Cellular Control of Protein Turnover via the Modification of the Amino Terminus
Abstract
1. Introduction
2. Methionine Aminopeptidase as a Processing Enzyme towards Protein Destabilization
3. Processing by Met Aminopeptidases: Exceptions from the Rule
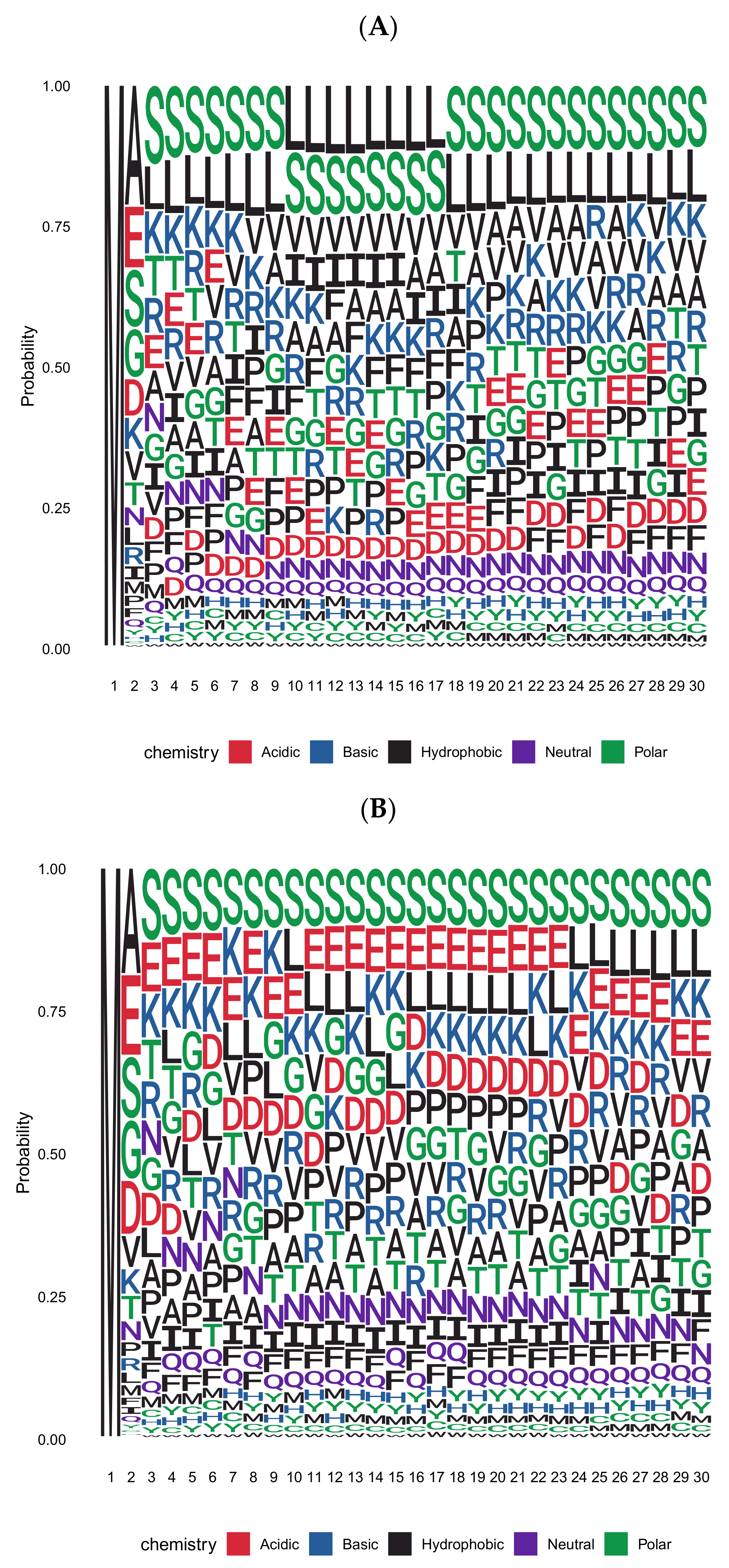
4. Cleavage of Signal Peptides Can Be Followed by Escape of Proteins from the ER
5. Caspases, Metacaspases, and Other Endoproteases Related to Cell Death Programs
6. Bacterial Effectors
7. Other Specialized Endoproteases
8. No Proteolytic Processing of Polypeptides Emerging from the Ribosome: Can Met Be a Destabilizing Residue If Followed by an Amino Acid with Hydrophobic Side Chain?
9. Blocking the Alpha-Amino Group Can Generate a Distinct Set of N-Degrons
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dissmeyer, N. Conditional protein function via N-degron pathway-mediated proteolysis in stress physiology. Annu. Rev. Plant Biol. 2019, 70, 83–117. [Google Scholar] [CrossRef]
- Millar, A.H.; Heazlewood, J.L.; Giglione, C.; Holdsworth, M.J.; Bachmair, A.; Schulze, W.X. The scope, functions, and dynamics of posttranslational modifications. Annu. Rev. Plant Biol. 2019, 70, 119–151. [Google Scholar] [CrossRef]
- Varshavsky, A. N-degron and C-degron pathways of protein degradation. Proc. Natl. Acad. Sci. USA 2019, 116, 358–366. [Google Scholar] [CrossRef]
- Tasaki, T.; Sriram, S.M.; Park, K.S.; Kwon, Y.T. The N-end rule pathway. Annu. Rev. Biochem. 2012, 81, 261–289. [Google Scholar] [CrossRef]
- Gibbs, D.J.; Bacardit, J.; Bachmair, A.; Holdsworth, M.J. The eukaryotic N-end rule pathway: Conserved mechanisms and diverse functions. Trends Cell Biol. 2014, 24, 603–611. [Google Scholar] [CrossRef]
- Perrar, A.; Dissmeyer, N.; Huesgen, P.F. New beginnings and new ends: Methods for large-scale characterization of protein termini and their use in plant biology. J. Exp. Bot 2019, 70, 2021–2038. [Google Scholar] [CrossRef] [PubMed]
- Bradshaw, R.A.; Brickey, W.W.; Walker, K.W. N-terminal processing: The methionine aminopeptidase and N-alpha-acetyl transferase families. Trends Biochem. Sci. 1998, 23, 263–267. [Google Scholar] [CrossRef]
- Giglione, C.; Boularot, A.; Meinnel, T. Protein N-terminal methionine excision. Cell Mol. Life Sci. 2004, 61, 1455–1474. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.; Giglione, C.; Pierre, M.; Espagne, C.; Meinnel, T. Functional and developmental impact of cytosolic protein N-terminal methionine excision in Arabidopsis. Plant Physiol. 2005, 137, 623–637. [Google Scholar] [CrossRef]
- Gibbs, D.J.; Lee, S.C.; Isa, N.M.; Gramuglia, S.; Fukao, T.; Bassel, G.W.; Correia, C.S.; Corbineau, F.; Theodoulou, F.L.; Bailey-Serres, J.; et al. Homeostatic response to hypoxia is regulated by the N-end rule pathway in plants. Nature 2011, 479, 415–418. [Google Scholar] [CrossRef]
- Licausi, F.; Kosmacz, M.; Weits, D.A.; Giuntoli, B.; Giorgi, F.M.; Voesenek, L.A.; Perata, P.; van Dongen, J.T. Oxygen sensing in plants is mediated by an N-end rule pathway for protein destabilization. Nature 2011, 479, 419–422. [Google Scholar] [CrossRef]
- Gibbs, D.J.; Tedds, H.M.; Labandera, A.M.; Bailey, M.; White, M.D.; Hartman, S.; Sprigg, C.; Mogg, S.L.; Osborne, R.; Dambire, C.; et al. Oxygen-dependent proteolysis regulates the stability of angiosperm polycomb repressive complex 2 subunit VERNALIZATION 2. Nat. Commun. 2018, 9, 5438. [Google Scholar] [CrossRef]
- Weits, D.A.; Kunkowska, A.B.; Kamps, N.C.W.; Portz, K.M.S.; Packbier, N.K.; Nemec Venza, Z.; Gaillochet, C.; Lohmann, J.U.; Pedersen, O.; van Dongen, J.T.; et al. An apical hypoxic niche sets the pace of shoot meristem activity. Nature 2019, 569, 714–717. [Google Scholar] [CrossRef] [PubMed]
- Weits, D.A.; Giuntoli, B.; Kosmacz, M.; Parlanti, S.; Hubberten, H.M.; Riegler, H.; Hoefgen, R.; Perata, P.; van Dongen, J.T.; Licausi, F. Plant cysteine oxidases control the oxygen-dependent branch of the N-end-rule pathway. Nat. Commun. 2014, 5, 3425. [Google Scholar] [CrossRef] [PubMed]
- Choi, W.S.; Jeong, B.C.; Joo, Y.J.; Lee, M.R.; Kim, J.; Eck, M.J.; Song, H.K. Structural basis for the recognition of N-end rule substrates by the UBR box of ubiquitin ligases. Nat. Struct. Mol. Biol. 2010, 17, 1175–1181. [Google Scholar] [CrossRef] [PubMed]
- White, M.D.; Kamps, J.J.A.G.; East, S.; Taylor Kearney, L.J.; Flashman, E. The plant cysteine oxidases from Arabidopsis thaliana are kinetically tailored to act as oxygen sensors. J. Biol. Chem. 2018, 293, 11786–11795. [Google Scholar] [CrossRef] [PubMed]
- Masson, N.; Keeley, T.P.; Giuntoli, B.; White, M.D.; Puerta, M.L.; Perata, P.; Hopkinson, R.J.; Flashman, E.; Licausi, F.; Ratcliffe, P.J. Conserved N-terminal cysteine dioxygenases transduce responses to hypoxia in animals and plants. Science 2019, 365, 65–69. [Google Scholar] [CrossRef]
- Hu, R.G.; Sheng, J.; Qi, X.; Xu, Z.; Takahashi, T.T.; Varshavsky, A. The N-end rule pathway as a nitric oxide sensor controlling the levels of multiple regulators. Nature 2005, 437, 981–986. [Google Scholar] [CrossRef]
- Gibbs, D.J.; Md Isa, N.; Movahedi, M.; Lozano-Juste, J.; Mendiondo, G.M.; Berckhan, S.; Marin-de la Rosa, N.; Vicente Conde, J.; Sousa Correia, C.; Pearce, S.P.; et al. Nitric Oxide Sensing in Plants Is Mediated by Proteolytic Control of Group VII ERF Transcription Factors. Mol. Cell 2014, 53, 369–379. [Google Scholar] [CrossRef]
- Hartman, S.; Liu, Z.; van Veen, H.; Vicente, J.; Reinen, E.; Martopawiro, S.; Zhang, H.; van Dongen, N.; Bosman, F.; Bassel, G.W.; et al. Ethylene-mediated nitric oxide depletion pre-adapts plants to hypoxia stress. Nat. Commun. 2019, 10, 4020. [Google Scholar] [CrossRef]
- Chen, S.-J.; Wu, X.; Wadas, B.; Oh, J.-H.; Varshavsky, A. An N-end rule pathway that recognizes proline and destroys gluconeogenic enzymes. Science 2017, 355, eaal3655. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Flockhart, I.; Vinayagam, A.; Bergwitz, C.; Berger, B.; Perrimon, N.; Mohr, S.E. An integrative approach to ortholog prediction for disease-focused and other functional studies. BMC Bioinform. 2011, 12, 357. [Google Scholar] [CrossRef]
- Chen, S.J.; Melnykov, A.; Varshavsky, A. Evolution of Substrates and Components of the Pro/N-Degron Pathway. Biochemistry 2020, 59, 582–593. [Google Scholar] [CrossRef] [PubMed]
- Timms, R.T.; Zhang, Z.; Rhee, D.Y.; Harper, J.W.; Koren, I.; Elledge, S.J. A glycine-specific N-degron pathway mediates the quality control of protein N-myristoylation. Science 2019, 365, eaaw4912. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y. A conserved motif: Potentiating apoptosis through IAP-binding. Cell Death Differ. 2002, 9, 93–95. [Google Scholar] [CrossRef] [PubMed]
- Kats, I.; Khmelinskii, A.; Kschonsak, M.; Huber, F.; Knieß, R.A.; Bartosik, A.; Knop, M. Mapping Degradation Signals and Pathways in a Eukaryotic N-terminome. Mol. Cell 2018, 70, 488–501. [Google Scholar] [CrossRef]
- Nguyen, K.T.; Kim, J.M.; Park, S.E.; Hwang, C.S. N-terminal methionine excision of proteins creates tertiary destabilizing N-degrons of the Arg/N-end rule pathway. J. Biol. Chem. 2019, 294, 4464–4476. [Google Scholar] [CrossRef]
- Vicente, J.; Mendiondo, G.M.; Pauwels, J.; Pastor, V.; Izquierdo, Y.; Naumann, C.; Movahedi, M.; Rooney, D.; Gibbs, D.J.; Smart, K.; et al. Distinct branches of the N-end rule pathway modulate the plant immune response. New Phytol. 2018, 221, 988–1000. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Yu, F.; Xie, Q. Insights into endoplasmic reticulum-associated degradation in plants. New Phytol. 2020, 226, 345–350. [Google Scholar] [CrossRef]
- Hwang, J.; Qi, L. Quality control in the endoplasmic reticulum: Crosstalk between ERAD and UPR pathways. Trends Biochem. Sci. 2018, 43, 593–605. [Google Scholar] [CrossRef]
- Cha-Molstad, H.; Sung, K.S.; Hwang, J.; Kim, K.A.; Yu, J.E.; Yoo, Y.D.; Jang, J.M.; Han, D.H.; Molstad, M.; Kim, J.G.; et al. Amino-terminal arginylation targets endoplasmic reticulum chaperone BiP for autophagy through p62 binding. Nat. Cell Biol. 2015, 17, 917–929. [Google Scholar] [CrossRef]
- Shim, S.M.; Choi, H.R.; Sung, K.W.; Lee, Y.J.; Kim, S.T.; Kim, D.; Mun, S.R.; Hwang, J.; Cha-Molstad, H.; Ciechanover, A.; et al. The endoplasmic reticulum-residing chaperone BiP is short-lived and metabolized through N-terminal arginylation. Sci. Signal. 2018, 11, eaan0630. [Google Scholar] [CrossRef] [PubMed]
- Kwon, D.H.; Park, O.H.; Kim, L.; Jung, Y.O.; Park, Y.; Jeong, H.; Hyun, J.; Kim, Y.K.; Song, H.K. Insights into degradation mechanism of N-end rule substrates by p62/SQSTM1 autophagy adapter. Nat. Commun. 2018, 9, 3291. [Google Scholar] [CrossRef] [PubMed]
- Ji, C.H.; Kim, H.Y.; Heo, A.J.; Lee, S.H.; Lee, M.J.; Kim, S.B.; Srinivasrao, G.; Mun, S.R.; Cha-Molstad, H.; Ciechanover, A.; et al. The N-Degron Pathway Mediates ER-phagy. Mol. Cell 2019, 75, 1058–1072. [Google Scholar] [CrossRef] [PubMed]
- Ji, C.; Zhou, J.; Guo, R.; Lin, Y.; Kung, C.-H.; Hu, S.; Ng, W.Y.; Zhuang, X.; Jiang, L. AtNBR1 Is a Selective Autophagic Receptor for AtExo70E2 in Arabidopsis. Plant Physiol. 2020, 184, 777–791. [Google Scholar] [CrossRef]
- Svenning, S.; Lamark, T.; Krause, K.; Johansen, T. Plant NBR1 is a selective autophagy substrate and a functional hybrid of the mammalian autophagic adapters NBR1 and p62/SQSTM1. Autophagy 2011, 7, 993–1010. [Google Scholar] [CrossRef]
- Zientara-Rytter, K.; Sirko, A. Significant role of PB1 and UBA domains in multimerization of Joka2, a slelctive autophagy cargo receptor from tobacco. Front. Plant Sci. 2014, 5, 13. [Google Scholar] [CrossRef]
- Almagro-Armenteros, J.J.; Sonderby, C.K.; Petersen, K.N.; Winther, O.; Brunak, S.; von Heijne, G.; Nielsen, H. SignalP 5.0 improves signal peptide predictions using deep neural networks. Nat. Biotechnol. 2019, 37, 420–423. [Google Scholar] [CrossRef]
- Willems, P.; Horne, A.; Van Parys, T.; Goormachtig, S.; De Smet, I.; Botzki, A.; Van Breusegem, F.; Gevaert, K. The Plant PTM Viewer, a central resource for exploring plant protein modifications. Plant J. 2019, 99, 752–762. [Google Scholar] [CrossRef] [PubMed]
- Ditzel, M.; Wilson, R.; Tenev, T.; Zachariou, A.; Paul, A.; Deas, E.; Meier, P. Degradation of DIAP1 by the N-end rule pathway is essential for regulating apoptosis. Nat. Cell Biol. 2003, 5, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Vasudevan, D.; Ryoo, H.D. Regulation of Cell Death by IAPs and Their Antagonists. Curr. Top. Dev. Biol. 2015, 114, 185–208. [Google Scholar]
- Domingues, C.; Ryoo, H.D. Drosophila BRUCE inhibits apoptosis through non-lysine ubiquitination of the IAP-antagonist REAPER. Cell Death Differ. 2012, 19, 470–477. [Google Scholar] [CrossRef]
- Kumar, S.; van Raam, B.J.; Salvesen, G.S.; Cieplak, P. Caspase cleavage sites of the human proteome: CaspDB, a database of predicted substrates. PLoS ONE 2014, 9, e110539. [Google Scholar] [CrossRef] [PubMed]
- Piatkov, K.I.; Brower, C.S.; Varshavsky, A. The N-end rule pathway counteracts cell death by destroying proapoptotic protein fragments. Proc. Natl. Acad. Sci. USA 2012, 109, E1839–E1847. [Google Scholar] [CrossRef] [PubMed]
- Tenev, T.; Ditzel, M.; Zachariou, A.; Meier, P. The antiapoptotic activity of insect IAPs requires activation by an evolutionarily conserved mechanism. Cell Death Differ. 2007, 14, 1191–1201. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.-M.; Butterwirth, M.; MacFarlane, M.; Dubiel, W.; Ciechanover, A.; Cohen, G.M. Caspase activation inhibits proteasome function during apoptosis. Mol. Cell 2004, 14, 81–93. [Google Scholar] [CrossRef]
- Coll, N.S.; Vercammen, D.; Smidler, A.; Clover, C.; Van Breusegem, F.; Dangl, J.L.; Epple, P. Arabidopsis type I metacaspases control cell death. Science 2010, 330, 1393–1397. [Google Scholar] [CrossRef]
- Salguero-Linares, J.; Coll, N.S. Plant proteases in the control of the hypersensitive response. J. Exp. Bot. 2019, 70, 2087–2095. [Google Scholar] [CrossRef]
- Tsiatsiani, L.; Timmerman, E.; De Bock, P.J.; Vercammen, D.; Stael, S.; van de Cotte, B.; Staes, A.; Goethals, M.; Beunens, T.; Van Damme, P.; et al. The Arabidopsis Metacaspase9 Degradome. Plant Cell 2013, 25, 2831–2847. [Google Scholar] [CrossRef]
- Yamada, K.; Basak, A.K.; Goto-Yamada, S.; Tarnawska-Glatt, K.; Hara-Nishimura, I. Vacuolar processing enzymes in the plant life cyle. New Phytol. 2020, 226, 21–31. [Google Scholar] [CrossRef]
- Vorster, B.J.; Culli, C.A.; Kunert, K.J. Plant vacuolar processing enzymes. Front. Plant Sci. 2019, 10, 479. [Google Scholar] [CrossRef] [PubMed]
- Chichkova, N.V.; Shaw, J.; Galiullina, R.A.; Drury, G.E.; Tuzhikov, A.I.; Kim, S.H.; Kalkum, M.; Hong, T.B.; Goshikova, E.N.; Torrance, L.; et al. Phytaspase, a relocalisable cell death promoting plant protase with caspase specificity. EMBO J. 2010, 29, 1149–1161. [Google Scholar] [CrossRef] [PubMed]
- Hara-Nishimura, I.; Hatsugai, N. The role of vacuole in plant cell death. Cell Death Differ. 2011, 18, 1298–1304. [Google Scholar] [CrossRef]
- Hatsugai, N.; Iwasaki, S.; Tamura, K.; Kondo, M.; Fuji, K.; Ogasawara, K.; Nishimura, M.; Hara-Nishimura, I. A novel membrane fusion-mediated plant immunity against bacterial pathogens. Genes Dev. 2009, 23, 2496–2506. [Google Scholar] [CrossRef]
- Yoshida, S.; Ito, M.; Callis, J.; Nishida, I.; Watanabe, A. A delayed leaf senescence mutant is defective in arginyl-tRNA: Protein arginyltransferase, a component of the N-end rule pathway in Arabidopsis. Plant J. 2002, 32, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-G.; Taylor, K.W.; Mudgett, M.B. Comparative analysis of the XopD type III secretion (T3S) effector family in plant pathogenic bacteria. Mol. Plant Pathol. 2011, 12, 715–730. [Google Scholar] [CrossRef] [PubMed]
- Mackey, D.; Belkhadir, Y.; Alonso, J.M.; Ecker, J.R.; Dangl, J.L. Arabidopsis RIN4 is a target of the type III virulence effector AvrRpt2 and modulates RPS2-mediated resistance. Cell 2003, 112, 379–389. [Google Scholar] [CrossRef]
- Takemoto, D.; Jones, D.A. Membrane release and destabilization of Arabidopsis RIN4 following cleavage by Pseudomonas syringae AvrRpt2. Mol. Plant Microbe Interact. 2005, 18, 1258–1268. [Google Scholar] [CrossRef]
- Goslin, K.; Eschen-Lippold, L.; Naumann, C.; Linster, E.; Sorel, M.; Klecker, M.; de Marchi, R.; Kind, A.; Wirtz, M.; Lee, J.; et al. Differential N-end rule degradation of RIN4/NOI fragments generated by the AvrRpt2 effector protease. Plant Physiol. 2019, 180, 2272–2289. [Google Scholar] [CrossRef]
- Kumar, R. Separase: Function beyond cohesin cleavage and an emerging oncogene. J. Cell Biochem. 2017, 118, 1283–1299. [Google Scholar] [CrossRef] [PubMed]
- Rao, H.; Uhlmann, F.; Nasmyth, K.; Varshavsky, A. Degradation of a cohesin subunit by the N-end rule pathway is essential for chromosome stability. Nature 2001, 410, 955–959. [Google Scholar] [CrossRef]
- Piatkov, K.I.; Oh, J.H.; Liu, Y.; Varshavsky, A. Calpain-generated natural protein fragments as short-lived substrates of the N-end rule pathway. Proc. Natl. Acad. Sci. USA 2014, 111, E817–E826. [Google Scholar] [CrossRef]
- Johnson, K.L.; Faulkner, C.; Jeffree, C.E.; Ingram, G.C. The phytocalpain defective kernel 1 is a novel Arabidopsis growth regulator whose activity is regulated by proteolytic processing. Plant Cell 2008, 20, 2619–2630. [Google Scholar] [CrossRef] [PubMed]
- Tran, D.; Galletti, R.; Neumann, E.D.; Dubois, A.; Sharif-Naeini, R.; Geitmann, A.; Frachisse, J.M.; Hamant, O.; Ingram, G.C. A mechanosensitive Ca2+ channel activity is dependent on the developmental regulator DEK1. Nat. Commun. 2017, 8, 1009. [Google Scholar] [CrossRef] [PubMed]
- Guerringue, Y.; Thomine, S.; Frachisse, J.M. Sensing and transducing forces in plants with MSL10 and DEK1 mechanosensors. FEBS Lett. 2018, 592, 1968–1979. [Google Scholar] [CrossRef]
- Mogk, A.; Schmidt, R.; Bukau, B. The N-end rule pathway for regulated proteolysis: Prokaryotic and eukaryotic strategies. Trends Cell Biol. 2007, 17, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Tasaki, T.; Zakrzewska, A.; Dudgeon, D.D.; Jiang, Y.; Lazo, J.S.; Kwon, Y.T. The substrate recognition domains of the N-end rule pathway. J. Biol. Chem. 2009, 284, 1884–1895. [Google Scholar] [CrossRef]
- Kim, H.K.; Kim, R.R.; Oh, J.H.; Cho, H.; Varshavsky, A.; Hwang, C.S. The N-Terminal Methionine of Cellular Proteins as a Degradation Signal. Cell 2014, 156, 158–169. [Google Scholar] [CrossRef]
- Hwang, C.S.; Shemorry, A.; Varshavsky, A. N-terminal acetylation of cellular proteins creates specific degradation signals. Science 2010, 327, 973–977. [Google Scholar] [CrossRef] [PubMed]
- Linster, E.; Stephan, I.; Bienvenut, W.V.; Maple-Grodem, J.; Myklebust, L.M.; Huber, M.; Reichelt, M.; Sticht, C.; Geir Moller, S.; Meinnel, T.; et al. Downregulation of N-terminal acetylation triggers ABA-mediated drought responses in Arabidopsis. Nat. Commun. 2015, 6, 7640. [Google Scholar] [CrossRef]
- Bailey, M.; Ivanauskaite, A.; Grimmer, J.; Akintewe, O.; Payne, A.C.; Osborne, R.; Labandera, A.M.; Etherington, R.D.; Rantala, M.; Baginsky, S.; et al. The Arabidopsis NOT4A E3 ligase promotes PGR3 expression and regulates chloroplast translation. Nat. Commun. 2021, 12, 251. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-M.; Seok, O.-H.; Ju, S.; Heo, J.-E.; Yeom, J.; Kim, D.-S.; Yoo, J.-Y.; Varshavsky, A.; Lee, C.P.; Hwang, C.-S. Formyl-Methionine as an N-degron of a eukaryotic N-end rule pathway. Science 2018, 362, eaat0174. [Google Scholar] [CrossRef] [PubMed]
- Kraft, E.; Bostick, M.; Jacobsen, S.E.; Callis, J. ORTH/VIM proteins that regulate DNA methylation are functional ubiquitin E3 ligases. Plant J. 2008, 56, 704–715. [Google Scholar] [CrossRef] [PubMed]
| Exposed Residue | Exposing Protease | Predicted Fate |
|---|---|---|
| Cys | Met aminopeptidase (Met AP) | Oxidation, arginylation, ubiquitylation, proteasomal degradation of protein |
| Pro | Met AP | Depending on sequence context: Pro N-degron |
| Gly | Met AP | Depending on sequence context: Gly N-degron |
| Ala | Met AP/mitochondrial protease | Depending on sequence context: Ala N-degron |
| Asn | Met AP (depending on sequence context; blocked by Met acetylation), other endoproteases? | De-amidation, arginylation, ubiquitylation, proteasomal degradation of protein |
| Gln | Met AP (depending on sequence context; blocked by Met acetylation), Metacaspases, other endoproteases? | De-amidation, arginylation, ubiquitylation, proteasomal degradation of protein |
| Asp | Caspase-like proteases, other endoproteases | Arginylation, ubiquitylation, proteasomal degradation of protein |
| Glu | Signal peptidase, other endoproteases? | Arginylation, ubiquitylation, proteasomal degradation of protein |
| Lys | Metacaspases | Ubiquitylation, proteasomal degradation of protein |
| Arg | Metacaspases | Ubiquitylation, proteasomal degradation of protein |
| Met Φ 1 | No processing | Depending on sequence context: ubiquitylation, proteasomal degradation of protein |
| Amino-Terminal Dipeptide | Abundance in the Compete Proteome | Abundance (%) | Abundance in Nuclear/Cytoplasmic Proteins | Abundance (%) | General Frequency of Amino Acid 2 in the Proteome |
|---|---|---|---|---|---|
| MA | 5891 | 21.5% | 1917 | 18.7% | 6.3% |
| ME | 3059 | 11.1% | 1469 | 14.3% | 6.7% |
| MS | 2898 | 10.6% | 1140 | 11.1% | 9.1% |
| MG | 2441 | 8.9% | 1090 | 10.6% | 6.4% |
| MD | 1874 | 6.8% | 988 | 9.6% | 5.4% |
| MK | 1629 | 5.9% | 479 | 4.7% | 6.4% |
| MV | 1519 | 5.5% | 598 | 5.8% | 6.7% |
| MT | 1216 | 4.4% | 458 | 4.5% | 5.1% |
| MN | 976 | 3.6% | 378 | 3.7% | 4.4% |
| ML | 951 | 3.5% | 225 | 2.2% | 9.5% |
| MR | 931 | 3.4% | 259 | 2.5% | 5.4% |
| MI | 738 | 2.7% | 162 | 1.6% | 5.3% |
| MM | 692 | 2.5% | 225 | 2.2% | 2.5% |
| MP | 601 | 2.2% | 285 | 2.8% | 4.8% |
| MF | 544 | 2.0% | 167 | 1.6% | 4.3% |
| MQ | 509 | 1.9% | 155 | 1.5% | 3.5% |
| MY | 340 | 1.2% | 101 | 1.0% | 2.8% |
| MH | 208 | 0.8% | 51 | 0.5% | 2.3% |
| MC | 205 | 0.7% | 68 | 0.7% | 1.9% |
| MW | 164 | 0.6% | 41 | 0.4% | 1.2% |
| Sum | 27,386 | 10,256 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Winter, N.; Novatchkova, M.; Bachmair, A. Cellular Control of Protein Turnover via the Modification of the Amino Terminus. Int. J. Mol. Sci. 2021, 22, 3545. https://doi.org/10.3390/ijms22073545
Winter N, Novatchkova M, Bachmair A. Cellular Control of Protein Turnover via the Modification of the Amino Terminus. International Journal of Molecular Sciences. 2021; 22(7):3545. https://doi.org/10.3390/ijms22073545
Chicago/Turabian StyleWinter, Nikola, Maria Novatchkova, and Andreas Bachmair. 2021. "Cellular Control of Protein Turnover via the Modification of the Amino Terminus" International Journal of Molecular Sciences 22, no. 7: 3545. https://doi.org/10.3390/ijms22073545
APA StyleWinter, N., Novatchkova, M., & Bachmair, A. (2021). Cellular Control of Protein Turnover via the Modification of the Amino Terminus. International Journal of Molecular Sciences, 22(7), 3545. https://doi.org/10.3390/ijms22073545






