Maternal and Postnatal High Linoleic Acid Diet Impacts Lipid Metabolism in Adult Rat Offspring in a Sex-Specific Manner
Abstract
1. Introduction
2. Results
2.1. Effect of Maternal and Postnatal Hla Diet on Body Weight and Organ Weight of Adult Offspring
2.2. Effect of Maternal and Postnatal HLA Diet on Plasma Fatty Acid Composition in Adult Offspring
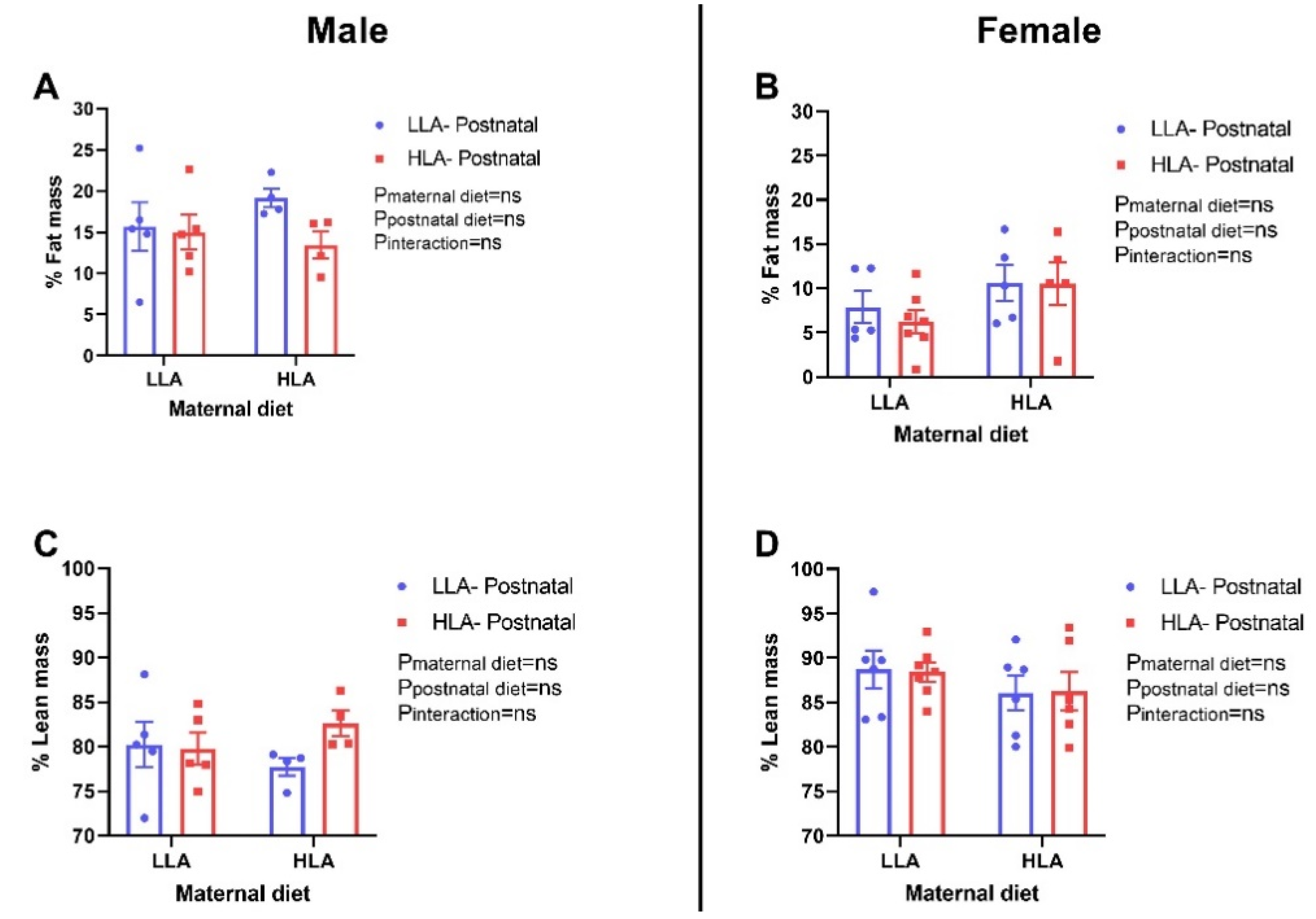
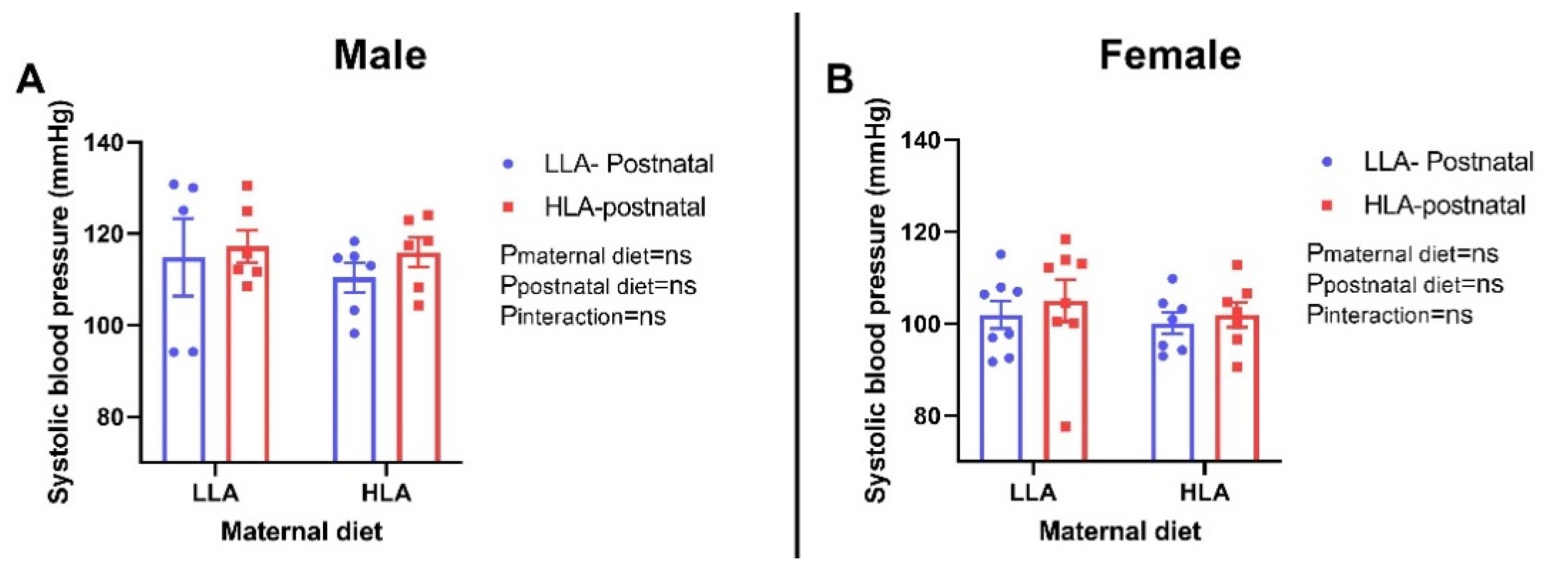
2.3. Effect of Maternal and Postnatal Hla Diet on Fat Mass, Lean Mass and Systolic Blood Pressure in Adult Offspring
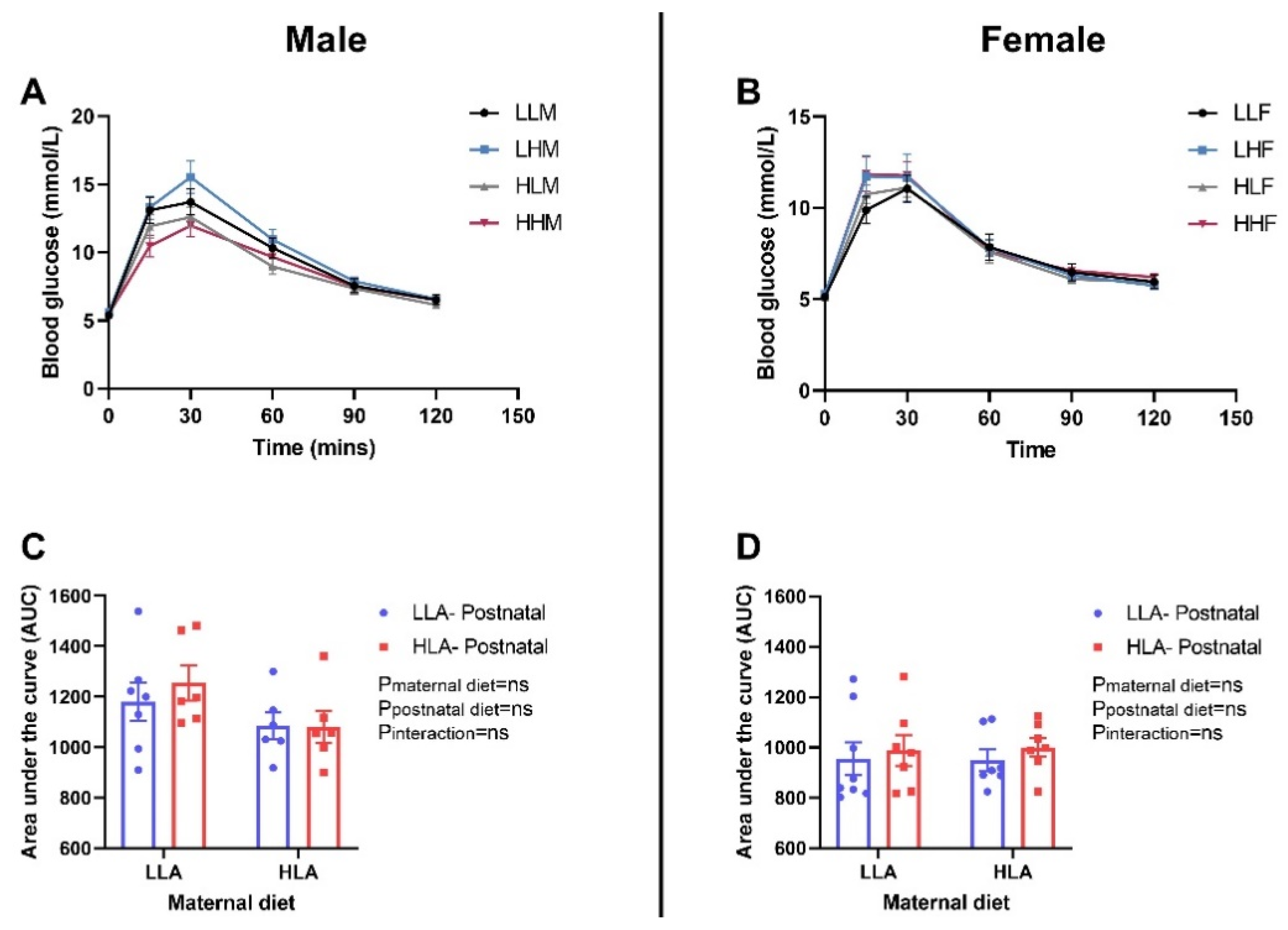
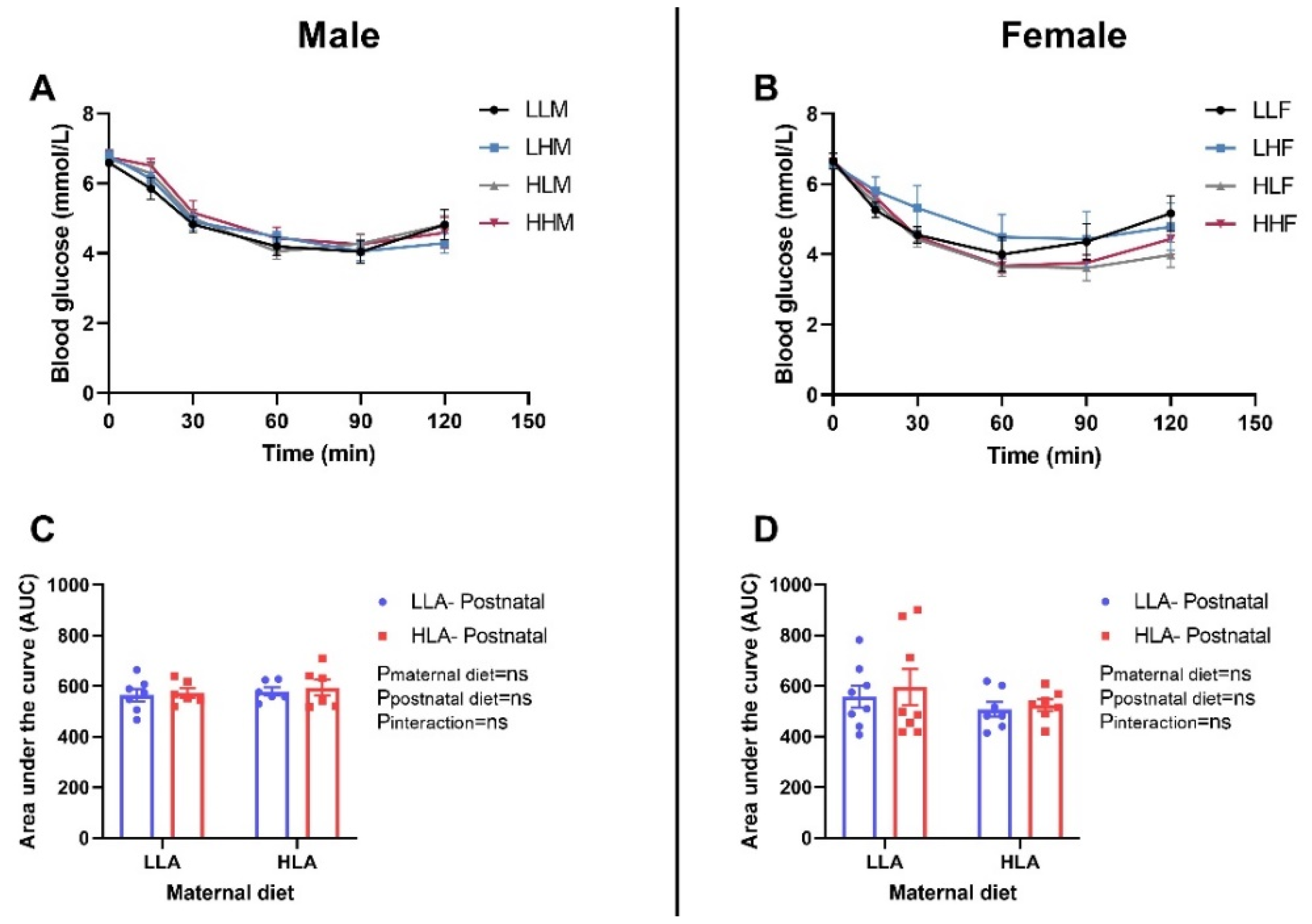
2.4. Effect of Maternal and Postnatal Hla Diet on Glucose Tolerance and Insulin Tolerance in Adult Offspring
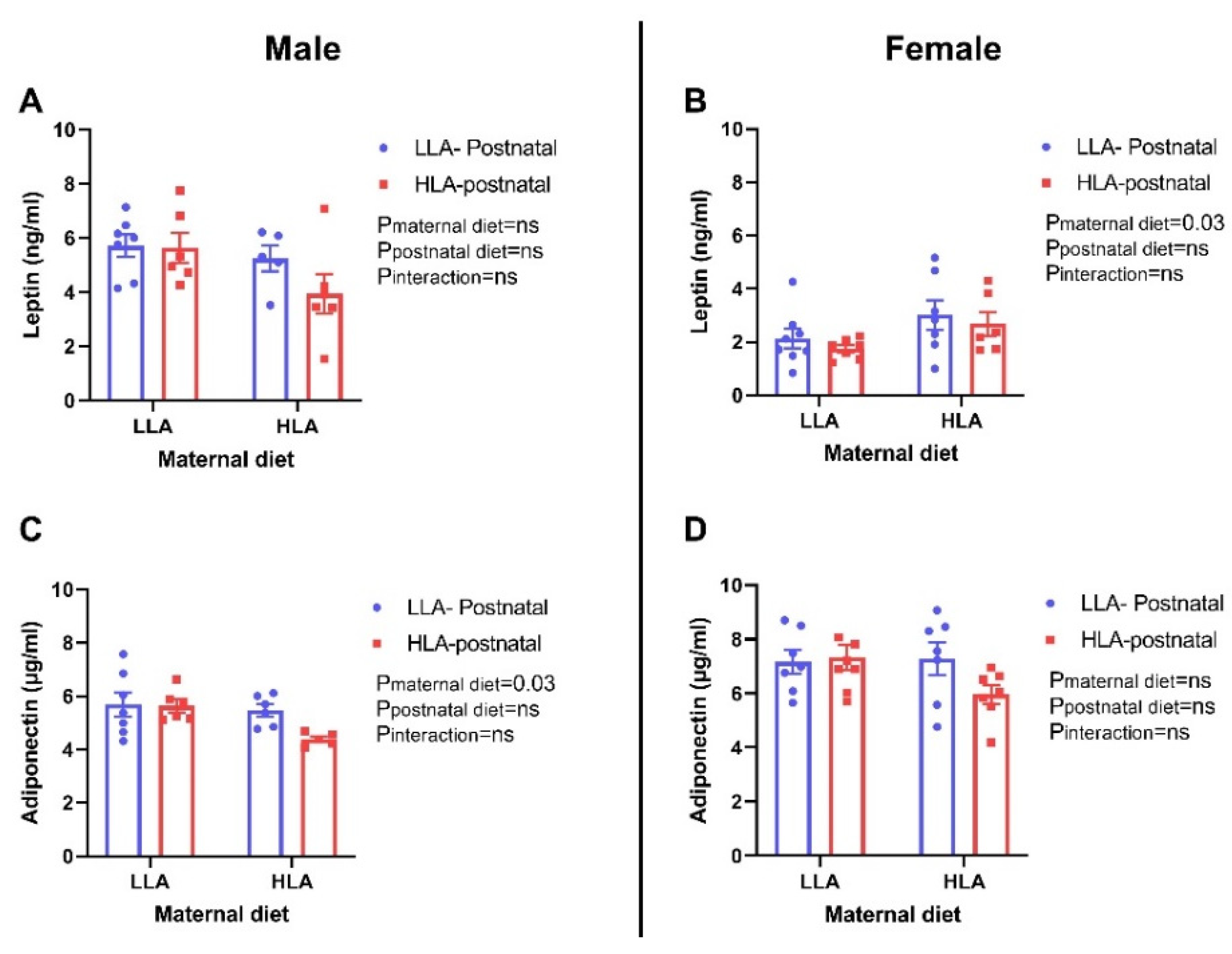
2.5. Effect of Maternal and Postnatal Hla Diet on Circulating Leptin and Adiponectin Concentrations in Adult Offspring
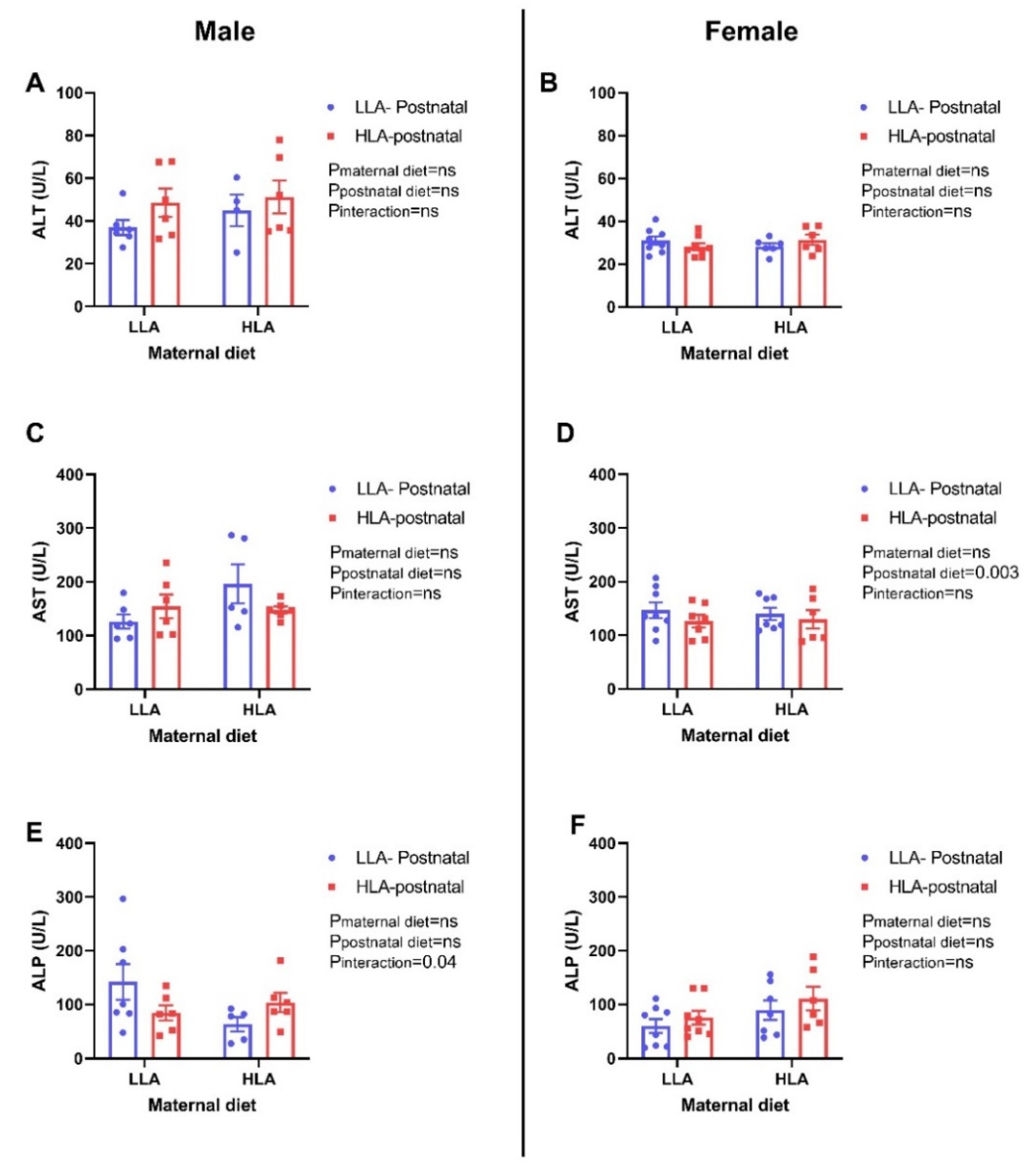
2.6. Effect of Maternal and Postnatal HLA Diet on Circulating Liver Enzymes in Adult Offspring
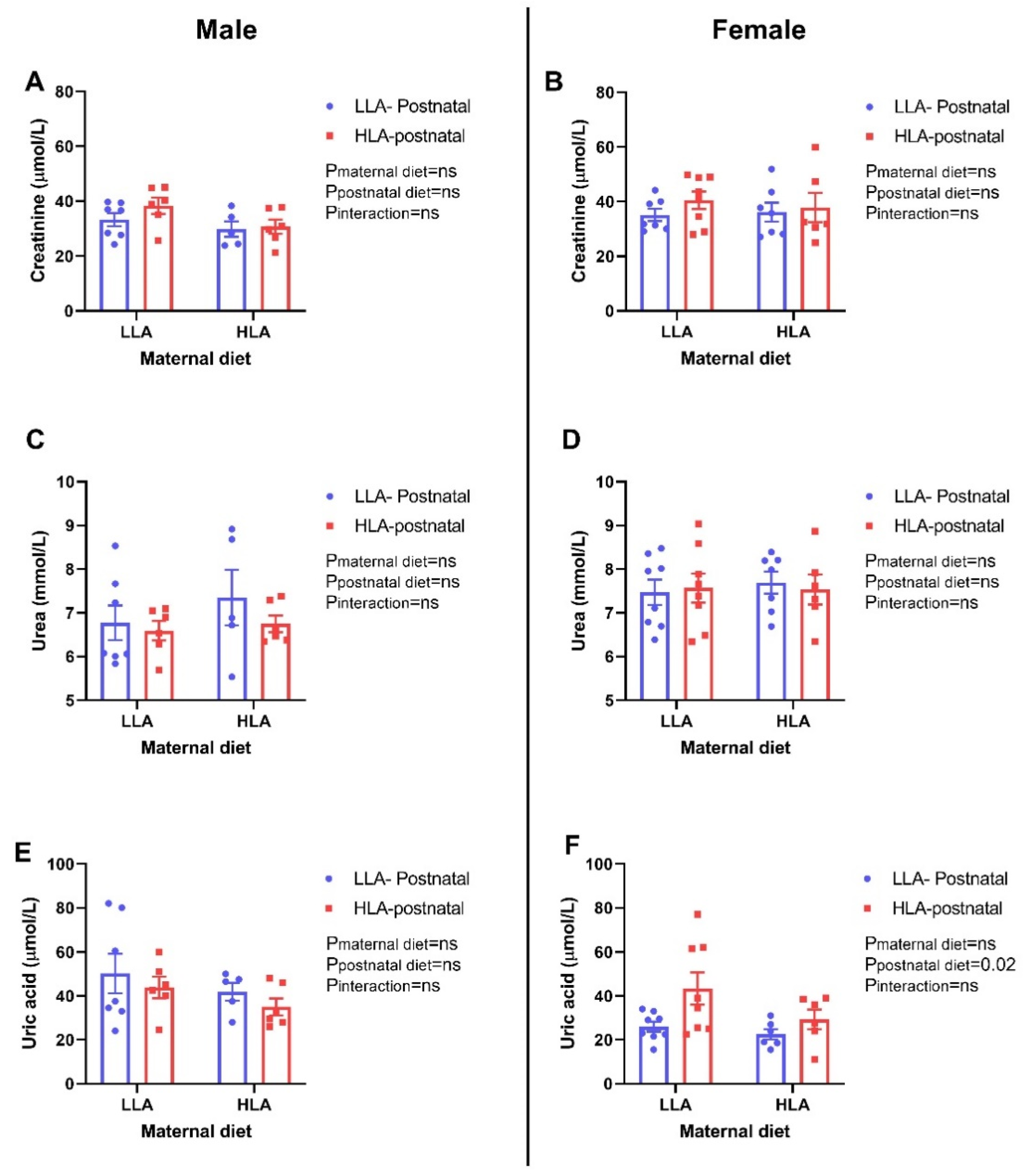
2.7. Effect of Maternal and Postnatal Hla Diet on Creatinine, Urea and Uric Acid in Plasma of Adult Offspring
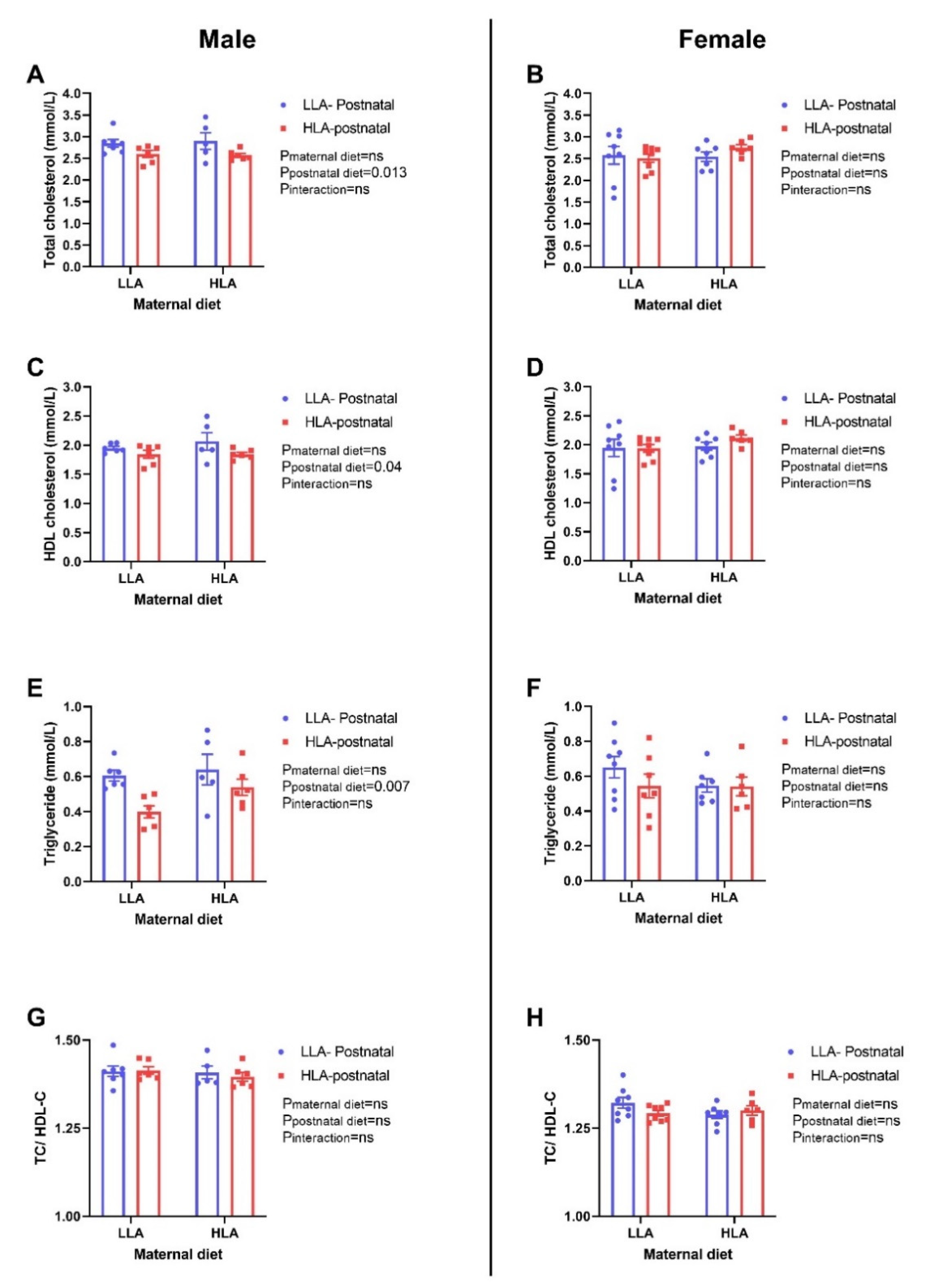
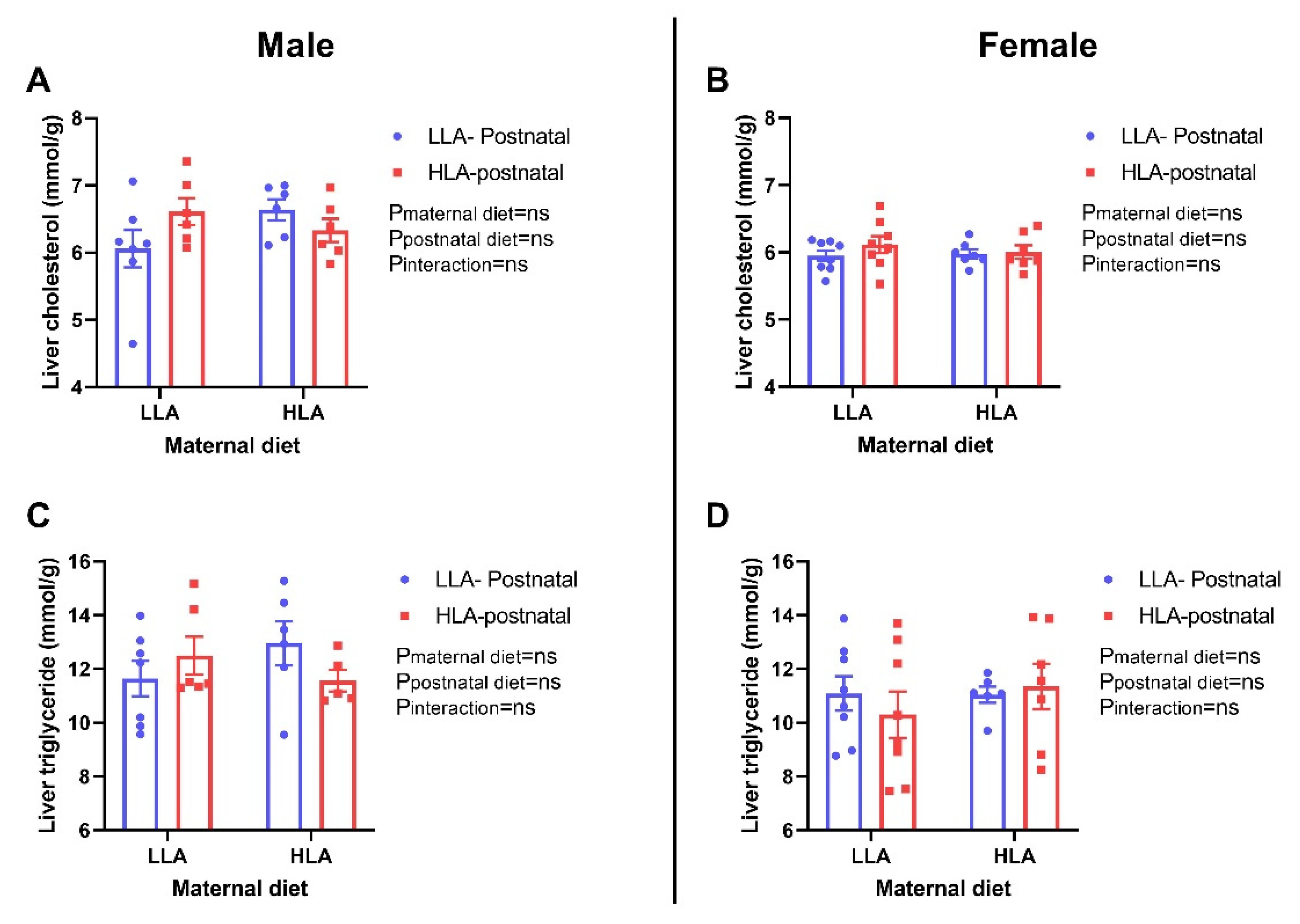
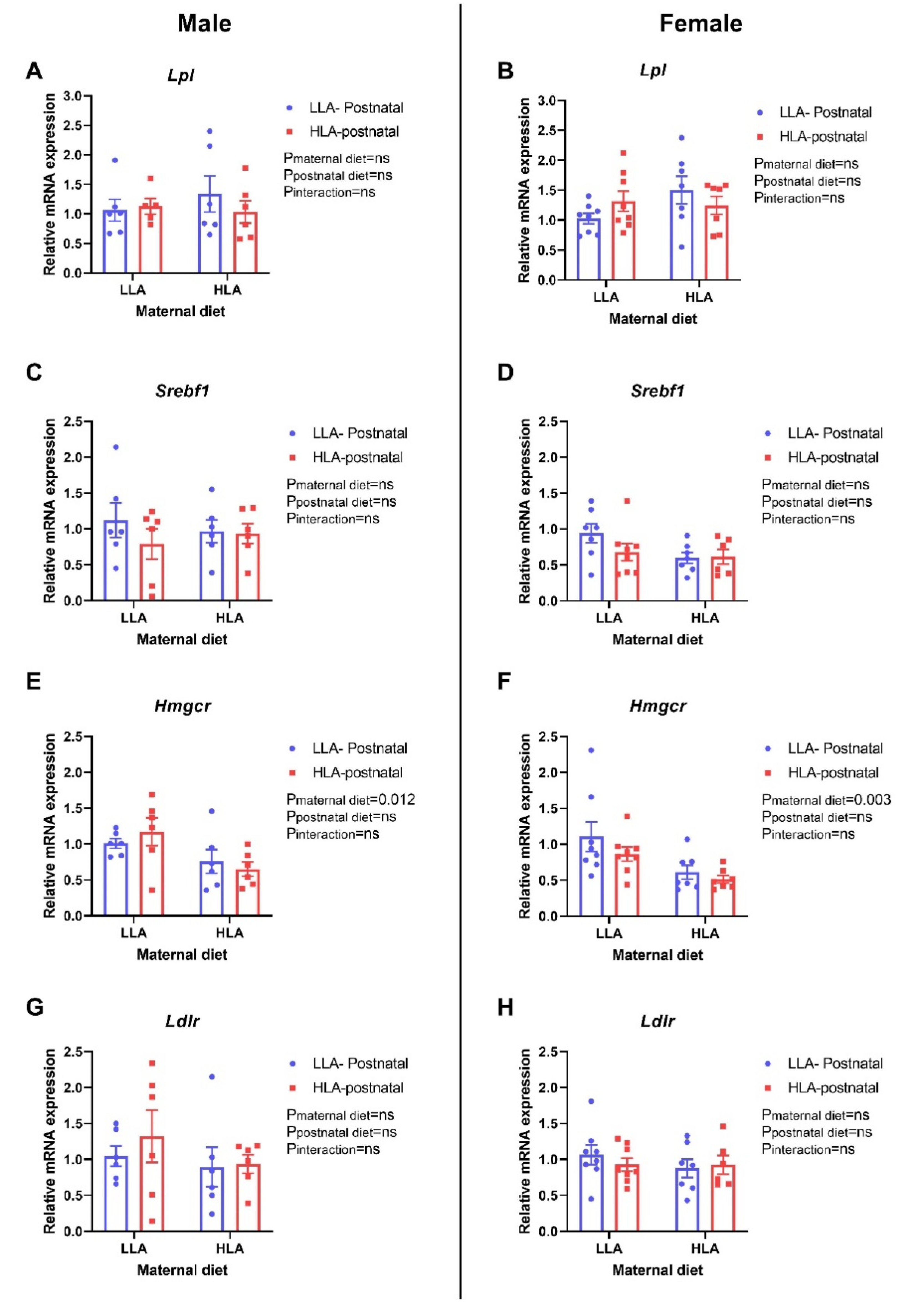
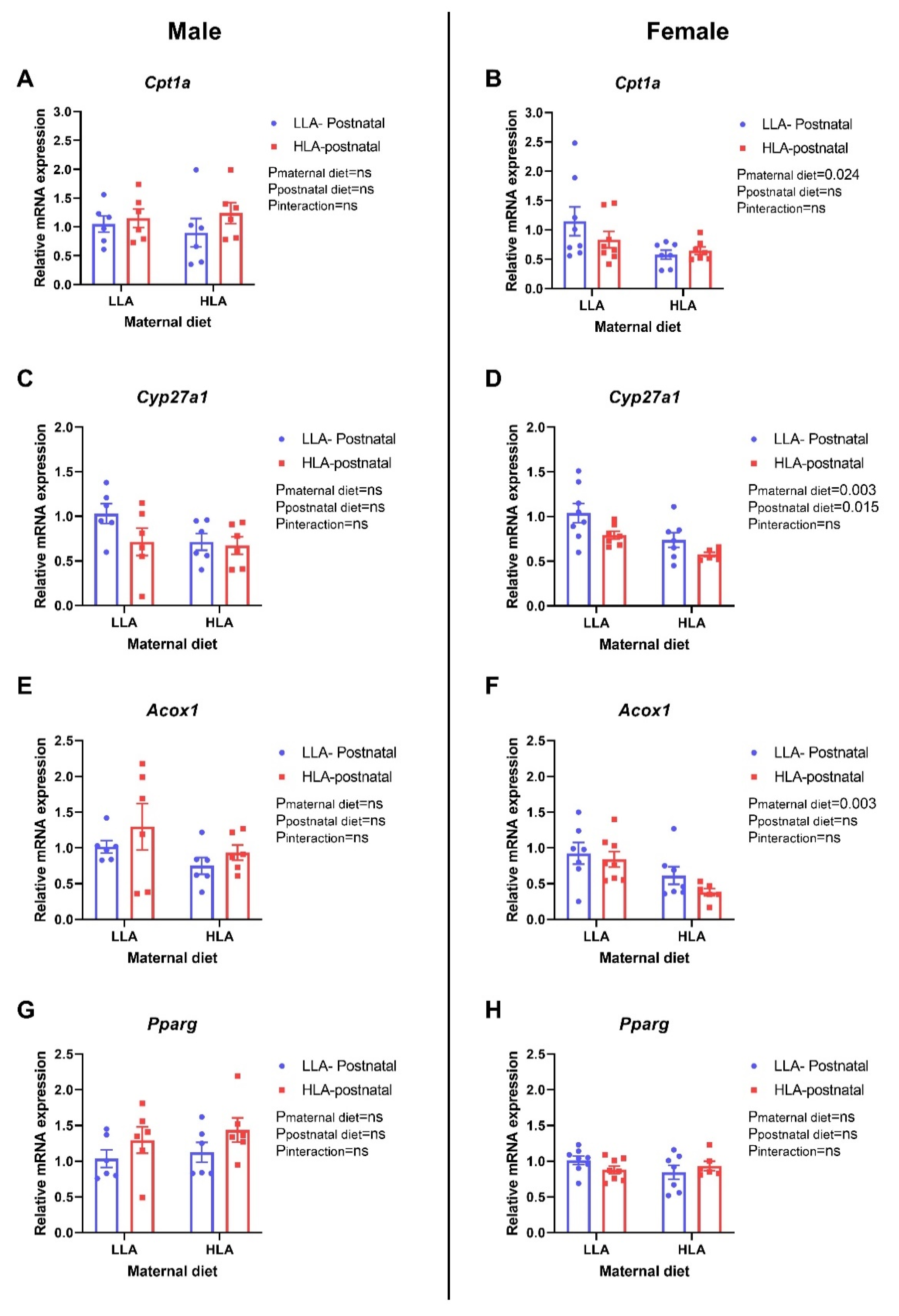
2.8. Effect of Maternal and Postnatal Hla Diet on Circulating and Hepatic Lipids in Adult Offspring
3. Discussion
4. Materials and Methods
4.1. Experimental Animal Model and Diet
4.2. Physiological Experiments in Adult Offspring
4.2.1. Non-Invasive Measurement of Systolic Blood Pressure in Pn180 Offspring
4.2.2. Intraperitoneal Glucose Tolerance Test in Pn180 Offspring
4.2.3. Intraperitoneal Insulin Tolerance Test in Adult Offspring
4.2.4. Body Composition Measurement in Adult Offspring
4.2.5. Fatty Acid Analysis in Offspring Plasma
4.2.6. Measurement of Circulating Leptin Concentration
4.2.7. Biochemical Analysis
4.2.8. Plasma Adiponectin Estimation in Adult Offspring
4.2.9. Cholesterol and Triglyceride Quantification in the Liver of Offspring
4.2.10. Quantitative Real Time Polymerase Chain Reaction (qPCR)
4.2.11. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Yki-Järvinen, H. Non-alcoholic fatty liver disease as a cause and a consequence of metabolic syndrome. Lancet Diabetes Endocrinol. 2014, 2, 901–910. [Google Scholar] [CrossRef]
- Ravelli, A.C.; van Der Meulen, J.H.; Osmond, C.; Barker, D.J.; Bleker, O.P. Obesity at the age of 50 y in men and women exposed to famine prenatally. Am. J. Clin. Nutr. 1999, 70, 811–816. [Google Scholar] [CrossRef] [PubMed]
- Portrait, F.; Teeuwiszen, E.; Deeg, D. Early life undernutrition and chronic diseases at older ages: The effects of the Dutch famine on cardiovascular diseases and diabetes. Soc. Sci. Med. 2011, 73, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Tiao, M.M.; Lin, Y.J.; Yu, H.R.; Sheen, J.M.; Lin, I.C.; Lai, Y.J.; Tain, Y.L.; Huang, L.T.; Tsai, C.C. Resveratrol ameliorates maternal and post-weaning high-fat diet-induced nonalcoholic fatty liver disease via renin-angiotensin system. Lipids Health Dis. 2018, 17, 178. [Google Scholar] [CrossRef]
- Bariani, M.V.; Correa, F.; Domínguez Rubio, A.P.; Marvaldi, C.; Schander, J.A.; Beltrame, J.S.; Cella, M.; Silberman, D.M.; Aisemberg, J.; Franchi, A.M. Maternal obesogenic diet combined with postnatal exposure to high-fat diet induces metabolic alterations in offspring. J. Cell. Physiol. 2020, 235, 8260–8269. [Google Scholar] [CrossRef] [PubMed]
- Wankhade, U.D.; Zhong, Y.; Kang, P.; Alfaro, M.; Chintapalli, S.V.; Piccolo, B.D.; Mercer, K.E.; Andres, A.; Thakali, K.M.; Shankar, K. Maternal High-Fat Diet Programs Offspring Liver Steatosis in a Sexually Dimorphic Manner in Association with Changes in Gut Microbial Ecology in Mice. Sci. Rep. 2018, 8, 16502. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.A.; Wakabayashi, H.; Izuishi, K.; Okano, K.; Yachida, S.; Maeta, H. The role of prostaglandins in liver ischemia-reperfusion injury. Curr. Pharm. Des. 2006, 12, 2935–2951. [Google Scholar] [CrossRef] [PubMed]
- Araya, J.; Rodrigo, R.; Videla, L.A.; Thielemann, L.; Orellana, M.; Pettinelli, P.; Poniachik, J. Increase in long-chain polyunsaturated fatty acid n-6/n-3 ratio in relation to hepatic steatosis in patients with non-alcoholic fatty liver disease. Clin. Sci. 2004, 106, 635–643. [Google Scholar] [CrossRef]
- Santoro, N.; Caprio, S.; Feldstein, A.E. Oxidized metabolites of linoleic acid as biomarkers of liver injury in nonalcoholic steatohepatitis. Clin. Lipidol. 2013, 8, 411–418. [Google Scholar] [CrossRef]
- Marchix, J.; Catheline, D.; Duby, C.; Monthéan-Boulier, N.; Boissel, F.; Pédrono, F.; Boudry, G.; Legrand, P. Interactive effects of maternal and weaning high linoleic acid intake on hepatic lipid metabolism, oxylipins profile and hepatic steatosis in offspring. J. Nutr. Biochem. 2020, 75, 108241. [Google Scholar] [CrossRef]
- Shrestha, N.; Cuffe, J.S.M.; Holland, O.J.; Perkins, A.V.; McAinch, A.J.; Hryciw, D.H. Linoleic Acid Increases Prostaglandin E2 Release and Reduces Mitochondrial Respiration and Cell Viability in Human Trophoblast-Like Cells. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 2019, 52, 94–108. [Google Scholar]
- Huot, P.S.; Ly, A.; Szeto, I.M.; Reza-López, S.A.; Cho, D.; Kim, Y.I.; Anderson, G.H. Maternal and postweaning folic acid supplementation interact to influence body weight, insulin resistance, and food intake regulatory gene expression in rat offspring in a sex-specific manner. Appl. Physiol. Nutr. Metab. 2016, 41, 411–420. [Google Scholar] [CrossRef]
- Gray, C.; Vickers, M.H.; Segovia, S.A.; Zhang, X.D.; Reynolds, C.M. A maternal high fat diet programmes endothelial function and cardiovascular status in adult male offspring independent of body weight, which is reversed by maternal conjugated linoleic acid (CLA) supplementation. PLoS ONE 2015, 10, e0115994. [Google Scholar]
- Lomas-Soria, C.; Reyes-Castro, L.A.; Rodríguez-González, G.L.; Ibáñez, C.A.; Bautista, C.J.; Cox, L.A.; Nathanielsz, P.W.; Zambrano, E. Maternal obesity has sex-dependent effects on insulin, glucose and lipid metabolism and the liver transcriptome in young adult rat offspring. J. Physiol. 2018, 596, 4611–4628. [Google Scholar] [CrossRef] [PubMed]
- De Almeida Faria, J.; Duque-Guimarães, D.; Carpenter, A.A.; Loche, E.; Ozanne, S.E. A post-weaning obesogenic diet exacerbates the detrimental effects of maternal obesity on offspring insulin signaling in adipose tissue. Sci. Rep. 2017, 7, 44949. [Google Scholar] [CrossRef] [PubMed]
- Rajia, S.; Chen, H.; Morris, M.J. Maternal overnutrition impacts offspring adiposity and brain appetite markers-modulation by postweaning diet. J. Neuroendocrinol. 2010, 22, 905–914. [Google Scholar] [CrossRef] [PubMed]
- Ojeda, N.B.; Intapad, S.; Alexander, B.T. Sex differences in the developmental programming of hypertension. Acta Physiol. 2014, 210, 307–316. [Google Scholar] [CrossRef]
- Dahlhoff, M.; Pfister, S.; Blutke, A.; Rozman, J.; Klingenspor, M.; Deutsch, M.J.; Rathkolb, B.; Fink, B.; Gimpfl, M.; Hrabě de Angelis, M.; et al. Peri-conceptional obesogenic exposure induces sex-specific programming of disease susceptibilities in adult mouse offspring. Biochim. Biophys. Acta 2014, 1842, 304–317. [Google Scholar] [CrossRef]
- Puri, P.; Baillie, R.A.; Wiest, M.M.; Mirshahi, F.; Choudhury, J.; Cheung, O.; Sargeant, C.; Contos, M.J.; Sanyal, A.J. A lipidomic analysis of nonalcoholic fatty liver disease. Hepatology 2007, 46, 1081–1090. [Google Scholar] [CrossRef] [PubMed]
- Jandacek, R.J. Linoleic Acid: A Nutritional Quandary. Healthcare 2017, 5, 25. [Google Scholar] [CrossRef]
- Simopoulos, A.P. An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef] [PubMed]
- Farvid, M.S.; Ding, M.; Pan, A.; Sun, Q.; Chiuve, S.E.; Steffen, L.M.; Willett, W.C.; Hu, F.B. Dietary linoleic acid and risk of coronary heart disease: A systematic review and meta-analysis of prospective cohort studies. Circulation 2014, 130, 1568–1578. [Google Scholar] [CrossRef]
- Harris, W.S. Linoleic acid and coronary heart disease. Prostaglandins Leukot. Essent. Fatty Acids 2008, 79, 169–171. [Google Scholar] [CrossRef]
- Marangoni, F.; Agostoni, C.; Borghi, C.; Catapano, A.L.; Cena, H.; Ghiselli, A.; La Vecchia, C.; Lercker, G.; Manzato, E.; Pirillo, A.; et al. Dietary linoleic acid and human health: Focus on cardiovascular and cardiometabolic effects. Atherosclerosis 2020, 292, 90–98. [Google Scholar] [CrossRef]
- Ramsden, C.E.; Zamora, D.; Leelarthaepin, B.; Majchrzak-Hong, S.F.; Faurot, K.R.; Suchindran, C.M.; Ringel, A.; Davis, J.M.; Hibbeln, J.R. Use of dietary linoleic acid for secondary prevention of coronary heart disease and death: Evaluation of recovered data from the Sydney Diet Heart Study and updated meta-analysis. BMJ 2013, 346, e8707. [Google Scholar] [CrossRef]
- Draycott, S.A.V.; Elmes, M.J.; Muhlhausler, B.S.; Langley-Evans, S. Omega-6:Omega-3 Fatty Acid Ratio and Total Fat Content of the Maternal Diet Alter Offspring Growth and Fat Deposition in the Rat. Nutrients 2020, 12, 2505. [Google Scholar] [CrossRef] [PubMed]
- Draycott, S.A.V.; George, G.; Elmes, M.J.; Muhlhausler, B.S.; Langley-Evans, S.C. The effect of maternal dietary fat content and n-6:n-3 ratio on offspring growth and hepatic gene expression in the rat. Br. J. Nutr. 2020, 123, 1227–1238. [Google Scholar] [CrossRef] [PubMed]
- Gali Ramamoorthy, T.; Allen, T.J.; Davies, A.; Harno, E.; Sefton, C.; Murgatroyd, C.; White, A. Maternal overnutrition programs epigenetic changes in the regulatory regions of hypothalamic Pomc in the offspring of rats. Int. J. Obes. (2005) 2018, 42, 1431–1444. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, N.; Cuffe, J.S.M.; Holland, O.J.; Bulmer, A.C.; Hill, M.; Perkins, A.V.; Muhlhausler, B.S.; McAinch, A.J.; Hryciw, D.H. Elevated maternal linoleic acid reduces circulating leptin concentrations, cholesterol levels and male fetal survival in a rat model. J. Physiol. 2019, 597, 3349–3361. [Google Scholar] [CrossRef]
- Vaughan, R.A.; Garrison, R.L.; Stamatikos, A.D.; Kang, M.; Cooper, J.A.; Paton, C.M. A high linoleic acid diet does not induce inflammation in mouse liver or adipose tissue. Lipids 2015, 50, 1115–1122. [Google Scholar] [CrossRef]
- Diniz, Y.S.A.; Cicogna, A.C.; Padovani, C.R.; Santana, L.S.; Faine, L.A.; Novelli, E.L. Diets rich in saturated and polyunsaturated fatty acids: Metabolic shifting and cardiac health. Nutrition 2004, 20, 230–234. [Google Scholar] [CrossRef]
- Lord, J.; Thomas, R.; Fox, B.; Acharya, U.; Wilkin, T. The central issue? Visceral fat mass is a good marker of insulin resistance and metabolic disturbance in women with polycystic ovary syndrome. BJOG Int. J. Obstet. Gynaecol. 2006, 113, 1203–1209. [Google Scholar] [CrossRef]
- Lang, P.-O.; Trivalle, C.; Vogel, T.; Proust, J.; Papazian, J.-P. Markers of metabolic and cardiovascular health in adults: Comparative analysis of DEXA-based body composition components and BMI categories. J. Cardiol. 2015, 65, 42–49. [Google Scholar] [CrossRef]
- Alferink, L.J.M.; Trajanoska, K.; Erler, N.S.; Schoufour, J.D.; de Knegt, R.J.; Ikram, M.A.; Janssen, H.L.A.; Franco, O.H.; Metselaar, H.J.; Rivadeneira, F. Nonalcoholic Fatty Liver Disease in The Rotterdam Study: About Muscle Mass, Sarcopenia, Fat Mass, and Fat Distribution. J. Bone Miner. Res. 2019, 34, 1254–1263. [Google Scholar] [CrossRef]
- Belury, M.A.; Cole, R.M.; Bailey, B.E.; Ke, J.Y.; Andridge, R.R.; Kiecolt-Glaser, J.K. Erythrocyte linoleic acid, but not oleic acid, is associated with improvements in body composition in men and women. Mol. Nutr. Food Res. 2016, 60, 1206–1212. [Google Scholar] [CrossRef] [PubMed]
- Oosting, A.; Kegler, D.; van de Heijning, B.J.; Verkade, H.J.; van der Beek, E.M. Reduced linoleic acid intake in early postnatal life improves metabolic outcomes in adult rodents following a Western-style diet challenge. Nutr. Res. 2015, 35, 800–811. [Google Scholar] [CrossRef] [PubMed]
- Francisco, V.; Pino, J.; Campos-Cabaleiro, V.; Ruiz-Fernández, C.; Mera, A.; Gonzalez-Gay, M.A.; Gómez, R.; Gualillo, O. Obesity, fat mass and immune system: Role for leptin. Front. Physiol. 2018, 9, 640. [Google Scholar] [CrossRef]
- Chehab, F.F.; Qiu, J.; Mounzih, K.; Ewart-Toland, A.; Ogus, S. Leptin and reproduction. Nutr. Rev. 2002, 60 (Suppl. 10), S39–S46. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Catteau, A.; Caillon, H.; Barrière, P.; Denis, M.; Masson, D.; Fréour, T. Leptin and its potential interest in assisted reproduction cycles. Hum. Reprod. Update 2016, 22, 320–341. [Google Scholar] [CrossRef]
- Gabriely, I.; Ma, X.H.; Yang, X.M.; Rossetti, L.; Barzilai, N. Leptin resistance during aging is independent of fat mass. Diabetes 2002, 51, 1016–1021. [Google Scholar] [CrossRef]
- Abella, V.; Scotece, M.; Conde, J.; Pino, J.; Gonzalez-Gay, M.A.; Gomez-Reino, J.J.; Mera, A.; Lago, F.; Gómez, R.; Gualillo, O. Leptin in the interplay of inflammation, metabolism and immune system disorders. Nat. Rev. Rheumatol. 2017, 13, 100. [Google Scholar] [CrossRef] [PubMed]
- Petersen, K.F.; Oral, E.A.; Dufour, S.; Befroy, D.; Ariyan, C.; Yu, C.; Cline, G.W.; DePaoli, A.M.; Taylor, S.I.; Gorden, P. Leptin reverses insulin resistance and hepatic steatosis in patients with severe lipodystrophy. J. Clin. Investig. 2002, 109, 1345–1350. [Google Scholar] [CrossRef] [PubMed]
- Fang, H.; Judd, R.L. Adiponectin regulation and function. Compr. Physiol. 2011, 8, 1031–1063. [Google Scholar]
- Pérez-Matute, P.; Martínez, J.; Marti, A.; Moreno-Aliaga, M. Linoleic acid decreases leptin and adiponectin secretion from primary rat adipocytes in the presence of insulin. Lipids 2007, 42, 913–920. [Google Scholar] [CrossRef] [PubMed]
- Standl, M.; Demmelmair, H.; Koletzko, B.; Heinrich, J. Cord blood n-3 LC-PUFA is associated with adiponectin concentrations at 10 years of age. Prostaglandins Leukot. Essent. Fatty Acids 2015, 96, 51–55. [Google Scholar] [CrossRef]
- Leamy, A.K.; Egnatchik, R.A.; Young, J.D. Molecular mechanisms and the role of saturated fatty acids in the progression of non-alcoholic fatty liver disease. Prog. Lipid Res. 2013, 52, 165–174. [Google Scholar] [CrossRef]
- Sekar, S.; Shafie, S.R.; Prasadam, I.; Crawford, R.; Panchal, S.K.; Brown, L.; Xiao, Y. Saturated fatty acids induce development of both metabolic syndrome and osteoarthritis in rats. Sci. Rep. 2017, 7, 46457. [Google Scholar] [CrossRef]
- Guo, X.-f.; Li, X.; Shi, M.; Li, D. n-3 polyunsaturated fatty acids and metabolic syndrome risk: A meta-analysis. Nutrients 2017, 9, 703. [Google Scholar] [CrossRef]
- Ghazali, R.; Mehta, K.J.; Bligh, S.A.; Tewfik, I.; Clemens, D.; Patel, V.B. High omega arachidonic acid/docosahexaenoic acid ratio induces mitochondrial dysfunction and altered lipid metabolism in human hepatoma cells. World J. Hepatol. 2020, 12, 84. [Google Scholar] [CrossRef]
- Sookoian, S.; Pirola, C.J. Liver enzymes, metabolomics and genome-wide association studies: From systems biology to the personalized medicine. World J. Gastroenterol. 2015, 21, 711. [Google Scholar] [CrossRef]
- Charatcharoenwitthaya, P.; Lindor, K.D.; Angulo, P. The spontaneous course of liver enzymes and its correlation in nonalcoholic fatty liver disease. Dig. Dis. Sci. 2012, 57, 1925–1931. [Google Scholar] [CrossRef]
- Shi, C.; Xue, W.; Han, B.; Yang, F.; Yin, Y.; Hu, C. Acetaminophen aggravates fat accumulation in NAFLD by inhibiting autophagy via the AMPK/mTOR pathway. Eur. J. Pharmacol. 2019, 850, 15–22. [Google Scholar] [CrossRef]
- Lima, W.G.; Martins-Santos, M.E.S.; Chaves, V.E. Uric acid as a modulator of glucose and lipid metabolism. Biochimie 2015, 116, 17–23. [Google Scholar] [CrossRef]
- Fathallah-Shaykh, S.A.; Cramer, M.T. Uric acid and the kidney. Pediatr. Nephrol. 2014, 29, 999–1008. [Google Scholar] [CrossRef]
- Hooper, L.; Al-Khudairy, L.; Abdelhamid, A.S.; Rees, K.; Brainard, J.S.; Brown, T.J.; Ajabnoor, S.M.; O’Brien, A.T.; Winstanley, L.E.; Donaldson, D.H.; et al. Omega-6 fats for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2018, 7, Cd011094. [Google Scholar] [PubMed]
- Torres-Castillo, N.; Silva-Gómez, J.A.; Campos-Perez, W.; Barron-Cabrera, E.; Hernandez-Cañaveral, I.; Garcia-Cazarin, M.; Marquez-Sandoval, Y.; Gonzalez-Becerra, K.; Barron-Gallardo, C.; Martinez-Lopez, E. High dietary ω-6: ω-3 PUFA ratio is positively associated with excessive adiposity and waist circumference. Obes. Facts 2018, 11, 344–353. [Google Scholar] [CrossRef] [PubMed]
- Marchix, J.; Choque, B.; Kouba, M.; Fautrel, A.; Catheline, D.; Legrand, P. Excessive dietary linoleic acid induces proinflammatory markers in rats. J. Nutr. Biochem. 2015, 26, 1434–1441. [Google Scholar] [CrossRef] [PubMed]
- Ness, G.C. Physiological feedback regulation of cholesterol biosynthesis: Role of translational control of hepatic HMG-CoA reductase and possible involvement of oxylanosterols. Biochim. Biophys. Acta 2015, 1851, 667–673. [Google Scholar] [CrossRef]
- Ohtani, H.; Hayashi, K.; Hirata, Y.; Dojo, S.; Nakashima, K.; Nishio, E.; Kurushima, H.; Saeki, M.; Kajiyama, G. Effects of dietary cholesterol and fatty acids on plasma cholesterol level and hepatic lipoprotein metabolism. J. Lipid Res. 1990, 31, 1413–1422. [Google Scholar] [CrossRef]
- Wang, S.; Matthan, N.R.; Wu, D.; Reed, D.B.; Bapat, P.; Yin, X.; Grammas, P.; Shen, C.L.; Lichtenstein, A.H. Lipid content in hepatic and gonadal adipose tissue parallel aortic cholesterol accumulation in mice fed diets with different omega-6 PUFA to EPA plus DHA ratios. Clin. Nutr. 2014, 33, 260–266. [Google Scholar] [CrossRef]
- Schlaepfer, I.R.; Joshi, M. CPT1A-mediated fat oxidation, mechanisms, and therapeutic potential. Endocrinology 2020, 161, bqz046. [Google Scholar] [CrossRef] [PubMed]
- Tobi, E.W.; Goeman, J.J.; Monajemi, R.; Gu, H.; Putter, H.; Zhang, Y.; Slieker, R.C.; Stok, A.P.; Thijssen, P.E.; Müller, F. DNA methylation signatures link prenatal famine exposure to growth and metabolism. Nat. Commun. 2014, 5, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Moody, L.; Xu, G.B.; Chen, H.; Pan, Y.-X. Epigenetic regulation of carnitine palmitoyltransferase 1 (Cpt1a) by high fat diet. Biochim. Biophys. Acta Gene Regul. Mech. 2019, 1862, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.F.; Tian, M.X.; Sun, R.Q.; Zhang, M.L.; Zhou, L.S.; Jin, L.; Chen, L.L.; Zhou, W.J.; Duan, K.L.; Chen, Y.J. SIRT 5 inhibits peroxisomal ACOX 1 to prevent oxidative damage and is downregulated in liver cancer. EMBO Rep. 2018, 19, e45124. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Sun, Q.; Zong, Y.; Liu, J.; Idriss, A.A.; Omer, N.A.; Zhao, R. Prenatal betaine exposure alleviates corticosterone-induced inhibition of CYP27A1 expression in the liver of juvenile chickens associated with its promoter DNA methylation. Gen. Comp. Endocrinol. 2017, 246, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Choque, B.; Catheline, D.; Delplanque, B.; Guesnet, P.; Legrand, P. Dietary linoleic acid requirements in the presence of α-linolenic acid are lower than the historical 2% of energy intake value, study in rats. Br. J. Nutr. 2015, 113, 1056–1068. [Google Scholar] [CrossRef]
- Liu, G.; Muhlhausler, B.S.; Gibson, R.A. A method for long term stabilisation of long chain polyunsaturated fatty acids in dried blood spots and its clinical application. Prostaglandins Leukot. Essent. Fatty Acids 2014, 91, 251–260. [Google Scholar] [CrossRef]
- Huang, T.H.; Peng, G.; Li, G.Q.; Yamahara, J.; Roufogalis, B.D.; Li, Y. Salacia oblonga root improves postprandial hyperlipidemia and hepatic steatosis in Zucker diabetic fatty rats: Activation of PPAR-alpha. Toxicol. Appl. Pharmacol. 2006, 210, 225–235. [Google Scholar]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef]
| Organ and Body Weight | LLA Maternal Diet | HLA Maternal Diet | Two-Way ANOVA | ||||
|---|---|---|---|---|---|---|---|
| LLA PN Diet | HLA PN Diet | LLA PN Diet | HLA PN Diet | Pmaternal | Ppostnatal | Pint | |
| Male | |||||||
| Body weight (gm) | 388.2 ± 5.4600 | 378.3 ± 6.5800 | 384.2 ± 6.3900 | 374.9 ± 5.0900 | ns | ns | ns |
| % Left kidney | 0.314 ± 0.0050 | 0.313 ± 0.0010 | 0.316 ± 0.0030 | 0.310 ± 0.0050 | ns | ns | ns |
| % Right kidney | 0.317 ± 0.0060 | 0.307 ± 0.0020 | 0.309 ± 0.0040 | 0.307 ± 0.0060 | ns | ns | ns |
| % Left adrenal | 0.005 ± 0.0001 | 0.005 ± 0.0005 | 0.004 ± 0.0001 | 0.004 ± 0.0002 | ns | ns | ns |
| % Right adrenal | 0.004 ± 0.0002 | 0.004 ± 0.0001 | 0.004 ± 0.0005 | 0.004 ± 0.0003 | ns | ns | ns |
| % Liver | 3.06 ± 0.0300 | 2.95 ± 0.0300 | 2.97 ± 0.0500 | 2.95 ± 0.0500 | ns | ns | ns |
| % Heart | 0.333 ± 0.0100 | 0.337 ± 0.0100 | 0.331 ± 0.0060 | 0.327 ± 0.0020 | ns | ns | ns |
| % Brain | 0.541 ± 0.0090 | 0.571 ± 0.0050 | 0.555 ± 0.0080 | 0.576 ± 0.0050 | ns | p = 0.003 | ns |
| % GAS | 0.554 ± 0.0100 | 0.548 ± 0.0090 | 0.558 ± 0.0100 | 0.580 ± 0.0200 | ns | ns | ns |
| % SOL | 0.035 ± 0.0010 | 0.036 ± 0.0009 | 0.035 ± 0.0010 | 0.036 ± 0.0010 | ns | ns | ns |
| % TA | 0.163 ± 0.0060 | 0.159 ± 0.0010 | 0.159 ± 0.0060 | 0.160 ± 0.0020 | ns | ns | ns |
| % EDL | 0.047 ± 0.0020 | 0.051 ± 0.0007 | 0.050 ± 0.0006 | 0.047 ± 0.0020 | ns | ns | ns |
| Female | |||||||
| Body weight (gm) | 230.6 ± 4.0000 | 240.2 ± 2.2800 | 241.4 ± 2.0200 | 238.1 ± 3.9400 | ns | ns | ns |
| % Left kidney | 0.304 ± 0.0040 | 0.313 ± 0.0010 | 0.305 ± 0.0040 | 0.296 ± 0.0040 | ns | ns | p = 0.03 |
| % Right kidney | 0.306 ± 0.0070 | 0.316 ± 0.0030 | 0.302 ± 0.0010 | 0.305 ± 0.0060 | ns | ns | ns |
| % Left adrenal | 0.01 ± 0.0010 | 0.01 ± 0.0006 | 0.01 ± 0.0005 | 0.01 ± 0.00010 | ns | ns | ns |
| % Right adrenal | 0.01 ± 0.0009 | 0.01 ± 0.0003 | 0.01 ± 0.0007 | 0.01 ± 0.00090 | ns | ns | ns |
| % Liver | 3.69 ± 0.0500 | 3.68 ± 0.0200 | 3.67 ± 0.0500 | 3.61 ± 0.0100 | ns | ns | ns |
| % Heart | 0.346 ± 0.0050 | 0.384 ± 0.0200 | 0.357 ± 0.0040 | 0.352 ± 0.0050 | ns | ns | ns |
| % Brain | 0.847 ± 0.0120 | 0.825 ± 0.0140 | 0.835 ± 0.0090 | 0.844 ± 0.0110 | ns | ns | ns |
| % GAS | 0.604 ± 0.0200 | 0.583 ± 0.0100 | 0.594 ± 0.0100 | 0.575 ± 0.010 | ns | ns | ns |
| % SOL | 0.042 ± 0.0010 | 0.037 ± 0.0010 | 0.041 ± 0.0008 | 0.043 ± 0.0010 | ns | ns | p = 0.011 |
| % TA | 0.174 ± 0.0020 | 0.172 ± 0.0020 | 0.165 ± 0.0010 | 0.164 ± 0.0040 | p = 0.005 | ns | ns |
| % EDL | 0.050 ± 0.0010 | 0.050 ± 0.0009 | 0.055 ± 0.0010 | 0.055 ± 0.0010 | p = 0.002 | ns | ns |
| % Left ovary | 0.015 ± 0.0008 | 0.016 ± 0.0010 | 0.017 ± 0.0008 | 0.017 ± 0.0010 | ns | ns | ns |
| % Right ovary | 0.017 ± 0.0008 | 0.016 ± 0.0008 | 0.017 ± 0.0008 | 0.018 ± 0.0005 | ns | ns | ns |
| Plasma Fatty Acid | LLA Maternal Diet | HLA Maternal Diet | Two-Way ANOVA | ||||
|---|---|---|---|---|---|---|---|
| LLA PN Diet | HLA PN Diet | LLA PN Diet | HLA PN Diet | Pmaternal | Ppostnatal | Pint | |
| Male | |||||||
| Total SFA | 33.41 ± 0.3600 | 32.51 ± 0.2100 | 33.85 ± 0.1800 | 32.61 ± 0.3300 | ns | p = 0.003 | ns |
| Total trans FA | 0.10 ± 0.0000 | 0.10 ± 0.0000 | 0.10 ± 0.0000 | 0.10 ± 0.0000 | ns | ns | ns |
| Total MUFA | 15.61 ± 1.4300 | 7.83 ± 0.3100 | 14.56 ± 0.9700 | 9.11 ± 0.5400 | ns | p < 0.0001 | ns |
| Total n-9 FA | 12.27 ± 1.1500 | 5.70 ± 0.2300 | 11.12 ± 0.8100 | 6.58 ± 0.3900 | ns | p < 0.0001 | ns |
| Total n-7 FA | 3.28 ± 0.3300 | 2.08 ± 0.1100 | 3.34 ± 0.3300 | 2.55 ± 0.1600 | ns | p = 0.0011 | ns |
| Total n-3 FA | 3.65 ± 0.1200 | 2.63 ± 0.0600 | 3.80 ± 0.0800 | 2.66 ± 0.0800 | ns | p < 0.0001 | ns |
| ALA (18:3n-3) | 0.32 ± 0.0400 | 0.21 ± 0.0100 | 0.26 ± 0.0400 | 0.20 ± 0.0000 | ns | p = 0.012 | ns |
| EPA (20:5n-3) | 0.25 ± 0.0600 | 0.10 ± 0.0000 | 0.20 ± 0.0000 | 0.10 ± 0.0000 | ns | p = 0.04 | ns |
| DPA (22:5n-3) | 0.44 ± 0.0200 | 0.35 ± 0.0200 | 0.50 ± 0.0300 | 0.40 ± 0.0000 | p = 0.019 | p = 0.0002 | ns |
| DHA (22:6n-3) | 2.64 ± 0.0600 | 2.03 ± 0.0800 | 2.86 ± 0.1100 | 2.01 ± 0.0700 | ns | p < 0.0001 | ns |
| Total n-6 FA | 47.21 ± 1.7400 | 56.93 ± 0.300 | 48.20 ± 0.8800 | 55.50 ± 0.6900 | ns | p < 0.0001 | ns |
| LA (18:2n-6) | 16.44 ± 0.6100 | 20.60 ± 0.5500 | 15.74 ± 1.0600 | 20.45 ± 0.6400 | ns | p < 0.0001 | ns |
| AA (20:4n-6) | 29.24 ± 1.0700 | 34.18 ± 0.5600 | 30.92 ± 0.6800 | 32.76 ± 0.7100 | ns | p = 0.0006 | ns |
| LA/ALA | 56.47 ± 9.1500 | 105.8 ± 5.9000 | 58.47 ± 4.5100 | 99.92 ± 3.6000 | ns | p < 0.0001 | ns |
| AA/DHA | 11.1 ± 0.5900 | 16.81 ± 0.5600 | 10.85 ± 0.2800 | 16.31 ± 0.3900 | ns | p < 0.0001 | ns |
| DHA/DPA | 5.96 ± 0.3100 | 6.02 ± 0.3200 | 5.98 ± 0.3800 | 5.37 ± 0.1100 | ns | ns | ns |
| Female | |||||||
| Total SFA | 34.55 ± 0.3100 | 34.90 ± 0.2500 | 35.02 ± 0.3800 | 35.02 ± 0.1900 | ns | ns | ns |
| Total trans FA | 0.12 ± 0.0100 | 0.10 ± 0.0000 | 0.10 ± 0.0000 | 0.10 ± 0.0000 | ns | ns | ns |
| Total MUFA | 13.74 ± 0.7800 | 8.82 ± 0.5600 | 12.45 ± 0.7300 | 7.81 ± 0.3700 | ns | p < 0.0001 | ns |
| Total n-9 FA | 11.20 ± 0.6900 | 6.63 ± 0.4700 | 9.94 ± 0.6400 | 5.75 ± 0.2900 | ns | p < 0.0001 | ns |
| Total n-7 FA | 2.45 ± 0.0900 | 2.13 ± 0.1000 | 2.40 ± 0.0800 | 1.95 ± 0.0900 | ns | p = 0.0005 | ns |
| Total n-3 FA | 4.94 ± 0.1500 | 3.86 ± 0.0500 | 4.92 ± 0.1500 | 3.96 ± 0.0500 | ns | p < 0.0001 | ns |
| ALA (18:3n-3) | 0.28 ± 0.0400 | 0.14 ± 0.0100 | 0.20 ± 0.0000 | 0.15 ± 0.0200 | ns | p = 0.0009 | ns |
| EPA (20:5n-3) | 0.15 ± 0.0300 | 0.10 ± 0.0000 | 0.10 ± 0.0000 | 0.04 ± 0.0200 | p = 0.014 | p = 0.014 | ns |
| DPA (22:5n-3) | 0.27 ± 0.0200 | 0.21 ± 0.0100 | 0.20 ± 0.0200 | 0.20 ± 0.0000 | p = 0.04 | ns | ns |
| DHA (22:6n-3) | 4.22 ± 0.1300 | 3.48 ± 0.0600 | 4.41 ± 0.1400 | 3.56 ± 0.0600 | ns | p < 0.0001 | ns |
| Total n-6 FA | 46.64 ± 0.7700 | 52.27 ± 0.6200 | 47.48 ± 0.6600 | 53.40 ± 0.4400 | ns | p < 0.0001 | ns |
| LA (18:2n-6) | 14.20 ± 0.9800 | 16.02 ± 0.8100 | 13.51 ± 0.9000 | 16.98 ± 0.7800 | ns | p = 0.006 | ns |
| AA (20:4n-6) | 30.94 ± 0.8500 | 34.01 ± 0.8300 | 32.38 ± 0.4400 | 34.25 ± 0.6000 | ns | p = 0.002 | ns |
| LA/ALA | 52.21 ± 4.8400 | 111.01 ± 5.3400 | 79.22 ± 6.2600 | 118.23 ± 2.8600 | p = 0.004 | p < 0.0001 | ns |
| AA/DHA | 7.34 ± 0.1400 | 9.77 ± 0.2400 | 7.34 ± 0.1500 | 9.31 ± 0.2300 | ns | p < 0.0001 | ns |
| DHA/DPA | 16.87 ± 1.0800 | 17.27 ± 1.1100 | 20.65 ± 1.0100 | 18.39 ± 0.6700 | p = 0.02 | ns | ns |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shrestha, N.; Vidimce, J.; Holland, O.J.; Cuffe, J.S.M.; Beck, B.R.; Perkins, A.V.; McAinch, A.J.; Hryciw, D.H. Maternal and Postnatal High Linoleic Acid Diet Impacts Lipid Metabolism in Adult Rat Offspring in a Sex-Specific Manner. Int. J. Mol. Sci. 2021, 22, 2946. https://doi.org/10.3390/ijms22062946
Shrestha N, Vidimce J, Holland OJ, Cuffe JSM, Beck BR, Perkins AV, McAinch AJ, Hryciw DH. Maternal and Postnatal High Linoleic Acid Diet Impacts Lipid Metabolism in Adult Rat Offspring in a Sex-Specific Manner. International Journal of Molecular Sciences. 2021; 22(6):2946. https://doi.org/10.3390/ijms22062946
Chicago/Turabian StyleShrestha, Nirajan, Josif Vidimce, Olivia J. Holland, James S. M. Cuffe, Belinda R. Beck, Anthony V. Perkins, Andrew J. McAinch, and Deanne H. Hryciw. 2021. "Maternal and Postnatal High Linoleic Acid Diet Impacts Lipid Metabolism in Adult Rat Offspring in a Sex-Specific Manner" International Journal of Molecular Sciences 22, no. 6: 2946. https://doi.org/10.3390/ijms22062946
APA StyleShrestha, N., Vidimce, J., Holland, O. J., Cuffe, J. S. M., Beck, B. R., Perkins, A. V., McAinch, A. J., & Hryciw, D. H. (2021). Maternal and Postnatal High Linoleic Acid Diet Impacts Lipid Metabolism in Adult Rat Offspring in a Sex-Specific Manner. International Journal of Molecular Sciences, 22(6), 2946. https://doi.org/10.3390/ijms22062946









