Molecular Docking Studies and Biological Evaluation of Berberine–Benzothiazole Derivatives as an Anti-Influenza Agent via Blocking of Neuraminidase
Abstract
1. Introduction
2. Results and Discussion
2.1. Cytotoxicity and Anti-Influenza Activity
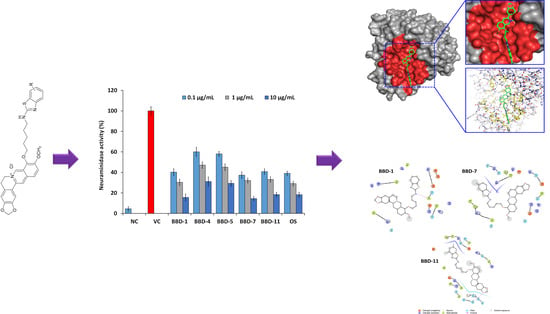
2.2. NA Inhibition Activity of BBDs
2.3. Molecular Interaction between BBD and NA Protein
3. Materials and Methods
3.1. Chemicals or Compounds
3.2. Reagents, Cells, and Viruses
3.3. Cytotoxicity
3.4. In vitro Antiviral Bioassay
3.5. Viral Neuraminidase Inhibition Assay
- NA activity (%) = (Treatment/Virus) × 100.
- Treatment: Virus + Compound
- Virus: Virus
3.6. Molecular Docking Study
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zambon, M.C. The pathogenesis of influenza in humans. Rev. Med. Virol. 2001, 11, 227–241. [Google Scholar] [CrossRef]
- World Health Organisation. Fact Sheet No. 211, Influenza. 2009. Available online: http://www.who.int/mediacentre/factsheets/fs211/en/ (accessed on 29 January 2021).
- World Health Organisation. Cumulative Number of Confirmed Human Cases for Avian Influenza A (H5N1) Reported to WHO, 2003–2013; World Health Organization: Geneva, Switzerland, 2013; pp. 1–2. [Google Scholar]
- Patel, M.; Dennis, A.; Flutter, C.; Khan, Z. Pandemic (H1N1) 2009 influenza. Br. J. Anaesth. 2010, 104, 128–142. [Google Scholar] [CrossRef]
- Fajardo-Dolci, G.; Gutierrez-Vega, R.; Arboleya-Casanova, H.; Villalobos, A.; Wilson, K.S.; García, S.G.; Sotelo, J.; Villalobos, J.A.C.; Díaz-Olavarrieta, C. Clinical characteristics of fatalities due to influenza A (H1N1) virus in Mexico. Thorax 2010, 65, 505–509. [Google Scholar] [CrossRef]
- Carrat, F.; Flahault, A. Influenza vaccine: The challenge of antigenic drift. Vaccine 2007, 25, 6852–6862. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation. Available online: http://www.who.int/news-room/fact-sheets/detail/influenza-(seasonal) (accessed on 29 January 2021).
- Wagner, R.; Matrosovich, M.; Klenk, H.D. Functional balance between haemagglutinin and neuraminidase in influenza virus infections. Rev. Med. Virol. 2002, 12, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Varghese, J.N.; Laver, W.G.; Colman, P.M. Structure of the influenza virus glycoprotein antigen neuraminidase at 2.9 Å resolution. Nature 1983, 303, 35–40. [Google Scholar] [CrossRef]
- Gong, J.; Xu, W.; Zhang, J. Structure and functions of influenza virus neuraminidase. Curr. Med. Chem. 2007, 14, 113–122. [Google Scholar] [CrossRef]
- Shen, Z.; Lou, K.; Wang, W. New small-molecule drug design strategies for fighting resistant influenza A. Acta Pharm. Sin. B 2015, 5, 419–430. [Google Scholar] [CrossRef]
- Shen, X.; Zhang, X.; Liu, S. Novel hemagglutinin-based influenza virus inhibitors. J. Thorac. Dis. 2013, 5 (Suppl. S2), S149–S159. [Google Scholar] [PubMed]
- Shin, H.B.; Choi, M.S.; Yi, C.M.; Lee, J.; Kim, N.J.; Inn, K.S. Inhibition of respiratory syncytial virus replication and virus-induced p38 kinase activity by berberine. Int. Immunopharmacol. 2015, 27, 65–68. [Google Scholar] [CrossRef]
- Song, S.; Qiu, M.; Chu, Y.; Chen, D.; Wang, X.; Su, A.; Wu, Z. Down regulation of cellular c-Jun N-terminal protein kinase and NF-kappa B activation by berberine may result in inhibition of herpes simplex virus replication. Antimicrob. Agents Chemother. 2014, 58, 5068–5078. [Google Scholar] [CrossRef]
- Gansukh, E.; Pandurangan, M.; Kim, D.H.; Mistry, B.M. Discovery of berberine based derivatives as anti-influenza agent through blocking of neuraminidase. Bioorg. Med. Chem. 2017, 25, 5185–5193. [Google Scholar]
- Gansukh, E.; Kim, D.H.; Gyun, S.P.; Pandurangan, M.; Daniel, A.N.; Moon, S.H.; Kadam, A.A.; Patel, R.V.; Shin, H.S.; Mistry, B.M. Berberine-piperazine conjugates as potent influenza neuraminidase blocker. Int. J. Biol. Macromol. 2018, 119, 1204–1210. [Google Scholar]
- Rabbani, G.H. Mechanism and treatment of diarrhoea due to vibrio cholerae and Escherichia coli: Roles of drugs and prostaglandins. Dan. Med. Bull. 1996, 43, 173–185. [Google Scholar]
- Chun, Y.T.; Yip, T.T.; Lau, K.L.; Kong, Y.C.; Sankawa, U. A biochemical study on the hypotensive effect of berberine in rats. Gen. Pharmacol. 1979, 10, 177–182. [Google Scholar] [CrossRef]
- Peng, L.; Kang, S.; Yin, Z.; Jia, R.; Song, X.; Li, L.; Li, Z.; Zou, Y.; Liang, X.; He, C.; et al. Antibacterial activity and mechanism of berberine against Streptococcus agalactiae. Int. J. Clin. Exp. Pathol. 2015, 8, 5217–5223. [Google Scholar]
- Liu, Q.; Xu, X.; Zhao, M.; Wei, Z.; Li, X.; Zhang, X.; Liu, Z.; Gong, Y.; Shao, C. Berberine induces senescence of human glioblastoma cells by downregulating the EGFR-MEK-ERK signaling pathway. Mol. Cancer Ther. 2015, 14, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Wang, J.; Hou, J.; Lin, C.; Bensoussan, A.; Chang, D.; Liu, J.; Wang, B. Berberine alleviates ox-LDL induced inflammatory factors by up-regulation of autophagy via AMPK/mTOR signaling pathway. J. Transl. Med. 2015, 13, 92. [Google Scholar] [CrossRef]
- Gansukh, E.; Muthu, M.; Paul, D.; Ethiraj, G.; Chun, S.; Gopal, J. Nature nominee quercetin’s anti-influenza combat strategy—Demonstrations and remonstrations. Rev. Med. Virol. 2017, 27, e1930. [Google Scholar] [CrossRef] [PubMed]
- Gansukh, E.; Anthonydhason, V.; Jung, S.; Kim, D.H.; Muthu, M.; Gopal, J.; Chun, S. Nanotherapeutic Anti-influenza Solutions: Current Knowledge and Future Challenges. J. Clust. Sci. 2018, 29, 933–941. [Google Scholar] [CrossRef]
- Xu, X.; Zhu, X.; Dwek, R.A.; Stevens, J.; Wilson, I.A. Structural characterization of the 1918 influenza virus H1N1 neuraminidase. J. Virol. 2008, 82, 10493–10501. [Google Scholar] [CrossRef] [PubMed]
- Mistry, B.M.; Patel, R.V.; Keum, Y.S.; Kim, D.H. Evaluation of the biological potencies of newly synthesized berberine derivatives bearing benzothiazole moieties with substituted functionalities. J. Saudi Chem. Soc. 2017, 21, 210–219. [Google Scholar] [CrossRef]
- Trott, O.; Olson, A.J. Software news and update AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [PubMed]




| BBD | “R” GROUP | MW | CC50 (µG/ML) a | A/PR/8/34 (H1N1) | A/VIC/3/75 (H3N2) | B/LEE/40 | B/MARYLAND/1/59 | AFFINITY (KCAL/MOL) | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IC50 (µg/mL) b | TI | IC50 (µg/mL) b | TI | IC50 (µg/mL) b | TI | IC50 (µg/mL) b | TI | |||||
| BBD 1 |  | 520 | 436.7 ± 2.635 | 25.20 ± 0.154 | 17.32 | 45.15 ± 1.75 | 9.672 | 55.46 ± 3.77 | 7.874 | 55.11 ± 2.65 | 7.924 | −7.9 |
| BBD 2 |  | 565 | 340.5 ± 1.180 | 30.92 ± 0.619 | 11.01 | 52.18 ± 2.07 | 6.525 | 61.72 ± 1.65 | 5.516 | 65.32 ± 1.88 | 5.212 | −6.9 |
| BBD 3 |  | 564 | 382.9 ± 2.964 | 35.94 ± 0.659 | 10.65 | 60.74 ± 1.06 | 6.303 | 53.89 ± 1.36 | 7.105 | 56.44 ± 1.06 | 6.784 | −7.1 |
| BBD 4 |  | 534 | 322.6 ± 2.543 | 37.27 ± 0.669 | 8.655 | 55.03 ± 0.87 | 5.862 | 43.24 ± 0.86 | 7.406 | 41.56 ± 0.67 | 7.762 | −7.5 |
| BBD 5 |  | 550 | 306.4 ± 3.612 | 39.54 ± 0.326 | 7.749 | 57.34 ± 2.13 | 5.343 | 36.94 ± 1.52 | 8.294 | 44.27 ± 2.06 | 6.921 | −7.6 |
| BBD 6 |  | 564 | 221.4 ± 1.180 | 42.77 ± 0.478 | 5.176 | 60.43 ± 1.35 | 3.663 | 38.76 ± 2.08 | 5.712 | 36.88 ± 2.15 | 6.003 | −5.7 |
| BBD 7 |  | 554 | 463.5 ± 3.386 | 24.28± 0.419 | 19.08 | 38.81 ± 2.51 | 11.94 | 81.42 ± 3.01 | 5.692 | 88.13 ± 1.16 | 5.259 | −8.4 |
| BBD 8 |  | 599 | 328.9 ± 1.065 | 68.02 ± 0.670 | 4.835 | 70.65 ± 0.94 | 4.65 | 74.82 ± 1.83 | 4.395 | 81.16 ± 0.91 | 4.052 | −7.1 |
| BBD 9 |  | 702 | 302.5 ± 1.771 | 54.53 ± 1.750 | 5.547 | 54.16 ± 2.09 | 5.585 | 69.84 ± 2.57 | 4.331 | 78.55 ± 2.31 | 3.851 | −7.1 |
| BBD 10 |  | 539 | 310.6 ± 1.629 | 30.50 ± 0.761 | 10.18 | 49.87 ± 1.31 | 6.228 | 64.64 ± 1.24 | 4.812 | 75.74 ± 2.06 | 4.100 | −6.8 |
| BBD 11 |  | 556 | 467.7 ± 2.647 | 27.20 ± 0.394 | 17.19 | 48.44 ± 2.20 | 9.655 | 85.12 ± 3.74 | 5.494 | 98.33 ± 2.21 | 4.75 | −8.0 |
| BBD 12 |  | 588 | 302.3 ± 2.446 | 28.73 ± 0.514 | 10.52 | 44.56 ± 1.65 | 6.784 | 67.65 ± 1.52 | 4.468 | 66.22 ± 1.82 | 4.565 | −6.0 |
| BBD 13 |  | 545 | 45.1 ± 0.864 | ND | - | ND | - | ND | - | ND | - | −6.7 |
| BBD 14 |  | 591 | 304.3 ± 1.261 | 40.34 ± 0.725 | 7.543 | 56.32 ± 2.06 | 5.403 | 70.32 ± 2.24 | 4.327 | 73.44 ± 2.17 | 4.143 | −6.7 |
| BERBERINE | 65.34 ± 1.92 | 36.12 ± 1.57 | 1.808 | 41.58 ± 2.04 | 1.571 | 60.83 ± 1.86 | 1.07 | 54.63 ± 1.45 | 1.196 | - | ||
| OSELTAMIVIR | 205.3 ± 1.78 | 11.56 ± 1.43 | 17.75 | 22.14 ± 1.07 | 9.27 | 55.87 ± 1.13 | 3.67 | 44.17 ± 1.83 | 4.64 | - | ||
| # | Affinity (kcal/mol) @ | RMSD L.B # | RMSD U.B * | |||
|---|---|---|---|---|---|---|
| BBD-7 | Ose | BBD-7 | Ose | BBD-7 | Ose | |
| 1 | −8.4 | −6.1 | 0 | 0 | 0 | 0 |
| 2 | −8.1 | −6.0 | 5.706 | 2.205 | 9.555 | 4.419 |
| 3 | −8 | −5.7 | 2.454 | 2.120 | 3.43 | 3.267 |
| 4 | −7.9 | −5.6 | 7.43 | 2.362 | 11.301 | 4.659 |
| 5 | −7.6 | −5.5 | 11.979 | 2.383 | 17.015 | 4.191 |
| 6 | −7.6 | −5.3 | 12.165 | 2.376 | 16.089 | 4.982 |
| 7 | −7.5 | −5.2 | 5.553 | 2.631 | 10.121 | 5.237 |
| 8 | −7.4 | −5.2 | 16.288 | 2.645 | 19.921 | 3.757 |
| 9 | −7.4 | −5.2 | 16.355 | 14.54 | 19.467 | 16.635 |
| Compounds | Residues |
|---|---|
| Oseltamivir | ARG116, ARG150, ARG368, ARG291, GLU276 |
| BBD-1 | ARG291, TYR344, TYR402, ARG399, ARG368, ILE427, LYS430, THR438, ARG223, GLU275, GLU276, GLU226, SER178, TRP177, GLU117, ARG116, LEU132, GLN134, ARG150, ARG154 |
| BBD-7 | SER367, ARG368, SER369, ARG399, TYR344, TYR402, ARG291, GLU276, ARG223, THR224, GLU226, TRP177, SER178, ARG150, ARG154, ARG116, GLU117, GLN134, ALA432, PRO431, LYS430, ILE427, THR438 |
| BBD-11 | ILE221, LEU222, ARG223, THR224, SER178, TRP177, GLU226, ARG150, SER151, TYR153, ARG154, GLU276, TYR402, GLU117, ARG116, PRO431, LYS430, THR438, TRP437, GLN134, GLY145, ASN144, SER143, SER136, HIS142 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, M.; Chung, S.-M.; Enkhtaivan, G.; Patel, R.V.; Shin, H.-S.; Mistry, B.M. Molecular Docking Studies and Biological Evaluation of Berberine–Benzothiazole Derivatives as an Anti-Influenza Agent via Blocking of Neuraminidase. Int. J. Mol. Sci. 2021, 22, 2368. https://doi.org/10.3390/ijms22052368
Kumar M, Chung S-M, Enkhtaivan G, Patel RV, Shin H-S, Mistry BM. Molecular Docking Studies and Biological Evaluation of Berberine–Benzothiazole Derivatives as an Anti-Influenza Agent via Blocking of Neuraminidase. International Journal of Molecular Sciences. 2021; 22(5):2368. https://doi.org/10.3390/ijms22052368
Chicago/Turabian StyleKumar, Manu, Sang-Min Chung, Ganuskh Enkhtaivan, Rahul V. Patel, Han-Seung Shin, and Bhupendra M. Mistry. 2021. "Molecular Docking Studies and Biological Evaluation of Berberine–Benzothiazole Derivatives as an Anti-Influenza Agent via Blocking of Neuraminidase" International Journal of Molecular Sciences 22, no. 5: 2368. https://doi.org/10.3390/ijms22052368
APA StyleKumar, M., Chung, S.-M., Enkhtaivan, G., Patel, R. V., Shin, H.-S., & Mistry, B. M. (2021). Molecular Docking Studies and Biological Evaluation of Berberine–Benzothiazole Derivatives as an Anti-Influenza Agent via Blocking of Neuraminidase. International Journal of Molecular Sciences, 22(5), 2368. https://doi.org/10.3390/ijms22052368