Specific Mycoparasite-Fusarium Graminearum Molecular Signatures in Germinating Seeds Disabled Fusarium Head Blight Pathogen’s Infection
Abstract
1. Introduction
2. Results and Discussion
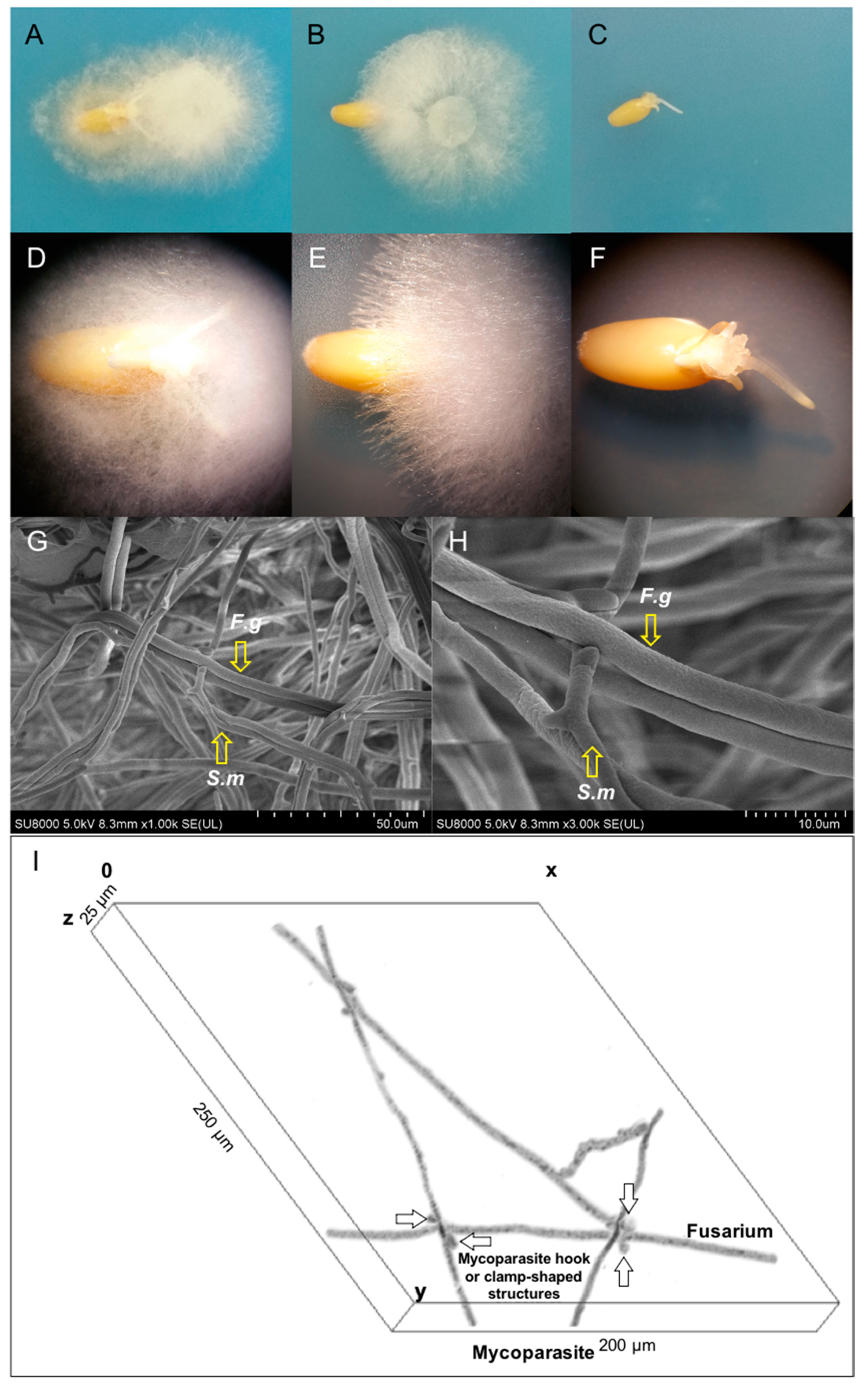
2.1. In Vitro Wheat Seed Germination and Growth
2.2. FTIR Spectra
2.2.1. Implication of Biochemical Changes in the Root Composition of Wheat Seed
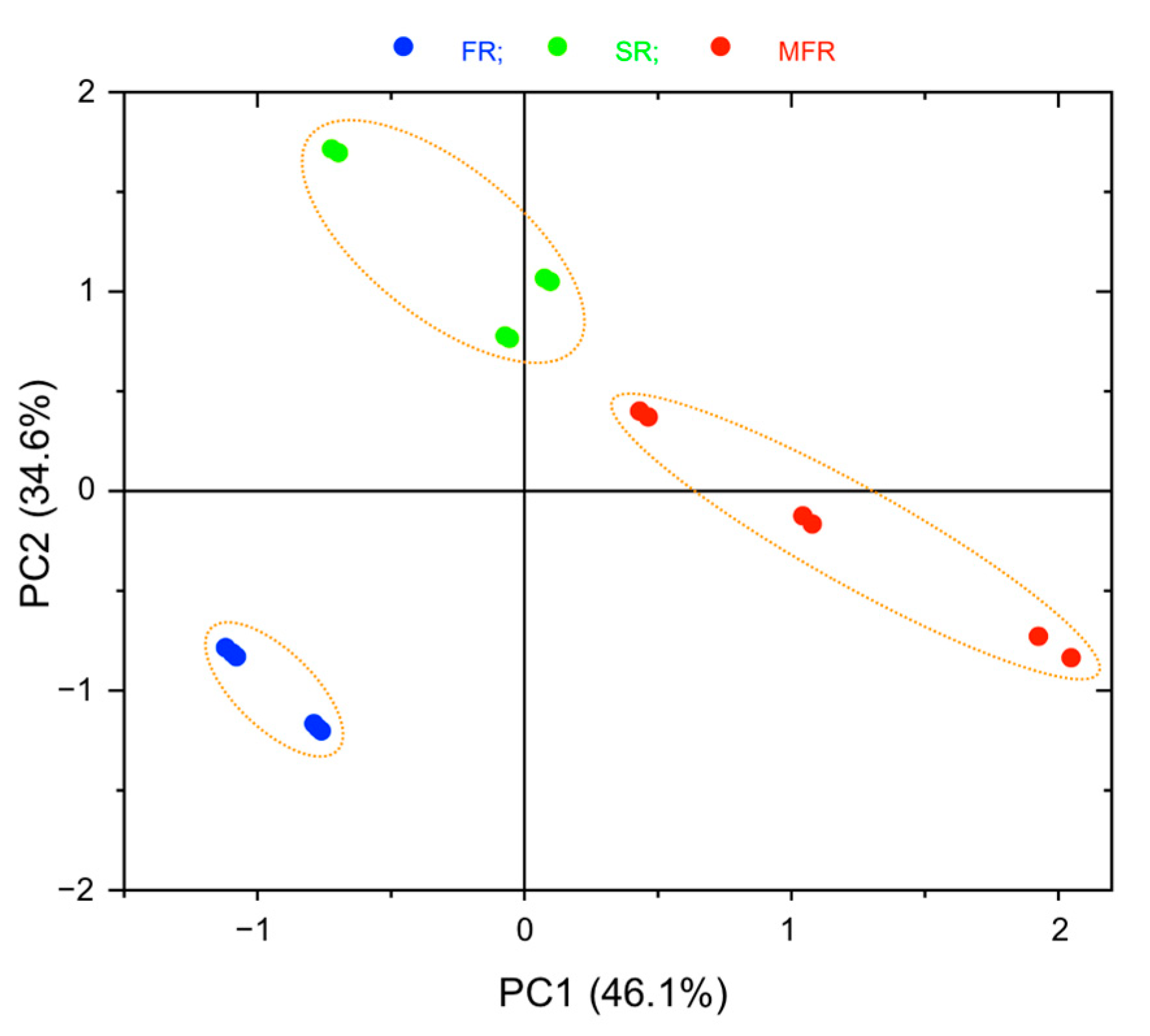
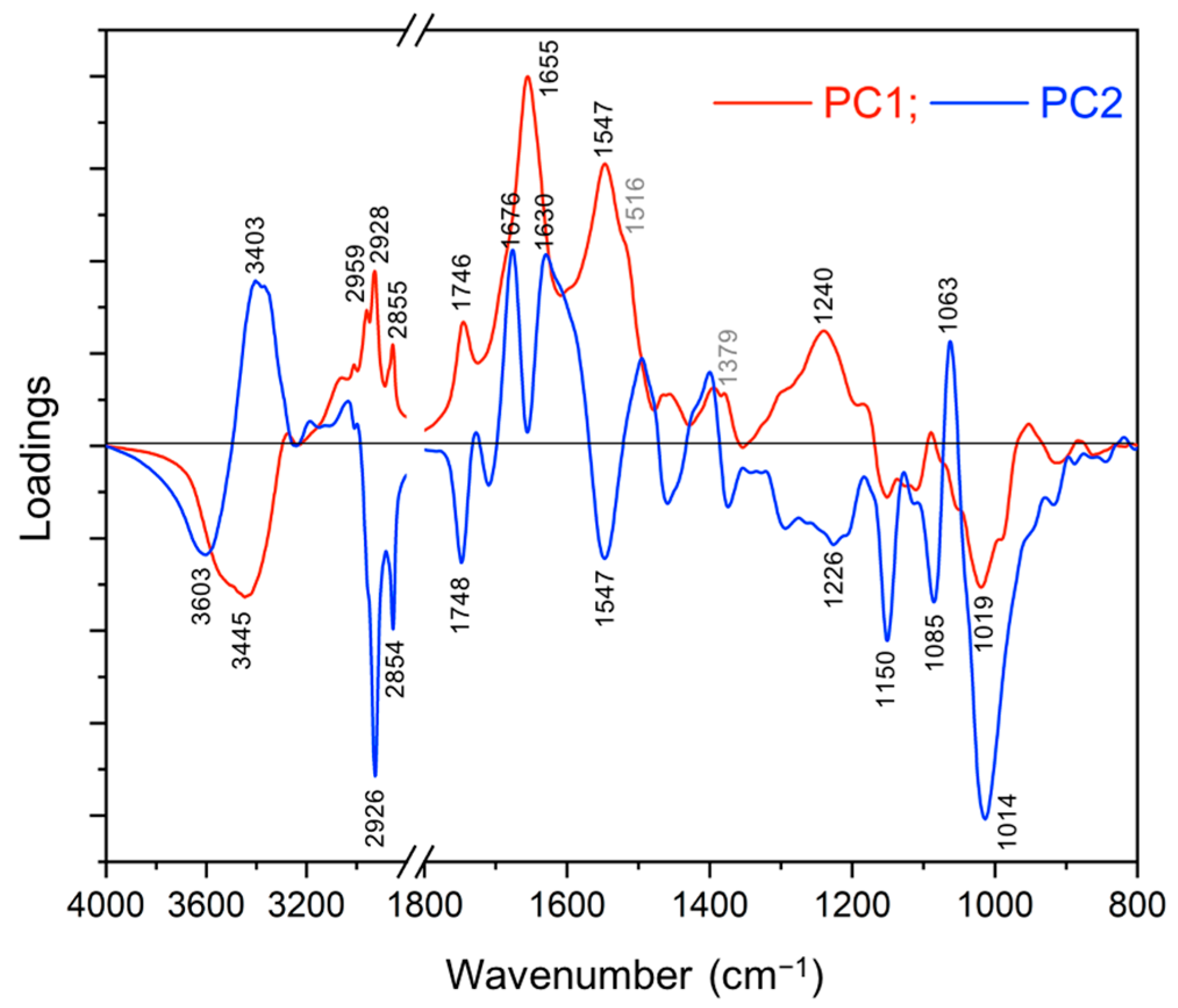
2.2.2. Principal Component Analysis (PCA)
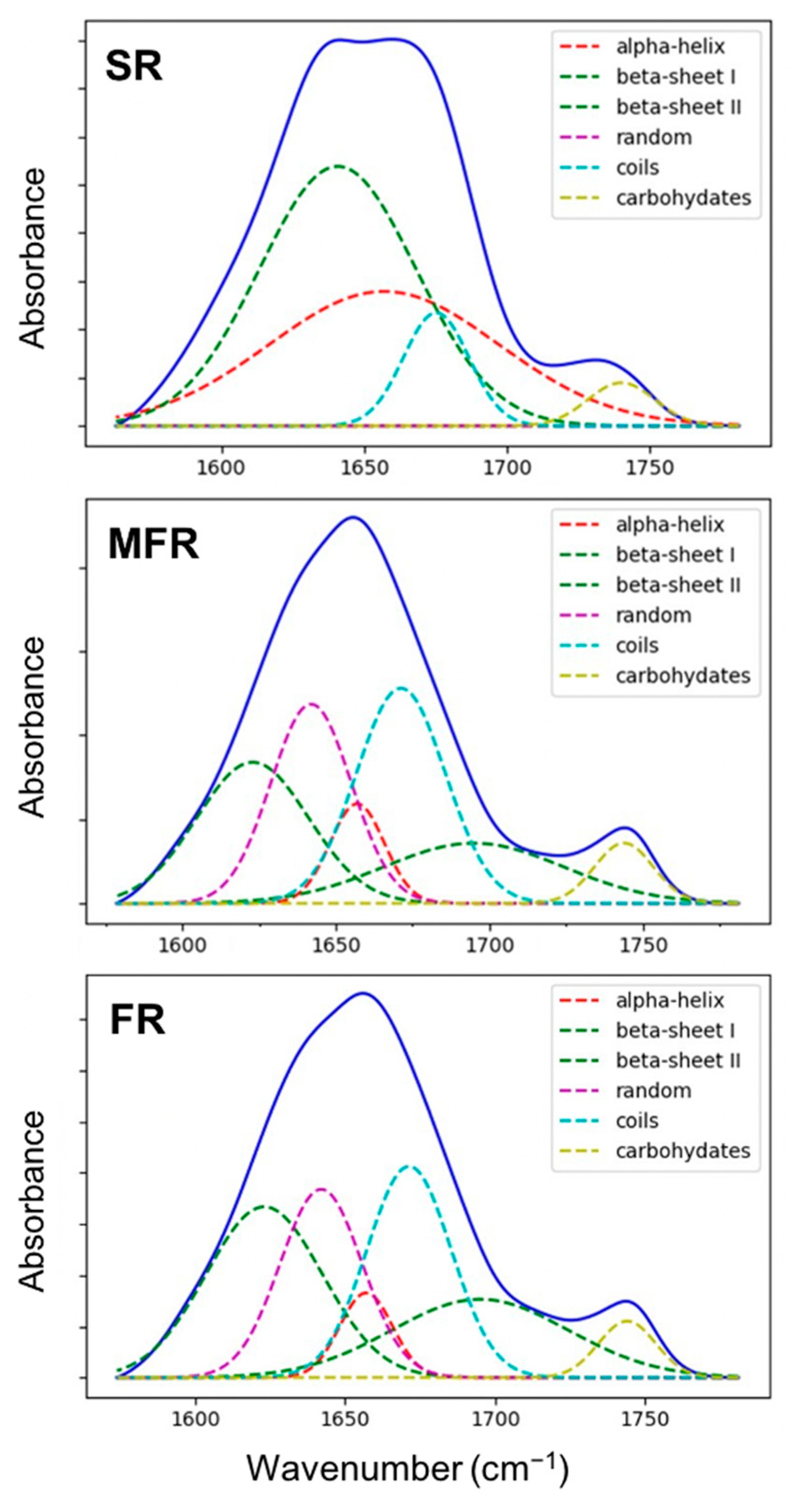
2.2.3. Estimation of Protein Secondary Structures in Tripartite System
3. Materials and Methods
3.1. Plant and Fungal Materials
3.2. In Vitro Wheat Seed Germination Assay
3.3. Scanning Electron and Confocal Laser Scanning Microscopy
3.4. Preparation of the Root Samples for FTIR Spectroscopy
3.5. FTIR Data Analysis
3.6. Protein Secondary Structure Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vujanovic, V.; Islam, M.N.; Daida, P. Transgenerational role of seed mycobiome—An endosymbiotic fungal composition as a prerequisite to stress resilience and adaptive phenotypes in Triticum. Sci. Rep. 2019, 9, 18483. [Google Scholar] [CrossRef]
- Bruinsma, J. World Agriculture: Towards 2015/2030 Summary Report; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2002. [Google Scholar]
- Vujanovic, V.; Germida, J.J. Seed endosymbiosis: A vital relationship in providing prenatal care to plants. Can. J. Plant Pathol. 2017, 97, 972–981. [Google Scholar] [CrossRef]
- Osborne, L.E.; Stein, J.M. Epidemiology of Fusarium head blight on small-grain cereals. Int. J. Food Microbiol. 2007, 119, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Schöneberg, A.; Musa, T.; Voegele, R.T.; Vogelgsang, S. The potential of antagonistic fungi for control of Fusarium graminearum and Fusarium crookwellense varies depending on the experimental approach. J. Appl. Microbiol. 2015, 118, 1165–1179. [Google Scholar] [CrossRef]
- Karlsson, M.; Durling, M.B.; Choi, J.; Kosawang, C.; Lackner, G.; Tzelepis, G.D.; Nygren, K.; Dubey, M.K.; Kamou, N.; Levasseur, A.; et al. Insights on the evolution of mycoparasitism from the genome of Clonostachys rosea. Genome Biol. Evol. 2015, 7, 465–480. [Google Scholar] [CrossRef]
- Vieira, P.M.; Coelho, A.S.; Steindorff, A.S.; de Siqueira, S.J.; Silva Rdo, N.; Ulhoa, C.J. Identification of differentially expressed genes from Trichoderma harzianum during growth on cell wall of Fusarium solani as a tool for biotechnological application. BMC Genomics 2013, 14, 177. [Google Scholar] [CrossRef] [PubMed]
- Nygren, K.; Dubey, M.; Zapparata, A.; Iqbal, M.; Tzelepis, G.D.; Durling, M.B.; Jensen, D.F.; Karlsson, M. The mycoparasitic fungus Clonostachys rosea responds with both common and specific gene expression during interspecific interactions with fungal prey. Evol. Appl. 2018, 11, 931–949. [Google Scholar] [CrossRef]
- Kim, S.H.; Vujanovic, V. Biodegradation and biodetoxification of Fusarium mycotoxins by Sphaerodes mycoparasitica. AMB Express 2017, 7, 145. [Google Scholar] [CrossRef]
- Vujanovic, V.; Korber, D.R.; Vujanovic, S.; Vujanovic, J.; Jabaji, S. Scientific prospects for cannabis-microbiome research to ensure quality and safety of products. Microorganisms 2020, 8, 290. [Google Scholar] [CrossRef]
- Pal, K.K.; Gardener, B.M. Biological control of plant pathogens. Plant Health Instr. 2006, 2, 1117–1142. [Google Scholar] [CrossRef]
- Kim, S.H.; Vujanovic, V. Relationship between mycoparasites lifestyles and biocontrol behaviors against Fusarium spp. and mycotoxins production. Appl. Microbiol. Biotechnol. 2016, 100, 5257–5272. [Google Scholar] [CrossRef]
- Vujanovic, V.; Kim, S.H. Adaptability of mitosporic stage in Sphaerodes mycoparasitica towards its mycoparasitic-polyphagous lifestyle. Mycologia 2017, 109, 701–709. [Google Scholar] [CrossRef]
- Vujanovic, V.; Goh, Y.K. qPCR quantification of Sphaerodes mycoparasitica biotrophic mycoparasite interaction with Fusarium graminearum: In vitro and in planta assays. Arch. Microbiol. 2012. [Google Scholar] [CrossRef] [PubMed]
- Vujanovic, V.; Kim, S.H.; Lahlali, R.; Karunakaran, C. Spectroscopy and SEM imaging reveal endosymbiont-dependent components changes in germinating kernel through direct and indirect coleorhiza-fungus interactions under stress. Sci. Rep. 2019, 9, 1665. [Google Scholar] [CrossRef]
- Ding, L.; Xu, H.; Yi, H.; Yang, L.; Kong, Z.; Zhang, L.; Xue, S.; Jia, H.; Ma, Z. Resistance to hemi-biotrophic F. graminearum infection is associated with coordinated and ordered expression of diverse defense signaling pathways. PLoS ONE 2011, 6, e19008. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, L.; Atanasova-Penichon, V.; Chéreau, S.; Richard-Forget, F. Metabolomics to decipher the chemical defense of cereals against Fusarium graminearum and deoxynivalenol accumulation. Int. J. Mol. Sci. 2015, 16, 24839–24872. [Google Scholar] [CrossRef]
- Lahlali, R.; Karunakaran, C.; Wang, L.; Willick, I.; Schmidt, M.; Liu, X.; Borondics, F.; Forseille, L.; Fobert, P.R.; Tanino, K.; et al. Synchrotron based phase contrast X-ray imaging combined with FTIR spectroscopy reveals structural and biomolecular differences in spikelets play a significant role in resistance to Fusarium in wheat. BMC Plant Biol. 2015, 15, 24. [Google Scholar] [CrossRef]
- Wachowska, U.; Tańska, M.; Konopka, I. Variations in grain lipophilic phytochemicals, proteins and resistance to Fusarium spp. growth during grain storage as affected by biological plant protection with Aureobasidium pullulans (de Bary). Int. J. Food Microbiol. 2016, 227, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, B.; Kohler, A. Infrared spectroscopy of pollen identifies plant species and genus as well as environmental conditions. PLoS ONE 2014, 9, e95417. [Google Scholar] [CrossRef] [PubMed]
- Lahlali, R.; Jiang, Y.; Kumar, S.; Karunakaran, C.; Liu, X.; Borondics, F.; Hallin, E.; Bueckert, R. ATR-FTIR spectroscopy reveals involvement of lipids and proteins of intact pea pollen grains to heat stress tolerance. Front. Plant Sci. 2014, 5. [Google Scholar] [CrossRef]
- Jiang, Y.; Lahlali, R.; Karunakaran, C.; Kumar, S.; Davis, A.R.; Bueckert, R.A. Seed set, pollen morphology and pollen surface composition response to heat stress in field pea. Plant Cell Environ. 2015. [Google Scholar] [CrossRef]
- Lahlali, R.; Song, T.; Chu, M.; Karunakaran, C.; Yu, F.; Wei, Y.; Peng, G. Synchrotron-based spectroscopy and imaging reveal changes in the cell-wall composition of barley leaves in defence responses to Blumeria graminis f. sp. tritici. Can. J. Plant Pathol. 2019, 41, 457–467. [Google Scholar] [CrossRef]
- Lahlali, R.; Song, T.; Chu, M.; Yu, F.; Kumar, S.; Karunakaran, C.; Peng, G. Evaluating changes in cell-wall components associated with clubroot resistance using Fourier Transform Infrared Spectroscopy and RT-PCR. Int. J. Mol. Sci. 2017, 18, 2058. [Google Scholar] [CrossRef]
- Hardoim, P.R.; Guerra, R.; Costa, A.M.R.D.; Serrano, M.S.; Sánchez, M.E.; Coelho, A.C.; Stenlid, J. Temporal metabolic profiling of the Quercus suber–Phytophthora cinnamomi system by middle-infrared spectroscopy. For. Pathol. 2016, 46, 122–133. [Google Scholar] [CrossRef]
- Thumanu, K.; Sompong, M.; Phansak, P.; Nontapot, K.; Buensanteai, N. Use of infrared microspectroscopy to determine leaf biochemical composition of cassava in response to Bacillus subtilis CaSUT007. J. Plant Interact. 2015, 10, 270–279. [Google Scholar] [CrossRef]
- Lahlali, R.; Kumar, S.; Wang, L.; Forseille, L.; Sylvain, N.; Korbas, M.; Muir, D.; Swerhone, G.; Lawrence, J.R.; Fobert, P.R.; et al. Cell wall biomolecular composition plays a potential role in the host type II resistance to Fusarium Head Blight in wheat. Front. Microbiol. 2016, 7, 910. [Google Scholar] [CrossRef]
- Kačuráková, M.; Wellner, N.; Ebringerová, A.; Hromádková, Z.; Wilson, R.H.; Belton, P.S. Characterisation of xylan-type polysaccharides and associated cell wall components by FT-IR and FT-Raman spectroscopies. Food Hydrocoll. 1999, 13, 35–41. [Google Scholar] [CrossRef]
- Lasch, P.; Naumann, D. Infrared Spectroscopy in Microbiology. In Encyclopedia of Analytical Chemistry; John Wiley & Sons: Hoboken, NJ, USA, 2015; pp. 1–32. [Google Scholar] [CrossRef]
- Perez-Guaita, D.; Kochan, K.; Martin, M.; Andrew, D.W.; Heraud, P.; Richards, J.S.; Wood, B.R. Multimodal vibrational imaging of cells. Vib. Spectrosc. 2017, 91, 46–58. [Google Scholar] [CrossRef]
- Durak, T.; Depciuch, J. Effect of plant sample preparation and measuring methods on ATR-FTIR spectra results. Environ. Exp. Bot. 2020, 169, 103915. [Google Scholar] [CrossRef]
- Mallamace, F.; Corsaro, C.; Mallamace, D.; Vasi, S.; Vasi, C.; Dugo, G. The role of water in protein’s behavior: The two dynamical crossovers studied by NMR and FTIR techniques. Comput. Struct. Biotechnol. J. 2015, 13, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Arrondo, J.L.; Goni, F.M. Infrared studies of protein-induced perturbation of lipids in lipoproteins and membranes. Chem. Phys. Lipids 1998, 96, 53–68. [Google Scholar] [CrossRef]
- Legner, N.; Meinen, C.; Rauber, R. Root Differentiation of agricultural plant cultivars and proveniences using FTIR Spectroscopy. Front. Plant Sci. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhu, J.; Huang, R.; Yang, Y. Investigation of cell wall composition related to stem lodging resistance in wheat (Triticum aestivum L.) by FTIR spectroscopy. Plant Signal. Behav. 2012, 7, 856–863. [Google Scholar] [CrossRef]
- Nguema-Ona, E.; Vicré-Gibouin, M.; Gotté, M.; Plancot, B.; Lerouge, P.; Bardor, M.; Driouich, A. Cell wall O-glycoproteins and N-glycoproteins: Aspects of biosynthesis and function. Front. Plant Sci. 2014, 5. [Google Scholar] [CrossRef]
- Barsberg, S.T.; Lee, Y.I.; Rasmussen, H.N. Development of C-lignin with G/S-lignin and lipids in orchid seed coats—An unexpected diversity exposed by ATR-FT-IR spectroscopy. Seed Sci. Res. 2018, 28, 41–51. [Google Scholar] [CrossRef]
- Sain, M.; Panthapulakkal, S. Bioprocess preparation of wheat straw fibers and their characterization. Ind. Crops Prod. 2006, 23, 1–8. [Google Scholar] [CrossRef]
- Siebers, M.; Brands, M.; Wewer, V.; Duan, Y.; Holzl, G.; Dormann, P. Lipids in plant-microbe interactions. Biochimica et Biophysica Acta 2016, 1861, 1379–1395. [Google Scholar] [CrossRef] [PubMed]
- Gronnier, J.; Germain, V.; Gouguet, P.; Cacas, J.-L.; Mongrand, S. GIPC: Glycosyl Inositol Phospho Ceramides, the major sphingolipids on earth. Plant Signal. Behav. 2016, 11, e1152438. [Google Scholar] [CrossRef]
- Mamode Cassim, A.; Gouguet, P.; Gronnier, J.; Laurent, N.; Germain, V.; Grison, M.; Boutté, Y.; Gerbeau-Pissot, P.; Simon-Plas, F.; Mongrand, S. Plant lipids: Key players of plasma membrane organization and function. Prog. Lipid Res. 2019, 73, 1–27. [Google Scholar] [CrossRef]
- Bagińska, K.; Makowska, J.; Wiczk, W.; Kasprzykowski, F.; Chmurzyński, L. Conformational studies of alanine-rich peptide using CD and FTIR spectroscopy. J. Pept. Sci. 2008, 14, 283–289. [Google Scholar] [CrossRef]
- Severcan, F.; Haris, P.I.; Press, I. Vibrational Spectroscopy in Diagnosis and Screening; IOS Press: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Abbas, O.; Compère, G.; Larondelle, Y.; Pompeu, D.; Rogez, H.; Baeten, V. Phenolic compound explorer: A mid-infrared spectroscopy database. Vib. Spectrosc. 2017, 92, 111–118. [Google Scholar] [CrossRef]
- El Semary, N.A. The characterisation of bioactive compounds from an Egyptian Leptolyngbya sp. strain. Ann. Microbiol. 2012, 62, 55–59. [Google Scholar] [CrossRef]
- Jensen, J.S.; Egebo, M.; Meyer, A.S. Identification of spectral regions for the quantification of red wine tannins with Fourier Transform Mid-Infrared Spectroscopy. J. Agric. Food Chem. 2008, 56, 3493–3499. [Google Scholar] [CrossRef]
- Wolkers, W.F.; Hoekstra, F.A. Aging of dry desiccation-tolerant pollen does not affect protein secondary structure. Plant Physiol. 1995, 109, 907–915. [Google Scholar] [CrossRef]
- Apaliya, M.T.; Zhang, H.; Zheng, X.; Yang, Q.; Mahunu, G.K.; Kwaw, E. Exogenous trehalose enhanced the biocontrol efficacy of Hanseniaspora uvarum against grape berry rots caused by Aspergillus tubingensis and Penicillium commune. J. Sci. Food Agric. 2018, 98, 4665–4672. [Google Scholar] [CrossRef] [PubMed]
- Imamura, K.; Ohyama, K.-I.; Yokoyama, T.; Maruyama, Y.; Kazuhiro, N. Temperature scanning FTIR analysis of secondary structures of proteins embedded in amorphous sugar matrix. J. Pharm. Sci. 2009, 98, 3088–3098. [Google Scholar] [CrossRef]
- Belton, P.S.; Gil, A.M. IR and Raman spectroscopic studies of the interaction of trehalose with hen egg white lysozyme. Biopolymers 1994, 34, 957–961. [Google Scholar] [CrossRef]
- Stankovic, M. Innovative Platform Technologies for Stabilization and Controlled Release of Proteins from Polymer Depots. Ph.D. Thesis, University of Groningen, Groningen, The Netherlands, 2014. [Google Scholar]
- Mouillon, J.-M.; Eriksson, S.K.; Harryson, P. Mimicking the plant cell interior under water stress by macromolecular crowding: Disordered dehydrin proteins are highly resistant to structural collapse. Plant Physiol. 2008, 148, 1925–1937. [Google Scholar] [CrossRef]
- Sowa, S.; Connor, K.F.; Towill, L.E. Temperature changes in lipid and protein structure measured by fourier transform infrared spectrophotometry in intact pollen grains. Plant Sci. 1991, 78, 1–9. [Google Scholar] [CrossRef]
- Abdellatif, L.; Bouzid, S.; Kaminskyj, S.; Vujanovic, V. Endophytic hyphal compartmentalization is required for successful symbiotic Ascomycota association with root cells. Mycol. Res. 2009, 113, 782–791. [Google Scholar] [CrossRef]
- Hubbard, M.; Germida, J.; Vujanovic, V. Fungal endophytes improve wheat seed germination under heat and drought stress. Botany 2012, 90, 137–149. [Google Scholar] [CrossRef]
- Clarke, J.M.; McLeod, J.G.; McCaig, T.N.; DePauw, R.M.; Knox, R.E.; Fernandez, M.R. AC Avonlea durum wheat. Can. J. Plant Pathol. 1998, 78, 621–623. [Google Scholar] [CrossRef]
- Vujanovic, V.; Goh, Y.K. Sphaerodes mycoparasitica sp. nov., a new biotrophic mycoparasite on Fusarium avenaceum, F. graminearum and F. oxysporum. Mycol. Res. 2009, 113, 1172–1180. [Google Scholar] [CrossRef] [PubMed]
- Goh, Y.K.; Vujanovic, V. Ascospore germination patterns revealed ascomycetous biotrophic mycoparasite specificity to Fusarium hosts. Botany 2010, 88, 1033–1043. [Google Scholar] [CrossRef]
- Vujanovic, V.; Goh, Y.K. Sphaerodes mycoparasitica biotrophic mycoparasite of 3-acetyldeoxynivalenol- and 15-acetyldeoxynivalenol-producing toxigenic Fusarium graminearum chemotypes. FEMS Microbiol. Lett. 2011, 316, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Fischer, E.R.; Hansen, B.T.; Nair, V.; Hoyt, F.H.; Dorward, D.W. Scanning Electron Microscopy. Curr. Protoc. Microbiol. 2012, CHAPTER, Unit2B.2. [Google Scholar] [CrossRef]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Toplak, M.; Birarda, G.; Read, S.; Sandt, C.; Rosendahl, S.M.; Vaccari, L.; Demšar, J.; Borondics, F. Infrared Orange: Connecting Hyperspectral Data with Machine Learning. Synchrotron Radiation News 2017, 30, 40–45. [Google Scholar] [CrossRef]

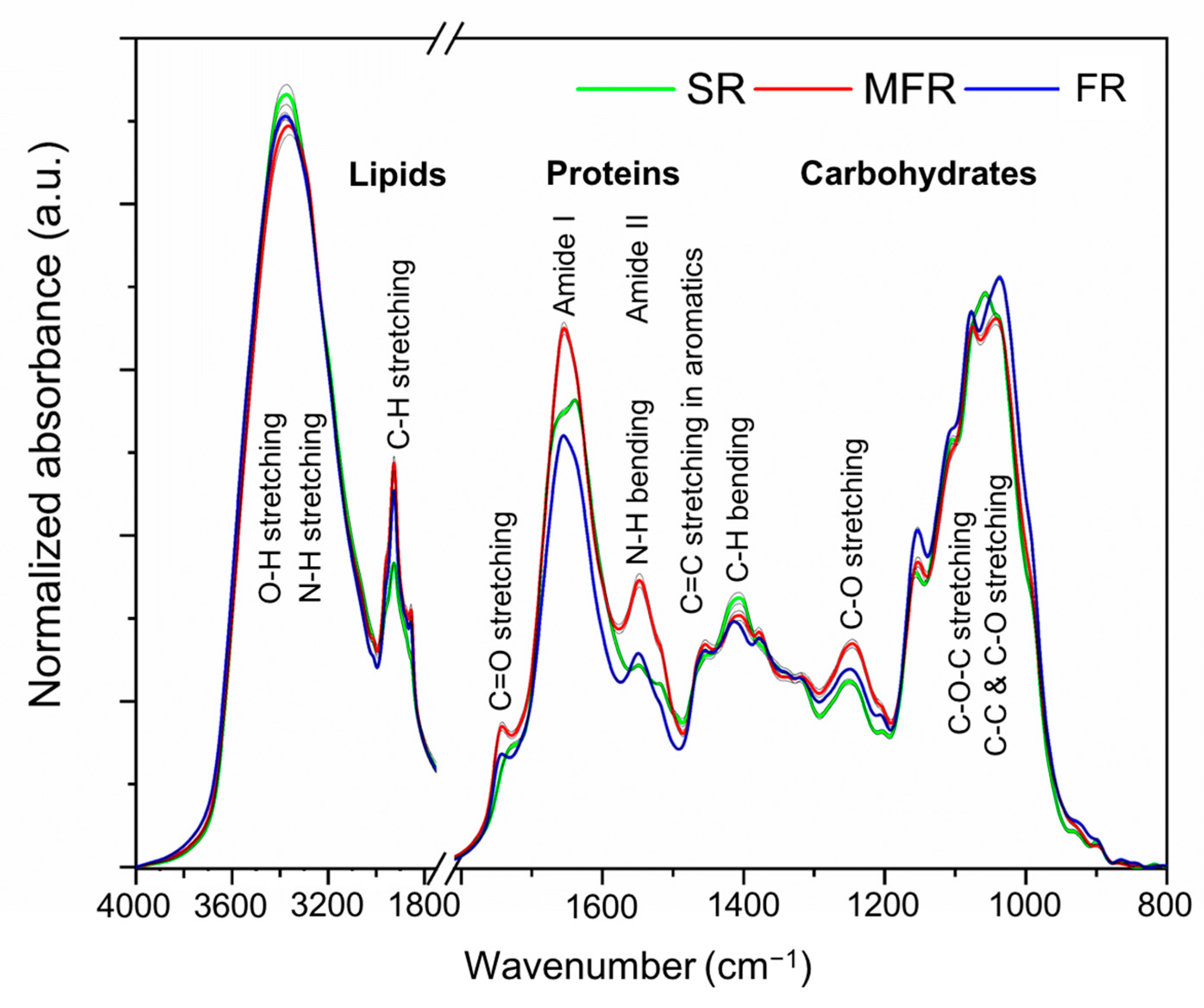
| Wavenumber (cm−1) | Vibration Modes of Functional Groups | Probable Components | References |
|---|---|---|---|
| 3430–3300 | O-H stretching of Alcohols and carboxylic acids | Carbohydrates (cellulose, hemicelluloses) and Glycoconjugates | [24,25] |
| N-H stretching of amide A | Proteins (amide A) | ||
| 2927–2924 2856–2853 | C-H stretching of asymmetric and symmetric CH2 | Lipids | [24,25] |
| 1744–1700 | C = O stretching of aldehyde, ketone, ester compounds, and carboxylic acid | Fatty acids, aliphatic and aromatic carbonyl compounds hemicelluloses, lignins, pectins | [24,25,26] |
| 1655–1638 | C = O stretching of -CO-NH | Proteins (amide Ⅰ) | [27] |
| 1549–1548 | N-H bending and C-N stretching | Proteins (amide Ⅱ) | [27] |
| 1520–1500 | C = C stretching of aromatic rings | Lignin related molecules; ferulic acid and aromatic ring containing compounds | [24,28,29] |
| 1456–1453 | C-H bending of CH2 and CH3 | Proteins and lipids | [30,31] |
| 1319–1318 | C-N stretching and N-H deformation or more complex vibrations | Proteins (amide Ⅲ) | [32] |
| 1250–1246 | C-O stretching Asymmetric P = O stretching (of PO2-) | Hemicellulose, Phospholipids and nucleic acids | [24,27,30,33] |
| 1157–1153 1077–1076 | Mainly C-O-C asymmetric stretching and C-O stretching Symmetric stretching of PO2- | Mainly Cellulose, Phospholipids and nucleic acids | [24,27,30,33] |
| 1058–1036 | C-O-C symmetric stretching and C-C, C-O stretching or C-OH bending | Mainly Cellulose, hemicellulose | [24,27,34,35] |
| Peak Position (cm−1) with Relative Identity | Integrated Absorption Peak | |||
|---|---|---|---|---|
| Wavenumber Range (cm−1) | SR | MFR | FR | |
| 3430–3300 OH stretching and Amide A | 3680–3000 | 19.58 ± 0.02 b | 19.18 ± 0.12 a | 19.54 ± 0.03 b |
| 2927–2924 2856–2853 Lipids | 3000–2800 | 2.73 ± 0.06 a | 3.11 ± 0.06 b | 2.96 ± 0.02 b |
| 1744–1743 Carbonyl | 1760–1720 | 0.23 ± 0.00 a | 0.30 ± 0.01 b | 0.24 ± 0.00 a |
| 1655–1638 Amide I | 1700–1620 | 1.92 ± 0.01 b | 2.06 ± 0.03 c | 1.69 ± 0.00 a |
| 1549–1548 Amide II | 1560–1530 | 0.35 ± 0.00 a | 0.49 ± 0.01 b | 0.37 ± 0.00 a |
| 1518–1516 C = C stretching of aromatic rings | 1520–1500 | 0.21 ± 0.00 b | 0.23± 0.01 c | 0.18 ± 0.00 a |
| Esterification (%) 1 | 10.70 ± 0.05 a | 12.56 ± 0.08 b | 12.47 ± 0.05 b | |
| Protein Structure | Treatment | Ratio | |||
|---|---|---|---|---|---|
| SR | MFR | FR | SR:MFR | SR:FR | |
| α-helices | 50.7 | 4.2 | 3.0 | 12.07 | 16.9 |
| β-sheets (I + II) | 45.6 | 53.1 | 61.2 | 0.86 | 0.75 |
| Coils | 3.5 | 24.3 | 19.4 | 0.14 | 0.18 |
| Random | 0.2 | 18.4 | 16.4 | 0.01 | 0.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.H.; Lahlali, R.; Karunakaran, C.; Vujanovic, V. Specific Mycoparasite-Fusarium Graminearum Molecular Signatures in Germinating Seeds Disabled Fusarium Head Blight Pathogen’s Infection. Int. J. Mol. Sci. 2021, 22, 2461. https://doi.org/10.3390/ijms22052461
Kim SH, Lahlali R, Karunakaran C, Vujanovic V. Specific Mycoparasite-Fusarium Graminearum Molecular Signatures in Germinating Seeds Disabled Fusarium Head Blight Pathogen’s Infection. International Journal of Molecular Sciences. 2021; 22(5):2461. https://doi.org/10.3390/ijms22052461
Chicago/Turabian StyleKim, Seon Hwa, Rachid Lahlali, Chithra Karunakaran, and Vladimir Vujanovic. 2021. "Specific Mycoparasite-Fusarium Graminearum Molecular Signatures in Germinating Seeds Disabled Fusarium Head Blight Pathogen’s Infection" International Journal of Molecular Sciences 22, no. 5: 2461. https://doi.org/10.3390/ijms22052461
APA StyleKim, S. H., Lahlali, R., Karunakaran, C., & Vujanovic, V. (2021). Specific Mycoparasite-Fusarium Graminearum Molecular Signatures in Germinating Seeds Disabled Fusarium Head Blight Pathogen’s Infection. International Journal of Molecular Sciences, 22(5), 2461. https://doi.org/10.3390/ijms22052461







