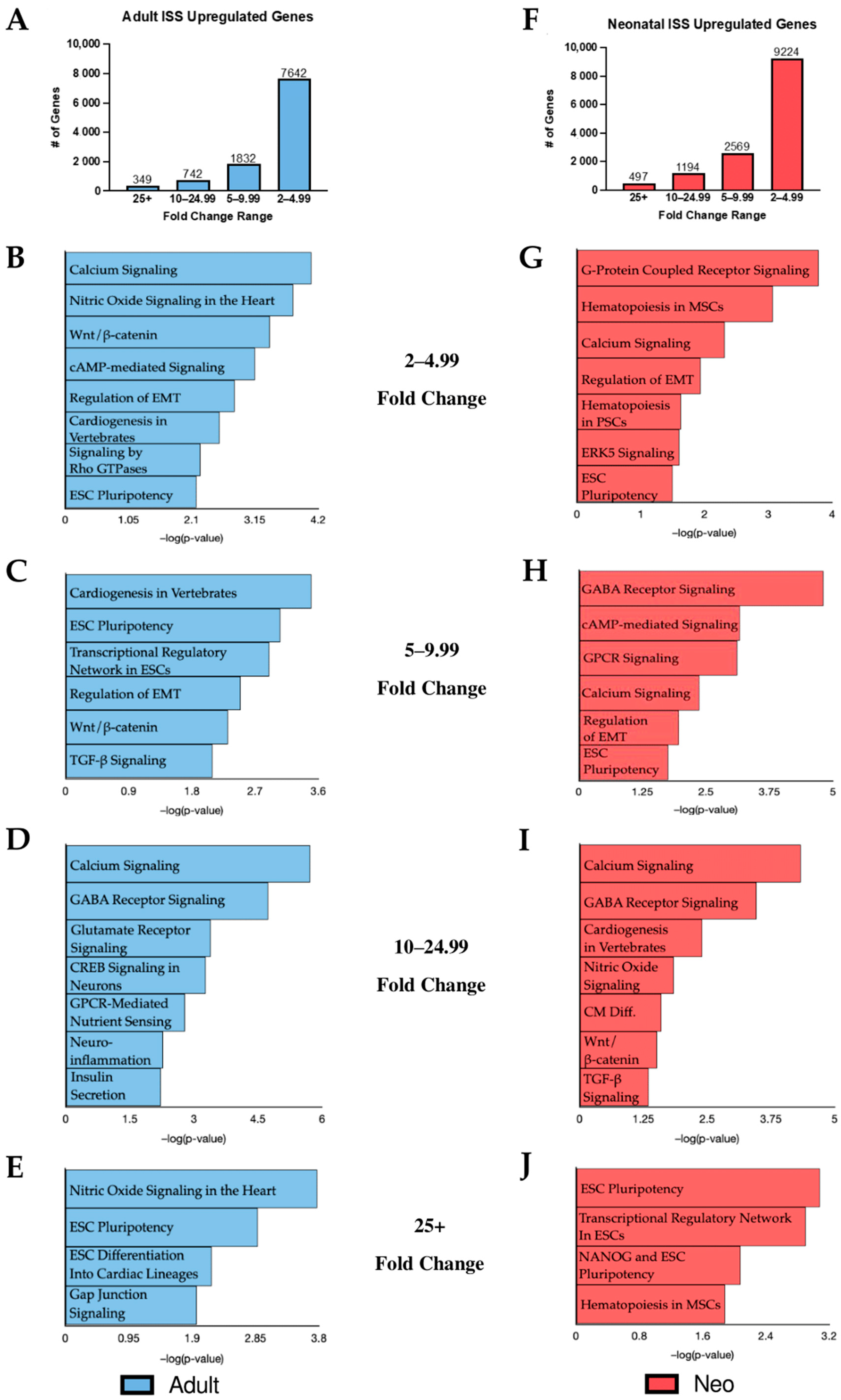
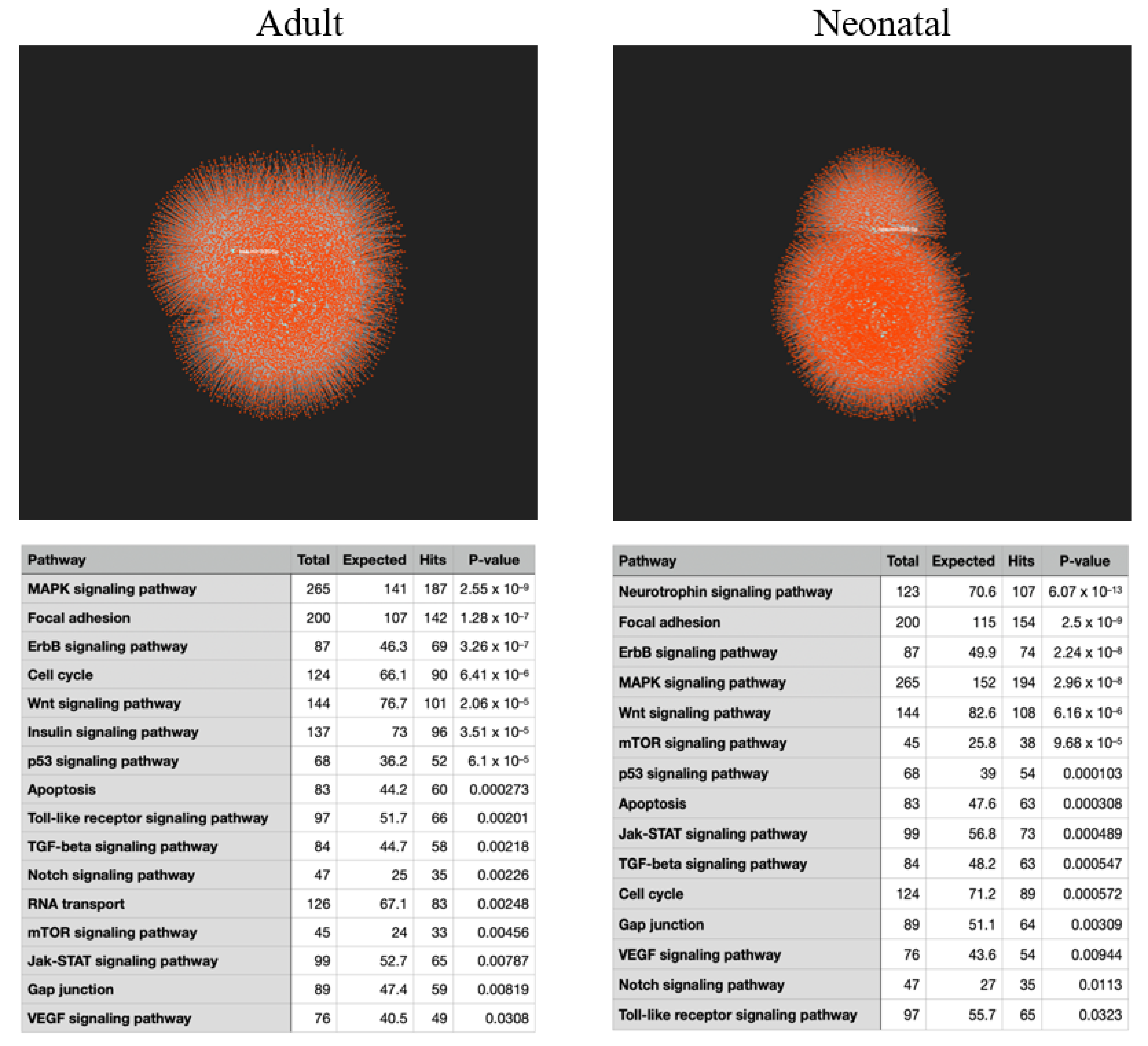
2.1. KEGG Analysis of Induced Gene Transcripts Reveals Broad Responses to Spaceflight
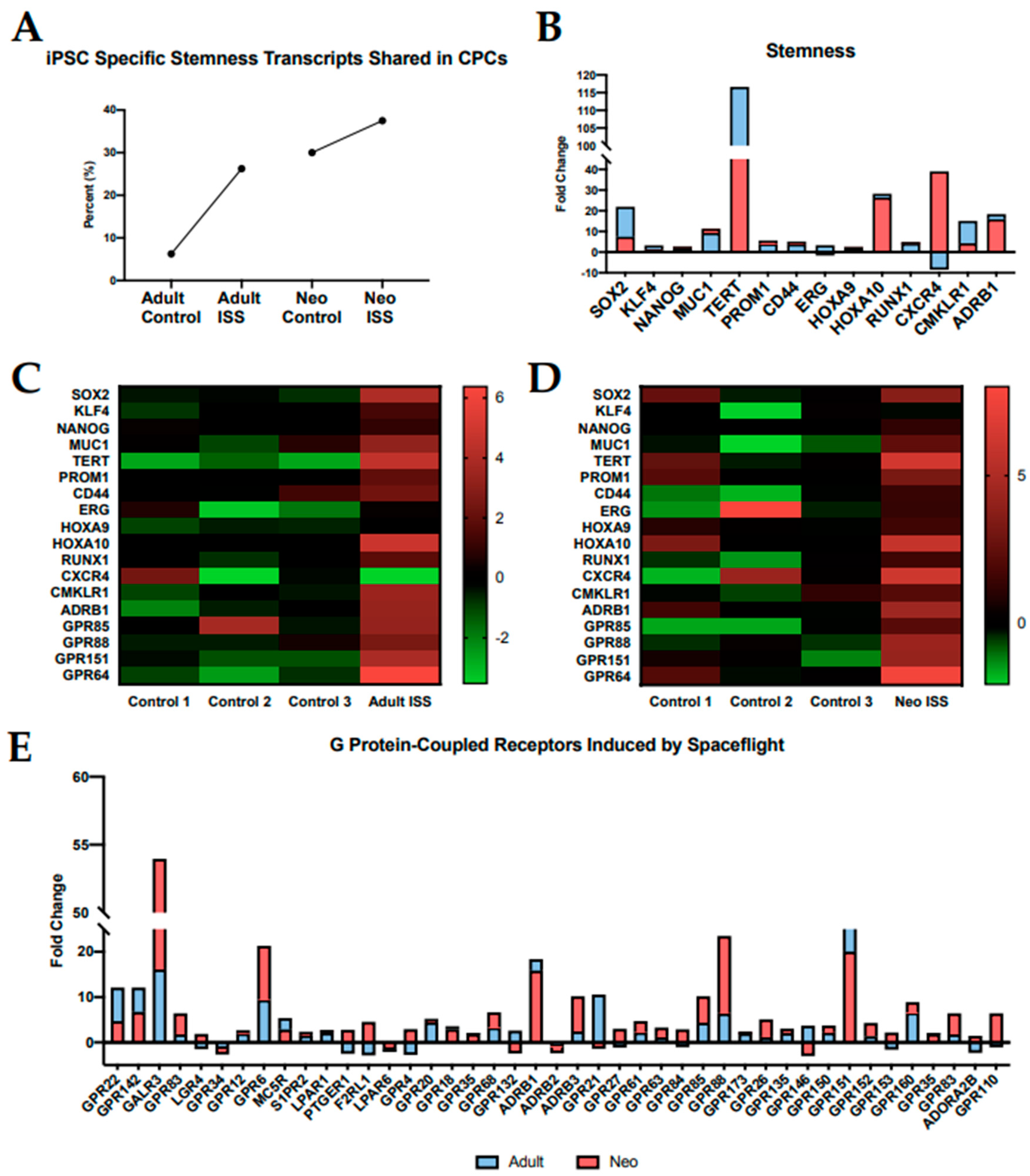
2.2. Spaceflight Induces Stemness
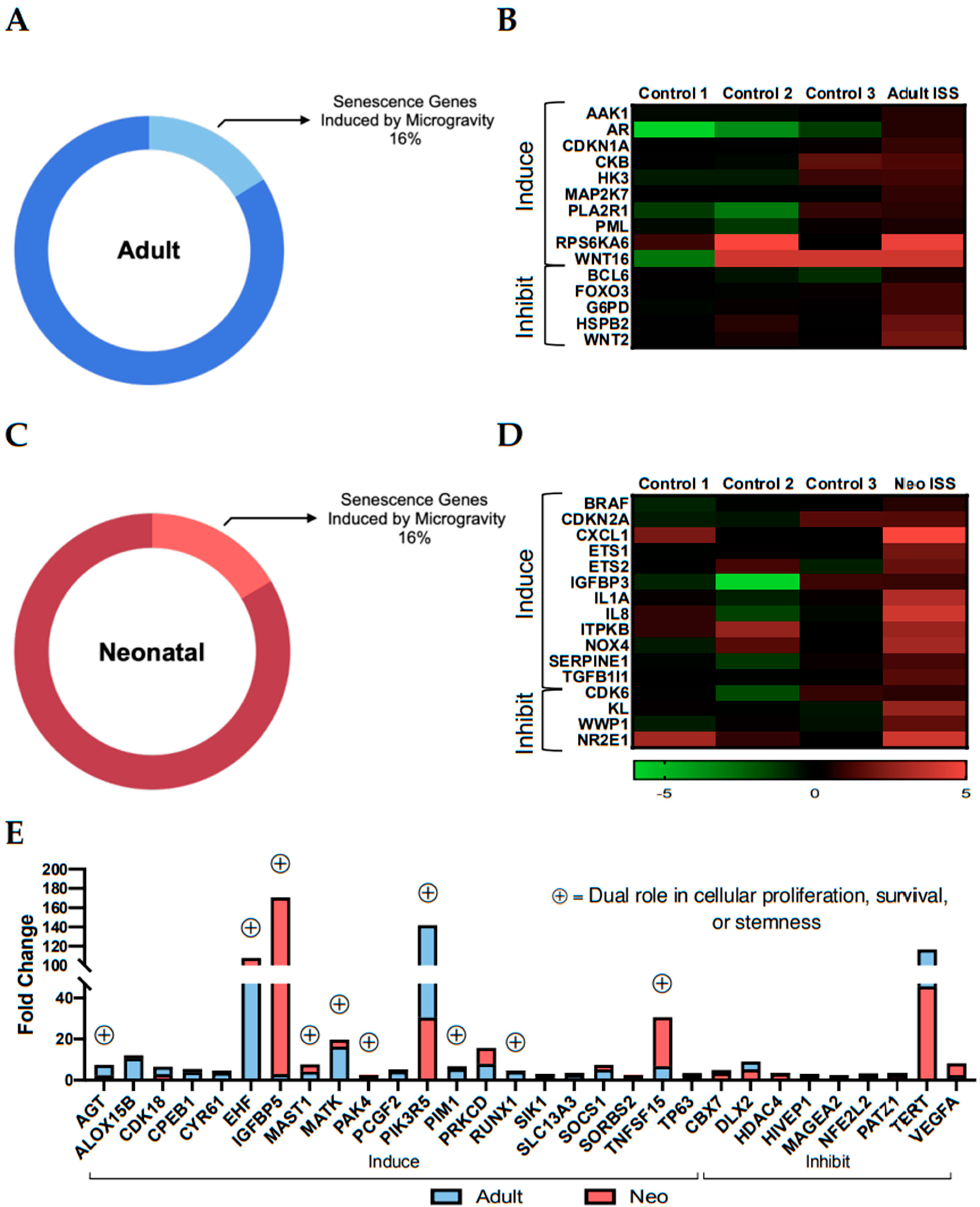
2.3. Culture Aboard the ISS for 30 Days Induced Transcripts with Dual Roles in Stemness and Senescence
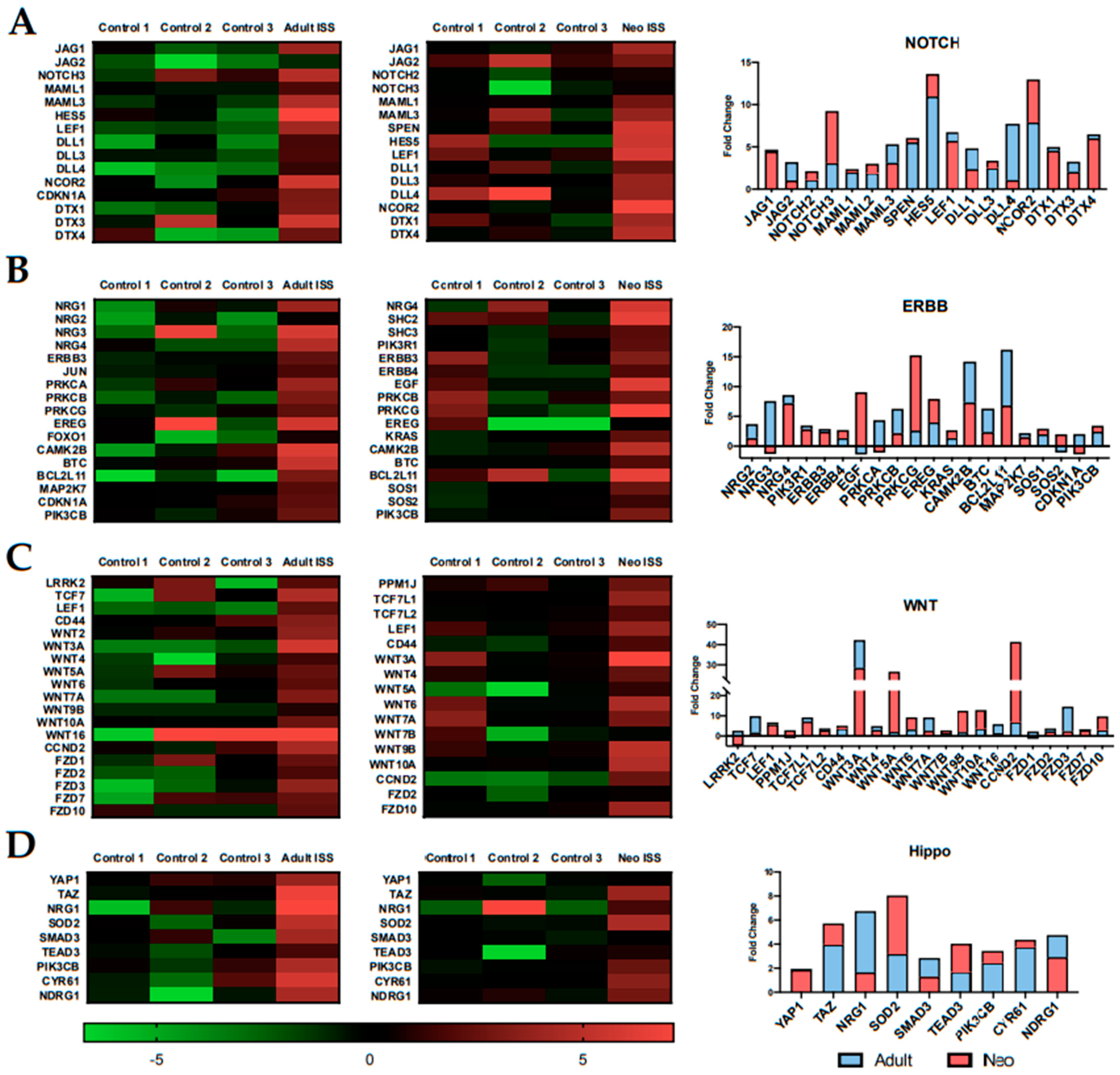
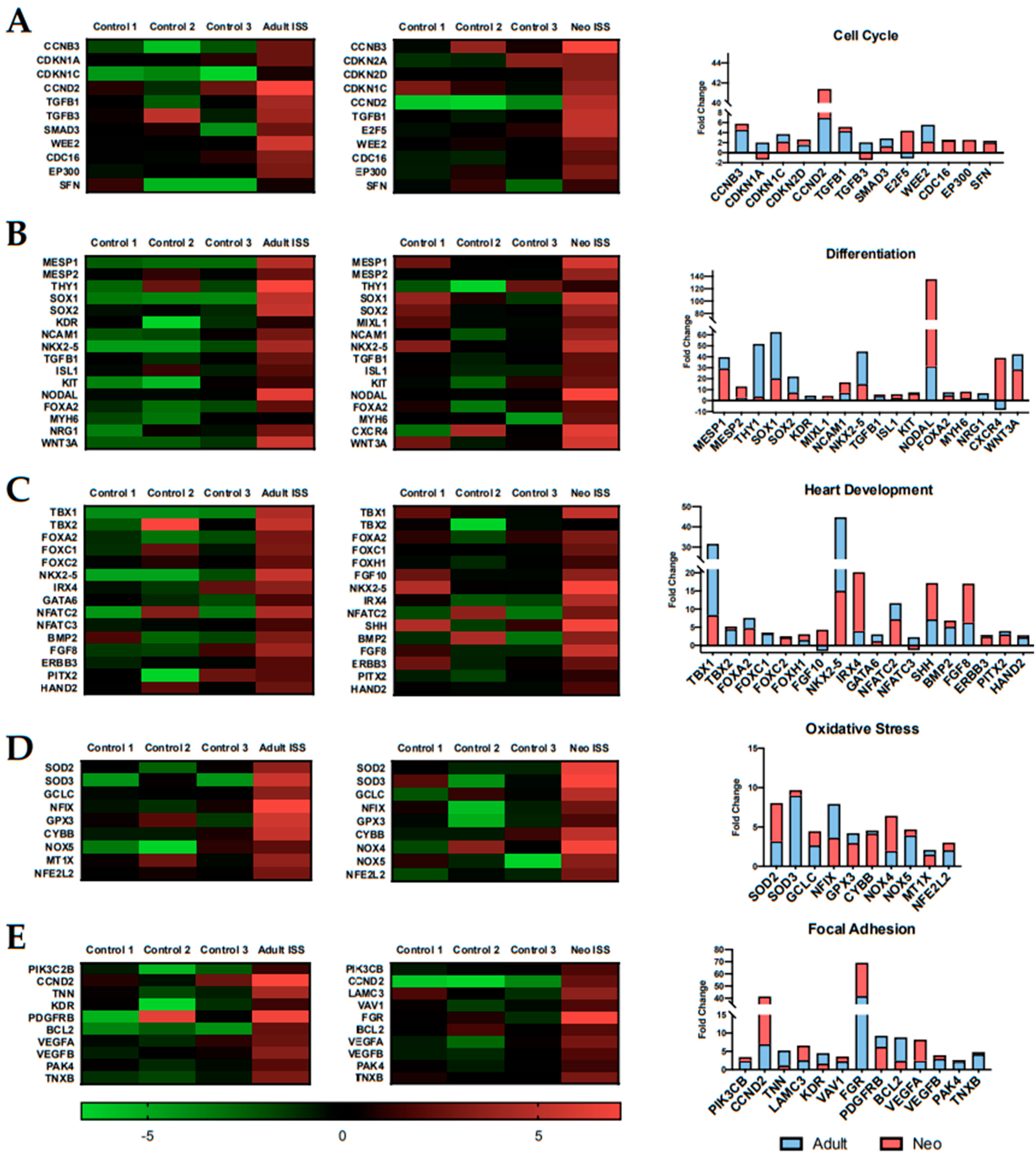
2.4. Spaceflight Alters the Expression of Specific Pathways Related to Proliferation and Survival
2.5. Spaceflight Alters the Expression of Genes Related to Specific Biological Processes
2.6. MicroRNA Analysis
3. Discussion
4. Materials and Methods
4.1. CPCs and Culture Aboard the ISS
4.2. RNA Sequencing
4.3. Transcriptomic Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CPC | Cardiovascular progenitor cell |
| DAVID | Database for Annotation, Visualization and Integrated Discovery |
| EMT | Epithelial–mesenchymal transition |
| ESC | Embryonic stem cell |
| GPCR | G protein-coupled receptor |
| iPSC | Induced pluripotent stem cell |
| IPA | Ingenuity Pathway Analysis |
| ISS | International Space Station |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| NASA | National Aeronautics and Space Agency |
| NRG | Neuregulin |
References
- Bellone, J.A.; Gifford, P.S.; Nishiyama, N.C.; Hartman, R.E.; Mao, X.W. Long-Term Effects of Simulated Microgravity and/or Chronic Exposure to Low-Dose Gamma Radiation on Behavior and Blood–Brain Barrier Integrity. NPJ Microgravity 2016, 2, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Nassef, M.Z.; Kopp, S.; Melnik, D.; Corydon, T.J.; Sahana, J.; Krüger, M.; Wehland, M.; Bauer, T.J.; Liemersdorf, C.; Hemmersbach, R.; et al. Short-Term Microgravity Influences Cell Adhesion in Human Breast Cancer Cells. Int. J. Mol. Sci. 2019, 20, 5730. [Google Scholar] [CrossRef]
- Bradbury, P.; Wu, H.; Choi, J.U.; Rowan, A.E.; Zhang, H.; Poole, K.; Lauko, J.; Chou, J. Modeling the Impact of Microgravity at the Cellular Level: Implications for Human Disease. Front. Cell Dev. Biol. 2020, 8. [Google Scholar] [CrossRef] [PubMed]
- Thiel, C.S.; Tauber, S.; Lauber, B.; Polzer, J.; Seebacher, C.; Uhl, R.; Neelam, S.; Zhang, Y.; Levine, H.; Ullrich, O. Rapid Morphological and Cytoskeletal Response to Microgravity in Human Primary Macrophages. Int. J. Mol. Sci. 2019, 20, 2402. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.-Y.; Guo, S.; Li, B.-B.; Jiang, N.; Li, A.; Yan, H.-F.; Yang, H.-M.; Zhou, J.-L.; Li, C.-L.; Cui, Y. Effect of Weightlessness on the 3D Structure Formation and Physiologic Function of Human Cancer Cells. Biomed. Res. Int. 2019, 2019. [Google Scholar] [CrossRef]
- Grimm, D.; Wehland, M.; Corydon, T.J.; Richter, P.; Prasad, B.; Bauer, J.; Egli, M.; Kopp, S.; Lebert, M.; Krüger, M. The Effects of Microgravity on Differentiation and Cell Growth in Stem Cells and Cancer Stem Cells. STEM CELLS Transl. Med. 2020, 9, 882–894. [Google Scholar] [CrossRef] [PubMed]
- Strauch, S.M.; Grimm, D.; Corydon, T.J.; Krüger, M.; Bauer, J.; Lebert, M.; Wise, P.; Infanger, M.; Richter, P. Current Knowledge about the Impact of Microgravity on the Proteome. Expert Rev. Proteom. 2019, 16, 5–16. [Google Scholar] [CrossRef]
- Garrett-Bakelman, F.E.; Darshi, M.; Green, S.J.; Gur, R.C.; Lin, L.; Macias, B.R.; McKenna, M.J.; Meydan, C.; Mishra, T.; Nasrini, J.; et al. The NASA Twins Study: A Multidimensional Analysis of a Year-Long Human Spaceflight. Science 2019, 364. [Google Scholar] [CrossRef]
- Wnorowski, A.; Sharma, A.; Chen, H.; Wu, H.; Shao, N.-Y.; Sayed, N.; Liu, C.; Countryman, S.; Stodieck, L.S.; Rubins, K.H.; et al. Effects of Spaceflight on Human Induced Pluripotent Stem Cell-Derived Cardiomyocyte Structure and Function. Stem Cell Rep. 2019, 13, 960–969. [Google Scholar] [CrossRef]
- Grigoryan, E.N.; Radugina, E.A. Behavior of Stem-Like Cells, Precursors for Tissue Regeneration in Urodela, Under Conditions of Microgravity. Stem Cells Dev. 2019. [Google Scholar] [CrossRef]
- Fuentes, T.I.; Appleby, N.; Raya, M.; Bailey, L.; Hasaniya, N.; Stodieck, L.; Kearns-Jonker, M. Simulated Microgravity Exerts an Age-Dependent Effect on the Differentiation of Cardiovascular Progenitors Isolated from the Human Heart. PLoS ONE 2015, 10, e0132378. [Google Scholar] [CrossRef]
- Baio, J.; Martinez, A.F.; Bailey, L.; Hasaniya, N.; Pecaut, M.J.; Kearns-Jonker, M. Spaceflight Activates Protein Kinase C Alpha Signaling and Modifies the Developmental Stage of Human Neonatal Cardiovascular Progenitor Cells. Stem Cells Dev. 2018, 27, 805–818. [Google Scholar] [CrossRef] [PubMed]
- Phuyal, S.; Farhan, H. Multifaceted Rho GTPase Signaling at the Endomembranes. Front. Cell Dev. Biol. 2019, 7. [Google Scholar] [CrossRef]
- Johnson, R.D.; Camelliti, P. Role of Non-Myocyte Gap Junctions and Connexin Hemichannels in Cardiovascular Health and Disease: Novel Therapeutic Targets? Int. J. Mol. Sci. 2018, 19, 866. [Google Scholar] [CrossRef] [PubMed]
- Lemcke, H.; Gaebel, R.; Skorska, A.; Voronina, N.; Lux, C.A.; Petters, J.; Sasse, S.; Zarniko, N.; Steinhoff, G.; David, R. Mechanisms of Stem Cell Based Cardiac Repair-Gap Junctional Signaling Promotes the Cardiac Lineage Specification of Mesenchymal Stem Cells. Sci. Rep. 2017, 7, 1–17. [Google Scholar] [CrossRef]
- Yu, O.M.; Brown, J.H. G Protein–Coupled Receptor and RhoA-Stimulated Transcriptional Responses: Links to Inflammation, Differentiation, and Cell Proliferation. Mol. Pharmacol. 2015, 88, 171–180. [Google Scholar] [CrossRef]
- Dolatshad, N.F.; Hellen, N.; Jabbour, R.J.; Harding, S.E.; Földes, G. G-Protein Coupled Receptor Signaling in Pluripotent Stem Cell-Derived Cardiovascular Cells: Implications for Disease Modeling. Front. Cell Dev. Biol. 2015, 3. [Google Scholar] [CrossRef]
- Gomez, N.; Erazo, T.; Lizcano, J.M. ERK5 and Cell Proliferation: Nuclear Localization Is What Matters. Front. Cell Dev. Biol. 2016, 4. [Google Scholar] [CrossRef]
- Pereira, D.M.; Gomes, S.E.; Borralho, P.M.; Rodrigues, C.M.P. MEK5/ERK5 Activation Regulates Colon Cancer Stem-like Cell Properties. Cell Death Discov. 2019, 5, 68. [Google Scholar] [CrossRef]
- Bianchi, M.E.; Mezzapelle, R. The Chemokine Receptor CXCR4 in Cell Proliferation and Tissue Regeneration. Front. Immunol. 2020, 11, 2109. [Google Scholar] [CrossRef]
- Wang, X.; Wang, H.; Wei, X.; Wang, A.; Wen, L.; Wang, L.; Huang, Q. Effect of CXCR4 Silencing with ShRNA on MAPK Signaling in Ovarian Cancer. Oncol. Lett. 2018, 15, 10026–10030. [Google Scholar] [CrossRef]
- Majidinia, M.; Aghazadeh, J.; Jahanban-Esfahlani, R.; Yousefi, B. The Roles of Wnt/β-Catenin Pathway in Tissue Development and Regenerative Medicine. J. Cell Physiol. 2018, 233, 5598–5612. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Unternaehrer, J.J. Epithelial-Mesenchymal Transition and Cancer Stem Cells: At the Crossroads of Differentiation and Dedifferentiation: EMT in Differentiation and Dedifferentiation. Dev. Dyn. 2019, 248, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Wang, Y.; Zhang, M.; Zhang, P.; Liang, H.; Bai, H.; Yu, X.; Yang, H. Functional Expression of the Ca2+ Signaling Machinery in Human Embryonic Stem Cells. Acta Pharmacol. Sin. 2017, 38, 1663–1672. [Google Scholar] [CrossRef] [PubMed]
- Pinto, J.P.; Kalathur, R.K.; Oliveira, D.V.; Barata, T.; Machado, R.S.R.; Machado, S.; Pacheco-Leyva, I.; Duarte, I.; Futschik, M.E. StemChecker: A Web-Based Tool to Discover and Explore Stemness Signatures in Gene Sets. Nucleic Acids Res. 2015, 43, W72–W77. [Google Scholar] [CrossRef]
- Bragança, J.; Lopes, J.A.; Mendes-Silva, L.; Almeida Santos, J.M. Induced Pluripotent Stem Cells, a Giant Leap for Mankind Therapeutic Applications. World J. Stem Cells 2019, 11, 421–430. [Google Scholar] [CrossRef]
- Choi, H.Y.; Saha, S.K.; Kim, K.; Kim, S.; Yang, G.-M.; Kim, B.; Kim, J.; Cho, S.-G. G Protein-Coupled Receptors in Stem Cell Maintenance and Somatic Reprogramming to Pluripotent or Cancer Stem Cells. BMB Rep. 2015, 48, 68–80. [Google Scholar] [CrossRef]
- Milanovic, M.; Fan, D.N.Y.; Belenki, D.; Däbritz, J.H.M.; Zhao, Z.; Yu, Y.; Dörr, J.R.; Dimitrova, L.; Lenze, D.; Monteiro Barbosa, I.A.; et al. Senescence-Associated Reprogramming Promotes Cancer Stemness. Nature 2018, 553, 96–100. [Google Scholar] [CrossRef]
- Sanada, F.; Taniyama, Y.; Muratsu, J.; Otsu, R.; Shimizu, H.; Rakugi, H.; Morishita, R. IGF Binding Protein-5 Induces Cell Senescence. Front. Endocrinol. (Lausanne) 2018, 9, 53. [Google Scholar] [CrossRef] [PubMed]
- Sureshbabu, A.; Okajima, H.; Yamanaka, D.; Tonner, E.; Shastri, S.; Maycock, J.; Szymanowska, M.; Shand, J.; Takahashi, S.-I.; Beattie, J.; et al. IGFBP5 Induces Cell Adhesion, Increases Cell Survival and Inhibits Cell Migration in MCF-7 Human Breast Cancer Cells. J. Cell Sci. 2012, 125, 1693–1705. [Google Scholar] [CrossRef]
- Yen, C.-F.; Wang, H.-S.; Lee, C.-L.; Liao, S.-K. Roles of Integrin-Linked Kinase in Cell Signaling and Its Perspectives as a Therapeutic Target. Gynecol. Minim. Invasive Ther. 2014, 3, 67–72. [Google Scholar] [CrossRef]
- Liu, J.-Y.; Souroullas, G.P.; Diekman, B.O.; Krishnamurthy, J.; Hall, B.M.; Sorrentino, J.A.; Parker, J.S.; Sessions, G.A.; Gudkov, A.V.; Sharpless, N.E. Cells Exhibiting Strong P16 INK4a Promoter Activation in Vivo Display Features of Senescence. Proc. Natl. Acad. Sci. USA 2019, 116, 2603–2611. [Google Scholar] [CrossRef]
- Hatzis, P.; Snippert, H.J.G. Long Noncoding RNAs in Gut Stem Cells. Nat. Cell Biol. 2018, 20, 1106–1107. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Ai, M.; Nie, M.; Zhao, L.; Deng, G.; Hu, S.; Han, Y.; Zeng, W.; Wang, Y.; Yang, M.; et al. EHF Promotes Colorectal Carcinoma Progression by Activating TGF-β1 Transcription and Canonical TGF-β Signaling. Cancer Sci. 2020, 111, 2310–2324. [Google Scholar] [CrossRef]
- Baio, J.; Martinez, A.F.; Silva, I.; Hoehn, C.V.; Countryman, S.; Bailey, L.; Hasaniya, N.; Pecaut, M.J.; Kearns-Jonker, M. Cardiovascular Progenitor Cells Cultured Aboard the International Space Station Exhibit Altered Developmental and Functional Properties. NPJ Microgravity 2018, 4. [Google Scholar] [CrossRef]
- Camberos, V.; Baio, J.; Bailey, L.; Hasaniya, N.; Lopez, L.V.; Kearns-Jonker, M. Effects of Spaceflight and Simulated Microgravity on YAP1 Expression in Cardiovascular Progenitors: Implications for Cell-Based Repair. Int. J. Mol. Sci. 2019, 20, 2742. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Fang, Y.; Wan, L.; Xu, Q.; Huang, H.; Zhu, R.; Wu, Q.; Liu, J. Notch Signaling Inhibits Cardiac Fibroblast to Myofibroblast Transformation by Antagonizing TGF-β1/Smad3 Signaling. J. Cell Physiol. 2019, 234, 8834–8845. [Google Scholar] [CrossRef] [PubMed]
- D’Uva, G.; Tzahor, E. The Key Roles of ERBB2 in Cardiac Regeneration. Cell Cycle 2015, 14, 2383–2384. [Google Scholar] [CrossRef]
- Totaro, A.; Castellan, M.; Biagio, D.D.; Piccolo, S. Crosstalk between YAP/TAZ and Notch Signaling. Trends Cell Biol. 2018, 28, 560–573. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Lv, X.; Hua, G.; Lele, S.M.; Remmenga, S.; Dong, J.; Davis, J.S.; Wang, C. YAP Forms Autocrine Loops with the ERBB Pathway to Regulate Ovarian Cancer Initiation and Progression. Oncogene 2015, 34, 6040–6054. [Google Scholar] [CrossRef]
- Li, N.; Lu, N.; Xie, C. The Hippo and Wnt Signalling Pathways: Crosstalk during Neoplastic Progression in Gastrointestinal Tissue. FEBS J. 2019, 286, 3745–3756. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.; Li, R.; Zhang, Q.; Wang, Y.; Jiang, Y.; Meng, Q.; Xi, Q.; Wu, G. Prognostic Role of Cyclin D2/D3 in Multiple Human Malignant Neoplasms: A Systematic Review and Meta-analysis. Cancer Med. 2019, 8, 2717–2729. [Google Scholar] [CrossRef]
- Granados-Aparici, S.; Hardy, K.; Franks, S.; Sharum, I.B.; Waite, S.L.; Fenwick, M.A. SMAD3 Directly Regulates Cell Cycle Genes to Maintain Arrest in Granulosa Cells of Mouse Primordial Follicles. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Heemskerk, I.; Burt, K.; Miller, M.; Chhabra, S.; Guerra, M.C.; Liu, L.; Warmflash, A. Rapid Changes in Morphogen Concentration Control Self-Organized Patterning in Human Embryonic Stem Cells. eLife 2019, 8, e40526. [Google Scholar] [CrossRef]
- Zhang, L.; Nomura-Kitabayashi, A.; Sultana, N.; Cai, W.; Cai, X.; Moon, A.M.; Cai, C.-L. Mesodermal Nkx2.5 Is Necessary and Sufficient for Early Second Heart Field Development. Dev. Biol. 2014, 390, 68–79. [Google Scholar] [CrossRef]
- Anderson, D.J.; Kaplan, D.I.; Bell, K.M.; Koutsis, K.; Haynes, J.M.; Mills, R.J.; Phelan, D.G.; Qian, E.L.; Leitoguinho, A.R.; Arasaratnam, D.; et al. NKX2-5 Regulates Human Cardiomyogenesis via a HEY2 Dependent Transcriptional Network. Nat. Commun. 2018, 9, 1–13. [Google Scholar] [CrossRef]
- Moris, D.; Spartalis, M.; Tzatzaki, E.; Spartalis, E.; Karachaliou, G.-S.; Triantafyllis, A.S.; Karaolanis, G.I.; Tsilimigras, D.I.; Theocharis, S. The Role of Reactive Oxygen Species in Myocardial Redox Signaling and Regulation. Ann. Transl. Med. 2017, 5. [Google Scholar] [CrossRef]
- Tseng, W.-L.; Chou, S.-J.; Chiang, H.-C.; Wang, M.-L.; Chien, C.-S.; Chen, K.-H.; Leu, H.-B.; Wang, C.-Y.; Chang, Y.-L.; Liu, Y.-Y.; et al. Imbalanced Production of Reactive Oxygen Species and Mitochondrial Antioxidant SOD2 in Fabry Disease-Specific Human Induced Pluripotent Stem Cell-Differentiated Vascular Endothelial Cells. Cell Transplant. 2017, 26, 513–527. [Google Scholar] [CrossRef]
- Yu, H.; Gao, M.; Ma, Y.; Wang, L.; Shen, Y.; Liu, X. Inhibition of Cell Migration by Focal Adhesion Kinase: Time-Dependent Difference in Integrin-Induced Signaling between Endothelial and Hepatoblastoma Cells. Int. J. Mol. Med. 2018, 41, 2573–2588. [Google Scholar] [CrossRef]
- Bueno, M.J.; Malumbres, M. MicroRNAs and the Cell Cycle. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2011, 1812, 592–601. [Google Scholar] [CrossRef]
- Mestdagh, P.; Boström, A.-K.; Impens, F.; Fredlund, E.; Van Peer, G.; De Antonellis, P.; von Stedingk, K.; Ghesquière, B.; Schulte, S.; Dews, M.; et al. The MiR-17-92 MicroRNA Cluster Regulates Multiple Components of the TGFβ-Pathway in Neuroblastoma. Mol. Cell 2010, 40, 762–773. [Google Scholar] [CrossRef] [PubMed]
- Diez-Cuñado, M.; Wei, K.; Bushway, P.J.; Maurya, M.R.; Perera, R.; Subramaniam, S.; Ruiz-Lozano, P.; Mercola, M. MiRNAs That Induce Human Cardiomyocyte Proliferation Converge on the Hippo Pathway. Cell Rep. 2018, 23, 2168–2174. [Google Scholar] [CrossRef]
- Lu, C.; Zhou, D.; Wang, Q.; Liu, W.; Yu, F.; Wu, F.; Chen, C. Crosstalk of MicroRNAs and Oxidative Stress in the Pathogenesis of Cancer. Oxid. Med. Cell Longev. 2020, 2020. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Wang, C. Dedifferentiation: Inspiration for Devising Engineering Strategies for Regenerative Medicine. NPJ Regen. Med. 2020, 5. [Google Scholar] [CrossRef]
- Transient Reprogramming of Neonatal Cardiomyocytes to a Proliferative Dedifferentiated State|BioRxiv. Available online: https://www.biorxiv.org/content/10.1101/801092v1 (accessed on 14 February 2021).
- Le, T.Y.L.; Pickett, H.A.; Yang, A.; Ho, J.W.K.; Thavapalachandran, S.; Igoor, S.; Yang, S.F.; Farraha, M.; Voges, H.K.; Hudson, J.E.; et al. Enhanced Cardiac Repair by Telomerase Reverse Transcriptase Over-Expression in Human Cardiac Mesenchymal Stromal Cells. Sci. Rep. 2019, 9, 1–17. [Google Scholar] [CrossRef]
- Smith-Sonneborn, J. Telomerase Biology Associations Offer Keys to Cancer and Aging Therapeutics. Curr. Aging Sci. 2020, 13, 11–21. [Google Scholar] [CrossRef]
- Muñoz-Lorente, M.A.; Cano-Martin, A.C.; Blasco, M.A. Mice with Hyper-Long Telomeres Show Less Metabolic Aging and Longer Lifespans. Nat. Commun. 2019, 10, 4723. [Google Scholar] [CrossRef] [PubMed]
- Othman, A.; Mubarak, R.; Sabry, D. Fibroblast Growth Factor-6 Enhances CDK2 and MATK Expression in Microvesicles Derived from Human Stem Cells Extracted from Exfoliated Deciduous Teeth. F1000Research 2020, 7. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Deng, Y.; Zhu, J.; Duan, Y.; Weng, W.; Wu, X. Pim1 Promotes Cell Proliferation and Regulates Glycolysis via Interaction with MYC in Ovarian Cancer. Onco Targets Ther. 2018, 11, 6647–6656. [Google Scholar] [CrossRef] [PubMed]
- Kulandavelu, S.; Karantalis, V.; Fritsch, J.; Hatzistergos, K.E.; Loescher, V.Y.; McCall, F.; Wang, B.; Bagno, L.; Golpanian, S.; Wolf, A.; et al. Pim1 Kinase Overexpression Enhances Ckit+ Cardiac Stem Cell Cardiac Repair Following Myocardial Infarction in Swine. J. Am. Coll. Cardiol. 2016, 68, 2454–2464. [Google Scholar] [CrossRef]
- Milanovic, M.; Yu, Y.; Schmitt, C.A. The Senescence–Stemness Alliance—A Cancer-Hijacked Regeneration Principle. Trends Cell Biol. 2018, 28, 1049–1061. [Google Scholar] [CrossRef]
- Triana-Martínez, F.; Loza, M.I.; Domínguez, E. Beyond Tumor Suppression: Senescence in Cancer Stemness and Tumor Dormancy. Cells 2020, 9, 346. [Google Scholar] [CrossRef]
- Ritschka, B.; Storer, M.; Mas, A.; Heinzmann, F.; Ortells, M.C.; Morton, J.P.; Sansom, O.J.; Zender, L.; Keyes, W.M. The Senescence-Associated Secretory Phenotype Induces Cellular Plasticity and Tissue Regeneration. Genes Dev. 2017, 31, 172–183. [Google Scholar] [CrossRef]
- Zheng, M.; Jacob, J.; Hung, S.-H.; Wang, J. The Hippo Pathway in Cardiac Regeneration and Homeostasis: New Perspectives for Cell-Free Therapy in the Injured Heart. Biomolecules 2020, 10, 1024. [Google Scholar] [CrossRef]
- Ferrari, R.; Rizzo, P. The Notch Pathway: A Novel Target for Myocardial Remodelling Therapy? Eur. Heart J. 2014, 35, 2140–2145. [Google Scholar] [CrossRef]
- Ramachandra, C.J.A.; Mehta, A.; Lua, C.H.; Chitre, A.; Ja, K.P.M.M.; Shim, W. ErbB Receptor Tyrosine Kinase: A Molecular Switch Between Cardiac and Neuroectoderm Specification in Human Pluripotent Stem Cells: ErbB RTKs Regulate Cardio- and Neurogenesis. Stem Cells 2016, 34, 2461–2470. [Google Scholar] [CrossRef] [PubMed]
- Geissler, A.; Ryzhov, S.; Sawyer, D.B. Neuregulins: Protective and Reparative Growth Factors in Multiple Forms of Cardiovascular Disease. Clin. Sci. 2020, 134, 2623–2643. [Google Scholar] [CrossRef]
- Rao, T.C.; Ma, V.P.-Y.; Blanchard, A.; Urner, T.M.; Grandhi, S.; Salaita, K.; Mattheyses, A.L. EGFR Activation Attenuates the Mechanical Threshold for Integrin Tension and Focal Adhesion Formation. J. Cell Sci. 2020, 133, jcs238840. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Xu, S.; Ji, Z.; Xu, H.; Zhao, W.; Xia, Z.; Xu, R. Mechanistic Study of MtROS-JNK-SOD2 Signaling in Bupivacaine-Induced Neuron Oxidative Stress. Aging (Albany NY) 2020, 12, 13463–13476. [Google Scholar] [CrossRef] [PubMed]
- D’Oria, R.; Schipani, R.; Leonardini, A.; Natalicchio, A.; Perrini, S.; Cignarelli, A.; Laviola, L.; Giorgino, F. The Role of Oxidative Stress in Cardiac Disease: From Physiological Response to Injury Factor. Oxid. Med. Cell Longev. 2020, 2020, 5732956. [Google Scholar] [CrossRef]
- Malkani, S.; Chin, C.R.; Cekanaviciute, E.; Mortreux, M.; Okinula, H.; Tarbier, M.; Schreurs, A.-S.; Shirazi-Fard, Y.; Tahimic, C.G.T.; Rodriguez, D.N.; et al. Circulating MiRNA Spaceflight Signature Reveals Targets for Countermeasure Development. Cell Rep. 2020, 33. [Google Scholar] [CrossRef]
- Kasiviswanathan, D.; Perumal, R.C.; Bhuvaneswari, S.; Kumar, P.; Sundaresan, L.; Philip, M.; Krishnankutty, S.P.; Chatterjee, S. Interactome of MiRNAs and Transcriptome of Human Umbilical Cord Endothelial Cells Exposed to Short-Term Simulated Microgravity. NPJ Microgravity 2020, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lenihan, D.J.; Anderson, S.A.; Lenneman, C.G.; Brittain, E.; Muldowney, J.A.S.; Mendes, L.; Zhao, P.Z.; Iaci, J.; Frohwein, S.; Zolty, R.; et al. A Phase I, Single Ascending Dose Study of Cimaglermin Alfa (Neuregulin 1β3) in Patients With Systolic Dysfunction and Heart Failure. JACC Basic Transl. Sci. 2016, 1, 576–586. [Google Scholar] [CrossRef]
- Nistri, S.; Sassoli, C.; Bani, D. Notch Signaling in Ischemic Damage and Fibrosis: Evidence and Clues from the Heart. Front. Pharmacol. 2017, 8. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, C.; Wang, C.; Hong, X.; Miao, J.; Liao, Y.; Zhou, L.; Liu, Y. An Essential Role for Wnt/β-Catenin Signaling in Mediating Hypertensive Heart Disease. Sci. Rep. 2018, 8, 1–14. [Google Scholar] [CrossRef]
- Fuentes, T.I.; Appleby, N.; Tsay, E.; Martinez, J.J.; Bailey, L.; Hasaniya, N.; Kearns-Jonker, M. Human Neonatal Cardiovascular Progenitors: Unlocking the Secret to Regenerative Ability. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Bioinformatics Enrichment Tools: Paths toward the Comprehensive Functional Analysis of Large Gene Lists. Nucleic Acids Res. 2009, 37, 1–13. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and Integrative Analysis of Large Gene Lists Using DAVID Bioinformatics Resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef]
- Vlachos, I.S.; Zagganas, K.; Paraskevopoulou, M.D.; Georgakilas, G.; Karagkouni, D.; Vergoulis, T.; Dalamagas, T.; Hatzigeorgiou, A.G. DIANA-MiRPath v3.0: Deciphering MicroRNA Function with Experimental Support. Nucleic Acids Res. 2015, 43, W460–W466. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Camberos, V.; Baio, J.; Mandujano, A.; Martinez, A.F.; Bailey, L.; Hasaniya, N.; Kearns-Jonker, M. The Impact of Spaceflight and Microgravity on the Human Islet-1+ Cardiovascular Progenitor Cell Transcriptome. Int. J. Mol. Sci. 2021, 22, 3577. https://doi.org/10.3390/ijms22073577
Camberos V, Baio J, Mandujano A, Martinez AF, Bailey L, Hasaniya N, Kearns-Jonker M. The Impact of Spaceflight and Microgravity on the Human Islet-1+ Cardiovascular Progenitor Cell Transcriptome. International Journal of Molecular Sciences. 2021; 22(7):3577. https://doi.org/10.3390/ijms22073577
Chicago/Turabian StyleCamberos, Victor, Jonathan Baio, Ana Mandujano, Aida F. Martinez, Leonard Bailey, Nahidh Hasaniya, and Mary Kearns-Jonker. 2021. "The Impact of Spaceflight and Microgravity on the Human Islet-1+ Cardiovascular Progenitor Cell Transcriptome" International Journal of Molecular Sciences 22, no. 7: 3577. https://doi.org/10.3390/ijms22073577
APA StyleCamberos, V., Baio, J., Mandujano, A., Martinez, A. F., Bailey, L., Hasaniya, N., & Kearns-Jonker, M. (2021). The Impact of Spaceflight and Microgravity on the Human Islet-1+ Cardiovascular Progenitor Cell Transcriptome. International Journal of Molecular Sciences, 22(7), 3577. https://doi.org/10.3390/ijms22073577




