Biomolecular Basis of Cellular Consciousness via Subcellular Nanobrains
Abstract
1. Cellular Nature of Life
2. Chimeric Nature of the Eukaryotic Cell
3. Structures and Processes Behind Cellular Consciousness—Evolution of Chimeric Consciousness of Eukaryotic Cell
4. Structures and Processes behind Cellular Consciousness—Two Types of Nanobrains Generating Consciousness of Eukaryotic Cell
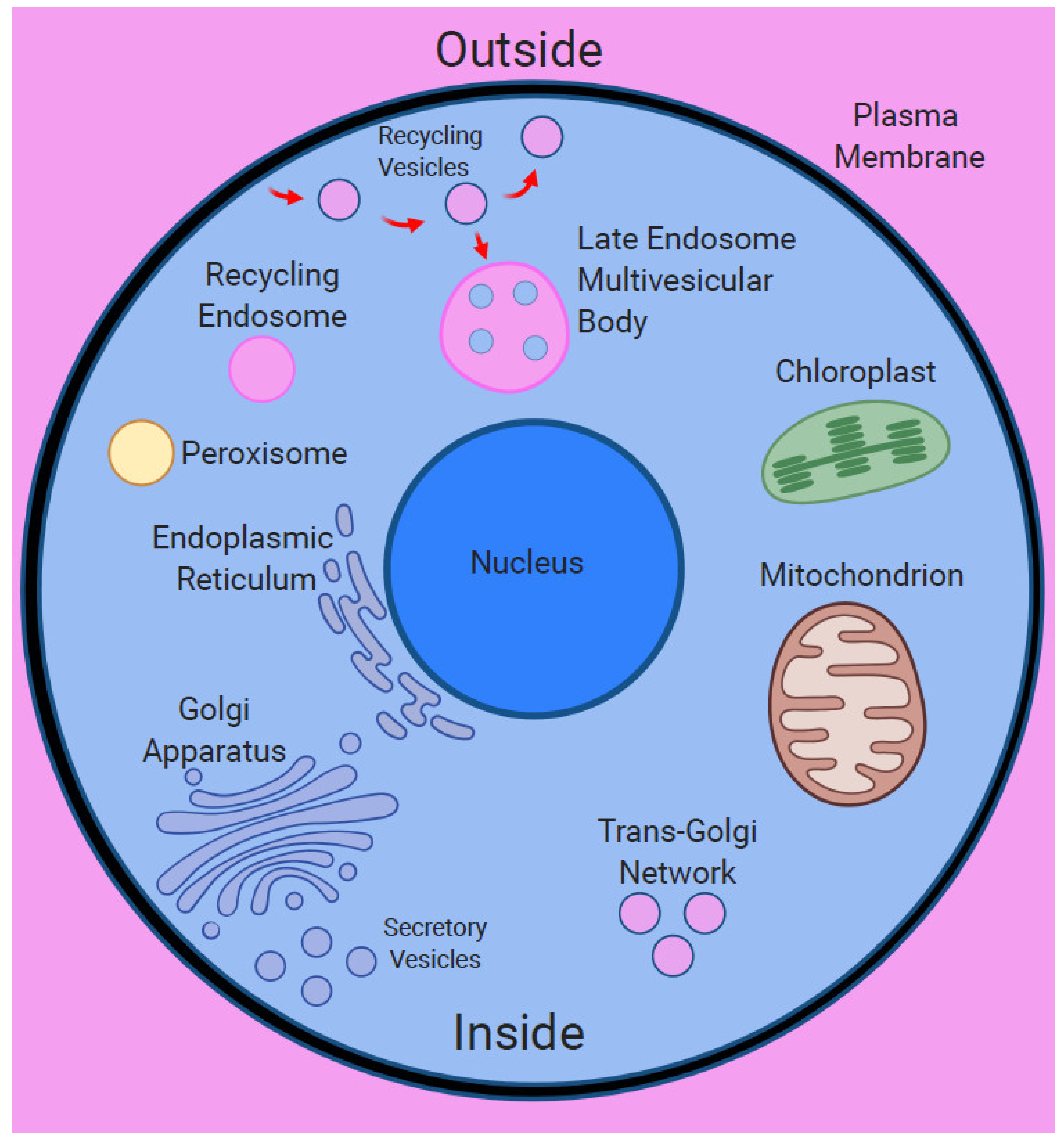
5. Plasma Membrane as Primary Nanobrain
6. Centrosomes and Nuclear Surfaces as Cell Body/Energide Nanobrains
7. Nano-Intentionality and Nano-Mind from Eukaryotic Nanobrains
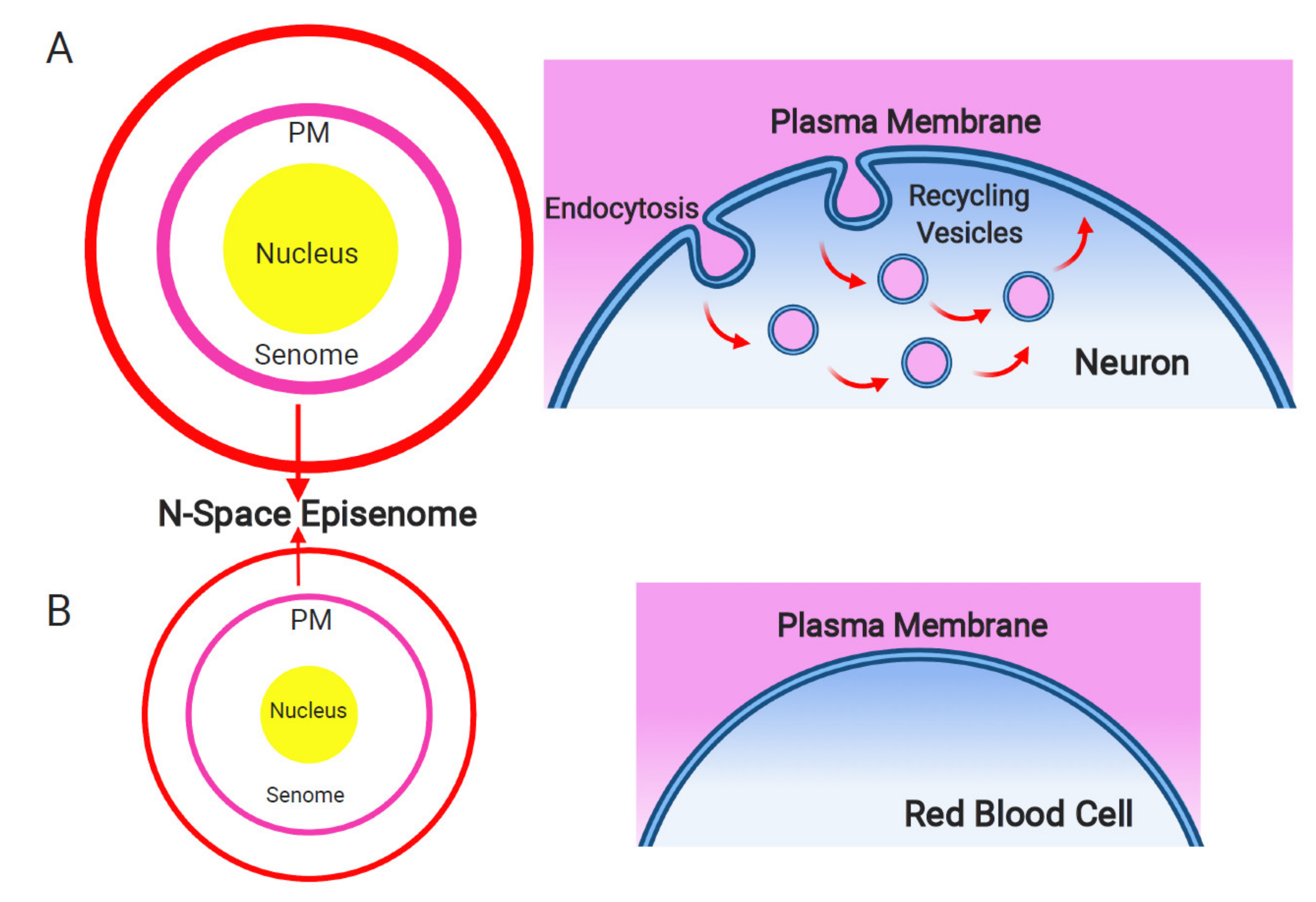
8. The N-Space Episenome as an Informational Matrix for Supra-Cellular Consciousness
9. Supra-Cellular Consciousness (Organismal Experience) Affects Cellular Structures
10. Life Is Electric: Bioelectric and Biomagnetic Nature of Life Processes
11. Membranes and Proteins as Bioelectric Devices—Proteins Dance to Senomic Tunes
12. Cells as Unitary Organisms: From Mechanicism to Organicism
13. Outlook
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nurse, P. What Is Life? Understanding Biology in Five Steps; David Fickling Book: Oxford, UK, 2020. [Google Scholar]
- Mazzarello, P. A unifying concept: The history of cell theory. Nat. Cell Biol. 1999, 1, E13–E15. [Google Scholar] [CrossRef]
- Richmond, M.L. Thomas Henry Huxley‘s developmental view of the cell. Nat. Rev. Mol. Cell Biol. 2002, 3, 61–65. [Google Scholar] [CrossRef]
- Nicholson, D.J. Biological atomism and cell theory. Stud. Hist. Phil. Biol. Biomed. Sci. 2010, 41, 202–211. [Google Scholar] [CrossRef]
- Lyons, S. From Cells to Organisms: Re-Envisioning Cell Theory; University of Toronto Press: Toronto, ON, Canada, 2020. [Google Scholar]
- Baluška, F.; Volkmann, D.; Barlow, P.W. Cell-cell channels and their implications for Cell Theory. In Cell-Cell Channels; Baluška, F., Volkmann, F., Barlow, D., Peter, W., Eds.; Springer: New York, NY, USA, 2006; pp. 1–18. [Google Scholar]
- Baluška, F. Cell-cell channels, viruses, and evolution: Via infection, parasitism, and symbiosis toward higher levels of biological complexity. Ann. N Y Acad. Sci. 2009, 1178, 106–119. [Google Scholar]
- Margulis, L. Serial endosymbiotic theory (SET) and composite individuality. Transition from bacterial to eukaryotic genomes. Microbiol. Today 2004, 31, 172–174. [Google Scholar]
- Margulis, L.; Dolan, M.F.; Guerrero, R. The chimeric eukaryote: Origin of the nucleus from karyomastigont in amitochondriate protist. Proc. Natl. Acad. Sci. USA 2000, 97, 6954–6999. [Google Scholar] [CrossRef] [PubMed]
- Baluška, F.; Volkmann, D.; Barlow, P.W. Cell bodies in a cage. Nature 2004, 428, 371. [Google Scholar] [CrossRef] [PubMed]
- Baluška, F.; Volkmann, D.; Barlow, P.W. Eukaryotic cells and their Cell Bodies: Cell Theory revisited. Ann. Bot. 2004, 94, 9–32. [Google Scholar] [CrossRef] [PubMed]
- Baluška, F.; Volkmann, D.; Menzel, D.; Barlow, P.W. Strasburger’s legacy to mitosis and cytokinesis and its relevance for the Cell Theory. Protoplasma 2012, 249, 1151–1162. [Google Scholar] [CrossRef]
- Shepherd, V.A.; Beilby, M.J.; Bisson, M.A. When is a cell not a cell? A theory relating coenocytic structure to the unusual electrophysiology of Ventricaria ventricosa (Valonia ventricosa). Protoplasma 2004, 223, 79–91. [Google Scholar] [CrossRef]
- Margulis, L. The conscious cell. Ann. N Y Acad. Sci. 2001, 929, 55–70. [Google Scholar] [CrossRef]
- Baluška, F.; Lyons, S. Energide-cell body as smallest unit of eukaryotic life. Ann. Bot. 2018, 122, 741–745. [Google Scholar] [CrossRef]
- Baluška, F.; Lyons, S. Symbiotic origin of eukaryotic nucleus—From cell body to Neo-Energide. In Concepts in Cell Biology—History and Evolution; Sahi, V., Baluška, F., Eds.; Plant Cell Monographs 23; Springer International Publishing: Cham, Switzerland, 2018; pp. 39–66. [Google Scholar]
- Baluška, F.; Lyons, S. Archaeal origins of eukaryotic cell and nucleus. Biosystems 2021, 203, 104375. [Google Scholar] [CrossRef]
- Wilson, E.B. The Cell in Development and Inheritance; MacMillan: New York, NY, USA, 1892. [Google Scholar]
- Wolpert, L. The evolution of ‘the cell theory’. Curr. Biol. 1996, 6, 225–228. [Google Scholar] [CrossRef]
- Harris, H. The Birth of the Cell; Yale University Press: New Haven, CT, USA, 1999. [Google Scholar]
- Reber, A.S. The First Minds: Caterpillars, Karyotes, and Consciousness; Oxford University Press: Oxford, UK, 2019. [Google Scholar]
- Baluška, F.; Reber, A. Sentience and consciousness in single cells: How the first minds emerged in unicellular species. BioEssays 2019, 41, e1800229. [Google Scholar] [CrossRef]
- Reber, A.S.; Baluška, F. Cognition is some surprising places. Biochem. Biophys. Res. Commun. 2021, in press. [Google Scholar] [CrossRef]
- Fitch, W.T. Nano-intentionality: A defense of intrinsic intentionality. Biol. Philos. 2008, 23, 157–177. [Google Scholar] [CrossRef]
- Shapiro, J.A. Evolution: A View from the 21st Century; FT Press Science: Saddle River, NJ, USA, 2011. [Google Scholar]
- Baluška, F.; Yokawa, K. Anaesthetics and plants: From sensory systems to cognition-based adaptive behaviour. Protoplasma 2021, in press. [Google Scholar]
- Sarai, C.; Tanifuji, G.; Nakayama, T.; Kamikawa, R.; Takahashi, K.; Yazaki, E.; Matsuo, E.; Miyashita, H.; Ishida, K.I.; Iwataki, M.; et al. Dinoflagellates with relic endosymbiont nuclei as models for elucidating organellogenesis. Proc. Natl. Acad. Sci. USA 2020, 117, 5364–5375. [Google Scholar] [CrossRef]
- Nakayama, T.; Takahashi, K.; Kamikawa, R.; Iwataki, M.; Inagaki, Y.; Tanifuji, G. Putative genome features of relic green alga-derived nuclei in dinoflagellates and future perspectives as model organisms. Commun. Integr. Biol. 2020, 13, 84–88. [Google Scholar] [CrossRef]
- Sibbald, S.J.; Archibald, J.M. Genomic insights into plastid evolution. Genome Biol. Evol. 2020, 12, 978–990. [Google Scholar] [CrossRef]
- De Duve, C. The origin of eukaryotes: A reappraisal. Nat. Rev. Genet. 2007, 8, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Zachar, I.; Boza, G. Endosymbiosis before eukaryotes: Mitochondrial establishment in protoeukaryotes. Cell. Mol. Life Sci. 2020, 77, 3503–3523. [Google Scholar] [CrossRef]
- Patron, N.J.; Waller, R.F.; Keeling, P.J. A tertiary plastid uses genes from two endosymbionts. J. Mol. Biol. 2006, 357, 1373–1382. [Google Scholar] [CrossRef]
- Hehenberger, E.; Gast, R.J.; Keeling, P.J. A kleptoplastidic dinoflagellate and the tipping point between transient and fully integrated plastid endosymbiosis. Proc. Natl. Acad. Sci. USA 2019, 116, 17934–17942. [Google Scholar] [CrossRef]
- Jacob, F. Evolution and tinkering. Science 1977, 196, 1161–1166. [Google Scholar] [CrossRef]
- Agnati, L.F.; Baluška, F.; Barlow, P.W.; Guidolin, D. Mosaic, self-similarity logic, and biological attraction principles: Three explanatory instruments in biology. Commun. Integr. Biol. 2009, 2, 552–563. [Google Scholar] [CrossRef] [PubMed]
- Agnati, L.F.; Barlow, P.W.; Baldelli, E.; Baluška, F. Are maternal mitochondria the selfish entities that are masters of the cells of eukaryotic multicellular organisms? Commun. Integr. Biol. 2009, 2, 194–200. [Google Scholar] [CrossRef]
- Baluška, F.; Miller, W.B., Jr. Senomic view of the cell: Senome versus Genome. Commun. Integr. Biol. 2018, 11, 1–9. [Google Scholar] [CrossRef]
- Miller, W.B., Jr.; Torday, J.S.; Baluška, F. The N-space Episenome unifies cellular information space-time within cognition-based evolution. Prog. Biophys. Mol. Biol. 2020, 150, 112–139. [Google Scholar] [CrossRef]
- Miller, W.B., Jr.; Baluška, F.; Torday, J.S. Cellular senomic measurements in cognition-based evolution. Prog. Biophys. Mol. Biol. 2020, 156, 20–33. [Google Scholar] [CrossRef]
- Tuszynski, J.A.; Portet, S.; Dixon, J.M.; Luxford, C.; Cantielo, H.F. Ionic wave propagation along actin filaments. Biophys. J. 2004, 86, 1890–1903. [Google Scholar] [CrossRef]
- Iglesias, P.A.; Devreotes, P.N. Biased excitable networks: How cells direct motion in response to gradients. Curr. Opin. Cell Biol. 2012, 24, 245–253. [Google Scholar] [CrossRef]
- Pal, D.S.; Li, X.; Banerjee, T.; Niao, Y.; Devreotes, P.N. The excitable signal transduction networks: Movers and shapers of eukaryotic cell migration. Int. J. Dev. Biol. 2019, 63, 407–416. [Google Scholar] [CrossRef]
- Beta, C.; Gov, N.S.; Yochelis, A. Why a large-scale mode can be essential for understanding intracellular actin waves. Cells 2020, 9, 1533. [Google Scholar] [CrossRef]
- Cifra, M.; Pokorný, J.; Havelka, D.; Kucera, O. Electric field generated by axial longitudinal vibration modes of microtubule. Biosystems 2010, 100, 122–131. [Google Scholar] [CrossRef]
- Havelka, D.; Cifra, M.; Kučera, O.; Pokorný, J.; Vrba, J. High-frequency electric field and radiation characteristics of cellular microtubule network. J. Theor. Biol. 2011, 286, 31–40. [Google Scholar] [CrossRef]
- Kučera, O.; Havelka, D. Mechano-electrical vibrations of microtubules—Link to subcellular morphology. Biosystems 2012, 109, 346–355. [Google Scholar] [CrossRef]
- Kalra, A.P.; Eakins, B.B.; Patel, S.D.; Ciniero, G.; Rezania, V.; Shankar, K.; Tuszynski, J.A. All wired up: An exploration of the electrical properties of microtubules and tubulin. ACS Nano 2020. [Google Scholar] [CrossRef]
- Baluška, F.; Reber, A. Senomic and ephaptic principles of cellular consciousness: The biomolecular basis for plant and animal sentience. J. Consc. Stud. 2021, 28, 31–49. [Google Scholar]
- Tuszynski, J.A. The bioelectric circuitry of the cell. In Brain and Human Body Modeling: Computational Human Modeling at EMBC 2018; Makarov, S., Horner, M., Noetscher, G., Eds.; Springer International Publishing: Basel, Switzerland, 2018; pp. 195–208. [Google Scholar]
- Tuszynski, J.A.; Friesen, D.; Freedman, H.; Sbitnev, V.I.; Kim, H.; Santelices, I.; Kalra, A.P.; Patel, S.D.; Shankar, K.; Chua, L.O. Microtubules as sub-cellular memristors. Sci. Rep. 2020, 10, 2108. [Google Scholar] [CrossRef]
- Baluška, F.; Volkmann, D.; Barlow, P.W. Nuclear components with microtubule-organizing properties in multicellular eukaryotes: Functional and evolutionary considerations. Int. Rev. Cytol. 1997, 175, 91–135. [Google Scholar]
- Oster, G.; Wang, H. Rotary protein motors. Trends Cell Biol. 2003, 13, 114–121. [Google Scholar] [CrossRef]
- Loewenstein, W.R. Physics in Mind: A Quantum View of the Brain; Basic Books: New York, NY, USA, 2013. [Google Scholar]
- Binder, P.M.; Danchin, A. Life’s demons: Information and order in biology: What subcellular machines gather and process the information necessary to sustain life? EMBO Rep. 2011, 12, 495–499. [Google Scholar] [CrossRef] [PubMed]
- Boël, G.; Danot, O.; de Lorenzo, V.; Danchin, A. Omnipresent Maxwell’s demons orchestrate information management in living cells. Micr. Biotechnol. 2019, 12, 210–242. [Google Scholar] [CrossRef]
- Hoffmeyer, J. Surfaces inside surfaces—On the origin of agency and life. Cybernet. Hum. Know. 1998, 5, 33–42. [Google Scholar]
- Baluška, F.; Mancuso, S.; Volkmann, D.; Barlow, P.W. The ‘root-brain’ hypothesis of Charles and Francis Darwin: Revival after more than 125 years. Plant Signal. Behav. 2009, 4, 1121–1127. [Google Scholar] [CrossRef]
- Baluška, F.; Mancuso, S. Root apex transition zone as oscillatory zone. Front. Plant Sci. 2013, 4, 354. [Google Scholar] [CrossRef]
- Lintilhac, P. Toward a theory of cellularity—Speculations on the nature of the living cell. Bioscience 1999, 49, 59–68. [Google Scholar] [CrossRef]
- Tan, T.H.; Liu, J.; Miller, P.W.; Tekant, M.; Dunkel, J.; Fakhri, N. Topological turbulence in the membrane of a living cell. Nat. Phys. 2020, 16, 657–662. [Google Scholar] [CrossRef]
- Matzke, A.J.M.; Lin, W.D.; Matzke, M. Evidence that ion-based signaling initiating at the cell surface can potentially influence chromatin dynamics and chromatin-bound proteins in the nucleus. Front. Plant Sci. 2019, 10, 1267. [Google Scholar] [CrossRef] [PubMed]
- Mazia, D. The chromosome cycle and the centrosome cycle in the mitotic cycle. Int. Rev. Cytol. 1987, 100, 49–92. [Google Scholar]
- Alvarado-Kristensson, M. Choreography of the centrosome. Heliyon 2020, 6, e03238. [Google Scholar] [CrossRef]
- Nygren, J.; Adelman, R.A.; Myakishev-Rempel, M.; Sun, G.; Li, J.; Zhao, Y. Centrosome as a micro-electronic generator in live cell. Biosystems 2020, 197, 104210. [Google Scholar] [CrossRef]
- Albrecht-Buehler, G. Rudimentary form of cellular ‘vision’. Proc. Natl. Acad. Sci. USA 1992, 89, 8288–8292. [Google Scholar] [CrossRef]
- Albrecht-Buehler, G. Cellular infrared detector appears to be contained in the centrosome. Cell Motil. Cytoskel. 1994, 27, 262–271. [Google Scholar] [CrossRef] [PubMed]
- Albrecht-Buehler, G. A long-range attraction between aggregating 3T3 cells mediated by near-infrared light scattering. Proc. Natl. Acad. Sci. USA 2005, 102, 5050–5055. [Google Scholar] [CrossRef]
- Kokkoris, V.; Stefani, F.; Dalpé, Y.; Dettman, J.; Corradi, N. Nuclear dynamics in the arbuscular mycorrhizal fungi. Trends Plant Sci. 2020, 25, 765–778. [Google Scholar] [CrossRef]
- Kuhn, G.; Hijri, M.; Sanders, I.R. Evidence for the evolution of multiple genomes in arbuscular mycorrhizal fungi. Nature 2001, 414, 745–748. [Google Scholar] [CrossRef]
- Young, J.P. Genome diversity in arbuscular mycorrhizal fungi. Curr. Opin. Plant Biol. 2015, 26, 113–119. [Google Scholar] [CrossRef]
- Marleau, J.; Dalpé, Y.; St-Arnaud, M.; Hijri, M. Spore development and nuclear inheritance in arbuscular mycorrhizal fungi. BMC Evol. Biol. 2011, 11, 51. [Google Scholar] [CrossRef]
- Ingber, D.E. Cellular tensegrity: Defining new rules of biological design that govern the cytoskeleton. J. Cell Sci. 1993, 104, 613–627. [Google Scholar]
- Bordoni, B.; Varacallo, M.A.; Morabito, B.; Simonelli, M. Biotensegrity or fascintegrity? Cureus 2019, 11, e4819. [Google Scholar] [CrossRef]
- Scarr, G. Biotensegrity: What is the big deal? J. Bodywork Moc. Ther. 2020, 24, 134–137. [Google Scholar] [CrossRef] [PubMed]
- Nuccitelli, R. A role for endogenous electric fields in wound healing. Curr. Top. Dev. Biol. 2003, 58, 1–26. [Google Scholar] [PubMed]
- Cifra, M.; Fields, J.Z.; Farhadi, A. Electromagnetic cellular interactions. Prog. Biophys. Mol. Biol. 2011, 105, 223–246. [Google Scholar] [CrossRef] [PubMed]
- Fels, D. The double-aspect of life. Biology 2018, 7, 28. [Google Scholar] [CrossRef] [PubMed]
- Burdick, R.K.; Villabona-Monsalve, J.P.; Mashour, G.A.; Goodson, T., 3rd. Modern anesthetic ethers demonstrate quantum interactions with entangled photons. Sci. Rep. 2019, 9, 11351. [Google Scholar] [CrossRef]
- Brugger, M.S.; Baumgartner, K.; Mauritz, S.C.F.; Gerlach, S.C.; Röder, F.; Schlosser, C.; Fluhrer, R.; Wixforth, A.; Westerhausen, C. Vibration enhanced cell growth induced by surface acoustic waves as in vitro wound-healing model. Proc. Natl. Acad. Sci. USA 2020, 117, 31603–31613. [Google Scholar] [CrossRef] [PubMed]
- Levin, M. The computational boundary of a “self”: Developmental bioelectricity drives multicellularity and scale-free cognition. Front. Psychol. 2019, 10, 2688. [Google Scholar] [CrossRef]
- Fields, C.; Levin, M. Scale-free biology: Integrating evolutionary and developmental thinking. BioEssays 2020, 42, 1900228. [Google Scholar] [CrossRef]
- Albrecht-Buehler, G. Is cytoplasm intelligent too? Cell Muscle Motil. 1985, 6, 1–21. [Google Scholar] [PubMed]
- Baluška, F.; Mancuso, S. Deep evolutionary origins of neurobiology: Turning the essence of ‘neural’ upside-down. Commun. Integr. Biol. 2009, 2, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Baluška, F.; Levin, M. On having no head: Cognition throughout biological systems. Front. Psychol. 2016, 7, 902. [Google Scholar] [CrossRef]
- Cooke, J.E. The living mirror theory of consciousness. J. Consc. Stud. 2020, 27, 127–147. [Google Scholar]
- Miller, W.B., Jr.; Torday, J.S.; Baluška, F. Biological evolution as defense of ‘self’. Prog. Biophys. Mol. Biol. 2019, 142, 54–74. [Google Scholar] [CrossRef]
- Ford, B.J. Cellular intelligence: Microphenomenology and the realities of being. Prog. Biophys. Mol. Biol. 2017, 131, 273–287. [Google Scholar] [CrossRef] [PubMed]
- Dexter, J.P.; Prabakaran, S.; Gunawardena, J. A complex hierarchy of avoidance behaviors in a single-cell eukaryote. Curr. Biol. 2019, 29, 4323–4329. [Google Scholar] [CrossRef] [PubMed]
- Marshall, W.F. Cellular cognition: Sequential logic in a giant protist. Curr. Biol. 2019, 29, R1303–R1305. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.B., Jr. Biological information systems: Evolution as cognition-based information management. Progr. Biophys Mol. Biol. 2018, 134, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Ford, B.J. Are cells ingenious? Microscope 2004, 52, 135–144. [Google Scholar]
- Ford, B.J. On intelligence in cells: The case for whole cell biology. Int. Sci. Rev. 2009, 34, 350–365. [Google Scholar] [CrossRef]
- Davis, M.M.; Krogsgaard, M.; Huse, M.; Huppa, J.; Lillemeier, B.F.; Li, Q.J. T cells as a self-referential, sensory organ. Annu. Rev. Immunol. 2007, 25, 681–695. [Google Scholar] [CrossRef] [PubMed]
- Baluška, F.; Mancuso, S. Plants and animals: Convergent evolution in action? In Plant–Environment Interactions: From Sensory Plant Biology to Active Plant Behaviour; Baluška, F., Ed.; Springer: Berlin, Germany, 2009; pp. 285–301. [Google Scholar]
- Trewavas, A.J.; Baluška, F. The ubiquity of consciousness: The ubiquity of consciousness, cognition and intelligence in life. EMBO Rep. 2011, 12, 1221–1225. [Google Scholar] [CrossRef] [PubMed]
- Trewavas, A. Plant Behaviour and Intelligence; Oxford University Press: Oxford, UK, 2014. [Google Scholar]
- Dodig-Crnkovic, G. Modeling life as cognitive info-computation. In Conference on Computability in Europe; Springer: Cham, Germany, 2014; pp. 153–162. [Google Scholar]
- Marshall, W.F. How cells measure length on subcellular scales. Trends Cell Biol. 2015, 25, 760–768. [Google Scholar] [CrossRef]
- Lyon, P. The cognitive cell: Bacterial behavior reconsidered. Front. Microbiol. 2015, 6, 264. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.B. Cognition, information fields and hologenomic entanglement: Evolution in light and shadow. Biology 2016, 5, 21. [Google Scholar] [CrossRef]
- Torday, J.S.; Miller, W.B., Jr. The resolution of ambiguity as the basis for life: A cellular bridge between Western reductionism and Eastern holism. Progr. Biophys. Mol. Biol. 2017, 131, 288–297. [Google Scholar] [CrossRef]
- Miller, W.B., Jr.; Torday, J.S. Four domains: The fundamental unicell and Post-Darwinian cognition-based evolution. Progr. Biophys. Mol. Biol. 2018, 140, 49–73. [Google Scholar] [CrossRef]
- Cárdenas-García, J.F. The process of info-autopoiesis—The source of all information. Biosemiotics 2020, 13, 199–221. [Google Scholar] [CrossRef]
- Lloyd, S. Computational capacity of the universe. Phys. Rev. Lett. 2002, 88, 237901. [Google Scholar] [CrossRef]
- Walker, S.I.; Kim, H.; Davies, P.C. The informational architecture of the cell. Phil. Trans. R. Soc. A 2016, 374, 20150057. [Google Scholar] [CrossRef]
- Kaech, S.; Brinkhaus, H.; Matus, A. Volatile anesthetics block actin-based motility in dendritic spines. Proc. Natl. Acad. Sci. USA 1999, 96, 10433–10437. [Google Scholar] [CrossRef] [PubMed]
- Matus, A.; Brinkhaus, H.; Wagner, U. Actin dynamics in dendritic spines: A form of regulated plasticity at excitatory synapses. Hippocampus 2000, 10, 555–560. [Google Scholar] [CrossRef]
- Platholi, J.; Herold, K.F.; Hemmings, H.C., Jr.; Halpain, S. Isoflurane reversibly destabilizes hippocampal dendritic spines by an actin-dependent mechanism. PLoS ONE 2014, 9, e102978. [Google Scholar] [CrossRef]
- Kätzel, D.; Miesenböck, G. Experience-dependent rewiring of specific inhibitory connections in adult neocortex. PLoS Biol. 2014, 12, e1001798. [Google Scholar] [CrossRef] [PubMed]
- Weaver, J. How brain circuits adapt to changes in sensory experience. PLoS Biol. 2014, 12, e1001802. [Google Scholar] [CrossRef]
- Yamada, T.; Yang, Y.; Valnegri, P.; Juric, I.; Abnousi, A.; Markwalter, K.H.; Guthrie, A.N.; Godec, A.; Oldenborg, A.; Hu, M.; et al. Sensory experience remodels genome architecture in neural circuit to drive motor learning. Nature 2019, 569, 708–713. [Google Scholar] [CrossRef]
- Tonn Eisinger, K.R.; West, A.E. Transcribing memories in genome architecture. Trends Neurosci. 2019, 42, 565–566. [Google Scholar] [CrossRef]
- Eliezer, Y.; Deshe, N.; Hoch, L.; Iwanir, S.; Pritz, C.O.; Zaslaver, A. A memory circuit for coping with impending adversity. Curr. Biol. 2019, 29, 1573–1583. [Google Scholar] [CrossRef]
- Katz, M.; Shaham, S. Learning and memory: Mind over matter in C. elegans. Curr. Biol. 2019, 29, R365–R367. [Google Scholar] [CrossRef]
- Hayek, F.A. The Sensory Order; The University of Chicago Press: Chicago, IL, USA, 1952. [Google Scholar]
- Torday, J.S.; Baluška, F. Why control an experiment? From empiricism, via consciousness, toward Implicate Order. EMBO Rep. 2019, 20, e49110. [Google Scholar] [CrossRef]
- Allchin, D. To err and win a Nobel Prize: Paul Boyer, ATP Synthase and the emergence of bioenergetics. J. Hist. Biol. 2002, 35, 149–172. [Google Scholar] [CrossRef] [PubMed]
- Grote, M. Surfaces of action: Cells and membranes in electrochemistry and the life sciences. Stud. Hist. Phil. Biol. Biomed. Sci. 2010, 41, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Hohmann-Marriott, M.F.; Blankenship, R.E. Evolution of photosynthesis. Annu. Rev. Plant Biol. 2011, 62, 515–548. [Google Scholar] [CrossRef] [PubMed]
- Nelson, N.; Junge, W. Structure and energy transfer in photosystems of oxygenic photosynthesis. Annu. Rev. Biochem. 2015, 84, 659–683. [Google Scholar] [CrossRef] [PubMed]
- Boyer, P.D. The ATP synthase—A splendid molecular machine. Annu. Rev. Biochem. 1997, 66, 717–749. [Google Scholar] [CrossRef] [PubMed]
- Morelli, A.M.; Ravera, S.; Panfoli, I. The aerobic mitochondrial ATP synthesis from a comprehensive point of view. Open Biol. 2020, 10, 200224. [Google Scholar] [CrossRef] [PubMed]
- Nelson, N.; Junge, W. ATP synthase. Annu. Rev. Biochem. 2015, 84, 631–657. [Google Scholar]
- Kováč, L. Life, chemistry and cognition. EMBO Rep. 2006, 7, 562. [Google Scholar] [CrossRef]
- Kováč, L. Information and knowledge in biology—Time for reappraisal. Plant Signal. Behav. 2007, 2, 65. [Google Scholar] [CrossRef]
- Kováč, L. Bioenergetics: A key to brain and mind. Commun. Integr. Biol. 2008, 1, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, P. Coupling of phosphorylation to electron and hydrogen transfer by a chemi-osmotic type of mechanism. Nature 1961, 191, 144–148. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, P.; Moyle, J. Chemiosmotic hypothesis of oxidative phosphorylation. Nature 1967, 213, 137–139. [Google Scholar] [CrossRef]
- Mitchell, P. Vectorial chemiosmotic processes. Annu. Rev. Biochem. 1977, 46, 996–1005. [Google Scholar] [CrossRef]
- Morange, M. What history tells us. XI. The complex history of the chemiosmotic theory. J. Biosci. 2007, 32, 1245–1250. [Google Scholar] [CrossRef]
- Kettenmann, H. Alexander von Humboldt and the concept of animal electricity. Trends Neurosci. 1997, 20, 239–242. [Google Scholar] [CrossRef]
- Piccolino, M. Animal electricity and the birth of electrophysiology: The legacy of Luigi Galvani. Brain Res. Bull. 1998, 46, 381–407. [Google Scholar] [CrossRef]
- Tseng, A.S.; Levin, M. Cracking the bioelectric code: Probing endogenous ionic controls of pattern formation. Commun. Integr. Biol. 2013, 6, e22595. [Google Scholar] [CrossRef] [PubMed]
- De Loof, A. The cell’s self-generated “electrome”: The biophysical essence of the immaterial dimension of life? Commun. Integr. Biol. 2016, 9, e1197446. [Google Scholar] [CrossRef]
- De Loof, A. The evolution of “life”: A metadarwinian integrative approach. Commun. Integr. Biol. 2017, 10, e1301335. [Google Scholar] [CrossRef]
- Levin, M.; Pezzulo, G.; Finkelstein, J.M. Endogenous bioelectric signaling networks: Exploiting voltage gradients for control of growth and form. Annu. Rev. Biomed. Eng. 2017, 19, 353–387. [Google Scholar] [CrossRef] [PubMed]
- Medvedeva, V.P.; Pierani, A. How do electric fields coordinate neuronal migration and maturation in the developing cortex? Front. Cell. Dev. Biol. 2020, 8, 580657. [Google Scholar] [CrossRef] [PubMed]
- Needleman, D.; Brugues, J. Determining physical principles of subcellular organization. Dev. Cell 2014, 29, 135–138. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mogilner, A.; Manhart, A. Intracellular fluid mechanics: Coupling cytoplasmic flow with active cytoskeletal gel. Annu. Rev. Fluid Mech. 2018, 50, 347–370. [Google Scholar] [CrossRef]
- Witzany, G.; Baluška, F. Can subcellular organization be explained only by physical principles? Commun. Integr. Biol. 2015, 8, e1009796. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Crick, F. Central dogma of molecular biology. Nature 1970, 227, 561–563. [Google Scholar] [CrossRef] [PubMed]
- Brenner, S. The revolution in the life sciences. Science 2012, 338, 1427–1428. [Google Scholar] [CrossRef] [PubMed]
- Szent-Györgyi, A. Oxidation, Energy Transfer, and Vitamins; Nobel Lecture; The Nobel Foundation: Stockholm, Sweden, 1937. [Google Scholar]
- Szent-Györgyi, A. Energy transmission in biological systems. Science 1956, 125, 751–752. [Google Scholar]
- Steele, R.H.; Szent-Györgyi, A. On excitation of biological substances. Proc. Natl. Acad. Sci. USA 1957, 43, 477–491. [Google Scholar] [CrossRef]
- Bay, Z.; Goodall, M.C.; Szent-Györgyi, A. The transmission of excitation from the membrane to actomyosin. Bull. Math. Biophys. 1956, 15, 1–13. [Google Scholar] [CrossRef]
- Avery, J.; Bay, Z.; Szent-Györgyi, A. On the energy transfer in biological systems. Proc. Natl. Acad. Sci. USA 1961, 47, 1742–1744. [Google Scholar] [CrossRef] [PubMed]
- Szent-Györgyi, A. Bioelectronics. Science 1968, 161, 988–990. [Google Scholar] [CrossRef]
- Szent-Györgyi, A. Bioelectronics. A Study in Cellular Regulations, Defense, and Cancer; Academic Press: Cambridge, MA, USA, 1968. [Google Scholar]
- Szent-Györgyi, A. Bioelectronics and cancer. J. Bioenerg. 1973, 4, 533–562. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H. Reactive oxygen species, oxidative signaling and the regulation of photosynthesis. Environ. Exp. Bot. 2018, 154, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Noctor, G. Redox homeostasis and signaling in a higher-CO2 world. Annu. Rev. Plant Biol. 2020, 71, 157–182. [Google Scholar] [CrossRef] [PubMed]
- Belevich, I.; Verkhovsky, M.I.; Wikström, M. Proton-coupled electron transfer drives the proton pump of cytochrome c oxidase. Nature 2006, 440, 829–832. [Google Scholar] [CrossRef]
- Horemans, N.; Asard, H.; Caubergs, R.J. The role of ascorbate free radical as an electron acceptor to cytochrome b-mediated trans-plasma membrane electron transport in higher plants. Plant Physiol. 1994, 104, 1455–1458. [Google Scholar] [CrossRef][Green Version]
- Picco, C.; Scholz-Starke, J.; Festa, M.; Costa, A.; Sparla, F.; Trost, P.; Carpaneto, A. Direct recording of trans-plasma membrane electron currents mediated by a member of the cytochrome b561 family of soybean. Plant Physiol. 2015, 169, 986–995. [Google Scholar] [CrossRef] [PubMed]
- Ly, J.D.; Lawen, A. Transplasma membrane electron transport: Enzymes involved and biological function. Redox Rep. 2003, 8, 3–21. [Google Scholar] [CrossRef] [PubMed]
- Lane, D.J.; Lawen, A. Ascorbate and plasma membrane electron transport—Enzymes vs efflux. Free Radic. Biol. Med. 2009, 47, 485–495. [Google Scholar] [CrossRef]
- Eccardt, A.M.; Bell, T.P.; Mattathil, L.; Prasad, R.; Kelly, S.C.; Fisher, J.S. Trans-plasma membrane electron transport and ascorbate efflux by skeletal muscle. Antioxidants 2017, 6, 89. [Google Scholar] [CrossRef] [PubMed]
- Schrenzel, J.; Serrander, L.; Bánfi, B.; Nüsse, O.; Fouyouzi, R.; Lew, D.P.; Demaurex, N.; Krause, K.H. Electron currents generated by the human phagocyte NADPH oxidase. Nature 1998, 392, 734–737. [Google Scholar] [CrossRef] [PubMed]
- Tsong, T.Y. Deciphering the language of cells. Trends Biochem. Sci. 1989, 14, 89–92. [Google Scholar] [CrossRef]
- Platre, M.P.; Jaillais, Y. Anionic lipids and the maintenance of membrane electrostatics in eukaryotes. Plant Signal. Behav. 2017, 12, e1282022. [Google Scholar] [CrossRef] [PubMed]
- Noack, L.C.; Jaillais, Y. Functions of anionic lipids in plants. Annu. Rev. Plant Biol. 2020, 71, 71–102. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Kundu, M.; Zhong, D. Effects of nonequilibrium fluctuations on ultrafast short-range electron transfer dynamics. Nat. Commun. 2020, 11, 2822. [Google Scholar] [CrossRef] [PubMed]
- Wolynes, P.G. Evolution, energy landscapes and the paradoxes of protein folding. Biochimie 2015, 119, 218–230. [Google Scholar] [CrossRef]
- Tsai, M.Y.; Zheng, W.; Balamurugan, D.; Schafer, N.P.; Kim, B.L.; Cheung, M.S.; Wolynes, P.G. Electrostatics, structure prediction, and the energy landscapes for protein folding and binding. Protein Sci. 2016, 25, 255–269. [Google Scholar] [CrossRef]
- Ferreira, P.H.B.; Freitas, F.C.; McCully, M.E.; Slade, G.G.; de Oliveira, R.J. The role of electrostatics and folding kinetics on the thermostability of homologous cold shock proteins. J. Chem. Inf. Model. 2020, 60, 546–561. [Google Scholar] [CrossRef]
- Hering, E. Memory as a General Function of Organized Matter; Address Delivered at the Imperial Academy of Sciences Vienna; Austrian Academy of Sciences: Vienna, Austrian, 30 May 1870. [Google Scholar]
- Hutton, F.W. Hering’s theory of heredity and its consequences. Nature 1904, 69, 366. [Google Scholar] [CrossRef][Green Version]
- Bray, D. Wetware: A Computer in Every Living Cell; Yale University Press: Yale, UK, 2009. [Google Scholar]
- Bray, D. Limits of computational biology. In Silico Biol. 2015, 12, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Witzany, G.; Baluška, F. Turing: A formal clash of codes. Nature 2012, 483, 541. [Google Scholar] [CrossRef] [PubMed]
- Baluška, F.; Witzany, G. Life’s code script does not code itself. The machine metaphor for living organisms is outdated. EMBO Rep. 2012, 13, 1054–1056. [Google Scholar]
- Witzany, G.; Baluška, F. Life is more than a computer running DNA software. World J. Biol. Chem. 2014, 5, 275–278. [Google Scholar]
- Koseska, A.; Bastiaens, P.I. Cell signaling as a cognitive process. EMBO J. 2017, 36, 568–582. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, J.A. Living organisms author their read-write genomes in evolution. Biology 2017, 6, 42. [Google Scholar] [CrossRef]
- Shapiro, J.A. All living cells are cognitive. Biochem. Biophys. Res. Commun. 2020. [Google Scholar] [CrossRef]
- McClintock, B. The Significance of Responses of the Genome to Challenge; Nobel Lecture; The Nobel Foundation: Stockholm, Sweden, 1983. [Google Scholar]
- McClintock, B. The significance of responses of the genome to challenge. Science 1984, 226, 792–801. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, D.J. The machine conception of the organism in development and evolution: A critical analysis. Stud. Hist. Philos. Biol. Biomed. Sci. 2014, 48, 162–174. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, D.J. Is the cell really a machine? J. Theor. Biol. 2019, 47, 108–126. [Google Scholar] [CrossRef]
- Jerman, I.; Krasovec, R.; Leskovar, R.T. Deep significance of the field concept in contemporary biomedical sciences. Electromagn. Biol. Med. 2009, 28, 61–70. [Google Scholar] [CrossRef]
- Skarja, M.; Jerman, I.; Ruzic, R.; Leskovar, R.T.; Jejcic, L. Electric field absorption and emission as an indicator of active electromagnetic nature of organisms. Electromagn. Biol. Med. 2009, 28, 85–95. [Google Scholar] [CrossRef]
- Liboff, A.R. Magnetic correlates in electromagnetic consciousness. Electromagn. Biol. Med. 2016, 35, 228–236. [Google Scholar] [CrossRef]
- De Ninno, A.; Pregnolato, M. Electromagnetic homeostasis and the role of low-amplitude electromagnetic fields on life organization. Electromagn. Biol. Med. 2017, 36, 115–122. [Google Scholar] [CrossRef] [PubMed]
- McFadden, J. Integrating information in the brain’s EM field: The cemi field theory of consciousness. Neurosci. Conscious. 2020, 6, niaa016. [Google Scholar] [CrossRef] [PubMed]
- Fabricant, A.; Iwata, G.Z.; Scherzer, S.; Bougas, L.; Rolfs, K.; Jodko-Władzińska, A.; Voigt, J.; Hedrich, R.; Budker, D. Action potentials induce biomagnetic fields in carnivorous Venus flytrap plants. Sci. Rep. 2021, 11, 1438. [Google Scholar] [CrossRef] [PubMed]
- Levin, M.; Martyniuk, C.J. The bioelectric code: An ancient computational medium for dynamic control of growth and form. Biosystems 2018, 164, 76–93. [Google Scholar] [CrossRef] [PubMed]
- Prindle, A.; Liu, J.; Asally, M.; Ly, S.; Garcia-Ojalvo, J.; Süel, G.M. Ion channels enable electrical communication in bacterial communities. Nature 2015, 527, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.X.; Bialecka-Fornal, M.; Weatherwax, C.; Larkin, J.W.; Prindle, A.; Liu, J.; Garcia-Ojalvo, J.; Süel, G.M. Encoding membrane-potential-based memory within a microbial community. Cell Syst. 2020, 10, 417–423. [Google Scholar] [CrossRef]
- Francis, N.R.; Wolanin, P.M.; Stock, J.B.; Derosier, D.J.; Thomas, D.R. Three-dimensional structure and organization of a receptor/signaling complex. Proc. Natl. Acad. Sci. USA 2004, 101, 17480–17485. [Google Scholar] [CrossRef]
- Webre, D.J.; Wolanin, P.M.; Stock, J.B. Modulated receptor interactions in bacterial transmembrane signaling. Trends Cell Biol. 2004, 14, 478–482. [Google Scholar] [CrossRef]
- Baker, M.D.; Wolanin, P.M.; Stock, J.B. Signal transduction in bacterial chemotaxis. Bioessays 2006, 28, 9–22. [Google Scholar] [CrossRef]
- Li, M.; Hazelbauer, G.L. Core unit of chemotaxis signaling complexes. Proc. Natl. Acad. Sci. USA 2011, 108, 9390–9395. [Google Scholar] [CrossRef]
- Stock, J.B.; Zhang, S. The biochemistry of memory. Curr. Biol. 2013, 23, R741–R745. [Google Scholar] [CrossRef][Green Version]
- Parvez, S.; Long, M.J.C.; Poganik, J.R.; Aye, Y. Redox signaling by reactive electrophiles and oxidants. Chem. Rev. 2018, 118, 8798–8888. [Google Scholar] [CrossRef]
- Kosmachevskaya, O.V.; Shumaev, K.B.; Topunov, A.F. Electrophilic signaling: The role of reactive carbonyl compounds. Biochemistry 2019, 84, S206–S224. [Google Scholar] [CrossRef]
- Kasamatsu, S. Persulfide-dependent regulation of electrophilic redox signaling in neural cells. Antioxid. Redox Signal. 2020, 33, 1320–1331. [Google Scholar] [CrossRef]
- Poganik, J.R.; Long, M.J.C.; Aye, Y. Getting the message? Native reactive electrophiles pass two out of three thresholds to be bona fide signaling mediators. Bioessays 2018, 40, e1700240. [Google Scholar] [CrossRef]
- Liu, X.; Long, M.J.C.; Aye, Y. Proteomics and beyond: Cell decision-making shaped by reactive electrophiles. Trends Biochem. Sci. 2019, 44, 75–89. [Google Scholar] [CrossRef]
- Zhang, X.C.; Li, H. Interplay between the electrostatic membrane potential and conformational changes in membrane proteins. Protein Sci. 2019, 28, 502–512. [Google Scholar] [CrossRef] [PubMed]
- Baluška, F.; Mancuso, S. Synaptic view of eukaryotic cell. Int. J. Gen. Syst. 2014, 43, 740–756. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baluška, F.; Miller, W.B., Jr.; Reber, A.S. Biomolecular Basis of Cellular Consciousness via Subcellular Nanobrains. Int. J. Mol. Sci. 2021, 22, 2545. https://doi.org/10.3390/ijms22052545
Baluška F, Miller WB Jr., Reber AS. Biomolecular Basis of Cellular Consciousness via Subcellular Nanobrains. International Journal of Molecular Sciences. 2021; 22(5):2545. https://doi.org/10.3390/ijms22052545
Chicago/Turabian StyleBaluška, František, William B. Miller, Jr., and Arthur S. Reber. 2021. "Biomolecular Basis of Cellular Consciousness via Subcellular Nanobrains" International Journal of Molecular Sciences 22, no. 5: 2545. https://doi.org/10.3390/ijms22052545
APA StyleBaluška, F., Miller, W. B., Jr., & Reber, A. S. (2021). Biomolecular Basis of Cellular Consciousness via Subcellular Nanobrains. International Journal of Molecular Sciences, 22(5), 2545. https://doi.org/10.3390/ijms22052545





