Neuromodulation of Astrocytic K+ Clearance
Abstract
1. Introduction
2. Results
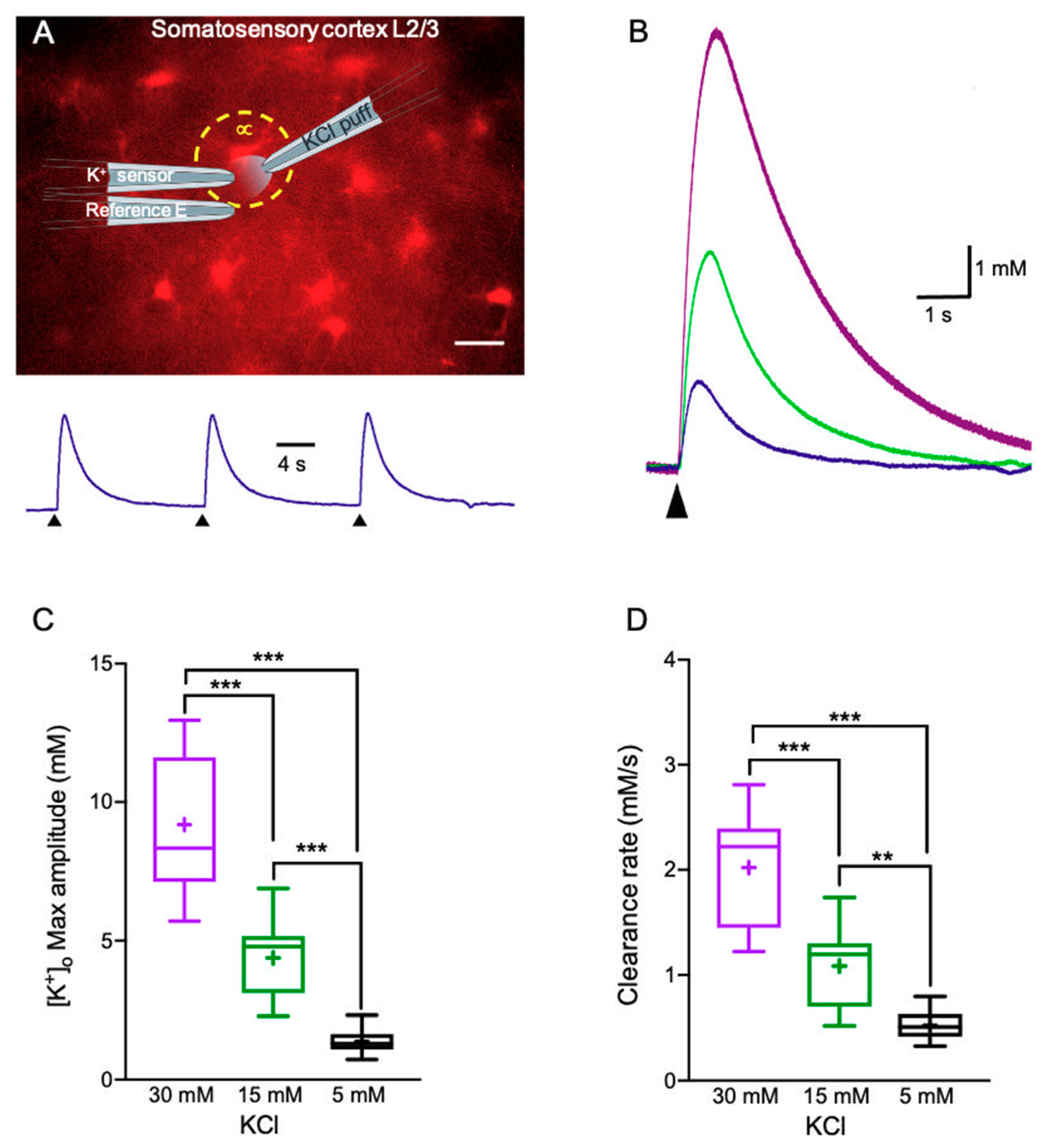
2.1. K+ Clearance Time Course in Acute Brain Slices
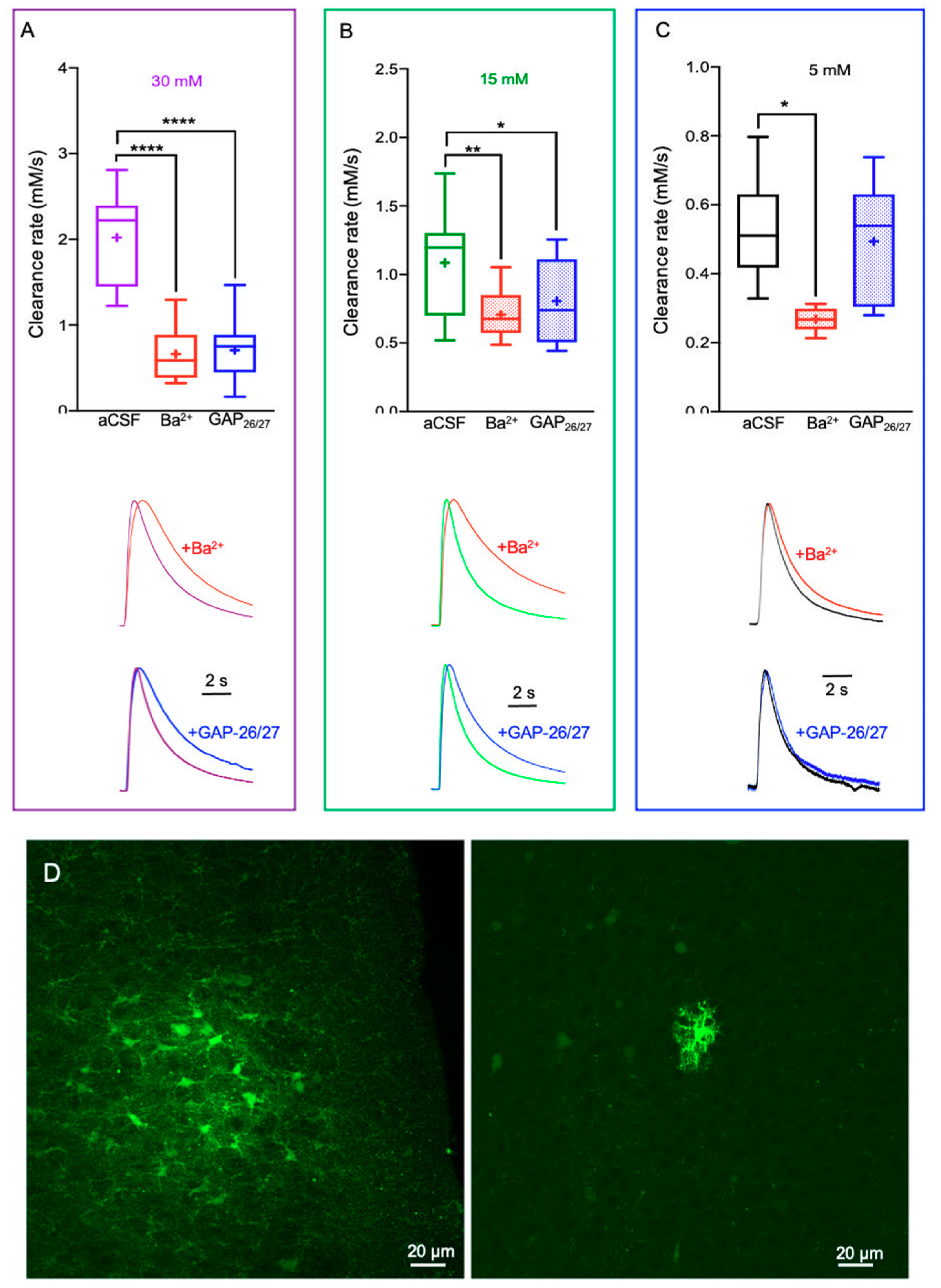
2.2. Alterations in Astrocytic K+ Uptake and Buffering Mechanisms Affect the [K+]o Clearance Rate
2.3. Neuromodulation of Astrocytic K+ Clearance
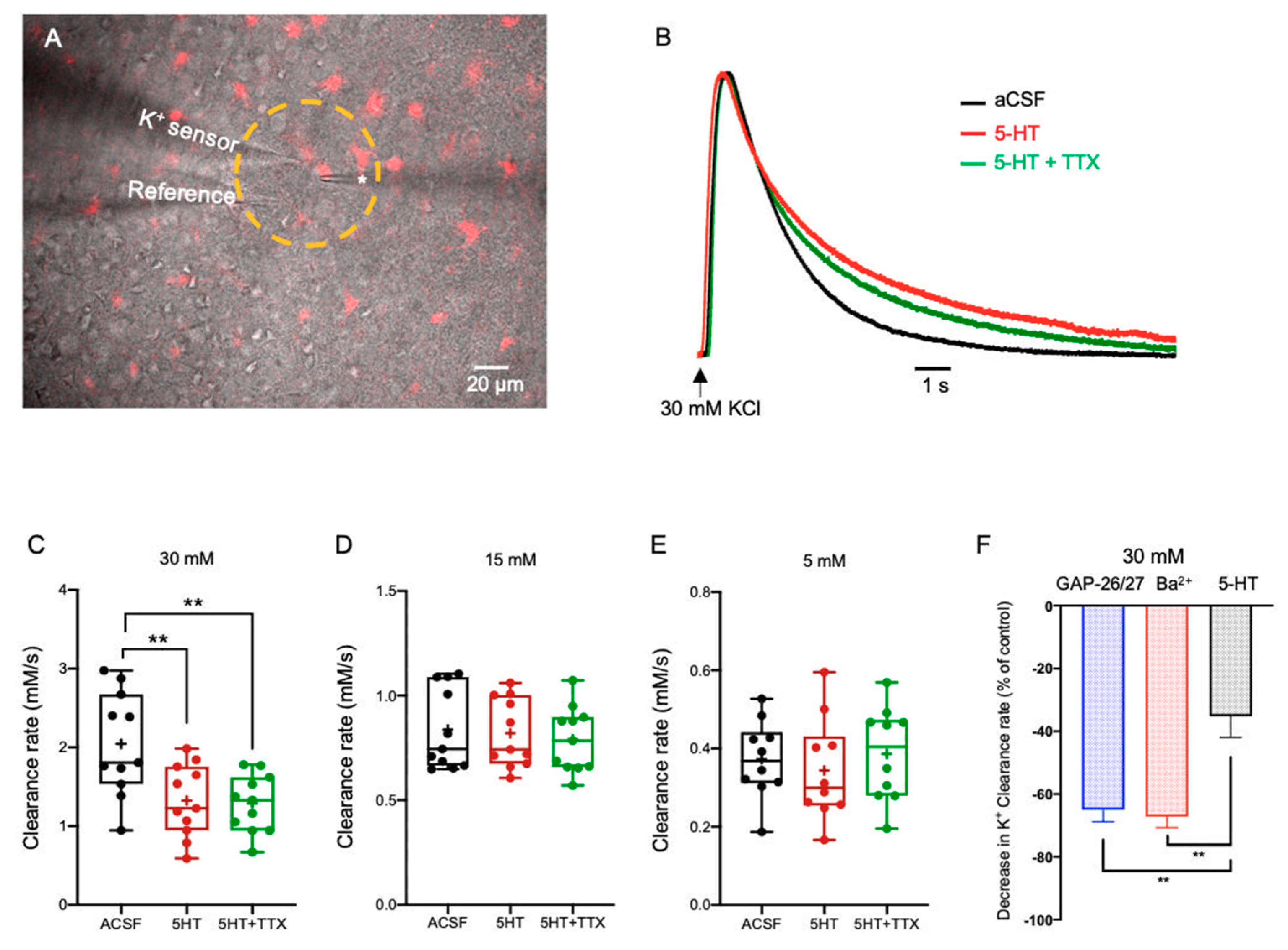
2.3.1. Serotonin (5-HT)
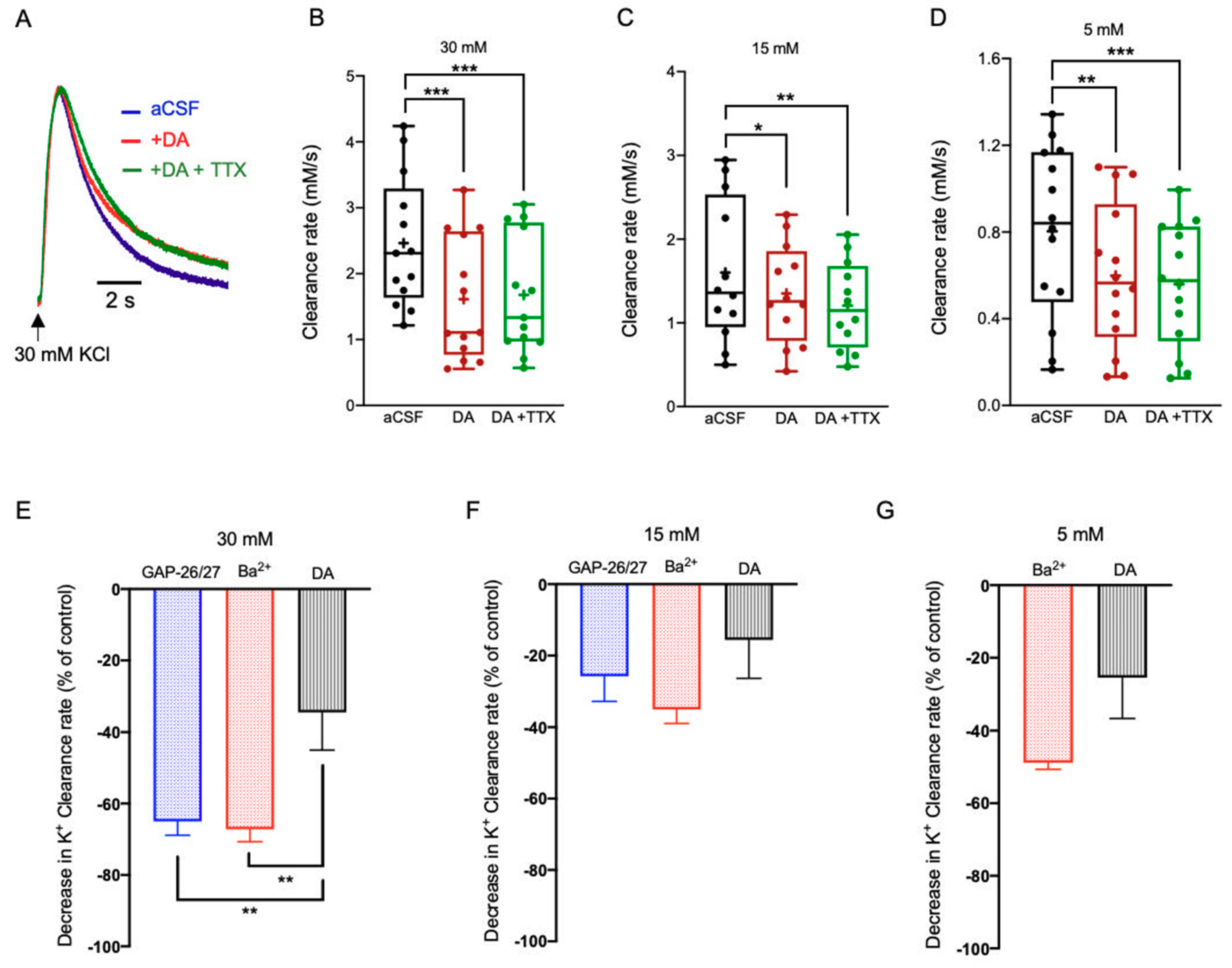
2.3.2. Dopamine (DA)
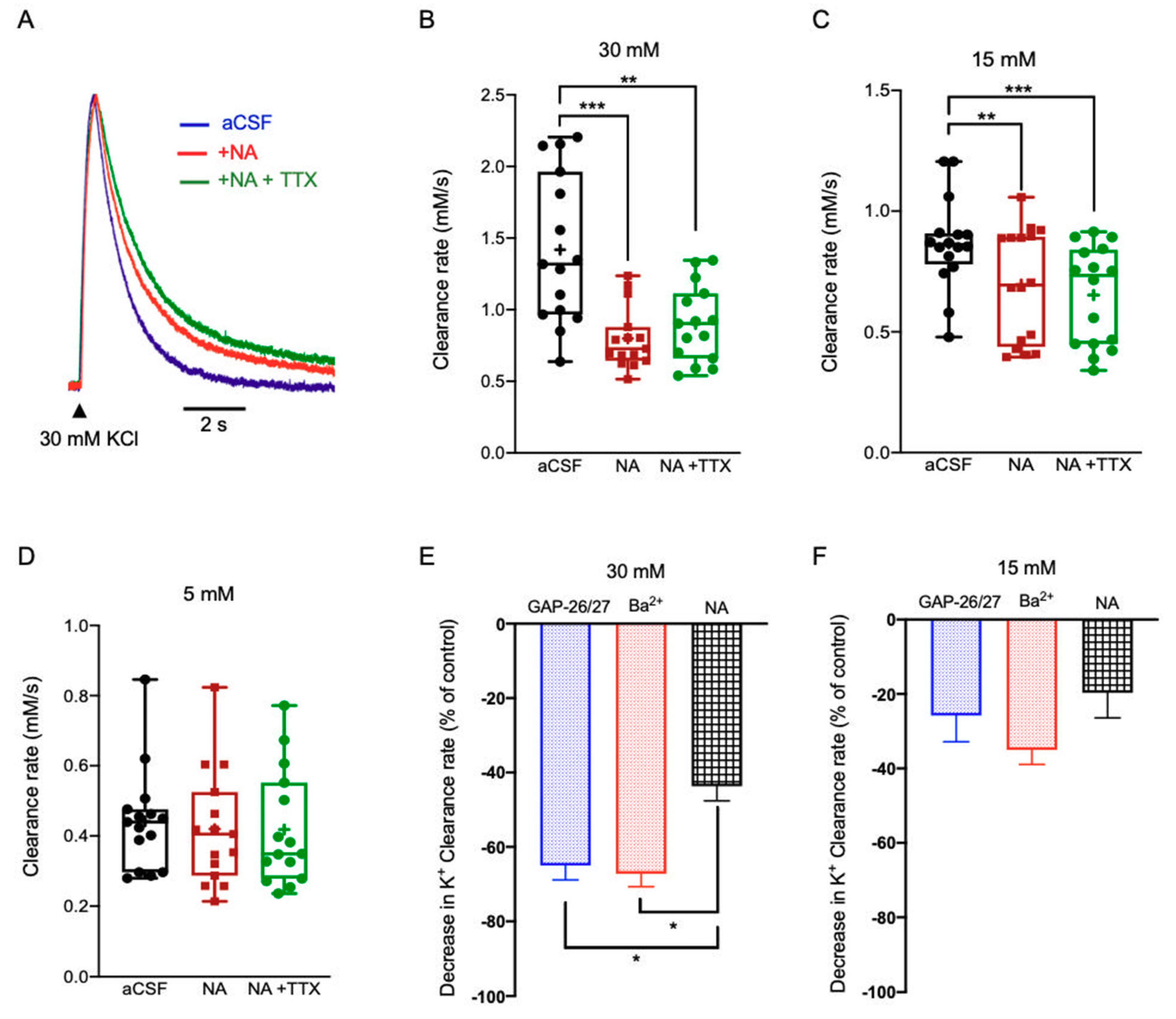
2.3.3. Noradrenaline (NA)
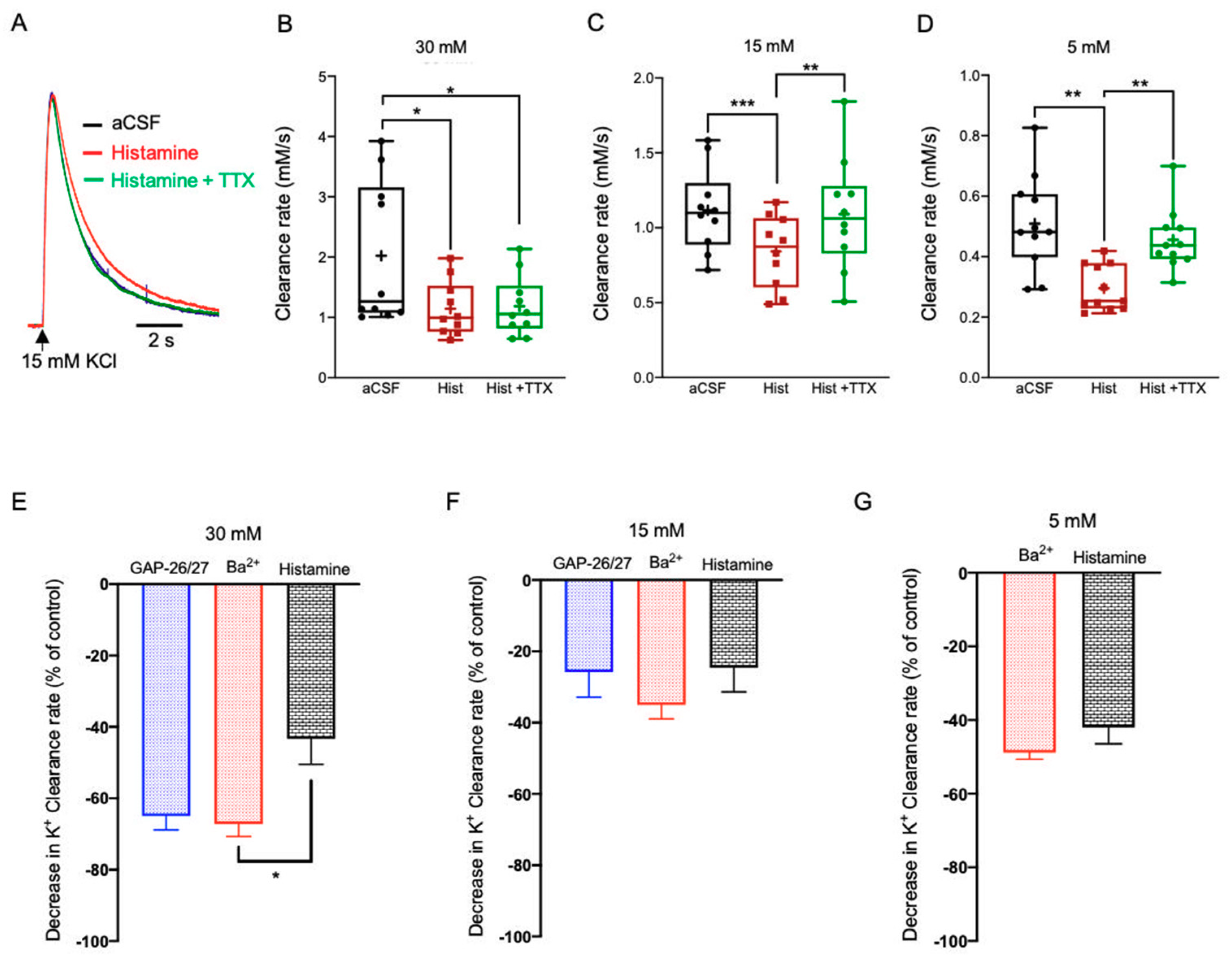
2.3.4. Histamine
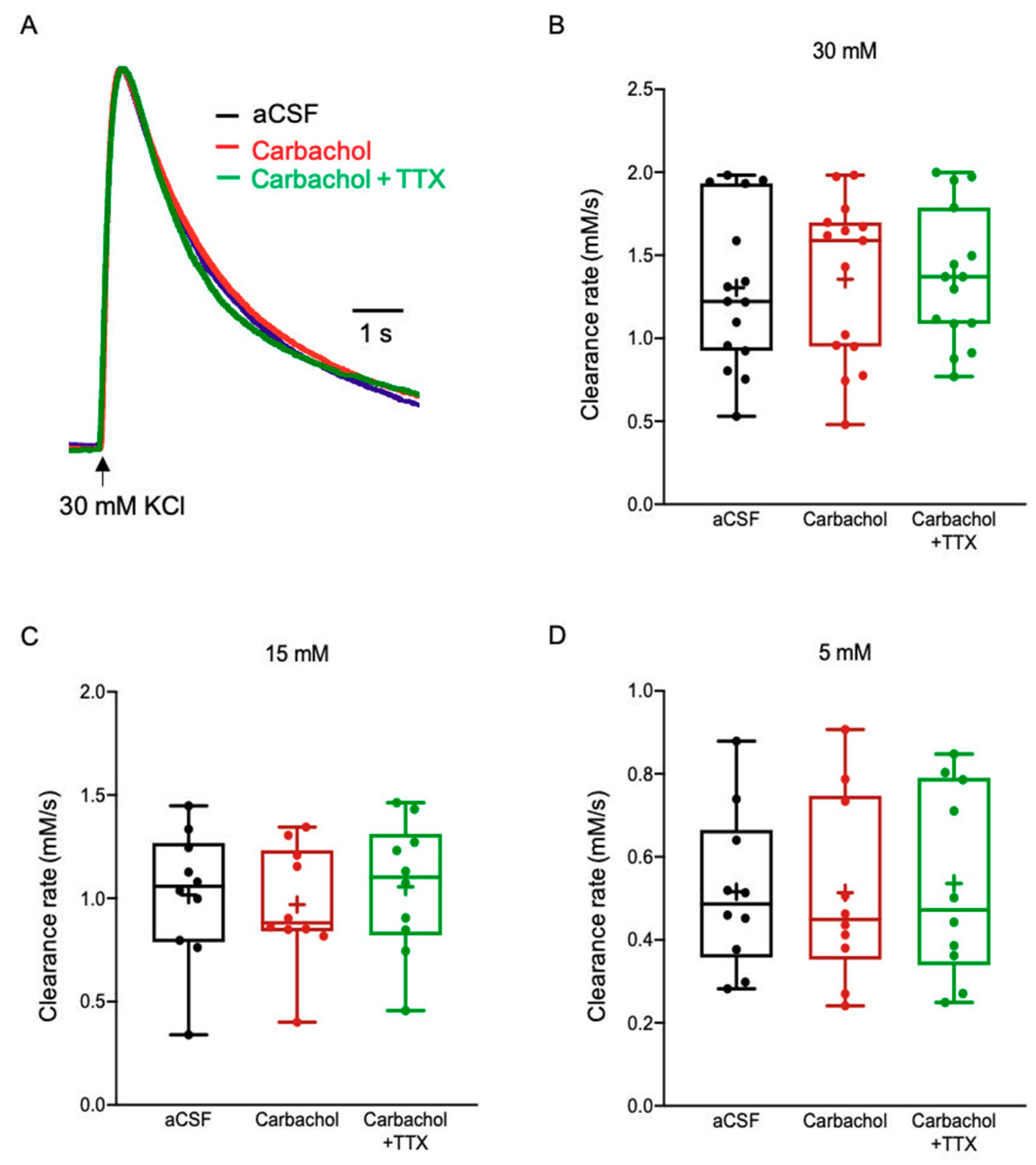
2.3.5. Acetylcholine (ACh)
3. Discussion
3.1. Mechanisms That Affect the K+ Clearance Rate
3.2. Neuromodulators Impact on the [K+]o Clearance Rate
4. Materials and Methods
4.1. Ethical Approval
4.2. Animals and Slice Preparation
4.3. Electrophysiological Recording and Stimulation
4.4. Drugs
4.5. Experimental Design and Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Buskila, Y.; Bellot-Saez, A.; Morley, J.W. Generating Brain Waves, the Power of Astrocytes. Front. Neurosci. 2019, 13, 1125. [Google Scholar] [CrossRef]
- Bellot-Saez, A.; Cohen, G.; Van Schaik, A.; Ooi, L.; Morley, J.W.; Buskila, Y. Astrocytic modulation of cortical oscillations. Sci. Rep. 2018, 8, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-H.; Dan, Y. Neuromodulation of Brain States. Neuron 2012, 76, 209–222. [Google Scholar] [CrossRef] [PubMed]
- Oikawa, H.; Nakamichi, N.; Kambe, Y.; Ogura, M.; Yoneda, Y. An increase in intracellular free calcium ions by nicotinic acetylcholine receptors in a single cultured rat cortical astrocyte. J. Neurosci. Res. 2005, 79, 535–544. [Google Scholar] [CrossRef] [PubMed]
- Amar, M.; Lucas-Meunier, E.; Baux, G.; Fossier, P. Blockade of different muscarinic receptor subtypes changes the equilibrium between excitation and inhibition in rat visual cortex. Neurosci. 2010, 169, 1610–1620. [Google Scholar] [CrossRef]
- Inagaki, N.; Fukui, H.; Taguchi, Y.; Wang, N.P.; Yamatodani, A.; Wada, H. Characterization of histamine H1-receptors on astrocytes in primary culture: [3H]mepyramine binding studies. Eur. J. Pharmacol. 1989, 173, 43–51. [Google Scholar] [CrossRef]
- Hirst, W.D.; Cheung, N.Y.; Rattray, M.; Price, G.W.; Wilkin, G.P. Cultured astrocytes express messenger RNA for multiple serotonin receptor subtypes, without functional coupling of 5-HT1 receptor subtypes to adenylyl cyclase. Mol. Brain Res. 1998, 61, 90–99. [Google Scholar] [CrossRef]
- Bekar, L.K.; He, W.; Nedergaard, M. Locus Coeruleus α-Adrenergic–Mediated Activation of Cortical Astrocytes In Vivo. Cereb. Cortex 2008, 18, 2789–2795. [Google Scholar] [CrossRef]
- Morin, D.; Sapena, R.; Zini, R.; Onteniente, B.; Tillement, J.-P. Characterization of β-adrenergic receptors of freshly isolated astrocytes and neurons from rat brain. Life Sci. 1996, 60, 315–324. [Google Scholar] [CrossRef]
- Khan, Z.U.; Koulen, P.; Rubinstein, M.; Grandy, D.K.; Goldman-Rakic, P.S. An astroglia-linked dopamine D2-receptor action in prefrontal cortex. Proc. Natl. Acad. Sci. USA 2001, 98, 1964–1969. [Google Scholar] [CrossRef]
- Wei, X.; Ma, T.; Cheng, Y.; Huang, C.C.; Wang, X.; Lu, J.; Wang, J. Dopamine D1 or D2 receptor-expressing neurons in the central nervous system. Addict. Biol. 2017, 23, 569–584. [Google Scholar] [CrossRef]
- Stevenson, R.; Samokhina, E.; Rossetti, I.; Morley, J.W.; Buskila, Y. Neuromodulation of Glial Function during Neurodegeneration. Front. Cell. Neurosci. 2020, 14, 1–23. [Google Scholar] [CrossRef]
- Jung, S.; Pfeiffer, F.; Deitmer, J.W. Histamine-induced calcium entry in rat cerebellar astrocytes: Evidence for capacitative and non-capacitative mechanisms. J. Physiol. 2000, 527, 549–561. [Google Scholar] [CrossRef]
- Fang, Q.; Hu, W.-W.; Wang, X.-F.; Yang, Y.; Lou, G.-D.; Jin, M.-M.; Yan, H.-J.; Zeng, W.-Z.; Shen, Y.; Zhang, S.-H.; et al. Histamine up-regulates astrocytic glutamate transporter 1 and protects neurons against ischemic injury. Neuropharmacology 2014, 77, 156–166. [Google Scholar] [CrossRef] [PubMed]
- Ding, F.; O’Donnell, J.; Thrane, A.S.; Zeppenfeld, D.; Kang, H.; Xie, L.; Wang, F.; Nedergaard, M. α1-Adrenergic receptors mediate coordinated Ca2+ signaling of cortical astrocytes in awake, behaving mice. Cell Calcium 2013, 54, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Schnell, C.; Negm, M.; Driehaus, J.; Scheller, A.; Hülsmann, S. Norepinephrine-induced calcium signaling in astrocytes in the respiratory network of the ventrolateral medulla. Respir. Physiol. Neurobiol. 2016, 226, 18–23. [Google Scholar] [CrossRef]
- Nuriya, M.; Takeuchi, M.; Yasui, M. Background norepinephrine primes astrocytic calcium responses to subsequent norepinephrine stimuli in the cerebral cortex. Biochem. Biophys. Res. Commun. 2017, 483, 732–738. [Google Scholar] [CrossRef]
- Jennings, A.; Tyurikova, O.; Bard, L.; Zheng, K.; Semyanov, A.; Henneberger, C.; Rusakov, D.A. Dopamine elevates and lowers astroglial Ca2+through distinct pathways depending on local synaptic circuitry. Glia 2016, 65, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Vaarmann, A.; Gandhi, S.; Abramov, A.Y. Dopamine Induces Ca2+ Signaling in Astrocytes through Reactive Oxygen Species Generated by Monoamine Oxidase. J. Biol. Chem. 2010, 285, 25018–25023. [Google Scholar] [CrossRef] [PubMed]
- Bosson, A.; Boisseau, S.; Buisson, A.; Savasta, M.; Albrieux, M. Disruption of dopaminergic transmission remodels tripartite synapse morphology and astrocytic calcium activity within substantia nigra pars reticulata. Glia 2014, 63, 673–683. [Google Scholar] [CrossRef]
- Blomstrand, F.; Khatibi, S.; Muyderman, H.; Hansson, E.; Olsson, T.; Rönnbäck, L. 5-hydroxytryptamine and glutamate modulate velocity and extent of intercellular calcium signalling in hippocampal astroglial cells in primary cultures. Neuroscience 1999, 88, 1241–1253. [Google Scholar] [CrossRef]
- Schipke, C.G.; Heuser, I.; Peters, O. Antidepressants act on glial cells: SSRIs and serotonin elicit astrocyte calcium signaling in the mouse prefrontal cortex. J. Psychiatr. Res. 2011, 45, 242–248. [Google Scholar] [CrossRef]
- Sandén, N.; Thorlin, T.; Blomstrand, F.; Persson, P.; Hansson, E. 5-Hydroxytryptamine2B receptors stimulate Ca2+ increases in cultured astrocytes from three different brain regions. Neurochem. Int. 2000, 36, 427–434. [Google Scholar] [CrossRef]
- Araque, A.; Martin, E.D.; Perea, G.; Arellano, J.I.; Buno, W. Synaptically Released Acetylcholine Evokes Ca2+ Elevations in Astrocytes in Hippocampal Slices. J. Neurosci. 2002. [Google Scholar] [CrossRef]
- Perea, G. Properties of Synaptically Evoked Astrocyte Calcium Signal Reveal Synaptic Information Processing by Astrocytes. J. Neurosci. 2005, 25, 2192–2203. [Google Scholar] [CrossRef] [PubMed]
- Ding, F.; O’Donnell, J.; Xu, Q.; Kang, N.; Goldman, N.; Nedergaard, M. Changes in the composition of brain interstitial ions control the sleep-wake cycle. Science (80-) 2016, 352, 550–555. [Google Scholar] [CrossRef]
- Wang, F.; Smith, N.A.; Xu, Q.; Fujita, T.; Baba, A.; Matsuda, T.; Takano, T.; Bekar, L.; Nedergaard, M. Astrocytes Modulate Neural Network Activity by Ca2+-Dependent Uptake of Extracellular K+. Sci. Signal. 2012, 5, ra26. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Stork, T.; Bergles, D.E.; Freeman, M.R. Neuromodulators signal through astrocytes to alter neural circuit activity and behaviour. Nature 2016, 539, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Bellot-Saez, A.; Kékesi, O.; Morley, J.W.; Buskila, Y. Astrocytic modulation of neuronal excitability through K+ spatial buffering. Neurosci. Biobehav. Rev. 2017, 77, 87–97. [Google Scholar] [CrossRef]
- Liang, Z.; Wang, X.; Hao, Y.; Qiu, L.; Lou, Y.; Zhang, Y.; Ma, D.; Feng, J. The Multifaceted Role of Astrocyte Connexin 43 in Ischemic Stroke through Forming Hemichannels and Gap Junctions. Front. Neurol. 2020, 11, 1–12. [Google Scholar] [CrossRef]
- Haack, N.; Durry, S.; Kafitz, K.W.; Chesler, M.; Rose, C.R. Double-barreled and Concentric Microelectrodes for Measurement of Extracellular Ion Signals in Brain Tissue. J. Vis. Exp. 2015, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Ransom, C.B.; Ransom, B.R.; Sontheimer, H. Activity-dependent extracellular K+ accumulation in rat optic nerve: The role of glial and axonal Na+pumps. J. Physiol. 2000, 522, 427–442. [Google Scholar] [CrossRef] [PubMed]
- Djukic, B.; Casper, K.B.; Philpot, B.D.; Chin, L.-S.; McCarthy, K.D. Conditional Knock-Out of Kir4.1 Leads to Glial Membrane Depolarization, Inhibition of Potassium and Glutamate Uptake, and Enhanced Short-Term Synaptic Potentiation. J. Neurosci. 2007, 27, 11354–11365. [Google Scholar] [CrossRef] [PubMed]
- Ransom, B.; Sontheimer, H. Biophysical and pharmacological characterization of inwardly rectifying K+ currents in rat spinal cord astrocytes. J. Neurophysiol. 1995, 73, 333–346. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Yang, Y.; Ni, Z.; Dong, Y.; Cai, G.; Foncelle, A.; Ma, S.; Sang, K.; Tang, S.; Li, Y.; et al. Astroglial Kir4.1 in the lateral habenula drives neuronal bursts in depression. Nat. Cell Biol. 2018, 554, 323–327. [Google Scholar] [CrossRef] [PubMed]
- Wallraff, A.; Köhling, R.; Heinemann, U.; Theis, M.; Willecke, K.; Steinhäuser, C. The Impact of Astrocytic Gap Junctional Coupling on Potassium Buffering in the Hippocampus. J. Neurosci. 2006, 26, 5438–5447. [Google Scholar] [CrossRef]
- Larsen, B.R.; Macaulay, N. Kir4.1-mediated spatial buffering of KC: Experimental challenges in determination of its temporal and quantitative contribution to KC clearance in the brain. Channels 2014, 8, 1–7. [Google Scholar] [CrossRef]
- Ma, B.; Xu, G.; Wang, W.; Enyeart, J.J.; Zhou, M. Dual patch voltage clamp study of low membrane resistance astrocytes in situ. Mol. Brain 2014, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Desplantez, T.; Verma, V.; Leybaert, L.; Evans, W.; Weingart, R. Gap26, a connexin mimetic peptide, inhibits currents carried by connexin43 hemichannels and gap junction channels. Pharmacol. Res. 2012, 65, 546–552. [Google Scholar] [CrossRef]
- Enkvist, M.O.K.; McCarthy, K.D. Astroglial Gap Junction Communication Is Increased by Treatment with Either Glutamate or High K+ Concentration. J. Neurochem. 2002, 62, 489–495. [Google Scholar] [CrossRef]
- Halnes, G.; Østby, I.; Pettersen, K.H.; Omholt, S.W.; Einevoll, G.T. Electrodiffusive Model for Astrocytic and Neuronal Ion Concentration Dynamics. PLoS Comput. Biol. 2013, 9, e1003386. [Google Scholar] [CrossRef] [PubMed]
- Bonhaus, D.W.; Bach, C.; DeSouza, A.; Salazar, F.R.; Matsuoka, B.D.; Zuppan, P.; Chan, H.W.; Eglen, R.M. The pharmacology and distribution of human 5-hydroxytryptamine2B (5-HT2b) receptor gene products: Comparison with 5-HT2a and 5-HT2c receptors. Br. J. Pharmacol. 1995, 115, 622–628. [Google Scholar] [CrossRef]
- Azmitia, E.; Gannon, P.; Kheck, N.; Whitakerazinitia, P. Cellular localization of the 5-HT receptor in primate brain neurons and glial cells. Neuropsychopharmacology 1996, 14, 35–46. [Google Scholar] [CrossRef]
- Carson, M.J.; Thomas, E.A.; Danielson, P.E.; Sutcliffe, J.G. The 5-HT(5A) serotonin receptor is expressed predomi-nantly by astrocytes in which it inhibits cAMP accumulation: A mechanism for neuronal suppression of reactive astrocytes. Glia 1996. [Google Scholar] [CrossRef]
- Maxishima, M.; Shiga, T.; Shutoh, F.; Hamada, S.; Maeshima, T.; Okado, N. Serotonin 2A receptor-like immunoreactivity is detected in astrocytes but not in oligodendrocytes of rat spinal cord. Brain Res. 2001, 889, 270–273. [Google Scholar] [CrossRef]
- Mahé, C.; Bernhard, M.; Bobirnac, I.; Keser, C.; Loetscher, E.; Feuerbach, D.; Dev, K.K.; Schoeffter, P. Functional expression of the serotonin 5-HT7 receptor in human glioblastoma cell lines. Br. J. Pharmacol. 2004, 143, 404–410. [Google Scholar] [CrossRef]
- Jennings, A.; Rusakov, D.A. Do astrocytes respond to dopamine? Opera. Med. Physiol. 2016, 2, 34–43. [Google Scholar]
- Castro, L.R.V.; Brito, M.; Guiot, E.; Polito, M.; Korn, C.W.; Hervé, D.; Girault, J.-A.; Paupardin-Tritsch, D.; Vincent, P. Striatal neurones have a specific ability to respond to phasic dopamine release. J. Physiol. 2013, 591, 3197–3214. [Google Scholar] [CrossRef]
- Wotton, C.A.; Cross, C.D.; Bekar, L.K. Serotonin, norepinephrine, and acetylcholine differentially affect astrocytic potassium clearance to modulate somatosensory signaling in male mice. J. Neurosci. Res. 2020, 98, 964–977. [Google Scholar] [CrossRef]
- Medrano, S.; Gruenstein, E.; Dimlich, R.V. Histamine stimulates glycogenolysis in human astrocytoma cells by increasing intracellular free calcium. Brain Res. 1992, 592, 202–207. [Google Scholar] [CrossRef]
- Hirase, H.; Iwai, Y.; Takata, N.; Shinohara, Y.; Mishima, T. Volume transmission signalling via astrocytes. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130604. [Google Scholar] [CrossRef] [PubMed]
- Hobson, J.A.; Goldberg, M.; Vivaldi, E.; Riew, D. Enhancement of desynchronized sleep signs after pontine microinjection of the muscarinic agonist bethanechol. Brain Res. 1983, 275, 127–136. [Google Scholar] [CrossRef]
- Orkand, R.K.; Nicholls, J.G.; Kuffler, S.W. Effect of nerve impulses on the membrane potential of glial cells in the cen-tral nervous system of amphibia. J. Neurophysiol. 1966, 29, 788–806. [Google Scholar] [CrossRef] [PubMed]
- Ito, H.T.; Schuman, E.M. Frequency-dependent gating of synaptic transmission and plasticity by dopamine. Front. Neural Circuits 2007, 1, 1. [Google Scholar] [CrossRef] [PubMed]
- Kirkwood, A.; Rozas, C.; Kirkwood, J.; Perez, F.; Bear, M.F. Modulation of Long-Term Synaptic Depression in Visual Cortex by Acetylcholine and Norepinephrine. J. Neurosci. 1999, 19, 1599–1609. [Google Scholar] [CrossRef]
- O’Donnell, J.; Zeppenfeld, D.; McConnell, E.; Pena, S.; Nedergaard, M. Norepinephrine: A Neuromodulator That Boosts the Function of Multiple Cell Types to Optimize CNS Performance. Neurochem. Res. 2012, 37, 2496–2512. [Google Scholar] [CrossRef]
- Constantinople, C.M.; Bruno, R.M. Effects and Mechanisms of Wakefulness on Local Cortical Networks. Neuron 2011, 69, 1061–1068. [Google Scholar] [CrossRef]
- Webster, H.H.; Jones, B.E. Neurotoxic lesions of the dorsolateral pontomesencephalic tegmentum-cholinergic cell area in the cat. II. Effects upon sleep-waking states. Brain Res. 1988, 458, 285–302. [Google Scholar] [CrossRef]
- Van Dort, C.J.; Zachs, D.P.; Kenny, J.D.; Zheng, S.; Goldblum, R.R.; Gelwan, N.A.; Ramos, D.M.; Nolan, M.A.; Wang, K.; Weng, F.-J.; et al. Optogenetic activation of cholinergic neurons in the PPT or LDT induces REM sleep. Proc. Natl. Acad. Sci. USA 2015, 112, 584–589. [Google Scholar] [CrossRef]
- Ni, K.-M.; Hou, X.-J.; Yang, C.-H.; Dong, P.; Li, Y.; Zhang, Y.; Jiang, P.; Berg, D.K.; Duan, S.; Li, X.-M. Selectively driving cholinergic fibers optically in the thalamic reticular nucleus promotes sleep. eLife 2016, 5, e10382. [Google Scholar] [CrossRef]
- Carter, M.E.; Yizhar, O.; Chikahisa, S.; Nguyen, H.; Adamantidis, A.; Nishino, S.; Deisseroth, K.; De Lecea, L. Tuning arousal with optogenetic modulation of locus coeruleus neurons. Nat. Neurosci. 2010, 13, 1526–1533. [Google Scholar] [CrossRef]
- Monti, J.M. Involvement of histamine in the control of the waking state. Life Sci. 1993, 53, 1331–1338. [Google Scholar] [CrossRef]
- Monti, J.; Jantos, H. The roles of dopamine and serotonin, and of their receptors, in regulating sleep and waking. Progress Brain Res. 2008, 172, 625–646. [Google Scholar] [CrossRef]
- Larsen, B.R.; Stoica, A.; Macaulay, N. Managing Brain Extracellular K+ during Neuronal Activity: The Physiological Role of the Na+/K+-ATPase Subunit Isoforms. Front. Physiol. 2016, 7, 1–10. [Google Scholar] [CrossRef]
- Rasmussen, R.; Nicholas, E.; Petersen, N.C.; Dietz, A.G.; Xu, Q.; Sun, Q.; Nedergaard, M. Cortex-wide Changes in Extracellular Potassium Ions Parallel Brain State Transitions in Awake Behaving Mice. Cell Rep. 2019, 28, 1182–1194. [Google Scholar] [CrossRef] [PubMed]
- Volnova, A.; Tsytsarev, V.; Ptukha, M.; Inyushin, M. In Vitro and In Vivo Study of the Short-Term Vasomotor Response during Epileptic Seizures. Brain Sci. 2020, 10, 942. [Google Scholar] [CrossRef] [PubMed]
- Gardner-Medwin, A.R. Analysis of potassium dynamics in mammalian brain tissue. J. Physiol. 1983, 335, 393–426. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Hu, B.; Zhang, H.; Boyle, C.A. Saobo Lei Roles of K+ and Cation Channels in ORL-1 Receptor-mediated Depression of Neuronal Excitability and Epileptic Activities in the Medial Entorhinal Cortex. Neuropharmacology 2019, 151, 144–158. [Google Scholar] [CrossRef] [PubMed]
- Thrane, V.R.; Thrane, A.S.; Wang, F.; Cotrina, M.L.; Smith, N.A.; Chen, M.; Xu, Q.; Kang, N.; Fujita, T.; Nagelhus, E.A.; et al. Ammonia triggers neuronal disinhibition and seizures by impairing astrocyte potassium buffering. Nat. Med. 2013, 19, 1643–1648. [Google Scholar] [CrossRef]
- Heinemann, U.; Lux, H.D. Ceiling of stimulus induced rises in extracellular potassium concentration in the cerebral cortex of cat. Brain Res. 1977, 120, 231–249. [Google Scholar] [CrossRef]
- Orkand, R.K. Introductory Remarks: Glial-interstitial fluid exchange. Neuronal Microenviron. 1986, 481, 269–272. [Google Scholar] [CrossRef] [PubMed]
- Larsen, B.R.; Assentoft, M.; Cotrina, M.L.; Hua, S.Z.; Nedergaard, M.; Kaila, K.; Voipio, J.; Macaulay, N. Contributions of the Na+/K+-ATPase, NKCC1, and Kir4.1 to hippocampal K+ clearance and volume responses. Glia 2014, 62, 608–622. [Google Scholar] [CrossRef] [PubMed]
- Hajek, I.; Subbarao, K.V.; Hertz, L. Acute and chronic effects of potassium and noradrenaline on Na+, K+-ATPase activity in cultured mouse neurons and astrocytes. Neurochem. Int. 1996, 28, 335–342. [Google Scholar] [CrossRef]
- Butt, A.M.; Kalsi, A. Inwardly rectifying potassium channels (Kir) in central nervous system glia: A special role for Kir4.1 in glial functions. J. Cell. Mol. Med. 2006, 10, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Pannasch, U.; Vargová, L.; Reingruber, J.; Ezan, P.; Holcman, D.; Giaume, C.; Syková, E.; Rouach, N. Astroglial networks scale synaptic activity and plasticity. Proc. Natl. Acad. Sci. USA 2011, 108, 8467–8472. [Google Scholar] [CrossRef]
- Eckart, C.; Woźniak-Kwaśniewska, A.; Herweg, N.A.; Fuentemilla, L.; Bunzeck, N. Acetylcholine modulates human working memory and subsequent familiarity based recognition via alpha oscillations. NeuroImage 2016, 137, 61–69. [Google Scholar] [CrossRef]
- Lowry, C.A.; Johnson, P.L.; Hay-Schmidt, A.; Mikkelsen, J.; Shekhar, A. Modulation of anxiety circuits by serotonergic systems. Stress 2005, 8, 233–246. [Google Scholar] [CrossRef] [PubMed]
- Cirelli, C.; Tononi, G. Differential Expression of Plasticity-Related Genes in Waking and Sleep and Their Regulation by the Noradrenergic System. J. Neurosci. 2000, 20, 9187–9194. [Google Scholar] [CrossRef]
- Ray, N.; Jenkinson, N.; Wang, S.; Holland, P.; Brittain, J.; Joint, C.; Stein, J.; Aziz, T. Local field potential beta activity in the subthalamic nucleus of patients with Parkinson’s disease is associated with improvements in bradykinesia after dopamine and deep brain stimulation. Exp. Neurol. 2008, 213, 108–113. [Google Scholar] [CrossRef]
- Panula, P.; Rinne, J.; Kuokkanen, K.; Eriksson, K.; Sallmen, T.; Kalimo, H.; Relja, M. Neuronal histamine deficit in Alzheimer’s disease. Neuroscience 1997, 82, 993–997. [Google Scholar] [CrossRef]
- Heneka, M.T.; Nadrigny, F.; Regen, T.; Martinez-Hernandez, A.; Dumitrescu-Ozimek, L.; Terwel, D.; Jardanhazi-Kurutz, D.; Walter, J.; Kirchhoff, F.; Hanisch, U.-K.; et al. Locus ceruleus controls Alzheimer’s disease pathology by modulating microglial functions through norepinephrine. Proc. Natl. Acad. Sci. USA 2010, 107, 6058–6063. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Shalinsky, M.H.; Alonso, A.; Dickson, C.T. Effects of serotonin on the intrinsic membrane properties of layer II medial entorhinal cortex neurons. Hippocampus 2007, 17, 114–129. [Google Scholar] [CrossRef]
- Rosenkranz, J.A.; Johnston, D. Dopaminergic Regulation of Neuronal Excitability through Modulation of Ih in Layer V Entorhinal Cortex. J. Neurosci. 2006, 26, 3229–3244. [Google Scholar] [CrossRef]
- Maccaferri, G.; McBain, C.J. The hyperpolarization-activated current (Ih) and its contribution to pacemaker activity in rat CA1 hippocampal stratum oriensalveus interneurones. J. Physiol. 1996, 497 Pt 1, 119–130. [Google Scholar] [CrossRef]
- Tseng, H.-A.; Martinez, D.; Nadim, F. The frequency preference of neurons and synapses in a recurrent oscillatory network. J. Neurosci. 2014, 34, 12933–12945. [Google Scholar] [CrossRef]
- Slezak, M.; Kandler, S.; Van Veldhoven, P.P.; Bonin, V.; Holt, M.G. Astrocytes integrate local sensory and brain-wide neuromodulatory signals. bioRxiv 2018, 381434. [Google Scholar] [CrossRef]
- Grundy, D. Principles and standards for reporting animal experiments in The Journal of Physiology and Experimental Physiology. J. Physiol. 2015, 593, 2547–2549. [Google Scholar] [CrossRef] [PubMed]
- Buskila, Y.; Morley, J.W.; Tapson, J.; Van Schaik, A. The adaptation of spike backpropagation delays in cortical neurons. Front. Cell. Neurosci. 2013, 7, 192. [Google Scholar] [CrossRef]
- Kekesi, O.; Buskila, Y. Method for Prolonged Incubation of Brain Slices. Bio-Protocol 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Breen, P.P.; Buskila, Y. Braincubator: An incubation system to extend brain slice lifespan for use in neurophysiology. In Proceedings of the 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, EMBC 2014, Chicago, IL, USA, 26–30 August 2014; pp. 4864–4867. [Google Scholar]
- Cameron, M.; Kekesi, O.; Morley, J.W.; Tapson, J.; Breen, P.P.; Van Schaik, A.; Buskila, Y. Calcium Imaging of AM Dyes Following Prolonged Incubation in Acute Neuronal Tissue. PLoS ONE 2016, 11, e0155468. [Google Scholar] [CrossRef] [PubMed]
- Buskila, Y.; Breen, P.P.; Tapson, J.; Van Schaik, A.; Barton, M.B.; Morley, J.W. Extending the viability of acute brain slices. Sci. Rep. 2015, 4, 5309. [Google Scholar] [CrossRef] [PubMed]
- Buskila, Y.; Bellot-Saez, A.; Kékesi, O.; Cameron, M.; Morley, J. Extending the Life Span of Acute Neuronal Tissue for Imaging and Electrophysiological Studies. In The Brain Reward System; Wright, N., Ed.; Springer: New York, NY, USA, 2019; Volume 152, pp. 235–259. ISBN 978-1-4614-9064-7. [Google Scholar]
- Haack, N.; Rose, C.R. Preparation, Calibration and Application of Potassium-Selective Microelectrodes. Microelectrodes 2014, 49, 87–105. [Google Scholar]
- Deveau, J.S.T.; Lindinger, M.I.; Grodzinski, B. An improved method for constructing and selectively silanizing double-barreled, neutral liquid-carrier, ion-selective microelectrodes. Biol. Proced. Online 2005, 7, 31–40. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bellot-Saez, A.; Stevenson, R.; Kékesi, O.; Samokhina, E.; Ben-Abu, Y.; Morley, J.W.; Buskila, Y. Neuromodulation of Astrocytic K+ Clearance. Int. J. Mol. Sci. 2021, 22, 2520. https://doi.org/10.3390/ijms22052520
Bellot-Saez A, Stevenson R, Kékesi O, Samokhina E, Ben-Abu Y, Morley JW, Buskila Y. Neuromodulation of Astrocytic K+ Clearance. International Journal of Molecular Sciences. 2021; 22(5):2520. https://doi.org/10.3390/ijms22052520
Chicago/Turabian StyleBellot-Saez, Alba, Rebecca Stevenson, Orsolya Kékesi, Evgeniia Samokhina, Yuval Ben-Abu, John W. Morley, and Yossi Buskila. 2021. "Neuromodulation of Astrocytic K+ Clearance" International Journal of Molecular Sciences 22, no. 5: 2520. https://doi.org/10.3390/ijms22052520
APA StyleBellot-Saez, A., Stevenson, R., Kékesi, O., Samokhina, E., Ben-Abu, Y., Morley, J. W., & Buskila, Y. (2021). Neuromodulation of Astrocytic K+ Clearance. International Journal of Molecular Sciences, 22(5), 2520. https://doi.org/10.3390/ijms22052520





