Activation of Apoptosis in a βB1-CTGF Transgenic Mouse Model
Abstract
1. Introduction
2. Results
2.1. Loss of Neurofilament H at 10 Weeks
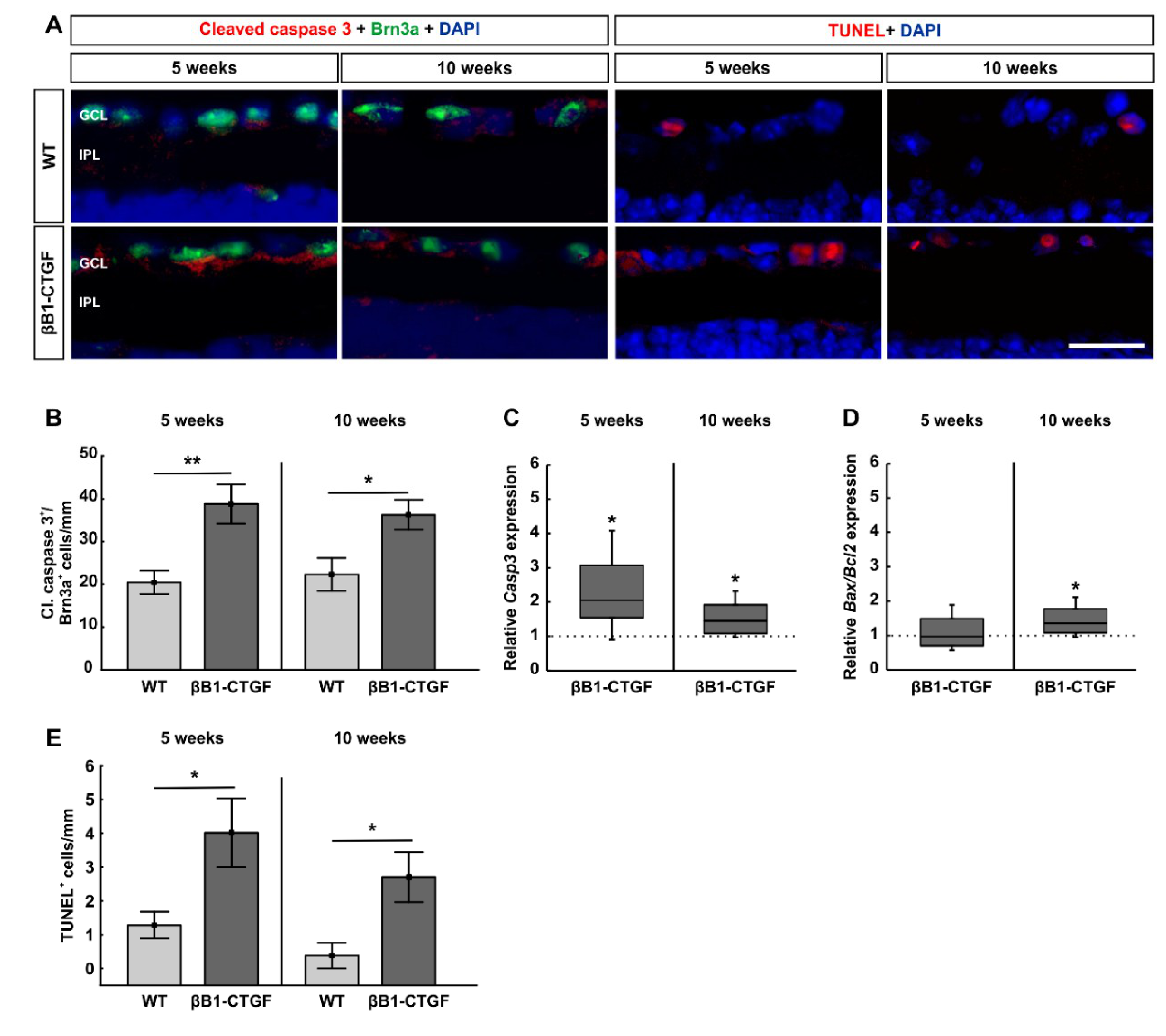
2.2. Increased Number of Apoptotic Retinal Ganglion Cells
2.3. Loss of Synapses
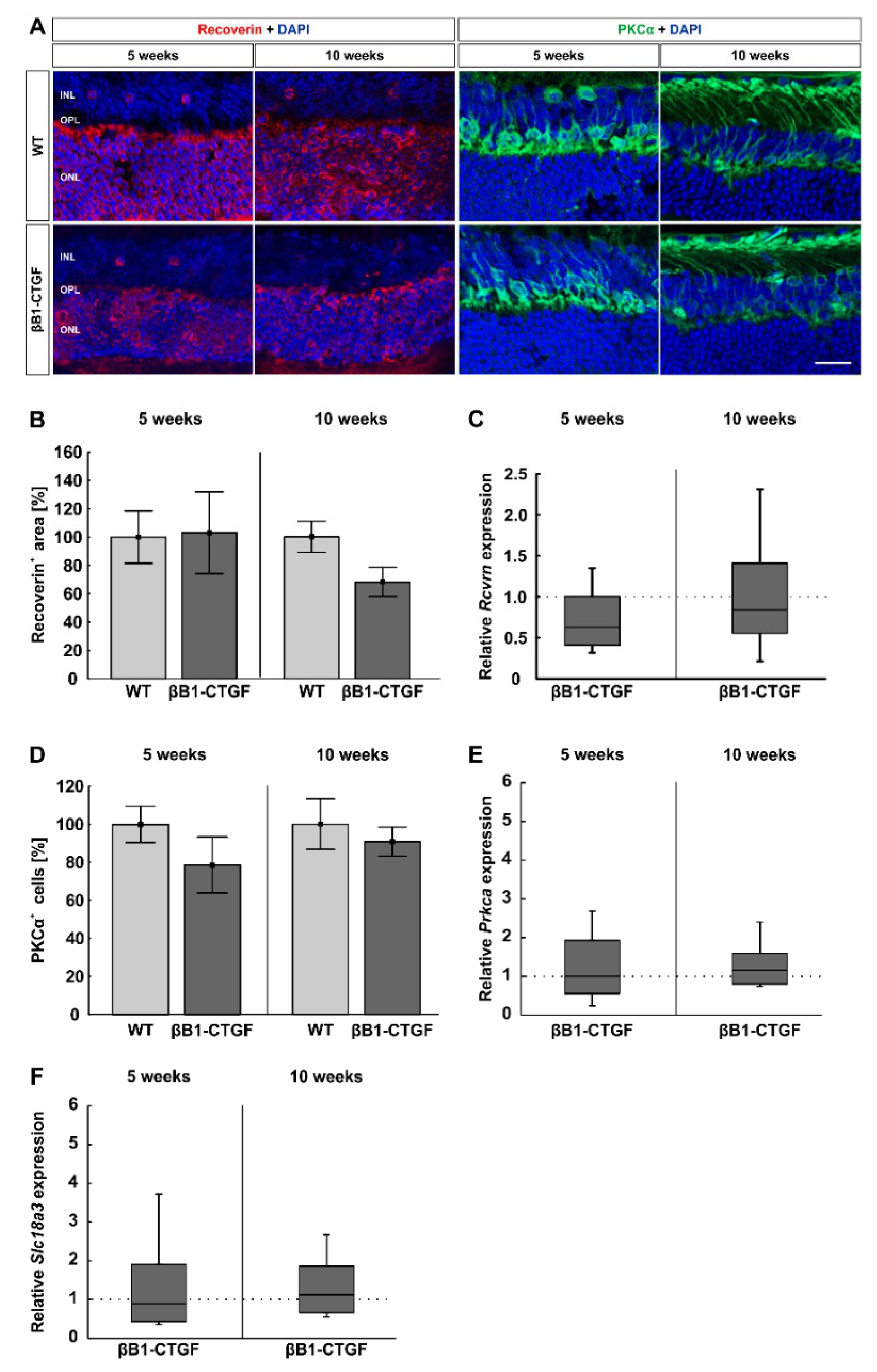
2.4. Similar Bipolar Cell Numbers
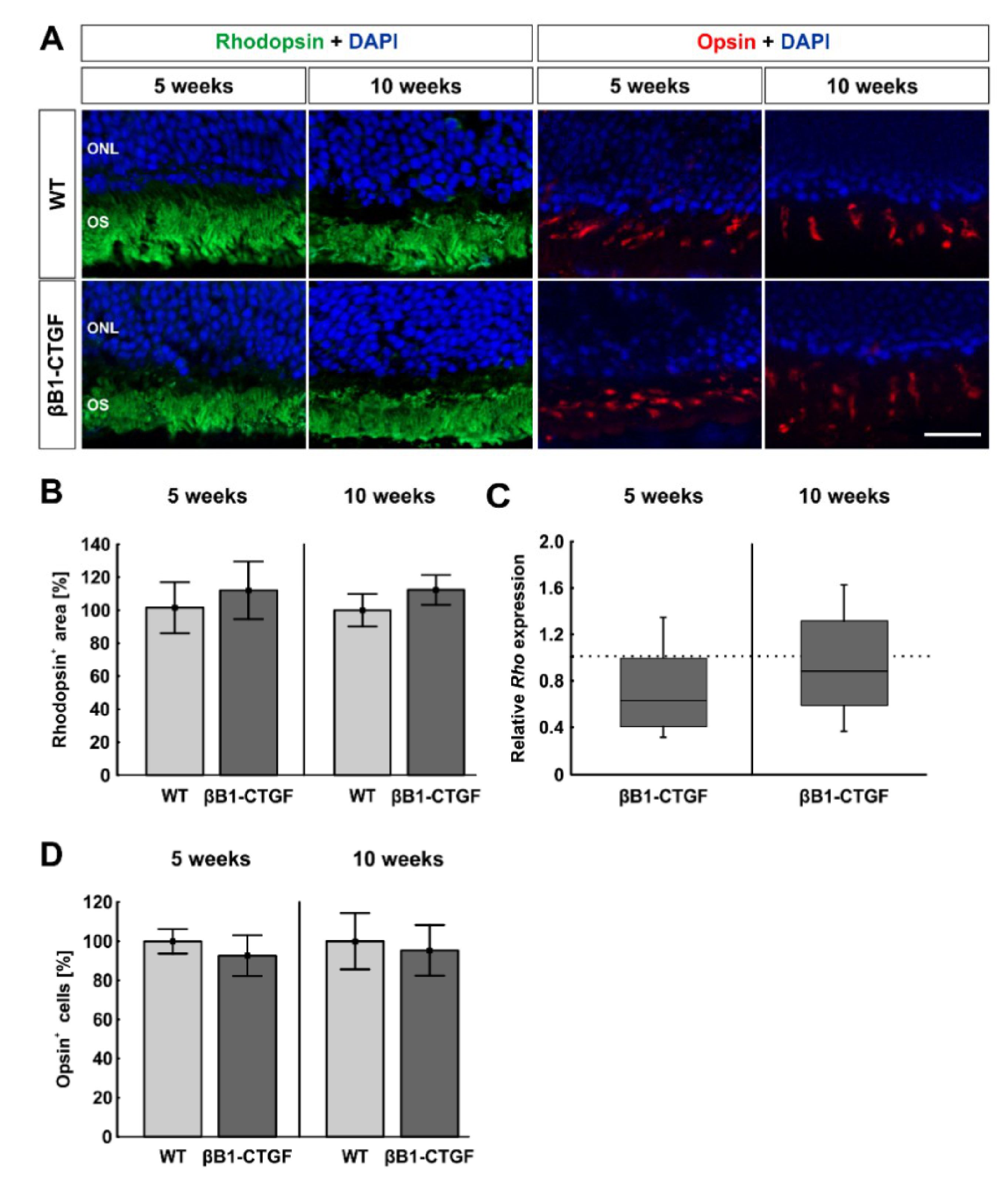
2.5. No Effects on Photoreceptors
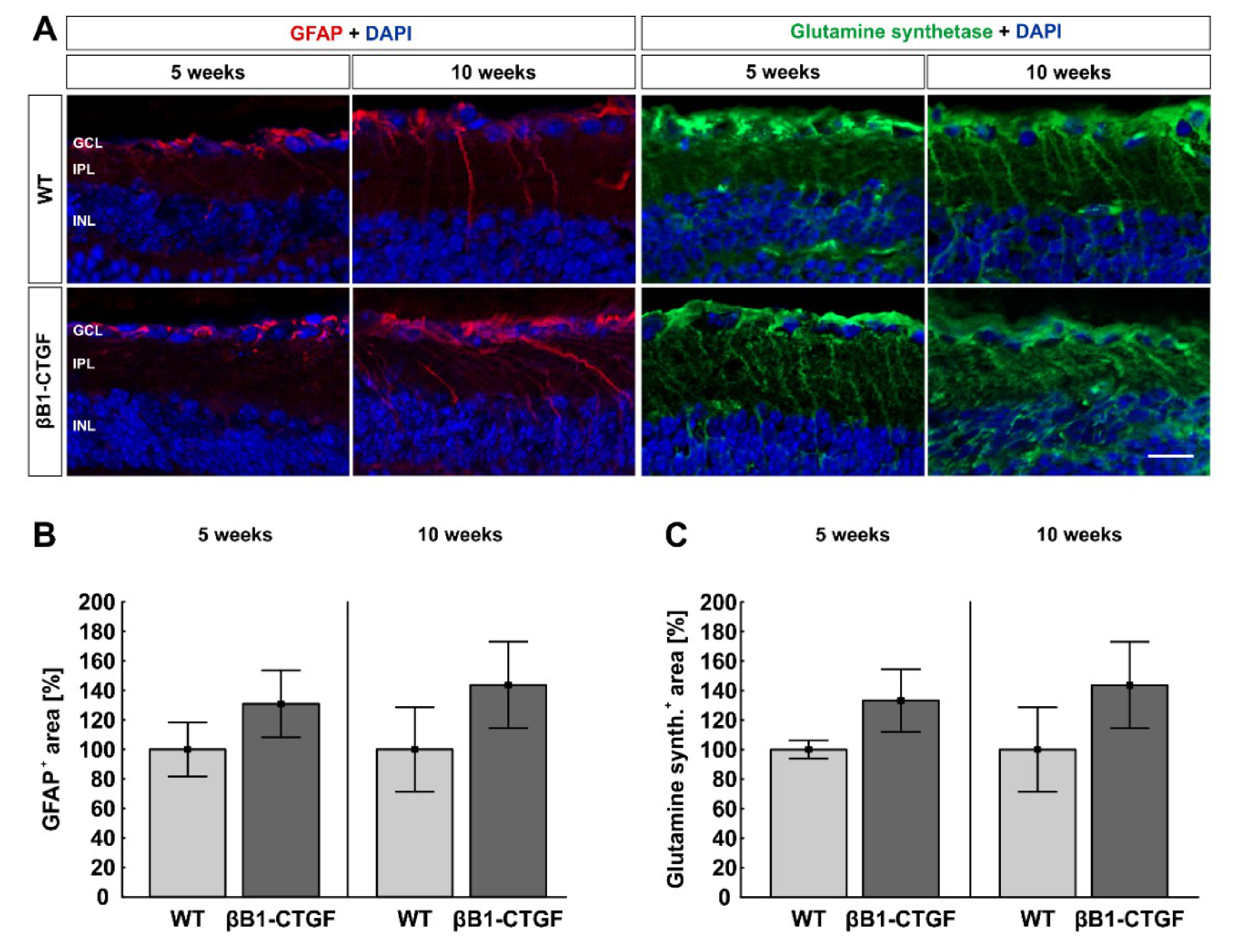
2.6. No Alteration in Macroglia
2.7. No Functional or Morphological alterations
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Optical Coherence Tomography
4.3. Electroretinogram Analyses
4.4. Tissue Preparation for Immunohistology
4.5. Immunohistology
4.6. Histological Examination
4.7. Quantitative Real-Time PCR
4.8. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Flaxman, S.R.; Bourne, R.R.A.; Resnikoff, S.; Ackland, P.; Braithwaite, T.; Cicinelli, M.V.; Das, A.; Jonas, J.B.; Keeffe, J.; Kempen, J.H.; et al. Global causes of blindness and distance vision impairment 1990–2020: A systematic review and meta-analysis. Lancet Global Health 2017, 5, e1221–e1234. [Google Scholar] [CrossRef]
- Tezel, G.; Wax, M.B. Glaucoma. Chem. Immunol. Allergy 2007, 92, 221–227. [Google Scholar]
- Kerrigan-Baumrind, L.A.; Quigley, H.A.; Pease, M.E.; Kerrigan, D.F.; Mitchell, R.S. Number of ganglion cells in glaucoma eyes compared with threshold visual field tests in the same persons. Invest. Ophthalmol. Vis. Sci. 2000, 41, 741–748. [Google Scholar] [PubMed]
- EGS. European Glaucoma Society Terminology and Guidelines for Glaucoma, 4th Edition—Chapter 2: Classification and Terminology. Br. J. Ophthalmol. 2017, 101, 73–127. [Google Scholar] [CrossRef]
- Harwerth, R.S.; Wheat, J.L.; Fredette, M.J.; Anderson, D.R. Linking structure and function in glaucoma. Prog. Retin Eye Res. 2010, 29, 249–271. [Google Scholar] [CrossRef]
- Quigley, H.A. Glaucoma. Lancet 2011, 377, 1367–1377. [Google Scholar] [CrossRef]
- Junglas, B.; Kuespert, S.; Seleem, A.A.; Struller, T.; Ullmann, S.; Bosl, M.; Bosserhoff, A.; Kostler, J.; Wagner, R.; Tamm, E.R.; et al. Connective tissue growth factor causes glaucoma by modifying the actin cytoskeleton of the trabecular meshwork. Am. J. Pathol. 2012, 180, 2386–2403. [Google Scholar] [CrossRef] [PubMed]
- Sommer, A.; Tielsch, J.M.; Katz, J.; Quigley, H.A.; Gottsch, J.D.; Javitt, J.; Singh, K. Relationship between intraocular pressure and primary open angle glaucoma among white and black Americans. The Baltimore Eye Survey. Arch. Ophthalmol. 1991, 109, 1090–1095. [Google Scholar] [CrossRef]
- Schaub, J.A.; Kimball, E.C.; Steinhart, M.R.; Nguyen, C.; Pease, M.E.; Oglesby, E.N.; Jefferys, J.L.; Quigley, H.A. Regional Retinal Ganglion Cell Axon Loss in a Murine Glaucoma Model. Invest. Ophthalmol. Vis. Sci. 2017, 58, 2765–2773. [Google Scholar] [CrossRef]
- Steinhart, M.R.; Cone-Kimball, E.; Nguyen, C.; Nguyen, T.D.; Pease, M.E.; Chakravarti, S.; Oglesby, E.N.; Quigley, H.A. Susceptibility to glaucoma damage related to age and connective tissue mutations in mice. Exp. Eye Res. 2014, 119, 54–60. [Google Scholar] [CrossRef]
- Reinehr, S.; Koch, D.; Weiss, M.; Froemel, F.; Voss, C.; Dick, H.B.; Fuchshofer, R.; Joachim, S.C. Loss of retinal ganglion cells in a new genetic mouse model for primary open-angle glaucoma. J. Cell. Mol. Med. 2019. [Google Scholar] [CrossRef]
- Wallace, D.M.; Murphy-Ullrich, J.E.; Downs, J.C.; O’Brien, C.J. The role of matricellular proteins in glaucoma. Matrix Biol. 2014, 37, 174–182. [Google Scholar] [CrossRef] [PubMed]
- John, S.W.; Smith, R.S.; Savinova, O.V.; Hawes, N.L.; Chang, B.; Turnbull, D.; Davisson, M.; Roderick, T.H.; Heckenlively, J.R. Essential iris atrophy, pigment dispersion, and glaucoma in DBA/2J mice. Invest. Ophthalmol. Vis. Sci. 1998, 39, 951–962. [Google Scholar] [PubMed]
- Yang, J.W.; Sakiyalak, D.; Krupin, T. Pigmentary glaucoma. J. Glaucoma 2001, 10 (Suppl 1), S30–S32. [Google Scholar] [CrossRef]
- Levkovitch-Verbin, H.; Quigley, H.A.; Martin, K.R.; Valenta, D.; Baumrind, L.A.; Pease, M.E. Translimbal laser photocoagulation to the trabecular meshwork as a model of glaucoma in rats. Invest. Ophthalmol. Vis. Sci. 2002, 43, 402–410. [Google Scholar] [PubMed]
- Sappington, R.M.; Carlson, B.J.; Crish, S.D.; Calkins, D.J. The microbead occlusion model: A paradigm for induced ocular hypertension in rats and mice. Invest. Ophthalmol. Vis. Sci. 2010, 51, 207–216. [Google Scholar] [CrossRef]
- Mirzaei, M.; Gupta, V.K.; Chitranshi, N.; Deng, L.; Pushpitha, K.; Abbasi, M.; Chick, J.M.; Rajput, R.; Wu, Y.; McKay, M.J.; et al. Retinal proteomics of experimental glaucoma model reveal intraocular pressure-induced mediators of neurodegenerative changes. J. Cell. Biochem. 2020. [Google Scholar] [CrossRef] [PubMed]
- Lakshmanan, Y.; Wong, F.S.Y.; Zuo, B.; Bui, B.V.; Chan, H.H. Longitudinal outcomes of circumlimbal suture model-induced chronic ocular hypertension in Sprague-Dawley albino rats. Graefes Arch. Clin. Exp. Ophthalmol. 2020. [Google Scholar] [CrossRef]
- Taylor, A.W. Primary Open-Angle Glaucoma: A Transforming Growth Factor-β Pathway-Mediated Disease. Am. J. Pathol. 2012, 180, 2201–2204. [Google Scholar] [CrossRef] [PubMed]
- Wallace, D.M.; Pokrovskaya, O.; O’Brien, C.J. The Function of Matricellular Proteins in the Lamina Cribrosa and Trabecular Meshwork in Glaucoma. J. Ocul. Pharmacol. Ther. 2015, 31, 386–395. [Google Scholar] [CrossRef] [PubMed]
- Joachim, S.C.; Mondon, C.; Gramlich, O.W.; Grus, F.H.; Dick, H.B. Apoptotic retinal ganglion cell death in an autoimmune glaucoma model is accompanied by antibody depositions. J. Mol. Neurosci. 2014, 52, 216–224. [Google Scholar] [CrossRef]
- Zhou, X.; Li, F.; Kong, L.; Tomita, H.; Li, C.; Cao, W. Involvement of inflammation, degradation, and apoptosis in a mouse model of glaucoma. J. Biol. Chem. 2005, 280, 31240–31248. [Google Scholar] [CrossRef] [PubMed]
- Mosinger Ogilvie, J.; Deckwerth, T.L.; Knudson, C.M.; Korsmeyer, S.J. Suppression of developmental retinal cell death but not of photoreceptor degeneration in Bax-deficient mice. Invest. Ophthalmol. Vis. Sci. 1998, 39, 1713–1720. [Google Scholar] [PubMed]
- Deckwerth, T.L.; Elliott, J.L.; Knudson, C.M.; Johnson, E.M., Jr.; Snider, W.D.; Korsmeyer, S.J. BAX is required for neuronal death after trophic factor deprivation and during development. Neuron 1996, 17, 401–411. [Google Scholar] [CrossRef]
- Libby, R.T.; Li, Y.; Savinova, O.V.; Barter, J.; Smith, R.S.; Nickells, R.W.; John, S.W. Susceptibility to neurodegeneration in a glaucoma is modified by Bax gene dosage. PLoS Genet. 2005, 1, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Kerrigan, L.A.; Zack, D.J.; Quigley, H.A.; Smith, S.D.; Pease, M.E. TUNEL-positive ganglion cells in human primary open-angle glaucoma. Arch. Ophthalmol. 1997, 115, 1031–1035. [Google Scholar] [CrossRef] [PubMed]
- Balaratnasingam, C.; Morgan, W.H.; Bass, L.; Kang, M.; Cringle, S.J.; Yu, D.Y. Time-dependent effects of focal retinal ischemia on axonal cytoskeleton proteins. Invest. Ophthalmol. Vis. Sci. 2010, 51, 3019–3028. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, M.F.; Guo, L.; Luong, V.; Harding, G.; Wang, W.; Jones, H.E.; Moss, S.E.; Sillito, A.M.; Fitzke, F.W. Real-time imaging of single nerve cell apoptosis in retinal neurodegeneration. Proc. Natl. Acad. Sci. USA 2004, 101, 13352–13356. [Google Scholar] [CrossRef]
- Morquette, J.B.; Di Polo, A. Dendritic and synaptic protection: Is it enough to save the retinal ganglion cell body and axon? J. Neuroophthalmol. 2008, 28, 144–154. [Google Scholar] [CrossRef] [PubMed]
- Reinehr, S.; Kuehn, S.; Casola, C.; Koch, D.; Stute, G.; Grotegut, P.; Dick, H.B.; Joachim, S.C. HSP27 immunization reinforces AII amacrine cell and synapse damage induced by S100 in an autoimmune glaucoma model. Cell Tissue Res. 2018, 371, 237–249. [Google Scholar] [CrossRef]
- Reinehr, S.; Reinhard, J.; Wiemann, S.; Hesse, K.; Voss, C.; Gandej, M.; Dick, H.B.; Faissner, A.; Joachim, S.C. Transfer of the Experimental Autoimmune Glaucoma Model from Rats to Mice-New Options to Study Glaucoma Disease. Int. J. Mol. Sci. 2019, 20, 2563. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Sanchez, L.; de Sevilla Muller, L.P.; Brecha, N.C.; Cuenca, N. Loss of outer retinal neurons and circuitry alterations in the DBA/2J mouse. Investig. Ophthalmol. Vis. Sci. 2014, 55, 6059–6072. [Google Scholar] [CrossRef]
- Tonnies, E.; Trushina, E. Oxidative Stress, Synaptic Dysfunction, and Alzheimer’s Disease. J. Alzheimer Dis. JAD 2017, 57, 1105–1121. [Google Scholar] [CrossRef] [PubMed]
- Oddo, S.; Caccamo, A.; Shepherd, J.D.; Murphy, M.P.; Golde, T.E.; Kayed, R.; Metherate, R.; Mattson, M.P.; Akbari, Y.; LaFerla, F.M. Triple-transgenic model of Alzheimer’s disease with plaques and tangles: Intracellular Abeta and synaptic dysfunction. Neuron 2003, 39, 409–421. [Google Scholar] [CrossRef]
- Jansen, D.; Janssen, C.I.; Vanmierlo, T.; Dederen, P.J.; van Rooij, D.; Zinnhardt, B.; Nobelen, C.L.; Janssen, A.L.; Hafkemeijer, A.; Mutsaers, M.P.; et al. Cholesterol and synaptic compensatory mechanisms in Alzheimer’s disease mice brain during aging. J. Alzheimer Dis. JAD 2012, 31, 813–826. [Google Scholar] [CrossRef] [PubMed]
- Scheff, S.W.; Price, D.A. Synaptic pathology in Alzheimer’s disease: A review of ultrastructural studies. Neurobiol. Aging 2003, 24, 1029–1046. [Google Scholar] [CrossRef] [PubMed]
- Boncristiano, S.; Calhoun, M.E.; Howard, V.; Bondolfi, L.; Kaeser, S.A.; Wiederhold, K.H.; Staufenbiel, M.; Jucker, M. Neocortical synaptic bouton number is maintained despite robust amyloid deposition in APP23 transgenic mice. Neurobiol. Aging 2005, 26, 607–613. [Google Scholar] [CrossRef]
- Arendt, T. Alzheimer’s disease as a disorder of mechanisms underlying structural brain self-organization. Neuroscience 2001, 102, 723–765. [Google Scholar] [CrossRef]
- Masliah, E.; Crews, L.; Hansen, L. Synaptic remodeling during aging and in Alzheimer’s disease. J. Alzheimer Dis. JAD 2006, 9 (Suppl. 3), 91–99. [Google Scholar] [CrossRef] [PubMed]
- Park, H.Y.; Kim, J.H.; Park, C.K. Alterations of the synapse of the inner retinal layers after chronic intraocular pressure elevation in glaucoma animal model. Mol. Brain 2014, 7, 53. [Google Scholar] [CrossRef]
- Johnson, J.; Fremeau, R.T., Jr.; Duncan, J.L.; Renteria, R.C.; Yang, H.; Hua, Z.; Liu, X.; LaVail, M.M.; Edwards, R.H.; Copenhagen, D.R. Vesicular glutamate transporter 1 is required for photoreceptor synaptic signaling but not for intrinsic visual functions. J. Neurosci. 2007, 27, 7245–7255. [Google Scholar] [CrossRef]
- Ly, A.; Scheerer, M.F.; Zukunft, S.; Muschet, C.; Merl, J.; Adamski, J.; de Angelis, M.H.; Neschen, S.; Hauck, S.M.; Ueffing, M. Retinal proteome alterations in a mouse model of type 2 diabetes. Diabetologia 2014, 57, 192–203. [Google Scholar] [CrossRef]
- Khurana, R.N.; Deng, P.F.; Epstein, D.L.; Vasantha Rao, P. The role of protein kinase C in modulation of aqueous humor outflow facility. Exp. Eye Res. 2003, 76, 39–47. [Google Scholar] [CrossRef]
- Milam, A.H.; Dacey, D.M.; Dizhoor, A.M. Recoverin immunoreactivity in mammalian cone bipolar cells. Vis. Neurosci. 1993, 10, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Moon, J.I.; Kim, I.B.; Gwon, J.S.; Park, M.H.; Kang, T.H.; Lim, E.J.; Choi, K.R.; Chun, M.H. Changes in retinal neuronal populations in the DBA/2J mouse. Cell. Tissue Res. 2005, 320, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Nork, T.M.; Ver Hoeve, J.N.; Poulsen, G.L.; Nickells, R.W.; Davis, M.D.; Weber, A.J.; Vaegan; Sarks, S.H.; Lemley, H.L.; Millecchia, L.L. Swelling and loss of photoreceptors in chronic human and experimental glaucomas. Arch. Ophthalmol. 2000, 118, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Matlach, J.; Mulholland, P.J.; Cilkova, M.; Chopra, R.; Shah, N.; Redmond, T.; Dakin, S.C.; Garway-Heath, D.F.; Anderson, R.S. Relationship between Psychophysical Measures of Retinal Ganglion Cell Density and In Vivo Measures of Cone Density in Glaucoma. Ophthalmology 2017, 124, 310–319. [Google Scholar] [CrossRef][Green Version]
- Hasegawa, T.; Ooto, S.; Takayama, K.; Makiyama, Y.; Akagi, T.; Ikeda, H.O.; Nakanishi, H.; Suda, K.; Yamada, H.; Uji, A.; et al. Cone Integrity in Glaucoma: An Adaptive-Optics Scanning Laser Ophthalmoscopy Study. Am. J. Ophthalmol. 2016, 171, 53–66. [Google Scholar] [CrossRef]
- Vincent, A.; Shetty, R.; Devi, S.A.; Kurian, M.K.; Balu, R.; Shetty, B. Functional involvement of cone photoreceptors in advanced glaucoma: A multifocal electroretinogram study. Doc. Ophthalmol. 2010, 121, 21–27. [Google Scholar] [CrossRef]
- Ortin-Martinez, A.; Salinas-Navarro, M.; Nadal-Nicolas, F.M.; Jimenez-Lopez, M.; Valiente-Soriano, F.J.; Garcia-Ayuso, D.; Bernal-Garro, J.M.; Aviles-Trigueros, M.; Agudo-Barriuso, M.; Villegas-Perez, M.P.; et al. Laser-induced ocular hypertension in adult rats does not affect non-RGC neurons in the ganglion cell layer but results in protracted severe loss of cone-photoreceptors. Exp. Eye Res. 2015, 132, 17–33. [Google Scholar] [CrossRef] [PubMed]
- Inman, D.M.; Horner, P.J. Reactive nonproliferative gliosis predominates in a chronic mouse model of glaucoma. Glia 2007, 55, 942–953. [Google Scholar] [CrossRef]
- Schneider, M.; Fuchshofer, R. The role of astrocytes in optic nerve head fibrosis in glaucoma. Exp. Eye Res. 2016, 142, 49–55. [Google Scholar] [CrossRef]
- Seitz, R.; Ohlmann, A.; Tamm, E.R. The role of Muller glia and microglia in glaucoma. Cell. Tissue Res. 2013, 353, 339–345. [Google Scholar] [CrossRef]
- Bagnis, A.; Izzotti, A.; Centofanti, M.; Saccà, S.C. Aqueous humor oxidative stress proteomic levels in primary open angle glaucoma. Exp. Eye Res. 2012, 103, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Li, F.; Kong, L.; Chodosh, J.; Cao, W. Anti-inflammatory effect of pigment epithelium-derived factor in DBA/2J mice. Mol. Vis. 2009, 15, 438–450. [Google Scholar] [PubMed]
- Ji, M.; Miao, Y.; Dong, L.D.; Chen, J.; Mo, X.F.; Jiang, S.X.; Sun, X.H.; Yang, X.L.; Wang, Z. Group I mGluR-mediated inhibition of Kir channels contributes to retinal Muller cell gliosis in a rat chronic ocular hypertension model. J. Neurosci. 2012, 32, 12744–12755. [Google Scholar] [CrossRef] [PubMed]
- Ashimatey, B.S.; King, B.J.; Swanson, W.H. Retinal putative glial alterations: Implication for glaucoma care. Ophthalmic Physiol. Opt. J. Br. Coll. Ophthalmic Opt. 2018, 38, 56–65. [Google Scholar] [CrossRef]
- Nutzi, C.; Schotzau, A.; Grieshaber, M.C. Structure and Function Relationship of Activated Retinal Glia in Primary Open-Angle Glaucoma Patients. J. Ophthalmol. 2017, 2017, 7043752. [Google Scholar] [CrossRef]
- Grieshaber, M.C.; Moramarco, F.; Schoetzau, A.; Flammer, J.; Orguel, S. Detection of retinal glial cell activation in glaucoma by time domain optical coherence tomography. Klin. Monbl. Augenheilkd. 2012, 229, 314–318. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, Y.; Chen, S.; Vernon, G.; Wong, W.T.; Qian, H. Light-Dependent OCT Structure Changes in Photoreceptor Degenerative rd 10 Mouse Retina. Invest. Ophthalmol. Vis. Sci. 2018, 59, 1084–1094. [Google Scholar] [CrossRef]
- Garcia Garrido, M.; Mühlfriedel, R.L.; Beck, S.C.; Wallrapp, C.; Seeliger, M.W. Scale Adjustments to Facilitate Two-Dimensional Measurements in OCT Images. PLoS ONE 2015, 10, e0131154. [Google Scholar] [CrossRef]
- Reinehr, S.; Gomes, S.C.; Gassel, C.J.; Asaad, M.A.; Stute, G.; Schargus, M.; Dick, H.B.; Joachim, S.C. Intravitreal Therapy Against the Complement Factor C5 Prevents Retinal Degeneration in an Experimental Autoimmune Glaucoma Model. Front. Pharmacol. 2019, 10, 1381. [Google Scholar] [CrossRef] [PubMed]
- Casola, C.; Reinehr, S.; Kuehn, S.; Stute, G.; Spiess, B.M.; Dick, H.B.; Joachim, S.C. Specific Inner Retinal Layer Cell Damage in an Autoimmune Glaucoma Model Is Induced by GDNF With or Without HSP27. Invest. Ophthalmol. Vis. Sci. 2016, 57, 3626–3639. [Google Scholar] [CrossRef] [PubMed]
- Reinehr, S.; Reinhard, J.; Gandej, M.; Kuehn, S.; Noristani, R.; Faissner, A.; Dick, H.B.; Joachim, S.C. Simultaneous Complement Response via Lectin Pathway in Retina and Optic Nerve in an Experimental Autoimmune Glaucoma Model. Front. Cell Neurosci. 2016, 10, 140. [Google Scholar] [CrossRef]
- Li, J.; Zhao, L.; Urabe, G.; Fu, Y.; Guo, L.W. Epigenetic intervention with a BET inhibitor ameliorates acute retinal ganglion cell death in mice. Mol. Vis. 2017, 23, 149–159. [Google Scholar] [PubMed]
- Reinehr, S.; Reinhard, J.; Gandej, M.; Gottschalk, I.; Stute, G.; Faissner, A.; Dick, H.B.; Joachim, S.C. S100B immunization triggers NFκB and complement activation in an autoimmune glaucoma model. Sci. Rep. 2018, 8, 9821. [Google Scholar] [CrossRef] [PubMed]
- Wilmes, A.T.; Reinehr, S.; Kuhn, S.; Pedreiturria, X.; Petrikowski, L.; Faissner, S.; Ayzenberg, I.; Stute, G.; Gold, R.; Dick, H.B.; et al. Laquinimod protects the optic nerve and retina in an experimental autoimmune encephalomyelitis model. J. Neuroinflammation 2018, 15, 183. [Google Scholar] [CrossRef]
- Palmhof, M.; Lohmann, S.; Schulte, D.; Stute, G.; Wagner, N.; Dick, H.B.; Joachim, S.C. Fewer Functional Deficits and Reduced Cell Death after Ranibizumab Treatment in a Retinal Ischemia Model. Int. J. Mol. Sci. 2018, 19, 1636. [Google Scholar] [CrossRef]
- Pfaffl, M.W.; Horgan, G.W.; Dempfle, L. Relative expression software tool (REST) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Res. 2002, 30, e36. [Google Scholar] [CrossRef]


| Primary Antibodies | Secondary Antibodies | ||||
|---|---|---|---|---|---|
| Antibody | Company | Dilution | Antibody | Company | Dilution |
| Anti-Brn3a | Santa Cruz | 1:100 | Donkey anti-goat Alexa Fluor 488 | Dianova | 1:500 |
| Anti-cleaved caspase 3 | Sigma-Aldrich | 1:300 | Donkey anti-rabbit Alexa Fluor 555 | Invitrogen | 1:500 |
| Anti-gephyrin | Synaptic Systems | 1:500 | Donkey anti-rabbit Alexa Fluor 488 | Thermo Fisher | 1:500 |
| Anti-GFAP | Millipore | 1:500 | Donkey anti-chicken Cy3 | Millipore | 1:500 |
| Anti-glutamine synthetase | Abcam | 1:500 | Goat anti-mouse Alexa Fluor 488 | Invitrogen | 1:500 |
| Anti-neurofilament H | Synaptic Systems | 1:500 | Donkey anti-chicken Cy3 | Millipore | 1:500 |
| Anti-opsin | Millipore | 1:1200 | Donkey anti-rabbit Alexa Fluor 555 | Invitrogen | 1:500 |
| Anti-PKCα | Santa Cruz | 1:300 | Goat anti-mouse Alexa Fluor 488 | Invitrogen | 1:500 |
| Anti-rhodopsin | Abcam | 1:400 | Goat anti-mouse Alexa Fluor 488 | Eugene | 1:500 |
| Anti-ribeye | Synaptic Systems | 1:500 | Donkey anti-guinea pig Alexa Fluor 488 | Jackson Immuno Research | 1:500 |
| Anti-recoverin | Millipore | 1:1000 | Donkey anti-rabbit A555 | Invitrogen | 1:400 |
| Anti-Vglut1 | Synaptic Systems | 1:500 | Donkey anti-chicken Cy3 | Millipore | 1:500 |
| Anti-rhodopsin | Abcam | 1:400 | Goat anti-mouse Alexa Fluor 488 | Eugene | 1:500 |
| Protein | Background | Lower Threshold | Upper Threshold |
|---|---|---|---|
| Gephyrin | 50 | 6.01 | 252.22 |
| GFAP | 50 | 8.97 | 246.72 |
| Glutamine synthetase | 50 | 10.47 | 262.82 |
| Neurofilament H | 50 | 10.22 | 84.63 |
| Recoverin | 50 | 9.95 | 264.02 |
| Rhodopsin | 50 | 9.02 | 265.00 |
| Vglut1 | 50 | 6.62 | 250.29 |
| Gene | Forward (F) and Reverse (R) Oligonucleotides | GenBank Accession Number | Amplicon Size |
|---|---|---|---|
| Actb-F Actb-R | ctaaggccaaccgtgaaag accagaggcatacagggaca | NM_007393.5 | 104 bp |
| Bax-F Bax-R | gtgagcggctgcttgtct gtgggggtcccgaagtag | NM_007527.3 | 73 bp |
| Bcl2-F Bcl2-R | agtacctgaaccggcatctg ggggccatatagttccacaaa | NM_009741.5 | 77 bp |
| Casp3-F Casp3-R | gaggctgacttcctgtatgctt aaccacgacccgtccttt | NM_001284409.1 | 77 bp |
| Gphn-F Gphn-R | tgatcttcatgctcagatcca ttgcaaatgttgttggcaag | NM_145965.2 | 68 bp |
| Nefh-F Nefh-R | cattgagattgccgcttaca actcggaccaaagccaatc | NM_010904.3 | 67 bp |
| Pou4f1-F Pou4f1-R | ctccctgagcacaagtaccc ctggcgaagaggttgctc | AY706205.1 | 98 bp |
| Ppid-F Ppid-R | ttcttcataaccacaagtcaagacc tccacctccgtaccacatc | M60456.1 | 95 bp |
| Prkca-F Prkca-R | caagggatgaaatgtgacacc cctcttctctgtgtgatccattc | NM_011101.3 | 96 bp |
| Rcvrn-F Rcvrn-R | caatgggaccatcagcaaa cctaggcttgatcattttga | NM_009038.2 | 71 bp |
| Rho-F Rho-R | tgtggtcttcacctggatcat gaacattgcatgccctcag | NM_145383.1 | 90 bp |
| Slc17a7-F Slc17a7-R | gtgcaatgaccaaggacaag agatgacaccgccgtagtg | NM_182993.2 | 103 bp |
| Slc18a3-F Slc18a3-R | agagccctaccctgatctctg caagtaggcgctggcattag | NM_021712.3 | 77 bp |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weiss, M.; Reinehr, S.; Mueller-Buehl, A.M.; Doerner, J.D.; Fuchshofer, R.; Stute, G.; Dick, H.B.; Joachim, S.C. Activation of Apoptosis in a βB1-CTGF Transgenic Mouse Model. Int. J. Mol. Sci. 2021, 22, 1997. https://doi.org/10.3390/ijms22041997
Weiss M, Reinehr S, Mueller-Buehl AM, Doerner JD, Fuchshofer R, Stute G, Dick HB, Joachim SC. Activation of Apoptosis in a βB1-CTGF Transgenic Mouse Model. International Journal of Molecular Sciences. 2021; 22(4):1997. https://doi.org/10.3390/ijms22041997
Chicago/Turabian StyleWeiss, Maximilian, Sabrina Reinehr, Ana M. Mueller-Buehl, Johanna D. Doerner, Rudolf Fuchshofer, Gesa Stute, H. Burkhard Dick, and Stephanie C. Joachim. 2021. "Activation of Apoptosis in a βB1-CTGF Transgenic Mouse Model" International Journal of Molecular Sciences 22, no. 4: 1997. https://doi.org/10.3390/ijms22041997
APA StyleWeiss, M., Reinehr, S., Mueller-Buehl, A. M., Doerner, J. D., Fuchshofer, R., Stute, G., Dick, H. B., & Joachim, S. C. (2021). Activation of Apoptosis in a βB1-CTGF Transgenic Mouse Model. International Journal of Molecular Sciences, 22(4), 1997. https://doi.org/10.3390/ijms22041997








