Therapeutic Potential for Regulation of the Nuclear Factor Kappa-B Transcription Factor p65 to Prevent Cellular Senescence and Activation of Pro-Inflammatory in Mesenchymal Stem Cells
Abstract
1. Introduction
2. Results
2.1. p65 Is Involved in the Cellular Senescence Establishment and Proinflammatory Activation in MSCs
2.2. Inhibition of p65 Prevents the Cellular Senescence Establishment and Proinflammatory Activation in MSCs
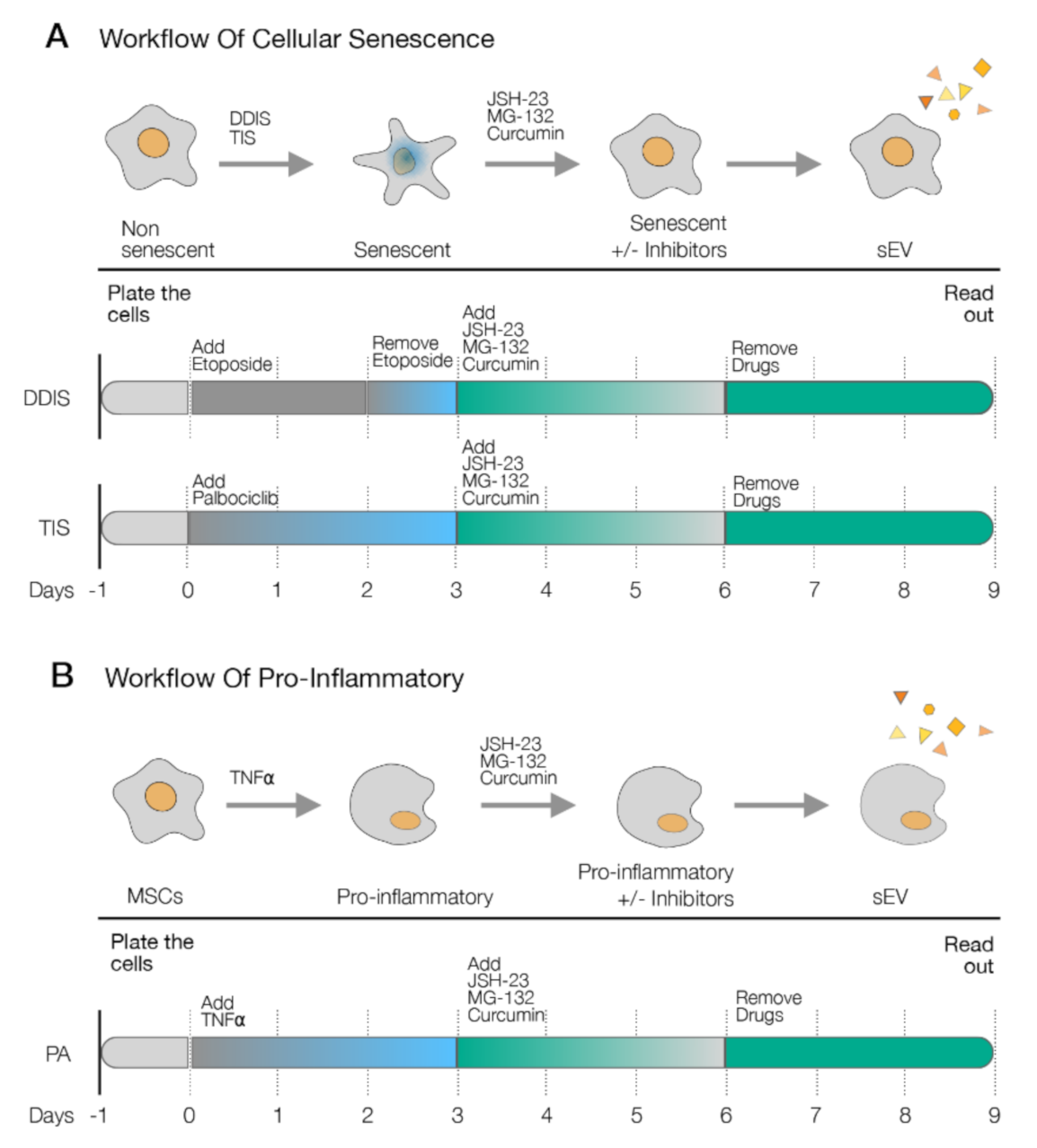
2.3. p65 Pathway Modulate the Production of sEV in Senescent and Pro-Inflammatory-Activated MSCs
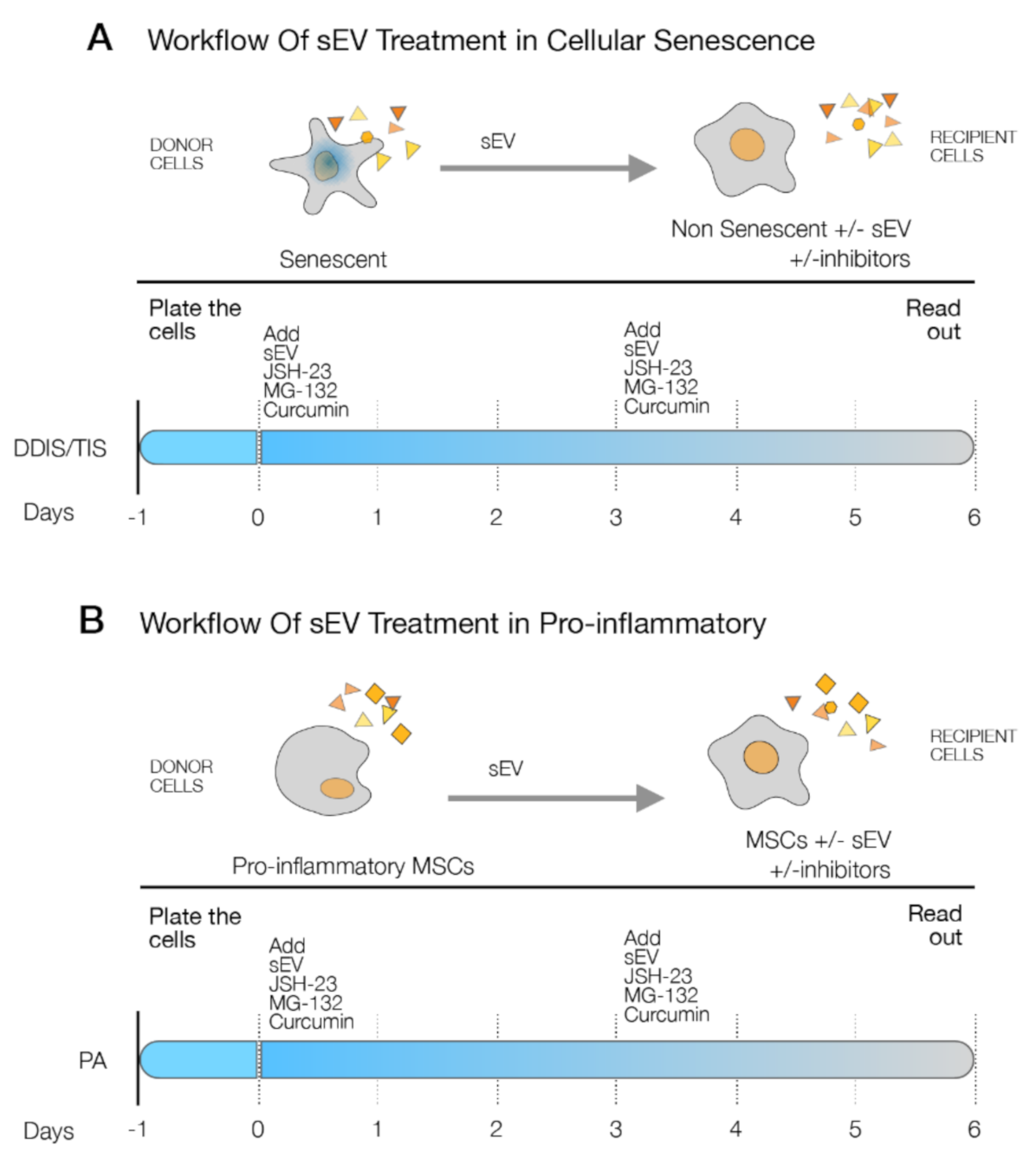
2.4. p65 Is a Common Pathway Involved in the Cellular Senescence and Proinflammatory Activation Paracrine through sEV
3. Discussion
4. Materials and Methods
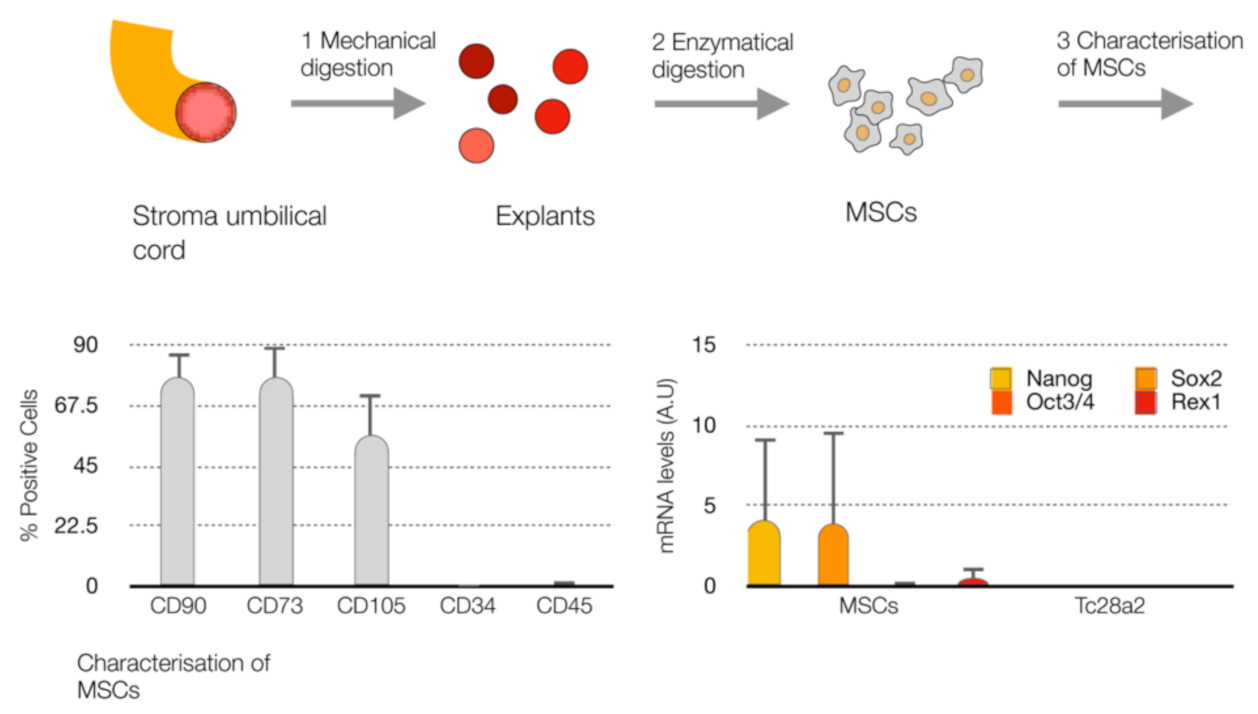
4.1. Cell Culture
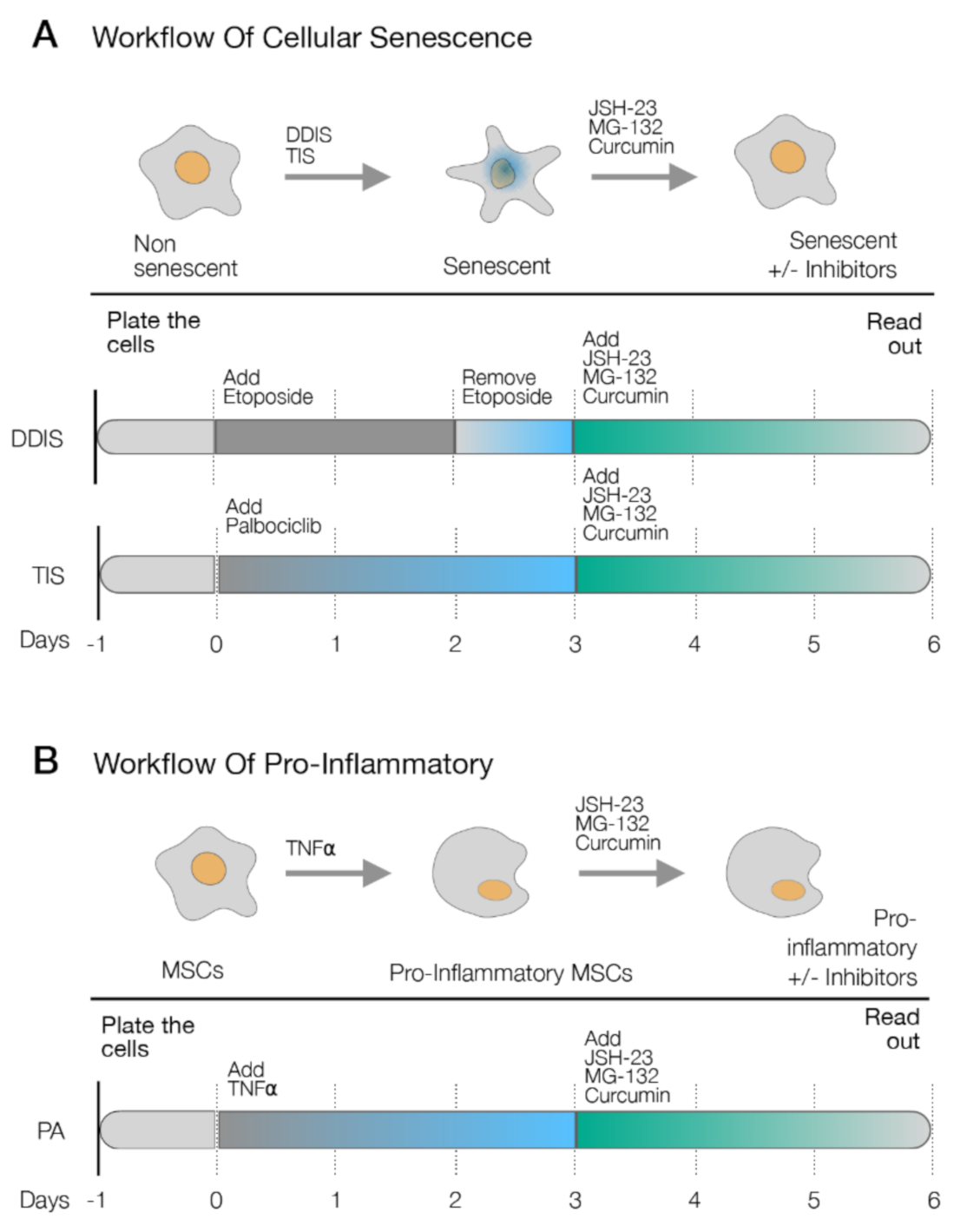
4.2. Cellular Senescence Induction in UC-MSCs
4.3. Pro-Inflammatory Activation in UC-MSCs
4.4. Treatment with Drugs
4.5. Treatment with sEV
4.6. Cell Proliferation Assay
4.7. Crystal Violet Staining
4.8. β-GALACTOSIDASE Assay
4.9. Isolation Extracellular Vesicles
4.10. RNA Isolation, Synthesis cDNA and qPCR-RT
4.11. Flow Cytometry
4.12. ELISA
4.13. Nanoparticle Tracking Analysis
4.14. Statistics
 p < 0.05 and
p < 0.05 and  p < 0.01 were considered statistically significant.
p < 0.01 were considered statistically significant.5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Drug | Concentration | Source | Cat. No |
|---|---|---|---|
| Etoposide | 1 µM | MedChemExpress | HY-13629 |
| Palbociclib (CDK4/6i) | 1 µM | MedChemExpress | HY-50767 |
| TNF-A α | 10 ng/mL | ImmunoTools | 11343013 |
| JSH-23 | 10 µM | MedChemExpress | HY-13982 |
| MG-132 | 10 µM | MedChemExpress | HY-13259 |
| Curcumin | 10 µM | Sigma-Aldrich | 458-37-7 |
| Antibody | Dilution | Source |
|---|---|---|
| CD90-FITC | 1:100 | BD Pharmigen |
| CD45-FITC | 1:100 | BD Pharmigen |
| CD73-PE | 1:100 | BD Pharmigen |
| CD105-FITC | 1:100 | BD Pharmigen |
| CD34-PE | 1:100 | BD Pharmigen |
| IgG1-PE | 1:100 | St. Cruz Biotechnology’s |
| IgG1-FITC | 1:100 | St. Cruz Biotechnology’s |
| Target | Species | Forward Primer | Reverse Primer |
|---|---|---|---|
| p65 | Human | ttcccgatctgagtccaggt | gcttgtctcgggtttctgga |
| Nanog | Human | atgcctcacacggagactgt | aagtgggttgtttgcctttg |
| Sox2 | Human | ctccgggacatgatcagc | ggtagtgctgggacatgtgaa |
| Rex1 | Human | cgcaatcgcttgtcctcagagt | gctctcaacgaacgctttccca |
| Oct4 | Human | ctcctggagggccaggaatc | atatacacaggccgatgtgg |
| β-actin | Human | agagctacgagctgcctgac | ggatgccacaggactcca |
References
- Fernández-Pernas, P.; Fafián-Labora, J.; Lesende-Rodriguez, I.; Mateos, J.; De la Fuente, A.; Fuentes, I.; Santos, J.D.T.; García, F.B.; Arufe, M.C. 3,3′,5-triiodo-L-thyronine Increases In Vitro Chondrogenesis of Mesenchymal Stem Cells from Human Umbilical Cord Stroma Through SRC2. J. Cell. Biochem. 2016, 117, 2097–2108. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Pernas, P.; Rodríguez-Lesende, I.; De La Fuente, A.; Mateos, J.; Fuentes, I.; De Toro, J.; Blanco, F.J.; Arufe, M.C. CD105+ mesenchymal stem cells migrate into osteoarthritis joint: An animal model. PLoS ONE 2017, 12, e0188072. [Google Scholar] [CrossRef] [PubMed]
- Fafián-Labora, J.A.; Fernández-Pernas, P.; Fuentes, I.; De Toro, J.; Oreiro, N.; Sangiao-Alvarellos, S.; Mateos, J.; Arufe, M.C. Influence of age on rat bone-marrow mesenchymal stem cells potential. Sci. Rep. 2015, 5, 16765. [Google Scholar] [CrossRef] [PubMed]
- Fafián-Labora, J.; Lesende-Rodriguez, I.; Fernández-Pernas, P.; Sangiao-Alvarellos, S.; Monserrat, L.; Arntz, O.J.; Van De Loo, F.A.J.; Mateos, J.; Arufe, M.C. Effect of age on pro-inflammatory miRNAs contained in mesenchymal stem cell-derived extracellular vesicles. Sci. Rep. 2017, 7, srep43923. [Google Scholar] [CrossRef] [PubMed]
- Fafián-Labora, J.A.; Morente-López, M.; Arufe, M.C. Effect of aging on behaviour of mesenchymal stem cells. World J. Stem Cells 2019, 11, 337–346. [Google Scholar] [CrossRef]
- Franceschi, C.; Garagnani, P.; Vitale, G.; Capri, M.; Salvioli, S. Inflammaging and ‘Garb-aging’. Trends Endocrinol. Metab. 2017, 28, 199–212. [Google Scholar] [CrossRef]
- Ou, H.; Hoffmann, R.; González-López, C.; Doherty, G.J.; Korkola, J.E.; Muñoz-Espín, D. Cellular senescence in cancer: From mechanisms to detection. Mol. Oncol. 2020. [Google Scholar] [CrossRef]
- Wang, B.; Kohli, J.; Demaria, M. Senescent Cells in Cancer Therapy: Friends or Foes? Trends Cancer 2020, 6, 838–857. [Google Scholar] [CrossRef]
- Fafián-Labora, J.A.; O’Loghlen, A. Classical and Nonclassical Intercellular Communication in Senescence and Ageing. Trends Cell Biol. 2020, 30, 628–639. [Google Scholar] [CrossRef]
- Borghesan, M.; Fafián-Labora, J.; Eleftheriadou, O.; Carpintero-Fernández, P.; Paez-Ribes, M.; Vizcay-Barrena, G.; Swisa, A.; Kolodkin-Gal, D.; Ximénez-Embún, P.; Lowe, R.; et al. Small Extracellular Vesicles Are Key Regulators of Non-cell Autonomous Intercellular Communication in Senescence via the Interferon Protein IFITM3. Cell Rep. 2019, 27, 3956–3971. [Google Scholar] [CrossRef]
- Russell, A.E.; Sneider, A.; Witwer, K.W.; Bergese, P.; Bhattacharyya, S.N.; Cocks, A.; Cocucci, E.; Erdbrügger, U.; Falcon-Perez, J.M.; Freeman, D.W.; et al. Biological membranes in EV biogenesis, stability, uptake, and cargo transfer: An ISEV position paper arising from the ISEV membranes and EVs workshop. J. Extracell. Vesicles 2019, 8, 1684862. [Google Scholar] [CrossRef]
- Fafián-Labora, J.; Morente-López, M.; Sánchez-Dopico, M.J.; Arntz, O.J.; Van De Loo, F.A.J.; De Toro, J.; Arufe, M.C. Influence of mesenchymal stem cell-derived extracellular vesicles in vitro and their role in ageing. Stem Cell Res. Ther. 2020, 11, 1–12. [Google Scholar] [CrossRef]
- Fafián-Labora, J.A.; Rodríguez-Navarro, J.A.; O’Loghlen, A. Small Extracellular Vesicles Have GST Activity and Ameliorate Senescence-Related Tissue Damage. Cell Metab. 2020, 32, 71–86. [Google Scholar] [CrossRef]
- Rodriguez-Navarro, J.A.; Pascual-Guerra, J.; Sacristan, S.; Nogales, M.D.C.; Fafián-Labora, J.A.; O’Loghlen, A. Extracellular vesicles as potential tools for regenerative therapy. Mol. Cell. Oncol. 2020, 7. [Google Scholar] [CrossRef]
- Gorlé, N.; Blaecher, C.; Bauwens, E.; Van den Driessche, C.; Balusu, S.; Vandewalle, J.; Van Cauwenberghe, C.; Van Wonterghem, E.; Van Imschoot, G.; Liu, C.; et al. The choroid plexus epithelium as a novel player in the stomach-brain axis during Helicobacter infection. Brain Behav. Immun. 2018, 69, 35–47. [Google Scholar] [CrossRef]
- Lin, Y.-H.; Kang, L.; Feng, W.-H.; Cheng, T.-L.; Tsai, W.-C.; Huang, H.-T.; Lee, H.-C.; Chen, C.-H. Effects of Lipids and Lipoproteins on Mesenchymal Stem Cells used in Cardiac Tissue Regeneration. Int. J. Mol. Sci. 2020, 21, 4770. [Google Scholar] [CrossRef]
- Zhang, Q.; Lenardo, M.J.; Baltimore, D. 30 Years of NF-κB: A Blossoming of Relevance to Human Pathobiology. Cell 2017, 168, 37–57. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 1–9. [Google Scholar] [CrossRef]
- Taniguchi, K.; Karin, M. NF-κB, inflammation, immunity and cancer: Coming of age. Nat. Rev. Immunol. 2018, 18, 309–324. [Google Scholar] [CrossRef]
- Wang, J.; Jacob, N.K.; Ladner, K.J.; Beg, A.; Perko, J.D.; Tanner, S.M.; Liyanarachchi, S.; Fishel, R.; Guttridge, D.C. RelA/p65 functions to maintain cellular senescence by regulating genomic stability and DNA repair. EMBO Rep. 2009, 10, 1272–1278. [Google Scholar] [CrossRef]
- Böcker, W.; Docheva, D.; Prall, W.C.; Egea, V.; Pappou, E.; Rossmann, O.; Popov, C.; Mutschler, W.; Ries, C.; Schieker, M. IKK-2 is required for TNF-α-induced invasion and proliferation of human mesenchymal stem cells. J. Mol. Med. 2008, 86, 1183–1192. [Google Scholar] [CrossRef] [PubMed]
- Flores, R.R.; Clauson, C.L.; Cho, J.; Lee, B.-C.; McGowan, S.J.; Baker, D.J.; Niedernhofer, L.J.; Robbins, P.D. Expansion of myeloid-derived suppressor cells with aging in the bone marrow of mice through a NF-κB-dependent mechanism. Aging Cell 2017, 16, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.-C.; Liao, J.-J.; Bonasera, S.; Longo, D.L.; Goetzl, E.J. Nuclear factor-κB-dependent reversal of aging-induced alterations in T cell cytokines. FASEB J. 2008, 22, 2142–2150. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, C.A.; Quintanilla, R.; Olate-Briones, A.; Venturini, W.; Mancilla, D.; Cayo, A.; Moore-Carrasco, R.; Brown, N.E. SASP-Dependent Interactions between Senescent Cells and Platelets Modulate Migration and Invasion of Cancer Cells. Int. J. Mol. Sci. 2019, 20, 5292. [Google Scholar] [CrossRef]
- Chen, X.; Liu, G.; Yuan, Y.; Wu, G.; Wang, S.; Yuan, L. NEK7 interacts with NLRP3 to modulate the pyroptosis in inflammatory bowel disease via NF-κB signaling. Cell Death Dis. 2019, 10, 1–12. [Google Scholar] [CrossRef]
- Qiu, B.; Xu, X.; Yi, P.; Hao, Y. Curcumin reinforces MSC-derived exosomes in attenuating osteoarthritis via modulating the miR-124/NF-kB and miR-143/ROCK1/TLR9 signalling pathways. J. Cell. Mol. Med. 2020, 24, 10855–10865. [Google Scholar] [CrossRef]
- Nakajima, S.; Kato, H.; Takahashi, S.; Johno, H.; Kitamura, M. Inhibition of NF-κB by MG132 through ER stress-mediated induction of LAP and LIP. FEBS Lett. 2011, 585, 2249–2254. [Google Scholar] [CrossRef]
- Van Deursen, J.M. The role of senescent cells in ageing. Nat. Cell Biol. 2014, 509, 439–446. [Google Scholar] [CrossRef]
- Cao, X.; Lin, W.; Liang, C.; Zhang, D.; Yang, F.; Zhang, Y.; Zhang, X.; Feng, J.; Chen, C. Naringin rescued the TNF-α-induced inhibition of osteogenesis of bone marrow-derived mesenchymal stem cells by depressing the activation of NF-κB signaling pathway. Immunol. Res. 2015, 62, 357–367. [Google Scholar] [CrossRef]
- Liu, T.; Long, T.; Li, H. Curcumin suppresses the proliferation of oral squamous cell carcinoma through a specificity protein 1/nuclear factor-κB-dependent pathway. Exp. Ther. Med. 2021, 21, 1. [Google Scholar] [CrossRef]
- Pandey, A.C.A.; Semon, J.; Kaushal, D.; O’Sullivan, R.P.; Glowacki, J.; Gimble, J.M.A.; Bunnell, B. MicroRNA profiling reveals age-dependent differential expression of nuclear factor κB and mitogen-activated protein kinase in adipose and bone marrow-derived human mesenchymal stem cells. Stem Cell Res. Ther. 2011, 2, 49. [Google Scholar] [CrossRef]
- Wyld, L.; Bellantuono, I.; Tchkonia, T.; Morgan, J.; Turner, O.; Foss, F.; George, J.; Danson, S.; Kirkland, J.L. Senescence and Cancer: A Review of Clinical Implications of Senescence and Senotherapies. Cancers 2020, 12, 2134. [Google Scholar] [CrossRef]
- Chien, Y.; Scuoppo, C.; Wang, X.; Fang, X.; Balgley, B.; Bolden, J.E.; Premsrirut, P.; Luo, W.; Chicas, A.; Lee, C.S.; et al. Control of the senescence-associated secretory phenotype by NF-B promotes senescence and enhances chemosensitivity. Genes Dev. 2011, 25, 2125–2136. [Google Scholar] [CrossRef]
- Worthington, E.N.; Hagood, J.S. Therapeutic use of Extracellular Vesicles for Acute and Chronic Lung Disease. Int. J. Mol. Sci. 2020, 21, 2318. [Google Scholar] [CrossRef]
- Van Esch, B.C.A.M.; Porbahaie, M.; Abbring, S.; Garssen, J.; Potaczek, D.P.; Savelkoul, H.F.J.; Neerven, R.J.J. The Impact of Milk and its Components on Epigenetic Programming of Immune Function in Early Life and Beyond: Implications for Allergy and Asthma. Front. Immunol. 2020, 11, 2141. [Google Scholar] [CrossRef]
- Alhamwe, B.A.; Miethe, S.; Von Strandmann, E.P.; Potaczek, D.P.; Garn, H. Epigenetic Regulation of Airway Epithelium Immune Functions in Asthma. Front. Immunol. 2020, 11. [Google Scholar] [CrossRef]
- Deng, W.; Tang, T.; Hou, Y.; Zeng, Q.; Wang, Y.; Fan, W.; Qu, S. Extracellular vesicles in atherosclerosis. Clin. Chim. Acta 2019, 495, 109–117. [Google Scholar] [CrossRef]
- Cavallo, C.; Merli, G.; Borzì, R.M.; Zini, N.; D’Adamo, S.; Guescini, M.; Grigolo, B.; Di Martino, A.; Santi, S.; Filardo, G. Small Extracellular Vesicles from adipose derived stromal cells significantly attenuate in vitro the NF-κB dependent inflammatory/catabolic environment of osteoarthritis. Sci. Rep. 2021, 11, 1–16. [Google Scholar] [CrossRef]

 p < 0.05 and
p < 0.05 and  p < 0.01 (D) levels of interleukin-8 (IL-8) and (E) interleukin-6 (IL-6) in the conditioned medium from senescent cells (DDIS and TIS) and proinflammatory-activated MSCs. Data shows the mean ± SD of three independent experiments. Nonparametric Kolmogorov–Smirnov test was used to calculate the significance, and it was represented as
p < 0.01 (D) levels of interleukin-8 (IL-8) and (E) interleukin-6 (IL-6) in the conditioned medium from senescent cells (DDIS and TIS) and proinflammatory-activated MSCs. Data shows the mean ± SD of three independent experiments. Nonparametric Kolmogorov–Smirnov test was used to calculate the significance, and it was represented as  p < 0.05 and
p < 0.05 and  p < 0.01. A.U: arbitrary units, NON-Se: proliferative or nonsenescent MSCs, Se: senescent (DDIS or TIS) MSCs, DMSO: MSCs treated with DMSO (dimethyl sulfoxide) and TNF-α: MSCs treated with tumor necrosis factor alpha.
p < 0.01. A.U: arbitrary units, NON-Se: proliferative or nonsenescent MSCs, Se: senescent (DDIS or TIS) MSCs, DMSO: MSCs treated with DMSO (dimethyl sulfoxide) and TNF-α: MSCs treated with tumor necrosis factor alpha.
 p < 0.05 and
p < 0.05 and  p < 0.01 (D) levels of interleukin-8 (IL-8) and (E) interleukin-6 (IL-6) in the conditioned medium from senescent cells (DDIS and TIS) and proinflammatory-activated MSCs. Data shows the mean ± SD of three independent experiments. Nonparametric Kolmogorov–Smirnov test was used to calculate the significance, and it was represented as
p < 0.01 (D) levels of interleukin-8 (IL-8) and (E) interleukin-6 (IL-6) in the conditioned medium from senescent cells (DDIS and TIS) and proinflammatory-activated MSCs. Data shows the mean ± SD of three independent experiments. Nonparametric Kolmogorov–Smirnov test was used to calculate the significance, and it was represented as  p < 0.05 and
p < 0.05 and  p < 0.01. A.U: arbitrary units, NON-Se: proliferative or nonsenescent MSCs, Se: senescent (DDIS or TIS) MSCs, DMSO: MSCs treated with DMSO (dimethyl sulfoxide) and TNF-α: MSCs treated with tumor necrosis factor alpha.
p < 0.01. A.U: arbitrary units, NON-Se: proliferative or nonsenescent MSCs, Se: senescent (DDIS or TIS) MSCs, DMSO: MSCs treated with DMSO (dimethyl sulfoxide) and TNF-α: MSCs treated with tumor necrosis factor alpha.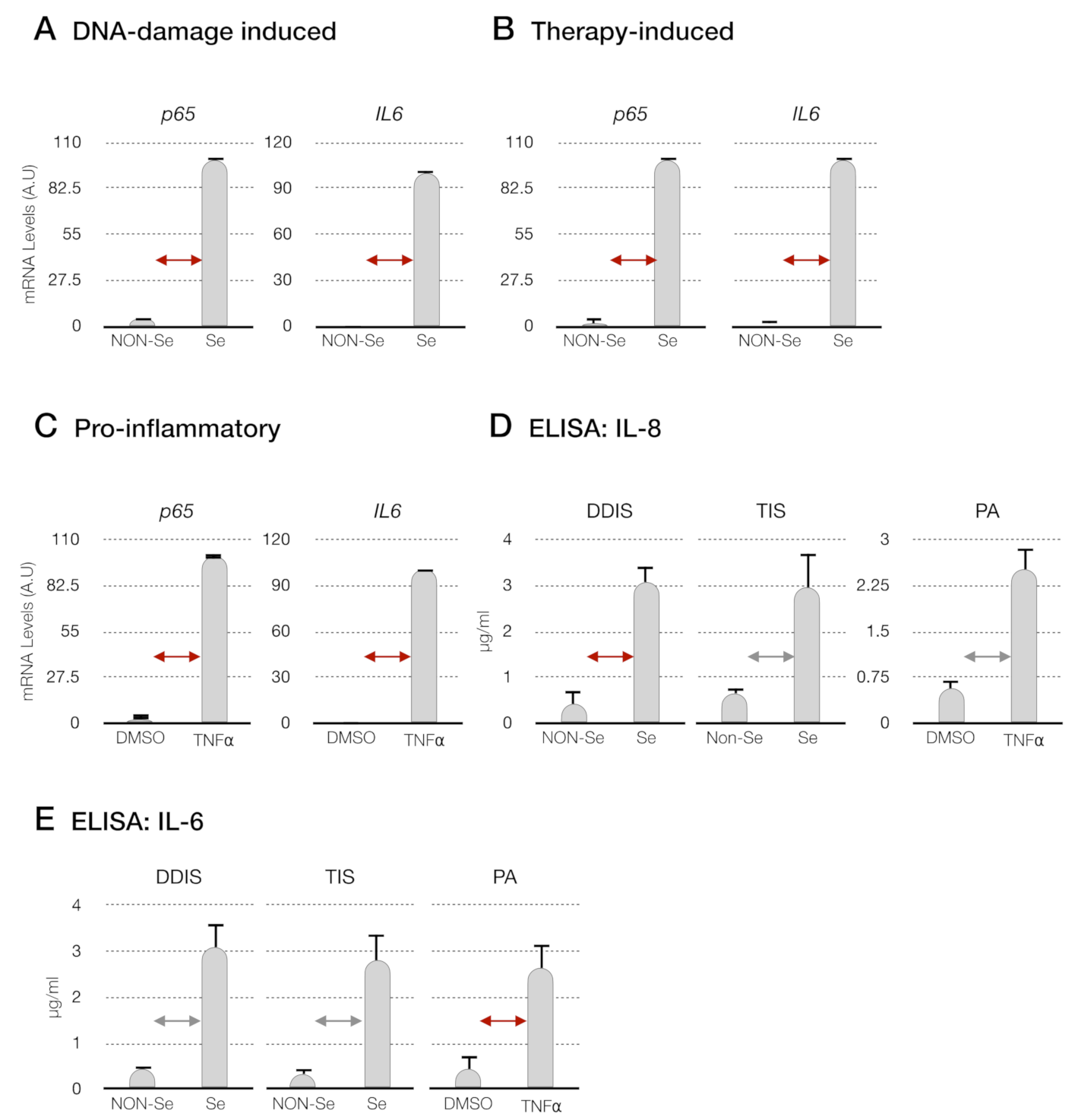
 p < 0.05 and
p < 0.05 and  p < 0.01. (C) Quantification of senescence-associated β-galactosidase activity in MSCs treated with the pharmacological inhibition of the p65 pathway using JSH-23, MG-132 and curcumin during the cellular establishment (DDIS and TIS). All data represent the mean ± SD of three independent experiments. Nonparametric Kolmogorov–Smirnov test was used to calculate the significance, and it was represented as
p < 0.01. (C) Quantification of senescence-associated β-galactosidase activity in MSCs treated with the pharmacological inhibition of the p65 pathway using JSH-23, MG-132 and curcumin during the cellular establishment (DDIS and TIS). All data represent the mean ± SD of three independent experiments. Nonparametric Kolmogorov–Smirnov test was used to calculate the significance, and it was represented as  p < 0.05 and
p < 0.05 and  p < 0.01. NON-Se: Proliferative or nonsenescent MSCs, Se: Senescent MSCs; TNF-α: MSCs treated with tumor necrosis factor alpha, JSH-23: Senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with JSH-23, MG-132: Senescent (DDIS and TIS) or proinflammatory activated (TNF-α) MSCs treated with MG-132, Curc.: Senescent (DDIS and TIS) or proinflammatory activated (TNF-α) MSCs treated with curcumin and A.U: arbitrary units.
p < 0.01. NON-Se: Proliferative or nonsenescent MSCs, Se: Senescent MSCs; TNF-α: MSCs treated with tumor necrosis factor alpha, JSH-23: Senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with JSH-23, MG-132: Senescent (DDIS and TIS) or proinflammatory activated (TNF-α) MSCs treated with MG-132, Curc.: Senescent (DDIS and TIS) or proinflammatory activated (TNF-α) MSCs treated with curcumin and A.U: arbitrary units.
 p < 0.05 and
p < 0.05 and  p < 0.01. (C) Quantification of senescence-associated β-galactosidase activity in MSCs treated with the pharmacological inhibition of the p65 pathway using JSH-23, MG-132 and curcumin during the cellular establishment (DDIS and TIS). All data represent the mean ± SD of three independent experiments. Nonparametric Kolmogorov–Smirnov test was used to calculate the significance, and it was represented as
p < 0.01. (C) Quantification of senescence-associated β-galactosidase activity in MSCs treated with the pharmacological inhibition of the p65 pathway using JSH-23, MG-132 and curcumin during the cellular establishment (DDIS and TIS). All data represent the mean ± SD of three independent experiments. Nonparametric Kolmogorov–Smirnov test was used to calculate the significance, and it was represented as  p < 0.05 and
p < 0.05 and  p < 0.01. NON-Se: Proliferative or nonsenescent MSCs, Se: Senescent MSCs; TNF-α: MSCs treated with tumor necrosis factor alpha, JSH-23: Senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with JSH-23, MG-132: Senescent (DDIS and TIS) or proinflammatory activated (TNF-α) MSCs treated with MG-132, Curc.: Senescent (DDIS and TIS) or proinflammatory activated (TNF-α) MSCs treated with curcumin and A.U: arbitrary units.
p < 0.01. NON-Se: Proliferative or nonsenescent MSCs, Se: Senescent MSCs; TNF-α: MSCs treated with tumor necrosis factor alpha, JSH-23: Senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with JSH-23, MG-132: Senescent (DDIS and TIS) or proinflammatory activated (TNF-α) MSCs treated with MG-132, Curc.: Senescent (DDIS and TIS) or proinflammatory activated (TNF-α) MSCs treated with curcumin and A.U: arbitrary units.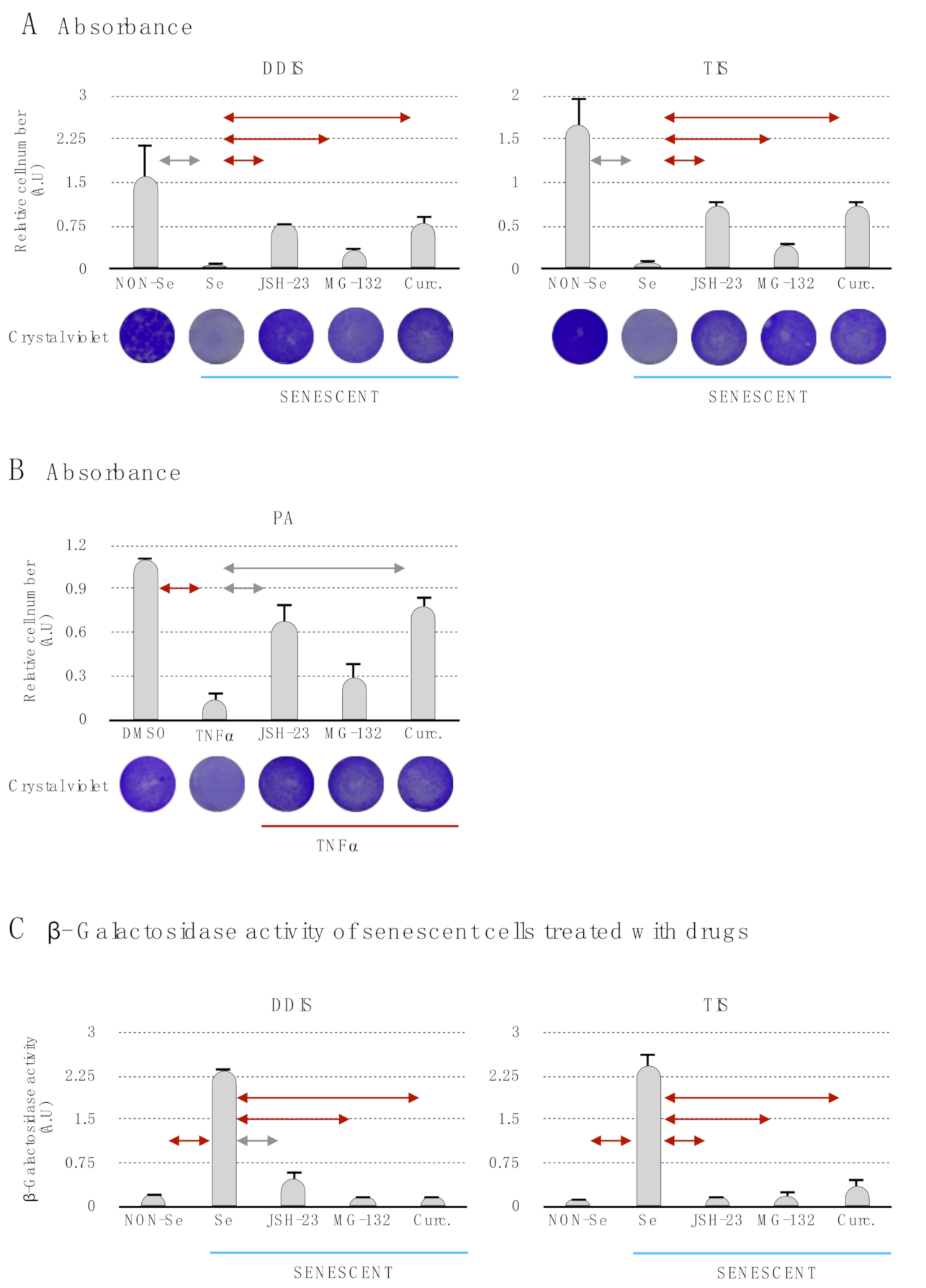
 p < 0.05 and
p < 0.05 and  p < 0.01. Quantification of the number of particles in (C) MSCs treated with JSH-23, MG-132 and curcumin during the cellular senescence induction (DDIS and TIS) and (D) PA using an NTA. The graphs shown the mean ± SD of three independent experiments. Nonparametric Kolmogorov–Smirnov test was used to calculate the significance, and it was represented as
p < 0.01. Quantification of the number of particles in (C) MSCs treated with JSH-23, MG-132 and curcumin during the cellular senescence induction (DDIS and TIS) and (D) PA using an NTA. The graphs shown the mean ± SD of three independent experiments. Nonparametric Kolmogorov–Smirnov test was used to calculate the significance, and it was represented as  p < 0.05 and
p < 0.05 and  p < 0.01. NON-Se: proliferative or nonsenescent MSCs, Se: senescent MSCs, TNF-α: MSCs treated with tumor necrosis factor alpha, JSH-23: senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with JSH-23, MG-132: senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with MG-132, Curc.: senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with curcumin, nm: nanometer and M:1000.
p < 0.01. NON-Se: proliferative or nonsenescent MSCs, Se: senescent MSCs, TNF-α: MSCs treated with tumor necrosis factor alpha, JSH-23: senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with JSH-23, MG-132: senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with MG-132, Curc.: senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with curcumin, nm: nanometer and M:1000.
 p < 0.05 and
p < 0.05 and  p < 0.01. Quantification of the number of particles in (C) MSCs treated with JSH-23, MG-132 and curcumin during the cellular senescence induction (DDIS and TIS) and (D) PA using an NTA. The graphs shown the mean ± SD of three independent experiments. Nonparametric Kolmogorov–Smirnov test was used to calculate the significance, and it was represented as
p < 0.01. Quantification of the number of particles in (C) MSCs treated with JSH-23, MG-132 and curcumin during the cellular senescence induction (DDIS and TIS) and (D) PA using an NTA. The graphs shown the mean ± SD of three independent experiments. Nonparametric Kolmogorov–Smirnov test was used to calculate the significance, and it was represented as  p < 0.05 and
p < 0.05 and  p < 0.01. NON-Se: proliferative or nonsenescent MSCs, Se: senescent MSCs, TNF-α: MSCs treated with tumor necrosis factor alpha, JSH-23: senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with JSH-23, MG-132: senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with MG-132, Curc.: senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with curcumin, nm: nanometer and M:1000.
p < 0.01. NON-Se: proliferative or nonsenescent MSCs, Se: senescent MSCs, TNF-α: MSCs treated with tumor necrosis factor alpha, JSH-23: senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with JSH-23, MG-132: senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with MG-132, Curc.: senescent (DDIS and TIS) or proinflammatory-activated (TNF-α) MSCs treated with curcumin, nm: nanometer and M:1000.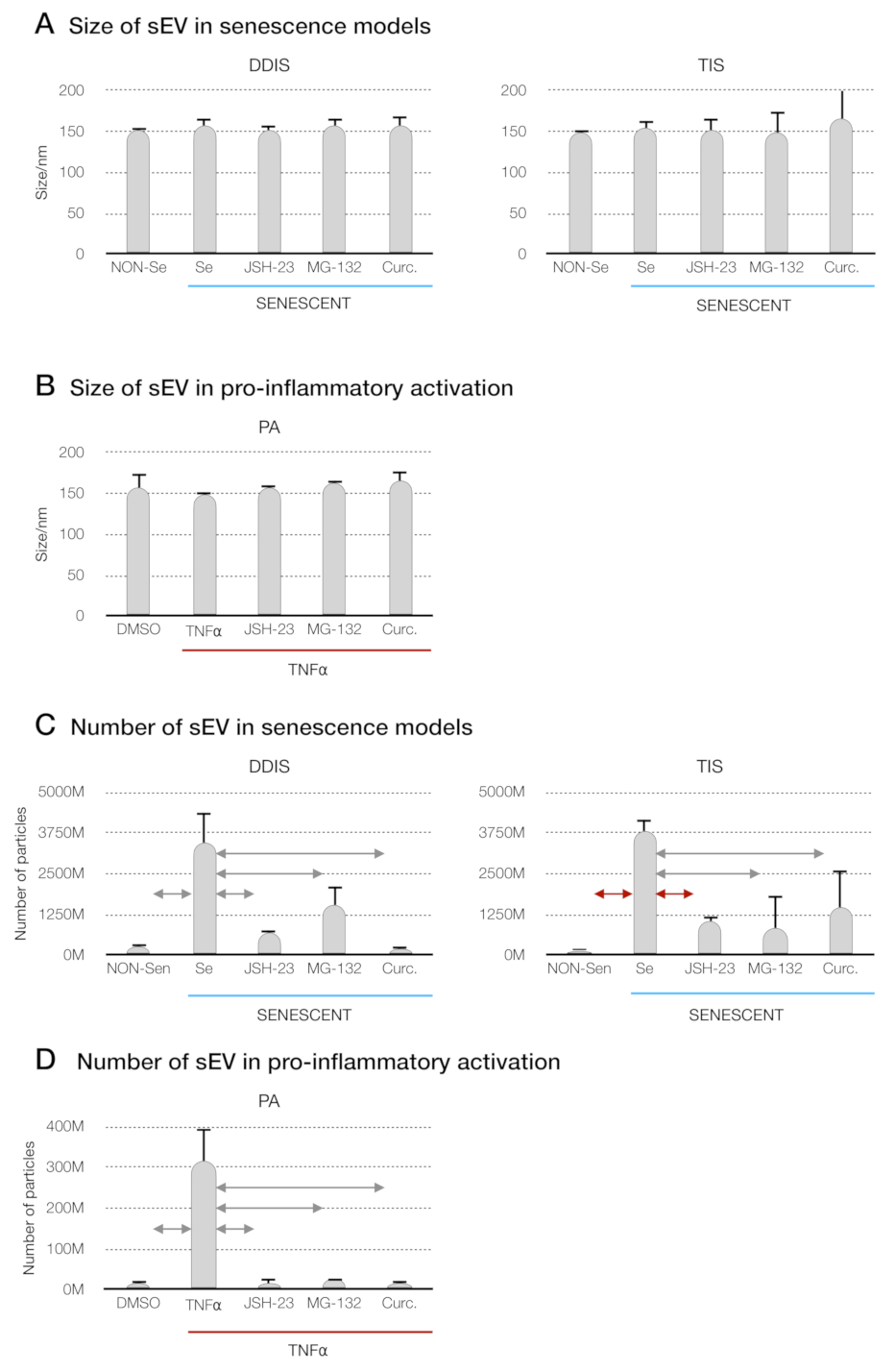
 p < 0.05 and
p < 0.05 and  p < 0.01. A.U: arbitrary units and MTT (bromide 3-(4,5-dimetiltiazol-2-il)-2,5-diphenyltetrazolium).
p < 0.01. A.U: arbitrary units and MTT (bromide 3-(4,5-dimetiltiazol-2-il)-2,5-diphenyltetrazolium).
 p < 0.05 and
p < 0.05 and  p < 0.01. A.U: arbitrary units and MTT (bromide 3-(4,5-dimetiltiazol-2-il)-2,5-diphenyltetrazolium).
p < 0.01. A.U: arbitrary units and MTT (bromide 3-(4,5-dimetiltiazol-2-il)-2,5-diphenyltetrazolium).

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mato-Basalo, R.; Morente-López, M.; Arntz, O.J.; van de Loo, F.A.J.; Fafián-Labora, J.; Arufe, M.C. Therapeutic Potential for Regulation of the Nuclear Factor Kappa-B Transcription Factor p65 to Prevent Cellular Senescence and Activation of Pro-Inflammatory in Mesenchymal Stem Cells. Int. J. Mol. Sci. 2021, 22, 3367. https://doi.org/10.3390/ijms22073367
Mato-Basalo R, Morente-López M, Arntz OJ, van de Loo FAJ, Fafián-Labora J, Arufe MC. Therapeutic Potential for Regulation of the Nuclear Factor Kappa-B Transcription Factor p65 to Prevent Cellular Senescence and Activation of Pro-Inflammatory in Mesenchymal Stem Cells. International Journal of Molecular Sciences. 2021; 22(7):3367. https://doi.org/10.3390/ijms22073367
Chicago/Turabian StyleMato-Basalo, Rocío, Miriam Morente-López, Onno J Arntz, Fons A. J. van de Loo, Juan Fafián-Labora, and María C. Arufe. 2021. "Therapeutic Potential for Regulation of the Nuclear Factor Kappa-B Transcription Factor p65 to Prevent Cellular Senescence and Activation of Pro-Inflammatory in Mesenchymal Stem Cells" International Journal of Molecular Sciences 22, no. 7: 3367. https://doi.org/10.3390/ijms22073367
APA StyleMato-Basalo, R., Morente-López, M., Arntz, O. J., van de Loo, F. A. J., Fafián-Labora, J., & Arufe, M. C. (2021). Therapeutic Potential for Regulation of the Nuclear Factor Kappa-B Transcription Factor p65 to Prevent Cellular Senescence and Activation of Pro-Inflammatory in Mesenchymal Stem Cells. International Journal of Molecular Sciences, 22(7), 3367. https://doi.org/10.3390/ijms22073367







