Cyanidin 3-O-galactoside: A Natural Compound with Multiple Health Benefits
Abstract
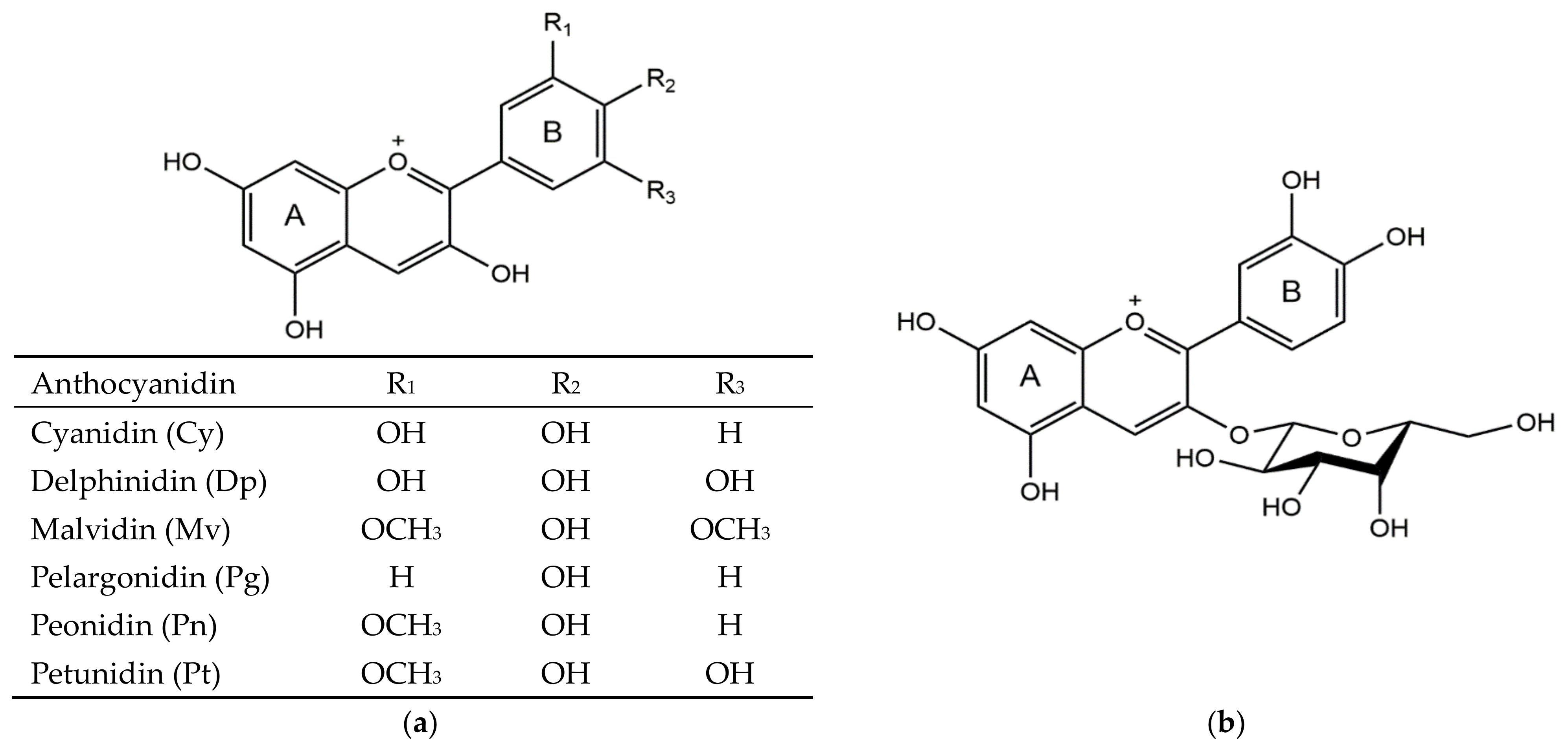
1. Introduction
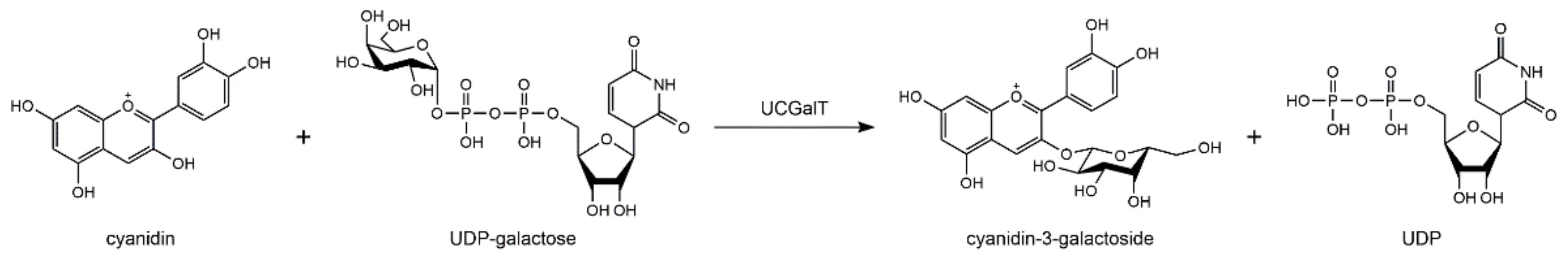
2. Biosynthesis
3. Chemical Stability
4. Natural Occurrences and Extracting Methods
5. Physiological Functions
5.1. In Vitro Studies
5.1.1. Acting Solely
5.1.2. Acting Synergistically
5.2. In Vivo Studies
5.2.1. Acting Solely
5.2.2. Acting Synergistically
6. Potential Applications
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Andersen, Ø.M.; Jordheim, M. Anthocyanins. In Encyclopedia of Life Sciences; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2002. [Google Scholar]
- Qiu, Z.; Wang, X.; Gao, J.; Guo, Y.; Huang, Z.; Du, Y.; Wu, K. The tomato hoffman’s anthocyaninless gene encodes a bHLH transcription factor involved in anthocyanin biosynthesis that is developmentally regulated and induced by low temperatures. PLoS ONE 2016, 11, e0151067. [Google Scholar]
- Breusegem, F.V.; Dat, J.F. Reactive oxygen species in plant cell death. Plant Physiol. 2006, 141, 384–390. [Google Scholar] [CrossRef]
- Merzlyak, M.N.; Chivkunova, O.B.; Solovchenko, A.E.; Razi, N.K. Light absorption by anthocyanins in juvenile, stressed, and senescing leaves. J. Exp. Bot. 2008, 59, 3903–3911. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L.; Wu, X. Anthocyanins: Structural characteristics that result in unique metabolic patterns and biological activities. Free Radic. Res. 2006, 40, 1014. [Google Scholar] [CrossRef] [PubMed]
- Kocic, B.; Filipovic, S.; Nikolic, M.; Petrovic, B. Effects of anthocyanins and anthocyanin-rich extracts on the risk for cancers of the gastrointestinal tract. J. Balk. Union Oncol. 2011, 16, 602–608. [Google Scholar]
- Aboonabi, A.; Singh, I. Chemopreventive role of anthocyanins in atherosclerosis via activation of Nrf2–ARE as an indicator and modulator of redox. Biomed. Pharmacother. 2015, 72, 30–36. [Google Scholar] [CrossRef]
- Cassidy, A. Berry anthocyanin intake and cardiovascular health. Mol. Aspects Med. 2018, 61, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Ling, W. The update of anthocyanins on obesity and type 2 diabetes: Experimental evidence and clinical perspectives. Rev. Endocr. Metab. Dis. 2015, 16, 1–13. [Google Scholar] [CrossRef]
- Galvano, F.; Fauci, L.L.; Vitaglione, P.; Fogliano, V.; Felgines, C. Bioavailability, antioxidant and biological properties of the natural free-radical scavengers cyanidin and related glycosides. Ann. Ist. Super. Sanita 2007, 43, 382–393. [Google Scholar] [PubMed]
- Sadilova, E.; Stintzing, F.C.; Carle, R. Chemical quality parameters and anthocyanin pattern of red-fleshed Weirouge apples. J. Appl. Bot. Food Qual. 2012, 80, 82–87. [Google Scholar]
- Rupasinghe, H.P.V.; Huber, G.M.; Embree, C.; Forsline, P.L. Red-fleshed apple as a source for functional beverages. Can. J. Plant Sci. 2010, 90, 95–100. [Google Scholar] [CrossRef]
- Liu, P.; Kallio, H.; Lü, D.; Zhou, C.; Yang, B. Quantitative analysis of phenolic compounds in Chinese hawthorn (Crataegus spp.) fruits by high performance liquid chromatography–electrospray ionisation mass spectrometry. Food Chem. 2011, 127, 1370–1377. [Google Scholar] [CrossRef] [PubMed]
- Baj, A.; Bombardelli, E.; Gabetta, B.; Martinelli, E.M. Qualitative and quantitative evaluation of Vaccinium myrtillus anthocyanins by high-resolution gas chromatography and high-performance liquid chromatography. J. Chromatogr. A 1983, 279, 365–372. [Google Scholar] [CrossRef]
- Ohnishi, R.; Ito, H.; Kasajima, N.; Kaneda, M.; Kariyama, R.; Kumon, H.; Hatano, T.; Yoshida, T. Urinary excretion of anthocyanins in humans after cranberry juice ingestion. Biosci. Biotech. Biochem. 2006, 70, 1681–1687. [Google Scholar] [CrossRef]
- Kokotkiewicz, A.; Jaremicz, Z.; Luczkiewicz, M. Aronia plants: A review of traditional use, biological activities, and perspectives for modern medicine. J. Med. Food 2010, 13, 255–269. [Google Scholar] [CrossRef] [PubMed]
- Lehtonen, H.M.; Rantala, M.; Suomela, J.P.; Viitanen, M.; Kallio, H. Urinary excretion of the main anthocyanin in lingonberry (Vaccinium vitis-idaea), cyanidin 3-O-galactoside, and its metabolites. J. Agric. Food Chem. 2009, 57, 4447–4451. [Google Scholar] [CrossRef]
- Skemiene, K.; Pampuscenko, K.; Rekuviene, E.; Borutaite, V. Protective effects of anthocyanins against brain ischemic damage. J. Bioenerg. Biomembr. 2020, 52, 1–12. [Google Scholar] [CrossRef]
- Wen, H.; Cui, H.; Tian, H.; Zhang, X.; Ma, L.; Ramassamy, C.; Li, J. Isolation of Neuroprotective Anthocyanins from Black Chokeberry (Aronia melanocarpa) against Amyloid-β-Induced Cognitive Impairment. Foods 2021, 10, 63. [Google Scholar] [CrossRef]
- Pervaiz, T.; Jiu, S.; Faghihi, F.; Haider, M.S.; Fang, J. Naturally occurring anthocyanin, structure, functions and biosynthetic pathway in fruit plants. J. Plant Biochem. Physiol. 2017, 5, 187. [Google Scholar] [CrossRef]
- Hassani, D.; Fu, X.; Shen, Q.; Khalid, M.; Rose, J.K.; Tang, K. Parallel transcriptional regulation of artemisinin and flavonoid biosynthesis. Trends Plant Sci. 2020, 25, 466–476. [Google Scholar] [CrossRef] [PubMed]
- Ban, Y.; Kondo, S.; Ubi, B.E.; Honda, C.; Bessho, H.; Moriguchi, T. UDP-sugar biosynthetic pathway: Contribution to cyanidin 3-galactoside biosynthesis in apple skin. Planta 2009, 230, 871–881. [Google Scholar] [CrossRef] [PubMed]
- Rose, A.; Gläßgen, W.E.; Hopp, W.; Seitz, H.U. Purification and characterization of glycosyltransferases involved in anthocyanin biosynthesis in cell-suspension cultures of Daucus carota L. Planta 1996, 198, 397–403. [Google Scholar] [CrossRef]
- Xu, Z.; Ma, J.; Wang, F.; Ma, H.; Wang, Q.; Xiong, A. Identification and characterization of DcUCGalT1, a galactosyltransferase responsible for anthocyanin galactosylation in purple carrot (Daucus carota L.) taproots. Sci. Rep. 2016, 6, 27356. [Google Scholar] [CrossRef] [PubMed]
- Feng, K.; Xu, Z.; Liu, J.; Li, J.; Wang, F.; Xiong, A. Isolation, purification, and characterization of AgUCGalT1, a galactosyltransferase involved in anthocyanin galactosylation in purple celery (Apium graveolens L.). Planta 2018, 247, 1363–1375. [Google Scholar] [CrossRef] [PubMed]
- Ginsburg, V. Comparative biochemistry of nucleotide-linked sugars. Prog. Clin. Biol. Res. 1978, 23, 595–600. [Google Scholar]
- Frey, P.A. The Leloir pathway: A mechanistic imperative for three enzymes to change the stereochemical configuration of a single carbon in galactose. FASEB J. 1996, 10, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Jaakola, L. New insights into the regulation of anthocyanin biosynthesis in fruits. Trends Plant Sci. 2013, 18, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Koes, R.; Verweij, W.; Quattrocchio, F. Flavonoids: A colorful model for the regulation and evolution of biochemical pathways. Trends Plant Sci. 2005, 10, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Jaakola, L.; Määttä, K.; Pirttilä, A.M.; Törrönen, R.; Kärenlampi, S.; Hohtola, A. Expression of genes involved in anthocyanin biosynthesis in relation to anthocyanin, proanthocyanidin, and flavonol levels during bilberry fruit development. Plant Physiol. 2002, 130, 729–739. [Google Scholar] [CrossRef]
- Pombo, M.A.; Martínez, G.A.; Civello, P.M. Cloning of FaPAL6 gene from strawberry fruit and characterization of its expression and enzymatic activity in two cultivars with different anthocyanin accumulation. Plant Sci. 2011, 181, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Cao, Y.; Ye, S.; Irshad, M.; Pan, T.; Qiu, D. Isolation of CHS gene from Brunfelsia acuminata flowers and its regulation in anthocyanin biosysthesis. Molecules 2017, 22, 44. [Google Scholar] [CrossRef]
- Wu, X.; Gong, Q.; Ni, X.; Zhou, Y.; Gao, Z. UFGT: The key enzyme associated with the petals variegation in Japanese apricot. Front. Plant Sci. 2017, 8, 108. [Google Scholar] [CrossRef] [PubMed]
- Griesser, M.; Hoffmann, T.; Bellido, M.L.; Rosati, C.; Fink, B.; Kurtzer, R.; Aharoni, A.; Munoz-Blanco, J.; Schwab, W. Redirection of flavonoid biosynthesis through the down-regulation of an anthocyanidin glucosyltransferase in ripening strawberry fruit. Plant Physiol. 2008, 146, 1528–1539. [Google Scholar] [CrossRef]
- Han, Y.; Vimolmangkang, S.; Soria-Guerra, R.E.; Rosales-Mendoza, S.; Zheng, D.; Lygin, A.V.; Korban, S.S. Ectopic expression of apple F3′ H genes contributes to anthocyanin accumulation in the Arabidopsis tt7 mutant grown under nitrogen stress. Plant Physiol. 2010, 153, 806–820. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Vimolmangkang, S.; Soria-Guerra, R.E.; Korban, S.S. Introduction of apple ANR genes into tobacco inhibits expression of both CHI and DFR genes in flowers, leading to loss of anthocyanin. J. Exp. Bot. 2012, 63, 2437–2447. [Google Scholar] [CrossRef]
- Allan, A.C.; Hellens, R.P.; Laing, W.A. MYB transcription factors that colour our fruit. Trends Plant Sci. 2008, 13, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Mato, M.; Ozeki, Y.; Itoh, Y.; Higeta, D.; Yoshitama, K.; Teramoto, S.; Aida, R.; Ishikura, N.; Shibata, M. Isolation and characterization of a cDNA clone of UDP-galactose: Flavonoid 3-O-galactosyltransferase (UF3GaT) expressed in Vigna mungo seedlings. Plant Cell Physiol. 1998, 39, 1145–1155. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Miller, K.D.; Guyon, V.; Evans, J.N.S.; Shuttleworth, W.A.; Taylor, L.P. Purification, cloning, and heterologous expression of a catalytically efficient flavonol 3-O-galactosyltransferase expressed in the male gametophyte of Petunia hybrida. J. Biol. Chem. 1999, 274, 34011–34019. [Google Scholar] [CrossRef]
- Liu, Y.; Zhou, B.; Qi, Y.; Liu, C.; Liu, Z.; Ren, X. Biochemical and functional characterization of AcUFGT3a, a galactosyltransferase involved in anthocyanin biosynthesis in the red-fleshed kiwifruit (Actinidia chinensis). Physiol. Plantarum 2018, 162, 409–426. [Google Scholar] [CrossRef]
- Cavalcanti, R.N.; Santos, D.T.; Meireles, M.A.A. Non-thermal stabilization mechanisms of anthocyanins in model and food systems—An overview. Food Res. Int. 2011, 44, 499–509. [Google Scholar] [CrossRef]
- Farr, J.E.; Sigurdson, G.T.; Giusti, M.M. Stereochemistry and glycosidic linkages of C3-glycosylations affected the reactivity of cyanidin derivatives. Food Chem. 2019, 278, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Chen, Z.; Bai, X.; Ding, C.; Long, T.; Wei, F.; Miao, K. Structure–activity relationships of anthocyanidin glycosylation. Mol. Divers. 2014, 18, 687–700. [Google Scholar] [CrossRef]
- Rehman, R.N.U.; You, Y.; Zhang, L.; Goudia, B.D.; Khan, A.R.; Li, P.; Ma, F. High temperature induced anthocyanin inhibition and active degradation in Malus profusion. Front. Plant Sci. 2017, 8, 1401. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.M.; Lee, K.M.; Jung, I.C. Changes in anthocyanin content of aronia (Aronia melancocarpa) by processing conditions. Korean J. Plant Resour. 2017, 30, 152–159. [Google Scholar] [CrossRef][Green Version]
- Oszmiański, J.; Wojdylo, A. Aronia melanocarpa phenolics and their antioxidant activity. Eur. Food Res. Technol. 2005, 221, 809–813. [Google Scholar] [CrossRef]
- Oszmianski, J.; Sapis, J.C. Anthocyanins in fruits of Aronia melanocarpa (chokeberry). J. Food Sci. 1988, 53, 1241–1242. [Google Scholar] [CrossRef]
- Lin, T.Y.; Koehler, P.E.; Shewfelt, R.L. Stability of anthocyanins in the skin of Starkrimson apples stored unpackaged, under heat shrinkable wrap and in-package modified atmosphere. J. Food Sci. 1989, 54, 405–407. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, G.; You, Y.; Yang, C.; Li, P.; Ma, F. Effects of relative air humidity on the phenolic compounds contents and coloration in the ‘Fuji’ apple (Malus domestica Borkh.) peel. Sci. Hortic. 2016, 201, 18–23. [Google Scholar] [CrossRef]
- Rein, M. Copigmentation Reactions and Color Stability of Berry Anthocyanins. Ph.D. Thesis, University of Helsinki, Helsinki, Finland, 2005; p. 87. [Google Scholar]
- Błaszczak, W.; Amarowicz, R.; Górecki, A.R. Antioxidant capacity, phenolic composition and microbial stability of aronia juice subjected to high hydrostatic pressure processing. Innov. Food Sci. Emerg. Technol. 2017, 39, 141–147. [Google Scholar] [CrossRef]
- Knebel, T.; Braun, P.; Dietrich, H. Degradation kinetics of anthocyanins and polyphenols during storage of red apple juice produced from red-fleshed apples. Eur. Food Res. Technol. 2018, 244, 1741–1750. [Google Scholar] [CrossRef]
- Su, F.; Xue, J.; Yang, X.; Deng, H.; Meng, Y.; Guo, Y. Effects of Phenolic Acids on Copigmentation and Stability of Anthocyanins in Red-Fleshed Apple. Sci. Agric. Sin. 2017, 50, 732–742. [Google Scholar]
- Saito, T.; Ishikura, H.; Hada, Y.; Fukui, K.; Kodera, Y.; Matsushim, A.; Inada, Y. Photostabilization of phycocyanin and anthocyanin in the presence of biopterin-α-glucoside from Spirulina platensis under ultraviolet ray. Dyes Pigments 2003, 56, 203–207. [Google Scholar] [CrossRef]
- Ngo, T.; Zhao, Y. Stabilization of anthocyanins on thermally processed red D’Anjou pears through complexation and polymerization. LWT-Food Sci. Technol. 2009, 42, 1144–1152. [Google Scholar] [CrossRef]
- Dussi, M.C.; Sugar, D.; Wrolstad, R.E. Characterizing and quantifying anthocyanins in red pears and the effect of light quality on fruit color. J. Am. Soc. Hortic. Sci. 1995, 120, 785–789. [Google Scholar] [CrossRef]
- Awad, M.A.; De Jager, A. Flavonoid and chlorogenic acid concentrations in skin of ‘Jonagold’ and ‘Elstar’ apples during and after regular and ultra low oxygen storage. Postharvest Biol. Technol. 2000, 20, 15–24. [Google Scholar] [CrossRef]
- Zheng, Y.; Wang, C.; Wang, S.; Zheng, W. Effect of high-oxygen atmospheres on blueberry phenolics, anthocyanins, and antioxidant capacity. J. Agric. Food Chem. 2003, 51, 7162–7169. [Google Scholar] [CrossRef] [PubMed]
- Starr, M.; Francis, F. Oxygen and ascorbic acid effect on relative stability of 4 anthocyanin pigments in cranberry juice. Food Technol. 1968, 22, 1293–1295. [Google Scholar]
- Attoe, E.; Von Elbe, J. Photochemial degradation of betanine and selected anthocyanins. J. Food Sci. 1981, 46, 1934–1937. [Google Scholar] [CrossRef]
- Ichiyanagi, T.; Oikawa, K.; Tateyama, C.; Konishi, T. Acid mediated hydrolysis of blueberry anthocyanins. Chem. Pharm. Bull. 2001, 49, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Sigurdson, G.T.; Robbins, R.J.; Collins, T.M.; Giusti, M.M. Impact of location, type, and number of glycosidic substitutions on the color expression of o-dihydroxylated anthocyanidins. Food Chem. 2018, 268, 416–423. [Google Scholar] [CrossRef]
- Farr, J.E.; Giusti, M.M. Investigating the interaction of ascorbic acid with anthocyanins and pyranoanthocyanins. Molecules 2018, 23, 744. [Google Scholar] [CrossRef]
- Leusink, G.J.; Kitts, D.D.; Yaghmaee, P.; Durance, T. Retention of antioxidant capacity of vacuum microwave dried cranberry. J. Food Sci. 2010, 75, C311–C316. [Google Scholar] [CrossRef]
- Liu, S.; Chang, X.; Liu, X.; Shen, Z. Effects of pretreatments on anthocyanin composition, phenolics contents and antioxidant capacities during fermentation of hawthorn (Crataegus pinnatifida) drink. Food Chem. 2016, 212, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Belwal, T.; Huang, H.; Li, L.; Duan, Z.; Zhang, X.; Aalim, H.; Luo, Z. Optimization model for ultrasonic-assisted and scale-up extraction of anthocyanins from Pyrus communis ‘Starkrimson’ fruit peel. Food Chem. 2019, 297, 124993. [Google Scholar] [CrossRef] [PubMed]
- Nowak, D.; Gośliński, M.; Szwengiel, A. Multidimensional comparative analysis of phenolic compounds in organic juices with high antioxidant capacity. J. Sci. Food Agric. 2017, 97, 2657–2663. [Google Scholar] [CrossRef] [PubMed]
- Kaack, K.; Austed, T. Interaction of vitamin C and flavonoids in elderberry (Sambucus nigra L.) during juice processing. Plant Food Hum. Nutr. 1998, 52, 187–198. [Google Scholar] [CrossRef]
- Cho, M.J.; Howard, L.R.; Prior, R.L.; Clark, J.R. Flavonoid glycosides and antioxidant capacity of various blackberry, blueberry and red grape genotypes determined by high-performance liquid chromatography/mass spectrometry. J. Sci. Food Agric. 2004, 84, 1771–1782. [Google Scholar] [CrossRef]
- Wu, X.; Gu, L.; Prior, R.L.; McKay, S. Characterization of anthocyanins and proanthocyanidins in some cultivars of Ribes, Aronia, and Sambucus and their antioxidant capacity. J. Agric. Food Chem. 2004, 52, 7846–7856. [Google Scholar] [CrossRef]
- Ubi, B.E.; Honda, C.; Bessho, H.; Kondo, S.; Wada, M.; Kobayashi, S.; Moriguchi, T. Expression analysis of anthocyanin biosynthetic genes in apple skin: Effect of UV-B and temperature. Plant Sci. 2006, 170, 571–578. [Google Scholar] [CrossRef]
- Štampar, F.; Bizjak, J.; Veberič, R.; Jakopič, J. Foliar application of phosphorus improves apple fruit color during ripening. Acta Univ. Agric. Silvic. Mendel. Brun. 2015, 63, 1195–1200. [Google Scholar] [CrossRef]
- Awad, M.A.; de Jager, A. Relationships between fruit nutrients and concentrations of flavonoids and chlorogenic acid in ‘Elstar’ apple skin. Sci. Hortic. 2002, 92, 265–276. [Google Scholar] [CrossRef]
- Escribano-Bailón, M.T.; Santos-Buelga, C.; Alonso, G.L.; Salinas, M.R. Anthocyanin composition of the fruit of Coriaria myrtifolia L. Phytochem. Analysis 2002, 13, 354–357. [Google Scholar] [CrossRef]
- Sato, H.; Otagaki, S.; Saelai, P.; Kondo, S.; Shiratake, K.; Matsumoto, S. Varietal differences in phenolic compounds metabolism of type 2 red-fleshed apples. Sci. Hortic. 2017, 219, 1–9. [Google Scholar] [CrossRef]
- Han, M.; Li, A.; Shen, T.; Meng, J.; Lei, Y.; Zhang, X.; Liu, P.; Gan, L.; Ao, L.; Li, H. Phenolic compounds present in fruit extracts of Malus spp. show antioxidative and pro-apoptotic effects on human gastric cancer cell lines. J. Food Biochem. 2019, 43, e13028. [Google Scholar] [CrossRef]
- Gómez-Cordovés, C.; Varela, F.; Larrigaudiere, C.; Vendrell, M. Effect of ethephon and seniphos treatments on the anthocyanin composition of starking apples. J. Agric. Food Chem. 1996, 44, 3449–3452. [Google Scholar] [CrossRef]
- Tsao, R.; Yang, R.; Young, J.C.; Zhu, H. Polyphenolic profiles in eight apple cultivars using high-performance liquid chromatography (HPLC). J. Agric. Food Chem. 2003, 51, 6347–6353. [Google Scholar] [CrossRef]
- Ogawa, K.; Sakakibara, H.; Iwata, R.; Ishii, T.; Sato, T.; Goda, T.; Shimoi, K.; Kumazawa, S. Anthocyanin composition and antioxidant activity of the crowberry (Empetrum nigrum) and other berries. J. Agric. Food Chem. 2008, 56, 4457–4462. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Zhang, F.; Zhang, M.; Wei, Z.; Yang, C.; Zhang, Y.; Tang, X.; Deng, Y.; Chi, J. Phenolic composition and antioxidant activity in seed coats of 60 Chinese black soybean (Glycine max L. Merr.) varieties. J. Agric. Food Chem. 2011, 59, 5935–5944. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Y.; Chen, C.-T.; Wang, S.Y. Changes of flavonoid content and antioxidant capacity in blueberries after illumination with UV-C. Food Chem. 2009, 117, 426–431. [Google Scholar] [CrossRef]
- Jakobek, L.; Šeruga, M.; Medvidović-Kosanović, M.; Novak, I. Antioxidant activity and polyphenols of Aronia in comparison to other berry species. Agric. Conspec. Sci. 2007, 72, 301–306. [Google Scholar]
- Kapci, B.; Neradová, E.; Čížková, H.; Voldřich, M.; Rajchl, A.; Capanoglu, E. Investigating the antioxidant potential of chokeberry (Aronia melanocarpa) products. J. Food Nutr. Res. 2013, 52, 219–229. [Google Scholar]
- Ochmian, I.D.; Grajkowski, J.; Smolik, M. Comparison of some morphological features, quality and chemical content of four cultivars of chokeberry fruits (Aronia melanocarpa). Not. Bot. Horti Agrobot. 2012, 40, 253–260. [Google Scholar] [CrossRef]
- Wangensteen, H.; Bräunlich, M.; Nikolic, V.; Malterud, K.E.; Slimestad, R.; Barsett, H. Anthocyanins, proanthocyanidins and total phenolics in four cultivars of aronia: Antioxidant and enzyme inhibitory effects. J. Funct. Foods 2014, 7, 746–752. [Google Scholar] [CrossRef]
- Martinović, A.; Cavoski, I. The exploitation of cornelian cherry (Cornus mas L.) cultivars and genotypes from Montenegro as a source of natural bioactive compounds. Food Chem. 2020, 318, 126549. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Yang, C.; Zhou, J.; Zhu, J.; Meng, J.; Shen, T.; Xin, Z.; Li, H. Analysis of flavonoids and anthocyanin biosynthesis-related genes expression reveals the mechanism of petal color fading of Malus hupehensis (Rosaceae). Braz. J. Bot. 2020, 43, 81–89. [Google Scholar] [CrossRef]
- Wang, S.Y.; Ballington, J.R. Free radical scavenging capacity and antioxidant enzyme activity in deerberry (Vaccinium stamineum L.). LWT-Food Sci. Technol. 2007, 40, 1352–1361. [Google Scholar] [CrossRef]
- Lee, J.; Finn, C.E. Lingonberry (Vaccinium vitis-idaea L.) grown in the Pacific Northwest of North America: Anthocyanin and free amino acid composition. J. Funct. Foods 2012, 4, 213–218. [Google Scholar] [CrossRef]
- Isaak, C.K.; Petkau, J.C.; Kamin, O.; Debnath, S.C.; Siow, Y.L. Manitoba lingonberry (Vaccinium vitis-idaea) bioactivities in ischemia-reperfusion injury. J. Agric. Food Chem. 2015, 63, 5660–5669. [Google Scholar] [CrossRef]
- Kerio, L.C.; Wachira, F.N.; Wanyoko, J.K.; Rotich, M.K. Characterization of anthocyanins in Kenyan teas: Extraction and identification. Food Chem. 2012, 131, 31–38. [Google Scholar] [CrossRef]
- Huang, W.; Zhang, S.; Qin, G.; Wenquan, L.; Wu, J. Isolation and determination of major anthocyanin pigments in the pericarp of P. communis L. cv. ‘Red Du Comices’ and their association with antioxidant activity. Afr. J. Agric. Res. 2012, 7, 3772–3780. [Google Scholar]
- Seeram, N.P.; Zhang, Y.; Henning, S.M.; Lee, R.; Niu, Y.; Lin, G.; Heber, D. Pistachio skin phenolics are destroyed by bleaching resulting in reduced antioxidative capacities. J. Agric. Food Chem. 2006, 54, 7036–7040. [Google Scholar] [CrossRef] [PubMed]
- Slatnar, A.; Mikulic-Petkovsek, M.; Veberic, R.; Stampar, F.; Schmitzer, V. Anthocyanin and chlorophyll content during poinsettia bract development. Sci. Hortic. 2013, 150, 142–145. [Google Scholar] [CrossRef]
- Abdel-Aal, E.M.; Hucl, P. Composition and stability of anthocyanins in blue-grained wheat. J. Agric. Food Chem. 2003, 51, 2174–2180. [Google Scholar] [CrossRef]
- Fang, J.; Huang, J. Accumulation of plasma levels of anthocyanins following multiple saskatoon berry supplements. Xenobiotica 2020, 50, 454–457. [Google Scholar] [CrossRef] [PubMed]
- Zatylny, A.M.; Ziehl, W.D.; St-Pierre, R.G. Physicochemical properties of fruit of 16 saskatoon (Amelanchier alnifolia Nutt.) cultivars. Can. J. Plant Sci. 2005, 85, 933–938. [Google Scholar] [CrossRef]
- Adhikari, D.P.; Francis, J.A.; Schutzki, R.E.; Chandra, A.; Nair, M.G. Quantification and characterisation of cyclo-oxygenase and lipid peroxidation inhibitory anthocyanins in fruits of Amelanchier. Phytochem. Anal. 2005, 16, 175–180. [Google Scholar] [CrossRef]
- Cerezo, A.B.; Cuevas, E.; Winterhalter, P.; Garcia-Parrilla, M.C.; Troncoso, A.M. Isolation, identification, and antioxidant activity of anthocyanin compounds in Camarosa strawberry. Food Chem. 2010, 123, 574–582. [Google Scholar] [CrossRef]
- Pallauf, K.; Rivas-Gonzalo, J.C.; Del Castillo, M.; Cano, M.P.; de Pascual-Teresa, S. Characterization of the antioxidant composition of strawberry tree (Arbutus unedo L.) fruits. J. Food Compos. Anal. 2008, 21, 273–281. [Google Scholar] [CrossRef]
- Li, J.; Hashimoto, F.; Shimizu, K.; Sakata, Y. Anthocyanins from red flowers of Camellia reticulata LINDL. Biosci. Biotech. Biochem. 2007, 71, 2833–2836. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pilaczynska-Szczesniak, L.; Skarpanska-Steinborn, A.; Deskur, E.; Basta, P.; Horoszkiewicz-Hassan, M. The influence of chokeberry juice supplementation on the reduction of oxidative stress resulting from an incremental rowing ergometer exercise. Int. J. Sport Nutr. Exerc. Metab. 2005, 15, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Broncel, M.; Kozirog, M.; Duchnowicz, P.; Koter-Michalak, M.; Sikora, J.; Chojnowska-Jezierska, J. Aronia melanocarpa extract reduces blood pressure, serum endothelin, lipid, and oxidative stress marker levels in patients with metabolic syndrome. Med. Sci. Monitor 2009, 16, CR28–CR34. [Google Scholar]
- Kardum, N.; Konić-Ristić, A.; Šavikin, K.; Spasić, S.; Stefanović, A.; Ivanišević, J.; Miljković, M. Effects of polyphenol-rich chokeberry juice on antioxidant/pro-oxidant status in healthy subjects. J. Med. Food 2014, 17, 869–874. [Google Scholar] [CrossRef] [PubMed]
- Lyu, S.Y.; Shin, A.H.; Hahn, D.R.; Park, W.B. Antioxidant activity of cyanidins isolated from Ogapy (Acanthopanax divaricatus var. albeofructus) fruits in U937 macrophages. Food Sci. Biotechnol. 2012, 21, 1445–1450. [Google Scholar] [CrossRef]
- Bellocco, E.; Barreca, D.; Laganà, G.; Calderaro, A.; El Lekhlifi, Z.; Chebaibi, S.; Smeriglio, A.; Trombetta, D. Cyanidin-3-O-galactoside in ripe pistachio (Pistachia vera L. variety Bronte) hulls: Identification and evaluation of its antioxidant and cytoprotective activities. J. Funct. Foods 2016, 27, 376–385. [Google Scholar] [CrossRef]
- Denev, P.; Číž, M.; Kratchanova, M.; Blazheva, D. Black chokeberry (Aronia melanocarpa) polyphenols reveal different antioxidant, antimicrobial and neutrophil-modulating activities. Food Chem. 2019, 284, 108–117. [Google Scholar] [CrossRef]
- Yan, X.; Murphy, B.T.; Hammond, G.B.; Vinson, J.A.; Neto, C.C. Antioxidant activities and antitumor screening of extracts from cranberry fruit (Vaccinium macrocarpon). J. Agric. Food Chem. 2002, 50, 5844–5849. [Google Scholar] [CrossRef] [PubMed]
- Lyu, S.Y.; Park, W.B. Photoprotective potential of anthocyanins isolated from Acanthopanax divaricatus Var. albeofructus fruits against UV irradiation in human dermal fibroblast cells. Biomol. Ther. 2012, 20, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Gabrielska, J.; Oszmiański, J. Antioxidant activity of anthocyanin glycoside derivatives evaluated by the inhibition of liposome oxidation. Z. Naturforsch. C 2005, 60, 399–407. [Google Scholar] [CrossRef]
- Strugała, P.; Dudra, A.; Gabrielska, J. Activity of blackcurrant and chokeberry extracts and two major cyanidin glycosides against lipid membrane oxidation and their binding properties to albumin. Acta Pol. Pharm. 2017, 74, 676–687. [Google Scholar]
- Hu, C.; Kwok, B.H.L.; Kitts, D.D. Saskatoon berries (Amelanchier alnifolia Nutt.) scavenge free radicals and inhibit intracellular oxidation. Food Res. Int. 2005, 38, 1079–1085. [Google Scholar] [CrossRef]
- Rugină, D.; Diaconeasa, Z.; Coman, C.; Bunea, A.; Socaciu, C.; Pintea, A. Chokeberry anthocyanin extract as pancreatic β-cell protectors in two models of induced oxidative stress. Oxid. Med. Cell. Longev. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Bonarska-Kujawa, D.; Pruchnik, H.; Kleszczyńska, H. Interaction of selected anthocyanins with erythrocytes and liposome membranes. Cell. Mol. Biol. Lett. 2012, 17, 289–308. [Google Scholar] [CrossRef] [PubMed]
- Isaak, C.K.; Petkau, J.C.; Blewett, H.; Karmin, O.; Siow, Y.L. Lingonberry anthocyanins protect cardiac cells from oxidative-stress-induced apoptosis. Can. J. Physiol. Pharm. 2017, 95, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, A.; Watanabe, J.; Sakaguchi, H.; Okazaki, Y.; Suzuki, T.; Chiji, H. Anthocyanin-enriched extracts from aronia (Aronia melanocarpa E.) and haskap (Lonicera caerulea L.) suppressed KBrO3-induced renal damage in rats. J. Jpn. Soci. Food Sci. Technol. 2015, 62, 235–241. [Google Scholar] [CrossRef][Green Version]
- Malinowska, J.; Babicz, K.; Olas, B.; Stochmal, A.; Oleszek, W. Aronia melanocarpa extract suppresses the biotoxicity of homocysteine and its metabolite on the hemostatic activity of fibrinogen and plasma. Nutrition 2012, 28, 793–798. [Google Scholar] [CrossRef] [PubMed]
- Olas, B.; Wachowicz, B.; Tomczak, A.; Erler, J.; Stochmal, A.; Oleszek, W. Comparative anti-platelet and antioxidant properties of polyphenol-rich extracts from: Berries of Aronia melanocarpa, seeds of grape and bark of Yucca schidigera in vitro. Platelets 2008, 19, 70–77. [Google Scholar] [CrossRef]
- Sikora, J.; Broncel, M.; Mikiciuk-Olasik, E. Aronia melanocarpa Elliot reduces the activity of angiotensin I-converting enzyme—in vitro and ex vivo studies. Oxid. Med. Cell. Longev. 2014, 2014. [Google Scholar] [CrossRef]
- Poreba, R.; Skoczynska, A.; Gac, P.; Poreba, M.; Jedrychowska, I.; Affelska-Jercha, A.; Turczyn, B.; Wojakowska, A.; Oszmianski, J.; Andrzejak, R. Drinking of chokeberry juice from the ecological farm Dzieciolowo and distensibility of brachial artery in men with mild hypercholesterolemia. Ann. Agric. Environ. Med. 2009, 16, 305–308. [Google Scholar]
- Naruszewicz, M.; Łaniewska, I.; Millo, B.; Dłużniewski, M. Combination therapy of statin with flavonoids rich extract from chokeberry fruits enhanced reduction in cardiovascular risk markers in patients after myocardial infraction (MI). Atherosclerosis 2007, 194, e179–e184. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Pang, W.; Lu, H.; Cheng, D.; Yan, X.; Cheng, Y.; Jiang, Y. Comparison of metabolic profiling of cyanidin-3-O-galactoside and extracts from blueberry in aged mice. J. Agric. Food Chem. 2011, 59, 2069–2076. [Google Scholar] [CrossRef]
- Auger, C.; Kim, J.H.; Trinh, S.; Chataigneau, T.; Popken, A.M.; Schini-Kerth, V.B. Fruit juice-induced endothelium-dependent relaxations in isolated porcine coronary arteries: Evaluation of different fruit juices and purees and optimization of a red fruit juice blend. Food Funct. 2011, 2, 245–250. [Google Scholar] [CrossRef]
- Hellström, J.K.; Shikov, A.N.; Makarova, M.N.; Pihlanto, A.M.; Pozharitskaya, O.N.; Ryhänen, E.-L.; Kivijärvi, P.; Makarov, V.G.; Mattila, P.H. Blood pressure-lowering properties of chokeberry (Aronia mitchurinii, var. Viking). J. Funct. Foods 2010, 2, 163–169. [Google Scholar] [CrossRef]
- Hu, C.; Cai, Y.; Li, W.; Corke, H.; Kitts, D.D. Anthocyanin characterization and bioactivity assessment of a dark blue grained wheat (Triticum aestivum L. cv. Hedong Wumai) extract. Food Chem. 2007, 104, 955–961. [Google Scholar] [CrossRef]
- Appel, K.; Meiser, P.; Millán, E.; Collado, J.A.; Rose, T.; Gras, C.C.; Carle, R.; Muñoz, E. Chokeberry (Aronia melanocarpa (Michx.) Elliot) concentrate inhibits NF-κB and synergizes with selenium to inhibit the release of pro-inflammatory mediators in macrophages. Fitoterapia 2015, 105, 73–82. [Google Scholar] [CrossRef]
- Qin, B.; Anderson, R.A. An extract of chokeberry attenuates weight gain and modulates insulin, adipogenic and inflammatory signalling pathways in epididymal adipose tissue of rats fed a fructose-rich diet. Brit. J. Nutr. 2012, 108, 581–587. [Google Scholar] [CrossRef] [PubMed]
- Malik, M.; Zhao, C.; Schoene, N.; Guisti, M.M.; Moyer, M.P.; Magnuson, B.A. Anthocyanin-rich extract from Aronia meloncarpa E. induces a cell cycle block in colon cancer but not normal colonic cells. Nutr. Cancer 2003, 46, 186–196. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Giusti, M.M.; Malik, M.; Moyer, M.P.; Magnuson, B.A. Effects of commercial anthocyanin-rich extracts on colonic cancer and nontumorigenic colonic cell growth. J. Agric. Food Chem. 2004, 52, 6122–6128. [Google Scholar] [CrossRef] [PubMed]
- Bermúdez-Soto, M.J.; Larrosa, M.; Garcia-Cantalejo, J.M.; Espín, J.C.; Tomás-Barberan, F.A.; García-Conesa, M.T. Up-regulation of tumor suppressor carcinoembryonic antigen-related cell adhesion molecule 1 in human colon cancer Caco-2 cells following repetitive exposure to dietary levels of a polyphenol-rich chokeberry juice. J. Nutr. Biochem. 2007, 18, 259–271. [Google Scholar] [CrossRef]
- Lala, G.; Malik, M.; Zhao, C.; He, J.; Kwon, Y.; Giusti, M.M.; Magnuson, B.A. Anthocyanin-rich extracts inhibit multiple biomarkers of colon cancer in rats. Nutr. Cancer 2006, 54, 84–93. [Google Scholar] [CrossRef]
- Yang, H.; Jiang, Y.; Pang, W.; Chen, D.; Lu, H.; Lu, S. Study on the effect of anthocyanin from blueberry on improving learning and memory in aged mice. Acta Nutr. Sin. 2009, 31, 583–587. [Google Scholar]
- Lee, H.Y.; Weon, J.B.; Jung, Y.S.; Kim, N.Y.; Kim, M.K.; Ma, C.J. Cognitive-enhancing effect of Aronia melanocarpa extract against memory impairment induced by scopolamine in mice. Evid.-Based Compl. Alt. Med. 2016, 2016. [Google Scholar] [CrossRef]
- Maslov, D.L.; Ipatova, O.M.; Abakumova, O.I.; Tsvetkova, T.A.; Prozorovskiĭ, V.N. Hypoglycemic effect of an extract from Aronia melanocarpa leaves. Vopr. Med. Khim. 2002, 48, 271–277. [Google Scholar] [PubMed]
- Valcheva-Kuzmanova, S.; Kuzmanov, K.; Tancheva, S.; Belcheva, A. Hypoglycemic and hypolipidemic effects of Aronia melanocarpa fruit juice in streptozotocin-induced diabetic rats. Methods Find. Exp. Clin. Pharmacol. 2007, 29, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Valcheva-Kuzmanova, S.; Marazova, K.; Krasnaliev, I.; Galunska, B.; Borisova, P.; Belcheva, A. Effect of Aronia melanocarpa fruit juice on indomethacin-induced gastric mucosal damage and oxidative stress in rats. Exp. Toxicol. Pathol. 2005, 56, 385–392. [Google Scholar] [CrossRef]
- Castañeda-Ovando, A.; de Lourdes Pacheco-Hernández, M.; Páez-Hernández, M.E.; Rodríguez, J.A.; Galán-Vidal, C.A. Chemical studies of anthocyanins: A review. Food Chem. 2009, 113, 859–871. [Google Scholar] [CrossRef]
- Jordheim, M.; Aaby, K.; Fossen, T.; Skrede, G.; Andersen, Ø.M. Molar absorptivities and reducing capacity of pyranoanthocyanins and other anthocyanins. J. Agric. Food Chem. 2007, 55, 10591–10598. [Google Scholar] [CrossRef]
- De Rosso, V.V.; Moran Vieyra, F.E.; Mercadante, A.Z.; Borsarelli, C.D. Singlet oxygen quenching by anthocyanin’s flavylium cations. Free Radic. Res. 2008, 42, 885–891. [Google Scholar] [CrossRef] [PubMed]
- Korte, G.; Dreiseitel, A.; Schreier, P.; Oehme, A.; Locher, S.; Hajak, G.; Sand, P.G. An examination of anthocyanins’ and anthocyanidins’ affinity for cannabinoid receptors. J. Med. Food 2009, 12, 1407–1410. [Google Scholar] [CrossRef]
- Kim, J.H.; Auger, C.; Kurita, I.; Anselm, E.; Rivoarilala, L.O.; Lee, H.J.; Lee, K.W.; Schini-Kerth, V.B. Aronia melanocarpa juice, a rich source of polyphenols, induces endothelium-dependent relaxations in porcine coronary arteries via the redox-sensitive activation of endothelial nitric oxide synthase. Nitric Oxide 2013, 35, 54–64. [Google Scholar] [CrossRef]
- Trumbeckaitė, S.; Burdulis, D.; Raudonė, L.; Liobikas, J.; Toleikis, A.; Janulis, V. Direct effects of Vaccinium myrtillus L. fruit extracts on rat heart mitochondrial functions. Phytother. Res. 2013, 27, 499–506. [Google Scholar] [CrossRef]
- Martin, D.A.; Taheri, R.; Brand, M.H.; Draghi, A., II; Sylvester, F.A.; Bolling, B.W. Anti-inflammatory activity of aronia berry extracts in murine splenocytes. J. Funct. Foods 2014, 8, 68–75. [Google Scholar] [CrossRef]
- Xu, J.; Mojsoska, B. The immunomodulation effect of Aronia extract lacks association with its antioxidant anthocyanins. J. Med. Food 2013, 16, 334–342. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Vareed, S.K.; Nair, M. Human tumor cell growth inhibition by nontoxic anthocyanidins, the pigments in fruits and vegetables. Life Sci. 2005, 76, 1465–1472. [Google Scholar] [CrossRef] [PubMed]
- Lila, M.A.; Burton-Freeman, B.; Grace, M.; Kalt, W. Unraveling anthocyanin bioavailability for human health. Annu. Rev. Food Sci. Technol. 2016, 7, 375–393. [Google Scholar] [CrossRef]
- Mazza, G.; Kay, C.D.; Cottrell, T.; Holub, B.J. Absorption of anthocyanins from blueberries and serum antioxidant status in human subjects. J. Agric. Food Chem. 2002, 50, 7731–7737. [Google Scholar] [CrossRef]
- Ichiyanagi, T.; Shida, Y.; Rahman, M.M.; Hatano, Y.; Konishi, T. Bioavailability and tissue distribution of anthocyanins in bilberry (Vaccinium myrtillus L.) extract in rats. J. Agric. Food Chem. 2006, 54, 6578–6587. [Google Scholar] [CrossRef] [PubMed]
- Andres-Lacueva, C.; Shukitt-Hale, B.; Galli, R.L.; Jauregui, O.; Lamuela-Raventos, R.M.; Joseph, J.A. Anthocyanins in aged blueberry-fed rats are found centrally and may enhance memory. Nutr. Neurosci. 2005, 8, 111–120. [Google Scholar] [CrossRef]
- Chen, T.Y.; Kritchevsky, J.; Hargett, K.; Feller, K.; Klobusnik, R.; Song, B.J.; Cooper, B.; Jouni, Z.; Ferruzzi, M.G.; Janle, E.M. Plasma bioavailability and regional brain distribution of polyphenols from apple/grape seed and bilberry extracts in a young swine model. Mol. Nutr. Food Res. 2015, 59, 2432–2447. [Google Scholar] [CrossRef]
- Fang, J. Bioavailability of anthocyanins. Drug Metab. Rev. 2014, 46, 508–520. [Google Scholar] [CrossRef] [PubMed]
- McGhie, T.K.; Ainge, G.D.; Barnett, L.E.; Cooney, J.M.; Jensen, D.J. Anthocyanin glycosides from berry fruit are absorbed and excreted unmetabolized by both humans and rats. J. Agric. Food Chem. 2003, 51, 4539–4548. [Google Scholar] [CrossRef]
- He, J.; Magnuson, B.A.; Lala, G.; Tian, Q.; Schwartz, S.J.; Giusti, M.M. Intact anthocyanins and metabolites in rat urine and plasma after 3 months of anthocyanin supplementation. Nutr. Cancer 2006, 54, 3–12. [Google Scholar] [CrossRef]
- Talavera, S.; Felgines, C.; Texier, O.; Besson, C.; Lamaison, J.L.; Rémésy, C. Anthocyanins are efficiently absorbed from the stomach in anesthetized rats. J. Nutr. 2003, 133, 4178–4182. [Google Scholar] [CrossRef] [PubMed]
- Duthie, S.J.; Jenkinson, A.M.; Crozier, A.; Mullen, W.; Pirie, L.; Kyle, J.; Yap, L.S.; Christen, P.; Duthie, G.G. The effects of cranberry juice consumption on antioxidant status and biomarkers relating to heart disease and cancer in healthy human volunteers. Eur. J. Nutr. 2006, 45, 113–122. [Google Scholar] [CrossRef]
- Qin, L.; Zhang, J.; Qin, M. Protective effect of cyanidin 3-O-glucoside on beta-amyloid peptide-induced cognitive impairment in rats. Neurosci. Lett. 2013, 534, 285–288. [Google Scholar] [CrossRef]
- Dos Santos, N.M.; Batista, P.B.; Batista, Â.G.; Júnior, M.R.M. Current evidence on cognitive improvement and neuroprotection promoted by anthocyanins. Curr. Opin. Food Sci. 2019, 26, 71–78. [Google Scholar] [CrossRef]
- Kim, B.; Park, Y.; Wegner, C.J.; Bolling, B.W.; Lee, J. Polyphenol-rich black chokeberry (Aronia melanocarpa) extract regulates the expression of genes critical for intestinal cholesterol flux in Caco-2 cells. J. Nutr. Biochem. 2013, 24, 1564–1570. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, J.; Yin, H.; Fan, Z.; Li, J. Variation of flower colors and their relationships with anthocyanins in cultivars of Camellia japonica. J. Ecol. Rural Environ. 2019, 35, 1307–1313. [Google Scholar]
- Türkyılmaz, M.; Özkan, M. Kinetics of anthocyanin degradation and polymeric colour formation in black carrot juice concentrates during storage. Int. J. Food Sci. Tech. 2012, 47, 2273–2281. [Google Scholar] [CrossRef]


| Direct Sources | Botanical Name | Content of Cy3Gal (mg/100g Material Weight) | Notes | Reference |
|---|---|---|---|---|
| Achene | Coriaria myrtifolia L. | 607.3 c | - | [74] |
| Apple juice | Malus pumila | 12.5 d | - | [12] |
| Malus marjorensis | 1.25 d | |||
| Apple red flesh | - | 6.765 b | - | [11] |
| Malus domestica | 0.45–47.63 b | - | [75] | |
| Malus spp. | 31.86–1246.36 c | The whole fruit was used in the study | [76] | |
| Apple white flesh | Malus domestica | 0.00 b | - | [75] |
| Malus micromalus | 0.00 c | The whole fruit was used in the study | [76] | |
| Apple peel | Malus domestica | 4.9321–85.6 c | - | [73,77] |
| 0–20.82 b | - | [11,78] | ||
| Bilberry | Vaccinium myrtillus | 370 b | - | [79] |
| Black soybean seed coat | Glycine max L. Merr. | 6.0 ± 3.6 b | The data are counted as the mean of 60 varieties | [80] |
| Blueberry | Vaccinium spp. | 28 b | - | [79] |
| Vaccinium corymbosum L. | 6.24 ± 0.81 b | - | [81] | |
| Chokeberry | Aronia melanocarpa Michx. | 168.0–636.0 b | - | [82,83,84,85] |
| Aronia prunifolia | 497 ± 10 b | - | [85] | |
| Chokeberry juice | - | 108.9–326.1 d | - | [67] |
| Cornelian cherry fruit | Cornus mas L. | 4.63–130.93 b | - | [86] |
| Crabapple flower petal | Malus hupehensis | 131.37–1430.75 c | - | [87] |
| Cranberry | Vaccinium macrocarpon | 5.7–123.4 c | Materials were dried using different methods | [64] |
| Vaccinium oxycoccos | 386 b | - | [79] | |
| Cranberry juice | Vaccinium macrocarpon Ait. | 64.20 d | - | [15] |
| Crowberry | Empetrum nigrum | 804 b | - | [79] |
| Deerberry | Vaccinium stamineum L. | 74.42–98.75 b | - | [88] |
| Hawthorn fruit | Crataegus pinnatifida var. major | 15 ± 1.10 c | The data are the mean from 10 cultivars | [13] |
| Crataegus brettschneideri | 37 ± 3.36 c | The data are the mean from 8 cultivars | ||
| Crataegus pinnatifida | 20 ± 2.89 c | The data are the mean from 3 cultivars | ||
| Crataegus scabrifolia | 0 c | - | ||
| Lingonberry | Vaccinium vitis-idaea L. | 18.7–35.5 b | - | [89] |
| 575 ± 20 c | - | [90] | ||
| Kenyan tea | Camellia sinensis L. | 0.951–15.544 d | - | [91] |
| Pear peel | Pyrus communis | 23.7 ± 3.2 b | - | [92] |
| Pistachio nuts skin | Pistacia Vera L. | 69.6 ± 4.5 b | - | [93] |
| Poinsettia bract | Euphorbia pulcherrima Willd. | 0.724–108.168 b | Only data of bracts in fully pigmented stage are presented | [94] |
| Purple wheat | Triticum aestivum | 0.098 ± 0.015 b | - | [95] |
| Saskatoon berry | Amelanchier alnifolia Nutt. | 20.5–155.0 b | - | [96,97] |
| Amelanchier arborea | 390 b | - | [98] | |
| Amelanchier canadensis | 165 b | |||
| Strawberry | Fragaria × ananassa | 0.214 ± 0.0014 b | - | [99] |
| Arbutus unedo L. | 2.84 ± 0.540 b | - | [100] | |
| Theaceae flower petal | Camellia reticulata | 19.5 c | The fresh petals were treated with boiling water for 4–6 s and were then dried at room temperature | [101] |
| Camellia pitardii var. yunnanica | 33.1 c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, Z.; Liang, H.; Guo, Y.; Yang, D. Cyanidin 3-O-galactoside: A Natural Compound with Multiple Health Benefits. Int. J. Mol. Sci. 2021, 22, 2261. https://doi.org/10.3390/ijms22052261
Liang Z, Liang H, Guo Y, Yang D. Cyanidin 3-O-galactoside: A Natural Compound with Multiple Health Benefits. International Journal of Molecular Sciences. 2021; 22(5):2261. https://doi.org/10.3390/ijms22052261
Chicago/Turabian StyleLiang, Zhongxin, Hongrui Liang, Yizhan Guo, and Dong Yang. 2021. "Cyanidin 3-O-galactoside: A Natural Compound with Multiple Health Benefits" International Journal of Molecular Sciences 22, no. 5: 2261. https://doi.org/10.3390/ijms22052261
APA StyleLiang, Z., Liang, H., Guo, Y., & Yang, D. (2021). Cyanidin 3-O-galactoside: A Natural Compound with Multiple Health Benefits. International Journal of Molecular Sciences, 22(5), 2261. https://doi.org/10.3390/ijms22052261







