An Extracellular Perspective on CNS Maturation: Perineuronal Nets and the Control of Plasticity
Abstract
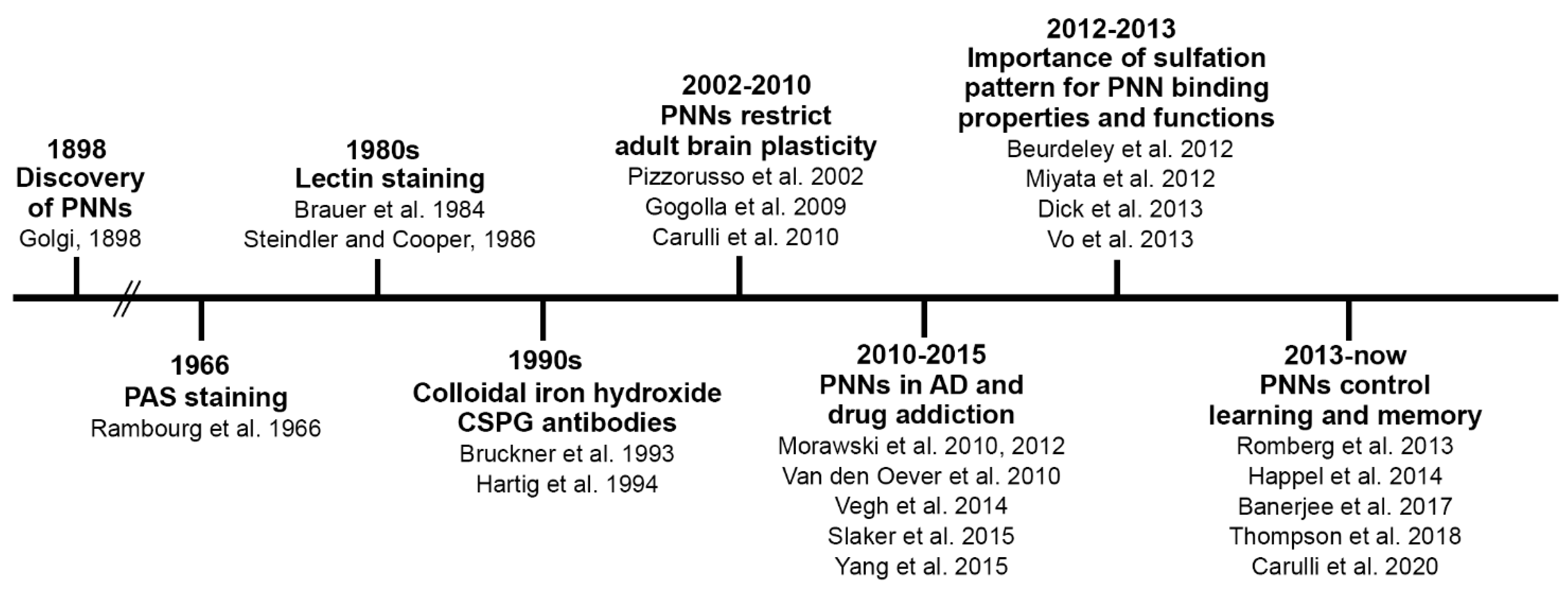
1. Introduction
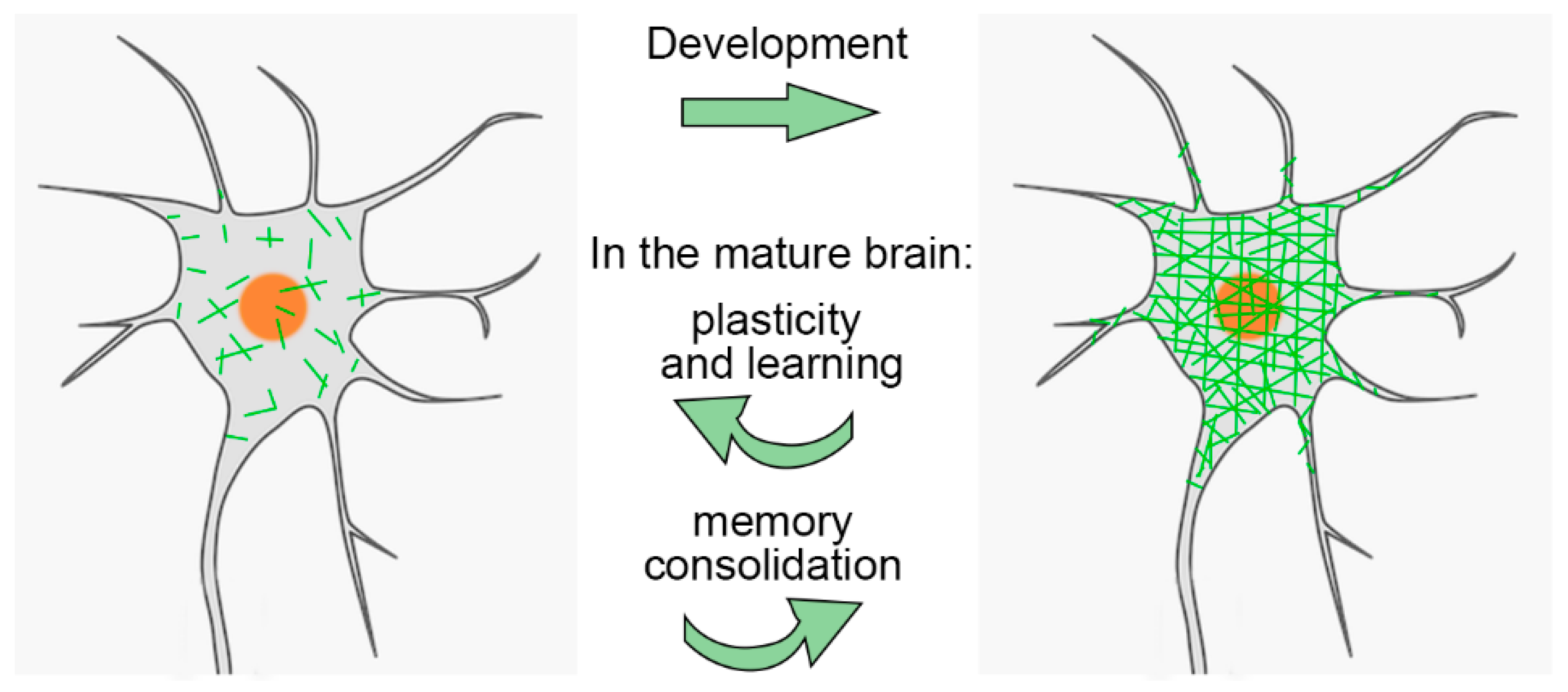
2. PNNs and Brain Maturation
2.1. Distribution of PNNs
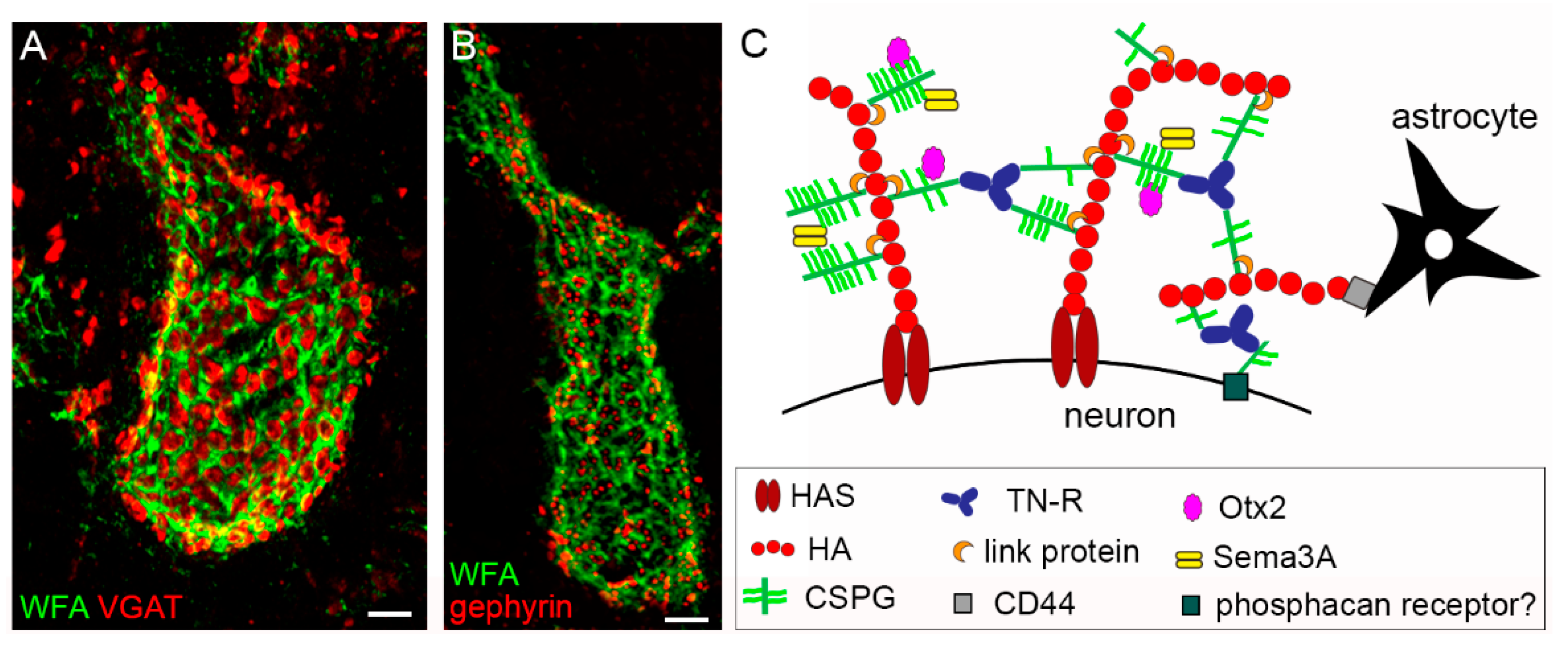
2.2. Composition of the PNN
2.3. PNNs and the Regulation of Critical Periods
3. PNNs and the Mature Brain
3.1. PNN Dynamics and Brain Physiology
3.2. PNNs during Learning and Memory
3.3. Role of Metalloproteinases
3.4. PNNs during Aging
4. PNNs and Memory-Related Diseases
4.1. PNNs in Alzheimer’s Disease
4.2. PNNs and Drug Addiction
5. Conclusions
Funding
Conflicts of Interest
References
- Reh, R.K.; Dias, B.G.; Nelson, C.A., 3rd; Kaufer, D.; Werker, J.F.; Kolb, B.; Levine, J.D.; Hensch, T.K. Critical period regulation acrossmultiple timescales. Proc. Natl. Acad. Sci. USA 2020, 117, 23242–23251. [Google Scholar] [CrossRef] [PubMed]
- Hubel, D.H.; Wiesel, T.N. Receptive fields, binocular interaction and functional architecture in the cat’s visual cortex. J. Physiol. 1962, 160, 106–154. [Google Scholar] [CrossRef]
- Hubel, D.H.; Wiesel, T.N. Binocular interaction in striate cortex of kittens reared with artificial squint. J. Neurophysiol. 1965, 28, 1041–1059. [Google Scholar] [CrossRef] [PubMed]
- Fifková, E. Changes in the visual cortex of rats after unilateral deprivation. Nature 1968, 220, 379–381. [Google Scholar] [CrossRef] [PubMed]
- Antonini, A.; Stryker, M.P. Plasticity of geniculocortical afferents following brief or prolonged monocular occlusion in the cat. J. Comp. Neurol. 1996, 369, 64–82. [Google Scholar] [CrossRef]
- Dews, P.B.; Wiesel, T.N. Consequences of monocular deprivation on visual behaviour in kittens. J. Physiol. 1970, 206, 437–455. [Google Scholar] [CrossRef]
- Callaghan, B.L.; Graham, B.M.; Li, S.; Richardson, R. From resilience to vulnerability: Mechanistic insights into the effects of stress on transitions in critical period plasticity. Front. Psychiatry 2013, 4, 90. [Google Scholar] [CrossRef]
- Southwell, D.G.; Froemke, R.C.; Alvarez-Buylla, A.; Gandhi, S.P. Cortical Plasticity Induced by Inhibitory Neuron Transplantation. Science 2010, 327, 1145–1148. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Stryker, M.P.; Alvarez-Buylla, A.; Espinosa, J.S. Cortical plasticity induced by transplantation of embryonic somatostatin or parvalbumin interneurons. Proc. Natl. Acad. Sci. USA 2014, 111, 18339–18344. [Google Scholar] [CrossRef]
- Morales, B.; Choi, S.Y.; Kirkwood, A. Dark rearing alters the development of GABAergic transmission in visual cortex. J. Neurosci. 2002, 22, 8084–8090. [Google Scholar] [CrossRef] [PubMed]
- Cancedda, L.; Putignano, E.; Sale, A.; Viegi, A.; Berardi, N.; Maffei, L. Acceleration of visual system development by environmental enrichment. J. Neurosci. 2004, 24, 4840–4848. [Google Scholar] [CrossRef]
- Sale, A.; Putignano, E.; Cancedda, L.; Landi, S.; Cirulli, F.; Berardi, N.; Maffei, L. Enriched environment and acceleration of visual system development. Neuropharmacology 2004, 47, 649–660. [Google Scholar] [CrossRef] [PubMed]
- Baroncelli, L.; Scali, M.; Sansevero, G.; Olimpico, F.; Manno, I.; Costa, M.; Sale, A. Experience affects critical period plasticity in the visual cortex through an epigenetic regulation of histone post-translational modifications. J. Neurosci. 2016, 36, 3430–3440. [Google Scholar] [CrossRef]
- Huang, Z.J.; Kirkwood, A.; Pizzorusso, T.; Porciatti, V.; Morales, B.; Bear, M.F.; Maffei, L.; Tonegawa, S. BDNF regulates the maturation of inhibition and the critical period of plasticity in mouse visual cortex. Cell 1999, 98, 739–755. [Google Scholar] [CrossRef]
- Gu, Y.; Huang, S.; Chang, M.C.; Worley, P.; Kirkwood, A.; Quinlan, E.M. Obligatory role for the immediate early gene NARP in critical period plasticity. Neuron 2013, 79, 335–346. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Tran, T.; Murase, S.; Borrell, A.; Kirkwood, A.; Quinlan, E.M. Neuregulin-dependent regulation of fast-spiking interneuron excitability controls the timing of the critical period. J. Neurosci. 2016, 36, 10285–10295. [Google Scholar] [CrossRef]
- Sugiyama, S.; Di Nardo, A.A.; Aizawa, S.; Matsuo, I.; Volovitch, M.; Prochiantz, A.; Hensch, T.K. Experience-Dependent Transfer of Otx2 Homeoprotein into the Visual Cortex Activates Postnatal Plasticity. Cell 2008, 134, 508–520. [Google Scholar] [CrossRef]
- Fawcett, J.W.; Oohashi, T.; Pizzorusso, T. The roles of perineuronal nets and the perinodal extracellular matrix in neuronal function. Nat. Rev. Neurosci. 2019, 20, 451–465. [Google Scholar] [CrossRef] [PubMed]
- Dityatev, A.; Schachner, M.; Sonderegger, P. The dual role of the extracellular matrix in synaptic plasticity and homeostasis. Nat. Rev. Neurosci. 2010, 11, 735–746. [Google Scholar] [CrossRef]
- Hockfield, S.; McKay, R.D.; Hendry, S.H.; Jones, E.G. A surface antigen that identifies ocular dominance columns in the visual cortex and laminar features of the lateral geniculate nucleus. Cold Spring Harb. Symp. Quant. Biol. 1983, 48, 877–889. [Google Scholar] [CrossRef]
- Seeger, G.; Brauer, K.; Härtig, W.; Brückner, G. Mapping of perineuronal nets in the rat brain stained by colloidal iron hydroxide histochemistry and lectin cytochemistry. Neuroscience 1994, 58, 371–388. [Google Scholar] [CrossRef]
- Brückner, G.; Schütz, A.; Härtig, W.; Brauer, K.; Paulke, B.R.; Bigl, V. Projection of non-cholinergic basal forebrain neurons ensheathed with perineuronal nets to rat mesocortex. J. Chem. Neuroanat. 1994, 8, 11–18. [Google Scholar] [CrossRef]
- Härtig, W.; Brückner, G.; Brauer, K.; Schmidt, C.; Bigl, V. Allocation of perineuronal nets and parvalbumin-, calbindin-D28k- and glutamic acid decarboxylase-immunoreactivity in the amygdala of the rhesus monkey. Brain Res. 1995, 698, 265–269. [Google Scholar] [CrossRef]
- Hausen, D.; Brückner, G.; Drlicek, M.; Härtig, W.; Brauer, K.; Bigl, V. Pyramidal cells ensheathed by perineuronal nets in human motor and somatosensory cortex. Neuroreport 1996, 7, 1725–1729. [Google Scholar] [CrossRef] [PubMed]
- Ojima, H.; Sakai, M.; Ohyama, J. Molecular heterogeneity of Vicia villosa-recognized perineuronal nets surrounding pyramidal and nonpyramidal neurons in the guinea pig cerebral cortex. Brain Res. 1998, 786, 274–280. [Google Scholar] [CrossRef]
- Brückner, G.; Grosche, J.; Hartlage-Rübsamen, M.; Schmidt, S.; Schachner, M. Region and lamina-specific distribution of extracellular matrix proteoglycans, hyaluronan and tenascin-R in the mouse hippocampal formation. J. Chem. Neuroanat. 2003, 26, 37–50. [Google Scholar] [CrossRef]
- Carulli, D.; Rhodes, K.E.; Brown, D.J.; Bonnert, T.P.; Pollack, S.J.; Oliver, K.; Strata, P.; Fawcett, J.W. Composition of perineuronal nets in the adult rat cerebellum and the cellular origin of their components. J. Comp. Neurol. 2006, 494, 559–577. [Google Scholar] [CrossRef]
- Vidal, E.; Bolea, R.; Tortosa, R.; Costa, C.; Domènech, A.; Monleón, E.; Vargas, A.; Badiola, J.J.; Pumarola, M. Assessment of calcium-binding proteins (Parvalbumin and Calbindin D-28K) and perineuronal nets in normal and scrapie-affected adult sheep brains. J. Virol. Methods 2006, 136, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Galtrey, C.M.; Kwok, J.C.F.; Carulli, D.; Rhodes, K.E.; Fawcett, J.W. Distribution and synthesis of extracellular matrix proteoglycans, hyaluronan, link proteins and tenascin-R in the rat spinal cord. Eur. J. Neurosci. 2008, 27, 1373–1390. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Leamey, C.A.; Sawatari, A. Perineuronal nets play a role in regulating striatal function in the mouse. PLoS ONE 2012, 7, e32747. [Google Scholar] [CrossRef]
- Yamada, J.; Jinno, S. Spatio-temporal differences in perineuronal net expression in the mouse hippocampus, with reference to parvalbumin. Neuroscience 2013, 253, 368–379. [Google Scholar] [CrossRef]
- Carstens, K.E.; Phillips, M.L.; Pozzo-Miller, L.; Weinberg, R.J.; Dudek, S.M. Perineuronal nets suppress plasticity of excitatory synapses on CA2 pyramidal neurons. J. Neurosci. 2016, 36, 6312–6320. [Google Scholar] [CrossRef] [PubMed]
- Fech, T.; Calderón-Garcidueñas, L.; Kulesza, R.J. Characterization of the superior olivary complex of Canis lupus domesticus. Hear. Res. 2017, 351, 130–140. [Google Scholar] [CrossRef]
- Mirzadeh, Z.; Alonge, K.M.; Cabrales, E.; Herranz-Pérez, V.; Scarlett, J.M.; Brown, J.M.; Hassouna, R.; Matsen, M.E.; Nguyen, H.T.; Garcia-Verdugo, J.M.; et al. Perineuronal net formation during the critical period for neuronal maturation in the hypothalamic arcuate nucleus. Nat. Metab. 2019, 1, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Marchand, A.; Schwartz, C. Perineuronal net expression in the brain of a hibernating mammal. Brain Struct. Funct. 2020, 225, 45–56. [Google Scholar] [CrossRef]
- Härtig, W.; Brauer, K.; Bigl, V.; Brückner, G. Chondroitin sulfate proteoglycan-immunoreactivity of lectin-labeled perineuronal nets around parvalbumin-containing neurons. Brain Res. 1994, 635, 307–311. [Google Scholar] [CrossRef]
- Alpár, A.; Gärtner, U.; Härtig, W.; Brückner, G. Distribution of pyramidal cells associated with perineuronal nets in the neocortex of rat. Brain Res. 2006, 1120, 13–22. [Google Scholar] [CrossRef]
- Baker, K.D.; Gray, A.R.; Richardson, R. The development of perineuronal nets around parvalbumin GABAergic neurons in the medial prefrontal cortex and basolateral amygdala of rats. Behav. Neurosci. 2017, 131, 289–303. [Google Scholar] [CrossRef]
- Morikawa, S.; Ikegaya, Y.; Narita, M.; Tamura, H. Activation of perineuronal net-expressing excitatory neurons during associative memory encoding and retrieval. Sci. Rep. 2017, 7, 46024. [Google Scholar] [CrossRef] [PubMed]
- Härtig, W.; Derouiche, A.; Welt, K.; Brauer, K.; Grosche, J.; Mäder, M.; Reichenbach, A.; Brückner, G. Cortical neurons immunoreactive for the potassium channel Kv3.1b subunit are predominantly surrounded by perineuronal nets presumed as a buffering system for cations. Brain Res. 1999, 842, 15–29. [Google Scholar] [CrossRef]
- Morawski, M.; Reinert, T.; Meyer-Klaucke, W.; Wagner, F.E.; Tröger, W.; Reinert, A.; Jäger, C.; Brückner, G.; Arendt, T. Ion exchanger in the brain: Quantitative analysis of perineuronally fixed anionic binding sites suggests diffusion barriers with ion sorting properties. Sci. Rep. 2015, 5, 16471. [Google Scholar] [CrossRef]
- Yamaguchi, Y. Lecticans: Organizers of the brain extracellular matrix. Cell. Mol. Life Sci. 2000, 57, 276–289. [Google Scholar] [CrossRef]
- Kwok, J.C.F.; Carulli, D.; Fawcett, J.W. In vitro modeling of perineuronal nets: Hyaluronan synthase and link protein are necessary for their formation and integrity. J. Neurochem. 2010, 114, 1447–1459. [Google Scholar] [CrossRef]
- Aruffo, A.; Stamenkovic, I.; Melnick, M.; Underhill, C.B.; Seed, B. CD44 is the principal cell surface receptor for hyaluronate. Cell 1990, 61, 1303–1313. [Google Scholar] [CrossRef]
- Fowke, T.M.; Karunasinghe, R.N.; Bai, J.-Z.; Jordan, S.; Gunn, A.J.; Dean, J.M. Hyaluronan synthesis by developing cortical neurons in vitro. Sci. Rep. 2017, 7, 44135. [Google Scholar] [CrossRef] [PubMed]
- Deepa, S.S.; Carulli, D.; Galtrey, C.; Rhodes, K.; Fukuda, J.; Mikami, T.; Sugahara, K.; Fawcett, J.W. Composition of perineuronal net extracellular matrix in rat brain: A different disaccharide composition for the net-associated proteoglycans. J. Biol. Chem. 2006, 281, 17789–17800. [Google Scholar] [CrossRef] [PubMed]
- Happel, M.F.K.; Niekisch, H.; Castiblanco Rivera, L.L.; Ohl, F.W.; Deliano, M.; Frischknecht, R. Enhanced cognitive flexibility in reversal learning induced by removal of the extracellular matrix in auditory cortex. Proc. Natl. Acad. Sci. USA 2014, 111, 2800–2805. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.Y.; Bozzelli, P.L.; Caccavano, A.; Allen, M.; Balmuth, J.; Vicini, S.; Wu, J.; Conant, K. Disruption of perineuronal nets increases the frequency of sharp wave ripple events. Hippocampus 2018, 28, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Matthews, R.T.; Kelly, G.M.; Zerillo, C.A.; Gray, G.; Tiemeyer, M.; Hockfield, S. Aggrecan Glycoforms Contribute to the Molecular Heterogeneity of Perineuronal Nets. J. Neurosci. 2002, 22, 7536–7547. [Google Scholar] [CrossRef]
- Sugahara, K.; Kitagawa, H. Recent advances in the study of the biosynthesis and functions of sulfated glycosaminoglycans. Curr. Opin. Struct. Biol. 2000, 10, 518–527. [Google Scholar] [CrossRef]
- Miyata, S.; Komatsu, Y.; Yoshimura, Y.; Taya, C.; Kitagawa, H. Persistent cortical plasticity by upregulation of chondroitin 6-sulfation. Nat. Neurosci. 2012, 15, 414–422. [Google Scholar] [CrossRef]
- Miller, G.M.; Hsieh-Wilson, L.C. Sugar-dependent modulation of neuronal development, regeneration, and plasticity by chondroitin sulfate proteoglycans. Exp. Neurol. 2015, 274, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Miyata, S.; Nadanaka, S.; Igarashi, M.; Kitagawa, H. Structural Variation of Chondroitin Sulfate Chains Contributes to the Molecular Heterogeneity of Perineuronal Nets. Front. Integr. Neurosci. 2018, 12, 1–12. [Google Scholar] [CrossRef]
- Kitagawa, H.; Tsutsumi, K.; Tone, Y.; Sugahara, K. Developmental regulation of the sulfation profile of chondroitin sulfate chains in the chicken embryo brain. J. Biol. Chem. 1997, 272, 31377–31381. [Google Scholar] [CrossRef] [PubMed]
- Carulli, D.; Pizzorusso, T.; Kwok, J.C.F.; Putignano, E.; Poli, A.; Forostyak, S.; Andrews, M.R.; Deepa, S.S.; Glant, T.T.; Fawcett, J.W. Animals lacking link protein have attenuated perineuronal nets and persistent plasticity. Brain 2010, 133, 2331–2347. [Google Scholar] [CrossRef] [PubMed]
- Foscarin, S.; Raha-Chowdhury, R.; Fawcett, J.W.; Kwok, J.C.F. Brain ageing changes proteoglycan sulfation, rendering perineuronal nets more inhibitory. Aging 2017, 9, 1607–1622. [Google Scholar] [CrossRef]
- Wang, H.; Katagiri, Y.; McCann, T.E.; Unsworth, E.; Goldsmith, P.; Yu, Z.X.; Tan, F.; Santiago, L.; Mills, E.M.; Wang, Y.; et al. Chondroitin-4-sulfation negatively regulates axonal guidance and growth. J. Cell Sci. 2008, 121, 3083–3091. [Google Scholar] [CrossRef]
- Lin, R.; Rosahl, T.W.; Whiting, P.J.; Fawcett, J.W.; Kwok, J.C.F. 6-Sulphated chondroitins have a positive influence on axonal regeneration. PLoS ONE 2011, 6, e21499. [Google Scholar] [CrossRef]
- Sahu, S.; Li, R.; Loers, G.; Schachner, M. Knockdown of chondroitin-4-sulfotransferase-1, but not of dermatan-4-sulfotransferase-1, accelerates regeneration of zebrafish after spinal cord injury. FASEB J. 2019, 33, 2252–2262. [Google Scholar] [CrossRef]
- Beurdeley, M.; Spatazza, J.; Lee, H.H.C.; Sugiyama, S.; Bernard, C.; Di Nardo, A.A.; Hensch, T.K.; Prochiantz, A. Otx2 Binding to Perineuronal Nets Persistently Regulates Plasticity in the Mature Visual Cortex. J. Neurosci. 2012, 32, 9429–9437. [Google Scholar] [CrossRef]
- Brown, J.M.; Xia, J.; Zhuang, B.Q.; Cho, K.S.; Rogers, C.J.; Gama, C.I.; Rawat, M.; Tully, S.E.; Uetani, N.; Mason, D.E.; et al. A sulfated carbohydrate epitope inhibits axon regeneration after injury. Proc. Natl. Acad. Sci. USA 2012, 109, 4768–4773. [Google Scholar] [CrossRef]
- Despras, G.; Bernard, C.; Perrot, A.; Cattiaux, L.; Prochiantz, A.; Lortat-Jacob, H.; Mallet, J.-M. Toward libraries of biotinylated chondroitin sulfate analogues: From synthesis to in vivo studies. Chem. A Eur. J. 2013, 19, 531–540. [Google Scholar] [CrossRef] [PubMed]
- Dick, G.; Tan, C.L.; Nuno Alves, J.; Ehlert, E.M.E.; Miller, G.M.; Hsieh-Wilson, L.C.; Sugahara, K.; Oosterhof, A.; van Kuppevelt, T.H.; Verhaagen, J.; et al. Semaphorin 3A binds to the perineuronal nets via chondroitin sulfate type E motifs in rodent brains. J. Biol. Chem. 2013, 288, 27384–27395. [Google Scholar] [CrossRef] [PubMed]
- Meyer-Puttlitz, B.; Milev, P.; Junker, E.; Zimmer, I.; Margolis, R.U.; Margolis, R.K. Chondroitin Sulfate and Chondroitin/Keratan Sulfate Proteoglycans of Nervous Tissue: Developmental Changes of Neurocan and Phosphacan. J. Neurochem. 1995, 65, 2327–2337. [Google Scholar] [CrossRef]
- Hirakawa, S.; Oohashi, T.; Su, W.D.; Yoshioka, H.; Murakami, T.; Arata, J.; Ninomiya, Y. The brain link protein-1 (BRAL1): cDNA cloning, genomic structure, and characterization as a novel link protein expressed in adult brain. Biochem. Biophys. Res. Commun. 2000, 276, 982–989. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bekku, Y.; Su, W.-D.; Hirakawa, S.; Fässler, R.; Ohtsuka, A.; Kang, J.S.; Sanders, J.; Murakami, T.; Ninomiya, Y.; Oohashi, T. Molecular cloning of Bral2, a novel brain-specific link protein, and immunohistochemical colocalization with brevican in perineuronal nets. Mol. Cell. Neurosci. 2003, 24, 48–159. [Google Scholar] [CrossRef]
- Ogawa, H.; Oohashi, T.; Sata, M.; Bekku, Y.; Hirohata, S.; Nakamura, K.; Yonezawa, T.; Kusachi, S.; Shiratori, Y.; Ninomiya, Y. Lp3/Hapln3, a novel link protein that co-localizes with versican and is coordinately up-regulated by platelet-derived growth factor in arterial smooth muscle cells. Matrix Biol. 2004, 23, 287–298. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Oohashi, T.; Edamatsu, M.; Bekku, Y.; Carulli, D. The hyaluronan and proteoglycan link proteins: Organizers of the brain extracellular matrix and key molecules for neuronal function and plasticity. Exp. Neurol. 2015, 274, 134–144. [Google Scholar] [CrossRef]
- Lundell, A.; Olin, A.I.; Mörgelin, M.; al-Karadaghi, S.; Aspberg, A.; Logan, D.T. Structural basis for interactions between tenascins and lectican C-type lectin domains: Evidence for a crosslinking role for tenascins. Structure 2004, 12, 1495–1506. [Google Scholar] [CrossRef] [PubMed]
- Weber, P.; Bartsch, U.; Rasband, M.N.; Czaniera, R.; Lang, Y.; Bluethmann, H.; Margolis, R.U.; Levinson, S.R.; Shrager, P.; Montag, D.; et al. Mice deficient for tenascin-R display alterations of the extracellular matrix and decreased axonal conduction velocities in the CNS. J. Neurosci. 1999, 19, 4245–4262. [Google Scholar] [CrossRef]
- Brückner, G.; Grosche, J.; Schmidt, S.; Härtig, W.; Margolis, R.U.; Delpech, B.; Seidenbecher, C.I.; Czaniera, R.; Schachner, M. Postnatal development of perineuronal nets in wild-type mice and in a mutant deficient in tenascin-R. J. Comp. Neurol. 2000, 428, 616–629. [Google Scholar] [CrossRef]
- Miller, B.; Sheppard, A.M.; Bicknese, A.R.; Pearlman, A.L. Chondroitin sulfate proteoglycans in the developing cerebral cortex: The distribution of neurocan distinguishes forming afferent and efferent axonal pathways. J. Comp. Neurol. 1995, 355, 615–628. [Google Scholar] [CrossRef]
- Pearlman, A.L.; Sheppard, A.M. Extracellular matrix in early cortical development. Prog. Brain Res. 1996, 108, 117–134. [Google Scholar]
- Carulli, D.; Rhodes, K.E.; Fawcett, J.W. Upregulation of aggrecan, link protein 1, and hyaluronan synthases during formation of perineuronal nets in the rat cerebellum. J. Comp. Neurol. 2007, 501, 83–94. [Google Scholar] [CrossRef]
- Bekku, Y.; Saito, M.; Moser, M.; Fuchigami, M.; Maehara, A.; Nakayama, M.; Kusachi, S.; Ninomiya, Y.; Oohashi, T. Bral2 is indispensable for the proper localization of brevican and the structural integrity of the perineuronal net in the brainstem and cerebellum. J. Comp. Neurol. 2012, 520, 1721–1736. [Google Scholar] [CrossRef]
- Rowlands, D.; Lensjø, K.K.; Dinh, T.; Yang, S.; Andrews, M.R.; Hafting, T.; Fyhn, M.; Fawcett, J.W.; Dick, G. Aggrecan Directs Extracellular Matrix-Mediated Neuronal Plasticity. J. Neurosci. 2018, 38, 10102–10113. [Google Scholar] [CrossRef]
- Eill, G.J.; Sinha, A.; Morawski, M.; Viapiano, M.S.; Matthews, R.T. The protein tyrosine phosphatase RPTPξ/phosphacan is critical for perineuronal net structure. J. Biol. Chem. 2020, 295, 955–968. [Google Scholar] [CrossRef] [PubMed]
- Canoll, P.D.; Barnea, G.; Levy, J.B.; Sap, J.; Ehrlich, M.; Silvennoinen, O.; Schlessinger, J.; Musacchio, J.M. The expression of a novel receptor-type tyrosine phosphatase suggests a role in morphogenesis and plasticity of the nervous system. Dev. Brain Res. 1993, 75, 293–298. [Google Scholar] [CrossRef]
- Shintani, T.; Watanabe, E.; Maeda, N.; Noda, M. Neurons as well as astrocytes express proteoglycan-type protein tyrosine phosphatase ζ/RPTPβ: Analysis of mice in which the PTPζ/RPTPβ gene was replaced with the LacZ gene. Neurosci. Lett. 1998, 247, 135–138. [Google Scholar] [CrossRef]
- Zhou, X.-H.; Brakebusch, C.; Matthies, H.; Oohashi, T.; Hirsch, E.; Moser, M.; Krug, M.; Seidenbecher, C.I.; Boeckers, T.M.; Rauch, U.; et al. Neurocan Is Dispensable for Brain Development. Mol. Cell. Biol. 2001, 21, 5970–5978. [Google Scholar] [CrossRef]
- Brakebusch, C.; Seidenbecher, C.I.; Asztely, F.; Rauch, U.; Matthies, H.; Meyer, H.; Krug, M.; Böckers, T.M.; Zhou, X.; Kreutz, M.R.; et al. Brevican-Deficient Mice Display Impaired Hippocampal CA1 Long-Term Potentiation but Show No Obvious Deficits in Learning and Memory. Mol. Cell. Biol. 2002, 22, 7417–7427. [Google Scholar] [CrossRef]
- Rauch, U.; Zhou, X.H.; Roos, G. Extracellular matrix alterations in brains lacking four of its components. Biochem. Biophys. Res. Commun. 2005, 328, 608–617. [Google Scholar] [CrossRef] [PubMed]
- Geissler, M.; Gottschling, C.; Aguado, A.; Rauch, U.; Wetzel, C.H.; Hatt, H.; Faissner, A. Primary Hippocampal Neurons, Which Lack Four Crucial Extracellular Matrix Molecules, Display Abnormalities of Synaptic Structure and Function and Severe Deficits in Perineuronal Net Formation. J. Neurosci. 2013, 33, 7742–7755. [Google Scholar] [CrossRef] [PubMed]
- Gottschling, C.; Wegrzyn, D.; Denecke, B.; Faissner, A. Elimination of the four extracellular matrix molecules tenascin-C, tenascin-R, brevican and neurocan alters the ratio of excitatory and inhibitory synapses. Sci. Rep. 2019, 9, 13939. [Google Scholar] [CrossRef]
- Apostolova, I.; Irintchev, A.; Schachner, M. Tenascin-R restricts posttraumatic remodeling of motoneuron innervation and functional recovery after spinal cord injury in adult mice. J. Neurosci. 2006, 26, 7849–7859. [Google Scholar] [CrossRef] [PubMed]
- Pizzorusso, T.; Medini, P.; Berardi, N.; Chierzi, S.; Fawcett, J.W.; Maffei, L. Reactivation of ocular dominance plasticity in the adult visual cortex. Science 2002, 298, 1248–1251. [Google Scholar] [CrossRef] [PubMed]
- McRae, P.A.; Rocco, M.M.; Kelly, G.; Brumberg, J.C.; Matthews, R.T. Sensory Deprivation Alters Aggrecan and Perineuronal Net Expression in the Mouse Barrel Cortex. J. Neurosci. 2007, 27, 5405–5413. [Google Scholar] [CrossRef] [PubMed]
- Ueno, H.; Suemitsu, S.; Okamoto, M.; Matsumoto, Y.; Ishihara, T. Sensory experience-dependent formation of perineuronal nets and expression of Cat-315 immunoreactive components in the mouse somatosensory cortex. Neuroscience 2017, 355, 161–174. [Google Scholar] [CrossRef]
- Kalb, R.G.; Hockfield, S. Molecular evidence for early activity-dependent development of hamster motor neurons. J. Neurosci. 1988, 8, 2350–2360. [Google Scholar] [CrossRef]
- Reinhard, S.M.; Abundez-Toledo, M.; Espinoza, K.; Razak, K.A. Effects of developmental noise exposure on inhibitory cell densities and perineuronal nets in A1 and AAF of mice. Hear. Res. 2019, 381, 107781. [Google Scholar] [CrossRef]
- Ueno, H.; Suemitsu, S.; Okamoto, M.; Matsumoto, Y.; Ishihara, T. Parvalbumin neurons and perineuronal nets in the mouse prefrontal cortex. Neuroscience 2017, 343, 115–127. [Google Scholar] [CrossRef]
- Kolb, B.; Gibb, R. Plasticity in the prefrontal cortex of adult rats. Front. Cell. Neurosci. 2015, 9, 15. [Google Scholar] [CrossRef]
- Sigal, Y.M.; Bae, H.; Bogart, L.J.; Hensch, T.K.; Zhuang, X. Structural maturation of cortical perineuronal nets and their perforating synapses revealed by superresolution imaging. Proc. Natl. Acad. Sci. USA 2019, 116, 7071–7076. [Google Scholar] [CrossRef] [PubMed]
- Richter, R.P.; Baranova, N.S.; Day, A.J.; Kwok, J.C. Glycosaminoglycans in extracellular matrix organisation: Are concepts from soft matter physics key to understanding the formation of perineuronal nets? Curr. Opin. Struct. Biol. 2018, 50, 65–74. [Google Scholar] [CrossRef]
- Gogolla, N.; Caroni, P.; Lüthi, A.; Herry, C. Perineuronal nets protect fear memories from erasure. Science 2009, 325, 1258–1261. [Google Scholar] [CrossRef] [PubMed]
- Nowicka, D.; Soulsby, S.; Skangiel-Kramska, J.; Glazewski, S. Parvalbumin-containing neurons, perineuronal nets and experience-dependent plasticity in murine barrel cortex. Eur. J. Neurosci. 2009, 30, 2053–2063. [Google Scholar] [CrossRef]
- Balmer, T.S.; Carels, V.M.; Frisch, J.L.; Nick, T.A. Modulation of perineuronal nets and parvalbumin with developmental song learning. J. Neurosci. 2009, 29, 12878–12885. [Google Scholar] [CrossRef]
- Cornez, G.; Collignon, C.; Müller, W.; Cornil, C.A.; Ball, G.F.; Balthazart, J. Development of perineuronal nets during ontogeny correlates with sensorimotor vocal learning in canaries. Eneuro 2020, 7. [Google Scholar] [CrossRef] [PubMed]
- Cornez, G.; Shevchouk, O.T.; Ghorbanpoor, S.; Ball, G.F.; Cornil, C.A.; Balthazart, J. Testosterone stimulates perineuronal nets development around parvalbumin cells in the adult canary brain in parallel with song crystallization. Horm. Behav. 2020, 119, 104643. [Google Scholar] [CrossRef]
- Pizzorusso, T.; Medini, P.; Landi, S.; Baldini, S.; Berardi, N.; Maffei, L. Structural and functional recovery from early monocular deprivation in adult rats. Proc. Natl. Acad. Sci. USA 2006, 103, 8517–8522. [Google Scholar] [CrossRef] [PubMed]
- Fawcett, J.W. The extracellular matrix in plasticity and regeneration after CNS injury and neurodegenerative disease. Prog. Brain Res. 2015, 218, 213–226. [Google Scholar]
- Pechtel, P.; Pizzagalli, D.A. Effects of early life stress on cognitive and affective function: An integrated review of human literature. Psychopharmacology 2011, 214, 55–70. [Google Scholar] [CrossRef] [PubMed]
- Teicher, M.H.; Ohashi, K.; Lowen, S.B.; Polcari, A.; Fitzmaurice, G.M. Mood dysregulation and affective instability in emerging adults with childhood maltreatment: An ecological momentary assessment study. J. Psychiatr. Res. 2015, 70, 1–8. [Google Scholar] [CrossRef]
- Page, C.E.; Coutellier, L. Adolescent Stress Disrupts the Maturation of Anxiety-related Behaviors and Alters the Developmental Trajectory of the Prefrontal Cortex in a Sex- and Age-specific Manner. Neuroscience 2018, 390, 265–277. [Google Scholar] [CrossRef]
- Santiago, A.N.; Lim, K.Y.; Opendak, M.; Sullivan, R.M.; Aoki, C. Early life trauma increases threat response of peri-weaning rats, reduction of axo-somatic synapses formed by parvalbumin cells and perineuronal net in the basolateral nucleus of amygdala. J. Comp. Neurol. 2018, 526, 2647–2664. [Google Scholar] [CrossRef]
- Riga, D.; Kramvis, I.; Koskinen, M.K.; van Bokhoven, P.; van der Harst, J.E.; Heistek, T.S.; Jaap Timmerman, A.; van Nierop, P.; van der Schors, R.C.; Pieneman, A.W.; et al. Hippocampal extracellular matrix alterations contribute to cognitive impairment associated with a chronic depressive-like state in rats. Sci. Transl. Med. 2017, 9, eaai8753. [Google Scholar] [CrossRef] [PubMed]
- Murthy, S.; Kane, G.A.; Katchur, N.J.; Mejia, P.S.L.; Obiofuma, G.; Buschman, T.J.; McEwen, B.S.; Gould, E. Perineuronal Nets, Inhibitory Interneurons, and Anxiety-Related Ventral Hippocampal Neuronal Oscillations Are Altered by Early Life Adversity. Biol. Psychiatry 2019, 85, 1011–1020. [Google Scholar] [CrossRef]
- Guadagno, A.; Verlezza, S.; Long, H.; Wong, T.P.; Walker, C.D. It is all in the right amygdala: Increased synaptic plasticity and perineuronal nets in male, but not female, juvenile rat pups after exposure to early-life stress. J. Neurosci. 2020, 40, 8276–8291. [Google Scholar] [CrossRef]
- Felt, B.T.; Lozoff, B. Brain iron and behavior of rats are not normalized by treatment of iron deficiency anemia during early development. J. Nutr. 1996, 126, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Felt, B.T.; Beard, J.L.; Schallert, T.; Shao, J.; Aldridge, J.W.; Connor, J.R.; Georgieff, M.K.; Lozoff, B. Persistent neurochemical and behavioral abnormalities in adulthood despite early iron supplementation for perinatal iron deficiency anemia in rats. Behav. Brain Res. 2006, 171, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.T.; Waldow, K.J.; Grove, W.M.; Salinas, J.A.; Georgieff, M.K. Dissociating the Long-Term Effects of Fetal/Neonatal Iron Deficiency on Three Types of Learning in the Rat. Behav. Neurosci. 2007, 121, 475–482. [Google Scholar] [CrossRef]
- Riggins, T.; Miller, N.C.; Bauer, P.J.; Georgieff, M.K.; Nelson, C.A. Consequences of low neonatal iron status due to maternal diabetes mellitus on explicit memory performance in childhood. Dev. Neuropsychol. 2009, 34, 762–779. [Google Scholar] [CrossRef] [PubMed]
- Jorgenson, L.A.; Wobken, J.D.; Georgieff, M.K. Perinatal Iron Deficiency Alters Apical Dendritic Growth in Hippocampal CA1 Pyramidal Neurons. Dev. Neurosci. 2003, 25, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Brunette, K.E.; Tran, P.V.; Wobken, J.D.; Carlson, E.S.; Georgieff, M.K. Gestational and neonatal iron deficiency alters apical dendrite structure of CA1 pyramidal neurons in adult rat hippocampus. Dev. Neurosci. 2010, 32, 238–248. [Google Scholar] [CrossRef]
- Fretham, S.J.B.; Carlson, E.S.; Wobken, J.; Tran, P.V.; Petryk, A.; Georgieff, M.K. Temporal manipulation of transferrin-receptor-1-dependent iron uptake identifies a sensitive period in mouse hippocampal neurodevelopment. Hippocampus 2012, 22, 1691–1702. [Google Scholar] [CrossRef]
- Umemori, J.; Winkel, F.; Castrén, E.; Karpova, N.N. Distinct effects of perinatal exposure to fluoxetine or methylmercury on parvalbumin and perineuronal nets, the markers of critical periods in brain development. Int. J. Dev. Neurosci. 2015, 44, 55–64. [Google Scholar] [CrossRef]
- Ko, M.C.; Lee, L.J.H.; Li, Y.; Lee, L.J. Long-term consequences of neonatal fluoxetine exposure in adult rats. Dev. Neurobiol. 2014, 74, 1038–1051. [Google Scholar] [CrossRef]
- Tzakis, N.; Holahan, M.R. Social Memory and the Role of the Hippocampal CA2 Region. Front. Behav. Neurosci. 2019, 13, 233. [Google Scholar] [CrossRef] [PubMed]
- Dudek, S.M.; Alexander, G.M.; Farris, S. Rediscovering area CA2: Unique properties and functions. Nat. Rev. Neurosci. 2016, 17, 89–102. [Google Scholar] [CrossRef]
- Giger, R.J.; Pasterkamp, R.J.; Heijnen, S.; Holtmaat, A.J.G.D.; Verhaagen, J. Anatomical distribution of the chemorepellent semaphorin III/collapsin- 1 in the adult rat and human brain: Predominant expression in structures of the olfactory-hippocampal pathway and the motor system. J. Neurosci. Res. 1998, 52, 27–42. [Google Scholar] [CrossRef]
- Vo, T.; Carulli, D.; Ehlert, E.M.E.; Kwok, J.C.F.; Dick, G.; Mecollari, V.; Moloney, E.B.; Neufeld, G.; de Winter, F.; Fawcett, J.W.; et al. The chemorepulsive axon guidance protein semaphorin3A is a constituent of perineuronal nets in the adult rodent brain. Mol. Cell. Neurosci. 2013, 56, 186–200. [Google Scholar] [CrossRef]
- Nadanaka, S.; Miyata, S.; Yaqiang, B.; Tamura, J.-I.; Habuchi, O.; Kitagawa, H. Reconsideration of the semaphorin-3a binding motif found in chondroitin sulfate using galnac4s-6st-knockout mice. Biomolecules 2020, 10, 1499. [Google Scholar] [CrossRef]
- Yang, S.; Hilton, S.; Nuno Alves, J.; Saksida, L.M.; Bussey, T.; Matthews, R.T.; Kitagawa, H.; Spillantini, M.G.; Kwok, J.C.F.; Fawcett, J.W. Antibody recognizing 4-sulfated chondroitin sulfate proteoglycans restores memory in tauopathy-induced neurodegeneration. Neurobiol. Aging 2017, 59, 197–209. [Google Scholar] [CrossRef]
- Boggio, E.M.; Ehlert, E.M.; Lupori, L.; Moloney, E.B.; De Winter, F.; Vander Kooi, C.W.; Baroncelli, L.; Mecollari, V.; Blits, B.; Fawcett, J.W.; et al. Inhibition of Semaphorin3A Promotes Ocular Dominance Plasticity in the Adult Rat Visual Cortex. Mol. Neurobiol. 2019, 56, 5987–5997. [Google Scholar] [CrossRef] [PubMed]
- Janssen, B.J.C.; Malinauskas, T.; Weir, G.A.; Zameel Cader, M.; Siebold, C.; Jones, E.Y. Neuropilins lock secreted semaphorins onto plexins in a ternary signaling complex. Nat. Struct. Mol. Biol. 2012, 19, 1293–1299. [Google Scholar] [CrossRef]
- Gutekunst, C.-A.; Steward, E.N.; Gross, R.E. Immunohistochemical distribution of PlexinA4 in the adult rat central nervous system. Front. Neuroanat. 2010, 4, 1–17. [Google Scholar] [CrossRef]
- Saywell, V.; Cioni, J.M.; Ango, F. Developmental gene expression profile of axon guidance cues in purkinje cells during cerebellar circuit formation. Cerebellum 2014, 13, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Holtmaat, A.J.G.D.; Gorter, J.A.; De Wit, J.; Tolner, E.A.; Spijker, S.; Giger, R.J.; Da Silva, F.H.L.; Verhaagen, J. Transient downregulation of Sema3A mRNA in a rat model for temporal lobe epilepsy: A novel molecular event potentially contributing to mossy fiber sprouting. Exp. Neurol. 2003, 182, 142–150. [Google Scholar] [CrossRef]
- Djerbal, L.; Vivès, R.; Lopin-Bon, C.; Richter, R.; Kwok, J.C.; Lortat-Jacob, H. Semaphorin 3A binding to chondroitin sulfate E enhances the biological activity of the protein, and cross-links and rigidifies glycosaminoglycan matrices. BioRxiv 2019. [Google Scholar] [CrossRef]
- De Wit, J.; Toonen, R.F.; Verhaagen, J.; Verhage, M. Vesicular trafficking of semaphorin 3A is activity-dependent and differs between axons and dendrites. Traffic 2006, 7, 1060–1077. [Google Scholar] [CrossRef] [PubMed]
- Pasterkamp, R.J.; Giger, R.; Ruitenberg, M.-J.; Holtmaat, A.; De Wit, J.; De Winter, F.; Verhaagen, J. Expression of the gene encoding the chemorepellent semaphorin III is induced in the fibroblast component of neural scar tissue formed following injuries of adult but not neonatal CNS. Mol. Cell. Neurosci. 1999, 13, 143–166. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.H.C.; Bernard, C.; Ye, Z.; Acampora, D.; Simeone, A.; Prochiantz, A.; Di Nardo, A.A.; Hensch, T.K. Genetic Otx2 mis-localization delays critical period plasticity across brain regions. Mol. Psychiatry 2017, 22, 680–688. [Google Scholar] [CrossRef] [PubMed]
- Bernard, C.; Vincent, C.; Testa, D.; Bertini, E.; Ribot, J.; Di Nardo, A.A.; Volovitch, M.; Prochiantz, A. A Mouse Model for Conditional Secretion of Specific Single-Chain Antibodies Provides Genetic Evidence for Regulation of Cortical Plasticity by a Non-cell Autonomous Homeoprotein Transcription Factor. PLoS Genet. 2016, 12, e1006035. [Google Scholar] [CrossRef] [PubMed]
- Johansson, P.A.; Irmler, M.; Acampora, D.; Beckers, J.; Simeone, S.; Götz, M. The transcription factor Otx2 regulates choroid plexus development and function. Development 2013, 140, 1055–1066. [Google Scholar] [CrossRef] [PubMed]
- Spatazza, J.; Lee, H.H.C.; Di Nardo, A.A.; Tibaldi, L.; Joliot, A.; Hensch, T.K.; Prochiantz, A. Choroid-Plexus-Derived Otx2 Homeoprotein Constrains Adult Cortical Plasticity. Cell Rep. 2013, 3, 1815–1823. [Google Scholar] [CrossRef]
- Hou, X.; Yoshioka, N.; Tsukano, H.; Sakai, A.; Miyata, S.; Watanabe, Y.; Yanagawa, Y.; Sakimura, K.; Takeuchi, K.; Kitagawa, H.; et al. Chondroitin Sulfate Is Required for Onset and Offset of Critical Period Plasticity in Visual Cortex. Sci. Rep. 2017, 7, 1–17. [Google Scholar] [CrossRef]
- Barreto, G.; Schäfer, A.; Marhold, J.; Stach, D.; Swaminathan, S.K.; Handa, V.; Döderlein, G.; Maltry, N.; Wu, W.; Lyko, F.; et al. Gadd45a promotes epigenetic gene activation by repair-mediated DNA demethylation. Nature 2007, 445, 671–675. [Google Scholar] [CrossRef]
- Ma, D.K.; Guo, J.U.; Ming, G.L.; Song, H. DNA excision repair proteins and Gadd45 as molecular players for active DNA demethylation. Cell Cycle 2009, 8, 1526–1531. [Google Scholar] [CrossRef] [PubMed]
- Gavin, D.P.; Akbarian, S. Epigenetic and post-transcriptional dysregulation of gene expression in schizophrenia and related disease. Neurobiol. Dis. 2012, 46, 255–262. [Google Scholar] [CrossRef]
- Leach, P.T.; Poplawski, S.G.; Kenney, J.W.; Hoffman, B.; Liebermann, D.A.; Abel, T.; Gould, T.J. Gadd45b knockout mice exhibit selective deficits in hippocampus-dependent long-term memory. Learn. Mem. 2012, 19, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Sultan, F.A.; Wang, J.; Tront, J.; Liebermann, D.A.; David Sweatt, J. Genetic deletion of gadd45b, a regulator of active DNA demethylation, enhances long-term memory and synaptic plasticity. J. Neurosci. 2012, 32, 17059–17066. [Google Scholar] [CrossRef]
- Apulei, J.; Kim, N.; Testa, D.; Ribot, J.; Morizet, D.; Bernard, C.; Jourdren, L.; Blugeon, C.; Di Nardo, A.A.; Prochiantz, A. Non-cell Autonomous OTX2 Homeoprotein Regulates Visual Cortex Plasticity Through Gadd45b/g. Cereb. Cortex 2019, 29, 2384–2395. [Google Scholar] [CrossRef]
- Djerbal, L.; Lortat-Jacob, H.; Kwok, J.C.F. Chondroitin sulfates and their binding molecules in the central nervous system. Glycoconjug. J. 2017, 34, 363–376. [Google Scholar] [CrossRef] [PubMed]
- Vallet, S.D.; Clerc, O.; Ricard-Blum, S. Glycosaminoglycan–Protein Interactions: The First Draft of the Glycosaminoglycan Interactome. J. Histochem. Cytochem. 2020, 69, 93–104. [Google Scholar] [CrossRef]
- Sugahara, K.; Mikami, T. Chondroitin/dermatan sulfate in the central nervous system. Curr. Opin. Struct. Biol. 2007, 17, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, C.S.; Gotthard, I.; Wyatt, E.V.; Bongu, S.; Mohan, V.; Weinberg, R.J.; Maness, P.F. Perineuronal Net Protein Neurocan Inhibits NCAM/EphA3 Repellent Signaling in GABAergic Interneurons. Sci. Rep. 2018, 8, 6143. [Google Scholar] [CrossRef] [PubMed]
- Orlando, C.; Ster, J.; Gerber, U.; Fawcett, J.W.; Raineteau, O. Perisynaptic chondroitin sulfate proteoglycans restrict structural plasticity in an integrin-dependent manner. J. Neurosci. 2012, 32, 18009–18017. [Google Scholar] [CrossRef] [PubMed]
- De Vivo, L.; Landi, S.; Panniello, M.; Baroncelli, L.; Chierzi, S.; Mariotti, L.; Spolidoro, M.; Pizzorusso, T.; Maffei, L.; Ratto, G.M. Extracellular matrix inhibits structural and functional plasticity of dendritic spines in the adult visual cortex. Nat. Commun. 2013, 4, 1484. [Google Scholar] [CrossRef]
- Tan, C.L.; Kwok, J.C.F.; Patani, R.; Ffrench-Constant, C.; Chandran, S.; Fawcett, J.W. Integrin activation promotes axon growth on inhibitory chondroitin sulfate proteoglycans by enhancing integrin signaling. J. Neurosci. 2011, 31, 6289–6295. [Google Scholar] [CrossRef]
- Favuzzi, E.; Marques-Smith, A.; Deogracias, R.; Winterflood, C.M.; Sánchez-Aguilera, A.; Mantoan, L.; Maeso, P.; Fernandes, C.; Ewers, H.; Rico, B. Activity-Dependent Gating of Parvalbumin Interneuron Function by the Perineuronal Net Protein Brevican. Neuron 2017, 95, 639–655. [Google Scholar] [CrossRef]
- Frischknecht, R.; Heine, M.; Perrais, D.; Seidenbecher, C.I.; Choquet, D.; Gundelfinger, E.D. Brain extracellular matrix affects AMPA receptor lateral mobility and short-term synaptic plasticity. Nat. Neurosci. 2009, 12, 897–904. [Google Scholar] [CrossRef]
- Senkov, O.; Andjus, P.; Radenovic, L.; Soriano, E.; Dityatev, A. Neural ECM molecules in synaptic plasticity, learning, and memory. Progr. Brain Res. 2014, 214, 53–80. [Google Scholar]
- Fisher, D.; Xing, B.; Dill, J.; Li, H.; Hiep Hoang, H.; Zhao, Z.; Yang, X.-L.; Bachoo, R.; Cannon, S.; Longo, F.M.; et al. Leukocyte common antigen-related phosphatase is a functional receptor for chondroitin sulfate proteoglycan axon growth inhibitors. J. Neurosci. 2011, 31, 14051–14066. [Google Scholar] [CrossRef] [PubMed]
- Lang, B.T.; Cregg, J.M.; DePaul, M.A.; Tran, A.P.; Xu, K.; Dyck, S.M.; Madalena, K.M.; Brown, B.P.; Weng, Y.-L.; Li, S.; et al. Modulation of the proteoglycan receptor PTPσ promotes recovery after spinal cord injury. Nature 2015, 518, 404–408. [Google Scholar] [CrossRef]
- Dickendesher, T.L.; Baldwin, K.T.; Mironova, Y.A.; Koriyama, Y.; Raiker, S.J.; Askew, K.L.; Wood, A.; Geoffroy, C.G.; Zheng, B.; Liepmann, C.D.; et al. NgR1 and NgR3 are receptors for chondroitin sulfate proteoglycans. Nat. Neurosci. 2012, 15, 703–712. [Google Scholar] [CrossRef]
- Golgi, C. Intorno alla struttura delle cellule nervose. Boll. Soc. Med. Chir. Pavia 1898, 13, 3–16. [Google Scholar]
- Ramón y Cajal, S. Histologie du Systeme Nerveux de l’Homme et Des Vertebres; Maloine: Paris, France, 1909. [Google Scholar]
- Rambourg, A.; Neutra, M.; Leblond, C.P. Presence of a ‘cell coat’ rich in carbohydrate at the surface of cells in the rat. Anat. Rec. 1966, 154, 41–71. [Google Scholar] [CrossRef]
- Glegg, R.E.; Pearce, R.H. Chemical extraction of metachromatic and periodic acid-Schiff positive carbohydrates from cerebral tissue. J. Comp. Neurol. 1956, 106, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Brauer, K.; Brückner, G.; Leibnitz, L.; Werner, L. Structural and cytochemical features of perineuronal glial nets in the rat brain. Acta Histochem. 1984, 74, 53–60. [Google Scholar] [CrossRef]
- Steindler, D.A.; Cooper, N.G.F. Wheat germ agglutinin binding sites in the adult mouse cerebellum: Light and electron microscopic studies. J. Comp. Neurol. 1986, 249, 170–185. [Google Scholar] [CrossRef]
- Brückner, G.; Brauer, K.; Härtig, W.; Wolff, J.R.; Rickmann, M.J.; Derouiche, A.; Delpech, B.; Girard, N.; Oertel, W.H.; Reichenbach, A. Perineuronal nets provide a polyanionic, glia-associated form of microenvironment around certain neurons in many parts of the rat brain. Glia 1993, 8, 183–200. [Google Scholar] [CrossRef]
- Sale, A.; Maya Vetencourt, J.F.; Medini, P.; Cenni, M.C.; Baroncelli, L.; De Pasquale, R.; Maffei, L. Environmental enrichment in adulthood promotes amblyopia recovery through a reduction of intracortical inhibition. Nat. Neurosci. 2007, 10, 679–681. [Google Scholar] [CrossRef]
- Foscarin, S.; Ponchione, D.; Pajaj, E.; Leto, K.; Gawlak, M.; Wilczynski, G.M.; Rossi, F.; Carulli, D. Experience-dependent plasticity and modulation of growth regulatory molecules at central synapses. PLoS ONE 2011, 6, e16666. [Google Scholar] [CrossRef]
- Donato, F.; Rompani, S.B.; Caroni, P. Parvalbumin-expressing basket-cell network plasticity induced by experience regulates adult learning. Nature 2013, 504, 272–276. [Google Scholar] [CrossRef]
- Sale, A.; Berardi, N.; Maffei, L. Environment and brain plasticity: Towards an endogenous pharmacotherapy. Physiol. Rev. 2014, 94, 189–234. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.C.; Mauricio, R.; Nobre, L.; Marsh, B.; Wüst, R.C.I.; Rossiter, H.B.; Ichiyama, R.M. Differential regulation of perineuronal nets in the brain and spinal cord with exercise training. Brain Res. Bull. 2015, 111, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Miyata, S.; Akagi, A.; Hayashi, N.; Watanabe, K.; Oohira, A. Activity-dependent regulation of a chondroitin sulfate proteoglycan 6B4 phosphacan/RPTPβ in the hypothalamic supraoptic nucleus. Brain Res. 2004, 1017, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Theodosis, D.T.; Poulain, D.A. Activity-dependent neuronal-glial and synaptic plasticity in the adult mammalian hypothalamus. Neuroscience 1993, 57, 501–535. [Google Scholar] [CrossRef]
- Maya-Vetencourt, J.F.; Sale, A.; Viegi, A.; Baroncelli, L.; De Pasquale, R.; O’Leary, O.F.; Castrén, E.; Maffei, L. The antidepressant fluoxetine restores plasticity in the adult visual cortex. Science 2008, 320, 385–388. [Google Scholar] [CrossRef]
- Guirado, R.; Perez-Rando, M.; Sanchez-Matarredona, D.; Castrén, E.; Nacher, J. Chronic fluoxetine treatment alters the structure, connectivity and plasticity of cortical interneurons. Int. J. Neuropsychopharmacol. 2014, 17, 1635–1646. [Google Scholar] [CrossRef] [PubMed]
- Popova, D.; Castrén, E.; Taira, T. Chronic fluoxetine administration enhances synaptic plasticity and increases functional dynamics in hippocampal CA3-CA1 synapses. Neuropharmacology 2017, 126, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Harauzov, A.; Spolidoro, M.; DiCristo, G.; De Pasquale, R.; Cancedda, L.; Pizzorusso, T.; Viegi, A.; Berardi, N.; Maffei, L. Reducing Intracortical Inhibition in the Adult Visual Cortex Promotes Ocular Dominance Plasticity. J. Neurosci. 2010, 30, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Pantazopoulos, H.; Gisabella, B.; Rexrode, L.; Benefield, D.; Yildiz, E.; Seltzer, P.; Valeri, J.; Chelini, G.; Reich, A.; Ardelt, M.; et al. Circadian rhythms of perineuronal net composition. Eneuro 2020, 7, 1–21. [Google Scholar] [CrossRef]
- Chaudhury, D.; Wang, L.M.; Colwell, C.S. Circadian regulation of hippocampal long-term potentiation. J. Biol. Rhythms 2005, 20, 225–236. [Google Scholar] [CrossRef]
- Rasch, B.; Born, J. About sleep’s role in memory. Physiol. Rev. 2013, 93, 681–766. [Google Scholar] [CrossRef] [PubMed]
- Havekes, R.; Park, A.J.; Tudor, J.C.; Luczak, V.G.; Hansen, R.T.; Ferri, S.L.; Bruinenberg, V.M.; Poplawski, S.G.; Day, J.P.; Aton, S.J.; et al. Sleep deprivation causes memory deficits by negatively impacting neuronal connectivity in hippocampal area CA1. Elife 2016, 5, e13424. [Google Scholar] [CrossRef]
- Raven, F.; Meerlo, P.; Van der Zee, E.A.; Abel, T.; Havekes, R. A brief period of sleep deprivation causes spine loss in the dentate gyrus of mice. Neurobiol. Learn. Mem. 2019, 160, 83–90. [Google Scholar] [CrossRef]
- Morawski, M.; Brückner, M.K.; Riederer, P.; Brückner, G.; Arendt, T. Perineuronal nets potentially protect against oxidative stress. Exp. Neurol. 2004, 188, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Cabungcal, J.-H.; Steullet, P.; Morishita, H.; Kraftsik, R.; Cuenod, M.; Hensch, T.K.; Do, K.Q. Perineuronal nets protect fast-spiking interneurons against oxidative stress. Proc. Natl. Acad. Sci. USA 2013, 110, 9130–9135. [Google Scholar] [CrossRef] [PubMed]
- Suttkus, A.; Rohn, S.; Weigel, S.; Glöckner, P.; Arendt, T.; Morawski, M. Aggrecan, link protein and tenascin-R are essential components of the perineuronal net to protect neurons against iron-induced oxidative stress. Cell Death Dis. 2014, 5, e1119. [Google Scholar] [CrossRef]
- Harkness, J.H.; Bushana, P.N.; Todd, R.P.; Clegern, W.C.; Sorg, B.A.; Wisor, J.P. Sleep disruption elevates oxidative stress in parvalbumin-positive cells of the rat cerebral cortex. Sleep 2019, 42, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Faralli, A.; Dagna, F.; Albera, A.; Bekku, Y.; Oohashi, T.; Albera, R.; Rossi, F.; Carulli, D. Modifications of perineuronal nets and remodelling of excitatory and inhibitory afferents during vestibular compensation in the adult mouse. Brain Struct. Funct. 2016, 221, 3193–3209. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, S.T.; Archibeque, I.; Luke, L.; Nolan, T.; Momiy, J.; Li, S. Growth-associated gene expression after stroke: Evidence for a growth-promoting region in peri-infarct cortex. Exp. Neurol. 2005, 193, 291–311. [Google Scholar] [CrossRef]
- Carulli, D.; Foscarin, S.; Faralli, A.; Pajaj, E.; Rossi, F. Modulation of semaphorin3A in perineuronal nets during structural plasticity in the adult cerebellum. Mol. Cell. Neurosci. 2013, 57, 10–22. [Google Scholar] [CrossRef]
- Sánchez-Ventura, J.; Giménez-Llort, L.; Penas, C.; Udina, E. Voluntary wheel running preserves lumbar perineuronal nets, enhances motor functions and prevents hyperreflexia after spinal cord injury. Exp. Neurol. 2021, 336, 113533. [Google Scholar] [CrossRef]
- Banerjee, S.B.; Gutzeit, V.A.; Baman, J.; Aoued, H.S.; Doshi, N.K.; Liu, R.C.; Ressler, K.J. Perineuronal Nets in the Adult Sensory Cortex Are Necessary for Fear Learning. Neuron 2017, 95, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Wei, X.; Wang, X.; Du, S.; Liu, W.; Song, J.; Wang, Y. Perineuronal nets protect long-term memory by limiting activity-dependent inhibition from parvalbumin interneurons. Proc. Natl. Acad. Sci. USA 2019, 116, 27063–27073. [Google Scholar] [CrossRef] [PubMed]
- Carulli, D.; Broersen, R.; de Winter, F.; Muir, E.M.; Mešković, M.; de Waal, M.; de Vries, S.; Boele, H.-J.; Canto, C.B.; De Zeeuw, C.I.; et al. Cerebellar plasticity and associative memories are controlled by perineuronal nets. Proc. Natl. Acad. Sci. USA 2020, 117, 6855–6865. [Google Scholar] [CrossRef]
- Lasek, A.W.; Chen, H.; Chen, W.Y. Releasing Addiction Memories Trapped in Perineuronal Nets. Trends Genet. 2018, 34, 197–208. [Google Scholar] [CrossRef] [PubMed]
- Miquel, M.; Vazquez-Sanroman, D.; Carbo-Gas, M.; Gil-Miravet, I.; Sanchis-Segura, C.; Carulli, D.; Manzo, J.; Coria-Avila, G.A. Have we been ignoring the elephant in the room? Seven arguments for considering the cerebellum as part of addiction circuitry. Neurosci. Biobehav. Rev. 2016, 60, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Sanroman, D.; Leto, K.; Cerezo-Garcia, M.; Carbo-Gas, M.; Sanchis-Segura, C.; Carulli, D.; Rossi, F.; Miquel, M. The cerebellum on cocaine: Plasticity and metaplasticity. Addict. Biol. 2015, 20, 941–955. [Google Scholar] [CrossRef]
- Vazquez-Sanroman, D.; Carbo-Gas, M.; Leto, K.; Cerezo-Garcia, M.; Gil-Miravet, I.; Sanchis-Segura, C.; Carulli, D.; Rossi, F.; Miquel, M. Cocaine-induced plasticity in the cerebellum of sensitised mice. Psychopharmacology 2015, 232, 4455–4467. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Hernandez, A.; Nicolas, C.; Gil-Miravet, I.; Guarque-Chabrera, J.; Solinas, M.; Miquel, M. Time-dependent regulation of perineuronal nets in the cerebellar cortex during abstinence of cocaine-self administration. Psychopharmacologym 2021. [Google Scholar] [CrossRef]
- Carbo-Gas, M.; Moreno-Rius, J.; Guarque-Chabrera, J.; Vazquez-Sanroman, D.; Gil-Miravet, I.; Carulli, D.; Hoebeek, F.; De Zeeuw, C.; Sanchis-Segura, C.; Miquel, M. Cerebellar perineuronal nets in cocaine-induced pavlovian memory: Site matters. Neuropharmacology 2017, 125, 166–180. [Google Scholar] [CrossRef]
- Van Den Oever, M.C.; Lubbers, B.R.; Goriounova, N.A.; Li, K.W.; Van der Schors, R.C.; Loos, M.; Riga, D.; Wiskerke, J.; Binnekade, R.; Stegeman, M.; et al. Extracellular matrix plasticity and GABAergic inhibition of prefrontal cortex pyramidal cells facilitates relapse to heroin seeking. Neuropsychopharmacology 2010, 35, 2120–2133. [Google Scholar] [CrossRef] [PubMed]
- Slaker, M.L.; Jorgensen, E.T.; Hegarty, D.M.; Liu, X.; Kong, Y.; Zhang, F.; Linhardt, R.J.; Brown, T.E.; Aicher, S.A.; Sorg, B.A. Cocaine Exposure Modulates Perineuronal Nets and Synaptic Excitability of Fast-Spiking Interneurons in the Medial Prefrontal Cortex. Eneuro 2018, 5. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; He, D.; Lasek, A.W. Repeated Binge Drinking Increases Perineuronal Nets in the Insular Cortex. Alcohol. Clin. Exp. Res. 2015, 39, 1930–1938. [Google Scholar] [CrossRef]
- Cunha-Oliveira, T.; Silva, L.; Silva, A.M.; Moreno, A.J.; Oliveira, C.R.; Santos, M.S. Mitochondrial complex I dysfunction induced by cocaine and cocaine plus morphine in brain and liver mitochondria. Toxicol. Lett. 2013, 219, 298–306. [Google Scholar] [CrossRef]
- Kochlamazashvili, G.; Henneberger, C.; Bukalo, O.; Dvoretskova, E.; Senkov, O.; Lievens, P.M.-J.; Westenbroek, R.; Engel, A.K.; Catterall, W.A.; Rusakov, D.A.; et al. The extracellular matrix molecule hyaluronic acid regulates hippocampal synaptic plasticity by modulating postsynaptic L-type Ca2+ channels. Neuron 2010, 67, 116–128. [Google Scholar] [CrossRef]
- Hylin, M.J.; Orsi, S.A.; Moore, A.N.; Dash, P.K. Disruption of the perineuronal net in the hippocampus or medial prefrontal cortex impairs fear conditioning. Learn. Mem. 2013, 20, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Thompson, E.H.; Lensjø, K.K.; Wigestrand, M.B.; Malthe-Sørenssen, A.; Hafting, T.; Fyhn, M. Removal of perineuronal nets disrupts recall of a remote fear memory. Proc. Natl. Acad. Sci. USA 2018, 115, 607–612. [Google Scholar] [CrossRef] [PubMed]
- Romberg, C.; Yang, S.; Melani, R.; Andrews, M.R.; Horner, A.E.; Spillantini, M.G.; Bussey, T.J.; Fawcett, J.W.; Pizzorusso, T.; Saksida, L.M. Depletion of Perineuronal Nets Enhances Recognition Memory and Long-Term Depression in the Perirhinal Cortex. J. Neurosci. 2013, 33, 7057–7065. [Google Scholar] [CrossRef]
- Hirono, M.; Watanabe, S.; Karube, F.; Fujiyama, F.; Kawahara, S.; Nagao, S.; Yanagawa, Y.; Misonou, H. Perineuronal Nets in the Deep Cerebellar Nuclei Regulate GABAergic Transmission and Delay Eyeblink Conditioning. J. Neurosci. 2018, 38, 6130–6144. [Google Scholar] [CrossRef] [PubMed]
- Bertocchi, I.; Mele, P.; Ferrero, G.; Oberto, A.; Carulli, D.; Eva, C. NPY-Y1 receptor signaling controls spatial learning and perineuronal net expression. Neuropharmacology 2021, 184, 108425. [Google Scholar] [CrossRef]
- Kaczmarek, L.; Lapinska-Dzwonek, J.; Szymczak, S. Matrix metalloproteinases in the adult brain physiology: A link between c-Fos, AP-1 and remodeling of neuronal connections? EMBO J. 2002, 21, 6643–6648. [Google Scholar] [CrossRef]
- Verslegers, M.; Lemmens, K.; Van Hove, I.; Moons, L. Matrix metalloproteinase-2 and -9 as promising benefactors in development, plasticity and repair of the nervous system. Prog. Neurobiol. 2013, 105, 60–78. [Google Scholar] [CrossRef]
- Howell, M.D.; Gottschall, P.E. Lectican proteoglycans, their cleaving metalloproteinases, and plasticity in the central nervous system extracellular microenvironment. Neuroscience 2012, 217, 6–18. [Google Scholar] [CrossRef] [PubMed]
- Stamenkovic, V.; Stamenkovic, S.; Jaworski, T.; Gawlak, M.; Jovanovic, M.; Jakovcevski, I.; Wilczynski, G.M.; Kaczmarek, L.; Schachner, M.; Radenovic, L.; et al. The extracellular matrix glycoprotein tenascin-C and matrix metalloproteinases modify cerebellar structural plasticity by exposure to an enriched environment. Brain Struct. Funct. 2017, 222, 393–415. [Google Scholar] [CrossRef] [PubMed]
- Murase, S.; Lantz, C.L.; Quinlan, E.M. Light reintroduction after dark exposure reactivates plasticity in adults via perisynaptic activation of MMP-9. Elife 2017, 6, e27345. [Google Scholar] [CrossRef]
- Ganguly, K.; Rejmak, E.; Mikosz, M.; Nikolaev, E.; Knapska, E.; Kaczmarek, L. Matrix metalloproteinase (MMP) 9 transcription in mouse brain induced by fear learning. J. Biol. Chem. 2013, 288, 20978–20991. [Google Scholar] [CrossRef]
- Dubey, D.; McRae, P.A.; Rankin-Gee, E.K.; Baranov, E.; Wandrey, L.; Rogers, S.; Porter, B.E. Increased metalloproteinase activity in the hippocampus following status epilepticus. Epilepsy Res. 2017, 132, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Yuan, W.; Matthews, R.T.; Sandy, J.D.; Gottschall, P.E. Association between protease-specific proteolytic cleavage of brevican and synaptic loss in the dentate gyrus of kainate-treated rats. Neuroscience 2002, 114, 1091–1101. [Google Scholar] [CrossRef]
- Rossier, J.; Bernard, A.; Cabungcal, J.-H.; Perrenoud, Q.; Savoye, A.; Gallopin, T.; Hawrylycz, M.; Cuénod, M.; Do, K.; Urban, A.; et al. Cortical fast-spiking parvalbumin interneurons enwrapped in the perineuronal net express the metallopeptidases Adamts8, Adamts15 and Neprilysin. Mol. Psychiatry 2015, 20, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.T.; Dorman, L.C.; Pan, S.; Vainchtein, I.D.; Han, R.T.; Nakao-Inoue, H.; Taloma, S.E.; Barron, J.J.; Molofsky, A.B.; Kheirbek, M.A.; et al. Microglial Remodeling of the Extracellular Matrix Promotes Synapse Plasticity. Cell 2020, 182, 388–403. [Google Scholar] [CrossRef]
- Bolós, M.; Perea, J.R.; Terreros-Roncal, J.; Pallas-Bazarra, N.; Jurado-Arjona, J.; Ávila, J.; Llorens-Martín, M. Absence of microglial CX3CR1 impairs the synaptic integration of adult-born hippocampal granule neurons. Brain. Behav. Immun. 2018, 68, 76–89. [Google Scholar] [CrossRef] [PubMed]
- Crapser, J.D.; Ochaba, J.; Soni, N.; Reidling, J.C.; Thompson, L.M.; Green, K.N. Microglial depletion prevents extracellular matrix changes and striatal volume reduction in a model of Huntington’s disease. Brain 2020, 143, 266–288. [Google Scholar] [CrossRef]
- Karetko-Sysa, M.; Skangiel-Kramska, J.; Nowicka, D. Aging somatosensory cortex displays increased density of WFA-binding perineuronal nets associated with GAD-negative neurons. Neuroscience 2014, 277, 734–746. [Google Scholar] [CrossRef] [PubMed]
- Végh, M.J.; Rausell, A.; Loos, M.; Heldring, C.M.; Jurkowski, W.; van Nierop, P.; Paliukhovich, I.; Li, K.W.; del Sol, A.; Smit, A.B.; et al. Hippocampal extracellular matrix levels and stochasticity in synaptic protein expression increase with age and are associated with age-dependent cognitive decline. Mol. Cell. Proteom. 2014, 13, 2975–2985. [Google Scholar] [CrossRef] [PubMed]
- Ueno, H.; Takao, K.; Suemitsu, S.; Murakami, S.; Kitamura, N.; Wani, K.; Okamoto, M.; Aoki, S.; Ishihara, T. Age-dependent and region-specific alteration of parvalbumin neurons and perineuronal nets in the mouse cerebral cortex. Neurochem. Int. 2018, 112, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Mizoguchi, K. Influence of aging on chondroitin sulfate proteoglycan expression and neural stem/progenitor cells in rat brain and improving effects of a herbal medicine, yokukansan. Neuroscience 2009, 164, 1224–1234. [Google Scholar] [CrossRef] [PubMed]
- Richard, A.D.; Tian, X.L.; El-Saadi, M.W.; Lu, X.H. Erasure of striatal chondroitin sulfate proteoglycan-associated extracellular matrix rescues aging-dependent decline of motor learning. Neurobiol. Aging 2018, 71, 61–71. [Google Scholar] [CrossRef]
- Gray, E.; Thomas, T.L.; Betmouni, S.; Scolding, N.; Love, S. Elevated matrix metalloproteinase-9 and degradation of perineuronal nets in cerebrocortical multiple sclerosis plaques. J. Neuropathol. Exp. Neurol. 2008, 67, 888–899. [Google Scholar] [CrossRef]
- Pantazopoulos, H.; Berretta, S. In sickness and in health: Perineuronal nets and synaptic plasticity in psychiatric disorders. Neural Plast. 2016, 2016, 9847696. [Google Scholar] [CrossRef]
- Testa, D.; Prochiantz, A.; Di Nardo, A.A. Perineuronal nets in brain physiology and disease. Semin. Cell Dev. Biol. 2019, 89, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Spijker, S.; Koskinen, M.K.; Riga, D. Incubation of depression: ECM assembly and parvalbumin interneurons after stress. Neurosci. Biobehav. Rev. 2020, 118, 65–79. [Google Scholar] [CrossRef] [PubMed]
- Braak, H.; Braak, E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 1991, 82, 239–259. [Google Scholar] [CrossRef] [PubMed]
- Goedert, M.; Sisodia, S.S.; Price, D.L. Neurofibrillary tangles and β-amyloid deposits in Alzheimer’s disease. Curr. Opin. Neurobiol. 1991, 1, 441–447. [Google Scholar] [CrossRef]
- Kobayashi, K.; Emson, P.C.; Mountjoy, C.Q. Vicia villosa lectin-positive neurones in human cerebral cortex. Loss in Alzheimer-type dementia. Brain Res. 1989, 498, 170–174. [Google Scholar] [CrossRef]
- Baig, S.; Wilcock, G.K.; Love, S. Loss of perineuronal net N-acetylgalactosamine in Alzheimer’s disease. Acta Neuropathol. 2005, 110, 393–401. [Google Scholar] [CrossRef]
- Crapser, J.D.; Spangenberg, E.E.; Barahona, R.A.; Arreola, M.A.; Hohsfield, L.A.; Green, K.N. Microglia facilitate loss of perineuronal nets in the Alzheimer’s disease brain. EBioMedicine 2020, 58, 102919. [Google Scholar] [CrossRef]
- Cattaud, V.; Bezzina, C.; Rey, C.C.; Lejards, C.; Dahan, L.; Verret, L. Early disruption of parvalbumin expression and perineuronal nets in the hippocampus of the Tg2576 mouse model of Alzheimer’s disease can be rescued by enriched environment. Neurobiol. Aging 2018, 72, 147–158. [Google Scholar] [CrossRef]
- Morawski, M.; Brückner, G.; Jäger, C.; Seeger, G.; Arendt, T. Neurons associated with aggrecan-based perineuronal nets are protected against tau pathology in subcortical regions in Alzheimer’s disease. Neuroscience 2010, 169, 1347–1363. [Google Scholar] [CrossRef] [PubMed]
- Morawski, M.; Brückner, G.; Jäger, C.; Seeger, G.; Matthews, R.T.; Arendt, T. Involvement of perineuronal and perisynaptic extracellular matrix in Alzheimer’s disease neuropathology. Brain Pathol. 2012, 22, 547–561. [Google Scholar] [CrossRef] [PubMed]
- Végh, M.J.; Heldring, C.M.; Kamphuis, W.; Hijazi, S.; Timmerman, A.J.; Li, K.W.; van Nierop, P.; Mansvelder, H.D.; Hol, E.M.; Smit, A.B.; et al. Reducing hippocampal extracellular matrix reverses early memory deficits in a mouse model of Alzheimer’s disease. Acta Neuropathol. Commun. 2014, 2, 76. [Google Scholar] [CrossRef][Green Version]
- Miyata, S.; Nishimura, Y.; Nakashima, T. Perineuronal nets protect against amyloid β-protein neurotoxicity in cultured cortical neurons. Brain Res. 2007, 1150, 200–206. [Google Scholar] [CrossRef]
- Brückner, G.; Hausen, D.; Härtig, W.; Drlicek, M.; Arendt, T.; Brauer, K. Cortical areas abundant in extracellular matrix chondroitin sulphate proteoglycans are less affected by cytoskeletal changes in Alzheimer’s disease. Neuroscience 1999, 92, 791–805. [Google Scholar] [CrossRef]
- Suttkus, A.; Holzer, M.; Morawski, M.; Arendt, T. The neuronal extracellular matrix restricts distribution and internalization of aggregated Tau-protein. Neuroscience 2016, 313, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Holmes, B.B.; DeVos, S.L.; Kfoury, N.; Li, M.; Jacks, R.; Yanamandra, K.; Ouidja, M.O.; Brodsky, F.M.; Marasa, J.; Bagchi, D.P.; et al. Heparan sulfate proteoglycans mediate internalization and propagation of specific proteopathic seeds. Proc. Natl. Acad. Sci. USA 2013, 110, E3138–E3147. [Google Scholar] [CrossRef]
- Yang, S.; Cacquevel, M.; Saksida, L.M.; Bussey, T.J.; Schneider, B.L.; Aebischer, P.; Melani, R.; Pizzorusso, T.; Fawcett, J.W.; Spillantini, M.G. Perineuronal net digestion with chondroitinase restores memory in mice with tau pathology. Exp. Neurol. 2015, 265, 48–58. [Google Scholar] [CrossRef]
- Gawin, F.H.; Kleber, H.D. Abstinence Symptomatology and Psychiatric Diagnosis in Cocaine Abusers: Clinical Observations. Arch. Gen. Psychiatry 1986, 43, 107–113. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, C.P.; Childress, A.R.; Ehrman, R.; Robbins, S.J. Conditioning factors in drug abuse: Can they explain compulsion? J. Psychopharmacol. 1998, 12, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Slaker, M.; Blacktop, J.M.; Sorg, B.A. Caught in the net: Perineuronal nets and addiction. Neural Plast. 2016, 2016, 7538208. [Google Scholar] [CrossRef]
- Slaker, M.; Churchill, L.; Todd, R.P.; Blacktop, J.M.; Zuloaga, D.G.; Raber, J.; Darling, R.A.; Brown, T.E.; Sorg, B.A. Removal of perineuronal nets in the medial prefrontal cortex impairs the acquisition and reconsolidation of a cocaine-induced conditioned place preference memory. J. Neurosci. 2015, 35, 4190–4202. [Google Scholar] [CrossRef]
- Blacktop, J.M.; Todd, R.P.; Sorg, B.A. Role of perineuronal nets in the anterior dorsal lateral hypothalamic area in the acquisition of cocaine-induced conditioned place preference and self-administration. Neuropharmacology 2017, 118, 124–136. [Google Scholar] [CrossRef] [PubMed]
- Blacktop, J.M.; Sorg, B.A. Perineuronal nets in the lateral hypothalamus area regulate cue-induced reinstatement of cocaine-seeking behavior. Neuropsychopharmacology 2019, 44, 850–858. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.-X.; Xue, L.-F.; Liu, J.-F.; He, J.; Deng, J.-H.; Sun, S.-C.; Han, H.-B.; Luo, Y.-X.; Xu, L.-Z.; Wu, P.; et al. Depletion of Perineuronal Nets in the Amygdala to Enhance the Erasure of Drug Memories. J. Neurosci. 2014, 34, 6647–6658. [Google Scholar] [CrossRef]
- Tsien, R.Y. Very long-term memories may be stored in the pattern of holes in the perineuronal net. Proc. Natl. Acad. Sci. USA 2013, 110, 12456–12461. [Google Scholar] [CrossRef]
- Duncan, J.A.; Foster, R.; Kwok, J.C.F. The potential of memory enhancement through modulation of perineuronal nets. Br. J. Pharmacol. 2019, 176, 3611–3621. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.R.; Muir, E.M.; Nuno Alves, J.; Rickman, H.; Allan, A.Y.; Kwok, J.C.F.; Roet, K.C.D.; Verhaagen, J.; Schneider, B.L.; Bensadoun, J.-C.; et al. Lentiviral vectors express chondroitinase ABC in cortical projections and promote sprouting of injured corticospinal axons. J. Neurosci. Methods 2011, 201, 228–238. [Google Scholar] [CrossRef]
- Bartus, K.; James, N.D.; Didangelos, A.; Bosch, K.D.; Verhaagen, J.; Yáñez-Muñoz, R.J.; Rogers, J.H.; Schneider, B.L.; Muir, E.M.; Bradbury, E.J. Large-Scale Chondroitin Sulfate Proteoglycan Digestion with Chondroitinase Gene Therapy Leads to Reduced Pathology and Modulates Macrophage Phenotype following Spinal Cord Contusion Injury. J. Neurosci. 2014, 34, 4822–4836. [Google Scholar] [CrossRef]
- James, N.D.; Shea, J.; Muir, E.M.; Verhaagen, J.; Schneider, B.L.; Bradbury, E.J. Chondroitinase gene therapy improves upper limb function following cervical contusion injury. Exp. Neurol. 2015, 271, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Burnside, E.R.; De Winter, F.; Didangelos, A.; James, N.D.; Andreica, E.-C.; Layard-Horsfall, H.; Muir, E.M.; Verhaagen, J.; Bradbury, E.J. Immune-evasive gene switch enables regulated delivery of chondroitinase after spinal cord injury. Brain 2018, 141, 2362–2381. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carulli, D.; Verhaagen, J. An Extracellular Perspective on CNS Maturation: Perineuronal Nets and the Control of Plasticity. Int. J. Mol. Sci. 2021, 22, 2434. https://doi.org/10.3390/ijms22052434
Carulli D, Verhaagen J. An Extracellular Perspective on CNS Maturation: Perineuronal Nets and the Control of Plasticity. International Journal of Molecular Sciences. 2021; 22(5):2434. https://doi.org/10.3390/ijms22052434
Chicago/Turabian StyleCarulli, Daniela, and Joost Verhaagen. 2021. "An Extracellular Perspective on CNS Maturation: Perineuronal Nets and the Control of Plasticity" International Journal of Molecular Sciences 22, no. 5: 2434. https://doi.org/10.3390/ijms22052434
APA StyleCarulli, D., & Verhaagen, J. (2021). An Extracellular Perspective on CNS Maturation: Perineuronal Nets and the Control of Plasticity. International Journal of Molecular Sciences, 22(5), 2434. https://doi.org/10.3390/ijms22052434




