Extracellular Vesicles as Biomarkers Carriers in Bladder Cancer: Diagnosis, Surveillance, and Treatment
Abstract
1. Introduction
2. Biology and Function of EVs
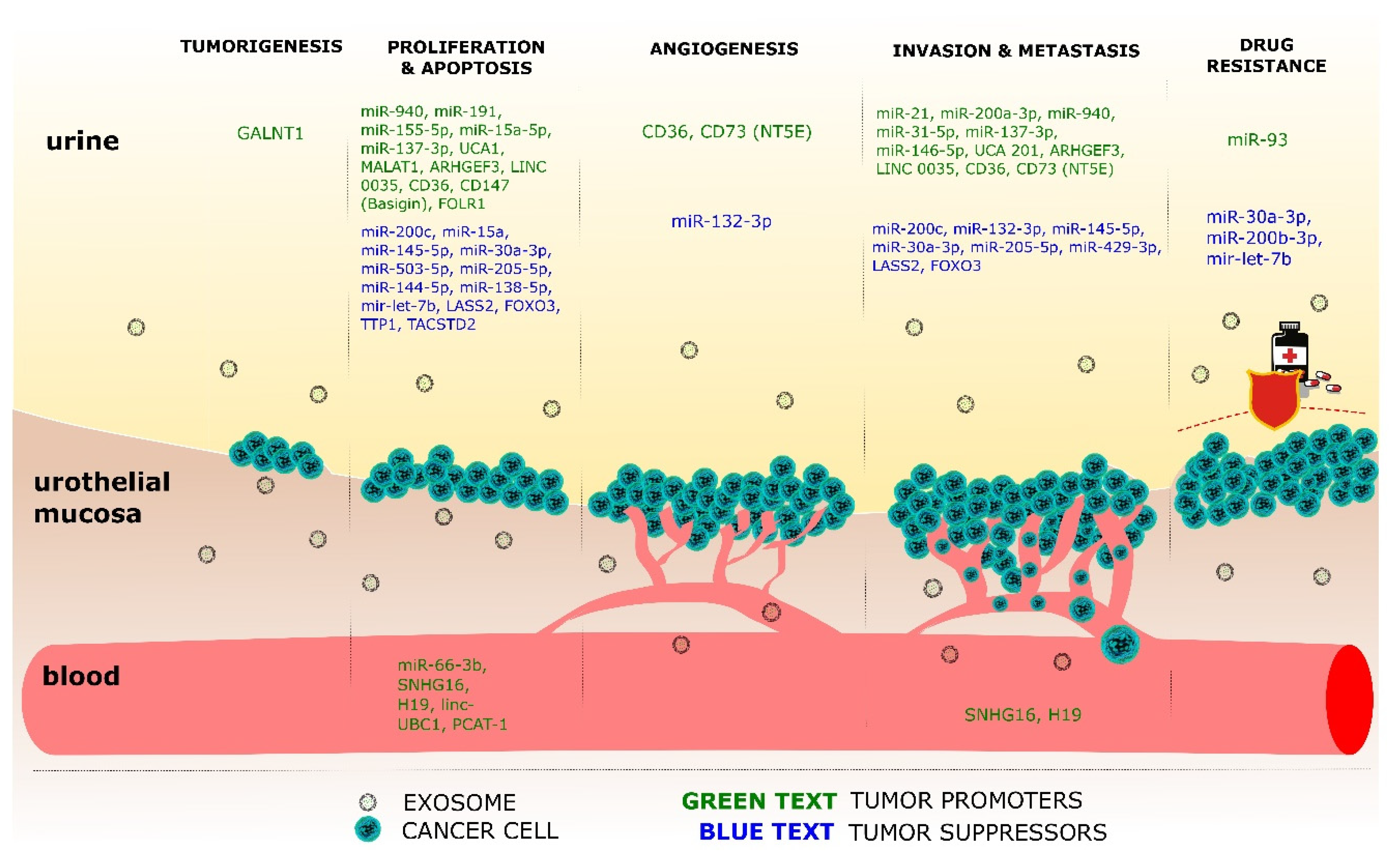
3. EVs in Bladder Cancer Pathogenesis
4. EVs as Biomarker Carriers for Bladder Cancer Diagnosis
4.1. Protein Biomarkers
4.2. MicroRNAs and IncRNAs
5. EVs as Biomarkers Carriers for Bladder Cancer Disease Progression and Recurrence
6. EVs as Potential Therapeutic Vectors
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012: Global Cancer Statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Dahm, P.; Gschwend, J.E. Malignant Non-Urothelial Neoplasms of the Urinary Bladder: A Review. Eur. Urol. 2003, 44, 672–681. [Google Scholar] [CrossRef]
- AiRtUM Working Group. Available online: www.epiprev.it (accessed on 26 November 2020).
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- DeGeorge, K.C.; Holt, H.R.; Hodges, S.C. Bladder Cancer: Diagnosis and Treatment. Am. Fam. Physician 2017, 96, 507–514. Available online: www.aafp.org/afp (accessed on 22 December 2020). [PubMed]
- Lotan, Y.; Roehrborn, C.G. Sensitivity and specificity of commonly available bladder tumor markers versus cytology: Results of a comprehensive literature review and meta-analyses. Urology 2003, 61, 109–118. [Google Scholar] [CrossRef]
- Amling, C.L. Diagnosis and management of superficial bladder cancer. Curr. Probl. Cancer 2001, 25, 224–278. [Google Scholar] [CrossRef] [PubMed]
- Mowatt, G.; N’Dow, J.; Vale, L.; Nabi, G.; Boachie, C.; Cook, J.A.; Fraser, C.; Griffiths, T.R.L. Photodynamic diagnosis of bladder cancer compared with white light cystoscopy: Systematic review and meta-analysis. Int. J. Technol. Assess. Heal. Care 2011, 27, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W. Cystoscopy. In Bladder Cancer; Elsevier BV: Amsterdam, The Netherlands, 2018; pp. 83–86. [Google Scholar]
- Duquesne, I.; Weisbach, L.; Aziz, A.; Kluth, L.A.; Xylinas, E. The contemporary role and impact of urine-based biomarkers in bladder cancer. Transl. Androl. Urol. 2017, 6, 1031–1042. [Google Scholar] [CrossRef]
- De Oliveira, M.C.; Caires, H.R.; Oliveira, M.J.; Fraga, A.; Vasconcelos, M.H.; Ribeiro, R. Urinary Biomarkers in Bladder Cancer: Where Do We Stand and Potential Role of Extracellular Vesicles. Cancers 2020, 12, 1400. [Google Scholar] [CrossRef]
- Sylvester, R.J. Natural History, Recurrence, and Progression in Superficial Bladder Cancer. Sci. World J. 2006, 6, 2617–2625. [Google Scholar] [CrossRef]
- Simeone, P.; Bologna, G.; Lanuti, P.; Pierdomenico, L.; Guagnano, M.T.; Pieragostino, D.; Del Boccio, P.; Vergara, D.; Marchisio, M.; Miscia, S.; et al. Extracellular Vesicles as Signaling Mediators and Disease Biomarkers across Biological Barriers. Int. J. Mol. Sci. 2020, 21, 2514. [Google Scholar] [CrossRef]
- Huyan, T.; Li, H.; Peng, H.; Chen, J.; Yang, R.; Zhang, W.; Li, Q. Extracellular Vesicles—Advanced Nanocarriers in Cancer Therapy: Progress and Achievements. Int. J. Nanomed. 2020, 15, 6485–6502. [Google Scholar] [CrossRef]
- Xu, R.; Rai, A.; Chen, M.; Suwakulsiri, W.; Greening, D.W.; Simpson, R.J. Extracellular vesicles in cancer—Implications for future improvements in cancer care. Nat. Rev. Clin. Oncol. 2018, 15, 617–638. [Google Scholar] [CrossRef] [PubMed]
- Van Niel, G.; D’Angelo, G.; Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 2018, 19, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Fabbiano, F.; Corsi, J.; Gurrieri, E.; Trevisan, C.; Notarangelo, M.; D’Agostino, V.G. RNA packaging into extracellular vesicles: An orchestra of RNA-binding proteins? J. Extracell. Vesicles 2020, 10, e12043. [Google Scholar] [CrossRef] [PubMed]
- Maas, S.L.; Breakefield, X.O.; Weaver, A.M. Extracellular Vesicles: Unique Intercellular Delivery Vehicles. Trends Cell Biol. 2017, 27, 172–188. [Google Scholar] [CrossRef] [PubMed]
- Masaoutis, C.; Al Besher, S.; Koutroulis, I.; Theocharis, S. Exosomes in Nephropathies: A Rich Source of Novel Biomarkers. Dis. Markers 2020, 2020, 8897833. [Google Scholar] [CrossRef]
- Zhang, X.; Yuan, X.; Shi, H.; Wu, L.; Qian, H.; Xu, W. Exosomes in cancer: Small particle, big player. J. Hematol. Oncol. 2015, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R. The biology and function of exosomes in cancer. J. Clin. Investig. 2016, 126, 1208–1215. [Google Scholar] [CrossRef] [PubMed]
- Han, L.; Lam, E.W.-F.; Sun, Y. Extracellular vesicles in the tumor microenvironment: Old stories, but new tales. Mol. Cancer 2019, 18, 1–14. [Google Scholar] [CrossRef]
- Ogorevc, E.; Hudoklin, S.; Veranič, P.; Kralj-Iglič, V. Extracellular vesicle-mediated transfer of membranous components from the highly malignant T24 urinary carcinoma cell line to the non-malignant RT4 urinary papilloma cell line. Protoplasma 2013, 251, 699–702. [Google Scholar] [CrossRef] [PubMed]
- Rosivatz, E.; Becker, I.; Bamba, M.; Schott, C.; Diebold, J.; Mayr, D.; Höfler, H.; Becker, K.-F. Neoexpression of N-cadherin in E-cadherin positive colon cancers. Int. J. Cancer 2004, 111, 711–719. [Google Scholar] [CrossRef]
- Gravdal, K.; Halvorsen, O.J.; Haukaas, S.A.; Akslen, L.A. A Switch from E-Cadherin to N-Cadherin Expression Indicates Epithelial to Mesenchymal Transition and Is of Strong and Independent Importance for the Progress of Prostate Cancer. Clin. Cancer Res. 2007, 13, 7003–7011. [Google Scholar] [CrossRef] [PubMed]
- Loh, C.-Y.; Chai, J.Y.; Tang, T.F.; Wong, W.F.; Sethi, G.; Shanmugam, M.K.; Chong, P.P.; Looi, C.Y. The E-Cadherin and N-Cadherin Switch in Epithelial-to-Mesenchymal Transition: Signaling, Therapeutic Implications, and Challenges. Cells 2019, 8, 1118. [Google Scholar] [CrossRef]
- Wu, C.-H.; Silvers, C.R.; Messing, E.M.; Lee, Y.-F. Bladder cancer extracellular vesicles drive tumorigenesis by inducing the unfolded protein response in endoplasmic reticulum of nonmalignant cells. J. Biol. Chem. 2019, 294, 3207–3218. [Google Scholar] [CrossRef] [PubMed]
- Ribatti, D.; Tamma, R.; Annese, T. Epithelial-Mesenchymal Transition in Cancer: A Historical Overview. Transl. Oncol. 2020, 13, 100773. [Google Scholar] [CrossRef] [PubMed]
- Gooding, A.J.; Schiemann, W.P. Epithelial–Mesenchymal Transition Programs and Cancer Stem Cell Phenotypes: Mediators of Breast Cancer Therapy Resistance. Mol. Cancer Res. 2020, 18, 1257–1270. [Google Scholar] [CrossRef] [PubMed]
- Franzen, C.A.; Blackwell, R.H.; Todorovic, V.; Greco, K.A.; Foreman, K.E.; Flanigan, R.C.; Kuo, P.C.; Gupta, G. Urothelial cells undergo epithelial-to-mesenchymal transition after exposure to muscle invasive bladder cancer exosomes. Oncogenesis 2015, 4, e163. [Google Scholar] [CrossRef]
- Goulet, C.R.; Champagne, A.; Bernard, G.; Vandal, D.; Chabaud, S.; Pouliot, F.; Bolduc, S. Cancer-associated fibroblasts induce epithelial–mesenchymal transition of bladder cancer cells through paracrine IL-6 signalling. BMC Cancer 2019, 19, 1–13. [Google Scholar] [CrossRef]
- Yang, L.; Wu, X.-H.; Wang, D.; Luo, C.-L.; Chen, L.-X. Bladder cancer cell-derived exosomes inhibit tumor cell apoptosis and induce cell proliferation in vitro. Mol. Med. Rep. 2013, 8, 1272–1278. [Google Scholar] [CrossRef]
- Masaoutis, C.; Mihailidou, C.; Tsourouflis, G.; Theocharis, S. Exosomes in lung cancer diagnosis and treatment. From the translating research into future clinical practice. Biochimie 2018, 151, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Masaoutis, C.; Korkolopoulou, P.; Theocharis, S. Exosomes in sarcomas: Tiny messengers with broad implications in diagnosis, surveillance, prognosis and treatment. Cancer Lett. 2019, 449, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Isola, A.L.; Chen, S. Exosomes: The Messengers of Health and Disease. Curr. Neuropharmacol. 2016, 15, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Witwer, K.W.; Buzás, E.I.; Bemis, L.T.; Bora, A.; Lässer, C.; Lötvall, J.; Hoen, E.N.N.; Piper, M.G.; Sivaraman, S.; Skog, J.; et al. Standardization of sample collection, isolation and analysis methods in extracellular vesicle research. J. Extracell. Vesicles 2013, 2, 20360. [Google Scholar] [CrossRef]
- Doyle, L.M.; Wang, M.Z. Overview of Extracellular Vesicles, Their Origin, Composition, Purpose, and Methods for Exosome Isolation and Analysis. Cells 2019, 8, 727. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Zhang, T.; Zheng, M.; Liu, Y.; Chen, Z. Exosomal proteins as potential markers of tumor diagnosis. J. Hematol. Oncol. 2017, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Welton, J.L.; Khanna, S.; Giles, P.J.; Brennan, P.; Brewis, I.A.; Staffurth, J.; Mason, M.D.; Clayton, A. Proteomics Analysis of Bladder Cancer Exosomes. Mol. Cell. Proteom. 2010, 9, 1324–1338. [Google Scholar] [CrossRef] [PubMed]
- Shvartsur, A.; Bonavida, B. Trop2 and its overexpression in cancers: Regulation and clinical/ therapeutic implications. Genes Cancer 2014, 6, 84–105. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Li, B.; Gao, N.; Xia, H.; Men, Y.; Liu, Y.; Liu, Z.; Chen, Q.; Li, L. Expression of tumor-associated calcium signal transducer 2 in patients with salivary adenoid cystic carcinoma: Correlation with clinicopathological features and prognosis. Oncol. Lett. 2014, 8, 1670–1674. [Google Scholar] [CrossRef][Green Version]
- Chen, C.-L.; Lai, Y.-F.; Tang, P.; Chien, K.-Y.; Yu, J.-S.; Tsai, C.-H.; Chen, H.-W.; Wu, C.-C.; Chung, T.; Hsu, C.-W.; et al. Comparative and Targeted Proteomic Analyses of Urinary Microparticles from Bladder Cancer and Hernia Patients. J. Proteome Res. 2012, 11, 5611–5629. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.-Y.; Chang, C.-H.; Wu, H.-C.; Lin, C.-C.; Chang, K.-P.; Yang, C.-R.; Huang, C.-P.; Hsu, W.-H.; Chang, C.-T.; Chen, C.-J. Proteome Profiling of Urinary Exosomes Identifies Alpha 1-Antitrypsin and H2B1K as Diagnostic and Prognostic Biomarkers for Urothelial Carcinoma. Sci. Rep. 2016, 6, 34446. [Google Scholar] [CrossRef] [PubMed]
- Yazarlou, F.; Mowla, S.J.; Oskooei, V.K.; Motevaseli, E.; Tooli, L.F.; Afsharpad, M.; Nekoohesh, L.; Sanikhani, N.S.; Ghafouri-Fard, S.; Modarressi, M.H. Urine exosome gene expression of cancer-testis antigens for prediction of bladder carcinoma. Cancer Manag. Res. 2018, 10, 5373–5381. [Google Scholar] [CrossRef] [PubMed]
- Wahid, F.; Shehzad, A.; Khan, T.; Kim, Y.Y. MicroRNAs: Synthesis, mechanism, function, and recent clinical trials. Biochim. Biophys. Acta Bioenerg. 2010, 1803, 1231–1243. [Google Scholar] [CrossRef]
- Pérez, A.; Loizaga, A.; Arceo, R.; Lacasa, I.; Rabade, A.; Zorroza, K.; Mosen-Ansorena, D.; Gonzalez, E.; Aransay, A.M.; Falcón-Pérez, J.M.; et al. A Pilot Study on the Potential of RNA-Associated to Urinary Vesicles as a Suitable Non-Invasive Source for Diagnostic Purposes in Bladder Cancer. Cancers 2014, 6, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Matsuzaki, K.; Fujita, K.; Jingushi, K.; Kawashima, A.; Ujike, T.; Nagahara, A.; Ueda, Y.; Tanigawa, G.; Yoshioka, I.; Ueda, K.; et al. MiR-21-5p in urinary extracellular vesicles is a novel biomarker of urothelial carcinoma. Oncotarget 2017, 8, 24668–24678. [Google Scholar] [CrossRef] [PubMed]
- De Long, J.; Sullivan, T.B.; Humphrey, J.; Logvinenko, T.; Summerhayes, K.A.; Kozinn, S.; Harty, N.; Summerhayes, I.C.; Libertino, J.A.; Holway, A.H.; et al. A non-invasive miRNA based assay to detect bladder cancer in cell-free urine. Am. J. Transl. Res. 2015, 7, 2500–2509. [Google Scholar]
- Yin, X.; Zheng, X.; Liu, M.; Wang, D.; Sun, H.; Qiu, Y.; Chen, J.; Shi, B. Exosomal miR-663b targets Ets2-repressor factor to promote proliferation and the epithelial–mesenchymal transition of bladder cancer cells. Cell Biol. Int. 2019, 44, 958–965. [Google Scholar] [CrossRef] [PubMed]
- Prensner, J.R.; Iyer, M.K.; Balbin, O.A.; Dhanasekaran, S.M.; Cao, Q.; Brenner, J.C.; Laxman, B.; Asangani, I.A.; Grasso, C.S.; Kominsky, H.D.; et al. Transcriptome sequencing across a prostate cancer cohort identifies PCAT-1, an unannotated lincRNA implicated in disease progression. Nat. Biotechnol. 2011, 29, 742–749. [Google Scholar] [CrossRef] [PubMed]
- Kogo, R.; Shimamura, T.; Mimori, K.; Kawahara, K.; Imoto, S.; Sudo, T.; Tanaka, F.; Shibata, K.; Suzuki, A.; Komune, S.; et al. Long Noncoding RNA HOTAIR Regulates Polycomb-Dependent Chromatin Modification and Is Associated with Poor Prognosis in Colorectal Cancers. Cancer Res. 2011, 71, 6320–6326. [Google Scholar] [CrossRef]
- Kung, J.T.Y.; Colognori, D.; Lee, J.T. Long Noncoding RNAs: Past, Present, and Future. Genetics 2013, 193, 651–669. [Google Scholar] [CrossRef] [PubMed]
- Gibb, E.A.; Brown, C.J.; Lam, W.L. The functional role of long non-coding RNA in human carcinomas. Mol. Cancer 2011, 10, 38. [Google Scholar] [CrossRef]
- Fang, Y.; Fullwood, M.J. Roles, Functions, and Mechanisms of Long Non-coding RNAs in Cancer. Genom. Proteom. Bioinform. 2016, 14, 42–54. [Google Scholar] [CrossRef]
- Yazarlou, F.; Modarressi, M.H.; Mowla, S.J.; Oskooei, V.K.; Motevaseli, E.; Tooli, L.F.; Nekoohesh, L.; Eghbali, M.; Ghafouri-Fard, S.; Afsharpad, M. Urinary exosomal expression of long non-coding RNAs as diagnostic marker in bladder cancer. Cancer Manag. Res. 2018, 10, 6357–6365. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Du, L.; Wang, L.; Jiang, X.; Zhan, Y.; Li, J.; Yan, K.; Duan, W.; Zhao, Y.; Wang, L.; et al. Evaluation of serum exosomal LncRNA-based biomarker panel for diagnosis and recurrence prediction of bladder cancer. J. Cell. Mol. Med. 2019, 23, 1396–1405. [Google Scholar] [CrossRef]
- Van Der Heijden, A.G.; Witjes, J.A. Recurrence, Progression, and Follow-Up in Non–Muscle-Invasive Bladder Cancer. Eur. Urol. Suppl. 2009, 8, 556–562. [Google Scholar] [CrossRef]
- Baumgart, S.; Meschkat, P.; Edelmann, P.; Heinzelmann, J.; Pryalukhin, A.; Bohle, R.; Heinzelbecker, J.; Stöckle, M.; Junker, K. MicroRNAs in tumor samples and urinary extracellular vesicles as a putative diagnostic tool for muscle-invasive bladder cancer. J. Cancer Res. Clin. Oncol. 2019, 145, 2725–2736. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yang, K.; Yuan, W.; Gao, Z. Determination of Serum Exosomal H19 as a Noninvasive Biomarker for Bladder Cancer Diagnosis and Prognosis. Med. Sci. Monit. 2018, 24, 9307–9316. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Y.; Li, Y.; Guan, B.; Wang, Z.; Peng, D.; Chen, Z.; He, A.; He, S.; Gong, Y.; Li, X.; et al. Long non-coding RNA HNF1A-AS1 promotes proliferation and suppresses apoptosis of bladder cancer cells through upregulating Bcl-2. Oncotarget 2017, 8, 76656–76665. [Google Scholar] [CrossRef] [PubMed]
- Mari, A.; Campi, R.; Tellini, R.; Gandaglia, G.; Albisinni, S.; Abufaraj, M.; Hatzichristodoulou, G.; Montorsi, F.; Van Velthoven, R.; Carini, M.; et al. Patterns and predictors of recurrence after open radical cystectomy for bladder cancer: A comprehensive review of the literature. World J. Urol. 2018, 36, 157–170. [Google Scholar] [CrossRef] [PubMed]
- Hiltbrunner, S.; Mints, M.; Eldh, M.; Rosenblatt, R.; Holmström, B.; Alamdari, F.; Johansson, M.; Veerman, R.E.; Winqvist, O.; Sherif, A.; et al. Urinary Exosomes from Bladder Cancer Patients Show a Residual Cancer Phenotype despite Complete Pathological Downstaging. Sci. Rep. 2020, 10, 5960. [Google Scholar] [CrossRef] [PubMed]
- Ostenfeld, M.S.; Jeppesen, D.K.; Laurberg, J.R.; Boysen, A.T.; Bramsen, J.B.; Primdal-Bengtson, B.; Hendrix, A.; Lamy, P.; Dagnaes-Hansen, F.; Rasmussen, M.H.; et al. Cellular Disposal of miR23b by RAB27-Dependent Exosome Release Is Linked to Acquisition of Metastatic Properties. Cancer Res. 2014, 74, 5758–5771. [Google Scholar] [CrossRef] [PubMed]
- Aldousari, S.; Kassouf, W. Update on the management of non-muscle invasive bladder cancer. Can. Urol. Assoc. J. 2013, 4, 56–64. [Google Scholar] [CrossRef]
- NIH. PubMed Health. A Service of the National Library of Medicine, Bladder Cancer Treatment (PDQ®). 23 September 2016. Available online: http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0032608/ (accessed on 22 December 2020).
- Cheung, G.; Sahai, A.; Billia, M.; Dasgupta, P.; Khan, M.S. Recent advances in the diagnosis and treatment of bladder cancer. BMC Med. 2013, 11, 13. [Google Scholar] [CrossRef]
- Dai, J.; Su, Y.; Zhong, S.; Cong, L.; Liu, B.; Yang, J.; Tao, Y.; He, Z.; Chen, C.; Jiang, Y. Exosomes: Key players in cancer and potential therapeutic strategy. Signal Transduct. Target. Ther. 2020, 5, 1–10. [Google Scholar] [CrossRef]
- Vakhshiteh, F.; Atyabi, F.; Ostad, S.N. Mesenchymal stem cell exosomes: A two-edged sword in cancer therapy. Int. J. Nanomed. 2019, 14, 2847–2859. [Google Scholar] [CrossRef] [PubMed]
- Barile, L.; Vassalli, G. Exosomes: Therapy delivery tools and biomarkers of diseases. Pharmacol. Ther. 2017, 174, 63–78. [Google Scholar] [CrossRef] [PubMed]
- Yong, T.; Zhang, X.; Bie, N.; Zhang, H.; Zhang, X.; Li, F.; Hakeem, A.; Hu, J.; Gan, L.; Santos, H.A.; et al. Tumor exosome-based nanoparticles are efficient drug carriers for chemotherapy. Nat. Commun. 2019, 10, 1–16. [Google Scholar] [CrossRef]
- Cai, X.; Qu, L.; Yang, J.; Xu, J.; Sun, L.; Wei, X.; Qu, X.; Bai, T.; Guo, Z.; Zhu, Y. Exosome–transmitted microRNA-133b inhibited bladder cancer proliferation by upregulating dual-specificity protein phosphatase 1. Cancer Med. 2020, 9, 6009–6019. [Google Scholar] [CrossRef]
- Gil-Araujo, B.; Lobo, M.-V.T.; Gutiérrez-Salmerón, M.; Gutiérrez-Pitalúa, J.; Ropero, S.; Angulo, J.C.; Chiloeches, A.; Lasa, M. Dual specificity phosphatase 1 expression inversely correlates with NF-κB activity and expression in prostate cancer and promotes apoptosis through a p38 MAPK dependent mechanism. Mol. Oncol. 2014, 8, 27–38. [Google Scholar] [CrossRef]
- Abraham, S.; Clark, A. Dual-specificity phosphatase 1: A critical regulator of innate immune responses. Biochem. Soc. Trans. 2006, 34, 1018–1023. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, J.-Y.; Kho, D.; Reiners, J.J., Jr.; Wu, G.S. Role for DUSP1 (dual-specificity protein phosphatase 1) in the regulation of autophagy. Autophagy 2016, 12, 1791–1803. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Huyan, T.; Cai, S.; Huang, Q.; Zhang, M.; Peng, H.; Zhang, Y.; Liu, N.; Zhang, W. The role of exosomal miR-375-3p: A potential suppressor in bladder cancer via the Wnt/β-catenin pathway. FASEB J. 2020, 34, 12177–12196. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.-D.; Wu, X.-H.; Fan, Y.-R.; Tan, B.; Quan, Z.; Luo, C.-L. Exosome-derived microRNA-29c Induces Apoptosis of BIU-87 Cells by Down Regulating BCL-2 and MCL-1. Asian Pac. J. Cancer Prev. 2014, 15, 3471–3476. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, X. miRDB: An online database for prediction of functional microRNA targets. Nucleic Acids Res. 2020, 48, D127–D131. [Google Scholar] [CrossRef] [PubMed]
- Kozomara, A.; Birgaoanu, M.; Griffiths-Jones, S. miRBase: From microRNA sequences to function. Nucleic Acids Res. 2019, 47, D155–D162. [Google Scholar] [CrossRef] [PubMed]
- Wishart, D.S.; Bartok, B.; Oler, E.; Liang, K.Y.H.; Budinski, Z.; Berjanskii, M.; Guo, A.; Cao, X.; Wilson, M. MarkerDB: An online database of molecular biomarkers. Nucleic Acids Res. 2021, 49, D1259–D1267. [Google Scholar] [CrossRef]
- Gao, Y.; Wang, P.; Wang, Y.; Ma, X.; Zhi, H.; Zhou, D.; Li, X.; Fang, Y.; Shen, W.; Xu, Y.; et al. Lnc2Cancer v2.0: Updated database of experimentally supported long non-coding RNAs in human cancers. Nucleic Acids Res. 2019, 47, D1028–D1033. [Google Scholar] [CrossRef]
- Batista, I.A.; Melo, S.A. Exosomes and the Future of Immunotherapy in Pancreatic Cancer. Int. J. Mol. Sci. 2019, 20, 567. [Google Scholar] [CrossRef] [PubMed]
| Protein Extracellular Vesicle Biomarkers in Bladder Cancer | |||
|---|---|---|---|
| Proteins | Effect | Mechanism of Action | Ref. |
| Promoters of Oncogenesis | |||
| CD36 a | ↑ migration, proliferation and angiogenesis | ↑ Fatty acid uptake | [37] |
| 5T4 * | ↑ EMT and migration | ↑ CXCL12/CXCR4 chemotaxis ↓ Normal WNT/β-catenin pathway | [37] |
| CD44 * | ↑ proliferation, migration and angiogenesis | Adhesion molecule Docking of proteases on cell membrane | [37] |
| CD73 (NT5E) a | ↑ angiogenesis, metastases and invasion | Regulates cellular signaling with extracellular matrix components such as fibronectin and laminin | [37] |
| CD147 (Basigin) a | ↑ proliferation | Regulates glycolytic metabolic pathways | [37] |
| Alpha 1-antitrypsin a,* | ↓ apoptosis | Immunity regulation | [41] |
| MAGEB4 a,* | ↑ tumorigenesis and proliferation | ↑ ubiquitination and degradation of various tumor suppressors (p53) | [42] |
| NMP-22 | ↑ proliferation | Part of nuclear mitotic apparatus | [42] |
| FOLR1 a | ↑ proliferation | ↑ folic uptake in tumor cells ↑ nucleic acid synthesis ↑ STAT3 activation | [60] |
| Suppressors of Oncogenesis | |||
| TACSTD2 a,** | ↑ apoptosis | Participates in TAp63-dependent apoptosis | [38,39,40] |
| H2B1K a,* | ↓ tumorigenesis | Regulates response to DNA damage ↑ Transcriptional expression of tumor suppressor genes | [41] |
| TTP1 a | ↑ apoptosis | mitochondrial toxin | [60] |
| miRNA Extracellular Vesicle Biomarkers in Bladder Cancer | |||
|---|---|---|---|
| miRNAs | Effect | Mechanism of Action | Ref. |
| Promoters of Oncogenesis | |||
| miR-15a-5p a | ↑ proliferation | post transcriptional regulation of MYB oncogene | [45] |
| miR-31-5p a | ↑ migration and invasion | MAGI2-AS3/miR-31-5p/TNS1 axis | [45] |
| miR-21 a | ↑ invasion | ↓ AKT and MAPK pathways | [45,46] |
| miR-155-5p a | ↓ apoptosis | ↓ TP53INP1 expression | [45,56] |
| miR-940 a | ↑ proliferation, migration and invasion | ↑ expression of INPP4A, GSK3b, c-MYC, cyclin D, β-catenin ↓ p-27 expression | [46] |
| miR-191 a | ↓ apoptosis | ↓ circ-FOXO-3 expression | [46] |
| miR-93 a | ↑ drug resistance | ↓ cisplatin-induced apoptosis and regulates LASS2 | [46] |
| miR-66-3b b | ↑ proliferation | ↓ TUSC2, p53 and p21 | [47] |
| miR-200a-3p a | ↑ invasion | ↑ MMP-2 expression through Dicer/miR-16/JNK2/MMP-2 axis | [46] |
| miR-146-5p a | ↑ invasion | ↑ ETS2-Mediated mmp2 mRNA transcription | [56] |
| Suppressors of Oncogenesis | |||
| miR-205-5p a,* | ↑ apoptosis ↓ EMT and invasion | regulates the expression of the tumor-suppressor protein PTEN targets the transcriptional repressors of E-cadherin, ZEB1 and ZEB2 | [11,46] |
| miR-132-3p a | ↑ angiogenesis, migration ↓ invasion and EMT | TGFβ1/Smad2 signaling pathway | [45] |
| miR-200c a | ↓ EMT, proliferation and invasion | ↓ ZEB1/2 ↑ E-cadherin ↓ LDHA-induced glycolysis | [46] |
| miR-15a a | ↓ proliferation | Targets the oncogene BCL2 | [46] |
| miR-30a-3p a | ↓ autophagy ↑ chemosensitivity to cisplatin ↓ invasion | ↓ autophagy-related genes (including ATG5, ATG12, and Beclin-1) ↓ MMP-2 and MMP-9 expression | [46] |
| miR-503-5p a | ↓ proliferation | interferes with the Rb/E2F signaling pathway | [46] |
| Mirlet7b a,** | ↑ apoptosis ↓ drug resistance | targets MTDH, CALU and MTDH | [46] |
| miR-138-5p a | ↑ apoptosis | ↓ Bcl-w and Akt1 protein expression | [56] |
| miR-144-5p a | ↓ proliferation | ↓ cell cycle-related genes expression (CCNE1, CCNE2, CDC25A, and PKMYT1) | [56] |
| miR-145-5p a | ↓ proliferation and migration | Targets TAGLN2 | [61] |
| miR-23b | ↓ EMT induces G0/G1 cell cycle arrest and apoptosis | ↓ expression of Zeb1 | [61] |
| miR-133b b | ↑ apoptosis | ↓ Bcl-w and Akt1 protein expression | [69] |
| miR-375-3p b | ↓ proliferation and invasion | ↓ expression of FZD8 and therefore blocks the Wnt/β-catenin pathway | [73] |
| miR-29c b | ↓ proliferation | suppresses the G1/S cell cycle transition inhibits AKT and GSK-3β phosphorylation | [75] |
| RNAs, lncRNAs Extracellular Vesicle Biomarkers in Bladder Cancer | |||
|---|---|---|---|
| RNAs, lncRNAs | Effect | Mechanism of Action | Ref. |
| Promoters of Oncogenesis | |||
| GALNT1 a | maintenance of bladder cancer stem cells and bladder tumorigenesis | Mediates O-linked glycosylation of SHH to promote its activation | [44] |
| ARHGEF3 a,* | ↑ proliferation and invasion | ↑ expression of Cyclin A2, Cyclin D1, and MMP2 | [44] |
| UCA1 a | ↑ proliferation | regulates CREB | [53] |
| MALAT1 a | ↓ apoptosis | antagonizes miR-125b | [53,58] |
| UCA 201 a | ↑ migration and invasion | ↑ the expression levels of ZEB1 and ZEB2 ↓ expression of hsa-miR-145 and its target gene, the actin-binding protein FSCN1 | [53] |
| LINC 0035 a,* | ↑ proliferation, migration and invasion | ↓ miR-466 and LYAR | [53] |
| SNHG16 b | ↑ proliferation, migration and invasion | activation of the Wnt/β-catenin pathway↑ expression of STAT3 by ↓ miR-98 expression↓ expression of Bax, cleaved-caspase-3 and cleaved-caspase-9 | [54] |
| linc-UBC1 b | ↑ proliferation | binds to PRC2 complex and regulates target gene expression | [54] |
| PCAT-1 b,** | ↑ proliferation | ↑ Myc and ↓ BRCA2 | [54,58] |
| H19 b | ↑ proliferation, EMT and metastasis | ↓ expression of E-cadherin↑ expression of ID2 | [57] |
| Suppressors of Oncogenesis | |||
| LASS2 a | ↓ cancer cell invasion and proliferation | ↓ V-ATPase activity, the extracellular hydrogen ion concentration and, in turn, the activation of secreted MMP-2 and MMP-9 | [44] |
| FOXO3 a | ↓ proliferation, migration and invasion | ↑ miR-9-5p expression and thus ↓ TGFBR2 expression | [44] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Georgantzoglou, N.; Pergaris, A.; Masaoutis, C.; Theocharis, S. Extracellular Vesicles as Biomarkers Carriers in Bladder Cancer: Diagnosis, Surveillance, and Treatment. Int. J. Mol. Sci. 2021, 22, 2744. https://doi.org/10.3390/ijms22052744
Georgantzoglou N, Pergaris A, Masaoutis C, Theocharis S. Extracellular Vesicles as Biomarkers Carriers in Bladder Cancer: Diagnosis, Surveillance, and Treatment. International Journal of Molecular Sciences. 2021; 22(5):2744. https://doi.org/10.3390/ijms22052744
Chicago/Turabian StyleGeorgantzoglou, Natalia, Alexandros Pergaris, Christos Masaoutis, and Stamatios Theocharis. 2021. "Extracellular Vesicles as Biomarkers Carriers in Bladder Cancer: Diagnosis, Surveillance, and Treatment" International Journal of Molecular Sciences 22, no. 5: 2744. https://doi.org/10.3390/ijms22052744
APA StyleGeorgantzoglou, N., Pergaris, A., Masaoutis, C., & Theocharis, S. (2021). Extracellular Vesicles as Biomarkers Carriers in Bladder Cancer: Diagnosis, Surveillance, and Treatment. International Journal of Molecular Sciences, 22(5), 2744. https://doi.org/10.3390/ijms22052744








