Neural Circuitry–Neurogenesis Coupling Model of Depression
Abstract
1. Introduction
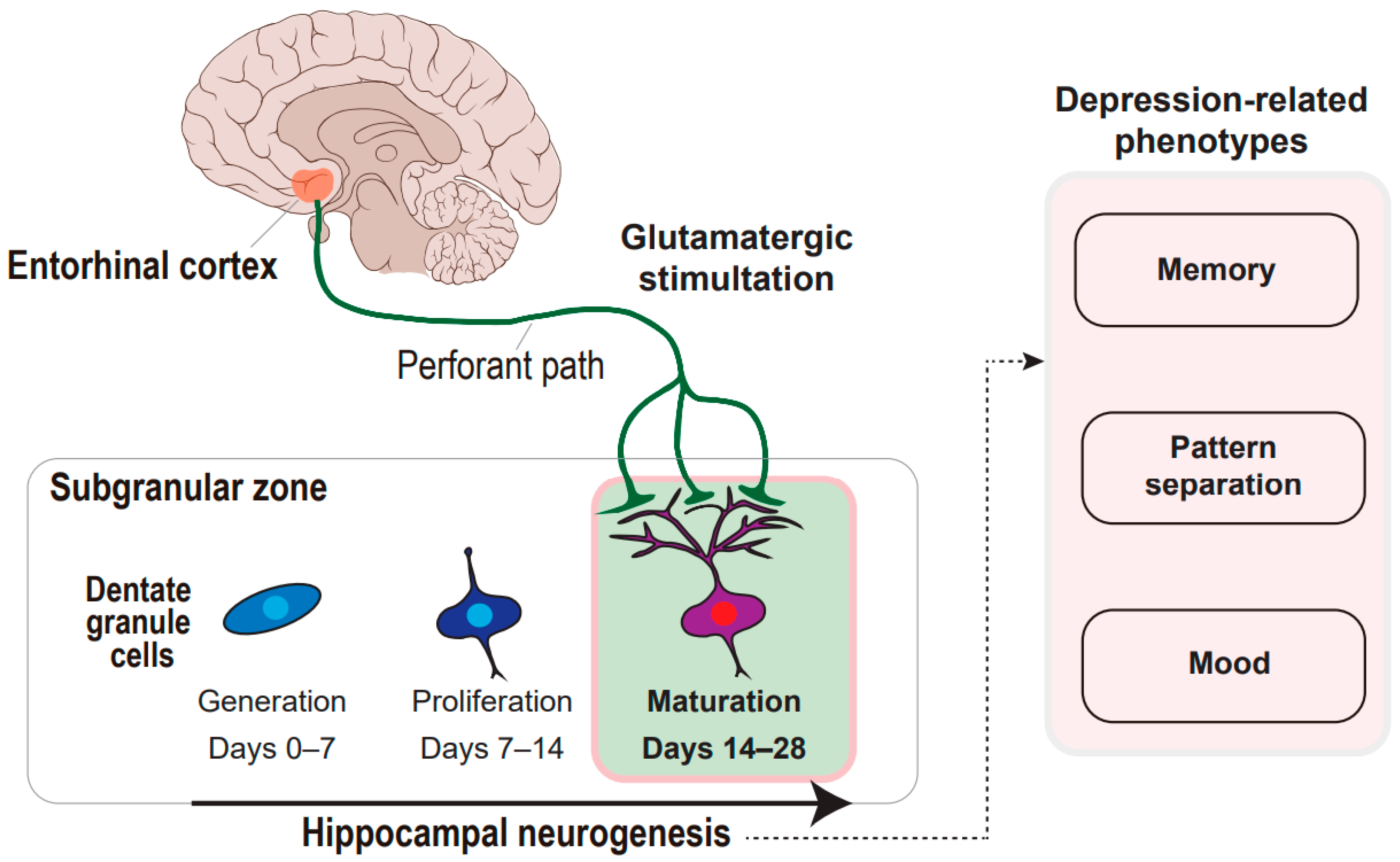
2. Activity-Dependent Adult Neurogenesis and Framework of Neural Circuitry–Neurogenesis Coupling Model of Depression
3. Entorhinal–Hippocampal Circuitry and Neurogenesis in Memory
3.1. Supportive Findings from Human Studies
3.2. Supportive Findings from Animal Studies
4. Entorhinal–Hippocampal Circuitry and Neurogenesis in Pattern Separation
4.1. Supportive Findings from Human Studies
4.2. Supportive Findings from Animal Studies
5. Entorhinal–Hippocampal Circuitry and Neurogenesis in Mood Regulation
Supportive Findings from Animal Studies
6. Suggestions for Advancing the Neural Circuitry–Neurogenesis Coupling Model of Depression
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lopez, A.D.; Mathers, C.D.; Ezzati, M.; Jamison, D.T.; Murray, C.J. Global Burden of Disease and Risk Factors; The World Bank: Washington, DC, USA, 2006. [Google Scholar]
- Hay, S.I.; Jayaraman, S.P.; Truelsen, T.; Sorensen, R.J.; Millear, A.; Giussani, G.; Beghi, E. GBD 2015 disease and injury incidence and prevalence collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: A systematic analysis for the global burden of disease study 2015 (vol 388, pg 1545, 2016). Lancet 2017, 389, E1. [Google Scholar]
- World Health Organization. Depression and Other Common Mental Disorders: Global Health Estimates; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Kupfer, D.J.; Frank, E.; Phillips, M.L. Major depressive disorder: New clinical, neurobiological, and treatment perspectives. Focus 2016, 14, 266–276. [Google Scholar] [CrossRef]
- Trivedi, M.H. Modeling predictors, moderators and mediators of treatment outcome and resistance in depression. Biol. Psychiatry 2013, 74, 2–4. [Google Scholar] [CrossRef]
- McGrath, P.J.; Khan, A.Y.; Trivedi, M.H.; Stewart, J.W.; Morris, D.W.; Wisniewski, S.R.; Miyahara, S.; Nierenberg, A.A.; Fava, M.; Rush, A.J. Response to a selective serotonin reuptake inhibitor (citalopram) in major depressive disorder with melancholic features: A STAR* D report. J. Clin. Psychiatry 2008, 69, 1847–1855. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-C.; Shinfuku, N.; Maramis, M.M.; Lee, M.-S.; Park, Y.C. Adjunctive antipsychotic prescriptions for outpatients with depressive disorders in Asia: The research on Asian psychotropic prescription patterns for antidepressants (REAP-AD) study. Am. J. Psychiatry 2015, 172, 684–685. [Google Scholar] [CrossRef] [PubMed]
- Insel, T.R. Next-generation treatments for mental disorders. Sci. Transl. Med. 2012, 4, ps119–ps155. [Google Scholar] [CrossRef] [PubMed]
- Kronmüller, K.-T.; Pantel, J.; Köhler, S.; Victor, D.; Giesel, F.; Magnotta, V.A.; Mundt, C.; Essig, M.; Schröder, J. Hippocampal volume and 2-year outcome in depression. Br. J. Psychiatry 2008, 192, 472–473. [Google Scholar] [CrossRef]
- Yun, S.; Reynolds, R.P.; Masiulis, I.; Eisch, A.J. Re-evaluating the link between neuropsychiatric disorders and dysregulated adult neurogenesis. Nat. Med. 2016, 22, 1239–1247. [Google Scholar] [CrossRef]
- Sahay, A.; Hen, R. Adult hippocampal neurogenesis in depression. Nat. Neurosci. 2007, 10, 1110–1115. [Google Scholar] [CrossRef]
- Ma, D.K.; Kim, W.R.; Ming, G.-L.; Song, H. Activity-dependent extrinsic regulation of adult olfactory bulb and hippocampal neurogenesis. Ann. N. Y. Acad. Sci. 2009, 1170, 664. [Google Scholar] [CrossRef]
- Kempermann, G. Regulation of adult hippocampal neurogenesis–implications for novel theories of major depression 1. Bipolar Disord. 2002, 4, 17–33. [Google Scholar] [CrossRef]
- Song, J.; Olsen, R.H.; Sun, J.; Ming, G.-l.; Song, H. Neuronal circuitry mechanisms regulating adult mammalian neurogenesis. Cold Spring Harb. Perspect. Biol. 2016, 8, a018937. [Google Scholar] [CrossRef]
- Stone, S.S.; Teixeira, C.M.; DeVito, L.M.; Zaslavsky, K.; Josselyn, S.A.; Lozano, A.M.; Frankland, P.W. Stimulation of entorhinal cortex promotes adult neurogenesis and facilitates spatial memory. J. Neurosci. 2011, 31, 13469–13484. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Hu, W.-H.; Wu, D.-L.; Zhang, K.; Zhang, J.-G. Behavioral effects of deep brain stimulation of the anterior nucleus of thalamus, entorhinal cortex and fornix in a rat model of Alzheimer’s disease. Chin. Med. J. 2015, 128, 1190. [Google Scholar] [CrossRef] [PubMed]
- Ronaghi, A.; Zibaii, M.I.; Pandamooz, S.; Nourzei, N.; Motamedi, F.; Ahmadiani, A.; Dargahi, L. Entorhinal cortex stimulation induces dentate gyrus neurogenesis through insulin receptor signaling. Brain Res. Bull. 2019, 144, 75–84. [Google Scholar] [CrossRef]
- Suthana, N.; Haneef, Z.; Stern, J.; Mukamel, R.; Behnke, E.; Knowlton, B.; Fried, I. Memory enhancement and deep-brain stimulation of the entorhinal area. N. Engl. J. Med. 2012, 366, 502–510. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.; Reynolds, R.P.; Petrof, I.; White, A.; Rivera, P.D.; Segev, A.; Gibson, A.D.; Suarez, M.; DeSalle, M.J.; Ito, N. Stimulation of entorhinal cortex–dentate gyrus circuitry is antidepressive. Nat. Med. 2018, 24, 658–666. [Google Scholar] [CrossRef]
- Witter, M.P. The perforant path: Projections from the entorhinal cortex to the dentate gyrus. Prog. Brain Res. 2007, 163, 43–61. [Google Scholar]
- Li, Y.; Mu, Y.; Gage, F.H. Development of neural circuits in the adult hippocampus. Curr. Top. Dev. Biol. 2009, 87, 149–174. [Google Scholar]
- Claiborne, B.J.; Amaral, D.G.; Cowan, W.M. A light and electron microscopic analysis of the mossy fibers of the rat dentate gyrus. J. Comp. Neurol. 1986, 246, 435–458. [Google Scholar] [CrossRef]
- Köhler, C. Intrinsic connections of the retrohippocampal region in the rat brain. II. The medial entorhinal area. J. Comp. Neurol. 1986, 246, 149–169. [Google Scholar] [CrossRef]
- Kempermann, G.; Jessberger, S.; Steiner, B.; Kronenberg, G. Milestones of neuronal development in the adult hippocampus. Trends Neurosci. 2004, 27, 447–452. [Google Scholar] [CrossRef]
- Ge, S.; Sailor, K.A.; Ming, G.l.; Song, H. Synaptic integration and plasticity of new neurons in the adult hippocampus. J. Physiol. 2008, 586, 3759–3765. [Google Scholar] [CrossRef] [PubMed]
- Ge, S.; Goh, E.L.; Sailor, K.A.; Kitabatake, Y.; Ming, G.-l.; Song, H. GABA regulates synaptic integration of newly generated neurons in the adult brain. Nature 2006, 439, 589–593. [Google Scholar] [CrossRef] [PubMed]
- Vivar, C.; Potter, M.C.; Choi, J.; Lee, J.-y.; Stringer, T.P.; Callaway, E.M.; Gage, F.H.; Suh, H.; Van Praag, H. Monosynaptic inputs to new neurons in the dentate gyrus. Nat. Commun. 2012, 3, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Hieber, C.; Jonas, P.; Bischofberger, J. Enhanced synaptic plasticity in newly generated granule cells of the adult hippocampus. Nature 2004, 429, 184–187. [Google Scholar] [CrossRef] [PubMed]
- Ge, S.; Yang, C.-H.; Hsu, K.-S.; Ming, G.-L.; Song, H. A critical period for enhanced synaptic plasticity in newly generated neurons of the adult brain. Neuron 2007, 54, 559–566. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.M.; Reif, A.; Schmitt, A.G. Major depression: A role for hippocampal neurogenesis? In Behavioral Neurobiology of Depression and Its Treatment; Springer: Berlin/Heidelberg, Germany, 2012; pp. 153–179. [Google Scholar]
- Wang, H.; Warner-Schmidt, J.; Varela, S.; Enikolopov, G.; Greengard, P.; Flajolet, M. Norbin ablation results in defective adult hippocampal neurogenesis and depressive-like behavior in mice. Proc. Natl. Acad. Sci. USA 2015, 112, 9745–9750. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; He, H.; Qiao, Y.; Zhou, T.; He, H.; Yi, S.; Zhang, L.; Mo, L.; Li, Y.; Jiang, W. Priming of microglia with IFN-γ impairs adult hippocampal neurogenesis and leads to depression-like behaviors and cognitive defects. Glia 2020, 68, 2674–2692. [Google Scholar] [CrossRef] [PubMed]
- Zakzanis, K.K.; Leach, L.; Kaplan, E. On the nature and pattern of neurocognitive function in major depressive disorder. Neuropsychiatry Neuropsychol. Behav. Neurol. 1998, 11, 111–119. [Google Scholar] [PubMed]
- Elderkin-Thompson, V.; Mintz, J.; Haroon, E.; Lavretsky, H.; Kumar, A. Executive dysfunction and memory in older patients with major and minor depression. Arch. Clin. Neuropsychol. 2007, 22, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Austin, M.-P.; Mitchell, P.; Goodwin, G.M. Cognitive deficits in depression: Possible implications for functional neuropathology. Br. J. Psychiatry 2001, 178, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Burt, D.B.; Zembar, M.J.; Niederehe, G. Depression and memory impairment: A meta-analysis of the association, its pattern, and specificity. Psychol. Bull. 1995, 117, 285. [Google Scholar] [CrossRef]
- Söderlund, H.; Moscovitch, M.; Kumar, N.; Daskalakis, Z.J.; Flint, A.; Herrmann, N.; Levine, B. Autobiographical episodic memory in major depressive disorder. J. Abnorm. Psychol. 2014, 123, 51. [Google Scholar] [CrossRef] [PubMed]
- Bäckman, L.; Forsell, Y. Episodic memory functioning in a community-based sample of old adults with major depression: Utilization of cognitive support. J. Abnorm. Psychol. 1994, 103, 361. [Google Scholar] [CrossRef]
- Lemogne, C.; Piolino, P.; Friszer, S.; Claret, A.; Girault, N.; Jouvent, R.; Allilaire, J.-F.; Fossati, P. Episodic autobiographical memory in depression: Specificity, autonoetic consciousness, and self-perspective. Conscious. Cogn. 2006, 15, 258–268. [Google Scholar] [CrossRef]
- Murray, B.D.; Holland, A.C.; Kensinger, E.A. Episodic Memory and Emotion; The Guilford Press: New York, NY, USA, 2013. [Google Scholar]
- Steffens, D.C.; Byrum, C.E.; McQuoid, D.R.; Greenberg, D.L.; Payne, M.E.; Blitchington, T.F.; MacFall, J.R.; Krishnan, K.R.R. Hippocampal volume in geriatric depression. Biol. Psychiatry 2000, 48, 301–309. [Google Scholar] [CrossRef]
- Lloyd, A.J.; Ferrier, I.N.; Barber, R.; Gholkar, A.; Young, A.H.; O’Brien, J.T. Hippocampal volume change in depression: Late-and early-onset illness compared. Br. J. Psychiatry 2004, 184, 488–495. [Google Scholar] [CrossRef]
- O’Brien, J.T.; Lloyd, A.; McKeith, I.; Gholkar, A.; Ferrier, N. A longitudinal study of hippocampal volume, cortisol levels, and cognition in older depressed subjects. Am. J. Psychiatry 2004, 161, 2081–2090. [Google Scholar] [CrossRef]
- Hickie, I.; Naismith, S.; Ward, P.B.; Turner, K.; Scott, E.; Mitchell, P.; Wilhelm, K.; Parker, G. Reduced hippocampal volumes and memory loss in patients with early-and late-onset depression. Br. J. Psychiatry 2005, 186, 197–202. [Google Scholar] [CrossRef]
- Bell-McGinty, S.; Butters, M.A.; Meltzer, C.C.; Greer, P.J.; Reynolds, C.F., III; Becker, J.T. Brain morphometric abnormalities in geriatric depression: Long-term neurobiological effects of illness duration. Am. J. Psychiatry 2002, 159, 1424–1427. [Google Scholar] [CrossRef]
- Fang, J.; Demic, S.; Cheng, S. The reduction of adult neurogenesis in depression impairs the retrieval of new as well as remote episodic memory. PLoS ONE 2018, 13, e0198406. [Google Scholar] [CrossRef] [PubMed]
- Becker, S.; Wojtowicz, J.M. A model of hippocampal neurogenesis in memory and mood disorders. Trends Cogn. Sci. 2007, 11, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Déry, N.; Pilgrim, M.; Gibala, M.; Gillen, J.; Wojtowicz, J.M.; MacQueen, G.; Becker, S. Adult hippocampal neurogenesis reduces memory interference in humans: Opposing effects of aerobic exercise and depression. Front. Neurosci. 2013, 7, 66. [Google Scholar] [CrossRef] [PubMed]
- Deng, W.; Aimone, J.B.; Gage, F.H. New neurons and new memories: How does adult hippocampal neurogenesis affect learning and memory? Nat. Rev. Neurosci. 2010, 11, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Tsao, A.; Sugar, J.; Lu, L.; Wang, C.; Knierim, J.J.; Moser, M.-B.; Moser, E.I. Integrating time from experience in the lateral entorhinal cortex. Nature 2018, 561, 57–62. [Google Scholar] [CrossRef]
- Kropff, E.; Carmichael, J.E.; Moser, M.-B.; Moser, E.I. Speed cells in the medial entorhinal cortex. Nature 2015, 523, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, W.A.; Miller, E.K.; Desimone, R. Object and place memory in the macaque entorhinal cortex. J. Neurophysiol. 1997, 78, 1062–1081. [Google Scholar] [CrossRef] [PubMed]
- Lipton, P.; Eichenbaum, H. Complementary roles of hippocampus and medial entorhinal cortex in episodic memory. Neural Plast. 2008, 2008. [Google Scholar] [CrossRef]
- Sugar, J.; Moser, M.B. Episodic memory: Neuronal codes for what, where, and when. Hippocampus 2019, 29, 1190–1205. [Google Scholar] [CrossRef]
- Sasaki, T.; Leutgeb, S.; Leutgeb, J.K. Spatial and memory circuits in the medial entorhinal cortex. Curr. Opin. Neurobiol. 2015, 32, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Buzsáki, G.; Moser, E.I. Memory, navigation and theta rhythm in the hippocampal-entorhinal system. Nat. Neurosci. 2013, 16, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, J.; Miller, J.; Lee, S.A.; Coffey, T.; Watrous, A.J.; Sperling, M.R.; Sharan, A.; Worrell, G.; Berry, B.; Lega, B. Direct electrical stimulation of the human entorhinal region and hippocampus impairs memory. Neuron 2016, 92, 983–990. [Google Scholar] [CrossRef] [PubMed]
- Kee, N.; Teixeira, C.M.; Wang, A.H.; Frankland, P.W. Preferential incorporation of adult-generated granule cells into spatial memory networks in the dentate gyrus. Nat. Neurosci. 2007, 10, 355–362. [Google Scholar] [CrossRef]
- Stone, S.S.; Teixeira, C.M.; Zaslavsky, K.; Wheeler, A.L.; Martinez-Canabal, A.; Wang, A.H.; Sakaguchi, M.; Lozano, A.M.; Frankland, P.W. Functional convergence of developmentally and adult-generated granule cells in dentate gyrus circuits supporting hippocampus-dependent memory. Hippocampus 2011, 21, 1348–1362. [Google Scholar] [CrossRef] [PubMed]
- Robinson, N.T.; Priestley, J.B.; Rueckemann, J.W.; Garcia, A.D.; Smeglin, V.A.; Marino, F.A.; Eichenbaum, H. Medial entorhinal cortex selectively supports temporal coding by hippocampal neurons. Neuron 2017, 94, 677–688.e676. [Google Scholar] [CrossRef]
- Eichenbaum, H.; Sauvage, M.; Fortin, N.; Komorowski, R.; Lipton, P. Towards a functional organization of episodic memory in the medial temporal lobe. Neurosci. Biobehav. Rev. 2012, 36, 1597–1608. [Google Scholar] [CrossRef]
- Neunuebel, J.P.; Yoganarasimha, D.; Rao, G.; Knierim, J.J. Conflicts between local and global spatial frameworks dissociate neural representations of the lateral and medial entorhinal cortex. J. Neurosci. 2013, 33, 9246–9258. [Google Scholar] [CrossRef] [PubMed]
- Nilssen, E.S.; Doan, T.P.; Nigro, M.J.; Ohara, S.; Witter, M.P. Neurons and networks in the entorhinal cortex: A reappraisal of the lateral and medial entorhinal subdivisions mediating parallel cortical pathways. Hippocampus 2019, 29, 1238–1254. [Google Scholar] [CrossRef]
- Kerr, K.M.; Agster, K.L.; Furtak, S.C.; Burwell, R.D. Functional neuroanatomy of the parahippocampal region: The lateral and medial entorhinal areas. Hippocampus 2007, 17, 697–708. [Google Scholar] [CrossRef]
- Fyhn, M.; Molden, S.; Witter, M.P.; Moser, E.I.; Moser, M.-B. Spatial representation in the entorhinal cortex. Science 2004, 305, 1258–1264. [Google Scholar] [CrossRef]
- Leutgeb, J.K.; Leutgeb, S.; Moser, M.-B.; Moser, E.I. Pattern separation in the dentate gyrus and CA3 of the hippocampus. Science 2007, 315, 961–966. [Google Scholar] [CrossRef] [PubMed]
- Bakker, A.; Kirwan, C.B.; Miller, M.; Stark, C.E. Pattern separation in the human hippocampal CA3 and dentate gyrus. Science 2008, 319, 1640–1642. [Google Scholar] [CrossRef] [PubMed]
- Rolls, E. The mechanisms for pattern completion and pattern separation in the hippocampus. Front. Syst. Neurosci. 2013, 7, 74. [Google Scholar] [CrossRef] [PubMed]
- Yassa, M.A.; Stark, C.E. Pattern separation in the hippocampus. Trends Neurosci. 2011, 34, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Clelland, C.; Choi, M.; Romberg, C.; Clemenson, G.D.; Fragniere, A.; Tyers, P.; Jessberger, S.; Saksida, L.; Barker, R.; Gage, F. A functional role for adult hippocampal neurogenesis in spatial pattern separation. Science 2009, 325, 210–213. [Google Scholar] [CrossRef] [PubMed]
- Gandy, K.; Kim, S.; Sharp, C.; Dindo, L.; Maletic-Savatic, M.; Calarge, C. Pattern separation: A potential marker of impaired hippocampal adult neurogenesis in major depressive disorder. Front. Neurosci. 2017, 11, 571. [Google Scholar] [CrossRef]
- Sahay, A.; Scobie, K.N.; Hill, A.S.; O’Carroll, C.M.; Kheirbek, M.A.; Burghardt, N.S.; Fenton, A.A.; Dranovsky, A.; Hen, R. Increasing adult hippocampal neurogenesis is sufficient to improve pattern separation. Nature 2011, 472, 466–470. [Google Scholar] [CrossRef]
- Berron, D.; Schütze, H.; Maass, A.; Cardenas-Blanco, A.; Kuijf, H.J.; Kumaran, D.; Düzel, E. Strong evidence for pattern separation in human dentate gyrus. J. Neurosci. 2016, 36, 7569–7579. [Google Scholar] [CrossRef]
- Gerritsen, L.; Comijs, H.C.; van der Graaf, Y.; Knoops, A.J.; Penninx, B.W.; Geerlings, M.I. Depression, hypothalamic pituitary adrenal axis, and hippocampal and entorhinal cortex volumes—The SMART Medea study. Biol. Psychiatry 2011, 70, 373–380. [Google Scholar] [CrossRef]
- Dickerson, B.C.; Feczko, E.; Augustinack, J.C.; Pacheco, J.; Morris, J.C.; Fischl, B.; Buckner, R.L. Differential effects of aging and Alzheimer’s disease on medial temporal lobe cortical thickness and surface area. Neurobiol. Aging 2009, 30, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Olsen, R.K.; Yeung, L.-K.; Noly-Gandon, A.; D’Angelo, M.C.; Kacollja, A.; Smith, V.M.; Ryan, J.D.; Barense, M.D. Human anterolateral entorhinal cortex volumes are associated with cognitive decline in aging prior to clinical diagnosis. Neurobiol. Aging 2017, 57, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Reagh, Z.M.; Noche, J.A.; Tustison, N.J.; Delisle, D.; Murray, E.A.; Yassa, M.A. Functional imbalance of anterolateral entorhinal cortex and hippocampal dentate/CA3 underlies age-related object pattern separation deficits. Neuron 2018, 97, 1187–1198.e1184. [Google Scholar] [CrossRef]
- Maurer, A.P.; Johnson, S.A.; Hernandez, A.R.; Reasor, J.; Cossio, D.M.; Fertal, K.E.; Mizell, J.M.; Lubke, K.N.; Clark, B.J.; Burke, S.N. Age-related changes in lateral entorhinal and CA3 neuron allocation predict poor performance on object discrimination. Front. Syst. Neurosci. 2017, 11, 49. [Google Scholar] [CrossRef]
- Leal, S.L.; Noche, J.A.; Murray, E.A.; Yassa, M.A. Disruption of amygdala–entorhinal–hippocampal network in late-life depression. Hippocampus 2017, 27, 464–476. [Google Scholar] [CrossRef]
- Kheirbek, M.A.; Tannenholz, L.; Hen, R. NR2B-dependent plasticity of adult-born granule cells is necessary for context discrimination. J. Neurosci. 2012, 32, 8696–8702. [Google Scholar] [CrossRef] [PubMed]
- Niibori, Y.; Yu, T.-S.; Epp, J.R.; Akers, K.G.; Josselyn, S.A.; Frankland, P.W. Suppression of adult neurogenesis impairs population coding of similar contexts in hippocampal CA3 region. Nat. Commun. 2012, 3, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.V.; Hen, R. Functional dissociation of adult-born neurons along the dorsoventral axis of the dentate gyrus. Hippocampus 2014, 24, 751–761. [Google Scholar] [CrossRef]
- Tronel, S.; Belnoue, L.; Grosjean, N.; Revest, J.M.; Piazza, P.V.; Koehl, M.; Abrous, D.N. Adult-born neurons are necessary for extended contextual discrimination. Hippocampus 2012, 22, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Mahar, I.; Bambico, F.R.; Mechawar, N.; Nobrega, J.N. Stress, serotonin, and hippocampal neurogenesis in relation to depression and antidepressant effects. Neurosci. Biobehav. Rev. 2014, 38, 173–192. [Google Scholar] [CrossRef]
- Paizanis, E.; Hamon, M.; Lanfumey, L. Hippocampal neurogenesis, depressive disorders, and antidepressant therapy. Neural Plast. 2007, 2007. [Google Scholar] [CrossRef]
- Tang, M.-M.; Lin, W.-J.; Pan, Y.-Q.; Guan, X.-T.; Li, Y.-C. Hippocampal neurogenesis dysfunction linked to depressive-like behaviors in a neuroinflammation induced model of depression. Physiol. Behav. 2016, 161, 166–173. [Google Scholar] [CrossRef]
- Sahay, A.; Drew, M.R.; Hen, R. Dentate gyrus neurogenesis and depression. Prog. Brain Res. 2007, 163, 697–822. [Google Scholar] [PubMed]
- Kubera, M.; Obuchowicz, E.; Goehler, L.; Brzeszcz, J.; Maes, M. In animal models, psychosocial stress-induced (neuro) inflammation, apoptosis and reduced neurogenesis are associated to the onset of depression. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2011, 35, 744–759. [Google Scholar] [CrossRef]
- Chesnokova, V.; Pechnick, R.N.; Wawrowsky, K. Chronic peripheral inflammation, hippocampal neurogenesis, and behavior. Brain Behav. Immun. 2016, 58, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, M.L.; Brachman, R.A.; Martinowich, K.; Schloesser, R.J.; Herkenham, M. Glucocorticoids orchestrate divergent effects on mood through adult neurogenesis. J. Neurosci. 2013, 33, 2961–2972. [Google Scholar] [CrossRef]
- Taliaz, D.; Stall, N.; Dar, D.; Zangen, A. Knockdown of brain-derived neurotrophic factor in specific brain sites precipitates behaviors associated with depression and reduces neurogenesis. Mol. Psychiatry 2010, 15, 80–92. [Google Scholar] [CrossRef]
- Lee, E.; Son, H. Adult hippocampal neurogenesis and related neurotrophic factors. BMB Rep. 2009, 42, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-C. Neurogenesis and antidepressant action. Cell Tissue Res. 2019, 1–12. [Google Scholar] [CrossRef]
- Pascual-Leone, A.; Rubio, B.; Pallardó, F.; Catalá, M.D. Rapid-rate transcranial magnetic stimulation of left dorsolateral prefrontal cortex in drug-resistant depression. Lancet 1996, 348, 233–237. [Google Scholar] [CrossRef]
- Padberg, F.; George, M.S. Repetitive transcranial magnetic stimulation of the prefrontal cortex in depression. Exp. Neurol. 2009, 219, 2–13. [Google Scholar] [CrossRef]
- George, M.S.; Nahas, Z.; Molloy, M.; Speer, A.M.; Oliver, N.C.; Li, X.-B.; Arana, G.W.; Risch, S.C.; Ballenger, J.C. A controlled trial of daily left prefrontal cortex TMS for treating depression. Biol. Psychiatry 2000, 48, 962–970. [Google Scholar] [CrossRef]
- Boggio, P.S.; Bermpohl, F.; Vergara, A.O.; Muniz, A.L.; Nahas, F.H.; Leme, P.B.; Rigonatti, S.P.; Fregni, F. Go-no-go task performance improvement after anodal transcranial DC stimulation of the left dorsolateral prefrontal cortex in major depression. J. Affect. Disord. 2007, 101, 91–98. [Google Scholar] [CrossRef]
- Nahas, Z.; Teneback, C.C.; Kozel, A.; Speer, A.M.; DeBrux, C.; Molloy, M.; Stallings, L.; Spicer, K.M.; Arana, G.; Bohning, D.E. Brain effects of TMS delivered over prefrontal cortex in depressed adults: Role of stimulation frequency and coil–cortex distance. J. Neuropsychiatry Clin. Neurosci. 2001, 13, 459–470. [Google Scholar] [CrossRef]
- Li, X.; Nahas, Z.; Kozel, F.A.; Anderson, B.; Bohning, D.E.; George, M.S. Acute left prefrontal transcranial magnetic stimulation in depressed patients is associated with immediately increased activity in prefrontal cortical as well as subcortical regions. Biol. Psychiatry 2004, 55, 882–890. [Google Scholar] [CrossRef]
- Levkovitz, Y.; Harel, E.V.; Roth, Y.; Braw, Y.; Most, D.; Katz, L.N.; Sheer, A.; Gersner, R.; Zangen, A. Deep transcranial magnetic stimulation over the prefrontal cortex: Evaluation of antidepressant and cognitive effects in depressive patients. Brain Stimul. 2009, 2, 188–200. [Google Scholar] [CrossRef]
- Hamani, C.; Diwan, M.; Macedo, C.E.; Brandão, M.L.; Shumake, J.; Gonzalez-Lima, F.; Raymond, R.; Lozano, A.M.; Fletcher, P.J.; Nobrega, J.N. Antidepressant-like effects of medial prefrontal cortex deep brain stimulation in rats. Biol. Psychiatry 2010, 67, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Fregni, F.; Boggio, P.S.; Nitsche, M.A.; Marcolin, M.A.; Rigonatti, S.P.; Pascual-Leone, A. Treatment of Major Depression with Transcranial Direct Current Stimulation. Bipolar Disorders 2006, 8, 203–204. [Google Scholar] [CrossRef] [PubMed]
- Kito, S.; Fujita, K.; Koga, Y. Regional cerebral blood flow changes after low-frequency transcranial magnetic stimulation of the right dorsolateral prefrontal cortex in treatment-resistant depression. Neuropsychobiology 2008, 58, 29–36. [Google Scholar] [CrossRef]
- Voineskos, D.; Blumberger, D.M.; Zomorrodi, R.; Rogasch, N.C.; Farzan, F.; Foussias, G.; Rajji, T.K.; Daskalakis, Z.J. Altered transcranial magnetic stimulation–electroencephalographic markers of inhibition and excitation in the dorsolateral prefrontal cortex in major depressive disorder. Biol. Psychiatry 2019, 85, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Brennan, S.; McLoughlin, D.M.; O’Connell, R.; Bogue, J.; O’Connor, S.; McHugh, C.; Glennon, M. Anodal transcranial direct current stimulation of the left dorsolateral prefrontal cortex enhances emotion recognition in depressed patients and controls. J. Clin. Exp. Neuropsychol. 2017, 39, 384–395. [Google Scholar] [CrossRef] [PubMed]
- Randver, R. Repetitive transcranial magnetic stimulation of the dorsolateral prefrontal cortex to alleviate depression and cognitive impairment associated with Parkinson’s disease: A review and clinical implications. J. Neurol. Sci. 2018, 393, 88–99. [Google Scholar] [CrossRef] [PubMed]
- Philip, N.S.; Barredo, J.; van’t Wout-Frank, M.; Tyrka, A.R.; Price, L.H.; Carpenter, L.L. Network mechanisms of clinical response to transcranial magnetic stimulation in posttraumatic stress disorder and major depressive disorder. Biol. Psychiatry 2018, 83, 263–272. [Google Scholar] [CrossRef]
- Baeken, C.; Marinazzo, D.; Everaert, H.; Wu, G.-R.; Van Hove, C.; Audenaert, K.; Goethals, I.; De Vos, F.; Peremans, K.; De Raedt, R. The impact of accelerated HF-rTMS on the subgenual anterior cingulate cortex in refractory unipolar major depression: Insights from 18FDG PET brain imaging. Brain Stimul. 2015, 8, 808–815. [Google Scholar] [CrossRef]
- Boes, A.D.; Uitermarkt, B.D.; Albazron, F.M.; Lan, M.J.; Liston, C.; Pascual-Leone, A.; Dubin, M.J.; Fox, M.D. Rostral anterior cingulate cortex is a structural correlate of repetitive TMS treatment response in depression. Brain Stimul. 2018, 11, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Berlim, M.T.; McGirr, A.; Van den Eynde, F.; Fleck, M.P.; Giacobbe, P. Effectiveness and acceptability of deep brain stimulation (DBS) of the subgenual cingulate cortex for treatment-resistant depression: A systematic review and exploratory meta-analysis. J. Affect. Disord. 2014, 159, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Johansen-Berg, H.; Gutman, D.; Behrens, T.; Matthews, P.; Rushworth, M.; Katz, E.; Lozano, A.; Mayberg, H. Anatomical connectivity of the subgenual cingulate region targeted with deep brain stimulation for treatment-resistant depression. Cereb. Cortex 2008, 18, 1374–1383. [Google Scholar] [CrossRef] [PubMed]
- Lozano, A.M.; Mayberg, H.S.; Giacobbe, P.; Hamani, C.; Craddock, R.C.; Kennedy, S.H. Subcallosal cingulate gyrus deep brain stimulation for treatment-resistant depression. Biol. Psychiatry 2008, 64, 461–467. [Google Scholar] [CrossRef]
- Riva-Posse, P.; Choi, K.S.; Holtzheimer, P.E.; McIntyre, C.C.; Gross, R.E.; Chaturvedi, A.; Crowell, A.L.; Garlow, S.J.; Rajendra, J.K.; Mayberg, H.S. Defining critical white matter pathways mediating successful subcallosal cingulate deep brain stimulation for treatment-resistant depression. Biol. Psychiatry 2014, 76, 963–969. [Google Scholar] [CrossRef]
- Choi, K.S.; Riva-Posse, P.; Gross, R.E.; Mayberg, H.S. Mapping the “depression switch” during intraoperative testing of subcallosal cingulate deep brain stimulation. JAMA Neurol. 2015, 72, 1252–1260. [Google Scholar] [CrossRef]
- Jing, Y.; Zhao, N.; Deng, X.P.; Feng, Z.J.; Huang, G.F.; Meng, M.; Zang, Y.F.; Wang, J. Pregenual or subgenual anterior cingulate cortex as potential effective region for brain stimulation of depression. Brain Behav. 2020, 10, e01591. [Google Scholar] [CrossRef]
- Bewernick, B.H.; Hurlemann, R.; Matusch, A.; Kayser, S.; Grubert, C.; Hadrysiewicz, B.; Axmacher, N.; Lemke, M.; Cooper-Mahkorn, D.; Cohen, M.X. Nucleus accumbens deep brain stimulation decreases ratings of depression and anxiety in treatment-resistant depression. Biol. Psychiatry 2010, 67, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Bewernick, B.H.; Kayser, S.; Sturm, V.; Schlaepfer, T.E. Long-term effects of nucleus accumbens deep brain stimulation in treatment-resistant depression: Evidence for sustained efficacy. Neuropsychopharmacology 2012, 37, 1975–1985. [Google Scholar] [CrossRef]
- Grubert, C.; Hurlemann, R.; Bewernick, B.H.; Kayser, S.; Hadrysiewicz, B.; Axmacher, N.; Sturm, V.; Schlaepfer, T.E. Neuropsychological safety of nucleus accumbens deep brain stimulation for major depression: Effects of 12-month stimulation. World J. Biol. Psychiatry 2011, 12, 516–527. [Google Scholar] [CrossRef]
- Falowski, S.M.; Sharan, A.; Reyes, B.A.; Sikkema, C.; Szot, P.; Van Bockstaele, E.J. An evaluation of neuroplasticity and behavior after deep brain stimulation of the nucleus accumbens in an animal model of depression. Neurosurgery 2011, 69, 1281–1290. [Google Scholar] [CrossRef]
- Nauczyciel, C.; Robic, S.; Dondaine, T.; Verin, M.; Robert, G.; Drapier, D.; Naudet, F.; Millet, B. The nucleus accumbens: A target for deep brain stimulation in resistant major depressive disorder. J. Mol. Psychiatry 2013, 1, 17. [Google Scholar] [CrossRef]
- Tronnier, V.M.; Rasche, D.; Thorns, V.; Alvarez-Fischer, D.; Münte, T.F.; Zurowski, B. Massive weight loss following deep brain stimulation of the nucleus accumbens in a depressed woman. Neurocase 2018, 24, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, F.; Velasco, F.; Salin-Pascual, R.; Hernández, J.A.; Velasco, M.; Criales, J.L.; Nicolini, H. A patient with a resistant major depression disorder treated with deep brain stimulation in the inferior thalamic peduncle. Neurosurgery 2005, 57, 585–593. [Google Scholar] [CrossRef]
- Jiménez, F.; Nicolini, H.; Lozano, A.M.; Piedimonte, F.; Salín, R.; Velasco, F. Electrical stimulation of the inferior thalamic peduncle in the treatment of major depression and obsessive compulsive disorders. World Neurosurg. 2013, 80, S30-e17.e25. [Google Scholar]
- Velasco, M.; Velasco, F.; Jiménez, F.; Carrillo-Ruiz, J.D.; Velasco, A.L.; Salín-Pascual, R. Electrocortical and behavioral responses elicited by acute electrical stimulation of inferior thalamic peduncle and nucleus reticularis thalami in a patient with major depression disorder. Clin. Neurophysiol. 2006, 117, 320–327. [Google Scholar] [CrossRef]
- Andy, O.J.; Jurko, F. Thalamic stimulation effects on reactive depression. Stereotact. Funct. Neurosurg. 1987, 50, 324–329. [Google Scholar] [CrossRef]
- Magdaleno-Madrigal, V.M.; Pantoja-Jiménez, C.R.; Bazaldúa, A.; Fernández-Mas, R.; Almazán-Alvarado, S.; Bolaños-Alejos, F.; Ortíz-López, L.; Ramírez-Rodriguez, G.B. Acute deep brain stimulation in the thalamic reticular nucleus protects against acute stress and modulates initial events of adult hippocampal neurogenesis. Behav. Brain Res. 2016, 314, 65–76. [Google Scholar] [CrossRef]
- Malone, D.A., Jr.; Dougherty, D.D.; Rezai, A.R.; Carpenter, L.L.; Friehs, G.M.; Eskandar, E.N.; Rauch, S.L.; Rasmussen, S.A.; Machado, A.G.; Kubu, C.S. Deep brain stimulation of the ventral capsule/ventral striatum for treatment-resistant depression. Biol. Psychiatry 2009, 65, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Dougherty, D.D.; Rezai, A.R.; Carpenter, L.L.; Howland, R.H.; Bhati, M.T.; O’Reardon, J.P.; Eskandar, E.N.; Baltuch, G.H.; Machado, A.D.; Kondziolka, D. A randomized sham-controlled trial of deep brain stimulation of the ventral capsule/ventral striatum for chronic treatment-resistant depression. Biol. Psychiatry 2015, 78, 240–248. [Google Scholar] [CrossRef]
- Pogarell, O.; Koch, W.; Pöpperl, G.; Tatsch, K.; Jakob, F.; Zwanzger, P.; Mulert, C.; Rupprecht, R.; Möller, H.-J.; Hegerl, U. Striatal dopamine release after prefrontal repetitive transcranial magnetic stimulation in major depression: Preliminary results of a dynamic [123I] IBZM SPECT study. J. Psychiatr. Res. 2006, 40, 307–314. [Google Scholar] [CrossRef]
- Kubu, C.S.; Brelje, T.; Butters, M.A.; Deckersbach, T.; Malloy, P.; Moberg, P.; Tröster, A.I.; Williamson, E.; Baltuch, G.H.; Bhati, M.T. Cognitive outcome after ventral capsule/ventral striatum stimulation for treatment-resistant major depression. J. Neurol. Neurosurg. Psychiatry 2017, 88, 262–265. [Google Scholar] [CrossRef]
- Kubu, C.S.; Malone, D.A.; Chelune, G.; Malloy, P.; Rezai, A.R.; Frazier, T.; Machado, A.; Rasmussen, S.; Friehs, G.; Greenberg, B.D. Neuropsychological outcome after deep brain stimulation in the ventral capsule/ventral striatum for highly refractory obsessive-compulsive disorder or major depression. Stereotact. Funct. Neurosurg. 2013, 91, 374–378. [Google Scholar] [CrossRef] [PubMed]
- Lozano, A.M.; Lipsman, N. Probing and regulating dysfunctional circuits using deep brain stimulation. Neuron 2013, 77, 406–424. [Google Scholar] [CrossRef]
- Kjelstrup, K.G.; Tuvnes, F.A.; Steffenach, H.-A.; Murison, R.; Moser, E.I.; Moser, M.-B. Reduced fear expression after lesions of the ventral hippocampus. Proc. Natl. Acad. Sci. USA 2002, 99, 10825–10830. [Google Scholar] [CrossRef] [PubMed]
- Fanselow, M.S.; Dong, H.-W. Are the dorsal and ventral hippocampus functionally distinct structures? Neuron 2010, 65, 7–19. [Google Scholar] [CrossRef]
- Stone, E.A.; Lin, Y. An anti-immobility effect of exogenous corticosterone in mice. Eur. J. Pharmacol. 2008, 580, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Murray, F.; Smith, D.W.; Hutson, P.H. Chronic low dose corticosterone exposure decreased hippocampal cell proliferation, volume and induced anxiety and depression like behaviours in mice. Eur. J. Pharmacol. 2008, 583, 115–127. [Google Scholar] [CrossRef]
- Casanova, E.; Fehsenfeld, S.; Mantamadiotis, T.; Lemberger, T.; Greiner, E.; Stewart, A.; Schütz, G. A CamKIIα iCre BAC allows brain-specific gene inactivation. Genesis 2001, 31, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, V.; Han, M.-H.; Graham, D.L.; Berton, O.; Renthal, W.; Russo, S.J.; LaPlant, Q.; Graham, A.; Lutter, M.; Lagace, D.C. Molecular adaptations underlying susceptibility and resistance to social defeat in brain reward regions. Cell 2007, 131, 391–404. [Google Scholar] [CrossRef] [PubMed]
- Toni, N.; Schinder, A.F. Maturation and functional integration of new granule cells into the adult hippocampus. Cold Spring Harb. Perspect. Biol. 2016, 8, a018903. [Google Scholar] [CrossRef] [PubMed]
- Cameron, H.A.; McEwen, B.S.; Gould, E. Regulation of adult neurogenesis by excitatory input and NMDA receptor activation in the dentate gyrus. J. Neurosci. 1995, 15, 4687–4692. [Google Scholar] [CrossRef]
- Schlett, K. Glutamate as a modulator of embryonic and adult neurogenesis. Curr. Top. Med. Chem. 2006, 6, 949–960. [Google Scholar] [CrossRef]
- Melzer, S.; Michael, M.; Caputi, A.; Eliava, M.; Fuchs, E.C.; Whittington, M.A.; Monyer, H. Long-range–projecting GABAergic neurons modulate inhibition in hippocampus and entorhinal cortex. Science 2012, 335, 1506–1510. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-T.; Kao, M.-H.; Hou, W.-H.; Wei, Y.-T.; Chen, C.-L.; Lien, C.-C. Causal evidence for the role of specific GABAergic interneuron types in entorhinal recruitment of dentate granule cells. Sci. Rep. 2016, 6, 36885. [Google Scholar] [CrossRef] [PubMed]
- Colgin, L.L. Theta–gamma coupling in the entorhinal–hippocampal system. Curr. Opin. Neurobiol. 2015, 31, 45–50. [Google Scholar] [CrossRef]
- Reagh, Z.M.; Yassa, M.A. Object and spatial mnemonic interference differentially engage lateral and medial entorhinal cortex in humans. Proc. Natl. Acad. Sci. USA 2014, 111, E4264–E4273. [Google Scholar] [CrossRef] [PubMed]
- Luna, V.M.; Anacker, C.; Burghardt, N.S.; Khandaker, H.; Andreu, V.; Millette, A.; Leary, P.; Ravenelle, R.; Jimenez, J.C.; Mastrodonato, A. Adult-born hippocampal neurons bidirectionally modulate entorhinal inputs into the dentate gyrus. Science 2019, 364, 578–583. [Google Scholar] [CrossRef] [PubMed]
- Acsady, L.; Kali, S. Models, structure, function: The transformation of cortical signals in the dentate gyrus. Prog. Brain Res. 2007, 163, 577–599. [Google Scholar] [PubMed]
- Liu, A.; Jain, N.; Vyas, A.; Lim, L.W. Ventromedial prefrontal cortex stimulation enhances memory and hippocampal neurogenesis in the middle-aged rats. Elife 2015, 4, e04803. [Google Scholar] [CrossRef] [PubMed]
- Kirby, E.D.; Friedman, A.R.; Covarrubias, D.; Ying, C.; Sun, W.G.; Goosens, K.A.; Sapolsky, R.M.; Kaufer, D. Basolateral amygdala regulation of adult hippocampal neurogenesis and fear-related activation of newborn neurons. Mol. Psychiatry 2012, 17, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Chamaa, F.; Sweidan, W.; Nahas, Z.; Saade, N.; Abou-Kheir, W. Thalamic stimulation in awake rats induces neurogenesis in the hippocampal formation. Brain Stimul. 2016, 9, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Winter, C.; Bregman, T.; Voget, M.; Raymond, R.; Hadar, R.; Nobrega, J.N.; Hamani, C. Acute high frequency stimulation of the prefrontal cortex or nucleus accumbens does not increase hippocampal neurogenesis in rats. J. Psychiatr. Res. 2015, 68, 27–29. [Google Scholar] [CrossRef]
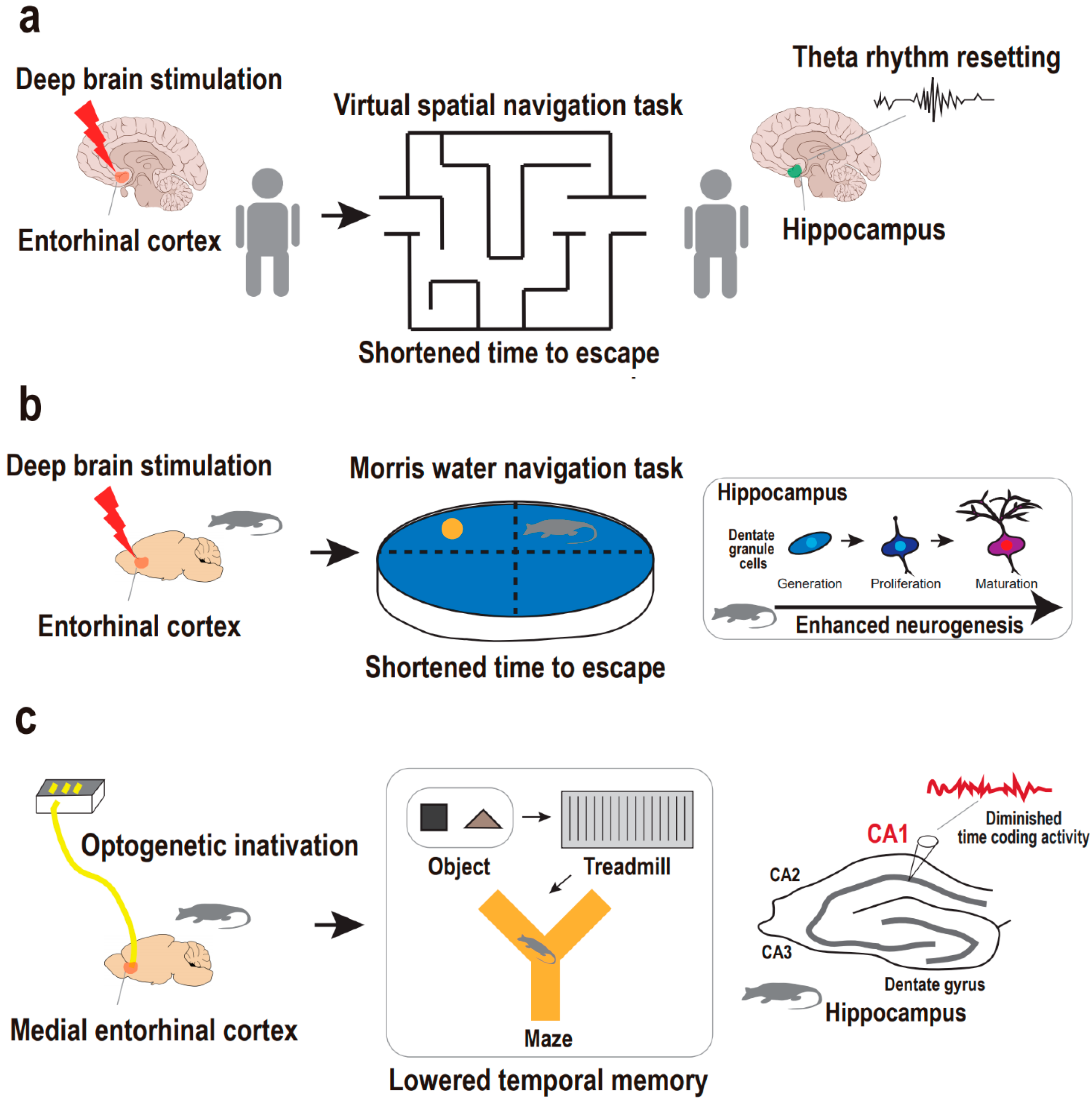
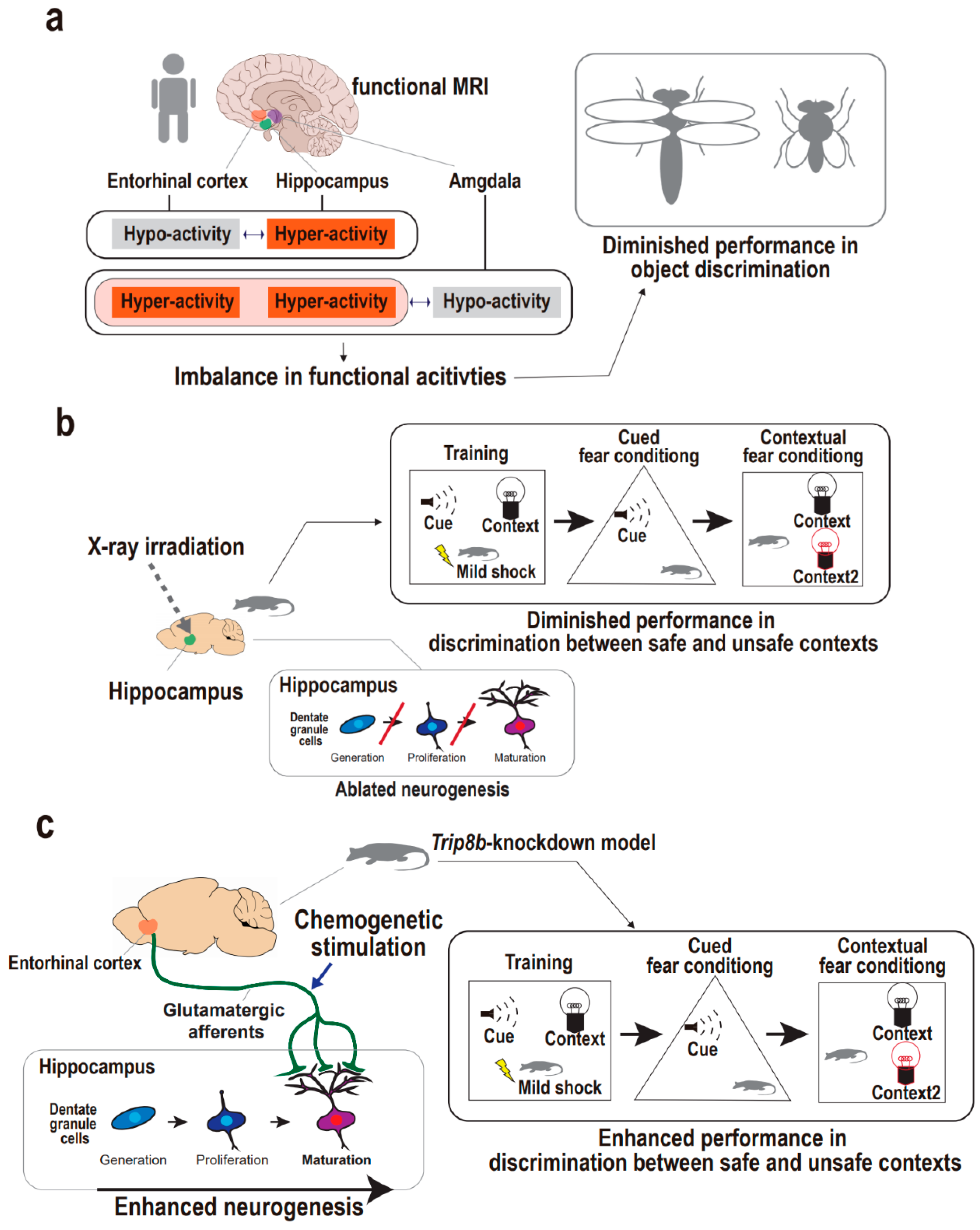
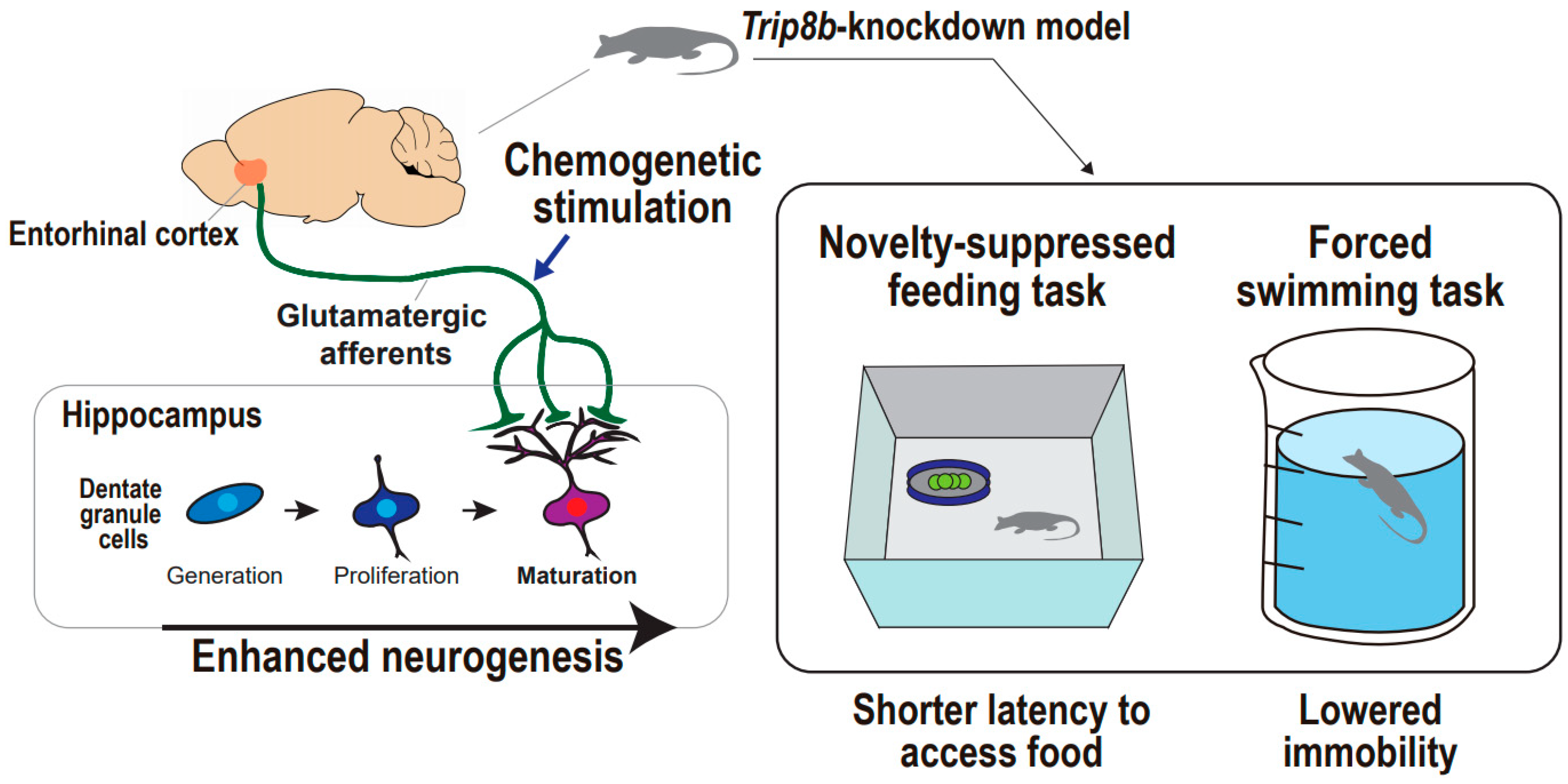
| Stimulation Approach | Subject | Depression-Related Phenotype | Target of Stimulation | Consequence in Hippocampus | Behavioral Task | Work * |
|---|---|---|---|---|---|---|
| DBS | Human | Spatial memory | Entorhinal cortex | Hippocampal theta rhythm, resetted | Spatial navigation test | 13 |
| DBS | Mouse | Spatial memory | Entorhinal cortex | Dentate gyrus neurogenesis, enhanced | Morris water maze test | 12 |
| DBS | Rat | Spatial memory | Entorhinal cortex | Dentate gyrus neurogenesis, enhanced | Morris water maze test | 15 |
| Optogenetics | Transgenic rat | Temporal memory | Medial entorhinal cortex | Hippocampal CA1 temporal coding activity, enhanced | Object–treadmill–maze test | 48 |
| Chemogenetics | Transgenic mouse | Pattern separation | Entorhinal glutamatergic afferents | Dentate gyrus neurogenesis, enhanced | Fear-context conditioning test | 16 |
| Chemogenetics | Transgenic mouse | Depressive-like behaviors | Entorhinal glutamatergic afferents | Dentate gyrus neurogenesis, enhanced | Forced swimming test Novelty-suppressed feeding test | 16 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, I.B.; Park, S.-C. Neural Circuitry–Neurogenesis Coupling Model of Depression. Int. J. Mol. Sci. 2021, 22, 2468. https://doi.org/10.3390/ijms22052468
Kim IB, Park S-C. Neural Circuitry–Neurogenesis Coupling Model of Depression. International Journal of Molecular Sciences. 2021; 22(5):2468. https://doi.org/10.3390/ijms22052468
Chicago/Turabian StyleKim, Il Bin, and Seon-Cheol Park. 2021. "Neural Circuitry–Neurogenesis Coupling Model of Depression" International Journal of Molecular Sciences 22, no. 5: 2468. https://doi.org/10.3390/ijms22052468
APA StyleKim, I. B., & Park, S.-C. (2021). Neural Circuitry–Neurogenesis Coupling Model of Depression. International Journal of Molecular Sciences, 22(5), 2468. https://doi.org/10.3390/ijms22052468







